| This page in the original is blank. |
Longitudinal Studies of Blood Availability
Douglas M. Surgenor
I will present some findings today from a survey of blood collections and transfusions in the United States during the 1992 calendar year.1
Blood is collected by two types of institutions in the United States: regional blood centers and hospitals. In 1992, estimated domestic blood collections included 12,035,000 units of allogeneic blood, i.e., blood donated by others; 1,117,000 units of autologous blood, i.e., blood donated by patients for their own expected use; and 436,000 units of directed blood, i.e., blood donated by others for use by designated patients (Table 1). Most of the domestic blood (approximately 90 percent) was collected by 176 regional and community blood centers. The remainder was collected by hospitals. Testing of blood for disease and other markers resulted in the exclusion of 4.5 percent of collected units.
When 1992 collections were compared against 1989 collections, some interesting trends were noted. Total estimated allogeneic collections decreased by 7.0 percent. On the other hand, autologous donations increased by more than 70 percent, while directed donations increased by almost 25 percent. The strong upward trend in autologous and directed donations reflects patient concerns about the safety of the blood supply. The high rates of nonutilization of these units are attributable to overdonation beyond expected need, lack of clear criteria for their transfusion, and hesitancy to "cross over" unused units for use by others. Unfortunately, there is a large economic cost to the nonutilization of these units; in many instances, the cost must be borne by the hospital in which the units were not transfused. We observed substantial differences between the rates of autologous and directed donations in different U.S. census regions. In New England, autologous units represented 3.2 percent of total collections, and in the Pacific region, autologous units represented 9.9 percent of total collections. The high rate in the Pacific region has been
attributed, in part, to legislation in California that requires counseling of patients about their transfusion options. Similar differences were observed in the rates of directed donation, from 0.8 percent in New England to 6.1 percent in the East South Central region.
TABLE 1 Units of Red Blood Cells (RBC) and Platelets Collected and Transfused in the United States in 1989 and 1992.
|
Blood Component |
1989 |
1992 |
% Change |
|
Collected |
|||
|
Allogeneic RBC |
12,939,000 |
12,035,000 |
-7.0 |
|
Autologous RBC |
655,000 |
1,117,000 |
70.5 |
|
Directed RBC |
350,000 |
436,000 |
24.6 |
|
Platelets |
2,190,000 |
3,828,000 |
74.8 |
|
Transfused |
|||
|
Allogeneic RBC |
11,532,000 |
10,491,000 |
-9.0 |
|
Autologous RBC |
356,000 |
566,000 |
59.0 |
|
Directed RBC |
97,000 |
136,000 |
40.2 |
|
Platelets (apheresis) |
2,112,000 |
3,642,000 |
72.4 |
|
Platelets (concentrate) |
5,146,000 |
4,688,000 |
-8.9 |
Transfusions of blood and its components were given almost exclusively to hospitalized patients. Transfusions of allogeneic blood fell by 9 percent between 1989 and 1992. This was the first time in over 20 years of surveillance of the national blood supply that such substantial declines in these indices of transfusion activities have been recorded. The U.S. red cell transfusion rate, measured in units transfused per thousand population, also fell substantially. The proportion of collected units of red cells that were transfused varied depending on donation category. Approximately 92 percent of collected allogeneic units remaining after testing were transfused. However, only 51 percent of autologous units were transfused to the patient who gave them. The remaining autologous units were not used. As for directed units, only 31 percent were transfused.
The findings from this survey indicate that over 1.5 million units of allogeneic red cells from the national blood resource were outdated, lost, or unaccounted for during 1992 (Table 1). Given a red cell dating period of about 35 days, this implies that 100,000 units of red cells became outdated every 35 days during the year. Most of those units were on hospital blood bank shelves when they became outdated. Assuming that every allogeneic unit had an equal chance of being transfused to a patient, this corresponds to an average of 20
units of various blood types becoming outdating every 35 days in each of 5,000 hospitals.
The high rate of non-use of autologous and directed units (49 percent) stems from the fact that such units were only rarely used for unrelated patient transfusions.
Our observations on the collection and transfusion of platelets are of particular interest. Collections of apheresis platelets increased by more than 72 percent between 1989 and 1992. Apheresis is a procedure in which, using a special machine, blood is collected, platelets are separated, and the remaining parts of the blood are returned to the donor. Apheresis platelets so obtained are called single donor platelets to distinguish them from pooled platelet concentrates that are separated from donated whole blood from many donors. The data in Table 1 also reveal substantial changes in the sources of the platelets that were transfused. Between 1989 and 1992, transfusions of single donor platelets increased by more than 72 percent. In the same period, transfusions of platelet concentrates decreased by almost 9 percent in the same interval. The result was a substantial increase in the proportion of platelets being transfused as single donor (apheresis) platelets. This trend benefits patients who need platelets by reducing donor exposure, risks of infection, and immunization and transfusion reactions.
Questions and Comments
Alvin Drake: Doug, of the people who give autologously in preparation for their surgery, any idea of how many of those people end up getting blood from the common supply anyway because the autologous donation wasn't enough?
Douglas Surgenor: It is an interesting problem. In a cohort of over 500 total hip replacement patients who had predeposited autologous units, we observed that the proportion of patients who completely avoided transfusion with allogeneic units increased as the number of units predeposited increased from one up to four units. About 80 percent of the patients who donated two units avoided any allogeneic transfusion. When three units had been donated, about 95 percent avoided allogeneic transfusion. However, there was an offsetting cost: the percentage of donated autologous units that were not needed rose from about 20 percent when two units were predeposited to 50 percent when three units were predeposited.
Toby Simon: If I followed your discussion correctly, the outdate or discard rate on allogeneic, undirected, and non-autologous blood is in the neighborhood of a million units. Not included in that is a discard rate from directed and autologous donations, so your actual discard rate is close to 1.9
million units a year?
Douglas Surgenor: Yes. That is about right.
Arthur Bracey: I am very interested in that 1.9 million units. Are there data reported by the hospital transfusion services to the AABB on outdates so that you can compare the calculated number with the observed number?
Douglas Surgenor: A large proportion of the data that we used to estimate the national activities that I have touched on came from reports submitted to AABB by its member hospitals.
William Sherwood: There seems to be a focus on that 1.9 million outdates and the study, I am assuming, did not number these by blood type. It would be my observation that these outdates are mostly Type A and Type B and if anyone is looking at these as a source to solve a nationwide problem of occasional shortages, that really won't be useful. Our shortages are usually Type O, and distributing more of those Type A's and Type B's around the country isn't going to help.
Douglas Surgenor: That is a good point. We found that if you take autologous and directed units out of the picture and just look at the difference between what allogeneic blood was collected and what was transfused in 1989 and 1992, the same net amount of blood was not used. It comes out at about a million units that were not used in each year. That suggests there is kind of an irreducible minimum.
Eve Lackritz: What I found interesting is you are seeing an increase in these directed and autologous donations, which is being driven, perhaps, by concerns about the safety of the general supply. But in actuality, you say the blood supply is getting progressively safer over the years.
Douglas Surgenor: That is a fact, but we don't know the factors that may be driving autologous and directed donations.
Ernest Simon: I think the point you made is that there were a million units that are unaccounted for. It is not clear that they are outdated. The reason I mention that is that I think in the Booz-Allen report of 1971, there was about 25 percent that was unaccounted for. And I am not sure that we should equate "unaccounted for" with "outdated."
Douglas Surgenor: No, the point being that you have got to realize that the
blood resource is spread over these several thousand hospitals in the United States. In addition to actual outdating, there is loss due to breakage, handling, and that sort of thing.
Celso Bianco: Dr. Surgenor, you implied that it was somewhat unfair for the patient's hospital to be charged for all of these units that are ultimately not used. Who should pay for them?
Douglas Surgenor: I don't know. All I know is that there is an inequity here that hasn't been addressed.
Harvey Klein: As you pointed out, these data were collected in 1992. There were enormous trend changes between 1989 and 1992. We don't know what is happening between 1992 and 1995. Do you know of any effort to collect such data? Are you going to be collecting them, or do you know of any efforts to get more rapid turnaround so that we can make decisions more reasonably, based on available data.
Douglas Surgenor: Yes. We have initiated a new survey of collections and transfusions. With the cooperation of AABB, we hope that the results can be made available by 1996.
| This page in the original is blank. |
Blood Supply Fluctuations
Jeffrey McCullough
This presentation will focus on fluctuations in the blood supply. First of all, why should we care about the fluctuation in the blood supply? An inadequate blood supply may pose a danger to patients if the proper amount and type of blood is not available when needed. It may lead to longer hospital stays if elective procedures must be postponed in order to wait for an adequate blood supply. This leads to increased costs, as well as potential risks for patients. On the other hand, an excess supply leads to wastage and increased costs, and it also calls into question whether we have served blood donors properly by obtaining blood that is not needed or was not used for patient therapy.
Is there a problem with the fluctuation in the blood supply, and if so, how big is the problem? Acquiring these kinds of data is fairly difficult, so the data are not as adequate as would be ideal in order to address this issue. Hospitals usually track blood utilization in fairly large blocks of time, quarterly or monthly. Although the data are available, it is not customary for hospitals to look at fluctuations in blood usage on a weekly or a daily basis. Thus, by looking at utilization data in a longer time frame, some of these fluctuations don't become apparent. What we are really concerned about is more short term fluctuations in the blood supply that will lead to temporary imbalances.
Today I am focusing on short-term fluctuations, where it is necessary to tailor the blood availability to specific patient needs. Over the longer term, there will be shifts in blood utilization based on changes in medical therapy and changes in the indications for transfusion, leading to variations in the blood supply. The blood supply that is necessary in a hospital is based on physician requests for blood to be made available for use rather than actual blood use. The size of the inventory is thus not necessarily based on how much patients use. If requests for blood are far in excess of actual usage, the hospital's inventory is inflated and the likelihood of blood wastage then increases. This is something that we dealt with over the last 15 years with the development of more structured approaches to the amount of blood that is cross-matched.
There is some need for an extra safety margin in the blood inventory. One of the issues is the size of this safety supply or safety margin. Is it possible to have this safety margin without some outdating or wastage? It may be that this kind of a safety stop is necessary in order to operate the system.
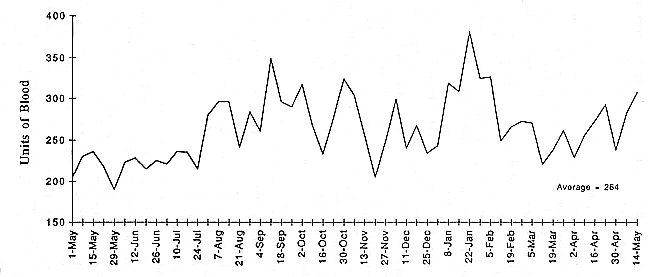
I want to look at the extent of this fluctuation and spend a few minutes talking about the reasons for these kinds of fluctuations and how well the system is organized to cope with these fluctuations. The average weekly blood usage from our hospital, the University of Minnesota Hospital, from May 1, 1994 through May 1995 was 264 units of red cells (Figure 1). The weekly usage range was from a low of about 195 units toward the end of May to 395 units around January or February. Thus, our blood supplier was expected to deal with an unplanned difference of blood use of 200 units a week or almost 100 percent of the lowest weekly use.
Some of these lower usage weeks can be accounted for by certain events, but others cannot. For Memorial Day week, which is a shorter work week, there was a decrease in blood utilization. On the other hand, during the Fourth of July week and Labor Day week there was not the kind of decrease that we saw with the Memorial Day week. Christmas week is not one of our lower weeks, although Thanksgiving week did show decreased blood use. So, holidays had some effect, but not always the effect that we might have expected.
I have no explanation for the two peaks in utilization in early September and mid-January. I believe with tracking of relevant patient-related activity it would be possible to determine the causes of the increased blood use. Thus, although we often think that holiday weeks affect this balance between supply and utilization, in our experience last year they didn't affect utilization as much as we might have thought.
What are some of the reasons for these fluctuations? The week that the American College of Surgeons meets is something that we notice. When most of the surgeons are out of town, the elective surgery schedule is decreased and the blood utilization that week is lower. Short work weeks affect the elective surgery schedule, and this also is noticeable, but as we have seen, not dependably so. Medical meetings other than surgical meetings usually don't have that much of an effect on blood utilization.
Another source of variation can be one or two particular patients, such as a major trauma case, who might use 50, 60, or 70 units of red cells. A unique patient mix may also affect blood use. For instance, if one or two organ donors become available and most of those organs match patients, our hospital might do multiple transplants in the course of 24–48 hours—a couple of
livers, several kidneys, and a heart. A unique set of circumstances can arise in this way in a period of a day or two and place a very substantial demand on red cells.
Another example is that sometimes groups of patients just fall together. For instance, a large bone marrow transplant program may have about 40 beds on the unit. There will be times when 10 or 12 patients may be discharged at about the same time. If you have about 12 new patients all coming on the transplant unit within a couple of days of each other, there is a huge change in the blood demands over a period of about a week because of this confluence of patient factors. Last on this list is a major disaster. This is something we talk about and think about, but, in fact, it doesn't turn out to be that much of a factor in all this because these are such rare events.
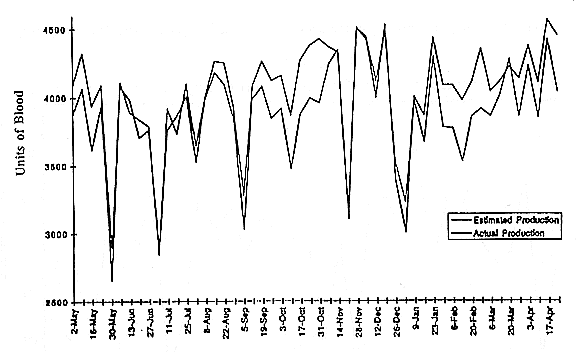
We have been discussing fluctuations in blood utilization. Now, let's consider the supply side. With supply, we have a more dependable change in blood availability. There are some periods of the year—in May, in October—when there is a substantial difference between what we actually collected and what we had planned to collect. For the most part, though, blood production coincides with the planned amount to be collected.
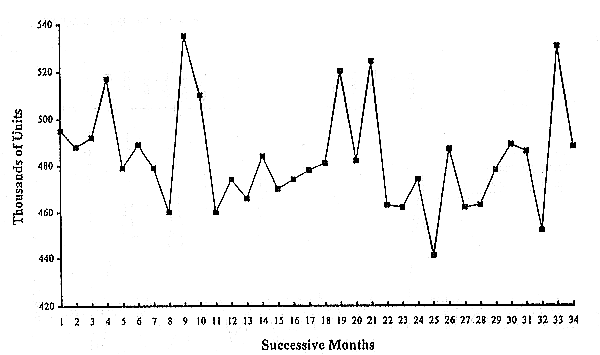
If these collection goals were tightly related to the projected blood needs, these time periods would be potential problems of lack of supply. The American Red Cross national data for June 1992 to April 1995 (Figure 2, kindly provided by Brian McDonough) reveals considerable monthly fluctuation. Monthly collections varied from about 440,000 to 540,000 units, a range of about 20 percent of the lowest monthly collection. If we look at the data from the North Central Region of the ARC, which includes Minnesota, we see collections averaging around 4,000 to 4,100 units a week (Figure 3). On a short week, it drops down to under 3,000, but most weeks it runs closer to 4,000. Toby Simon could show you similar data from Phoenix and Chicago, including some of the same kinds of differences between budgeted and actual collections.
Another way to consider these same data, the fluctuation in blood availability, is based on deviation from the projected blood collections. In one blood collection center, it ranged from a high of 130 percent of what they planned to collect to a low of around 75 percent of what they planned to collect, which has been our experience in St. Paul.
Many of the factors causing this fluctuation are local. One of these is the weather. It is too hot, or it is too cold. It may be that it rained, or that it didn't rain and it has been dry for two weeks. It may be that there was a blizzard. It seems as if whatever is happening out there is the reason that is used to account for why we didn't collect the blood that we thought we needed. The weather is not a common factor that affects blood availability,
but when it does, it certainly can be a very major issue.
Time of year is another factor in blood collections. We find in general that spring is a much more effective time to operate a blood drive in high schools and colleges. High school students age during the year, so you have a larger proportion of students toward the end of the school year old enough to donate blood than in the fall.
Another consideration is work or manufacturing plant schedules, depending on how the manufacturing plant is running and whether or not they change their schedules. Probably the most important factor, however, that determines blood availability and consistency of supply is the donor recruitment staff at the blood center. A qualified, knowledgeable staff can have an enormous effect on increasing blood availability.
There are no solid answers to the issue of fluctuations in blood use and supply, partly because the factors that may influence utilization and collection are not things that we ordinarily document. Thus, we can't go back to identify at these periods of peaks and valleys what exactly was happening in the hospital to account for changes in utilization or exactly what was happening in the blood collection system to account for changes in availability. As with so many things, improved data would be valuable.
Questions/Comments
Steve Bradshaw: I recently entered this area of expertise from the orthopedic business, and it was interesting to me to see the seasonality of blood usage vary with the orthopedic schedule of seasonality factors. There are over a half a million procedures of joint replacement annually, and they use as much as three to five units of blood. Seasonally, it occurs more toward the colder time of the year and ties into some of the other points you have made.
Alvin Drake: What is the rate of surgery in your hospital? Is the blood there when it is needed? Is the issue that it is difficult to get it? Or is the issue that it is not there when you need it?
Jeffrey McCullogh: We don't have a problem with blood availability. In 20 years, I can't remember being unable to do surgical procedures because of the lack of blood. The issue is more to try to match supply with demand, avoid wastage as well as shortages, and be optimally cost-effective.
Harvey Klein: But in all fairness, you are an exporter. How about some people who are importers?
Jeffrey McCullough: Yes, we are quite unusual because we collect twice as
much as we use locally, and the rest of it is exported to other parts of the country.
William Sherwood: I'm with the Red Cross in Philadelphia, and we are an importer. We do have very little delay of elective surgery. Virtually all of our surgery is taken care of. There are only a very small number of cases per year where there is cancellation of elective surgery, and it is at some of those times that were pointed out. There is another practical matter. A blood center can't build in a capacity to have an enormous collection at one peak and then people sitting on their hands for a very short period of time. So, the capacity planning often limits what you can collect at any given point in time. We plan capacity for a little bit over what that median would be, so there are times that we can collect from all the donors we want to provide for and times that we have more capacity available than donors. So, capacity planning and the capacity of a blood center are as important for the short-term fluctuations as the real availability of the blood supply.
S. Gerald Sandler: From a hospital perspective, I would like to point out that the inference that it is bad to have a shortage isn't always true. When there is a shortage in Washington, for example, on platelets, we will drop the trigger for obtaining platelets from 20,000 mm3 to 5,000 mm3 at the hospital level and get to where we probably should be anyway. If there is a shortage of red cells, we will be forced to go back to the level we probably should use. No one has bled to death in Washington during a shortage. We have probably come closer to the levels that we should be using for our triggers.
Jeffrey McCullough: There is a related theme that I would like to mention. A friend and colleague of mine in Minneapolis, Herb Polesky, got into a fairly vigorous debate at the state medical association meeting a few years ago when he took the position that it was okay periodically to have blood shortages because it brought to the attention of the physicians that blood is not just a casual resource out there in unlimited supply. He noticed that during and shortly after blood shortages, physicians took their decisions about transfusions much more seriously and that it was actually easier to get them involved in helping to recruit blood donors.
Ronald Gilcher: A comment and a question. The comment is that it is very clear to me that if we had a steady flow of donations, it would be much better from the standpoint of staffing and not having to pay overtime at the times when you overcollect. My question is when we look at the valleys that you mentioned, they were both budgeted and actual. Was it budgeted because your recruitment staff thought that they couldn't collect or was it because you
decided you didn't want to collect at that time? The reason I make that comment is we actually tend to overcollect around the holidays because our center gets requests from all over the country because of shortages, and, in fact, we do budget and we are able to overcollect at the time of the holidays.
Jeffrey McCullough: I think a lot of the things that some donor recruitment people take as givens of what they can and can't do are probably not really givens at all, and for the most part, those low weeks were planned low weeks because people thought that they couldn't get the blood.
Celso Bianco: I think that we are all beginning to realize that many of these valleys and peaks are management issues that are very difficult to deal with. I remember the New York Times article that was very critical of our shortages about a year or two ago. We can plan for Christmas. Christmas happens every year, and I think that we and most of the blood centers have learned how to manage. I think that we cannot plan for certain events that occur in the country. For some reason donors decide either that they want to donate a lot—and the Gulf War is a good example, along with the bombing in Oklahoma City—or they don't, and the blood supply dries up. I think that that is the part of the problem on which we have to focus. In New York we learned for instance that when we need to import, we can do that. We oscillate; in certain months, such as May and June, we will collect 30,000 units a month, but we will collect 40,000 units a month in July and August and we learn how to manage that.
Thomas Zuck: There is a corollary to the argument you and Herb had. You looked at it from the usage side. Ron Franzmeier, who most of us knew, used to say that it wasn't bad to have an appeal every now and then because the general public knows you are still there and still need blood.
Harvey Klein: I would like to ask you two questions. The first is the data that you presented were for red cells. Is there a problem with platelets as well, which clearly are different with the increase in growth of single donor platelets? Second, couldn't we totally eliminate the issue of shortages by using frozen depots in the United States? Couldn't we eliminate that problem at a cost?
Jeffrey McCullough: Platelets are a more difficult issue because of the shorter shelf life. As Bill Sherwood mentioned, with 42 days of red cells, there is a lot more latitude, whereas long weekends can be difficult for platelets. I also agree with Dr. Bianco that we are supposed to be the professionals and we are supposed to be able to plan and manage for this. I
think we have allowed more excuses to be foisted on us than is probably appropriate.
Regarding frozen red cells, I suppose the answer is "Yes, it could be done if you don't care how much it costs." To try to plan for a system in which for a few days you could deglycerolize huge numbers of red cells would be very costly and inefficient. I don't think it is a practical alternative.
Paul McCurdy: Jeff, your hospital weekly utilization data were very interesting and probably not too surprising. I suspect that if you look at the same data from the standpoint of the so-called blood center you may find they don't fluctuate a great deal because when one hospital is up, another one is down. The exceptions would be smaller blood centers and places where there is one major hospital that uses almost all of the blood supply, so that their fluctuation goes to the blood center.
When Should Physicians Transfuse?
Jeffrey L. Carson
My job today is to tell you whether we in the practice of clinical medicine are appropriately using red cell transfusions. I will try to create for you a framework for a transfusion decision and what approaches have been used to determine what the appropriate use of transfusions has been. I will then show you some relevant animal data, and finally I will turn to two studies that we are in the process of completing: one we call Anemia and Surgery and the second is Surgical Blood Transfusion Variation and Outcome. I will try to convince you that we need large randomized clinical trials to answer this question definitively and that we will never know what the appropriate use is unless we get more data.
I think about transfusions in the following way. The first question I want to know is what are the mortality and morbidity levels associated with different levels of hemoglobin and how are those affected by age, comorbidity, the nature of the surgical procedure, and operative blood loss. These are issues that I think need to be incorporated into any transfusion decision. Second, if you understand what these mortality and morbidity levels are, how much can they be reduced by transfusion? Third, of course, is if you can reduce the immediate mortality and morbidity, at what risk is the patient from side effects of transfusion?
Now, this can be looked at in a slightly different way. If we were to take a surgical environment, where you are trying to make a decision to transfuse or not transfuse, and you decide to transfuse that patient, there is going to be a mortality associated with surgery that may or may not be influenced by the transfusion. The patient may survive. If the patient survives, then he or she is at risk for complications of transfusion, which include HIV, hepatitis, and perhaps bacterial infections, which may, in fact, be the most important adverse event related to transfusion.
You can add up the risks and benefits and compare them with those in the group you don't transfuse. Then you try to decide whether those who were transfused do better than those who were not transfused. And then, of course, you need to look at this in subgroups, such as those with underlying heart
disease, how old they are, and the nature of the blood loss in surgical procedures. Finally, we must consider the economic consequences of transfusion decisions.
What approaches have been used to try to determine whether there is appropriate blood use? It has been mostly expert opinion. For example, the criteria for transfusion have changed during the past decade. For many years, we were using the 10–30 rule (hemoglobin below 10 g/dl and/or hematocrit below 30 percent). Then in 1988, NIH's consensus conference rejected the 10–30 rule and said that this doesn't make sense, that we ought to not be using specific hemoglobin criteria, and that we ought to be looking at other factors as well. The consensus conference suggested that perhaps 7 to 8 g/dl is a more appropriate hemoglobin level for deciding to transfuse. Of course, this is unsupported as well.
There have been several studies examining the appropriateness of transfusions. I will review one today that was published by Soumerai and colleagues2 in 1993. In this study a group of experts from the Boston area decided on the appropriate indicators for transfusion. These included a pre-transfusion hematocrit of 24 percent and a hemoglobin of 8 g/dl; a fall of hematocrit of greater than or equal to 6 percent in those who had hematocrits between 24 and 30 percent, if they had a history of angina within 24 hours; a myocardial infarction (MI) within the past six weeks; an electrocardiogram which showed ischemic changes; or 1,000 cc of blood loss.
What they found was that about 40 percent of their transfusions were not appropriate by their criteria. About 20 to 25 percent were appropriate and the remaining were a so-called indeterminate group, with hematocrits between 24 and 30 percent, for whom they couldn't decide if the transfusion was appropriate.
They then tried to educate the doctors. They gave lectures and also met with these doctors face to face. They were able to increase somewhat the number of appropriate transfusions, up into the range of 40 percent or so, and they were able to reduce the number of inappropriate transfusions. In the control groups there was no change in the proportion of appropriate transfusions.
This study shows that after an educational program, the proportion of appropriate transfusions increases. The question remaining is whether these are truly the appropriate criteria for transfusion. I think they are quite reasonable, but I am not sure they are the best ones.
What other data are there to guide transfusion decisions? There is very
limited experimental evidence in humans to guide therapy. There have been a number of observational studies, but my view is that they are either seriously flawed or not large enough to answer this question. There has never been a randomized trial large enough to adequately evaluate blood transfusions in any clinical setting.
There are some animal data to draw upon, and if we examine how well animals tolerate anemia, what you find is that normal animals can tolerate anemia down to about about 5 g/dl. At a hemoglobin level of 5 g/dl, changes on the ST segment of the electrocardiogram begin, and below 3 g/dl, lactate production and decreased ventricular function begin and the animals begin to die.
The second point is that when coronary artery disease is experimentally induced by tying off animals' coronary arteries, reducing blood flow 50 to 70 percent, these animals are much less tolerant of anemia than normal animals. Animals with coronary obstruction develop ST segment changes at between 7 and 10 grams.
Obviously, such experiments cannot be done with humans, but I would now like to talk about two studies we are doing that ask similar questions in an ethical approximation. The first I call Anemia and Surgery. The second is a variation of a transfusion practice study. I need to emphasize that we are not done with these studies and what I am going to share with you are preliminary results.
The aims of the Anemia and Surgery study were to estimate the risk of death associated with pre- and postoperative hemoglobin levels and to determine the level of hemoglobin at which mortality begins to rise, controlling for things such as age and morbidity of surgical procedures.
We performed a retrospective cohort study of patients who refused blood transfusion for religious reasons. We were able to recruit surgeons from throughout the country who would allow us to copy their patients' charts, eliminate identifying information, and then have these medical records reviewed by nurse abstractors. We collected information on demographics, comorbidity, and preop hemoglobin levels. Our main outcome measure was 30-day mortality, which was assessed by medical record review and the National Death Index (NDI) Search.
For patients to be eligible for our study, we required evidence in the medical record that the patient would refuse blood transfusion if offered and that the patient underwent a surgical procedure in the operating room. We excluded open heart cases because of the nature of the procedure and blood use there, and we required a preop hemoglobin level in their chart. We restricted the study to those 18 years of age or older because the courts could have forced younger Jehovah's Witnesses to take blood. We had a total of 2,102 patients, of which 1,958 were ages 18 or older. I want to emphasize one point, which is that we have a variable that we call cardiovascular disease,
which includes a history of MI, angina, congestive heart failure, or a history of peripheral vascular disease.
What were some of our results? In this group of 1,958 patients, only 3.2 percent died. We have concluded from this preliminary analysis that surgical mortality was very low in this population who declined blood transfusion, and that it increases with declining hemoglobin levels in patients with cardiovascular disease. In patients with a preoperative hemoglobin of 10 or less, mortality is much more sensitive to preop hemoglobin in patients with cardiovascular disease than in those without such disease. These results suggest that the hemoglobin trigger for transfusion should be at a higher level in patients with cardiac disease than in those without cardiac disease. However, I think a randomized trial is needed to establish whatever the transfusion trigger should be.
We are not nearly as far along on the second study I am going to tell you about. This is a study called Surgical Blood Transfusion Variation and Outcome. It is funded by the Agency for Health Care Policy and Research (AHCPR), and the aims of this particular study are to understand the independent predictors of transfusion practices. We will determine if surgeons are transfusing for comorbidity, age, or other patient characteristics or whether they are using a transfusion trigger. The second aim of the study is to describe the effect of transfusion on postoperative mortality and morbidity. We will study these questions in cases of hip fractures undergoing surgical repair, using 30-day mortality as our primary outcome. Secondary outcomes include mortality up to one year, in-hospital morbidity, bacterial infections, length of stay, and disposition as well as some very simple functional status measures.
The study will include 10,000 hip fracture patients, from four geographic regions, who underwent surgery during the past ten years. So far, we have only collected the data. We are about two weeks away from having our mortality data all defined. It is probably not going to be until the end of the summer that I have some good preliminary results. What we plan to do is to describe the mortality and morbidity in patients who are transfused compared to those in patients who are not transfused.
How do these studies all fit together? How do they try to get at the answer to this question of who we should be transfusing? I think the Anemia and Surgery study identifies the hemoglobin levels at which the death rate begins to rise, above which you clearly wouldn't consider transfusing. However, it doesn't tell you whether transfusion is going to impact that increased mortality we were seeing. We expect that the variation study will evaluate the effect of blood transfusion on this death rate and how at different hemoglobin levels transfusion affects postoperative mortality and morbidity.
Both of these studies are observational, however, and we will never know for sure if we completely control for confounding variables. Therefore, a
randomized trial will absolutely be needed to define the transfusion trigger. The basic idea of such a study is to contrast two transfusion triggers. I am waiting to see what our observational studies show before we decide what these comparison groups should be. We are proposing to do a very large simple randomized clinical trial in hip fracture patients using 30-day mortality as our primary outcome. The real unknown at this point is the number of patients that we need in this trial. Until I finish the hip fracture study and get my 30-day mortality data, I can't do my sample size calculations.
I would summarize my formal remarks by saying we currently have very few data to guide transfusion decisions. One observational study suggests that patients with cardiovascular disease should be transfused at a higher hemoglobin level than those without cardiovascular disease. However, additional data are required to determine who truly should be transfused, and I think a randomized trial is clearly needed. Without such data, it is very hard for me to answer the question of whether red cells are being appropriately used. My answer is that I don't know. I think there is evidence to suggest that maybe they are not, but I think until you can provide doctors with clear evidence about what they should and shouldn't do, it is hard to convince them that they are doing things inappropriately.
Questions/Comments
Eve Lackritz: What proportion of blood is being used in the surgical setting?
Jeffrey Carson: My sense is that it is probably 80 percent.
Douglas Surgenor: I don't think anybody has a firm number in a large sense.
Jeffrey Carson: I suspect the blood centers have a sense that it is most of the blood supply. It is certainly well over 50 percent.
William Sherwood: Mortality is a nice, clean end point. I am wondering if there are other parameters, though, that are useful to evaluate.
Jeffrey Carson: I couldn't agree more with you. I would be pleased to talk to you later about the problems in trying to do a study of this sort. The basic problem is that with mortality outcome, you don't need the blinded trial. Think about how you might try to blind doctor and patient to who is getting a unit of blood. I think you can't do that practically. You could blind hemoglobin levels to the physicians and the patients and so still keep their assigned groups blinded. The practical part of that is that it is going to require more money to pull that off.
Let's create a scenario in which you show no difference in mortality and morbidity, you show no difference in functional status, but you show a prolonged length of stay or perhaps that more of these older folks are going to nursing homes if they don't get transfused. If I showed that one variable that was significant and nothing else, that would be almost enough reason for me to consider transfusing someone.
I absolutely agree with you that those secondary outcomes would be very important to include in your analysis. It is an issue of how you can do this when you have morbidity as the outcome, which in principle is subject to bias. It is harder to do, but I don't believe that it is an unsurmountable problem.
C. Robert Valeri: The issue that you should look at is whether or not the hemoglobin concentration per se has an important impact on hemostasis.
Jeffrey Carson: They are correlated. Those who bleed have lower hemoglobins. How do you dissect that out? I don't think you can dissect that.
C. Robert Valeri: There are good data to suggest that with anemia, you produce platelet dysfunction. If you are very anemic, you may make the platelets dysfunction, and therefore, you are going to bleed more. So, it would be important to consider surgical patients who are bleeding to be in need of transfused red cells, regardless of their hemoglobin level, to help platelet function.
Harvey Klein: I think one of the points we wanted to make here is that we all assume that too much blood is being transfused in the United States, and it is clear that we really don't know that for certain. In some instances it may be that too little is being transfused.
International Perspectives on Blood Availability
Robert G. Westphal
I've been asked to talk to you today about the international perspective because I just returned from spending three years in Geneva, Switzerland as a Medical Advisor in the Blood Programme Department of the International Federation of Red Cross and Red Crescent Societies. However, I didn't spend much time in Geneva looking at Western European blood programs. I spent a lot of time looking at developing programs, so I don't consider myself an expert on blood transfusion in Western Europe. However, I do have some items I think we can focus on and possibly learn from.
Comparing Europe with the United States, I think that we in the United States have some problems with the way we handle our donors and consequently with getting our donors back. To put it a different way, I think we have reagent problems in our blood industry.
It wasn't until the end of the 1980s that we began to actually regulate blood component production as a drug manufacturing process. We have overdone it a bit, and it is not entirely the fault of the regulatory agencies. When the regulatory agencies said to build a 12-foot wall, in many cases we built a 20-foot wall. We actually now have a category of "biohazardous donors" whom we treat in an absurdly onerous fashion.
Our donors are the only reagents I know of that change their shape. They forget their birthdays and other identifying numbers. They change their names. They move. They have conjugal relationships with other reagents. They are just not reagents in the classical sense. We should take another look at how we treat them and develop a different view of them.
There is a totally unreported infectious disease that is present in our blood programs today and that is a transfusion-related infection I call the bipolar AA virus. I think we create so much anger and so much apathy—and that is why it is bipolar—in our donors that we are having a lot of trouble. A lot of these remarks are really directed to the panels that will be coming up to discuss some of these issues. We spread this virus fairly readily, and I think we need to look at how we are treating our donors if we want them to keep coming back. I think the compounding of autologous and directed collections in this has not helped us.
What are some of the causes of this AA virus infection? Most of you could promulgate a list as easily as I could. First, we have a tremendous number of donors who are deferred for unnecessary reasons. The American Red Cross alone defers 1.2 million donors per year because of elevated liver enzymes (ALT), mostly false positives. In the Northeast Region in two states, Massachusetts and Maine, after hepatitis C virus (HCV) tests became available in 1990, we lost about 20,000 donors to generally falsely positive ALT tests. To that can be added roughly the same number from false-positive hepatitis B core antibody tests and 3,000 nonconfirmed HIV-positive tests. This is not an insignificant number of donors. Furthermore, they have friends and their friends look at these letters that they get and they are quite concerned.
We also have what Merlyn Sayers described as the donor interrogation or donor inquisition, as some of us have come to call it. I am sure that all of you have seen this letter from a donor in the CCBC Newsletter . I will read some of it in case you haven't seen it.
This is from a 66-year-old man, who has given 11 gallons of blood. He wrote, "Thirty years ago, I got in the habit of donating blood regularly, averaging three times a year. Unfortunately, in recent years, my blood center has made the process so ugly and repugnant, that I have quit. In the case of my blood center, protecting the regulators and the blood center against lawsuits has displaced gathering blood as the primary function. At the predonation interview, a nurse young enough to be my granddaughter asked me a massive series of ugly questions, including those I wouldn't discuss with my male friends. At the end she tells me she knows there is still a good chance that I might have lied in answering all those questions, so she is going to give me one last chance to not withhold the truth. She will give me a moment of privacy where I can relent and fix the bar code warning that I did, in fact, lie."
"Wouldn't it be reasonable to expect that after 30 years of testing and transfusing my blood, over and over again verifying that it is clean and safe, that the blood center's computer system would know that, know me, trust me, and want me? I should have an ID card with a magnetic strip they could run through a reader. After verification of my identity, the question they should ask is, 'Any change since the last time?' If 'No,' skip the next 100 grossly intrusive questions."
He continues in this vein and as a caveat, he says, "I am sure you are thinking that I am just a grumpy old man, but ask yourself how many other grumpy old men and women have stopped coming to give blood to you for those reasons."
He makes two points. One is about the donor interrogation, and the other is that confidential unit exclusion is ambiguous for donors because on the one hand, they are all expected to tell the truth, but some of them can lie if they want and then check another box. We have learned that it is not a very
satisfactory means, at least these days, in most centers for us to screen donors.
There are many examples of the confusing, vague, and frightening letters we send to donors. The country's largest blood collector has now abandoned the whole idea of reentering donors who have false-positive tests. We are also proposing questions to eliminate donors who might be at risk for a very unusual illness, Creutzfeldt-Jakob disease (CJD), caused by something we have not completely identified and cannot confidently say is infectious in the blood transfusion sense. To ask donors questions to prevent something with which they might become infected in the future, or may have in their family but don't know about, is quite a leap into chaos.
Many countries don't do direct questioning of donors. Many countries don't do some of the tests we do, and therefore, they don't have some of the results that we do. For example, ALT testing, HBV core testing, and HTLV-1 and -2 testing are not done in most of the major European countries. The Europeans have also made the absolutely marvelous discovery that the best place to store blood is in the human body. They do very little autologous collection, and they do virtually no directed donations. And the reason they don't is because the patients believe their doctors who tell them that it is not always needed (autologous) and that it is not safer blood (directed).
Major shipments come from several countries in Europe to a well-known blood center in the United States. It is a very good idea. I used to be in a region where we had the opportunity to collect an excess amount of blood and ship it to other regional centers. We were able to help regions that had some shortages. However, when you ship competitively to hospitals, it is disruptive to planning and meeting needs. Then the costs of providing services tend to go up within that region. This doesn't happen in Europe. There are shipments between national blood program centers to other national blood program centers and just a few shipments from one country to another, but really very little of that.
Let me share with you some numbers from the Council of Europe for the year 1989. That same year in Japan and the United States the collections per 1,000 population were 65 and 54, respectively. In Belgium, Denmark, France, to some degree in Luxembourg, and to a tremendous degree in Switzerland, there was a much greater rate of donation. The numbers on Eastern Europe for 1992 get worse. From 1992 to 1993, these have gone down in countries such as Albania, Bulgaria, Romania, and Czech Republic. You see massive declines in the number of blood donations there (See Table 2).
TABLE 2 Number of Voluntary, Nonremunerated Blood Donations per 1,000 Inhabitants in 1989, by European Country
|
Country |
Donations/1,000 |
Country |
Donations/1,000 |
|
Albania |
44 |
Lithuania |
35 |
|
Belgium |
68 |
Luxembourg |
56 |
|
Bulgaria |
24 |
Netherlands |
52 |
|
Czech Republic |
36 |
Poland |
23 |
|
Denmark |
83 |
Portugal |
18 |
|
Estonia |
51 |
Romania |
26 |
|
France |
70 |
Slovakia |
31 |
|
Germany |
44 |
Slovenia |
51 |
|
Greece |
42 |
Spain |
25 |
|
Hungary |
47 |
Switzerland |
100 |
|
Ireland |
37 |
United Kingdom |
44 |
|
Italy |
31 |
|
|
But the real question is why are some of these countries in Western Europe doing so well? How can they collect so much blood? I don't know what the answer is exactly. I can tell you that after talking with several people over there, I have a somewhat different picture of the blood recruitment and collection process than what we have in the United States.
First of all, in general, the collection and/or public education responsibility in Western European countries is given to a single authority in the country, generally under the auspices of some kind of national blood program and policy. Another major difference is that the people responsible for collection and/or donor recruitment invest a major effort at the very highest levels to meet with their counterparts in other organizations and get the recruitment message out to the public. In the United States, this work tends to devolve down to a fairly low common denominator of a donor recruitment person going out and talking with someone at their level at a plant or company, school, or college or university.
There aren't enough big shots involved in donor recruitment in the United States compared to Europe. In Europe there is a great deal of effort, much of which goes back through the history of World War II, to make the program a part of each community. Each community, then, has a real commitment to themselves to provide for their blood needs. In Switzerland, for example, there is a group of almost paramilitary, paramedical personnel, with traditions dating back to World War II, who have a membership of about a million. These
million people are one-seventh of the population of Switzerland, and they comprise a major recruitment group. They go out and run blood drives and they do it at a very grassroots level. Sometimes they do it at a somewhat socially coercive level, but it is very effective.
In Europe, many cities have a sense of community, which I think we lack in many of the cities in the United States. We are just too big. Our messages get totally diluted because of the size and because there are so many other things that are going on. It is a little bit like the rural ethic and the urban ethic. If you live in the country, people know if they don't take care of it, it won't get done, whereas in the larger areas, people think someone else is going to do it.
The other thing in Europe is they don't talk so much about donor recruitment. They talk about donor retention. They want to get their old donors back, the ones that they know, who are safer and easier to find. This donor retention is a multidisciplinary effort, and they all work at it, from the top guy in the Swiss Red Cross right on down.
In addition, young people are viewed in Europe as the key to the future of blood transfusion. Blood needs are part of the biology curriculum in some European schools. The blood service operates as a public trust, and the public expects a tremendous amount of accountability. They ask for it and they generally get it.
So, in summary, in comparing my long experience in the States with my brief experience in Europe, one of the biggest differences has to do with how we treat our donors. Generally speaking, in Europe the donors are treated much more charitably and much more gratefully. In the United States, the donor is viewed with suspicion. He is lying. Otherwise, we would have no confidential unit exclusion (CUE). Donors who have positive infectious disease results that are confirmed positives are generally not notified by letter in Europe. They receive an invitation to visit the doctor, their own or the blood center's. In the United States, they are judged, sentenced, and practically put away by mail with some of these awful letters that all of you know about.
There is much more local autonomy, not so much in regulated areas but in how you organize a blood drive, how you set it up, the rules for collecting and who can be a donor, and what the minimum standards should be. These are not the very precisely defined standards that some of us try to spread across the country, which can be very difficult because communities are different and they expect different things.
Most Europeans don't do direct questioning of donors. They give them the form. They review it carefully. If they have questions about it later, they may call them or if the form isn't filled out right, they will go back to them, but they don't do the donor inquisition. The European forms are simpler in general, but most of the forms look very much like ours. The questions asked
in the countries that export blood to the United States, obviously, include all of the questions that we ask in the United States. They don't do much CUE, and in general, their donors are valued and honored more than ours are.
Jim Mosley noted in 1991 in an article in Transfusion: "The intricacy and complexity which one can get into can overwhelm an honest donor and overwhelm a conscientious interviewer."3 We have exceeded the limits of sensitivity and specificity of the donor questionnaire in excluding hazardous donations. What we are doing to our donors is really unconscionable. I think it is one of the reasons we have trouble with our blood supply. People are saving it up. They don't want the hassle. They will give their own blood when they need it. If a friend asks them to give, maybe they will. And I think that something can be done about that.
Questions/Comments
Arthur Caplan: One of the things you said near the end of your talk was that public accountability is important. I spent the past two days thinking about Mickey Mantle because the public these days is very concerned about issues of equity and fairness in the distribution of organs. One of the issues that comes up with respect to Mr. Mantle and his liver transplant is whether or not the distribution system that is out there is fair and equitable. We have some recent studies that point directly to distrust and mistrust of the system's distribution patterns as reasons for people not to participate in organ donation and tissue donation. This is particularly true for poor and minority people, who basically look at the system and say, "If I can't benefit, I won't donate to it and I won't carry a card and I am not going to be interested in what happens with respect to making my organs or tissues available upon my death."
That is a lesson that may or may not carry into the blood world as well. I know it is true in the organ and tissue world. It comes up with people who either are celebrities or have the ability to command resources out of the system that others can't. Thus, I offer the speakers on the next panel the hypothesis that accountability with respect to distribution and allocation of resources is crucial to understanding how to enhance and find ways to encourage both donor participation and donor retention.