B8 Ethylbenzene
Hector D. Garcia, Ph.D.
Johnson Space Center Toxicology Group
Biomedical Operations and Research Branch
National Aeronautics and Space Administration
Houston, Texas
PHYSICAL AND CHEMICAL PROPERTIES
Ethylbenzene (EB) is a clear, colorless, flammable liquid with an aromatic odor (Windholz, 1976; Sandmeyer, 1981; ACGIH, 1991a).
|
Synonyms: |
Phenylethane, ethylbenzol, ethyl benzene |
|
Formula: |
C8H10; C6H5(CH2CH3) |
|
CAS number: |
100414 |
|
Molecular weight: |
106.18 |
|
Boiling point: |
136.2°C |
|
Melting point: |
-94.9°C |
|
Lower explosive limit: |
1.2% |
|
Upper explosive limit: |
6.8% |
|
Autoignition temperature: |
810°F |
|
Flash point (closed cup): |
59°F |
|
Specific gravity: |
0.8669 at 20°, referred to water at 4°C |
|
Vapor pressure: |
10 mm at 25.9°C |
|
Vapor density: |
3.66 |
|
Solubility: |
Insoluble in water or ammonia; miscible in alcohol, ether, and some organic solvents; soluble in SO2 |
|
Conversion factors |
1 ppm = 4.34 mg/m3 |
|
at 25ºC, 1 atm: |
1 mg/m3 = 0.23 ppm |
|
Human odor threshold: |
10 ppm |
OCCURRENCE AND USE
EB is present in crude petroleum oil and is produced commercially by the alkylation of benzene with ethylene. Its principal use is as a starting material for the production of styrene by catalytic dehydrogenation. It also is used in gasoline and many industrial solvents. It is present in substantial amounts (~ 15%) in technical-grade xylene.
EB has been identified as a trace component of many volatile flavor compounds, such as those found in honey, jasmine, papaya, olive oil, and cheese flavors and in the neutral component of roast-beef flavor isolate (Min et al., 1979).
EB is not used in spacecraft during flight; however, occasionally it is found in the spacecraft atmosphere at concentrations of up to 0.12 ppm, probably due to off-gassing from nonmetallic materials (Liebich et al., 1973; Rippstein and Coleman, 1983).
TOXICOKINETICS AND METABOLISM
The rates of absorption, distribution, and excretion of EB are functions of its solubilities in the various compartments of body tissues and fluids. EB is taken up from the lungs at a moderate rate by the blood and tends to partition preferentially into fat, as indicated by its partition coefficients of 28.4, 1.7, and 3790 for blood, water, and oil, respectively (Sato and Nakajima, 1987). Although no studies were found on the rate of uptake of inhaled EB into the blood in humans exposed to known concentrations of EB, a good estimate of the time needed to reach steady-state blood concentrations can be made based on data available for xylene, a structural isomer of EB. For m-xylene, the concentration in the blood reached a steady state in about 1 h in eight subjects exposed to EB at 100 ppm (Riihimäki and Savolainen, 1980).
Humans exposed for 8 h to EB at 23, 43, 46, and 86 ppm retain 4964% of the EB inhaled (Bardodej and Bardodejova, 1970). Only 4-5% of the retained EB is estimated to be exhaled without transformation (Åstrand et al., 1978). The half-life of EB in the exhaled breath is 0.5 to 3 h (Wolf, 1976).
In humans exposed to EB at up to 85 ppm for 8 h, only traces of EB were found in the expired air after termination of exposure, and only a negligible amount of the unchanged hydrocarbon was excreted in the urine (Bardodej and Bardodejova, 1970). Absorbed EB is excreted mainly in the urine as mandelic and phenylglyoxylic acids (Bardodej and Bardodejova, 1970; Engström et al., 1984; Gromiec and Piotrowski, 1984). Urinary elimination of mandelic acid is biphasic, with half-lives of 3.1 and 24.5 h (Gromiec and Piotrowski, 1984). In four volunteers exposed to EB at 150 ppm, mandelic acid and phenylglyoxylic acid amounted to 90% of the metabolites, and 4% consisted of 4-ethylphenol, p-hydroxyacetophenone, and m-hydroxyacetophenone (Engström et al., 1984).
Rats differ from humans in metabolism of EB, as shown in Table 8-1 (Bardodej and Bardodejova, 1970). It should be noted, however, that the exposure conditions differed: male Wistar rats were exposed at 0, 300, or 600 ppm for 6 h (Engström et al., 1985), and humans were exposed at up to 85 ppm for 8 h. In rats, quantitation of the total output of EB by measurement of urinary metabolites is complicated by the fact that some EB metabolites also are formed endogenously.
The rat study cited above (Engström et al., 1985) also examined elimination kinetics. At 600 ppm, 6% of the amount absorbed (assuming 60% retention) was excreted in the urine during exposure, and 59% was excreted within 48 h of onset of exposure. At 300 ppm, the amounts were 13% during exposure and 83 % within 48 h of exposure (Engström, 1984). In rats inhaling 14C-labeled EB at 230 ppm for 6 h, about 80-90% of absorbed 14C label was excreted in the urine and about 10% was exhaled in breath (Chin et al., 1978).
TABLE 8-1 Metabolism of EB in Humans and Rats
|
Urinary Metabolites |
Humans |
Rats |
|
Mandelic acid |
64% |
25% |
|
Phenylglyoxylic acid |
25% |
- |
|
Methylphenylcarbinol |
5% |
- |
|
1-Phenylethanol |
- |
25% |
|
Benzoic acid |
- |
27% |
In rats exposed 6 h/d for 5 d to various mixtures of xylene and EB in air, metabolite excretion rates abruptly increased from the second day onward for rats exposed to a mixture of m-xylene at 600 ppm and EB at 200 ppm, an exposure concentration that also increased microsomal drug-metabolizing activity in the liver. That mixture also resulted in interactive metabolism; the metabolism of EB was specifically enhanced at that exposure concentration but not at an exposure concentration of m-xylene at 300 ppm and EB at 100 ppm.
TOXICITY SUMMARY
The acute toxicity of EB is low. The main effect of exposure to EB vapors is irritation of eyes, nose, and mucous membranes at concentrations of about 200 ppm; higher concentrations cause central-nervoussystem (CNS) depression and transient liver and kidney injury (Von Burg, 1992). Exposure to more than 460 ppm can cause acute poisoning in laboratory animals (Aldyreva, 1983). Toxic signs include irritation of mucous membranes, conjunctivitis, difficulty in breathing, pulmonary irritation, cramps, coordination disorders, narcosis, convulsions, and death due to respiratory center paralysis (Aldyreva, 1983). The main pathological findings are marked edema of brain and lungs, foci of epithelial necrosis in renal tubules, and hepatic dystrophy (Aldyreva, 1983).
Acute and Short-Term Exposures
Lethality
Smyth et al. (1962) reported a 4-h LC50 of EB at 4000 ppm for rats. Yant et al. (1930) reported EB vapor to be lethal at 5000 ppm to guinea pigs in 30 to 60 min; 10,000 ppm was lethal in ''a few minutes.''
Eye and Respiratory Irritation
Six volunteers exposed to EB at 1000 ppm experienced initial severe
eye irritation and lacrimation that decreased after 1 or 2 min to scarcely noticeable (Yant et al., 1930). The same six volunteers found EB at 2000 ppm almost intolerable initially but less irritating upon continued exposure, and one volunteer who remained in the chamber for 5 min found that the eye and throat irritation gradually disappeared, but vertigo developed (Yant et al., 1930). Exposure at 2000 ppm was accompanied by a feeling of constriction of the chest (Yant et al., 1930). Four volunteers exposed for 6 min to EB accumulating in a chamber to a concentration of 2000 ppm noticed moderate nasal irritation and moderate-to-strong eye irritation; all complained of dizziness upon leaving the chamber. Three volunteers entering a 5000-ppm EB atmosphere found it intolerably irritating to the eyes, nose, and throat (Yant et al., 1930).
Bardodej and Bardodejova (1961) investigated the metabolism of inhaled EB in humans. During 8-h exposures to EB vapors, they found that nine subjects exposed at 100 ppm did not complain of any problems, but 11 subjects exposed at 180 ppm complained of irritation of the respiratory tract and conjunctiva.
Yant et al. (1930) reported that groups of three to six guinea pigs exposed to EB at 1000 ppm showed signs of nasal irritation (rubbing nose) after 3 min and signs of eye irritation and lacrimation after 8 min of exposure. Comparing the responses of guinea pigs with those of humans reported by Yant et al. (1930), guinea pigs and humans appear to suffer similar signs at 1000 ppm, but the effects in guinea pigs might be delayed slightly more than those in humans.
CNS Effects
In the report by Bardodej and Bardodejova (1961) in which the metabolism of EB was studied in humans, nine subjects exposed at 100 ppm for 8 h did not complain of any problems, but toward the end of the exposure time at 180 ppm, 11 subjects complained of headaches and sleepiness. Because blood concentrations probably reached steady state early (~ 1 h) in the exposure, the kinetics suggest that headaches and sleepiness are due to metabolites of EB. Transient sensations of slight drunkenness also were reported.
In the Yant et al. (1930) study of groups of three to six guinea pigs
exposed to various concentrations of EB, a 480-min exposure at 1000 ppm produced only transient slight nasal irritation and slight lacrimation, which disappeared within 30 min, but no vertigo or ataxia. At 2000 ppm, guinea pigs showed signs of moderate eye and nasal irritation after 1 min, vertigo after 390 min, and ataxia after 480 min of exposure (Yant et al., 1930). At 5000 ppm, vertigo was seen after 26 min, ataxia after 30 min, apparent unconsciousness after 160 min, tremors of the extremities after 178 min, and shallow respiration after 215 min of exposure (Yant et al., 1930). At 10,000 ppm, vertigo and ataxia were seen after 4 to 10 min, unconsciousness after 18 min, tremors of the extremities after 5 to 18 min, rapid jerky respiration after 21 min, shallow respiration after 57 min, and very slow gasping respiration after 260 min (Yant et al., 1930).
Molnár et al. (1986) reported that narcosis occurred in groups of eight rats exposed for 4 h to EB at concentrations as low as 2180 ppm but not at 1500 ppm. Concentrations of 400 to 1500 ppm induced a moderate increase in group motility in rats exposed for 1, 2, 3, or 4 h.
Subchronic Exposures
Lethality
Cragg et al. (1989) reported a 4-d LC100 of EB at 2400 ppm for 6 h/d for rats and mice and an LC80 at 1200 ppm (four of five mice died). Chan (1992) reported a no-observed-adverse-effect level (NOAEL) at 1000 ppm for lethality in rats and mice for 13 w of exposure for 6 h/d, 5 d/w.
NOAELs for All Measured Effects
In rats and mice exposed 6 h/d, 5 d/w, for 4 w, the NOAEL for clinical chemistry, hematological, urological, and histological toxicity was reported to be 382 ppm; in similarly exposed rabbits, the NOAEL was 782 ppm (Cragg et al., 1989). In a 13-w National Toxicology Program (NTP) study of rats and mice, the highest exposure concentration tested (1000 ppm) was found to be a NOAEL for mutagenicity,
clastogenicity, and clinical or histopathological changes; effects observed were increases in weights of kidney, liver, and lung in rats and weights of livers in mice (Chan, 1992).
Elovaara et al. (1985) showed that exposure of rats to EB at concentrations up to 600 ppm for 6 h/d, 5 d/w, for up to 16 w increased the levels of liver proteins and increased the activity of selected liver and kidney enzymes (Elovaara et al., 1985). Glutathione levels remained constant in the liver and increased 30% in the kidney. Ultrastructural changes included slight proliferation of smooth endoplasmic reticulum, slight degranulation and splitting of the rough endoplasmic reticulum, and enlarged mitochondria. These data are not used to set acceptable concentrations (AC) for two reasons: first, all of the changes can be considered adaptive rather than adverse; second, the results could not be easily extrapolated to humans because metabolism of EB in humans has been shown to be qualitatively and quantitatively different from metabolism of EB in rats.
Respiratory Distress, CNS Effects
In a range-finding study, Cragg et al. (1989) exposed male mice, rats, and rabbits (4-5 animals per species) 6 h/d for 4 d to EB at 400, 1200, and 2400 ppm. At 1200 ppm, all rats showed marked clinical signs, including respiratory distress, salivation, prostration, and anogenital staining. The signs were similar in one surviving mouse of five mice exposed at 1200 ppm. No effects were noted in mice or rats exposed at 400 ppm. Rabbits showed no signs of respiratory distress or CNS effects at concentrations up to 2400 ppm (Cragg et al., 1989). In a recent NTP study (Chan, 1992), however, no adverse effects were seen in rats or mice exposed at 1000 ppm (the highest exposure concentration tested) for 6 h/d, 5 d/w, for 13 w (equivalent to ~ 16 d if exposure were continuous). Because those effects are less sensitive indicators than eye and respiratory irritation in humans, no ACs were set on the basis of those end points. ACs based on eye and respiratory irritation should protect against the more severe effects.
Neurochemical Changes
Exposure of rabbits to EB at 750 ppm for 12 h/d for 7 d caused a
marked depletion of striatal and tubero-infundibular dopamine (Mutti et al., 1988). Similar effects were achieved using mandelic and phenylglyoxylic acid, which are metabolites of EB. Experiments in vitro suggest that dopamine condenses nonenzymatically with reactive carbonylic groups of such α-keto acids, thus becoming ineffective as a neurotransmitter. Although those results might help explain the mechanism of EB's CNS effects, the neurochemical changes are not correlated to a functional deficit; thus, those results cannot be used to set an AC.
Chronic Exposures
Sleepiness, Fatigue, Eye, and Respiratory Irritation and Headache
Bardodej and Bardodejova (1970) measured exposures of industrial workers to EB, styrene, and α-methylstyrene. The study showed that some symptoms were reported by the workers if the exposure exceeded the threshold limit value of 100 ppm for EB. "The main complaints were fatigue, sleepiness, and headache, and mild irritation of the eyes and respiratory tract, particularly in the case of styrene." No detailed data were reported on the range of exposure concentrations, the duration of the exposures necessary to produce the reported effects, how EB concentrations in the workplace were determined, the numbers of individuals exposed, or whether any or all of the symptoms correlated with EB exposure concentrations.
Carcinogenicity
Bardodej and Cirek (1988) conducted biomonitoring of occupational EB exposures for 20 y (1964-1985) in approximately 200 Czechoslovakian production workers. No excess malignancies were seen in the workers for the last 10 y of the study. None of the exposed workers showed excess damage to hematopoiesis or liver tissue (Bardodej and Cirek, 1988). Although levels of urinary metabolites were monitored, atmospheric concentration measurements were not reported except to say that they were below the Czechoslovakian maximum allowable concentration (MAC) (46 ppm time-weighted average) after 1974.
Kidney and Liver Changes
Wolf et al. (1956) reported that a slight increase in liver weights was induced by inhalation of EB by male and female rats at concentrations of 400, 600, and 1250 ppm; by male rats at 2200 ppm; by guinea pigs at 600 ppm, but not at 400 or 1250 ppm; and by monkeys at 600 ppm, but not at 400 ppm, for 7-8 h/d, 5 d/w, for 6 mo (144 d at 2200 ppm). They also reported that slight "cloudy swelling" in the liver occurred in rats inhaling 1250 ppm for 186 d or 2200 ppm for 144 d (Wolf et al., 1956).
In the same study, rats, but not guinea pigs, rabbits, or monkeys, exposed to EB at concentrations of 400, 600, 1250, and 2200 ppm for 186 d (144 d at 2200 ppm) were found to have slightly increased kidney weights. Rats exposed at 1250 or 2200 ppm also exhibited slight "cloudy swelling" of the tubular epithelium of the kidney (Wolf et al., 1956).
Testicular Toxicity
Wolf et al. (1956) exposed 10 to 25 rats to EB at concentrations of 0, 400, 600, 1250, or 2200 ppm; 5 to 10 guinea pigs and 1 rabbit at 0, 400, 600, and 1250 ppm; and 1 to 2 monkeys at 0, 400 or 600 ppm for 7-8 h/d, 5 d/w, for 186 d. In the rabbit and monkey, 600 ppm induced slight histopathological changes described as degeneration of the germinal epithelium of the testes (concentration x time (C × T) = 900,000 ppm·h. No male monkeys were tested at exposure concentrations below 600 ppm, but no effects on the testes were seen in one male rabbit exposed at 400 ppm (C × T = 600,000 ppm·h). No testicular effects were seen in guinea pigs or rats at any of the tested exposure concentrations (up to 2200 ppm in rats). Although these results may be due to species differences in metabolism, it is not known whether the metabolism of humans is more similar to that of monkeys or rabbits than of rats.
The preceding results can be compared with the results of Cragg et al. (1989) who found no gross or microscopic changes in rabbit testes (in five rabbits per group) attributable to EB exposure at concentrations of 382, 782, or 1610 ppm for 6 h/d, 5 d/w, for 4 w (20 expo-
sures) (C × T = 46,000 to 190,000 ppm-h). Although no effects were seen, the total dose in these experiments was 4.7-fold below that used by Wolf et al. (1956), a dose that led to testicular toxicity.
Ivanov (1964, as cited by Cragg et al., 1989) exposed rabbits to EB at 2.3, 23, and 230 ppm for 4 h/d for 7 mo. Further protocol details were not provided. Assuming exposure for 7 d/w, the maximum dose for the rabbits (expressed as C × T) was 190,000 ppm h.
Genotoxicity
EB injected intraperitoneally into mice did not induce micronuclei in their bone-marrow cells (Mohtashamipur et al., 1985). EB was not mutagenic in Salmonella typhimurium strains TA100, TA1535, TA97, and TA98 when tested at doses up to 1000 µg per plate, which was toxic to the bacteria, in the presence or absence of S9 (Dean et al., 1985; Chan, 1992). EB was not mutagenic in strains TA1537 or TA1538, in Escherichia coli WP2 or WP2uvrA (Nestmann et al., 1980; Dean et al., 1985), or in Saccharomyces cerevisiae JD1 (Dean et al., 1985). EB exposure did not induce sister chromatid exchanges (SCEs) or chromosomal aberrations in CHO cells in the presence or absence of S9 (Chan, 1992), but it did induce a slight increase in SCEs in cultured human lymphocytes in the presence of S9 (Norppa and Vainio, 1983).
Reproductive and Developmental Toxicity
The offspring of rats inhaling EB continuously from gestation d 7 to 15 showed moderately increased rates of malformations at 552 ppm compared with controls and increased rates of skeletal retardation and dead or resorbed fetuses at 138, 276, and 552 ppm (Ungváry and Tátrai, 1985). The offspring of mice inhaling EB at 115 ppm continuously from gestation d 6 to 15 showed only moderately increased rates of malformations compared with controls (Ungváry and Tátrai, 1985). EB caused spontaneous abortion in rabbits exposed at 230 ppm continuously from gestation d 7 to 20 (Ungváry and Tátrai, 1985). No ACs are based on those effects.
Interactions with Other Chemicals
In rats exposed to EB at a concentration of 650 ppm, coexposure to ethyl acetate at 1000 or 4000 ppm lowered the concentrations of EB in the blood by 26% (Freundt et al., 1989). In rats exposed at 180 ppm, coexposure to ethanol at 20 mmol/kg increased the blood concentration of EB by 139%.
TABLE 8-2 Toxicity Summary
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
101 |
8 h |
Human (n = 9) |
NOAEL for eye and respiratory irritation, sleepiness, headache, and transient sensations of slight drunkenness |
Bardodej and Bardodejova, 1970 |
|
150 |
4 h |
Human (n = 4) |
No toxic effects reported; study examined urinary metabolites of EB |
Engström et al., 1984 |
|
184 |
8 h |
Human (n = 11) |
LOAEL for eye and respiratory irritation; transient sensations of slight drunkenness and, toward the end of exposure, sleepiness and headache |
Bardodej and Bardodejova, 1970 |
|
200 |
NS |
Human |
Transient eye irritation |
Gerarde, 1963 |
|
1000 |
A few min |
Human (n = 6) |
Initial severe eye irritation and lacrimation, decreasing to very slight within 2 min |
Yant et al., 1930 |
|
2000 |
5 min |
Human (n = 1) |
Initial extreme eye, nose, and throat irritation and feeling of chest constriction, gradually disappearing with development of vertigo |
Yant et al., 1930 |
|
5000 |
A few breaths |
Human (n = 3) |
Intolerable irritation of eyes and nose |
Yant et al., 1930 |
|
115 |
6 h/d, 5 d/w |
Mouse |
Moderately increased fetal malformation |
Ungváry and Tátrai, 1985 |
|
138 |
6 h/d, 5 d/w |
Rat |
Increased "skeletal retardation," dead or resorbed fetuses |
Ungváry and Tátrai, 1985 |
|
230 |
6 h/d, 5 d/w |
Rabbit |
Spontaneous abortion |
Ungváry and Tátrai, 1985 |
|
382 |
6 h/d, 5 d/w, 4 w |
Rat, mouse |
NOAEL |
Cragg et al., 1989 |
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
400 |
6 h/d, 4 d |
Rat, mouse |
NOAEL for salivation, prostration, anogenital staining |
Cragg et al., 1989 |
|
552 |
6 h/d, 5 d/w |
Rat |
Increased rate of fetal malformations |
Ungváry and Tátrai, 1985 |
|
600 |
7-8 h/d, 5 d/w, 6 mo |
Rat |
Clouding and swelling of kidney tubular epithelium and hepatocytes |
Wolf et al., 1956 |
|
600 |
6 h/d, 5 d/w, 16 w |
Rat |
Increased levels of proteins and increased liver and kidney enzyme activities |
Elovaara et al., 1985 |
|
600 |
186 d |
Guinea pig |
No testicular histopathological changes |
Wolf et al., 1956 |
|
600 |
186 d |
Monkey, rabbit |
Slight testicular histopathological changes |
Wolf et al., 1956 |
|
750 |
12 h/d, 7 d |
Rabbit |
Marked depletion of striatal and tuberoinfundibular dopamine |
Mutti et al., 1988 |
|
782 |
6 h/d, 5 d/w, 4 w |
Rabbit |
NOAEL |
Cragg et al., 1989 |
|
1000 |
6 h/d, 5 d/w, 13 w |
Rat, mouse |
NOAEL for histopathological and clinical effects |
Chan, 1992 |
|
1000 |
3 min |
Guinea pig |
Nasal irritation-rubbing nose |
Yant et al., 1930 |
|
1000 |
8 min |
Guinea pig |
Eye irritation-squinting and lacrimation |
Yant et al., 1930 |
|
1000 |
480 min |
Guinea pig |
Slight transient nasal irritation and slight lacrimation that disappeared within 30 min |
Yant et al., 1930 |
|
1200 |
6 h/d, 4 d |
Rat |
Salivation, prostration, anogenital staining |
Cragg et al., 1989 |
|
1200 |
6 h/d, 4 d |
Mouse |
LC80 (4 of 5 mice died) |
Cragg et al., 1989 |
|
1250 |
214 d |
Rat |
No testicular histopathological changes |
Wolf et al., 1956 |
|
1500 |
4 h |
Rat |
NOAEL for anesthesia |
Molnár et al., 1986 |
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
1610 |
6 h/d, 5 d/w |
Rabbit |
NOAEL for testicular toxicity |
Cragg et al., 1989 |
|
2000 |
1 min |
Guinea pig (n = 3 to 6) |
Eye irritation—squinting and lacrimation |
Yant et al., 1930 |
|
2000 |
1 min |
Guinea pig (n = 3 to 6) |
Nasal irritation—rubbing nose |
Yant et al., 1930 |
|
2000 |
390 min |
Guinea pig (n = 3 to 6) |
Vertigo—unsteadiness |
Yant et al., 1930 |
|
2000 |
480 min |
Guinea pig (n = 3 to 6) |
Static and motor ataxia |
Yant et al., 1930 |
|
2180 |
4h |
Rat |
LOAEL for anesthesia |
Molnár et al., 1986 |
|
2200 |
144 d |
Rat |
No testicular histopathological changes |
Wolf et al., 1956 |
|
2400 |
6 h/d, 4 d |
Rabbit |
NOAEL for clinical signs |
Cragg et al., 1989 |
|
2400 |
6 h/d, 4 d |
Mouse, rat |
LC100 |
Cragg et al., 1989 |
|
4000 |
4 h |
Rat |
LC50 |
Smyth et al., 1962 |
|
5000 |
26 min |
Guinea pig (n = 3 to 6) |
Vertigo |
Yant et al., 1930 |
|
5000 |
30 min |
Guinea pig (n = 3 to 6) |
Ataxia |
Yant et al., 1930 |
|
5000 |
160 min |
Guinea pig (n = 3 to 6) |
Apparent unconsciousness |
Yant et al., 1930 |
|
5000 |
178 min |
Guinea pig (n = 3 to 6) |
Tremors of extremities |
Yant et al., 1930 |
|
5000 |
215 min |
Guinea pig (n = 3 to 6) |
Shallow respiration |
Yant et al., 1930 |
|
8000 |
4 h |
Rat |
LC50 |
Smyth et al., 1962 |
|
10,000 |
4-10 min |
Guinea pig |
Vertigo and ataxia |
Yant et al., 1930 |
|
10,000 |
18 min |
Guinea pig |
Apparent unconsciousness |
Yant et al., 1930 |
|
10,000 |
5-18 min |
Guinea pig |
Tremors of extremities |
Yant et al., 1930 |
|
10,000 |
21 min |
Guinea pig |
Rapid jerky respiration |
Yant et al., 1930 |
|
10,000 |
57 min |
Guinea pig |
Shallow respiration |
Yant et al., 1930 |
|
10,000 |
260 min |
Guinea pig |
Very slow gasping type of respiration |
Yant et al., 1930 |
|
NS, not specified. |
||||
TABLE 8-3 Exposure Limits Set or Recommended by Other Organizations
|
Agency or Organization |
Exposure Limit, ppm |
Reference |
|
ACGIH's TLV (1985) |
100 |
ACGIH, 1991b |
|
ACGIH's STEL |
125 |
ACGIH, 1991b |
|
NIOSH's REL |
100 (TWA) |
ACGIH, 1991b |
|
OSHA's PEL |
100 |
ACGIH, 1991b |
|
TLV, Threshold Limit Value; STEL, short-term exposure limit; REL, recommended exposure limit; TWA, time-weighted average; PEL, permissible exposure limit. |
||
TABLE 8-4 Spacecraft Maximum Allowable Concentrations
|
Exposure Durationa |
Concentration, ppm |
Concentration, mg/m3 |
Target Toxicity |
|
1 h |
180 |
780 |
Eye and respiratory irritation, headache, sleepiness |
|
24 h |
60 |
260 |
Headache, sleepiness |
|
7 db |
30 |
130 |
Eye and respiratory irritation, testicular toxicity |
|
30 d |
30 |
130 |
Eye and respiratory irritation, testicular toxicity |
|
180 d |
12 |
50 |
Testicular toxicity |
|
a Ceiling limits. b The former 7-d SMAC is 20 ppm (86.8 mg/m3). |
|||
RATIONALE FOR ACCEPTABLE CONCENTRATIONS
The evaluation of and setting priorities for the toxicological effects listed in Table 8-2 took into account various factors. For EB, rodent toxicity was not heavily weighted because rat metabolism of EB has
been shown to yield products qualitatively and quantitatively different from those of humans. The human metabolic products of EB, however, caused neurochemical effects in rabbits similar to those caused by EB. Because differences in metabolism in humans and in lower primates have not been reported, data on toxicity in monkeys could not be discounted. Fetal toxicity was not considered in setting ACs because the National Aeronautics and Space Administration will not knowingly send a pregnant astronaut into space.
Of the toxic effects attributed to EB, ACs were not set for the following: lethality (adequate protection would be achieved by ACs for less severe effects); genotoxicity (minimal effect demonstrated; quantitation of dose-response relationship is not currently possible); respiratory distress (adequate protection would be achieved by ACs for less severe effects); neurochemical changes (no correlation to a functional deficit); developmental toxicity (not applicable to astronauts); carcinogenicity (none observed); and kidney and liver changes (demonstrated effects at high exposure concentrations were not judged to be adverse). ACs were set for the remaining effects: eye and respiratory irritation, testicular toxicity, and sleepiness and headache.
Calculation of the highest AC for each major end point and exposure duration is documented below. The resulting ACs for the various end points are listed in Table 8-5. SMAC values set at each exposure duration are based on the end point that yielded the lowest AC at that duration, following the guidelines of the National Research Council (NRC, 1992).
Eye and Respiratory Irritation
Bardodej and Bardodejova (1961) reported that 100 ppm was an 8-h NOAEL for 9 volunteers and 180 ppm was a lowest-observed-adverse-effect level (LOAEL) for 11 volunteers for eye and respiratory irritation and, toward the end of the 8-h exposure, for sleepiness and headache. Because irritation of the eye and respiratory system is generally independent of exposure duration and sleepiness and headache might increase in intensity with longer exposures, these end points will be dealt with separately. Mild eye and respiratory irritation would be acceptable
for emergency situations and the intensity should not increase with longer exposures; therefore, the 1-h and 24-h ACs are set equal to the 8-h LOAEL:
1-h and 24-h ACs = 8-h LOAEL = 180 ppm.
For exposures longer than 24 h, no irritation is acceptable. Thus, these ACs are based on the 8-h NOAEL and adjusted for the low number of subjects tested:
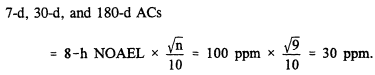
Testicular Toxicity
In a study by Wolf et al. (1956), 10-25 rats per group were exposed to EB at 2200, 1250, 600, or 400 ppm, 5-10 guinea pigs per group were exposed at 1250, 600, or 400 ppm, one male rabbit per exposure concentration was exposed at 400, 600, and 1250 ppm, and one male monkey per exposure concentration was exposed at 400 and 600 ppm for 7-8 h/d, 5 d/w, for 186 d. In the rabbit and monkey, EB at 600 ppm for 7-8 h/d, 5 d/w, for 186 d induced slight histopathological changes described as degeneration of the germinal epithelium of the testes. No testicular toxicity was seen in the one male rabbit tested at 400 ppm. No testicular effects were seen in guinea pigs or in rats at any of the tested exposure concentrations (up to 2200 ppm in rats). No other chronic inhalation studies on monkeys have been found that could be used to confirm or discredit these findings. One Russian study (Ivanov, 1964) cited by Cragg et al. (1989) exposed rabbits to EB at 2.3, 23, and 230 ppm for 4 h/d for 7 mo. That study reported hematological changes, changes in blood protein and cholinesterase levels, and dystrophic changes in kidneys and liver but did not mention effects on the testes. It is not known if the testes were examined. Thus, some evidence points to toxicity of EB to the testes, but that toxicity certainly has not been firmly established. Nevertheless, because an effect was
reported in a primate, and at the risk of erring on the conservative side, ACs were calculated by dividing the 400-ppm NOAEL by a factor of 10 for possible species differences. Haber's rule was used to extrapolate from 54 d (i.e., where 7 h/d ÷ 24 h/d × 186 d = 54 d) but was not used to increase ACs for exposures shorter than 54 d.
180-d AC = NOAEL/10 (species) × (54 d/180 d) = 400 ppm/10 × 0.3 = 12 ppm.
30-d and 7-d AC = 400 ppm/10 (species) = 40 ppm.
No ACs were set for 1 h and 24 h because it would require a time extrapolation of greater than 10-fold.
Sleepiness and Headache
The AC for 1-h and 24-h exposures can be set on the basis of the 180-ppm 8-h LOAEL for sleepiness and headache in human volunteers. Although narcosis is not acceptable, slight sleepiness, headache, and fatigue would be acceptable for emergency 1-h and 24-h exposures. Thus, the ACs for a 1-h exposure is set equal to the 8-h LOAEL (which is a NOAEL for 1 h), and the AC for a 24-h exposure is reduced threefold to ensure that the slight headaches do not become severe.
1-h AC = 8-h NOAEL = 180 ppm.
24-h AC = 8-h LOAEL/3 = 180/3 = 60 ppm.
ACs were not set for exposure periods of 7 d or more because that would require more than a 10-fold time extrapolation from the data.
Spaceflight Considerations
None of the toxic effects known to be induced by exposure to EB would be affected by launch, microgravity, or re-entry; thus, no spaceflight factor was used.
RECOMMENDATIONS
-
Additional research is needed to confirm or disprove the reported testicular toxicity of long-term exposure to EB in rabbits and possibly in monkeys and the potential of EB to cause testicular toxicity in humans.
-
Testing of a large number (near 100) of volunteers for irritation at the 100-ppm exposure concentration for 4 h or more might permit the ACs for 7 d and 30 d to be increased from 30 to 100 ppm.
-
Testing of volunteers for more than 8 h at several concentrations is needed to examine the effect of long (greater than 24 h) continuous exposures on headache and sleepiness in humans. Performance testing would be a necessary part of this experiment.
TABLE 8-5 Acceptable Concentrations
|
|
|
Uncertainty Factors |
||||||||
|
|
|
To NOAEL |
|
|
Spaceflight |
Acceptable Concentrations, ppm |
||||
|
Effect, Data, Reference |
Species |
Species |
Time |
1 h |
24 h |
7 d |
30 d |
180 d |
||
|
Eye and respiratory irritation |
|
|||||||||
|
LOAEL, 184 ppm, 8 h (Bardodej and Bardodejova, 1970) |
Human (n = 11) |
1 |
1 |
1 |
1 |
180 |
180 |
- |
- |
- |
|
NOAEL, 101 ppm, 8 h (Bardodej and Bardodejova, 1970) |
Human (n = 9) |
0.3 = √(n/100) |
1 |
1 |
1 |
- |
- |
30 |
30 |
30 |
|
Testicular toxicity |
|
|||||||||
|
NOAEL, 400 ppm, 7-8 h/d, 5 d/w, 186 d (Wolf et al., 1956) |
Rabbit, monkey |
1 |
10 |
HR |
1 |
- |
- |
40 |
40 |
12 |
|
Sleepiness and headache |
|
|||||||||
|
8-h LOAEL, 184 ppm (Bardodej and Bardodejova, 1970) |
Human (n = 11) |
1 |
1 |
1 or HR |
1 |
180 |
60 |
- |
- |
- |
|
SMACs |
|
|
|
|
|
180 |
60 |
30 |
30 |
12 |
|
—, Data not considered applicable to the exposure time; HR, Haber's rule. |
||||||||||
REFERENCES
ACGIH. 1991a. Documentation of the Threshold Limit Values and Biological Exposure Indices, 6th Ed. American Conference of Governmental Industrial Hygienists, Cincinnati, Ohio.
ACGIH. 1991b. P. 50 in Guide to Occupational Exposure Values—1991. American Conference of Governmental Industrial Hygienists, Cincinnati, Ohio.
Aldyreva, M. V. 1983. Styrene and ethylbenzene. Pp. 2114-2115 in Encyclopedia of Occupational Health and Safety, Vol. 2, L. Parmeggiani, ed. International Labour Organization, Geneva, Switzerland.
Åstrand, I., J. Engstrom, and P. Ovrum. 1978. Exposure to xylene and ethylbenzene. I. Uptake, distribution and elimination in man. Scand. J. Work Environ. Health 4:185.
Bardodej, Z., and E. Bardodejova. 1961. Usefulness and application of exposure tests. Cesk. Hyg. 6:537-545.
Bardodej, Z., and E. Bardodejova. 1970. Biotransformation of ethylbenzene, styrene, and alpha-methylstyrene in man. Am. Ind. Hyg. Assoc. J. 31:206-209.
Bardodej, Z., and A. Cirek. 1988. Long-term study on workers occupationally exposed to ethylbenzene. J. Hyg. Epidemiol. Microbiol. Immunol. 32:1-5.
Chan, P. 1992. NTP Report on the Toxicity Studies of Ethylbenzene in F344/N Rats and B6C3F1 Mice (Inhalation Studies). National Institutes of Health, National Toxicology Program, Research Triangle Park, N.C.
Chin, B. H., L. J. Sullivan, S. J. Kozbelt, and L. J. Calisti. 1978. Excretion and urinary metabolic profiles of ethylbenzene, ethylcyclohexane, and methylethylbenzene in rats and dogs. Toxicol. Appl. Pharmacol. 45:240.
Cragg, S. T., E. A. Clarke, I. W. Daly, R.R. Miller, J. B. Terrill, and R. E. Ouellette. 1989. Subchronic inhalation toxicity of ethylbenzene in mice, rats, and rabbits. Fundam. Appl. Toxicol. 13:399408.
Dean, B. J., T. M. Brooks, G. H. Walker, and D. H. Hutson. 1985. Genetic toxicity testing of 41 industrial chemicals. Mutat. Res. 153:57-77.
Elovaara, E., K. Engström, J. Nickels, A. Aitio, and H. Vainio. 1985. Biochemical and morphological effects of long-term inhalation
exposure of rats to ethylbenzene. Xenobiotica 15:299-308.
Engström, K. M. 1984. Metabolism of inhaled ethylbenzene in rats. Scand. J. Work Environ. Health 10:83-87.
Engström, K., V. Riihimäki, and A. Lies. 1984. Urinary disposition of ethylbenzene and m-xylene in man following separate and combined exposure. Int. Arch. Occup. Environ. Health 54:355-363.
Engström, K., E. Elovaara, and A. Aitio. 1985. Metabolism of ethylbenzene in the rat during long-term intermittent inhalation exposure. Xenobiotica 15:281-286.
Freundt, K. J., K. G. Römer, and R. J. Federsel. 1989. Decrease of inhaled toluene, ethyl benzene, m-xylene, or mesitylene in rat blood after combined exposure to ethyl acetate. Bull. Environ. Contam. Toxicol. 42:495-498.
Gerarde, H. W. 1963. The aromatic hydrocarbons. Pp. 1219-1240 in Industrial Hygiene and Toxicology, 2nd Revised Ed., F. A. Patty, ed., Vol. 2, Toxicology, D. W. Fassett and D. D. Irish, eds. New York: Wiley-Interscience.
Gromiec, J. P., and J. K. Piotrowski. 1984. Urinary mandelic acid as an exposure test for ethylbenzene. Int. Arch. Occup. Environ. Health 55:61.
Ivanov, S. V. 1964. Toxicology and hygienic rating of ethylbenzene content in the atmosphere of industrial areas. Gig. Tr. Prof. Zabol. 8:9-14.
Liebich, H. M., W. Bertsch, A. Zlatkis, and H. J. Schneider. 1973. Volatile organic components in the Skylab 4 spacecraft atmosphere. Aviat. Space Environ. Med. 46:1002-1007.
Min, D. B. S., K. Ina, R. J. Peterson, and S. S. Chang. 1979. Preliminary identification of volatile flavor compounds in the neutral fraction of roast beef. J. Food Sci. 44:639-642.
Mohtashamipur, E., K. Norpoth, U. Woelke, and P. Huber. 1985. Effects of ethylbenzene, toluene, and xylene on the induction of micronuclei in bone marrow polychromatic erythrocytes of mice. Arch. Toxicol. 58:106-109.
Molnár, J., K. A. Paksy, and M. Náray. 1986. Changes in the rat's motor behavior during 4-hr inhalation exposure to prenarcotic concentrations of benzene and its derivatives. Acta Physiol. Hung. 67:349-353.
Mutti, A., M. Falzoi, A. Romanelli, M. C. Bocchi, C. Ferroni, and I. Franchini. 1988. Brain dopamine as a target for solvent toxicity:
Effects of some monocyclic aromatic hydrocarbons. Toxicology 49:77-82.
Nestmann, E. R., E. G.-H. Lee, T. I. Matula, G. R. Douglas, and J. C. Mueller. 1980. Mutagenicity of constituents identified in pulp and paper mill effluents using the Salmonella/mammalian microsome assay. Mutat. Res. 79:203-212.
Norppa, H., and H. Vainio. 1983. Induction of sister chromatid exchanges by styrene analogues in cultured human lymphocytes. Mutat. Res. 116:379-387.
NRC. 1992. Guidelines for Developing Spacecraft Maximum Allowable Concentrations for Space Station Contaminants. Washington, D.C.: National Academy Press.
Riihimäki, V., and K. Savolainen 1980. Human exposure to m-xylene. Kinetics and acute effects on the central nervous system. Ann. Occup. Hyg. 23:411-122.
Rippstein, W. J., and M. Coleman. 1983. Toxicological evaluation of the Columbia spacecraft. Aviat. Space Environ. Med. 54(Suppl. 1):S60-S67.
Sandmeyer, E. E. 1981. Ethylbenzenes. Pp. 3303-3304 in Patty's Industrial Hygiene and Toxicology, 3rd Revised Ed., Vol. 2B, Toxicology, G. D. Clayton and F. E. Clayton, eds. New York: Wiley-Interscience.
Sato, A., and T. Nakajima. 1987. Pharmacokinetics of organic solvent vapors in relation to their toxicity. Scand. J. Work Environ. Health 13:81-93.
Smyth, H. F., Jr., C. A. Carpenter, C. S. Weil, U. C. Pozzani, and J. A. Striegel. 1962. Range finding toxicity data, List VI. Am. Ind. Hyg. Assoc. J. 23:95.
Ungváry, G., and E. Tátrai 1985. On the embryotoxic effects of benzene and its alkyl derivatives in mice, rats and rabbits. Arch. Toxicol. Suppl. 8:425-430.
Von Burg, R. 1992. Toxicology update: Ethylbenzene. J. Appl. Toxicol. 12:69-71.
Windholz, M. 1976. Ethylbenzene. P. 3694 in Merck Index. Rahway, N.J.: Merck & Co.
Wolf, M. S. 1976. Evidence for existence in human tissues of monomers for plastics and rubber manufacture. Environ. Health Perspect. 17:183.
Wolf, M. A., V. K. Rowe, R. L. McCollister, and F. Oyen. 1956.
























