B12 Xylene
Hector D. Garcia, Ph.D.
Johnson Space Center Toxicology Group
Biomedical Operations and Research Branch
National Aeronautics and Space Administration
Houston, Texas
PHYSICAL AND CHEMICAL PROPERTIES
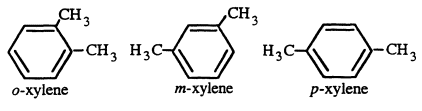
Xylene is a clear, colorless, flammable liquid at room temperature with an ''aromatic'' odor similar to that of benzene and toluene (Sax, 1984). The three isomers of xylene are ortho-(1, 2-dimethylbenzene), meta-(1, 3-dimethylbenzene), and para-(1, 4-dimethylbenzene). Commercial xylene is a mixture of the three forms, meta- (m-) being the major component and ortho- (o-) and para- (p-) present at minor concentrations. The commercial mixture often contains substantial amounts of ethylbenzene. The three isomers of xylene are very similar in physical, chemical, and toxicological properties. The odor threshold for vapors of mixed xylenes was determined by a panel of six people to be on the order of 1 ppm (Carpenter et al., 1975).
|
Synonyms: |
Dimethylbenzene, xylol |
|||
|
Formula: |
C8H10; C6H4(CH3)2 |
|||
|
|
ortho- |
meta- |
para- |
Mixture |
|
CAS number: |
95476 |
108383 |
106423 |
1330207 |
|
Molecular weight: |
106.18 |
106.18 |
106.18 |
106.18 |
|
Boiling point: |
144.4°C |
139°C |
138.3°C |
138.5°C |
|
Melting point: |
-25.2°C |
-47.9°C |
13-14°C |
|
|
Lower explosive limit: |
1.0% |
1.1% |
1.1% |
|
|
Upper explosive limit: |
6.0% |
7.0% |
7.0% |
|
|
Autoignition temperature: |
|
563°C |
530°C |
|
|
Flash point (closed cup): |
17°C |
25°C |
25°C |
37.8°C |
|
Specific gravity (20/4°C): |
0.880 |
0.864 |
0.8611 |
0.864 |
|
Vapor pressure in mmHg at 28°C: |
10 |
10 |
6.72 |
|
|
Vapor density: |
3.66 |
3.66 |
|
|
|
Solubility: |
Insoluble in water; miscible in alcohol, ether, and some organic solvents |
|||
|
Conversion factors at 25°C, 1 atm: |
1 ppm = 4.34 mg/m3 1 mg/m3 = 0.23 ppm |
|||
Commercial xylene is synthesized from petroleum and obtained from the destructive distillation of coal tar. Both products are mixtures of three isomers. Commercial xylene frequently contains ethylbenzene and traces of toluene, phenol, thiophene, pyridine, and small amounts of nonaromatic hydrocarbons.
Xylene is used as a solvent, especially in the paint, printing, rubber, and leather industries, and in the manufacture of mirrors. It is used as a cleaning agent and degreaser and as a constituent of aviation and automotive fuels (gasoline). It is used in the chemical industry as a starting material and intermediate for the manufacture of phthalic and terephthalic acids. Those acids are used in the manufacture of plastics and resins, including textile fabrics. Xylenes are used in the coating and impregnation of fabric and paper. Other uses for xylene include dyes, inks, adhesives, cements, and thinners.
Xylene is not used in spacecraft during flight. Xylenes are occasion-
ally found, however, in the spacecraft atmosphere at concentrations up to 0.48 ppm, probably due to off-gassing from nonmetallic materials (Liebich et al., 1975; Huntoon, 1987).
TOXICOKINETICS AND METABOLISM
Humans retain an average of 63.6 ± 4.2% of inhaled xylene, independent of the atmospheric concentration within the range of 46 to 200 ppm (Sedivec and Flek, 1976). Retention is also independent of the duration of exposure for exposure times of 15 min to 8 h in a single day (Sedivec and Flek, 1976) or after 5 d of 6 h/d exposure (Riihimäki et al., 1979a, b). The percentage of retention varies only slightly between individuals and between the three isomers of xylene (Sedivec and Flek, 1976). The values for retention after 15 min of exposure were practically the same as the values between the first and third hours and those between the fifth and seventh hours. Even in repeated experiments performed after a few days or even weeks, retention in a given individual was similar to retention seen at 15 min (Sedivec and Flek, 1976; Riihimäki et al., 1979a, b). Retention might decrease somewhat as ventilation rates increase (Sedivec and Flek, 1976; Riihimäki et al., 1979a, b).
Measurement of xylene in exhaled air after exposure to 46 or 92 ppm for 8 h continuously revealed that a total of about 4% to 6% of the retained xylene is eventually excreted unchanged through the lungs with three-phase elimination kinetics (Sedivec and Flek, 1976; Riihimäki et al., 1979a, b). A trace amount of xylene can be observed in the exhaled air even 48 h after exposure to xylene at 46 or 92 ppm (Sedivec and Flek, 1976). In humans exposed at 100 ppm, the half-lives for elimination of xylene were 0.8 h for the initial phase, 7.7 h for the intermediate phase, and 17.7 h for the slowest phase (Low et al., 1989).
Only trace amounts (0.0047% of body burden) of unchanged xylene appear in the urine after exposure at concentrations of 46 or 92 ppm (Sedivec and Flek, 1976). At retained body burdens of 0.019 g/kg in humans, essentially all the conjugated xylene in the urine was in the form of glycine-bound toluic acid (methylhippuric acid) (Sedivec and Flek, 1976; Riihimäki et al., 1979a, b). In rabbits exposed at doses of
up to 0.6 g/kg of body weight, a portion of the xylene in the urine was conjugated to glucuronic acid, presumably because of overload of the glycine-conjugating system (Bray et al., 1949).
In another study in which 10 human volunteers were exposed to m-xylene at 23, 69, and 138 ppm, results contradicted those reported above. In that study, m-xylene vapor retained in the lungs tended to decrease at the end of the 8 h exposure, and the amount of the decrease was dependent on the inhaled concentration (Senczuk and Orlowski, 1978). At 139 ppm, the initial retention was 78%, and the retention at the end of the exposure was 65 %; at 69 ppm, the values were 83 % and 67%; and at 23 ppm, the values were 87% and 84% (Senczuk and Orlowski, 1978). The 8-h exposures were interrupted by two 0.5-h breaks-at 2.5 h and 5.5 h. Metabolism of m-xylene to m-methylhippuric acid was found to be very rapid compared with the rate of excretion of the m-methylhippuric acid into the urine (Senczuk and Orlowski, 1978).
Under steady-state conditions and conditions of increased xylene absorption (physical exercise), the blood xylene concentration in humans was found to be related directly to the rate of xylene uptake (Riihimäki and Savolainen, 1980). Acute effects on the central nervous system (CNS) were correlated with blood xylene concentrations and with a rapid rise in blood xylene concentrations. Thus, exposure to a fluctuating xylene concentration with high uptake rates during the peak concentration caused more pronounced effects than a corresponding exposure to a constant concentration. Physical exercise markedly raised the uptake rates and enhanced the effects (Riihimäki et al., 1979a; Riihimäki and Savolainen, 1980). Kinetic data in humans showed that well-perfused tissues, such as the brain, should reach xylene equilibrium within minutes and muscles within a few hours. Calculation of partition coefficients suggests that brain xylene concentrations should closely follow blood xylene concentrations (Riihimäki and Savolainen, 1980). In adipose tissue, repeated daily exposure results in xylene accumulation over a period of a few weeks; with continuous exposure, steady-state concentrations are reached after a few days (Riihimäki and Savolainen, 1980). Concentrations in gluteal subcutaneous fat reached 10-fold higher concentrations than those in blood after 6 exposure days (5-d exposure, weekend with no exposure, and 1-d exposure). Postexposure excretion of xylene from most tissues initially takes place rapidly. Elimination half-times were reported to be about 0.5 to 1.0 h during the first 2 to 3
h after exposure (Riihimäki and Savolainen, 1980). Consequently, acute xylene effects are probably short-lived after the end of the exposure. Because xylene stored in adipose tissue has an elimination half-life of about 58 h for subcutaneous fat, xylene mobilization results in only low blood concentrations (Riihimäki and Savolainen, 1980).
In mice, whole-body autoradiography reveals that metabolites of inhaled xylene (methylhippuric acid) accumulate in the nasal mucosa. These metabolites also are transported, probably by axonal flow, into the olfactory bulb of the brain (Ghantous et al., 1990). These localizations are interesting in view of case reports of a deteriorated sense of smell after occupational exposure to solvents (Emmet, 1976).
No reports have been found that document synergistic effects between xylene and any other agents.
TOXICITY SUMMARY
Early reports on the toxicity of xylene in humans are often difficult to assess because of impurities (including benzene) present in commercial xylene. In fact, the term "xylene" was often used as a general term for various solvents, including "pure" toluene and "pure" benzene as well as mixtures of various solvents.
Liquid xylene is a skin irritant causing erythema and dryness due to its fat solubilizing action. Xylene vapor is anaesthetic in atmospheric concentrations exceeding about 5000 ppm (Carpenter et al., 1975). At lower concentrations, probably in the range of a few hundred parts per million, workers sometimes have experienced a variety of subjective symptoms. Fatigue, drowsiness, headache, dizziness, dyspepsia, and nausea have been reported during repetitive daily exposure to unmeasured atmospheric concentrations (Browning, 1965).
Acute Exposures
Seven human volunteers reported minimal degrees of subjective responses (eye, nose, or throat irritation; dizziness or light-headedness; and tearing) to a 15-min inhalation exposure to mixed xylenes at 230 ppm (Carpenter et al., 1975). One of the seven experienced mild throat discomfort during the first minute and again during the seventh minute
of inhalation at 106 ppm, as well as during the first minute of inhalation at 230 ppm, but no discomfort at 460 ppm. Four of the seven volunteers reported either intermittent or continuous mild eye irritation while inhaling 690 ppm (Carpenter et al., 1975).
The cardiac toxicity of a 2-min inhalation of m-xylene vapors was examined in dogs. A concentration of m-xylene at 0.35% (3500 ppm) was the threshold required to induce a decrease in peak dp/dt (the first derivative of left-ventricular pressure) (Kobayashi et al., 1989).
In mice, tests of operant and motor performance were performed after 30-min exposures to xylenes at 500 to 7000 ppm. The results suggested a CNS stimulant action at concentrations of 1400 to 2400 ppm, 1400 ppm being the lowest significantly effective concentration for operant performance (lever pressing) for each xylene isomer (Moser et al., 1985). Minor differences between isomers were seen, however, in the concentrations that produced half-maximal decreases in the response rate (m-xylene, 6176 ppm; p-xylene, 5611 ppm; o-xylene, 5179 ppm) (Moser et al., 1985). Differences between isomers also were seen in their minimally effective concentrations (m-xylene at 3000 ppm, p-xylene at 2000 ppm, and o-xylene at 3000 ppm) and in the concentrations that produced half maximal decreases in motor performance (m-xylene at 3790 ppm, p-xylene at 2676 ppm, and o-xylene at 3640 ppm) (Moser et al., 1985). Thus, the differences between isomers were less than twofold in all cases.
In male rats, slightly increased group motility (movement about the exposure chamber) was observed during a 4-h exposure to m-xylene at concentrations between 130 and 1500 ppm; narcosis was observed at 2100 ppm (Molnár et al., 1986).
The LC50 for rats in a 4-h inhalation period was 29 mg/L (6700 ppm) (Carpenter et al., 1975). Cats succumbed within 2 h at 41 mg/L (9500 ppm) with signs suggesting CNS effects: the sequential development of salivation, ataxia, tonic and clonic spasms, anesthesia, and death (Carpenter et al., 1975). The only lesions reported that were considered significant in any of the acute inhalation studies were atelectasis, hemorrhage, and interlobular edema of the lung in rats that died after inhaling mixed xylenes at 9900 ppm for several hours (Carpenter et al., 1975). In rats, the oral LD50 was 4.0 g/kg.
In humans, a single report (Morley et al., 1970) of a lethality was found that appeared to be attributable to xylene exposure. Three painters who were working in a confined space on a ship were found uncon-
scious after at least 15 h of exposure to paint fumes consisting of > 90% xylene with traces of toluene. One painter died; the other two recovered. Lack of oxygen was not considered a factor in the death. Histological examination of the dead man's lungs showed severe congestion with focal intra-alveolar hemorrhage and acute pulmonary edema. The liver was congested with swelling and vacuolation of many cells. The brain showed microscopic petechial hemorrhages in both gray and white matter and hemorrhages in Virchow-Robin spaces. There was evidence of neuronal damage with swelling and loss of Nissl substance. The probable xylene concentration in the confined space in which they were working was estimated to be 10,000 ppm. One of the survivors remembered nothing after beginning work about 10:30 a.m. The other survivor remembered walking home (about 3 miles) for lunch and returning to work about 1:15 p.m., but he remembered nothing after returning until recovering consciousness in the hospital. The three men were discovered by rescuers at 5:00 a.m. the next morning. The two survivors completely recovered within a few days, except for persistent amnesia for events that occurred during the 24 h before their collapse.
Changes in human visual-and auditory-evoked potentials were seen in nine male volunteers exposed by inhalation to m-xylene at either a stable 200 ppm or fluctuating 135 to 400 ppm for 3 h in the morning and 40 min in the afternoon with a 40-min lunch break (Seppäläinen et al., 1989). The subjects were either sedentary or exercised at 100 watts for 10 min at the beginning of the exposure. The results suggest some activation of arousal level (decrease in the latency of the evoked potentials) of the subjects after the most intensive exposure situations (i.e., with physical exercise during exposure) (Seppäläinen et al., 1989). The magnitude of these changes was minor compared with the effects seen in general anesthesia or in long-term occupational exposure to hexane (Seppäläinen et al., 1989).
In a complicated study, inhalation of m-xylene at 100 to 400 ppm was reported to cause impairment of body balance and increase of reaction times in eight human volunteers (Riihimäki and Savolainen, 1980). Testing was done with constant or fluctuating concentrations of xylene and with subjects either sedentary or exercising on a bicycle ergometer. It appears that no impairment of reaction times was seen at 140 and 280 ppm in this study. With repetitive exposures, tolerance for most of these effects developed within a few days (Riihimäki and Savolainen,
1980). This study was very difficult to evaluate because of the lack of details about the methods used and the results obtained and thus was not used as a basis for setting acceptable exposures for xylene.
This same group examined the effect on sense of balance in humans of inhalation of m-xylene either at a fixed concentration of 200 ppm or at fluctuating concentrations of 135 to 400 ppm with or without exercise (Savolainen et al., 1985a, b). Sense of balance was measured by a strain gauge platform three times a day (before entering the chamber and 15 to 20 min into the morning and afternoon exposures). Two reports published within 1 mo of each other in different journals reached contradicting conclusions. The first paper reported a negative correlation between atmospheric m-xylene concentration and changes in body sway and a lack of correlation between m-xylene blood concentrations and changes in body sway. The second paper reported a positive correlation between m-xylene blood concentrations and changes in the eyes-closed-to-eyes-open ratios of both average and maximal body sway. In the first study, Savolainen et al. (1985b) reported that "the increase of body sway was always most pronounced during the sham exposure (control) at rest, declining with the increased atmospheric air m-xylene concentration." They also reported that m-xylene did not have any effects on manual or pedal reaction times to visual stimuli. The effect of m-xylene is to improve the overall human sense of balance, although the ratio of eyes closed to eyes open might suggest a slight impairment in balance with eyes closed. Physical exercise during exposure to m-xylene improved rather than impaired body sway (Savolainen et al., 1985b). This study also was not used as a basis for setting acceptable exposure concentrations for xylene because of inconsistencies in the reported results and the difficulty in interpreting them.
Subchronic Exposures
The no-observed-adverse-effect level (NOAEL) for rats and dogs exposed by inhalation to mixed xylenes 6 h/d, 5 d/w, for 13 w was at least 810 ppm. The criteria for a toxic response in this study were changes in body weight, urine chemistry, or blood analyses (Carpenter et al., 1975). Exposure at 1800 ppm caused a slight loss of coordination in rats by 2 h and definite lacrimation in the dog by 1 h and persisted throughout a 4-h exposure (Carpenter et al., 1975).
In general, most subchronic studies have focused on occupational intermittent exposure schedules. Jenkins et al. (1970), however, exposed rats, guinea pigs, monkeys, and dogs continuously for 90 d to o-xylene at 78 ppm. They reported that 1 of 15 rats died on d 56 (Jenkins et al., 1970). In the same study, intermittent exposures (8 h/d, 5 d/w, 30 exposures) to o-xylene at 780 ppm resulted in mortality in 3 of 15 rats, 0 of 15 guinea pigs, 0 of 2 dogs, and 1 of 4 monkeys (Jenkins et al., 1970). One of the two dogs was reported to have tremors of varying severity throughout the exposure. Histopathological examination of tissue sections of heart, lung, liver, spleen, and kidney of all species and brain and spinal cord of dogs and monkeys were essentially negative. Although this study appeared to involve considerable effort, the quality of the study cannot be established easily. The published report was very brief, and the two-paragraph methods section and the single-paragraph results section did not provide enough information to evaluate the quality of the study adequately.
No credible data were found implicating xylenes as reproductive toxins. Xylene has been reported to inhibit development in the in vitro hydra assay, but only at doses close to those that were toxic to adults (Johnson et al., 1986). In mice, m-xylene given by gavage at 2000 mg/kg (toxic to dam) was classified as negative in the Chernoff-Kavlock developmental toxicity screen (Seidenberg and Becker, 1987). In a Hungarian study using rats, however, exposure to o-, m-, or p-xylene at 35, 350, or 700 ppm for 24 h/d from d 7 to 14 of pregnancy was reported to produce the following effects for the three isomers: toxic effects in the dams exposed at 700 ppm and maternal deaths of 4 of 30 rats seen only for m-xylene at 700 ppm; reduced fetal weight at 700 ppm for all isomers and at 350 ppm for o-xylene; increased incidence of skeletal retardation for o-xylene at 700 ppm, for p-xylene at all three concentrations, but not for m-xylene; and decreased activity of several liver and thymus enzymes at the highest dose for all the isomers (Ungváry et al., 1980). Preimplantation fetal death was reported to be increased 31% by exposure to m-xylene at 700 ppm but not by exposure to o- or p-xylene. Postimplantation fetal death was increased by exposure to p-xylene at 700 ppm but not by exposure to o- or m-xylene. However, because the developmental toxicity of xylene is seen only at doses that are overtly toxic to the dams, xylene is not classified as a developmental toxin. In addition, the reported differences between isomers in their developmental toxicity is suspect because the xylene tested
was described to be from the U.S.S.R. and have "analytical purity"; the studies cited to support the assertion that differences exist between isomers did not provide the support. Thus, no credible evidence was found to support the existence of toxicity differences between xylene isomers.
Several groups of investigators have examined the neurochemical effects of xylene exposure. Male rats exposed to m-xylene vapor at concentrations of 49, 393, and 735 ppm for 6 h/d, 5 d/w, for 2 w showed evidence of accumulation of m-xylene in some tissues. Xylene concentrations in brain and perirenal fat increased in proportion to the exposure concentrations between w 1 and 2 of exposure (Savolainen and Pfäffli, 1980). An increase in brain NADPH-diaphorase and azoreductase activities was seen after 2 w at the two highest exposure concentrations, and superoxide dismutase activity decreased in a dose-related manner. Analyses 2 w after exposure ended indicated that the biochemical effects were largely abolished within that time. Cerebral RNA, however, remained above the control value at the two highest concentrations. Mice exposed to m-xylene at 1600 ppm for 4 h/d, 5 d/w, for 7 w had decreased concentrations of binding of 3H-clonidine to alphaadrenergic receptors in the hypothalamus region of the brain (Rank, 1985). During exposure at 1600 ppm, the mice ate and drank more than the control group and gained more weight than the controls (Rank, 1985). Increases in food and water consumption had been noted in preliminary experiments at xylene concentrations as low as 100 ppm (Rank, 1985).
Chronic Exposures
Carcinogenicity and Genotoxicity
The National Toxicology Program found no evidence of carcinogenicity in studies of mixed xylenes (commercial mixture containing 60% m-xylene, 14% p-xylene, 9% o-xylene, and 17% ethylbenzene) in male and female F344/N rats given 250 or 500 mg/kg and male and female mice given 500 or 1000 mg/kg; doses were administered by oral gavage with corn oil 5 d/w for 103 w (NTP, 1986).
TABLE 12-1 Toxicity Summary
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
200 |
3.7 h |
Human |
Increased sensory arousal (evoked potential) |
Seppäläinen et al., 1989 |
|
200 |
3-7 h |
Human |
NOAEL for reaction time and flicker fusiona |
Ogata et al., 1971 |
|
200 |
4 h |
Human |
LOAEL for impaired sense of balance |
Savolainen et al., 1985a |
|
230 |
15 min |
Human |
NOAEL for eye, nose, or throat discomfort |
Carpenter et al., 1975 |
|
10,000 |
~16 h |
Human |
Death in one of three painters; narcosis in other two with amnesia for events during exposure period |
Morley et al., 1970 |
|
78b |
90 d, continuous |
Monkey |
NOAEL for hematological, biochemical, and histopathological effects |
Jenkins et al., 1970 |
|
780b |
8 h/d, 5 d/w, 30 exposures |
Monkey |
Death in one of four monkeys |
Jenkins et al., 1970 |
|
780b |
8 h/d, 5 d/w, 30 exposures |
Dog, guinea pig, monkey |
NOAEL for lethality in 0 of 2 dogs, 0 of 15 guinea pigs, 0 of 3 monkeys |
Jenkins et al., 1970 |
|
49 |
6 h/d, 5 d/w, 2 w |
Rat |
NOAEL for changes in brain enzyme activities |
Johnson et al., 1986 |
|
78b |
90 d, continuous |
Dog |
NOAEL for hematological, biochemical, and histopathological effects |
Jenkins et al., 1970 |
|
78b |
90 d, continuous |
Rat |
Death in 1 of 15 rats |
Jenkins et al., 1970 |
|
78b |
90 d, continuous |
Guinea pig |
NOAEL for hematological, biochemical, and histopathological effects |
Jenkins et al., 1970 |
|
130 |
4 h |
Rat |
LOAEL for slightly increased motility |
Molnár et al., 1986 |
|
Concentration, ppm |
Exposure Duration |
Species |
Effects |
Reference |
|
780b |
8 h/d, 5 d/w, 30 exposures |
Rat |
Death in 3 of 15 rats |
Jenkins et al., 1970 |
|
780b |
8 h/d, 5 d/w, 30 exposures |
Dog |
Tremors in one of two dogs |
Jenkins et al., 1970 |
|
810 |
6 h/d, 5 d/w, 2 w |
Rat, dog |
NOAEL |
Carpenter et al., 1975 |
|
1400 |
30 min |
Mouse |
LOAEL CNS operant performance |
Moser et al., 1985 |
|
1600 |
4 h/d, 5 d/w 7 w |
Mouse |
Increased feeding and drinking; decreased alpha adrenergic receptor binding to hypothalamus |
Rank, 1985 |
|
100 |
Not stated |
Mouse |
Increased feeding and drinking |
Rank, 1985 |
|
2100 |
4 h |
Rat |
LOAEL narcosis |
Molnár et al., 1986 |
|
3500 |
2 min |
Dog |
LOAEL for decrease in left ventricular dp/dt |
Kobayashi et al., 1989 |
|
5984 |
6 h |
Rat |
LC50 |
Bonnet et al., 1982 |
|
6700 (mixed xylenes) |
4 h |
Rat |
LC50 |
Carpenter et al., 1975 |
|
9500 (mixed xylenes) |
2 h |
Cat |
Salivation, ataxia, tonic and clonic spasms, and anesthesia followed by death in four of four cats |
Carpenter et al., 1975 |
|
a Flicker fusion refers to the minimum frequency at which a flickering light no longer appears to flicker. b Exposure was to o-xylene. |
||||
TABLE 12-2 Exposure Limits Set or Recommended by Other Organizations
|
Agency or Organization |
Exposure Limit, ppm |
Reference |
|
ACGIH's TLV |
100 |
ACGIH, 1991 |
|
ACGIH's STEL |
150 |
ACGIH, 1991 |
|
OSHA's PEL |
100 |
ACGIH, 1991 |
|
NIOSH's REL |
100 |
ACGIH, 1991 |
|
NRC's 1-h EEGL |
200 |
NRC, 1984 |
|
NRC's 24-h EEGL |
100 |
NRC, 1984 |
|
NRC's 90-d CEGL |
50 |
NRC, 1984 |
|
TLV, Threshold Limit Value; STEL, short-term exposure limit; PEL, permissible exposure limit; REL, recommended exposure limit; EEGL, emergency exposure guidance level; CEGL, continuous exposure guidance limit. |
||
TABLE 12-3 Spacecraft Maximum Allowable Concentrations
|
Exposure Duration |
Concentration, ppm |
Concentration, mg/m3 |
Target Toxicity |
|
1 h |
100 |
435 |
Throat irritation, narcosis |
|
24 h |
100 |
435 |
Throat irritation, narcosis |
|
7 da |
50 |
217 |
Throat irritation |
|
30 d |
50 |
217 |
Throat irritation |
|
180 d |
50 |
217 |
Throat irritation |
|
a Previous 7-d SMAC = 20 ppm (86.8 mg/m3). |
|||
RATIONALE FOR ACCEPTABLE CONCENTRATIONS
Calculation of the highest acceptable concentration (AC) for each major end point and exposure duration is documented below. The guidelines established by the National Research Council (NRC, 1992) were used to compare the resulting ACs for the various end points, and the lowest AC at each exposure duration was selected as the SMAC.
Lethality
1-h and 24-h ACs in Humans
Three workers painting in a confined space on a ship were found unconscious and taken to a hospital One was dead on arrival. The men were discovered unconscious 15.75 h after their lunch. The atmospheric concentration of xylene in the confined space was estimated to be 10,000 ppm on the basis of the volume of paint applied to the walls, the volume of the confined space, and the poor ventilation. Despite the poor ventilation, Morley et al. (1970) believed that oxygen was adequately diffused into the area through the open access hole. ACs for 1-h and 24-h exposures are based on 10,000 ppm as the lowest-observed-adverse-effect level (LOAEL) for lethality for a 15.75-h exposure. Therefore,
1-h AC = 10,000 ppm/10 (to NOAEL) = 1000 ppm; and applying Haber's rule for a 24-h exposure,
24-h AC = 10,000 ppm/10 (to NOAEL) × (16 h/24 h) = 670 ppm.
1-h and 24-h ACs in Rats
The LC50 in rats for a 4-h exposure to xylene is approximately 6700 ppm (Carpenter et al., 1975). Comparing the data for rats with the data above for humans shows that rats have no less sensitivity to the lethal effects of xylene than do humans; thus, the use of a 10-fold interspecies safety factor is inappropriate in this case. The data used to calculate the LC50 comprise two concentrations: one yielded no deaths and the other higher concentration yielded 9 deaths of 10 exposed rats. Therefore, the data do not permit the use of the ''benchmark dose'' methodology for calculating the lower 95% bound on the dose that gives a 0.1 % risk of death. To calculate 1-h and 24-h ACs, the 6700-ppm LC50 was divided by 10 to extrapolate to a NOAEL for lethality and adjusted for exposure duration using Haber's rule. For the 1-h ex-
posure, the 6700-ppm 4-h LC50 was not increased. Extrapolations beyond 24 h are not appropriate. Thus, on the basis of rat lethality, the 1-h and 24-h ACs are
1-h AC = 6700 ppm/10 (to NOAEL) = 670 ppm.
24-h AC = 6700 ppm/10 (to NOAEL)/(24 h/4 h) = 110 ppm.
Long-term continuous exposure data are not available for m-xylene. Jenkins et al. (1970), however, studied the effects of continuous exposure to o-xylene for 90 d in several animal species. Of 15 rats exposed continuously to o-xylene at 78 ppm, one died on d 56 of a 90-d exposure. It was not stated whether the death was believed to be exposure related; however, because no rats died after 56 d and no guinea pigs, monkeys, or dogs died that were similarly exposed, the one rat death is considered spurious and not related to xylene exposure. Thus, ACs for lethality were not calculated for exposure times greater than 24 h.
Throat Irritation
One of seven human volunteers exposed for 15 min to m-xylene reported mild throat discomfort during the first minute and again during the seventh minute of inhalation at 106 ppm and during the first minute of inhalation at 230 ppm, but none reported discomfort while inhaling 460 ppm (Carpenter et al., 1975).
1-h and 24-h ACs
In general, irritation is highly dependent on atmospheric concentration and much less dependent on exposure duration. The degree of irritation reported in this study was minor and was inversely dose related. Such mild irritation would be acceptable for a brief contingency exposure. Thus, in calculating the ACs for 1-h and 24-h exposures, the 106-ppm value was not adjusted to a NOAEL value and was not adjusted for exposure duration. The value was rounded to 100 ppm.
1-h and 24-h AC = 106 ppm, rounded to 100 ppm.
7-d 30-d, and 180-d ACs
Although the irritation reported in the study was mild and intermittent, the 7-d, 30-d, and 180-d ACs were set to prevent even that degree of discomfort. Thus, the 1-h AC, based on the LOAEL, was divided by 2 to estimate a NOAEL:
7-d, 30-d, 180-d AC = 106 ppm/2 = 53 ppm, rounded to 50 ppm.
Eye Irritation
One of seven volunteers exposed for 15 min to m-xylene reported intermittent mild eye irritation at 230 ppm (Carpenter et al., 1975). The degree of irritation reported in this study was minor but dose related. Such mild irritation would be acceptable for a brief contingency exposure. Thus, in calculating the ACs for 1-h and 24-h exposures, the 230-ppm LOAEL value was not adjusted to a NOAEL value and was not adjusted for exposure duration.
For a 1-h or 24-h exposure,
AC = 230 ppm, rounded to 250 ppm.
Again, although the irritation reported in the study was mild and intermittent, the 7-d, 30-d, and 180-d ACs were set to prevent even that degree of discomfort. Thus, the 230-ppm LOAEL was divided by 2 to estimate the NOAEL and rounded to a value of 100 ppm.
For 7-d, 30-d, or 180-d exposures,
AC = 230 ppm/2 = 115 ppm, rounded to 100 ppm.
Histopathological Changes
A well-designed and executed subchronic intermittent inhalation study of mixed xylenes was reported by Carpenter et al. (1975). Nine-
teen rats and four dogs exposed for 6 h/d, 5 d/w, for 65 d to mixed xylenes (65% m-xylene, 19.3% ethylbenzene, 7.8% p-xylene, 7.6% o-xylene) at 810, 460, or 180 ppm were monitored for exposure-related changes (Carpenter et al., 1975). Examination revealed no differences at any dose from air-exposed controls in body weight, blood and urine analyses, and histopathological changes of tissues from the adrenal, brain, pituitary, trachea, thyroid, parathyroid, lung, heart, liver, kidney, spleen, stomach, duodenum, pancreas, ileum, jejunum, colon, skeletal muscle, sciatic nerve, and bone-marrow impression smear (Carpenter et al., 1975). To calculate the ACs for 7 d and 30 d, the 810-ppm value was divided by 10 for species extrapolation, multiplied by 16.25 (= 6 h/d × 65 d ÷ 24 h/d), and divided by the exposure duration in days to apply Haber's rule. Use of Haber's rule in extrapolation to shorter times (24 h and 1 h) was not justified because of possible threshold effects in which the body's defense mechanisms (e.g., metabolism) could be overwhelmed. Extrapolation to 180 d was also inappropriate from a total exposure of only 390 h (6 h/d × 65 d). Therefore,
For a 7-d exposure,
AC = 810 ppm/10 × 16.25/7 = 188 ppm, rounded to 200 ppm.
For a 30-d exposure,
AC = 810 ppm/10 × 16.25/30 = 44 ppm, rounded to 50 ppm.
Narcosis
Groups of eight rats were exposed to m-xylene for 4 h at concentrations up to 2100 ppm (Molnár et al., 1986). Narcosis occurred only at the highest concentration (2100 ppm). The next lower concentration tested was 1000 ppm. This was considered the NOAEL for narcosis. In deriving an AC, the NOAEL was not adjusted for the exposure duration because, as was mentioned above, effects on the CNS depend on concentrations of xylene in the blood but do not increase with exposure duration (Savolainen et al., 1985a). Thus, the ACs for narcosis for all
exposure durations were calculated from the NOAEL using only a species extrapolation factor of 10:
AC (1 h through 180 d) = 1000 ppm/10 = 100 ppm.
Reduced Cardiac Output
A threshold value of 0.35% (3500 ppm) m-xylene was required to produce a statistically significant decrease in the peak left-ventricular dp/dt in 25 dogs anesthetized with sodium pentobarbital and exposed for 2 min to xylene-vapor concentrations up to 2.0% (20,000 ppm) (Kobayashi et al., 1989). A dose response was observed at higher concentrations. A decrease of 10% or more in the dp/dt would be of clinical concern due to reduced cardiac output (R. Billica, chief, Medical Operations, NASA Johnson Space Center, Houston, Tex., personal commun., 1992). Although there was considerable scatter in the dose-response data, Kobayashi et al. (1989) calculated a regression line that indicates that a 10% reduction in the dp/dt would occur at a concentration of about 1.4% (14,000 ppm) m-xylene. Cardiac effects have been shown to depend on the concentration of an organic solvent in the blood and to be independent of the exposure duration (G. D. Whedon, consultant, Shriners Hospital Headquarters, Tampa, Fla., personal commun., 1982); therefore, a calculated AC value should apply for all exposure times from 1 h to 180 d. The assumption must be made that the blood concentrations of xylene achieved in a 2-min exposure approach the concentrations that would be achieved in exposures of 1 h or longer. To calculate an AC based on reduced cardiac output, the 14,000-ppm LOAEL is divided by 10 for interspecies extrapolation and again by 10 to estimate the NOAEL from the LOAEL. Thus, the AC for all exposure durations is
14,000 ppm/10/10 = 140 ppm.
Although spaceflight has been suspected as a factor in increasing the risk of cardiac arrhythmias, a reduction in cardiac output, such as that potentially induced by xylene exposure, would reduce the risk of cardiac arrhythmia (G. D. Whedon, consultant, Shriners Hospital Headquarters, Tampa, Fla., personal commun., 1982).
SPACEFLIGHT CONSIDERATIONS
Of the end points induced by exposure to xylenes, only cardiac output possibly would be affected by launch, microgravity, or re-entry. The decrease in the force of contraction of the left ventricle would tend to counteract the increased susceptibility to arrhythmia induced by spaceflight. Thus, no spaceflight factor was used in calculating the AC for cardiac depression.
TABLE 12-4 Acceptable Concentrations
|
|
|
Uncertainty Factors |
||||||||
|
|
|
To NOAEL |
|
|
Spaceflight |
Acceptable Concentrations, ppm |
||||
|
Effect, Data, Reference |
Species |
Species |
Time |
1 h |
24 h |
7d |
30 d |
180 d |
||
|
Lethality, LC50, 6700 ppm (Carpenter et al., 1975) |
Rat |
10 |
1 |
HR |
1 |
670 |
110 |
|||
|
Lethality, LOAEL, 10,000 ppm (Morley et al., 1970) |
Human |
10 |
1 |
HR |
1 |
1000 |
670 |
|||
|
Mild throat irritation, LOAEL, 10,000 ppm (Carpenter et al., 1975) |
Human |
1, 2 |
1 |
1 |
1 |
100 |
100 |
50 |
50 |
50 |
|
Eye irritation, LOAEL, 230 ppm (Carpenter et al., 1975) |
Human |
1, 2 |
1 |
1 |
1 |
250 |
250 |
100 |
100 |
100 |
|
Tissue histology, clinical chemistry, hematology, urine analysis, body and organ weights, |
Rat, mouse |
1 |
10 |
HR |
1 |
200 |
50 |
|||
|
EKGs, food consumption; NOAEL, 810 ppm, 6 h/d, 5 d/w, 65 d (Carpenter et al., 1975) |
|
|||||||||
|
Narcosis, NOAEL, 1000 ppm (Molnár et al., 1986) |
Rat |
1 |
10 |
HR |
1 |
100 |
100 |
100 |
100 |
100 |
|
Cardiac depression, LOAEL, 14,000 ppm (Kobayashi et al., 1989) |
Dog |
10 |
10 |
1 |
1 |
140 |
140 |
140 |
140 |
140 |
|
SMACs |
|
|
|
|
|
100 |
100 |
50 |
50 |
50 |
|
—, Data not considered applicable to the exposure time; HR, Haber's rule. |
||||||||||
REFERENCES
ACGIH. 1991. Guide to Occupational Exposure Values—1991. American Conference of Governmental Industrial Hygienists, Cincinnati, Ohio.
Bonnet, P., Y. Morale, G. Raoult, D. Zissu, and D. Gradiski. 1982. Détermination de la concentration léthale50 des principaux hydrocarbons aromatiques chez la rat. Arch. Mal. Prof. 43:261-265.
Bray, H. G., B. G. Humpris, and W. V. Thorpe. 1949. Metabolism of derivatives of toluene. 3. o-, m-, and p-xylenes. Biochem. J. 45:241.
Browning, E. 1965. Xylene. Pp. 77-89 in Toxicity and Metabolism of Industrial Solvents. Amsterdam: Elsevier.
Carpenter, C. P., E. R. Kinkead, D. L. Geary, Jr., L. J. Sullivan, and J. M. King. 1975. Petroleum hydrocarbon toxicity studies. V. Animal and human response to vapors of mixed xylenes. Toxicol. Appl. Pharmacol. 33:543-558.
Emmet, E. A. 1976. Paraosmia and hyosmia induced by solvent exposure. Br. J. Ind. Med. 33:196-198.
Ghantous, H., L. Denker, J. Gabrielson, B. R. G. Danielsson, and K. Bergman. 1990. Accumulation and turnover of metabolites of toluene and xylene in nasal mucosa and olfactory bulb in the mouse. Pharmacol. Toxicol. 66:87-92.
Jenkins, L. J., Jr., R. A. Jones, and J. Siegel. 1970. Long-term inhalation screening studies of benzene, toluene, oxylene, and cumene on experimental animals. Toxicol. Appl. Pharmacol. 16:818-823.
Johnson, E. M., B. E. G. Gabel, M. S. Christian, and E. Sica. 1986. The developmental toxicity of xylene and xylene isomers in the hydra assay. Toxicol. Appl. Pharmacol. 82:323-328.
Kobayashi, H., R. Hobara, and T. Sakai. 1989. Effects of inhalation of several organic solvents on left-ventricular dp/dt. Jpn. J. Ind. Health 31:136-141.
Liebich, H. M., W. Bertsch, A. Zlatkis, and H.J. Schneider. 1975. Volatile organic components in the Skylab. Aviat. Space Environ. Med. 46:1002-1007.
Low, L. K., J. R. Meeks, and C. R. Mackerer. 1989. Health effects of the alkylbenzenes. II. Xylenes. Toxicol. Ind. Health 5:85-105.
Molnár, J., K. A. Paksy, and M. Náray. 1986. Changes in the rat's motor behaviour during 4 hr inhalation exposure to prenarcotic con-
centrations of benzene and its derivatives. Acta Physiol. Hungarica 67:349-354.
Morley, R., D. W. Eccleston, C. P. Douglas, W. E. J. Greville, D. J. Scott, and J. Anderson. 1970. Xylene poisoning: A report of one fatal case and two cases of recovery after prolonged unconsciousness. Br. Med. J. 3:442-443.
Moser, V. C., E. M. Coggeshall, and R. L. Balster. 1985. Effects of xylene isomers on operant responding and motor performance in mice. Toxicol. Appl. Pharmacol. 80:293-298.
NRC. 1984. Emergency and Continuous Exposure Guidance Levels for Selected Airborne Contaminants, Vol. 2. Washington, D.C.: National Academy Press.
NRC. 1992. Guidelines for Developing Spacecraft Maximum Allowable Concentrations for Space Station Contaminants. Washington, D.C.: National Academy Press.
NTP. 1986. Toxicology and Carcinogenesis Studies of Xylenes (Mixed) in F344/N Rats and B6C3F1 Mice. NTP TR 327. National Institutes of Health, National Toxicology Program, Research Triangle Park, N.C.
Ogata, M., Y. Takatsuka, K. Tomokuni, and K. Muroi. 1971. Excretion of hippuric acid and m- or p-methylhippuric acid in the urine of persons exposed to vapours of toluene and m- or p-xylene in a exposure chamber and in workshops, with specific reference to repeated exposures. Br. J. Ind. Med. 28:382-385.
Rank, J. 1985. Xylene induced feeding and drinking behavior and central adrenergic receptor binding. Neurobehav. Toxicol. Teratol. 7:421-426.
Riihimäki, V., P. Pfäffli, and K. Savolainen. 1979a. Kinetics of m-xylene in man: Influence of intermittent physical exercise and changing environmental concentrations on kinetics . Scand. J. Work Environ. Health 5:232-248.
Riihimäki, V., P. Pfäffli, K. Savolainen, and K. Pekari. 1979b. Kinetics of m-xylene in man: General features of absorption, distribution, biotransformation and excretion in repetitive inhalation exposure. Scand. J. Work Environ. Health 5:217-231.
Riihimäki, V., and K. Savolainen. 1980. Human exposure to m-xylene. Kinetics and acute affects on the central nervous system. Ann. Occup. Hyg. 23:411-422.
Huntoon, C. L. 1987. Introduction Summary Report of Postflight At-
mospheric Analysis for STS 41-D to 61-C. Doc. SD4/87-253. Internal NASA JSC Memorandum from SA/Director, Space and Life Sciences, to GA/Deputy Manager, National Space Transportation System, Johnson Space Center, Houston, Tex.
Savolainen, H., and P. Pfäffli. 1980. Dose-dependent neurochemical changes during short-term inhalation exposure to m-xylene. Arch. Toxicol. 45:117-122.
Savolainen, K., V. Riihimäki, R. Luukkonen, and O. Muona. 1985a. Changes in the sense of balance correlate with concentrations of xylene in venous blood. British J. of Industrial Medicine 42:765-769.
Savolainen, K., V. Riihimäki, O. Muona, J. Kekoni, R. Luukkonen, and A. Laine. 1985b. Conversely exposure-related effects between atmospheric m-xylene concentrations and human body sense of balance. Acta Pharmacol. Toxicol. 57:67-71.
Sax, N. I., ed. 1984. Dangerous Properties of Industrial Materials, Sixth Ed. New York: Van Nostrand Reinhold.
Sedivec, V., and J. Flek. 1976. The absorption, metabolism, and excretion of xylenes in man. Int. Arch. Occup. Environ. Health 37: 205-217.
Seidenberg, J. M., and R. A. Becker. 1987. A summary of the results of 55 chemicals screened for developmental toxicity in mice. Teratogen. Carcinogen. Mutagen. 7:17-28.
Senczuk, W., and J. Orlowski. 1978. Absorption of m-xylene vapours through the respiratory tract and excretion of m-methylhippuric acid in urine. Br. J. Ind. Med. 35:50-55.
Seppäläinen, A. M., A. Laine, T. Salmi, V. Riihimäki, and E. Verkkala. 1989. Changes induced by short-term xylene exposure in human evoked potentials. Int. Arch. Occup. Environ. Health 61:443-449.
Ungváry, G., E. Tátrai, A. Hudák, G. Barcza, and M. Lorincz. 1980. Studies on the embryotoxic effects of ortho-, meta-, and para-xylene. Toxicology 18:61-74.
























