| This page in the original is blank. |
Introduction
Robert Newcomer
Empirical investigation of the health outcomes and quality of care in managed care systems for people with chronic health conditions has a short history. It traces back to the mid-1980s with the initiation of the Medical Outcomes Study (MOS) and various health maintenance organization (HMO) demonstration evaluations. These studies have generally been designed as comparisons between managed care patients and patients with similar conditions receiving care under fee-for-service reimbursement (FFS). The FFS system is used as the frame of reference to define appropriate access to tests and procedures, and patient outcomes such as functionality, readmission rates, and mortality levels. Issues such as these are given particular attention because of the patterns of lower service use—particularly hospitals, home health care, and specialist referrals—found among managed care members. A fundamental concern is whether reduced access to care (and the presumed lower expenditures) produces an adverse effect on health status or mortality.
The analysis comparing health outcomes between fee for service and managed care usually reports results for either nonelderly or only elderly patients. Although there are exceptions, the general pattern has been to find that managed care members are somewhat healthier at the time of enrollment in these plans than those remaining in fee for service and that health outcomes between these two groups are similar. Such findings have been reported for both elderly and nonelderly members. Many of these findings have come from the Medical Outcomes Study.
Alvin Tarlov presents new findings from the MOS, which compares physical health status change over four years. This measure is a composite index weighted by physical functioning, role performance, pain, general health
perception, social functioning, change in health, sense of vitality, and mental health. Three sets of comparisons between managed care and FFS members are shown for the nonelderly, elderly, and poor versus nonpoor. The elderly, those in poverty, and those who were the most ill at the outset of the four-year period show less favorable change in status than comparable persons in fee for service. This finding is at variance with previous MOS and other analyses that have looked at more discrete outcomes. Nevertheless, it raises an important hypothesis about the relationship between health care expenditures (or utilization) and outcomes. Dr. Tarlov and his colleagues depict this relationship as an S-shaped curve, with expenditures or utilization on the horizontal axis and health gain on the vertical. They posit that many in the population (i.e., the nonelderly) are on the flat part of the S, meaning that expenditures and utilization can be reduced without adverse effects on health status. Other groups such as the elderly, as suggested by these findings, may be closer to the rising portions of the S—which makes them more vulnerable to reductions in expenditures or utilization.
One implication from these findings is that the formulas for reimbursing managed health care may have to be more sensitive to differences in historical patterns of utilization (and the marginal gains from this utilization) between the general population and those from more vulnerable segments of the population. This conclusion likely extends to a variety of chronic conditions such as rheumatoid arthritis and system lupus erythematosus. A second implication builds on recognition that the quality and the appropriateness of care are wholly determined by reimbursement. There remain the other concerns addresses by this conference: self-management, care coordination among physicians and ancillary providers, and ensuring access to prevention and rehabilitation. These objectives emerge as important priorities for medical education and health delivery refinement. Testing the efficacy of these refinements similarly emerges as the next stage in outcomes research.
Invited Address
Alvin R. Tarlov
This morning I am going to present data85 from the Medical Outcomes Study. Begun in 1986, MOS is a longitudinal observational study of about 2,300 patients with chronic disease in three large metropolitan areas. The patients were selected from 28,000 doctor-patient interactions in those cities, based on having moderately severe or worse diseases in five different categories—hypertension, recovery from acute myocardial infarction, congestive heart failure, adult onset diabetes mellitus, and clinical depression. At entry into the study, patients were being cared for in either the fee-for-service system or some variation of the HMO system. The latter category was actually a combination of the different systems that John Eisenberg showed. The definition of HMO in the Medical Outcomes Study is a system in which patient care is prepaid and capitated, and in which there is some form of restraint on utilization. The MOS has already proven to be a valuable source of high-quality information.86
In the MOS, functional health data were collected on the same patients in 1986, 1987, 1988, and 1990, and deaths up to 1994. The data presented today are four-year longitudinal data in patients with five chronic diseases aggregated. A focus is on baseline and four-year data (1986 and 1990).
The presentation concentrates on specific subgroups within the total patient population of 2,235. Patients are divided into those in HMO care and those in fee-for-service care; they are then subdivided further into elderly and nonelderly groups and into poverty and nonpoverty groups. Elderly is defined simply as 65 years old or greater. Poverty means income within 200 percent of the official federal poverty line.
As primary measures of physical and mental health, we used summary indices from the self-reported Short-Form Health Survey (SF-36).87 The physical health score is a compilation of eight differentially weighted scales: physical functioning, role performance, physical pain, general health perception, social functioning, sense of vitality, role performance, emotional and mental health. For the physical health score, the scales are differentially
|
|
262:3298–3302. Stewart, AL, S Greenfield, RD Hays, et al. (1989). Functional status and well-being of patients with chronic conditions: Results from the Medical Outcomes Study. Journal of the American Medical Association, 262: 907–913. Kravitz RL, S Greenfield, WH Rogers, WG Manning, Jr., M Zubkoff, EC Nelson, AR Tarlov, JE Ware, Jr. (1992). Differences in the mix of patients among medical specialties and systems of care: Results from the Medical Outcomes Study. Journal of the American Medical Association, 267: 1617–1623. Greenfield S, EC Nelson, M Zubkoff, WG Manning, W Rogers, RL Kravitz, A Keller, AR Tarlov, JE Ware, Jr. (1992). Variations in resource utilization among medical specialties and systems of care: Results from the Medical Outcomes Study. Journal of the American Medical Association, 267: 1624–1630. McHorney, CA, JE Ware, AE Raczek (1993). The MOS 36-Item Short-Form Health Survey (SF-36), II: Psychometric and clinical tests of validity in measuring physical and mental health constructs. Medical Care, 31: 247–263. Rubin, H, B Gandek, WH Rogers, M Kosinski, C McHorney, JE Ware (1993). Patient's ratings of outpatient visits in different practice settings: Results from the Medical Outcomes Study. Journal of the American Medical Association, 207: 836–840. Rogers, WH, KB Wells, LS Meredith. R Sturm, A Burnam (1993). Outcomes for adult outpatients with depression under prepaid or fee-for-service financing. Archives of General Psychiatry, 50: 517–525. Safran, D, AR Tarlov, WH Rogers (1994). Primary care performances in fee-for-service and prepaid health care systems: Results from the Medical Outcomes Study. Journal of the American Medical Association, 271: 1579–1586. Ware, JE, M Kosinski, SK Keller (1994). SF-36 Physical and Mental Health Summary Scales: A User's Manual. Boston: The Health Institute, New England Medical Center. Ware, JE, M Kosinski, MS Bayliss, CA McHorney, WH Rogers, A Raczek (1995). Comparison of methods for scoring and statistical analysis of SF-36 Health Profiles and Summary Measures: Summary of Results from the Medical Outcomes Study. Medical Care, 33:4 AS264–AS279. Greenfield, S, WH Rogers, M Mangotich, MF Carney, AR Tarlov (1995). Outcomes of patients with hypertension and non-insulin-dependent diabetes mellitus treated by different systems and specialties: Results from the Medical Outcomes Study. Journal of the American Medical Association, 274: 1436–1474. |
weighted to give greater importance to the physical health end of the scales and progressively less importance to the mental health end of the scales. Only the physical health data are given in this presentation. All data have been adjusted for sociodemographic factors, disease, and disease severity.
Baseline scores, change in scores in four years, and categorical change in four years are presented. Categorical change is the percentages of patients whose physical health scores worsened, stayed the same, and got better in four years. Worse physical health is defined as a decrease of at least 1 standard deviation in score at the end of four years. Better is defined as a gain of at least 1 standard deviation at the end of four years.
The U.S. population—a general, national probability sample—scores 50 on physical health because the instrument was calibrated that way, with a standard deviation of about 7. The MOS patient mean of 45 indicates that MOS patients are sicker than the general U.S. population, as expected from our selection of patients having chronic disease. In fact, a score of 45 in physical health places these patients in the 24th percentile of the U.S. population. Their mean score of 48.5 on mental health places these patients in the 35th percentile.
In tracking through the four years, the physical health score decreases by 3 points for the average MOS patient—a drop from the 24th to the 19th percentile. Mental health, on the other hand, increases—there is a rise in mental health with age with this instrument—from the 35th to the 38th percentile. The categorical change scores in physical health indicate that 15 percent of the total MOS population improved and 29 percent got worse.
The power of the Medical Outcomes Study is such that a change from 1 to 1.5 points, on either the physical or the mental health scale, is significant statistically and clinically. To put these scores in perspective, a patient whose SF-36 physical functioning score increases by 6.5 points, has had an improvement in function equivalent to a patient who received an aortic valve replacement to relieve congestive heart failure or a total hip arthroplasty. Patients who get worse on physical health are one-third more likely to lose their job in the next year because of health than patients whose scores are unchanged. MOS patients who declined on their physical health in four years—29 percent of them—are more likely to have experienced a new myocardial infarction, have a 50 percent chance of having had a weight loss sufficient to prompt a visit to a physician, and have a 60 percent chance of having experienced chest pain of sufficient severity to warrant hospitalization. The improvement in mental health scores by patients who improved is equivalent to a recovery from depression. The calibration is mentioned to indicate that the discerning power of becoming worse or getting better by using this methodology is powerful and meaningful and should be given appropriate anention, despite what appear to be small changes in the raw scores.
Turning to the main focus of the study, we found that HMO and fee-for-service patients on average were equivalent on physical health entry to the study, that their four-year change (decline of 3 points) is about equal, and that the categorical changes—worse, same, and better—were similar in the HMO and the FFS groups. This is essentially what Shelly Greenfield and colleagues reported in the Journal of the American Medical Association in 1995, with patients in HMO and FFS systems who have either hypertension or adult onset diabetes showing equivalent changes over time.88
The data from the 2,235 patients were separated into elderly and nonelderly groups (Table 6-1). As expected, at entry the elderly as a group score lower—a decrease of 5.8 points versus 1.9 for the nonelderly. The more rapid decline of the elderly in physical health is also reflected in categorical change scores, with fewer of the elderly getting better and more becoming worse over the four-year period.
TABLE 6-1 Physical Health Changes in MOS Patients over Four Years
The four-year transitions of elderly patients in HMOs showed a greater decline in physical functioning than those in the FFS system—a 7.0 point drop versus a 5.0-point drop for the fee-for-service group. That difference in the four-year change is statistically significant. For the nonelderly, we saw no such difference. The categorical change data (Table 6-2) are especially impressive. Physical health was more stable (the same percentage) by a factor of two in the fee-for-service system than in the prepaid system. Roughly twice as many HMO compared to FFS patients got worse in a four-year period, a statistically significant difference at the .01 level. We saw no such difference,
at least not a very impressive one with the sample size that we had, in the nonelderly in those two systems of care.
TABLE 6-2 Physical Changes in Elderly Patients over Four Years, by Type of Health Plan
|
Measure |
HMO (n = 346) |
FFS (n = 476) |
|
Percent worse |
54 |
28 |
|
Percent same |
37 |
63 |
|
Percent better |
9 |
9 |
|
NOTE: All differences are statistically significant (p < 0.001). |
||
According to the physical health scores and categorical changes of poor patients, 18 percent of patients in the FFS system improved over the four-year period, whereas only 9 percent improved in the HMO system (Table 6-3). This was significant at the .05 level. The nonpoverty groups in HMO and in FFS didn't show that difference.
Poor patients who were most sick at the onset of the study in 1986 (bottom third on physical health score) were analyzed separately. At baseline, the physical functioning of the sickest was about 10 points lower than the average poor patient.
TABLE 6-3 Physical Changes in MOS Patients over Four Years, by Income Level and Type of Health Plan
Of more interest to the committee's deliberations, HMO patients got worse (-2.0), whereas the FFS patients improved (+5.4 points), significant at a probability level of < 0.001 (Table 6-4). A similar pattern is seen in the categorical change data: in the FFS system, roughly two-and-a-half times as many patients improved in the HMO system, 33 percent of patients in HMOs got worse, and 5 percent of those in fee for service system, a differential of six-or sevenfold (Table 6-4).
TABLE 6-4 Physical Changes in the Sickest Third of Poor Patients over Four Years, by Type of Health Plan
|
Measure |
HMO (n = 90) |
FFS (n = 126) |
|
SF-36 change |
-2.0 |
5.4 |
|
Percent worse |
33 |
5 |
|
Percent same |
45 |
38 |
|
Percent better |
22 |
57 |
|
All differences are statistically significant (p < 0.05). |
||
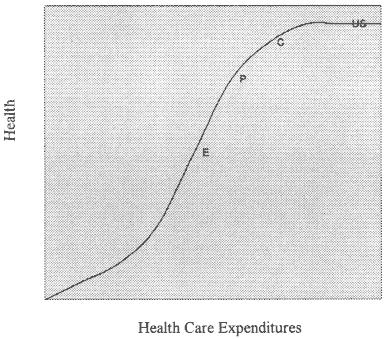
I'd like to close by summarizing my subjective sense of the state of health care in a hypothetical curve (Figure 6-1) describing the relationship between health utilization or expenditures on the horizontal axis and health gain on the vertical axis. This relation, I think, is expressed in an S-shaped curve with a long flat plateau at the top. The average U.S. population is on the flat of the curve, I speculate, perhaps $100 billion dollars or more from the point of inflection. If the total U.S. health care expenditure was reduced 10 percent, there would be no change in the average health gain or average health of the entire population. However, that subset of the U.S. population with chronic disease is likely to be at the point of inflection of this curve (C), so reduction in utilization of its medical services is likely to result in a decline in health. The elderly with chronic disease (E) and the poor with chronic disease (P) may be further to the left, on the downslope of the curve, perhaps needing more services than they are presently receiving and perhaps more vulnerable to decrements in health services. I think that the MOS data are telling us that although for the average American, decreases in health care services will not be detrimental to health, elderly and poor people with chronic disease should be monitored separately to ensure that their health is not being jeopardized by cost-restraining mechanisms.
| This page in the original is blank. |
Invited Reaction
Carolyn Clancy
I want to share some good news and some bad news about where we stand right now in terms of tools available to assess performance and quality in organized systems of care. The good news is that, along with a growing public concern that what some people call the epidemic of managed care may get us to an end point where less is not more but simply less, has come an intense interest in actually developing and using tools to assess quality. This really is good news. The bad news is that it is a lot harder to do than we thought. I would challenge any of you to try to come up with some tools that are easily used but would reflect the quality of care given a population of patients with any condition in rheumatology.
I know Matt Liang is working on some tools for the Foundation for Accountability, but the number of tools that are ready to use right now is actually quite limited. For the past couple of years, an effort that grew out of health care reform to actually have a list of conditions and performance measures has grown into a database that we will make publicly available soon. Clinicians, health care organizations, and others can use this database to identify tools and even create their own measures for assessing quality. This is also good news.
Again, the bad news is actually making this happen in the real world. Some of the problems that arise are fairly obvious: How do you identify the right eligible patient population? You have to use diagnostic coding data for the most part, which are quite flawed in many systems of care. The diagnostic codes were not created to identify clinical conditions, they were meant to simplify billing.
Frankly, the list of conditions for which there is a very clear link between
the process of care and the outcomes of that care is very short. I think this is an important challenge for all of you, as we start to think about the impact of changing health care organizations on rheumatologic diseases.
Many of you are familiar with the report cards that exist now. I heard these recently described as being something like the Wright brothers airplane: simultaneously an astonishing achievement, but astonishingly primitive as well. We are working very hard with National Committee for Quality Assurance (NCQA) and other groups to try to push the science in this field, but it will remain a challenge, I think, for the foreseeable future.
What is the fuel, if you will, for these performance measures? That, I think, very much comes out of our outcomes research program. As many of you are probably aware, the Agency for Health Care Policy and Research (AHCPR) has had sort of a politically disastrous year this past year, but we are still here. That is the good news. The not-so-good news, of course, is that we are functioning on a much lower budget and trying to prioritize as carefully as possible. After six years, a fairly substantial investment in outcomes research, I think the good news is that we have built a tremendous capacity to perform this kind of research and have tried to establish a growing interest in it. The Institute of Medicine now tells us that there is actually a shortage of outcomes researchers, which I think is all very, very good news. I certainly hope that resources will be forthcoming to keep the researchers busy and productive.
The bad news is that we find ourselves in a situation with which, I think, the field of rheumatology has been familiar for a long time. That is to say, although our knowledge has expanded, we find ourselves in a situation with more questions than answers.
One of the questions that challenges us as we try to assess quality of care for rheumatology and other fields is, What are the best kinds of measures? Are they disease-specific or general measures? I am sure that Dr. Tarlov has a very clear opinion about that, but there are many people doing research in this area who believe that clinicians are much more persuaded by disease-specific measures.
Another big issue relates to the timing of assessments. How often do you use the instruments you have? I often run into people at meetings who say, ''We think the SF-36 is the greatest thing we have ever found, but we actually don't know how often to give it out and whom we should ask to take it.''
Invited Reaction
Matthew H. Liang
Dr. Tarlov's data are very important. In fact, they probably underestimate the true magnitude of the problem. I say this because generic health status measures were used, and disease-specific measures would be even more sensitive to change for groups of patients and would show greater differences. After 20 years of work, partly funded by the National Institutes of Health (NIH) and the Arthritis Foundation, we have a collection of fine, disease-specific instruments that are valid in measuring what they purport to measure, are reproducible, and are sensitive in clinical trials of medications, surgery, or rehabilitation.89 Disease-specific measures for lupus, for example, would include the following:
1. Activity-related:
SLE—Disease Activity Index
SLE—Activity Measure
British Isles Lupus Assessment Group
European Consensus Lupus Activity Measure
Lupus Activity Index
2. Organ damage related:
Systemic Lupus International Collaborative Clinics
A list of disease-specific measures that have been developed for rheumatoid arthritis would include the following:
- Arthritis Impact Measurement Scales (AIMS)
- Health Assessment Questionnaire (HAQ)
- Functional Status Index (FSI)
- MACTAR Questionnaire
- Lee Functional Status Instrument
- Toronto Functional Capacity Questionnaire
- Convery Polyarticular Disability Index
- Modified Health Assessment Questionnaire
- Shortened Arthritis Impact Measurement Scale
The best measures we have, however, are frequently insensitive at the patient level. One example involves grip strength. When one compares pounds of pressure generated in a grip strength test to a response to a question about how difficult it is to do a power grip, a significant number of people who by objective physical measurement are unable to do a power grip say they have no problems.
Another example is a 70-year-old gentleman seen on house call for arthritis. His apartment was completely denuded except for a cockpit of items around the sofa where he sat. Many years before my visit his wife had died. Shortly thereafter he was admitted to a hospital with a urinary tract infection, came home in a weakened condition, and stumbled as he went into the apartment. He was convinced that something bad had happened and basically lived on his couch for 17 years, developing severe flexion contractures of the hip and knee. Nevertheless, on a reliable, valid measure of function he reported no functional problems. New measures work in groups but are insensitive at the individual level because of the multiple determinants of self-expressed dysfunction or disability.
Tarlov's data are extremely important and probably underestimate the extent of the problem for those with chronic rheumatic diseases—particularly at the individual level. For individuals at risk, we are trying to get scientific answers to what I think are basically political and business questions. We have the tools, although we can not really capture the true impact on individuals, but we don't have the time to study the situation in a changing environment.
Discussion
MARK ROBBINS: One of the challenges to outcome studies is that health care delivery systems and the incentives that work at the system or provider level are changing so rapidly that by the time we actually get a measure and follow diseases long enough to be able to make some conclusions, the incentives have already changed. Hence, some of the studies that have been done with staff and group model HMOs may not be relevant now.
The second issue is that we have no idea how patients select health plans and what kind of report cards they are going to need. This is even more problematic for outcomes measures. What kind of outcomes measures will make a difference to patients in the way they select things?
CAROLYN CLANCY: I have noticed that when a study reports news that sounds very good to the industry the study is praised, whatever analysis is being presented. When the news is not so good or might be interpreted in a different light somebody says, "What do you want, this was in the late 1980s?" as if the study were conducted 100 years ago.
AHCPR actually has a fairly large investment in trying to understand how consumers make choices about health care plans, which I agree is an enormously difficult problem. Right now in some markets the big problem is that even if you had perfect measures and tools, with the turnover in plans it is very difficult to actually assess care for the very small population that stays in one place long enough to be measured twice.
ROBERT EPSTEIN: Given the plethora of both generic and disease-specific measures that have been published in this field—at least nine different generic measures have been used—what are your feelings about people coming to some consensus for folks like me who are administering benefits to millions
of individuals and would like to use the same methodology and the same instruments?
A second question is somewhat related to Jack Rowe's comment about patient report cards. What evidence is there to suggest that within-person variability is so large that collecting information on individuals and tracking them over time isn't as good as group-level information?
MATTHEW LIANG: I don't think we should spend much time on this question of which is the preferred measure. Any one of these measures is fine. I think the question is more important than the measure.
ROBERT EPSTEIN: What about the variability issue within person versus within group? This is a big problem, and I wonder if people feel comfortable with reporting individual scores back to patients?
CAROLYN CLANCY: I think that is an open question, and it is very unclear what the right level of accountability is. Right now there is a lot of interest in tracking accountability at the plan level. However, if a plan gets a score that is the equivalent of a B-minus and wants to fix that score, the question is where the problem is. A lot of our measures and current methods aren't very good at helping plans determine figure that. We assume they will figure it out internally, but exactly how you would look at the level of the provider or another level, is, I think, a big challenge.
In terms of consensus, I do think that this is the value of the report card movement. It will create some consensus. I think the greater challenge is the actual state of data systems. For most of the measures that exist in rheumatology now, you would have a very hard time going to most health care systems and actually being able to get the data that you need.
MATTHEW LIANG: Could I just add one comment about the Health Plan Employer Data and Information Set (HEDIS), which is the industry standard? If you look at it, it is very frightening for people who are interested in rheumatic and musculoskeletal disease. The measures are mostly process oriented, and there is not one single item that reflects how people on a population level might be doing in terms of getting care for arthritis and musculoskeletal disease. I think that really must be corrected given the morbidity of the population.
ROBERT NEWCOMER: Dr. Tarlov, your S-shaped curve is a very appealing concept to me. My question is whether you have any optimism about risk-adjusted reimbursement as a way to solve what may be the cost and access to care issue indicated by your data?
ALVIN TARLOV: I think that when the nation established the managed care system it was to solve a problem, runaway costs. Managed care has done that. Now I think managed care has become the normative system, we are stuck with it, and it is going to take a couple of decades to work out the problems with it and to arrive at a health services state in which we are confident that we are working for quality and improved health outcomes.
Will risk-adjusted reimbursement do that? I don't think so. I think that the methodology does exist right now to risk-adjust, but it is complex and subject to perversion. From a practical point of view I don't think that this is a worthwhile approach to improving the nation's health. I do think that outcomes-driven systems with proper incentives to achieve good results are desirable.
| This page in the original is blank. |