1
Introduction
The long-range plans of NASA include possible human exploratory missions to the moon and Mars within the next 25 years. There are three potentially serious health effects for crew members that need to be controlled or mitigated before such relatively long-term missions beyond low Earth orbit can be initiated:1 (1) the effects of microgravity on human physiology and the effects, if any, on cell biology and biochemistry; (2) the psychosocial aspects of long-duration confinement in microgravity with no escape possible; and (3) the biological effects of exposure to radiation in space. The last concern is a serious one because the levels of radiation in space are high enough and the missions are long enough that adequate shielding is necessary to minimize carcinogenic, cataractogenic, and possible neurologic effects for crew members. A question still to be answered is what will provide the necessary protection, for the extent of a mission, against the biological effects of high-energy galactic cosmic ray particles ranging from energetic protons with low mean linear energy transfer (LET) to nuclei of high atomic number and very high energies (HZE) with high LET, and against the effects of transient radiation in solar particle events.*
This report summarizes current knowledge of the types and levels of radiation to which crews will be exposed in space and discusses the range of possible human health effects that need to be protected against (Chapters 1 and 2). It points out that recent reductions in facilities for radiation research raise concerns about how best to acquire needed new knowledge. The report goes on to suggest other steps to be taken and the types of experiments needed to reduce significantly the level of uncertainty regarding health risks to human crews in space (Chapter 3). In Chapter 4 the task group recommends priorities for research from which NASA can obtain the information needed to evaluate the biological risks faced by humans exposed to radiation in space and to mitigate such risks. It outlines, in general terms, the commitment of resources that NASA should make to carrying out these experiments in order to accomplish an effective shielding design in time for a possible mission launch to Mars in 2018, a year allowing for energetically favorable flight trajectories. The final chapter of the report addresses additional issues pertinent to carrying out studies on the effects of radiation, and the report appendixes provide additional details and clarification as appropriate.
Statement of Problem
The knowledge needed to design adequate shielding has both physical and biological components. Knowledge is needed of the distribution and energies of radiation particles present behind a given shielding material as a
result of the shield being struck by a given type and level of incident radiation. Equally important is knowledge of the effects of a given dose on relevant biological systems for different radiation types. Each of these components involves significant uncertainty that must be reduced to permit effective design of shielding, given that the level of uncertainty governs the amount of shielding. It is only prudent to design shielding that will protect space crew members from the predicted, but uncertain, high levels of biological effects from their exposure to radiation. At the same time, excess shielding, based on current cost estimates, would impose an excess expenditure at the level of tens of billions of dollars.2
An understanding of the scope of needed biological and physical data requires an explanation of certain aspects of radiation behavior and the biological impact. HZE particles impinging on shielding, or on human tissue, result in very dense ionization tracks (high LET) with numerous fragments that produce a spectrum of other energetic nuclei, protons, neutrons, and heavy fragments. The numbers of these other nuclei depend on the nature of the shielding and its mass per unit area. The energy loss of the individual particles depends on their types and energies. Thus each particle contributes to the radiation dose and biological response, which are dependent on the number of particles, their types, and their energies. The theoretical calculations of doses per particle type obtained thus far for relevant shielding materials must be verified by ground-based experiments, because the radiation field rate in space is too complex for sufficient experimental analysis. At the present time, the uncertainties in these measurements amount to a factor of ~2 or more (see “Estimates of Uncertainty in Radiation Risk Factors,” Chapter 2).
Ionizing radiation either directly affects cellular macromolecules or reacts with water to produce free radicals that affect these macromolecules by so-called indirect effects. These effects are mitigated somewhat by the presence of free radical scavengers in the surrounding medium. The scavengers are useful in reducing the effects of low-LET radiation but do not seem to result in any significant decrease in the damage caused by high-LET radiation.
The biological effects of fast charged particles depend on the nature of the particle (its charge and velocity) and on the specific biological end point under observation (e.g., cell killing, mutation at a specific genetic locus, chromosomal alterations, cell transformation in vitro, and tumor induction). The relative biological effectiveness (RBE) is taken as the ratio of the dose of gamma rays required to produce a specific effect to the dose of particle radiation required to produce the same level of effect. The RBE depends on the type of particle and the biological effect under consideration and may vary with the magnitude of the biological effect. More importantly, RBE varies greatly with the LET of the particle. For example, high-energy protons may have an RBE value approaching 1.0, whereas high-energy iron nuclei may have an RBE value approaching 40. For tumor induction in animals exposed at lower doses, the relationship between RBE and LET is known for only one tumor site—the Harderian gland in mice. As there are no equivalent data for tumor induction in humans for different LET values, it is necessary to extrapolate from cell and scanty mouse data to evaluate human risk.
Human radiation risk data, still being collected, are available from the analysis of cancer induction in the Japanese individuals exposed to acute doses of radiation resulting from the atomic bombs.3 These doses are not known precisely. As this radiation was primarily low LET, in order to estimate risks to humans in spaceflight conditions one must extrapolate from the RBE vs. LET data for cells in culture and small mammals to humans. In addition, one must extrapolate from the risks from acute exposures of humans to the low-dose-rate chronic exposures involved in space missions (except for the relatively acute exposures from solar particle events). As a general rule, as the dose rate decreases, the biological effect from a given dose also decreases. This dose reduction, in going from acute to chronic exposure, also depends on the biological system and may range from a factor of 2 to 10.4 The dose rate reduction factor for HZE particles is not well known but is probably closer to 1.5 Two other factors that must be considered, but whose impacts are currently unknown, are the effects of biochemical or cellular repair reactions following exposure to HZE particles and the effects of microgravity on such reactions. Thus, in estimating the risks to humans exposed to radiation in space, the uncertain factors are the radiation fields behind the shielding and the extrapolation, via cell culture and animal experiments, from the uncertain risks posed by acute low-LET exposure to risks posed by chronic high-LET exposure.
To quote Curtis, Nealy, and Wilson, “Uncertainties in these numbers are difficult to estimate but a rough analysis leads to a 10–15% uncertainty in the initial charged particle spectra, a 50% uncertainty in the radiation transport calculation, a factor of 2–3 uncertainty in the risk coefficients for low-LET radiation (most of which is due to uncertainty in the dose and dose rate effectiveness factor) and perhaps a factor of 2–5 uncertainty in the risk cross sections at high-LET. Thus, an overall uncertainty in the risk of radiation-induced cancer of a factor of 4–15 for a space crew in the galactic cosmic ray environment appears to exist at the present state of our knowledge.” 6 Obviously, these uncertainties have, themselves, large uncertainties.
Contributions and Use of Past Radiation Research
In 1961, an ad hoc working group was appointed by the Space Science Board of the National Research Council (NRC) to provide scientific advice on the radiation environment in space associated with a manned lunar landing. A report was prepared for NASA and the Department of Defense (DOD).7 In 1964, this group, known as the Space Radiation Study Panel, was reconstituted with the charge to (1) establish a scientific and philosophical basis for determining radiation-protection criteria for manned spaceflight operations; (2) identify the biological responses in humans relevant to mission success or failure and determine their relative order of importance; (3) propose, where possible, interim estimates of dose-response relationships for those responses of greatest importance to missions of up to 3 years' duration; and (4) recommend research programs required to fill gaps representing deficiencies in current knowledge relative to accomplishment of the above objectives. The panel's report, Radiobiological Factors in Manned Space Flight, was published in 1967 and still remains an essential reference.8 It was the main source of information for the guidelines recommended in 1970 by the NRC to NASA9 for establishing radiation exposure limits for space crew members. The recommendation was that the career limit for whole-body exposure to radiation be 4 Sv.10 Additional limits were suggested to prevent or limit the effects of radiation on the skin (12 Sv), the testes (2 Sv), and the lens of the eye (6 Sv).11 These recommendations were the basis of NASA's radiation protection program for the next 20 years.
In the 1967 report it was concluded that “the present knowledge of man's responses to radiation, particularly under the conditions anticipated in space, does not permit establishment of dose-effect relationships to the degree of accuracy desired for spacecraft design and operational planning” (p. 267).12 The panel predicted correctly that it would take many decades before such accuracy would be achieved. On a more hopeful note, it surmised that observations on humans and radiobiological research would result in a better assessment of radiation risks in space.
In succeeding years, considerable progress has been made toward improved understanding of the risks posed by exposure of humans to ionizing radiation. Most of this information has been gained from studies of the effects of radiation in general and not from investigations aimed directly at answering questions about the effects of radiation in space. For example, the 1970 NRC-recommended radiation protection exposure limit of 4 Sv to the whole body was based on estimates for the risk of excess leukemia because there were no adequate risk estimates for solid cancers.13
In the late 1980s it was clear that a reexamination of the radiation exposure limits was required. Both the missions and the makeup of the astronaut corps were changing, and perhaps most importantly, there were more data about radiation risks. In 1989, the National Council on Radiation Protection and Measurements (NCRP) issued its report Guidance on Radiation Received in Space Activities, which had been requested by NASA.14 This report introduced different career limits depending on gender and the age of onset of exposure in space. The career equivalent dose limits that were recommended were based on a lifetime excess risk of cancer mortality of 3 × 10-2 per 1 Gy of low-LET, acute radiation.15 The report's scope was limited to low-Earth-orbit missions, although it considered the radiation environment in deep space and the biological effects of high-Z high-energy (HZE) particles. Recommendations for protection against deterministic effects were also made in 1989. The career limit of 12 Sv recommended in 1979 by the NRC for skin was reduced by the NCRP in 1989 to 6 Sv, and the limit for the lens of the eye was reduced from 6 Sv to 4 Sv.16 Since 1989, the estimates of cancer risk based on studies of atomic bomb survivors have been increased significantly, and the NCRP will issue new recommendations in the near future.
Current Understanding of Biological Effects of Radiation
Types of Effects
For settling radiation protection standards, the division of biological effects that are important for human health into stochastic and deterministic effects has been useful.
Stochastic effects are considered to be due to radiation-induced changes randomly distributed in the DNA of single cells that may lead to cancer or genetically transmissible effects, depending on the target cells. Cancer occurs after a long latent period: after 2 or more years in the case of leukemia and, in the case of solid cancers, within a period ranging from 2 years to decades. It is assumed that the frequency of such effects increases with dose without a threshold and that the severity of the effect is independent of dose. Stochastic effects are the most important consideration in setting protection limits for human populations exposed to radiation at low doses. It is important to note that factors such as radiation weighting factors or quality factors apply only to stochastic effects in the dose range pertinent to radiation protection. Based on studies of atomic bomb survivors at Hiroshima who were exposed to acute levels of mainly gamma rays but also fission neutrons at very high dose rates, estimates for the risk of contracting leukemia have been refined, 17 and there are also data on mortality and the incidence of solid cancers at more than 20 sites in the human body.18,19 (The precise contribution of the fission neutrons to the total dose at Hiroshima is poorly known but is not considered to be a major contributor to the risk of cancer to those exposed at Hiroshima.) In 1991, the International Commission on Radiological Protection (ICRP)20 included leukemia and eight specific sites of solid cancers in its estimates of the probability of an individual contracting a fatal cancer after whole-body exposure to low-LET radiation at 1 Gy and at a high dose rate. The estimated probabilities were 7.12 × 10-2 per person based on a multiplicative projection model and 4.16 × 10-2 per person using an additive model. 21
Deterministic effects, previously termed nonstochastic effects, occur only after exposure to relatively high doses and affect cell populations to the detriment of specific organs or whole organisms. These effects can range from acute radiation sickness to hair loss or nausea. In contrast to stochastic effects, deterministic effects are dose dependent in both frequency and severity. Deterministic effects may occur early, in a matter of hours or days, or late, after many months or even later. Radiation protection standards are set to prevent deterministic effects, whereas standards to protect against stochastic effects are selected to limit effects to an “acceptable” level.
Effects Induced by Protons.
While the estimated risks of adverse biological effects calculated from the data on atomic bomb survivors are the basis for current radiation protection limits, the types of radiation received by the atomic bomb survivors differ markedly from the types of radiation to which space crews would potentially be exposed. In deep space, the radiation environment consists mainly of galactic cosmic radiation (GCR) at a low fluence rate. In the energy range from 100 MeV per nucleon to 10 GeV per nucleon, the GCR consists of 87 percent protons, 12 percent helium ions, and 1 percent heavier ions.22
Protons are also the major component of solar particle events (SPEs), with a smaller contribution by helium and heavier ions emitted from the sun. A major difference between SPE radiation and the GCR is the much greater transient fluence in SPE radiation, which in very large SPEs can be 1010 protons cm-2 with energies greater than 10 MeV.23
No data are available for most of the deterministic effects induced in humans by exposure to protons, and very limited data are available from studies done on animals. One such study was carried out from 1963 to 1969 by the U.S. Air Force (USAF) and NASA to determine the RBE of various types of radiation found in space.24 About 2000 rhesus monkeys and 5000 mice were irradiated with protons of energies ranging from 32 to 2300 MeV obtained using cyclotrons at various institutions. An attempt to simulate exposure to SPEs was made using 10 MeV plus 110 MeV protons at NASA's Space Radiation Effects Laboratory synchrocyclotron at Langley Research Center. Exposures to electrons and x rays were also carried out to enable comparison of the effects of radiation of different qualities. The study showed that the biological effects of the higher-energy and penetrating protons (>138 MeV) were similar to those caused by 2-MeV x rays and 60Co gamma
rays. The RBE for acute mortality was about 1.0 to 1.1. Other studies on the acute biological effects of high-energy protons suggest an RBE of about 1 compared with that for x rays. In the case of 160-MeV protons, Urano et al. found RBE values ranging between 0.8 and 1.3 for killing of jejunal crypt cells, skin damage, and effects on the lens of the eye in exposed mice.25 An exception to these RBE values of about 1 was the indirect finding by Storer et al. of higher RBE values, namely 2.4 and 4.9 for 30-day lethality and testicular atrophy, respectively, in mice.26
In the treatment of human cancer with irradiation by protons, an RBE of 1.1 has been used for planning purposes, and this value does not appear to underestimate the effectiveness of the protons.
A subpopulation of the primates studied in the USAF/NASA project has been monitored for almost 30 years for late effects such as cancer, cataracts, and shortening of life. The follow-up of these animals has been especially important in assessing the risk of cataractogenesis, because no estimates exist of the risk of cataract induction in humans following proton irradiation. Significant lenticular opacifications have occurred in monkeys about 20 to 24 years after exposure to 55-MeV protons at 1.25 Gy and higher levels.27 Results obtained from these experiments suggest that the dose-response relationship for induction of cataracts by protons are similar to that seen with low-LET radiation.
In the case of stochastic effects, there are no data for cancer induction in humans exposed to protons. The USAF/NASA study of primates discussed above has found life shortening and cancer induction to be dependent on dose but not on proton energy.28 In the groups exposed to 138 to 2300 MeV protons, leukemia was not a major cause of death. In males, solid cancers were the major cause of life shortening. The dose-response relationships that could be derived were consistent with those found in other experimental studies of the effects of low-LET radiation, namely curvilinear. In females, endometriosis was a major effect that contributed to radiation-induced life shortening. Another finding of note was the increased incidence of malignant brain tumors in the group exposed at 55 MeV.29 The increase can be attributed to the unusual dose distribution in the brain that resulted from the rotation of the monkeys during exposure, the limited penetration of protons with this level of energy, and probably the young age of the animals. There is no reason to believe that the finding of increased brain tumors is associated with the types or energies of the radiation, but it is related to the doses incurred. In a study of the induction of tumors in mice exposed to 60-MeV protons, Clapp et al. found no RBE values greater than about 1.0.30 Burns et al. reported an RBE of about 3 for the induction of skin tumors in rats exposed to 10-MeV protons compared with electrons.31
When considering astronaut safety it should be remembered that the studies discussed above were carried out with single high-dose-rate exposures that are very different from the exposures occurring in space. Burns et al. noted a reduction in the tumorogenic effect with the increasing fractionation of 10-MeV proton irradiation, an indication of recovery.32 Furthermore, the curvilinear response to single doses was similar to the response to low-LET radiation, indicating that the protons have attributes of both low-LET radiation and, because the RBE is ~3, of high-LET radiation. Obviously, more data, using protons of several energy ranges, are needed to estimate human cancer risks from galactic protons.
Effects Induced by Heavy Ions
The deterministic effects of exposure to heavy ions have been studied in experimental animals. The RBE of various ions was determined for effects that result from cell killing in the gut, testes, and bone marrow, and in vitro systems.33 As Figure 1.1 indicates, the RBE increases with increasing LET, reaches a maximum between 100 and 200 keV/µm, and decreases rapidly at higher LET values. The maximum RBE values for effects involving cell killing have been found to be between 2.0 and 3.0.34 In the case of cataract induction, which is discussed in Chapter 2, the RBE values obtained for rats and mice by Merriam35 suggest much higher RBE values, 40 to 50 at low doses. Although the work of Lett and coworkers and of Worgul and coworkers suggests that it may be possible, with further data, to extrapolate across species to obtain RBE values for cataract induction, current data do not allow reliable estimation of the risk of cataract induction occurring in humans as a result of exposure to radiation in deep space.36,37
Observations on radiotherapy patients indicate that very high doses of low-LET radiation give rise to deterministic-type damage. HZE particles produce high-dose ionization tracks and kill the cells they traverse. The concern about such microlesions in the central nervous system resulting from traversal of cells by heavy
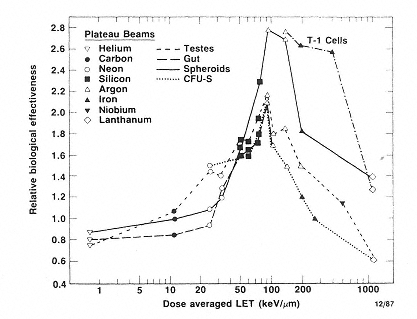
Figure 1.1
RBE-LET relationship for inactivation of CFU-S, intestinal crypt clonogenic cells, cells in spheroids, human T-1 cells, and the loss of testis weight. SOURCE: National Council on Radiation Protection and Measurements (NCRP). 1989. Guidance on Radiation Received in Space Activities. Recommendations of the National Council on Radiation Protection and Measurements. NCRP Report No. 98. National Council on Radiation Protection and Measurements, Bethesda, Md. Reprinted by permission.
charged particles, such as iron, has not been eliminated, nor has any evidence been produced to show that the concern is justified. The question of the effects of heavy ions on the central nervous system is discussed in detail in Chapter 2.
The main concern about stochastic effects is the risk of cancer induction. It is agreed that the RBE for carcinogenesis increases with the increasing LET of the radiation. The evidence comes largely from animal experiments with fission neutrons but also from data on induction of lung tumors in humans exposed to alpha particles from radon.38 There has been only one systematic study of the relationship between the LET of heavy ions and the RBE values of the ions for tumor induction, which was carried out on the Harderian gland of mice.39 Although this gland is a suitable epithelial system, it is the only tumor model that has been examined over the range of LET values encountered in space. Hence, it is not possible to generalize on the basis of these data about RBE values for induction of cancer in important sites of the human body such as the breast, lung, and bone marrow. The data on the Harderian gland tumors show a rise in RBE with increasing LET, reaching a maximum and a plateau of about 30 at about 100 to 200 keV/µm.40 However, unlike the case of the RBE-LET relationship for cell killing and mutation,41 there is no evidence of a rapid decrease in RBE at higher LET values. This fact raises an important question, because radiation protection standards are based on dose equivalents described by the quality factor Q as a function of LET, such that dose equivalent = Q × dose. The function Q is established by consensus and is restricted to stochastic effects. The latest consensus Q-LET relationship, adopted by the ICRP in 1991, conforms to the data for cell killing, but it is not yet clear that the relationship holds for the induction of tumors.42 Similar data, namely, those showing the initial slopes of the dose-response curves for cancer induction in relevant tissues as a result of exposure to representative heavy ions, are required for application of the method suggested by Curtis 43 for estimating of the risks to humans. Furthermore, the proton irradiations associated with those data were performed with a (slightly) different animal model, adding to the level of uncertainty about the biological effects of the heavier ions relative to the effects of protons. The data for induction of skin tumors by argon ions44 support the expectation of a high RBE for induction of tumors by heavy ions but do not allow any more precise estimate of what
quality factor should be used in estimates of the risk of cancer induction in humans. Because of the importance of establishing the precise Q-LET relationship for cancer induction, more experimental studies are required.
References
1. Space Studies Board, National Research Council. 1993. Scientific Prerequisites for the Human Exploration of Space. National Academy Press, Washington, D.C.
2. Wilson, J.W., Cucinotta, F.A., Shinn, J.L., Kim, M.H., and Badavi, F.F. 1997. Shielding Strategies for Human Space Exploration: Introduction. Chapter 1 in: Shielding Strategies for Human Space Exploration: A Workshop (John W. Wilson, Jack Miller, and Andrei Konradi, eds.). NASA, Washington, D.C., forthcoming.
3. Board on Radiation Effects Research, National Research Council. 1990. Health Effects of Exposure to Low Levels of Ionizing Radiation: BIER V. National Academy Press, Washington, D.C.
4. Board on Radiation Effects Research, National Research Council, 1990, Health Effects of Exposure to Low Levels of Ionizing Radiation.
5. Blakely, E.A., Ngo, F.Q.H., Curtis, S.B., and Tobias, C.A. 1984. Heavy-ion radiobiology: Cellular studies. Adv. Radiat. Biol. 11: 295–389.
6. Curtis, S.B., Nealy, J.E., and Wilson, J.W. 1995. Risk cross sections and their application to risk estimation in the galactic cosmic-ray environment. Radiat. Res. 141: 57–65.
7. Space Science Board, National Research Council. 1961. First Summary Report. Man in Space Committee, Working Group on Radiation Problems, National Academy of Sciences, Washington, D.C.
8. Space Science Board, National Research Council. 1967. Radiobiological Factors in Manned Space Flight. National Academy of Sciences, Washington, D.C.
9. Space Science Board, National Research Council. 1970. Radiation Protection Guides and Constraints for Space-Mission and Vehicle-Design Studies Involving Nuclear Systems. Radiobiological Advisory Panel, Committee on Space Medicine. National Academy of Sciences, Washington, D.C.
10. Space Science Board, National Research Council, 1970, Radiation Protection Guides and Constraints for Space-Mission and Vehicle-Design Studies Involving Nuclear Systems.
11. Space Science Board, National Research Council, 1970, Radiation Protection Guides and Constraints for Space-Mission and Vehicle-Design Studies Involving Nuclear Systems .
12. Space Science Board, National Research Council, 1967, Radiobiological Factors in Manned Space Flight.
13. Space Science Board, National Research Council, 1967, Radiobiological Factors in Manned Space Flight.
14. National Council on Radiation Protection and Measurements (NCRP). 1989. Guidance on Radiation Received in Space Activities. Recommendations of the National Council on Radiation Protection and Measurements. NCRP Report No. 98. National Council on Radiation Protection and Measurements, Bethesda, Md.
15. NCRP, 1989, Guidance on Radiation Received in Space Activities.
16. NCRP, 1989, Guidance on Radiation Received in Space Activities.
17. Preston, D.L., Kusumi, S., Tomonaga, M., Izumi, S., Ron, E., Kuramato, A., Kamada, N., Dohy, H., Matsuo, T., Nonaka, H., Thompson, D.E., Soda, M., and Mabuchi, K. 1994. Cancer incidence in atomic bomb survivors. Part III. Leukemia, lymphoma and multiple myeloma, 1950–1987. Radiat. Res. 137: S68–S97.
18. Pierce, D.A., Shimizu, Y., Preston, D.L., Vaeth, M., and Mabuchi, K. 1996. Studies of the mortality of Atomic bomb survivors. Report 12, Part I. Cancer: 1950–1990. Radiat. Res. 146: 1–27.
19. Thompson, D.E., Mabuchi, K., Ron, E., Soda, M., Tokunaga, M., Ochikubo, S., Sugimoto, S., Ikeda, T., Terasaki, M., Izumi, S., and Preston, D.L. 1994. Cancer incidence in atomic bomb survivors. Part II: Solid Tumors, 1958–1987. Radiat. Res. 137: S17–S67.
20. International Commission on Radiological Protection (ICRP). 1991. 1990 Recommendations of the International Commission on Radiological Protection. ICRP Publication 60. Annals of the ICRP 21. Pergamon Press, Elmsford, N.Y.
21. ICRP, 1991, 1990 Recommendations of the International Commission on Radiological Protection.
22. Simpson, J.A. 1983. Introduction to the Galactic Cosmic Radiation. Composition and Origin of Cosmic Rays (M.M. Shapiro, ed.). Reidel Publishing, Dordrecht, Netherlands.
23. Simpson, 1983, Introduction to the Galactic Cosmic Radiation.
24. Dalrymple, G.V., Lindsay, J.R., Mitchell, J.C., and Hardy, K.A. 1991. A review of the USAF/NASA proton bioeffects project: Rationale and acute effects. Radiat. Res. 126: 117–119.
25. Urano, M., Verkey, L.J., Guitein, M., Lepper, J.E., Suit, H.D., Mendrondo, O., Gragoudos, E., and Koehler, A. 1984. Relative biological effectiveness of modulated proton beams in various murine tissues. Int. J. Oncol. Biol. Phys. 10: 509–514.
26. Storer, J.B., Harris, P.S., Furchner, J.E., and Langham, W.H. 1957. The relative biological effectiveness of various ionizing radiations in mammalian systems. Radiat. Res. 6: 188–288.
27. Dalrymple et al., 1991, A review of the USAF/NASA proton bioeffects project.
28. Dalrymple et al., 1991, A review of the USAF/NASA proton bioeffects project.
29. Yochmowitz, M.G., Wood, D.M., and Salmor, Y.L. 1985. Seventeen-year mortality experience of proton radiation in Macaca mulatta. Radiat. Res. 102: 14–34.
30. Clapp, N.K., Darden, D.B., Jr., and Jernigan, M.C. 1974. Relative effects of whole-body sublethal doses of 60-Me V protons and 300-kVp x rays on disease incidence in RF mice. Radiat. Res. 57: 158–186.
31. Burns, F.J., Hosselet, S., and Garte, S.J. 1989. Extrapolations of rat skin tumor incidence: Dose, fractionation and linear energy transfer. Pp. 571–582 in: Low Dose Radiation: Biological Bases of Risk Assessment (K.F. Baverstock and J.W. Stather, eds.). Taylor and Francis, London.
32. Burns, F.J., Albert, R.E., Vanderlaan, M., and Strickland, P. 1975. The dose-response curve for tumor induction with single and split doses of 10 MeV protons. Radiat. Res. 62: 598 (abstract).
33. NCRP, 1989, Guidance on Radiation Received in Space Activities.
34. Ainsworth, E.J. 1986. Early and late mammalian responses to heavy charged particles. Adv. Space Res. 6: 153–165.
35. Merriam, G.R., Jr., Worgul, B.V., Medvedovsky, C., Zaider, M., and Rossi, H.H. 1984. Accelerated heavy particles and the lens. I. Cataractogenic potential. Radiat. Res. 98: 1, 129–140.
36. Lett, J.T., Lee, A.C., and Cox, A.B. 1991. Late cataractogenesis in rhesus monkeys irradiated with protons and radiogenic cataract in other species. Radiat. Res. 126: 147–156.
37. Worgul, B.V., Medvedovsky, C., Huang, Y., Marino, S.A., Randers-Pehrson, G., and Brenner, D.J. 1996. Quantitative assessment of the cataractogenic potential of very low doses of neutrons. Radiat. Res. 145: 343–349.
38. Committee on the Biological Effects of Ionizing Radiation, National Research Council. 1988. Health Effects of Radon and Other Internally Deposited Alpha-Emitters: BEIR IV. National Academy Press, Washington, D.C.
39. Alpen, E.L., Power-Risius, P., Curtis, S.B., DeGuzman, R., and Fry, R.J.M. 1994. Fluence-based relative biological effectiveness for charged particle carcinogenesis in mouse harderian gland. Adv. Space Res. 14: 573–581.
40. Alpen et al., 1994, Fluence-based relative biological effectiveness for charged particle carcinogenesis in mouse harderian gland.
41. NCRP, 1989, Guidance on Radiation Received in Space Activities. See also Blakely et al., 1984, Heavy ion radiobiology: Cellular studies.
42. ICRP, 1991, 1990 Recommendations of the International Commission on Radiological Protection.
43. Curtis, S.B. 1994. Single-track effects and new directions in GCR risk assessment. Adv. Space Res. 14: 855–894.
44. Burns et al., 1989, Extrapolations of rat skin tumor incidence.








