3
Neurobiology of Addiction: An Overview
This chapter presents a brief overview of the underlying neurobiology of drug addiction and suggests a perspective with which to put the neurobiological findings in context with the social, psychological, and environmental aspects of drug addiction. The chapter is not intended to be a comprehensive review of these topics. Rather, its intent is to demonstrate for new investigators, students, university administrators, and policymakers the richness of basic research regarding abuse of alcohol and other drugs and to provide the lay reader with a greater understanding of the role of the brain in the transition from drug use to addiction. Some of the key advances in research into the neurobiology of addiction are also summarized in Appendix F.
The role of dopamine as a neurotransmitter in the brain reward circuitry is highlighted in this chapter, because it relates to the four classes of drugs discussed in this report—nicotine, alcohol, stimulants, and opioids. Although there are some important differences among them, these drugs share common reward pathways in the brain. Fundamental neurobiological research as outlined in this chapter, together with findings from clinical and behavioral and social science research (see Chapter 4), are providing researchers with neurobiological answers that offer better ways to understand, prevent, or treat drug addiction.
At the heart of our current understanding of addiction is the idea that in vulnerable individuals, the disease of addiction is produced by the interaction of the drugs themselves with genetic, environmental, psychosocial, behavioral, and other factors, which causes long-lived alterations in the biochemical and functional properties of selected groups of neurons in the brain. In particular, addictive drugs, when taken with adequate dose, frequency, and chronicity, appear to
commandeer circuits intimately involved in the control of emotion and motivation, thus impairing the insight and volitional control of the addicted person. At the same time, the alterations produced by chronic drug use facilitate the formation of deeply ingrained emotional memories that predispose to drug craving and hence to relapse.
Thus, in developing a conceptual understanding of addiction, it must be acknowledged that compulsive drug use cannot be understood from any single level of analysis. The development of drug addiction, at a minimum, involves the properties of the drugs involved, the neural circuits on which they operate,
|
BOX 3.1 Animal Models: Examples from Alcohol Research Experimental laboratory animal models remain an essential resource in modern biomedical research because large segments of investigation require the intact animal. This is particularly true for behavioral disorders, including the addictive disorders, and self-administration models are particularly useful for studying all psychoactive substances in animals. Self-administration models, although they vary in specific aspects, all involve making the animals capable of administering drugs to themselves by performing some action, such as pressing a bar or entering a chamber. Consistent self-administration of alcohol can be established in primates, rodents, and other animals. These techniques have contributed to better understanding of the behavioral and biological conditions of ethanol intake. In many aspects, these procedures mimic the "learning to drink" phenomenon evident in humans. These procedures are used to study the pharmacodynamic effects of ethanol, such as acute intoxication, tolerance, and dependence, and to study the medical consequences of chronic ingestion; for example, fetal alcohol syndrome (FAS), brain damage, and liver disease. Self-administration models are particularly useful in studying the behavioral patterns and consequences of ethanol ingestion. The finding that food deprivation leads to increased ethanol intake in ethanol self-administration models provides a better understanding of the relationship between drug use and eating disorders. Alcohol is also commonly taken with other drugs and the self-administration model, where animals, like humans, take in substantial quantities of drugs and become dependent, also provides a means to examine the outcomes of multiple drug use. Further, animal self-administration models provide opportunities to examine the effects of potential medications for the treatment of alcohol dependence (e.g., naltrexone). Test medications are also evaluated to determine whether they sustain drug taking or might be abused. The strength of this model is such that the Food and Drug Administration (FDA) and Drug Enforcement Administration (DEA) rely heavily on self-administration evaluations to determine the "abuse liability" of drugs for purposes of scheduling under the Controlled Substances Act. |
an individual's genetic makeup and developmental experiences, the presence or absence of psychological distress or psychiatric illness, and the sociocultural context in which drugs might be used. A sophisticated understanding of how the brain works recognizes that an individual's life experience and social context exert powerful effects on the brain and, therefore, on behavior.
THE BRAIN'S COMMON "REWARD" PATHWAY
Two lines of investigation led to the conclusion that addictive drugs, although chemically different from one another, all affect a brain system involved in the control of motivated behavior. The first set of experiments were performed in rats in the 1950s and involved stimulating discrete brain regions (Olds and Milner, 1954). In these experiments, the discovery was made that there were a small number of brain regions in which stimulation was "pleasurable" or "rewarding" because the rats would press a lever tens of thousands of times in succession, ignoring normal needs for food, water, and rest, to gain electrical stimulation (self-stimulation; see also Box 3.1). In the popular literature these regions were called "the pleasure center" but scientifically they are better described as "brain reward regions" (i.e., regions in which electrical activation profoundly reinforces the lever-pressing behavior) (Wise, 1978).
A great deal of research since the 1950s has established that the major brain circuit mediating this type of reinforcement is a pathway extending from the ventral tegmental area (VTA), an area within the midbrain (a part of the brainstem), to a structure called the nucleus accumbens (NAc) (Figure 3.1). The NAc is particularly interesting because it occupies the crossroads between a group of brain structures, called the limbic system, that are primarily involved in the control of emotion, and another part of the brain, the striatum, that is involved in the initiation and control of movement. Nerve cells (neurons) in the brain communicate with each other using diverse chemical substances called neurotransmitters. One of the major neurotransmitters used in the brain reward circuit is dopamine (Wise, 1978).
The depiction of the interaction of a nerve cell (neuron) in the VTA with a neuron in the NAc (Figure 3.1) represents a general model of neuronal communication. Neurons communicate using chemical neurotransmitters such as dopamine—one of more than 100 chemical substances that are used for cell-to-cell communication in the nervous system. The neurotransmitter is stored in the transmitting neuron and released when the neuron is stimulated. The transmitter diffuses across a specialized divide called a synapse and binds to receptors, which are specialized recognition sites. Depending on the transmitter, the receiving or "postsynaptic" neurons will be excited or inhibited, or will undergo more complex biochemical alterations (Cooper et al., 1996).
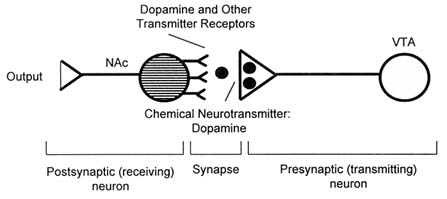
FIGURE 3.1 Schematic diagram of the brain reward circuitry. NOTE: NAc = nucleus accumbens; VTA = ventral tegmental area.
The second set of clues concerning the substrates of addiction came from more recent work which showed that each of these highly addictive drugs mimics or enhances the actions of one or more neurotransmitters in the brain that are involved in the control of the brain reward circuit (Cooper et al., 1996). The opioids mimic endogenous opioid-like compounds called endorphins; cocaine and related drugs enhance the actions of dopamine itself; nicotine mimics the action of acetylcholine (another transmitter) at its nicotinic receptors; and alcohol, among its many effects, facilitates the activation of a particular receptor for gamma-aminobutyric acid (GABA), one of the most prevalent neurotransmitters throughout the brain. Although each of these four mimicked neurotransmitters has many actions in the brain, they all share one common property: all regulate the activity of the brain reward pathway that extends from the VTA to the NAc (Di Chiara and Imperato, 1988).
Each of these abused drugs also interacts with other neurotransmitter systems and molecules within neurons that act as "second messengers" that effectively translate input to neurons into a variety of intracellular chemical and molecular changes (signal transduction), some of which affect gene expression. In addition, other types of neurotransmitters can be affected in direct and indirect ways. For example, one way neurons regulate the amount of neurotransmitter acting at a second neuron is through reuptake mechanisms, that is, the reabsorption of the transmitter into the presynaptic neuron via specialized channels called "transporters." Cocaine inhibits the neuronal reuptake of serotonin and norepinephrine, as well as dopamine, leaving these neurotransmitters in the synapse and effectively increasing their actions within the synapse by increasing their action on their receptors. These additional actions contribute to the unique properties of each class of abused agents and may enhance or reduce the behavioral
reinforcement associated with activation of dopamine pathways (Koob, 1996).
As described, the major neurotransmitter released by VTA neurons in the NAc is dopamine. Experiments in which dopamine neurons are destroyed, or other experiments in which dopamine receptors are blocked, confirm that this neurotransmitter is necessary for significant brain reward. Cocaine and the related synthetic compound, amphetamine, both facilitate the action of dopamine while also affecting the balance of other neurotransmitters.
DRUG EFFECTS ON BRAIN REWARD SYSTEMS
Normally, following the release of dopamine, its action is terminated by a presynaptic reuptake transporter that takes the dopamine back into the neuron that released it (the presynaptic neuron). Cocaine mimics dopamine enough to bind to the dopamine reuptake transporter, but not enough to be taken up. Hence, cocaine blocks the transporter, causing a buildup of dopamine in the synapse because it is not inactivated by reuptake. Amphetamine causes the presynaptic neuron to release more dopamine, essentially by putting the transporter into "reverse." Thus, both cocaine and amphetamine produce strong stimulation of the dopamine pathway leading to the brain reward centers (Hyman, 1996) (Figure 3.2).
Opioids are hypothesized to act on the brain reward circuitry in at least two ways. There are opioid receptors on neurons in the limbic system, including the NAc; thus opioids may act on these brain regions directly. In addition, opioids can indirectly cause VTA neurons to release more dopamine. The firing rate of VTA dopamine neurons is held at a certain "tonic" or stable level by inhibitory neurons. These inhibitory neurons, in turn, possess opioid receptors. Because opioids are inhibitory, endogenous opioids (e.g., enkephalins) or opioid drugs (e.g., morphine) essentially turn off or decrease the action of these inhibitory neurons (Nicoll et al., 1980) and thereby disinhibit the midbrain dopamine neurons. This results in enhanced release of dopamine and a stronger stimulation of the brain reward centers and imbalance with other neurotransmitters (Johnson and North, 1992; Izenwasser et al., 1993).
Although less well understood, both alcohol (ethanol) (Tabakoff and Hoffman, 1996) and nicotine (Dani and Heinemann, 1996) appear to disinhibit VTA neurons and to cause dopamine release in this same circuit. Ethanol increases the firing of dopamine neurons in the ventral tegmental area (VTA) and increases extracellular dopamine levels in the nucleus accumbens (NAc). The sites at which ethanol exerts it action may be multiple and include a number of modulators of dopaminergic activity in the VTA and the NAc (See Box 3.2). These (and other) neurotransmitter systems and sites may also be involved in mediating negative reinforcement by ethanol, as with drinking for relief of withdrawal symptoms and anxiety (Koob, 1992a).
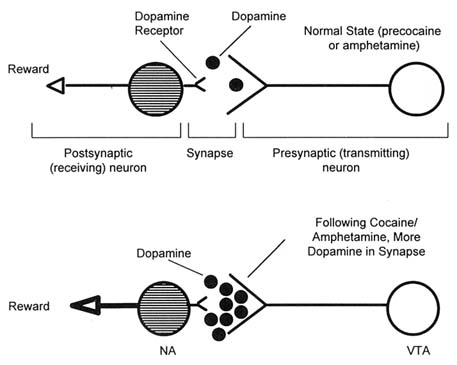
FIGURE 3.2 The effect of cocaine and amphetamine.
NOTE: NAc = nucleus accumbens; VTA = ventral tegmental area.
EMOTION, MEMORY, AND THE TRANSITION FROM USE TO ADDICTION
In addition to the reward pathways, the human brain contains multiple circuits that are involved in the processing of emotion, learning, and conditioning. These "emotional circuits" are adaptive survival circuits; they assign significance (e.g., dangerous, edible, desirable) to individuals, things, and events in the world, and then regulate the functioning of both the input and output circuits of the brain to produce adaptive responses, including escape, approach, and aggression. Connections between brain circuits mediating emotional states and those that mediate the encoding of memories underlie the development of memories with strong or weak emotional components. If strong enough, such memory can be quite simple, as when a child burns his or her finger, and a single experience will dominate subsequent behavior. However, most memories are complex and involve multiple emotional components.
|
BOX 3.2 Effects of Alcohol on Neurotransmission Ethanol differs from other psychotropic drugs in that it produces primary-level effects on multiple receptor systems, ion channels, and signal transduction pathways both in the brain and in peripheral organ systems. Recent evidence has shown that ethanol potentiation of the GABAA receptor requires phosporylation of the gamma 2L splice variant of the GABAA receptor by the gamma isoform of protein kinase C (PKC) (Harris et al., 1995; Wafford and Whiting, 1992; Wafford et al., 1991). Mutant knockout mice without the PKC? isozyme lack ethanol responsive actions on cerebellar membrane preparations and show reduced behavioral and physiological responses to alcohol while retaining sensitivity to barbiturate and benzodiazepine drugs (Harris et al., 1995). However, alcohol also affects the functioning of other systems in a less direct way, which results in an enhancement or decrease in the activity of many other neurotransmitters, ions, enzymes, and other cellular processes in the brain and elsewhere. Because it is freely permeable across cell membranes, ethanol also disrupts intracellular metabolic processes and signal transduction pathways in neuronal cells as well as cells in other organs (e.g., liver, heart, pancreas) (Li, 1983). Studies in the last 15 years have shown that ethanol (in concentrations that produce behavioral effects) affects the function of several membrane receptors which regulate the flow of ions through other membrane molecules that serve as ion channels (ligand-gated ion channels). In this way, ethanol causes an increase in the flow of chloride ions mediated by a specific type of GABA receptor (the GABAA-benzodiazepine receptor complex) and decreases calcium ion flow mediated by other receptor types (the NMDA-glutamate receptor system). Ethanol also affects a specific type of serotonin receptor that mediates ion channel function to enhance the flow of sodium and potassium ions. Cell-to-cell differences as well as regional differences in sensitivity of these ligand-gated ion channels to ethanol have been discovered. Such differences help explain some of the varied behavioral effects produced by ethanol, for example, release of inhibition, sedation, and memory impairment. Chronically, ethanol causes long-term neuroadaptive changes in the function of the GABAA-benzodiazepine receptor complex and the NMDA-glutamate receptor complex, which contribute to the production of chronic tolerance. A second group of signal transduction pathways that ethanol affects are the G-protein coupled and adenylate cyclase related receptors, whose activating ligands include the opioid peptides, dopamine, serotonin, and adenosine. Chronically, ethanol, through actions on G proteins, causes changes in the activity of neurons in pathways mediated by the opioid peptides, serotonin, and dopamine, which also contribute to the development of tolerance (Fitzgerald and Nestler, 1995). |
The dopaminergic pathway that extends from the VTA to the NAc is part of an emotional circuit that likely evolved to motivate behaviors required for survival
and reproductive success, such as sexual behaviors. When something is highly rewarding, memories of that experience are likely to be vivid, complete with positive emotional overtones, and a remembrance of the circumstances under which it was encountered. Thus, the dopaminergic reward pathway likely plays a role in assigning novel stimuli to specific adaptive behavioral repertoires. By perverse serendipity, this adaptive emotional circuit makes brains vulnerable to drug addiction because certain addictive drugs mimic or enhance the actions of neurotransmitters used within it. Thus, drugs of abuse and addiction tap into and ''short circuit" powerful emotional circuitry in the brain (Koob, 1996). An important implication is that drug addiction research may be one of the most promising windows on the neurobiology of emotions elicited by non-drug stimuli. Indeed, addictive drugs provide very important information about how the brain normally functions.
All addictive drugs have actions outside the dopaminergic brain reward circuit as well as within it. The sum total of a drug's actions in the brain is determined by where receptors for that drug are found, and what those receptors do. Because all addictive drugs have both a common site of action—the brain reward pathway—and unshared sites of action, their subjective and objective behavioral properties differ. Thus, for example, both cocaine and opioids are reinforcing, but opioids are sedating, while cocaine is a powerful stimulant (O'Brien, 1996).
Some of the actions of cocaine depend on interactions with serotonergic systems. Cocaine increases extracellular serotonin levels by inhibiting the reuptake of previously released serotonin in much the same way that it inhibits the reuptake of dopamine. There is a diffuse network of serotonin-releasing neurons projecting to the VTA and NAc (Phelix and Broderick, 1995); activation of this system probably facilitates dopamine release in the NAc. The activity of the VTA to NAc neurons that appear to be so important in reward processes are also subject to regulation by several neuropeptides, including the endogenous opioids (as discussed above) and two other brain peptides that function as neuromodulators, cholecystokinin-8 (CCK-8) (Hamilton and Freeman, 1995) and neurotensin (Steinberg et al., 1995). Treatments that affect the release or actions of these neuropeptides may also alter the functions of reward pathways.
The neural locations of the receptors or transporters through which drugs act are obviously of primary importance in determining their effects, but other properties of a drug also appear to be important determinants of the abuse potential of drugs. Drugs are most reinforcing when their level in the brain rises very rapidly. Thus, drugs that are smoked or injected produce far more powerful effects than drugs taken orally, in part because they reach the brain so rapidly when administered in this way. Yet, it is clear, and well represented by the case of alcohol, that orally delivered drugs also can produce profound effects and dependence. It is an obvious but important point that purer or more potent forms of drugs also have more powerful effects on the brain. Purity and route of administration explain why smoked cocaine free base (e.g., "crack" cocaine,
which when smoked, produces volatilized pure drug exposed to the lungs' massive surface area) is far more quickly addictive than powdered cocaine hydrochloride absorbed via the nasal mucosa, which is, in turn, far more quickly addictive than chewed coca leaves, where the cocaine is absorbed more slowly from the mouth and gastrointestinal tract (and partially destroyed by digestive enzymes) (Wilkinson et al., 1980). The form in which a drug is used, however, may also create barriers to use. For example, smoking is not initially natural and may cause coughing and other discomfort. Thus a certain amount of social pressure or support for smoking is needed to get young people over this initial barrier before they find tobacco smoking pleasurable and put themselves at risk for addiction. Taking alcohol or other drugs orally in liquid or tablet form does not present this type of barrier, so initial use may be easier to achieve.
People report that they take drugs to gain pleasure, to produce alterations in consciousness, to conform to the behavior of their peers, and to relieve stress and other negative emotions. However, the fact that certain drugs produce subjective euphoria or relieve dysphoria in humans and are reinforcing in animal models does not necessarily mean that they will produce addiction (i.e., inability to control use despite serious negative consequences; see Chapter 1). Indeed, over time, the addicted person's enjoyment of drug-taking is decreased because tolerance develops or medical complications ensue. Despite diminished enjoyment, the life of the addicted person revolves around the obtaining, using, and recovering from the effects of the drug despite problems at home and work and failure in life roles. The effects of the drug and circumstances of use are somehow so important that the addicted individual may go to great lengths to deny that its use is causing any difficulty. The denial, manipulation, and dishonesty that the addicted person frequently exhibits in the service of ongoing drug use may frustrate and anger family members, colleagues, and caregivers.
A key point in our current understanding of the neurobiology of drug abuse is that there is good evidence that use of a drug at an adequate dose with adequate frequency and chronicity produces long-lived changes in brain functioning. Many of these changes represent compensatory adaptations (homeostatic responses) to excessive bombardment by the drug. It is these drug-induced changes in brain function that produce addiction in some individuals.
The types of long-term changes that addictive drugs produce in the brain now can be divided conceptually into three categories. First, opioids and ethyl alcohol, but not cocaine, produce compensatory adaptations in brain regions that control somatic functions, thus producing physical dependence. As a result, discontinuation of opioids or alcohol can produce a physical withdrawal syndrome, such as the well-known alcohol withdrawal syndrome that includes hypertension, rapid heartbeat, tremor, nystagmus, insomnia, and grand mal seizures (O'Brien, 1996).
Second, all addictive drugs appear to produce adaptations within the brain reward circuitry itself. These are quite complex and far from fully understood. A subset of these adaptations contributes to tolerance, decreasing some of the reinforcing
effects of the drug and therefore contributing to increases in the dosage needed to achieve a desired level of effect. The same types of adaptations that contribute to tolerance also contribute to dependence, that is, putting the brain in a state that will lead to emotional and motivational symptoms of withdrawal (e.g., depressed mood, inability to experience pleasure, and drug craving) following drug cessation. At the same time there is evidence for other types of adaptations in brain reward circuits that may produce sensitization (i.e., the concept that some drug effects may actually get stronger with repeated stimulation). Some aspects of sensitization increase craving for the drug; others, like the syndrome of cocaine-induced paranoia that may develop with chronic use, create novel difficulties for drug users (Satel et al., 1991).
A third category of long-term alteration in brain function that may be produced by addictive drugs in some individuals is the production of powerful "emotional memories" of drug use. There is increasing evidence to suggest that things learned in the context of strong emotional activation are most indelibly etched into distinct memories. Among the most dramatic examples of this phenomenon are seen in individuals suffering from posttraumatic stress disorder (PTSD); for a PTSD survivor, even minor cues can bring the entire traumatic episode flooding back into consciousness, including the related emotions. The dopaminergic circuitry activated by drugs of abuse may be directly involved in setting the strength of memories associated with reinforcement. Thus, places, persons, and bodily feelings that have been connected with drug use may be stored in memory in such a way that cues recalling drug experiences may elicit strong drug cravings (O'Brien, 1976; Wilker, 1973). Well-known examples include the craving for a cigarette produced by a large meal or the reexperiencing of some withdrawal symptoms by detoxified heroin addicts who return to a site at which they had previously used drugs. The relationship of these cue-dependent memories to relapse in previously detoxified individuals is currently a matter of study (IOM, 1996). In addition, behavioral therapies (e.g., cognitive-behavioral psychotherapy, supportive-expressive psychotherapy, interpersonal psychotherapy, 12-step facilitation, and motivational enhancement therapy; also see Chapter 4) have been developed to help detoxified individuals cope with circumstances that elicit craving (Beck et al., 1990; Carroll et al., 1991; Luborsky et al., 1995; Marlatt and Gordon, 1985; NIAAA, 1994a,b, 1995; Prochaska and DiClemente, 1986).
These different types of long-term changes in the brain have varied time courses of onset and decay. Somatic withdrawal may last from days to one or two weeks; the motivational aspects of withdrawal may last from several weeks to months; emotional memories related to drug use may last a lifetime. Drug abuse researchers are now beginning to uncover many of the details of the mechanisms by which drugs alter the functioning of the brain.
VULNERABILITIES TO ADDICTION
In this brief chapter it is not possible to address all the reasons why some individuals who use addictive drugs become dependent and others do not, and why some stop using and others do not. Risk factors for becoming addicted are as yet poorly understood but can be divided into factors that increase consumption and factors that increase the likelihood that the individual will be captured by the drug. Both genetic and environmental factors likely influence both the willingness of an individual to experiment with drugs and alcohol as well as the risk of developing dependence.
There is very little known currently about potential genetic contributions to vulnerability to opiate, cocaine, or nicotine addiction. However, most researchers agree, based on three types of investigations, that there are clear genetic components to vulnerability to alcoholism. These investigations are twin and adoption studies, genetic studies of certain human populations, and animal studies. Twin and adoption studies have demonstrated that early onset alcoholism is strongly influenced in males and females by genetic factors. Later onset alcoholism seems less influenced by genetic factors and more strongly influenced by environmental and emotional factors (Cloninger et al., 1989; Heath et al., 1991a,b; Kendler et al., 1992).
Genetic studies have focused on individuals in populations in which many people exhibit a strong aversive physiological reaction to alcohol (alcohol flush reaction), which is sometimes inaccurately called an allergy to alcohol. It includes facial flushing, rapid heartbeat, and headaches. The alcohol flush reaction, which if severe enough can cause loss of consciousness, occurs in roughly half of people with Chinese and Japanese ancestry and about a third of people with Korean ancestry. The flush reaction may represent a physiological "protective" factor that tends to prevent affected individuals from developing an alcohol addiction. The biological basis of this flush reaction is a variation in two genes that code for two principal enzymes of alcohol metabolism, alcohol dehydrogenase (ADH) and an aldehyde dehydrogenase (ALDH2) located in a specific part of cells called mitochondria (Thomasson et al., 1991). The most alcohol-sensitive individuals in these populations are those who inherit from both parents (homozygous) the gene variant that causes ALDH2 deficiency; these individuals cannot drink even small amounts of alcohol. If only one such gene is inherited (heterozygous), the individual can drink but is less able to drink large quantities and is 4-6 times less likely to become addicted. Individuals with the variant gene for ADH are also less likely to become addicted, but the effect is not as great. Interestingly, Caucasians, Alaskan Natives, and Native American populations exhibit very low prevalence rates for these genetic variations (Thomasson et al., 1994).
Genetic vulnerability to alcohol addiction can also be studied in animal models, including rats, mice, and even fruit flies (see Appendix D). Several rat strains have been developed through selective breeding that exhibit high or low
alcohol preference in self-administration models (Li et al., 1993). These animal models have characteristics that mirror in many respects human alcohol addiction; the animals show tolerance development, and pharmacological agents (e.g., naltrexone, an opiate antagonist) that reduce alcohol consumption in humans also reduce consumption in the animals (Li and McBride, 1995; Schuckit, 1994). Inbred and recombinant strains of mice have also been developed to assess the contributions of multiple genes to alcohol- (and drug-) related responses. Using a method called quantitative trait loci gene mapping (QTL), specific parts of genes have been identified that are associated with a variety of alcohol-related traits, including alcohol preference, sensitivity, tolerance, and withdrawal (Crabbe et al., 1994).
The search for specific genes that may confer enhanced risk of alcoholism has already progressed significantly, and much interest is focused on variant genes that may increase vulnerability to other types of addiction. The study of environmental factors that produce risk versus resilience with respect to drug addiction, however, must contend with multiple behavioral, psychological, and social factors that complicate analysis. In addition, other factors influence drug taking, including drug availability, acceptability in the individual's subculture, behavioral alternatives to drug use, and presence of other psychiatric illness. Factors that influence the "capture rate" include the intrinsic addictiveness of the drug used and, as mentioned above, the form in which it is administered (e.g., smoked crack cocaine).
In the model that has been outlined here, addiction is a disease of the nervous system, interacting with other biological systems and behavioral and environmental factors, that markedly impairs a person's ability to control his or her drug-seeking behavior; in fact, it makes resumption of drinking or drug-taking a compelling risk for the individual despite the highly negative effects of drug addiction on the individual's health and well-being.
Without building a greater context, however, such a biological model could contribute to the common misconception that if alcoholism or drug addiction is not simply willful misbehavior, addicted individuals cannot be held responsible for their actions. Individuals predisposed to developing coronary artery disease, for example, are not castigated by their physicians, but they are asked to follow a certain diet, to exercise, and to comply with medication regimens. Yet, for many individuals, the behavioral changes required to cope with heart disease are difficult and relapses to well-ingrained patterns are common. The same concept should be applied by physicians and society to those addicted to alcohol, nicotine, or illicit drugs. As in the treatment of heart disease, the best therapeutic results are obtained when addicted persons are given responsibility for themselves once a diagnosis is made and treatment recommended.
The clinically critical difference between addiction and coronary artery disease, however, is that the disease of addiction markedly diminishes the ability of the patient to follow through on medical advice. Hard as it is for patients with coronary artery disease to comply with treatment it is even harder for addicted
patients to comply. Thus, much research is devoted to finding medications to treat drug addiction (e.g., methadone) or to reduce drug cravings (e.g., naltrexone) (IOM, 1995). In addition, the clinician, as well as the patient's family, friends, and employers, must serve as a bulwark for the patient, against the intense drive for another drink, one more cigarette, or a few puffs of crack cocaine.
The idea that addictive drugs may commandeer key motivational circuits in the brain should also help researchers, physicians, and others to understand some of the otherwise inexplicable behavior of addicted people, who continue to use drugs either openly or surreptitiously despite, in many cases, tremendously negative personal and social consequences. This model also implies the importance of prevention and underlines the fact that, for those who are most severely affected, recovery may never be complete when environmental cues can always engender intense cravings and risk of relapse.
CONCLUSION
Recent discoveries have turned addiction research into a mature field that should attract the very best scientists interested in both basic and translational research. Researchers have cloned the brain receptors (i.e., the immediate molecular targets) for all significant drugs of abuse and have defined their locations in the brain. Of great significance, there is now general agreement on the importance of the dopaminergic brain reward pathway as one of the key common sites of action of addictive drugs. For example, because of the positive correlation between the potencies of cocaine as dopamine reuptake blockers and their ability to maintain self-administration behavior, it has been suggested that the action of cocaine at its binding site mediates the effects that contribute to abuse (Fischman and Johanson, 1996; Bergman et al., 1989; Ritz et al., 1987). Opioids and nicotine also stimulate the release of dopamine triggering the dopaminergic brain reward pathway (Corrigall et al., 1992; Koob, 1992b). Some aspects of treatment for all drugs are beginning to capitalize on the identification of this common pathway and the systems that regulate it. For example, naltrexone, an orally effective and long-acting opioid antagonist, has been shown effective in preventing relapse to opiate dependence in extremely motivated people (i.e., under strong external pressure to remain opiate free) (Brahen et al., 1978). Naltrexone has been shown to be effective in reducing relapse to alcohol dependence (O'Malley et al., 1996; Volpicelli et al., 1992). Nalmefene, a newer opioid antagonist, is currently undergoing testing and appears to have positive effects similar to those of naltrexone (Mason et al., 1994).
Researchers can now turn to the very difficult problems of understanding the precise brain mechanisms by which drugs alter brain function and come to dominate behavior. Table 3.1 lists some of these compelling research questions for the future. In the process, a great deal will be learned about the normal control
of motivation and emotion in the brain. With such discoveries, understanding about other human diseases and illnesses can also increase. For example, dopamine systems are not only the substrates of drug abuse and addiction, but also of psychotic symptoms that occur in a variety of psychiatric disorders and of some movement disorders, such as Parkinson's disease.
The importance of these areas and findings notwithstanding, it must be emphasized that drug addiction is the result of interacting biological, behavioral, social, and environmental factors. Thus successful treatment can come only from integrative research efforts involving multiple disciplines.
TABLE 3.1 Some Future Challenges in Basic Research
|
• |
Identification of factors leading to susceptibility or resistance to drug abuse in animals |
|
• |
Analysis of the inputs to and outputs from brain reward pathways: How does stimulation of these pathways produce reinforcement? |
|
• |
Development of better animal models, suitable for simultaneous genetic analysis, genetic manipulation, and behavioral analysis |
|
• |
Development of targeted, conditional gene knockout technologies and continued development of conditional gene activation technologies in transgenic animals (both are broad needs in neuroscience) |
|
• |
Dopamine: identification of which of the five distinct dopamine receptors are involved in mediating reward responses |
|
• |
Animal models: production of genetically engineered animals in which the genes for individual dopamine receptors or the dopamine transporter can be selectively inactivated in an inducible fashion; assess susceptibility of animals to drug consumption and drug addiction before and after genetic manipulations |
|
• |
Determination of the neurobiological basis of nicotine reinforcement |
|
• |
Molecular studies identifying genes, posttranslational modifications, or signal transduction pathways that produce functional subtypes of mu, kappa, and delta opioid receptors |
|
• |
Development of selective agonists or antagonists of kappa and delta opioid receptors for use in humans |
|
• |
Further characterization and evaluation of vaccine approaches to cocaine addiction |
|
• |
Identification of genes and quantitative tract loci associated with alcohol-related phenotypes in animal models |
|
• |
Elucidation of neurobiology and molecular biology underlying alcohol tolerance |
|
• |
Use of transgenic and gene knockout animal models to evaluate alcohol responses on brain neurotransmitter and reward systems |
|
• |
Evaluation of the role of N-methyl-D-aspartate and nitric oxide pathways on alcohol neurotoxicity, tolerance, and cognitive impairment |
|
• |
Elucidation of neuropeptide effects (e.g., corticotropin-releasing factor [CRF], neurotensin, arginine vasopressin [AVP]) and neurosteroid effects on alcohol actions and alcohol withdrawal |
REFERENCES
Beck AT, Wright FD, Newman CF. 1990. Cognitive Therapy of Cocaine Abuse. Philadelphia: Center for Cognitive Therapy.
Bergman, J, Madras BK, Johnson SE, Spealman RD. 1989. Effects of cocaine and related drugs in nonhuman primates. Part III. Self-administration by squirrel monkeys. Journal of Pharmacology and Experimental Therapeutics 251:150–155.
Brahen LS, Capone T, Bloom S, et al. 1978. An alternative to methadone for probationer addicts: Narcotic antagonist treatment. Contemporary Drug Issues 13:117–132.
Carroll ME, Carmona GG, May SA. 1991. V. Cocaine self-administration in rats: Effects of non-drug alternative reinforcers on acquisition. Psychopharmacology 110:5–12.
Cloninger CR, Sigvardsson S, Gilligan SB, Von Knorring AL, Reich T, Bohman M. 1989. Genetic heterogeneity and the classification of alcoholism. Advances in Alcoholism and Substance Abuse 7(3/4):3–16.
Cooper JR, Bloom FE, Roth RH. 1996. Cellular foundation of neuropharmacology. In: The Biochemical Basis of Neuropharmacology. 7th Edition. New York: Oxford University Press. Pp. 9–48.
Corrigall WA, Franklin KBJ, Coen KM, Clarke PBS. 1992. The mesolimbic dopamine system is implicated in the reinforcing effects of nicotine. Psychopharmacology (Berl) 107:285–289.
Crabbe JC, Belknap JK, Buck KJ. 1994. Genetic animal models of alcohol and drug abuse. Science 264:1715–1723.
Dani JA, Heinemann S. 1996. Molecular and cellular aspects of nicotine abuse. Neuron 16:905–908.
Di Chiara G, Imperato A. 1988. Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proceedings of the National Academy of Sciences of the United States of America 85:5274–5278.
Fischman MW, Johanson CE. 1996. Cocaine. In: Schuster CR, Gust SW, Kuhar MJ, eds. Pharmacological Aspects of Drug Dependence: Towards an Integrated Neurobehavioral Approach. Handbook of Experimental Pharmacology 118:159–195.
Fitzgerald LW, Nestler EJ. 1995. Molecular and cellular adaptations in signal transduction pathways following ethanol exposure. Clinical Neuroscience 3:165–173.
Hamilton ME, Freeman AS. 1995. Effects of administration of cholecystokinin into the VTA on DA overflow in nucleus accumbens and amygdala of freely moving rats. Brain Research 688:134–142.
Harris RA, McQuilkin SJ, Paylor R, Abeliovich A, Tonegawa S, Wehner JM. 1995. Mutant mice lacking the gamma-isoform of protein kinase C show decreased behavioral actions of ethanol and altered function of gamma-aminobutyrate type A receptors. Proceedings of the National Academy of Sciences of the United States of America 92(9):3658–3662.
Heath AC, Meyer J, Eaves LJ, Martin NG. 1991a. The inheritance of alcohol consumption patterns in a general population twin sample: I. Multidimensional scaling of quantity/frequency data. Journal of Studies on Alcohol 52(4):345–352.
Heath AC, Meyer J, Jardine R, Martin NG. 1991b. The inheritance of alcohol consumption patterns in a general population twin sample: II. Determinants of consumption frequency and quantity consumed. Journal of Studies on Alcohol 52(5):425–433.
Hyman SE. 1996. Addiction to cocaine and amphetamine. Neuron 16:901–904.
IOM (Institute of Medicine). 1995. The Development of Medications for the Treatment of Opiate and Cocaine Addictions. Washington, D.C.: National Academy Press.
IOM. 1996. Pathways of Addiction: Opportunities in Drug Abuse Research . Washington, D.C.: National Academy Press.
Izenwasser S, Búzás B, Cox BM. 1993. Differential regulation of adenylyl cyclase activity by mu- and delta-opioids in rat caudate-putamen and nucleus accumbens. Journal of Pharmacology and Experimental Therapeutics 267:145–152.
Johnson SW, North RA. 1992. Opioids excite dopamine neurons by hyperpolarization of local interneurons. Journal of Neuroscience 12:483–488.
Kendler KS, Heath AC, Neale MC, Kessler RC, Eaves LJ. 1992. A population-based twin study of alcoholism in women. Journal of the American Medical Association 268(14):1877–1882.
Koob GF. 1992a. Drugs of abuse: Anatomy, pharmacology, and function of reward pathways. Trends in Pharmacological Sciences 13:177–184.
Koob GF. 1992b. Dopamine, addiction and reward. Seminars in the Neurosciences 4:139–148.
Koob GF. 1996. Drug addiction: The yin and yang of hedonistic homeostasis. Neuron 16:893–896.
Li T-K. 1983. The absorption, distribution and metabolism of ethanol and its effects on nutrition and hepatic function. In: Tabakoff B, Sutker PB, Randall CL, eds. Medical and Social Aspects of Alcohol Abuse. New York: Plenum Publishing. Pp. 44–77.
Li T-K, McBride WJ. 1995. Pharmacogenetic models of alcoholism. Clinical Neuroscience 3:182–188.
Li T-K, Lumeng L, Doolittle DP. 1993. Selective breeding for alcohol preference and associated responses. Behavior Genetics 23:163–170.
Luborsky L, Woody GE, Hole A, Velleco A. 1995. Supportive-expressive dynamic psychotherapy for treatment of opiate drug dependence. In: Barber JP, Crits-Christoph P, eds. Dynamic Therapies for Psychiatric Disorders (Axis I). New York: Basic Books. Pp. 131–160.
Mason BJ, Ritvo EC, Morgan RO, Salvato FR, Goldberg G, Welch B, Mantero-Atienza E. 1994. A double-blind, placebo-controlled pilot study to evaluate the efficacy and safety of oral nalmefene HCl for alcohol dependence. Alcoholism: Clinical and Experimental Research 18:1162–1167.
Marlatt GA, Gordon JR. 1985. Relapse Prevention: Maintenance Strategies in the Treatment of Addictive Behaviors. New York: Guilford Press.
NIAAA (National Institute on Alcohol Abuse and Alcoholism). 1994a. Twelve step facilitation therapy manual. Project MATCH Series, Volume 1. NIH Publication No. 94-3722. Bethesda, MD: NIAAA.
NIAAA. 1994b. Motivational enhancement therapy manual. Project MATCH Series, Volume 2. NIH Publication No. 94-3723. Bethesda, MD: NIAAA.
NIAAA. 1995. Cognitive-behavioral coping skills therapy manual. Project MATCH Series, Volume 3. NIH Publication No. 95-3911. Bethesda, MD: NIAAA.
Nicoll RA, Alger BE, Jahar C. 1980. Enkephalins block inhibitory pathways in mammalian CNS. Nature 287:22–25.
O'Brien CP. 1976. Experimental analysis of conditioning factors in human narcotic addiction. Pharmacological Reviews 27:533–543.
O'Brien CP. 1996. Drug addiction and drug abuse. In: Hardman JG, Limbird L, eds. Goodman and Gilman's The Pharmacologic Basis of Therapeutics . 9th Edition. New York: McGraw-Hill. Pp. 557–577.
Olds ME, Milner P. 1954. Positive reinforcement produced by electrical stimulation of septal area and other regions of the rat brain. Journal of Comparative and Physiological Psychology 47:419–427.
O'Malley SS, Jaffe AJ, Chang G, Rode S, Schottenfeld R, Meyer RE, Rounsaville B. 1996. Six-month follow-up of naltrexone and psychotherapy for alcohol dependence. Archives of General Psychiatry 53:217–224.
Phelix CF, Broderick PA. 1995. Light microscopic immunocytochemical evidence of converging serotonin and dopamine terminals in ventrolateral nucleus accumbens. Brain Research Bulletin 37:37–40.
Prochaska JO, DiClemente CC. 1986. Toward a comprehensive model of change. In: Miller WR, Heather N, eds. Treating Addictive Behaviors: Process of Change. New York: Plenum Press.
Ritz MC, Lamb RJ, Goldberg SR, Kuhar MJ. 1987. Cocaine receptors on dopamine transporters are related to self-administration of cocaine. Science 237:1219–1223.
Satel SL, Southwick SM, Gawin FH. 1991. Clinical features of cocaine-induced paranoia. American Journal of Psychiatry 148:495–498.
Schuckit MA. 1994. Low level of response to alcohol as a predictor of future alcoholism. American Journal of Psychiatry 151:184–189.
Steinberg R, Brun P, Souilhac J, Bougault I, Leyris R, Le Fur G, Soubrie P. 1995. Neurochemical and behavioral effects of neurotensin vs. [D-Tyr11]neurotensin on mesolimbic dopaminergic function. Neuropeptides 28:42–50.
Tabakoff B, Hoffmann PL. 1996. Alcohol addiction: An enigma among us. Neuron 16:909–912.
Thomasson HR, Edenberg HJ, Crabb DW, Mai X-L, Jerome PE, Li T-K, Wang SP, Liu YT, Lu RB, Yin SJ. 1991. Alcohol and aldehyde genotypes and alcoholism in Chinese men. American Journal of Human Genetics 48:677–681.
Thomasson HR, Zeng D, Mai X-L, McGarvey S, Deka R, Li T-K. 1994. Population distribution of ADH2 and ALDH2 alleles. Alcoholism: Clinical and Experimental Research 18:60A.
Volpicelli JR, Alterman AI, Hayashida M, O'Brien CP. 1992. Naltrexone in the treatment of alcohol dependence. Archives of General Psychiatry 46:876–880.
Wafford KA, Whiting PJ. 1992. Ethanol potentiation of GABAA receptors requires phosphorylation of the alternatively spliced variant of the gamma 2 subunit. FEBS Letters 313(2):113–117.
Wafford KA, Burnett DM, Leidenheimer NJ, Burt DR, Want JB, Kofuji P, Dunwiddie TV, Harris RA, Sikela JM. 1991. Ethanol sensitivity of the GABAA receptor expressed in Xenopus oocytes required 8 amino acids contained in the gamma 2L subunit. Neuron 7(1):27–33.
Wilker A. 1973. Dynamics of drug dependence: Implications of a conditioning theory for research and treatment. Archives of General Psychiatry 28:611–616.
Wilkinson P, Van Dyke C, Jatlow P, Barash P, Byck R. 1980. Intranasal and oral cocaine kinetics. Clinical Pharmacology and Therapeutics 27:386–394.
Wise RA. 1978. Catecholamine theories of reward: A critical review. Brain Research 152:215–247.


















