21
New Approaches to the Study of Abnormal Immune Function
Gabriel Virella,1Candace Enockson, and Mariano La Via
INTRODUCTION
While the definition and diagnosis of congenital and acquired immunodeficiencies have been relatively well established for over a decade, the study of more subtle functional alterations of the immune system, which are believed (with more-or-less supporting evidence) to be associated with malnutrition or deficient nutrition, psychological stress, and aging has been slowed down by the lack of well-defined end points (Virella, 1993a). Parameters that are measurable in samples that are easy to obtain, store, and transport and that can be assayed using automated methodologies, providing highly reproducible measurements at low cost, would be ideally suited for the purpose. Unfortunately, few functional parameters related to the immune system fulfill these conditions.
One of the major factors complicating the selection of immune function parameters is the complexity of the immune system and the existence of regulatory networks, which tend to involve most defined cell populations and effector molecules in a complex set of interactions (Figure 21-1) and are difficult to dissect into separate measurable units with well-defined rationales. For example, classically, the effector mechanisms of the immune system are subdivided into two arms, antibody mediated and cell mediated. Antibody synthesis is a property of plasma cells that emerges after the antigenic stimulation of B-lymphocytes. The protective role of antibodies relates to (1) promoting phagocytosis or complement-mediated lysis or microorganisms and (2) blocking the interaction of microorganisms or their toxic products with their respective receptors, thus avoiding infection or the pathogenic consequences of toxin re-
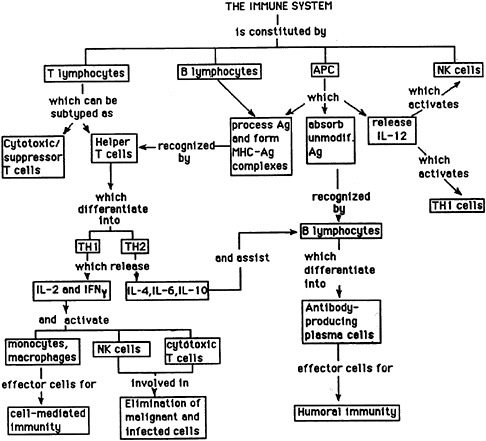
FIGURE 21-1 A simplified diagram of the main components and interactions of the human immune system. Ag, antigen; APC, antigen-presenting cell; IFNγ, interferon gamma; IL, interleukin; MHC, major histocompatability; NK, natural killer; TH1, T-helper 1 cell.
SOURCE: Adapted from Virella (in press).
lease. Cell-mediated immunity is most important for the elimination of intracellular microorganisms. This can be achieved by (1) activating the intrinsic antimicrobial mechanisms of the infected cells and (2) causing the death of the infected cells (cytotoxicity, mediated primarily by CD8+ lymphocytes and natural killer [NK] cells) (Virella, 1993b). However, the direct protective effects of antibodies are limited to blocking the absorption or penetration of infective agents or of their exotoxins to the respective receptors and targets (Virella, 1993c). Other anti-infectious mechanisms primarily dependent on the synthesis of antibodies require the cooperation of other humoral systems—such as the complement system (a complex of 11 plasma proteins that interact to mediate several functions of the inflammatory response)—or of cells able to recognize target-bound antibodies through membrane Fc receptors (cell membrane receptors that interact with the terminal domains of the heavy-chain constant regions of immunoglobulin molecules), and promote the destruction of those targets (Virella, 1993c). The humoral response itself cannot be triggered without the assistance of helper T-cells and antigen-presenting cells, such as macrophages (Virella, 1993d). To add to all these problems is the fact that a great deal of uncertainty remains regarding to what extent measurements made with circulating lymphocytes or assays of circulating cytokines reflect the true physiological conditions that exist at the level of a lymphoid organ engaged in the initial stages of an active immune response.
These questions may remain unanswered for some time, but this should not stand in the way of continuing efforts to apply basic knowledge to the definition of end points that may help define subtle abnormalities of the immune system. These abnormalities are not expected to be of the magnitude associated with full-fledged immune deficiencies and may not appear to be significant by themselves. However, even relatively minor functional abnormalities are likely to play an important role as cofactors determining poor resistance to infections and malignancies.
EVALUATION OF HUMORAL IMMUNE RESPONSES
Measurement of Serum Immunoglobulin Concentrations and Antibody Levels
Humoral immune responses are the easiest and cheapest to assess, particularly when serum or secretory immunoglobulins are the parameters being measured. Serum is easy to obtain, store, and ship, and immunoglobulin levels can be measured with great accuracy using automated techniques. Thus, an immunoglobulin assay would fulfill all criteria for an ideal parameter, adequate even for field studies.
On the negative side is the exhaustive documentation of patients with normal immunoglobulin levels who fail to mount specific responses against infectious agents and are therefore classified as functionally immunocompromised
(Virella, 1993a, e; Virella and Hyman, 1991). It was a considerable surprise to researchers, in a study that was conducted on the effects of dietary fish oil supplementation in a group of healthy normal individuals, when they found that changes in serum immunoglobulin levels were the most reproducible parameter in the assessment of humoral immunity (Virella et al., 1991; Unpublished data, G. Virella, Medical University of South Carolina, Charleston, S.C., 1994). As shown in Figures 21-2 to 21-4, there was a significant reduction in the serum levels of the three major immunoglobulin classes at the end of 6 weeks of fish oil supplementation (10 g/dl), which was not seen in controls who received an identical dose of olive oil. All levels measured in the participating volunteers were within the normal ranges for the respective immunoglobulin levels at all time points, and the differences between the baseline levels of volunteers receiving fish oil and those receiving olive oil also were nonsignificant due to the large individual variations in serum immunoglobulin levels. However, there was considerably less variability in any given subject when followed longitudinally. The techniques for immunoglobulin quantitation are highly reproducible, and the variations related to the technique virtually can be eliminated if all samples can be assayed on the same run. Under these circumstances, relatively small differences associated with fish oil ingestion reached statistical significance when paired analysis was performed.
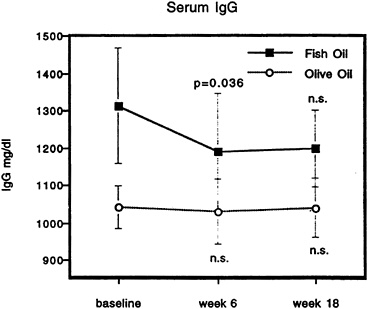
FIGURE 21-2 Longitudinal evolution of serum IgG levels in two groups of human volunteers, one whose diet was supplemented with fish oil (n = 7) and the other whose diet was supplemented with olive oil (n = 6). Statistical analysis was performed by the paired t-test, comparing week 6 and week 18 with baseline values.
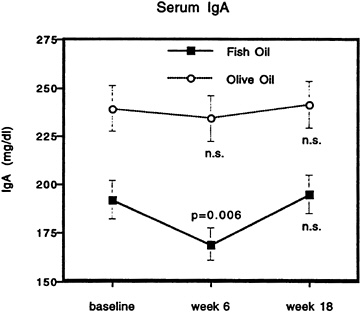
FIGURE 21-3 Longitudinal evolution of serum IgA levels in two groups of human volunteers, one whose diet was supplemented with fish oil (n = 7) and the other whose diet was supplemented with olive oil (n = 6). Statistical analysis was performed by the paired t-test, comparing week 6 and week 18 with baseline values.
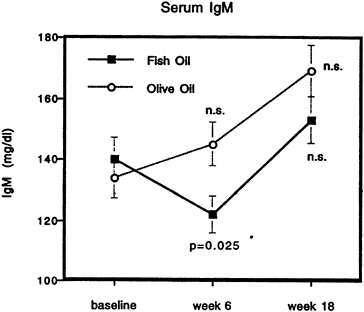
FIGURE 21-4 Longitudinal evolution of serum IgM levels in two groups of human volunteers, one whose diet was supplemented with fish oil (n = 7) and the other whose diet was supplemented with olive oil (n = 6). Statistical analysis was performed by the paired t-test, comparing week 6 and week 18 with baseline values.
When the ability to mount an immune response is evaluated, two approaches are possible. One is to study the response to an antigen that is unlikely to have been encountered previously by the subject (primary response). The other is to study the recall response to an antigen that has been experienced previously by the subject (secondary, recall, or anamnestic response). Studies in leukemia patients following bone marrow transplant have demonstrated that patients with normal immunoglobulin levels may not be able to mount either primary or secondary immune responses (Virella, 1993e; Virella and Hyman, 1991). Thus, it appears that the measurement of a specific antibody immune response should be a more sensitive end point for the assessment of functional impairment of the immune system. However, this approach also has some drawbacks. To test for a primary immune response, it is necessary to choose an antigen that is not likely to have been encountered previously by the individual, and this trivial aspect can become extremely frustrating because of previous immunizations, inapparent infections, and cross-reactions (particularly significant when polysaccharide antigens are considered as candidates for this type of study). To this day, the best candidate antigen to study the primary immune response is a phage (øX174), which has been used successfully in studies of both primary and secondary immunodeficiencies (Ochs et al., 1993; Virella, 1993e). The disadvantages of bacteriophage immunization are at least two-fold: the antigen is not easily accessible in a form approved for human use, and the assay of antibodies relies on a neutralization technique that is performed only in a few select laboratories.
Secondary immune responses to antigens, such as tetanus toxoid, are easy to assay with reproducible techniques, although these are not fully automated. The assays are carried out in serum samples collected before and after immunization. These assays are easy to apply to field studies. One aspect that needs to be considered is that, by its own dynamic nature, the response to immunization is not a parameter that can be repeatedly measured. Thus, any attempt to use an active immune response as an end point requires a proper cross-sectional design in which the response will be compared in two groups, one in which the variable with possible influence on the immune response will be applied and a control group. The timing of the immunization also needs to be considered very carefully to maximize the probabilities of observing an effect that is of sufficient magnitude so as not to be obliterated by individual variations. In addition, there are excellent data collected during the National Health and Nutrition Examination Survey III concerning the distribution of antibody titers in the general population (Gergen et al., 1995); these could allow a comparison of levels of preformed antibody or a comparison of the magnitude of response to a toxoid booster in individuals exposed to a stressor, relative to an age-and gender-matched cohort of control individuals. Of course, these participants would need to be matched for the time period elapsed since their last tetanus toxoid booster.
On the minus side, the high degree of individual variability observed in the response of humans to any kind of immunization is a significant obstacle to the
use of active immune responses as parameters in studies concerned with abnormalities in immunoregulation. In the groups of healthy volunteers in which the response to immunization with tetanus toxoid has been studied, there always have been subjects who fail to respond (Virella et al., 1978, 1991), and the frequency of nonresponders seems to be relatively high, estimated between 1 and 5 percent. Also, when the studied individuals have preimmunization antibody levels in excess of 1.5 U/mL (10-fold over the lower limit of protection), there is a good chance that immunization may trigger suppressor circuits, and a reduction in the concentration of serum antitetanus antibodies may be observed after a booster (Virella and Hyman, 1991; Virella et al., 1978). There is no indication that this negative response represents a risk for those who exhibit it, but it is certainly a significant confounding factor. Probably as a consequence of all these variables, the measurement of the response to a tetanus toxoid booster in volunteers receiving fish oil or olive oil dietary supplementation failed to reveal any significant difference (Figure 21-5), while the same group had revealed a significant decline in immunoglobulin levels in association with fish oil supplementation.
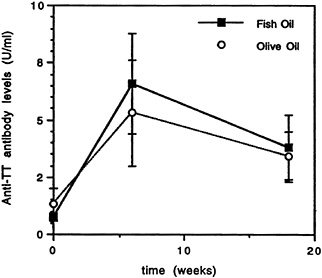
FIGURE 21-5 Longitudinal evolution of serum antitetanus toxoid antibody levels in two groups of human volunteers, one whose diet was supplemented with fish oil and the other whose diet was supplemented with olive oil. The increase in antibody concentration from baseline to week 6 was significant by the Mann-Whitney test for both groups of volunteers (p < 0.05), but there was no statistically significant difference between the levels of antibody reached at weeks 6 and 18 between the two groups.
Study of Humoral Immune Responses Elicited In Vitro
In clinical studies of patients with an unexpectedly high frequency of infectious episodes, it has been observed that a lack of in vitro immunoglobulin synthesis stimulation may be the only abnormality detected. However, in studies of the effects of dietary supplementation with fish oil, consistent differences could not be detected using in vitro immunoglobulin or antibody synthesis as end points (Virella et al., 1991). Thus, this approach may not be sensitive enough to detect low-level abnormalities in a heterogeneous population with a high degree of variability in the magnitude of their immune responses. Two additional problems with this approach are, first, the need for viable cells, which complicates sample collection, storage, and transport, and second, the nature of the assays, which are entirely manual and require specialized and experienced personnel. These factors result in higher cost and suboptimal reproducibility.
Attempts to simplify the protocols for study of mitogenic responses by using whole blood instead of mononuclear cells have the drawback of adding an additional variable—the red cells. Studies have demonstrated that adding autologous red cells to purified mononuclear cells enhances B-cell responses (Rugeles et al., 1987), probably through the delivery of costimulating signals to T-cells through cell-cell interactions that are mediated by a variety of complementary membrane molecules, including the CD2 molecule expressed by T-lymphocytes and CD58 (leukocyte function antigen-3 [LFA-3]) expressed by most nucleated cells as well as by human erythrocytes (Virella et al., 1988). The lymphocyte-erythrocyte ratio is critically important for the elicitation of this effect, and this ratio cannot be maintained absolutely constant if unfractionated blood is used in mitogenic stimulation studies. An additional problem with whole blood is that it contains plasma inhibitory compounds that interfere with cytokine activity, and these compounds are likely to vary from individual to individual.
EVALUATION OF CELL-MEDIATED IMMUNE FUNCTION
Lymphocyte Populations and Subpopulations
The parallel development of the monoclonal antibody and flow cytometry techniques has resulted in the definition of a large number of lymphocyte membrane antigens, allowing for the easy enumeration of lymphocyte subpopulations in the peripheral blood. The sample requirements are easy to fulfill—a few milliliters of unclotted blood are usually sufficient—and the techniques are semiautomated and relatively easy to perform, although both reagents and equipment are expensive and specialized personnel are essential. In spite of these limitations, the enumeration of lymphocyte subpopulations is very amenable to studies in the field.
However, the enumeration of lymphocyte subpopulations has not fulfilled the overoptimistic expectations that were raised by the studies reporting the identification of membrane markers and the definition of functional subsets based on membrane marker phenotypes. Part of the problem results from the less-than-perfect correlation between membrane markers and function. To illustrate the point, CD4+ T-cells, defined as ''helper" by their phenotype, actually can have cytotoxic functions (Rahelu et al., 1993; Smyth and Ortaldo, 1993). "Help" can be mediated by CD4- cells (Locksley et al., 1993), and the clinical significance of CD4-mediated "help" has been put to test by the finding by several groups of a less-than-perfect correlation between CD4 cell counts and clinical evolution of HIV-infected patients (Dormont, 1994; Keet et al., 1994; MAPW, 1994). The extensive data concerning CD4 counts in HIV-AIDS also have illustrated a second problem with the enumeration of T-lymphocyte subpopulations, which is the variability of these subpopulations (Hughes et al., 1994). Single, isolated enumerations of CD4+ lymphocytes may have very little meaning.
Recently, the reports of the predominant role played by T-helper 1 (Th1) cells in the activation of cell-mediated immune processes, including macrophage activation (Romagnani, 1991), and the possible significance of imbalances in the activity of helper T-lymphocytes with Th1 or Th2 cytokine release patterns in the progression of HIV-infected individuals toward full-blown AIDS (Clerici and Shearer, 1993) have raised considerable interest in their assessment. Most investigators rely on the characterization of the interleukin-release pattern of T-cell clones differentiated from patients' peripheral blood lymphocytes (Romagnani, 1991; van der Pouw-Kraan et al., 1993), as well as on the assay of interleukins released by peripheral blood lymphocytes in vitro, spontaneously and after mitogenic stimulation (Clerici et al., 1993; van der Pouw-Kraan et al., 1993). Some reports of attempts to define membrane markers for Th1 and Th2 cells have been published recently. CD4+ and CD56+ appear to define the Th1 subpopulation (Barnaba et al., 1994) while CD4+, CD27-, and CD30+ define the Th2 subpopulation (Del Prete et al., 1995; Elson et al., 1994). Whether these preliminary reports will hold and whether the enumeration of cells with these phenotypes will be informative about the state of immune responsiveness of a given individual remain to be seen. It must be emphasized that no one can be sure about the meaning of changes in the distribution of T-lymphocyte subpopulations in the peripheral blood. It is likely that the most significant changes in lymphocyte subpopulations occur in lymphoid tissues, and sampling peripheral blood may be irrelevant.
In studies conducted in this laboratory on the effects of fish-oil dietary supplementation on lymphocyte distribution, consistent changes in lymphocyte subpopulations could not be identified, even when two-color analysis was performed to define activated subpopulations through the coexpression of CD25 (IL-2 receptor) and other markers. Others have reported changes of the NK subpopulation in association with stress and with malnutrition in cancer patients
(Schedlowski et al., 1992; Sieber et al., 1992; Villa et al., 1991). The significance of these findings is diminished by the fact that the biological significance of the NK subpopulation has been established on the basis of data obtained in vitro, which have yet to be corroborated by clinical data, such as the description of patients with immunodeficiency clearly related to a lack of NK cells.
Assay of Circulating Cytokines and Soluble Receptors
At the time of writing this review, a total of 15 interleukins, a large number of growth factors and cytokines, and three types of interferons have been characterized. All of them seem to play some role in the activation and/or differentiation of different types of immune cells. However, some of these soluble molecules appear to have more important roles than others: IL-1β, IL-8, and TNF-α predominantly are involved in inflammatory reactions, particularly in septic shock (Goust and Bierer, 1993). IL-12 plays a significant role in the initial activation of NK cells and helper T-cells and in the differentiation of the Th1 subpopulation, which produces large amounts of IL-2 and interferon-γ (Manetti, 1993). IL-2 plays a significant role in the expansion of the Th1 subpopulation through autocrine and paracrine circuits and, together with IL-12 and interferon-γ, activates NK cells (Goust and Bierer, 1993). Interferon-γ, in turn, activates macrophages (Virella, 1993c). Th2 cells, which mainly are involved in assisting the activation of B-lymphocytes, secrete, among others, IL-4, which activates B-cells, particularly those producing IgE (Gascan et al., 1991; Goust and Bierer, 1993) and IL-10, which downregulates Th1 cells (Fiorentino et al., 1991). One additional interleukin with major biological significance is IL-8, which is a chemotactic factor for neutrophils and lymphocytes (Ribeiro, 1994). It also has been demonstrated that upon activation, several membrane molecules are shed by lymphocytes, including IL-2 receptors (Bock et al., 1994; Campen et al., 1988) and the CD4 and CD8 molecules (Kurane et al., 1991). Some of those cytokines and receptors have been found in relatively large concentrations in the urine (Bock et al., 1994, Newstead et al., 1993).
All the above-mentioned cytokines and shed molecules can be assayed by enzyme-linked immunoabsorption (ELISA), and providing that samples are properly collected, stored, and shipped, field studies have become relatively simple. One significant problem is the cost of the assays, usually ranging from $5 to $10 per sample per cytokine or soluble molecule for reagents, plus the costs of personnel. Also, assays performed in serum tend to be less reproducible than those performed in cell culture supernatants, perhaps because of the presence of soluble molecules in circulation, which may interfere with the assays. Such interfering molecules are apparently less of a problem in urine. Assays of urinary cytokines have dual advantages: ease of sample collection and accuracy of determination. The counterpoint is the variation that may be associated with diuresis, for which reason it is necessary either to collect 24-h urine or to correct
the data by expressing them as a function of urine osmolarity or some other parameter that may reflect variations in urinary volume.
Information is still somewhat limited concerning the possibility of finding significant correlations between the levels of circulating cytokines or soluble membrane receptors-markers and the status of the immune system. It also can be argued that the circulating cytokines are, in general, meaningless by-products, only a very pale reflection of the most significant lymphokine traffic that happens in lymphoid tissues. Direct interleukin traffic between cooperating cells probably involves intimate contact and even membrane fusion (Goust and Bierer, 1993). In spite of these questions, and until experimental data prove otherwise, the assay of circulating or urinary levels of selected lymphokines and lymphokine receptors, with their relatively well-established physiological roles, appears as one of the most promising avenues for functional studies of the immune system.
In Vitro Assays of Lymphocyte Function
The in vitro assays of lymphocyte function can be based on the incorporation of DNA precursors, expression of antigenic or mitogenic activation markers, release of lymphokines, suppression of B-or T-cell activation, and cytotoxicity (Virella et al., 1993). All the corresponding assays have the same basic problem, which is the requirement for freshly isolated cells, as discussed earlier in the chapter in the section on in vitro tests for B-cell function.
Mitogenic Assays
The response to mitogenic stimulation is the classic test used to evaluate lymphocyte responsiveness. In the most widely used format, the tests require incubation of mononuclear cells in the presence of mitogens for 3 days, at which time tritiated thymidine is added to the culture, and its incorporation into nascent DNA is determined 6 to 8 hours later. Other techniques have been developed that avoid the use of radioisotopes but require equally expensive technologies, such as sensitive colorimeters or flow cytometers (Huong et al., 1991; Kuhn et al., 1995). So in the end, there are few technological solutions that would allow simplification of these assays to the point of making them easy to perform in the field. Other problems faced when performing these assays are difficulties in reproducibility (changes in medium composition, source or batch of animal serum or serum replacements; incubation conditions; isolation methodology; anticoagulants; and culture vessels) and relative insensitivity.
In recent years, there has been a trend to replace DNA synthesis by what appear to be more physiological end points—such as IL-2 release or expression of IL-2 receptors—in mitogenic response assays (Virella et al., 1993). The release of IL-2 can be easily and reproducibly measured after 24 hours of incuba-
tion with pokeweed mitogen or phytohemagglutinin using standard ELISA methodology. In the authors' experience, it is not unusual to find individuals with normal or even elevated mitogenic indices when release of IL-2 is below normal (Table 21-1), suggesting that the release of IL-2 is a sensitive index of regulatory abnormalities affecting the initial stages of Th1 proliferation and differentiation.
The expression of IL-2 receptors (CD25) also has been found to be a good end point in studies evaluating the effects of stress and psychological depression on the immune system (La Via et al., 1996). As illustrated in Figure 21-6, the expression of CD25 after mitogenic stimulation was depressed significantly in a group of students tested immediately after taking their medical board examinations (Unpublished manuscript, M. F. La Via, Medical University of South Carolina, Charleston, S.C., 1994). These students also had a considerably higher
TABLE 21-1 Comparative Results of Mitogenic Responses and IL-2 Assay in Supernatants of Mitogen-Stimulated Mononuclear Cells*
|
|
|
Mitogenic Response† |
IL-2 Release |
||
|
Subject |
Diagnosis |
PHA‡ |
PWM§ |
PHA‖ |
PWM§ |
|
|
Normal controls |
33–160* |
13–40* |
9–39* |
|
|
A |
Immunodeficiency |
619 |
108 |
7 |
5 |
|
B |
BM transplant |
367 |
104 |
19 |
< 3 |
|
C |
BM transplant |
88 |
58 |
< 3 |
< 3 |
|
D |
BM transplant |
138 |
16 |
7 |
< 3 |
|
E |
BM transplant |
121 |
18 |
3 |
3 |
|
F |
BM transplant |
151 |
34 |
10 |
6 |
|
G |
Unknown |
31 |
25 |
< 3 |
not done |
|
H |
Healthy volunteer |
151 |
37 |
16 |
6 |
|
NOTE: PHA, phytohemagglutinin; PWM, pokeweed mitogen; BM, bone marrow. * Values above the upper limit of normality are shown in bold; values below the lower limit of normality are shown in italics. † Stimulation index [(cpm stimulated/cpm baseline) × 100]. ‡ 10 μg/ml. § 1:200 dilution. ‖ 2 μg/ml. # Values expressed in U/ml. |
|||||
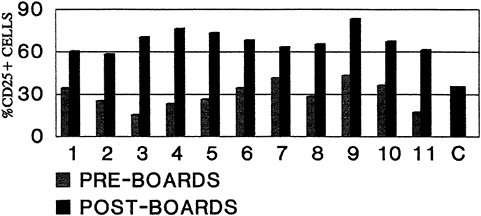
FIGURE 21-6 Comparison of the percentage of lymphocytes from 11 medical students and 10 control subjects expressing the CD25+ after 72 hours of culture in the presence of anti-CD3. The students were studied during the week before medical board examinations (cross-hatched bars) and 6 weeks after boards (black bars). The bar labeled "C" represents the average value for the 10 controls.
stress index than the respective controls. A similar depression in the percentage of cells expressing CD25 after stimulation with anti-CD3 was observed in patients with generalized anxiety disorder (La Via et al., 1996) and in another group of patients with panic disorder (PD). As shown in Table 21-2, PD patients also had a significantly higher stress intrusion score and a higher frequency of upper respiratory infections (URI) (Personal communication, G. Virella, Medical University of South Carolina, Charleston, 1997). Interestingly, a significant inverse correlation existed between the frequency of URI and CD25 expression. However, this type of study may be difficult to perform because it requires access to a sufficient number of flow cytometers to handle a large volume of samples in a short window of time, so as not to introduce significant errors due to time variations between adding the mitogen and testing for CD25 expression.
Functional Assays
In vitro mitogenic responses to antigenic stimulation using soluble proteins as antigens can be tested using thymidine incorporation as an end point. However, the results are highly variable, depending on the preceding history of exposure to the antigen in question. This variability, combined with the low magnitude of the expected responses makes this approach impractical for cross-sectional studies. A different approach that could yield interesting data would be to compare in vitro responses shortly after immunization in two groups of individuals, one exposed to conditions that could interfere with immunity and a
TABLE 21-2 CD25 Expression, Stress Intrusion Score, and Frequency of Upper Respiratory Infections in 19 Panic Disorder Patients and 18 Normal Controls
|
Variable |
Control Group Mean (SD) |
PD Group Mean (SD) |
p Value* |
|
% change in CD25+† |
44.6 (24.4) |
12.2 (12.2) |
<0.0005 |
|
SIS‡ |
3.7 (2.8) |
9.1 (5.0) |
0.0010 |
|
URI§ |
1.0 (0.6) |
1.8 (1.5) |
0.040 |
|
NOTE: SD, standard deviation; PD, panic disorder; SIS, stress intrusion score; URI, upper respiratory infection. * Calculated by the nonpaired student's t-test. † Percentage change is the difference between the percent of CD25+ cells at day 0 and day 3 after stimulation with anti-CD3 monoclonal antibody. ‡ Stress intrusion score (La Via et al., 1996; Zilberg et al., 1982). § Number of upper respiratory infections over a 6-mo period. |
|||
well-matched control group. Such studies, however, are likely to be affected by individual variations in the immune response (high responders vs. low responders) and may not have a significant advantage over the assay of serum antibodies elicited under similar circumstances, which is considerably simpler to perform.
Functional assays for helper or suppressor activity and cytotoxicity assays, although informative, are rather complex and difficult to standardize. The assay of helper/suppressor activity requires isolation of mononuclear cells and separation of T-cell-and B-cell-enriched populations and cocultures and determination of adequate end points, such as cytokine release or immunoglobulin synthesis. Cytotoxicity assays also involve isolation of lymphocytes or mononuclear cells and preparation of adequate targets. The simplest approach to studying T-cell cytotoxicity involves preparing mixed lymphocyte cultures and adding 51Cr-labeled target cells to the mixed culture after 4 to 5 days of incubation (Virella et al., 1993). This becomes impractical for studies of large groups of individuals. NK-cell activity can be easily measured by exposing adequate targets (such as the K562 cell line) to NK-enriched lymphocyte preparations. A major problem with this assay is the uncertainty that surrounds the physiological role of NK cells, which has been alluded to earlier.
PHAGOCYTIC CELL ASSAYS
Most phagocytic cell function assays are carried out with polymorphonuclear (PMN) leukocytes because of their abundance in the peripheral blood and the relative ease of their isolation. As a rule, peripheral-blood PMN leukocyte
preparations are constituted largely by neutrophils, and any functional tests carried out with these preparations are essentially neutrophil function tests. Acquired neutrophil abnormalities are most frequently a consequence of the depletion of this important cell population (acquired agranulocytosis). Rarely, neutrophils may be normal in number, but their function may be impaired. Neutrophil functional abnormalities have been reported in patients with severe trauma (particularly burns) and severe malnutrition (Virella, 1993a) but appear to be rather infrequent in patients with less dramatic insults to the integrity of their defenses. One of the most significant parameters of neutrophil function is the generation of superoxide and other oxygen active radicals (respiratory burst) as a consequence of the ingestion of antibody and/or complement-coated microbes or inert particles. Quantitative methodologies to measure the respiratory burst have been developed by the authors (Virella et al., 1990) and others (Metcalf, 1986, 96–114). Usually they require sophisticated equipment and trained personnel, but more importantly, they require freshly isolated cells and are not easy to adapt to the study of large numbers of individuals. Given the lack of data suggesting that the superoxide burst may be affected by physical or psychological stress, the investment in resources does not appear justified by such unlikely returns.
AUTHORS' CONCLUSIONS AND RECOMMENDATIONS
In spite of the progress that has been made in the basic knowledge of the immune system, there is a considerable gap separating that knowledge from the current methodologic approaches used to test immune abnormalities in human subjects. The choice of adequate end points for prospective studies, aimed at determining the influence of factors such as physical or psychological stress on the immune response, is particularly difficult because of the multiple components involved in a normal immune response. Pragmatic choices have to be based on the availability and reproducibility of techniques and on a good rationale supporting the decision to measure any given parameter. Longitudinal studies of serum immunoglobulin concentrations, measurement of humoral responses to vaccines and boosters, and determination of serum and urinary levels of select cytokines involved in inflammatory processes and immunoregulatory processes (particularly those cytokines that define Th1 vs. Th2 activity) are relatively easy-to-obtain parameters whose measurement can be supported by currently available basic and clinical research data. In vitro function assays, such as immunoglobulin synthesis, release of IL-2, or expression of CD25 in response to mitogenic stimulation, also appear to have the potential of revealing abnormalities in immunoregulation when other parameters, such as immunoglobulin levels and distribution of lymphocyte subpopulations in the peripheral blood, are within normal limits. Functional assays are more sophisticated and difficult to scale up for large population studies, but the potential rewards suggested by the authors' small-scale studies suggest that they should not be ruled out unless
it is absolutely impossible to devise a protocol in which they could be incorporated.
In conclusion, many variables need to be considered when choosing the parameters to measure immune function in relation to variables such as nutritional factors. In principle, functional assays would be ideal, but practical considerations may preclude their consideration. In that case, the choice of tests should be based on their physiological relevance (e.g., expression of lymphocyte activation markers) or measurements that have been shown to be affected by dietary interventions (e.g., serum immunoglobulin levels).
REFERENCES
Barnaba, V., A. Franco, M. Paroli, R. Benvenuto, G. De Petrillo, V.L. Burgio, I. Santilio, C. Balsano, M.S. Bonavita, and G. Cappelli 1994 Selective expansion of cytotoxic T lymphocytes with a CD4+CD56+ surface phenotype and a T helper 1 profile of cytokine secretion in the liver of patients clinically infected with hepatitis B virus. J. Immunol. 152:3074–3087.
Bock, G.H., L. Neu, C. Long, L.T. Patterson, S. Korb, J. Gelpi, and D.L. Nelson 1994 An assessment of serum and urine soluble interleukin-2 receptor concentrations during renal transplant rejection. Am. J. Kidney Dis. 23:421–426.
Campen, D.H., D.A. Horwitz, F.P. Quismorio, Jr., G.R. Ehresmann, and W.J. Martin 1988 Serum levels of interleukin-2 receptor and activity of rheumatic diseases characterized by immune cell activation. Arthritis Rheum. 31:1358–1364.
Clerici, M., and G.M. Shearer 1993 A Th1-Th2 switch is a critical step in the etiology of HIV infection. Immunol. Today 14:107–111.
Clerici M., F.T. Haki, D.J. Venzon, S. Blatt, C.W. Hendrix, T.A. Wynn, and G.M. Shearer 1993 Changes in interleukin-2 and interleukin-4 production in asymptomatic, human immunodeficiency virus-seropositive individuals. J. Clin. Invest. 91:759–765.
Del Prete, G., M. De Carli, F. Almerigogna, C.K. Daniel, M.M. D'Elios, G. Zancuoghi, F. Vinante, G. Pizzolo, and S. Romagnani 1995 Preferential expression of CD30 by human CD4+ T cells producing Th2-type cytokines. FASEB J. 9:81–86.
Dormont, J. 1994 Future treatment strategies in HIV infection. AIDS 8 (suppl. 3):S31–S33.
Elson, L.H., S. Shaw, R.A. Van Liwer, and T.B. Nutman 1994 T cell subpopulation phenotypes in filarial infections: CD27 negativity defines a population greatly enriched for Th2 cells. Int. Immunol. 6:1003–1009.
Fiorentino, D.F., A. Zlotnik, P. Vieira, T.R. Mosmann, M. Howard, K.W. Moore, and A. O'Garra 1991 IL-10 acts on the antigen-presenting cell to inhibit cytokine production by Th1 cells. J. Immunol. 146:3444–3451.
Gascan, H., J.F. Gauchat, M.G. Roncarolo, H. Yssel, H. Spits, and J.E. deVries 1991 Human B cell clones can be induced to proliferate and to switch to IgE and IgG4 synthesis by interleukin 4 and a signal provided by activated CD4+ T cell clones. J. Exp. Med. 173:747–750.
Gergen, P.J., G.M. McQuillan, M. Kiely, T.M. Ezzati-Rice, R.W. Sutter, and G. Virella 1995 A population-based serologic survey of immunity to tetanus in the United States. N. Engl. J. Med. 332:761–766.
Goust, J.M., and B. Bierer 1993 Cell-mediated immunity. Pp. 187–208 in Introduction to Medical Immunology, 3d ed., G. Virella, ed. New York: Marcel Dekker.
Hughes, M.D., D.S. Stein, H.M. Gundacker, F.T. Valentine, J.P. Phair, and P.A. Volberding 1994 Within-subject variation in CD4 lymphocyte count in asymptomatic human immunodeficiency virus infection: Implications for patient monitoring. J. Infect. Dis. 169:28–36.
Huong, P.L., A.H. Kolk, T.A. Eggelte, C.P. Verstijnen, N. Gilis, and J.T. Hendriks 1991 Measurement of antigen-specific lymphocyte proliferation using 5-bromo-deoxyuridine incorporation. An easy and low cost alternative to radioactive thymidine incorporation. J. Immunol. Meth. 140:243–248.
Kett, I.P., A. Krol, M. Koot, M.T. Roos, F. de Wolf, F. Miedema, and R.A. Coutinho 1994 Predictors of disease progression in HIV-infected homosexual men with CD4+ cells 200 × 106/L but free of AIDS-defining clinical disease. AIDS 8:1577–1583.
Kuhn, U., U. Lempertz, J. Knop, and D. Becker 1995 A new method for phenotyping proliferating cell nuclear antigen positive cells using flow cytometry: Implications for analysis of the immune response in vivo. J. Immunol. Methods 179:215–222.
Kurane, I., B.L. Innis, S. Nimmannitya, A. Nisalak, A. Meager, J. Janus, and F.A. Ennis 1991 Activation of T lymphocytes in dengue virus infections. High levels of soluble interleukin 2 receptor, soluble CD4, soluble CD8, interleukin 2, and interferon-gamma in sera of children with dengue. J. Clin. Invest. 88:1473–1480.
La Via, M.F., J. Munno, R.B. Lydiard, E.A. Workman, J.R. Hubbard, Y. Michel, and E. Pauling 1996 The influence of stress intrusion on immunodepression in generalized anxiety disorder patients and controls. Psychosom. Med. 58:138–142.
Locksley, R.M., S.L. Reiner, F. Hatam, D.R. Litman, and N. Killeen 1993 Helper T-cells without CD4: Control of leishmaniasis in CD4-deficient mice. Science 261:1448–1451.
Manetti, R. 1993 Natural killer cell stimulatory factor (interleukin 12 [IL-12]) induces T helper type 1 (Th1)-specific immune responses and inhibits the development of IL-4-producing Th cells. J. Exp. Med. 177:1199–1204.
MAPW (Multicohort Analysis Project Workshop—Part I) 1994 Immunologic markers of AIDS progression: Consistency across five HIV-infected cohorts. AIDS 8:911–921.
Metcalf, J.A. 1986 Laboratory Manual of Neutrophil Function. New York: Raven Press.
Newstead, C.G., W.R. Lamb, P.E. Brenchley, and C.D. Short 1993 Serum and urine IL-6 and TNF-alpha in renal transplant recipients with graft dysfunction. Transplantation 56:831–835.
Ochs, H.D., S. Nonoyama, Q. Zhu, M. Farrington, and R.J. Wedgwood 1993 Regulation of antibody responses: The role of complement and adhesion molecules. Clin. Immunol. Immunopathol. 67(suppl. 2):S33–S40.
Rahelu, M., G.T. Williams, D.S. Kumararatne, G.S. Eaton, and J.S. Gaston 1993 Human CD4+ cytolitic T cells kill antigen-pulsed target T cells by induction of apoptosis. J. Immunol. 150:4856–4866.
Ribeiro, R.A. 1994 IL-8 causes in vivo neutrophil migration by a cell-dependent mechanism. Immunology 73:472–477.
Romagnani, S. 1991 Type 1 T-helper and Type 2 T-helper cells: Functions, regulation and role in protection and disease. Int. J. Clin. Lab. Res. 21:152–158.
Rugeles, M.T., M. La Via, J-M. Goust, J.M. Kilpatrick, B. Hyman, and G. Virella 1987 Autologous RBC potentiate antibody synthesis by unfractionated human mononuclear cell cultures. Scand. J. Immunol. 26:119–127.
Schedlowski, M., R. Jacobs, G. Stratman, S. Richter, A. Hadicke, U. Tewes, T.D. Wagner, and R.E. Schmidt 1992 Changes in natural killer cells during acute psychological stress. J. Clin. Immunol. 13:119–126.
Sieber, B.J., J. Rodin, L. Larson, S. Ortega, N. Cummings, S. Levy, T. Whiteside, and R. Herberman 1992 Modulation of human natural killer cell activity by exposure to uncontrollable stress. Brain Behav. Immunol. 6:141–156.
Smyth, M.J., and J.R. Ortaldo 1993 Mechanisms of cytotoxicty used by human peripheral blood CD4+ and CD8+ T-cell subsets. The role of granule exocytosis. J. Immunol. 151:40–47.
van der Pouw-Kraan, T., R. deJong, and L. Aarden 1993 Development of human Th1 and Th2 cytokine responses: The cytokine production profile of T cells is dictated by the primary in vitro stimulus. Eur. J. Immunol. 23:1–5.
Villa, M.L., E. Ferrario, F. Bergamasco, F. Bozzetti, L. Cozzaglio, and E. Clerici 1991 Reduced natural killer cell activity and IL-2 production in malnourished cancer patients. Br. J. Cancer 63:1010–1014.
Virella, G., ed. 1993a Immunodeficiency diseases. Pp. 543–573 in Introduction to Medical Immunology, 3d ed. New York: Marcel Dekker.
1993b Introduction. Pp. 1–8 in Introduction to Medical Immunology, 3d ed. New York: Marcel Dekker.
1993c Infections and immunity. Pp. 233–246 in Introduction to Medical Immunology, 3d ed. New York: Marcel Dekker.
1993d Antigenicity and immune recognition. Pp. 49–70 in Introduction to Medical Immunology, 3d ed. New York: Marcel Dekker.
1993e Diagnostic evaluation of humoral immunity. Pp. 275–285 in Introduction to Medical Immunology, 3d ed. New York: Marcel Dekker.
Virella, G. In Press Introduction to Medical Immunology, 4th ed. New York: Marcel Dekker.
Virella, G., and B. Hyman 1991 Quantitation of anti-tetanus and anti-diphtheria antibodies by enzymoimmunoassay: Methodology and applications. J. Clin. Lab. Anal. 5:43–48.
Virella, G., H.H. Fudenberg, K.U. Kyong, J.P. Pandey, and R.M. Galbraith 1978 A practical method for quantitation of antibody responses to tetanus and diphtheria toxoids in normal young children. Zeitschrift fur Immunitatsforschung 155:80–86.
Virella, G., M.T. Rugeles, B. Hyman, M. La Via, J.M. Goust, M. Frankis, and B.E. Bierer 1988 The interaction of CD2 with its LFA-3 ligand expressed by autologous erythrocytes results in enhancement of B cell responses. Cell. Immunol. 116:308–319.
Virella, G., T. Thompson, and R. Haskill-Stroud 1990 A new quantitative Nitroblue Tetrazolium Reduction Assay based on kinetic colorimetry. J. Clin. Lab. Anal. 4:86–89.
Virella, G., K. Fourspring, B. Hyman, R. Haskill-Stroud, L. Long, I. Virella, M. La Via, A.J. Gross, and M. Lopes-Virella 1991 Immunosuppressive effects of fish oil in normal human volunteers: Correlation with the in vitro effects of eicosapentanoic acid on human lymphocytes. Clin. Immunol. Immunopathol. 61:161–176.
Virella, G., C. Patrick, and J.M. Goust 1993 Diagnostic evaluation of lymphocyte functions and cell-mediated immunity. Pp. 291–306 in Introduction to Medical Immunology, 3d ed., G. Virella, ed. New York: Marcel Dekker.
Zilberg, N.J., D.S. Weiss, and M.J. Horowitz 1982 Impact of events scale: A cross-validation study and some empirical evidence supporting a conceptual model of stress response syndromes. J. Consult. Clin. Psychol. 50:407–414.
DISCUSSION
TIM KRAMER: I would like to direct a comment toward your last statement about using whole blood. I am the one who is responsible for those data. I think when we used the whole blood, we set it up for several reasons. One is that we wanted a physiologic response in its natural milieu.
Now, I agree with you, the red blood cell has an interaction with the T lymphocyte in its responsiveness, and that is the way it is in vivo.
GABRIEL VIRELLA: Well, except that in vivo you do not have responses in the peripheral blood. You have responses in the lymph nodes, where the number of red cells is very small.
TIM KRAMER: That is true, and I agree with you that if you take the red blood cells and if you do add them to separated mononuclear cells, you will cause the lymphocyte responses to go up.
GABRIEL VIRELLA: Not only that, but you cause an increased B-cell response that is IL-2 independent, which may reflect a predominant stimulation of Th2-type T-cells.
TIM KRAMER: This is directed to that and to your comment about recommending looking at IL-2 as being possibly better than looking at mitogenic response. I have probably run close to 7,000 to 8,000 whole-blood samples, both in the field and in the laboratory, with studies ranging from nutrition studies that are conducted in house within the U.S. Department of Agriculture, to field stud-
ies with the Army or in China, and what we have come to find out now is that if we optimize our sensitivity in a mitogen-induced whole-blood system, the CV [coefficient of variation] values are down at 17 or 20 percent in comparison to the cytokines produced from whole blood in the cultures, which are up in the 50 to 60 percent range.
Actually, we find that IL-2 gives us the poorest data in comparison with interferon gamma and IL-10. So I think when you start looking at the whole area of immunology, if you try to look at the whole big picture it becomes too complex. That is why we try to use the lymphocyte as a sort of cell from the immune system in order to look at its response in vitro. We have found that we get less variation when we take a CBC [complete blood count]; thus, we can calculate the data per volume of blood and also per lymphocyte. Our data actually come out tighter that way than with standard.
GABRIEL VIRELLA: It would be tighter just because you have been standardizing your technique through the years, and obviously that happens. The more we run a technique, the better trained and the more experienced our technicians are, the tighter the coefficient of variation becomes. That is fine.
But I have a problem with this being physiological. I do not think testing whole blood is a physiological way to test immune response. I mean, it does not happen in whole blood. It does not happen that way.
TIM KRAMER: That is right, but if you look at it the other way, I have a question about the physiology when you take lymphocytes and standardize them to a constant number in vitro. You are looking at the activity of the cells on a per cell basis, but then the differences between individuals are hard to interpret because their white blood cell counts are different. So I think you have big flaws [with both methods].
GABRIEL VIRELLA: But in the IL-2 assay, I have tighter results than you do, so each one of us has tighter results in what we like to run, you know.
(Laughter)




















