5
CENTRALIZATION OF RESEARCH CHIMPANZEE MANAGEMENT AND DEVELOPMENT OF A NATIONAL CHIMPANZEE RESOURCE
Chimpanzees constitute a national resource for biomedical research that can be perpetuated only by breeding animals already in the captive population. However, there are more chimpanzees than are now needed for research, and the high cost of maintaining them makes it difficult to meet the special requirements for their care and diverts funds from research. A major concern of the committee is the fragmentation of financial support and decision-making related to the research chimpanzee population in the U.S. There is a critical need for better organization, coordination, responsibility, and oversight at a national level for this important animal model resource, which is used by various federal agencies in programs that ultimately benefit humanity—the biomedical research activities of the National Institutes of Health (NIH), the drug development programs of the pharmaceutical industry and the Food and Drug Administration (FDA), and the emerging infectious disease programs of the Centers for Disease Control and Prevention (CDC) and the Department of Defense (DOD).
The committee recommends that
-
Chimpanzee breeding and research programs be centralized in a
-
Chimpanzee Management Program (ChiMP) and its Advisory Council, to be based in a national office, preferably in the office of the director of NIH or a suitable alternative that has the autonomy, infrastructure, and expertise to manage the program (figure 5.1).
-
A single entity in a government agency (ChiMP) be assigned ownership, or lifetime care, with responsibility and authority for stewardship of this vital resource, which is to be supported by and used in partnership with other federal agencies.
This national program (designated the National Chimpanzee Resource, or NCR) consists of the ChiMP office, the ChiMP Advisory Council, and chimpanzees owned or supported by the government. ChiMP should be an autonomous body with sole responsibility and authority for coordinating the management of a US government-owned population of chimpanzees for use in biomedical research by any government agency or department, irrespective of whether an investigator is employed by the government, receives research funding from a governmental source, or represents private enterprise. Because most biomedical research with chimpanzees is supported by the NIH intramural and extramural programs, it is logical that ChiMP be housed at NIH to facilitate transfer of information between ChiMP and the intramural NIH institutes and centers and the extramural chimpanzee-using institutions. Although the office is proposed to be at NIH, it is intended that it be supported by a consortium of federal agencies that use chimpanzees in research (such as FDA, CDC, and DOD) and therefore not be subject solely to NIH operating and budgetary constraints.
Elsewhere we have recommended that scientific review of protocols in which chimpanzees are proposed to be used be conducted by the principal investigator's institution and approved by that institution's animal care and use committee (IACUC). It is therefore the intent that ChiMP not serve this function; rather, its goal should be to expedite and simplify the assignment of chimpanzees to IACUC-approved protocols. The ChiMP office would serve as a resource for information on government-sponsored chimpanzee studies and seek to avoid unnecessary duplication of research, maximize efficiency of use of the chimpanzees, and ensure the implementation of the best practices of animal care and careful attention to the welfare of the chimpanzees. Requests
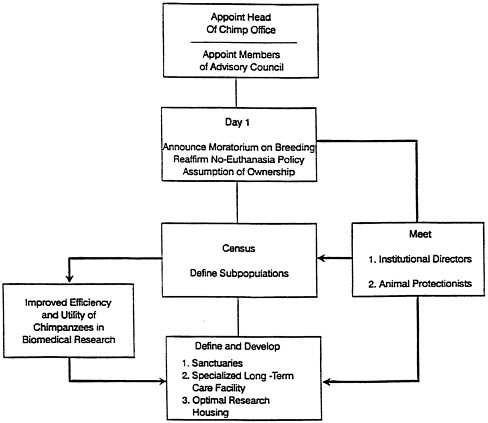
FIGURE 5.1 Comprehensive Care and Utilization of Chimpanzees in Biomedical Research—Implementation by Chimpanzee Management Office
for use of chimpanzees by NIH-funded investigators and those at other federal agencies and commercial organizations—such as FDA, CDC, DOD, and pharmaceutical companies—would be ranked by ChiMP on the basis of scientific merit; urgency of the public health-related problem; availability of animals with specific requirements regarding their age, sex, and past history; and preservation of the resource.
Because of ChiMP's broad range of responsibility, it is the committee's intent that it would operate with the full backing of the leadership of the NIH and other user agencies.
About 570 research chimpanzees in the United States are owned by government agencies, and about 700 are maintained by government
entities, including those in the breeding program and in research protocols. Some 600 animals housed in biomedical facilities are owned by private and state institutions. The fragmentation of support, ownership, and management has resulted in inefficiencies and inconsistencies in the use of animals for research and in the maintenance of optimal numbers and breeding capacity.
The committee recommends that a Chimpanzee Management Program (ChiMP) be created and given direct administrative and fiscal responsibility for about 1,000 government-owned or government-supported animals, which would constitute the NCR. The NCR would be comprised of all chimpanzees judged necessary to serve critical national needs. Also, the NCR would be responsible for animals used in government-supported research that might represent a public-health threat. Monitoring the NCR would lead to more-efficient use of chimpanzees in research and facilitate strategic planning for the long-term care of chimpanzees in the breeding population, available for research, on research studies, not needed for research and breeding, and posing public health threat. Such monitoring could include surveillance for disease conditions that might threaten the NCR population as a whole. To these ends, the ChiMP office would
-
Serve as responsible steward and custodian of the NCR.
-
Facilitate communication between institutions housing government-owned or government-supported and privately owned chimpanzees.
-
Communicate the availability of chimpanzees to appropriate scientists.
-
Be well informed of the status of all chimpanzees used in biomedical and behavioral research, both government and private.
-
Have budgetary authority for administering Public Health Service (PHS) and other government funds for maintaining the resource.
-
Articulate the principles and objectives of breeding, demography, and long-term care to appropriate institutional animal care and use committees (IACUCs) to promote coordination with research needs.
-
Establish a plan (before reinitiating breeding) for chimpanzees
-
no longer needed in research that adequately recognized ethical and moral obligations.
-
Ensure that government-owned or government-supported chimpanzees were used in federally supported biomedical research, except when scientific factors dictate the use of animals available from other sources.
-
Assign chimpanzees to approved and funded protocols.
The last responsibility requires specific attention. At present, the paperwork and time required for second-level peer review by the Interagency Animal Models Committee delay and limit the use of chimpanzees. This special second-level peer-review process was instituted because of concern that HIV-related research might rapidly deplete the available supply of research chimpanzees—a scenario that has not materialized. Active NIH control and restriction of the use of chimpanzees in research will no longer be needed and should be discontinued. Use of chimpanzees in research protocols should be approved or disapproved by IACUCs, which should apply the principles and objectives of ChiMP in their reviews. Under those circumstances, ChiMP's function would be to monitor the supply, demand, and specific uses of the chimpanzees, and to assign the most appropriate animals for specific purposes.
Elsewhere in this report, the committee recommends that use fees not apply to federally funded use of government-owned or government-supported chimpanzees. High use fees have substantially impeded the use of chimpanzees in biomedical research. The result of these recommendations would be to remove unnecessary expense, paperwork, and delay in the use of chimpanzees for important research activities.
OWNERSHIP
The fragmentation of ownership of chimpanzees and of oversight of their use in research has resulted in inefficiency and added immeasurably to the associated costs. The issue of ''surplus" chimpanzees is problematic and raises both practical and ethical concerns with respect to who should be responsible for such animals. Over 500 chimpanzees
are available for research, of which at least 200 are supported by the NIH National Center for Research Resources (in addition to those dedicated to breeding) and have not been used in any infectious disease research. PHS agencies have supported both the production of additional chimpanzees at the facilities that hold PHS-supported animals available for research and the leasing of chimpanzees from non-federal-government sources. The consequence of the combination of high productivity of the breeding program and less-than-anticipated research use has resulted in a surplus of animals, which has led to overcrowding and a risk of contamination of the breeding population with infectious disease. Use of animals in some studies, such as those involving the hepatitis viruses and HIV, might result in contaminated animals that have little prospects of future financial support. To offset that possibility, payment of endowments or use fees up to $55,000-66,000 per animal have been assessed. These fees could be eliminated from federal budgets if provisions for long-term support of chimpanzees as recommended in this report were made by the federal government, thereby making the chimpanzee model more accessible to investigators and substantially increasing its use.
Centralization of management of a specified number of chimpanzees as a critical national resource for ensuring the public health must include the outright ownership or lifetime support of the animals by the federal government. Because responsibility for long-term care is a major determinant of the cost of using chimpanzees, the committee believes that at least a minimum number of government-owned and-supported animals should be maintained for research and breeding. To reduce research costs and achieve the oversight required, it is recommended that ownership or lifetime care of selected animals, including those now owned by the DOD, be transferred to NIH. Management of their long-term support and use would be the responsibility of the ChiMP office.
Many animals requiring long-term management and care are owned by the government, others are supported but not owned by the government, and some of both of these categories were used in infectious disease research in the past and pose unknown health risks to humans. This committee recommends that ChiMP assume or retain ownership
or establish a mechanism to provide lifetime support of chimpanzees.
|
• Owned by NIH (excluding 99 in NIH breeding program) |
|
341 |
|
• Owned by the US Air Force |
|
135 |
|
• In NIH breeding program (including 99 owned by NIH) |
538 |
|
|
• That are breeding adults and offspring not in NIH program |
35 |
|
|
• That make up the total breeding population |
|
573 |
|
• That were previously used in infectious disease protocols |
|
350 |
|
• That are in research protocols or available for research |
|
100 |
|
Total |
|
1,499 |
|
• Privately owned, not expected to transfer to government |
|
-500 |
|
• Recommended for ChiMP ownership or support |
|
999 |
It is recommended that ChiMP assume ownership of life-time care of approximately 1,000 of these animals, as recommended in chapters 3 and 4. The committee derived the number 1,000 from two different calculations. In addition to the above listing, table 3.2 derives the same recommendation through categorization of population subgroups, excluding ownership. This number is thought to represent animals for which current owners would agree to transfer ownership or lifetime care to ChiMP. Approximately 500 animals distributed in all categories of the above listing are privately owned and used in nongovernment research. It is not recommended that ChiMP seek ownership of these animals. It should be noted that the figures are the best available to the committee but are not exact and that some categories overlap considerably. The data should be considered as general estimates to serve as guidance to ChiMP in managing the population. Current owners of selected animals should be provided with a one-time option to transfer ownership or responsibility for lifetime support to the government. Animals not transferred to the government under either scenario should remain the responsibility of the nongovernment owner.
A key question to be addressed by ChiMP is how many of the 1,000 animals designated for government ownership or life-time support might be removed to lower-cost facilities. On the basis of identification of the number needed for research use, the number needed for breeding to sustain this use, and the number considered to present a public health hazard, this figure can then be calculated. The determination will be based on the breeding model selected (e.g., the number of
animals needed in the breeding colony), the numbers needed for research, and the number that might present a public health threat. For example (using data from table 3.2 and breeding colony models in chapter 4):
|
Crisis Breeding Model |
168 |
|
Now in research (e.g., "needed for research") |
360 |
|
Potential public health threat |
260 |
|
Total |
788 |
Thus, 212 of the 1,000 animals might be released to public sanctuaries or other long-term care facilities. A different model with different assumptions and population sizes will have a different result. It is likely that careful review of those chimpanzees currently used in research and those considered infectious will reveal considerable overlap, and the number on active research will be considerably smaller. In addition, it should be recognized that animals considered to pose a public health threat might continue to be useful for research and be included in the number "needed for research" although they might be held in a lower-cost long-term care facility with experience in housing infectious chimpanzees and returned to research later. These details and the categorization of individual animals must be resolved by ChiMP before there can be a definitive response to the above question. If nongovernment sanctuaries become available, a substantial number of animals in each category might be designated as no longer needed for research or breeding and transferred to such facilities. Animals infected with agents that could pose a public health hazard or a risk for contamination of native chimpanzees (category D-3) require supervision by the ChiMP program and should be transferred to long-term care facilities that maintain existing NIH-supported chimpanzee colonies. They might be transferred to public facilities that have suitable experience in housing and handling such animals. The number of animals in D-3 underestimates the animals that pose a public health threat, in that it primarily includes animals used only in HIV and hepatitis infectious protocols.
In the aggregate, most of the remaining animals would serve as the breeding nucleus and research pool for use in biomedical research sponsored by the government. Other investigators, such as those in
pharmaceutical and biotechnology industries, should be encouraged to use animals from this pool at a cost determined by the ChiMP office. The additional revenues should be applied to the lifetime maintenance of animals in the NCR.
FINANCIAL SUPPORT OF THE NATIONAL CHIMPANZEE RESOURCE (NCR)
The committee recommends that ChiMP, in concert with the ChiMP Advisory Council, use this report as an aid in determining the short-term and long-term requirements for maintaining the NCR in ways that are consistent with the projected research needs and the long-term care of the animals. The costs for support of these animals can be met in part through appropriate payments from the various government agencies that support research with chimpanzees or for which maintenance of an adequate population of animals in readiness is essential. Several components of NIH, FDA, CDC, DOD and other agencies now use and fund research with chimpanzees although the responsibility for their long-term care and facilities has rested primarily with NIH. The current and long-term national need for chimpanzees in research might involve emergencies to which any agencies must respond. Therefore, the committee further recommends that
-
Funding for chimpanzee facilities, maintenance, and long-term care be sought and directed to chimpanzee use jointly by NIH, DOD, FDA, and CDC.
-
The agency responsible for coordinating chimpanzee budgets and procurements be NIH (or a suitable alternative that has the autonomy, infrastructure, and expertise to manage the program), which should delegate responsibility to ChiMP.
The committee foresees the need for congressional appropriations to provide full support for ChiMP, the facilities, and research to understand chimpanzee biology, management, and welfare better and emphasizes that these would be new funds—the current and future biomedical research base is already overextended. The committee does not view it
justifiable to expect the biomedical research community to bear the entire cost of the NCR when its existence is motivated by broader societal values, such as the demand for biomedical research and special ethical considerations for chimpanzees.
In the long term, one can expect that the improved coordination of the management of the entire US research chimpanzee population by the ChiMP office and its Advisory Council to result in a reduction in federal funds for supporting chimpanzees. In the short term, additional funds will be needed to maintain ChiMP and its Advisory Council, to institute ways to optimize efficiency of use of facilities and support services, and to meet housing standards recommended herein. The committee believes that these efforts will receive the support of the public, the animal protection community, and scientists who use chimpanzees in research, and it urges that the ChiMP office seek implementation of its recommendations through development of appropriate public and private partnerships.
PRIVATELY OWNED CHIMPANZEES IN BIOMEDICAL RESEARCH
Owners of privately owned research chimpanzees should be encouraged to include their animals in the ChiMP inventory because their numbers and availability will affect the demand for government-owned animals. Information on these chimpanzees should be considered in the evaluation of all requests for animals so that ChiMP can act as a clearinghouse to facilitate the most effective use of the entire US research chimpanzee population and so that ChiMP standards for care and housing can be disseminated to all chimpanzee colonies and institutions that use government-supported chimpanzees. The importance of privacy and confidentiality for industrial product development suggests that industry might prefer to use privately owned research chimpanzees as negotiated directly with holding institutions. However, requests for the use of government-owned chimpanzees by private industry should be considered and be accompanied by an appropriate use fee. Although it was suggested to the committee that private industry (e.g., pharmaceutical companies) might not contribute directly to the joint funding of the
NCR by government agencies, preferring instead to pay use fees as required for each study, the possibility should not be discounted. Privately owned animals could also be valuable to federally supported research if they fulfill requirements that cannot be met by the pool of government-owned animals; an example would be very young animals for studies with respiratory syncytial virus. As noted earlier, ChiMP must require strong scientific justification for government use of privately owned animals and be prepared for the short-term and long-term consequences related to the disposition of such animals. Even though ChiMP would not automatically become responsible for the lifetime care of these animals, it should anticipate the possibility of such responsibility if an infectious agent that poses a public-health threat is involved.
CHIMPANZEE SANCTUARIES
Sanctuaries and other long-term care facilities constitute a mechanism for removing surplus animals (defined as animals for which there is no current or projected need in research or breeding) from the core population needed to meet current and future research requirements. Sanctuaries offer an opportunity for substantially reducing costs of long-term maintenance of chimpanzees without compromising high standards of well-being. There are at least three scenarios for long-term maintenance of chimpanzees no longer needed for breeding and research: surplus animals could be permanently removed from the national resource pool and sent to sanctuaries, possibly national sanctuaries supported by a combination of congressional and private sources; they could be sent to existing private sanctuaries; and they could be maintained at US government-owned or government-supported chimpanzee facilities remodeled for cost-effective long-term housing. This committee enthusiastically supports the principal of retiring chimpanzees not needed for research or breeding to a low-cost, high-quality life. It does not recommend any of the three above alternatives over the others; each probably has a role in a strategy to enhance their welfare, and reduce the cost of maintaining them.
However, the committee strongly underscores the need for owners
to guarantee lifetime care of animals designated for retirement. Procedures for retirement of surplus animals must include a detailed evaluation of the financial stability of the sanctuary or long-term-care facility and an assessment of current or proposed animal-care programs. The ChiMP office should coordinate the transfer of all pertinent animal records between the donor and receiving facilities and oversee the legal transfer of ownership when government-owned animals are moved to sanctuaries. Creating standards for sanctuary and long-term-care facilities should be encouraged, and it should be recognized that high-quality care of chimpanzees can be achieved with standards less rigorous than those now used in biomedical facilities. The minimal standards are those of the Animal Welfare Act and those detailed in chapter 3. The committee strongly encourages the ChiMP office to be proactive in assisting with strategic planning, development, and maintenance of private sanctuaries. Careful planning and communication are needed to build a base of mutual respect in preparation for meetings with animal protectionist organizations.
THE NATIONAL CHIMPANZEE MANAGEMENT PROGRAM ADVISORY COUNCIL
In this report, the committee has assessed the present and future role of chimpanzees in biomedical and behavioral research, proposed guidelines for the long-term care of the captive-chimpanzee population with appropriate consideration of ethical and moral obligations, and recommended an operational structure that centralizes management of this national resource. The committee believes it to be an absolute necessity that appropriate and continuing oversight of such an enterprise be instituted. A national Advisory Council (AC), in concert with ChiMP, would provide continuity in the management of this valuable resource and ensure that quality care of the animals is maintained in a cost-effective and ethically acceptable manner.
CHARGE
The ChiMP AC should consist of representatives of the disciplines
discussed in this report. Its charge should be to oversee ChiMP's implementation of and adherence to this committee's recommendations, including ensuring that an adequate number of chimpanzees for use in biomedical research be maintained and housed in approved facilities where high-quality veterinary care and behavioral enrichment programs are provided as economically as possible. This charge could be facilitated by the establishment of continuing discussions with all institutions and facilities that house chimpanzees and the maintenance of a regularly updated census of animals being used or available for use in research protocols. It is hoped that non-government-supported facilities will comply voluntarily with recommendations and policies established by ChiMP and cooperate fully with ChiMP and the Advisory Council.
DUTIES
The duties of the proposed AC are in five categories: implementation, adherence, monitoring, reassessment, and recommendation. As indicated above, the first responsibility of the AC is to provide guidance to all facilities through ChiMP and ensure proper implementation of specific recommendations that are adopted. The AC should also promote a strategic plan for the use and breeding of chimpanzees and for periodic review of each facility.
Second, it should be the duty of the AC to ensure that all facilities that house government-owned or-supported chimpanzees or chimpanzees in studies supported by federal grants adhere to the policies and standards established in this report for the long-term care and use of chimpanzees in biomedical research. These policies and standards would apply to all facilities, irrespective of whether resident chimpanzees are being used for research or breeding or are "retired." To accomplish this goal, the AC should ensure an ongoing dialogue between ChiMP and all facility managers.
Third, to remain well informed about the numbers of animals both in use and available for use in research protocols, the AC should oversee the monitoring of the number of chimpanzees in each facility and the specific research in which they are or were used. Because a hierarchy of use has been established for different infectious agents (for example, chimpanzees are considered appropriate for entry into some
HIV-1 protocols only after completion of hepatitis studies), the knowledge gained through such monitoring would be important if a new infectious disease emerged that required the use of animals exposed to specific agents. Because of the need to maintain accurate census records at the national level and the fact that chimpanzees are considered an endangered species in their native habitats, a close liaison should be established with the International Species Information System (ISIS) or an equivalent. It would also be advantageous if the AC could obtain relevant demographic information from international facilities that house chimpanzees for research purposes, such as the Biomedical Primate Research Center in the Netherlands. Such information would assist in monitoring genetic diversity in the world's captive population.
Fourth, because of changing needs for chimpanzees in biomedical research, the AC should periodically reassess the recommendations made by the present committee and the initial strategic plans of ChiMP. The overall reassessment should be made at least once every two-three yr. Although we recommend that breeding of chimpanzees be stopped for the next five yr, this recommendation should be reevaluated annually to ensure that sufficient numbers will be available for up to 10-yr for continuing research programs or in case of a public health emergency. Any policy regarding euthanasia that is adopted or recommended should also be reviewed periodically.
Finally, on the basis of its periodic reassessments not only of the need for chimpanzees, but also of the plan to maintain the quality of their long-term care, the AC should advise and make new recommendations to ChiMP.
MEMBERSHIP
The AC should include persons with a wide range of expertise: colony management, veterinary medicine, reproductive biology, demography, population genetics, biomedical research, animal welfare, ethics, and chimpanzee health and behavior. The majority of AC members should not be federal employees. The AC might also include representatives of the privately supported facilities that house chimpanzees, such as members of their advisory boards. It should also include at















