3
Simulant Section
IMPORTANCE OF DERMAL PENETRATION
The importance of protective ensembles to prevent penetration of vapors is based on the consequence of dermal exposure. Dermal penetration has been shown to play a significant role in exposure to both chlorinated pesticides and organophosphates. In an attempt to account for both dermal penetration and inhalation, Finland and the United States now monitor chlorophenols in urine for setting air standards for workers exposed to chlorinated compounds. The importance of dermal exposure has also been shown in the treatment of wood (Fenske et al., 1987; Kauppinen and Lindross, 1985). Dermal exposure to organophosphates has also been found to be significant (Environmental Protection Agency, 1992).
The effects of changes in the skin barrier can be critical. For example, although parquet is not known to be absorbed by the skin, a fatal case involving skin penetration has been reported (Newhouse et al., 1978). In this case, the patient had numerous scratches on his arms and legs, and skin absorption over a period of time was fatal. Besides cuts and scratches, other conditions that enhance dermal penetration include skin hydration and dermatitis. Fenske and co-workers (Fenske et al., 1987) reported on skin contamination by tetrachlorophenol (detected by a fluorescent tracer) in timber workers who wore polyvinyl chloride gloves. More than 86 percent of the contamination was detected on the palms (in the case of one worker, a cut through the glove material was found). As all of these and other studies have shown, dermal penetration by chemical agents can be significant. Consequently, evaluating the potential penetration of protective ensembles is necessary for determining the amount of chemical that might be deposited on the skin and/or absorbed through the skin.
Because of the acute toxicity of the agents being considered, simulant compounds must be used for testing. Regardless of the permeability of the simulant, passive dosimitry is an effective testing method for determining the relative distribution of the chemical to assess potential problem areas of the suit. Most problem areas appear to be at closures or around tears. If a simulant is used to predict actual skin penetration, the simulant should have similar physical and chemical characteristics as the agent of concern. Otherwise, comparative absorption rates, specific to each body region for each agent and simulant, must be developed.
USE OF SIMULANT TO PREDICT DERMAL PENETRATION
Protective garments may be required for an array of chemical agents. These include the organophosphate nerve agents, GA, GB, and VX, as well as the vesicant blister formulations of sulfur mustard, H, HD, and HT. Protection against these agents may be needed during wars, during terrorist attacks (such as the Japanese subway incident).
The World Health Organization (WHO) (1990), has reported that each year approximately 3 million people worldwide are poisoned by pesticides (nerve agents) resulting in 220,000 deaths. (According to the WHO, acute poisonings, including suicide attempts, mass poisonings from contaminated food, chemical accidents in industry, and occupational exposure in agriculture constitute the most serious health hazards from agricultural pesticides). Operations like Desert Shield and Desert Storm revealed the need for protective clothing. Because of the aggressive toxicity of nerve agents, the highest quality of protection is critical. At the same time, the soldier's operational capability must not be impaired.
In the 1940s, mustard gas was used on human test subjects. Observations showed that infiltration occurred at the head, neck, and ankles. The suits that were tested performed well except for closure areas. However, testing on human subjects with real agents has been discontinued. Therefore, an appropriate surrogate compound must now be used to test protective clothing.
There are two basic types of surrogate compounds for chemical warfare agents (CWAs): analogs and simulants. Analogs are not classified as CWAs but are structurally similar and are considerably toxic. Examples are chloroethyl ethyl sulfide, a powerful vesicant, and diisopropyl fluorophosphonate, a potent cholinesterase inhibitor.
Simulants have similar physical properties to CWAs but are essentially nontoxic. Simulants for fabric testing include methyl salicylate (MeS) for HD, dimethyl methylphosphonate for VX, and di-isopropyl methyl phosphonate for GB. Challenges from a surrogate should reveal weaknesses in protective gear. For example, in a 1990 test evaluating vapor protection capabilities of the jacket and trouser interface of a chemical protective ensemble, the mean vapor level measured in the abdominal area was 17 percent of the outside level. This demonstrated that a significant amount of vapor infiltrated the CPE through the interfaces and could pose a threat to the wearer (Scott and Pointer, 1990).
The military has used simulants of chemical agents in a variety of studies where it was important to estimate the disposition or movement of chemical agents and where a toxic endpoint was either unnecessary or undesirable. A simulant was selected on the basis of low toxicity and similarity of certain physical properties to the chemical agent. Chemical structure could be quite different. For example, MeS has a structure that bears no similarity to HD or VX (Figure 3-1), but its vapor pressure, density, and water solubility are similar to those of HD (Table 3-1). The general assumption is that chemicals with similar chemical and physical properties will behave in similar ways. For example, it is known that at low to moderate pressures, binary diffusion coefficients vary inversely with pressure or density (Reid et al., 1987). These coefficients would be important for selecting chemical agent simulants for MIST.
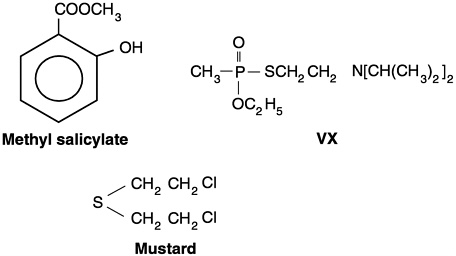
Figure 3-1 Chemical structures of methyl salicylate, VX, and mustard.
TABLE 3-1 Characteristics of Chemical Agents and Methyl Salicylatea
|
Chemical Agent |
GB |
VX |
H, HD |
HT |
MeS |
|
Common Name |
Sarin |
- |
Sulfur mustard |
Sulfur mustard |
Methyl salicylate |
|
CAS No. |
107-44-8 |
50782-69-9 |
505-60-2 |
Blend |
119-36-8 |
|
Chemical Formula |
C4H10FO2P |
C11H26NO2PS |
C4H8Cl2S |
Blend |
C803H8 |
|
Molecular weight |
- |
267 |
159 (HD) |
- |
152 |
|
Vapor Pressure (@ 25°C mm Hg) |
2.9 |
0.0007 |
0.08 |
0.104 |
0.091c (20°C) |
|
Liquid Density (@ 25°C g/cm3) |
1.089 |
1.008 |
1.27 |
1.27 |
1.18b |
|
Freezing Point (°C) |
-56 |
-39 |
8–12(H) |
- |
-8.3c |
|
Water Solubility (g/m @25°C) |
∞ d |
3.1d |
0.09d |
- |
0.07e |
|
Mode of Action |
Nerve agent |
Nerve agent |
Blister agent |
Blister agent |
Relatively nontoxic |
|
a Characteristics of agents from Daughtery et al., 1992. b Conkle et al., 1986. c Arca, 1996. d Reifenrath, 1980. e The Merck Index, 1996. |
|||||
A gas can move through protective clothing by sorption onto the ensemble surface, diffusion into the material, and desorption of the molecules from the inner surface of the fabric. A gas can also move through closures, seams, and imperfections in protective clothing. Component level tests of fabric swatches showed that penetration by chemical agents and MeS occurred only after three or four days of exposure (Dugway Proving Ground, 1994). Therefore, MeS penetration in the two hour system level MIST test was probably due to
movement of the gas through discontinuities in the ensembles. It follows that the value of the MIST conducted with MeS was in predicting ensemble fit and closure integrity.
SELECTION OF A SIMULANT
A number of simulant compounds have been used to predict agent penetration of ensembles. In 1994, the Army attempted to determine the relationship between fabric penetration by the vesicant agent HD and the simulant MeS in order to correlate the penetration of these two chemicals. The results of those tests were the basis for choosing MeS as a simulant for the MIST program (Dugway Proving Ground, 1994).
The Army has conducted penetration tests of various fabrics with both HD and MeS vapors. Vapor breakthrough was evaluated by plotting penetration curves (breakthrough concentration vs. cumulative Ct). Ct accounts for minor fluctuations in concentration during the test and represents the true loading of agent on fabric.
The resulting penetration curves indicated that MeS penetrates the fabrics about 30 percent slower than HD. It appears that the initial breakthrough occurs after three or four days of challenge (Dugway Proving Ground, 1994). Breakthrough levels increase gradually to about 5 percent, after which there is a dramatic increase. The data reveals a large amount of scatter. However, breakthrough curves were similar enough to support MeS as a simulant for evaluating the ensembles to protect against HD. Some similarities between the physical and chemical characteristics of MeS and other agents suggest that MeS may also be an appropriate surrogate for organophosphates. However, the significant difference in vapor pressure between MeS and nerve agents suggests that MeS would not be as good a simulant for nerve agents (see Table 3-1).
Because vapor penetration of the fabric was reported only after three or four days after onset of challenge, the two-hour testing period can be expected to test only penetration through seams and closure areas. It is not possible to conclude from the MIST that the most significant penetration of CPEs in actual use occurs around closures because the MIST results would logically be skewed toward penetration through closure areas. The MIST can, however, predict closure/seam areas of greatest penetration. This information is valuable for the immediate protection of personnel under attack.
Because penetration during the first few days of exposure are most significant at closure regions, any simulant might be able to provide the necessary information. In this case, both closure areas and folds could change because test subjects don ensembles slightly differently on different days. The mean suit penetration is the most important measurement for comparing garments.
The similar physical properties of MeS and HD support the use of MeS in the MIST. However, MeS and the nerve agents (satin and VX) do not have similar physical properties. Additional data would be required to support the use of MeS as a simulant for measuring the skin penetration of blister or nerve agents.
TOXICITY OF METHYL SALICYLATE
MeS is an oily liquid with wintergreen flavor. It is used in perfumes; as a flavoring in foods, beverages, and pharmaceuticals; and as an ultraviolet absorber in sunburn lotions. MeS is not acutely toxic (lethal doses are reported as 30 ml for adults and 10 ml for children) and does not cause dermal irritation in humans (Sax and Lewis, 1987). It has been used historically as a simulant and has been approved for use with humans by the Army's surgeon general. The environmental toxicity and persistence of MeS have also been studied (Cataldo et al., 1994), including the interaction of MeS with foliage and soils.
FINAL CHOICE OF A SIMULANT FOR MIST
The Army's choice of MeS as a simulant was based on the following information:
-
Tests for MeS and HD have been conducted with protective suit fabrics. Penetration curves indicate that MeS penetrates fabrics about 30 percent slower than HD, but the differences in penetration rates are not as significant as they first appear because the amount of vapor that penetrates incomplete closures or tears far exceeds the amount of vapor that penetrates intact fabric (Hanzelka et al., 1996).
-
The MIST is a system level test of chemical protective ensembles (CPEs). The MIST and BRHA require a simulant only for generating protection factors for the CPEs.
-
MeS has low toxicity.
MeS was chosen as the simulant for the MIST program. A single challenge level for the simulant was set at 100 mg/m3. However, comparisons with other surrogates should also be developed to confirm that MeS is the most appropriate simulant.
MeS can provide a relative ranking of the vapor protection of various CPEs. However, comparative skin penetration rates for MeS and chemical agents, which would be required to relate MIST data to physiological endpoints, cannot be determined.







