I
THE DISEASE AND TRANSMISSION
The same species of B. abortus occurs in cattle, bison, elk, and sometimes other wildlife species, and transmission of B. abortus among cattle, bison, and elk has occurred in captivity, which documents their common susceptibility and the potential for transmission in the wild. B. abortus probably is not native to North America but was introduced with the importation of European breeds of domestic cattle (Meagher and Meyer 1994).
Much research has been devoted to B. abortus transmission in cattle (Bos taurus), but considerably less has been conducted on transmission between wildlife species and cattle. Two of the most difficult issues to address concern the probability and mode of transmission of brucellosis among wildlife species and the probability of transmission among free-ranging animals and between wildlife and cattle.
Although some studies are available and others are under way, almost no controlled research has been done on those subjects. The available evidence is essentially anecdotal and inferential, and apparent transmission in wild and free-ranging populations has been interpreted by extrapolation from what is known about transmission in domestic livestock, particularly cattle.
The following sections describe the evolutionary history of bison and factors that are important to understanding transmission of B. abortus in free-ranging populations. Relationships between serology and culture used to determine infection are discussed, as are environmental and animal behavior factors that affect transmission of brucellosis among species.
BISON AND CATTLE
Bison and cattle are considered to belong not only to different species, but
different genera (Bison and Bos). Recent studies of mtDNA (Janecek et al. 1996) suggest that bison and cattle are sufficiently closely related that they should be placed in the same genus—Bos—but that revision has not yet been accepted by the Nomenclature Committee of the American Society of Mammalogists, the body that sanctions such changes. Bison and cattle do have anatomic and physiologic similarities and are capable of interbreeding. They also are susceptible to similar diseases.
That bison and cattle are classified as separate species is less important than the length of time since they diverged in evolutionary history. That determines the period over which effects of natural selection, genetic drift, and, in the case of cattle, artificial selection accumulate: the longer the divergence time, the greater the expected differences between bison and cattle. The traditional view, based on paleontology and morphology, placed the diversion time at about 2 million years ago (McDonald 1981), but the more-precise DNA molecular-clock approach suggests that it is substantially greater. Examination of mtDNA control-region sequences (691 base pairs) revealed divergence of 0.09 (Bison bison versus Bos taurus) and 0.093 (Bison bison versus Bos indicus). Assuming a divergence over time of 2% per million years (Brown et al. 1979), those values imply an approximate time since divergence of 4.5 million years (J. Derr, Texas A&M University, pers. commun., 1997). That interval encompasses the evolutionary speciation of most of the currently recognized ungulate species (see, e.g., Georgiadis et al. 1990; Cronin 1991). Thus, although bison and cattle share many genes because of their common ancestry, each has been isolated for a long period during which independent mutation and selection could result in differences in physiology that equal or exceed those in morphology.
There is also differentiation below the species level, and two subspecies are ordinarily recognized (McDonald 1981): plains bison (Bison bison bison) and mountain or wood bison (B. b. athabaska). This distinction based on morphologic characters is supported by modern DNA analysis. Studies of bison at Elk Island National Park, Alberta, show that wood bison and plains bison are genetically distinct populations, based on genomic DNA restriction fragment length polymorphisms (Bork et al. 1991), and they are estimated to have diverged from a common stock around 5,000 years ago (Wilson 1969).
B. ABORTUS INFECTION AND TRANSMISSION
Much has been made of the difference in disease between bison and cattle. Certainly, B. abortus induces disease in bison and elk that differs from the
disease in cattle. Biologic differences in antibody production, T-lymphocyte action, antibacterial proteins in normal serum, steroid-hormone synthesis, and genes for macrophage cytokines, integrins, and susceptibility genes in bison and elk all are different from those in cattle. However, the differences have not been shown unequivocally to underlie a major difference in pathogenesis that should shift our view of the pathobiology of brucellosis in the species.
Animal species vary in their clinical and tissue responses to B. abortus. Most bovids do not suffer marked fever, anorexia, or other signs of disease when infected. Disease is manifest only at the cellular or tissue level, such as replication of bacteria in lymphoid tissue with chronic inflammatory lesions referred to as granulomatous inflammation. The only external clinical signs might be slight swelling of lymph nodes that drain the site of infection. However, the pregnant female typically develops placental infection and can lose the fetus or experience premature labor. Even during abortion, the pain and other symptoms appear to be no greater than those encountered in normal parturition. In contrast with bovids, humans and other primates have a high, transient, fluctuating febrile response, the basis of the term ''undulant fever." Lipopolysaccharide molecules on the surfaces of B. abortus underlie many clinical signs that occur in disease and stimulate the precipitating antibodies that are the basis of most serologic diagnostic tests.
Several major factors characterize the evolving disease in wild mammals. First, brucellosis in YNP bison is a chronic, active process. Chronicity implies a substantial degree of immunity in many individuals that blunts the course of disease and allows calves to clear B. abortus from their bodies before they reach sexual maturity. Second, many subpopulations of animals are present in the YNP. At one extreme are the immune calves that inherit immunity from their mothers; at the other extreme are the immunologically naive female calves—calves that, when later infected as young heifers, will carry B. abortus into their first pregnancy and abort a highly infected placenta. It is the latter group, typically small in a chronically infected herd, that will sustain brucellosis in the herd.
The general principles of etiologic-agent transmission in intracellular infections (Miller et al. 1994) surely apply to brucellosis. In bison herds of high endemicity, the disease occurs most often in the young; in herds of low endemicity, the disease affects all ages. The patterns of B. abortus pathology depend on the degree of endemicity. Where endemicity is high, even though females are still infected after the age of 5 yr, the frequencies of disease and abortion are greatly reduced. Similar immune protection from disease in older animals is never reached in herds where there is low exposure to B. abortus.
Pregnant females, even if previously immune, will have a substantially higher risk for developing pathologic lesions and abortion.
B. abortus is most likely to be found in the organs and tissues of the reproductive system and mammary gland, associated lymph nodes, and lymph nodes of the head and neck. If not present in those sites, B. abortus in bison is largely confined to lymph nodes, spleen, and other lymphoid organs, sites with fewer bacteria per gram of tissue than would be the case in the reproductive tract. Bison with non-reproductive-tract infection rarely develop bacteremia, nor do they shed bacteria in saliva, urine, or other body secretions. Thus, even when such animals are in direct contact, bison-to-cattle transmission and even bison-to-bison transmission will occur rarely, if ever. Bison with non-reproductive-tract infection do not generally pose a risk of transmission to elk or cattle (E. Williams, U. Wyom., pers. commun., 1997).
That characterization of low risk is based on the small amount of data on bison, considerable evidence of transmission of B. abortus in cattle and other ruminant species, and knowledge of other Brucella species in mammals. Although more data on bison are needed, it is imprudent to assume a high risk of transmission in bison that are not infected in the reproductive tract or mammary gland.
The overriding importance of pregnancy and the reproductive system in the life cycle of B. abortus must be understood. Brucellosis in bovids and other ruminant animals is maintained in nature by the capacity of Brucella species to grow in the female reproductive tract, particularly in membranes and fluids that surround the developing fetus (see plates 1 and 2). The natural spread of brucellosis in ruminants is highly associated with abortion and the birthing process. During the perinatal period, several events come together to define the prevalence and survival of the disease in nature.
During late pregnancy, bacteria efficiently replicate in specific epithelial cells of the developing fetal trophoblast (Figure I-3). The association of two factors drives the transmission of B. abortus and ensures perpetuation of the disease:
-
Massive numbers of B. abortus in the placental fluids and genital exudates from the aborting female.
-
The strong attractant effect of expelled fetal membranes.
At the time of abortion or birth of an infected calf, transmission of B. abortus to other animals occurs through contact of oropharyngeal tissues of a susceptible host with contaminated materials, usually by touching, licking, or ingestion of placental membranes and fluids. In particular, male and

FIGURE I-3. Electron micrograph of B. abortus-infected chorioallantoic cytotrophoblast. Inside these cells, bacterial growth occurs in a distinct cellular compartment, the rough endoplasmic reticulum (RER), an organelle designed for highly efficient protein production. The production of peptides and glycosylation reactions in this organelle may be required for the massive growth of B. abortus that produces the billions of bacteria that are required for transmission to be sustained in nature (Anderson et al. 1986). Bacteria stained with an immunogold procedure to identify the genus Brucella.
female 2-year-olds in the nursery herd sniff and lick the expelled placenta and its fluids. Once in contact with mucous membranes of the eye, nose, or oral cavity, bacteria are taken into the body through several portals of entry, including tonsils, oronasal lymphoid tissues, and tear ducts. After the bacteria have passed the epithelial barriers of the oropharynx, they drain from the sites of initial infection into the lymphatic system and bloodstream. During travel through the lymph nodes and lymphatics and blood vessels, bacteria are taken into white blood cells, where they persist. Survival and dissemination in circulating white blood cells lead to infection of other tissues. In sexually mature animals, brucellae and brucella-infected cells have a strong tropism for the genital tissues and mammary glands—tissues that are needed for efficient replication.
It is unlikely that large numbers of bison in a herd would be infected in lymphoid tissues but not in the uterus or mammary glands. However, five important cases that increase the risk of transmission when reproductive tract or mammary gland infection is absent must be noted: persistence in pregnancy, chronic infection, transmission to scavengers and predators, shedding in mammary glands and milk, and shedding in feces.
Persistence in Pregnancy
The most important exception to the rule of low-risk infections is that female bison infected only in non-reproductive tissues constitute a population of
animals that can change to a high-risk population during pregnancy. The small numbers of B. abortus in lymphoid organs are stimulated to replicate during pregnancy and to infect the reproductive tract. Here they replicate to high numbers, and the affected female bison has a very high likelihood of transmitting B. abortus. The percentage of chronically infected female bison that will develop infections of the placenta and fetus during pregnancy is unknown, but the percentage cannot be assumed to be insignificant and does represent a major way in which brucellosis might continue in a population of bison.
Periodic Bacteremia in Chronic Infection
Chronically infected female cattle periodically and transiently become bacteremic and shed B. abortus in genital infections (Manthei et al. 1950; Lambert et al. 1960) (Figure I-4). The small numbers of positive bacterial cultures that are obtained from a chronically infected herd do not come from the same individuals; that clearly indicates that the number of infected females is always greater than the number that are shedding or bacteremic at a given time. The assumption is that the reproductive tract is infected, and the rise and fall of genital bacterial numbers are related to unknown stimuli or stress factors. However, whether a female bison must be infected in reproductive tissues for transient bacteremia to occur is unknown. If external stimuli can activate B. abortus growth in non-reproductive tissue that leads to asymptomatic bacteremia and genital infection in bison, the risk of transmitting B. abortus increases markedly for females with non-reproductive-tract infections.
Transmission to Scavengers and Predators
The risk of transmission of B. abortus in non-reproductive tissues from bison or elk to predators is high. Scavengers that eat infected tissues of dead bison can become infected and then, in turn, shed B. abortus (see Part II). Hunters and butchers are also at some risk from these animals. Predators or scavenging animals are particularly at risk of infection if they have contact with or ingest a heavily infected non-reproductive organ, such as a B. abortus-infected granuloma, spleen, or lymph nodes. As hosts, predators and scavengers tend
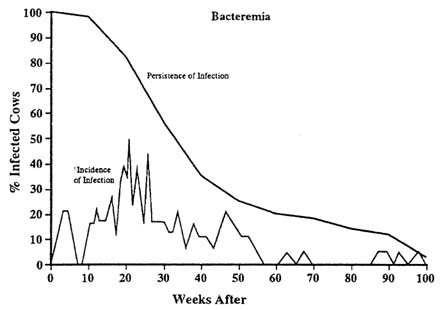
FIGURE I-4. Incidence and persistence of bacteremia in 18 experimentally infected cows (Bos taurus) over a 2-year period. Cows were exposed via the conjunctiva at doses of 7.4 × 105B. abortus strain 2308 (Manthei and Carter 1950). Each sample consisted of 100 ml. blood. Incidence is the percent of cows that had positive blood samples. Persistence is the percent of cows that were known to be infected at a particular point (note that not all were bacteremic).
to be only short-term shedders of B. abortus and are not important in the epidemiology of brucellosis (see discussion below). The infecting doses in the tissues of non-reproductive organs are small, and the predators involved are generally viewed as "dead-end" hosts—they do not shed organisms in amounts or at sites that are likely to infect ruminant species of animals. Thus, the risk of transmission of B. abortus from bison or elk that are infected only in the lymphoid system to an animal population that can maintain the disease in its own species and transmit it to other species is very low. Transmission, if it occurs, might be insufficient to maintain brucellosis in any animal population.
Although fetal membranes and fluids can contain the massive amounts of bacteria that effectively promote transmission, tissues of aborted bison fetuses are also highly infectious for predators. Unlike tissues of infected adults, the lungs and gastric contents of bison fetuses typically contain the
most bacteria, probably from fluids moved into the alimentary and respiratory tract during intrauterine life. In natural infections of cattle, meninges and choroid plexus of the fetus are tropic for bacteria (Hong et al. 1991), although the incidence of brucellar meningitis is not known and the mechanisms of infection are not understood.
Shedding in Mammary Glands and Milk
An important exception to the low risk of transmission concerns the mammary gland, which is not anatomically part of the reproductive tract. B. abortus in the mammary gland of lactating females replicates to high numbers in mammary tissue and in the lymphoid tissues of the regional lymph node—the supramammary lymph node—that drains the mammary gland. Although bacterial numbers are lower than in the infected placenta, they are typically high enough to present a serious risk of transmission. Any susceptible calf that suckles can become infected (vertical transmission1) even though horizontal transmission in this scenario would be highly unlikely (D. Davis, Texas A&M, pers. commun., 1997). In suckling bison calves, the relationship between oral infection via bacteria in milk and immunity from colostral antibodies is not known. Whether bacterial antigens and antibodies in intramammary milk interact in some unknown way to create complexes that increase either infection or immunity is an important pathogenesis issue that has not been well researched. Milk is an important route of transmission; further information is needed to understand the role it has in transmission.
In most lactating ruminant species, B. abortus is shed frequently in the milk when bacteria are localized in the mammary gland or supramammary lymph node (Morgan and McDiarmid 1960; Duffield et al. 1984). Bacterial numbers in milk are increased by a failure to suckle; the ensuing milk stasis leads to a marked increase in numbers of B. abortus in the mammary gland of goats and cattle (Meador et al. 1989) (Figure I-5). The association of milk stasis with a marked increase in B. abortus in mammary tissue and milk is an important possibility in bison, but needs to be confirmed (Rhyan et al. 1997).
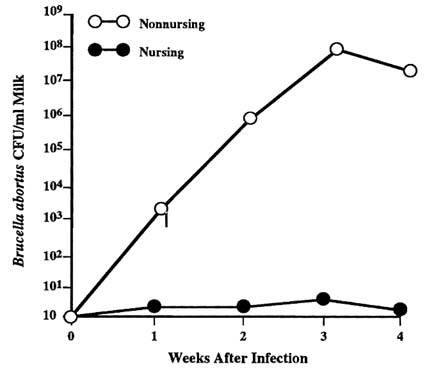
FIGURE I-5. Kinetics of bacterial numbers in mammary gland. Mean colony-forming units (CFU) of B. abortus isolated from milk of infected, nursing and nonnursing goats (data from Meador et al. 1989).
Shedding in Feces
Fecal shedding of B. abortus has been documented in bison. It is believed to occur only transiently in females that have ingested highly infected placenta and fetal fluids. In cattle, fecal shedding in the period after abortion has been reported (Fitch et al. 1932) and is thought to occur only in females consuming placenta during abortion. Because copraphagous animals could be infected by eating bison feces, fecal excretion that results from infection of intestinal tissue— particularly the lymphoid tissue of the ileum and colon—cannot be ruled out.
INFECTION IN MALES
Bison bulls develop orchitis, epididymitis, and seminal vesiculitis when infected with B. abortus (Tunnicliff and Marsh 1935; Williams et al. 1993; Rhyan et al. 1998); this suggests that excretion of bacteria from the testes, epididymides, and seminal vesicles into semen and urine could pose a risk of venereal transmission. However, shedding of B. abortus in semen of bison is reportedly rare (M. Stewart, APHIS, pers. commun., 1997), and the presence of B. abortus in bison semen and urine is not adequately documented.
In a current surveillance study of bison that were killed while leaving YNP in the winter of 1997, B. abortus biovar 1 has been isolated from testes, epididymides, and seminal vesicles of 50% of culture-positive animals; in this survey, 57% of the YNP male bison are culture positive for B. abortus to date. B. abortus biovar 1 also has been isolated from testicular abscesses in two YNP bison (D. Ewalt, Nat. Vet. Serv. Lab pers. commun., 1997).
In some captive herds, seropositive male bison have a high incidence of infection, that is, have culture-positive tissue (Rhyan et al. 1997). In studies on a captive herd of approximately 3,500 bison in central South Dakota, tissue and blood specimens were obtained from seven 3- and 4-year-old male bison that had been seronegative but had recently become seropositive for brucellosis. Six bulls had high serum titers and lesions in genital tissues, and B. abortus was cultured from all six. One bull had no observable tissue lesions and a low serum antibody titer, and B. abortus was not cultured from genital tissues.
Painful lesions in genital organs of bull bison appear to affect fertility and libido; males with painful testes do not compete successfully in breeding. That, with lameness that can result from brucellar arthritis and bursitis (Tessaro 1987), reduces the ability of males to breed.
Although it can occur, venereal transmission probably does not play a large role in the maintenance of brucellosis in bison in nature. Nonetheless, potential transmission through copulation does need to be considered for bison cows and domestic cows, given that bison bulls will serve domestic cows if confined with them. Most of the original hybrids between bison and cattle (beefalo) were the result of natural breeding rather than artificial insemination, the usual practice now (P. Rebich, Bigsky Beefalo, pers. commun. 1997). Rebich believes that cross breeding is unlikely to happen on the open range. Some bison bulls occasionally remain on the winter range during the summer in the northern Yellowstone area, but this can be prevented by hazing stragglers back to the summer range.
Bulls are not considered to be a likely source of infection either in the literature or in questionnaire results. Robison (1994) reported that venereal transmission by an infected bison did not occur, but the sample size was small.
No studies of transmission of B. abortus during copulation have been done on bison. Studies of possible transmission in cattle have been carried out over many years because at the turn of the century brucellosis was considered to be a venereal disease (Buck et al. 1919). Later work on penned brucellosis-infected bulls and clean cows has shown that transmission from bulls is usually not through copulation, but through nasal-oral contact or consumption of contaminated feed. Robison (1994) reported that venereal transmission by an infected bison bull in service of cows in a captive group failed.
Artificial insemination studies have shown that infected semen placed in the uterus often leads to transmission, whereas semen placed in the vagina or cervix usually does not (Manthei et al. 1950). Anatomy might limit the deposition of semen from bison bulls to the anterior vagina of bison cows, although that has not been established. The epithelial lining of the uterus differs from that of the vagina. Uterine epithelium is more susceptible to bacterial infection and has cellular mechanisms for bacterial uptake that are absent in vaginal epithelium. The low numbers of B. abortus shed in semen probably influence the likelihood of transmission (L. Corbeil, Univ. Calif., pers. commun., 1997).
Semen of some persistently infected male cattle does contain B. abortus , and bison semen might be a source of transmission. In cattle, B. abortus was isolated from 90 of 93 consecutive semen ejaculates from one bull over 5 years (Lambert et al. 1963). Despite that presence in semen, venereal transmission was not viewed as important. B. abortus was not transmitted in cattle during the use of an infected bull in natural service for 3 years, although B. abortus was present in each of 80 ejaculates collected over 18 months; the number of organisms varying from 100 to 49,500 per milliliter of semen (Manthei et al. 1950). When the same semen was experimentally inoculated directly into the uterus, infections were induced in six of six first-calf heifers and in two of six second-calf cows.
If B. abortus in bison follows transmission patterns of other ruminant species, venereal transmission alone would not be sufficient to sustain the persistence in the herd. The transmission of B. abortus by male cattle is better understood (King 1940; Manthei et al. 1950; Rankin 1965); it is capricious and not viewed as important in the disease at the herd level (although the mechanism of persistence in male genital tissues remains unclear).
As a practical matter, brucellosis commonly is eliminated from cattle herds by vaccinating cows without regard to bulls, which suggests that venereal transmission is rare. Brucellosis has been eliminated from many managed bison herds using the same protocols as are applied to cattle (i.e., vaccination and culling).
Practical experience suggests that transmission by bulls to cows by service among bison in the wild is unlikely, but more research data are needed on the role of the male in transmitting infection, particularly to determine the age, route, and persistence of B. abortus in genital tissues of the male and the reliability of serologic testing to detect infections. But given the spatial separation that usually results from management and the behavioral barriers to copulation between species, transmission of brucellosis between bison bulls and domestic cows in the GYA appears to be vanishingly small.
DETECTING INFECTED ANIMALS
Like tubercle bacilli and other intracellular bacteria, Brucella species have the capacity to survive in the face of strong host immune defenses by entering and surviving inside the cell. The capacity for a few brucellae to persist in lymphoid tissues in chronic infections and to be reactivated to grow again is closely related to their ability to survive in nature and explains many of the seemingly capricious events that occur in serologic and bacteriologic studies. The cell in the host's lymphoid tissues that perpetuates bacterial growth in chronically infected males and nonpregnant females is not known but is often referred to as the macrophage. Coupled with long-term survival in the host is the capacity to undergo striking growth, when activated by progesterone or some other hormone associated with developing pregnancy, and to move to and replicate in the developing placenta.
Serology
The presence of antibrucellar antibodies in serum of an infected animal indicates that infection is present or that infection has occurred recently. Because they detect antibodies, not living bacteria, serologic tests are indirect evidence of infection—evidence of an immune response to live B. abortus or to brucellar antigens of dead bacteria that have remained in the host. In brucellosis, humoral immune responses fail to eliminate or even control infection in the host but do limit the extent of infection.
Antibrucellar antibodies in serum of bison and elk are detected by a battery of tests that include the standard tube agglutination test, complement fixation test, CARD test, rivanol test, and particle concentration fluorescence immunoassay (PCFIA). All of these use brucellar lipopolysaccharides (LPS) as the test antigen, and thus detect antibodies to the perosamine residues (O side-chains) of bacterial LPS. Rough strains of B. abortus, such as the mutant vaccine strain RB51, are deficient in the O side-chain of LPS and do not induce antibodies that are detected in the standard serologic tests for brucellosis. None of the LPS-based serologic tests discriminate between vaccine strain 19 and field strains of B. abortus; they may detect differences in magnitude or persistence of brucella antibodies induced by those strains but not qualitatively different responses.
Specific serologic data from bison and elk are being analyzed to confirm the adequacy of guidelines that are used to interpret serologic reactions. The Brucellosis Scientific Advisory Subcommittee of the U.S. Animal Health Association is evaluating data on two new tests: the rapid automated presumptive (RAP) test and the fluorescent polarization assay. The subcommittee also is reviewing an APHIS-sponsored analysis of data from sera collected from cervids (M. Gilsdorf, APHIS, pers. commun., 1997). Sections of Brucellosis Eradication: Uniform Methods and Rules (USDA 1984) are being updated with rules that apply to commercial bison herds, interstate shipment of bison, and bison quarantine facilities.
Serologic tests of blood and milk are used to identify infected individuals and to characterize a herd. A positive serologic test in one individual is taken as indirect evidence that the herd is infected. An infected herd is one in which at least one animal has been shown to be infected; the diagnosis is based on results of several serologic tests, bacterial culture results, and information regarding herd history, clinical signs, and epidemiology. Minimal criteria for a diagnostically positive reaction to various serologic tests in cattle are provided by APHIS. Diagnostic criteria for bison and elk have been proposed but not approved (M. Gilsdorf, APHIS, pers. commun., 1997).
Serologic responses in bison might develop more slowly than in cattle or other species. Experiments have shown that although vaccinated bison challenged intraconjunctivally with a virulent B. abortus strain seroconvert on brucellosis-surveillance tests (Olsen et al. 1997), the antibody responses of bison to B. abortus challenge lagged approximately 2 to 3 wk behind that seen in cattle at the same time (Davis et al. 1990). An animal with natural resistance to Brucella spp. that has been infected with B. abortus and has cleared the bacterium generally will have a short-lived antibody response.
The False-Negative Serologic Test—Serologic tests can be falsely negative in infected bison when there is an absence of antibodies in an infected animal, when the test is insufficiently sensitive to detect low antibody titers, or when the test being used is itself defective.
Bison can lack antibodies in their serum but still be infected. In acute infection, that occurs when serum is taken during the incubation stage when antibodies have not developed. Not all actively infected animals have serologic evidence of exposure (R. Zarnke, Alaska Dept. Fish and Game, pers. commun., 1997), and that is most often the case for serologic tests taken in early stages of incubation. In chronic infection, false-negative reactions occur when bacteria are sequestered in lymphoid tissues in a state that does not induce antibody formation.
One problem of brucellosis serology is that some animals do not clear bacteria but retain them in their lymphoid tissues in very small numbers or in an inactive state that does not stimulate precipitating antibodies sufficient to react in serologic tests. Such animals often are seronegative but must be considered infected. It is clear that bison can be infected but be seronegative on all standard LPS-based serologic tests (Figure 1-6). Thus, for bison-management plans, seronegative bison cannot be assumed to be free of brucellosis.
The False-Positive Serologic Test—Serologic tests can be read as positive when no antibodies resulting from exposure have occurred. A false-positive test might be due to cross-reacting antigens or to defective test procedures. Tests also can be positive in the recovery phases in young animals that have no live bacteria.
Bacterial Culture
The isolation of B. abortus by bacterial culture of animals tissue or body fluids is the definitive indication of infection. Failure to culture bacteria that are present in small numbers in tissue can be due to inappropriate sampling, improper storage of specimens, or failure to use sufficient amounts of tissue. Correct techniques involve use of special growth media (Alton et al. 1988). In chronic infections in which very few bacteria are present, the use of subculture techniques might be required (Jensen et al. 1995). Any failure to follow established bacteriologic techniques will diminish the capacity to isolate B. abortus and, in turn, in the ability to identify all infected animals in a herd.
Other pitfalls in the interpretation of bacterial culture data are related to
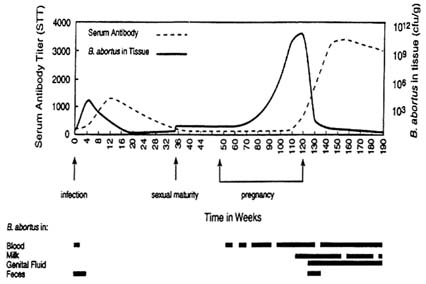
FIGURE I-6. Hypothetical titers (based on limited data) of serum antibodies and tissue bacteria in a bison calf from birth to 190 wks of age. Early phases are characterized by bacteria in lymphoid tissues of the head and neck. There is a small peak at sexual maturity as the reproductive system is infected and a large increased in late pregnancy as the placenta, fetus, and mammary gland are infected.
the sample. Even with use of proper technique, brucellae grow very slowly; in field specimens, overgrowth of more rapidly growing pathogens and sapro phytes in the sample easily can obscure more slowly growing brucellae.
The amount of body fluid or tissue in a sample correlates directly with the detection of B. abortus. In acute infections, especially of the reproductive tract and mammary tissues, that might not be critical, because large numbers of organisms are present. But in chronic infections with few organisms per gram of tissue, large samples must be used, e.g., 100 mL of blood, entire lymph nodes, and large sections of organs. Bacteria can reside only in one small part of a lymph node, so the entire lymph node must be sampled for appropriate detection.
Tissues collected for culture are those in which B. abortus is most likely to be found: organs and tissues of the genital system and its associated lymph nodes and lymph nodes of the head and neck. The placenta, if present, should be carefully examined to note the extent of lesions in cotyledons
(tufted areas of the placenta) and intercotyledonary placenta. Numbers of B. abortus present will be greatest in portions of the placenta that have exudates and tissue lesions. If the objective is to identify infection, then the simple determination of the presence or absence of bacteria is sufficient. Some studies, however, require precise knowledge of the extent of infection; in that case, the number of bacteria per gram of tissue should be determined, and this requires titration of tissue suspensions.
In chronically infected bison and elk, B. abortus is usually present in one or more lymph nodes. The specific lymph nodes to be harvested for bacterial culture should include bronchial, hepatic, internal iliac, mandibular, mesenteric, parotid, popliteal, prefemoral, retropharyngeal, superficial cervical (prescapular), and scrotal or supramammary.
The number of samples needed to detect infection in adult bison is unknown. One report from tests in adult cattle suggests that 100% detection can result from sampling of uterine caruncle and supramammary, mandibular, and medial iliac lymph nodes (Alton et al. 1988) but most laboratories do not achieve this detection rate. Young animals clear B. abortus quickly, and they require greater tissue sampling than adult bison.
Young adults of most species are most often culture positive because of more recent exposure and other factors. For example, B. abortus is stimulated to grow at sexual maturity and typically disseminates to the mammary gland, reproductive tissues, and associated lymph nodes. The growth stimulates a rise in antibody during and after sexual maturity. Thereafter, the serum antibody rises and falls according to the persistence or clearance of bacteria from tissue.
Detection of one or two B. abortus in lymphoid tissues of chronically infected animals remains a problem in that defects in sampling or culture technique often lead to false-negative results. A newly developed polymerase chain reaction can detect very small amounts of bacterial DNA (Bricker and Halling 1995), but it has not been established for official use.
If a highly sensitive test to detect all B. abortus cannot be developed, a test that will induce exacerbation of persisting bacteria might allow positive culture with current techniques—exacerbation mimics natural phenomena in brucellosis, in which unknown factors by unknown mechanisms stimulate latent B. abortus to replicate and spread in the host.
Correlation of Serology with Bacterial Culture
Multiple serologic and bacteriologic culture tests done over time are the only
reliable method to determine infection in live animals. A culture-positive animal always has the potential of transmitting the disease, but false-negative culture results can be obtained if inappropriate tissues are selected for culture or if tissues are mishandled during collection or laboratory processing.
High serologic responses correlate well with isolation of B. abortus on bacterial cultures; that is, bison infected with large numbers of bacteria typically have high serologic titers. A serologic result can be a good but not infallible indicator that an animal is infected. It is unlikely that a serologic response positive for B. abortus will provide a strong indication of whether an individual animal is infectious (S. Olsen, USDA, pers. commun., 1997).
The percentage of animals in a herd with serum antibodies (''positive" serology) is referred to as seroprevalence. It is widely believed that seroprevalence overestimates the prevalence of B. abortus infection, but that fact has not been established. Current knowledge of serologic reactions to organisms of the genus Brucella suggests that although we know that the presence of antibodies in an animal having had brucellosis lasts beyond the point where bacteria have been cleared from the host, it is more probable that any discrepancy between a clearly positive serologic test and bacterial culture is due to culture techniques.
Tests designed for cattle have been used for years to detect seropositivity in bison, but diagnostic tests used now for cattle have not been validated in bison. Current official tests are based on LPS of B. abortus. Data on the serology of bison with those tests are insufficient to support dogmatic statements regarding known relationships among serology, culture-test results, and likelihood of infectiousness (Olsen et al. 1998). S. Olsen (USDA, pers. commun., 1997) stated that "as is the case in cattle, it is unlikely that a strong correlation will be found between positive responses by bison on Brucella serologic tests and culture-positive status."
In summary, negative serologic test results do not equate with the absence of infection. Individuals can falsely test positive or negative on serologic tests. Serologic tests are evaluated in terms of sensitivity, specificity, and predictive values instead of absolutes, and all have some degree of imperfection. It is important to recognize that discrepancies between serologic and bacteriologic data might be real or artifactual. The discrepancy in GYA bison between the high percent of seropositivity and the much lower percent of bacterial isolations is most likely due to culture techniques.
Immunity
Immunity to brucellosis could be measured in bison and elk by means other than serology. Cell-mediated immunity, as measured by lymphocyte proliferation assays from naturally infected and vaccinated cattle and bison plays a role in recovery from B. abortus infection (Davis et al. 1990; Olsen et al. 1997). Cutaneous delayed hypersensitivity reactions against brucellin, an extract of B. abortus, are used in Europe to diagnose brucellosis in cattle of seronegative herds. In the course of bovine brucellosis, cutaneous reactivity develops later than do antibody responses but persist long after serologic evidence of infection has disappeared (Bercovich et al. 1990; Cheville et al. 1994). The current recommendation is intradermal injection of 0.1 mL brucellin in the neck or tail fold and gross examination of skin lesions at 24-72 hours later. The intensity of the reaction is based on the degree of skin thickness. This test is complicated by the occurrence of false-negative reactions and by variability in evaluations; it has never been tested in bison or elk.
LIKELIHOOD OF INFECTIOUSNESS
To be infectious, an infected animal must release B. abortus from its body in a way that will infect another animal. To place infectiousness in appropriate context, attention must be given to its precise definition and to the definitions of infection and disease:
Disease: any deviation from or interruption of the normal structure or function of any part, organ, or system of the body that is manifested by a characteristic set of symptoms and signs and whose etiology, pathology, and prognosis may be known or unknown.
Infection: 1. invasion and multiplication of microorganisms in body tissues, which may be clinically inapparent or result in local cellular injury due to competitive metabolism, toxins, intracellular replication, or antigen-antibody responses. 2. an infectious disease. Cf. infestation.
Infectiousness: ability to transmit a pathogenic agent from an infected individual to another susceptible individual. (Dorland's Medical Dictionary, 28th edition)
Brucellosis, particularly the chronic form in which there are few bacteria and no obvious clinical signs, is a disease. Bacteria are present in the animal, microscopic pathologic tissue changes are present, and, despite their lack of effect, immune systems are at work. Chronic forms of the disease have been called subclinical, latent, or inapparent infections.
Although controlled studies have not been done, it is unlikely that prediction
of infectiousness can be based on serology alone. Clearly, bison and elk may have serologic titers to B. abortus yet not be infectious. It is also highly probable that those species can have serologic titers and not be infected—when the animals are in a postrecovery period in which all live bacteria have been cleared but the animals are still responding to brucellar antigens that remain in their tissue. It is dangerous to assume that large numbers of seropositive animals do not carry live B. abortus. The occurrence of latent carriers among cattle (heifer syndrome) is widely accepted, and experimental evidence indicates that they occur among bison and elk (Thorne and Morton 1978).
MINIMUM INFECTIOUS DOSE
The minimum infectious dose (MID) of B. abortus in bison—the smallest number of bacteria that can initiate infection or disease—is not known. The smallest dose that will infect 100% of cattle is reported to be 15.6 × 106 live bacteria; below that dose, the infection rate is correlated with the dose (Manthei and Carter 1950) (Table I-1). In natural conditions, infection occurs with very large doses present in placenta, placental exudates, and milk. Even in cattle, the MID has never been established unequivocally. Experimentally, the same dose will typically infect a different percentage of the animals. The definitive study in cattle used five groups of pregnant heifers (10 per group) to estimate infective dose of virulent B. abortus given conjunctivally. The extent of placental infection was greatest in heifers given the largest inoculum (McEwen et al. 1939).
The value of the MID is compromised by two important characteristics: the validity of data used to establish it, and lack of full knowledge of the factors that skew the MID one way or the other. For example, animals with intercurrent disease that stimulates marked antibody production or cell-mediated responses show a significantly increased immune response to virulent Brucella spp.: calves with severe cutaneous fungal infections, such as ringworm, develop strikingly greater antibody responses to virulent B. abortus and clear the bacteria from the site of infection and draining lymph node much more quickly (Cheville et al. 1993) than animals without fungal infections do.
The capacity of B. abortus to survive in soil and debris at varied temperature, acidity, and relative humidity has a great bearing on numbers of bacteria available for transmission. Little is known about how long bacteria survive after abortion or birth events under natural conditions in the YNP. E. Williams (Univ. Wyo., pers. comm., 1997) stated that "preliminary studies indicate
prolonged survival of the bacteria in the Wyoming environment with the titer of bacteria remaining high for weeks in the late winter. … In spring, numbers of bacteria decrease rapidly along with accelerated decomposition of fetuses."
TABLE I- 1. Incidence of infection and abortion in normal nonvaccinated pregnant bovine heifers given different conjunctival doses of virulent B. abortus. Challenge strains were 544 (McEwan et al. 1939) or 2308 (Manthei and Carter 1950).
|
Authors |
Dose, no. bacteria |
Proportion infected, % |
Percent aborted |
|
McEwen et al. |
1.46 × 105 |
45 |
22 |
|
|
1.46 × 106 |
50 |
30 |
|
|
1.46 × 107 |
90 |
90 |
|
|
1.46 × 109 |
100 |
77 |
|
Manthei and Carter |
3.5 × 105 |
78 |
56 |
|
7.0 × 107 |
87 |
74 |
|
|
|
1.5 × 107 |
100 |
100 |
|
|
2.5 × 107 |
97 |
91 |
|
|
7.5 × 107 |
100 |
92 |
|
|
1.0 × 108 |
100 |
97 |
The value of MIDs for bison is questionable. Experiments to obtain MIDs for bison will be expensive; once derived, they will be limited in application, because differences in bacterial dose, route, temperature, and host factors (age, sex, stress, and disease) will cause variations that exceed the value of the data.
TRUE PREVALENCE OF B. ABORTUS IN GYA BISON AND ELK
The true prevalence of brucellosis in GYA bison and elk is unknown. Insufficient sampling (in regard to both number and reliability) has been done to
establish reliable data. Furthermore, in previous studies, some of the methods used were imprecise and led to false-negative or false-positive results of serologic and bacteriologic tests. The data that are available suggest only that the true prevalence is not zero and might vary from 12% to 100% in bison and from 1% to 38% in elk.
Bison
Bison populations in YNP and Grand Teton National Park (GTNP) can be considered to be chronically infected with B. abortus. Many bison develop immune responses but do not clear the bacteria (Olsen et al. 1997). B. abortus-induced abortion in free-ranging bison was first proved by bacterial culture and pathology in March 1989 in the Jackson herd of about 120 bison. B. abortus biovar 1 was isolated from reproductive tissues, and the pathologic description of disease was clear: "endometritis was characterized by lymphoplasmacytic infiltrates in the lamina propria and neutrophils in uterine glands and within necrotic debris and exudate in the uterine lumen" (Williams et al. 1993). B. abortus has since been shown to cause abortion in bison from YNP (Rhyan et al. 1994). The Jackson herd is infected with B. abortus; seroprevalence in 35 bison sampled in 1989-90 was 77%, and B. abortus was cultured from 4 of 11 (36%) seropositive bison (GYIBC 1997).
In 1985, B. abortus was cultured from 6 of 88 bison (Clark and Kopec 1985), and in 1991-92, from 26 of 218 bison (Aune and Schladweiler 1992). Recently, B. abortus biovar 1 was isolated from an aborted fetus found near Old Faithful and a stillborn calf located outside the YNP; both were heavily infected with B. abortus (billions of organisms per gram of tissue). Data on a retained placenta from a bison shot and sampled on the north side of YNP established that B. abortus infects the placenta and causes abortion in bison in a manner similar to its effects on cattle. Furthermore, vegetation and soil were sampled at two bison birth sites (Lamar area samples taken May 8 and 22, 1996; North Gate area samples taken May 16, 1996); B. abortus remained viable in soil for 14-18 days after abortion occurred (Roffe et al. 1997).
Serologic evidence of brucellosis in YNP bison was first reported in 1917, and 40-54% of YNP bison tested have been reported as seropositive since then (GYIBC 1997). Serologic evidence indicates that as many as 60% of YNP bison (Tunnicliff and Marsh 1935, Pac and Frey 1991, Aune and Schladweiler 1992) and 77% of GTNP bison (Williams et al. 1993) contain serum antibodies against B. abortus. Those data do not include animals that are infected but do not have a measurable antibody response at the time of sampling.
Immune responses from natural infection might induce some degree of
protection against B. abortus, inasmuch as substantial fetal loss or infertility has not been reported for the bison populations in the GYA (Olsen et al. 1998). Experimental studies suggest that bison are more susceptible to brucellosis than are cattle or elk (Davis et al. 1990, 1991): nearly all infected female bison aborted their first calf.
Elk
In North America, substantial brucellosis in wild elk occurs only in the GYA. Seroprevalence among adult female elk in the western Wyoming feeding-ground complex has averaged 37% since 1970.2 During herd reductions of the 1960s, 1.7% of 6,027 elk on the northern range were brucellosis test reactors; YNP elk in the northern herd have not been tested recently. Elsewhere, seropositivity (Morton et al. 1981; Smith and Roffe 1992; Rhyan et al. 1997) was
-
Six of 126 elk trapped northwest of the YNP during spring of 1988.
-
Less than 0.03% of 3,833 elk tested in Colorado from 1967-1976.
-
None of 170 Idaho elk.
-
Three of 113 Utah elk.
-
Two of 178 Wyoming elk not associated with feeding grounds.
In the GYA, B. abortus is unlikely to be maintained in elk in the absence of the bison reservoir and if the elk winter feeding grounds are closed. Elk have much lower seropositive rates on natural winter ranges in Wyoming in comparison with elk on feeding grounds. Over time, elk would not serve as reservoirs for brucellosis in the absence of elk feeding grounds.
Research at the Sybille Research Unit in Wyoming has shown that 50-70% of female elk that become infected with B. abortus lose their first calf (Thorne et al. 1979); retained placentae and infertility do not occur in elk as they do in cattle (Thorne and Herriges 1992).
Transmission from infected elk in captive herds to susceptible cattle occurred when the two species were in close contact and pregnant elk gave birth or aborted (Thorne and Herriges 1992). Such transmission was believed to be extremely unlikely in normal calving on traditional elk calving ranges
and probably occurs only in the close confines of elk feeding grounds. "Elk do not seem to be capable of sufficient intraspecific transmission of brucellosis to maintain the disease in the population when not concentrated on feeding grounds" (Thorne and Herriges 1992).
Serologic evidence of brucellosis has been found in blood samples from elk corralled in 18 of 23 feeding grounds with seropositive rates averaging 37% in adult females (Herriges et al. 1991). Blood samples from hunter-killed elk outside the GYA that were tested in 1970-1992 were negative (Thorne and Herriges 1992). Normal calving behavior—calving in seclusion and clearing the placenta—almost completely removes the likelihood that an elk will transmit brucellosis to another animal (see p. 45, "Bison and Elk Behavior and Transmission") (Thorne and Herriges 1992).
Infection in Other Mammals in the GYA
Canids
The assessment of risk of carnivore transmission involves three separate issues: the risk of transmission from bison or elk to the carnivore, the probability that infection will be maintained in that species, and the frequency of transmission back to bison or elk.
B. abortus has been isolated from wild carnivores in areas that contain infected bison and elk (Tessaro 1986). Those predators consume infected elk and bison meat, especially during the winter and early spring, and frozen infectious material distributed by scavengers cannot be overlooked (A. Clark, Ore. Dept. Agricult., pers. commun., 1997). Coyotes (Canis latrans) can be infected with B. abortus (Davis et al. 1979, 1988), but serologic surveys of coyotes have not revealed exposed animals in the GYA (Gese et al. 1997). No surveys of coyotes on feeding grounds have been reported, but exposure might occur. There is no evidence that coyotes are important in the epidemiology of brucellosis in the GYA, although coyotes elsewhere have been found to be infected and able to shed the organisms for a short period (Davis et al. 1979, 1988).
Dogs infected with B. abortus typically do not develop clinical signs of disease, although dogs with brucellosis do suffer abortion, epididymitis, and lameness associated with joint lesions (Forbes 1990). Seroconversion can occur 4-14 days after exposure, and a positive serotiter can be maintained for as long as 2 years (Kiok et al. 1978). Seronegative, culture-positive dogs have been reported. In a study of 14 dogs from farms with B. abortus-infected cattle,
B. abortus was cultured from lymph nodes draining the head, neck, lungs, and intestines, and urogenital infection with shedding was seen in one bitch (Forbes 1990). The maximal duration of infection was 539 days; that suggests that dogs have the potential to infect bison and elk. Dogs have also been experimentally infected orally with vaccine strains of B. abortus, and in pregnant females, the placenta was infected, although abortion did not occur (Palmer and Cheville 1997).
Moose
From 1937 to 1985, four cases of brucellosis in wild moose (Alces alces) were reported, each proved by isolation of B. abortus from multiple tissues. In three cases, the moose had been in contact with cattle that had brucellosis. Clinical signs in all four were weakness and debility. Lesions in the carcasses included swollen lymph nodes, pneumonia, and fibrinous exudates on the pleura, the pericardium, and other serous surfaces. Fibrinous exudates from the lungs and serous surfaces are characteristic of pasteurellosis; Pasteurella multocida was isolated from one of the moose (Corner and Connell 1958), so it is questionable whether brucellosis was the cause of death.
Despite the presence of brucellosis in moose, substantial titers of antibody against B. abortus have not been found in free-ranging moose in North America, even in areas where moose are in contact with infected cattle. That might be the origin of the widely held belief that infection with B. abortus is generally fatal in moose. All moose tested in the GYA and in Montana have been serologically negative, and that finding might have buttressed the conclusion that all infected moose die, leaving no residuum of seropositive, chronically infected moose.
To determine susceptibility, Forbes et al. (1996) experimentally inoculated four moose conjunctivally with B. abortus biovar 1. No acute phase of disease developed—no clinical signs, no abnormal blood values, no abnormal serum chemistry. Two moose were killed at day 70 after inoculation, one died at day 85, and one was killed at day 166. None of the moose had clinical signs of disease, except the moose that died, which had fever; clinical pathology data remained normal. B. abortus was isolated from several tissues, most notably lymph nodes, where bacterial counts often exceeded 4 × 104 CFU/g of tissue (Forbes et al. 1996). Although the authors suggest that "lesions seen in all moose were indicative of endotoxemia," no data presented supported that conclusion or established that any tissue change was caused by B. abortus.
If brucellosis is generally fatal in moose, it must be unusual. Acute death from brucellosis would be unique; infected ruminants do not develop life-threatening febrile disease. It has been widely claimed that moose die of a septicemic brucellosis with tissue responses that include peritonitis, pleuritis, and other acute inflammatory lesions. The evidence that such lesions are directly due to B. abortus is not convincing, and the pathogenesis of brucellosis in moose needs to be resolved.
It is not likely that moose play a major role in sustaining infections with B. abortus in wildlife in the GYA. Moose are also susceptible to B. suis (Dieterich et al. 1991); if they are in contact with swine or caribou, that bacterial species should be investigated.
Horse
Horses can be infected with several Brucella spp. and have been reported to be a source of B. abortus infection in cattle (White and Swett 1935). B. abortus has a predilection for joints, bursae, and tendons, and the common clinical features of equine brucellosis are suppurative spinous bursitis ("fistulous withers") and suppurative atlantal bursitis ("poll evil"). Although mares infected with B. abortus have been reported to abort, experimentally infected mares have produced normal, noninfected foals at full term (MacMillan et al. 1982). B. melitensis and B. suis (from feral pigs) have also been reported in horses (Cook and Kingston 1988). A 9-year-old pregnant mare that had contact with elk in a winter feeding ground 5 miles south of Jackson, Wyoming, developed fistulous withers that contained pus from which B. abortus was isolated. The mare later foaled, and neither the foal nor the placenta had evidence of B. abortus. A second horse used to pack out hunted elk also had fistulous withers and positive serologic tests; bacterial cultures did not grow B. abortus, but the horse had been treated with tetracycline before culture was attempted. Horses used by rangers and those brought into the GYA by park visitors for recreation are susceptible on contact with tissues or fluids of infected elk or bison.
Bear, Deer, and Other Big Game
Records from the Montana Livestock Sanitary Board laboratories that tabulate blood tests in big-game animals from 1932 to 1968 show that seroreactors were not found in antelope (Antilocapra americana), bighorn sheep (Ovis canadensis), mule deer (Odocoileus hemionus), moose, mountain goat (Oreamnos
americanus), and grizzly bear (Ursus arctos); one seroreactive black bear (Ursus americanus) was found (Barmore 1968). Brucellosis recently was detected in black bear and grizzly bear in the greater Yellowstone ecosystem (K. Aune, Mont. Fish, Wildlife, and Parks, pers. commun., 1997). The extent of infection in bear is not known, but bear are unlikely to play a major role in the persistence of brucellosis in YNP (see Part II, "Transmission Among and Between Species").
Mule deer outside YNP have been shown to be seropositive, but deer in YNP have not been shown to carry B. abortus, and it is widely assumed that deer are not a major host for it. Brucellosis has not been detected in Montana in mule deer or white-tailed deer (O. virginianus).


























