| This page in the original is blank. |
Overview
JANIE M. FOUKE
National Science Foundation
Arlington, Virginia
Photographs surfaced recently of a creature that can serve as an emblem of the paradigm we are about to enter in biomechanics. It was a mouse, about 4 inches long, and on his back was what appeared to be a surgically implanted human ear. The ear was at least 75 percent as large as the mouse himself.
That alone is remarkable—a huge appendage on the back of a mouse. What is even more remarkable is that this implanted tissue was viable by every known measure. Our colleagues at the Massachusetts Institute of Technology and the University of Massachusetts had built a human ear from human cartilage cells. They grew it in the laboratory and implanted it surgically in the back of a mouse.
This is an emblem of where medical science and engineering may take us in the new millennium. We call this field tissue engineering, and it is nurtured by, founded on, and supported by biomechanics. The field did not exist 15 years ago.
Many biological materials can be coaxed to grow in a culture dish—skin, bone, and cartilage are being generated artificially, constructed, and tested in clinical settings right now. Skin is already on the market; one can buy artificially constructed skin. Certainly many problems remain, including issues associated with growing three-dimensional rather than two-dimensional structures and issues associated with vascularization and innervation. Nonetheless, the progress has been exciting.
The focus of these papers from the Biomechanics session of the Third Annual Symposium on Frontiers of Engineering is on bone and cartilage and their mechanical properties. It is important to realize that implanted cells do not function in isolation. For them to be biologically active, they have to
coalesce and respond as normal tissues do in many respects. That is, not only are they driven by and able to drive mechanical events, but they also interact with their chemical and physical environments. Biological consequences result from and can trigger the mechanical activity discussed in the papers in this volume.
The role of biomechanics in biological systems is not surprising if one thinks about it on a macrolevel. One of the earliest-known mechanoreceptors in the human body is the baroreceptor—tiny structures that exist near the carotid artery, which supplies blood to the brain. They are exquisitely sensitive to pressure. In fact, they are the mechanical sensor in the mechanical feedback loop controlling blood pressure. A familiar scene on certain TV shows and movies is that of someone coming up behind a character and applying a sudden pressure to his neck, triggering an abrupt change in blood pressure and blood flow to the brain, which causes the person to pass out. This is the baroreceptor response, which is a mechanoreceptor in the body.
There are other places where mechanoreceptors play important roles. There is a lot of evidence to indicate that the cells that line the arteries are mechanosensitive—in other words, sensitive to shear, stress, and strain. As a consequence, through mechanisms that are not yet well understood, lipids are deposited. Thus, there is a relationship between the mechanical response of the cell on the artery and the development of atherosclerosis.
The first paper in this section, ''Biomechanics of Cells and Cell-Matrix Interactions," discusses mechanosensing properties of cells and the ability of cells to generate mechanical responses. It focuses on cartilage and, to some extent, bone, and reminds us that changes in the mechanical environment alter the homeostatic set point. All mechanisms in our bodies strive toward a stasis, a balance. One example is the balance between bone creation and bone resorption. When bone is stressed and consequently the cells are stressed, regrowth occurs. In the absence of that we get deterioration.
More examples of this type are described in the second paper, "Mechanical Influences on Bone Development and Adaptation." That paper focuses on the mechanical environment in which bone thrives and in which the adaptive responses elicited by physiochemical factors amplify and supplement the responses to mechanical loading. Genetic factors play into this; however, the interplay between the physicochemical biological milieu and the mechanical milieu is critically important.
The third paper, "Implant Design and Technology," covers the design of structures to work with, supplement, and in some instances, replace bones and joints. There have been spectacular successes in the past 20 years with artificially implanted mechanical structures. There have also been spectacular failures. For instance, if 15 years ago we had known about the role of mechanical events in building bone, we probably never would have designed an artificial implant that removes the load from the bone. This is because once
the load is removed, the bone deteriorates, creating a hostile environment for the implant.
The papers in this section, which cover cellular, tissue, and constructed environments for bone structures, have some common themes. One is the interplay between the biochemical and the mechanical environments and how one feeds and triggers the other. Another theme is that the advances discussed could not have occurred without major developments in technology, in particular the ability to measure exquisitely tiny forces. Technological developments such as new imaging modalities, microscopic modalities, adaptively analytical finite element models, large-scale numerical modeling ability, and computational power have been critical factors in the intellectual advances in physiology and biomechanics discussed herein.
Biomechanics of Cells and Cell-Matrix Interactions
FARSHID GUILAK
Duke University Medical Center
Durham, North Carolina
Cells are the fundamental structural and functional entities that form the basis of life. The physiological activity of a cell is a tightly controlled process that is regulated by the genetic programming of the cell in combination with signals derived from the chemical and physical environments. In the human body the interaction of living cells with physical forces, such as mechanical stress, is critical to the normal health and function of most organ systems. Cells not only have extraordinary capabilities to generate mechanical forces but also can possess exquisite abilities to detect and respond to mechanical signals in their environment. In many cases the sensitivity and efficiency of living cells in interconverting chemical and mechanical energy surpass those of the best man-made systems.
However, the mechanisms through which mechanical forces interact with cells are not fully understood. The study of the biomechanics of living cells is a rapidly growing, multidisciplinary field that brings together expertise from several different areas to study problems in medicine and biology. The field of cell mechanics utilizes concepts of engineering, medicine, biology, chemistry, and physics to further our understanding of the biological and biophysical mechanisms involved in bone and joint disease, cardiovascular disease, wound healing, reproduction, and cancer. Recent advances in engineering technology have had a significant impact on this field by introducing new experimental and theoretical techniques for the study of cellular biomechanics (Mow et al., 1994).
Mechanical Signal Transduction
Many cells of the body are specialized in their ability to detect mechanical signals for sensory purposes such as touch and hearing (Hudspeth, 1992).
However, most nonsensory cells also have the ability to perceive mechanical signals in their environment and to transduce them into an intracellular chemical signal. Cells of the musculoskeletal, cardiovascular, and respiratory systems are particularly sensitive to their mechanical environment and depend on mechanical signals to properly regulate their activity. This capability of mechanical signal transduction is necessary due to the large metabolic demand required to maintain and regenerate living tissues of the body; tissues that are not adequately utilized are not regenerated. For example, bone is normally in a balanced physiological state such that the rate of tissue resorption equals the rate of new tissue formation. Changes in the mechanical environment of the cells can shift the balance of synthesis and degradation to adapt to increased or decreased mechanical requirements by producing or resorbing surrounding tissue (Rubin et al., 1994; van der Meulen, 1998). Similarly, muscle cells rapidly grow in response to the physical demands of exercise and atrophy with lack of use (Costill et al., 1979). In many cases, mechanical loads are required for the maintenance of healthy tissue, and insufficient loading can lead to a pathological condition of tissue loss (e.g., osteoporosis).
The mechanisms by which single cells or ensembles of cells are able to transduce relatively small forces and deformations involve a complex cascade of biophysical and biochemical events that are not fully understood. Many recent advances in this field have been made through the application of new theoretical and experimental engineering technologies in combination with techniques in cell biology. In particular, new microscopy and imaging techniques coupled with large-scale numerical modeling have been very useful in providing new information on the micromechanical environment of single cells.
One paradigm system that will be reviewed is the process of mechanical signaling in articular cartilage, the tissue that serves as the bearing surface for the joints of the body. Cartilage cells (chondrocytes) perceive and respond to the cyclic mechanical loads generated by the activities of daily living such as walking and running. Under normal conditions, the chondrocytes are able to maintain the integrity of this tissue over decades of cyclic loading at peak stresses of up to 18 megapascals (˜2600 psi) with little wear or degeneration. However, under abnormal circumstances, mechanical loading can lead to progressive joint degeneration with such diseases as osteoarthritis. Therefore, knowledge of the specific mechanisms of signal transduction would provide new insight into the causes of osteoarthritis and may lead to the development of new techniques for the prevention or treatment of the disease.
Large-scale numerical computing has been utilized to theoretically model the mechanical and biological behavior of living cells and their interaction with the extracellular matrix. These models require a detailed knowledge of the mechanical properties of cells and their various components, as well as information on the constitutive laws governing their behavior. New techniques using micropipette manipulation as shown in Figure 1 (Guilak et al.,
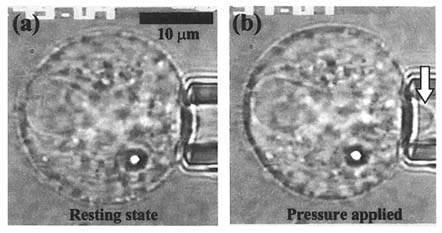
FIGURE 1 The micropipette aspiration test for determining the elastic and viscoelastic properties of living cells. A glass micropipette (inner diameter˜5 µm) is used to apply pressure to a single cell, and the resulting deformation of cell is recorded using video microscopy (arrow). A theoretical analysis is combined with these experimental measurements to determine the properties of the cell.
1996; Hochmuth, 1993) and atomic force microscopy (Shroff et al., 1995) have been successful in quantifying the mechanical properties of individual cells and their components. The major engineering contribution of these techniques has been the ability to measure forces on the order of piconewtons and displacements on the order of nanometers. These measurements, in conjunction with a continuum viscoelastic model of the cell in its extracellular matrix (Guilak and Mow, 1992), have provided new information on the spatial and temporal distributions of stress and strain around a single cell under both normal and disease conditions. Validation of these theoretical predictions has been made using three-dimensional confocal scanning laser microscopy to quantify the changes in shape and volume that living chondrocytes undergo as articular cartilage is subjected to physiological levels of compression (Guilak, 1995). Confocal scanning laser microscopy has also been used to elucidate the earliest intracellular events that occur in response to mechanical stimuli. With this technique, it has been shown that cells can respond to mechanical stress within milliseconds by rapidly altering the concentration of intracellular ions such as calcium (Guilak et al., 1994).
Mechanical Force Generation
The ability of cells to generate force is clearly a necessity for bodily functions such as locomotion, respiration, and blood circulation. On a
smaller scale, however, many important cellular and subcellular activities require the generation of force by "molecular motors" within the cell. For example, contractile forces and controlled intermolecular motion drive the locomotion of single cells, which is involved in numerous physiological processes such as wound healing, reproduction, and cellular invasion (e.g., metastasis of cancer cells). The generation of internal forces is also necessary for critical cellular processes such as cell division and intracellular transport of various molecules.
Cellular force generation in these cases is a series of reactions by which chemical energy is used to translocate one molecule relative to another (e.g., actin and myosin in the case of skeletal muscle). The magnitude of the forces involved in the mechanics of single cells are on the order of nano–to piconewtons. These forces often represent the interactions between individual molecules and are at the limit of detection using current technologies. New advances in engineering technology have enabled measurement and application of extremely low forces in this range using such techniques as atomic force microscopy, optical gradient traps, micropipette manipulation, and microelectromechanical systems.
For example, most cells migrate by extending the leading edge of their plasma membrane as a pseudopod ("false foot"), which is then attached to the underlying substrate and used to pull the cell forward. These gross cellular motions are driven by forces generated by mechanochemical enzymes such as the molecule kinesin. Recent studies have been able to quantify the force generated by a single molecular motor. These forces were measured with a laser-induced, single-beam optical gradient trap, also known as optical tweezers. This device can be used to generate forces of a few piconewtons using a gradient in laser light that traps a latex microsphere. In this experiment it was found that the motion of kinesin could be stopped by exerting a force of approximately 2 piconewtons (Kuo and Sheetz, 1993). The ability to measure forces generated by single macromolecules now permits determination of the fundamental thermodynamic mechanisms of mechanochemical force transduction by living cells.
Cell-Matrix Interactions and Cell Adhesion
The transmission of force between a cell and its extracellular matrix occurs through adhesion molecules on the cell membrane that bind to specific matrix proteins. Thus, an understanding of the processes involved in the formation and dissociation of these bonds is an important aspect of the inter-action of cells with mechanical forces. Several recent studies have sought to measure the force of a single molecular bond between a cell membrane receptor and an extracellular protein using novel transducers with piconewton force resolution.
Development of the atomic force microscope has revolutionized the quantification of intermolecular forces by allowing accurate and precise measurement of extremely low forces (10 to 1,000 piconewtons) at frequencies of up to 20 kiloherz (Chilkoti et al., 1995; Engel, 1997). Several other novel techniques based on micropipette manipulation have been developed for quantifying the force of adhesion of a single molecular bond. These techniques are based on fluid pressures being applied through a glass micropipette with a diameter of 1 to 5 microns. One technique has involved using a blood cell that has been pressurized by micropipette suction as an ultrasensitive force transducer. Deformation of the cell can be measured with nanometer resolution using interference microscopy. By altering the tension, the sensitivity of the transducer can be tuned through a range of forces from 0.01 to approximately 1,000 piconewtons (Evans et al., 1995). By coating the transducer and the target region with specific molecules, the kinetics and strength of individual bonds can be studied.
Future Directions
With the development of such techniques as atomic force microscopy, optical gradient traps, and micropipette manipulation, it is now possible to measure the extremely low forces and displacements generated by molecular interactions in living cells. Simultaneously, rapid advances in computational speed and power have made it possible to develop large-scale continuum and structural models of the interactions between tissues and cells. With improved imaging and microscopy techniques, recent studies have been able to decrease the geometric scale of these models while increasing the level of molecular detail that is modeled. As this trend continues, we approach the fundamental limits of continuum mechanics, and new approaches are incorporating structural models of cells and tissues at the molecular level. It is now becoming necessary to acquire information on the intrinsic mechanical properties and behavior of single molecules and the interactions between individual molecules.
Acknowledgments
A segment of the research presented in this paper was supported by grants from the Whitaker Foundation and the National Institutes of Health (AR43876).
References
Chilkoti, A., T. Boland, B. D. Ratner, and P. S. Stayton. 1995. The relationship between ligandbinding thermodynamics and protein-ligand interaction forces measured by atomic force microscopy. Biophysical Journal 69:2125–2130.
Costill, D. L., E. F. Coyle, W. F. Fink, G. R. Lesmes, and F. A. Witzmann. 1979. Adaptations in skeletal muscle following strength training. Journal of Applied Physiology: Respiratory, Environmental & Exercise Physiology 46:96–99.
Engel, A. 1997. A closer look at a molecular motor by atomic force microscopy. Biophysical Journal 72(3):988.
Evans, E., K. Ritchie, and R. Merkel. 1995. Sensitive force technique to probe molecular adhesion and structural linkages at biological interfaces. Biophysical Journal 68:2580–2587.
Guilak, F. 1995. Compression-induced changes in the shape and volume of the chondrocyte nucleus. Journal of Biomechanics 28:1529–1542.
Guilak, F., and V. C. Mow. 1992. Determination of chondrocyte mechanical environment using finite element modeling and confocal microscopy. ASME Advances in Bioengineering BED–20:21–23.
Guilak, F., H. J. Donahue, R. Zell, D. A. Grande, K. J. McLeod, and C. T. Rubin. 1994. Deformation-induced calcium signaling in articular chondrocytes. Pp. 380–397 in Cell Mechanics and Cellular Engineering, V. C. Mow, F. Guilak, R. Tran-Son-Tay, and R. M. Hochmuth, eds. New York: Springer-Verlag.
Guilak, F., H. P. Ting-Beall, W. R. Jones, G. M. Lee, and R. M. Hochmuth. 1996. Mechanical properties of chondrocytes and chondrons. ASME Advances in Bioengineering BED-33:253–254.
Hochmut, R. M. 1993. Measuring the mechanical properties of individual human blood cells. Journal of Biomechanical Engineering 115:515–519.
Hudspeth, A. J. 1992. Hair-bundle mechanics and a model for mechanoelectrical transduction by hair cells. Society of General Physiologists Series 47:357–370.
Kuo, S. C., and M. P. Sheetz. 1993. Force of single kinesin molecules measured with optical tweezers. Science 260:232–234.
Mow, V. C., F. Guilak, R. Tran-Son-Tay, and R. M. Hochmuth, eds. 1994. Cell Mechanics and Cellular Engineering. New York: Springer-Verlag.
Rubin, C., T. Gross, H. J. Donahue, F. Guilak, and K. McLeod. 1994. Physical and environmental influences on bone formation. Pp. 61–78 in Bone Formation and Repair, C. T. Brighton, G. Friedlaender, and J. M. Lane, eds. Rosemont, Ill.: American Academy of Orthopaedic Surgeons.
Shroff, S. G., D. R. Saner, and R. Lal. 1995. Dynamic micromechanical properties of cultured rat atrial myocytes measured by atomic force microscopy. American Journal of Physiology 269:C286-C292.
van der Meulen, M. C. H. 1998. Mechanical influences on bone development and adaptation. Pp. 12–15 in Frontiers of Engineering: Reports on Leading Edge Engineering from the 1997 Symposium on Frontiers of Engineering. Washington, D.C.: National Academy Press.
Mechanical Influences on Bone Development and Adaptation
MARJOLEIN C. H. VAN DER MEULEN
Cornell University
Ithaca, New York
The vertebrate skeleton is a wonderfully adaptive structure whose form is determined in part by its structural function. The bone tissue of the skeleton consists of a hard mineralized matrix that has living cells embedded in it. These cells produce the extracellular matrix around themselves and enable the skeleton to respond to its environment by altering their matrix production. Many biophysical factors influence bone cell function, including mechanical loading, hormones, growth factors, and intrinsic genetic composition. For centuries the ability of the skeleton to adapt its structural form has fascinated scientists, including Galileo and Darwin.
Mechanical regulation of skeletal biology begins during embryogenesis and continues throughout postnatal life. Most skeletal elements form initially as cartilage prepatterns in the embryo. In the human embryo, involuntary muscle contractions begin 6 weeks postconception, coinciding with the initiation of ossification in the cartilage rudiments. These muscle contractions result in joint motions and tissue strains that guide the ongoing skeletal development. After birth, continued skeletal growth is strongly influenced by mechanical forces that affect both bone size and density. Mechanical loading-based influences are particularly evident in the diametrical growth of the dense cortical bone found at the midshaft and in the structure and anisotropy of the porous trabecular bone at the epiphyses of the long slender bones of the appendicular skeleton (Carter et al., 1996).
Because normal growth is mechanically regulated, alterations in mechanical loading due to physical activity, injury, or implants induce an adaptive skeletal response. Experimental studies examining bone functional adaptation to mechanical loading have been performed for over a century. In the adult, increased
mechanical loading results in increased bone formation; decreases in loading result in bone loss, or resorption (Biewener and Bertram, 1993). Analytical engineering-based approaches are becoming more common and need to be combined with experimental data. In the past decade, adaptive analytical and finite element models have been developed to simulate the functional adaptation of both the continuum and microstructural features of bone (Beaupréet al., 1990; Mullender et al., 1994; van der Meulen et al., 1993).
Whereas the overall nature of the adaptive response of bone has been well documented, the mechanisms that transduce a mechanical stimulus to a biological signal to increase or decrease bone mass are not understood, including the stimulus, signal transduction pathway, and response process (Duncan and Turner, 1995). Without this knowledge, our ability to quantitatively predict adaptation is very limited. Important questions that need to be addressed include possible physical stimuli associated with mechanical loading, including cell deformation, fluid flow, streaming potentials, and fatigue damage. We also need to understand how bone cells sense these stimuli and respond to their environment, such as the research described in the previous paper, Biomechanics of Cells and Cell-Matrix Interactions. However, the results of such in vitro cell culture studies must be applied to understanding the in vivo behavior of cells, tissues, and organs.
Technological advances in imaging, computing, and molecular biology have started to impact our understanding of the skeleton, and these techniques not only need to be implemented but, more importantly, integrated. Emerging techniques for imaging are allowing us to observe and ask questions about structures and phenomena never before visualized. The architecture of trabecular bone has traditionally been evaluated by reconstructing two-dimensional serial sections. In the rat, trabecular widths and separations are around 50 and 150µm, respectively. Micro-computed tomography (micro-CT) is a noninvasive method for assessing three-dimensional trabecular bone structure. Micro-CT scans of bone samples can nominally obtain resolutions of 14µm (Müller and Rüeggsegger, 1997). Current limitations include high radiation doses and sample size restrictions. These systems are slowly becoming widely available and are considered the standard for trabecular morphometry. Further improved resolution can be obtained using monochromatic synchrotron radiation (Kinney et al., 1993). Synchrotron-based x-ray tomography provides spatial resolutions of 2 to 8 µm and can be performed in vivo but is clearly not widely available. In addition to detailed structural data, the tomographic intensities also provide the bone mineral density locally at the same resolution.
Increased computational speed and availability have led to more complex models and algorithms. Both continuum and cellular-level models of bone have been used, but each level of analysis has traditionally been examined independently. Enhanced computational power coupled with improved imaging resolution has led to the development of models incorporating continuum,
tissue, and cellular details. Using micro-CT imaging, very detailed models of trabecular microstructure have been developed (van Rietbergen et al., 1997). These models convert the micro-CT voxels directly to cubic finite elements, producing finite element models with 10 5–106 elements. Comparisons between continuum apparent behavior can be made with tissue-level predictions.
Genetic factors underlie the development of bone and cartilage, but external physicochemical factors, such as mechanical loading, control and modulate gene expression. Improved molecular biology techniques allow us to essentially map cells, genes, and proteins throughout tissues. Specific questions about protein expression during bone adaptation can now be addressed, particularly in embryological experiments. In experiments on bone growth and development, the presence and patterns of expression of proteins believed to play a role in skeletal tissue formation can be followed. In addition, the ability to deactivate, or knock out, genes has enabled us to begin to examine the contributions of individual genes (Mikic´ et al., 1996). To date, these studies have primarily been used to map normal gene expression, and we are beginning to address the interaction between gene expression and epigenetic factors (van der Meulen and Allen, 1997).
The difficulty in combining these technologies arises from the interdisciplinary nature of this research. Measurements made in the laboratory need to be related to the inputs required for engineering models. Validation of analytical models is critical to their success and can only be achieved by better integration with experimental technology. Experiments need to be designed with measurements that can be incorporated into models. Observations made on cells in culture need to be translated to in vivo organ responses. Communication and interaction between engineers, biologists, clinicians, and other is critical and will determine future advances.
The potential impact of understanding bone adaptation to biophysical stimuli is significant: mechanical factors are implicated in many orthopedic and endocrine skeletal disorders, including total joint replacements, fracture healing, arthritis, and osteoporosis. Osteoporosis is a severe worldwide health problem. In the United States, 9.4 million (30 percent) postmenopausal Caucasian women are estimated to have osteoporosis when the condition is defined by low bone mass; half of these women are estimated to have severe osteoporosis, based on experiencing one or more fractures. For 1986 the economic cost of osteoporosis and associated fractures in the United States was estimated to be $7 billion to $10 billion (U.S. Department of Health and Human Services, 1991). Fractures experienced by osteoporotic individuals are the direct consequence of bone loss, which compromises skeletal ability to withstand loads. Therapies for increasing bone mass include exercise and pharmacological agents; therefore, an understanding of the relationship between mechanical loading and bone mass changes during bone adaptation is important. Osteoarthritis is the end-stage of cartilage degeneration and is
associated with many factors, including mechanical loading. Osteoarthritic cartilage damage affects nearly half of all individuals over age 65 and often requires costly total joint replacements, as discussed in more depth in the following paper, Implant Design and Technology . As the elderly population worldwide increases in the coming decades, the incidence and cost to society of load-related skeletal conditions also will increase, motivating the need to better understand skeletal functional adaptation.
References
Beaupré, G. S., T. E. Orr, and D. R. Carter. 1990. An approach for time-dependent modeling and remodeling—theoretical development. Journal of Orthopaedic Research 8:651–661.
Biewener, A. A., and J. E. A. Bertram. 1993. Mechanical loading and bone growth in vivo. Pp. 1–36 in Bone, Volume 7: Bone Growth-B, B. K. Hall, ed. Boca Raton, Fla.: CRC Press.
Carter, D. R., M. C. H. van der Meulen, and G. S. Beaupré. 1996. Mechanical factors in bone growth and development. Bone 18(S1):5–10.
Duncan, R. L., and C. H. Turner. 1995. Mechanotransduction and the functional response of bone to mechanical strain. Calcified Tissue International 57:344–358.
Kinney, J. H., T. M. Breunig, T. L. Starr, D. Haupt, M. C. Nichols, S. R. Stock, M. D. Butts, and R. A. Saroyan. 1993. X-ray tomographic study of chemical vapor infiltration processing of ceramic composites. Science 260:789–792.
Mikic', B., M. C. H. van der Meulen, D. M. Kingsley, and D. R. Carter. 1996. Mechanical and geometric changes in the growing femora of BMP-5 deficient mice. Bone 18:601–608.
Mullender, M. G., R. Huiskes, and W. Wienans. 1994. A physiological approach to the simulation of bone remodeling as a self-organizational control process. Journal of Biomechanics 27:1389–1394.
Müller, R, and P. Rüeggsegger. 1997. Micro-tomographic imaging for the nondestructive evaluation of trabecular bone architecture. Pp. 61–79 in Bone Research in Biomechanics, G. Lowet, P. Rüegsegger, H. Weinans, and A. Meunier, eds. Amsterdam: IOS Press.
U.S. Department of Health and Human Services. 1991. Objective 17.18 in Healthy People 2000: National Health Promotion and Disease Prevention Objectives. Report No. 91–50212. Washington, D.C.: U.S. Government Printing Office.
van der Meulen, M. C. H., and W. A. Allen. 1997. BMP-5 and bone mechanobiology. Transactions of the Orthopaedic Research Society 22:403.
van der Meulen, M. C. H., G. S. Beaupré, and D. R. Carter. 1993. Mechanobiologic influences in long bone cross-sectional growth. Bone 14:635–642.
van Rietbergen, B., R. Müller, D. Ulrich, P. Rüeggsegger, and R. Huiskes. 1997. Quantitative assessment of tissue loading in a proximal femur, using a full scale microstructural FE-model . Transactions of the Orthopaedic Research Society 22:62.
Implant Design and Technology
AVRAM ALLAN EDIDIN
Osteonics Corporation
Allendale, New Jersey
Mechanical replacement of diseased or traumatized joints with engineered structures is one of the great medical triumphs of the past 30 years. Over time the indications for joint replacement surgery have broadened faster, and to include even younger patients, than has science's ability to engineer longer-lived devices. Society's reliance on increasingly more technological solutions to the challenges of aging has cast into the field of implant design the holy grail of a lifetime prosthesis coupled with a minimum alteration of the patient's lifestyle. To this end the engineer, scientist, and clinician must synthesize their ideas and experiences with an eye to understanding that hypothesis testing can take many years, if not decades.
Until about 1960 the normal degradation of joint function with increasing age, or due to systemic disease such as rheumatoid arthritis, was considered a regrettable but essentially nontreatable chronic condition. In about 1960 Sir John Charnley of the United Kingdom developed the idea of replacing the articulating mechanism of the hip with an artificial construct consisting of a stainless steel ball affixed to an implantable stem articulating with a plastic (polyethylene) bearing. In addition, he pioneered the idea of using a polymer grout (methylmethacrylate) to fix the stem within the femoral canal. The true breakthrough, however, was in the choice of a bearing that was substantially smaller than the natural head in order to reduce the torque generated by the mechanism. Thus, the kinematics of the hip joint were restored using a design that varied from the apparent optimum design provided by biology.
In subsequent years, as the artificial hip became an accepted treatment for natural joint degradation, attention turned to the other large weight-bearing joint in the body—the knee. In France a group of designers developed what
became known as the Guepar knee, which took its design cues from the natural knee's suggestion that a hinge coupling the femur and tibia would best replace a damaged knee. While moderately successful in patients with limited physical activity, the design neglected to permit the rotatory motion about the natural knee, and the torque thus generated in the artificial construct tended to tear the components out of their bone bed. Once again, it took a sideways path away from the natural biology to develop a kinematically successful knee replacement. Eventually designs that mimicked the bearing surfaces of the natural knee while permitting the still-intact ligaments to dictate the kinematics were developed and used successfully beginning in the early to mid-1970s.
While other joints in the body, including the shoulder, elbow, and wrist to name a few, can be replaced with man-made constructs, hip and knee replacements dominate the field, and much research today is focused on improving the longevity and, to a lesser degree, the function of these two joint replacements. If one momentarily assumes that the kinematics of the designs are roughly optimized, the research questions that arise tend to be almost exclusively in the domains of material and interface improvements.
Early hip and knee prostheses were fixed to the bone using an in situ polymerizing polymer. Within about a decade following widespread acceptance of hip replacement in the medical community, patients began to present with lesions about the stemmed portion of the implant in the femoral canal, caused by localized osteolysis. Thin lesions, while not destroying the bone per se, had the effect of decoupling the bone-implant construct, which led to pain and necessitated reimplantation of a second stem after cleaning out the femoral canal. Larger lesions increased the risk of fracture of the femur itself due to thinning of the femoral tube's walls and thus posed an ongoing problem even if the original stem was exchanged. In time these osteolytic changes were attributed to the release of microscopic amounts of methylmethacrylate in particulate form, leading to a localized macrophage response. Design focus then shifted to development of femoral stems that could be implanted without the polymer grout and thus development of a direct interface between the living bone tissue and the (almost always) metal stem. These implant designs, known as cementless implants, came into clinical use in the early 1980s.
Early cementless implants were made of a cobalt-chromium alloy surfaced with tiny beads to create pores into which the living bone would grow and thus form a mechanical bond and load transferring interface between the femur and the implant. Strategies for placement of the beads varied by design, but it became apparent that in order to achieve bony integration with the implant the relative motion between the implant and the bone had to be reduced to below about 100 µm. For a time research efforts focused on optimizing the stem design to make it as motionless within the femoral canal as possible, a constraint that had previously been nulled out by the use of the polymer grout. In time one old and one new problem arose with the use of
these cementless stems: osteolysis in the femoral canal and loss of bone density about the implant. The first problem was the one supposedly solved by the use of a cementless construct, and the second was a problem already understood to be caused by a transfer of load from the relatively low stiffness bone to the very stiff implant, thus bypassing or shielding the bone from load and causing an adaptive response leading to lowered bone density.
Further development that continues today in femoral stem design has chiefly been oriented toward reducing the structural stiffness of the implantable stem, thus increasing the bone's share of the joint loads and reducing the adaptive response leading to bone resorption. Titanium alloy stems were developed that had the chief advantage of being twice as flexible on a size-for-size basis as an equivalent cobalt-chromium stem. They had a second advantage of naturally passivating to a coating of titanium dioxide, which biological studies had shown was more biointegrable than the cobalt-chromium surface of the previous generation of implants. Unfortunately, because of the notch sensitivity of the material, the beads used previously to encourage bonding of the bone to the stem could not be used without risk of fatigue fracture of the stem itself. Attempts to implant the stems directly without any mechanically roughened interface were generally unsuccessful. Attempts to encourage bone ingrowth into pads of fibrous titanium affixed to the stem were more successful, but results were still much more varied than the uniformly good record of cemented implants in the early to midterm.
If the interface was the problem, the solution lay in finding a material that would be both biologically compatible and mechanically feasible. Hydroxylapatite, a calcium phosphate that is the naturally occurring mineral phase of bone, was chosen as a natural biologically compatible material. However, since it is a ceramic with very low bulk strength, attempts to apply the material to stems in large amounts led to delamination. Application of a very thin layer using a plasma spray process was much more successful in maintaining a bond between the metal and the ceramic while still presenting a biologically compatible surface to the host bone. Hydroxylapatite-coated stems came onto the U.S. market in the late 1980s and to date have shown a significantly lowered propensity toward bone resumption, presumably because of the enhanced load transfer to the bone as a function of the reduced structural stiffness of the stem coupled with an enhanced interface between the living bone and the calcium phosphate layer.
The second failure mode, osteolysis, is now known to be caused by small particles of almost any material, be it titanium, cobalt-chromium, or more especially polyethylene. As younger and more active patients are fitted with prostheses because of their overwhelming clinical success, more wear of the plastic bearing is seen. Research since the early 1990s has focused extensively on both characterizing polyethylene as it exists today and on ways of increasing its resistance to abrasive wear while reducing the polymer's natural
tendency to degrade through oxidation. Development of inert gas packages and enhanced cross-linking through high-energy irradiation are some of the techniques being developed to reduce the level of particulates generated in the artificial joint. Alternative bearing couples, such as ceramic-ceramic bearings that wear almost imperceptibly, also are being developed in the United States, although earlier designs somewhat more prone to fracture because of larger grain sizes have been used in Europe for over a decade.
The kinematics of implant function have come under scrutiny as newer methods of in vivo visualization, such as full-motion cinefluoroscopy, have permitted scientists to compare the functions of natural and replaced joints, especially knees. Recently, nearly 100-year-old ideas regarding how the femur bends and rotates about the tibia have been challenged as a result of data emanating from fluoroscopy studies, and devices incorporating some of these findings are now entering the market. Recognition that the plastic bearing in knee devices is subject to very different loads than that in hip devices has led to a bifurcation in studies on how to improve this material. Specifically, while polyethylene destined for hip bearings may need to be designed to resist abrasive wear, the same material used in a knee bearing may need to resist microcracking arising from high cyclic contact stresses.
The overall goal of research into the design and fabrication of implants today is to produce man-made devices that last the natural life of the recipient. While in many areas people no longer believe that technology will rescue them from whatever challenge befalls, in medicine the same people place a trust on the engineer, scientist, and physician that all ailments can be cured or at least substantially mitigated—hence, the continued study of artificial prostheses, their material composition, and their interaction with the living body.
| This page in the original is blank. |




















