2
Process Description and Scientific Principles
PROCESS OVERVIEW
The general scheme for the neutralization of VX and treatment of the neutralization residues is shown in Figure 2-1 (Stone and Webster, 1997a). The block diagram indicates a number of discrete operations in the overall process of VX destruction. Each block in the diagram represents more than one unit operation. For example, the left block (TCC/Neutralization) represents activities associated with removing VX from its storage cylinders, decontaminating the cylinders, and neutralizing the agent.
The ton containers (TCs) in which the agent is stored are transported to a ton container cleanout (TCC) facility where they are perforated with a mechanical punch and drained of liquid agent (U.S. Army, 1996b). This operation is very similar to the “punch and drain” operation at the chemical stockpile destruction facilities at Johnston Island and Tooele, Utah. However, in contrast to the thermal decontamination process used to decontaminate the drained containers at incineration sites, the Newport TCC process involves cutting the drained containers into two sections and using steam and hot water sprays to rinse and decontaminate the metal surfaces. The liquid collected from the decontamination process, which contains small amounts of VX as well as some agent hydrolysis products, will be collected and treated by the agent neutralization process. The Army intends to thermally treat and smelt the decontaminated metal parts at another Army facility.
The liquid VX drained from the TCs is treated with hot (90°C) 13.6 wt. percent aqueous NaOH solution in
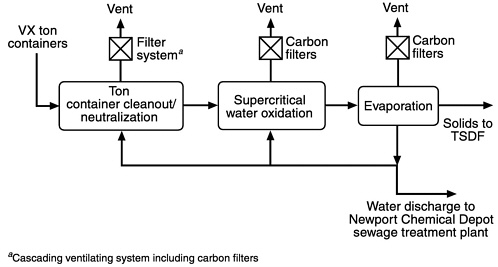
FIGURE 2-1 Block diagram of the overall neutralization and SCWO treatment process. Source: Adapted from Stone and Webster, 1997a.
a vigorously stirred reactor for three to four hours, during which time the VX is almost completely hydrolyzed (described in Chapter 1). The hot reaction mixture is then transferred to a second stirred reactor that contains rinse water from the TCC operation. This mixture is heated for an additional four to five hours to destroy the VX and EA-2192 to concentrations below their analytical detection limits (Stone and Webster, 1997a). Prior to release from the neutralization process step to subsequent treatment steps, both VX and EA-2192 must be nondetectable in the hydrolysate, with detection limits established at 20 ppb for VX and 5 ppm for EA-2192. After completion of the neutralization reaction, the hydrolysate is held in the reactor for several hours to allow time to certify by chemical analysis that it is suitable for release from the TCC/neutralization process area to the SCWO process unit (see Figure 2-1 ). The residual EA-2192, which could be present in higher concentration than VX, poses no vapor hazard because this highly acidic compound will be present only as its sodium salt in the alkaline hydrolysate. However, the liquid does have some toxic characteristics.
After analysis has verified that adequate destruction of VX and EA-2192 has been achieved, the VX hydrolysate is transferred to the SCWO unit, which is the focus of this report. The strongly alkaline hydrolysate is heated and pumped into a SCWO reactor along with an oxidizing agent (air or oxygen) and heated to 600 to 650C° under about 275 bar pressure. In the course of about 30 seconds, the organic components are largely (about 99.99 percent) oxidized to water and sodium carbonate, phosphate, and sulfate, as well as gaseous nitrogen-containing products (e.g., N2 and NOx). This mixture of materials is cooled by adding quench water and through heat exchange and then released from the SCWO reactor through a pressure reduction system. The resulting effluent is a mixture of gases (O2, N2, CO2), a concentrated aqueous salt solution, and entrained solid salts (Stone and Webster, 1997b). Trace concentrations of partially oxidized organic constituents (e.g., MPA and acetic acid) may also be present. Complete analyses are shown in Tables 3-7 and 3-8 .
The aqueous products from the SCWO reactor, including entrained solids, are then fed to the evaporation unit, where the mixture is heated to distill excess water. At this point, the salts that have crystallized from solution are filtered and packaged for disposal in a secure landfill. A large portion of the water distilled from the SCWO effluent is recycled back to the TCC and SCWO units, which reduces the amount of fresh water needed for process operations. In the case of VX, the amount of water produced by oxidation is generally greater than the amount required to satisfy process needs; consequently, some water will be discharged to the Newport Chemical Depot sewage treatment facility or another acceptable water treatment facility.
Each major unit in the process will have a vent for the release of gaseous materials. Each vent will pass through a bed of activated carbon that will collect residual traces of organic vapors prior to releasing the gas into the atmosphere.
SCIENTIFIC PRINCIPLES
SCWO (supercritical water oxidation) is a hydrothermal process for the oxidative destruction of organic wastes using air or oxygen in the presence of high concentrations of water heated above the critical temperature and pressure of pure water (Tc = 374°C, Pc = 221 bar). In the last two decades, SCWO technology has been the subject of several reviews (Modell, 1989; Shaw et al., 1991; Gopalan et al., 1995; Tester et al., 1993). The reaction mechanisms for the destruction of organic compounds generally involve free radical (mostly •OH and •OOH) chain reactions with oxidative radicals. Thermal bond cleavage and polar or ionic reactions, including hydrolysis, also occur under the severe reaction conditions. Very high destruction efficiencies (> 99 percent to 99.9999 percent) have been reported for the destruction of a large number of organic compounds when they are treated for residence times of seconds to minutes at temperatures of 400 to 650 °C. Aqueous organic wastes with sufficient heating value can be autogenic and can sustain the necessary reaction temperatures for extensive oxidation without the addition of supplemental fuel. Tests by General Atomics showed that the VX hydrolysate was autogenic under test conditions (General Atomics, 1997b). Although fuel was added during some tests, it appears to have been unnecessary for sustaining the reaction (Stone and Webster, 1997b).
In a SCWO process like the one in Figure 2-1 , the feed stream (aqueous waste to be treated) to the SCWO reactor is heated, pressurized, and mixed with oxidant (as air, pure oxygen, or hydrogen peroxide). It is then pumped through a flow reactor at supercritical conditions designed to provide the required residence time (seconds to one minute typically). Heat produced by the oxidation reactions can be recovered (or must be
removed) based on the heating value of the feed stream. If the feed stream has inadequate heating value to heat the reactor, supplemental fuel can be added. The Army will choose one of three oxidants. Liquid oxygen can be pumped, whereas air must be compressed; compressors are noisy and complicated (for the high pressure required here). There is little experience with the third choice, hydrogen peroxide.
Downstream of the reactor, the system is depressurized (“let down”), either before or after cooling. Solids produced from oxidation reactions can be recovered prior to or following pressure let-down. Cooling prior to pressure let-down often results in the redissolution of salts, which can be precipitated later via solvent evaporation. Alternatively, pressure let-down at high temperature can result in the direct removal of salts as a concentrated brine stream or solid. A number of pressure let-down schemes have been devised—mostly valves or capillary tubes that restrict the release of fluids from the process equipment. The effluent is then passed through gas/liquid separators, and the gas stream and aqueous streams can be treated further, as required, for responsible disposal. With sufficient salt removal, the water effluent can often be recycled within the system.
In general, organic carbon is oxidized to carbon dioxide or carbonate. Under the strong oxidizing conditions of this medium (assuming sufficient oxidant is available), most heteroatoms in organic material (e.g., Cl, P, and S) are converted to their oxidized mineral acids (HCl, H3PO4 and H2SO4, respectively) or the corresponding salts, depending on effluent pH and cation concentration. The fate of nitrogen in organics is unclear; it has been reported to yield mixtures of N2 and nitrogen oxides, including N2O, with the possible formation of nitrate as well (Killilea et al., 1992). Nitrate has been shown to be an effective oxidant in SCWO systems reactions to yield N2 and N2O (Proesmans et al., 1997; Dell'Orco et al., 1995). In the treatment of VX hydrolysate, the oxidation would produce large quantities of inorganic salts:
![]()
Very hot water, near or above its critical point, behaves much differently from water at standard conditions. The density of water at its critical point (Tc = 374°C, Pc = 221 bar) is about 0.3 g/cm3. As water is heated and pressurized, its dielectric constant decreases precipitously (from 88 at 0°C to less than 20 at 300°C and significantly less than 10 at much higher temperatures). For a pure substance at its critical point, the density of the vapor and the liquid is identical, and the two fluids become indistinguishable. At temperatures above the critical point, a single phase of the substance is formed, which is known as a “supercritical fluid.” Supercritical water is highly compressible; its density depends on both temperature and pressure. Its solubility properties and most other physical properties (e.g., diffusivity, dielectric constant, viscosity) depend on its density.
A significant factor is that the solvent properties of very hot water differ greatly from those of normal water. Simple gases like oxygen or nitrogen are miscible at relatively high concentrations. Organic compounds often are soluble or miscible in supercritical water because of its low dielectric constant. At pressures much greater than at the critical point, and therefore at high density, salts can be soluble and can even dissociate. However, at the lower densities (0.1-0.2 g/cm3) common to most SCWO conditions (206 bar and > 600°C), most salts are practically insoluble.
KINETICS
The oxidation kinetics of some simple organic compounds under homogeneous SCWO conditions have been measured and modeled (Li et al., 1991; Li et al., 1993; Gopalan and Savage, 1995). Phenomenologically, a near zero order dependence on oxygen concentration is observed if sufficient oxygen is present. The oxidation of certain compounds, such as methane, show nonintegral dependence on oxygen. Most organic compounds can be destroyed to concentrations below detection limits within short reaction times at temperatures of 400 to 650°C. However, some small molecules, notably ammonia and acetate ion, are more recalcitrant and require higher temperatures or longer reaction times for complete oxidation. MPA (methylphosphonic acid) has been demonstrated to react only slowly in bench and pilot test studies of VX hydrolysate, suggesting slow cleavage of the carbon-phosphorus bond (Gloyna and Li, 1997).
The complete oxidation of even simple organic compounds requires a large number of elementary reactions. Although a number of reactions have been measured and modeled extensively in simple systems, most organic wastes have been described best by phenomenological kinetic models. For more complex systems, only empirical models specifying the residence times required to achieve certain destruction efficiencies at the
specified temperature and pressure have been developed. Moreover, the reaction mechanisms for complex wastes in SCWO systems are poorly understood, including the elementary steps, the interplay or relative contribution of homogeneous and heterogeneous reactions, and pyrolytic or hydrolytic chemical pathways.
PHASE BEHAVIOR
Understanding the phase behavior of the reaction medium within a SCWO reactor is essential for confident scale-up and smooth operation. A number of attempts have been made to model or predict phase behavior for ideal systems (Chiavlo et al., 1995; Bischoff and Rosenbauer, 1988; Bischoff and Pitzer, 1989; Marshall et al., 1981; Valyashko, 1976). They have generally assumed the miscibility of water, organics, and air and, on that basis, assumed homogeneous reactions near or above the critical point. However, this simple assumption is true only for dilute solutes in water. The phase behavior of concentrated systems (even systems with just a few percent solute like VX hydrolysate) can be very complex and is probably multiphase because the critical point of water depends strongly on the concentration of solutes (e.g., gases, organic compounds, and salts). Thus, the critical point of a concentrated stream could be significantly higher than the critical point of pure water (Armellini et al., 1994; Bischoff and Rosenbauer, 1988; Bischoff and Pitzer, 1989), suggesting that vapor phases or concentrated and dilute aqueous phases could persist considerably above the critical temperature. Salt precipitation would lead to additional phases. The presence of more than one phase or the presence of subcritical phases in the reactor can significantly alter reaction mechanisms, kinetics, and destruction efficiencies, as well as solids retention and corrosion rates.




