This paper was presented at a colloquium entitled “Neuroimaging of Human Brain Function,” organized by Michael Posner and Marcus E.Raichle, held May 29–31, 1997, sponsored by the National Academy of Sciences at the Arnold and Mabel Beckman Center in Irvine, CA.
Neural components of topographical representation
GEOFFREY K.AGUIRRE, ERIC ZARAHN, AND MARK D’ESPOSITO*
Department of Neurology, University of Pennsylvania Medical Center, Philadelphia, PA 19104–4283
ABSTRACT Studies of patients with focal brain damage suggest that topographical representation is subserved by dissociable neural subcomponents. This article offers a condensed review of the literature of “topographical disorientation” and describes several functional MRI studies designed to test hypotheses generated by that review. Three hypotheses are considered: (i) The parahippocampal cortex is critically involved in the acquisition of exocentric spatial information in humans; (ii) separable, posterior, dorsal and ventral cortical regions subserve the perception and long term representation of position and identity, respectively, of landmarks; and (iii) there is a distinct area of the ventral occipitotemporal cortex that responds maximally to building stimuli and may play a role in the perception of salient landmarks. We conclude with a discussion of the inferential limitations of neuroimaging and lesion studies. It is proposed that combining these two approaches allows for inferences regarding the computational involvement of a neuroanatomical substrate in a given cognitive process although neither method can strictly support this conclusion alone.
The topographical domain may be defined as that space that is beyond our immediate perceptual horizon. Topographical spaces include the interior of a building or the town in which the building is located. We routinely generate internal representations of these spaces, as well as their contents, which suffice to guide us from place to place. There are, perhaps, as many different kinds of representations as there are possible solutions to the problem of way-finding, ranging from inflexible, route-based lists of right and left turns to map-like representations that encode the metric distance relationships between landmarks (1). These representations can be cast within different spatial frames, including egocentric (body-centered) and exocentric (world-centered) coordinates (2). There is evidence that these different types of representations can be brought to bear in combination and in isolation, depending on the features of the environment (3) and the means (4) and goals (5) of exploration. The aim of this article is to review some recent studies regarding the neural basis of these types of topographical representations.
There are three primary ideas that will be presented here. All three fall under the more general proposal that distinct functional, neuroanatomical components, which normally operate in concert to provide the seamless percept of orientation, differentially contribute to the representational processes mentioned above. Each of these hypotheses are related to the performance deficits from which topographically disoriented brain-damaged patients suffer, and these hypotheses have been tested explicitly in intact subjects by using functional magnetic resonance imaging (fMRI).
The first hypothesis is that the medial-temporal lobes are regionally subspecialized for the acquisition of topographic information. The hippocampus holds a privileged position (1) in theories regarding exocentric spatial representation based on the identification of place cells (neurons with receptive fields for current position in the environment) within the rat hippocampus (6) and the demonstration that hippocampal lesions impair place learning in the rat (7). As will be described, however, there is little evidence that lesions of the hippocampus in the human lead to selective impairments in topographical ability. Instead, the evidence from lesion studies (8–10) and recent neuroimaging studies (11–13) suggests that the parahippocampus subserves neural computations that are predominantly required for the acquisition of novel topographical knowledge.
Second, we propose that distinct neocortical structures represent conceptually distinct components of topographical knowledge. Environmental psychology studies of normal subjects suggest that knowledge of topographic space is not behaviorally unitary. Instead, it appears that people can acquire separate representations of the appearance of places and their relative spatial positions (4, 14, 15). A review of the literature of topographical disorientation (provided below) suggests that these behavioral components are subserved by separable, neuroanatomical components although the existence of this division has been questioned by other workers in the field. A recent neuroimaging study in intact subjects (16) explicitly tested this idea and demonstrated distinct dorsal and ventral areas that respond to the consideration of, respectively, landmark position and identity.
Third and finally, we offer the possibility of yet another functional domain within inferior temporal cortex. There is now considerable evidence (see, e.g., refs. 17–19) that the inferior temporal cortex contains an anatomically segregated population of neurons that are specialized for face processing. As will be described, patients suffering from a variety of topographical disorientation, termed “landmark agnosia,” appear specifically unable to recognize salient features of their environment, particularly buildings. The frequent, but not absolute, co-occurrence of this disorder with prosopagnosia (inability to recognize faces) suggests that there may be an area of cortex near the “fusiform face area” (19) that is specialized for the perception of buildings.
Throughout this article, we will draw on the rich, but at times disordered, neurological literature of topographical disorientation. Over the last century, several dozen cases of these patients who are unable to find their way within their locomotor environment have been reported. The interpretation of these cases has been debated, and the neuroimaging studies described here have been designed to address some of these controversies.
The Medial Temporal Lobes and Place Learning
Studies of Place Learning in Rats, Monkeys, and Humans. Because of impressive findings from electrophysiology (6) and
© 1998 by The National Academy of Sciences 0027–8424/98/95839–8$2.00/0
PNAS is available online at http://www.pnas.org.
|
|
Abbreviation: fMRI, functional MRI. |
|
* |
To whom reprint requests should be addressed. e-mail: despo@mail.med.upenn.edu. |
lesion studies (7) in the rodent, the hippocampus has been offered (1) as the site of a “cognitive map” responsible for the flexible representation of exocentric position. The specificity of the role played by the hippocampus (i.e., Ammon’s horn, the dentate gyrus, and the subiculum) in spatial representation subsequently has been debated at length (see, e.g., ref. 20). At the very least, it is clear that selective (neurotoxic), bilateral lesions of this structure in the rodent greatly impair performance on “place” learning tasks such as the water maze (21, 22).
The importance of the hippocampus for exocentric spatial representation in the human has been more difficult to demonstrate. Spatial memory tests that present fixed stimulus arrays to a stationary patient (see, e.g., refs. 23 and 24) are not strictly relevant because the cognitive map theory proposes a flexible, map-like representational role for the hippocampus. In contrast to other sites (described below), unilateral lesions of the hippocampus do not produce any appreciable real world way-finding impairments in humans (25). The existence of anterograde way-finding deficits in patients with general anterograde amnesia after bilateral lesions of the hippocampus (and adjacent structures) (26–28) has not been commented on explicitly. However, if present, any topographical difficulties obviously would be accompanied by memory impairments in other areas. Retrograde loss of way-finding knowledge in these patients is not apparently disproportionate to losses in other areas (27) and can be preserved (29). Based on these findings, if the hippocampus is indeed necessary for the representation of topographical space in humans, then it must be said (i) that way-finding in previously learned places can be accomplished in its absence (29) and (ii) that place learning is but one of many kinds of knowledge for which it is necessary [i.e., place learning is a type of declarative memory (30)].
fMRI Studies of Topographical Learning. We have attempted to examine the neural correlates of exocentric spatial learning in the human by using fMRI (11). The purpose of this study was to create conditions under which neuroanatomical structures that increase their activity during the acquisition of place knowledge could be identified. This information could then be used to either support or question assertions regarding the role of different medial-temporal lobe structures for these kinds of memory tasks. Nine subjects were studied with fMRI during their free exploration of a “virtual reality” maze (Fig. 1A and B). The signal obtained during these periods was compared with that obtained while subjects repetitively traversed a simple corridor. Subjects were tested both on their ability to produce sketch maps of the maze (Fig. 1C) and to perform way-finding tasks within the environment from novel start points. The ability of our subjects to perform these tasks strongly suggests, but does not absolutely prove (31), the acquisition of exocentric representations. The interpretation of this experiment relied on two assumptions. The first was that the navigation of a virtual reality environment engages the same cognitive processes used during real world way-finding (32). The second was the “cognitive subtraction” assumption, which allows one to attribute differences in signal between the two conditions to the putatively isolated cognitive process of exocentric learning (33).
The primary finding of the study was that, within the medial-temporal lobes, activity was confined to the parahippocampal gyrus (Fig. 2A).† The laterality of this activity varied among the nine subjects, with three each demonstrating left, right, and bilateral signal changes. Differences in signal between the two conditions also were noted in several other areas of the brain, including (bilaterally) the posterior-parietal cortex, retrosplenial cortex, and medial-occipital cortex. No
|
† |
The parahippocampal gyrus is comprised of several, distinct cortical fields, including the entorhinal cortex, the parahippocampal cortex (areas TH and TF) and perirhinal cortex (34). Our use of the specific term “parahippocampal cortex” and the general term “parahippocampal gyrus” (when more specific anatomical statements cannot be made) is deliberate. |
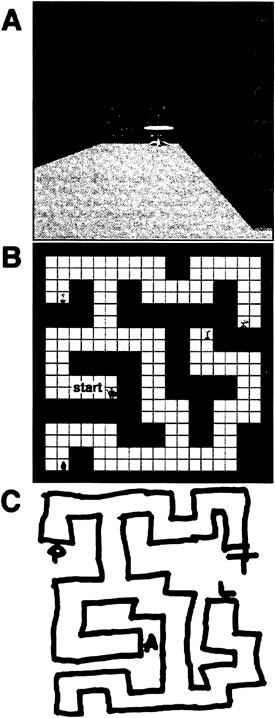
FIG. 1. Stimuli from an fMRI study of topographical learning. (A) Subject view. Actual stimuli were in full color and were several times wider in the horizontal plane than pictured. (B) Aerial view of maze. (C) Representative subject rendition of maze.
signal changes were observed within the hippocampus in any subject.
This finding of parahippocampal activity associated with topographical learning has received additional support from subsequent positron emission tomography (PET) studies. In one study (13), subjects explored a virtual reality environment with and without useful landmarks. A comparison of these two conditions revealed activity in the right parahippocampal gyrus whereas comparison of either condition to baseline control tasks (involving joystick movements in response to textured and colored images) did not identify signal changes within the hippocampus. This study was interpreted as demonstrating a role for the parahippocampus in embedding landmarks in a spatial framework. In a separate study (12), signals measured while subjects viewed (and attempted to learn a route from) a video-taped trip through a town was compared with that obtained while subjects watched a film of urban activity taken from a stationary position. Bilateral parahippocampal gyrus activity was found although extension of activity into the most posterior portion of the right hippocampus also was noted. Thus, although parahippocampal activity has been associated with the acquisition of novel topographical knowledge, activity within the hippocampus has not been observed consistently.
Are we able to conclude from these studies alone that the parahippocampus is necessary for exocentric learning and that the hippocampus is not involved? In truth, the inferential power of these studies (11–13) in isolation is limited because the behaviors under study are enormously complicated, thus making any attempt to isolate them by “subtraction” questionable. It always will be possible that activity in the parahippocampus (or any other area) is the result of confounding and uncontrolled behaviors that differ between the two conditions. The presence of activity in this structure across several different experiments and tasks (11–13) is reassuring but cannot be taken as absolute evidence for the involvement of the parahippocampus in topographical learning. The claim that necessity cannot be demonstrated by using neuroimaging techniques is discussed in greater detail below.
Anterograde Topographical Disorientation and the Parahippocampus. When these neuroimaging results are considered along with studies of brain-damaged patients, however, an inferentially grounded account emerges. After having suffered isolated lesions of the right parahippocampal gyrus (8), patients are unable to learn their way around new environments but have preserved way-finding in places known several months to years before their injury. These deficits are accompanied, if at all, by only minor anterograde memory impairments, generally confined to the visual domain, and in contrast to other categories of topographically disoriented patients described later, these patients are not grossly disoriented in egocentric space or agnosic. Almost exclusively, they are unable to acquire exocentric topographical information. The lesion site that has been reported to produce this disorder accords well with the site of neuroimaging signal change seen in our study of topographical learning (Fig. 2B).
Recent reports also have examined the effect of lesions in this area. Twenty patients with unilateral medial-temporal lesions (half on either side) were tested on a video-taped route learning task (9). Although these patients denied retrograde topographical disorientation and did not have any measurable general memory impairments, they were impaired relative to controls on tests of route learning and exocentric position judgments. All 20 patients had surgical resections of the parahippocampal gyrus. The authors did not report any difference in performance or clinical presentation between the patient groups with small (excluding hippocampus) or large (including hippocampus) resections. Of interest, patients with
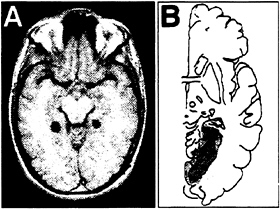
FIG. 2. (A) Single axial slice showing voxels with significant fMRI signal changes during putative topographical learning in one subject. Right and left are reversed. Signal changes were observed, among other areas, bilaterally within the parahippocampal gyrus. (B) Common lesion site in four patients with anterograde topographical disorientation (8). Areas of lesion overlap are indicated by progressively darker shades of gray. The common lesion site is in the right parahippocampus.
left or right excisions had roughly equivalent impairments. Another recent report (10) found different effects of laterality although with much smaller sample sizes. Fourteen patients with well defined thermocoagulation lesions of the medial-temporal lobes were tested on a human analog of the Morris water maze task. Patients with lesions confined to the right parahippocampal cortex were impaired relative to those with lesions of the right or left hippocampus and epileptic controls. This report is particularly helpful because (i) the critical lesion site was well defined within the parahippocampal cortex, (ii) it provides a within-task comparison of hippocampal and parahippocampal lesions, and (iii) it uses a well defined test of exocentric spatial learning.
Thus, in concert with evidence that lesions of the parahippocampus produce an anterograde topographical disorientation, the neuroimaging results reviewed here suggest that the parahippocampus subserves some computational function necessary for the acquisition of novel route-based and exocentric topographical knowledge. Moreover, and in contrast to the hippocampus, interruption of this function seems to produce fairly selective deficits in topographical learning.
A specialized role in spatial learning (broadly defined) has been proposed previously for the parahippocampal cortex based on the neocortical inputs to the area (including inferior caudal visual areas, retrosplenial cortex, and the superior parietal lobule) (34). As is described below, some of these sources of input have been implicated (16) in the representation of components of environmental information. Thus, the parahippocampal cortex is in a position to associate particular landmarks (represented in ventral occipito-temporal regions) with particular spatial relationships (represented in posterior parietal cortex) (35). A recent positron emission tomography (PET) study, which reported right parahippocampal gyrus activity (albeit localized to the entorhinal cortex) during an object-place memory task but not during object-only or place-only memory conditions, is intriguing in this context (36). Finally, the relationship of this proposed function of the parahippocampus with the demonstrated importance of the perirhinal and parahippocampal cortices for general memory in monkey models of amnesia (37, 38) requires further consideration. It is certainly possible that damage to the parahippocampal cortex interrupts some basic associative/computa-
tional function to which exocentric learning is exquisitely sensitive.
Neocortical Representations of Environmental Space
Several types of information are necessary inputs to an exocentric representation of space. Knowledge of the visual features and constituent elements of the environment must be combined with information regarding the position of those features with respect to oneself as well as an integrated representation of one’s movement through the space (39). What is the neural basis of the representation of these components of environmental knowledge? Further consideration of the topographical disorientation literature is potentially instructive. Disoriented patients with parahippocampal lesions are only a small subset of a larger group. There also is a fairly heterogeneous collection of case studies of neocortical damage, which vary widely in lesion site and nature of deficit. The interpretation of these cases has been, however, an issue of some debate. Some have proposed that these subjects divide into those with an inability to represent the identity of environmental landmarks and those with a primarily “spatial” deficit (40–42). This division of environmental knowledge is reminiscent of the well studied “what vs. where” framework of visual processing (43–45). Other authors have argued against the existence of such a cognitive (8) or anatomical (46) division. Instead, a unitary representation of both the identity and allocentric location of environmental features has been proposed to exist in a ventral cortical area (47).
We have considered the extant cases of topographical disorientation and have found evidence for the existence of different types of impairment. Our taxonomy of this disorder is similar in several respects to the basic landmark/spatial divisional model that has been proposed previously. There are, however, some nuances and additions to the account offered here. We propose that patients traditionally labeled as topographically disoriented with neocortical damage divide into several major classes, which are briefly described below. (This review is, of necessity, cursory and does not consider in detail contentious cases. A comprehensive review is forthcoming). We also describe two neuroimaging experiments based on the ideas generated by this review. The first fMRI study tests the divisional model of representation, and the second attempts to identify an area of ventral occipitotemporal cortex specialized for landmark representation.
Landmark Agnosia. The first class of topographically disoriented patients can be described as “landmark agnosics,” in that the primary aspect of their impairment is an inability to use prominent, salient environmental features for the purposes of orientation (40, 48–53). Although able to produce maps and describe routes, suggesting intact spatial representation, these patients are unable to recognize previously familiar landmarks. In particular, these patients seem unable to recognize specific buildings, leading some to dub this disorder a specific building agnosia. Typically, they rely on small environmental details for orientation, such as the mailbox in front of their house, thus demonstrating some measure of intact spatial representation and object recognition.
Other visual deficits have been noted to co-occur with landmark agnosia, such as prosopagnosia and achromatopsia. These impairments do not invariably accompany landmark agnosia (48, 50), however, and are known to occur without accompanying topographical disorientation (54). Thus, it is unlikely that these ancillary impairments are actually the causative factor of disorientation. Where detailed lesion information has been available, the lesion has been on the right side, within ventral occipitotemporal cortex, sparing the structures of the hippocampal system. Most commonly, these impairments are produced by infarctions of the posterior cerebral artery, which supplies this area.
Egocentric Disorientation. A second group of patients who traditionally have been labeled “topographically disoriented” do not, in fact, have selective deficits within the exocentric spatial sphere (41, 42, 55–57). Although able to identify salient environmental objects, they are unable to represent the spatial relationship between these objects and themselves. A frequent demonstration of this disability is that, although the patient can point to a visualized object, they are no longer able to do so with their eyes closed. Thus, patients with egocentric disorientation become topographically disoriented as a consequence of this more basic impairment. Lesions of the right posterior parietal cortex are characteristic of this deficit.
Because of the level on which these patients are disoriented, it is not possible to determine from these cases alone whether the exocentric impairments are a consequence of the more basic egocentric disorientation or whether the damaged neuroanatomical site normally supports both functions. Notably, cells with exocentric firing properties have not been identified in the rodent parietal cortex although cells responsive to complex conjunctions of stimulus egocentric position and ego-motion have been reported (58). Therefore, a likely possibility is that, although the right parietal cortex does not explicitly represent exocentric spatial information, the computations that take place there are a necessary component of exocentric representations developed elsewhere. The last division of topographical disorientation speaks to this possibility.
Heading Disorientation. A final group of patients raise the interesting possibility that exocentric spatial representations can be damaged selectively. A few cases describe patients who are both able to recognize salient landmarks and do not have the dramatic egocentric disorientation described above. Instead, these patients are unable to derive directional information from landmarks that they recognize. It seems that they have lost a sense of exocentric direction, or “heading,” within their environment. Because there are so few of these cases and because they have not been subjected to extensive testing, these observations must be regarded as tentative. It is interesting, however, that the few cases that have been reported possess lesions within the retrosplenial (i.e., posterior cingulate) cortex (59–60). Studies in rodents (61) have identified cells within this area that fire only when the rat is maintaining a certain heading or orientation within the environment. These cells have been dubbed head direction cells (62) and likely generate their signals based on a combination of landmark, vestibular, and idiothetic (i.e., self-motion) cues.
fMRI Study of Divisional Representation. The taxonomy provided above makes different predictions from those in which a single cortical area is proposed to represent landmark position and identity (47). One such prediction of the divisional model is that a ventral area specialized for representation of environmental features will be recruited to a lesser extent during judgments regarding the position of places compared with periods in which the appearance of places is considered. We designed and conducted an fMRI study to test this prediction (16).
Four subjects explored, over a period of several days, a detailed virtual reality town. The town was comprised of 16 distinctive, named “places” interconnected by a variety of roads and paths and arranged in a 4×4 grid, with a river that divided the town roughly in half (Fig. 3A). fMRI was used to observe regional brain activity while subjects recalled information regarding the environment. During two alternating conditions, subjects were presented with snapshots (Fig. 3B) of the town. During one condition, subjects were asked whether the name of the presented place matched the
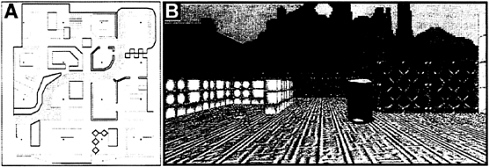
FIG. 3. Virtual reality town in which subjects were trained before scanning. (A) Aerial view. Subjectively, the town was ≈140 meters in width. (B) View of one location within the town. Each location was designated as such by the presence of a marker in the ground.
snapshot shown. In the other condition, subjects were given the name and picture of a location and were asked to determine the spatial (survey) direction of a second, named target location. The two tasks used identical stimuli, differing only in the aspect of topographical knowledge that the subject was required to recall. A direct comparison of these two conditions was hypothesized to reveal a posterior dorsal/ ventral dissociation. In addition, activity observed during judgments of appearance and position was compared with a visuo-motor control task in which subjects alternated button presses in response to scrambled snapshots. The purpose of this last comparison was to identify neural substrates common to both tasks. Those regions for which only one task condition produced significant changes above baseline might be considered areas with “specific” responses to the recall of identity or position.
A direct comparison of the appearance and position conditions revealed a consistent dorsal/ventral dissociation across subjects (Fig. 4A). The superior and inferior parietal lobule possessed significantly greater activity during the position task compared with the appearance task. The lingual and inferior fusiform gyri were found to have greater signal during the appearance condition. Compared with the baseline condition (Fig. 4B), both tasks activated an extensive area of posterior cortex, including parietal, retrosplenial, and lingual cortices. The parahippocampal gyrus bilaterally was activated also by both tasks relative to the baseline condition. Only the inferior fusiform gyrus and inferior parietal lobule bilaterally were activated by a single task condition. For these two areas, we may state that not only was there a dissociation of activity between the two tasks but a specificity of response.
These findings are in agreement with the proposal that distinct neural areas represent different components of environmental knowledge and that subjects might draw on these components selectively. The observed areas of selective response are in good agreement with the lesion literature reviewed above. It seems reasonable to propose that these neocortical areas provide input to the parahippocampus where an integrated representation is generated. Notably, greater parahippocampal signal was observed during both the appearance and position tasks relative to the control task whereas neither comparison revealed hippocampal activity. Potentially, the parahippocampus is involved in the retrieval of recently acquired topographical knowledge as well as its acquisition.
fMRI Study of Building Perception. We might further ask whether the fusiform gyrus site we have identified is involved in general visual feature analysis or whether it plays a specific role in the representation of environmental landmarks. The location of this site (i.e., within inferior occipitotemporal cortex) agrees with the reported lesion sites responsible for
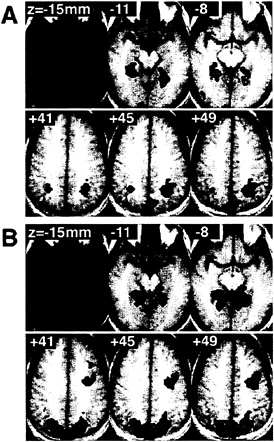
FIG. 4. Sites of replicated significant activity across subjects. Right and left are reversed. (A) Shown are three inferior and three superior axial slices through a brain in standard space. In shades of blue are those voxels where multiple subjects had significantly greater signal during the appearance task compared with the position task; in shades of red are those voxels with greater signal during the position task compared with the appearance task. (B) In shades of green are shown those voxels that had significantly greater activity in both the appearance and position tasks compared with the baseline task over multiple subjects.
landmark agnosia. Additionally, and as might have been predicted from the lesion literature, the area is in the vicinity of that reported for face-processing tasks, as detected by depth electrode recording and neuroimaging studies (19, 62, 63). Because some degree of prosopagnosia frequently (50), but not invariably (54), co-occurs with topographical agnosia, we have proposed that there might exist separable areas of inferior temporal cortex that are specialized for the perception of faces and landmark information. In particular, the deficits that landmark agnosic patients display might be explained by damage to a cortical area responsible for representing buildings, a class of objects highly confounded with landmarks. We might further expect that cells in such a region would be “tuned” to respond maximally to building stimuli in a manner analagous to “face” areas (19). We are conducting fMRI experiments to test this idea and present here the results of one of these studies.
The experiment was designed to detect the neural correlates of perception of exemplars from different stimulus categories. Before scanning, subjects studied one picture from each of three categories: faces, buildings, and general inanimate objects. During scanning, subjects viewed blocks of 10 grayscale pictures from each category, each picture presented for 3000 ms. If the subjects detected a picture that matched one studied, they were to make a bilateral button press on a fiber optic game pad. If the picture was new, the subjects were instructed to not make any overt response. Targets were infrequent (≈1 in 10 stimuli). The order of the three stimulus blocks, and a fixation condition, were fixed within subjects but varied across subjects. Data were analyzed using the general linear model for serially correlated error terms (65), modified to accommodate the null-hypothesis distribution of power observed in our laboratory (66, 67). α was controlled at the map-wise level at 0.05 by
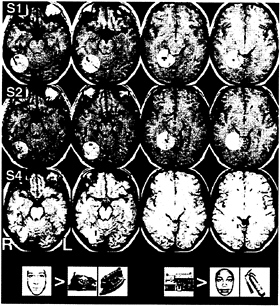
FIG. 5. Putative “face” and “building” areas in three subjects. Shown are 4-mm axial slices in standard space, arranged from inferior to superior. Superimposed in red are those voxels that responded with significantly greater signal to the presentation of faces than to the presentation of buildings or objects. In blue are those areas with a greater response to building than to faces or objects. The circles of lighter contrast are intended to illustrate the anatomical correspondence of the suprathreshold voxels across subjects.
Bonferroni correction for the number of voxels examined for each contrast.
For four of five subjects, voxels within ventral occipitotemporal cortex were identified that had a significantly greater response to faces than to buildings or general objects. These areas were similar in location to those reported in a recent, comprehensive study of the “fusiform face area” (19). Additionally, for these four subjects, voxels were identified that responded with greater signal to buildings than to faces or objects. These areas were located medial and superior to the “face” voxels. Fig. 5 presents the results from three of these subjects.
These findings are consistent with the existence of an area that is specialized for the perception and representation of buildings. We are currently conducting additional studies with other classes of stimuli to further test the specificity of this putative “building” area. A future area of investigation might be to determine whether the responses of this region generalize to other classes of landmarks.
DISCUSSION
We have reviewed here some experimental forays into the neural basis of human representation of topographical space. The interpretation of this work has drawn on both neuroimaging studies of intact subjects as well as consideration of the deficits that follow specific brain damage. We suggest that both of these types of studies are necessary to provide an inferentially sound basis for the conclusions drawn. Because these issues are infrequently considered in the interpretation of neuroimaging data, we close with a brief discussion of the importance of combining lesion and neuroimaging studies.
The Utility of “Converging” Evidence. In the experiments and reviews described in this article, we frequently attempted to compare and contrast the results of neuroimaging experiments with the findings of studies of brain-damaged patients. We propose that, because of inferential limitations of lesion and neuroimaging studies in isolation, it is preferable to obtain compatible evidence from both types of studies. This convergence is suggested to be necessary to establish that a neuroanatomical region performs some computation necessary for a given cognitive process. Note that this brief commentary does not concern itself with the challenges and pitfalls of ascribing a specific computation to a neuroanatomical region (68, 69).
Unlike the lesions produced in animal models, which can be precisely created with neurotoxins, lesions in human patients are often wide ranging and damaging not only to local neurons but also to “fibers of passage” (21). It is also possible that connections from region “A” support the continued metabolic function of region “B” but that region “A” is not computationally involved in certain processes undertaken by region “B”. Mechanisms that might be imagined to produce such an effect include diaschisis (70) or retrograde transsynaptic degeneration (71). As a result, lesions of the kind often studied in humans cannot conclusively demonstrate that the neurons within a specific area are themselves critical to the computational support of an impaired cognitive process.
Neuroimaging studies, on the other hand, are able to demonstrate computational involvement (i.e., differential neural activity) of a specific neuroanatomical region. However, the technique is unable to prove that the observed activity is necessary for a putatively isolated cognitive process. This is because one never has perfect control over the cognitive processes in which a subject engages. Although an experiment may control the conditions or tasks to which a subject is exposed, it cannot demonstrate conclusively that a
subject is engaging differentially a single, identified cognitive process. It should be noted that “a more sensitive behavioral test” cannot solve this problem because it is always possible that the subject engages unnecessary cognitive processes that either have no overt, measurable effects or are perfectly confounded with the process of interest. As a result, observed neural activity may be the result of some confounding neural computation that is not itself necessary for the execution of the cognitive process ostensibly under study. An equivalent formulation of these statements is to note that, essentially, neuroimaging is an observational, correlative method (72).
When combined, however, a stronger level of inference results from lesion and neuroimaging studies. One type of combination might be that (i) lesions to a cortical area impair a given cognitive process and that (ii) the cognitive process, when engaged by intact subjects, evokes neural activity in the same cortical area. The inference that the neuroanatomical area is computationally necessary for the cognitive process is now rendered less vulnerable to the faults detailed above for each method in isolation although it is still possible to conceive of failures. As a result, neuroimaging and lesion studies are complementary, in that each provides inferential support that the other lacks.
This work was supported by grants from the National Institute of Health (NS01762 and AG13483) and the McDonnell-Pew Program in Cognitive Neuroscience.
1. O’Keefe, J. & Nadel, L. (1978) The Hippocampus as a Cognitive Map (Oxford Univ. Press, Oxford).
2. Brewer, B. & Pears, J. (1993) in Spatial Representation, eds. Eilan, N., McCarthy, R. & Brewer, B. (Blackwell, Oxford), pp. 25–30.
3. Heft, H. (1979) Environ. Psychol. Nonverbal Behav. 3, 172–185.
4. Thorndyke, P.W. & Hayes, R.B. (1982) Cognit. Psychol. 14, 560–589.
5. Magliano, J.P., Cohen, R., Allen, G.L. & Rodrigue, J.R. (1995) J. Environ. Psychol. 15, 65–75.
6. O’Keefe, J. & Dostrovsky, J. (1971) Brain Res. 34, 171–175.
7. Morris, R.G., Garrud, P., Rawlins, J.N. & O’Keefe, J. (1982) Nature (London) 297, 681–683.
8. Habib, M. & Sirigu, A. (1987) Cortex 23, 73–85.
9. Maguire, E.A., Burke, T., Phillips, J. & Staunton, H. (1996) Neuropsychologia 34, 993–1001.
10. Bohbot, V., Kalina, M., Stepankova, K., Spackova, N., Petrides, M. & Nadel, L. Neuropsychologia, in press.
11. Aguirre, G.K., Detre, J.A., Alsop, D.C. & D’Esposito, M. (1996) Cereb. Cortex 6, 823–829.
12. Maguire, E.A., Frackowiak, R.S.J. & Frith, C.D. (1996) Proc. R. Soc. London 263, 1745–1750.
13. Maguire, E.A., Burgess, N., Donnett, J.G., O’Keefe, J. & Frith, C.D. (1998) J. Cognit. Neurosci., in press.
14. Siegel, A.W. & White, S.H. (1975) in Advances in Child Development and Behavior, eds. Reese, H.W. (Academic, New York), pp. 9–55.
15. Thorndyke, P. (1981) in Cognition, Social Behavior, and the Environment, ed. Harvey, J. (Erlbaum, Hillsdale, NJ).
16. Aguirre, G.K. & D’Esposito, M. (1997) J. Neurosci. 17, 2512– 2518.
17. Farah, M.J. (1990) Visual Agnosia (MIT Press, Cambridge, MA).
18. Desimone, R. (1991) J. Cognit. Neurosci. 3, 1–8.
19. Kanwisher, N., McDermott, J. & Chun, M.M. (1997) J. Neurosci. 17, 4302–4311.
20. Cohen, N.J. & Eichenbaum, H. (1993) Memory, Amnesia and the Hippocampal System (MIT Press, Cambridge, MA).
21. Jarrard, L.E. (1993) Behav. Neural Biol. 60, 9–26.
22. Morris, R.G.M., Schenk, F., Tweedie, F. & Jarred, L. (1990) Eur. J. Neurosci. 2, 1016–1028.
23. Smith, M.L. & Milner, B. (1981) Neuropsychologia 19, 781–793.
24. Cave, C.B. & Squire, L.R. (1991) Hippocampus 1, 329–340.
25. DeRenzi, E. (1982) Disorders of Space Exploration and Cognition (Wiley, New York).
26. Zola-Morgan, S., Squire, L.R. & Amaral, D.G. (1986) J. Neurosci. 6, 2950–2967.
27. Rempel-Clower, N.L., Zola, S.M., Squire, L.R. & Amaral, D.G. (1996) J. Neurosci. 16, 5233–5255.
28. Scoville, W.B. & Milner, B. (1957) J. Neurol. Neurosurg. Psychiatry 20, 11–21.
29. Milner, B., Corkin, S. & Teuber, H.-L. (1968) Neuropsychologia 6, 215–234.
30. Squire, L.R. & Zola, S.M. (1996) Proc. Natl. Acad. Sci. USA 93, 13515–13522.
31. Pick, H.L. (1993) in Spatial Representation, eds. Eilan, N., McCarthy, R. & Brewer, B. (Blackwell, Oxford), pp. 31–42.
32. Regian, J.W. & Shebilske, W.L. (1992) J. Communication 42, 136–149.
33. Friston, K.J., Price, C.J., Fletcher, P., Moore, C., Frackowiak, R.S.J. & Dolan, R.J. (1996) NeuroImage 4, 97–104.
34. Suzuki, W.A. & Amaral, D.G. (1994) J. Comp. Neurol. 350, 497–533.
35. McNaughton, B.L., Leonard, B. & Chen, L. (1989) Psychobiology 17, 230–235.
36. Owen, A.M., Milner, B., Petrides, M. & Evans, A.C. (1996) J. Cognit. Neurosci. 8, 588–602.
37. Zola-Morgan, S., Squire, L.R., Amaral, D.G. & Suzuki, W.A. (1989) J. Neurosci. 9, 4355–4370.
38. Suzuki, W.A., Zola, M.S., Squire, L.R. & Amaral, D.G. (1993) J. Neurosci. 13, 2430–2451.
39. Anderson, R.A., Snyder, L.H., Li, C.-S. & Stricanne, B. (1993) Curr. Opin. Neurobiol. 3, 171–176.
40. Whiteley, A.M. & Warrington, E.K. (1978) J. Neurol. Neurosurg. Psychiatry 41, 575–578.
41. Levine, D., Warach, J. & Farah, M. (1985) Neurology 35, 1010– 1018.
42. Hanley, J.R. & Davies, A.D. (1995) in Broken Memories, eds. Campbell, R. & Conway, M.A. (Blackwell, Oxford), pp. 195–208.
43. Mishkin, M., Ungerleider, L.G. & Macko, K.A. (1983) Trends Neurosci. 6, 414–417.
44. Ungerleider, L.G. & Haxby, J.V. (1994) Curr. Opin. Neurobiol. 4, 157–165.
45. Moscovitch, C., Kapur, S., Kohler, S. & Houle, S. (1995) Proc. Natl. Acad. Sci. USA 92, 3721–3725.
46. McCarthy, R. (1993) in Spatial Representation, eds. Eilan, N., McCarthy, R. & Brewer, B. (Blackwell, Oxford), pp. 373–399.
47. Farrell, M.J. (1996) Neurocase 2, 509–520.
48. Hécaen, H., Tzortzis, C. & Rondot, P. (1980) Cortex 16, 525–542.
49. Rocchetta, A.I., Cipolotti, L. & Warrington, E.K. (1996) Cortex 32, 727–735.
50. Landis, T., Cummings, J.L., Benson, D.F. & Palmer, E.P. (1986) Arch. Neurol. 43, 132–136.
51. Takahashi, N., Kawamura, M., Hirayama, K. & Tagawa, K. (1989) No to Shinkei Brain Nerve 41, 703–710.
52. McCarthy, R.A., Evans, J.J. & Hodges, J.R. (1996) J. Neurol. Neurosurg. Psychiatry 60, 318–325.
53. Pallis, C.A. (1955) J. Neurol. Neurosurg. Psychiatry 18, 218–224.
54. Tohgi, H., Watanabe, K., Takahashi, H., Yonezawa, H., Hatano, K. & Sasaki, T. (1994) J. Neurol. 241, 470–474.
55. Holmes, G. & Horrax, G. (1919) Arch. Neurol. Psychiatr. 1, 385–407.
56. Kase, C.S., Troncoso, J.F., Court, J.E., Tapia, J.F. & Mohr, J.P. (1977) J. Neurol. Sci. 34, 267–278.
57. Stark, M., Coslett, B. & Saffran, E.M. (1996) Cognit. Neuropsychol. 13, 481–523.
58. McNaughton, B.L., Mizumori, S.J. & Barnes, C. (1994) Cereb. Cortex 4, 27–39.
59. Cammalleri, R., Gangitano, M., D’Amelio, M., Raieli, V., Raimondo, D. & Camarda, R. (1996) Neuropsychologia 34, 321–326.
60. Takahashi, N., Kawamura, M., Shiota, J., Kasahata, N. & Hirayama, K. (1997) Neurology 49, 464–469.
61. Chen, L.L., Lin, L.H., Green, E.J., Barnes, C.A. & McNaughton, B.L. (1994) Exp. Brain Res. 101, 8–23.
62. Taube, J.S., Goodridge, J.P., Golob, E.J., Dudchenko, P.A. & Stackman, R.W. (1996) Brain Res. Bull. 40, 477–486.
63. Allison, T., Ginter, H., McCarthy, G., Nobre, A.C., Puce, A., Luby, M. & Spencer, D.D. (1994) J. Neurophysiol. 71, 821–825.
64. Haxby, J.V., Horwitz, B., Ungerleider, L.G., Maisog, J.M., Pietrini, P. & Grady, C.L. (1994) J. Neurosci. 14, 6336–6353.
65. Worsley, K.J. & Friston, K.J. (1995) NeuroImage 2, 173–182.
66. Zarahn, E., Aguirre, G.K. & D’Esposito, M. (1997) NeuroImage 5, 179–197.
67. Aguirre, G.K., Zarah, E. & D’Esposito, M. (1997) NeuroImage 2, 199–212.
68. Damasio, H. & Damasio, A. (1989) Lesion Analysis in Neuropsychology (Oxford Univ. Press, New York).
69. Farah, M.J. (1994) Behav. Brain Sci. 17, 43–103.
70. Feeney, D.M. & Baron, J.-C. (1986) Stroke 17, 817–830.
71. Jacobson, M. (1978) Developmental Neurobiology (Plenum, New York), pp. 256–258.
72. Sarter, M., Berntson, G. & Cacioppo, J. (1996) Am. Psychol. 51, 13–21.








