4
Results of an End State Based Analysis of Technology Development Needs for the Hanford Tanks
A limited application of the end state approach, exemplified by the three scenarios for Hanford tanks remediation described in Chapter 3, can bring into focus the technology development needs to meet functional requirements. It is not the intent of this report to discuss the technology needs of each processing function shown or implied in the scenarios. Rather, four important functions have been selected, and these will be discussed in some detail to illustrate the type of review and analysis for evaluating technology development needs to support deployment of remediation systems. This illustrates the final steps of the end state approach shown in Figure 3.
The Hanford tanks end state remains to be negotiated with the state, its citizens, tribal nations, and the U.S. Environmental Protection Agency (USEPA). At present, the Tank Waste Remediation System (TWRS) has adopted some target end states to provide a focus for work. The committee was unable to determine whether those end states had been formally adopted by the Department of Energy (DOE). Although a recent Hanford report (Acree, 1998) discusses end states, the committee found little indication that the Office of Environmental Management (EM) program plans to explicitly address end state issues in the near term (i.e., before fiscal year 2000), even though some of the issues (e.g., chemically active barriers to migration of radioactive species) will require innovative technologies for their solution. Comprehensive performance and cost assessments would identify the nature and priority of technology development needs, or, in the case of the DOE Environmental Management Science Program (EMSP), the need for more basic research.
The committee found the lack of well defined end states a potential EM remediation program deficiency that increases the risk of failure to meet schedules, budgets, and technical requirements. Hence, end state definitions and associated technology development needs are not completely defined and integrated in the Office of Science and Technology (OST) programs. As noted above, there are tentative criteria for some of the end states for the Hanford remediation system. However, to thoroughly identify waste remediation process technology needs, the end states must be completely defined and pertinent attributes of the end states must be described in terms that can be related to process steps.
The committee recognizes that its reference scenario, while very similar to the Hanford baseline (U.S. Department of Energy, 1997b), may not be adequate to uncover all technology development needs for remediation of all the Hanford tanks. Nevertheless, the approach described herein identifies gaps and needs for improvements in planned remediation processes, as well as technology needs to address and alleviate those deficiencies. Also, it is likely to
uncover deficiencies and needs in the Hanford baseline scenario, even when remediation activities are well under way. The committee believes that the analysis presented below for the four processing functions selected for review demonstrates the process that DOE should use to identify waste remediation technology needs.
By comparing scenarios such as the three presented in this report, it is possible to identify common functions for the scenarios, as well as those that are relevant to only one or two of the scenarios. In this way, a comprehensive set of technology gaps and improvement needs can be readily identified without duplication of effort, and once the results of performance and cost assessments are in hand, a judgment can be made about the relative priorities of the technology needs.
Various technologies required for tank waste remediation have been developed by TWRS program elements, DOE national laboratories, industry, and university laboratories. This development work was carried out over a period of many years, and some of the work was done before the tank contents were adequately characterized and the complexity of the remediation task fully appreciated. Therefore, the evaluation of technology deficiencies and needs should include a review of previously developed technologies to determine if they are still relevant and adequate.
Technology needs refers to the actions required to obtain specified information or to carry out specified functions. As used in this report, it does not refer to the acquisition of data or information through the use of well-defined or established practices or techniques. Technology needs arise when existing processes are inadequate for one or more reasons, such as safety and health risk, inability to perform the functions required to meet end state criteria, economics, and limitations on applicability or scale-up to the required levels.
Technology Development Program for Hanford Tank Wastes
The Site Technology Coordinating Group (STCG) for Hanford, which includes TWRS program representatives, identifies technology development needs for tank remediation. The EM technology development programs that directly address or have some potential relevance to address these needs consist of three major components, listed in order of decreasing funding: (1) a significant portion of the Tank Focus Area portfolio (Pacific Northwest National Laboratory, 1997a), (2) the Hanford Tank Initiative (HTI) (Root, 1997), and (3) a number of miscellaneous technology development and technical support activities funded directly by Hanford TWRS (D. Wodrich, DOE Richland, personal communication, 1997). Needs being addressed by these three components are summarized as follows:
- Storage—monitoring tank integrity, in situ waste characterization, reduction of waste streams designated for tank storage.
- Retrieval—mobilization and retrieval of bulk waste, retrieval of heels, saltcake dissolution, blending of wastes, transfer of slurries in pipelines, leak detection and mitigation, establishing the technical basis for retrieval performance criteria.
- Processing—radionuclide removal, solids washing, enhanced sludge washing, behavior of wash solutions, solid-liquid separation, maintaining water balances.
- Immobilization—testing of low-activity waste (LAW) waste forms performance in support of risk assessment and development of waste acceptance criteria, optimization of high-level waste (HLW) glass formulations.
- Closure—measuring the quantity of radioactive waste residue in a tank, sampling the plume of contaminants in soil, establishing the technical basis for closure criteria.
The Hanford privatization contractors are also presumably performing technology development activities, especially in the areas related to radionuclide separations and immobilization.
Technology Assessment for Selected Functions of the Scenarios
The three example Hanford waste tank remediation scenarios presented in Figure 4 in Chapter 3 were used to select four functions to illustrate the definition of requirements and technology development needs. The four functional process steps selected for further analysis are (see bold outlined boxes in Figure 5) (1) enhanced sludge washing, (2) offgas processing, (3) stabilization of tanks and unretrieved contents, and (4) enhanced barriers for tanks from which the contents have not been removed. They were selected partly for their process importance and partly to provide an example of technology development needs not currently in DOE's program.
Enhanced Sludge Washing
Some of the chemicals that can have a negative effect on costs or processing operations, especially on vitrification, will certainly not be removed when tank sludges are washed with water or dilute caustic solution. Therefore, an enhanced washing step is included in Hanford's baseline and the committee's reference scenario. Compounds of particular interest are those containing chromium, phosphate, aluminum, and sodium, because they will either remain in large quantities in washed sludges or have a disproportionately large impact on HLW volume (Beahm et al., 1997). The diverse nature of the tank solids suggests that no single enhanced sludge washing procedure will be effective for all of the tank waste.
Enhanced sludge washing is described in Chapter 3 and shown in Figure 5. The general functional requirement of enhanced sludge washing is to reduce the total volume of the vitrified HLW product by solubilizing non-radioactive chemicals that are not soluble in near-neutral water and routing the remaining insoluble solids, which contain most of the radionuclides, to the HLW vitrifier. A product of the enhanced washing step is a solution containing primarily non-radioactive, amphoteric, bulk chemicals. This solution is routed to a cesium removal step. The technology development needs relate to removing potentially deleterious and voluminous bulk chemicals from the waste solids going to the HLW vitrifier while retaining important radionuclides, such as transuranic elements and strontium, in the HLW stream.
Specific functional requirements for the removal of key species (i.e., aluminum, chromium, sodium, phosphates) to achieve a HLW end state volume target of about 15,000 m3 (4 million gallons) are given by Boston (1997) and are used for this example. There is also a requirement that transuranic elements not be solubilized to an extent that would result in the immobilized LAW being a transuranic waste (i.e., greater than 100 nCi/g of long-lived alpha radioactivity). More desirably, the waste should meet the U.S. Nuclear Regulatory Commission's criteria for Class A waste of ¬ 10 nCi/g. Based on values given in the TWRS Environmental
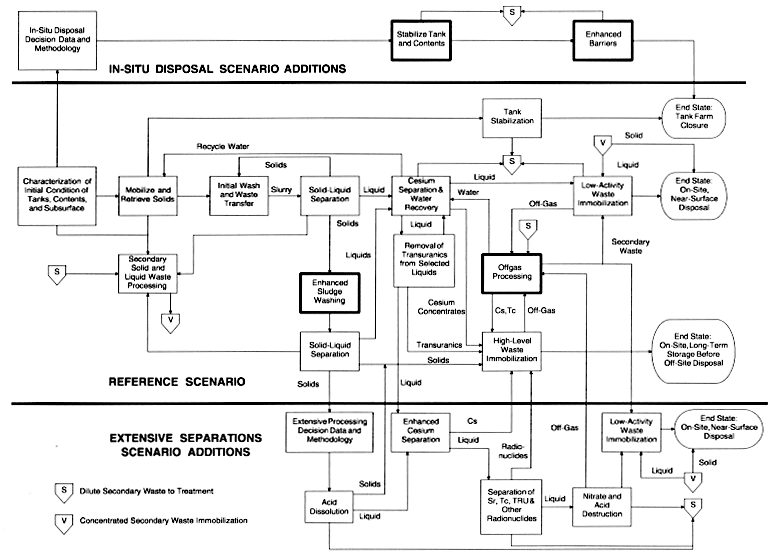
Figure 5.
Committee's Reference and Plausible Bounding Scenarios and Functional Flowsheets for Hanford Site Tanks and Associated Wastes (same as Figure 4), with Functions Selected for Discussion in This Report Shown in Bold Outlined Boxes
Impact Statement (EIS) (U.S. Department of Energy and Washington State Department of Ecology, 1996), the average concentration of transuranics in the immobilized LAW if the entire inventory is solubilized is about 144 nCi/g. Thus, on average, in order to meet a limit of 10 nCi/g in the immobilized LAW, at most 10/144 or about 7 percent of the important transuranic elements (americium and plutonium) could be solubilized.
The ultimate determinant of permissible technetium content of the LAW is the dose from technetium to the critical group in the vicinity of the Hanford Site. The actual dose has yet to be determined by confirmed modeling studies for technetium release from LAW and transport to the geographical point where the dose must meet the standard, especially that for ground water contamination. In the absence of a regulatory dose standard and an accepted modeling study for technetium release, prudence dictates that methods for technetium removal from the LAW stream be developed to ensure a capability for meeting eventual regulatory requirements. Technology needs relate to removing technetium to levels that meet these requirements.
Use of caustic solutions to leach sludges can lead to formation of colloids, gels, and precipitates that can plug pipelines or interfere with subsequent processes. Even though there appears to be little evidence to date of problems from colloid, gel, and precipitate formation caused by enhanced sludge washing, the potential for such formation remains a concern. Colloidal forms of the actinides such as plutonium and americium could remain with the LAW stream, potentially changing the waste classification of some wastes. Gels and precipitates from combinations of the many chemicals in the wastes could easily form, especially as the wastes cool in pipelines. As a result, a qualitative functional requirement is to sufficiently understand this operation so that these undesirable species are not formed in unacceptable amounts.
Enhanced sludge washing has received a great deal of attention at Hanford because of its cost benefits and the avoidance of process complications (McConville, Johnson, and Derby, 1995; Orme, 1995). Study of chromium chemistry and caustic recovery was identified as being important by the Hanford STCG (Pacific Northwest National Laboratory, 1997a, b). Chemical solubility data in the highly caustic wash solutions expected to be used for enhanced sludge washing are available on some existing Hanford tank waste (Peretrukhin, Shilov, and Pikaev, 1995; Peretrukhin et al., 1996), and on some compounds that are expected to be formed during processing. There appear to be little data available on the rates at which compounds will be formed or dissolved, although there is some information on the kinetics of valence changes and their effects on solubility (Shilov et al., 1996). This may cause some unsuspected effects, such as the formation of insoluble compounds that may cause problems in transfer lines. In addition, there is evidence that some of these processes will be quite slow, occurring over days or weeks (Beahm, et al., 1998). Slow reaction rates could have a large effect on flowsheet design, process efficiency, plant throughput, and production scheduling. Also, there appear to be few systematic and relevant studies on the effects of temperature on formation and solubility of compounds such as aluminum hydroxide and various oxides of aluminum, phosphates and fluorophosphates, and carbonates. All of these could cause problems with formulation and consistent production of an acceptably homogeneous glass, and with the feasibility of achieving the desired volume reductions, both of which are important aspects of the HLW end state. It is important to continue the work on adverse consequences of precipitation reactions to the waste treatment process.
Changing valence states of the ions of concern to improve their removal by enhanced sludge washing is another important issue. For example, it is possible to dramatically change chromium chemistry (and enhance its removal) through valence state adjustments using oxidizing chemicals such as permanganate or ozone. This process is being studied for tanks in which the chromium has proven difficult to remove by enhanced sludge washing (Rapko, Delegard, and Wagner, 1997; Lumetta and Rapko, in press). Other elements in the tank wastes
(e.g., plutonium and neptunium) are also subject to valence changes under redox conditions similar to those being considered for chromium valence adjustment. However, recent evidence (Rapko, 1998) indicates that while the highest potential concentration of transuranics in LAW glass observed in laboratory scale experiments is about 10 nCi/g, more typical concentrations would be on the order of 1 nCi/g, both of which are probably low enough to be acceptable.
In light of the diverse chemistry of the wastes and the need to mix wastes from various tanks to partially homogenize the feed to the vitrifier, the sludge washing chemistry and accompanying physical and chemical changes must be predictable. Enhanced sludge washing is identified by the DOE Tank Focus Area (TFA) (Pacific Northwest National Laboratory, 1997b) to be at the engineering development stage, and only parametric tests are planned. Table 6 summarizes the OST technology development projects needed for this process step. Enhanced sludge washing has also been studied by TWRS, and presumably by the Hanford tank privatization contractors.
As shown in the table, OST is or has been pursuing several projects related to enhanced sludge washing. The committee was unable to determine from information received from OST whether enhanced sludge washing needs are being identified and pursued systematically at a level required for basic understanding, and whether the projects selected will contribute to meeting the processing function's requirements.
Vitrifier Offgas Processing
Offgas processing will be required at various process steps throughout the Hanford baseline scenario, and also in the committee's reference scenario shown in Figure 5. Offgases from the HLW and LAW vitrifiers present formidable cleanup problems because of the high temperatures and the resultant decomposition and vaporization of a variety of chemicals (International Atomic Energy Agency, 1988). These chemicals include radionuclides (such as cesium and technetium) in the form of hydroxides or oxides, as well as other volatile metallic compounds, oxides of nitrogen, and organic compounds. In addition to these chemicals, there will also be large quantities of vaporized water and entrained materials to be treated at many points in the process. Ruthenium, which contributes little to the total radioactivity in tank wastes but is present in appreciable quantities, will likely appear in the offgas stream and must also be managed.
The qualitative functional requirement for offgas processing is a stream that is acceptable for release to the atmosphere, plus internal streams (usually aqueous) suitable either for the secondary waste processing system or, if suitable, for recycling to the vitrifier. The offgas streams contain the chemical compounds of ruthenium, sodium, and boron, which could clog the offgas processing system. In particular, the functional requirements for offgas processing are not driven by a scenario end state criteria, but rather they are largely defined by acceptance requirements for other functions in the flowsheet. Qualitative functional requirements are the following:
- removal of a variety of semivolatile compounds of radionuclides such as cesium, iodine, and technetium,
- destruction or removal of nitrogen oxides resulting from decomposition of nitrate and nitrite salts in the vitrifier, and
- removal of sulfur oxides resulting from decomposition of sulfates in the vitrifier.
Table 6 Department of Energy Office of Science and Technology Projects on Enhanced Sludge Washing
|
Technology Needed |
Technical Task Plan |
Title |
|
Removal of troublesome chemicals |
AL16WT41-A |
Parametric studies of Hanford sludge |
|
|
OR16WT41-C |
Prevention of solids formation |
|
Solubility data for selected compounds |
RL08WT41-A |
Saltcake dissolution |
|
|
OR16WT41-B |
Saltcake dissolution |
|
Kinetic data for compound formation |
OR16WT3 1-C |
Control of leachate solids formation |
|
Colloid, gel, and precipitate formation |
|
|
|
Temperature coefficient of solubility for hydroxides of aluminum, iron, and zirconium, phosphates and fluorophosphates |
|
No specific projects identified |
|
SOURCE: Pacific Northwest National Laboratory (1997b). |
||
In addition, problems may be caused by the presence of mercury and similar chemicals. The technology development needs relate to retaining radioactive and toxic wastes in the vitrified product and preventing deleterious buildups of solids and radioactivity in the offgas system.
Recovery and recycling of cesium and technetium will be required because the volatilities of their oxides are such that it may be difficult to retain them completely in the glass melt. This may result in a relatively large inventory of these elements in the offgas stream, either in gaseous form or as aerosols formed in the gas stream leaving the vitrifier. Buildup of radioactivity from cesium could lead to high radiation levels in parts of the offgas system. Contamination by technetium is a concern because of the volatility of its oxides (Vida, 1994) and its very long half-life. This contamination could become, for example, an important problem during equipment decontamination for repair or maintenance. Since the French vitrification experience suggests that the volatility of both technetium and ruthenium can be reduced during vitrification of HLW (Jouan, Moncouyoux, and Halaszovich, 1985), a similar reduction in the volatilities of technetium and ruthenium compounds could occur during vitrification of Hanford waste, although this needs to be validated for the vitrifier and unique waste compositions at Hanford. The committee notes that the Hanford vitrifier is not likely to operate under the more desirable reducing conditions that would inhibit chemical volatility because of the presence of nitrates, air, and other oxidizing substances.
Entrained particles or aerosols of compounds of cesium, technetium, and ruthenium, as well as volatilized compounds, can adhere to pipes and eventually obstruct gas flow (Yonega et al., 1985). This is also a problem in the LAW vitrifier, where volatilization of boric acid and alkali borates is likely to occur. Deposited particles can also lead to maintenance problems. The extent of the volatilization problems depends to a large extent on the design and operating conditions of the vitrifier (Wilson, 1996; Whyatt et al., 1996).
In general, condensers and scrubbers are used to remove vaporized and entrained materials from offgas streams (International Atomic Energy Agency, 1988, p. 31-39). Catalytic destruction of nitrogen oxides is also an option for the LAW vitrifier offgas, but catalyst poisoning may be a problem and must be addressed by technology development. The water produced in the reaction will also need to be treated to remove contaminants.
Chloride and fluoride, which can be present in the vitrifier offgas from some of the tank wastes, can cause corrosion problems, as can oxides of sulfur in the presence of water. Sulfur
oxides can be removed by sorption on reactive sorber beds. More generally, corrosive components of the offgas stream will need to be considered either in equipment design or removed and stabilized in the vitrified wastes.
Processing of secondary wastes from the offgas system will be required. Some work on this is being supported at the Savannah River Site with its small experimental vitrifiers (Pacific Northwest National Laboratory, 1997b), but its relevance to the Hanford wastes for the privatization work is unknown to the committee. Tests of the LAW vitrifier with attention to offgas cleanup to meet regulatory and internal recycling criteria will need to be completed to verify performance.
The application of large vitrifiers that function reliably in a radioactive environment will require offgas treatment systems capable of performing their required functions over a sustained period with a variety of feed compositions. A generalized flowsheet for the functions of various components of an offgas system has been issued (U.S. Department of Energy and Washington State Department of Ecology, 1996, p. B-96) based largely on conventional hot-gas quenching by aqueous spray treatments and with a focus on the bulk of the offgas chemicals (e.g., nitrogen oxides, sulfur dioxide).
The committee found no Hanford-designated technology needs identified by the STCG for this function, nor are there relevant projects on these offgas topics evident in the TFA program or elsewhere in OST. The privatization contractors responsible for the vitrification may complete technology development necessary to deploy a reliable offgas system, but information on privatization contractor activities in this regard were not available to the committee. However, the committee found no documentation that specifies or ensures any role for DOE in this matter. Treatment of some offgas from highly radioactive operations is a mature engineering practice, and it is only in the cases of unusual materials in the offgas or of extraordinary throughputs where special attention may be needed. Both unusual materials and exceptional throughputs may be expected from the Hanford vitrifiers. The Savannah River Site vitrifier has an offgas system that required special attention because of (1) its unique design, (2) the unusual components of the feed that could reach the offgas system and require management, (3) issues of scaling up from small experiments, and (4) unusual (high or low) throughput. Accordingly, there is reason to believe that special attention will also be needed at Hanford.
Stabilize Tanks Containing Unretrieved Waste
In the in situ scenario, tanks and their contents are presumed to be disposed of in place. Stabilizing tanks containing waste is a necessary function in the committee's in situ disposal scenario. Stabilizing the entire contents of tanks from which all pumpable liquid has been removed is an alternative considered in the recent Hanford TRWS EIS (U.S. Department of Energy and Washington State Department of Ecology, 1996), but it was not adopted in the Record of Decision (U.S. Department of Energy, 1997b).
This function will require filling the tanks containing a mixture of soft and hard materials with stabilizing agents. Unless changes to the waste are made, the stabilization agents must be compatible with highly alkaline waste, retain radionuclides, prevent tank collapse, and allow stabilized tanks to meet applicable performance criteria. Specific functional requirements have not yet been developed or postulated. In principle, the end state criterion (such as dose limits for the immobilized LAW in near-surface disposal as defined in the committee's reference scenario in Chapter 3 and set by the U.S. Nuclear Regulatory Commission and DOE Orders) should suffice for planning purposes. Translating dose limits into quantitative requirements (e.g.,
maximum radionuclide release rates for the stabilization agents, required degree of homogenization) requires a detailed performance assessment. Therefore, the discussion of technology needs and status here is based on generic functional requirements.
Technology development needs relate to the retention of waste components in the tanks. An additional need is the development of stabilization agents that have the ability to resist degradation by ground water while immobilizing radionuclides. Immobilization of such radionuclides as technetium may be particularly challenging. Achieving this may require that the stabilization agent be impermeable and insoluble, as well as capable of maintaining an environment that reduces the mobility of important radionuclides. Some technologies, such as reducing grouts used in nearly empty tanks at the Savannah River Site, have been developed and might meet this need, but more development and demonstration would be needed for the Hanford application.
Stabilizing agents such as grout must also adequately mix with substantial amounts of waste in the tanks. This involves mixing large amounts of material, some of which may not be readily mobile, and working through relatively small tank penetrations without further degrading the integrity of the tank. Technology development to meet this need is not now underway nor planned by OST. Technology is also needed to characterize the stabilized tank to assure that the functional requirements have been met without adversely impacting the stabilization function. Technology development to meet this need is neither underway nor planned.
In summary, the technology required to implement in situ stabilization does not appear to exist in OST, and there is essentially no ongoing technology development in this arena. This finding is not unexpected because DOE and local public stakeholders do not currently support consideration of the in situ disposal scenario.
Enhanced Barriers for Unretrieved Tanks and Waste
The provision of enhanced barriers for unretrieved tanks containing stabilized waste is a necessary function of the committee's in situ disposal scenario. Use of barriers on stabilized unretrieved tanks was considered in the in situ alternative in the recent Hanford TRWS EIS (U.S. Department of Energy and Washington State Department of Ecology, 1996), although the exact nature of the barriers was not described.
This function will require placing physical barriers around a stabilized tank to prevent intrusion of water, humans, and biota, as well as reactive barriers to inhibit migration of radioactive and toxic constituents. Because of the greater inventory of radioactive toxic materials in the in situ disposal scenario than in the nearly empty tanks of the committee's reference scenario, we expect the functional requirements for the enhanced barrier system to be much more stringent than for the barrier system in the committee's reference scenario. As with stabilization of unretrieved tanks, translating this into specific quantitative functional requirements (e.g., maximum allowable water ingress and radionuclide release rates as a function of time) requires a detailed performance assessment. Therefore, the discussion of the technology needs and status here is based on generic functional requirements. The utility of barriers, both on and beneath the surface of the ground, in diminishing the effects of tank leakage and retarding subsequent transport of radionuclides has been discussed in a number of publications and reports, including a report of a recent workshop by the National Research Council (1997).
A large amount of design (Myers and Duranceau, 1994), engineering study (Skelly et al., 1996), and experimental work (Gee et al., 1994) has been done on surface barriers to prevent
ingress of water. Follow-up studies on the performance of existing surface barriers are continuing.
A study of the use of subsurface barriers to mitigate leakage from Hanford tanks has been prepared (Treat et al., 1995). However, subsurface barriers are considered by DOE and the Hanford TWRS to provide only marginal reduction with regard to radionuclide release and transport at relatively high cost (J. Honeyman, personal communication, November 12, 1997). Consequently, the Tri-Party Agreement milestones for development of subsurface barriers have been deleted from the tank waste remediation program.
Barriers may be physical, chemical, or a combination of the two. Achieving highly effective chemical barriers depends on finding chemical substances that remain effective over long periods of time to incorporate into the soil or other materials surrounding tanks or tank farms and that will react with the radionuclides of concern to stop or greatly retard their movement (Balsley et al., 1997). Effective and economically acceptable barrier materials could substantially diminish the risk from tank leakage. The OST program for this process step is summarized in Table 7.
Leak mitigation for single-shell tanks was identified as a technology need, but was assigned a low priority. Hanford had identified a technical need for getter materials (especially for technetium and selenium) for LAW storage and disposal. The TFA combined this issue with evaluation and modeling of moisture flow for performance assessment and assigned the combined project a low priority. It is important to note that, even if the needs in Table 7 are funded, they relate to barriers for the tanks that are nearly empty. No programs relevant to enhanced barriers for closed tanks from which waste has not been removed were identified.
The committee believes that enhanced barriers could contribute significantly to retention of important mobile radionuclides on site and to making cost effective alternatives to the Hanford baseline flowsheet feasible. Enhanced barriers have been considered for use in the Hanford baseline, but apparently only research and development on the standard Hanford cap has been funded.
TABLE 7 Department of Energy Office of Science and Technology Projects on Enhanced Barriers
|
Problem Area |
Technical Task Title |
Status |
|
Inhibit further leakage |
Tank leak detection and mitigation |
Low priority and not funded |
|
|
Tank leak mitigation/repair |
Low priority and not funded |
|
Surface barriers |
Several projects completed, especially standard Hanford cap |
No work on enhanced barriers. |
|
Subsurface barriers |
|
No activity or plans |
|
Technetium engineered barriers |
Getter material for technetium and selenium, related to LAW disposal |
Low priority and not funded |
|
SOURCE: Pacific Northwest National Laboratory (1997b). |
||
Summary and Conclusions
Four important functions were selected from the three example scenarios developed in Chapter 3. Realistic but hypothetical functional requirements were then developed for each of the example functions. The current status of technology in each of the four areas was compared to the requirements, yielding technology development needs. The needs were then compared to the present DOE EM technology development program to evaluate the adequacy of the program to support the four selected functions.
Based on this example, the committee finds that the end state based approach to specifying an appropriate technology development program can systematically and efficiently identify technology needs and necessary changes in ongoing technology development efforts. The committee inferred, from the absence of programs on some technologies needed for the baseline remediation system, that DOE's technology development program appears to have been substantially limited by the potential for privatization of some tank remediation functions. This is an appropriate approach in cases where applicable technologies are known, or where similar problems have been successfully addressed by the private sector under competitive circumstances. In the case of the unique Hanford situation, where only one private sector contractor remains, this approach should be reassessed with a view toward providing incentives for private sector efficiency, improved technology for potential use by the private sector, and maintaining DOE expertise for the purpose of being able to better manage the privatization work and to provide a contingency option in case privatization fails.
The committee also notes that technology development to meet the requirements of alternative scenarios is essentially non-existent. Such development is highly desirable to facilitate a more flexible approach to tank remediation that likely could also balance risk and cost.











