7
Nutrition and Immune Responses: What Do We Know?
Ranjit Kumar Chandra1
Introduction
Twenty-five years ago, the question ''What do we know about nutrition and immunity?'' could have been answered in a few minutes and written up on a few pages. The mutually aggravating interaction of malnutrition and infection has been known for centuries, but the concept of impaired immune responses mediating this interaction is relatively recent.
Three separate forces have driven the study of the immune system in nutritional deficiencies (Anonymous, 1987; Scrimshaw et al., 1968): (1) epidemiologic data showing an interaction, usually synergistic but sometimes antagonistic, between malnutrition and infection, (2) emerging new concepts and techniques in immunology, and (3) dramatic human interest cases. In the early 1970s, the results of the first systematic comprehensive studies were published (Chandra, 1972; Smythe et al., 1971). In young children with protein-calorie malnutrition, that is, both marasmus and kwashiorkor, alterations in a number of immune responses were shown. These alterations include
histomorphology of lymphoid tissues, delayed hypersensitivity skin reactions, lymphocyte antibody production, and complement activity. Subsequently, these observations have been extended by experimentation in laboratory animals and in work on human subjects. It is now recognized that nutritional deficiencies are associated with impaired cell-mediated immunity; reduced number of circulating T-lymphocytes, particularly CD4+ helper T-cells and CD3+ CD25+ T-cells that bear the interleukin (IL)-2 receptor; decreased lymphocyte stimulation response to mitogens and antigens; altered production of cytokines; lower secretory IgA antibody response on mucosal surfaces; decreased antibody affinity; and phagocyte dysfunction. Similar alterations in immune responses have been reported with deficiencies of individual nutrients, such as protein, essential fatty acids, vitamin A, vitamin E, pyridoxine, folic acid, zinc, iron, copper, and selenium (Chandra, 1991, 1992a; Meydani and Hayek, 1992).
Today, nutritional immunology forms the basis of semester-long graduate courses, week-long symposia, and expansive monographs. Thus, to provide a complete answer to the question "What do we know about nutrition and immune responses?" is not easy or simple. Instead, a selective review and some recent observations are provided below.
General Concepts
Several general principles and conclusions on nutrition and immunity can be stated:
- Protein-calorie malnutrition and deficiencies of individual nutrients, even subclinical deficits, are associated with impaired immune responses and altered risk of infection.
- Excessive intake of some nutrients may result in reduced immune responses.
- Dose-response curves should form the basis of recommendations for optimal nutrient intake.
- Immune responses are sensitive and functional indices of nutritional status and can aid in assessing prognosis in medical and surgical patients.
- Several factors other than nutrition can modulate immunocompetence.
- Basic knowledge of nutrition and immune interactions can be utilized to formulate nutritional recommendations and interventions that may reduce illness and improve chances of survival.
Immune Responses in Altered Nutritional Status
Nutrient Deficiencies
Several review articles and monographs have analyzed data on nutrition and immunity (Alexander, 1995; Beisel, 1982, 1992; Bendich and Chandra, 1991; Chandra, 1991, 1992b, 1996; Chandra and Newberne, 1977; Chowdhury and Chandra, 1987; Gershwin et al., 1985; Good and Lorenz, 1992; Keusch et al., 1983; Santos, 1995; Suskind, 1977; Watson, 1984). Malnutrition penetrates many host defense mechanisms (Figure 7-1). For example, the proportion and absolute number of T-cells is reduced, particularly CD4+ helper cells (Figure 7-2), and the CD4+/CD8+ (helper/suppressor T-cell) ratio is decreased. There is reduced production of IL-1 and IL-2, interferon-γ, and tumor necrosis factor. Several antigen-nonspecific defenses also are altered, such as the microbicidal activity of phagocytes, levels of hemolytic complement and components C3 and Factor B, production of mucin, ciliary movement, and lysozyme.
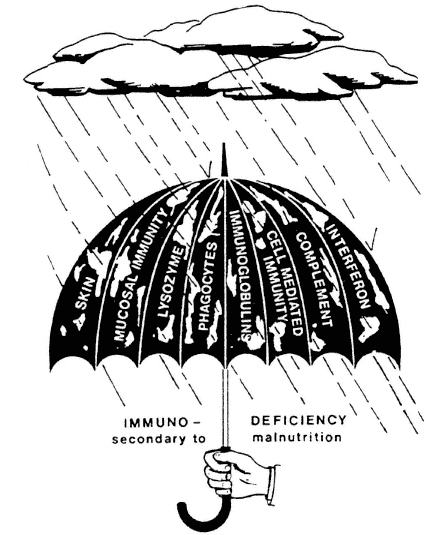
FIGURE 7-1
Immunity in malnutrition. A single view of host defenses as a protective umbrella, consisting of physical barriers (skin, mucous membranes), nonspecific mechanisms (complement, lysozyme, phagocytes), and antigen-specific processes (antibodies, cell-mediated immunity). In protein-energy malnutrition and in deficiencies of various nutrients, many of the host defenses are breached, allowing microbes to invade and produce clinical infection that is more severe and prolonged. SOURCE: Chandra, 1992b. Copyright ARTS Biomedical Publishers, reproduced with permission.
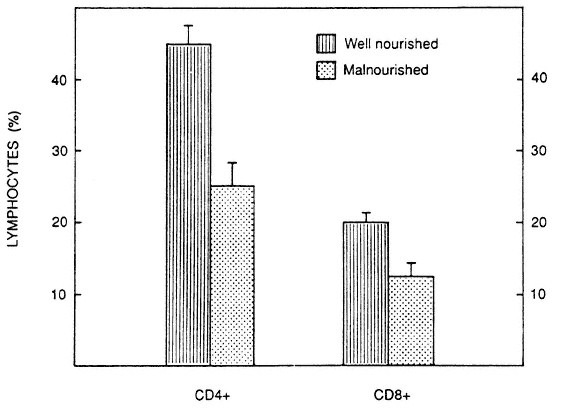
FIGURE 7-2
Proportion of T-lymphocyte subsets in malnourished and well-nourished subjects. There is a marked reduction in CD4+ cells.
Profound changes in immune responses have been observed in micronutrient deficiencies. For example, zinc deficiency results in lymphoid atrophy, impaired delayed cutaneous hypersensitivity, reduced lymphocyte response to mitogens, lower helper/suppressor T-cell ratio, decreased IL-2 production, and most importantly, reduced serum thymulin activity. Copper deficiency reduces the antibody response to T-cell-dependent antigens and impairs phagocyte function (Failla and Bala, 1992). Altered immune responses are readily reversed within weeks or months when nutritional counseling and appropriate supplements are provided; the one exception is the case of intrauterine growth retardation that is associated with prolonged reduction in cell-mediated immunity. In animal models of nutritional deficiency produced before and/or during gestation, the adverse effects on immune responses are observed in the first- and second-generation offspring (Chandra, 1975).
Serum thymulin activity is the prime example of a nutrient-specific immunologic function test that correlates significantly with zinc intake and therefore can serve as a specific functional measure par excellence of zinc deficiency. Other immunologic tests that may be used singly or in combination as indicators of nutrition are listed in Table 7-1. The choice will depend upon the clinical and population setting where the study is done (Sarchielli and Chandra, 1991).
Table 7-1 Immunologic Tests as Indicators of Nutritional Status
|
Lymphocyte number (CD3, CD4) |
|
Terminal deoxynucleotidyl transferase activity |
|
Lymphocyte response to mitogens and antigens |
|
Complement C3 and Factor B concentration |
|
Interleukin-2 production |
|
Delayed cutaneous hypersensitivity |
|
Serum Thymulin activity |
Nutrient Excesses
If reduced intake of an essential nutrient impairs immunity, this does not imply that large amounts will be beneficial. In fact, there may be a negative impact on immune responses and even infectious morbidity as has been shown for zinc and vitamin A (Chandra, 1984, 1991; Semba et al., 1995). Doseresponse curves (Figure 7-3) should be constructed to assess the amount of nutrient intake associated with optimal high immune response.
There is much recent work on lipid modulation of immune responses, and this is reviewed by other authors in this volume (see Kelley, Chapter 14). Briefly, dietary intake of large quantities of fats impairs immune responses. The results of studies are conflicting on the effects of individual dietary fatty acids on various immune responses, such as lymphocyte stimulation in the presence of mitogen. The confounding variables include the amount and degree of unsaturation of fatty acid, age of subjects, species and genetic background of animals, source of mononuclear cells tested, nature and dose of infectious or tumor challenge, and the time and duration of observation (Erickson et al., 1992). Modest amounts of omega-3 fatty acids contained in marine oils reduce inflammatory responses (Endres et al., 1993) and may decrease the severity of autoimmune arthritis, improve survival following endotoxin challenge, and slow the growth of cancer (Fernandes et al., 1992; Venkatraman and Fernandes, 1992). In one study, feeding of flax seed oil rich in α-linolenic acid (18:3n-3) to healthy young adults suppressed the lymphocyte proliferation response to phytohemagglutinin (PHA) and concanavalin A and decreased the delayed hypersensitivity response to seven recall antigens. However, serum concentrations of immunoglobulins, complement C3, C4, salivary IgA, the number of T- and B-cells, and their subsets were not affected (Kelley et al., 1991).

FIGURE 7-3
Lymphocyte stimulation response to optimal dose of phytohemagglutinin (PHA) in adult men with a range of zinc intakes from dietary and supplemental sources. Results are shown as stimulation index calculated as counts per minute in PHA-stimulated culture divided by counts per minute in unstimulated cultures. Bars represent mean ± standard deviation. There were 6 to 10 subjects in each group.
Other Factors Influencing Immune Responses
The evaluation of the effects of nutrition on immunity demands an awareness of other confounding variables that also influence immune responses. The prominent factors are listed in Table 7-2. Physical and thermal trauma alters a variety of immune responses; most are decreased, and this change may be beneficial to the host since clones of immunocompetent cells that react against the host are suppressed transiently. Both extreme cold and hot environmental temperatures, which in turn would change the body temperature, impair some immune responses, such as phagocyte function, and can contribute to the increased risk of infections seen at extremes of temperature. Infection itself can suppress immune responses and worsen malnutrition. The prime examples of this are measles and acquired immunodeficiency syndrome. Measles is associated with impaired cell-mediated immunity for 12-16 weeks. Emotional stress, such as bereavement on the part of the elderly (Table 7-3) and fear of scholastic examinations on the part of medical school students, is associated with reductions
TABLE 7-2 Confounding Nonnutritional Factors Influencing Immune Responses
|
Genetics |
|
Trauma: physical, thermal |
|
Environmental and body temperature |
|
Infection |
|
Emotional stress |
|
Physical activity |
in immune responses such as natural killer (NK) cell activity, lymphocyte stimulation response to mitogens, and IL-2 production. Physical activity has an interesting correlation with immune response (Gleeson et al., 1995) and susceptibility to infection. Moderate, graded exercise is associated with enhanced immune responses and decreased incidence of infection, particularly among those groups such as the elderly who show compromised immunity and experience frequent infections (Table 7-4). In contrast, strenuous exercise—both severe and prolonged exercise—reduces immunity and increases the incidence of infection in the short term. Thus, a reverse J-shaped curve best describes the relationship between physical activity and immunocompetence (Figure 7-4).
TABLE 7-3 Immunologic Responses in 11 Elderly Subjects
|
Immunologic Response |
Prebereavement |
Postbereavement |
P |
|
Lymphocyte stimulation response to PHA* |
47 ± 9 |
23 ± 12 |
< 0.05 |
|
Interleukin-2 production, U/ml |
10 ± 2 |
7 ± 2 |
< 0.05 |
|
Natural killer cell activity, % |
43 ± 4 |
28 ± 7 |
< 0.01 |
|
NOTE: P was calculated by paired Student's t-test. Prebereavement tests were done 1 to 3 months prior to loss of spouse as part of a longitudinal study. Postbereavement assessment was conducted 1 to 2 weeks after loss of spouse. U, units. The data represent means ± SD. * PHA, phytohemagglutinin. Results were expressed as counts per minute in PHA-stimulated culture/counts in unstimulated cultures. |
|||
TABLE 7-4 Immunologic Responses in 14 Elderly Subjects Enrolled in an Exercise Program
|
Immunologic Response |
Pre-exercise Period |
Post-exercise Period |
P |
|
Lymphocyte stimulation response to PHA* |
39 ± 8 |
63 ± 13 |
< 0.05 |
|
Interleukin-2 production, U/ml |
7 ± 3 |
16 ± 2 |
< 0.01 |
|
Natural killer cell activity, % |
32 ± 6 |
49 ± 8 |
< 0.01 |
|
NOTE: P was calculated by paired Student's t-test. After base-line assessment, the subjects were enrolled in a graded program of gentle exercise, consisting of walking and low-impact aerobic activity, ~40 minutes, 4 times a week for a total of 6 months. Blood was withdrawn on a nonexercise day. U, units. The data represent means ± SD. * PHA, Phytohemagglutinin. Results were expressed as counts per minute in PHA-stimulated culture/counts in unstimulated cultures. |
|||

FIGURE 7-4
Schematic relationship among intensity of exercise, immune responses, and incidence of respiratory infection. Strenuous and/or prolonged physical activity lowers immunity, and regular moderate exercise for short periods enhances immunity. ●, young subjects (n=8–12 in each group); ■, elderly subjects (n=8–11 in each group).
Nutritional Supplements for Boosting Immunity
In population groups with a high prevalence of nutritional deficiencies, supplementation to prevent or treat specific nutrient deficiency may be expected to decrease morbidity and improve survival. This has been documented for vitamin A, iron, and zinc (Sazawal et al., 1996; Sommer et al., 1986; Vyas and Chandra, 1984), although a few studies have failed to show beneficial results (Arthur et al., 1992; Rahmathullah et al., 1991), and in others, the effect on diarrheal disease was negated by an increase in respiratory illness. The potential significance of the negative effect of massive dosing of single nutrients should be borne in mind. Nevertheless, population-wide efforts to eradicate highly prevalent nutritional deficiencies can have a major impact on immunity and risk of illness and death. A significant correlation between serum retinol levels and long-term morbidity has been observed in infants with respiratory syncytial virus bronchiolitis2 (Figure 7-5). It should be noted that none of the serum retinol levels were below 20 µg/dl, a threshold commonly accepted to demarcate clinically significant deficiency of vitamin A. Thus, even within the "normal"
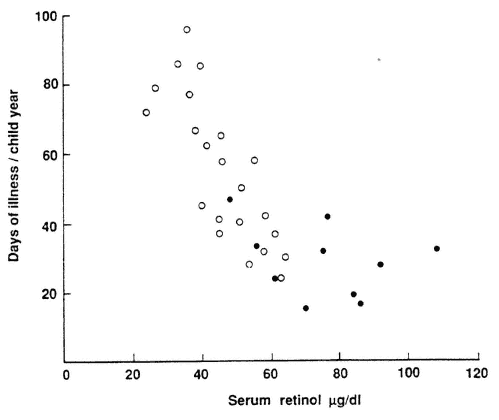
FIGURE 7-5
Relationship between serum retinol concentration and respiratory morbidity in 32 infants with acute bronchiolitis
caused by respiratory syncytial virus. Blood was drawn approximately 7 to 10 days after recovery from the acute illness, and the infants were followed monthly for 1 year. Morbidity data were recorded on a questionnaire, completed daily at home and reviewed by the study team every month. Retinol was estimated by high performance liquid chromatography. ●, infants (n=10) receiving multivitamin supplements at home; ○, infants (n=22) who had not been given any multivitamin supplement (r = 0.87).
TABLE 7-5 Immunostimulant Nutrients
|
Vitamins: vitamin A, β-carotene, vitamin E |
|
Minerals: zinc, selenium |
|
Amino acids: glutamine, arginine |
range of retinol values, there was a correlation between nutrient levels and the burden of illness. A limited trial with physiologic amounts of vitamin A—5,600 retinol equivalents once a week for 1 year—was associated with reduced respiratory morbidity (Unpublished data, R. K. Chandra, Memorial University of Newfoundland, Janeway child Health Centre, St. John's, 1994).
Whereas deficiencies of most nutrients impair immune responses, a modest increase in the intake of some micronutrients may be associated with an enhancement of selected immune responses. The principal nutrients for which such evidence exists are listed in Table 7-5. Thus, a feeding regimen that includes an extra amount of some or all of these micronutrients can enhance immunity particularly in individuals with a compromised immune system. This concept has been used successfully to devise feeding formulas with purported immunostimulant properties, for example, IMPACT® (Sandoz) and IMMUNAID® (McGaw). Both these products contain additional amounts of arginine, glutamine, omega-3 fatty acids and micro-nutrients. Indeed, rodents fed on these special formulas have higher immune responses and improved survival after challenge with Listeria monocytogenes (Chandra et al., 1991, 1992). Limited clinical data such as incidence of wound infections, other postoperative complications, and length of hospital stay support these observations (Bower, 1995). The era of designer feeding cocktails has just dawned.
Another population group in whom nutrient deficiencies are common is the elderly (Chandra, 1991, 1992a; Herbeth et al., 1992; Lesourd et al., 1992; Meydani et al., 1995). As many as 40 percent of elderly have reduced dietary intake and/or low blood levels of one or more nutrients. This is associated with impaired immune responses and a high incidence of infections. In a randomized, double-blind, placebo-controlled study of noninstitutionalized elderly, supplementation with all micronutrients in physiological amounts or modest excess was associated with enhanced cell-mediated immune responses, higher antibody level after immunization with influenza virus vaccine, higher NK cell activity, and increased IL-2 production (Chandra, 1992a). Most importantly, those receiving the micronutrient supplement experienced fewer respiratory infections and therefore were prescribed fewer antibiotics.
Concluding Remarks
Several exciting advances have contributed to an understanding of interactions among nutrition, immunity, and infection. A new facet of host-parasite interaction includes the possibility of viral mutation with altered virulence brought about by nutrient deficiency. It is now established that undernutrition is associated with consistent changes in immune responses such as the number of T-cells, lymphocyte response to mitogens and antigens, phagocyte function, secretory IgA antibody response, complement activity, NK cell activity, and production of cytokines. Immunocompetence is a sensitive and functional index of nutrition. Nutrient-specific immunological tests, such as thymulin for zinc status, hold promise for estimating optimum intake. The prevention and correction of nutrient deficiencies, even subclinical ones, can reduce the burden of illness and decrease mortality.
References
Alexander, J.W. 1995. Specific nutrients and the immune response. Nutrition 11(suppl. 2):229-232.
Anonymous. 1987. This week's Citation Classic. Current Contents 30:15.
Arthur, P., B. Kirkwood, D. Ross, J. Gyapong, A. Tomkins, and A. Hutton. 1992. Impact of vitamin A supplementation on childhood morbidity in northern Ghana. Lancet 339:361-362.
Beisel, W.R. 1982. Single nutrients and immunity. Am. J. Clin. Nutr. 35:417-468.
Beisel, W.R. 1992. History of nutritional immunology. J. Nutr. 122:591-596.
Bendich, A., and R.K. Chandra. 1990. Micronutrients and Immune Functions. New York: New York Academy of Sciences.
Bower, R.H., F.B. Cerra, B. Bershadksy, J.J. Licari, D.B. Hoyt, G.L. Jensen, C.T. Van Buren, M.M. Rothkopf, J.M. Daly and B.R. Adelsberg. 1995. Early enteral administration of a formula (Impact) supplemented with arginine, nucleotides, and fish oil in intensive care unit patients: results of a multicenter, prospective, randomized, clincial trial. Crit. Care Med. 23(3):436-449.
Chandra, R.K. 1972. Immunocompetence in undernutrition. J. Pediatr. 81:1194-1200.
Chandra, R.K. 1975. Antibody formation in the first and second generation offspring of nutritionally deprived rats. Science 19:289-290.
Chandra, R.K. 1984. Excessive intake of zinc impairs immune responses. J. Am. Med. Assoc. 252:1443-1446.
Chandra, R.K. 1991. 1990 McCollum Award lecture. Nutrition and immunity: lessons from the past and new insights into the future. 53(5):1087-101.
Chandra, R.K. 1992a. Effect of vitamin and trace-element supplementation on immune responses and infection in elderly subjects. Lancet 340:1124-1127.
Chandra, R.K. 1992b. Experience of an old traveller and recent observations. Pp. 9-43 in The First INIG Award Lecture: Nutrition and Immunology. St. John's, Newfoundland: ARTS Biomedical Publishers and Distributors.
Chandra, R.K. 1996. Nutrition, immunity and infection: From basic knowledge of dietary manipulation of immune responses to practical application of ameliorating suffering and improving survival . Proc. Natl. Acad. Sci. USA 93:14304-14307.
Chandra, R.K., and P.M. Newberne. 1977. Nutrition, Immunity, and Infection: Mechanisms of Interactions. New York: Plenum.
Chandra, R.K., M. Baker, S. Whang, and B. Au. 1991. Effect of two feeding formulas on immune responses and mortality in mice challenged with Listeria monocytogenes. Immunol. Lett. 27:45-48.
Chandra, R.K., S. Whang, and B. Au. 1992. Enriched feeding formula and immune responses and outcome after Listeria monocytogenes challenge in mice. Nutrition 8:426-429.
Chowdhury, B.A., and R.K. Chandra. 1987. Biological and health implications of toxic heavy metal and essential trace element interactions. Prog. Food Nutr. Sci. 11:55-113.
Endres, S., S.N. Meydani, R. Ghorbani, R. Schindler, and C.A. Dinarello. 1993. Dietary supplementation with n-3 fatty acids suppresses interleukin-2 production and mononuclear cell proliferation. J. Leukoc. Biol. 54:599-603.
Erickson, K.L., N.E. Hubbard, and S.D. Sommers. 1992. Dietary fat and immune function. Pp. 81-104 in Nutrition and Immunology, R.K. Chandra, ed. St. John's, Newfoundland: ARTS Biomedical Publishers.
Failla, M., and S. Bala. 1992. Cellular and biochemical functions of copper in immunity. Pp. 129-142 in Nutrition and Immunology, R.K. Chandra, ed. St. John's, Newfoundland: ARTS Biomedical Publishers.
Fernandes, G., J.T. Venkatraman, and N. Mohan. 1992. Effect of omega-3 lipids in delaying the growth of human breast cancer cells in nude mice. Pp. 283-296 in Nutrition and Immunology, R.K. Chandra, ed. St. John's, Newfoundland: ARTS Biomedical Publishers.
Gershwin, M.E., R.S. Beach, and L.S. Hurley. 1985. Nutrition and Immunity. New York: Academic Press.
Gleeson, M., W.A. McDonald, A.W. Cripps, D.B. Pyne, R.L. Clancy, and P.A. Fricker. 1995. The effect on immunity of long-term intensive training in elite swimmers. Clin. Exp. Immunol. 102:210-216.
Good, R.A., and E. Lorenz. 1992. Nutrition and cellular immunity. Int. J. Immunopharmacol. 14:361-366.
Herbeth, B., A. Lemoine, B.P. Zhu, and M. Chavance. 1992. Vitamin status, immunity and infections in the elderly. Pp. 225-237 in Nutrition and Immunology, R.K. Chandra, ed. St. John's, Newfoundland: ARTS Biomedical Publishers.
Kelley, D.S., L.B. Branch, J.E. Love, P.C. Taylor, Y.M. Rivera, and J.M. Iacono. 1991. Dietary α-linolenic acid and immunocompetence in humans. Am. J. Clin. Nutr. 53:40-46.
Keusch, G.T., C.S. Wilson, and S.D. Waksall. 1983. Nutrition, host defenses and the lymphoid system. Arch. Host Def. Mech. 2:275-359.
Lesourd, B.M., R. Moulias, M. Favre-Beffone, and C.H. Rapin. 1992. Nutritional influences on immune responses in the elderly. Pp. 211-224 in Nutrition and Immunology, R.K. Chandra, ed. St. John's, Newfoundland: ARTS Biomedical Publishers.
Meydani, S.N., and M. Hayek. 1992. Vitamin E and the immune response. Pp. 105-128 in Nutrition and Immunology, R.K. Chandra, ed. St. John's, Newfoundland: ARTS Biomedical Publishers.
Meydani, S.N., D. Wu, M.S. Santos, and M.G. Hayek. 1995. Antioxidants and immune response in aged persons. Overview of present evidence. Am. J. Clin. Nutr. 62(suppl.):1462S-1476S.
Rahmathullah, L., B.A. Underwood, R.D. Thulasiraj, and R.C. Milton. 1991. Diarrhoea, respiratory infections and growth are not affected by a weekly low dose vitamin A supplement: A masked controlled field trial in children in Southern India. Am. J. Clin. Nutr. 54:568-577.
Santos, J.I. 1995. Nutrition, infection, and immunocompetence. Infect. Dis. Clinics North Am. 8:243-267.
Sarchielli, P., and R.K. Chandra. 1991. Immunocompetence methodology. Pp. 425-545 in Nutrition Status Assessment, F. Fidanza, ed. London: Chapman and Hall.
Sazawal, S., R.E. Black, M.K. Bhan, S. Jalla, N. Bhandari, A. Sinha, and S. Majumdar. 1996. Zinc supplementation reduces the incidence of persistent diarrhea and dysentery among low socioeconomic children in India. J. Nutr. 126(2):443-450.
Scrimshaw, N.S., C.E. Taylor, and J.E. Gordon. 1968. Interactions of nutrition and infection. Monograph. Geneva: World Health Organization.
Smythe, P.M., M. Schonland, G.G. Brereton-Stiles, H.M. Coovadia, H.J. Grace, W.E.K. Loening, A. Mafoyne, M.A. Parent, and G.H. Vos. 1971. Thymolymphatic deficiency and depression of cell-mediated immunity in protein-calorie malnutrition. Lancet 2(7731):939-943.
Sommer, A., I. Tarwotjo, E. Djunaedi, K.P. West Jr., A.A. Loeden, R. Tilden, L. Mele. 1986. Impact of vitamin A supplementation on childhood mortality. A randomised controlled community trial. 1(8491):1169-1173.
Suskind, R.M., ed. 1977 Malnutrition and the Immune System. New York: Raven Press.
Venkatraman, J.T., and G. Fernandes. 1992. Mechanisms of delayed autoimmune disease in B/W mice by omega-3 lipids and food restriction. Pp. 309-323 in Nutrition and Immunology, R.K. Chandra, ed. St. John's, Newfoundland: ARTS Biomedical Publishers.
Vyas, D. and R.K. Chandra. 1984. Functional implications of iron deficiency. In Iron Nutrition in Infancy and Childhood, A. Stekel, ed. New York: Raven Press.
Watson, R.R., ed. 1984. Nutrition, Disease Resistance and Immune Functions. New York: Marcel Dekker.
Discussion
RONALD SHIPPEE: That was great, Dr. Chandra. In our human model in the field, we can only look at one compartment or lymphoid system as well as peripheral blood. I often wonder if we are trying to fix something that is not broken. We have already had some comments questioning the Army's interpretation of the PHA stimulation data for peripheral blood lymphocytes. Maybe we are just looking at the effect of margination of blood vascular leukocytes in these soldiers, and maybe it is a protective mechanism, and maybe we should not be in there trying to change it.
RANJIT CHANDRA: I think that Dr. Wilmore certainly addressed this as well: that in the surgical patient or in some of the acute stress patients, sometimes for a short period it is beneficial for the host to turn down immune responses. I think there may be some stressful situations where it is useful to let immune response stay depressed for a period of time. But there are other aspects of immunity which I think need to be brought up to normal. So, once again, I cannot overemphasize that your end point has to be what the biological outcome will be, even if you are looking at low lymphocyte responses to mitogens. If you are seeing no change in infection, then what does it mean?
RONALD SHIPPEE: I worked for 8 years at the Burn Center in San Antonio. Studies employing a burned rat model have demonstrated that if you have a 30
percent scald burn on a rat, the circulating blood [lymphocytes] show a decrease in ConA [concanavalin A]-stimulated proliferation, but proliferation in the draining lymph node and the spleen is actually decreased.
HARRIS LIEBERMAN: Thank you for a wonderful presentation. My question is, you had noted at the beginning of your talk that there appeared to be some association between the progression of AIDS and nutritional status. Can you indicate how strong that association typically has been found to be, and what sort of populations have been studied with that issue in mind, that is, in developed world or third-world kinds of populations?
RANJIT CHANDRA: There are three groups [that are studying the relationship of the progression of AIDS and nutritional status] in three areas of the world that I am aware of. The first group of studies are those originating in the United States—one from the Baltimore area, led by Neil Graham and his group in epidemiology. I have been associated with a couple of those studies. Secondly, there is also a group from Johns Hopkins working in Uganda, working mostly on maternal and maternal-child transmission. I know that Dr. Semba has worked on vitamin A status on maternal child transmission. Then there are studies coming from Florida. Dr. Beach, among others, has shown that the micronutrient levels in the blood correlate with some progression indices. Now, these studies are in an early phase. As you might expect, different studies are coming up with different nutrients that correlate best. Also, I think one earlier study from the Hopkins group suggested some B vitamins as being very important, and lastly, another study from Africa showing that a higher B12 level is, in fact, more ominous than a low B12 level. So I think we are in an early phase. But there seems to be some indication that there may be a relationship between the progression of AIDS and nutritional status that we need to look at carefully.
MELVIN MATHIAS: I have been very interested in following this literature and making the transition from the deficiency model to a supplemental model. I am not sure that the biochemical mechanism is continuous. You have done a lot of work with zinc in immunity. Is there a transposition from deficiency—that is, is there a change in the biochemical mechanism in the deficient state to the supplemental state?
RANJIT CHANDRA: I think there is. I really appreciate this question because we know very little. For example, we know that zinc deficiency affects several enzymes that are key for protein synthesis and for cellular proliferation, and certain aspects of immune responses. But when you go beyond deficiency to
large amounts, other mechanisms come into play. I am thinking of the membrane transport of nutrients. Even calcium transport across the lymphocyte cell membrane is affected by large amounts of zinc. I am looking at stability of lymphocyte membranes. I am looking at the composition of lymphocyte membrane in terms of fatty acid composition, which is affected by zinc intake.
We know, from earlier studies, that even serum levels of fatty acids are affected, which, in turn, might impair or influence immune responses. So you are perfectly correct to say that the mechanisms whereby a zinc deficiency impairs immune responses are probably quite different from mechanisms where very large amounts of zinc may also impair immune response.
DOUGLAS WILMORE: The committee is faced with different types of questions, and I wonder whether you would reflect on them. My view really jives very much with the English church records, that is, malnutrition results in immunological impairment and infection. But what we are faced with are several sorts of objectives. One is to keep the military personnel well nourished before they go into a stress situation. We are asked, can we take those people shortly before they go into a stress situation and give them something, possibly a supplement, to make them superimmune or superresponsive, or superresistant? Then I guess the last thing that we are asked is when soldiers are in the field and in these other stress situations and probably suboptimally nourished, is there some way we can alter nutrient intake in that situation to enhance immunologic response?
We have a fourth concern, and that is, once they are injured or infected how to return them to function, but let's stay away from that for right now. Give us your sense about where we can realistically accomplish some of these goals, sort of along these various pathways.
RANJIT CHANDRA: To my mind, the answers to these questions are yes, yes, and yes. That is, there are things that one can do to optimize nutritional status and, therefore, immunity prior to anybody going into a stressful situation, even with the best optimal military diet. First, I am sure that, if you assess the nutritional status of these individuals going into the Ranger training, not all of them will be at an optimum level in all nutrients. If, for a period of time, be it 2 weeks, or 4 weeks, or 4 months, you can not only assess but optimize their status, it will prepare them for a period of stress, maybe increased utilization and need for nutrients, and, therefore, minimize the decline that will occur, not only in nutritional status but also in functional consequences like immunity.
Secondly, certainly during a stressful period, there could be elements that we can look at which are more important than others in sustaining immune response.
To your last question, there is no doubt, as we have seen in many other scenarios, that once you are depleted, there are certain ways in which you can
not only replete nutrient status more rapidly but also replete immune responses. So I think what we need is to fine-tune these ''yeses'' to identify the nutrients, the amounts, and the period over which such repletion has to take place.
DOUGLAS WILMORE: Those may all be different in different situations.
RANJIT CHANDRA: Certainly.
ROBERT NESHEIM: I think we need to take our coffee break. Actually, I think that those questions you pose are ones that the committee is going to have to deal with in the next couple of days, and hopefully, from an analysis of all of the information we have, we can make some recommendations. That certainly spells out our task.
















