Chapter 2
Etiology, Diagnosis, and Detection of Tuberculosis
Cattle are considered to be the definitive hosts of M. bovis although the prevalence of M. bovis infection in cattle in the United States has been greatly reduced by eradication efforts (Timoney et al., 1988; Konyha et al., 1980; Konyha, 1986; Essey, 1991).
Most M. bovis infected cattle appear normal. They carry and may shed the organism for years without showing signs of disease. Clinical signs occur only in the advanced stages of the disease, thus most infected cattle are slaughtered or culled for other reasons before they reach the advanced stages. In situations where clinical disease is observed, signs include emaciation, fever, coughing, labored breathing, and infrequently, diarrhea. Superficial lymph nodes may be swollen, or rupture and drain to the outside.
INFECTION
The bacterium usually enters by inhalation, multiplies locally, and causes primary lesions in the lungs and the lymph nodes that drain the respiratory tract (Collins and Grange, 1983; Pritchard, 1988). Infection also occurs through ingestion of contaminated milk, water, or feed, resulting in primary lesions in the lymph nodes associated with the alimentary tract and the intestine. Calves may become infected with M. bovis in utero or by ingestion of milk from a tuberculous udder. Infection rarely occurs through the skin (wounds) or genital tract (at breeding). Inhaled or ingested M. bovis can enter the tissue through breeches in the epithelium lining the respiratory or alimentary tracts or through specialized transporting epithelial cells, called M cells. Bacteria in tissue are engulfed by neutrophils and macrophages. Some of the infected macrophages migrate carrying the infection to the regional lymph nodes. Occasionally, infected macrophages migrate into the blood and the infection is disseminated throughout the body. Thus, M. bovis can infect multiple organ systems or become localized in any organ in the body.
Mycobacterium bovis survives in, multiplies in, and ultimately destroys the neutrophils and macrophages that engulf it (Johnston et al., 1988; Kaufmann, 1988). Growth of M. bovis in generations of macrophages accounts for the chronic progressive course of the disease. The characteristic lesion (tubercle or granuloma) contains debris from necrotic host cells and possibly free M. bovis cells at its core. The core is surrounded by M. bovis infected macrophages (epithelioid and giant cells), which are in turn surrounded by fibrous connective tissue and lymphocytes. Resolved or inactive lesions may be walled off by an extensive connective tissue capsule, have calcified debris at the core, and contain few if any viable M. bovis organisms. Active, rapidly progressing lesions tend to have extensive cores with semisolid (caseous) contents, numerous viable M. bovis, and comparatively little capsule. Expansion of these tubercles until they rupture (cavitary lesions) into air passages of the lung, blood vessels, mammary gland, intestinal tract, or through the skin facilitates spread of the organism within and between animals. Tubercles range from microscopic size to several centimeters in diameter. Thus, in the early stages or in mild infections, the lesions may not
be detectable by visual examination (gross postmortem) of the carcass (Corner et al., 1990), and samples collected from infected animals for microscopic examination or culture may not always include the infected tissue.
Disease Processes
Small numbers of M. bovis remain viable for years within macrophages at the center of encapsulated, inactive tubercles in some cattle. The lesions sometimes become active again later in life, perhaps caused by a number of factors including stress, concurrent disease, or old age (Griffin, 1989).
Mycobacterium bovis contains an extensive array of complex lipids that enable the bacteria to survive in macrophages (Thoen and Himes, 1986). These lipids are thought to inhibit fusion of lysosomes (macrophage organelles that contain enzymes that kill and digest bacteria) with phagosomes (macrophage organelles that contain bacteria). The lipids also protect M. bovis against the effects of lysosomal enzymes and the array of other antimicrobial substances produced by macrophages. Mycobacterium bovis also produces an array of proteins (called stress or heat-shock proteins) that further protect the bacteria from destruction by phagocytes and enable them to compete with host cells for the limited nutrients available within phagosomes (Munk and Kaufmann, 1991; Young et al., 1988).
Although production of specific endotoxins by M. bovis has not been established, the metabolic products of the bacteria are assumed to be toxic for neutrophils and macrophages. Tissue necrosis is mediated in part by the direct effects of bacterial products and in part by the immune response of the host (Rook, 1988). Once immunity has been established, cytotoxic T-lymphocytes kill macrophages bearing M. bovis antigens. The enzymes, cytokines (for example, tumor necrosis factor, and interleukin 1), and other products released by lymphocytes and activated or necrotic macrophages are toxic to some host cells and may result in expansion of the necrotic core of the granuloma. They can also cause systemic manifestations of disease (fever, emaciation, and changes in the concentration of some blood proteins such as fibrinogen and haptoglobin). Histologically, giant cells, epithelioid cells, and acid-fast organisms characterize tuberculous abscesses. Culture and identification of M. bovis confirms the diagnosis (Beatson, 1985).
Transmission
Mycobacterium bovis can be shed from virtually any body orifice. The respiratory route is consistently described as the major route of infection although oral infection is also common. This inference is largely based on location of lesions, although early experimental work indicated that it was more difficult to infect via the oral route than via the aerosol route. Respiratory shedders also tend to shed organisms in their feces subsequent to swallowing bronchial exudate. Shedding, particularly by the respiratory route, can precede the development of grossly identifiable lesions.
Introduction and maintenance of bovine tuberculosis in a herd is greatly influenced by herd or population factors such as size (number of animals), confinement density, and exposure to individuals from the outside. The risk of introduction is lowest in populations that do not introduce new animals from the outside and have no contact with other herds or populations of susceptible animals. Large herds provide the greatest number of opportunities for contact of an infected individual with individuals that are susceptible to the disease. High density and confinement enhance the efficiency
(dose and frequency of exposure) of aerosol transmission, increase the number of nose-to-nose contacts, and increase the proportion of animals that contact contaminated feces, exudates, feed, or water. Housing, perhaps due to increased density of animals as well as contamination of the air and or environment, aids transmission of the disease (Blood et al., 1989).
The major source of transmission is other infected Bovidae (or, in more recent cases, infected Cervidae) sharing the same premises. Often a simple wire fence is sufficient to prevent pasture-to-pasture spread; this principle was used in early campaigns where a clean herd was started on the same premises as an infected herd (the Bang Method; Pritchard, 1988). On infected premises, other infected animals can include dogs and cats, which serve as reservoirs for infecting other cattle, and the general recommendation is that they be destroyed if they have had contact with infected cattle and the herd is to be depopulated. Farm workers infected with either M. tuberculosis or M. bovis can be a source of infection for cattle (Konyha et al., 1980). Such spread may arise from respiratory aerosols, urinary contamination of hay or straw bedding as well as fecal contamination of the housing/pasture area.
The organism does not multiply outside of the animal's body but can survive outside the body for extended periods, especially if the environment is damp (18 days in stagnant water; 6 to 8 weeks under contaminated feces) and warm. Sunlight and dryness shorten its environmental survival.
Cleaning infected premises, especially feedtroughs and waterers, followed by disinfection with 5 percent phenol or equivalent cresol is recommended. Pastures may be harrowed and allowed to dry in sunlight to reduce contamination (Blood et al., 1989).
Routes of Transmission
A number of experiments have been performed to infect cattle and/or contaminate their environment (pasture) and then study the spread of disease in susceptible cattle. Little is known of the period between becoming infected and being able to transmit infection to others; one study indicated that this period might be approximately 87 days (Neill et al., 1991).
Overall, the results of most studies seem to indicate that cattle-to-cattle transmission does not occur rapidly unless the shedding cattle are releasing large numbers of organisms, as might be expected in cases of clinical disease. Given the virtual absence of clinical disease in cattle in North America, it would seem reasonable to conclude that the risk of transmission from infected cattle is low. For example, in one study 22 naturally infected cattle failed to transmit infection to contact cattle in a 125-to-257-day period (O'Reilly and Costello, 1988). The slowness of spread would also be consistent with the observed success of well-performed test and slaughter campaigns in virtually all countries without a wildlife or other animal reservoir.
Although there is limited data available on the transmission of infection by Cervidae, the nature of the lesions (which are frequently large and with more liquified purulent matter than in cattle) in many cervids would lead one to speculate that they may be more, if not highly, contagious. Certainly the spread of infection from cervids to humans under the conditions of a postmortem examination are consistent with this hypothesis (Fanning and Edwards, 1991). Likewise, badgers and possums may be most contagious in the terminal stages of tuberculosis.
Nontraditional farmed ruminants such as deer, elk, other cervids, llamas, antelope, and wild cattle may serve as sources for infecting cattle or may be at risk of being infected by tuberculous cattle. Both groups may be potential infection sources for susceptible native wildlife species, for example, deer, elk, moose, and badgers. Wild populations of mule deer, white-tailed deer, and elk
have undergone remarkable growth in the United States since 1900. The population densities of these species in the United States now exceeds those in other countries where outbreaks of bovine tuberculosis have occurred in wild cervids (U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Veterinary Service, 1992a). Infected wildlife could in turn become reservoirs for tuberculosis in cattle.
Infections with other species of Mycobacterium are important because they may interfere with testing for M. bovis. Mycobacterium avium infections frequently confound the tuberculin skin test and serologic tests [for example, the enzyme-linked immunosorbent assay (ELISA)] in cattle, cervids, wild ruminants, and nonhuman primates by causing nonspecific immune sensitization. Similarly, M. paratuberculosis causes a chronic debilitating enteritis (Johne's disease) in cattle, sheep, goats, camelids, and some wild ruminants. Mycobacterium paratuberculosis is closely related to M. avium and can sensitize an infected host to tuberculin (Timoney et al., 1988). It is likely that a number of the cattle, cervids, and zoo ungulates that have been culled because of positive tuberculin skin tests, but were free of visible lesions at necropsy, were sensitized by infection with M. avium, M. paratuberculosis, or in some cases saprophytic soil mycobacteria.
IMMUNE RESPONSE
Genetic variability contributes substantially to the differences in susceptibility to M. bovis that occur among cattle. It also contributes to the wide variations in immune response to M. bovis that occur even among individual cattle within the same herd (Dowling and Schleehauf, 1991; Fifis et al., 1992). Some of those infected develop protective immunity and readily resolve the infection, others partially resolve the infection but retain viable organisms within inactive lesions, while some develop little or no protective immunity and have active progressive disease.
Genetic control and mode of inheritance of innate resistance to tuberculosis has been well defined in mice. There is also some evidence that these variations in resistance are hereditary in humans (Skamene, 1986). Macrophages are the principal site for expression of genetically controlled innate resistance to tuberculosis (Schurr et al., 1991). Macrophages from resistant animals are more readily activated and more effective at killing bacteria and presenting bacterial antigens to the immune system in a manner that will initiate a protective immune response. Macrophages from individuals resistant to mycobacteria are also resistant to other intracellular bacterial pathogens such as Listeria, Brucella, and Salmonella.
When an organism such as M. bovis invades the tissues of the immunocompetent animal, the immune system recognizes it as being structurally different from the makeup of normal tissue and responds to these determinants or antigens. Lymphocytes are the major responder cells of the immune system, and are classified as B cells or T cells. B cells differentiate to daughter cells that produce specific immunoglobin (antibody) when presented with a new antigen. T cells differentiate and produce specific receptors for that antigen. The T-cell response is usually referred to as cell-mediated immunity (CMI); the B- cell response is referred to as an humoral immunity.
Mycobacterium bovis antigens are presented to T cells by macrophages in a form that is recognized by and stimulates some of the T cells to initiate specific immune responses to the array of antigens presented (Kaufmann, 1988). The T-cell responses involve proliferation and dissemination of lymphocytes throughout the body that recognize mycobacterial antigens. One population of lymphocytes called T helper or CD4+ cells is primarily regulatory and produces lymphokines [such as
gamma interferon (γ-IFN)] that stimulate effector cells of the immune system (Huygen et al., 1992). These effector cells include B cells (which differentiate to plasma cells and produce antibody), macrophages (which activate to enhance killing of bacteria and increase production of cytokines), and cytotoxic (killer T and null) lymphocytes that kill host cells bearing mycobacterial antigens. This latter population of lymphocytes, called T suppressor/cytotoxic or CD8+ cells, can act directly as effector cells or as regulatory cells through the release of lymphokines that suppress lymphocyte responses to mycobacterial antigens. Once the mixed populations of M. bovis-committed T cells have been disseminated throughout the body, some of them may persist for years as memory cells and are capable of initiating a rapid immune response on subsequent contact with antigens. Alternatively, this “memory” may be the result of persistent presence of stimulating antigen. This general state of reactivity is known as cell-mediated immunity (CMI) and when the response is stimulated within the skin, it is called delayed-type hypersensitivity (DTH). The net effect of DTH can be protective, by keeping tubercles in the inactive state or by destruction of M. bovis cells encountered in a subsequent external exposure. The net effect can also be destructive if large doses of antigen are presented to a hypersensitive individual through rupture or expansion of an active tubercle. In this case immune-mediated tissue destruction can be severe, it can accelerate progression of the disease, and precipitate clinical signs.
Both protective and destructive components of the immune response depend primarily on CMI (Thorns and Morris, 1983). Antibody-mediated mechanisms (enhanced phagocytosis, complement fixation, antibody-dependent cytotoxicity) play a comparatively minor role in tuberculosis. In advanced stages of the disease, suppressor T-cell effects can predominate. The net effect of suppressor cells combined with the stress of advanced disease can result in immunologic anergy. Anergy is characterized by the lack of response by committed lymphocytes from sensitized individuals when mycobacterial antigens are encountered either in vivo or in vitro. Anergy can be a nonspecific stress-mediated state or a specific immune-mediated state regulated through lymphokines produced by suppressor T cells.
DIAGNOSIS AND DETECTION OF MYCOBACTERIUM BOVIS INFECTION
For nearly a century, tuberculosis has been diagnosed primarily by the detection of acid-fast bacilli in postmortem examinations and the use of the intradermal tuberculin test in live animals (Pritchard, 1988). Numerous diagnostic tests have been proposed, evaluated, and discarded during the past century; most control efforts have depended on the original diagnostic tests (Lepper and Corner, 1983; Pritchard, 1988), which are the tests officially recognized and approved by the Office International des Epizooties. This section will present methods available for diagnosis and detection of M. bovis infection and discuss the use of these diagnostic tests in a surveillance program.
Interpretation of Diagnostic Tests
The availability of an efficient screening test is an essential feature for a successful disease control and eradication program such as the Bovine Tuberculosis Eradication Program (U. S. Department of Agriculture, 1988). Ideally a screening test should be easy to perform, relatively inexpensive, and easy to interpret as positive or negative. It should be possible to incorporate the test
easily into the routine management and production system of the animal population under surveillance.
An ideal screening test must be sensitive so that it identifies all the diseased animals tested, and it must avoid identifying nondiseased animals as diseased. Thus, the ideal diagnostic test would detect signals with the following characteristics:
-
a signal that is present in the animal throughout the course of M. bovis infection;
-
a signal that can be detected in a reproducible manner;
-
a signal of such intensity that it can be consistently detected or amplified to detectable
-
levels without distracting levels of background noise;
-
a signal distributed uniformly throughout the animal or localized in specifically predictable sites so that choice of specimen collection does not lead to incorrect results; and
-
a signal that is uniquely and specifically associated with M. bovis infection.
Performance Characteristics
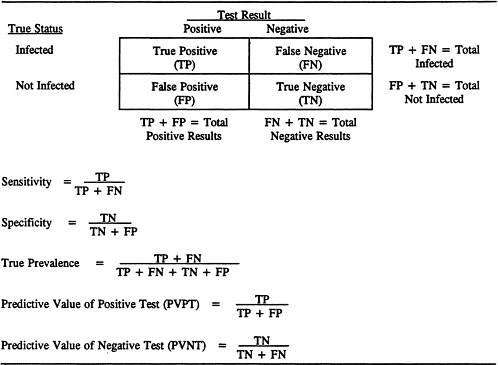
The epidemiological performance characteristics of diagnostic tests can be described by the sensitivity, specificity, and predictive values for positive and negative tests (Vecchio, 1966; Gerstman and Cappucci, 1986; Smith, 1991). Formulas for calculating these characteristics are defined in Table 2-1. Sensitivity is the probability that an infected animal will have a positive test result and specificity is the probability that a noninfected animal will have a negative test result in the assay being evaluated. These two characteristics estimate the probability for the outcome of a test given that the status of the animal is known in advance. These parameters are the primary characteristics used in epidemiological evaluation of test performance. However, when applying tests in a field situation, it is not the status of the animal that is known but rather the result of a test, and the health status then must be interpreted. In this instance, one is interested in knowing the predictive value of a positive test (PVPT) or the predictive value of a negative test (PVNT) result. PVPT is the probability that a positive test result has identified an infected animal and PVNT is the probability that a negative test result has identified a noninfected animal.
Predictive values are influenced by the prevalence of disease and by the sensitivity and specificity of the test. When the disease is present at a low prevalence, PVPT is influenced more by specificity than by sensitivity of the test (Vecchio, 1966). Unfortunately, the proportion of false-positives increases (that is, a lower PVPT) with a decrease in prevalence within a herd (Rosworm and Konyha, 1973). Hence, the proportion of positive tests that are false-positive will become higher as the disease prevalence is reduced. Knowledge of this simple relationship can explain much of the criticism of the present tuberculosis testing program. For example, with a test having a sensitivity of 85 percent and a specificity of 99 percent with a 5 percent true prevalence (which is near the estimated prevalence of tuberculosis in 1917), the PVPT is 81.7 percent. In other words, approximately four of five test-positive animals would be truly infected. Today, however, with the prevalence much less than 1 percent, more than one-half of the positive test results are expected to be false-positive. Thus, in reading the literature it should be noted that apparent prevalence (percent that test positive) probably is higher than the true prevalence (percent actually infected/diseased) (Good, 1987).
It is also noteworthy that in most cases, specificity and sensitivity of diagnostic tests are inversely related. To achieve 100 percent specificity, the test may have such a low sensitivity that it cannot detect a high proportion of infected animals.
Specificity and sensitivity of diagnostic tests are probability estimates based on the samples used. One should characterize sensitivity and specificity of tests based on the total potential population of animals to be tested. Hence, it is crucial that the animals used to evaluate the test be representative of the relative proportion of infected and noninfected animals in the population.
Diagnosis by Direct Detection
By definition, the presence of M. bovis in an animal is considered to be infection, and finding infected animals is a principal goal of an eradication program. Therefore, detection of M. bovis or
products of the microorganism as evidence of infection in the animal meets some requirements of an ideal diagnostic test. The organism, however, may be present in numbers too low for efficient isolation or in sites inaccessible to sampling techniques; so direct detection has not been an efficient approach. Sensitivity of culture is low and the procedure is time consuming and expensive.
Figure 2-1 displays the course of the infection over time. The course must also be considered when trying to detect a single infected animal at various stages of the disease process. When an animal is initially infected with M. bovis, viable, cultivatable bacteria are present in low numbers. As the disease progresses over time, the bacteria multiply and spread to other body sites. Now, the presence of multiple, easily observable lesions leads to collection of multiple samples. Therefore, the probability becomes greater for locating a sample that contains cultivatable bacteria and for isolating the bacteria from the sample when it arrives in the laboratory. Nonetheless, the number of viable organisms in optimally selected samples can be highly variable. Tissue samples must be handled in a manner that preserves the viability of mycobacterial organisms and prevents contamination of the
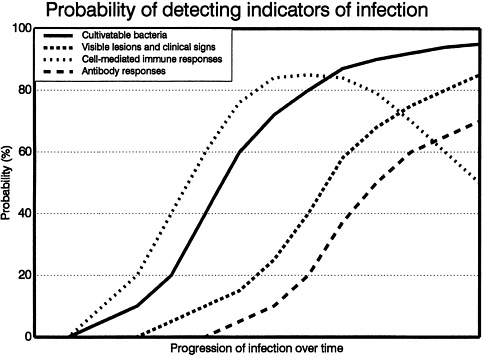
FIGURE 2-1 Probability of detecting indicators of infection. A variable period of time extending from weeks to years following exposure to M. bovis is necessary before the infection progresses to the extent that lesions develop and can be recognized during the postmortem examination. An arbitrary number (10 weeks or years) has been used here to aid in a visual representation of the course of the disease over time. CB, cultivatable bacteria; LS, visible lesions and clinical signs; CMI, cell-mediated immune responses; AR, antibody response.
sample with other bacteria and fungi that could overgrow the organism during the culture period in the laboratory.
Several processing steps are required for optimal isolation of mycobacteria (Roberts et al., 1991). First, the non-M. bovis bacterial flora of the sample must be eliminated or significantly reduced. Several standard decontamination procedures have been described (Collins and Grange, 1983; Corner and Trajstman, 1988; Roberts et al., 1991). Even under the best conditions, however, most decontamination procedures eliminate all but 10 to 20 percent of the mycobacteria contained in the specimen (Roberts et al., 1991).
A variety of media are recommended for the primary isolation of M. bovis (Corner and Nicolacopoulos, 1988; Thoen, 1990). Recent studies of media suggest that modifications of historically favored methods provide enhanced recovery of the organism (Veerman et al., 1986; Cousins et al., 1989; Martin et al., 1989; Jarnagin, 1991). It takes 3 to 12 weeks for the small colonies of M. bovis to grow on primary isolation. Then the isolate must be definitively identified by means of criteria such as growth rate, colony morphology, biochemical behavior, and drug susceptibility (Collins and Grange, 1983).
Diagnosis by Detection of Lesions
Many diagnostic tests detect a signal that is associated with the pathophysiological process of M. bovis infection, for example, detection of granulomatous lesions or an immunological response to mycobacterial antigen. These indirect tests have been referred to as “surrogate tests” (Feinstein, 1974) because they measure substitute factors that are relatively easy to detect and quantitate but only indirectly implicate the presence of M. bovis infection.
Clinical diagnosis of M. bovis infection rarely occurs in cattle because they are usually sent to slaughter at a young age or culled from herds because of low productivity before pathophysiological processes become clinically apparent.
An accurate postmortem diagnosis of M. bovis infection requires a thorough necropsy examination of the animal carcass. A variable period of weeks to years following exposure to M. bovis is necessary for the infection to progress to the point that lesions develop and can be recognized during postmortem examination. Mycobacterium bovis usually infects certain primary sites; thus subsequent tuberculous lesions are not randomly distributed throughout the animal carcass (Lepper and Corner, 1983; Rivas et al., 1985). Further, lesions may vary from a single primary lesion to multiple secondary lesions and range in size from microscopic foci of infection to large readily identifiable tubercles. A thorough systematic necropsy is more likely to detect infection than routine slaughter inspection of carcasses (Corner et al., 1990).
The nonspecific nature of granulomas establishes a tubercle-like lesion as a surrogate test. It becomes a two-stage process requiring histological and microbiological examination of suspected lesions. Special stains must be applied to the tissue section to detect mycobacterial organisms (Roberts et al., 1991). As noted earlier, definitive diagnosis requires cultural isolation and identification of M. bovis from the tissues. In addition, nonviable M. bovis could be present in lesions and lead to a false-negative culture result. New approaches to identify specific bacteria include immunoassay or immunohistochemical detection of mycobacterial antigens, the use of nucleic acid probes for recognition of specific DNA or RNA base sequences, and chemical analysis for the detection of specific metabolic by-products or structural components of M. bovis.
Intensive slaughter surveillance investigations suggest that infected animals may be presented that harbor cultivatable M. bovis in their tissues and do not demonstrate lesions (Duffield et al., 1985; Neill et al., 1986; de Kantor et al., 1987; Margatho et al., 1989; Ritacco et al., 1990). Therefore, the probability of diagnosing infection by culture of suspected exposed animals could be greater than by lesion detection. Diagnosing infection could be enhanced by routinely collecting a predetermined set of tissue samples from reactors for pooling and culture.
Diagnosis Using Immunological Signals of Infection
Because immunological responses depend on antigen presentation followed by lymphocyte differentiation, there will be a variable length of time following infection when immunological response cannot be detected (Figure 2-1). The first immunological response or signal detected in diagnostic tests is weak. With repeated antigenic stimulation, provided by growth of M. bovis in the animal, the intensity of the response generally increases.
In addition to the variable latent period, there are other reasons why immune responses may not be detectable. Differential gene expression during the course of infection might affect which antigens are recognized by the immune system (Young and Mehlert, 1989). Biological variability, which may be genetically based, can lead to major differences in the antigens that are recognized by different classes, species, or breeds of animals (Thorns and Morris, 1983; Nikonenko et al., 1988; Pritchard, 1988). Differences in immunological responses may also be secondary to other pathophysiological processes that alter immune function including viral infections of lymphoid tissues, environmental intoxicants specific for lymphocytes, stress, and immune-modulating chemotherapy. Another problem occurs when infected cattle become “anergic” and lose responsiveness to a particular antigen. The frequency of occurrence and mechanism of anergy in bovine tuberculosis is poorly defined (Plackett et al., 1989).
Other causes of false-negative tuberculin tests include tuberculin losing its activity through improper storage, handling, or contamination and improper test administration. Injection of too little antigen or injecting it too deeply may not produce a recognizable positive test response. Also, if test responses are not observed or not recorded, results will be skewed. Downey (1992) concluded that “defective and careless testing have undoubtedly contributed to the lack of progress ” in the Irish program. Another factor is residual effects from tuberculin testing. The animal may develop a stronger response to subsequent tests by producing antibodies or it may have a diminished response to tuberculin at the same site within the next 60 days (Radunz and Lepper, 1985; Harboe et al., 1990).
Nonspecific or cross-reactive false-positive responses may be caused by previous stimulation of the immune system by antigenic determinants presented from sources other than M. bovis. For example, the mycobacteria have genus-specific antigens and some antigens in common with Nocardia and other actinomycetes (Stanford, 1983; Monaghan et al., 1991). Therefore, stimulation of the immune system by bacteria other than M. bovis frequently leads to the development of immunological responses that are detected in diagnostic tests.
Cell-Mediated Immunological Tests
The standard test for detection of tuberculosis is the intradermal tuberculin test that has been used for nearly a century to detect M. bovis infection in cattle (Pritchard, 1988). The test relies on the intradermal injection of mycobacterial antigens. Two to 3 days after injection of the antigens into
a previously exposed animal, a characteristic inflammatory response usually occurs that includes erythema, swelling, and induration at the site. The tuberculin test in cattle has been reported to show a positive response as early as 10 to 14 days (Thoen and Himes, 1986) or as late as 8 weeks (Harboe et al., 1990) after experimental infection; as early as 8 days (Kleeberg, 1960) or as late as 4 to 6 weeks (Blaha, 1989) in naturally acquired infections. Field investigations of new outbreaks show that it may be as long as several years before some cattle become positive. Clearly there is a highly variable period of infection when the test might produce a false-negative result.
Two basic types of tuberculin preparations are in use today. Old tuberculin (OT) is made from concentrated heat-sterilized culture filtrates of mycobacteria (Lepper and Corner, 1983). Because it is a crude preparation with extraneous material from the culture medium, this product may produce nonspecific inflammatory responses (Benoit et al., 1989). The other type of tuberculin is purified protein derivative (PPD) prepared as the protein precipitant fraction of OT from cultures of M. bovis (bovine PPD) and M. avium (avian PPD) (U.S. Department of Agriculture, 1988a). Production of PPD preparations has become standardized (Zsidai and Joo, 1986).
Serological Tests
Mycobacterium bovis infection of cattle induces a humoral immune response although a prolonged latent period of infection may be present during which antibodies cannot be detected. Studies have indicated that this latent period may range up to 20 weeks in experimentally infected cattle (Harboe et al., 1990). Antibody production appears to be proportional to the antigenic load in the animal and reciprocal to CMI responses (Plackett et al., 1989; Harboe et al., 1990; Ritacco et al., 1991). Therefore, serological tests should not be considered as a substitute diagnostic test for the detection of bovine tuberculosis but rather as a complementary test to detect animals in the more advanced stages of infection. This application would be particularly valuable because some of these animals become anergie to the intradermal test. Based on present knowledge serological screening of young animals presented for import holds little promise.
Serological approaches to the diagnosis of bovine tuberculosis have undergone a renaissance with the introduction of ELISA for detection of antibody to mycobacterial antigens (Thoen et al., 1980; Auer, 1987; Ritacco et al., 1987, 1988; Auer and Schleehauf, 1988; Ritacco et al., 1988; Hanna et al., 1989; Plackett et al. 1989; Harboe et al., 1990; Dowling and Schleehauf, 1991; Ritacco et al., 1990, 1991). ELISA tests can be standardized and packaged in kits with stable reagents for low-cost testing. They have demonstrated good reproducibility and do not require elaborate instrumentation. In addition to the ELISA procedures, agglutination and precipitation tests, complement fixation tests, hemagglutination, latex agglutination, bentonite flocculation, indirect fluorescent antibody, and radioimmunoassay procedures have been used to detect antimycobacterial antibodies (Lepper and Corner, 1983; Thoen and Himes, 1986).
Epidemiological Characteristics of Current Diagnostic Tests
The varying probability of detecting the signals of infection (as illustrated in Figure 2-1) is a major reason for differences in reports of sensitivity and specificity for these tests. Each report estimates sensitivity and specificity based on a sample population of animals. Because each sample usually consists of some unknown blending of animals at various stages of the disease instead of a
random sample of the diseased and “normal” populations, they become weighted averages of the probabilities and can vary considerably from study to study.
The intradermal tuberculin test in its various forms has been the mainstay of antemortem diagnosis of M. bovis infection in cattle throughout the eradication program. It is somewhat surprising that firm estimates of sensitivity and specificity are not available, given the widespread and long-term use of this test. Regulatory officials have known the test “works” but not how well (quantitatively). The sensitivity of intradermal tuberculin tests has been reported to range from 70 percent to 90 percent with an average sensitivity of 85 percent (Lepper and Corner, 1983) but this is extremely dependent on the skill and commitment of the diagnostician (Suther et al., 1974). Much less variable than sensitivity, specificity can be influenced by geographic location and environmental exposure of animals to other mycobacteria and related organisms. Therefore, sample size or origin of samples can greatly influence specificity calculations. A crude estimate of the specificity of the caudal fold tuberculin test could be based on the total tested cattle population in the United States. For example, in 1990, 1.49 million cattle were tested with only 3,474 suspect or reactor results reported (U.S. Department of Agriculture, 1990a), leading to an estimated specificity of 99.8 percent. However, if any of the 3,474 responses is a true positive-test result, then the false-positive error rate is reduced and the specificity of the testing program may be greater than 99.8 percent. As a worst-case scenario, in large dairy herds of 4,000 to 8,000 animals that are currently suspected to be infected with M. bovis, the reactor rate to the caudal fold test is less than 2 percent (Good, 1987). Therefore, the specificity of the caudal fold test can be estimated to be greater than 98 percent.
Data are not readily available for calculation of the sensitivity and specificity of the single cervical test. It is generally agreed that it is more sensitive (Suther et al., 1974) but less specific than the caudal fold tuberculin test. The test is presently applied only to a preselected sample of animals at high risk of exposure and infection.
The comparative cervical tuberculin test was reported to have a sensitivity of 74.4 percent and specificity, when used as a primary test, of 97.3 percent (Roswurm and Konyha, 1973). In practical application, however, this test is used as a second stage test and its specificity and sensitivity when used in this manner are reported to be 64.5 percent and 74.1 percent, respectively (Suther et al., 1974).
The sensitivity and specificity of the γ-IFN assay has been reported recently from a large field study in Australia (Wood et al., 1991). The sensitivity was estimated to range from 77 percent to 94 percent; the specificity was estimated to range from 96.2 percent to 98.1 percent. Therefore, the accuracy of the γ-IFN assay appears to be comparable to other tests of CMI responses in cattle.
In general, serological tests are unlikely to have sufficient sensitivity for use as a single screening test to detect infection at all stages of disease. The sensitivity of an ELISA was recently estimated as 73.6 percent (Ritaco et al., 1990). In another study (Dowling and Schleehauf, 1991), of a sample of 150 M. bovis infected cattle, 28.7 percent of the animals did not produce antibodies detectable by ELISA. Thus serology seems destined to remain as a supplemental test in M. bovis control programs.
Slaughter surveillance is now the primary means of detecting tuberculous cattle in the United States. Visual inspection of the carcass for lesions suggestive of tuberculosis is economically efficient because it is included in the routine inspection of meat for human consumption. Suspect lesions are collected and sent to the laboratory for histological and microbiological examination.
The slaughter surveillance program has been successful in finding the majority of infected herds (Hosker, 1987). There are some potential drawbacks, however.
-
The sensitivity of routine inspection in an individual animal is low. Only 19 percent of routinely inspected tuberculin test-positive cattle were found to have lesions; after detailed examination, however, 52 percent were found to have lesions. Many M. bovis- infected animals, roughly one-half the lesioned animals, have only a single lesion and thus may be missed during routine inspection (Corner et al., 1990). The poor sensitivity is offset by inspecting many animals per herd (see subsequent section on Testing Animal Populations). Nonetheless, there has been concern that the number of lesions detected by Food Safety Inspection Service (FSIS) and submitted for laboratory examination is not adequate to detect bovine tuberculosis when present at very low prevalence. Therefore, there is a need for more detailed inspection and collection of more nonspecific granulomatous lesions as prevalence of tuberculosis decreases in order to detect the few remaining infected animals (Hosker, 1987).
-
Individual suspect lesions have a low predictive value because most lesions submitted are not tuberculous; this can lead to complacency among inspectors. There has also been some concern about the effectiveness of this program because the delay between identification of a suspect and initiation of an investigation was approximately 1 month (Hosker, 1987).
Laboratory examination of the lesion is considered the definitive diagnostic test and is used for identifying which animals should be the basis for further epidemiological studies to detect foci of M. bovis infection. In an eradication program, however, the lack of sensitivity of slaughter inspection can be a major hinderance to detecting all cases of infection. Inspector training and efficiency in detecting lesions can be highly variable and limit the effectiveness of this method. Data from field studies suggest that the sensitivity of slaughter inspection ranges from 33 percent to 67 percent (Corner et al., 1990; de Kantor et al., 1987). These estimates may be high, however, because they assume isolation and identification of M. bovis as being 100 percent sensitive. Critical evaluation of M. bovis isolation efficiency from cattle is not available in the scientific literature; however, Neill and colleagues (1986) were able to isolate the organism from 92.1 percent of visible lesions from reactor cattle with histopathologically confirmed tuberculosis.
Use of Multiple Diagnostic Tests
Often the technical and biological limitations of a single diagnostic test limit the potential specificity of the procedure. Further refinement of the test to increase the specificity may not be possible without sacrificing the sensitivity of the assay to the point that the test becomes worthless. A common approach to increasing the specificity of the testing protocol and the PVPT is known as sequential testing. This concept is readily demonstrated by slaughter surveillance for lesions of tuberculosis. The detection of lesions is easily incorporated into routine carcass inspection, but it is nonspecific. Thus the carcass in which a lesion is found is then subjected to an additional set of tests, including histopathological and microbiological examination of the tissue. This sequential testing procedure greatly increases the specificity of the overall diagnostic procedure. Another example of sequential testing is the use of the caudal fold tuberculin test to screen a herd of animals for responders. All responders are then subjected to the comparative cervical test as an ancillary test. Only those animals that continue to react are classified as infected. Ideally the comparative cervical test reclassifies animals as negative that nonspecifically reacted to the bovine PPD as noted earlier; the extent to which this ideal is realized remains unknown.
Although sequential tests reduce the number of false-positives, there is the potential for reduction in sensitivity because of false-negative results on the second test. A unique approach to sequential testing that has been proposed to increase sensitivity of testing programs is the administration of the intradermal PPD test followed by a serological test. Preliminary evidence suggests that infected animals would be stimulated to increase antibody production and be more likely to test positive on the subsequent serological test (Harboe et al., 1990; Dowling and Schleehauf, 1991).
As noted earlier, deer are considered to be more susceptible to M. bovis than are cattle. The immune response of deer to M. bovis infection appears to differ somewhat from that of cattle. The tuberculin skin test may be both less sensitive (more false-negative reactions) and less specific (more false-positive reactions) when used in deer (single cervical test) than it is when used in cattle (caudal fold test). Therefore, detection and control of cervid tuberculosis is not likely to be achieved simply by applying the same procedures and technology used in cattle. Recently a combination of tests including an ELISA to detect antibodies, differential lymphocyte transformation responses to M. bovis and M. avium antigens, and assays of blood proteins (fibrinogen concentration, haptoglobin concentration, plasma viscosity), which increase in acute inflammatory reactions, have been used to provide an immunologic profile of suspect deer. This combination of tests is referred to as the blood tuberculosis (BTB) test and is used primarily in New Zealand as an ancillary test procedure for individual deer that have reacted positively to the tuberculin skin test and in herds where tuberculin skin test reactors have occurred. Ancillary testing, via the blood tuberculosis test has been reported to greatly improve the sensitivity and specificity of the diagnosis of tuberculosis in farmed deer (Griffin et al., 1992; Griffin and Cross, 1986). Unfortunately, data needed to thoroughly analyze and interpret this test were not available to the committee. If valid, the test appears to have similar potential for use in other Cervidae and in nondomestic ungulates. However, the complexity and cost of the test, combined with the fact that the test is patented and the assays have been conducted in only one laboratory in the world (Griffin et al., 1992) have restricted use and evaluation of the test in the United States. Recently, however, Texas A&M University has been licensed to run the test.
Testing Populations of Animals
Given the low probability of most animals actually having tuberculosis, combined with the occurrence of false-negative and false-positive test results, current tuberculin skin tests have considerable limitations for individual or small numbers of animals. Nonetheless, these tuberculin skin tests frequently are referred to as good “herd tests,” but this is not necessarily true. The herd-level sensitivity and specificity of a test depend on the individual level of sensitivity and specificity of the test plus the number of animals tested in a herd and the percentage of these animals that are actually infected. This may range from 0 percent in a noninfected herd to 100 percent in a completely infected herd; usually the range will be between 0 and 30 percent (Martin et al., 1992).
Often what is meant when the procedure is referred to as a good herd test is that the herd sensitivity will be improved over the individual level sensitivity. That is, the ability to declare a herd with at least one infected animal “diseased” is assumed to be greater than the ability to declare an individual infected animal as “diseased.” However, this may not be a valid assumption. As an example, given a test sensitivity of 84 percent and a specificity of 96 percent, the herd sensitivity does not exceed 84 percent, with 10 percent of the herd infected, unless at least 15 animals are tested (see Table 2-2). Fortunately, this would be a common scenario when testing most herds in the United
TABLE 2-2 Herd-Level Performance of Testing Regimes by Individual Sensitivity and Specificity and Number Tested
|
Test Sensitivity 84 percent |
Test Sensitivity 61 percent |
|||||
|
Test Specificity 96 percent |
Test Specificity 99.8 percent |
|||||
|
Number Tested |
Herd Sensa |
Herd Specb |
Pred Valuec |
Herd Sensa |
Herd Specb |
Pred Valuec |
|
2 |
0.23 |
0.92 |
0.13 |
0.12 |
0.99 |
0.62 |
|
5 |
0.47 |
0.82 |
0.12 |
0.28 |
0.99 |
0.59 |
|
10 |
0.72 |
0.67 |
0.10 |
0.48 |
0.98 |
0.56 |
|
15 |
0.85 |
0.54 |
0.09 |
0.62 |
0.97 |
0.53 |
|
20 |
0.92 |
0.44 |
0.08 |
0.73 |
0.96 |
0.49 |
|
40 |
0.99 |
0.20 |
0.06 |
0.93 |
0.92 |
0.39 |
|
100 |
1.00 |
0.017 |
0.05 |
1.00 |
0.82 |
0.23 |
|
500 |
1.00 |
0.00 |
0.05 |
1.00 |
0.34 |
0.08 |
|
NOTE: Assumed within-herd prevalence of infection 10 percent. aHerd Sens = probability of an infected herd having at least one reactor to the test. bHerd Spec = probability that a non-infected herd will have no animals denoted as reactors to the test. cPred Value = predictive value of a positive test. |
||||||
States. The drawback to the reliance on testing a large number of animals to improve the sensitivity of the herd test is that as the number tested increases, the chance of a false-positive test result also increases--thus the herd-level specificity decreases. In this example the herd specificity is only 54 percent, whereas the individual level specificity is 96 percent (Table 2-2). With this herd-level specificity, if only 5 percent of herds actually were infected, only 30 percent of herds with positive reactors would contain infected animals. Of course in the United States, the level of herd infection is less than 1 per 10,000 so the predictive value at the herd level would be very low. Besides cost, this was a major reason for abandoning routine whole-herd testing and shifting to an increased reliance on slaughterhouse surveillance.
Although one never knows, in advance, the true state of nature, with respect to tuberculosis, the following approach can prove useful (Sackett et al., 1985). Three different scenarios can be considered, relating to the probability of tuberculosis in a herd, in which testing is carried out: first, suspect herds (for example, herds receiving animals from known infected herds) in which the pretest likelihood of infection is high (perhaps 50 percent); second, “in-contact” herds in which the pretest probability of infection is relatively low (perhaps 5 percent); third, area or annual certified herd testing in which the pretest probability of infection might be very low (perhaps 0.01 percent). The
predictive values could then be modeled in each of these situations: in the first two scenarios, only the caudal fold test would be used (sensitivity = 82 percent; specificity = 96 percent) to identify reactors; in the third, the animals that tested positive with the caudal fold test would be retested by the comparative cervical test to establish their reactor status, thus changing the performance characteristics of the testing procedure (combined sensitivity = 61 percent; specificity = 99.8 percent).
The above discussion focuses on the performance of the disease detection processes. It may prove instructive to examine how this might relate to the ability of the testing process to “clean-up” infected herds, particularly in view of the historical evidence that the process appeared to work better in small herds but appears to be failing, currently, in large herds. According to the Reed-Frost model of herd immunity (Martin et al., 1987), the spread of infection/disease in a population is a function of the number of infective cases, the number of susceptible animals, and the adequate contact rate between infected and susceptible animals in the population. As any one, or combination, of these factors increases in value, so does the rapidity and extent of spread of infection (see Table 2-3 comparing the total cases and shape of the epidemic curve--number of cases by time period--across population sizes from 50 to 300 animals).
The model indicates that for any rate of transmission (chiefly by respiratory route in bovine tuberculosis and modeled as “adequate contact rate”), the infection spread greatly depends on population size; larger herds have a greater probability of allowing the infection to persist and cause an “epidemic.” Further, the rate of spread and extent of infection in a large herd can be much greater than in a small herd, given the same adequate contact rate.
Obviously, one way to decrease the spread is to identify infected animals and remove them from the population as is done in a test and slaughter program. The ability of the process to detect infection depends on herd size, sensitivity of tests, nature of infection, and management factors. For example, the model predicts that a test having a sensitivity of 65 percent will clear infection from a small herd (N = 50), but not from a larger herd (N > 100); a test with sensitivity of 85 percent will also fail to clear a herd of 300 animals. As modeled here (Table 2-3), the infection persists for a protracted period at a low level. Although the Reed-Frost model is simplistic and should not be used to predict outcomes in specific herds, the findings of this model have been shown to be of general value in a variety of diseases. Whether this scenario accurately explains the current tuberculosis situation in the El Paso area is unknown, but this and other herd-level phenomena should not be ignored. Clearly, ongoing detailed epidemiologic investigations will be required to clarify the situation in any event. Some of the newer technologies for identifying and tracing microorganisms also should prove helpful in solving this problem.
Improved Diagnostic Methods
Rapid advances in molecular biological techniques for disease detection have been focused on the M. tuberculosis complex due to concerns over the increased incidence of human tuberculosis in recent years and the resulting infusion of research dollars. The new and improved diagnostic methods discussed below have also been used to study M. bovis, but many problems are yet to be resolved before practical application to field detection of M. bovis is possible.
TABLE 2-3 Number of Cases by Sensitivity of Test (Removal Process) and Population (Herd) Size
Culture
Culture methods using improved formulations of media have been shown to be as reliable as, or more reliable than, animal inoculation, although there is still reluctance to yield this traditional practice to less costly, safer, and reduced animal-use procedures (Pallen, 1987; Neill et al., 1988; Martin et al., 1989; Ovdienko et al., 1990). For example, mycobacteria in cultures can now be detected much earlier using a radiometric detection system (Heifets, 1986; Crawford et al., 1989; Roberts et al., 1991). This has significantly improved the recovery rates and decreased the time for isolation of M. tuberculosis and M. bovis from clinical specimens by one-half (Neill et al., 1986; Anargyros et al., 1990). The organism can be
speciated by performing a battery of radiometric growth-detection tests (Collins and Levett, 1989) or conventional identification tests, or by using some of the new identification technologies described below.
Other approaches used to shorten the time necessary to isolate and identify M. bovis include chemical and antigenic identification of the organism. The chemical structure of the organism can be analyzed to recognize unique lipid or carbohydrate components (Jarnagin et al., 1983; Jarnagin and Colgrove, 1986; Jantzen et al., 1989; Sisson et al., 1991) or proteins (De Jong et al., 1991). Monoclonal antibodies have been produced that are specific for M. bovis (Corner et al., 1988; Androsova, et al., 1989; Veerman et al., 1990; Verstijnen et al., 1991). These methods can also be modified to detect the unique structural chemicals or antigens of M. bovis directly in tissue specimens. Preliminary studies suggest that these approaches have promise (Humphrey and Weiner, 1987; Cambiaso et al., 1990; Hara et al., 1991; Maliwan et al., 1991); however, an amplification step may be essential to improve the sensitivity of the assay.
In 1987 nucleic acid probes for the identification of the M. tuberculosis complex, including M. bovis, became commercially available (Gonzalez and Hanna, 1987). They have also been applied to the identification of Mycobacterium sp. (Crawford et al., 1989). These probes were for the specific identification of mycobacteria isolated in culture. Probe technology also requires amplification of the signal nucleic acid to provide sufficient mass for reaction with the probe. Originally, the amplification step consisted of growing the organism on a medium to produce colonies. As probe technology has been refined, probes have been applied to enrichment cultures for rapid identification of mycobacterial growth as well as the accurate identification of species (Ellner et al., 1988; Peterson et al., 1989; Kusunoki et al., 1991). In addition, the probes carry a radioactive marker that further enhances detection of weak signals. Recently, the polymerase chain reaction (PCR) has been used to amplify DNA for probe detection (Boddinghaus et al., 1990; Sjobring et al., 1990; Cousins et al., 1991, 1992; Fiss et al., 1992). This remarkable advancement has provided the means to directly detect mycobacteria in clinical specimens (De Wit et al., 1990; Eisenach et al., 1991; Manjunath et al., 1991; Pierre et al., 1991; Altamirano et al., 1992; van der Giessen et al., 1992).
Standardization of procedures and consistent sensitivity in clinical specimens are still the major challenges for routine use of probes. Most probes are highly specific for the M. tuberculosis complex of organisms, which includes M. bovis (Wayne and Kubica, 1986). In several of the above cited studies, M. bovis was accurately identified within the complex. Del Portillo and colleagues (1991) reported the discovery of a specific DNA fragment that is present in M. tuberculosis but not M. bovis that will allow rapid differentiation of these organisms.
Present limitations to the implementation of probe diagnostic tests on a routine basis include the cost of equipment and reagents, extensive laboratory personnel services and expertise, use of radioisotope labels on the probes, and problems with cross-contamination between samples or within the laboratory that will produce false-positive results. Nucleic acid probes have recently been developed with detection systems that do not include a radioactive label, so they can be incorporated into laboratory procedures more readily (Goto et al., 1991; Lim et al., 1991; Evans et al., 1992; Lebrun et al., 1992).
Although the direct detection of microbial components and products is revolutionizing the field of microbiological diagnosis, it has not displaced the need for isolation of the organism for epidemiological purposes. Detailed subspecies typing provides a “fingerprint” of the organism of interest. Repeated isolation of organisms with the same fingerprint from different animals suggests that the organism may have originated from a single clone. Such findings may indicate transmission from a common source or by a common mechanism. The ability to identify and trace the spread of specific strains is an important aid in the rational development of effective measures to control and prevent transmission of M. bovis. Conventional, phenotypic typing methods that have been applied to Mycobacterium sp. include antimicrobial susceptibility profiles, biochemical profiles, serological typing, and bacteriophage susceptibility patterns (Barrow, 1986; Roberts et al., 1991; Ross et al., 1991). Distinct serotype differences between field isolates ofM. bovis have not been reported (Morris and Ivanyi, 1985). The discovery of mobile genetic elements in strains of M. tuberculosis complex has resulted in a rapid method for typing M. tuberculosis and M. bovis isolates using a combination of restriction endonuclease analysis and pulsed-field electrophoresis (Shoemaker
et al., 1986; Mazurek et al., 1991; van Soolingen et al., 1991; Zhang et al., 1992). At least 33 unique strains of M. bovis from outside of North America have been differentiated using restriction endonuclease methods (Collins and de Lisle, 1985; Collins et al., 1986, 1988; de Lisle et al., 1990; Collins and Stephens, 1991).
DNA-based typing methods are relatively rapid and simple to perform, highly reproducible, stable over time, capable of identifying a large number of polymorphisms, and valid in terms of distinguishing epidemiologically related versus unrelated isolates. Despite these many advantages, several disadvantages remain that must be addressed. These include variations in the final profile caused by problems with DNA extraction, differences in the conditions of electrophoresis, difficulties in analyzing complex DNA patterns, and lack of standardization with respect to methodology and reference strains.
New Cell-Mediated Immunity-Based Tests
The lymphocyte transformation (LT) test is a promising in vitro correlate of delayed-type hypersensitivity (DTH) (Outteridge and Lepper, 1973; Muscoplat et al., 1974, 1978). The LT test, also known as lymphocyte blastogenic test or lymphocyte stimulation test, is performed by collecting a sample of heparinized blood from the animal. The blood is transported to the laboratory and the lymphocytes separated out by differential centrifugation. The enriched mononuclear cell fraction is placed in tissue culture and challenged with a battery of antigens. After 3 to 5 days incubation, the cultures are pulsed with a radioisotope-labeled nucleotide. After further incubation, the cells are harvested and the incorporation of the radioisotope-labeled nucleotide is determined. Incorporation of the marker reflects rapid proliferation of cells known as blastogenesis. Lymphocytes in the control cultures remain at a resting metabolic rate throughout the incubation period. In cultures stimulated with antigen, however, those lymphocytes that have previously been primed and differentiated to recognize the antigen will begin proliferating in response to the presence of the antigen. Recent advances in immunology such as T-cell cloning and identification of T-cell receptors are providing the means to identify specific T-cell antigens and may make the LT and related laboratory tests of T-cell activity more promising in the future (Billman-Jacobe et al., 1990).
When lymphocytes recognize and respond to antigen, they begin producing a number of intercellular messenger molecules known as lymphokines in addition to undergoing blastogenesis. To differentiate nonspecific blastogenesis from antigen specific stimulation, the detection of specific messenger lymphokines may be a more accurate in vitro test of CMI responses. The release of bovine γ-IFN as a measure of CMI responsiveness to specific antigen has been shown to correlate with the conventional lymphocyte transformation assay. This approach has been exploited in the development of a new bovine tuberculosis diagnostic test (Wood et al., 1990).
The γ-IFN test developed and tested in Australia, however, did not appear to perform better than the skin test; although this technology was cited as a potential aid to eradication in the Australian Program Review. Despite its supposed benefit of one-time handling, herds with reactors had to be mustered again to identify the positive reactors. The γ-IFN test has been used in pilot studies in the El Paso milkshed; preliminary indications are that it performs similarly to the skin tests in terms of sensitivity and specificity (U.S. Department of Agriculture, 1992).
A sandwich ELISA using monoclonal antibodies for the specific detection of bovine γ-IFN was developed to detect the release of γ-IFN in response to PPD stimulation in a whole-blood culture (Rothel et al., 1990). A drawback is that this test requires the collection of a blood sample that must be handled in a manner that maintains the viability and functional capacity of the lymphocytes. The sample must be carefully and quickly transported to the laboratory and divided into multiple aliquots; antigen and nonantigen control solutions are then added to the cultures and incubated for 24 hours. Thus there must be careful coordination between the field sample collection crew and laboratory personnel to accommodate the requirements of this test.
Suitable data are not available to determine whether the various assays of CMI responses, including intradermal tuberculin tests, LT tests, and γ-IFN tests, are capable of detecting different subpopulations of infected animals or different stages of the disease.
New Serologic Tests
Mycobacterium bovis specific epitopes can be identified through the use of monoclonal antibodies and immunoblotting techniques (Fifis et al., 1989, 1991; Billman-Jacobe et al., 1990; Radford et al., 1990). Specific epitopes can be mass produced through cloning technology (Radford et al., 1988) or synthesis of the epitope (Brennan, 1989). Presently, research activities are focusing on a variety of mycobacterial proteins (Haslov et al., 1987; Radford et al., 1988, 1990; Billman-Jacobe et al., 1990, Fifis et al., 1989, 1991) and glycolipid antigens (Chatterjee et al., 1989; Sada et al., 1990; VanTeim, 1992). VanTeim (1992), using an ELISA test, conducted a preliminary study using a panel of antigens, including bovine PPD, crude culture filtrate, lipoarabinomannan, and MPB70 to develop a decision model for diagnosis of bovine tuberculosis in cattle, elk, and llamas.
Other promising approaches to serological diagnosis include the development of competitive ELISA tests using monoclonal antibodies (Wilkins et al., 1991) and latex agglutination tests (Wu et al, 1990) that are specific for M. bovis epitopes.
VACCINATION
Under some circumstances the immune response can protect the recovered individual from reinfection. Protective immunity can be stimulated by vaccines and extends beyond the species of mycobacteria contained in the vaccines. The live attenuated strain of M. bovis called bacillus Calmette-Guèrin (BCG) used as a vaccine in children can increase their resistance to M. tuberculosis and M. leprae (leprosy) infection. Live vaccines are more effective than killed vaccines. The safety of the live BCG vaccine for humans is well documented and generally accepted. The overall efficacy of BCG vaccination in controlling human tuberculosis, however, is controversial for two reasons. First, field trials have repeatedly demonstrated that while it protects in some populations, it does not protect in others. Second, the vaccine-induced DTH causes vaccinated people to react positively to the tuberculin skin test used to detect tuberculosis. Thus vaccination reduces the value of the tuberculin skin test in control programs. For these reasons, BCG is not used in the United States.
BCG vaccination does not provide a useful or cost-effective level of protection in cattle. There have been numerous attempts to develop safe, practical, cost-effective, live or killed vaccines to protect cattle against M. bovis infection (Lepper and Corner, 1983), but none has been successful. In theory, vaccines could be developed that protect but do not interfere with the diagnostic tests used for detection of infection. Such vaccines would be strategically useful in the final stages of eradication and in responding to reintroduction of the disease after it has been eradicated from the United States. For example, the vaccine could be used in exposed cattle in herds around an outbreak (ring vaccination). It could be used within infected herds where financial or legal constraints necessitated repeated test and slaughter rather than complete depopulation. It could be used to enhance the value of U.S. seedstock exported to regions of the world with a high incidence of bovine tuberculosis, and it could also be used in farmed bison and cervids, endangered ungulates in zoos, or infected herds of free-roaming wildlife. Rational development of such a vaccine would involve determining which antigens of M. bovis are required for protective immunity. This knowledge would allow construction of vaccines that contain optimal combinations and amounts of the protective antigens. Marker deletion vaccines could be constructed that would contain the protective antigens but lack one or more antigens recognized by the bovine immune system during M. bovis infection and not required for protective immunity. Diagnostic tests based on antigens deleted from the vaccine would allow differentiation between immune responses generated by vaccination and infection; thus vaccination and eradication programs could proceed simultaneously.
TREATMENT OF TUBERCULOSIS
In humans treatment of pulmonary tuberculosis requires the use of multiple antimicrobial drugs for prolonged periods (6 to 12 months). In populations with a large percentage of drug resistant infections, concurrent treatment with four drugs is recommended (Hoeprich, 1977; Berkow, 1987; Centers for Disease Control, 1993b).
Within the past 5 years an increased frequency of M. tuberculosis strains resistant to one or more antimicrobial drugs has been reported. Poor patient compliance with drug treatment regimens, especially among homeless people, drug addicts, and others living in poverty, are considered largely responsible for the emergence of the drug resistant strains (Culliton, 1992). Drug resistant M. bovis isolates have rarely been reported. Although drug resistant strains are not likely to emerge from farm animals because they are not treated for tuberculosis, the treatment of animals in zoological parks and humans infected with M. bovis could provide sufficient selective pressure for the emergence of resistant strains.
Successful treatment in humans with newer antimicrobial drug combinations offers some hope that similar success might be achieved in treating valuable or endangered animals, but research to develop suitable therapeutic regimens will be needed. Chemotherapy is warranted in valuable and endangered captive wild animals infected with tuberculosis. It has been successful when multiple drug therapy was used for 9 to 12 months (L. Durran, unpublished data, 1989; Fowler, 1978; Bush et al., 1990). The chemotherapeutic agents and treatment regimes that are effective in some humans have been shown to be effective in cattle (Konyha, 1986; Konyha et al., 1980; Timoney et al., 1988).
The treatment of tuberculosis in cattle is not cost effective because it requires prolonged periods (4 to 12 months) of daily dosing. An additional drawback in therapy is the possibility of relapse after removal from the treatment regimen. Recrudescence of active infection in previously treated animals has been reported in many species including humans (Kleeburg et al., 1966; Wolf et al., 1988; Fowler, 1978).
In addition, a treatment strategy for food animals must consider the problem of drug residues in meat, milk, and other consumed products (such as medicinals prepared from velvet antlers); therefore, a treatment strategy is contraindicated from a public health standpoint. The treatment of M. bovis infection in cattle and other farmed animals is not recommended, because the selective pressure of treatment could enhance the emergence of antimicrobial resistant strains of M. bovis which would be unresponsive to treatment should they be transmitted to humans.