3
MODIFICATIONS OF THE SPENT FUEL PROCESSING FLOW SHEET
As noted in Chapter 2, the basic unit operations for electrometallurgical processing of spent nuclear fuel are the same as those for the IFR technology. Some changes were incorporated to allow the introduction of feed streams other than metallic fuel, and further modifications of the flow sheet have been proposed for the addition of excess weapons plutonium as a potential feed stream.
The essential steps of the fuel processing flow sheet are:
-
spent fuel element chopping,
-
lithium reduction (for fuel other than metallic)/lithium regeneration,
-
electrorefining,
-
disposition of uranium metal product, and
-
disposition of chloride waste streams.
This is the basic flow sheet that was the focus of the ANL processing development activities under the IFR program. The lithium reduction step has been incorporated to allow application of the technology to a broader range of fuels.
The head-end fuel pin chopping operations have been demonstrated for a variety of fuels. The lithium reduction of oxides and the associated regeneration of lithium from the by-product lithium oxide have been less well demonstrated. ANL has performed experiments that indicate the feasibility of the lithium reduction technology, including the reduction of the oxides of uranium and plutonium. However, the inclusion of this step to allow the processing of non-metallic fuels introduces additional quantities of salt that must be maintained in a molten state and transferred between reaction vessels. Further complications may arise from incomplete reduction of oxides or solubility problems with some of the reduced chemical species. The solubility of the by-product lithium oxide in the lithium chloride flux has been established, and the planned throughput requires maintaining the lithium oxide content of the salt well below the solubility limit. The large differences in the densities and melting points of the metallic lithium and the salt flux may lead to problems in transporting the mixture and in recovering the elemental lithium for reuse. This step in the flow sheet needs to be fully demonstrated.
Considerable research and development have been done on the electrorefining aspect of the proposed technology. Electrorefining has been demonstrated at laboratory scale (10 kg heavy metal/day), but in the absence of significant levels of radioactivity. The next step of scale-up to production-size equipment and throughputs (200 kg heavy metal/day) has yet to be fully demonstrated. The collection of the uranium product on a steel cathode has been carried out. Whether this uranium product is recycled for further energy production, denatured with natural uranium to a lower enrichment, or stored pending disposition decisions, the technology for separating it from the parent feed appears to be viable.
The recovery and disposal of the waste streams, which include the metallic fuel cladding and the fission products, take two paths. The metallic constituents can be separated easily and disposed of as solid waste in either the metallic or a less reactive oxidized form. A clean metal/salt separation is required for any metals coming from the electrorefiner. If any entrained chloride salt were present, its high vapor pressure at the elevated process temperatures (1000 to 1400°C) could result in fouling of equipment and difficult process operation. The zeolite sorption portion of the flow sheet is discussed in Chapter 4, “Waste Forms and Characteristics.”
For possible electrometallurgical treatment of plutonium, the flow sheet preferred by ANL contains only three effluent streams and is essentially the one proposed for treating DOE spent nuclear fuels (see Figure 2 in Chapter 2).
These streams include (1) the relatively pure uranium metal fraction; 1 (2) miscellaneous metal wastes containing the noble metal fission products, cladding, and reactor assembly hardware; and (3) zeolites containing the transuranics and the alkali metal, alkaline earth, and rare-earth and other fission products. In the ANL process, the zeolites would be converted to a glass-bonded zeolite waste form, and the miscellaneous metals would be formed into ingots of a corrosion-resistant metal waste form (a zirconium-iron alloy). ANL believes that both waste forms would be suitable for disposal in a geologic repository. However, ANL is also investigating an alternative flow sheet (Fig. 3) in which the transuranic elements in the spent fuels would be combined with the undissolved material remaining in the anode baskets and incorporated in the Fe/Zr metallic waste form.
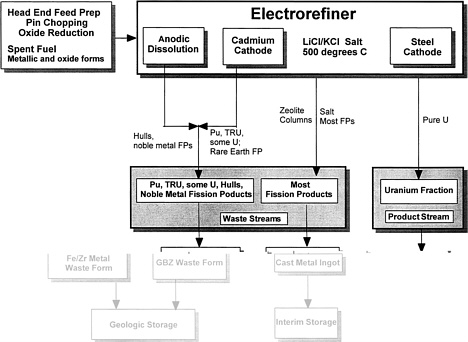
Figure 3. Alternative ANL electrometallurgical process scheme to generate a single metallic waste form. In adaptation for treatment of plutonium, the plutonium would be introduced at the point denoted spent fuel, metallic and oxide forms.
With respect to the disposition of surplus WPu, the primary features of the electrometallurgical spent fuel treatment process that are applicable to immobilization are:
-
the Li reduction step for the treatment of TRU-rich oxide residue;
|
1 |
The uranium metal produced in the electrorefining step has up to the present time been designated as a product to be stored, but not as a waste. Its final disposition remains to be decided. |
-
electrorefining of metallic fuel for the separation of a uranium fraction, and either individual TRU and fission product fractions, or a single TRU-fission product fraction;
-
incorporation of the fission products in the GBZ waste form; and
-
casting of the metal waste forms. If a molten cadmium cathode were to be used in the electrorefiner, the transuranic elements could be collected and disposed of in the Fe/Zr metal waste form (see Figure 3). Choosing to allow the transuranic elements to accumulate with most of the fission products in the salt would permit their removal from the salt by ion exchange with anhydrous zeolite and subsequent stabilization in the GBZ waste form (see Figure 2).
For the application of the electrometallurgical treatment technology to plutonium disposition, ANL proposes that the addition of CsCl from the Hanford capsules would provide most of the radiation field to create the radiation barrier, allowing the waste forms to meet the “spent fuel standard.” For the treatment of the large majority of surplus WPu, which is in the form of plutonium metal pits, ANL has proposed converting the metal to the hydride and then chlorinating the hydride in the electrorefiner vessel.2 That part of the plutonium inventory already in the chloride form could also be added directly to the electrorefiner vessel. The dissolution of the CsCl could likewise be carried out in the electrorefiner vessel. The resulting molten salt solution of plutonium trichloride and CsCl, which would also contain some rare-earth fission products from the spent fuel processing, could be fed directly to the zeolite sorption step followed by GBZ waste form fabrication. Plutonium-rich oxides, if they do not contain materials harmful to the electrorefining process, such as magnesium oxide and silicon dioxide, could be sent directly to the lithium reduction step.
Much of the proposed process shown in the flow sheet has reasonable data to support it, but additional R&D is needed. The regeneration of lithium metal from lithium oxide produced during the reduction of oxide fuels is not well demonstrated. The large differences in the densities and melting points of the metallic lithium and the salt flux may lead to problems in transporting the mixture and in recovering the elemental lithium for reuse. This portion of the process flow sheet needs to be fully demonstrated with simulated fuel as part of developing the step for reduction of nonmetallic plutonium. The technology for separating uranium from the parent feed appears to be viable and would allow the uranium to be recycled for further energy production, denatured with natural uranium to a lower enrichment, or stored pending disposition decisions. If any entrained chloride salt were present, its high vapor pressure at the elevated process temperatures (1000 to 1400°C) could result in fouling of equipment and difficult process operation.
PLUTONIUM DISPOSITION FLOW SHEET OPTION
ANL has indicated that “the plutonium immobilization process is visualized as being conducted in the same facilities, in the same electrorefiner, and at the same time as the spent fuel treatment” (Appendix B, p. 39). In order to incorporate the disposal of excess weapons plutonium into the flow sheet for spent fuel treatment, several additions and modifications have been made (see Figure 3). Because of the attractiveness of WPu as a target for terrorist use or other diversion to weapons manufacture, the use of spent fuel, or some radiological equivalent, has been deemed necessary to meet CISAC's “spent fuel standard” for the final waste form. A number of combinations of feeds and flow sheets have been proposed that would meet the spent fuel criteria.
Plutonium metal, either from weapons components or other sources, is compatible with the electrometallurgical technique. The process is designed for metallic feed, and all of the development work
|
2 |
If the spent fuel process were not available, this would become a “stand-alone” chlorination step. |
has been aimed at separation of various constituents from metal feed stock. If plutonium is to be added to the feed stream as metal, the proposed flow sheet shows it going through the electrorefiner. The pretreatment for removal of plutonium from weapons components would consist of hydriding followed by either dehydriding or chlorination. The hydriding step would eliminate security issues associated with weapons pits, including their classified shape. If the hydride were converted to metal, the resulting metallic plutonium would be fed directly into the electrorefiner. If it were chlorinated, the plutonium chloride could be fed directly to the salt blend tank prior to sorption on zeolite.
Because plutonium is more reactive than uranium, molten plutonium metal would reduce the uranium chloride. In the electrorefiner, the resulting plutonium chloride would be dissolved in the salt blanket and could be incorporated ultimately into the zeolite. Because the salt mixture in the electrorefiner would be continually enriched in plutonium as the uranium was depleted, either through reaction with the plutonium or reduction and collection of uranium onto the cathode, a bleed stream would be needed to keep the plutonium concentrations relatively low. The current plans are to remove all or part of the salt when the plutonium concentration reaches 12 to 15 wt. percent. 3 The proposed feed rate of weapons plutonium is approximately 20 kg/day. However, the EBR-II fuel would contribute approximately 11 kg/day of plutonium at the proposed fuel processing rates.
Another source of plutonium metal to be fed into the electrorefiner would be the lithium reduction of plutonium compounds. This process is similar to the reduction of oxide fuels. However, there are important differences between oxide fuel and the plutonium compounds available from DOE waste holdings. Fuel can be reasonably well characterized by determining the burn-up and the physics of the reactor core. Unfortunately, this is not the case for the oxide materials such as ash (the resultant residue of combustible waste contaminated with Pu), low-assay plutonium oxide, and other DOE materials. The poorly characterized plutonium compounds would require significant pretreatment before they would be suitable for lithium reduction. For waste such as ash, the pretreatment required to make the resulting material compatible with the lithium reduction process has not been defined. Large amounts of impurities such as silica, magnesia, and many other materials are present in some of the DOE material. The effects of these impurities must be evaluated if plutonium oxides and oxide-like materials are to be considered as potential feed materials. Note that processing would be necessary to convert impure oxide, ashes, and salts to a form appropriate for vitrification or use in MOX fuel.
An option has also been presented by ANL for adding plutonium trichloride into the salt as it is fed into the zeolite ion exchange step shown on the flow sheet. The PuCl3 would come either from the conversion of plutonium metal in weapons pits via hydriding followed by chlorination, or from the direct addition of plutonium chloride from other waste forms such as pyrochemical processing salts from other DOE sites. Most of the pyrochemical salts in the DOE holdings are composed of either a NaCl/KCl eutectic matrix with a relatively high assay of actinides, or CaCl2 with relatively small amounts of dispersed metallic plutonium and some plutonium oxide. The proposed process would introduce this additional salt feed stream via a salt blending tank. This would require including in the pretreatment some type of chlorination operations to assure that all of the actinides in the salt are present as chlorides. If the plutonium were to be introduced in the form of salts, additional development work would be needed to determine the effects of the salt matrices on the KCl/LiCl salt used in the processing operations.
All of these scenarios are predicated on the concurrent processing of spent fuel, which would provide the requisite radiation barrier. However, there is insufficient EBR-II fuel to provide the fission products to achieve the “spent fuel standard” for the majority of the excess weapons plutonium. Therefore, ANL has proposed that the electrochemical treatment option be significantly expanded such that a variety of types of
|
3 |
The flow sheets presented by ANL when its representatives briefed the committee appear to indicate that the Pu content of the salt from the electrorefiner would be closer to 22 wt. percent. |
fuels could be processed. ANL has suggested an additional scenario that does not depend solely on spent fuel to provide the radiation barrier. The use of radioactive cesium that has been stockpiled by DOE could allow the electrorefining portion of the flow sheet to be bypassed, although the process equipment would be employed. Cesium chloride would be added to the salt, which would then be contacted with the zeolite. This option requires that all of the plutonium be introduced into the process in the chloride form, and pretreatment would be needed to assure that all of the plutonium has been chlorinated.
Overall, the use of the entire suite of operations in the electrometallurgical technique for the disposition of excess plutonium appears to be dependent on the processing of spent fuel. The other options that have been advanced do not seem to be consistent with the equipment or the process conditions that exist in the present processing flow sheet (see Figure 2 in Chapter 2).
MODIFIED FLOW SHEET: CONCLUSIONS
-
Metallic plutonium from weapons components is compatible with the several flow sheets for electrometallurgical processing.
-
Significant pretreatment would be required for all forms of plutonium other than metal from weapons components before the material would be suitable for further electrometallurgical processing.
-
Interactions between the electrometallurgical process salt and non-metallic plutonium feed streams, specifically pyrochemical salts, might adversely affect electrometallurgical processing and/or waste form performance.
-
The lithium reduction/regeneration portion of the proposed process has not been adequately demonstrated on the different fuels to allow adoption as a pretreatment step for plutonium disposition.
MODIFIED FLOW SHEET: RECOMMENDATIONS
-
Pretreatment requirements for the nonmetal plutonium feed streams should be determined and, if possible, R&D should be started to validate the treatment and subsequent compatibility with the electrometallurgical process.
-
The effects of major impurities such as additional salts (NaCl/KCl and CaCl2) and other impurities such as Si, Mg, and C on the performance of the electrometallurgical treatment operations should be evaluated.





