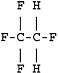3
Exposure Guidance Levels for Hydrofluorocarbon-134a
INTRODUCTION
Hydrofluorocarbon (HFC)-134a or 1,1,1,2-tetrafluoroethane is a gaseous halocarbon that is being considered as a prime candidate for replacing other halocarbon materials, such as Freon 12 (dichlorodifluoromethane) and Freon 22, for use in air conditioning and refrigeration systems and possibly as an aerosol propellant or foam expansion agent. Due to a federal regulation that mandates switching from CFCs to other suitable compounds that either do not damage the ozone layer or damage the ozone layer less than CFCs, the U.S. Navy requested that the NRC review the toxicity data on HFC-134a and recommend 1-hr and 24-hr EEGLs. The Navy also requested that the NRC recommend a 90-day CEGL for HFC-134a and identify appropriate research to fill data gaps. The remainder of this chapter consists of the supporting documentation assembled and evaluated by the subcommittee in support of its recommendations.
BACKGROUND INFORMATION
Physical and Chemical Properties
Occurrence and Use
The major uses of HFC-134a are in mobile air conditioning and
refrigeration; it may also be used as a propellant in medications, such as anti-asthmatic preparations (Olson et al., 1990).
TOXICOKINETICS
Absorption of fluorocarbons and bromofluorocarbons via the inhalation route is rapid; the maximal blood concentrations of the substances develop within 5 min and equilibrium is achieved within the next 15 min of exposure (Azar et al., 1973; Trochimowicz et al., 1974; Mullin et al., 1979). Blood concentrations do not increase further with increasing durations of exposure for a given concentration of these substances. The time course of absorption of HFC-134a via the inhalation route is likely to be similar.
Toxic effects observed in animals following oral and inhalation exposures to HFC-134a indicate that it is absorbed by the lungs and gastrointestinal (GI) tract (Salmon et al., 1980). Studies conducted in rats exposed to high concentrations of HFC-134a, either orally or via inhalation, indicate that it is rapidly excreted, mostly as the unchanged parent compound (Salmon et al., 1980). Analysis of the urine, feces, and expired air of rats exposed to HFC-134a at 10,000 ppm (1.0%) of HFC-134a for 1 hr showed that only 0.34-0.40% was metabolized. This study also provides evidence that some HFC-134a is retained in the liver and that relatively high amounts are retained in the adrenal gland.
Studies with rat-liver microsomes show that HFC-134a is oxidized by the cytochrome-P-450 system; that implies that cytochrome-P-450-containing tissues, such as nasal mucosa, liver, and lungs, might convert HFC-134a to trifluoracetic acid, a toxic metabolite (Olson et al., 1990).
TOXICITY INFORMATION
No toxicity data are available on humans following exposure to
HFC-134a. Animal studies indicate that HFC-134a has a low level of systemic toxicity following acute, subacute, subchronic, and chronic exposures. For example, neurotoxicity and cardiac sensitization occur after acute exposures to HCF-134a at high concentrations. HFC-134a at high concentrations appears to be a developmental toxicant (slight delays in skeletal ossification and lower body weights). The toxicity of HFC-134a in animals is discussed in more detail in the following discussion.
Acute Toxicity
HFC-134a has low acute toxicity via the inhalation route. Its approximate lethal concentration (ALC) in rats is 567,000 ppm following 4 hr of inhalation exposure. Silber and Kennedy (1979a) exposed six groups of rats (each group containing six animals) to HFC-134a at concentrations ranging from approximately 80,000 ppm to 653,000 ppm. Clinical signs of toxicity included lethargy, labored and rapid respiration, foaming at the nose, tearing, salivation, and weight loss.
Rissolo and Zapp (1967) exposed rats to HFC-134a at concentrations of 750,000 ppm for 30 min. Two of four animals died. During exposure to the chemical, animals showed incoordination, pumping respiration, darkening of the eyes, unresponsiveness, cyanosis, convulsions, and death. Upon removal from exposure to HFC-134a, the surviving animals became coordinate within 5 min and appeared normal. Pulmonary congestion and edema were observed during necropsy of the two rats who died during exposure. Surviving rats were necropsied 14 days after exposure and showed no abnormalities.
In summary, HFC-134a has very low acute toxicity. Lethal concentrations of HFC-134a range from 567,000 to 750,000 ppm in rats. Table 3-1 summarizes the acute toxicity information for HFC-134a.
TABLE 3-1 Acute Lethality of HFC-134a in Rats
|
Exposure Duration |
Exposure Concentration, ppm |
End Point |
Reference |
|
4 hr |
567,000 |
Approximate lethal concentration |
Siber and Kennedy, 1979a |
|
30 min |
750,000 |
2 of 4 died |
Rissolo and Zapp, 1967 |
Cardiac Sensitization
Mullin and Hartgrove (1979) evaluated the cardiac sensitization potential of HFC-134a in a standard epinephrine challenge test. Healthy male dogs were exposed to various concentrations of HFC-134a in the following manner. An intravenous control injection of epinephrine (8 µg/kg) was given before exposure. Following administration of the injection, the animals were exposed via inhalation for 5 min to HFC-134a at one of the following concentrations: 50,000, 75,000, or 100,000 ppm. A challenge dose of epinephrine (same as the pretest dose) was given immediately after cessation of exposure. Heart rate and wave dynamics were monitored with an electrocardiogram throughout the experiment. A “marked response” was scored when a life-threatening cardiac arrhythmia developed (multiple consecutive ventricular beats) or when ventricular fibrillation occurred and ended in cardiac arrest.
No dogs exhibited “marked responses” (life-threatening cardiac arrhythmias such as multiple ventricular beats or ventricular fibrillation) at exposures of 50,000 ppm. Two of 10 dogs exhibited multiple ventricular beats upon exposure at 75,000 ppm of the test compound. Two of 4 dogs exposed at 100,000 ppm showed marked responses, one with multiple consecutive ventricular beats, the other with ventricular fibrillation leading to cardiac arrest.
In summary, Mullin and Hartgrove's (1979) results indicate that HFC-134a is a weak cardiac sensitizer when tested in an epi-
nephrine challenge test in dogs. Epinephrine-induced cardiac arrhythmias were seen at doses of 75,000 ppm and greater. The noobserved-adverse-effect level (NOAEL) for this response was 50,000 ppm.
Hardy et al. (1991) exposed beagle dogs to HFC-134a at concentrations of 40,000, 80,000, 160,000, and 320,000 ppm. The test involved intravenous injection of epinephrine before and during gas inhalation. The dose of epinephrine administered to each dog was adjusted to result in a few ectopic beats in the absence of the test chemical. Dogs were either administered 2, 4, or 8 µg/kg, depending on their individual sensitivity to epinephrine. Using this paradigm, no cardiac sensitization was observed in dogs at concentrations of 40,000 ppm, whereas concentrations of 80,000 ppm and higher induced cardiac sensitization. Therefore, the NOAEL for this study is 40,000 ppm.
Subacute Toxicity
Subacute toxicity studies in rats indicate that the only pathological changes following 14 days or 28 days of exposure (6 hr/day, 5 days/wk) to HFC-134a at 50,000 or 100,000 ppm were changes in the lung, indicative of focal interstitial pneumonitis (Silber and Kennedy, 1979b). Some changes in organ weights (liver, kidney, and gonads) were seen after 28 days of exposure at 50,000 ppm. Increased liver weight was also seen at an exposure concentration of 10,000 ppm in males. None of the organ-weight changes were associated with histological changes. Male albino rats exposed to HCF-134a by inhalation at approximately 100,000 ppm 10 times over a 2-week period for 6 hr/day, 5 days/wk showed no compound-related hematological, blood chemistry, pathological, and urinary changes (Silber and Kennedy, 1979b). Urinary fluoride concentrations were increased after the ninth exposure, suggesting metabolic conversion of the retained halocarbon (Silber and Kennedy, 1979b). Groups of 16 male and 16 female rats were exposed
to HCF-134a by inhalation at 1,000, 10,000, or 50,000 ppm 20 times in a 28-day period for 6 hr/day (Riley et al., 1979). Changes in liver, kidney, and gonad weights were noted; these were confined to male rats exposed at 50,000 ppm except for a liver-weight increase, which was also seen at 10,000 ppm. There were no pathological changes in these tissues, and the liver- and kidney-weight increases are considered to be due to a physiological adaptation to treatment. The reduced testicular weight is not considered to be of toxicological importance.
In summary, the only pathological change of possible relationship to treatment in these subacute tests was in the lungs of several male rats receiving 50,000 ppm. This lesion was a focal interstitial pneumonitis. The NOAEL for this study is 10,000 ppm (Silber and Kennedy, 1979b).
Subchronic Toxicity
Hext (1989) exposed groups of 20 male and 20 female Wistarderived albino rats to daily concentrations of HFC-134a at 0, 2,000, 10,000, or 49,500 ppm for 6 hr/day, 5 days/wk for 13 weeks. Half of the animals from each group were killed following termination of exposure and the remaining animals were killed following a 4-week “recovery” period.
Body-weight gains, in general, tended to be lower in exposed groups than in controls; however, the differences were relatively small and were not dose-related. A few statistically significant changes in urine, blood chemistry, and hematological values and in the weights of the liver, lung, and heart did not appear to be either dose-related or consistent with repeated sampling. A statistically significant decrease in brain weights was reported; however, this change was not dose-related and was not accompanied by either positive histological findings or by transient central-nervoussystem (CNS) depression. Similar decreases in brain weight were not reported in animals allowed to recover after 4 weeks of sub-
chronic exposures to HFC-134a at up to 49,500 ppm or in animals sacrificed following 12 months of exposure to HFC-134a (Hext, 1989). In addition, histopathological findings were negative in animals allowed to recover following 4 weeks of exposure. Thus, the weight of evidence suggests that brain-weight decreases observed in female rats might be a statistical artifact. However, it is possible that brain lesions might be present in those areas of the brain that did not undergo histopathological examination. A decrease in brain weight could be representative of a significant loss of structure and function. In addition, cell loss within the CNS is often followed by replacement with connective tissue, minimizing the likelihood of measuring a decrease in overall brain weight. More specific information from ongoing studies is needed to evaluate fully the potential neurotoxicity of this compound.
Developmental Toxicity
Three inhalation studies (two in rats and one in rabbits) were conducted in animals to examine the developmental effects of HFC-134a.
Lu and Staples (1981) exposed pregnant CD rats to HFC-134a at concentrations of 30,000, 100,000, and 300,000 ppm for 6 hr/day from days 6 to 15 of gestation. There was a significant reduction in fetal weight and significant increases in several skeletal variations following exposure of dams to HFC-134a at 300,000 ppm. Maternal toxicity was observed following exposure of dams at both 100,000 ppm (effects were reduced responses to noise stimuli and uncoordinated movements) and 300,000 ppm (effects were significant reductions in food consumption and body-weight gain, no responses to noise stimuli, severe tremors, and uncoordinated movements). No developmental effects were observed following exposure of dams at 30,000 or 100,000 ppm.
Hodge et al. (1979a) exposed a group of pregnant rats (29-30 per group) to HFC-134a at concentrations of 0, 1,000, 10,000 or
50,000 ppm for 6 hr/day on days 6-15 of gestation. No statistically significant evidence of maternal toxicity was observed following exposure of dams to concentrations of HFC-134a as high as 50,000 ppm. However, at this concentration, developmental toxicity was evident. Fetal body weight was significantly reduced, and skeletal ossification was significantly delayed.
Wickramaratne (1989a,b) exposed pregnant New Zealand rabbits (18-23 per group) to HFC-134a at concentrations of 0, 2,500, 10,000, or 40,000 ppm, 6 hr/day on days 6-18 of gestation. Does were killed on day 29, and fetuses were weighed and examined for external, internal, and skeletal abnormalities. Inhalation exposure of rabbits in the 10,000- or 40,000-ppm group resulted in statistically significant increases in the fetal incidence of unossified seventh-lumbar transverse processes. The incidence in control animals also increased with time. Therefore, the incidence was not considered a significant chemical-related effect. In addition, maternal toxicity was observed in rabbits exposed at both concentrations of HFC-134a. Specifically, there was a statistically significant reduction in food consumption and body-weight gains. There was also a dose-related increase in the incidence of gaseous distention of the stomach, which was statistically significant at 40,000 ppm. However, this increased incidence (12.7%) was only slightly outside the range for historical controls (2.3-11.5%). The reasons for the increase in gaseous distention is unclear, but it could be indicative of lung problems or nonrandom handling of the animals. No maternal or developmental effects were seen at 2,500 ppm.
Reproductive Toxicity
In a 28-day inhalation study, 16 male rats were exposed to HFC-134a at 0, 1,000, 10,000, 50,000 ppm for 6 hr/day, 5 days/wk (Riley et al., 1979). The rats exhibited decreased gonad weights at the 50,000-ppm exposure concentration. However, no effect was
noted on the gonads of male rats exposed at 50,000 ppm (including organ-weight change) in a 90-day inhalation study; thus, the significance of this effect in the 28-day study is not clear. In a 2-year study conducted in male rats exposed to HFC-134a at 50,000 ppm for 6 hr/day, 5 days/wk, there was an increase in testicular weight (Hext and Parr-Dobrzanski, 1993). Leydig-cell hyperplasia and benign Leydig-cell tumors were also reported. The authors reported that none of these effects occurred following exposure of male rats at 10,000 ppm.
Genotoxicity
There is no evidence to suggest that HFC-134a induces either genetic or chromosomal mutations, and hence, there is no reason to suspect that HFC-134a might induce heritable effects in humans. HFC-134a is reported to be nonmutagenic when tested in the Ames assay (Litton Bionetics, 1976; Callandar and Priestley, 1990). It also did not alter DNA synthesis in rat hepatocytes (Trueman, 1990), induce chromosomal aberrations in human lymphocytes (Mackay, 1990), or alter micronucleus formation in the femoral bone marrow of exposed mice (Mueller and Hoffman, 1989). HFC-134a also appears to be nonmutagenic when tested in a dominant lethal assay (Hodge et al., 1979b). The results of the chromosomal aberrations in rat bone-marrow cells are inconclusive (Anderson and Richardson, 1979).
Carcinogenicity
Four groups of 85 male and 85 female rats were exposed to HFC-134a by inhalation at concentrations of 2,500, 10,000, and 50,000 ppm for 6 hr/day for up to 104 weeks (Hext and ParrDobrzanski, 1993). These exposures were whole-body exposures. No toxic effects were observed in the rats at concentrations of up
to 10,000 ppm. At 50,000 ppm, there were increases in testicular weight, Leydig-cell hyperplasia, and Leydig-cell tumors. The Leydig-cell tumors are common in rats and are induced by a wide variety of chemicals in rats. They are usually induced in rats by epigenetic or secondary mechanisms and are related to some unique aspect of rodent metabolism. These tumors are not applicable to humans and thus not considered to be an adverse effect. Thus, the NOAEL or upper limit of animal exposure for nontoxic effects of HFC-134a is about 50,000 ppm.
In a 52-week study, male and female rats were exposed to HFC-134a. The rats were exposed for 1 year by gavage 5 days/wk with HFC-134a dissolved in corn oil at a daily dose of 300 mg/kg of body weight. No carcinogenicity was observed in this investigation. However, only one concentration was used, and it is possible that the route of administration and the dose of the compound used were incapable of detecting a weak carcinogen (Longstaff et al., 1984).
SUMMARY
Table 3-2 summarizes the studies on HFC-134a for cardiac sensitization and subacute, developmental, and reproductive toxicity. Table 3-3 summarizes the studies on HFC-134a for genotoxicity and carcinogenicity.
RECOMMENDATIONSFOR EXPOSURE GUIDANCE LEVELS
The 1-hr EEGL recommended by the subcommittee is 4,000 ppm. This concentration is based on a NOAEL of 40,000 ppm identified in cardiac sensitization tests in which male beagles were exposed to HFC-134a at 40,000, 80,000, 160,000 and 320,000 ppm and simultaneously injected with epinephrine (Hardy et al.,
1991). Cardiac sensitization was seen in dogs exposed at the highest concentrations but was not observed in dogs exposed at 40,000 ppm. Although fetotoxic effects were observed in rats and rabbits at lower exposure concentrations, the NOAELs (20,000 ppm for rabbits exposed for 78 hr and 10,000 ppm for rats exposed for 240 hr) identified in these tests cannot be used to establish a 1-hr EEGL because animals were exposed to HFC-134a for much longer periods than would be appropriate for setting 1-hr EEGLs. Using the NOAEL of 40,000 ppm identified in dog cardiac sensitization studies, the subcommittee determined the 1-hr EEGL to be 4,000 ppm (40,000 ppm divided by an uncertainty factor of 10 to account for interspecies variability). Because blood concentrations of halogenated hydrocarbons are not likely to increase with increasing duration of exposure beyond 5 min, the NOAEL identified for cardiac sensitization following a 10-min exposure can be used without time extrapolation to set a 1-hr EEGL. The subcommittee selected the lower NOAEL (40,000 ppm) from the Hardy et al. (1991) study rather than the higher NOAEL (50,000 ppm) from the Mullin and Hartgrove (1979) study of cardiac sensitization, because Hardy et al. (1991) adjusted the dose of epinephrine administered to each dog individually, whereas Mullin and Hartgrove (1979) used the same dose of epinephrine for all dogs.
TABLE 3-2 Summary of Non-cancer Toxicity Information for HCF-134a
|
Species |
Exposure Frequency and Duration |
End Point |
NOAEL, ppm |
LOAEL, ppm |
Reference |
|
Acute toxicity |
|||||
|
Dog |
10 min |
Cardiac sensitization |
50,000 |
75,000 |
Mullin and Hartgrove, 1979 |
|
Dog |
10 min |
Cardiac sensitization |
40,000 |
80,000 |
Hardy et al., 1991 |
|
Subacute toxicity |
|||||
|
Rat |
14-28 days for 6 hr/day, 5 days/wk |
Interstitial pneumonitus |
10,000 |
50,000 |
Silber and Kennedy, 1979b |
|
Developmental toxicity |
|||||
|
Rat |
Gestation days 6-15, 6 hr/day |
Maternal toxicity |
30,000 |
100,000 |
Lu and Staples, 1981 |
|
Fetal toxicity |
100,000 |
300,000 |
|||
|
Rat |
Gestation days 6-15, 6 hr/day |
Maternal toxicity |
50,000 |
Hodge et al., 1979a |
|
|
Fetal toxicity |
10,000 |
50,000 |
|||
|
Rabbit |
Gestation days 6-18, 6 hr/day |
Maternal toxicity |
2,5000 |
10,000 |
Wickramaratne, 1989a,b |
|
Fetal toxicity |
10,000 |
||||
|
Reproductive toxicity |
|||||
|
Male rat |
28 days for 6 hr/day, 5 days/wk |
Reduced gonad weighta |
50,000 |
Riley et al., 1979 |
|
|
Male rat |
90 days for 6 hr/day, 5 days/wk |
50,000 |
Hext, 1989 |
||
|
Male rat |
104 days for 6 hr/day, 5 days/wk |
Increased testicular weighta |
50,000 |
Hext and Parr-Dobrzanski, 1993 |
|
|
aEnd points were observed at the NOAEL, but are not considered adverse effects. |
|||||
TABLE 3-3 Summary of Carcinogenicity Information for HCF-134aa
|
Species |
Exposure Frequency and Duration |
Tumors |
Reference |
|
Rat |
Inhalation for 104 wk for 6 hr/day, 5 days/wk |
Lydig-cell tumorsb |
Hext and Parr-Dobrzanski, 1993 |
|
Rat |
Oral (gavage) for 52 wk, once per day |
No tumors observed |
Longstaff et al., 1984 |
|
aNo studies have shown any mutagenicity, unschedules DNA synthesis, or chromosomal aberrations. bLeydig-cell tumors are not considered indicative of adverse effects in humans because they are not applicable to humans. |
|||
Results from the cardiac-sensitization test using a 10-min exposure should not be used to set 24-hr EEGLs or 90-day CEGLS. Therefore, the 24-hr EEGL is based on the fetotoxicity effects observed in rats (Hodge et al., 1979a). Using a NOAEL of 10,000 ppm, the subcommitte determined the 24-hr EEGL to be 1,000 ppm (10,000 ppm divided by an uncertainty factor of 10 to account for interspecies variability). The maternal toxicity effects observed in rabbits were not used to determine the 24-hr EEGL because the effects observed were body-weight changes, and the subcommittee did not consider those effects to be adverse.
The 90-day CEGL is based on the 2-year chronic toxicity study conducted in male rats exposed to HFC-134a at 2,500, 10,000, or 50,000 ppm for 6 hr/day, 5 days/wk (Hext and Parr-Dobrzanski, 1993). The NOAEL identified in the study is 50,000 ppm. At that dose, increases in testicular weight and benign Leydig tumors were reported. However, the subcommittee does not consider the increase in testicular weight to be an adverse effect in itself, and the increase in Leydig tumors is not applicable to humans because those tumors are related to some unique aspect of rodent metabolism. Therefore, the subcommittee identified 50,000 ppm as the NOAEL. Using the NOAEL of 50,000 ppm, the subcommittee determined the 90-day CEGL to be 900 ppm (50,000 ppm divided by 10 to account for interspecies variability, a factor of 4 for a 24-hr/day exposure (vs. a 6-hr/day exposure), and a factor of 5/7 for a 7-days/wk exposure (vs. a 5-days/wk exposure)). Although the recommended 24-hr EEGL (1,000 ppm) and 90-day CEGL (900 ppm) are similar, they are based on different end points or target toxicities. The fetotoxicity observed in rats during gestation was used to develop the 24-hr EEGL, whereas the 90-day CEGL was recommended on the basis of no adverse effects observed in rats chronically exposed to 50,000 ppm. The similarity of the exposure guidance levels recommended for 24-hr and 90-day exposures is consistent with the fact that blood concentrations of this class of compounds reach maximal levels within minutes of the onset of expo-
sure, and continuing exposure for many hours or days does not increase blood concentrations further (Mullin et al., 1979).
Proposed Recommendations for HFC-134a
|
Exposure |
Concentration, ppm |
|
1-hr EEGL |
4,000 |
|
24-hr EEGL |
1,000 |
|
90-day CEGL |
900 |
REFERENCES
Anderson, D., and C.R. Richardson. 1979. Arcton-134a: A Cytogenetic Study in the Rat. ICI Rep. No. CTL/P/444. Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Azar, A., H.J. Trochimowicz, J.B. Terrill, and L.S. Mullin. 1973. Blood levels of fluorocarbon related to cardiac sensitization. Am. Ind. Hyg. Assoc. J. 34:102-109.
Callander, R.D., and K.P. Priestley. 1990. HFC-134a: An Evaluation Using the Salmonella Mutagenicity Assay. ICI Rep. No. CTL/P/2422. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Hardy, C.J., I.J. Sharman, and G.C. Clark. 1991. Assessment of Cardiac Sensitisation Potential in Dogs. Rep. No. CTL/C/ 2521. Huntingdon Research Centre , Huntingdon, Cambridgeshire, U.K.
Hext, P.M. 1989. HFC 134a: 90-Day Inhalation Toxicity Study in the Rat. ICI Rep. No. CTL/P/2466. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Hext, P.M., and R.J. Parr-Dobrzanski. 1993. HFC-134a: A 2-Year Inhalation Toxicity Study in the Rat. ICI Rep. No. CTL/P/3841. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Hodge, M.C.E., M. Kilmartin, R.A. Riley, T.M. Weight, and J. Wilson.
1979a. Arcton 134a: Teratogenicity Study in the Rat. ICI Rep. No. CTL/P/417. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Hodge, M.C.E., D. Anderson, I.P. Bennett, and T.M. Weight. 1979b. Arcton-134a: Dominant Lethal Study in the Mouse. ICI Rep. No. CTL/P/437. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Litton Bionetics. 1976. Mutagenicity Evaluation of Genetron-134a. LBI Project No. 2683. Final Report. Prepared for Allied Chemical Corp. by Litton Bionetics, Kensington, Md.
Longstaff, E., M. Robinson, C. Bradbrook, J.A. Styles, and I.F.H. Purchase. 1984. Genotoxicity and carcinogenicity of fluorocarbons: Assessment by short-term in vitro tests and chronic exposure in rats. Toxicol. Appl. Pharmacol. 72:15-31.
Lu, M., and R. Staples. 1981. 1,1,1,2-Tetrafluoroethane (FC-134a): Embryo-fetal toxicity and teratogenicity study by inhalation in the rat. Report No. 317-81. Haskell Laboratory , Wilmington, Del.
Mackay, J.M. 1990. HFC-134a: An Evaluation in the in Vitro Cytogenetic Assay in Human Lymphocytes. ICI Rep. No. CTL/P/2977. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Müller, W., and T. Hoffmann. 1989. CFC-134a Micronucleus Test in Male and Female Mice after Inhalation . Rep. No. 89.0015, Study No. 88.1244. Hoechst Aktiengesellschaft , Frankfurt, Germany.
Mullin, L.S., and R.W. Hartgrove. 1979. Cardiac Sensitization. Rep. No. 42-79. Haskell Laboratory , Wilmington, Del.
Mullin, L.S., C.F. Reinhardt, and R.E. Hemingway. 1979. Cardiac arrhythmias and blood levels associated with inhalation of Halon 1301. Am. Ind. Hyg. Assoc. J. 40:653-658.
Olson, M.J., C.A. Reidy, J.T. Johnson, and T.C. Pederson. 1990. Oxidative defluorination of 1,1,1,2-tetrafluoroethane by rat liver microsomes. Drug Metab. Dispos. 18:992-998.
Riley, R.A., I.P. Bennett, I.S. Chart, C.W. Gore, M. Robinson, and T.M. Weight. 1979. Arcton-134a: Subacute Toxicity to the Rat by Inhalation. ICI Rep. No. CTL/P/463. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Rissolo, S.B., and J.A. Zapp. 1967. Acute Inhalation Toxicity. Rep. No. 190-67. Haskell Laboratory , Wilmington, Del.
Salmon, A.G., J.A. Nash, M.F.S. Oliver, and A. Reeve. 1980. Arcton134a: Excretion, Tissue Distribution, and Metabolism in the Rat. ICI Rep. No. CTL/P/ 513. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Silber, L.S., and G.L. Kennedy. 1979a. Acute Inhalation Toxicity of Tetrafluoroethane. Rep. No. 422-79. Haskell Laboratory , Wilmington, Del.
Silber, L.S., and G.L. Kennedy. 1979b. Subacute Inhalation Toxicity of Tetrafluoroethane (HFC 134a). Rep. No. 228-79. Haskell Laboratory , Wilmington, Del.
Trochimowicz, H.J., A. Azar, J.B. Terrill, and L.S. Mullin. 1974. Blood levels of fluorocarbon related to cardiac sensitization: Part II. Am. Ind. Hyg. Assoc. J. 35:632-639.
Trueman, R.W. 1990. HFC-134a: Assessment for the Induction of Unscheduled DNA Synthesis in Rat Hepatocytes In Vivo. ICI Rep. No. CTL/P/2550. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Wickramaratne, G.A. 1989a. HFC-134a: Teratogenicity Inhalation Study in the Rabbit. ICI Rep. No. CTL/P/2504. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.
Wickramaratne, G.A. 1989b. HFC-134a: Embryotoxicity Inhalation Study in the Rabbit. ICI Rep. No. CTL/P/2380. Central Toxicology Laboratory, Imperial Chemical Industries , Alderley Park, Macclesfield, Cheshire, U.K.