1
INTRODUCTION
In September 1994, the Department of Energy (DOE1) asked the National Research Council to undertake a technical evaluation of the electrometallurgical processing technology proposed by Argonne National Laboratory (ANL) as a potential approach for treating DOE spent nuclear fuel (SNF). A brief description of DOE's SNF holdings and ANL's rationale for proposing an electrometallurgical approach can be found in ANL's January 1995 “Proposal for Development of Electrometallurgical Technology for Treatment of DOE Spent Nuclear Fuel,” which is included in Appendix A.
COMMITTEE TASK
The DOE's request led to the formation of the Committee on Electrometallurgical Techniques for DOE Spent Fuel Treatment, which was charged with carrying out a fast-track study of the proposed electrometallurgical techniques as potential technologies for the treatment of DOE SNF. The committee was specifically charged with two tasks:
-
Hold a first meeting to receive briefings from representatives of the DOE and ANL, additional experts identified by the committee, and representatives of other relevant activities of the National Research Council and National Academies of Sciences and Engineering, and then prepare an interim report to address the question, “Do electrometallurgical techniques represent a potentially viable technology for DOE spent fuel treatment that warrants further research and development?”
-
Study in more depth the advantages and disadvantages of continued R&D into electrometallurgical processing as a candidate technology for disposition of DOE spent nuclear fuel, specifically addressing the issues of technical feasibility, cost-effectiveness, suitability of the metallic waste form for long-term storage or geologic disposal, and nonproliferation implications, and write a report on the committee 's assessments.
In accord with its charge, the committee focused on the technological aspects of electrometallurgical processing: the committee discussed all effluent streams that result from electrometallurgical processing, including the metallic waste form singled out in the charge. Neither its February 1995 interim report2 nor this final report addresses the issue of whether or not this technology should be adopted as a component of the national strategy for handling nuclear materials. The committee evaluated the electrometallurgical approach as one of several technical options available to the DOE but refrained from suggesting which option should be pursued. That decision, which lies outside the scope of this study, must be made by the DOE on the basis of a broader range of concerns than are addressed here.
|
1 |
The acronyms used in this report are listed in Appendix C |
|
2 |
A Preliminary Assessment of the Promise of Continued R&D into an Electrometallurgical Approach for Treating DOE Spent Fuel, National Research Council, Washington, D.C., 1995. |
COMMITTEE ACTIVITIES
The committee held its first meeting on January 9-11, 1995, at the National Research Council's facilities in Washington, D.C., during which it received briefings from representatives of the DOE and additional experts identified by the committee. The information obtained from these briefings enabled the committee to carry out its preliminary assessment of the ANL's R&D plan (included in Appendix A). Additional information was provided by discussions with technical experts from ANL, Westinghouse Hanford Co., Westinghouse Savannah River Co., and Sandia National Laboratories. The committee also received input from representatives of several interested public citizens groups. The agenda and attendance list for this meeting are included in Appendix B.
The interim report was released by the National Research Council in February 1995 to meet the first of the committee's tasks. The committee concluded that the technology of electrometallurgical processing appears sufficiently promising for treating a variety of DOE spent fuels that continued R&D would be warranted in federal FY96.
The committee held three additional meetings in the period of February through April 1995, during which it received additional technical briefings and had the opportunity to carry on extended discussions with individuals who are knowledgeable in various aspects of electrometallurgical technology. All meetings were unclassified. Agendas and attendance lists for these meetings are presented in Appendix B.
The committee's second meeting was held in Chicago on February 13-14. This meeting included a site visit to ANL-East on February 13, where the committee toured the laboratories involved in electrometallurgical R&D and directly discussed the program with those individuals conducting the R&D effort. The remainder of the meeting was held in the conference facilities of the O'Hare Hilton Hotel. The committee took the opportunity to address unresolved questions with ANL experts who attended the open parts of the meeting. The committee also heard from a representative of a public citizens group.
The third committee meeting took place in Idaho Falls, Idaho, on March 22-24 and included a site visit to ANL-West on March 23. The site visit provided committee members an opportunity to view the facilities that are being used to carry out the development phase for electrometallurgical processing, including the demonstration project for treating spent fuel from the Experimental Breeder Reactor II (EBR-II). The site visit also provided an opportunity for the committee to ask questions of technical staff directly involved with operating the facility. The open sessions of this committee meeting provided a further opportunity for the committee to discuss unresolved issues and questions with members of the ANL technical staff. The committee was also briefed by a representative of a regional public citizens group. During the executive session, the committee formulated an outline of the proposed final report, and writing assignments were given to individual committee members.
The fourth meeting of the committee took place on April 27-28 at the National Research Council's facilities in Washington, D.C. A short open session permitted an additional briefing and discussion with DOE representatives and a representative of a public citizens group. During its executive session, the committee discussed the materials written by individual members and reached consensus on the contents of the final report and its recommendations.
INTERIM REPORT
For completeness, the committee's February 1995 interim report2 is summarized below; the text of the report, including the ANL proposal as an appendix, appears in Appendix A. For the interim report, the committee focused on the technical merit of the proposed R&D in the near term, and the phrase “potentially viable technology” was taken in its limited sense, to refer to “technology for which the underlying basic science is reasonably understood, the laboratory work to date is generally promising, and the anticipated problems of scale-up and eventual maintenance do not appear to be technically insurmountable.”
On the basis of the technical presentations to the committee, discussions with the presenters, additional written materials reviewed by the committee,3 and the extensive technical experience of the committee members, no “showstoppers” were identified in the ANL technical proposal that would suggest discontinuing the planned R&D. The committee concluded that the technology of electrometallurgical processing appears sufficiently promising for treating a variety of DOE spent fuels that continued R&D would be warranted in federal FY96. The committee did not evaluate the associated funding levels, estimated by the DOE at $12 million annually.4
The committee recommended that the DOE should closely follow the progress of the proposed R&D program during the next 12 months to determine whether it should be continued beyond FY96, noting that critical input from experts outside the program and parallel evaluation of alternatives would be beneficial to the DOE in making that decision. The progress and status of the planned demonstration of the process as applied to irradiated EBR-II fuel were specifically noted as an essential factor in making this determination.
The committee's conclusion in favor of continued R&D through federal FY96, explicitly intended as neither an endorsement nor a rejection of the overall ANL proposal (included as an appendix of the interim report), was based on the following rationale.
-
First, the electrometallurgical technique is not a new technology. Progress made to this point in Argonne's R&D program indicates that the ANL group has understood and addressed a number of challenges in developing the electrometallurgical process.5 ANL personnel have already demonstrated, on an engineering scale, electrometallurgical separations with simulated metal fuel. They have also demonstrated, on a laboratory scale, the lithium reduction of simulated mixed oxide (MOX) fuel, which is suggestive of the applicability of that technique to treating DOE's oxide spent fuels.
-
Second, the electrometallurgical technology has been developed sufficiently to be proposed for treating EBR-II spent fuels, and the DOE apparently has sufficient confidence in the process to have approved this application. The electrometallurgical technique needs to be demonstrated with actual spent fuel, which could provide data on issues of scale-up, remote operation and
3
These materials included ANL's proposal, the sections on separations technology from the draft National Research Council report Separations Technology and Transmutation Systems (National Research Council, in preparation), which discusses pyroprocessing for the now-terminated Integral Fast Reactor (IFR) program, and the 1994 document “Pyrochemical Processing—Accomplishments and Applications” (Special Advisory Committee for the Integral Fast Reactor, University of Chicago, 1994).
4
Treatment of the EBR-II spent fuel is being funded separately.
5
“Pyrochemical Processing—Accomplishments and Applications,” Special Advisory Committee for the Integral Fast Reactor, University of Chicago, 1994.
-
maintenance in an inert atmosphere, and effects of high radiation levels. The scheduled demonstration of EBR-II spent fuel treatment will enable a practical near-term evaluation of important components of ANL's proposed electrometallurgical treatment technology. The committee emphasizes that the DOE should follow this demonstration project closely, in order to evaluate the efficacy of the technology.
The committee's recommendation for continuation of the R&D through FY96 was not intended to imply an endorsement of the incentives described by ANL for electrometallurgical treatment of DOE spent fuels (see Appendix A, pp. A-4 to A-6) nor of any particular disposal option for any of the product streams. Instead, the rationale for carrying the proposed R&D through its current phase of development was viewed by the committee as independent of the specific incentives described by ANL. Evaluation of such incentives was viewed as appropriate at a later stage when the DOE makes a future policy choice among technological options for the treatment of DOE spent fuel.
The committee further concluded that continued electrometallurgical technology R&D should not be taken as an endorsement of the Integral Fast Reactor (IFR) program that has been terminated by the DOE. The committee recommended that such R&D should be conducted exclusively to evaluate this technology's effectiveness for treating DOE spent fuel, and funding decisions about electrometallurgical R&D for DOE spent fuel treatment should be independent of issues related to the IFR program.
The committee noted that it was aware of other possible technologies for spent fuel treatment, but it did not attempt a comparative evaluation of the electrometallurgical approach against other potentially applicable technologies. The committee did seek to assess where the electrometallurgical treatment technology would fit into the overall DOE scheme for disposal of its spent fuel, but no overall “road map” for DOE's approach to spent fuel treatment was available to the committee. DOE representatives advised that a generic environmental impact statement is in preparation that would seek to address the anticipated options for DOE spent fuel disposition. The committee urged that prior to making a final decision about widespread implementation of the ANL electrometallurgical technology for treating DOE spent fuel, the DOE should determine how the electrometallurgical technology, and other treatment technologies, fit into its overall strategy for spent fuel disposal and the closely related strategy for the management of high-level and transuranic wastes.
2
ARGONNE NATIONAL LABORATORY'S ELECTROMETALLURGICAL TECHNOLOGY
DESCRIPTION
The electrometallurgical technology under development at ANL is derived from many years of R&D on molten salt systems related to the production of materials for nuclear weapons as well as from battery development activities. The proposed technology (summarized in ANL's draft proposal (see Appendix A) and in briefings to the committee (see Appendix B) for treating DOE's SNF consists of several unit operations, as shown in Figure 1. They include:
-
Head-end treatment, including fuel disassembly and steps such as oxide reduction, if required;
-
Electrorefining; and
-
Treatment of effluent electrorefining streams, including processing of the uranium metal deposited at the steel cathode.
The heart of the process is the electrorefining step, which employs a metallic feed, molten alkali metal salts as the reaction medium, and two cathodes, one steel and the other an immiscible pool of molten cadmium, to separate actinides from fission products and other nuclear reactor fuel materials. This separation process may be of value in the treatment of some DOE SNFs, particularly for reactive metallic fuels that are not expected to be amenable to geologic disposal in their present form.
Head-End Treatment
In ANL's proposed approach, processing of SNF would begin by dismantling the irradiated fuel assembly, removing the fuel elements, and chopping them into pieces for subsequent processing. If the fuel is metallic, the chopped fuel elements would be sent directly to electrorefining. If the fuel is an oxide, the oxide first must be reduced to metal (see below), which would then go to electrorefining. If the fuel is neither metal nor oxide (e.g. a carbide, nitride, etc.) or if it has protective layers of graphite, other steps can be envisioned that would convert the fuel to an oxide that would then be compatible with the oxide reduction step. These other steps have not yet been developed in most cases.
The purpose of the oxide reduction process, which is not required for metallic fuels, is to convert oxide fuels such as UO2 to metal, which can then be processed by electrorefining. The reaction is
UO2(s) + 4 Li(l) → U(s) + 2 Li2O(l)
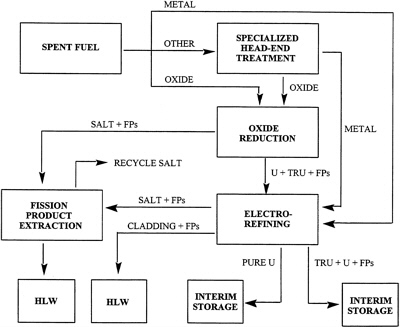
FIGURE 1 Schematic representation of the ANL electrometallurgical technology for treatment of spent nuclear fuel. HLW is high-level waste, FPs are fission products, and TRU denotes transuranic elements.
The reduction would be carded out in molten LiCl at about 500ºC because Li2O is more soluble in LiCl (8.7 wt % Li2O) than in LiCl-KCl. The unique aspect of the process is the direct production of solid uranium metal, which can be separated from the molten salt. To date, this separation of solid uranium particles from the molten salt of LiCl-Li2O has been done only on a 20-kg scale, by sieving with metal screens. In progress at ANL is development of a submersible centrifuge that may facilitate this separation. The Li2O in the spent salt would be reduced electrolytically at a graphite or inert metal electrode to produce lithium metal and pure LiCl for recycle to the oxide reduction process.
Electrorefining
Most of the chemical separations of the proposed SNF treatment would take place in the electrorefining step. Electrorefining utilizes molten LiCl-KCl as a solvent, at an operating temperature of about 500ºC, to effect the separations shown in Figure 2.
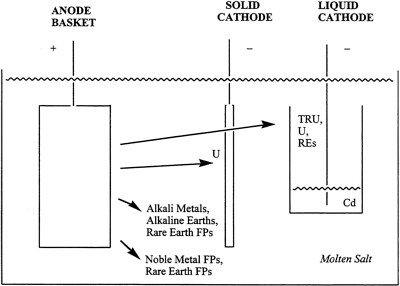
FIGURE 2 Schematic of ANL's electrochemical separations in ANL's proposed approach to treating SNF. REs are rare-earth elements, FPs are fission products, and TRU denotes transuranic elements.
Thus, pure uranium metal would be deposited at a solid steel cathode, and a metallic mixture of plutonium, americium, neptunium, curium, uranium, and some rare-earth fission products would be deposited in a liquid cadmium cathode. Major fission products such as cesium and strontium, however, would remain in the LiCl-KCl salt, which would be processed further to remove the residual actinides and produce a high-level waste salt that is depleted of transuranic (TRU) waste. If a decision is made to place the TRU elements in the ceramic waste form, treatment of the salt would not be required, nor would the liquid cadmium cathode be needed. The TRUs and fission products would simply be left in the salt for later immobilization in the zeolite waste form. The noble metal fission products and cladding hulls would remain in the anode basket. A large database of thermodynamic free energies of reaction and activity coefficients
has been developed from years of ANL experience, permitting predictions about many of the proposed reactions.
The actual sequence of electrorefining operations would take place as follows:
-
An oxidant, depleted UCl3 or CdCl2, would be added to the initial batch of melt to oxidize some of the spent fuel constituents and cause their dissolution in the molten LiCl-KCl salt. For example:
3 Cs + UCl3 → 3 CsCl + U
2 Cs + CdCl2 → 2 CsCl + Cd
The final distribution of uranium, TRUs, and fission products among the three electrodes depends on the concentration of UCl3 in the salt. The spent fuel feedstock would be treated with sufficient UCl3 or CdCl2 to produce a uranium chloride concentration in the KCl-LiCl of about 2 mole percent. Once this is achieved, upon initial startup of the electrorefiner, there would be no need for further oxidant additions until the electrolytic salt was removed for treatment to extract the accumulated fission products.
-
The spent fuel would be dissolved anodically and uranium deposited at the steel cathode. For example:
Anode reaction: U → U+3 +3 e−
Cathode reaction: U+3 + 3 e− → U
-
Liquid cadmium cathode: Additional fuel elements are dissolved anodically into the initial batch of molten salt until the ratio of PUCl3 to UCl3 reaches 3.0. Transuranic elements, rare-earth elements, and some uranium would then be deposited into a molten cadmium cathode. For example:
Anode reaction: M → M+x + x e−
Cathode reaction: Pu+3 + 6 Cd + 3e− → PuCd6
Data on free energies of formation for the chlorides (Table 1) permit estimation of the behavior of SNF elements in operations 1 and 2 above. In the oxidation process (operation 1), elements whose chlorides have a free energy of formation more negative than that of UCl3 or CdCl2 (i.e., U, Np, Cs, Sr, etc.) would be oxidized and would dissolve in the molten salt phase following the initial addition of UCl 3 or CdCl2. Elements with less negative free energy values (i.e., iron, etc.) would remain mostly unreacted in the anode basket. Similar considerations apply to the anodic dissolution process (operation 2).
TABLE 1 Free Energies of Formation of SNF Chlorides (−∆G0, in units of kcal/g-eq at 500ºC)
|
Elements That Remain in Salt (very stable chlorides) |
Elements That Can Be Electrotransported Efficiently |
Elements That Remain As Metals (less stable chlorides) |
|||
|
BaCl2 |
87.9 |
CmCl3 |
64.0 |
CdCl2 |
32.3 |
|
CsCl |
87.8 |
PuCl3 |
62.4 |
FeCl2 |
29.2 |
|
RbCl |
87.0 |
AmCl3 |
62.1 |
NbCl5 |
26.7 |
|
KCl |
86.7 |
NpCl3 |
58.1 |
MoCl4 |
16.8 |
|
SrCl2 |
84.7 |
UCl3 |
55.2 |
TcCl4 |
11.0 |
|
LiCl |
82.5 |
ZrCl2 |
46.6 |
RhCl3 |
10.0 |
|
NaCl |
81.2 |
PdCl2 |
9.0 |
||
|
CaCl2 |
80.7 |
RuCl4 |
6.0 |
||
|
LaCl3 |
70.2 |
||||
|
PrCl3 |
69.0 |
||||
|
CeCl3 |
68.6 |
||||
|
NdCl3 |
67.9 |
||||
|
YCl3 |
65.1 |
||||
|
SOURCE: Data from James Laidler, ANL, in presentation to the committeeon Jan. 9, 1995. |
|||||
In the cathode deposition process, chlorides with the least negative free energy values would be deposited preferentially. Thus, uranium would be deposited on the steel cathode, while the transuranic elements, rare earths, alkaline earths, and alkali metals would remain in the salt phase. While “textbook standard” free energies of formation are helpful in predicting behavior in electrorefining, such estimates are only approximate. Accurate predictions require measurements employing the molten salt as solvent under actual processing conditions.
At concentrations in excess of their solubility in cadmium, plutonium and the other transuranic elements will form intermetallic compounds with cadmium at the electrorefining cell temperature of 500ºC. The uranium-cadmium intermetallic compound U-Cd11 is not stable above 473ºC. At the steel cathode, uranium is deposited in preference to plutonium and the other transuranic elements, as can be inferred from the free energies in Table 1. At the cadmium electrode, however, the reduction potential for plutonium and the other transuranic elements is greatly increased by formation of the intermetallic compounds with cadmium. As a result, these elements, along with some uranium, are deposited at the cadmium cathode. The same holds true for the rare earths.
The above discussion of the expected fate of SNF elements in ANL's electrorefining process are summarized in Figure 3.
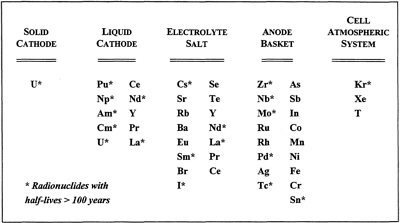
FIGURE 3 Separation of spent nuclear fuel elements during the ANL electrorefining process.
Treatment of Electrorefining Effluents
The electrorefining process is designed to produce several effluent streams. Each of these is a potential waste stream consisting of material that would need to be disposed of or stored pending some alternative disposition.
-
A uranium solid with some entrained electrolyte (i.e., LiCl/KCl) deposited on a steel cathode.
-
A metallic stream (from the cadmium cathode) containing most of the TRUs and some uranium and rare-earths.
-
A molten-salt stream containing salts of the alkali metals, alkaline earths, some uranium and transuranics, and the reactive metal fission products (including cesium, strontium, and most of the rare-earth elements).
-
A metallic stream, remaining at the anode in the dissolver basket, that would contain cladding hulls and noble metal fission products.
The solid uranium cathode deposit would go to the cathode processor for vacuum removal of volatile salts and casting into ingot form. Cadmium would be volatilized from the TRU stream and be recycled to the electrorefiner, and the TRU fraction also would be cast into ingot form for interim storage.
After processing of multiple batches of the spent fuel, fission products such as Cs and Sr and the rare-earth elements would build up in the molten salt. When the heat load from the radioactive decay of these fission products reached a specified limit in the electrorefiner, the electrolyte salt would have to be removed for waste treatment. At this stage, the molten salt would still contain actinide elements that would also need to be removed. The proposed removal involves reduction with Li-Cd to remove the bulk of uranium and TRUs, followed by reduction with depleted U-Cd to remove residual quantities of uranium and TRUs. Two countercurrent pyrocontactor stages would be employed for reduction using Li-Cd, and six countercurrent pyrocontactor stages would be used for U-Cd reduction. The TRU-depleted salt then would be passed through zeolite beds to remove fission products, and the effluent salt would be returned to the electrorefiner. The uranium and TRUs extracted into the cadmium in the pyrocontactors would be returned to the electrorefiner for subsequent electrotransport by oxidizing the mixture (U and TRU in Cd) with CdCl2. This would produce UCl3 and (TRU)Cl3, which would be added to the clean salt from the zeolite column and returned to the electrorefiner. The cadmium metal would be recycled to the pyrocontactors. For processing EBR-II fuel, where sodium was used for bonding and increased heat transfer between the cladding and fuel, a small portion of salt would be bled off periodically and replaced with LiCl/KCl. If NaCl were allowed to build up, the melting point of the molten salt reaction medium would increase.
When the zeolite had become nearly saturated with rare-earth fission products, it would be mixed with additional anhydrous zeolite and a glass binder and consolidated into a proposed final waste form. The process effluent consisting of cladding and noble metal fission products would undergo a melting process prior to being readied for disposal as high-level waste.
PRESENT STATUS AND FUTURE PLANS
Head-End Treatment
The head-end treatment for irradiated EBR-II fuels will be developed in the Fuel Conditioning Facility (FCF) at ANL-West during the processing of EBR-II driver and blanket fuels. Since EBR-II driver and blanket fuels are metallic, the head-end treatment should be straightforward and consists primarily of chopping the fuel elements to a size appropriate for introduction into the anode basket of the electrorefiner.
For the treatment of DOE SNFs that are in the oxide form (and do not contain aluminum as a component of the fuel matrix), the oxide reduction process would require additional development. To date the oxide reduction process has been demonstrated successfully on an engineering scale, using 20 kg of simulated spent light water reactor (LWR) oxide fuel. Laboratory experiments with spent Commonwealth Edison LWR fuel (0.5 kg) have also been done successfully. Little or no head-end development work has been carried out to this point for treating by the ANL electrometallurgical technique of more complicated fuel types such as metal-hydride, metal-aluminum, or metal-silicon alloys, carbide, and graphite or fuels with aluminum cladding.
Additional R&D that must be carried out to enable application of the ANL electrometallurgical technology to nonmetallic DOE fuels thus includes:
-
Development of an electrolytic process for the recovery of the Li2O produced in the oxide reduction process;
-
Development of a submersible centrifuge to replace the metal screens used for collection of uranium metal product;
-
Demonstration of oxide reduction processes in the FCF; and
-
Development of procedures for treating rubble from the Three Mile Island reactor, U-Zr hydride fuels, and spent or damaged fuels from the Power Burst Facility (PBF) and Loss of Fluid Test Reactor (LOFT).
Electrorefining
Electrorefining has been carried out by ANL on an engineering scale (10 kg of heavy metal/day) with unirradiated metal fuel. For the upcoming EBR-II demonstration, this equipment will be used in the FCF to process about 1 metric ton of EBR-II driver fuel. A high-throughput electrorefiner (200 kg heavy metal/day), currently being developed at ANL-East, will be used to process about 16 metric tons of EBR-II blanket fuel in the FCF. The EBR-II campaign should demonstrate the applicability or nonapplicability of the electrometallurgical process to the production-scale treatment of irradiated metallic fuels.
Treatment of Electrorefining Effluents
Laboratory experiments at ANL-East have shown that a TRU-depleted zeolite waste form could be produced from the electrorefiner's salt effluent and that the purified salt could be returned to the electrorefiner for reuse. However, this waste form is at a very early stage of development.
Treatment of the metal waste stream that consists of cladding and noble metal fission products is still under study. This proposed waste form would be prepared by combining the anode waste stream with stainless steel filter cartridges from the bulk fluid handling system. This mixture would be melted with a small amount of zirconium to produce a relatively lower melting eutectic.
The uranium from the electrorefiner's steel cathode requires processing to remove entrained salt and produce a uranium ingot. This is a major process step that would take place in an inert atmosphere in the cathode processor. The process has been tested with unirradiated uranium cathode deposits only, and it must be demonstrated at a production scale under inert-atmosphere conditions with actual spent fuel.
ANL's program for federal FY1995 through FY1997 calls for intensive R &D efforts to develop effluent processing procedures on an engineering scale and to demonstrate the processes in the FCF for EBR-II fuel.












