1
Introduction
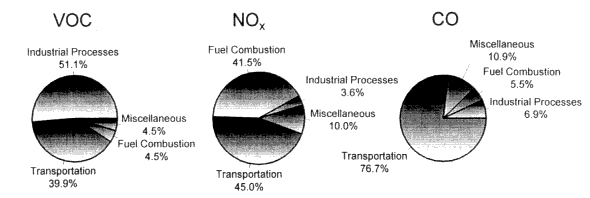
Photochemical smog, and its concomitant high concentrations of ground-level ozone (O3) and other noxious compounds, is caused by a complex series of chemical reactions involving the oxidation of volatile organic compounds (VOCs)1 and carbon monoxide (CO) in the presence of nitrogen oxides (NOx) and sunlight (Figure 1-1). As illustrated in Figure 1-2, the transportation sector is responsible for a large fraction of VOC, CO, and NOx emissions in the United States. On-road gasoline-fueled motor vehicles are estimated to account for about 26% of the VOC emissions from all source categories, about 56% of the CO emissions, and about 22% of the NOx emissions in 1997 (EPA 1998). As a result, motor vehicles have been a primary target for emission controls in the
|
1 |
An organic compound is a compound containing carbon combined with atoms of other elements, commonly hydrogen, oxygen, and nitrogen. Simple carbon-containing compounds such as carbon monoxide (CO) and carbon dioxide (CO2) are usually classified as inorganic compounds. A volatile organic compound (VOC) is an organic compound that exists as a gas under typical atmospheric conditions. A large number of acronyms are used to denote various categories of volatile organic compounds; a listing of some of the more common acronyms and their meanings is presented in Chapter 3. In this report, organic compounds in the gas phase are referred to as "VOCs" unless noted otherwise. |
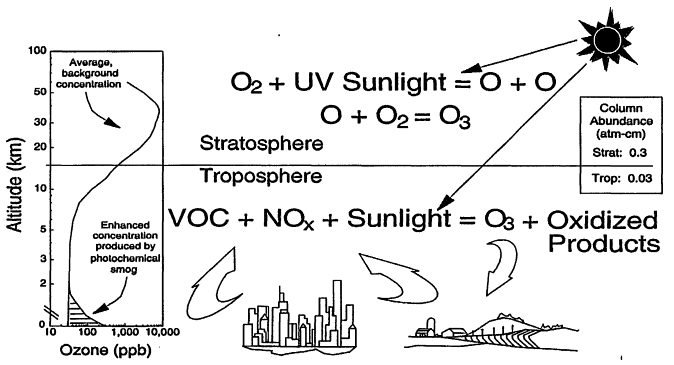
Figure 1-1
Atmospheric ozone (O3). Left-handed graphic: average O3 mixing ratio (in parts per billion by volume) as a function of altitude; center: photochemical processes responsible for producing O3 in the stratosphere and troposphere; right-handed inset: column abundance of O3 in atm-cm (i.e., the height of the O3 column if it were compressed to 1 atmosphere pressure). Although only VOCs and NOx are shown as ozone precursors, carbon monoxide (CO) can participate, in much the same way as VOCs, in the sequence of reactions leading to ozone formation. Source: Adapted from EPA 1998.
nation's strategy for mitigation of the ozone pollution problem.2,3 As part of this effort, the Clean Air Act Amendments of 1990 called for the development and use of reformulated gasoline (RFG)4 in light-duty motor vehicles to reduce the ozone precursor emissions from those vehicles.
Through the Clean Air Act Amendments of 1990, Congress mandated that RFG contain at least 2.0% oxygen by weight to decrease the emissions of ozone-precursors and air toxics. To meet that requirement, RFG blends typically contain small amounts of additives referred to as oxygenates, which are organic compounds that contain some chemically bound oxygen. The use of these oxygenates in RFG has given rise to a complex and often contentious debate concerning the relative benefits of one oxygenated compound over another (e.g., methyl tertiary-butyl ether (MTBE) versus ethanol). Oxygenates can affect the amounts and types of ozone precursors emitted by motor vehicles in different and potentially offsetting ways. Is oxygenated gasoline preferable from an air-quality point of view over nonoxygenated gasoline? Is one oxygenated compound clearly preferable from an air-quality point of view over another? Should some oxygenated additives be allowed to be used in RFG whereas others should not be allowed? In the traditional approach to ozone mitigation in the United States, these questions are addressed in a straightforward and simple manner: the mass of precursor emissions from the use of various RFG blends in motor vehicles are assessed; and,
|
2 |
EPA estimates that non-road gasoline-fueled motor vehicles account for about 9% of the VOC emissions from all source categories, about 19% of the CO emissions, and about 19% of the NOx emissions in 1997 (EPA 1998). Because emission test data from non-road vehicles fueled by RFG were not available to the committee, consideration of such emissions were not included in this study. |
|
3 |
Uncertainties associated with mobile source emission estimates are discussed in Chapter 4. |
|
4 |
In this report, "RFG" is used in the most generic sense to refer to blends of gasoline that have been reformulated to change any of a multitude of gasoline-blend characteristics (e.g., blend content, oxygen content, sulfur content, and vapor pressure) and motor-vehicle-emissions characteristics. Such use of the term "RFG" should not be confused with the more narrow regulatory definitions of RFG as a gasoline blend that is compliant with the specific requirements of the federal RFG program or the California RFG program. Those regulatory definitions represent a subset of the range of possible reformulated gasolines. |
if the mass of emissions from a blend exceeds sortie specified amount, it is disallowed. In this report, the committee assesses the utility and accuracy of an alternate approach based on evaluating the ozone-forming potential of ozone-precursor emissions. With such an approach, an RFG blend with a high rate of emissions based on mass might be considered acceptable if those emissions were of relatively low ozone-forming potential. It should be noted that this report is limited to the scientific and technical aspects of this issue; the possible design or implementation of regulations based on ozone-forming potential are not addressed.
The Ozone-Pollution Problem
For every billion atmospheric molecules, there are, on average, only about 300 ozone molecules. Despite this minute concentration, atmospheric ozone has a major impact on the environment. In the stratosphere, where about 90% of the atmosphere's ozone resides, it protects life from harmful ultraviolet radiation from the sun. In the upper part of the troposphere, ozone is critical to the oxidation process in the atmosphere by which a wide range of pollutants is removed from the air we breathe.
Ground-level ozone (i.e., at the lowest part of the troposphere) represents a small, but important, component of the total burden of ozone found in the troposphere. Although ground-level ozone concentrations in remote regions of the atmosphere are about 20 to 40 parts per billion by volume (ppb), those concentrations often exceed 100 ppb during episodes of increased air pollution and can rise above 200 ppb during severe episodes in urban areas.5 When ozone concentrations reach increased levels, they can harm rather than sustain organisms.
Adverse effects include impairment of lung function in humans and other animals, and damage to agricultural crops, forests, and other vegetation. It is this aspect of tropospheric ozone, as a ground-level pollutant, that forms the backdrop for this report.
History of U.S. Policies to Mitigate Ozone Pollution
The phenomenon known as photochemical smog was first documented in the 1940s when air pollutants were found to be causing damage to vegetable crops grown in the Los Angeles area (Middleton et al. 1950). Soon after, Haagen-Smit and others showed that ozone was the primary oxidant in photochemical smog causing crop damage and that it was produced by photochemical reactions requiring the participation of two types of precursors: VOCs and NOx (Haagen-Smit et al. 1951, 1953; Haagen-Smit 1952; Haagen-Smit and Fox 1954, 1955, 1956).
Subsequent observations revealed that photochemical smog and the concomitant high concentrations of ground-level ozone that accompanied the smog were not unique to Los Angeles but were common to most of the major metropolitan areas of the United States and elsewhere in the world. Those observations, along with medical and epidemiological studies documenting the adverse health effects of ozone at concentrations encountered during air-pollution episodes, provided the impetus for the promulgation of regulations designed to control or even eliminate the problem. Passage of the Clean Air Act of 1970 (Public Law 91-604) established National Ambient Air Quality Standards (NAAQS) for ozone and other criteria pollutants as well as a federally coordinated program to reach attainment of these standards within specific deadlines. With the persistence of the ozone problem, ever more stringent and costly air-pollution controls were promulgated by the Glean Air Act Amendments of 1977 (Public Law 95095) and 1990 (see Table 1-1).
In retrospect, it appears that the ozone mitigation policies our nation has embarked upon over the past 3 decades have had a positive impact. On average, peak ozone concentrations in urban areas of the United States appear to be on a downward trend (Figure 1-3) and the problem would undoubtedly be considerably more severe if controls had not been implemented (see e.g., Harley et al. 1997). The U.S. Environmental Protection Agency (EPA) reports that ozone concentrations decreased for the 1-hr and 8-hr averaging times (shown in Figure 1-3)
TABLE 1-1 Milestones in Ozone Pollution and Its Control in the United States
|
Year |
Milestone |
Notes |
|
1840s |
Ozone molecule discovered |
Schoenbein 1840 |
|
1850s |
Ozone presence in atmosphere documented |
Schoenbein 1854 |
|
1874 |
Ozone shown to be toxic to animals |
Andrews 1874 |
|
1940s |
Photochemical smog found to be causing crop damage |
Middleton 1950 |
|
1950s |
Ozone found to be major oxidant in photochemical smog VOC's and NOx shown to be ozone photochemical precursors |
Haagen-Smit 1952 |
|
1961 |
Basic science of ozone pollution documented in monograph |
Leighton 1961 |
|
1970 |
Clean Air Act of 1970 (CAA-70) establishes national program for the mitigation of ozone pollution in the United States. Sets 1975 as deadline for attainment of NAAQS |
|
|
1975 |
CAA-70 attainment deadline not met |
|
|
1977 |
Clean Air Act Amendments of 1977 (CAAA-77) establishes 1982 and 1987 as new deadlines for attainment |
|
|
1987 |
CAAA-77 attainment deadlines still not met |
|
|
1990 |
Clean Air Act Amendments of 1990 establishes new attainment deadlines extending into the 21st century and authorizes implementation of a reformulated gasoline program |
|
|
1997 |
New 8-hr, 80-ppb NAAQS for ozone promulgated |
EPA 1997a |
on average by about 1% per year from 1986 to 1997 (EPA 1998). On the other hand, the problem remains far from solved. In 1997, about 48 million people lived in 77 counties where ozone concentrations exceeded the second daily maximum 0.12-ppm, 1-hr NAAQS for ozone (EPA 1998). Of the 29 urban areas required by the Clean Air Act Amendments of 1990 to submit State Implementation Plans, 27 were unable to submit plans that showed attainment by the mandated date of 1998. Moreover, the promulgation of a new 8-hr, 80-ppb NAAQS for ozone in 1997 is expected to approximately triple the number of nonattainment counties
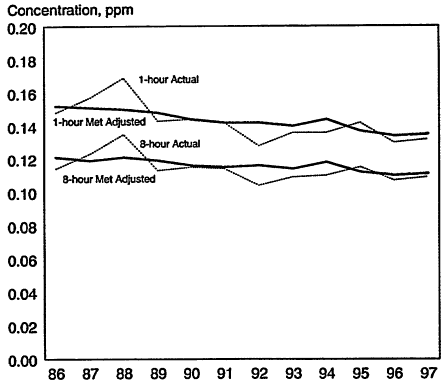
Figure 1-3
Comparison of actual (dotted lines) and meteorologically adjusted (solid lines) ozone trends in 1-hr and 8-hr 99th percentile ozone concentrations for the period of 1986-1997 across 41 metropolitan areas. Source: Adapted from EPA 1998.
and lead to widespread nonattainment in rural as well as urban areas of the eastern United States (Wolff 1996; Chameides et al. 1997). With the persistence of the ozone-pollution problem comes the need to develop new and innovative approaches to lowering ozone-precursor emissions. The federal RFG program is but one example of a new approach that is being promulgated to address this need.
In the formulation and testing of various blends of RFG, it became apparent that these blends could affect motor-vehicle emissions in various and subtle ways (AQIRP 1990; OTA 1990). In addition to affecting the total mass of VOC emissions, different RFGs could have different effects on the amounts of NOx and CO emitted by motor vehicles. They
could also affect the relative amounts of evaporative and exhaust emissions from motor vehicles and thus the chemical composition of the VOCs emitted by these vehicles. Controversy arose over whether the nation's traditional approach to assessing emissions, based on the mass of VOC emitted, was adequate to assess and regulate various RFG blends. With the use of ethanol as an oxygenated additive, such regulation proved to be especially contentious (e.g., EPA 1994). When compared with typical RFG blends using MTBE, blends using ethanol tend to have more evaporative VOC emissions but, it was argued, with a lower net ozone-forming potential. Accordingly, Senator Richard G. Lugar suggested that EPA establish a procedure to certify ethanol blends of RFG as equivalent to non-ethanol blends based on ozone-forming potential (see Appendix B, letter from Senator Richard G. Lugar dated October 17, 1995). EPA has not done so because it was unsure that there was an appropriate method for making such an assessment. Instead, EPA has commissioned this report to address the scientific and technical bases for such an assessment.
Charge to National Research Council Committee
Does RFG with ethanol as the oxygenate result in vehicular emissions with a lower ozone-forming potential than a blend with MTBE? Can a metric based on ozone-forming potential be reliably and robustly used to quantify the relative impacts of different RFG blends with different oxygenates on ozone pollution in the United States? As outlined above, these are the questions that motivated the formation of the National Research Council Committee on Ozone-Forming Potential of Reformulated Gasoline and this report. More specifically, the committee was given the task to assess whether the existing body of scientific and technical information is sufficient to permit a robust evaluation and comparison of the emissions from motor vehicles using different RFG blends based on their relative ozone-forming potentials; and the concomitant impact on air-quality benefits of the RFG program. The committee was asked to focus its assessment on the use of oxygenates in RFG, with specific attention to RFG blends using MTBE and ethanol.
The committee was asked to address the following specific issues:
- Assessment of the technical soundness of various approaches
- for evaluating and comparing the relative ozone-forming potentials of RFGs. Two prominent methods for assessing relative ozone impacts include relative reactivity factors and grid-based airshed models. Determine whether there is sound scientific basis for the use of reactivity factors, models, and/or any other approach(es) for evaluating the ozone-forming potential of RFGs in a nationwide program.
- Assessment of technical aspects of various air-quality issues when evaluating the relative ozone-forming potentials of RFGs. Air-quality issues to be considered include assessment of the ozone-forming potentials of RFGs for both peak (1-hr) and average (8-hr) ozone levels, inclusion or exclusion of CO as an ozone precursor, and consideration of changes in NOx emissions and the corresponding impact on ozone levels resulting from the use of different levels and/or types of oxygenates or other fuel composition changes.
- Assessment of the sensitivity of evaluations of the relative ozone-forming potentials of RFGs to factors related to fuel properties and the variability of vehicle technologies and driving patterns. Factors to be considered include assessment of effects of the fuel blending method (i.e., splash blending versus match blending), "distillation impact" and/or the "commingling effect," variability in fuel composition, engine operating conditions as they pertain to emissions, and changes in the exhaust-to-evaporative emissions ratio.6
The committee was asked to identify any gaps in the existing scientific and technical information, recommend how such gaps might be
filled, and identify the types of emission data that would be needed to continuously evaluate the ozone-forming potential of emissions from vehicles using RFG.
It is important to note that the committee was not asked to consider other issues related to the choice and use of various blends of RFG. Therefore, the committee has not addressed issues such as balance of trade, energy and cost requirements for fuel production, domestic sources of fuel versus foreign sources, human health and global environmental impacts, and use of renewable fuels versus nonrenewable fuels. In addition, it should be noted that this report is limited to the scientific and technical aspects of this issue; the possible design or implementation of regulations based on ozone-forming potential are not within the scope of this study.
Report Structure
In response to its charge, the committee's report addresses (1) how the ozone-forming potential of emissions from light-duty motor vehicles might be affected by the use of RFG blends with and without various types and concentrations of oxygenates; and (2) the extent to which current scientific and technical understanding and information are adequate to quantify these effects robustly. Although the focus is on the impacts of RFGs on ground-level ozone concentrations, RFG and the oxygenates added to these gasolines can also have impacts on other air-quality issues (e.g., toxics, carbon monoxide, and particulate matter); these other impacts are mentioned when they are relevant or potentially significant.
To provide a technical foundation for the assessment, the report provides overviews of the photochemistry of ozone, the concept of atmospheric reactivity and ozone-forming potential, motor vehicles as a source of ozone precursors, and RFGs in Chapters 2, 3, 4, and 5, respectively. In Chapter 6, the report assesses the likely magnitude of the air-quality benefits of the federal and California RFG programs (in total) based on observations. Chapter 6 also outlines the characteristics of a measurements program that could more robustly quantify the air-quality benefits and the changes in the ozone-forming potential of vehicular emissions arising from Phase II of the federal RFG program. The committee's assessment of RFG's overall impact on ozone serves as a calibration
point for the discussion in Chapter 7, which focuses on eight specific RFG blends to illustrate the methodology of, as well as the advantages and problems associated with, using ozone-forming potential m evaluate the relative impacts of these blends. Appendix A contains biographical information on the committee. Appendix B contains a letter from Senator Richard G. Lugar suggesting that EPA establish a procedure m certify ethanol blends of RFG as equivalent m methanol blends based on ozone-forming potential. Appendix C presents the equation set for EPA's Complex Model of Phase II of the federal RFG program, and Appendix D presents motor-vehicle-emissions data evaluated by the committee.