7
Using Ozone-Forming Potential to Evaluate the Relative Impacts of Reformulated Gasolines: A Case Study
As discussed in Chapter 6, reformulation of gasoline has the potential to substantially reduce the light-duty motor-vehicle (LDV) mass emissions of VOCs, NOx, and CO, as well as air toxics. Moreover, the emissions reductions resulting from the use of many of these formulations are sufficiently large to satisfy the requirements of the federal Phase II and California Phase 2 Reformulated Gasoline (RFG) programs. Thus, it is believed that the federal and California RFG programs will have a mitigating impact on ozone pollution, although various analyses suggest that the magnitude of the effect is not likely to be large (i.e., on the order of a few parts per billion) even if emissions from LDVs are underestimated by a factor of 2 or so.
This chapter turns to a more-subtle and more-difficult issue: namely, discerning the relative air-quality benefits of RFG blends using different amounts and types of oxygenated compounds. Because the mass of VOC emissions can be a misleading indicator of the ozone-forming potential of these emissions, the committee assessed the air-quality benefits of various RFG blends on the basis of the reactivity of these emissions as well as their mass. It should be noted at the outset, however, that this is a difficult task. Recall from Chapter 6, that the overall reduction in the reactivity of LDV emissions from the use of RFGs (over
that from conventional gasoline) is at most about 20%. The variation in the reactivity of emissions arising from various RFGs that differ in relatively minor ways (e.g., in oxygen content) is likely to be substantially smaller. On the other hand, recall from Chapter 3 that the uncertainty in the reactivities of a composite set of VOCs arising from a single source, such as motor vehicles, is probably also generally about 20%. Thus, a major challenge in this analysis was determining whether the difference in the reactivities of LDV emissions derived for two or more RFGs is statistically significant. In the analysis presented here, the committee adopted the so-called "paired t test"1 to make this determination.
In the sections that follow, a brief overview of the paired t test and its relationship to statistical uncertainty is provided. This methodology was applied to assess the statistical significance of differences in the LDV emissions arising from a subset of fuels studied by Auto/Oil Air Quality Improvement Research Program (AQIRP) and the California Air Resources Board (CARB). These fuels and their general properties are listed in Table 7-1 (and more detailed fuel properties are given in Table 6-1). Two approaches are used to estimate the LDV emissions from these fuels: one based solely on the experimental data arising from the emissions studies themselves, and the other using the Complex and Predictive Models. In order to assess the role of oxygenates and, more specifically, the relative roles of MTBE and ethanol, the subset of fuels included in this analysis was selected to provide a range of oxygen contents from 0 to 3.4% by weight (recall that the federal RFG program calls for a minimum oxygen content of 2% by weight), with this oxygen coming from MTBE or ethanol.
The subset of fuels used in this study were chosen to look for the effects of substituting MTBE by ethanol in otherwise closely similar fuels. Clearly, it would be preferable to use data on MTBE-containing and ethanol-containing fuels with the same fuel oxygen content or similar oxygenate volume percent, with all other chemical and physical properties (other than the presence of MTBE or ethanol) being the same. However,
TABLE 7-1 Properties of the Fuels Selected for the Case Studya
|
Fuel |
Ethanol (vol%) |
MTBE (vol%) |
Oxygen (wt%) |
RVP (psi)b |
|
AQIRP Phase Ic |
||||
|
F |
0 |
0 |
0 |
8.8 |
|
S |
0 |
0 |
0 |
8.0 |
|
U |
9.7 |
0 |
3.4 |
9.6 |
|
T |
9.7 |
0 |
3.4 |
9.3 |
|
N2 |
0 |
14.5 |
2.6 |
8.8 |
|
MM |
0 |
14.8 |
2.7 |
8.0 |
|
AQIRP Phase IId |
||||
|
C1 |
0 |
0 |
0 |
6.9 |
|
C2 |
0 |
11.2 |
2.0 |
6.8 |
|
California Ethanol Testing Programc |
||||
|
63 |
0 |
11.6 |
2.1 |
6.9 |
|
64 |
11.2 |
0 |
3.9 |
7.8 |
|
a See Table 6-1 for a more detailed tabulation of the fuel properties. b RVP (psi), Reid vapor pressure (pounds per square inch). c Fuel benzene, 1.4 ± 0.1 vol%; aromatics, 19.1-22.2 vol%; alkenes, 3.1-5.4 vol%; sulfur, 246-345 parts per million (ppm by wt). d Fuel benzene, 0.93-0.94 vol%; aromatics, 22.7-25.4 vol%; alkenes, 4.1-4.6 vol%; sulfur, 31-38 ppm by wt. e Fuel benzene, 0.82-0.83 vol%; aromatics, 23.3 vol%; alkenes, 4.8-4.9 vol%; sulfur, 32-34 ppm by wt. |
||||
the available database did not allow such a straightforward comparison; the fuels chosen were the best available to the committee and differ in the percent (by weight) of oxygen and the percent (by volume) of ethanol compared with MTBE (see Table 7-1).
Assessing Whether Emissions and Reactivity Differences are Statistically Significant
As discussed in Chapter 3, the calculation of reactivity for any given VOC or combination of VOCs can be in error for any number of reasons (e.g., errors in the chemical mechanism used to calculate the reactivity factors,
or errors in the speciation assumed for the VOC mixture). As a result, there is an uncertainty associated with the reactivity calculated for the emissions from any source, including those arising from the individual LDV using various blends of RFGs. The magnitude of the uncertainty in these reactivities is a crucial piece of information needed to decide Whether one RFG blend is preferable over another from an air-quality point of view.
The uncertainty in any measured parameter, including those related to LDV emissions, can arise from both random and systematic errors. Systematic error is defined as the difference between the true value of the quantity of interest and the value w which the mean of the measurements converges as more measurements are taken. These types of errors can arise from faulty experimental protocols or incorrect model assumptions, and introduce a bias into the results. Scientists and engineers always seek to eliminate all systematic errors. Nevertheless, the possibility of unidentified systematic errors can rarely be totally eliminated and, because they are often unidentified, they are difficult to quantify.
Random errors are somewhat easier to characterize by adopting a probabilistic or statistical approach. For example, take fuel a and fuel b and suppose that each fuel is tested on m different vehicles. On the basis of these m tests, the mean (or average) reactivity for each fuel can be calculated from
where ![]() is the mean reactivity for fuel x (x = a or b), and (Rx)i is the reactivity for fuel x obtained from test i. The variance is estimated by
is the mean reactivity for fuel x (x = a or b), and (Rx)i is the reactivity for fuel x obtained from test i. The variance is estimated by
Together ![]() and (sx)2 describe the probability that a new measurement of Rx will have a specific value, with
and (sx)2 describe the probability that a new measurement of Rx will have a specific value, with ![]() being the most probable value and sx describing the spread of values about
being the most probable value and sx describing the spread of values about ![]() . When the probability can be described by a probability density function (as in Figure 7-1),
. When the probability can be described by a probability density function (as in Figure 7-1),
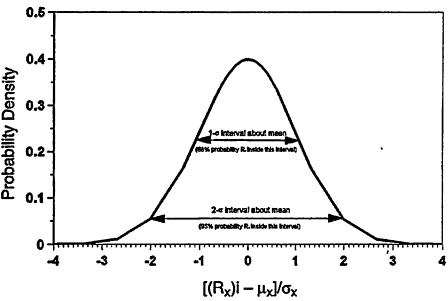
Figure 7-1
The probability density distribution for a population of reactivities, Rx
for fuel X about the population mean (μx) with a variance given by (σx)2.
there is a 68% probability that an additional measurement of Rx will lie between ![]() and
and ![]() and a 95% probability the measurement will lie between
and a 95% probability the measurement will lie between ![]() and
and ![]() .
.
Although sx defines the spread in the population of measured values of Rx, it does not define the uncertainty with which the mean reactivity, ![]() , is defined. To do this, the standard deviation of the mean2 is used:
, is defined. To do this, the standard deviation of the mean2 is used: ![]()
The 1-s confidence interval (i.e., the interval between ![]() and
and ![]() ) will contain the true or actual reactivity of fuel x 68% of the time and the 2sm,x confidence interval will contain the true reactivity 95% of the time.
) will contain the true or actual reactivity of fuel x 68% of the time and the 2sm,x confidence interval will contain the true reactivity 95% of the time.
For a decision maker confronted with choosing between two RFG blends on the basis of their reactivity, a critical question is whether the difference in the two reactivities is statistically significant. The answer to this question is closely tied to the magnitude of the standard deviation of the mean, sm,x,, for the two fuels. The smaller the values for sm,x,, the greater the likelihood of being able to establish that a small difference in reactivities is statistically significant. Thus, by inspection of Equation 7-3, we see that the most useful emissions studies for this purpose are those that involve a large number of (vehicle) tests and minimize the sources of random experimental error (e.g., from temporal fluctuations in laboratory conditions).
However, simply knowing the magnitudes of the sm,x values does not in and of itself provide the answer to the question of statistical significance. A set of rules must be adopted for deriding whether any similarity or difference in the reactivities of two RFG blends is in fact statistically significant. Typically, these rules include an appropriate type of statistical test and a selection of the level of confidence that will be required to certify statistical significance. Although the statistical test is an objective procedure, the setting of the level of confidence is a more-subjective exercise that relates to the concerns and priorities of the decision maker. In general, the decision maker must decide whether it is more important to avoid falsely concluding that a difference exists or to avoid falsely concluding that no difference exists. If a decision maker uses a difference in the mean reactivities measured for two fuels to implement a given control policy (e.g., choosing fuel a over fuel b on the basis of experimental data) but, in fact, there is no difference in the real world, the decision maker has committed a Type I error (falsely concluding that a difference exists). Such an error might not have a negative impact on air quality, but it could very well incur unnecessary economic costs. If on the other hand, the decision maker decides that the two reactivities are not significantly different and thus does not choose fuel a over fuel b when in fact the true reactivities are different, the decision maker has made a Type II error (falsely concluding that no difference exists). In this case, the error could have an unintended negative air-quality impact. Choosing which error is more important to avoid and
setting an acceptable level of risk of committing either error are policy decisions. The following discussion illustrates how one statistical approach can incorporate such choices when assessing fuels based on ozone-forming potential.
Consider an experiment in which motor vehicles are randomly selected for emissions testing. Each vehicle will be used to combust fuel a and fuel b, and the reactivities of the emissions are obtained, Ra and Rb respectively. The null hypothesis (typically denoted by statisticians as "Ho") is that there is no difference in the reactivities of emissions from a sampling of vehicles using fuel a versus fuel b, that is, μa = μb. The alternative hypothesis (denoted by statisticians as "Ha"), specifies that ![]() .
.
The two-tailed paired t test provides a methodology for determining the confidence or statistical probability that Ho can be rejected in favor of Ha or vice versa. One of the parameters calculated in a paired t test is the so-called "ρ value." This parameter can vary between 0 and 1 and increases as the difference in the emissions between two fuels becomes smaller and/or less statistically significant. It is defined as the probability that the null hypothesis, Ho is true, and it thus equal to (1 - probability) that Ho is false. Representative ρ values and the various probabilities implied by these values are listed in Table 7-2. For example, if the ρ value for a given paired t test is 0.05, there is a 5% probability that the null hypothesis is correct and a 95% probability that the null hypothesis is incorrect. (Another way of stating this is to say that the two reactivities are statistically different at the 95% confidence level.) On the other hand, if the ρ value for a given test is 0.95, there is 95% probability that the null hypothesis is correct, and so forth.
Because the ρ value is the probability that the null hypothesis is true, it is equivalent to the probability of making a Type I error (i.e., incorrectly choosing one fuel over another when there is in fact no difference in their emissions). Thus, when a small ρ value (reflecting large and significant differences in the reactivities of two fuels) is obtained, there is a relatively small probability of making a Type I error. In this case, the decision maker could choose the lower reactivity fuel with a high degree of confidence. On the other hand, when a large ρ value is obtained, a decision maker is likely to make a Type I error by choosing the fuel with the apparent, but not statistically significant lower reactivity.
In general, as the probability of making a Type I error increases, the probability of making a Type II error (i.e., not choosing the lower reac-
Table 7-2 Representative ρ Values and Associated Probabilities for a Two-Tailed t Test on Reactivities Ra and Rb
|
ρ Value |
Probability (%) That |
Probability (%) of Making Type I Error by Choosing One Fuel over the Other |
Probability (%) of Making Type I Error by Not Choosing One Fuel over the Other |
|
0.01 |
99 |
1 |
High |
|
0.05 |
95 |
5 |
|
|
0.1 |
90 |
10 |
|
|
0.15 |
85 |
15 |
|
|
0.2 |
80 |
20 |
|
|
0.4 |
60 |
40 |
Moderate |
|
0.6 |
40 |
60 |
|
|
0.8 |
20 |
80 |
Low |
tivity fuel) decreases. Thus, low r values imply a high probability of a Type II error if a decision maker decides to not choose the lower reactivity fuel, while high r values imply a low probability of a Type II error.
Fuels and Emissions Data from the AQIRP Study
As indicated in Table 7-1, eight fuels from the AQIRP study were selected for detailed analysis here: six from AQIRP Phase I and two from AQIRP Phase II. Collectively, the eight fuels provide a range of properties related to oxygen content and type of oxygenate. Fuel F, used in Phase I of the AQIRP, was an RFG with low aromatic content, low alkene content, low T90, and no oxygen. Fuel S was similar to fuel F, but with less butane, which resulted in a lower Reid vapor pressure (RVP). Approximately 10% ethanol was splash-blended into fuels F and S to form fuels U and T, respectively. As a result of this splash blending, the RVPs for fuels U and T were about I pound per square inch (psi) higher than the RVPs of fuels F and S. Fuels N2 and MM, on the other hand, contained oxygen but in the form of MTBE instead of ethanol. The MTBE was fully blended to the specifications of fuels F and S, respectively. As a result, no dilution effect on aromatic content, alkene content, or T90 was produced and the RVPs of fuels N2 and MM were identical to those of fuels F and S, respectively (Table 7-1). Fuel C2, used in AQIRP Phase II, was a low-sulfur RFG that contained MTBE and met the
1996 California Phase 2 regulatory requirements (see Chapter 5), whereas fuel C1 was a fuel blended to essentially the same requirements, but without MTBE.
Emissions for all Phase I fuels (F, S, U, T, N2, and MM) were measured using current-fleet vehicles. The Phase II fuels (C1 and C2), on the other hand, were tested using current-fleet vehicles, federal Tier I vehicles, and advanced-technology vehicles (see Chapter 4). It also should be noted that all of the vehicles in the AQIRP study were well-maintained and properly functioning and thus the data do not address the probable substantial contributions from high-emitting vehicles to overall precursor emissions.
Tables listing the LDV emissions from each of these fuels derived from the AQIRP data are presented in Appendix D. These data were gathered using the Federal Test Procedure (FTP) for exhaust and evaporative emissions according to the procedures described by Rueter et al. (1992) for the Phase I fuels and Burns et al. (1995) for the Phase II fuels. In the case of the Phase I fuels, data for exhaust, diurnal, and hot-soak emissions are presented. Although running-loss emissions were also measured for the Phase I study fuels, only a small number of tests were carried out (n = 6 for fuel F; 9 for fuel U; and 2 each for fuels S, T, N2 and MM) and the observed variations were very large (e.g., running-loss mass VOC emissions for the six vehicles tested with fuel U varied by a factor of ˜2,000). Accordingly, it is unlikely that these data are representative of the on-road vehicle fleet, and thus the running-loss data for these fuels are not considered here. Nevertheless, it should be borne in mind that high running losses due to fuel leakages and improper vehicular maintenance can be an important or even dominant source of VOC emissions from modem vehicles. In the case of the Phase II fuels, diurnal and running-loss emissions were not measured. Moreover, hot-soak emissions from fuel C1 were measured on only one advanced-technology vehicle and only three advanced-technology vehicles for fuel C2. Given this small sample size, the results of the hot-soak-emissions tests for this class of vehicles are not discussed here.
In addition to the mass of emissions, the tables in Appendix D indicate the total and specific reactivities3 of the emissions. The exhaust-
|
3 |
All reactivities discussed here are based on the maximum incremental reactivity (or MIR) scale and are derived using reactivity factors calculated from the SAPRC 1997 chemical mechanism. Similar conclusions are obtained using the SAPRC 1990 and SAPRC 1993 chemical mechanisms (see Chapter 3). |
emissions reactivities include carbon monoxide (CO). The committee has found that CO typically contributes 15% to 25% of the total exhaust emissions reactivity independent of the fleet type (i.e., current, federal Tier 1, or advanced technology). Thus, the contribution of CO to the exhaust reactivity is quite substantial and should not be neglected.
Before turning to an analysis of the differences in the emissions from the various fuels, it is relevant to note the rather large variability in the data from the AQIRP study. Inspection of the tables in Appendix D reveals that the mass of emissions (in units of grams per mile) measured for a given fuel often varied from one vehicle test to another by a factor of two or more and sometimes by more than a factor of five. This variability is perhaps not surprising in light of earlier discussions in this report of the myriad factors that can influence LDV emissions. Nevertheless, this large variability—compounded with the relatively small number of independent tests carried out for each fuel (typically less than 10)—tended to produce relatively large variances in the mean emissions for each fuel.
Given the substantial variability in emissions of the various vehicles tested with fuels A and B, the committee used logarithm[(emissions using fuel A) ÷ (emissions using fuel B)] for each vehicle used in the paired t test. The use of such an approach assumes, reasonably, that substituting fuel A for fuel B causes a constant fractional (or percentage) change in the emissions being considered (CO, NOx, VOC, etc.). When a number of tests was available for a given vehicle-fuel combination, an arithmetic mean was used for input into the logarithm [(emissions using fuel A) ÷ (emissions using fuel B)]. Obviously, only vehicles for which emissions tests were carried out using both fuels could be used in the paired t-test statistical analysis.
Effect of Reid Vapor Pressure
In addition to affecting the oxygen content, the presence of oxygenates (and especially ethanol) in gasoline can increase the fuel's RVP. Moreover, a primary effect of increasing the RVP of gasoline is to increase the evaporative emissions from LDVs. In the committee's assessment of the impact of oxygenates on RFG emissions, it would be useful, therefore, if one could separate out the effect of RVP increases from that of the addition of oxygen. Toward that end, it is instructive to assess what
effect increased RVP in the AQIRP fuels had on the emissions measured during that study. Inspection of Table 7-1 indicates that there are three fuel pairs with very similar properties except for their RVPs; comparison of the emissions from these pairs thus provides an opportunity to assess the effect of RVP observed by AQIRP. The fuel pairs are
- Fuel S (oxygen = 0%, RVP = 8.0 psi) vs. fuel F (oxygen = 0%, RVP = 8.8 psi).
- Fuel MM (oxygen = 2.7% using MTBE, RVP = 8.0 psi) vs. fuel N2 (oxygen = 2.6% using MTBE, RVP = 8.8 psi).
- Fuel T (oxygen = 3.4% using ethanol, RVP = 9.3 psi) vs. fuel U (oxygen = 3.4% using ethanol, RVP = 9.6 psi).
The reader will note that while the first two fuel pairs have a 0.8-psi difference in RVP, the third pair has only a 0.3-psi difference in RVP. Thus, if RVP has an effect on emissions, one might expect to find a larger difference in the emissions from the first two pairs compared with the third.
A comparison of the exhaust, diurnal, and hot-soak emissions of these fuel pairs, and the statistical significance of the differences in terms of the ρ values are presented in Tables 7-3, 7-4, and 7-5, respectively. Little evidence of a statistically significant effect of RVP is seen from these tables. In most cases, the r values were well above the 0.05 threshold to establish 95% confidence. However, Table 7-3 indicates a consistent decrease in CO emissions for the use of lower-RVP fuels. This observation is in agreement with the findings of Reuter et al. (1992), whose findings were based on eleven fuels in the AQIRP Phase I study (including the six used here) selected to investigate the effects of RVP and oxygenates on vehicular emissions. Reuter et al. (1992) found that, after combining the results from nonoxygenated fuels with fuels containing MTBE or ethanol, a 1.0-psi reduction in RVP resulted in a reduction in exhaust CO emissions of 9.1% (significant at the 95% confidence levels). On the other hand, some unexpected (even curious) results appear. For example, although the major effect of lower RVP is thought to be to lower evaporative emissions, the data presented here by no means confirm this trend. In fact, for each emissions category, lower RVP is associated with higher diurnal or hot-soak emissions in at least one of the three fuel pairs considered here. In the case of hot-soak emissions, a lower RVP fuel produced a higher reactivity that was significant at the 93% confidence level.
TABLE 7-3 Effect of RVP on Exhaust Emissions from Three AQIRP Fuel Pairs
|
Fuel Pair |
% Decrease in Emissions Attributable to Lower RVP1 |
ρ Value2 |
Summary |
|
A. Effect on mass of VOC emissions (g/mi) |
|||
|
S/F |
-9 |
0.2 |
Data from fuel pairs inconsistent. In first case, ˜80% probability that higher RVP fuel has lower emissions; in the second, >95% probability that lower RVP has lower emissions; and in the third, >60% probability that RVP has no effect on emissions. |
|
MM/N2 |
11 |
0.04 |
|
|
T/U |
1 |
0.6 |
|
|
B. Effect on mass of CO emissions (g/mi) |
|||
|
S/F |
7 |
0.3 |
Data indicate >40% probability that lower RVP fuel has lower emissions. Probability of Type I error small. |
|
MM/N2 |
19 |
0.009 |
|
|
T/U |
10 |
0.4 |
|
|
C. Effect on total reactivity (g O3/mi) |
|||
|
S/F. |
-7 |
0.5 |
No consistent, statistically significant effect apparent. |
|
MM/N2 |
12 |
0.2 |
|
|
T/U |
1 |
0.70 |
|
|
D. Effect on mass of NOx emissions (g/mi) |
|||
|
S/F |
-7 |
0.1 |
No significant and consistent effect apparent. |
|
MM/N2 |
6 |
0.8 |
|
|
T/U |
˜0 |
0.2 |
|
|
1 % decrease = [(emissions with low RVP) - (emissions with high RVP)] ÷ (emissions with high RVP). Negative value indicates an emissions or reactivity increase with lower RVP. 2 Based on logarithms of means. |
|||
Effect of Oxygenates Using MTBE
Inspection of Table 7-1 indicates that there are three fuel pairs that can be used to assess the effect of adding MTBE to gasoline, two from AQIRP Phase I and one from AQIRP Phase II. In each case, the fuel pairs have essentially identical RVPs, and as a result, the comparisons are well-
TABLE 7-4 Effect of RVP on Diurnal Emissions from Three AQIRP Fuel Pairs
|
Fuel Pair |
% Decrease in Emissions Attributable to Lower RVP1 |
ρ Value2 |
Summary |
|
A. Effect on mass of VOC emissions (g/test) |
|||
|
S/F |
36 |
0.02 |
Nonoxygenated fuels show significant decrease in emissions with lower RVP. However, data for oxygenated fuels do not consistently confirm this trend. |
|
MM/N2 |
-3 |
0.3 |
|
|
T/U |
16 |
0.2 |
|
|
B. Effect on total reactivity (g O3/test) |
|||
|
S/F |
29 |
0.01 |
Nonoxygenated fuels show significant decrease in reactivity of emissions with lower RVP. However, data for oxygenated fuels do not consistently confirm this trend. |
|
MM/N2 |
-8 |
0.1 |
|
|
T/U |
13 |
0.3 |
|
|
1 % decrease = [(emissions with low RVP) - (emissions with high RVP)] ÷ (emissions with high RVP). Negative value indicates an emissions or reactivity increase with lower RVP. 2 Based on logarithms of means. |
|||
TABLE 7-5 Effect of RVP on Hot-Soak Emissions from Three AQIRP Fuel Pairs
|
Fuel Pair |
% Decrease in Emissions Attributable to Lower RVP1 |
ρ Value2 |
Summary |
|
A. Effect on mass of VOC emissions (g/test) |
|||
|
S/F |
16 |
0.7 |
No consistent statistically significant effect of RVP on emissions is apparent. |
|
MM/N2 |
-20 |
0.1 |
|
|
T/U |
9 |
0.03 |
|
|
B. Effect on total reactivity (g O3/test) |
|||
|
S/F |
16 |
0.8 |
No consistent statistically significant effect of RVP on reactivity of emissions is apparent. |
|
MM/N2 |
-31 |
0.07 |
|
|
T/U |
7 |
0.1 |
|
|
1 % decrease = [(emissions with low RVP) - (emissions with high RVP)] ÷ (emissions with high RVP). Negative value indicates an emissions or reactivity increase with lower RVP. 2 Based on logarithms of means. |
|||
suited to isolating the effect of the oxygenated additive. The fuel pairs are
- Fuel N2 (oxygen = 2.6% using MTBE, RVP = 8.8 psi) vs. fuel F (oxygen = 0%, RVP = 8.8 psi).
- Fuel MM (oxygen = 2.7% using MTBE, RVP = 8.0 psi) vs. fuel S (oxygen = 0%, RVP = 8.0 psi).
- Fuel C2 (oxygen = 2% using MTBE, RVP = 6.8 psi) vs. fuel C1 (oxygen = 0%, RVP = 6.9 psi).
With regard to fuel pair C2 and C1, it should be noted that (1) comparisons can be made with three types of vehicles—current fleet, Tier I, and advanced technology—and (2) no data are available for diurnal emissions.
A comparison of the exhaust, diurnal, and hot-soak emissions of these fuel pairs, and the statistical significance of the differences are presented in Tables 7-6, 7-7, and 7-8, respectively. As in the previous comparisons, there is little evidence here to suggest a statistically significant effect of MTBE. In most of the emissions categories, the various fuel pairs produced conflicting results, with the MTBE fuel having lower emissions (or reactivity) in some cases and higher emissions (or reactivity) in other cases. The most consistent effect is an increase in NOx exhaust emissions from MTBE (Table 7-6D). There are also suggestions of an increase in the mass and reactivity of hot-soak VOC emissions (Table 7-8B), as well as a decrease in CO exhaust emissions (Table 7-6B) from the addition of MTBE.
Effect of Ethanol vs. MTBE
There are two fuel pairs (both from AQIRP Phase I) that provide an indication of the relative effects of using MTBE or ethanol as an oxygenate in RFG. These are
- Fuel T (oxygen = 3.4% using ethanol, RVP = 9.3 psi) vs. fuel MM (oxygen = 2.7% using MTBE, RVP = 8.0 psi).
- Fuel U (oxygen = 3.4% using ethanol, RVP = 9.6 psi) vs. fuel N2 (oxygen = 2.6% using MTBE, RVP = 8.8 psi).
Note that in both cases, the ethanol-blended fuel had about the same
TABLE 7-6 Effect of MTBE on Exhaust Emissions from Three AQIRP Fuel Pairs
|
Fuel Pair |
% Decrease in Emissions Attributable to MTBE1 |
ρ Value2 |
Summary |
|
A. Effect on mass of VOC emissions (g/mi) |
|||
|
N2/F |
-2 |
0.9 |
Fuel-pair MM/S indicates a significant benefit of adding MTBE. However, no statistically significant effect is apparent from other fuel pairs. |
|
MM/S |
16 |
0.005 |
|
|
C2/C1 |
|
|
|
|
Current fleet |
-1 |
0.5 |
|
|
Tier I |
2 |
0.6 |
|
|
Adv. technol. |
6 |
0.3 |
|
|
B. Effect on mass of CO emissions (g/mi) |
|||
|
N2/F |
-2 |
0.2 |
No consistent, statistically significant effect is apparent. |
|
MM/S |
11 |
0.4 |
|
|
C2/C1 |
|
|
|
|
Current fleet |
10 |
0.7 |
|
|
Tier I |
1 |
0.2 |
|
|
Adv. technol. |
7 |
0.3 |
|
|
C. Effect on total reactivity (g O3/mi) |
|||
|
N2/F |
-5 |
0.8 |
No consistent, statistically significant effect is apparent. |
|
MM/S |
14 |
0.07 |
|
|
C2/C1 |
|
|
|
|
Current fleet |
1 |
0.2 |
|
|
Tier I |
3 |
0.6 |
|
|
Adv. technol. |
6 |
0.2 |
|
|
D. Effect on mass of NOx emissions (g/mi) |
|||
|
N2/F |
-17 |
0.05 |
Data suggest an increase in NOx emissions from the addition of MTBE in all but advanced-technology vehicles. Likelihood of Type I error is small. |
|
MM/S |
-3 |
0.4 |
|
|
C2/C1 |
|
|
|
|
Current fleet |
-6 |
0.7 |
|
|
Tier I |
-11 |
0.1 |
|
|
Adv. technol. |
2 |
0.9 |
|
|
1 % decrease = [(emissions with MTBE) - (emissions without MTBE)] ÷ (emissions without MTBE). Negative value indicates an emissions or reactivity increase with the addition of MTBE. 2 Based on logarithms of means. |
|||
TABLE 7-7 Effect of MTBE on Diurnal Emissions from Three AQIRP Fuel Pairs
|
Fuel Pair |
% Decrease in Emissions Attributable to MTBE1 |
ρ Value2 |
Summary |
|
A. Effect on mass of VOC emissions (g/test) |
|||
|
N2/F |
29 |
0.03 |
Data are not consistent. |
|
MM/S |
-15 |
0.3 |
|
|
C2/C1 |
No data |
- |
|
|
B. Effect on total reactivity (g O3/test) |
|||
|
N2/F |
13 |
0.1 |
Data do not indicate a consistent effect. |
|
MM/S |
-33 |
0.06 |
|
|
C2/C1 |
No data |
- |
|
|
1 % decrease = [(emissions with MTBE) - (emissions without MTBE)] ÷ (reactivity without MTBE). Negative value indicates an emissions or reactivity increase with addition of MTBE. 2 Based on logarithms of means. |
|||
oxygen content as that of the MTBE-blended fuel and thus less volume percent oxygenate (see Table 5-2). A comparison of ethanol and MTBE-blended fuels with similar volume percent oxygenate, but different oxygen content, is provided by the data from the California Ethanol Test Program discussed in the next section. It is also relevant to note that the ethanol-blended fuels had about a 1-psi higher RVP than the MTBE-blended fuel.
A comparison of the exhaust, diurnal, and hot-soak emissions of these fuel pairs, and the statistical significance of the differences are presented in Tables 7-9, 7-10, and 7-11, respectively. As in the previous two comparisons, the data presented here from the AQIRP study on the relative benefits of ethanol and MTBE are by no means conspicuous or striking. There is a suggestion that ethanol (or the higher RVP it engendered in the fuels considered here) caused somewhat higher VOC exhaust and evaporative emissions. In each case, however, the effect of ethanol on the reactivity of the emissions was less than its effect on the mass of the VOC emissions. Finally it is relevant to note that an analysis of air toxic emissions from the AQIRP fuels considered here suggests that there are advantages and disadvantages related to the use of either oxygenate (see Text Box 7-1). The above conclusions concerning the
TABLE 7-8 Effect of MTBE on Hot-Soak Emissions from Three AQIRP Fuel Pairs
|
Fuel Pair |
% Decrease in Emissions Attributable to MTBE1 |
ρ Value2 |
Summary |
|
A. Effect on mass of VOC emissions (g/test) |
|||
|
N2/F |
8 |
0.10 |
Data are not consistent but, overall, indicate a small probability that addition of MTBE causes an increase in hot-soak emissions. |
|
MM/S |
-32 |
0.02 |
|
|
C2/C1 |
|
|
|
|
Current fleet |
-23 |
0.4 |
|
|
Tier I |
-17 |
0.8 |
|
|
Adv. technol. |
Insufficient |
data |
- |
|
B. Effect on total reactivity (g O3/test) |
|||
|
N2/F |
12 |
0.7 |
Data are not consistent but, overall, indicate a small probability that addition of MTBE causes an increase in the reactivity of hot-soak emissions. |
|
MM/S |
-38 |
0.02 |
|
|
C2/C1 |
|
|
|
|
Current fleet |
-11 |
0.9 |
|
|
Tier I |
-8 |
0.5 |
|
|
Adv. technol. |
Insufficient data |
- |
|
|
1 % decrease ÷ [(emissions with MTBE) - (emissions without MTBE)] ÷ (emissions without MTBE). Negative value indicates an emissions or reactivity increase with addition of MTBE. 2 Based on logarithms of means. |
|||
effects of RVP, MTBE, and ethanol on vehicle emissions are generally consistent with those reported by Dunker et al. (1996) from a modeling study of the impacts of different gasoline fuels on ozone levels in the Los Angeles, Dallas-Ft. Worth, and New York urban areas using the AQIRP data and the Urban Airshed Model.
Fuels and Emissions Data from the California Ethanol Testing Program
Because of the limited number of tests made in AQIRP that directly compared emissions from MTBE-containing and ethanol-containing fuels, the data from that study provide only fragmentary information on the
TABLE 7-9 Effect of Ethanol vs. MTBE on Exhaust Emissions from Two AQIRP Fuel Pairs
|
Fuel Pair |
% Decrease in Emissions Attributable to Ethanol1 |
ρ Value2 |
Summary |
|
A. Effect on mass of VOC emissions (g/mi) |
|||
|
T/MM |
-11 |
0.04 |
Some indication that ethanol might cause higher VOC mass emissions. |
|
U/N2 |
˜ |
0.08 |
|
|
B. Effect on mass of CO emissions (g/mi) |
|||
|
T/MM |
-11 |
0.6 |
No consistent, statistically significant effect is apparent. |
|
U/N2 |
1 |
0.4 |
|
|
C. Effect on total reactivity (g O3/mi) |
|||
|
T/MM |
-8 |
0.4 |
No consistent, statistically significant effect is apparent. |
|
U/N2 |
5 |
0.4 |
|
|
D. Effect on mass of NOx emissions (g/mi) |
|||
|
T/MM |
2 |
0.4 |
No statistically significant effect is apparent. |
|
U/N2 |
7 |
0.4 |
|
|
1 % decrease = [(emissions with ethanol) - (emissions with MTBE)] ÷ (emissions with MTBE). Negative value indicates an emissions or reactivity increase with the addition of ethanol. 2 Based on logarithms of means. |
|||
TABLE 7-10
Effect of Ethanol vs. MTBE on Diurnal Emissions from Two AQIRP Fuel Pairs
|
Fuel Pair |
% Decrease in Emissions Attributable to Ethanol1 |
ρ Value2 |
Summary |
|
A. Effect on mass of VOC emissions (g/mi) |
|||
|
T/MM |
-12 |
0.43 |
Data indicate probability that ethanol causes higher mass emissions. |
|
U/N2 |
-38 |
0.01 |
|
|
B. Effect on total reactivity (g O3/mi) |
|||
|
T/MM |
5 |
0.81 |
Data are not consistent, but most likely effect is an increase in reactivity of emissions from ethanol. |
|
U/N2 |
-18 |
0.02 |
|
|
1 % decrease = [(emissions with ethanol) - (mission with MTBE)] ÷ (emissions with MTBE). Negative value indicates an emissions or reactivity increase with the addition of ethanol. 2 Based on logarithms of means. |
|||
TABLE 7-11 Effect of Ethanol vs. MTBE on Hot-Soak Emissions from Two AQIRP Fuel Pairs
|
Fuel Pair |
% Decrease in Emissions Attributable to Ethanol1 |
ρ Value2 |
Summary |
|
A. Effect on mass of VOC emissions (g/mi) |
|||
|
T/MM |
-14 |
0.29 |
Data indicate > 30% probability that causes higher mass emissions. |
|
U/N2 |
-50 |
0.003 |
|
|
B. Effect on total reactivity (g O3/mi) |
|||
|
T/MM |
1 |
0.72 |
Data are not consistent, but the most likely effect is an increase in reactivity of emissions from ethanol. |
|
U/N2 |
-40 |
0.002 |
|
|
1 % decrease = [(emissions with ethanol) - (emissions with MTBE)] ÷ (emissions with MTBE). Negative value indicates an emissions or reactivity increase with the addition of ethanol. 2 Based on logarithms of means. |
|||
|
TEXT BOX 7-1 Effect of Oxygenates on Toxic Air Contaminant Emissions Exhaust and evaporative emissions of selected air toxics from LDVs using the six AQIRP Phase I fuels are listed in Table 7-12. The data suggest that the fuels result in similar emissions of 1-3 butadiene and benzene (i.e., they fall within the observed variability as indicated by the standard deviations of the means). However; there appear to be differences in acetaldehyde and formaldehyde emissions that at least border on being, larger than the observed variability. In the case of acetaldehyde exhaust emissions, the ethanol-containing fuels produce about a factor of 2 larger exhaust emissions than that of the MTBE-containing and Oxygen-free fuels. On the Other hand, the ethanol-containing fuels tend to result in somewhat lower exhaust emissions of formal dehyde. It is also interesting to note that while MTBE-containing fuels are generally thought to result in enhanced exhaust emissions of formaldehyde (see Chapter 6), this trend is not reflected in the data presented in Table 7-12. |
TABLE 7-12 Toxic Air Contaminant Emissions from AQIRP Phase I Fuels
|
Fuel |
Exhaust Formaldehyde (mg/mi) |
Exhaust Acetaldehyde (mg/mi) |
Exhaust 1,3-Butadiene (mg/mi) |
Exhaust Benzene (mg/mi) |
Diurnal Benzene (mg/test) |
Hot-Soak Benzene (mg/test) |
|
F (0% oxygen, RVP = 8.8 psi) |
1.65 ± 0.6 |
0.88 ± 0.3 |
0.70 ± 0.2 |
8.1 ± 3 |
7.8 ± 4 |
15.0 ± 4 |
|
S (0% oxygen, RVP = 8.0 psi) |
1.40 ± 0.6 |
0.88 ± 0.6 |
0.82 ± 0.3 |
8.8 ± 5 |
6.2 ± 4 |
11.2 ± 5 |
|
U (3.4% oxygen using EtOH, RVP = 9.6 psi) |
0.99 ± 0.3 |
1.38 ± 0.4 |
0.73 ± 0.3 |
7.7± 4 |
7.8 ± 6 |
16.2 ± 6 |
|
T (3.4% oxygen using EtOH, RVP = 9.3 psi) |
1.13 ± 0.4 |
2.07 ± 1.0 |
0.76 ± 0.3 |
7.8 ± 4 |
7.0 ± 6 |
15.6 ± 6 |
|
N2 (2.6% oxygen using MTBE, RVP = 8.8 psi) |
1.28 ± 0.6 |
0.74 ± 0.5 |
0.81 ± 0.4 |
8.3 ± 4 |
5.5 ± 6 |
10.1 ± 6 |
|
MM (2.7% oxygen using MTBE, RVP = 8.0 psi) |
1.71 ± 0.9 |
0.84 ± 0.6 |
0.73 ± 0.3 |
6.7 ± 3 |
5.7 ± 4 |
12.1 ± 5 |
|
Uncertainties are 1 standard deviation of the mean. |
||||||
relative benefits of the two types of oxygenates in RFG. Fortunately, the California Ethanol Testing Program produced a considerably more detailed database on this issue. During the program, multiple tests were made to characterize the emissions from LDVs using a fuel with MTBE (fuel 63) and a comparable fuel with ethanol (fuel 64). As indicated in Table 7-1, fuel 63 contained 2.1% oxygen by weight from MTBE and had an RVP of 6.9 psi; fuel 64 contained 3.9% oxygen by weight from ethanol and had an RVP of 7.8 psi. In all other respects (e.g., benzene, aromatic, and sulfur content), the fuels were essentially identical. Thus, a comparison of the two fuels directly addresses the question of whether the tendency for ethanol to increase RVP can be overcome by the addition of more oxygen.
Exhaust emissions in the program were measured using the FTP (Calvert et al. 1993) and the Rep05 test procedure.4 The evaporative emissions measured were hot-soak and 0-24-hr and 24-48-hr diurnal emissions (see Chapter 4). No running-loss emissions were measured but were estimated using an emissions model. Because this model is proprietary, its performance could not be assessed by this committee, and as a result, these emissions estimates are not included in this report. In addition to measurements of the mass of VOC, NOx, CO, and toxic emissions, the emissions of hydrocarbons, alcohols, carbonyls, and aldehydes were speciated; thus making possible reactivity calculations for the nonmethane organic gases (NMOGs). A detailed summary of the reactivities of the exhaust and evaporative emissions from fuels 63 and 64 are presented in Appendix D. These reactivities were calculated using the MIR scale. The reactivity factors used are found in the California Test Procedure, adopted by CARB in July 1992 and last amended June 24, 1996.
Fourteen vehicles of model years 1990 to 1995 were selected and classified according to engine family, evaporative family, and emissions-control technologies and then used to characterize emissions from the fuels. Acceptance criteria for each vehicle were based upon a protocol developed by the In-Use Compliance Section of CARB. This protocol consisted of a telephone questionnaire, a 10-point inspection of the vehicle, and restorative maintenance. The purpose of restorative maintenance was to bring the vehicle into manufacturer's specification and to ensure that all electrical and mechanical controls are functioning prop-
erly. Further requirements were that the vehicles pass a smog check, not exceed specified mileage limits set for the different model years included in the study, and it be obtained from rental fleets rather than private owners if possible.
Exhaust and evaporative emissions from each of the 14 vehicles with both fuels 63 and 64 were measured two times, and in some cases, three times. (However, tests involving two of the vehicles were discarded due to nonstatistical errors.) Thus, the mean emissions from each of the fuels for each emissions category were derived from almost 30 separate tests, a much larger number than that typically used to derive the mean emissions from the AQIRP data discussed in the previous sections. The larger number of tests in the California Ethanol Testing Program should make these data more amenable to discerning subtle differences in the emissions from each fuel.
The analyses suggest that the reactivity of the exhaust emissions for the ethanol-blended fuel was about 4% less than that of the MTBE-blended fuel. That decrease is essentially all attributable to an approximate 10% decrease in the mass of CO exhaust emissions for the ethanol-blended fuel. However, this relatively small decrease in the reactivity of the exhaust emissions was overwhelmed by the much larger increase in the mass and reactivity of the evaporative VOC emissions arising from the use of ethanol-blended fuel. As a result, the reactivity of the combined exhaust and evaporative emissions using the ethanol-blended fuel was estimated by CARB to be about 17% larger than those using the MTBE-blended RFG. The committee analyzed data obtained from the California Ethanol Testing Program before publication of CARB's (1998) analysis and before data on the reactivity of CO emissions were available. The committee compared the reactivities of emissions from fuels 63 and 64 using a two-sample t test (see Table 7-13 and Appendix D). Since the committee completed its analysis, CARB published its results of a more-comprehensive analysis of the data from the California Ethanol Testing Program. Although the results of CARB's analysis are somewhat different from those of the committee, the overall conclusions are the same.
Is there A Difference Between Conclusions Drawn on the Basis of Voc-Mass Emissions and the Reactivity of the Emissions?
In Chapter 3, we noted that because of the wide range of VOC species typically emitted by LDVs and the highly variable chemistry of these
TABLE 7-13 Effect of Ethanol vs. MTBE on Total Reactivity (g O3/mi or g O3/test) of Emissions from Fuels 63 and 64 of the California Ethanol Testing Program
|
Emissions Type |
% Decrease in Reactivity Attributable to Ethanol1 |
ρ Value2 |
Statistical Summary |
|
Exhaust based on FTP composite |
-9 |
0.50 |
No consistent, statistically significant effect is apparent |
|
Exhaust based on Rep-05 |
5 |
0.3 |
|
|
Hot Soak |
-73 |
0.002 |
>99% probability that difference in reactivity is significant. |
|
0-24 Diurnal |
-60 |
0.004 |
>99% probability that difference in reactivity is significant. |
|
24-48 Diurnal |
-82 |
0.002 |
>99% probability that difference in reactivity is significant. |
|
1 % decrease = [(emissions with ethanol) - (emissions with MTBE)] ÷ (emissions with MTBE). Negative value indicates an emissions or reactivity increase with the addition of ethanol. 2 Exhaust reactivities did not include CO. |
|||
compounds, the mass of VOC emissions might be a poor metric for the ozone-forming potential of these emissions. Under some circumstances a reactivity scale might provide a more-reliable measure. In light of this situation, it is interesting to consider whether the conclusions drawn above with regard to the relative benefits of ethanol and MTBE are affected by which metric is used.
Inspection of the data in Tables 7-3 through 7-13, as well as those provided in Appendix D, suggest that the two metrics did in fact produce some differing results. For example, note in Tables 7-10 and 7-11 that the mass of evaporative emissions from AQIRP fuels with ethanol are greater than those from fuels with MTBE. However, for one of the fuel pairs considered, the difference is cut by more than a factor of 2 when measured on the basis of reactivity; in the case of the other fuel pair, the reactivity from the ethanol-containing fuel is actually found to be less than that of the MTBE-containing fuel. However, in this latter case, the difference in both the mass and reactivity of emissions was not statistically significant.
A contrasting result was obtained for hot-soak emissions from the fuels in the California Ethanol Testing Program. In this case, the ethanol-
containing fuel also has a larger mass of emissions than the MTBE-containing fuel. However, this difference was further enhanced when the reactivity of the emissions was considered.
Despite these differences, however, it is important to note that, in none of these instances did the use of the reactivity metric fundamentally alter the conclusions that would have been reached if the mass-emissions metric had been adopted. For example, note in Table 7-10 that fuel U (containing ethanol) was found to produce higher emissions than fuel N2 (containing MTBE) at a greater than 95% confidence level regardless of the metric used; the inconsistency between the two metrics is only in the magnitude of the difference between the fuels. In the case of fuels T and MM on the other hand, the mass-emissions metric indicates higher emissions for the ethanol-containing fuel while the reactivity metric indicates lower emissions for the ethanol-containing fuels. However, in these cases the ρ values are relatively large, and thus the differences in the mass and reactivity of the emissions from two fuels are not statistically significant.
Analysis Using the Complex and Predictive Models
The analyses presented in the preceding two sections could perhaps be criticized for being based on test results from a limited number of fuels, and thus not representative of a fleet-wide response to changes in fuel composition. Indeed, in the case of the AQIRP study the smallness of the sample size limited the ability to unequivocally conclude that oxygenates had, or did not have, an impact. Other researchers (Mayotte et al. 1994) also find some indications of an impact, but warn that their sample size was limited as well.
As noted in Chapter 4, both EPA and CARB have conducted statistical analyses of a much larger number of tests to develop models to predict how the mass of VOC and NOx emissions respond to fuel-composition changes. (Recall that EPA's model is called the Complex Model, and CARB's is called the Predictive Model). The databases used to develop both models are similar. The major differences are in the statistical treatment of the data, and that the Complex Model has a separate segment for high-emitting vehicles (CARB 1991).
As a final check on the applicability of the results discussed above, the properties of the 10 fuels listed in Table 7-1, as well as the California Phase 2 reference fuel, were input into the Complex and Predictive
Models. The resulting exhaust and evaporative emissions predicted by the Complex Model are given in Table 7-14 and the percentage decrease in exhaust emissions predicted by the Predictive Model, relative to the reference fuel, are listed in Table 7-15.5 Because neither the Complex nor the Predictive Models estimate the composition of the emissions, these models cannot be used to predict changes in the reactivity of the emissions.
TABLE 7-14
VOC and NOx Emissions for Various Fuels Predicted by EPA's Complex Modela
TABLE 7-15 VOC, CO, and NOx Exhaust Emissions Changes for Various Fuels Predicted by CARB's Predictive Model and Draft CO Modela
Turning first to the results from the Complex Model, one finds two striking results: (1) the sizable reductions in exhaust emissions arising from low sulfur fuels; and (2) the increase in evaporative emissions with ethanol-containing fuels (presumably from the increased RVP of these fuels). This later result is far more definitive than, although not inconsistent with, the effect of ethanol discerned from the direct analysis of the AQIRP data discussed earlier in this chapter. On the other hand, the small and borderline significant increases in NOx exhaust emissions and evaporative VOC emissions, as suggested in the AQIRP data, associated with the addition of MTBE are not reflected in the results of the Complex Model.
Like the Complex Model results, the Predictive Model indicates that reducing sulfur content reduces emissions of all components. The model also projects a decrease in CO emissions from the addition of oxygen—an effect that was also seen in the analysis of the emissions data from the AQIRP fuels.
Summary
An analysis of emissions data and regression-model predictions for a limited set of RFG blends with a range of properties, that include different oxygen contents of MTBE and ethanol, suggests that:
- The differences inferred between the VOC emissions of two fuels using the mass of emissions as a metric varied on occasion with that inferred using reactivity as a metric. In some cases, consideration of reactivity decreased the apparent emissions difference and in other cases it enhanced the difference. However, in no case did the fundamental conclusion concerning the choice of one fuel over another (for the fuels studied here), on the basis of statistically significant air-quality benefits, change as a result of using a mass-emissions or reactivity-weighted metric.
- CO emissions account for 15% to 25% of the reactivity of exhaust emissions from LDVs and thus should be included in reactivity assessments because CO contributes to ozone formation due to its large amount of emissions.
- The addition of MTBE or ethanol appears to have only a small effect on the exhaust emissions of RFGs. The most substantial of these appears to be related to the emissions of CO and air toxics. Data from AQIRP suggest that ethanol-containing fuels lead to greater exhaust emissions of acetaldehyde than do fuels with MTBE, but less formaldehyde. Data from the California Ethanol Testing Program indicate that the exhaust emissions from vehicles using ethanol-containing fuels are about 10% lower than those arising from vehicles using fuels with MTBE. There is also some indication that oxygenates in fuels lead to somewhat higher emissions of NOx—an effect that could have undesired impacts on air quality in rural areas and on regional scales.
- Ethanol-containing fuels tended to have significantly higher evaporative emissions (on both a total-mass basis and a reactivity-weighted basis) than MTBE-containing fuels. This is likely due, at least in part, to the fact that ethanol fuels tend to have an approximate 1-psi-higher RVP than the equivalent MTBE fuel. Moreover, the increase in the evaporative emissions from the ethanol-containing fuels was significantly larger than the slight benefit obtained from the lowering of the CO exhaust emissions using the ethanol-containing fuel.
- Based on the findings presented above, the committee concludes that the use of commonly available oxygenates in RFG has little impact on improving ozone air quality. Also, use of an ethanol-containing RFG with a 1-psi-higher RVP is likely to produce a negative air-quality impact. This conclusion is consistent with CARB's evaluation in 1998 that led to its decision to not allow a 1-psi waiver for ethanol-containing fuels (CARB 1998).
Two important caveats should be noted. The first relates to the fact that the analysis presented here is based solely on data gathered from well-maintained vehicles with properly working catalytic converters. As noted in Chapters 4 and 6, there is substantial evidence to suggest that high-emitting motor vehicles (perhaps because of malfunctioning catalytic converters or faulty evaporative controls) can contribute disproportionately to the VOC and CO emissions arising from a fleet of LDVs, and the response of high-emitting vehicles to ethanol-blended and MTBE-blended RFG has yet to be fully characterized. For example, one might speculate that oxygen in the fuel would provide a greater emissions benefit for high emitters with faulty catalytic converters than for ordinary vehicles. Because ethanol fuels often contain more oxygen than the equivalent MTBE fuel, this might tend to offset the disadvantages of ethanol-containing fuels implied in the committee's analysis. However, the few data on this subject that are currently available are inconclusive (e.g., see Knepper et al 1993; Mayotte et al. 1994). Moreover, for high-emitting vehicles with faulty evaporative controls, the use of ethanol-blended RFG with a higher RVP would most likely lead to elevated evaporative emissions. For these reasons, the committee recommends that the effect of RFG on emissions from high-emitting vehicles be studied in greater detail.
The other caveat relates to the overall effect on ozone pollution that might arise from the emissions differences projected here for MTBE-containing and ethanol-containing RFG blends. Recall from the committee's earlier analyses that the overall effect of RFGs might be an approximate 20% reduction in the reactivity of LDV emissions and a few parts-per-billion reduction in peak ozone concentrations. After combining exhaust and evaporative emissions, the use of ethanol, as opposed to MTBE, as an oxygenate would lead to a decrease in the effectiveness of RFGs but not a total cancellation. The net effect on ozone concentrations would be extremely small and almost certainly not discernable from the ambient ozone concentration data.




























