◆ ◆ ◆ ◆ ◆
Executive Summary
After extensive consultations with key Russian officials and scientific leaders and drawing on the experience gained through the initiation of six pilot projects at two Russian facilities to investigate the practical aspects of cooperation, the National Academy of Sciences Committee on U.S.-Russian Cooperation on Dangerous Pathogens recommends a five-year Pathogens Initiative, followed by a second phase of sustained joint U.S.-Russian research and related efforts. The program will support collaboration on the epidemiology, prevention, diagnosis, and therapy of diseases associated with dangerous pathogens that pose serious public health threats, as well as related fundamental research. The Pathogens Initiative will engage a substantial number of highly qualified specialists from the former Soviet biological weapons complex and will serve important U.S. national security and public health goals.
CONTEXT
Rapid advances in the biological sciences and biotechnology hold the promise of dramatically improving human health, agriculture, and other aspects of life. The broad diffusion of knowledge and capabilities enables many countries to benefit from these advances.
The spread of biotechnology, however, is also accompanied by significant risks. The capabilities associated with research on dangerous human, animal, and plant pathogens represent a complex dual-use technology; some of the knowledge of medicine, agriculture, and biotechnology overlaps with the knowledge necessary to use pathogens for hostile purposes. In addition, some equipment and facilities are versatile. Certain types of vaccine facilities, for example, could be converted to produce biological agents for use by military forces or terrorists.
The international community has responded to the threat of biological weapons (BW) by constructing an international regime—based on the Geneva Protocol and the Biological Weapons Convention (BWC) and supplemented by the Australia Group's export control guidelines—to ban their use, development, stockpiling, and production and to prevent countries and subnational groups from acquiring them. Activities focused on a particular country, such as Iraq, also are part of the international effort to forestall or respond to the proliferation of BW.
The BWC, however, lacks verification provisions and contains only limited procedures for addressing a suspected violation. Achieving a broad consensus on strengthening the international regime in this critical area is impeded by a number of factors, not least of which is a deep-seated lack of trust between Western countries and Russia concerning BW-related activities. In 1992, Russia acknowledged that the Soviet Union had maintained a BW program involving activities that violated the BWC, thereby confirming long-standing Western suspicions. At that time, President Yeltsin declared that illegal activities had ceased and future work in violation of the BWC was prohibited, but the Russian government has been unable to convince the United States that Russia is now in complete compliance with its treaty obligations.
Adding to these uncertainties is the sheer size of the former Soviet BW complex, which Russia is finding difficult to maintain financially, whatever the intended purpose of the facilities may now be.
Although in disrepair in many respects, this complex remains, raising fears in the West that dangerous materials, equipment, and know-how could be misused or could leak to parties of proliferation concern. Encouraging Russia to reconfigure some of these facilities to carry out peaceful work on dangerous pathogens and to convert the others to peaceful use not connected with dangerous pathogens is thus an important aspect of U.S. nonproliferation policy.
THE IMPORTANCE OF U.S.-RUSSIAN COOPERATION ON DANGEROUS PATHOGENS
Russia will undoubtedly continue to support legitimate research and related activities on dangerous pathogens. U.S. involvement in these activities through cooperative programs will provide opportunities to build confidence that prohibited research is not being pursued under the guise of legitimate undertakings. Also, from a public health viewpoint, Russian scientists who participated in former Soviet BW program have a unique knowledge of many aspects of naturally occurring pathogens as well as those that could be used by terrorist groups.
The committee believes that appropriately structured U.S.-Russian cooperation on dangerous pathogens—featuring direct laboratory-to-laboratory contacts and based on the principle of broad transparency—will serve the interests of both countries. Such cooperation could contribute significantly to the following objectives:
-
National security benefits
-
Providing greater mutual confidence about compliance with the BWC than would otherwise be possible
-
Reducing proliferation incentives
-
Serving as a stepping stone to dismantlement opportunities
-
Reconfiguring former Soviet BW-related activities
-
Enhancing capabilities to combat bioterrorism
-
-
Public health benefits
-
Improving understanding of the prevalence and characteristics of pathogens that pose threats to public health
-
Strengthening capabilities to prevent, diagnose, and treat outbreaks of infectious diseases
-
Enhancing international communications concerning disease trends and outbreaks
-
-
Economic benefits
-
Improving the stability of Russian research institutes by increasing the commercial viability of their research products
-
Leveraging limited national financial and human resources to serve national security public health
-
Providing new opportunities for the U.S. private sector to become more active in Russia
-
-
Scientific benefits
-
Enhancing the base of fundamental knowledge about pathogenesis
-
Increasing the international availability of research results.
-
Even the most extensive collaboration between Russian and U.S. scientists will not provide incontrovertible assurance that all research activities on dangerous pathogens are devoted to legitimate purposes. Recognizing this risk, the committee has concluded that, governed by appropriate rules of transparency, a cooperative program can be carried out in a manner to ensure that the risk of abuse of such a program is reduced to an acceptable level.
Expanded arrangements to provide sufficient transparency should include mutual agreement on a project-by-project basis concerning the activities that are legitimate; regular and agreed-upon access to
facilities, personnel, and information; and commitment to the principle that providing assurance is an active rather than a passive responsibility. Moreover, during the evaluation of any joint research project, its potential contributions to health and national security must be judged to outweigh the risk that the project might contribute to the development or improvement of offensive BW capabilities.
The committee recommends that the proposed program be undertaken in close coordination with related bilateral activities (see Table E-1).
Table E-1 Selected Organizations with Program Interests Related to the Pathogens Initiative
|
Organization |
Description |
|
International Science and Technology Center (ISTC) |
This international organization, established by the United States, the European Union, Japan, and Russia, has supported a number of projects at a variety of institutes involved in the former Soviet BW program, including projects that address dangerous pathogens. ISTC has received dozens of other well-developed, but yet-to-be-funded proposals in this area. In addition, it has sponsored several symposia on related topics. |
|
Initiatives for Proliferation Prevention (IPP) |
This program of the U.S. Department of Energy recently announced its intention to support biotechnology proposals at Russian institutes, particularly Biopreparat institutes, that have potential for commercial markets. IPP has received dozens of proposals from a variety of Russian institutions, including proposals for research on dangerous pathogens. |
|
U.S. Civilian Research and Development Foundation (CRDF) |
This private foundation, established by Congress in 1992 and set up by the National Science Foundation in 1995, has solicited basic research proposals from interested Russian investigators in the biomedical field. CRDF currently supports eight projects at institutes that were part of the former Soviet BW complex. The future of additional competitions is subject to further funding. |
|
National Aeronautics and Space Administration (NASA) |
NASA has funded several projects at Biopreparat institutes in support of its space science activities. The future of these types of projects in Russia is uncertain. |
|
Centers for Disease Control and Prevention (CDC) |
The CDC has long-standing relationships with several Russian institutes, some of which participated in the former Soviet BW program. It has participated in activities in Russia supported by the U.S. Agency for International Development and frequently provides training for Russian scientists. |
|
National Institutes of Health (NIH) |
NIH provides grant supplements to its U.S. investigators to involve international scientists in their projects. NIH also provides training opportunities for international specialists in its laboratories. Russian researchers who participated in former Soviet BW activities can apply to these programs. |
|
U.S. Department of Agriculture (USDA) |
The USDA supports a limited number of biotechnology projects in Russia that are directly linked to agriculture development. Its current Russian portfolio does not include research on dangerous pathogens. |
ESTABLISHING THE BASIS FOR EXPANDED COOPERATION
The committee's consultations with a range of Russian officials, research managers, and laboratory scientists helped ensure that assessments of the technical basis for cooperation were authoritative and realistic. In addition, these interactions resulted in Russian specialists, acquiring a sense of genuine partnership in the development of the recommended program. Consultations included scientific visits to Russian research institutes in Koltsovo and Obolensk, where pilot projects were later established; one round of consultations and two joint planning meetings in Moscow; and an international symposium in the Kirov region involving 30 Russian specialists, sponsored by the National Academy of Sciences (NAS) and the International Science and Technology Center (ISTC).
Of particular importance is the reported endorsement by the Russian Defense Council of the NAS initiative. Such support would be critical to the future success of a cooperative program involving defense scientists.
Biopreparat—an organization originally established by the Soviet government to provide a wide range of BW-related research, production, and support services—was the principal point of contact in Russia for the NAS committee. Specialists from many Biopreparat facilities and other Russian organizations participated in these consultations.
The committee was not successful in its efforts to meet with specialists from the Russian Ministry of Defense (MOD), but efforts to engage MOD should continue. Several Russian officials have expressed optimism that MOD will eventually participate in bilateral cooperative activities, either directly or in partnership with Biopreparat institutes. Although MOD participation is highly desirable, the Biopreparat complex provided much of the critical research and development support for the Soviet program; thus, the committee believes that cooperation with Biopreparat in and of itself will make a valuable contribution to achieving the benefits mentioned above.
In parallel with these consultations, the committee initiated six pilot projects at two Russian facilities and is developing two more (see Box E-1) to gain experience conducting collaborative research projects. The projects have contributed useful insights at the scientist-to-scientist level into the capabilities of the two countries. The pilot projects also were important in convincing Russian colleagues that the NAS undertaking was a serious endeavor with strong backing from the U.S. government, thereby encouraging them to participate actively in planning a long-term program.
|
Box E-1 Pilot Projects Initiated by NAS and Financed by DOD The following projects were under way as of July 1997, with funds committed to Russian institutionsa : At the State Research Center for Virology and Biotechnology, “Vector,” Koltsovo
The following projects were being processed by ISTC as of October 1997: At the State Research Center for Virology and Biotechnology, “Vector,” Koltsovo
At the State Research Center for Applied Microbiology, Obolensk:
|
NOTE: CDC = Centers for Disease Control and Prevention; DOD = Department of Defense; USAMRIID = U.S. Army Medical Research Institute of Infectious Diseases.
a Funds committed to U.S. collaborating institutions are CDC, $47,000; USAMRIID, $20,000; Harvard University, $9,000.
The NAS committee used the following criteria in selecting the pilot projects:
-
Scientific importance of the topic;
-
Quality of the proposal;
-
Quality or capacity of the principal investigator, research team, and facilities;
-
Provision for strong U.S. collaboration;
-
Engagement of former Soviet BW expertise; and
-
Promotion of transparency.
The committee also made the judgment that each project's potential contributions to public health or U.S. national security interests outweigh the risk that the project might contribute to the development or improvement of offensive BW capabilities.
The pilot projects were limited efforts, and the committee concluded that the following additional criteria should be considered in the selection of projects within the larger program recommended in this report:
-
Likelihood of sustaining the research by attracting the interest of other organizations with financial capabilities to continue work in the general field after completion of the project and
-
Promotion of linkages between Russian scientists working in institutions that had been involved in BW activities and those that were not involved in such activities.
The committee also strongly recommends that if future joint activities are pursued, U.S. specialists should adopt a more proactive role in identifying possible research topics and proposals for funding. The framework for collaboration presented in this report is designed to promote this goal.
The NAS became a partner of the ISTC in Moscow, which provides an important administrative framework for processing and reviewing proposals, monitoring projects, and dispensing funds within Russia. Of special importance are ISTC procedures for distributing funds for salaries directly to individual researchers, thereby circumventing opportunities for intermediaries to divert a portion of the funds for unintended uses.
Drawing on this first-hand experience, the committee developed three overarching principles for guiding bilateral activities:
-
Projects should be collaborative in design and conduct.
-
Only projects that are of interest to specialists in both countries should be undertaken.
-
All projects should be conducted on the basis of cooperation, not assistance, with each side making intellectual, financial, and in-kind contributions.
-
All relevant constituencies in both countries should be able to apply for participation in the program.
-
-
Projects should be designed and conducted in a way that maximizes transparency.
-
Activities should be carried out in an environment of openness.
-
Direct contacts among specialists should be stressed.
-
A central coordination point within each government should be apprised of cooperative activities.
-
-
Results of cooperative projects should be disseminated to the widest possible interested audience.
-
Whenever possible, research results should be promptly published or made available to international audiences through other channels.
-
Intellectual property and sensitive findings should be protected.
-
Intellectual property rights resulting from cooperative activities should be shared by the participating institutions on fair and equitable terms.
-
PHASE 1: A PATHOGENS INITIATIVE
Although Russian interest in cooperation in this field is increasing, the future political course in Russia remains difficult to predict. As cooperation becomes more ingrained in the Russian scientific community, joint efforts are more likely to survive political shocks, thus underscoring the importance of establishing and broadening cooperation while the window of opportunity is open.
The core of a Pathogens Initiative should be joint research projects directed to the epidemiology, prophylaxis, diagnosis, and therapy of diseases associated with dangerous pathogens as well as related fundamental research. According to Russian colleagues, if the U.S. government decides to support such an initiative, early intergovernment endorsement of the program could encourage MOD to participate. In addition, such political support could help resolve many policy, implementation, and budget issues confronting Biopreparat and other interested organizations in both countries.
The committee recommends seven program areas as the initial framework for the program. The first five area—anthrax, melioidosis and glanders, plague, orthopox virus, and viral hemorrhagic fevers—are agents or diseases that have been linked with BW activities for many years. In each of these areas the Soviet government is believed to have invested substantial financial resources to carry out
research that is largely unknown outside that country. Organizations in the United States also have good research capabilities to help combat the infectious diseases of interest.
Two additional program areas will provide opportunities to address other pathogens or diseases of public health concern and to carry out related fundamental research. These two categories are particularly important in both providing support for key Russian scientists who are interested in pursuing careers not tied directly to potential BW agents and expanding the pool of potential collaborators in the United States.
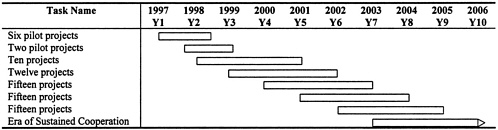
A five-year program that builds to a level at which 15 three-year projects are initiated each year, involving an average of 10 full-time Russian specialists per project, could engage a substantial number of leading Russian specialists in the field and most of the key Russian research facilities. See Table E-2 for the phasing of collaborative research projects.
Projects will be selected on a competitive basis by using the criteria set forth above. The resources devoted to each program area should depend on the quality of project proposals across all areas. The pilot projects fall into several areas, and the possibility of expanding these limited efforts should be considered if the results are promising.
Several supporting activities could effectively complement the research programs. Specifically, upgrading the communications capabilities of selected Russian institutes, improving the safekeeping and utilization of strain collections used as national reference standards, and expanding exchanges of information on biosafety requirements and practices are appropriate areas of cooperation.
In addition to annual reviews of all projects, the overall approach will be evaluated in depth at the end of the second year and adjusted as necessary.
PHASE 2: AN ERA OF SUSTAINED COOPERATION FOLLOWING THE PATHOGENS INITIATIVE
Recognizing that sustained cooperation must be accompanied by rules of disclosure and other measures designed to provide assurance that work is devoted strictly to legitimate purposes, the committee developed a model for progressive development of suitable transparency arrangements. The model is intended to emphasize the importance of transparency and to stimulate the official deliberations necessary to work out agreed-upon provisions. The model calls for an intergovernment mechanism to provide direction for collaborative efforts on a broad front.
Joint research projects would continue to be the core of long-term cooperation. Expanded cooperation in epidemiology and rapid response to outbreaks of infectious disease would promote trust between the two countries. Related to the expanded international exchange of data, the internal capabilities of Russia to assess and process epidemiologic information would have to be strengthened. During outbreaks of diseases, specialists from the two countries should collaborate in providing their
most relevant information and offering technical support to each other. Also, Russian specialists who are involved in field investigations should be encouraged to apply for participation in the training programs of the Centers for Disease Control and Prevention (CDC).
In addition, the promotion of effective national regulatory approaches to controlling dangerous pathogens appears to be essential to ensure responsible handling of these pathogens on a broad front. Effective enforcement procedures and sharing of experiences are particularly important in developing mutual trust.
At the same time, the need to prevent the dissemination of sensitive information to parties of proliferation concern and to protect intellectual property rights would continue to be important. The Russian and U.S. governments should adopt appropriate procedures to ensure that these issues are addressed in a manner that does not undercut the broader transparency objectives of cooperative endeavors.
COSTS
To build on current momentum, the committee recommends that the U.S. Department of Defense (DOD) promptly provide financial support for the Pathogens Initiative. Sustained funding for the longer-term Phase 2 program would undoubtedly require agreement between the executive branch and Congress about a line item in the budget of a selected agency. It is premature to speculate which department or agency should have long-term financial responsibility.
Pathogens Initiative
The projected costs to the United States of the Pathogens Initiative are as follows: fiscal year (FY) 1998, $6 million; FY 1999, $7 million; FY 2000, $8.5 million; FY 2001, $8.5 million; and FY 2002, $8.5 million. Most of these funds are earmarked for direct project support, as shown in Table E-3. When the Pathogens Initiative is fully developed in FY 2000, the costs will be $4.5 million (53 percent) for the Russian research teams, $2.5 million (29 percent) for the U.S. collaborators, $500,000 (6 percent) for project development activities, and $1 million (12 percent) for program evaluation, financial management, and related support activities.
Table E-3 Allocation of Funds per Fiscal Year During the Pathogens Initiative (in millions of dollars)
|
FY |
Budget |
New Projects |
Total Projectsa |
Funds to Russian Research Teams |
Funds to U.S. Collaborator |
Funds to Project Management |
|
1998 |
6.0 |
10 |
10 |
3.5 |
1.5 |
1.0 |
|
1999 |
7.0 |
12 |
22 |
4.0 |
2.0 |
1.0 |
|
2000 |
8.5 |
15 |
37 |
4.5 |
2.5 |
1.5 |
|
2001 |
8.5 |
15 |
42 |
4.5 |
2.5 |
1.5 |
|
2002 |
8.5 |
15 |
45 |
4.5 |
2.5 |
1.5 |
|
a This total does not include pilot projects. |
||||||
Phase 2: An Era of Longer-Term Sustained Cooperation
As indicated above, it is assumed that the size of the program will grow steadily and then level off during the era of sustained cooperation. Under the model suggested as a goal for expanded efforts, the estimated annual costs to the United States beginning in FY 2003 are $5 million (50 percent) to support U.S. collaborators; $2 million (20 percent) to support selected aspects of Russian participation in activities of special interest to the United States; $1 million (10 percent) for project development,
evaluation, and related activities; and $2 million (20 percent) for support for the intergovernment mechanism and related specialized committees to oversee the entire activity. The total U.S. contribution would be $10 million per year and Russian institutions would be expected to cover most of their own costs.
ANTICIPATED RESULTS OF THE PROGRAM
The proposed joint efforts could build a considerable level of trust between the scientific communities of Russia and the United States in a way that would help change the tone of diplomacy on the international security aspects of dangerous pathogens. Such efforts could have profound effects–both direct and indirect–in reducing the threats of proliferation and terrorism. Also, the program will make many contributions to combating dangerous infectious diseases, while serving as a model for global efforts when the dangers of new and reemerging diseases are being recognized more fully in many countries.
One likely effect of such a multiyear program in Russia is a structural adjustment of its research enterprise dealing with dangerous pathogens. Research projects will be increasingly concentrated at a handful of the best institutions, which would become centers of excellence. To the extent that other institutions remain viable, they should be motivated to find work outside the area of dangerous pathogens.
Thus, there is a high probability this program will help achieve DOD objectives of nonproliferation and reconfiguration of the former Soviet BW complex into a less diffuse, less uncertain, and more public health oriented establishment.