10
Biofilms in Drinking Water Distribution Systems: Significance and Control
Mark W. LeChevallier
A biofilm is a collection of organic and inorganic, living and dead material collected on a surface. It may be a complete film or, more commonly in water systems, small patches on pipe surfaces. Biofilms in drinking water pipe networks can be responsible for a wide range of water quality and operational problems. Biofilms can be responsible for loss of distribution system disinfectant residuals, increased bacterial levels, reduction of dissolved oxygen, taste and odor changes, red or black water problems due to iron or sulfate-reducing bacteria, microbial-influenced corrosion, hydraulic roughness, and reduced materials life (Characklis and Marshal, 1990).
Microorganisms in biofilms can include bacteria (including coccoid round, rod-shaped, filamentous, and appendaged bacteria), fungi, and higher organisms like nematodes, larvae, and Crustacea. Recently, researchers have shown that viruses and parasites like Cryptosporidium can be trapped in biofilms. Although viruses and Cryptosporidium do not grow in a biofilm, they can attach to biofilms after a contamination event. Therefore, it is important to thoroughly flush the distribution system to remove these organisms following a contamination event.
A primary reason that many water utilities become concerned with biofilms in drinking water systems is due to growth of coliform bacteria in the pipe network. In 1993 in the United States alone, nearly 4,400 water systems affecting 21 million people violated drinking water standards for total coliform bacteria (Pontius, 1995). Similar trends were noted for 1994 and 1995, with over 12,000 systems exceeding accepted coliform levels. Of concern are the nearly 2,000 systems every quarter that are significant noncompliers and repeatedly detect coliform bacteria in finished drinking water. Although some of these systems experience coliform occurrences due to cross connections and other operational defects, a large proportion of the systems can trace their problems to regrowth of the bacteria in distribution system biofilms.
Factors Related to Coliform Occurrences
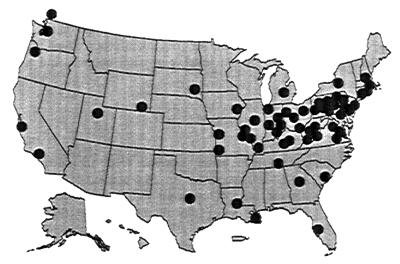
Recent studies have examined data from over 90 water systems (see Figure 10-1) to determine the factors that contribute to the occurrence of coliform bacteria in drinking water (LeChevallier et al., 1996; Volk et al., 1996). These studies have shown that the occurrence of coliform bacteria can be related to the following factors: filtration, temperature, disinfectant type and residual, assimilable organic carbon (AOC) level, corrosion control, and pipe material selection.
Filtration
Four unfiltered surface water systems were included in one study (LeChevallier et al., 1996) and accounted for 26.6 percent of the total number of bacterial samples collected but 64.3 percent (1,013 of 1,576) of the positive coliform samples. Although the results do not suggest that treatment was inadequate (e.g., coliforms were not related to breakthrough of treatment barriers), the data suggested that filtration may be an important factor in preventing coliform regrowth. Following the study, one of the systems installed filtration and distribution system coliform levels were reduced by a factor of three over the following 18-month interval.
Temperature
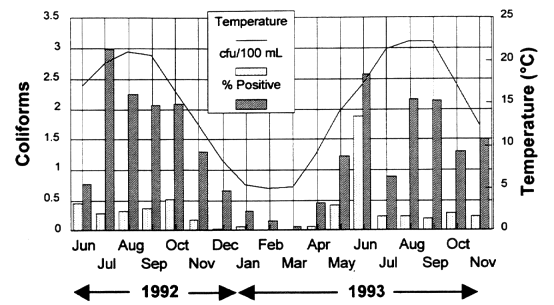
On average, the occurrence of coliform bacteria was significantly higher when water temperatures were >15ºC (See Figure 10-2). However, the minimum temperature at which microbial activity was observed varied from system to system. Systems that typically experienced cold water had increases in coliform occurrences when water temperatures ranged near 10ºC. The strains of coliform bacteria in these systems may be better adapted to grow at lower temperatures (psychrophiles).
Disinfectant Residual and Disinfectant Level
For filtered systems there was a difference between systems that maintained a free chlorine residual and systems that used chloramines. For systems that used free chlorine, 0.97 percent of 33,196 samples contained coliform bacteria, while 0.51 percent of 35,159 samples from chloraminated systems contained coliform bacteria (statistically different at p < .0001). The average density of coliform bacteria was 35 times higher in free chlorinated systems as compared to chloraminated water (0.60 colony-forming units CFUs/100 ml for free chlorinated water, compared to 0.017 CFUs/100 ml for
chloraminated water). Previous research (LeChevallier et al., 1990; LeChevallier, 1991) has hypothesized that chloramines may be able to better penetrate into distribution system biofilms and inactivate attached bacteria. The fact that different disinfectants may interact differently with biofilms can be related to their different mechanisms of action. Free chlorine, for example, is known to react with natural organic matter to form trihalomethanes (Rook, 1974). Chloramines do not form these products to the same degree. Free chlorine attacks the cytoplasmic membrane of gram-negative bacteria to produce a cellular lesion (injury) that results in an increased sensitivity to surfactants (Zaske et al., 1980). Chloramines do not produce the same type of injury as free chlorine, and the chloramine lesion can be reversed with a reducing agent (sodium sulfite) (Watters et al., 1989). The penetration of free chlorine into a biofilm has been modeled and shown to be limited by its fast reaction rate (LeChevallier, 1991; DeBeer et al., 1994). Essentially free chlorine is consumed before it can react with the bacterial components of the film (Chen and Stewart, 1996). Chloramines, on the other hand, are slower reacting and can diffuse into the biofilm and eventually inactivate attached bacteria. This mechanism has been elegantly demonstrated by researchers at Montana State University using an alginate bead model (Chen and Stewart, 1996). Stewart and colleagues (in press) showed that free chlorine effectively did not penetrate alginate beads containing bacterial cells but that chloramines did penetrate into the alginate material and reduced bacterial levels nearly 1 million-fold over a 60-minute interval (2.5 mg/L chloramines, pH 8.9).
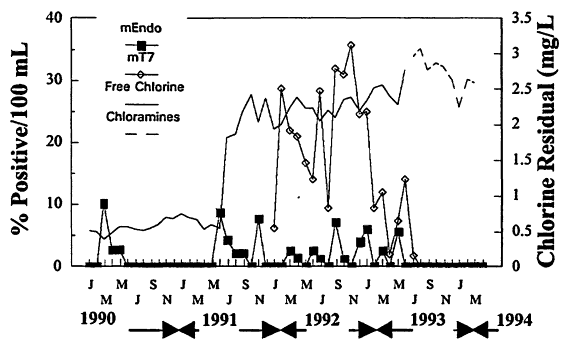
The effectiveness of a chloramine residual for controlling coliform occurrences suspected to be the result of biofilm growth in distribution pipelines is shown in Figure 10-3. The system experienced coliform occurrences even when free chlorine residuals averaged between 2 and 2.5 mg/L in the distribution system. Use of m-T7 medium, a technique that recovers injured bacteria (LeChevallier et al., 1983), showed coliform occurrence rates ranged between 10 and 40 percent even during months when coliforms were not recovered on the standard m-Endo medium. Conversion of the disinfectant to chloramines in June 1993 resulted in dramatic decreases in coliform occurrences measured by both m-Endo and m-T7 media, and the bacteria have not been detected in the finished drinking water for the three years following the change (Norton and LeChevallier, 1997).
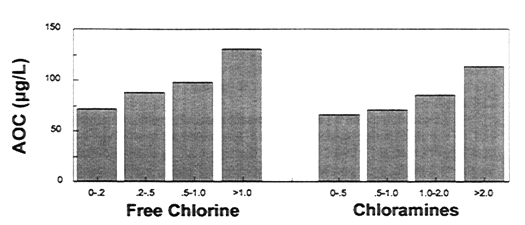
In addition to the type of disinfectant used, the residual maintained at the end of the distribution system was also associated with coliform occurrences (LeChevallier et al., 1996). Systems that maintained dead-end free chlorine levels <0.2 mg/L, or monochloramine levels <0.5 mg/L, had substantially more coliform occurrences than systems maintaining higher disinfectant residuals. However, systems with high AOC levels needed to maintain high disinfectant residuals to control coliform occurrences (see Figure 10-4). Therefore, maintenance of a disinfectant residual alone did not ensure that treated waters would be free of coliform bacteria.
AOC Level
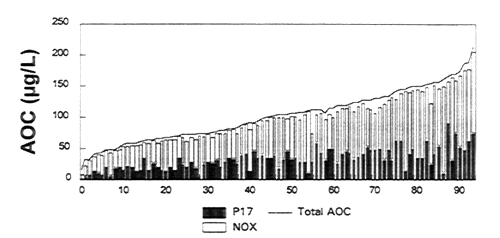
The combined results from two surveys of AOC levels in North American drinking water systems (LeChevallier et al., 1996; Volk et al. 1996) are shown in Figure 10-5. The levels (summarized as the geometric mean based on 12 to 36 samples) range from 20 to 214 μg/L. The results also indicate that the majority of the total AOC results from the growth of the test organism, Spirillum sp. strain NOX. This AOCNOX fraction is influenced by disinfection practices (chlorine, ozone, etc.) and suggests that changes in these practices (i.e., type of disinfectant, point of application, dose) can impact AOC levels in finished drinking water.
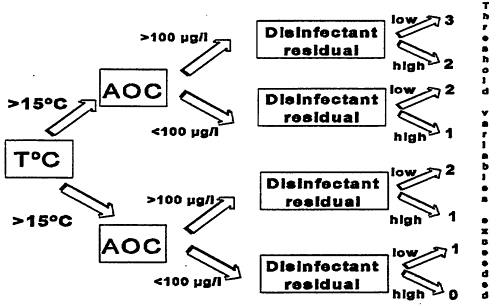
High levels of AOC can stimulate bacterial growth in distribution system biofilms (LeChevallier et al., 1996; Volk et al., 1996). On average, free chlorinated systems with AOC levels greater than 100 μg/L had 82 percent more coliform-positive samples, and the coliform densities were 19 times higher than free-chlorinated systems with average AOC levels less than 99 μg/L. However, high levels of AOC alone do not dictate the occurrence of coliform bacteria in drinking water but are only one factor. Figure 10-6 illustrates a decision tree that graphically depicts combinations of threshold values above which the probability of coliform occurrence is increased (Volk et al., 1996). As more of the threshold values are exceeded, the probability of coliform occurrences is increased. Data summarized in Table 10-1 show that the frequency of coliform occurrence was less than 2 percent when no threshold criteria were exceeded and increased to 16 percent when all three criteria were exceeded. The magnitude (number of positive samples per event) also increased with a greater exceedance of threshold criteria. Similar models developed for specific systems have yielded higher predictive probabilities (Volk and Joret, 1992).
TABLE 10-1
Relationship Between Threshold Criteria and Coliform Occurrence
|
No. Positive Criteria |
Total No. of Events |
Coliform-Positive Samples |
No. of Coliform Episodes |
Frequency of Coliform Observation (%) |
|
0 |
160 |
3 |
3 |
1.9 |
|
I |
292 |
18 |
15 |
5 |
|
2 |
191 |
24 |
16 |
8.4 |
|
3 |
62 |
26 |
10 |
16 |
Corrosion Control and Pipe Materials
Most systems do not measure corrosion rates on a daily basis, so this parameter is difficult to evaluate full-scale. However, recent research has demonstrated that corrosion of iron pipes can influence the effectiveness of chlorine-based disinfectants for inactivation of biofilm bacteria (LeChevallier et al., 1990, 1993). Therefore, the choice of pipe material and the accumulation of corrosion products can dramatically impact the ability to control the effects of biofilms in drinking water systems.
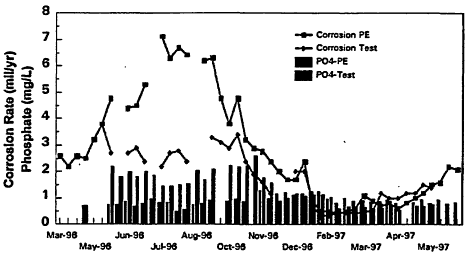
Figure 10-7 shows average monthly corrosion rates (in milles "thousandth of an inch" per year) from a system in Illinois. The conventional plant effluent corrosion rate showed marked seasonal variations. Corrosion rates were the highest during the summer months when, traditionally, the incidence of coliform occurrences are the highest (Figure 10-2). Similar seasonal variations have been observed in other systems (Norton and LeChevallier, 1997).
This variation in corrosion rates is important because the corrosion products react with residual chlorine, preventing the biocide from penetrating the biofilm and controlling bacterial growth. Studies have shown that free chlorine is impacted to a greater extent that monochloramine, although the effectiveness of both disinfectants is impaired if corrosion rates are not controlled (LeChevallier et al., 1990, 1993). Increasing the phosphate-based corrosion inhibitor dose, especially during the summer months, can help reduce corrosion rates (Figure 10-7). In full-scale studies, systems that used a phosphate-based corrosion inhibitor had lower coliform levels than systems that did not practice corrosion control (LeChevallier et al., 1996). In addition to the level of generalized corrosion, localized pitting can provide a protective habitat for bacterial proliferation. The pitting of certain metal pipes can be accelerated by high levels of chloride and sulfate. The ratio of chloride and sulfate to bicarbonate levels is known as the Larson index and can indicate the propensity for pitting corrosion. Research has shown that consideration of the level of generalized corrosion, Larson index, corrosion inhibitor, and disinfectant residual is necessary to accurately predict the inactivation of biofilm bacteria (see Table 10-2) (LeChevallier et al., 1993).
Studies have shown that the Larson index can vary seasonally in drinking water systems, with the highest levels occurring during the summer months (LeChevallier et al., 1993). Factors that can influence the Larson index include anything that increases chloride levels (chlorine disinfection, aluminum or ferric salts) or that changes the alkalinity of the water (lime, soda ash, sodium bicarbonate have a positive influence; hydrofluosilicic acid, chlorine gas, certain coagulants depress alkalinity).
The pipe surface itself can influence the composition and activity of biofilm populations. Studies have shown that biofilms developed more quickly on iron pipe surfaces than on plastic PVC pipes, despite the fact that adequate corrosion control was applied, the water was biologically treated to reduce AOC levels, and chlorine residuals were consistently maintained (Haas et al., 1983; Camper, 1996). This
TABLE 10-2
Multiple Linear Regression Modela for Monochloramine Disinfection of Biofilm Bacteria
TABLE 10-3
Detection Frequency of Slow-Growing Mycobacteria in Water Samples
|
|
|
Percent Samples Containing Slow-Growing Mycobacteria |
|||
|
Disinfectant Type |
Nutrient Level |
Raw |
Plant/Well Effluent |
Midpoint |
Deadend |
|
O3/free |
High |
24 |
0 |
0 |
0 |
|
Free |
Medium |
6 |
0 |
0 |
6 |
|
Free |
Low |
11 |
0 |
0 |
6 |
|
Free |
Groundwater |
NA |
0 |
29 |
24 |
|
Chloramine |
High |
47 |
6 |
6 |
12 |
|
Chloramine |
Medium |
39 |
11 |
0 |
33 |
|
Chloramine |
Medium/low |
39 |
17 |
11 |
50 |
|
Chloramine |
Groundwater |
NA |
11 |
6 |
0 |
stimulation of microbial communities on iron pipes has been observed by other investigators.
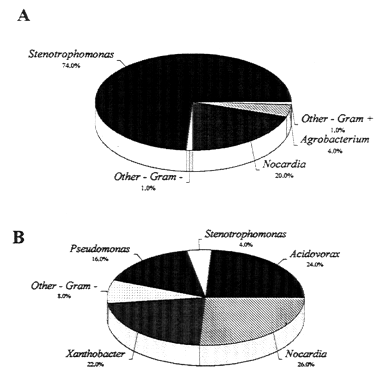
In addition to influencing the development of biofilms, the pipe surface has also been shown to affect the composition of the microbial communities present in the biofilm (see Figure 10-8) (Haas et al., 1983). Iron pipes supported a more diverse microbial population than did PVC pipes. The purpose of these studies is not to indicate that certain pipe materials are preferred over another but to demonstrate the importance of considering the type of materials that come into contact with potable water.
Public Health Significance of Biofilm Control
Growth of coliform bacteria in distribution system biofilms could be considered a nuisance if they had no public health significance. Coliform bacteria have traditionally been used to indicate the adequacy of drinking water treatment. A new interpretation of this indicator concept implies that drinking water is not adequately treated if coliform bacteria can proliferate in distribution system biofilms. One concern is whether opportunistic pathogens such as Legionella pneunophila, Mycobacterium avium complex (MAC), or other microbes can also proliferate.
Members of the M. avium complex (i.e., M. avium and Mycobacterium intracellulare) have been shown to occur in drinking water distribution systems with levels ranging between 0.08 and 4S,000 CFUs/ml (Haas et al., 1983; duMoulin and Stottmeier, 1986; duMoulin et al., 1988; Carson et al., 1988a; Fischeder et al., 1991; von Reyn et al., 1993, 1994; Glover et al., 1994). The greatest increase in M. avium complex infections has been with acquired immunodeficiency syndrome (AIDS) patients, approximately 25 to 50 percent of whom suffer debilitating and life-threatening MAC infections (Horsburgh, 1991; Nightingale et al., 1992). The organism infects the gastrointestinal or pulmonary tract, suggesting that food or water may be important routes of transmission for AIDS patients.
In an ongoing research study, examination of eight, well-characterized, drinking water systems showed that slow-growing mycobacteria were frequently detected in raw water and in distribution system samples using a chloramine residual (see Table 10-3). Either free-chlorine or ozone treatment appeared to be sufficient to eliminate mycobacteria to below detectable levels in plant effluent levels. The conclusion that chloramines provide a selective advantage for mycobacteria may be premature because of the higher rates of mycobacteria detection in raw water and possible elimination of bacteria that overgrow the selective medium. The reason for the frequent detection of slow-growing mycobacteria in the free-chlorinated groundwater site is unclear but may be due to the low chlorine residuals (average of 0.15 mg/L) observed at this location.
The levels of slow-growing mycobacteria detected in raw water samples and in distribution system samples are shown in Table 10-4. The results show that high levels were detected in a small number of samples. These levels could occur following flushing or other activities that could dislodge biofilm samples or resuspend distribution system sediments. Biofilm Mycobacterium levels ranged from nondetectable to >1,500 CFUs/cm2.
Using a nested PCR method (Kulski et al., 1995), 304 of the 708 (43 percent) of the water isolates, and 337 of 747 (45 percent) biofilm were identified as members of the genus Mycobacterium. Using both the nested PCR method and a PCR-based technique involving amplification of the 65-kDa heat shock protein gene (hsp-65) followed by digestion of the PCR product with restriction endonucleases (PCR-RE) (Telenti et al., 1993; Steingrube et al., 1995), 20 percent of the water isolates and 64 percent of the biofilm isolates were identified as M. avium or M. intracellulare. Additionally, eight percent of the water isolates were identified as M. kansasii. Ongoing biofilm studies are examining the role of nutrients, pipe material, disinfectant species, and temperature in an effort to better control mycobacteria in drinking water.
TABLE 10-4
Average Density of Slow-Growing Mycobacteria in Water Samples
|
|
|
Slow-Growing Mycobacteria/mLa |
|||
|
Disinfectant Type |
Nutrient Level |
Raw |
Plant Effluent |
Midpoint |
Deadend |
|
O3/free |
High |
0.46 |
<0.01 |
<0.01 |
<0.01 |
|
Free |
Medium |
<0.01 |
<0.01 |
<0.01 |
<0.01 |
|
Free |
Low |
2.44 |
<0.01 |
<0.01 |
3.41 |
|
Free |
Ground |
NA |
<0.01 |
15.68 |
130.08 |
|
Chloramine |
High |
20.65 |
<0.01 |
812.70 |
33.65 |
|
Chloramine |
Medium |
4.94 |
0.52 |
<0.01 |
5.13 |
|
Chloramine |
Medium/low |
7.79 |
0.86 |
6.25 |
101.70 |
|
Chloramine |
Ground |
NA |
13.08 |
7.28 |
<0.01 |
|
a For samples where slow-growing mycobacteria were detected. |
|||||
Conclusion
The occurrence of coliform regrowth in distribution systems depends on a complex interaction of chemical, physical, operational, and engineering parameters. No single factor could account for all coliform occurrences, so the water utility operator must consider all of the above parameters in devising a solution to the regrowth problem. Even systems that do not experience coliform problems may want to more closely examine biofilm control strategies and a means of limiting the occurrence of opportunistic pathogens such as Mycobacterium avium complex in drinking water supplies.
References
Camper, A. 1996. Factors Limiting Microbial Growth in Distribution Systems: Laboratory and Pilot-Scale Experiments. Denver, Colo.: American Water Works Association Research Foundation.
Carson, L. A., L. A. Bland, L. B. Cusick, M. S. Favero, G. A. Bolan, A. L. Reingold, and R. C. Good. 1988. Prevalence of nontuberculous mycobacteria in water supplies of hemodialysis centers. Applied Environmental Microbiology 54:3122-3125.
Characklis, W. G., and K. C. Marshal. 1990. Biofilms. New York: John Wiley & Sons, Inc.
Chen, X., and P. S. Stewart. 1996. Chlorine penetration into artificial biofilm is limited by a reaction-diffusion interaction. Environmental Science and Technology 30:2078.
De Beer, D., R. Srinivasan, and P. S. Stewart. 1994. Direct measurement of chlorine penetration into biofilms during disinfection. Applied Environmental Microbiology 60:4339.
duMoulin, G. C., and K. D. Stottmeier. 1986. Waterborne mycobacteria: An increasing threat to health. American Society of Microbiology News 52:525-529.
duMoulin, G. C., K. D. Stottmeier, P. A. Pelletier, A. Y. Tsang, and J. Hedley-Whyte. 1988. Concentration of Mycobacterium avium by hospital hot water systems. Journal of the American Medical Association 260:1599-1601.
Fischeder, R., R. Schulze-Röbbecke, and A. Weber. 1991. Occurrence of mycobacteria in drinking water samples. Zbl. Hyg. 192:154-158.
Glover, N., A. Holtzman, T. Aronson, S. Froman, O. G. W. Berlin, P. Dominguez, K. A. Kunkel, G. Overturf, G. Stelma, Jr., C. Smith, and M. Yakrus. 1994. The isolation and identification of Mycobacterium avium complex (MAC) recovered from Los Angeles potable water: A possible source of infection in AIDS patients. International Journal of Environmental Health Research 4:63-72.
Haas, C. N., M. A. Meyer, and M. S. Paller. 1983. The ecology of acid-fast organisms in water supply, treatment, and distribution systems. Journal of the American Water Works Association 75:139-144.
Horsburgh, C. R. 1991. Mycobacterium avium complex infection in the acquired immunodeficiency syndrome. New England Journal of Medicine 324:1332-1338.
Kulski, J. K., C. Khinsoe, T. Pryce, and K. Christiansen. 1995. Use of multiplex PCR to detect and identify Mycobacterium avium and M. Intracellulare in blood culture fluids of AIDS patients. Journal of Clinical Microbiology 33: 668-674.
LeChevallier, M. W. 1991. Biocides and the Current Status of Biofouling Control in Water Systems. Berlin: Springer-Verlag.
LeChevallier, M. W., C. D. Lowry and R. G. Lee. 1990. Disinfecting biofilms in a model distribution system. Journal of the American Water Works Association 82(7):87.
LeChevallier, M. W., C. D. Lowry, R. G. Lee, and D. L. Gibbon. 1993. Relationship between iron corrosion and disinfection of biofilm bacteria. Journal of the American Water Works Association 85(7):111.
LeChevallier, M. W., S. C. Cameron, and G. A. McFeters. 1983. New medium for the improved recovery of coliform bacteria from drinking water. Applied Environmental Microbiology 45(2):484.
LeChevallier, M. W., N. J. Welch, and D. B. Smith. 1996. Full scale studies of factors related to coliform regrowth in drinking water. Applied Environmental Microbiology 62:2201
Nightingale, S. D., L. T. Byrd, P. M. Southern, J. D. Jockusch, S. X. Cal, and B. A. Wynne. 1992. Mycobacterium avium-intracellulare complex bacteremia in
human immunodeficiency virus positive patients. Journal of Infectious Diseases 165:1082-1085.
Norton, C. D., and M. W. LeChevallier. 1997. Chloramination: Its effect on distribution system water quality. Journal of the American Water Works Association. 89(7):66-77.
Pontius, F. 1995. SDWA Advisor on CD. Denver, Colo.: American Water Works Association.
Rook, J. J. 1974. Formation of haloforms during chlorination of natural water. Water Treatment and Examination 23:234-243.
Steingrube, V. A., J. L. Gibson, R. A. Brown, Y. Zhang, R. W. Wilson, M. Rajagopalan, and R. J. Wallace, Jr. 1995. PCR amplification and restriction endonuclease analysis of a 65-kilodalton heat shock protein gene sequence for taxonomic separation of rapidly growing mycobacteria. Journal of Clinical Microbiology 33:149-153.
Stewart, P. S., S. Sanderson, X. Xu, J. Raquepas, and N. Cogan. In press. Biofilm control by antimicrobial agents. In Biofilms, 2nd edition, J. D. Bryers, ed. New York: John Wiley & Sons.
Telenti, A., F. Marchesi, M. Balz, R. Bally, E. C. Böttger, and T. Bodmer . 1993. Rapid identification of mycobacteria to the species level by polymerase chain reaction and restriction enzyme analysis. Journal of Clinical Microbiology 31:175-178.
Volk, C., C. Renner, and J. C. Joret. 1992. The measurement of BDOC: An index of bacterial regrowth potential in water. Revue Des Sciences De L'Eau 5(n special):189-205.
Volk, C., N. Welch and M. W. LeChevallier. 1996. Survey of Assimilable Organic Carbon and Coliform Regrowth in the American Water System. Voorhees, N.J.: American Water Works Service Company, Inc.
von Reyn, C. F., J. N. Maslow, T. W. Barber, J. O. Falkinham III, and R. D. Arbeit. 1994. Persistent colonization of potable water as a source of Mycobacterium avium infection in AIDS. Lancet 343:1137-1141.
von Reyn, C. F., R. D. Waddell, T. Eaton, R. D. Arbeit, J. N. Maslow, T. W. Barber, R. J. Brindle, C. F. Gilks, J. Lumio, J. Lähdevirta, A. Ranki, D. Dawson, and J. O. Falkinham III. 1993. Isolation of Mycobacterium avium complex from water in the United States, Finland, Zaire, and Kenya. Journal of Clinical Microbiology 31:3227-3230.
Watters, S. K., B. H. Pyle, M. W. LeChevallier, and G. A. McFeters. 1989. Enumeration of Enterobacter cloacae after chloramine exposure. Applied Environmental Microbiology 55:322.
Zaske, S. K., W. S. Dockins, and G. A. McFeters. 1980. Cell envelope damage in E. coli caused by short term stress in water. Applied Environmental Microbiology 40(2):386-390.