Protein and Amino Acids, 1999
Pp. 137-154. Washington, D.C.
National Academy Press
7
Effects of Protein Intake on Renal Function and on the Development of Renal Disease
Mackenzie Walser1
INTRODUCTION
One of the major functions of the kidney is the elimination Of the products of protein metabolism. It is not surprising, therefore, that protein intake exerts many diverse effects on the kidney. Most attention has been directed toward the role of protein intake in chronic renal failure. For at least a century, it has been known that reducing protein intake will ameliorate the symptoms of chronic renal failure, provided the reduction is not severe enough to induce protein deficiency. In recent years, much effort has been directed toward defining the optimum protein intake for patients with chronic renal failure, with the aim of slowing the rate of progression toward the end stage. Despite many reports, it is still not firmly established that protein restriction slows disease progression, although the bulk of available evidence supports this conclusion (for example, Fouque et al., 1992; Levey et al., 1996). In this review, however, attention will be limited to the effects of protein intake on the initiation of renal disease.
There are three ways in which protein intake may play a role in the development of renal disease: promotion of nephrolithiasis, enhancement of the morbidity of acute renal failure, and acceleration of glomerulosclerosis.
NEPHROLITHIASIS
During recent decades, nephrolithiasis has been increasing in frequency in developed countries in association with improved nutrition (Goldfarb, 1988; Robertson et al., 1979a). In Japan, for example, the incidence of renal stone disease has increased threefold since the period preceding World War II (Iguchi et al., 1990). Estimates of the prevalence of nephrolithiasis in the general population range from I percent to 15 percent (Johnson et al., 1979). Approximately 12 percent of the U.S. population will have a kidney stone at some time (Johnson et al., 1979; Sierakowski et al., 1978). One contributory factor is rising intake of protein-rich foods, which may promote urolithiasis (uric acid stone formation) in several ways (Robertson et al., 1979b).
Calcium Excretion and Calcium-Containing Stones
Calcium Intake
Calcific nephrolithiasis occurs when the activity product2 of free calcium times free oxalate or phosphate exceeds a certain level, which is in turn modified by crystallization inhibitory factors. Hence, a reduction in urinary calcium excretion might be expected to reduce the occurrence of calcific nephrolithiasis. Indeed, restriction of calcium intake has been a mainstay of dietary advice for the prevention of recurrent nephrolithiasis. However, as pointed out by Goldfarb (1988), this approach is inappropriate and even potentially dangerous for several reasons: first, negative calcium balance may ensue because gastrointestinal calcium losses continue, with resultant osteoporosis during long-term application (Lalau et al., 1992); second, retrospective data indicate that the incidence of stone recurrence is negatively correlated with spontaneous calcium intake, probably because ingested calcium precipitates oxalate in the gut (Curhan et al., 1993; Lemann et al., 1996). Whatever the explanation, it is clear that attempts to reduce renal clearance of calcium will be more fruitful than reducing calcium intake. In this respect the high calcium intake typically associated with a high protein intake may be protective against calcific nephrolithiasis.
Determinants of Calcium Clearance
As first reported by this laboratory (Walser, 1961a), calcium clearance in the dog is approximately equal to sodium clearance during a variety of experimental maneuvers (Figure 7-1), including the administration of diuretics (with the exception of thiazides) (Walser and Trounce, 1961). Many workers have confirmed these relationships in humans. Thus, a high sodium intake augments and a low sodium intake diminishes calcium clearance. A second
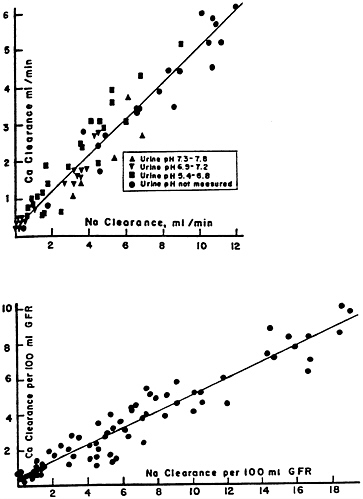
FIGURE 7-1 Calcium clearance in the dog as a function of sodium clearance during saline and osmotic diuresis, expressed as ml/min (above) and as ml/100 ml of glomerular filtration rate (below). Alkaline, neutral and acidic urines, indicated by different symbols, yield the same results. The correlations coefficients, r, are 0.95 (above) and 0.93 (below). A small but statistically significant (p < 0.01) intercept is present. Source: Reprinted by permission from Walser (1961a).
determinant of calcium clearance is sulfate excretion. Walser and Browder (1959) reported that infusion of sodium sulfate in dogs caused a pronounced increase in calcium clearance (Figure 7-2)—greater than the attendant increase in sodium clearance. This study showed that ion-pair formation between calcium and sulfate accounted for this effect, since the clearance of free calcium ions remained approximately equal to the clearance of sodium (Figure 7-3) (Walser, 1961b). This was the first demonstration of the biological role of ion pairs, which are electrostatic complexes between oppositely charged ions.
Subsequent work has confirmed that sodium excretion and sulfate excretion are the most important determinants of calcium excretion. For example, Tschope and Ritz (1985) measured 24-hour urinary excretion rates of calcium, sodium, and sulfate (as well as other constituents) in normal individuals and found that the ratio of calcium to creatinine was closely correlated with the ratio of sulfate to creatinine (r = 0.61, p < 0.001), and also correlated with the ratio of sodium to creatinine (r = 0.48, p < 0.01). When multiple regression analysis was
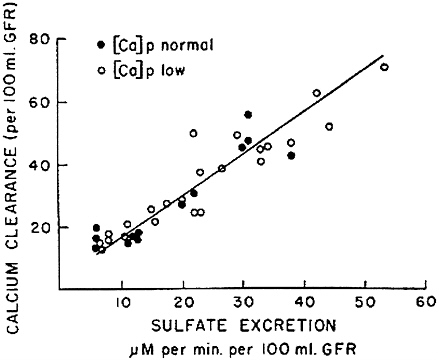
FIGURE 7-2 Calcium clearance per 100 ml of glomerular filtration rate as a function of sulfate excretion is dogs infused with sodium sulfate. Source: Reproduced from The Journal of Clinical Investigation, 1959, 38:1404-1411.
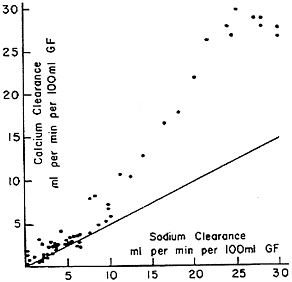
FIGURE 7-3 Calcium clearance as a function of sodium clearance in dogs infused with sodium sulfate. The diagonal line represents the relationship seen during saline or mannitol diuresis, as depicted in Figure 7-1. When calcium clearance is calculated as free calcium ion clearance, the relationship becomes the same as the diagonal line; the excess represents the calcium sulfate ion-pair complex. Source: Reprinted by permission from Walser (1961b).
performed, calcium:creatinine remained highly correlated with sulfate:creatinine (r = 0.76, p < 0.001). Similar findings were reported by Puche et al. (1987). Tschope and Ritz (1985) also administered methionine (40 mmol/d for 5 days) to nine normal individuals and noted that calcium excretion increased significantly in conjunction with the increase in sulfate excretion and fall in urine pH (Table 7-1). Forty mmol sulfur is the amount present as cystine and methionine in I pound of sirloin.
Urinary acidification also increases calcium clearance (Lemann et al., 1967), independently of sodium excretion. Thus, part of the effect of sulfate loads on calcium excretion is attributable to the attendant decrease in urine pH. Lutz (1984) found that two thirds of the increment in calcium excretion that resulted from an increase in protein intake in normal subjects was eliminated if sufficient sodium bicarbonate was concurrently administered to prevent any fall in urine pH.
Potassium supplementation may prevent the calciuretic action of a protein load (Kaneko et al., 1990). Lemann et al. (1991) have demonstrated that potassium administration reduces and potassium deprivation increases urinary calcium, possibly because of associated changes in sodium excretion or in calcitriol synthesis.
TABLE 7-1
Effect of Methionine Ingestion (40 rumor/day) in Normal Subjects; Average Urinary Exretion During Five Control Days and Five Experimental Days
|
Urinary Constituent |
Before |
During |
p |
|
Calcium, rag/day |
189 ±-94 |
276 ± 128 |
< 0.01 |
|
Magnesium, mg/day |
122±26 |
124 ± 24 |
n.s. |
|
Sulfate, mg/day |
2,382 ± 778 |
6,177 ± 893 |
< 0.01 |
|
Urea, g/day |
23 ± 4 |
23 ± 3 |
n.s. |
|
Citrate, mg/day |
880 ± 231 |
327 ± 168 |
< 0.06 |
|
PH |
5.57-6.70 |
5.02-6.37 |
<0.01 |
|
SOURCE: Reprinted by permission from Tschope and Ritz, 1995. |
|||
Contrary to the assertion of Trilok and Draper (1989), sulfur amine acids do not account entirely for the increase in net acid production associated with protein oxidation: phosphorylated amine acids and dibasic amine acids also contribute to endogenous acid production (Walser, 1986). This may be why other workers have found that sulfur amine acid content of dietary protein cannot account entirely for its calciuretic effect (Schuette et al., 1981; Zemel et al., 1981).
Patterns of Urinary Excretion in Stone Formers
In stone formers, urinary excretion of calcium and oxalate is often higher than in normal subjects, even though their diets may not differ in weekly intake of protein or calcium (Iguchi et al., 1990; Martini et al., 1993; Rao et al., 1982). Others have found, on the contrary, that stone formers consume more animal protein than controls (Iguchi et al., 1989; Robertson et al., 1979b; Trinchieri et al., 1991). Higher salt intake also may be a factor (Jaeger et al., 1988). The calciuric effect of dietary protein may be exaggerated in stone formers (Goldfarb, 1988; Jungers et al., 1993). Hypercalciuric patients tend to eat more protein at dinner and on weekends (Iguchi et al., 1989; Martini et al., 1993). Oxalate excretion after ingesting oxalate and the increase in calcium oxalate supersaturation of the urine are greater than in control subjects, provided that both groups are ingesting a high protein diet (Urivetzky et al., 1987).
Animal protein causes more calciuria than vegetable protein but, unlike the latter, does not increase urinary oxalate (Marangella et al., 1989). As is well known, animal protein contains a greater proportion of sulfur amine acids and therefore leads to a greater increment in urinary sulfate and net acid excretion.
Thus, it appears that sulfate excretion (and its effect on acid excretion) and sodium excretion, which are in turn functions of sulfur-amino acid intake and salt intake, respectively, are the principal determinants of calcium excretion by the kidney. Intake of calcium itself is of lesser importance. Phosphorus intake is also a factor: if phosphate intake increases at the same time as protein intake, less calciuria results (Hegsted et al., 1981, Linkswiler et al., 1981). Several other studies (Puche and Feldman, 1992; Puche et al., 1987; Schneider and Menden, 1988; Singh et al., 1993) have reached the same conclusions, although in one older study (Block et al., 1980), sulfur amino acid ingestion had no effect on calcium excretion in normal subjects.
It would seem to follow that dietary treatment of calcific nephrolithiasis should include restriction of the intake of salt and protein, particularly animal protein.
Uric Acid Stones
Acidification of the urine, which results from sulfur intake, increases the likelihood of uric acid stone formation. Furthermore, careful study of the ion products in the urine by Breslau et al. (1988), who compared subjects receiving a vegetable protein diet with subjects consuming an animal protein diet, found that the latter increased the tendency of the urine to form uric acid stones, but not calcium oxalate or calcium phosphate stones. Fellström et al. (1983, 1984) also reported that a high animal-protein diet increased urinary supersaturation with respect to uric acid and ammonium urate but not calcium oxalate. Thus although high animal protein diets may promote osteoporosis when consumed for decades, they are more likely to lead to uric acid stones than calcific nephrolithiasis. Nevertheless, uricosuria is associated with calcium oxalate urolithiasis (Coe, 1978), and uricosuria is increased by high protein intake (Robertson et al., 1979c).
Prophylactic Treatment of Nephrolithiasis
Allopurinol administration reduces the incidence of calcific nephrolithiasis in subjects who have hyperuricosuria but are not hypercalciuric (Ettinger et al., 1955). Protein restriction augments citrate excretion in hypercalciuric stone formers and reduces stone incidence but may not affect uric acid excretion (Goldfarb, 1988). Sodium restriction reduces urinary calcium in hypercalciuric subjects (Muldowney et al., 1982). As Churchill (1987) pointed out a decade ago, there are few scientifically adequate trials of any form of treatment in preventing recurrence of urinary stones. This remains true today.
ACUTE RENAL FAILURE
Andrews and Bates (1986) reported that the response of rat kidney to a 45 minute period of ischemia was dramatically affected by prior protein intake. For example, 93 percent of rats fed a high-protein diet for the preceding 2 weeks died of renal ischemia within 3 days, compared with 12 percent of rats fed a low-protein diet. One hundred percent of rats fed a zero protein diet survived, and most of them exhibited normal serum creatinine levels by the fourth day after ischemia.
Instituting protein restriction after a period of ischemia provided no protection, nor did switching from a zero-protein diet to a high-protein diet immediately after the ischemic insult cause any increase in mortality. Approximately I week of prior protein restriction was required to produce this remarkable effect.
The authors suggested that patients who are to be subjected to surgical procedures with a high risk of acute renal failure might benefit from protein restriction in the pre-operative period.
It is not clear how these findings might be applicable to soldiers, since their risk of acute renal failure, even in combat, is low. It is clear, however, that at least with regard to the risk of morbidity from acute renal failure, dietary protein restriction is more likely to confer benefit than to increase risk.
PROTEIN INTAKE AND THE DEVELOPMENT OF GLOMERULOSCLEROSIS
Glomerular filtration rate (GFR) declines with age for people over 40 in approximately two-thirds of persons without frank kidney disease (Lindeman et al., 1985), especially if they are hypertensive (Lindeman et al., 1984); in the remaining one third, GFR remains constant or increases with age (Lindeman, 1990). According to Fliser et al. (1997), about two-thirds of elderly subjects who are not in heart failure and not on diuretics (even if hypertensive) have GFRs within the range of younger subjects (Figure 7-4). Kidneys examined at autopsy of persons dying of causes other than kidney disease show a progressive, though extremely variable, increase in the fraction of glomeruli that are sclerotic (Figure 7-5) (Kaplan et al., 1975). Circulatory impairment may be another factor, since there may be localized areas of reduced flow within the kidneys of older subjects (Friedman et al., 1972).
In rats, the development of chronic renal failure with age is nearly universal (Coleman et al., 1977; Hayashida et al., 1986). The predominant lesion is glomerulosclerosis. Because this process was apparently attenuated by protein restriction (see review by Masoro and Yu, 1989), Anderson and Brenner (1987) suggested that the high protein intake of Western societies plays a central role in
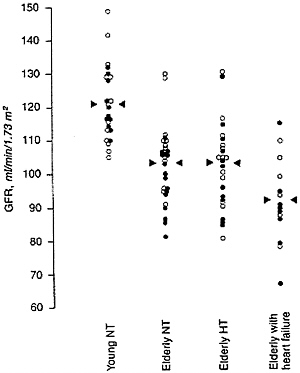
FIGURE 7-4 Glomerular filtration rate, measured as inulin clearance, in four groups of subjects. Young NT: healthy subjects 26 ± 3 years of age with normal blood pressure. Elderly NT: healthy subjects 68 ± 7 years of age with normal blood pressure. Elderly HT: subjects 70 ± 6 years of age with hypertension but not receiving diuretics. Elderly with heart failure: subjects 69 ± 6 years of age with heart failure. Symbols: open circles, men; solid circles, women.
Source: Used with permission from Kidney International, 1997, 51:1196-1204.
the decline of renal function with age and that restriction of dietary protein might prevent this decline.
This recommendation cannot be supported for a number of reasons as pointed out in a recent review (Walser, 1992). First, caloric restriction is more effective in rats than is protein restriction in retarding the age-associated decline in renal function (Tapp et al., 1989). Furthermore, caloric restriction without protein restriction markedly retarded the progression of glomerulosclerosis. Rats prefer rations containing higher proportions of protein, and the earlier studies indicating that protein restriction retarded renal damage failed to monitor food intake.
Second, protein restriction tends to lower GFR rather than increase it. Lew and Bosch (1991) recorded the dependence of creatinine clearance on spontaneous protein intake in subjects aged 22 to 50 years and in subjects aged
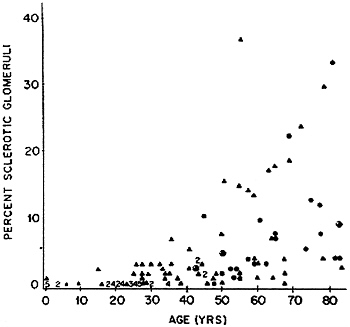
FIGURE 7-5 Percent sclerotic glomeruli in kidneys from 122 autopsied patients without known renal disease. Source: Reprinted by permission from Kaplan et al. (1975).
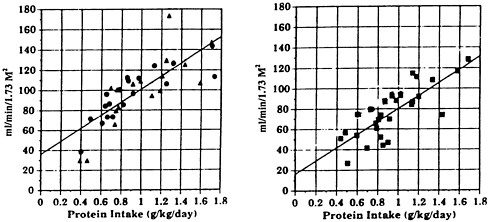
FIGURE 7-6 Dependence of creatinine clearance, in ml/min/1.73 M2 on protein intake in healthy subjects aged 22-50 years (left) and 55-88 years (right). The slopes are not different, but the intercepts (43 refrain end 31 ml/min, respectively) differ significantly. SOURCE: S.Q. Lew end J.P. Bosch, 1991, Effect of diet on creatinine clearance end excretion in young end elderly healthy subjects end in patients with renal disease, J. Am. Soc. Nephrol., 2:856-865.
55 to 88 years (Figure 7-6). In both groups, there was a similar pronounced dependence of clearance on protein intake; in the older subjects, a lower intercept was seen, reflecting the effect of age on GFR. If protein intake is not controlled, the apparent decline in GFR with age will be exaggerated, because older subjects consume less protein than younger subjects (Kerr et al., 1982).
Third, Tobin and Spector (1986) measured creatinine clearance in 198 normal men on two occasions, 10 to 18 years apart, and correlated the decline in clearance during this interval with protein intake: no relationship was detected. They found no evidence that a high intake of protein caused a progressive reduction in renal function.
Fourth, high-protein feeding (60%) for 2 years in rats had no effect on the percentage of sclerotic glomeruli (Collins et al., 1990).
Fifth, the progressive decline in renal function seen in rats after partial nephrectomy (Brenner et al., 1982) may be unique to this species. In dogs with 75 percent nephrectomy, GFR does not decline progressively with time for the ensuing 4 years, whether protein intake is high or low (Bovée, 1991). In baboons followed for 5 years after subtotal nephrectomy and on either 8 percent protein or 25 percent protein diets, renal failure did not occur; GFR, measured as inulin clearance, increased sharply in baboons fed 25 percent protein, and this difference tended to disappear with time (curiously, creatinine clearances did not decrease with time in either group). Even in baboons fed 8 percent protein, a slow decline in GFR with time was seen (5% per year). Proteinuria did not differ between the two groups and did not progress (Bourgoignie et al., 1994). In human kidney donors, mild proteinuria (but not albuminuria) is often seen, and the incidence of hypertension may be increased, but progressive renal failure rarely if ever occurs, and there is no correlation between protein intake and proteinuria (Anderson et al., 1985; Hakim et al., 1984). Such individuals are not generally advised to restrict their protein intake (Rocher and Swartz, 1987), although some clinicians have recommended that these individuals do so until the question is settled (Bay and Hebert, 1987). Likewise, protein restriction is not advised for persons with a solitary normal kidney (Mitch, 1989).
From these observations, it is clear that protein restriction does not prevent the decline in renal function with age and, in fact, is the major cause of that decline. A better way to prevent the decline would be to increase protein intake. Indeed, protein malnutrition in the elderly is a far more prevalent problem (Morgan et al., 1956; Rudman et al., 1989) than is nitrogen retention as a consequence of an age-related decline in renal function.
AUTHOR'S CONCLUSIONS AND RECOMMENDATIONS
Concerns about an adverse effect of high protein intake on renal function, and in particular on its decline with age, appear to be ill advised. Putting aside the potential adverse effects of protein intake on nephrolithiasis or on morbidity
of acute renal failure, detailed earlier, there is no reason to restrict protein intake in healthy individuals in order to protect the kidney. Nevertheless, there is also no evidence that a more restricted protein intake has any harmful long-term effects on the kidney.
REFERENCES
Anderson, C.F., J.A. Velosa, P.P. Frohnert, V.E. Torres, K.P. Offord, J.P. Vogel, J.V. Donadio, Jr., and D.M. Wilson. 1985. The risks of unilateral nephrectomy: Status of kidney donors 10 to 20 years postoperatively. Mayo Clin. Proc. 60:367-374.
Anderson, S., and B.M. Brenner. 1987. The aging kidney: structure, function, mechanisms, and therapeutic implications. J. Am. Geriatr. Soc. 35:590-593.
Andrews, P.M., and S.B. Bates. 1986. Dietary protein prior to renal ischemia dramatically affects postischemic kidney damage. Kidney Int. 30:299-303.
Bay, W.H., and L.A. Hebert. 1987. Kidney donors and protein intake [letter to the editor]. Ann. Intern. Med. 107:427.
Block, G.D., R.J. Wood, and L.H. Allen. 1980. A comparison of the effects of feeding sulfur amine acids and protein on urine calcium in man. Am. J. Clin. Nutr. 33:2128-2136.
Bourgoignie, J.J., G. Gayellas, S.G. Sabnis, and T.T. Antonovych. 1994. Effect of protein diets on the renal function of baboons (Papio hamadryas) with remnant kidneys: A 5-year follow-up. Am. J. Kidney Dis. 23:199-204.
Bovée, K.C. 1991. Influence of dietary protein on renal function in dogs. J. Nutr. 121:S128-S139.
Brenner, B.M., T.W. Meyer, and T.W. Hostetter. 1982. Dietary protein intake and the progressive nature of kidney disease: the role of hemodynamically moderated glomerular injury in the pathogenesis of progressive glomerular sclerosis in aging, renal ablation and intrinsic renal disease. New Engl. J. Med. 307:652-659.
Breslau, N.A., L. Brinkley, K.D. Hill, and C.Y.C. Pak. 1988. Relationship of animal protein-rich diet to kidney stone formation and calcium metabolism. J. Clin. Endocrinol. Metab. 66:140-146.
Churchill, D.N. 1987. Medical treatment to prevent recurrent calcium urolithiasis. Miner. Electrolyte Metab. 13:294-304.
Coe, F.L. 1978. Calcium-uric acid nephrolithiasis. Arch. Intern. Med. 138:1090-1093.
Coleman, G.L., W. Barthold, G.W. Osbaldiston, S.L Foster, and A.M. Jonas. 1977. Pathological changes during aging in barrier-reared Fischer 344 male rats. J. Gerontol. 32:258-278.
Collins, D.M., C.T. Rezzo, J.B. Kopp, P. Ruiz, T.M. Coffman, and P.E. Klotman. 1990. Chronic high protein feeding does not produce glomerulosclerosis or renal insufficiency in the normal rat. J. Am. Soc. Nephrol. 1:624.
Curhan, G.C., W.C. Willett, E.B. Rimm, and M.J. Stampfer. 1993. A prospective study of dietary calcium and other nutrients and the risk of symptomatic kidney stones. New Engl. J. Med. 328:833-838.
Ettinger, B., J.T. Citron, A. Tang, and B. Livermore. 1985. Prophylaxis of calcium oxalate stones; clinical trials of allopurinol, magnesium hydroxide, and chlorthalidone. Pp. 549-552 in Urolithiasis and Related Clinical Research, P.O. Schwille, L.H. Smith, W.G. Robertson, and W. Vahlensieck, eds. New York: Plenum.
Fellström, B., B.G. Danielson, B. Karlström, H. Lithell, S. Ljunghall, and B. Vessby. 1983. Dietary animal protein and urinary supersaturation in renal stone formers. Proc. Eur. Dial. Transplant. Assoc. 20:411-416.
Fellström, B., B.G. Danielson, B. Karlström, H. Lithell, S.L. Ljunghall, B. Vessby, and L. Wide. 1954. Effects of high intake of dietary animal protein on mineral metabolism and urinary supersaturation of calcium oxalate in renal stone formers. Brit. J. Urol. 56:263-269.
Fliser, D., E. Franek, M. Joest, S. Block, E. Mutschler, and E. Ritz. 1997. Renal function in the elderly: Impact of hypertension and cardiac function. Kidney Int. 51:1196-1204.
Fouque, D., M. Laville, J.P. Boissel, R. Chifflet, M. Labeeuw, and P.Y. Zech. 1992. Controlled low protein diets and chronic renal insufficiency: Meta-analysis. Br. Med. J. 304:216-220.
Friedman, S.A., A.E. Raizner, H. Rosen, N.A. Solomon, and W. Sy. 1972. Functional defects in the aging kidney. Ann. Intern. Med. 76:41-45.
Galdfarb, S. 1988. Dietary factors in the pathogenesis and prophylaxis of calcium nephrolithiasis. Kidney Int. 34:544-555.
Hakim, R.M., R.C. Goldszer, and B.M. Brenner. 1984. Hypertension and proteinuria: Long-term sequelae of uninephrectomy in humans. Kidney Int. 25:930-936.
Hayashida, M., B.P. Yu, E.J. Masoro, K. Iwasaki, and T. Ikeda. 1986. An electron microscopic examination of age-related changes in the rat kidney: The influence of diet. Exp. Gerontol. 21:535-553.
Hegsted, M., S.A. Schuette, M.B. Zemel, and H.M. Uinkswiler. 1981. Urinary calcium and calcium balance in young men as affected by level of protein and phosphorus intake. J. Nutr. 111:553-562.
Iguchi, M., T. Umekawa, Y. Ishikawa, Y. Katayama, M. Kodama, M. Takada, Y. Katoh, K. Kataoka, K. Kohri, and T. Kurita. 1989. [Dietary habits of Japanese stone formers and clinical effects of prophylactic dietary treatment]. Hinyokika Kiyo 35:2115-2128.
Iguchi, M., T. Umekawa, Y. Ishikawa, Y. Katayama, M. Kodama, M. Takada, Y. Katoh, K. Kataoka, K. Kohri, and T. Kurita. 1990. Dietary intake and habits of Japanese renal stone patients. J. Urol. 143:1093-1095.
Jaeger, P., L. Portmann, J.M. Ginalski, and P. Burckhardt. 1988. [So-called ''renal" idopathic hypercalciuria most often has a dietary origin]. Schweiz. Med. Wochenschr. 118:15-17.
Johnson, C.M., D.M. Wilson, W.M. O'Fallon, R.S. Malek, and L.T. Kurland. 1979. Renal stone epidemiology: A 25-year study in Rochester, Minnesota. Kidney Int. 16:624-631.
Jungers, P., M. Daudon, C. Hennequin, and B. Lacour. 1993. [Correlations between protein and sodium intake and calciuria in calcium [ithiasis]. Nephrologie 14:287-290.
Kaneko, K, U. Masaki, M. Aikyo, K. Yabuki, A. Haga, C. Matoba, H. Sasaki, and G. Koike. 1990. Urinary calcium and calcium balance in young women affected by high protein diet of soy protein isolate and adding sulfur-containing areinc acids and/or potassium. J. Nutr. Sci. Vitaminol. (Tokyo) 36:105-116.
Kaplan, C., S. Pasternack, H. Shah, and G. Gallo. 1975. Age-related incidence of sclerotic glomeruli in human kidneys. Am. J. Pathol. 80:227-234.
Kerr, G.R., E. S. Lee, M.-K.M. Lam, R.J. Lorimor, E. Randall, R.N. Forthofer, M.A. Davis, and S.M. Magnetti. 1982. Relationships between dietary and biochemical measures of nutritional status in HANES I data. Am. J. Clin. Nutr. 35:294-308.
Lalau, J.D., J.M. Achard, P. Bataille, C. Bergot, I. Jans, B. Boudailliez, J. Petit, G. Henon, P.F. Westeel, N. El Esper, M.A. Laval-Jeantet, R. Bouillon, J.L. Sebert, and A. Fournier. 1992. [Vertebral density in hypercalciuric lithiasis: relationship with calcium and protein consumption and vitamin D metabolism]. Aun. Med. Interne 143:293-298.
Lemann, J. Jr., J.R. Litzow, and E.J. Lennon. 1967. Studies of the mechanism by which chronic metabolic acidosis augments urinary calcium excretion in man. J. Clin. Invest. 46:1318-1328.
Lemann, J., Jr., J.A. Pleuss, R.W. Gray, and R.G. Hoffmann. 1991. Potassium administration reduces and potassium deprivation increases urinary calcium excretion in healthy adults. Kidney Int. 39:973-993.
Lemann, J., Jr., J.A. Pieuss, E.M. Worcester, L. Hornick, D. Schrab, and R.G. Hoffmann. 1996. Urinary oxalate excretion increases with body size and decreases with increasing dietary calcium intake among healthy adults. Kidney Int. 49:200-208.
Levey, A.S., S. Adler, A.W. Caggiula, B.K. England, T. Greene, L.G. Hunsicker, J.W. Kusek, N.L. Rogers, and P.E. Teschan for the Modification of Diet in Renal Disease Study Group. 1996. Effects of dietary protein restriction on the progression of advanced renal disease in the Modification of Diet in Renal Disease Study. Am. J. Kidney Dis. 27:652-663.
Lew, S.Q., and J.P. Bosch. 1991. Effect of diet on creatinine clearance and excretion in young and elderly healthy subjects and in patients with renal disease. J. Am. Soc. Nephrol. 2:856-865.
Lindeman, P.D. 1990. Overview: Renal physiology and pathophysiology of aging. Am J. Kidney Dis. 16:276-282.
Lindeman, R.D., J. Tobin, and N.W. Shock. 1984. Association between blood pressure and the decline in renal function with age. Kidney Int. 26:861-268.
Lindeman, P.D., J. Tobin, and N.W. Shock. 1985. Longitudinal studies on the rate of decline in renal function with age. J. Am. Geriatr. Soc. 33:278-285.
Linkswiler, H.M., M.B. Zemel, M. Hegsted, and S. Schuette. 1981. Protein-induced hypercalciuria. Fed. Proc. 40:2429-2433.
Lutz, J. 1984. Calcium balance and acid-base status of women as affected by increased protein intake and by sodium bicarbonate ingestion. Am. J. Clin. Nutr. 39:281-288.
Marangella, M., O. Blanco, C.V. Martini, M. Petrarulo, C. Vitale, and F. Linari. 1989. Effect of animal and vegetable protein intake on oxalate excretion in idiopathic calcium stone formers. Br. J. Urol. 63:348-351.
Martini, L.A., I.P. Heilberg, L. Cuppari,. F.A. Medeiros, S.A. Draibe, H. Ajzen, and N. Schor. 1993. Dietary habits of calcium stone formers. Braz. J. Med. Biol. Res. 26:805-812.
Masoro, E.J., and B.P. Yu. 1989. Diet and nephropathy [editorial]. Lab. Invest. 60:165-167.
Mitch, W.E. 1989. Dietary restrictions for a single normal kidney. Pediatr. Nephrol. 3:129.
Morgan, D.B., H.M. Newton, C.J. Schorah, M.A. Jewitt, M.P. Hancock, and R.P. Hullin. 1986. Abnormal indices of nutrition in the elderly: A study of different clinical groups. Age Ageing 15:65-76.
Muldowney, F.P., R. Freaney, and M.F. Moloney. 1982. Importance of dietary sodium in the hypercalciuria syndrome. Kidney Int. 22:292-296.
Puche, R.C., and S. Feldman. 1992. Relative importance of urinary sulfate and net acid excretion as determinants of calciuria in normal subjects. Medicina (B Aires) 52:220-224.
Puche, P.C., A.F. Carlomagno, A. Gonzalez, and A. Sanchez. 1987. A correlation and path coefficient analysis of components of calciuria in normal subjects and idiopathic stone formers. Bone Miner. 2:405-411.
Rao, P.N., V. Prendiville, A. Buxton, D.G. Moss, and N.J. Blacklock. 1982. Dietary management of urinary risk factors in renal stone formers. Br. J. Urol. 54:578-583.
Robertson, W.G., M. Peacock, and A. Hodgkinson. 1979a. Dietary changes and the incidence of urinary calculi in the U.K. between 1958 and 1976. J. Chronic Dis. 32:469-476.
Robertson, W.G., P.J. Heyburn, M. Peacock, F.A. Hanes, and R. Swaminathan. 1979b. The effect of high animal protein intake on the risk of calcium-stone-formation in the urinary track Clin. Sci. 57:285-288.
Robertson, W.G., M. Peacock, P.J. Heyburn, F.A. Hanes, A. Rutherford, E. Clementson, R. Swaminathan, and P.B. Clark. 1979c. Should recurrent calcium oxalate stone formers become vegetarians? Brit J. Urol. 51:427-431.
Rocher, L.L., and R.D. Swartz. 1987. Kidney donors and protein intake [letter to the editor]. Ann. Intern. IVied. 107:427.
Rudman, D., D.E. Mattson, A.G. Feller, R. Cotter, and R.C. Johnson. 1989. Fasting plasma amino acids in elderly men. Am. J. Clin. Nutr. 49:559-566.
Schneider, W., and E. Menden. 1988. [The effect of long-term increased protein administration on mineral metabolism and kidney function in the rat. I. Renal and enteral metabolism of calcium, magnesium, phosphorus, sulfate and acid]. Z. Ernährungwiss. 27:170-185.
Schuette, S.A., M. Hegsted, M.B. Zemel, and H.M. Linkswiler. 1981. Renal acid, urinary cyclic AMP, and hydroxyproline excretion as affected by the level of protein, sulfur amino acid, and phosphorus intake. J. Nutr. 111:2106-2116.
Sierakowski, R., B. Finlayson, R.R. Landes, C.D. Finlayson, and N. Sierakowski. 1978. The frequency of urolithiasis in hospital discharge diagnoses in the United States. Invest. Urol. 15:438-441.
Singh, P.P., F. Hussain, R.C. Gupta, A.K. Pendse, R. Kiran, and R. Ghosh. 1993. Effect of dietary methionine and inorganic sulfate with and without calcium supplementation on urinary calcium excretion of guinea pigs (Covia porcellus). Indian J. Exp. Biol. 31:96-97.
Tapp, D.C., W.G. Wortham, J.F. Addison, D.N. Hammonds, J.L. Barnes, and M.A. Venkatachalam. 1999. Food restriction retards body growth and prevents end-stage renal pathology in remnant kidney of rats regardless of protein intake. Lab. Invest. 60:184-195.
Tobin, J, and D. Spector. 1986. Dietary protein has no effect on future creatinine clearance. Gerontologist 26(SI):59A.
Trilok, G., and H.H. Draper. 1989. Sources of protein-induced endogenous acid production and excretion by human adults. Calcif. Tissue Int. 44:335-338.
Trinchieri, A., A. Mandressi, P. Luongo, and E. Pisani. 1991. The influence of diet on urinary risk factors for stones in healthy subjects and idiopathic renal calcium stone formers. Brit. J. Urol. 67:230-236.
Tschope, W., and E. Ritz. 1985. Sulfur-containing amino acids are the major determinant of urinary calcium. Mineral Electrolyte Metab. 11:137-139.
Urivetzky, M., J. Motola, S. Braverman, and A.D. Smith. 1987. Dietary protein levels affect the excretion of oxalate and calcium in patients with absorptive hypercalciuria type II. J. Urol. 137:690-692.
Walser, M. 1961a. Calcium clearance as a function of sodium clearance in the dog. Am. J. Physiol. 200:1099-1104.
Walser, M. 1961b. Ion association. VII. Dependence of calciuresis on natriuresis during sulfate infusion. Am. J. Physiol. 201:769-773.
Walser, M. 1986. The roles of urea production, ammonium excretion, and amino acid oxidation in acid-base balance. Am. J. Physiol. 250:F181-F188.
Walser, M. 1992. Dietary proteins and their relationship to kidney disease. Pp. 168-178 in Dietary Proteins in Health and Disease, G.U. Liepa, ed. Champaign: American Oil Chemists' Society.
Walser, M., and A.A. Browder. 1959. Ion association. III. The effect of sulfate infusion on calcium excretion. J. Clin. Invest. 38:1404-1411.
Walser, M., and J.R. Trounce. 1961, The effect of diuresis and diuretics upon the renal tubular transport of alkaline earth cations. Biochem. Pharmacol. 8:157.
Zemel, M.B., S.A. Schuette, M. Hegsted, and H.M. Linkswiler. 1981. Role of sulfurcontaining amino acids in protein-induced hypercalciuria in man. J. Nutr. 111:545-552.
DISCUSSION
JOAN CONWAY: Throughout your presentation this morning, you mentioned high and low protein intake. Can you quantitate those intakes so we have some ability to refer that to what we have been told military members consume?
MACKENZIE WALSER: Each study I reported to you would have a different answer to that question. I am not sure how you want me to answer that question because, as I say, no two studies are alike.
JOAN CONWAY: Well, if you say high and you say low, what would those be?
MACKENZIE WALSER: I think one gram per kilogram is pretty high. Some people would say that is not, but I think it is pretty high, and 0.6 is pretty low.
JOAN CONWAY: Okay. Thank you.
HARRIS LIEBERMAN: One gram per kilogram is much lower than what most people consume.
MACKENZIE WALSER: No. That is a myth. I know we are going to get into an argument about that. You are right about young medical students, but you are not right about older folks and women.
HARRIS LIEBERMAN: I am also right about soldiers.
MACKENZIE WALSER: Oh, that may well be. I have to re-orient my thinking in that regard.
STEVEN HEYMSFIELD: There were studies of protein restriction in people with chronic renal failure I believe. Is that right?
MACKENZIE WALSER: Yes.
STEVEN HEYMSFIELD: What ever happened with those studies?
MACKENZIE WALSER: Well, that work is going on and on. It looks like it is never going to end. I think that the bottom line at the moment is that protein restriction probably does slow progression. It certainly reduces symptoms, there is no question. That has been known for a hundred years. I cannot believe there are still people—telling their patients to go ahead and eat what they wish. That is a mistake. There is no question about that.
ROBERTNESHEIM: Dr. Millward.
D. JOE MILLWARD: Is there any information from the epidemiology of disease patterns in those groups of people who traditionally have had very high-protein diets, such as the Inuit Indians?
MACKENZIE WALSER: Is there any information about the incidence of renal disease?
D. JOE MILLWARD: Yes, they have low incidence of some chronic cardiovascular disease.
MACKENZIE WALSER: They do?
D. JOE MILLWARD: Yes, and, hence, all of the omega-three fatty acids are elevated. Traditionally, they have what is probably the highest protein intake of any group on the planet.
MACKENZIE WALSER: Yes. I have heard that. In fact, I learned somewhere that the reason that the Eskimos eat blubber is because if they eat just whale meat they get meat intoxication. They get up to about 400 grams a day of protein. That is why they eat some blubber to attenuate the protein intake. Have you read that?
D. JOE MILLWARD: Who knows why they do that?
MACKENZIE WALSER: You do not think they do so to prevent ammonia intoxication?
D. JOE MILLWARD: There are substantial long-term studies of their health.
MACKENZIE WALSER: I do not know the answer to your question. That is the straight answer.
ROBERT NESHEIM: Johanna?
JOHANNA DWYER: This was wonderful. With respect to the Brenner Hypothesis (Brenner et al, 1982) and later work that suggested maybe there was an energy deficit in the subjects on a low protein diet, how strong is the evidence?
MACKENZIE WALSER: Yes. Well, the progressive renal damage, according to Tap et al. (1989), is attributable to calories and not protein. That is right.
JOHANNA DWYER: Yes. If it is attributable to calories and not protein, but the reality of many patients with renal disease is that their energy intakes are also extremely low, do you think the Brenner hypothesis still has relevance to the human situation?
MACKENZIE WALSER: I think that it certainly must have relevance. I should add that those observations stimulated a great deal of research in this field without which the research never would have been done. Exactly where it fits in I am not sure.
For example, people born with one kidney or a small kidney, people with renal hypogenesis, do occasionally develop renal failure. So I am sure that there are instances in which renal failure results from partial nephrectomy or absence thereof and that probably could be ameliorated by protein restriction.
ROBERT NESHEIM: Thank you. We need to move on. It is time for a break here.


















