Protein and Amino Acids, 1999
Pp. 155-167. Washington, D.C.
National Academy Press
8
Infection and Injury: Effects on Whole Body Protein Metabolism
Douglas W. Wilmore1
Injury and infection elicit a rather stereotypic metabolic response characterized by hypermetabolism, accelerated gluconeogenesis, increased fat oxidation, and negative nitrogen balance (Wilmore, 1997). These responses are particularly evident in young, previously healthy, well-nourished individuals, such as members of the military, who have a well-developed lean body mass and no associated diseases or dysfunctional organs. Because body protein represents an indispensable structural and functional element of the body, the mechanisms that regulate the accelerated proteolysis during these catabolic states have received much attention. This is because significant erosion of lean body mass results in immunosuppression, poor wound healing, decreased strength and activity, and prolonged convalescence. Methods of attenuating the catabolic response or enhancing protein anabolism are now being evaluated in
patients in an effort to shorten convalescent recovery and decrease the length of hospital stay (Wilmore, 1991).
This chapter outlines some of the changes that occur in whole body protein metabolism following infection and injury. Evidence will be presented that these catabolic states result in marked translocation of protein from the carcass to visceral tissues, resulting in a net loss of skeletal muscle mass. Such loss results in decreased strength and activity.
BACKGROUND AND GENERAL RESPONSE CHARACTERISTICS
Over 200 years ago, John Hunter, the British surgeon and biologist, recognized that accidental injury initiated a series of responses in the host that presumably aided tissue repair and general recovery (Hunter, 1794). In the late 1800s, the concept of nitrogen balance was established by Voit and his German colleagues working in this area of physiological biochemistry (Munro, 1964). They noted that nitrogen balance could be related to the body's protein economy and was affected positively by the increased ingestion of both energy and protein and affected negatively by sepsis-induced fever. These concepts were confirmed and extended in the early 1900s, when Coleman and DuBels (1915) studied both energy and protein balance in a group of patients with typhoid fever. Using both direct calorimetric techniques and whole body balance methodology, they studied patients during the acute and convalescent phases of their illness. The investigators described the increased net loss of nitrogen that was associated with the febrile episodes of typhoid fever; the negative nitrogen balance could not be offset by the ingestion of large quantities of energy at the moderate level of protein intake that was utilized.
In 1932, Cuthbertson (1932) described a group of patients who had either undergone orthopedic surgical procedures on their lower extremities or who had sustained long bone fractures. He noted that these injuries to the long bone were associated with an enhanced loss of nitrogen from the body. This negative nitrogen balance was maximal during the first week postinjury and persisted for up to I month. Cuthbertson divided the metabolic response to injury into two phases: an early, acute "ebb" or shock phase and a latter "flow" or hyperdynamic phase. During the ebb phase, there was decreased metabolic activity, which was followed within 12 to 48 hours by the flow phase, a hypermetabolic state in which metabolic rate, temperature, and urinary nitrogen excretion all increased. Others have since contributed to the field of postinjury metabolism, with classic contributions being made by John Howard, Oliver Cope, Francis Moore, John Kinney, and many others (Wilmore, 1997).
During the early 1900s, researchers realized that infection was related to increased loss of protein from the body, but it was not until the 1960s that the systematic study of the metabolic response to infection was undertaken and published. William Beisel and colleagues (1967), working with the Army to
understand the impact of infectious disease on the host, studied normal individuals who were infected under experimental conditions with tularemia, sand fly fever, or Q fever. The individuals received a fixed diet of known composition, and after metabolic baseline was established, the infectious agent was administered. A well-defined sequence of events occurred over time: phagocyte function increased; serum amino acids, zinc, and iron concentrations fell; glucocorticoids and growth hormone concentrations increased in the blood stream, and acute-phase protein levels were also altered. The patients subsequently became febrile, retained salt and water, but excreted more nitrogen in the form of urea. The negative nitrogen balance was related to the decreased dietary intake of energy and protein and also to the fever-related increases in metabolism. In a separate experiment, exogenous glucocorticoids were administered to noninfected controls. Although this mimicked in part the adrenal cortical response to infection, this perturbation did not simulate the negative nitrogen and mineral balance observed in the febrile volunteers. Likewise, when noninfected normals consumed a caloric and protein intake similar to that ingested by the subjects who had experimental tularemia, the negative nitrogen balance was slight (18 g cumulative), demonstrating only about one-third of the losses observed in the infected patients (52 g cumulative). Because of the extent of the protein loss, it often took weeks for the infected individuals to replete their lean body mass.
From these and related studies, some general conclusions can be made concerning the whole body protein catabolic response following injury or infection:
-
The increased nitrogen loss occurs via the urine. Urea represents the major component of the nitrogen lost and contributes 85 to 90 percent to the negative nitrogen balance.
-
The response pattern generally shows dose-response characteristics; that is, the greater the infection or injury, the more extensive the nitrogen loss (Table 8-1).
-
The response follows a time course, with nitrogen excretion increasing in the first several days, peaking for several days or weeks, and then gradually returning to equilibrium as the inflammation resolves, food intake increases, and spontaneous physical activity occurs (Figure 8-1).
-
More nitrogen is lost from a well-nourished individual than from a depleted patient following a comparable catabolic insult. Thus, a relationship exists between the size of the lean body mass and the extent of protein catabolism, and this may account for variation in nitrogen balance that occurs with age and gender. Both older individuals and women are thought to excrete less nitrogen following a standard stimulus than do their appropriate controls.
TABLE 8-1 Estimates of Nitrogen Loss Following Catabolic Illness (First 10 Days, Ad Lib Feedings)
|
Precipitating Factor |
Cumulative Nitrogen Loss (g) |
|
Injury |
|
|
Major bum |
170 |
|
Multiple injury |
150 |
|
Peritonitis |
136 |
|
Simple fracture |
115 |
|
Major operation |
50 |
|
Minor operation |
24 |
|
Infection |
|
|
Typhoid fever (untreated) |
116 |
|
Pneumonia (untreated) |
59 |
|
Tularemia (treated) |
52 |
|
Q fever (treated) |
40 |
|
Sandfly fever (untreated) |
16 |
|
SOURCE: Adapted in part from Wannamacher, 1975. |
|
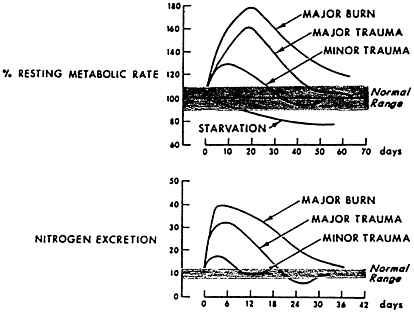
FIGURE 8-1 Hypermetabolism and nitrogen excretion are closely related, show a dose-response relationship, and resolve with time. Patients received 12 g of dietary nitrogen per day. Source: Adapted from Kinney, 1966.
TABLE 8-2 Alterations in Rates of Protein Synthesis and Catabolism That May Affect Hospitalized Patients
Negative nitrogen balance represents the net loss of nitrogen from the body. The nitrogen lost reflects a balance between protein synthesis and protein breakdown, which are simultaneously occurring in all tissues of the body. A variety of stable isotopic studies have attempted to measure these absolute rates. A summary of these studies is shown in Table 8-2. Injury and infection result in the increased whole body flux of amino acids throughout the body. Catabolic rates outstrip rates of protein synthesis in the partially fed state; feeding enhances protein synthesis and attenuates the negative nitrogen balance that occurs. Despite feeding, protein flux rates remain high in these catabolic states, and these rates do not return to normal until the inflammation is resolved.
THE TRANSLOCATION OF NITROGEN FROM CARCASS TO VISCERA
Although the protein-catabolic response may appear to be a generalized effect of net protein breakdown throughout the body, this is not the case. Skeletal muscle is known to become catabolic; arterial-venous measurements of amino acid concentration across noninjured extremities of injured patients have documented the accelerated release of nitrogen that occurs from skeletal muscle during the flow phase (Aulick and Wilmore, 1979). However, catheterization of vessels supplying and draining visceral organs in similar patients has demonstrated enhanced uptake of amino acids (Wilmore et al., 1980), and these rates of uptake generally match the rates of peripheral amino acid release. That translocation of nitrogen occurs from the carcass to visceral organs is also supported by the following phenomena:
-
In addition to the nitrogen lost in the urine, there is increased excretion of potassium and phosphorous. All of these elements are lost in proportion to their concentrations found in skeletal muscle.
-
Following a catabolic event, creatinuria occurs. In addition, there is increased excretion of 3-methyl histidine. Both of these substances are found predominantly in skeletal muscle.
-
A common, clinical observation is that wasting of skeletal muscle occurs during catabolic states, and this is accompanied by muscle weakness.
A characteristic pattern of amine acids released from catabolic muscle has been described: alanine and glutamine constitute 50 to 70 percent of the amine acid nitrogen released (Muhlbacher et al., 1984). This represents accelerated de nero synthesis of these amine acids; hydrolysis and analysis of skeletal muscle protein reveals that both alanine and glutamine contribute less than 10 percent to the overall amine acid residues. Translocation of these amine acids to visceral tissues presumably occurs for a purpose; both amine acids serve as important glucose precursors and support the enhanced rate of gluconeogenesis. In the liver, nitrogen residue from this reaction is converted to other nonessential nitrogen compounds or converted to urea and excreted in the urine. Glutamine is also extracted by the kidneys, where it contributes ammonia to combine with hydrogen ions to form ammonium (NH4+), which is excreted in the urine. This is an important component of acid-base homeostasis, for the loss of every H+ in the urine is accompanied by the secretion of a bicarbonate ion into the bloodstream. Glutamine is also taken up by enterocytes, lymphocytes, and macrophages, where it serves as a primary fuel source for these tissues, supporting proliferation and signaling or supporting other important responses.
Thus, skeletal muscle serves as an important source of substrate during catabolic states to support vital structure and functions of the visceral tissue necessary for host survival. However, the increased loss of skeletal muscle protein occurs with a cost—the price paid is loss of muscle mass and the disturbance of normal muscle function. These alterations in composition of organs in catabolic states have been observed in both animal models (Artuson, 1961) and human patients following injury or infection (Bararc-Nieto et al., 1978) (Figure 8-2).
REGULATORS OF THE TRANSLOCATION OF PROTEIN
Various factors have been associated with the increased skeletal muscle proteolysis and translocation of nitrogen from the carcass to visceral organs. Anorexia commonly accompanies infection, and the diminished food intake causes hormonal changes, such as a fall in insulin and insulin-like growth factor-1, which decrease skeletal muscle protein synthesis. Infection and injury are also associated with a variety of constitutional symptoms including pain, fever, myalgia, headache, and fatigue. As activity decreases, or bed rest is imposed, the dynamics of net skeletal muscle protein synthesis are altered, enhancing net skeletal muscle proteolysis.
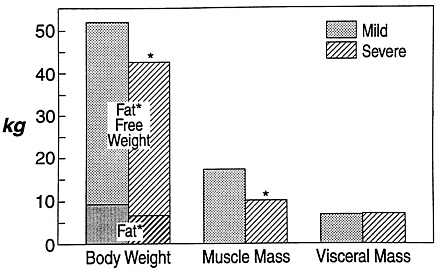
FIGURE 8-2 Body composition in patients with mild and severe chronic infection. Note the loss of fat-free tissue and muscle with severe infection, with maintenance of visceral tissues. *, P < 0.05. Source: Adapted from M. Bararc-Nieto et al., 1978.
Other factors that control intermediate metabolism are known to be associated with the altered protein metabolism that occurs during injury and infection. A characteristic hormonal response pattern has been observed during catabolism. Early in the time course, insulin levels are low, after which the blood levels generally rise to normal or supranormal levels. However, the counterregulatory hormones glucagon, glucocorticoids, and catecholemines are all elevated, and they generally remain so throughout the period of catabolism.
To evaluate the role of this altered hormonal environment, Bessey and associates (1984) and Watters and colleagues (1986) infused these catabolic hormones into normal volunteers with or without the addition of a pyrogen. Hormonal infusion caused many, but not all, of the responses observed following infection and injury. Specifically, when glucagon, glucocorticoids, and catecholemines were infused to achieve blood levels similar to those that occur in catabolic states, negative nitrogen balance, hypermetabolism, and accelerated gluconeogenesis were observed (Bessey et al., 1984). The administration of a pyrogen added fever and activation of many components of acute-phase response to this simple complex (Watters et al., 1986).
In subsequent studies, Bessey and Lowe (1993) used somatostatin to block insulin elaboration, as occurs during the early phases of injury or infection. Under these conditions, the triple hormone infusion resulted in much greater nitrogen losses, reminiscent of excretion rates observed in bum or seriously injured patients. Thus, the hormonal environment plays a major role in determining body protein balance. Hormonal changes during critical illness may
explain much of the negative nitrogen balance observed, especially if food intake is provided by enteral or parenteral support
Alterations in the hormonal environment are triggered by yet another set of signals, those of the cytokines and other pro-inflammatory mediators. Cytokines are glycoproteins synthesized by inflammatory cells and elsewhere in the body in response to inflammation and other noxious stimuli. Although these mediators primarily signal other cells in the local environment, they may be produced in abundance and can, on occasion, be detected in the blood stream (Cannon et al., 1990). Those cytokines central to the pro-inflammatory response are interleukin (IL-1), tumor necrosis factor (TNF), and IL-8 (a potent chemo-attractant). These and other pro-inflammatory mediators (e.g., complement, fatty acid metabolites, vascular endothelial factors) initiate both cellular (Hill et al., 1996) and hormonal changes (Hill et al., 1996; Michie et al., 1988) that induce skeletal muscle proteolysis (Mitch and Goldberg, 1996), alter carbohydrate metabolism, and stimulate hypermetabolism, all of which are components of the metabolic responses observed following injury and infection (Ling et al., 1997).
These pro-inflammatory cytokines are balanced by other endogenous factors. The elaboration of glucocorticoids is a result of cytokines signaling the central nervous system to initiate a pituitary-adrenal cortical response. Glucocorticoid elaboration is an important mechanism that attenuates cytokine effects and thus modulates catabolism (Santos et al., 1993). Other anti-inflammatory cytokines are elaborated (IL-4, IL-10, and IL-13), and these substances dampen or attenuate the inflammatory responses and enhance tissue viability and/or promote repair. Nutritional elements also serve as important antioxidants to diminish the effect of free radicals, which are also generated during the inflammatory process. Of particular importance during bacterial infections are vitamins A, C, and E and the minerals zinc and selenium (Grimble, 1993). Glutathione is the most important intracellular antioxidant, and the provision of the amine acids cystine (as N-acetyl-cystine or methionine) and glutamine is necessary to support the ongoing synthesis of this tripeptide. Other nutrients, such as omega-3 fatty acids and arginine may also be beneficial to ameliorate inflammation.
CONCLUSIONS
The mediators of the protein catabolic response are decreased food intake, reduced exercise, and a set of hormonal and cytokine signals that favor protein breakdown. Only now are researchers learning how to modify some of these factors in order to maintain the beneficial aspects of the protein catabolic response while attenuating those elements that appear to be deleterious to the host.
REFERENCES
Artuson, G. 1961. Pathophysiological aspects of the burn syndrome. Acta Chirg. Scand. 274:7-64.
Aulick, L.H., and D.W. Wilmore. 1979. Increased peripheral araino acid release following hum injury. Surgery 30:196-197.
Bararc-Nieto, M., G.B. Spurr, H. Lotere, M.G. Maksud. 1978. Body composition in chronic under nutrition. Am. J. Clin. Nut. 31:23-41.
Beisel, W.R., W.D. Sawyer, E.D. Ryll, and D. Crozier. 1967. Metabolic effects of intracellular infection in man. Ann. Int. Med. 67:744-779.
Bessey, P.Q., and K.A. Lowe. 1993. Early hormonal changes effect the catabolic response to trauma. Ann. Surg. 218:476-489.
Bessey, P.Q., J.M. Watters, T.T. Aoki, and D.W. Wilmore. 1984. Combined hormonal infusions simulate the metabolic response to injury. Ann. Surg. 200:264-281.
Cannon, J.G., R.G. Tompkins, J.A. Gelfand, H.R. Michie, G.G. Stanford, J.W.M. van der Meer, S. Endres, G. Lonnemann, J. Corsetti, B. Chernow, D.W. Wilmore, S.M. Wolff, J.F. Burke, and C.A. Dinarello. 1990. Circulating interleukin-1 and tumor necrosis factor in septic shock and experimental endotoxin fever. J. Infect. Dis. 161:79-84.
Coleman, W., and E.F. DuBels. 1915. Clinical calorimetry. VII calorimetric observations on the metabolism of typhoid patients with and without food. Ann. Int. Med. 15:887-938.
Cuthbertson, D.P. 1932. Observations on disturbance of metabolism produced by injury to the limbs. Quart. J. Med. 25:233-246.
Grimble, R.F. 1993. The maintenance of antioxidant defenses during inflammation. Pp. 347-366 in Metabolic Support of the Critically Ill Patient, D.W. Wilmore and Y.A. Carpentier, eds. New York: Springer-Verlag.
Hill, A.G., L. Jacobson, J. Gonzalez, J. Rounds, J.A. Majzoub, and D.W. Wilmore. 1996. Chronic central nervous system exposure to interleukin-1β causes catabolism in the rat. Am. J. Physiol. 271:R1142-R1148.
Hunter, J. 1794. A Treatise on the Blood Inflammation and Gunshot Wounds. London.
Kinney, J.M. 1966. Energy deficits in acute illness and injury. P. 174 in Proceedings of a Conference on Energy Metabolism and Body Fuel Utilization. A.P. Morgan, ed. Cambridge: Harvard University Press.
Ling, P.R., J.H. Schwartz, and B.R. Bistrian. 1997. Mechanisms of host wasting induced by administration of cytokines in rats. Am. J. Physiol. 272:E333-E339.
Michie, H.R., K.R. Manogue, D.R. Spriggs, A. Revhaug, S. O'Dwyer, C.A. Dinarello, A. Cerami, S.M. Wolff, and D.W. Wilmore. 1988. Detection of circulating tumor necrosis factor after endotoxin administration. N. Engl. J. Med. 318: 1481-1486.
Mitch, W.E., and A.L. Goldberg. 1996. Mechanisms of muscle wasting. N. Engl. J. Med. 335:1997-1905.
Muhlbacher, F., C.R. Knpadia, M.F. Colpys, R.J. Smith, and D.W. Wilmore. 1984. Effects of glucocorticoids on glutamine metabolism in skeletal muscle. Am. J. Physiol. 247:E75-E83.
Munro, H.N. 1964b. Historical introduction: The origin and growth of our present concept of protein metabolism. Pp. 1-29 in Mammalian Protein Metabolism, H.N. Munro and J.B. Allison, ed. New York: Academic Press.
Santos, A.A., M.R. Scheltinga, E. Lynch, E.F. Brown, P. Lawton, E. Chambers, J. Browning, and C.A. Dinarello. 1993. Receptor antagonist is not attenuated by glucocorticoids after endotoxemia. Arch. Surg. 128(2): 138-143.
Wannamacher, R.W., Jr. 1975. Protein metabolism (applied biochemistry). P. 133 in Total Parenteral Nutrition: Premises and Promises, H. Ghadimi, ed. New York: John Wiley.
Watters, J.M., P.Q. Bessey, C.A. Dinarello, S.M. Wolf, and D.W. Wilmore. 1996. Both inflammatory and endocrine mediatom stimulate host responses to sepsis. Arch. Surg. 121:179-190.
Wilmore, D.W., C.W. Goodwin, L.H. Aulick, M.C. Powanda, A.D. Mason, Jr., and B.A. Pruitt, Jr. 1980. Effect of injury and infection on visceral metabolism and circulation. Ann. Surg. 192:491-504.
Wilmore, D.W. 1991. Catabolic illness: Strategies for enhancing recovery. N. Engl. J. Med. 325(10):695-702.
Wilmore, D.W. 1997b. Homeostasis: Bodily changes in trauma and surgery. Pp. 55-67 in Textbook of Surgery, 15th ed., D.C. Sabiston, ed. Philadelphia: W.B. Saunders.
Wilmore, D.W. 1977. The Metabolic Management of the Critically Ill. New York: Plenum Medical.
Discussion
ROBERT NESHEIM: Thank you, Doug, for that very excellent and interesting presentation. It is open for questions.
JOHANNA DWYER: Just a quick question, Doug, on the dietary aspects. You mentioned a lot of antioxidants, some of which are nutrients. What about BHA and BHT? I remember that those are very potent antioxidants.
DOUGLAS WILMORE: I think there are a whole variety of antioxidants that can be utilized, Johanna. The reports on those are really a mixed bag simply because it is probably going to be difficult to see effects with single anti-oxidants. There is a very nice study, for example, of high-doses of vitamin C in long-distance runners who over-train, for example. But it is rare to find reports of single nutrients alone working as antioxidants that have major types of effects.
ROBERT NESHEIM: Wanda.
WANDA CHENOWETH: You have talked about antioxidants. What about pro-oxidants for example?
DOUGLAS WILMORE: For example?
WANDA CHENOWETH: Iron?
DOUGLAS WILMORE: Iron? Well, it is a good point. Bill may be able to make some comments about this. The classic teaching is that iron is the single
nutrient that should not be administered during infection and injury. If it is fixed in food and supplemented, in general, in a very controlled situation, you would take it out. You would not give it. This is simply because iron is redistributed after infection and your iron level in your blood stream becomes very low because it really is a very important substance for bacteria and some viruses to use for replication. So we would take it out or not give it. This, again, gets back to the problem of having fixed rations and all of the components in a ration, and the assumption that a ration that is appropriate for well people is appropriate for sick people, which is a concept that I do not embrace.
ROBERTNESHEIM: Dr. Millward.
D. JOE MILLWARD: I was just going to make the point in respect to BHT. I think that one of the interesting things about those sorts of antioxidants is that intakes of them are extremely high and we generally disregard them as important components of our food because they are there as additives. But, actually, we now take in large quantities Of these compounds. It may well be that they are in fact a major component of our antioxidant defense mechanism. I think that we should be thinking about them to a much greater extent than we do.
ROBERT NESHEIM: John Vanderveen.
JOHN VANDERVEEN: Doug, the question is whether, on a long-term basis or a short-term basis, we can build up body stores that are going to help us in the case of an infection or injury event, or is it something that is best handled in terms of a short preliminary type of preparative activity?
DOUGLAS WILMORE: That is a very strategic question from the standpoint of preparing people to go into maneuvers or go to do a task. I guess that one way we could start thinking about it, and this may not be the only way to think about it, would be to think in terms of water soluble and fat soluble substances. Because we can pretty well store many of the fat-soluble substances, but we would probably have to provide many of the water-soluble substances. When we talk about genetic predisposition or phenotype changing, it may well be that omega-three fats could be fed over the period of about three to four weeks, if you will, and we could change responses in just that short a period of time.
Vitamin E loading, in adequate doses, for example, takes about four weeks. And there are nice clinical data now from heart biopsies of patients undergoing cardiac surgery where various doses and time intervals have been used for vitamin E loading as an antioxidant.
The University of Toronto group has reported that you need about four weeks before you can get adequate vitamin E loading in cardiac muscle. So that is one way to think about it.
There is another way to think about it too and that is—it is not all that bad for many of these people to have a surfeit of adipose tissue which is their energy supply and an adequate skeletal muscle mass, because history has really told us that you can draw on those stores and that will enhance your survival as long as you are within some sort of a functional zone. So there may be some strategies that we can think about that will have to do with loading or preparing people. Mackenzie Walser and others may recall the interest people had with using odd-chain fats. Odd-chain fats would then provide you with gluconeogenic precursors. If you were starving for a long period of time, these fats would be drawn from your adipose tissue stores, but if you were provided with gluconeogenic precursors and you would not have to break down muscle as fast. So there have been talks about that in the past.
ROBERTNESHEIM: Dr. Walser.
MACKENZIE WALSER: Is the mechanism of that known? Does it have etiological value?
DOUGLAS WILMORE: Presumably it is there to protect circulating blood volume and oxygen delivery capacity. The mechanisms are through a lot of normal pathways that conserve salt and water.
ROBERT NESHEIM: Dr. Lieherman.
HARRIS LIEBERMAN: Doug, do we know anything about patterns of change in amine acids as a consequence of either acute or chronic illness, particularly those that are important to the brain, such as tryptophan or tyrosine?
DOUGLAS WILMORE: In general, all of the indispensable amine acids are lowered, I mean, as a sort of a general rule without knowing the specific pattern for studies. In general, all essential amine acids are low. The dispensable amine acids are generally normal or slightly increased. Joe, are there other thoughts? (No response.)
ROBERT NESHEIM: Steve Heymsfield, and then we will need to go on. We have a discussion period following the next presentation so we can cover many of these things then. Steve.
STEVEN HEYMSFIELD: You mentioned genetics as being one factor. Do gender or age affect the metabolic response to injury in any way?
DOUGLAS WILMORE: They must, but the age issue is exceedingly difficult to study because you introduce progressive organ dysfunction or change; you bring in other disease processes and things like that.
But when I was in the Army Burn Unit in the 1970s, we were able to sort surviving from nonsurviving patients by just their response to a cool environment. A cool environment, incidentally, was about this temperature (e.g. the temperature of an air conditioned room). It is one of the issues of why hospitals should not be cold. The older patients responded more rapidly to cold exposure than the younger individuals.
MICHAEL RENNIE: Can I just say something about that metabolic response? We have just done some studies on very young children, neonates. In fact, as far as we can tell, they do not show a metabolic response to injury. So that appears to be a developmentally-acquired response.
DOUGLAS WILMORE: And that is compatible with the fact that babies can actually undergo complete hormonal blockade with heart surgery and actually do better in terms of their overall response when compared with non blockaded patients.
ROBERT NESHEIM: Thank you very much. That was very interesting.














