Protein and Amino Acids, 1999
Pp. 217-242 Washington, D.C.
National Academy Press
10
Amino Acid Flux and Requirements: Counterpoint; Tentative Estimates Are Feasible and Necessary
Vernon R. Young
INTRODUCTION
This chapter serves as a counterpoint to that by Millward (see Chapter 9), in which he largely evaluates my views and those of my colleagues concerning the requirements for nutritionally indispensable amino acids in healthy adults. His points are multiple and well articulated, although he presents little direct experimental data from his own laboratories that are directly relevant to the quantitative determination of adult human amino acid requirements or that are in contrast to our own findings. Nevertheless, I agree with a number of important elements in his argument; indeed, I accept some of his criticisms of our work, which is overviewed briefly below. However, other major components of his position lack strength and/or validity. Of course, he, too, has made a similar statement about some of my ideas, concepts, and experimental data, while
attempting to reassess the quantitative needs for indispensable amine acids in adult humans and to assess their practical significance in human protein nutrition.
At the outset, I share his view that there are inherent difficulties in defining amine acid requirements. Indeed, Millward has provided an excellent backdrop against which I will make the case that the current international amine acid requirement values (FAO/WHO/UNU, 1985) for adults are ''no longer acceptable or nutritionally relevant" (Clugston et al.1996). Further, I will argue that the tentative requirement values that we have proposed (Young 1991, 1992; Young and El-Khoury, 1995; Young and Pellett, 1990) should be used for evaluating the amine acid adequacy of diets and/or for planning diets intended to meet adequately the physiological needs of consumers in healthy populations. With respect to the issue of indispensable amine acid requirements and dietary protein quality, the most recent UK expert panel (Department of Health, 1991), of which Millward was a member, stated: "DRVs (dietary reference values) assume that the dietary protein pattern includes sufficient variety of different protein-containing foods, or sufficient high quality protein sources to provide for indispensable amine acid (IAA) requirements." This assessment or advice would not be at all necessary if the IAA requirement estimates proposed by FAO/WHO/UNU (1985) and assumed to be adequate by Millward were valid. It would seem worthwhile then, to make our case for a significant revision in the current UN amine acid requirement values, while addressing a number of Millward's points and before concluding with a few comments about dietary protein and amine acids in relation to performance and exercise.
As Millward reminds us, much of the argument he makes has been reported previously. Similarly, my colleagues and I have also published our assessments and views elsewhere (Young et al., 1989; Young and El-Khoury, 1995, 1996). Hence, this chapter concentrates on many, but not all, of the particular issues raised by him. R is agreed, as he states: "The task in hand then is to define the amounts and amine acid pattern of the maintenance requirement."
Proposed Massachusetts Institute of Technology Amine Acid Requirement Pattern
In 1989, following up and extending on an idea concerning obligatory nitrogen (N) losses that was originally proposed by Millward and Rivers (1989), we adopted the values for the estimated obligatory amine acid losses (OAALs) as a partial basis for deriving the Massachusetts Institute of Technology Amine Acid Requirement Pattern (MIT-AARP). We also applied some of our data from 13C-amino acid tracer studies in deriving the AARP (Young et al., 1989). This pattern is summarized in Table 10-1, together with the proposed 1985 FAO/WHO/UNU patterns for preschool children and adults. Millward points out that the approach we adopted meant that we had defined the maintenance pattern
TABLE 10-1 The MIT Amino Acid Requirement Pattern (MIT-AARP) Compared with the 1985 FAO/WHO/UNU Patterns for Adults and Preschool Children
with the same amino acid composition of the mixed proteins in the body. He is correct except that the values for lysine, sulfur amino acids, and threonine, in particular, were derived more from the results of our tracer studies, which were limited in scope. This detail becomes important when we respond to the issues raised by Millward in his chapter.
Millward has three objections to the approach we followed in applying the OAAL. First, he criticizes the approach, because the OAAL for most amino acids were greater by 2 or more times than the FAO/WHO/UNU (1985) requirement values, with the exception of the total sulfur amino acids (methionine + cystine; SAA) for which the losses and FAO/WHO/UNU (1985) requirement values were quite close. He interprets this comparison by suggesting that the SAA "drives" the obligatory nitrogen loss. Further, this appears to be why Millward objects to our use of OAAL for purposes of deriving a tentative amino acid requirement pattern. However, the power of his argument depends, in the first instance, on an assumption of the validity and acceptance of the nutritional significance of the FAO/WHO/UNU amino acid requirement estimates. Because the latter can be legitimately questioned, his reasoning is problematical. Furthermore, it has been shown that the presence of a source of dietary cystine spares the methionine requirement, possibly by as much as 90 percent (Rose, 1957; Williams et al., 1974). Hence, in theory, the OAAL for methionine could be reduced to the equivalent of about one-quarter of the methionine loss under conditions of a high cystine intake. I am not aware, however, that dietary cystine, in contrast to methionine (Yoshida, 1986), reduces N excretion when experimental animals are given a protein-free diet. Furthermore, not only are short-term animal feeding studies of this kind difficult to interpret for their human nutritional significance, but similar experiments in adult humans are lacking. Thus, it is a matter of speculation whether the SAAs are rate determining for mobilization of whole body proteins under conditions of protein-free or amino acid-inadequate diets.
Second, in contrast to the position taken by Millward (1998, see Chapter 9), I conclude that there is a broad similarity between the amine acid pattern of mixed body proteins and the adult maintenance pattern. Indeed, the comparison that we made previously for the 1985 FAO/WHO/UNU preschool age pattern and body protein shows a close similarity (Young and El-Khoury, 1995). I recognize that this comparison may be complicated by the fact that the children in the studies that provide amine acid requirement values appear to have been retaining body protein at a much higher-than-normal rate (cf. FAO/WHO/UNU, 1985; Pineda et al., 1981). Thus, the dietary retention of indispensable amine acids may well have been more efficient than for a fully repleted child who was gaining body protein at a more usual rate. The latter rate accounts for a relatively small proportion of the total daily requirement (Young, 1991), but this does not necessarily mean that the amine acid requirement pattern would be that different.
Additionally, body protein maintenance in the adult involves depletion of body proteins during the fasting period of the day and their repletion during the fed period of the day. Thus, the pattern and composition of the retained amine acids must be that of mixed body proteins. The question then is whether this prandial gain would require, under steady state conditions of N balance and at intakes of N that met but did not significantly exceed the physiological need, an amine acid pattern (also at limiting and not excessive intakes) of IAA that is similar to that of body mixed proteins. Using leucine as an example, our tracer studies indicated that the leucine needed to achieve this balance, per unit of N intake, is in proportion to leucine in body proteins (El-Khoury et al., 1994a). Lysine metabolism shows a significant adaptive capacity when intakes are limiting. Thus, the question is whether the conclusion we have drawn for leucine also applies to lysine. Although we do not have the same extensive data for lysine, that we do for leucine, the fasting loss of lysine at a generous (77 mg/kg/day) lysine intake appears to be about 70 or 90 percent of that which we would predict from leucine oxidation, depending upon whether the route of 13C-lysine administration was intravenous or oral (El-Khoury et al., in press). At a limiting, probably inadequate, intake of lysine (12mg/kg/d) the fasting state rate of oxidation appears to be about 50 to 110 percent of the rate of leucine loss at an equivalent low leucine intake (Unpublished data, A.E. El-Khoury and V.R. Young, 1998). Hence, there appears to be some conservation of the lysine liberated during the postabsorptive period that may later be used for retention of protein and replenishment of earlier losses during the prandial phase. However, the difference between the measured and predicted oxidative loss of lysine and its replenishment during the prandial phase does not seem to be profoundly different from that for leucine. On this basis, a general similarity between the requirement pattern of amine acids and that of the mixed proteins in the body (with some differences that may not be large due to the specific characteristics of metabolism of the individual amine acids) appears to be a reasonable hypothesis. For lysine, the difference between our proposed lysine requirement (expressed per unit of protein requirement) and the concentration of lysine in whole body mixed
proteins amounts to about 30 percent, which is consistent with the lysine oxidation data mentioned above. This matter will be raised again below, in some detail, and with respect to the diurnal cycling of protein retention and loss.
Third, Millward concludes, from the animal data, that the maintenance pattern differs from that of growth, and criticizes our estimates for adult human amino acid requirements. I have said before that maintenance in a growing animal is a very different metabolic condition than that of nutritional maintenance in an adult and probably also that of a pre-school child. Further, the data for requirements for adult maintenance in different animal species are not only limited but also contradictory, as we both recognize (McLarney et al., 1996; Millward, 1998, see Chapter 9).
Millward's three positions are not supported adequately. A reasonable initial and tentative definition of the pattern and level of requirements for the IAA in adult humans can be made from estimates of OAALs (Young and El-Khoury, 1995).
Adaptive Aspects of Amino Acid Metabolism
Millward appropriately emphasizes that the adaptive component of amino acid metabolism brings complexity to the assessment of maintenance amino acid needs. The important practical question, however, is how long it takes the integrative aspects of whole body amino acid metabolism to adjust to a new level of amino acid, or total protein, intake so that a reasonable estimate of the steady state amino acid needs for that intake level and status of nutriture can be made by either N balance or tracer techniques. In our studies, we have used relatively short experimental diet periods of 6 to 7 days prior to the conduct of 13C-amino acid oxidation studies (Young, 1994). Millward (Chapter 9) concludes that the adaptation of oxidative catabolism to a change in protein intake is relatively slow and that the extent of postprandial oxidative catabolism reflects mainly the habitual rather than the actual or present protein intake supplied in meals. The validity of this conclusion depends on whether the metabolic responses and adaptations to altered protein or amino acid intakes are indeed as slow as Millward concludes. He bases his argument on his own studies of N balance and leucine oxidation following the transition from a high- to a low-protein diet in healthy adults.
The following six points are relevant, therefore, to the question of whether a 6-day period of "adaptation" is long enough to permit a suitable derivation of nutritionally meaningful amino acid requirement values:
-
Based on the earlier N excretion and balance studies carried out in our laboratories (Rand et al., 1976, 1979, 1985) and on our evaluation of the data published by others (Rand et al, 1981) it is entirely reasonable to propose that a 6 day adjustment period is suitable for purposes of establishing the amino acid needs of initially well-nourished subjects. Further, relatively short-term
-
experimental diet studies provide the database used by the UN group to establish adult amine acid requirements for populations worldwide (FAO/WHO/UNU, 1985). Quevedo et al. (1994), from their study on the transition of N balance from a high (292 mg N/kg/day) to lower (124 mg N/kg/day) protein diet, suggested that it takes longer and possibly more than 2 weeks for a new N equilibrium to be reached. However, their nitrogen excretion results are confounded by the apparently low energy intake supplied by their experimental diet. My calculations suggest that the energy supply in that experiment approximated 34 Kcal/kg/day, which means a physical activity level (PAL; total energy expenditure expressed as a multiple of the basal metabolic rate) of about 1.4. This value is much lower than the expected value of approximately 1.8 and above for young healthy adults (Goran et al., 1993; Roberts et al., 1990) and 1.6 to 1.8 for free-living elderly men (Roberts et al., 1995). On this basis, it appears likely that energy intake was inadequate and this raises doubts about whether their nitrogen balance data can be used to help answer adequately the question of the time required to achieve a new steady state of whole body protein metabolism when amine acid/protein intakes are changed.
-
A study was carried out in our laboratories to determine the rates of leucine oxidation (Marchini et al., 1993) and phenylalanine hydroxylation (Marchini et al., 1994) at the end of 1 and 3 weeks after giving healthy subjects diets supplying adequate L-amine acid mixtures patterned after (a) the FAO/WHO/UNU (1985) adult amine acid requirements, (b) the MIT tentative requirement values, and (c) whole hen's egg proteins. The kinetics of whole body leucine and phenylalanine catabolism during the fast and fed states did not differ between the 1- and 3-week time periods, again supporting the premise that a 6-day diet adjustment period is suitable for exploring aromatic amine acid kinetics, balances, and requirements in healthy adults. In addition, Zello et al. (1990) found that phenylalanine flux and oxidation rates at test intakes of phenylalanine ranging from 5 to 60 mg/kg/day (in the presence of generous tyrosine) were not affected by giving subjects a diet supplying 4.2 or 14 mg of phenylalanine per kg per day for up to 9 days prior to the kinetic measurements.
-
It could be argued that even a 3-week dietary period might not be long enough to reach a full degree of adaptation. However, this becomes an exceedingly difficult issue to resolve both conceptually and with respect to the implications of adaptive change for the determination of functional state and nutritional requirements.
-
Equally important to ask in this context of adaptive oxidative losses is whether the amine acid requirements of individuals in populations in developing regions of the world, particularly where protein and/or dietary lysine are likely to be more limiting and lower than for the U.S. diet, are similar to or different from those for U.S. subjects. This question is raised despite the fact that the current international FAO/WHO/UNU (1985) amine acid requirement values, based largely on studies conducted mainly in young adult American subjects (Irwin and Hegsted, 1971) are recommended for application world-wide.
-
To date, no major research effort outside of North America has carried out 13C-tracer studies of amino acid requirement comparable to those calculated at MIT or the University of Toronto. We have completed an initial study with our collaborators at St. John's Medical College, Bangalore, India (Kurpad et al., 1998), using the indicator amino acid oxidation technique (Zello et al., 1995) to assess the lysine requirement of healthy Indians whose long-term lysine intake appears to be about 60 percent of the level characteristic of the North American subjects. Results from this laboratory do not suggest that adequately nourished Indian subjects require a different lysine intake for maintenance as compared to MIT subjects. Further studies will need to be conducted to strengthen this initial but potentially very important conclusion.
-
There are few relevant data that can be used to predict whether the indispensable amino acid needs and the lysine requirement, in particular, are similar to or different among various population groups who have "adapted" to different dietary conditions. Studies of obligatory nitrogen losses in American (Calloway and Margen, 1971; Scrimshaw et al., 1972; Young and Scrimshaw, 1968), Chinese (Huang et al., 1972), Indian (Gopalan and Narasinga Rao, 1966), Nigerian (Atinmo et al., 1985; Nicol and Philips, 1976a) and Japanese men (Inoue et al., 1974) reveal that they are remarkably uniform (Bodwell et al., 1979). This implies similar obligatory amino acid losses (OAAL) and, by implication a probable similarity in the dietary requirements for indispensable amino acids (Young and El-Khoury, 1995), unless the efficiency of specific amino acid retention at requirement intake differs among apparently similar subjects in different population groups. Also, according to FAO/WHO/UNU (1955), nitrogen balance studies have not revealed any striking differences in estimates of total protein requirements in relation to body cell mass for well nourished subjects in different countries. The earlier studies by Nicol and Phillips (1976b), which might have suggested Nigerian men of low income utilize dietary protein more efficiently than, for example, U.S. students, are not appropriate to answer the question of whether requirements differ among populations. The N balance results in these Nigerian studies indicated that the subjects were depleted and were undergoing body protein repletion in response to the adequate diet given during the experiments. This confounds the nutritional interpretation of these studies and a later series carried out in Nigerian adult males (Atinmo et al., 1988) indicates that at maintenance nitrogen intakes, the efficiency of dietary protein utilization is the same as that for Caucasian and Asian subjects.
-
From our collaborative studies with Maroni and coworkers (Masud et al., 1994; Tom et al., 1995), it can be concluded that the postabsorptive rate of whole body leucine oxidation at the end of a 16 ± 2 month period during which non-acidotic chronic renal failure patients consumed a very low protein diet (0.28 g protein per kg per day) plus an amino acid-keto acid supplement was not different from that at the early phase of the low-protein intake. Furthermore, the leucine oxidation rate for these subjects was comparable to that reported in
-
healthy adults given either 14 or 38 mg leucine per kg per day for 6 days prior to the measurement of 13C=leucine kinetics (El-Khoury et al., 1994b).
In summary, it is proposed that a 6-day period of dietary "lead-in" permits an appropriate adjustment, or adaptation, to variable intakes of specific IAAs. This hypothesis should now be further validated through metabolic studies of varying duration and, preferably, in population groups in different geographic regions of the world.
Diurnal Cycling and Amine Acid Requirements
Millward and coworkers (1996) have conducted interesting and important studies on the relationships between the postabsorptive N losses and prandial N gains and how the magnitude of this diurnal cycle of protein metabolism is affected by the habitual and prevailing intake dietary protein. Millward also points out in Chapter 9 that the key question is the extent to which this diurnal cycle". . . influences the IAA composition of the adaptive metabolic demand" or, in other words, "the amounts and amine acid pattern of the maintenance requirement."
We accept, as concluded by Millward, that it does not necessarily follow that all of the IAAs liberated via tissue proteolysis are quantitatively oxidized. Therefore, a conservation of lysine (and perhaps threonine) may occur in the free amine acid pools, whereas there may be little conservation of other amine acids, such as leucine.
Regarding this metabolic issue, discussed above, evidence suggests some apparent conservation of lysine during the postabsorptive phase of amine acid metabolism. Millward uses the data of Bergstrom et al. (1990) to make his case for a significant contribution made by the free pool of lysine in muscle to the dietary retention of amine acids as body protein. Unfortunately, specific data on muscle free amine acid concentrations for the postabsorptive state are not given by Bergstrom et al. (1990). These data would be the most useful for evaluating the possibility of a postabsorptive conservation of lysine at different lysine intakes and in reference to estimations of the lysine requirement based on 13C-tracer techniques.
We have, however, considered this postabsorptive retention of lysine in relation to the fasted:fed ratio of lysine oxidation at a generous intake and whether this is far lower than that for leucine. If so, this would indicate that a significantly greater proportion of the lysine released from protein breakdown during the fasting state is retained within the flee lysine pool compared with that for leucine. Such a retention might occur, particularly in skeletal muscle and perhaps in other tissues and organs; the free lysine pool of muscle is relatively large and it responds to ingestion of protein-free and protein-containing meals (Bergstrom et al., 1990). Hence, there is a theoretical capacity to accommodate, or store, some free lysine that is liberated via proteolysis during the fasting
period. However, for a daily balance condition to be achieved, this "retained" lysine must contribute, in effect, to the total dietary intake during the fed period. Thus, it would have to be removed from the free lysine pool either via oxidation or re-incorporation into protein during the fed phase. A key issue, therefore, is the nature and extent to which changes occur in the size of the free lysine pool with feeding and fasting, under varying conditions of lysine intake.
Unfortunately, few direct experimental data have been published on changes in the concentration of free lysine and of leucine in the "metabolic free amino acid pool." Bergstrom et al. (1990) showed that a protein-rich meal increased the free lysine and leucine concentrations in muscle by 30 percent and 60 percent, respectively, at 3 hours after beginning a meal. When a protein-free meal was given, lysine and leucine concentrations declined by about 30 to 40 percent. Although these data are limited, they help us to interpret our estimates of lysine oxidation that are derived from plasma 13C-lysine values, in comparison with leucine oxidation-expected N losses (El-Khoury et al., in press). Thus, as summarized in Table 10-2, the measured rate of lysine oxidation during the 12-hour fast appeared to be close to 90 percent of that which would be predicted from leucine kinetics. This being so, the conclusion is that a relatively small retention of lysine causes a rise in the free lysine concentration in muscle tissue. The expected or predicted rise would fall well within the changes observed by Bergstrom et al. (1990) in subjects fed a protein meal. However, to maintain daily mass balance of lysine within the body free lysine pool, a fasting-state increase in muscle free lysine would have to be followed by an equivalent fall during the fed phase. This pattern of change is inconsistent with the data of Bergstrom et al. (1990), which again suggests that there might not be a significant retention of lysine arising from proteolysis in the free amino acid pool of muscle. Clearly, it would be worthwhile to obtain data on changes in the free amino acid concentrations in muscle tissue under the relevant dietary conditions of our 13C-amino acid tracer studies. However, technical difficulties and ethical constraints make this an unlikely research activity in the near future.
TABLE 10-2 Comparison of Rates of 13C-Lysine and 13C-Leucine Oxidation (expressed as IPNL) in Fast and Fed States at Generous IAA Intakes
Nitrogen Balance and Estimations of IAA Requirements
We have argued that N balance studies in adults as an assessment of amine acid requirements are of questionable value (Young 1987; Young and Marchini, 1990; Young et al., 1989). However, one problem with N balance data is illustrated simply by reference to those of Fisher et al. (1969) from which an estimate of the lysine requirement of college women was attempted. As summarized in Table 10-3 and according to these N balance results, body N balance can be achieved at lysine intakes of between 0 and 50 rag/day. This level of intake is so much lower than (a) our minimal estimates of postabsorptive lysine oxidation losses (Young and El-Khoury, 1995) and (b) the ileal losses of lysine reported by Fuller et al. (1994), that it would seem to be an invalid determination of the lysine requirement. Clearly, this points out a serious limitation of the N balance technique. A second problem with N balances is that if they are not determined with accuracy, then the estimate of the requirement for a specific amine acid will be in error as illustrated, for example, by the data of Rose et al. (1955b) and summarized in Table 10-4. If the reported N balance data shown here are recalculated to account for unmeasured N losses that are assumed to be about 8 mg/kg/day (FAO/WHO/UNU, 1985), then following the interpretation of the N balance results as used by these investigators, it must be concluded that the phenylalanine requirement of this subject exceeded 40 mg/kg/d. This contrasts with the conclusion of Rose et al. (1955b) that the phenylalanine requirement for the subject was 11 mg/kg/day.
A further problem that is related to the specific design of the N balance experiment, as well as the determination of body N balance per se, is illustrated in Figure 10-1. This figure depicts the sequence and length of a series of experimental diet periods providing different levels of lysine (Rose et al., 1955a). The lysine requirement was determined by these investigators to be 6.25 mg/kg/d (0.4 g lysine daily). However, the balance period that was used to derive this requirement immediately followed an earlier 5-day lysine-free diet period. This design would be expected to enhance the retention of lysine during the "requirement" phase of the study. For this reason, as well as the fact that only at
TABLE 10-3 An Illustration of the Limitation of Nitrogen Balance as a Basis for Estimation of Lysine Requirements in Five College Women
|
Time on Diet (days) |
Lysine Intake (rag/d) |
N balance (g/d)1 |
|
4 |
0 |
-0.67 ± 0.44 |
|
7 |
50 |
0.06 ± 0.08 |
|
5 |
0 |
-0.10 ± 0.15 |
|
5 |
50 |
0.43 ± 0.09 |
|
1 Mean values ± SE for five subjects. SOURCE: Adapted from Fisher et al., 1969. |
||
TABLE 10-4 Pbenylalanine Requirement of an Adult Human (H.I.E.)
the 1-g lysine intake level was N balance found to be positive when unmeasured losses of N were included in the determination of balance, it is reasonable to conclude that this requirement estimate of about 6 mg/kg is flawed.
In addition, even assuming that the design of the N balance study by Rose et al. (1955a) was appropriate, it is difficult to accept, on metabolic grounds, a lysine requirement of 6.25 mg/kg/d. The argument is as follows: if the plasma lysine flux is minimally about 70 to 100 μmol/kg/h (or 245 to 350 mg/kg/d), then at requirement intakes, the lysine oxidation would only amount to 1.7 to 2.6 percent of flux. At a lysine intake of 15 mg/kg/d, or a level that Millward
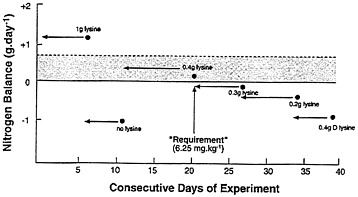
FIGURE 10-1 Rose's Estimate of the Lysine Requirement of Subject R.L.B. Graphic depiction of the design and results of an N balance study conducted by Rose et al (1955b) to estimate the minimum requirement for lysine in Subject RLB. The dashed line refers to the mean N balance for that period of days and the shaded area represents the difference between the measured and estimated N balance if miscellaneous losses of N had been taken into account. In this latter case the only period of zero or positive N balance was during the first period. The requirement for this subject was judged by Rose et al (1995b) to be 0.4g daily (6.25 mg.kg--l).
(Chapter 9) considers sufficient to meet a minimum obligatory metabolic demand, we estimate lysine oxidation to be approximately 21 mg/kg/d or about 10 percent of flux (unpublished data, A.E. El-Khoury and V.R. Young, 1998). On this basis, the estimate of Rose et al. (1955a) would appear to be far too low.
Millward and colleagues' (Millward et al., 1996) estimation (discussed earlier) of the magnitude of the prandial retention of protein necessary to balance subsequent post-absorptive losses appears to be at odds with estimates of the minimum requirement level for lysine; this observation is based on the following line of reasoning. First, for purposes of this argument, the minimum needs for total protein in adults can be estimated from the response of body nitrogen balances measured over relatively short experimental diet periods (e.g., 10 to 14 days) as discussed earlier. Second, at the minimum requirement intake level for total protein, the net prandial retention of protein would be just sufficient to balance the loss of protein during the postabsorptive period. Third, the magnitude of the prandial retention of N (protein = N x 6.25) for subjects consuming 0.77 g/kg/day for 10 days or more was reported to be equivalent to 251 mg/kg/12h (21 mg/kg/h) of protein (Price et al., 1994). These subjects, however, were in a daily negative balance of-9 mg N per kg, so it is uncertain whether a prandial retention of the magnitude observed is actually sufficient to maintain protein homeostasis. Nevertheless, from earlier 13C-leucine tracer studies (Motil et al., 1981), we found an approximate, net whole-body protein synthesis (protein synthesis minus protein breakdown) of about 23 mg/kg/h of protein during the prandial period in healthy adults with an intake of 0.8 g/kg/day of egg protein (Figure 10-2). This
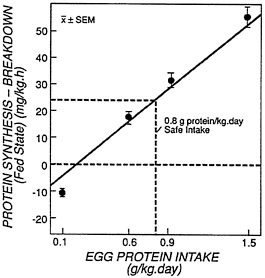
FIGURE 10-2 Net Protein Synthesis at a ''Safe" Intake of Protein. The relationship between protein intake and whole body net protein synthesis (protein synthesisprotein breakdown) in healthy young adult men during the absorptive phase of amine acid metabolism. Drawn from Motil et al (1981).
tracer-derived estimate is remarkably close to the N-balance data of Price et al. (1994), and thus a prediction might be made of the amount of dietary lysine minimally required to achieve this level of postprandial protein retention. Assuming that the lysine content of the protein retained is 78 mg/g of crude protein (Reeds, 1990) and that at a requirement level of lysine intake, the efficiency of dietary lysine retention is 80 percent, then the minimum necessary lysine intake to support this gain and subsequent loss of body protein would appear to be 27 mg/kg/d. This value is also comparable to most of the alternative lysine requirement estimates we have summarized (Young and El-Khoury, 1996) except for the far lower estimates based on N balance and the 1985 FAO/WHO/UNU recommendations.
For these reasons, we have not used N balance data to enhance interpretation of the results of our own tracer experiment that was concerned with testing the 1985 FAO/WHO/UNU amino acid requirement pattern (Marchini et al., 1993, 1994). We anticipated that the balance data would be difficult to assess for their nutritional significance and chose, instead, to evaluate the amino acid kinetic and plasma amino acid data. This does not mean, as Millward has incorrectly concluded, that we do not accept N balance data in adults under any circumstances. Rather it implies that we find the N balance technique in relatively short-term studies involving constant N intakes with a variable IAA composition to be of limited value. In contrast, when N intakes are varied over the submaintenance range of total protein needs during different periods, while possibly varying levels and/or sources of IAA, useful N balance data can be obtained. It is also in this context that our N balance data are consistent with our revised estimates of the lysine requirement.
The revised estimate of the lysine requirement in adults, 30 mg/kg/d (Table 10-1), has implications for the nutritional quality of wheat proteins (Young and Pellett, 1985). Thus, the lysine content of wheat products is summarized in Table 10-5, together with the lysine content of a number of FAO/W O amino acid
TABLE 10-5 Lysine Content of Wheat Flour Compared with other Foods or Amino Acid Requirement Patterns
|
Food Requirement Pattern |
Lysine Content (mg/g protein) |
|
Whole wheat flour |
24 |
|
Wheat flour (70 percent-80 percent extraction rate) |
20 |
|
Wheat bran |
16 |
|
Animal proteins |
85 ± 9 |
|
Legumes |
65 ± 7 |
|
1985 FAO/WHO/UNU pattern |
|
|
Adults |
16 |
|
School children |
44 |
|
Preschool children |
58 |
|
1991 FAO/WHO pattern |
58 |
|
MIT-AARP |
50 |
TABLE 10-6. Lysine Content of Whole Wheat Flour in Relation to an Estimate of Protein Quality
|
Amino Acid Pattern |
Amino Acid Score |
|
1985 FAO/WHO/UNU for adults |
>100 (L) |
|
1991 FAO/WHO |
41 (u) |
|
MIT-AARP |
48 (L) |
|
1985 FAO/WHO/UNU preschool child |
41 (L) |
|
NOTE: L, lysine, first limiting amino acid, not corrected for digestibility. |
|
scoring patterns. In addition, a usual concentration of lysine in most animal proteins and legumes and that for the MIT requirement pattern are also given for comparison in this table. Hence, if an amino acid score [(amino acid content in the food protein/amino acid content in the reference amino acid requirement pattern) x 100] were calculated for wheat flour, it would be greater than 100 when the 1985 FAO/WHO/UNU amino acid requirement pattern for the adult is used as the reference pattern (Table 10-6). This means that the nutritional value of wheat would be equal to that of high-quality, animal protein foods such as milk, egg, or beef. However, for scoring purposes, the FAO/WHO/UNU (1985) preschool amino acid pattern (or the 1991 FAO/WHO pattern) predicts a relative nutritional quality of 41 percent and with the MIT pattern, the score predicts a slightly higher value of 48 percent. In each case, lysine is predicted to be the most limiting amino acid. These preceding and lower estimates of the nutritional quality of wheat proteins in adults are consistent with the results of nitrogen balance experiments in healthy adults carried out at MIT approximately 20 years ago (Young and Pellett, 1985; Young et al., 1975).
The N balance response to graded intakes of test dietary protein in healthy adults, expressed as relative protein value (RPV = N balance slope using wheat/N balance reference protein x 100), was 54 for whole wheat protein, using beef protein as a reference (Table 10-7). Expressed as relative N requirement (RNR = 1/[amount of wheat protein to achieve N balance in 97.5 percent of population ÷ equivalent amount of beef protein] x 100), the response was about 56 (Table 10-7).
TABLE 10-7 Biological Assessment of the Nutritional Quality of Whole Wheat Proteins in Young Adults1
The MIT amino acid requirement pattern predicted a value of 48 (Tables 10-6 and 10-7). Hence, there is clearly very good agreement between these experimentally derived (nitrogen balance) values and the predicted nutritional quality of whole wheat proteins. In contrast, use of the 1985 FAO/WHO/UNU adult amino acid pattern gives an invalid estimate of the nutritional value of wheat protein, in that this pattern makes wheat proteins nutritionally equivalent to beef proteins. Notwithstanding the problems faced when attempting to aggregate N balance data across studies carried out in different laboratories or within the same laboratory on different occasions (e.g., Millward et al., 1989), our observations support the view that the 1985 FAO/WHO/UNU lysine requirement value of 12 mg/kg/d for the adult should be discarded. Further, they provide additional justification for the tentative requirement value of 30 mg/kg/d, proposed above (or 50 mg lysine per g of protein), and they strengthen our recommendation that this figure should be used until additional data become available that may make a further change in the recommendation both necessary and desirable.
Millward (Chapter 9) refers to "several long-term trials of both body weight and N balance maintenance on wheat" in support of his view that there is a relatively low requirement for lysine in adult maintenance. He refers to the Minnesota bread study as providing strong support for his proposition. Conducted by Bolourchi et al. (1968), the study reported that N balance was achieved in adults who were given daily, for 50 days, a 12-g N .diet (approximately 1g/kg/d of protein) in which wheat proteins supplied about 90 to 95 percent of total N intake. I estimate, from the data presented in their paper, a mean lysine intake of about 18 mg/kg/d. However, to prevent weight loss, these investigators found it necessary to provide their subjects a daily energy intake of 54 kcal/kg. This high energy intake confounds the interpretation of their N balance data, as previously discussed (Young 1987; Young and Marchini, 1990). Hence, this is a serious limitation of this study just as it was in the studies by Rose (1957).
In the North Carolina wheat study, Edwards et al. (1971) measured N balances in adults who were given a diet, for 15 to 29 days, that was based largely on wheat protein but also supplemented with other plant foods so that the level of lysine in the diet approximated 41 mg/g of protein or an intake of about 26 mg/kg/d (not 17 mg/kg as stated by Millward [1997]). Subjects in the study maintained body N equilibrium; these findings support our conclusions if we recognize that the daily lysine intake in this experiment exceeded the FAO/WHO/UNU requirement value (12 mg/kg) by about twofold. It also exceeded the mean requirement estimate (8.8 mg/kg/d) suggested by Rose et al. (1955a) by as much as threefold. For these reasons, I do not find that these N balance studies adequately support Millward's concept of "a relatively low metabolic demand for IAA in the human maintenance requirement."
Stable Isotope Estimates of Requirement Values
The 13C-tracer studies that we conducted to reassess and arrive at new tentative estimates of IAA requirements have been exhaustively examined by Waterlow (1996), who concluded, "Since I have failed to find any source of error large enough to account for the 2-3 fold difference from Rose's estimates, it is logical to look for sources of error in the old rather than the new figures." Millward (Chapter 9) lists the main problems related to the use of 13C-amino acid tracer studies for measurements of amino acid oxidation rates and amino acid balance. Two of the problems are less tractable and possibly insoluble, namely (1) the fact that the amount of tracer given is not massless and may influence balance and (2) the true precursor amino acid enrichment as compared with that measured to determine the oxidation rate.
With respect to the first problem, Fuller and Garlick (1994) and Millward (Chapter 9) appropriately point out that the distribution of the tracer and test diet amino acid over the 24-hour period might well affect the estimate of amino acid requirement. Furthermore, Fuller and Garlick (1994) conclude that this problem might give rise to an appreciable overestimate; this is potentially a real problem.
For example, in our 24-hour tracer studies with labeled tyrosine and phenylalanine (Basile-Filho et al., in press), we gave various tracers in equal amounts during both of the 12-hour fast and fed periods. At the low phenylalanine test intake, this meant that a significant fraction of the total dally intake was given during the postabsorptive phase. This raises the question about the relative utilization of phenylalanine when given in this way versus when it is supplied together with other amino acids. Again, as discussed earlier, the tissue free amino acid pools, particularly muscle, can serve as a reservoir for IAAs when intakes exceed immediate needs for protein synthesis (Bergstrom et al., 1990). Thus, these "stored" amino acids can later be used when there is an intake of the other IAAs. Millward accepts this possibility in his assessment of the recycling of free lysine liberated during the postabsorptive state. However, the extent to which the input of the 13C-labeled tracers during the fast actually determined the whole body fasting-state rate of tyrosine (and aromatic amino acid) oxidation or of leucine and lysine oxidation cannot be easily judged form the data we have gathered to date. Nevertheless, the rate of tyrosine oxidation during the fasting phase of the 24-hour day amounted to 10 μmol/kg/h for a tyrosine-free and an "intermediate" intake level of phenylalanine (38 mg/kg/d) and so this observation would seem to be appropriate for further evaluation. Thus, if it can be assumed that the obligatory loss of aromatic amino acids during file fasting period approximates about 7 μmol/kg/h or more (see Table I in Millward, Chapter 9), then it does not appear that the two tyrosine and one phenylalanine tracers given simultaneously in this study (total 7.7 μmol/kg/h) had any profound impact on the fasting rate of whole body aromatic amino acid oxidation. Again, the studies by Bergstrom et al. (1990) on the changes in muscle free amino acids in adult subjects following protein-free and protein-rich meals suggest that the tracer
doses of tyrosine and phenylalanine given during the fast are retained, at least in part, in tissue free amino acid pools. This effect is also implied in stable isotope tracer studies of amino acid oxidation, protein turnover, and calculated N balance, such as those by Millward and his colleagues (Quevedo et al., 1994), referred to earlier. Our studies with leucine as a tracer further support this contention, in view of the agreement obtained between measured and leucine-derived estimates of N excretion (El-Khoury et al., 1994a). This applies also to the postabsorptive rate of leucine oxidation (El-Khoury et al., 1995). Therefore, Millward's concern about the uncertainty of whether our estimates of leucine requirement are due to excessive postabsorption oxidative loss seems to be lessened by these considerations of the data. However, it remains for future studies to fully resolve this problem of level of isotope administration given during the fast period as well as how best to provide the daily test amino acid intake level during the 24-hour stable isotope tracer protocol.
The second of the less-tractable problems in the 13C-amino acid tracer studies concerns the enrichment of the amino acid precursor pool undergoing oxidation. This is not a problem with respect to leucine and the other branched-chain amino acids since the oxidation is measured in terms of the enrichment of their intracellularly derived keto acids in plasma. This is supported by the agreement between measured N excretion and that predicted from leucine oxidation when the level of trace amino acid intake in the dietary protein (percent w/w) matches that in mixed proteins of the body (El-Khoury et al., 1994a). The lack of agreement between the leucine oxidation and N balance data mentioned by Millward (Chapter 9) and in reference to his study with Price et al. (1994) was very likely due to (1) the choice of the timing of the amino acid oxidation measurements in relation to the daily N excretion determinations, and (2) a mismatch (uncorrected for) between the tracer plus dietary leucine intake per unit of N and the leucine concentration in mixed proteins in the body.
The indicator amino acid oxidation technique (Zello et al., 1990, 1993, 1995) involving 13C-phenylalanine (Zello et al., 1993) and 13C-leucine (Kurpad et al., 1998), as labeled indicators, potentially reduces the severity of the two problems noted above. Using 13C-phenylalanine as an indicator, the Toronto group has estimated the lysine requirement to be on the order of 40 mg/kg/d, using a "break point" analysis of the 13CO2 output data (Duncan et al., 1996; Zello et al., 1993). These findings support our tentative new requirement estimates for lysine (Table 10-1). Millward (Chapter 9) criticizes the Toronto studies in human subjects, even though they were designed in relation to the various indicator amino acid studies conducted in experimental animals, which have given amino acid requirement estimates that are consistent with those derived from growth studies (Zello et al., 1995). In a recent paper, Millward (1997) also faulted the lysine requirement values reported by Zello et al. (1993), which were obtained by the indicator oxidation method, because they made no attempt to allow for any adaptation in lysine oxidation due to a reduced intake. The dietary design used by Zello et al. (1993), as well as by Duncan et al. (1996) in a similar followup study,
involved giving test intakes of lysine on only one day during a dietary period when subjects were otherwise consuming an experimental diet that supplied a reasonably generous amount of lysine (60 mg/kg/day). Hence, it seems possible that this design would lead to a lower, rather than higher, break point on the lysine intake-indicator amine acid oxidation curve. This is because there could well be a "replete" free lysine pool that would serve as an unaccounted source of utilizable lysine, in addition to the actual intake supplied by the six small hourly meals given beginning 2 hours before and during the 4-hour isotope tracer study. It is also of possible interest that in estimating protein requirements in healthy elderly and young adults from 13C-leucine balance studies, Millward and colleagues (Fereday et al., 1997) followed a somewhat analogous design. They conducted their 13C-leucine balance estimates in subjects who had continued consuming their usual protein intakes until the night before the 9-hour tracer infusion protocol began the following morning.
Finally, in an unpublished study, conducted in collaboration with Anura Kurpad in Bangalore, India, we determined the daily (24-hour) oxidation rate of leucine at graded lysine intakes. Our preliminary results, depicted in Figure 10-3, suggest, once again, an approximate lysine requirement of 30 mg/kg/day.
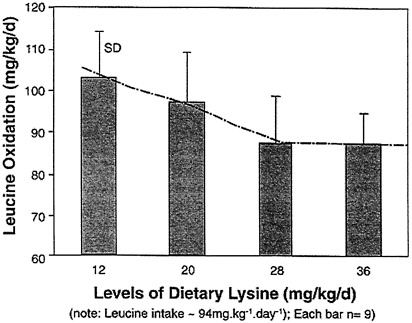
FIGURE 10-3 Relationship between 24h leucine oxidation and graded dietary lysine intakes (at constant N and other IAAs) in healthy Indian adult males. Unpublished, preliminary data of A. Kurpad and coworkers (Bangalore, India). For other experimental details see Kurpad et al. (1998).
The MIT Amino Acid Requirement Pattern and Physical Activity
With respect to physical activity, two final points need to be emphasized regarding the perspective of Millward and coworkers on the MIT nmino acid requirement pattern. First, in a critique of one of our papers published in support of our tentative MIT amino acid requirement pattern, Millward (1997) concluded that". . . given (1) the low minimal obligatory needs for indispensable amino acids,( 2) the fact that their metabolic demand reflects the extent to which adaptive changes in oxidation occur, and (3) the growing evidence for the availability of indispensable amino acids, including lysine, deriving from colonic microbial de novo amino acid synthesis from salvaged urea . . ., in my view, definition of adult indispensable amino acid requirements for protein quality scoring is not currently possible or likely to be useful in the future." We (Merges et al., 1997) and others (Gibson et al., 1997; Tanaka et al., 1980; Tollardona et al., 1994; Yeboah et al., 1996) have obtained evidence for the uptake into body tissues of lysine that is derived from intestinal microbial synthesis. However, the extent to which this source of lysine serves as a net contribution to the total lysine intake, supplied from dietary sources, is still to be determined. Assuming there is also a microbial synthesis and uptake of the other IAAs, as well as lysine, our studies with leucine (El-Khoury et al., 1994a, 1996) and those of Millward and colleagues (Price et al., 1994), suggest that this microbial source of IAAs compensates, in large part, for the losses of IAAs via the turnover and secretion of intestinal proteins and the subsequent oxidative catabolism of the liberated amino acids by the activity of the microflora and possibly intestinal tissues. In this case, a cycle of lysine (or IAA) synthesis within the gut lumen and the oxidative catabolism of lumen IAAs could be envisaged as a metabolic cycle that contributes to IAA homeostasis but that would not necessarily complicate estimation of the whole body kinetics and oxidation of the IAAs with the 13C-tracer techniques as currently employed in our laboratories and those of others, including Millward and collaborators.
Second, the above conclusion by Millward reminds me of an editorial by Yates (1983), in which he quotes Murphy (1982), as follows: "The growing point of science is discrepancy... the next best thing to uttering the truth is to say something so sufficiently definite that it can be constructively disagreed with." Indeed, I believe that the definitive conclusion drawn by Millward is neither acceptable nor helpful, despite the many real difficulties that he has noted appropriately in relation to defining the amino acid requirements in humans, and I have tried to be constructive in my disagreement.
If a requirement is defined, according to the definition proposed by FAO/IAEA/WHO (1996), as "the lowest continuing level of nutrient intake, that at a specified efficiency of utilization, will maintain the defined level of nutriture in the individual" then it should be possible to define a requirement for any one of the IAAs, just as is the case for many of the trace elements and vitamins. I agree with Millward that the requirement would best be established on the basis
of longer term experimental studies and on the functional consequences of variable intakes of protein and/or its constituent IAAs. This is an ideal goal for the future. In the interim, reasonable estimates of intakes minimally necessary to achieve body amino acid balance, after a suitable period of adaptation as discussed above, can be made. These would permit rational and safe decisions and policy concerning the maintenance of a satisfactory state of body protein and amino acid nutriture in both individuals and in defined population groups, including the military, which is the particular focus of concern in this report. Accordingly, it is prudent to accept the MIT amino acid requirement pattern (Table 10-1) in practical considerations of human protein nutrition, until strong data to the contrary are forthcoming. Millward's model (Chapter 9), which views amino acid metabolism and oxidative losses according to (a) a minimum obligatory metabolic demand and (b) an adaptive component, is certainly interesting and possibly might aid in the further scrutiny of the metabolic aspects of amino acid homeostasis at differing intakes of protein or IAA. It does not help solve the problems that are of greatest concern, namely (a) the practical nutritional relevance of the amino acid requirement values proposed initially by Rose and used together with comparable data from other investigators to arrive at the 1985 FAO/WHO/UNU adult indispensable amino acid requirement pattern and (b) determination of amino acid requirement values that are likely to be minimally sufficient to maintain body amino acid homeostasis, and by implication adequate body function, over the long term.
Millward (Chapter 9) refers to the studies by Chittenden (1904), which I have also found to be interesting and relevant (Young, 1997). As I have reviewed (Young, 1997), Millward (Chapter 9) concludes, "Chittenden's views were controversial at the time and have been given less prominence than they deserve in the intervening years of continued controversy. However, the data speak for themselves." I agree that they do. Thus, it might be of interest that the (calculated) lysine intake by Chittenden himself, who consumed a diet supplying about 0.75 g protein per kg body weight daily, approximated 46 mg/kg/day. This intake was possibly somewhat higher than for the soldiers, whose lysine intakes might be estimated to have been around 30 to 40 mg/kg/d, as judged from the limited dietary data given in Chittenden's extensive summary of his now classic experiments (Chittenden, 1904). One might wonder whether the performance of the soldiers, which actually improved over the 5-month experiment, would have deteriorated had the IAA intakes approximated those proposed by the international group (FAO/WHO/UNU, 1985) or those low values that Millward (Chapter 9) proposes as being sufficient to meet the minimum obligatory metabolic demand.
We have not examined this latter question specifically in relation to a further evaluation of the status of amino acid metabolism and balance at IAA intakes that either meet our tentative requirements or those approximating the 1985 FAO/WHO/UNU values. However, I am prepared to speculate from the experiments described in Marchini et al. (1993), that there would be a
deterioration in amino acid balance and, consequently, in function at intakes not exceeding the 1985 FAO/WHO/UNU requirement figures or those of Millward that are estimated to meet the minimum obligatory metabolic demand. At least, we can say, based on El-Khoury et al. (1997), that at a more generous IAA and leucine intake (diet and tracer contributing about 126 mg/kg/d) for two 90 rain periods of exercise at 45 percent VO2max, during both the fasting and fed periods of the day, resulted in a small overall increase in the rate of leucine oxidation, amounting to about 5 mg/kg/d. It would be interesting and highly worthwhile to repeat this detailed metabolic/exercise study but at dietary IAA intakes that are far more limiting.
Author's Conclusions and Recommendations
I have addressed a number of the issues raised by Millward (Chapter 9) in order to weigh the merits of our respective views. It is my judgment that the minimum physiological requirements for amino acids to maintain health and body function should be defined and that it would be prudent to apply the tentative requirement values proposed herein for the protein and amino acid nutrition of healthy adults. Clearly, there are difficulties involved in estimating amino acid requirements and limitations to the experiments that have been conducted to date, both by ourselves and by others. I concur with Rose (1957), who stated in his review article: "In view of the difficulties of such studies, it is to be hoped that many more will be made in both men and women and in different laboratories." This is crucial, given the importance of nutrient requirement data for the appropriate planning of world food and dietary protein needs (Young et al., 1998), not to mention the rational design of safe and effective diets for maximizing and maintaining physical performance.
REFERENCES
Atinmo, T., C.M.F. Mbofung, M.A. Hussain, and B.O. Osotimehin. 1985. Human protein requirements: Obligatory urinary and faecal nitrogen losses and the factorial estimation of protein needs of Nigerian male adults. Br. J. Nutr. 54:605-611.
Atinmo, T., C.M.F. Mbofung, G. Eggum, and B.O. Osotimehin. 1988. Nitrogen balance study in young Nigerian adult males using four levels of protein intake. Br. J. Nutr. 60:451-458.
Basile-Filho, A., L. Beaumier, A.E. El-Khoury, Y.-M. Yu, M. Kenneway, R.E. Gleason, and V.R. Young. 1998. Twenty-four hour L-[l-13C]tyrosine and L-[3,3-2H]phenylalanine oral tracer aromatic amino acid requirements in adults. Am. J. Clin. Nutr. 67:640-659
Bergstrom, J., P. Fürst, and E. Vinnars. 1990. Effect of a test meal, without and with protein, on muscle and plasma free amino acids. Clin. Sci. 79:331-337.
Bodwell, C.E., E.M. Schuster, E. Kyle, B. Brooks, M. Womack, P. Steele, and R. Ahrens. 1979. Obligatory urinary and fecal nitrogen losses in young women, older men and young men: Factorial estimation of adult human protein requirements. Am. J. Clin. Nutr. 32:2450-2459.
Bolourchi, S., C.M. Friedeman, and O. Mickelson. 1968. Wheat flour as a source of protein for adult human subjects. Am. J. Clin. Nutr. 21:827-835.
Calloway, D.H., and S. Margen. 1971. Variation in endogenous nitrogen excretion and dietary nitrogen utilization as determinants of human protein requirements. J. Nutr. 101:205-216.
Chittenden, R.H. 1904. Physiological Economy of Nutrition. N.Y.: Frederick A. Stokes Co.
Clugston, G., K.G. Dewey, C. Field, J. Millward, P. Reeds, N.S. Scrimshaw, K. Tontisirin, J.C. Waterlow, and V.R. Young. 1996. Report of the working group on protein and amino acid requirements. Europ. J. Clin. Nutr. 50:S193-S195.
Department of Health, Panel on Dietary Reference Values of the Committee on Medical Aspects of Food Policy. 1991. Report on Health and Social Subjects, Number 41. Dietary Reference Values for Food Energy and Nutrients for the United Kingdom. London: Her Majesty's Stationary Office (HMSO).
Duncan, A.M., R.O. Ball, and P.B. Pencharz. 1996. Lysine requirement of adult males is not affected by decreasing dietary protein intake. Am. J. Clin. Nutr. 64:718-725.
Edwards, C.H., L.K. Booker, C.H. Rmphg, W.G. Wright, and S. Ganapathy. 1971. Utilization of wheat by adult man: Nitrogen metabolism, plasma amino acids and lipids. Am. J. Clin. Nutr. 24:181-193.
El-Khoury, A.E., N.K. Fukagawa, M. Sanchez, R.H. Tsay, R.E. Gleason, T.E. Chapman, and V.R. Young. 1994a. Validation of the tracer balance concept with reference to leucine: 24-h intravenous tracer studies with L-[l-13C]leucine and [15N-15N]urea. Am. J. Clin. Nutr. 59:1000-1011.
El-Khoury, A.E., N.K. Fukagawa, M. Sanchez, R.H. Tsay, R.E. Gleason, T.E. Chapman, and V.R. Young. 1994b.. The 24-h pattern and rate of leucine oxidation, with particular reference to tracer estimates of leucine requirements in healthy adults. Am. J. Clin. Nutr. 59:1012-1020.
El-Khoury, A.E., M. Sanchez, N.K. Fukagawa, and V.R. Young. 1995b. Whole body protein synthesis in healthy adult humans; 13CO2 technique vs. plasma precursor approach. Am. J. Physiol. 268:E174-E184.
El-Khoury, A.E., A.M. Ajami, N.K. Fukagawa, T.E. Chapman, and V.R. Young. 1996. Diurnal pattern of the interrelationship among leucine oxidation, urea production, and hydrolysis in humans. Am. J. Physiol. 271:E563-E573.
El-Khoury, A.E., A. Forslund, R. Olson, S. Branth, A. Sjodin, A. Andersson, A. Atkinson, A. Selvaraj, L. Hambraeus, and V.R. Young. 1997. Moderate exercise at energy balance does not affect 24h leucine oxidation or nitrogen retention in healthy men. Am. J .Physiol. 273:E394-E407.
El-Khoury, A.E., A. Basile, L. Beaumier, S.Y. Wang, H.A. AI-Amiri, A. Selvaraj, S. Wong, A. Atkinson, A.M. Ajami, and V.R. Young. 1998. Twenty-four hour intravenous and oral tracer studies with L-]l-13C]-2-aminoadipic acid and L-[l-13C]lysine as tracers at generous nitrogen and lysine intakes in healthy adults. Am. J. Clin. Nutr. (in press).
FAO/IAEA/WHO (Food and Agriculture Organization of the United Nations/International Atomic Energy Agency/World Health Organization). 1996. Trace Elements in Human Nutrition and Health. Geneva: WHO.
FAO/WHO (Food and Agriculture Organization of the United Nations/World Health Organization). 1991. Protein Quality Evaluation. Report of a Joint FAO/WHO Expert Consultation. FAO Food and Nutrition Paper 51. Rome, Italy: FAO.
FAO/WHO/UNU ((Food and Agriculture Organization of the United Nations/World Health Organization)/United Nations University). 1985. Energy and Protein Requirements. Technical Report Series Number 724. Geneva: WHO.
Fereday A., N.R. Gibson, M. Cox, P.J. Pacy, and D.J. Millward. 1997b. Protein requirements and aging: Metabolic demand and efficiency of utilization. Brit. J. Nutr. 77:635-702.
Fisher H., M.K. Brush, and P. Griminger. 1969. Reassessment of amino acid requirements of young women on low nitrogen diets. 1. Lysine and tryptophan. Am. J. Clin. Nutr. 22:1190-1196.
Fuller, M.F., and P.J. Gatlick. 1994. Human amino acid requirements: Can the controversy be resolved. Ann. Rev. Nutr. 14:217-241.
Fuller, M.F., A. Milne, C.I. Harris, T.M.S. Reid, and R. Keenan. 1994. Amino acid losses in ileostomy fluid on a protein-free diet. Am. J. Clin. Nutr. 59:70-73.
Gibson, N., E. Al-Sing, A. Badalloo, T. Forrester, A. Jackson, and D.J. Millward. 1997. Transfer of 15N from urea to the circulating dispensable and indispensable nmino acid pool in the human infant. Proc. Nutr. Soc. 56:79A.
Gopalan, C., and B.S. Narasinga Rao. 1966. Effect of protein depletion on urinary nitrogen excretion in undernourished subjects. J. Nutr. 90:213-218.
Goran, M.I., W.H. Beer, R.R. Wolfe, E.T. Poehlman, and V.R. Young. 1993. Variation in total energy expenditure in young healthy free-living men. Metabolism 42:487-496.
Huang, P.C., H.E. Chong, and W.M. Rand. 1972. Obligatory urinary and fecal nitrogen losses in young Chinese men. J. Nutr. 102:1605-1614.
Inoue, G., Y. Fujita, K. Kishi, S. Yamamoto, and Y. Niyama. 1974. Nutritive values of egg protein and wheat gluten in young men. Nutr. Pep. Intl. 10:201-207.
Irwin, M.I., and D.M. Hegsted. 1971. A conspectus of research on amino acid requirements of man . J. Nutr. 101:539-566.
Kurpad, A.V., A.E. El-Khoury, L. Beaumier, A. Srivatsa, R. Kuriyon, T. Raj, S. Borgonha, A.M. Ajami, and V.R. Young. 1998. An initial assessment, using 24-hour 13C-leucine kinetics, of the lysine requirement of healthy adult Indian subjects. Am. J. Clin. Nutr. 67:58-66.
Marchini, J.S., J. Cortiella, T. Hiramatsu, T.E. Chapman, and V.R. Young. 1993. The requirements for indispensable amino acids in adult humans: A longer-term amino acid kinetic study, with support for the adequacy of the Massachusetts Institute of Technology amino acid requirement pattern. Am. J. Clin. Nutr. 58:670-683.
Marchini, J.S., J. Cortiella, T. Hiramatsu, L. Castillo, T.E. Chapman, and V.R. Young. 1994. Phenylalanine and tyrosine kinetics for different patterns and indispensable amino acid in adult humans. Am. J. Clin. Nutr. 60:79-86.
Masud, T., V.R. Young, T. Chapman, and B.J. Maroni. 1994. Adaptive responses to very low protein diets: The first comparison of ketoacids and essential amino acids . Kidney Intl. 45:1182-1192.
McLarney, M.J., P.L. Pellett, and V.R. Young. 1996. Pattern of amino acid requirements in humans: An across-species comparison of published amino acid requirement recommendations. J. Nutr. 126:1871-1882.
Metges, C.C., A.E. El-Khoury, K.J. Petzke, S. Bedri, M.F. Fuller, and V.R. Young. 1997. The quantitative contribution of microbial lysine to lysine flux in healthy male subjects. Abstract. FASEB J. 11(3):A149. Millward, D.J. 1997. Human amino acid requirements. J. Nutr. 127:1842-1846.
Millward, D.J., and J.P. Rivers. 1989. The nutritional role of indispensable amino acids and the metabolic basis for their requirements. Europ. J. Clin. Nutr. 42:367-393.
Millward, D.J., A.A. Jackson, G. Price, and J.P.W. Rivers. 1989. Human amino acid and protein requirements: Current dilemmas and uncertainties. Nutr. Res. Rev. 2:109-132.
Millward, D.J., A. Fereday, N. Gibson, and P.J. Pacy. 1996. Postprandial protein metabolism. Bailliere's Clin. Endocrin. Metab. 10:533-549.
Motil, K.J., D.E. Matthews, D.M. Bier, J.F. Burke, H.N. Munro, and V.R. Young. 1981. Whole-body leucine and lysine metabolism: Response to dietary protein in young men. Am. J. Physiol. 240:E712-E721.
Murphy, E.A. 1982. Muddling, meddling and modeling. Pp. 333-348 in Genetic Basis of the Epilepsies, V.E. Anderson, W.A. Hauser, J.K. Penry, and C.F. Sing, eds. New York: Raven.
Nicol, B.M., and P.G. Phillips. 1976a. Endogenous nitrogen excretion and utilization of dietary protein. Br. J. Nutr. 35:181-193.
Nicol, B.M., and P.G. Phillips. 1976b. The utilization of dietary protein by Nigerian men. Br. J. Nutr. 36:337-351.
Pineda, O., B. Torun, F.E. Viteri, and G. Arroyave. 1981. Protein quality in relation to estimates of essential amine acid requirements. Pp. 29-42 in Protein Quality in Humans: Assessment and In Vitro Estimation, C.E. Bodwell, J.S. Adkins, and D.T. Hopkins, eds. Westport, Conn.: AVI Publishers.
Price, G.M., D. Halliday, P.J. Pacy, M.R. Quevedo, and D.J. Millward. 1994. Nitrogen homeostasis in man: Influence of protein intake on the amplitude of diurnal cycling of body nitrogen. Clin. Sci. 86:91-102.
Quevedo, M.R., G.M. Price, D. Halliday, P.J. Pacy, and D.J. Millward. 1994. Nitrogen homeostasis in man: Diurnal changes in nitrogen excretion, leucine oxidation and whole body leucine kinetics during a reduction from a high to a moderate protein intake. Clin. Sci. 86:185-193.
Rand, W.M., V.R. Young, and N.S. Scrimshaw. 1976. Change of urinary nitrogen excretion in response to low protein diets in adults. Am. J. Clin. Nutr. 29:639-644.
Rand, W.M., N.S. Scrimshaw, and V.R. Young. 1979. An analysis of temporal patterns in urinary nitrogen excretion of young adults receiving constant diets at two nitrogen intakes for 8 to 11 weeks. Am. J. Clin. Nutr. 32:1408-1414.
Rand, W.M., N.S. Scrimshaw, and V.R. Young. 1981. Conventional ("long-term") nitrogen balance studies for protein quality evaluation in adults: Rationale and limitations. Pp. 61-94 in Protein Quality in Humans: Assessment and In Vitro Estimation, C.E. Bodwell, J.S. Adkin, and D.T. Hopkins, eds. Westport, Conn.: AVI Publishing.
Rand, W.M., N.S. Scrimshaw, and V.R. Young. 1985. A retrospective analysis of long-term metabolic balance studies: Implications for understanding dietary nitrogen and energy utilization. Am. J. Clin. Nutr. 42:1329-1350.
Reeds, P.J. 1990. Amine acid needs and protein scoring patterns. Proc. Nutr. Sec. 49:489-497.
Roberts, S.B., M.B. Heyman, W.J. Evans, P. Fuss, R. Tsay, and V.R. Young. 1990. Dietary requirements of young adult men, determined using doubly labeled water method. Am. J. Clin. Nutr. 54:499-505.
Roberts, S.B., P. Fuss, M.B. Heyman, and V.R. Young. 1995. Influence of age on energy requirements. Am. J. Clin. Nutr. 62:1053S-1058S.
Rose, W.C. 1957. The amine acid requirements of adult man. Nutr. Abstr. Rev. 27:631-647.
Rose, W.C., A. Borman, M.J. Coon, and G.F. Lambert. 1955a. The amine acid requirements of man. X. The lysine requirement. J. Biol. Chem. 214:579-587.
Rose, W.C., B.E. Leach, M.J. Coon, and G.F. Lambert. 1955b. The amine acid requirements of man. IX. The phenylalanine requirement. J. Biol. Chem. 213:913-922.
Scrimshaw, N.S., M.A. Hussain, E. Murray, W.M. Rand, and V.R. Young. 1972. Protein requirements of man: Variations in obligatory urinary and fecal nitrogen losses in young men. J. Nutr. 102:1595-1604.
Tanaka, N., K. Kubo, K. Shiraki, H. Koishi, and H. Yoshimura. 1980. A pilot study on protein metabolism in the Papuan New Guinea Highlanders. J. Nutr. Sci. Vitaminol. 26:247-259.
Tollardona, D., C.I. Harris, E. Milne, and M.F. Fuller. 1994. The contribution of intestinal microflora to amine acid requirements in pigs. Pp. 245-248 in Proceedings of the 6th International Symposium on Digestive Physiology in Pigs, W.B. Souffran, and H. Hagemeister, eds. Dummerstorf, Germany: Forsch. Inst. Biol. Landwirtsch. Nutztiere.
Tom K., V.R. Young, T. Chapman, T. Masud, L. Akpele, and B.J. Maroni. 1995. Long-term adaptive responses to dietary restriction in chronic renal failure. Am. J. Physiol. 268:E668-E677.
Waterlow, J.C. 1996. The requirements of adult man for indispensable amino acids. Europ. J. Clin. Nutr. 50:S151-S179.
Williams, H.H., A.E. Harper, D.M. Hegsted, G. Arroyave, L.E. Hold, Jr. 1974. Nitrogen and amino acid requirements. Pp. 23-63 in Improvement of Protein Nutritive Value. Washington, D.C.: National Academy Press.
Yates, F.E., 1983. Contribution of statistics to ethics of science. Am. J. Physiol 13:R3-R5.
Yeboah, N., E. Al-Sing, A. Bodalloo, T. Forrester, A. Jackson, and D.J. Millward. 1996. Transfer of 15N from urea to the circulating lysine pool in the human infant. Proc. Nutr. Soc. 55:37A.
Yoshida, A. 1986. Nutritional aspects of amino acid oxidation. Pp. 378-382 in Proceedings of the XIII International Congress of Nutrition, T.G. Taylor and N.K. Jenkins, eds. London: John Libbey and Co., Ltd.
Young, V.R. 1987. McCollum Award Lecture: Kinetics of human amino acid metabolism: Nutritional implications and some lessons. Am. J. Clin. Nutr. 46:709-725.
Young, V.R. 1991a. Nutrient interactions with reference to amino acid and protein metabolism in nonruminants: Particular emphasis on protein-energy relations in man. Z. Ernährungwiss 30:239-267.
Young, V.R. 1992. Protein and amino acid requirements in humans: Metabolic basis and current recommendations. Scand. J. Nutr./Naringsforskning 36:47-56.
Young, V.R. 1994. Adult amino acid requirements: The case for a major revision in current recommendations. J. Nutr. 124:1517S-1523S.
Young, V.R. 1997. Paper 5. Dietary protein standards can be halved (Chittenden, 1904). J. Nutr. 127:1025S-1027S.
Young, V.R., and A.E. El-Khoury. 1995a. Can amino acid requirements for maintenance in adult humans be approximated from the amino acid composition of body mixed proteins? Proc. Natl. Acad. Sci. USA 92:300-304.
Young, V.R., and A.E. El-Khoury. 1996. Human amino acid requirements: A re-evaluation. Food Nutr. Bull. 17:191-203.
Young, V.R., and J.S. Marchini. 1990. Mechanisms and nutritional significance of metabolic responses to altered intakes of protein and amino acids, with reference to nutritional adaptation in humans. Am. J. Clin. Nutr. 51:270-289.
Young, V.R., and P.L. Pellett. 1985. Wheat proteins in relation to protein requirements and availability of amino acids. Am. J. Clin. Nutr. 41:1077-1090.
Young, V.R., and P.L. Pellett. 1990. Current concepts concerning indispensable amino acid needs in adults and their implications for international nutrition planning. Food. Nutr. Bull. 12:289-300.
Young, V.R., and N.S. Scrimshaw. 1968. Endogenous nitrogen metabolism and plasma amino acids in young adults given a "protein-free" diet. Br. J. Nutr. 22:9-20.
Young, V.R., L. Fajardo, E. Murray, W.M. Rand, and N.S. Scrimshaw. 1975. Protein requirements of man: Comparative nitrogen balance response within the submaintenance-to-maintenance range of intakes of wheat and beef proteins. J. Nutr. 105:534-542.
Young, V.R., D.M. Bier, and P.L. Pellett. 1989. A theoretical basis for increasing current amino acid requirements in adult man, with experimental support. Am. J. Clin. Nutr. 50:80-92.
Young, V.R., N.S. Scrimshaw, and P.L. Pellett. 1998. Significance of dietary protein source in human nutrition: Animal or plant proteins. Pp. 205-212 in Feeding a World Population of More than Eight Billion People: A Challenge to Science, J.C. Waterlow, D.G. Armstrong, L. Fowden and R. Riley, eds. Oxford: Oxford University Press.
Zello, G.A., P.B. Pencharz, and R.O. Ball. 1990. Phenylalanine flux, oxidation and conversion to tyrosine in humans studied with [l-13C]phenylalanine. Am. J. Physiol. 259:E835-E843.
Zello, G.A., P.B. Pencharz, and R.O. Ball. 1993. Dietary lysine requirement of young adult males determined by oxidation of L-[l-13C]phenylalanine. Am. J. Physiol. 264:E677-E685.
Zello, G.A., L.J. Wykes, R.O. Ball, and B. Pencharz. 1995. Recent advances in methods of assessing dietary amine acid requirements for adult humans. J. Nutr. 125:2907-2915.


























