1
Introduction
Advances in medical science and technology have made solid organ transplantation an increasingly successful and common medical procedure, a literal ''second chance at life." Greater experience in performing transplantation and the development of better immunosuppressive regimens have increased the survival rates for transplant recipients. Since the enactment of the National Organ Transplant Act of 1984, the number of people receiving organs has increased annually. In 1998, nearly 21,000 Americans—about 57 people a day—were transplanted with a kidney, liver, heart, lung, or other organ (see Table 1-1).
More people are benefiting from organ transplants and their survival rates are steadily improving. Comparing data for transplantations performed in 1988 with data for transplantations performed in 1995, one-year patient survival rates increased from 81 to 87 percent for persons receiving a liver; from 83 to 85 percent for persons receiving a heart; and from 50 to 77 percent for persons receiving a lung (DHHS, 1998b). In addition, technological advances, such as the Belzer UW solution, have made it possible to preserve organs outside the body for longer periods of time.
A primary determinant of organ viability is "cold ischemic time," the time from when blood flow to the organ is stopped in the donor to the time that blood flow to the organ is restored in the recipient. The shorter the cold ischemic time, the more likely a transplant is to be successful. Ischemic injury results from prolonged lack of blood flow, and, at some point, affects organ function following transplantation. The amount of ischemia that compromises organ function differs by organ type and other factors, for example, donor age.
In the past 15 years, the national transplantation system has extended the lives of more than 200,000 individuals (Meier, 1999). Approximately 62,000 people wait for an organ on any given day and every 16 minutes a new name is added to the national waiting list (UNOS, 1999) (see Table 1-2). Each year the
number of patients added to the waiting list grows (see Table 1-3). Also, although the number of donors has increased steadily since 1988, it is not growing as quickly as the demand for organs (GAO, 1997). Roughly 4,000 Americans die each year (11 people per day) waiting for organs (UNOS, 1999). Organs are obtained for transplantation from less than 1 percent of U.S. deaths.
In an effort to increase donation rates, the federal government announced the National Organ and Tissue Donation Initiative in December 1997. As a part of the initiative, the Department of Health and Human Services (DHHS) issued a regulation requiring all Medicare-participating hospitals to refer all deaths and imminent deaths to organ procurement organizations (OPOs). The regulation went into effect in August 1998. In April 1999, DHHS announced a 5.6 percent increase in donation in 1998 (DHHS, 1999a).
TABLE 1-1 Number of Transplants Performed in 1998
OPOs are subject to certification and recertification by the federal Health Care Financing Administration (HCFA), which issues performance standards. These standards are designed to promote the efficiency of OPOs. HCFA also approves waivers that permit a hospital to have an arrangement with a different OPO than the one assigned to its area.
EVOLUTION OF THE FEDERAL SYSTEM OF ORGAN TRANSPLANTATION
The current arrangement of 62 organ procurement organizations nationally evolved gradually, reflecting improvements in transplantation science, organ preservation, and other factors. Historically, the hospital in which the donor resided was responsible for locating a recipient. Thus the earliest days of solid
organ transplantation, the donor and recipient were often in the same building. Gradually, a system of independent organizations, Organ Procurement Organizations (OPOs), developed to optimize matching of patients with donated organs. OPOs identified donors, retrieved organs, and found recipients within a reasonable time frame.
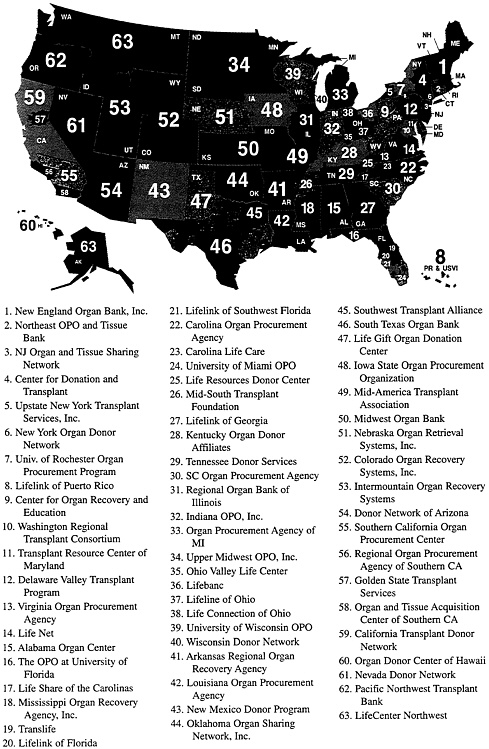
The designated geographic areas served by the various OPOs range in size from a few counties, to entire states, to multi-state areas covering parts or all of several states (see Figure 1-1).* The populations of these areas range from approximately 700,000 to 11,000,000 (DHHS, 1999b). In each area, only one OPO coordinates activities relating to organ procurement and allocation, and that OPO is required to have a working arrangement with all hospitals in its designated area. OPOs evaluate potential donors, discuss donation with family members, and arrange for the surgical removal of donated organs. OPOs also are responsible for preserving organs and arranging for their distribution according to nationally, regionally, or locally agreed upon organ-sharing policies.
TABLE 1-2 UNOS National Patient Waiting List for Organ Transplants
|
Type of Transplant |
Registrations for Transplanta |
Patients Waiting for Transplantb |
|
Kidney |
43,734 |
41,833 |
|
Liver |
13,181 |
12,987 |
|
Heart |
4,267 |
4,248 |
|
Lung |
3,299 |
3,250 |
|
Kidney-pancreas |
1,915 |
1,847 |
|
Pancreas |
453 |
442 |
|
Heart-lung |
248 |
244 |
|
Intestine |
120 |
120 |
|
Pancreas islet cell |
118 |
118 |
|
Total |
67,335 |
63,219 |
|
a UNOS policies allow patients to be listed with more than one transplant center (multiple listing); thus, the number of registrations is greater than the actual number of patients. b Some patients are waiting for more than one organ; therefore, the total number of patients is less than the sum of patients waiting for each organ. SOURCE: Based on UNOS Scientific Registry data as of May 12, 1999. |
||
|
* |
Figure 1-1 shows the 63 OPOS as they were in 1997. There are presently 62 OPOs, but the precise boundaries had not been determined at the time of publication of this report. |
TABLE 1-3 Number of Registrations on the National Transplant Waiting List by Organ at Year End, 1988-1998
|
|
Year |
|||||||||||
|
Organ |
1988 |
1989 |
1990 |
1991 |
1992 |
1993 |
1994 |
1995 |
1996 |
1997 |
1998 |
10-Year Percentage Increase |
|
Kidney |
13,943 |
16,294 |
17,883 |
19,352 |
22,376 |
24,973 |
27,498 |
31,045 |
34,550 |
38,236 |
42,364 |
204 |
|
Liver |
616 |
827 |
1,237 |
1,676 |
2,323 |
2,997 |
4,059 |
5,691 |
7,467 |
9,637 |
12,056 |
1,857 |
|
Pancreas |
163 |
320 |
473 |
600 |
126 |
183 |
222 |
285 |
323 |
361 |
455 |
179 |
|
Kidney-pancreas |
0 |
0 |
0 |
0 |
778 |
923 |
1,067 |
1,234 |
1,463 |
1,591 |
1,841 |
N/A |
|
Heart |
1,030 |
1,320 |
1,788 |
2,267 |
2,690 |
2,834 |
2,933 |
3,468 |
3,698 |
3,897 |
4,185 |
306 |
|
Heart-lung |
205 |
240 |
225 |
154 |
180 |
202 |
205 |
208 |
237 |
236 |
257 |
25 |
|
Intestine |
0 |
0 |
0 |
0 |
0 |
42 |
75 |
83 |
83 |
94 |
100 |
100 |
|
Lung |
69 |
94 |
308 |
670 |
942 |
1,240 |
1,625 |
1,923 |
2,309 |
2,664 |
3,165 |
447 |
|
Total |
16,026 |
19,095 |
21,914 |
24,719 |
29,415 |
33,394 |
37,684 |
43,937 |
50,130 |
56,716 |
64,423 |
302 |
|
SOURCE: Based on United Network for Organ Sharing, and Organ Procurement and Transplantation Network waiting lists on the last day of each year (UNOS, 1999). |
||||||||||||
Donated organs are transferred to one of 272 transplant centers in the United States (UNOS, 1998). Within these centers are 891 organ-specific transplantation programs (Hauboldt, 1996). The number of transplantation centers in the UNOS registry has increased by nearly 85 percent since 1988, an average increase of almost 10 percent per year (Hauboldt, 1996). For livers, the number of transplant center programs grew from 56 in 1988 to 125 in 1998.
In addition, the current system divides the country into 11 geographic regions, the second level of organization in the organ allocation system. The regions generally follow state boundaries: one region includes only a single state; the others consist of several states. The populations of these regions range from 9.6 million to more than 40 million (DHHS, 1999a). Very recently, UNOS' policies were modified to establish the 11 regions as a second tier for liver allocation.
The origin of the current system is the National Organ Transplant Act of 1984, which created a national transplant system to be operated by transplant professionals, with oversight by DHHS to ensure an equitable allocation system. The act created the Organ Procurement and Transplantation Network (OPTN), a nonprofit private-sector network to be operated by a contractor to DHHS. The United Network for Organ Sharing, or UNOS, has held this contract since 1984.
The statute did not impose a rigid system. Instead, each OPO was allowed to develop its own relationships with the local medical community, while maintaining considerable uniformity with other OPOs in the way they operate (see Chapter 2). Thus, although the system originated in federal statute and corresponding intent, it has over time accommodated local preferences and institutional variation in practice. Under the current system, local patients (i.e., within the OPO, or in some instances within agreed to areas of sharing) are given priority for organs that are procured in that local area. If a matching patient is not found, the search is broadened to the region.
Originally, OPTN membership and policies were voluntary, but with enactment of the Omnibus Budget Reconciliation Act of 1986, which added Section 1138 of the Social Security Act, all hospitals that perform transplants were required to abide by the rules and requirements of the OPTN to be eligible to participate in the Medicare and Medicaid programs.
This act has been amended twice to encourage the development of a fair, national system of organ allocation. The original statute (P.L. 98-507, Title II, Sec. 201, formerly codified at 42 U.S.C. 274(b)(2)(C)) required the OPTN to "assist organ procurement organizations in the distribution of organs, which cannot be placed within the service areas of the organizations." Congress changed the language in 1988 "so as to remove any statutory bias respecting the important question of criteria for the proper distribution of organs among patients" (P.L. 100-607, Title IV, Sec. 403, formerly codified at 42 U.S.C. 274(b)(2)(D)). In 1990, the language was again rewritten, this time to require that the OPTN "assist organ procurement organizations in the nationwide distribution of organs equitably among transplant patients" (P.L. 101-616, Title II, Sec. 202, now codified at 42 U.S.C. 274(b)(2)(D)). The language was also revised to redefine the definition of an OPO service area to be "of sufficient size
to ensure the maximum effectiveness of organ procurement and equitable organ allocation."
In December 1989, DHHS issued a Federal Register notice indicating that all OPTN rules and requirements would remain voluntary until the secretary promulgated regulations to define the roles and policy making procedures of the OPTN and DHHS (HCFA, 1989). A Notice of Proposed Rule Making containing these definitions was published on September 8, 1994. The issue of appropriate federal oversight of the system—including procedures for joining the OPTN, the federal review processes, procedures and standards for information collection and dissemination, OPTN membership requirements and compliance procedures, and the criteria for allocation of each of the solid organs—has been a subject of debate since that time. The DHHS proposal, 42 CFR Part 121, "Organ Procurement and Transplantation Network; Final Rule" (DHHS, 1998b) was intended to bring closure to the Notice of Proposed Rule Making, but instead reactivated the debate.
THE LATEST CALL FOR CHANGE: THE "FINAL RULE"
Because human organs are a scarce medical resource, organ transplantation policies and practices have become the subject of controversy about supply, demand, access, rationing, and equity. A major concern expressed by DHHS is that the current system appears to have resulted in large disparities in the total amount of time a patient waits for an organ (i.e., the time from registration to transplant), depending on where he or she lives. For example, a patient with type O blood in need of a liver transplant in New York City may wait 511 days for a new organ, while the same category patient might wait only 56 days in Newark, New Jersey, just a short distance away (DHHS, 1998b).
These disparities have been attributed to a variety of factors. These include variability in the listing criteria; varying supply of and demand for organs among service areas; differences in criteria used in accepting a donated organ (e.g., age, medical factors); and limitations in the categorization schema used (i.e., the criteria used to determine the status of patients and their survivability pre- and posttransplant). Concerns have also been raised that minorities and the poor have less access to some organ transplants, specifically kidneys, than do whites of higher socioeconomic status (Alexander and Sehgal, 1998).
The Final Rule does not establish specific policies to guide transplantation. Rather, it requires that sharing areas be broadened, if necessary, to give priority to those whose needs are medically urgent, that listing criteria be standardized, and that policy and procedural reforms be enacted. The primary objectives are to address disparities in waiting times and in the allocation of organs to low-income and minority patients. A brief description of the major provisions follows.
Major Provisions of the Final Rule
The Final Rule provides a framework within which the OPTN, its members, and other participants in organ procurement and transplantation will operate. The stated principles underlying the final regulation include the need for oversight in a system that permits variance in individual medical practice. A major impetus is the creation of a "level playing field" in organ allocation, where organs are allocated based on patients' medical need and less emphasis is placed on keeping organs in the local area where they are procured. A primary focus is to equalize waiting times among patients similarly situated medically, regardless of residence or location of transplant program. In addition, the Final Rule calls for standardized medical criteria to be used to determine the status of a person's illness and when the person can be placed on a waiting list. Finally, the Final Rule aims to improve data collection and analysis so that patients, their physicians, and the public have timely, accurate, and user-friendly, center-specific data on the performance of transplantation programs to measure quality and make transplant decisions. In announcing the Final Rule, Secretary Shalala said,
Patients who need an organ transplant should not have to gamble that an organ will become available in their local area, nor should they have to travel to transplant centers far from home simply to improve their chances of getting an organ. Instead, patients everywhere in the country should have an equal chance to receive an organ, based on their medical condition and the judgment of their physicians. (DHHS, 1998a)
DHHS charged that despite technological advances in preserving organs, the system for allocating scarce organs (especially livers) remains weighted to local organ allocation, instead of broader regional or national allocation according to medical need. Thus, a patient who is less ill in one geographic area with a short waiting list may get a matching organ before a patient whose condition is more medically urgent in another area with a longer waiting time. In the Final Rule, DHHS claims that by allocating organs primarily at the local level, current policies give some of the most medically urgent patients a substantially lower chance of being promptly matched to a suitable organ (and thereby receiving a potentially lifesaving transplant) than would be the case with broader geographic sharing.
In addition, DHHS maintains that medical criteria for listing patients and assessing their status vary from one transplantation center to another, making it difficult to objectively compare the medical need of patients awaiting organ transplantation in different centers and different areas of the country. Furthermore, although many data are available, DHHS asserts that there is still a need for more current and usable data collection and dissemination to help patients and doctors in measuring quality and making transplantation decisions.
In general, the Final Rule was developed to establish a framework within which both the OPTN and the DHHS will operate. It delineates the roles of each, providing a basis for the OPTN to act and the department to monitor and review
these actions. Major provisions address policy development, allocation of organs, and procedural requirements. Appendix D of this report presents the complete text of the Final Rule; Appendix C presents the current liver allocation policies. The remainder of this section summarizes the following components of the Final Rule: procedural requirements, policy development, and performance goals.
Procedural Requirements
Procedural requirements contained in the Final Rule include:
Transition to New Policies-When the OPTN initially revises organ allocation policies, it must propose transition policies ensuring that people who are already on the national waiting list for transplantation do not receive less favorable treatment than they would have under previous policies.
Board Composition-The Final Rule modifies the composition of the OPTN Board of Directors. At least six public members must come from fields such as behavioral science, computer science, economics, ethics, heath care financing, law, policy analysis, sociology, statistics, or theology. Another eight members, at least 25 percent of the board, must represent transplant candidates, transplant recipients, organ donors, and family members. No more than 50 percent of the members are to be transplant surgeons or transplantation physicians.
Public Access to Data-The Final Rule pays special attention to public access to data. When the secretary determines that information will serve the public's interest, the secretary may release it. The Final Rule requires that the OPTN make performance data available to the public, and that such data be updated every 6 months and be available no more than 6 months later than the period to which they apply. The data are to include the characteristics of individual transplant programs as well as rates of nonacceptance of organs and waiting times, and other data useful to patients, their families, and physicians in making transplantation decisions.
Review and Evaluation-The secretary or her or his designee may review and evaluate member OPOs and transplant hospitals where there is evidence of noncompliance with the OPTN rule or actions that risk patients' health or compromise public safety. Sanctions may include removal of transplantation program designation, termination of the transplant hospital's participation in Medicare or Medicaid, or termination of an OPO's Medicare and Medicaid reimbursement.
Policy Development
The OPTN Board of Directors is responsible for developing organ allocation policies. The National Organ Transplant Act requires that members of the "general public" serve on the OPTN Board of Directors. The Final Rule, as described previously under procedural requirements, expands on this provision by
requiring that "eight individuals representing transplant recipients, organ donors, and family members" be included on the board, as well as six members of the general public (42CFR 121.3(a)(1)(ii), (iv)). Proposed policies may be reviewed by the secretary and, if determined appropriate, published in the Federal Register for public comment.
In addition to policies for the equitable allocation of organs, the OPTN's policymaking role includes policies on the training and experience of transplant surgeons and physicians; policies for nominating OPTN board members; and other policies as directed by the secretary. Of particular note, given concerns regarding access to transplantation services for low-income populations and racial and ethnic minority groups, is the Final Rule's requirement that the OPTN board develop policies to reduce inequities resulting from socioeconomic status, including access to transplantation waiting lists (42 CFR 121.4(a)(3)).
Performance Goals
The OPTN Board of Directors is responsible for developing organ-specific policies for equitable organ allocation among potential recipients (including policies for combination transplants, e.g., heart-lung). The Final Rule sets three broad performance goals for organ allocation:
-
standardized listing criteria for placing patients on waiting lists, using to the extent possible objective and measurable medical criteria;
-
standardized criteria for determining medical status, also based, to the extent possible, on objective and measurable medical criteria, sufficient to differentiate patients from least to most medically urgent; and
-
organ allocation policies that give priority to those whose needs are most medically urgent, in accordance with sound medical judgment, with the result that differences in waiting times for patients of like medical status will be reduced.
Finally, the Final Rule requires the OPTN board to focus first on appropriate revisions to its current liver allocation policy and to propose a new liver allocation policy to the secretary within 60 days of the regulation's effective date. Other organ-specific policies must be provided to the secretary within 1 year of the regulation's effective date.
THE ENSUING CONTROVERSY
The issuance of the Final Rule raised a storm of controversy, with patient groups, transplant surgeons, OPOs, and UNOS bringing to the debate their reasons for either supporting or objecting to the proposed changes. Those who support the Final Rule argue that the current system is inequitable, that is, with allo-
cation units that are too small for equitable organ sharing. Proponents argue that a system of broader sharing will alleviate the circumstances in which a very sick patient might die waiting for a liver transplant while a healthier patient just a few miles away (but outside the OPO territory) might receive a transplant (DHHS, 1998b). In addition, advocates for the Final Rule claim that a national system would hinder the ability of patients or physicians to take advantage of defects in the current rules in order to gain personal advantage, a practice often alleged but not substantiated with data.
Those opposed to the Final Rule claim that organ referral and retrieval will be hampered and organ donation rates will drop, if organs are not offered locally first. They argue that OPOs, donor hospitals (the hospital where the donation takes place), and transplant centers have developed working relationships that achieve good results and that disruption of these relationships will result in fewer organs being available for transplantation and, therefore, more deaths. They claim that current variations in waiting time are related to differing listing criteria (a problem being addressed in the current system), differing geographic demand, and differing levels of organ availability, not true inequities in the allocation system (DHHS, 1998b). In addition, they argue, a broader sharing system will result in the closure of some small transplantation centers.
Opponents of the Final Rule assert that ''a sickest first policy would create a system that is wasteful and dangerous, resulting in fewer patients transplanted, increased death rates, increased retransplantation due to poor organ function, and increased overall cost of transplantation" (Benjamin, 1999). Some opponents of the Final Rule object in particular to the proposed ability of the DHHS Secretary to make OPTN allocation policies and direct the OPTN to implement them, claiming this will replace medical judgment with political judgment.
Opposition to broader organ sharing is exemplified by the efforts in several states in the last 18 months-Oklahoma, Louisiana, South Carolina, Tennessee, Texas, Arizona, Nevada, New Mexico, Ohio, and Wisconsin-to keep donated organs within their state borders. Some states enacted laws permitting an organ to be sent out of the state only on the condition that the state has entered into a reciprocal organ-sharing agreement with the receiving state. Others adopted resolutions urging Congress to oppose the Final Rule by pushing OPOs to use their best efforts to use organs within a state and by having potential donors stipulate that their organs be first offered to in-state recipients.
CHARGE TO THE COMMITTEE
In October 1998, Congress suspended implementation of the Final Rule for 1 year to allow further study of its potential impact (Omnibus Consolidated and Emergency Supplemental Appropriations Act, 1999, 11 USC, §213, 1999). The Institute of Medicine (IOM) was asked by Congress, through the General Accounting Office, to conduct a study to review the current policies of the OPTN and the potential impact of the Final Rule on:
-
access to transplantation services for low-income populations and racial and ethnic minority groups, including the impact of state policies (under Title XIX of the Social Security Act) regarding payment for services for patients outside of the states in which the patients reside;
-
organ donation rates, reasons for differences in organ donation rates, and impact of broader sharing (i.e., based on medical criteria instead of geography) on donation rates;
-
waiting times for organ transplants, including: (a) determinations specific to the various geographic regions of the United States and, if practicable, waiting times for each transplant center by organ and medical status category, and (b) impact of recent changes made by the OPTN in patient listing criteria and in measures of medical status;
-
patient survival rates and organ failure rates leading to retransplantation, including variances by income status, ethnicity, gender, race, or blood type; and
-
costs of organ transplantation services.
The legislation that called for this study included two additional areas for review: (1) confidentiality of information about the program, and (2) the possible legal liability of OPTN members arising from their peer review activities. As agreed, the GAO addressed these two issues in a separate report (GAO, 1999). Also as agreed in response to the legislation, GAO assisted IOM by providing data to the committee regarding costs of organ transplantation services.
COMMITTEE METHODS AND FOCUS OF THE REPORT
The committee held three meetings between March and May 1999. At two meetings (March and April) time was devoted to invited public participants and presentations. Additional data were requested, as needed, from UNOS. To complete its work, the IOM committee conducted an independent assessment of the issues to prepare an evidence-based report. Appendix A describes in greater detail the data sources and methods used by the committee.
Focusing the Analysis on Livers
At the committee's first meeting, consideration of several factors suggested that the analysis would be most useful, and more practically conducted, if the committee were to focus on studying the policies, practices, and data concerning liver procurement and allocation. Although many concepts and principles that have to be addressed are not organ-specific (e.g., consistency in criteria for listing, donor motivation), there are some elements specific to livers that justify this focused effort. Among these are:
-
Disparities in median waiting times for liver transplants was a primary factor in DHHS's rationale for developing the Final Rule;
-
Liver allocation policies have been especially contentious, with the OPTN making several changes in policies in the recent past;
-
Because the maximum desirable cold ischemic time for hearts and lungs is 3-4 hours, there is less opportunity to make significant changes in the current allocation rules for them than there is for livers, which have a longer ischemic time; and
-
The medical urgency of transplanting livers differs from that for some of the other solid organs, e.g., kidneys (those waiting for livers are often terminally ill with no alternative therapy, while those waiting for kidneys have the potential backup of using dialysis).
The focus on policies and practices related to liver transplantation was also necessitated by the extremely limited amount of time that the committee had for conducting its assessment and preparing its report.
For these reasons, this report focuses on policies and practices related to liver transplantation, unless otherwise noted in the text. As noted above, however, some of the general principles (e.g., consistency in listing criteria) apply in general to all organs.
ORGANIZATION OF THE REPORT
Following this introductory chapter, Chapter 2 describes the current policies and practices in organ procurement and transplantation. The remainder of this report is organized around the five tasks that were received from Congress as follows: Chapter 3 discusses the issue of access to transplantation. Chapter 4 focuses on donation and the possible effects of the Final Rule on donation rates. Chapter 5 provides an analysis of data related to waiting times. Chapter 6 addresses patient survival rates and organ failure. Chapter 7 appraises the effect of the Final Rule on costs of transplantation. Chapter 8 addresses crosscutting issues related to government oversight and review. Several appendixes are included to assist the reader with additional information: Appendix A—Data Sources and Methods; Appendix B—Summary data tables from Chapter 5 regarding waiting time for liver transplantation; Appendix C—Current UNOS Liver Allocation Policies; Appendix D—DHHS Final Rule; and Appendix E—Committee and Staff Biographies.