HEALTH RISK ASSESSMENT FOR LEWISITE
DRAFT REPORT
September 1996
(editorial corrections made April 1997)
Prepared for
U.S. Department of the Army
Army Environmental Center
under Interagency Agreement No. 1769-1769-A1
Prepared by
Life Sciences Division
OAK RIDGE NATIONAL LABORATORY*
Oak Ridge, Tennessee 37831
Submitted to
Material Chemical Risk Assessment Working Group
Advisory and Coordinating Committee
Environmental Risk Assessment Program
Strategic Environmental Research Development Program
PREFACE
This report assesses the potential non-cancer and cancer effects of chemical agent lewisite (CAS No. 541-25-3).
This document supports the activities of the Material/Chemical Risk Assessment Working Group of the Environmental Risk Assessment Program, a cooperative endeavor of the Department of Defense, Department of Energy, and Environmental Protection Agency. This working group is developing toxicity values for selected chemicals of concern at federal facilities. Toxicity values will be submitted for consideration by the EPA's IRIS Consensus Process for inclusion on IRIS (EPA's Integrated Risk Information System). The Material/Chemical Risk Assessment Working Group consists of Drs. Jim Cogliano (chair) and Harlal Choudhury (U.S. EPA), Dr. Bruce Briggs (Geo-Centers); Lt. Cmdr. Warren Jederberg and Dr. Robert L. Carpenter (U.S. Naval Medical Research Institute); Dr. Elizabeth Maull and Mr. John Hinz (U.S. Air Force Occupational and Environmental Health Directorate); Drs. Glenn Leach and Winnie Palmer (U.S. Army Center for Health Promotion and Preventive Medicine); Drs. Robert Young and Po-Yung Lu (Oak Ridge National Laboratory).
This document was written by Dr. Robert A. Young, Life Sciences Division, Oak Ridge National Laboratory, Oak Ridge, TN. Internal peer review was provided by Dr. Annetta Watson, and Mr. Robert Ross. External review of the toxicity data was provided by Dr. Thomas J. Bucci, Integrated Services, White Hall, AR and Dr. I. K. Ho of the U. of Mississippi Medical Center, Jackson MS. External review of the derivation of the RfDs was provided by Drs. Michael Dourson and Susan Velazquez of Toxicology Excellence for Risk Assessment, Cincinnati, OH, and Dr. William Hartley of Tulane Medical Center, New Orleans LA. Additional reviews were provided by Mr. Joe King, Dr. Jack Heller, Ms. Veronique Hauschild, Ms. Bonnie Gaborek, Mr. Maurice Weeks, Maj. Robert Gum, and Mr Kenneth Williams of the U.S Army.
1.
INTRODUCTION
In response to the FY Defense Authorization Act [Public Law (PL) 102–484, Sect. 176], review and evaluation of data were conducted to derive RfDs for non-stockpile chemical materiel (NSCM) in an effort to develop control limits for NSCM in soil and water. Lewisite (CAS No. 541-25-3) was identified as a priority chemical at non-stockpile materiel sites.
1.1 PHYSICAL/CHEMICAL PROPERTIES
Lewisite [dichloro(2-chlorovinyl)arsine] is an organic arsenical known for its vesicant properties (Rosenblatt et al., 1975). It has a molecular weight of 207.32, vapor pressure of 0.58 mm HG at 25°C, a liquid density of 1,89 g/cm3 at 25°C, freezing point of -18°C, boiling point of 190°C, and is negligibly soluble in water (DA, 1974). The chemical structure of lewisite is shown below. Lewisite may occur as a trans-isomer and as a cis-isomer. In aqueous solutions, the cis-isomer undergoes photoconversion to the trans-isomer (Clark, 1989). In the presence of moisture, lewisite is rapidly converted to the more stable but highly toxic lewisite oxide (2-chlorovinylarsenous acid) (Cameron et al., 1946).
Lewisite
[dichloro(2-chlorovinyl)arsine]
1.2 ENVIRONMENTAL FATE
Regardless of the method of lewisite degradation (combustion, hydrolysis, or other environmental degradation), the arsenic component will not be eliminated and, therefore, at least some combustion product or other degradation products may be some form of arsenical. The recognized degradation products of lewisite are listed in Table 1.
1.2.1 Air
Although no data on its fate in the atmosphere are available, UV absorption spectrum of lewisite at 200 to 350 nm indicates that some photodegradation may take place. Rapid hydrolysis may occur in the gas phase (MacNaughton and Brewer, 1994).
1.2.2 Water
Although lewisite is only slightly soluble in water, 0.5 g/L (Rosenblatt et al., 1975), hydrolysis, resulting in the formation of lewisite oxide and HCl is rapid. Cis-Lewisite must be heated to over 40°C to react with NaOH to yield vinyl chloride, sodium arsenite, and acetylene (Rosenblatt et al., 1975). In aqueous solution, the cis isomer undergoes a photoconversion to the trans isomer (Rosenblatt et al., 1975). Upon standing in water, the toxic trivalent arsenic of lewisite oxide is converted to the less toxic pentavalent arsenic (Epstein, 1956).
Table 1. Lewisite Degradation Products
|
Product |
Formula |
CAS No. |
|
Hydrolysis Product: |
||
|
Chlorovinyl arsenous oxide |
C2H2CIAsO |
3088-37-7 |
|
Combustion Products: |
||
|
Acetylene |
C2H2 |
74-86-2 |
|
Acetylene monochloride |
C2HCl |
593-63-5 |
|
Arsenic trichloride |
AsCl3 |
7784-34-1 |
|
Arsenic trioxide |
As2O3 |
1327-53-3 |
|
Chlorine |
Cl2 |
7782-50-5 |
|
Methyl chloride |
CH3Cl |
74-87-3 |
|
Vinyl chloride |
C2H3Cl |
75-01-4 |
|
Acetylene dichloride |
C2H2Cl2 |
540-59-0 |
|
Arsenic oxychloride |
AsOCl |
Not found |
|
Chlorovinyl arsenous oxide |
C2H2ClAsO |
3088-37-7 |
|
Sources: DA, 1974, 1988; Small, 1984; HEAST, 1993 |
||
1.2.3 Soil
Lewisite applied to soil may rapidly volatilize and/or be converted to lewisite oxide through exposure to soil moisture (Rosenblatt et al., 1975). However, its low water solubility indicates intermediate persistence in moist soil (Watson and Griffin, 1992). Both lewisite and lewisite oxide may be slowly oxidized to 2-chlorovinylarsonic acid (Rosenblatt et al., 1975). Suggested pathways of microbial degradation in soil include epoxidation of the C=C bond and reductive dehalogenation and dehydrohalogenation (Morrill et al., 1985). In addition, residual hydrolysis would result in arsenical compounds. Although lewisite will probably not bioaccumulate through food chains, arsenic (an elemental poison) may (Rosenblatt et al., 1975).
2.
MECHANISM OF ACTION
The toxicological effects of lewisite are ultimately due to its interaction with thiol groups of biologically active proteins such as enzymes. The interaction with sulfhydryl groups of enzymes may result in inhibition of enzyme function by the formation of stable cyclic structures with arsenic (As+3) as a result of the reaction of the arsenic with the sulfhydryl groups of organic compounds such as those occurring in dihydrolipoic acid and in reduced keratin (De Bruin 1976). Dihydrolipoic acid is a dithiol cofactor active in several important enzyme systems (required for cellular respiration) including alphaketoacid oxidases such as pyruvate oxidase, 2-oxoglutarate oxidase, and aldehyde dehydrogenase. Lewisite combines with the dihydrolipoic acid to form stable six-member ring structures (cyclic thioarsenite
complexes), thereby inactivating the enzymes. Overall, the end result of these interactions and the ultimate mechanism of lewisite toxicity appear to be energy depletion which, in turn, results in cell death. Organochloroarsines, of which lewisite is an example, are also potent alkylating agents; this feature suggests carcinogenic potential.
3.TOXICOLOGY
3.1 Introduction
Lewisite is a lethal vesicant and systemic poison. The toxicology of lewisite has recently been reviewed by Goldman and Dacre (1989), Watson and Griffin (1992), and Trammell (1992) and will, therefore, only be briefly discussed in this section. Lewisite may be lethal following inhalation or dermal exposure, or by ingestion. Its lethality is due primarily to vapor inhalation, although lewisite is much less potent than neurotoxic chemical warfare agents. Generally, the toxic effects of lewisite are of rapid onset and result from acute exposures. The vesicant properties of lewisite result from direct skin contact; it has been estimated that as little as 2 ml to an adult human (equivalent to 37.6 mg/kg) can be fatal within several hours (Sollman, 1957). Being lipophilic, percutaneous absorption of lewisite is rapid and may be associated with systemic toxicity characterized by pulmonary edema, diarrhea, agitation, weakness, hypothermia, and hypotension (IOM, 1993). The threshold for severe systemic effects in humans following dermal exposure to lewisite is approximately 10 mg/kg (9.1 – 13.4 mg/kg) (Sollman, 1957). It has been hypothesized that fatalities following dermal exposure to lewisite may be due to blood plasma loss resulting from extensive capillary damage (i.e., lewisite shock) (Cameron et al., 1946). Ingestion of trivalent arsenicals may also cause death due to fluid loss resulting from intestinal epithelium damage. The vesicant properties of lewisite are characterized by immediate onset of pain and, for ocular exposure, possible corneal necrosis. Studies in animals have shown that the target tissues and organs for systemic toxicity of lewisite include the liver, gall bladder, urinary bladder, lung, and kidneys (Cameron et al., 1946; Snider et al., 1990). It is important to note that the gaps in knowledge regarding the toxic effects and dose response for lewisite are extensive.
3.2 Short-term Toxicity
Liquid lewisite applied by eye-dropper to the forearms of men caused blanching and discoloration of the skin followed by extensive erythema within 15 to 30 minutes and vesication within 12 hours or less (Wardell, 1941, as cited in Goldman and Dacre, 1989). The pain associated with these dermal exposures reportedly occurred within two minutes and considerable discomfort persisted for about one week. Other tests with human subjects and clinical reports also indicate a similar temporal sequence of events. Exposure to lewisite vapor (0.06 to 0.33 mg/L) caused discoloration and blistering with the maximum effect occurring by 36 to 48 hours after exposure (Wardell, 1941). At a concentration of 0.01 mg/L, lewisite vapor caused inflammation of the eyes and swelling of the eyelids after 15 minutes of exposure, and inhalation of 0.5 mg/L for five minutes is considered to be potentially lethal.
Short-term exposure (10 to 30 minutes) of dogs to lewisite vapor (0.05 to 0.12 mg/L) produced vomiting, urination, defecation, and severe respiratory distress that resulted in the death of 80% of the dogs within 3 to 48 hours (Goldman and Dacre, 1989). It was not reported whether the exposures were whole body or head only.
Acute oral toxicity values for lewisite have been summarized by Watson and Griffin (1992). The only available oral LD50 is that for the rat (50 mg/kg). Lethality values for other routes of exposure indicate some species variability but the values differ by less than an order of magnitude for any particular exposure route.
3.3 Subchronic Toxicity
A drinking water exposure study in rats was reported by Leitch et al. (1941). In this study, 10 rats were administered lewisite in drinking water (10 or 16 mg/L) for 19 weeks (133 days). The treatment did not affect consumption of food or water and had no effect on animal growth. Additionally, there were no treatment-related histopathological findings. Based on this report, a lewisite concentration of 16 mg/L drinking water would represent a NOAEL. However, this study has some deficiencies, as noted by Daniels (1990). The study neither defined an effect level, nor monitored the actual concentration of lewisite in drinking water consumed. Additionally, the report did not provide information regarding water consumption by the test animals. These data would be critical in determining an actual or estimated dose of lewisite. It is also possible that the consumed concentration may have varied from the target concentration because of test article degradation. Daniels (1990), however, suggested that these data would probably provide an estimate of a 7-day NOEL equivalent to 1.4 mg lewisite/kg.
In a dose range-finding study for a teratology study in rats and rabbits, lewisite was administered by gavage to rats (10 per group) on gestation days 6–15 at doses of 0, 0.5, 1.0, 2.0, or 2.5 mg/kg, and by gastric intubation to rabbits (8 per group) on gestation days 6–19 at doses of 0, 0.5, 1.0, 1.5, or 2.0 mg/kg. For rats, deaths attributed to lewisite occurred in the 2.5 mg/kg group (2/10) and in the 2.0 mg/kg group (1/10). Dosing trauma deaths were also reported (1/10, 2/10 and 1/10 in the 1.0, 2.0, and 2.5 mg/kg groups). For rabbits, deaths attributed to lewisite were reported in the 1.0 mg/kg group (6/8), 1.5 mg/kg group (5/8) and 2.0 mg/kg group (8/8). Dosing trauma deaths were also noted; 1/8 and 3/8 in the 1.0 mg/kg group and 1.5 mg/kg group, respectively.
A 90-day subchronic toxicity study of lewisite in rats was conducted by Sasser et al. (1989a). In this study, groups of 10 male and 10 female rats were given lewisite in sesame oil by gastric intubation at doses of 0.01, 0.1, 0.5, 1.0, or 2.0 mg/kg. Dosing protocol was 5 days per week for 13 weeks or approximately 65 dosing days. Vehicle controls received sesame oil at a dose of 1.67 ml/kg. Deaths were observed in the three highest dose groups; three males and seven females of the 2.0 mg/kg dose groups, eight males and six females of the 1.0 mg/kg dose group, and two males and three females of the 0.5 mg/kg dose group. Although all of the deaths occurred in the three highest dose groups, the response was not dose-dependent. Forestomach lesions were observed in the two highest dose groups (8/10 males and 4/10 females in the 2.0 mg/kg group, and 1/10 males in the 1.0 mg/kg group) and were attributed to the test article. These lesions were characterized by necrosis of the stratified squamous epithelium accompanied by infiltration of numerous neutrophils and macrophages, hemorrhage, and edema. In some instances, hyperplasia of adjacent areas was noted. There was no evidence that the lesions were precancerous, but the duration of exposure and observation was insufficient to assess carcinogenic responses. Lesions were also present in the glandular stomach but to a lesser degree. The presence of the lesions was consistent with the irritant effect of lewisite. No lesions were observed in the lower dose groups.
Some of the animals died without exhibiting any clinical signs of toxicity; drooling or wetness around the mouth and chin, and labored respiration were noted among other rats immediately preceding death.
Gross pathology findings attributed all deaths, except one, to severe inflammatory lesions characterized by edema and epithelial necrosis of the respiratory tract. Respiratory lesions were most likely due to aspiration of the test material or induced reflux of stomach contents into the pharynx with subsequent aspiration into the airways. Inflammatory lesions observed in the respiratory tract of surviving rats were also indicative of accidental deposition or induced reflux of the test material. No significant treatment-related effects on body weights or organ weights were observed for any of the dose groups.
Clinical chemistry evaluations revealed a significant (p<0.05) decrease in total serum protein, serum creatinine, and serum SGOT and SGPT in male rats of the highest dose (2.0 mg/kg) group at 13 weeks. Lowered serum enzyme activity was also observed in male rats of the other lewisite dose groups. Females of the highest dose group exhibited significantly increased lymphocyte and platelet counts; the former at 6 weeks but not at 13 weeks and the latter only at 13 weeks. The biological/toxicological significance of these findings is, however, uncertain. The investigators noted that the no-effect dose was greater than 0.5 mg/kg and less than 1.0 mg/kg. The 0.5 mg/kg dose may be considered an estimate of the NOAEL for short-term oral exposure to lewisite.
3.4 Chronic Toxicity
No human or animal studies examining the effects of lewisite following chronic exposure were located in the searched literature.
3.5 Developmental and Reproductive Effects
In a teratogenicity study by Hackett et al. (1987), lewisite was administered by gavage to pregnant rats on gestation days 6 through 15 at doses of 0.5, 1.0, and 1.5 mg/kg and by gastric intubation to pregnant rabbits on gestation days 6 through 19 at doses of 0.07, 0.2, and 0.6 mg/kg. For rabbits, the mortality rates were 13%, 46%, and 69% for the 0.07, 0.2, and 0.6 mg/kg dose groups, respectively. The mortality rates were corrected for death from other causes (e.g., dose-delivery trauma, accidental delivery of the dose to the lungs, handling trauma, pregnancy complications unrelated to the test article) and, therefore, represent a significant dose-related frank effect. Surviving rabbits in the highest dose group exhibited decreased body weight gain relative to controls and other dose groups. However, the study authors noted more frequent incidences of anorexia in the high-dose rabbits when compared to controls and other dose groups. For those rabbits whose deaths were not attributed to the extraneous causes previously noted, gastric lesions (mucosal inflammation, edema, necrosis, and mucosal sloughing) were observed at all dose levels. The only statistically significant developmental effects were a significant increase in the incidences of fetal stunting and supernumerary ribs in the high-dose (0.6 mg/kg) group. Fetal weight and crown-rump length were somewhat lower in the 0.6 mg/kg dose group but these differences were not statistically significant. Maternal toxicity (13%) was also associated with the low-dose group, thereby indicating a NOAEL for this study to be <0.07 mg/kg/day for maternal toxicity and 0.2 mg/kg/day for developmental toxicity. The LOAEL based on maternal effects is 0.07 mg/kg/day and for developmental effects is approximately 0.6 mg/kg/day. The increased mortality of the does (13%) and the occurrence of gastric lesions in the low-dose group (0.07 mg/kg/day) suggest that the rabbit is the most sensitive of the species for which data are available.
The use of increased mortality as the critical effect for derivation of a reference dose is not appropriate. Furthermore, the intragastric intubation technique used for rabbits in this study concentrates the test article on the gastric mucosa more effectively than simple gavage administration thereby making the apparent increased sensitivity of rabbits more an artifact of administration than actual toxicodynamics. In the discussion of the study, Hackett et al. noted that the fetal toxicity observed in the rabbits appeared to be
occurring at doses above those required to induce increased maternal mortality. The findings of this study are, however, statistically compromised by the low number of pregnant survivors (9/12, 6/11, 5/13, and 3/15 for the control, 0.07, 0.2 and 0.6 mg/kg dose groups, respectively). In a dose range-finding study for this experiment (see Section 3.3.), significant mortality was observed in the 1.0 mg/kg group (6/8), 1.5 mg/kg group (5/8) and 2.0 mg/kg group (8/8).
Another phase of the Hackett et al. (1987) study investigated the potential teratogenicity of lewisite in rats. In this phase of the study, no maternal toxicity or teratogenic effects were observed, thereby identifying 1.5 mg/kg as a NOAEL. However, it must be noted that in a dose range-finding study in rats (Hackett et al., 1987; see Section 3.3), doses of 2.0 mg/kg and 2.5 mg/kg resulted in 10% and 20% maternal mortality, respectively.
A two-generation reproductive study in rats was conducted by Sasser et al. (1989b). In this study, lewisite (in sesame oil) was administered intragastrically at doses of 0.10, 0.25, or 0.60 mg/kg/day to groups of 25 male and 25 female rats five days per week for 13 weeks prior to mating and 7 days per week during gestation (21 days), and at least four days per week during lactation (21 days). The doses were selected based upon the findings of the subchronic toxicity study by Sasser et al. (1989a) which identified a NOAEL between 0.5 and 1.0 mg/kg/day, and the teratogenicity study by Hackett et al. (1987) in which 1.5 mg/kg/day was a NOAEL. In the dose range-finding phase of this report, 20% mortality (corrected for deaths due to dosing trauma) was observed in the 2.5 mg/kg/day group. At the time of birth of the F1 generation, the F0 male rats were sacrificed. Dams continued treatment (minimum of four doses per week) throughout lactation (3 weeks). A vehicle control group was given equivalent volumes of sesame oil (1.67 ml/kg). After weaning, 20 male and 25 female offspring were selected for the F1 phase of the study. The treatment protocol for these animals was as described for the F0 generation. Mortality was high among both the F0 and F1 females. The cause of death for most of these animals appeared to be associated with aspiration of the test article resulting in fatal respiratory tract lesions. Exposure of rats to lewisite did not adversely affect reproductive performance, fertility, or reproductive organ weights. The treatment had no significant effect on litter weights, sex ratio, mean pup weight, or offspring survival for either generation. Although this study revealed no toxic effects, arsenic is known to be embryotoxic and teratogenic, and the possibility exists that inorganic arsenic could be metabolically derived from lewisite.
An unpublished USSR study analyzed by the U.S. Army Research Institute of Chemical Defense (Solana, 1992) provided data indicating that preconception maternal exposure of rats to 0.045 or 0.002 mg lewisite/cm3,4 hours/day, 5 days/week for 4 months did not affect numbers of corpora lutea or implantations, number and physical dimensions of fetuses, increased intrauterine mortality or ossification of long bones. Approximately 140 litters of rats were used in this study.
Human data regarding reproductive/developmental effects due to lewisite exposure are inconclusive because of confounding factors such as concurrent exposure to other agents such as sulfur mustards and incomplete exposure data. Yamakido et al. (1985) studied workers from the Okuno-jima (Japan) factory where mustard and lewisite were manufactured in the World War II era, and noted no evidence of agent-induced mutations.
3.6 Carcinogenic and Mutagenic Effects
In a long-term follow-up study, Krause and Grussendorf (1978) reported the formation of a malignant lesion at the site of contact eight years following a single, acute dermal exposure to lewisite. A German soldier had been accidentally exposed to liquid lewisite on his lower right leg in 1940. In 1948, the lesion was diagnosed as malignant. Thirty-eight years after exposure, the area around the contact site was still ulcerated and diagnosed as Bowen's disease (intradermal squamous cell carcinoma). Bowen's disease was also diagnosed in workers at a Japanese facility that produced lewisite. These latter findings, however, were not conclusive because these workers were exposed concurrently to diphenylcyanoarsine and mustard agent and no quantitative estimates of dose or exposure rates were available (Inada et al., 1978).
There is only anecdotal evidence for the potential carcinogenicity of lewisite. These data are not definitive and do not support classifying lewisite as a suspected carcinogen. As such, quantitative assessment of the potential carcinogenicity of lewisite is not currently possible. Although the available evidence is not of sufficient quality to label lewisite a suspected carcinogen, the position maintained by CDC (CDC, 1988) that ''some evidence suggests that lewisite might also be a carcinogen" seems tenable. However, for environmental exposure and remediation concerns, the arsenic component and/or arsenic-containing degradation products would warrant concern.
Although the carcinogenicity of lewisite per se is equivocal and cannot be assessed quantitatively, several of its degradation products are known carcinogens. Lewisite combustion produces the inorganic arsenicals arsenic trichloride and arsenic trioxide, as well as vinyl chloride. Inorganic arsenic is carcinogenic in humans and animals and is classified as a Group A carcinogen for both oral and inhalation exposure (U.S. EPA, 1989). Arsenic trioxide and vinyl chloride are both considered Group A carcinogens by the U.S. EPA (U.S. EPA, 1984, 1988) and Group 1 carcinogens by IARC (IARC, 1987). Additionally, compounds such as arsenic trichloride, sodium arsenite (a lewisite hydrolysis product), arsenic oxychloride, and inorganic arsenicals in general are of concern to EPA as potential carcinogens (U.S. EPA, 1988). However, there are no human epidemiologic data or data from animal studies that show organic arsenicals to be carcinogenic. A review by the World Health Organization (WHO, 1981) stated that "There is no conclusive evidence that any of the organoarsenic compounds tested for carcinogenicity in laboratory animals are carcinogenic." IARC (1987) concluded that adequate data were not available for evaluating the carcinogenicity of organic arsenic compounds.
Data from genotoxicity studies do not indicate a carcinogenic potential for lewisite. Genotoxicity studies in Salmonella typhimurium strains TA97, TA98, TA100, and TA102 were negative with and without S9 activation at lewisite concentrations <1.0 µg/plate (Stewart et al., 1989). At 1.0 µg/plate and higher, lewisite was cytotoxic. Jostes et al. (1989) reported on the effects of lewisite in one mutation assay (hypoxanthine-guanine phyosphoribosyl transferase [HGPRT] locus) and two cytogenetic assays (chromosomal aberration and sister chromatid exchange [SCE]) using Chinese ovary cells. At concentrations ranging from 0.12 to 2.0 µM, the mutagenic response at the HGPRT locus was not significantly different from control values. The SCE assay resulted in a weakly positive response from 0.25 to 1.0 µM concentrations, but the values were not significantly different from control values. However, chromosome aberrations were induced at 0.50, 0.75, and 1.0 µM that were significantly greater than control values. The investigators concluded that lewisite was cytotoxic and clastogenic but SCE and mutation at the HGPRT locus was insignificant. Assays to determine sex-linked lethal mutations and chromosomal rearrangements in Drosophila melanogaster yielded negative results (Auerbach and Robson, 1946, 1947).
A dominant lethal study using CD rats was conducted by Bucci et al. (1993). In this study, male CD rats (20/group) were given lewisite in sesame oil by gavage for five days at doses of 0.375, 0.75, or 1.5 mg/kg. Vehicle controls received an equivalent volume of the vehicle and positive controls were given the vehicle followed by 100 mg ethyl methanesulphonate/kg, i.p. on day five. Each male was mated with two females over the next 10 weeks. With the exception of the positive controls, no significant differences were observed in reproductive indices and there were no histopathologic findings that could be attributed to lewisite treatment. Under the conditions of this study there were no dominant lethal mutations resulting from exposure to lewisite.
4.
ORAL REFERENCE DOSE FOR LEWISITE
The U.S. EPA has not adopted any Reference Doses for lewisite; consequently, an RfD for lewisite will be derived using available data. No controlled studies are available that have evaluated the oral toxicity of lewisite in humans; therefore, extrapolation from animal data is necessary.
The effects levels for the available studies are summarized in Table 2. A NOAEL of between 0.5 and 1.0 mg/kg/day was obtained from the rat 90-day oral subchronic toxicity study of Sasser et al. (1989a). The oral teratogenicity study by Hackett et al. (1987) provided data indicating a NOAEL of 1.5 mg/kg for teratogenic effects in rats with maternal toxicity occurring at 2.0 mg/kg/day. The report by Hackett et al. (1987) also reported that gestational exposure of rabbits at a dose 0.6 mg/kg/day resulted in maternal toxicity and fetal stunting. Doses as low as 0.07 mg/kg/day also resulted in 13% maternal toxicity (excluding deaths from extraneous causes) and were accompanied by marked gastric lesions. These data indicate a LOAEL of 0.07 mg/kg based upon gastric lesions and increased mortality. However, the results of this study are statistically compromised by the low numbers of surviving animals. A NOAEL of 0.6 mg/kg was obtained from the multigeneration-reproduction study in rats reported by Sasser et al. (1989b). Although the data from the 90-day subchronic toxicity study by Sasser et al. (1989a) only identified the NOAEL as being between 0.5 mg/kg and 1.0 mg/kg, the 0.5 mg/kg dose would provide a conservative estimate of the NOAEL.
Based upon the limited available data, the rabbit appears to represent the most sensitive species as indicated by the occurrence of gastric lesions concurrent with increased mortality following 14-day administration of lewisite by gastric intubation. The rabbit data are, however, statistically compromised by the small number of survivors in each treatment group. Studies assessing reproductive/developmental endpoints in rats were negative, and the data from rabbits indicated that developmental effects occurred at doses exceeding those that induce significant maternal mortality.
Table 2. Summary of Effect Levels for Lewisite Toxicity Studies
|
Study Typea |
Species |
NOAEL |
LOAEL (Critical Effect) |
Reference |
|||
|
Subchronic |
rat |
1.4 mg/kg (est.) |
None |
Leitch et al., 1941 |
|||
|
90-day |
rat |
0.5 mg/kg/day |
1.0 mg/kg (gastric lesions) |
Sasser et al., 1989a |
|||
|
Multigeneration |
rat |
0.6 mg/kg/day (0.44 mg/kg/day TWA) |
None |
Sasser et al., 1989b |
|||
|
Developmentalb |
rat |
1.5 mg/kg/day |
None |
Hackett et al., 1987 |
|||
|
Developmentalb |
rabbit |
<0.07 mg/kg/day |
0.07 mg/kg/day (gastric lesions, increased mortality) |
Hackett et al., 1987 |
|||
|
Range-findingb |
rat |
1.0 mg/kg/day |
2.0 mg/kg/day (increased mortality) |
Hackett et al., 1987 |
|||
|
Range-findingb |
rabbit |
0.5 mg/kg/day |
1.0 mg/kg/day (increased mortality) |
Hackett et al., 1987 |
|||
|
a Route of administration is gavage/gastric intubation, except Leitch et al. (1941) which was drinking water. b Test article administered on gestation days 6–15 (rats) and 6–19 (rabbits). |
|||||||
For the derivation of RfD for lewisite, both a 90-day study (Sasser et al., 1989a) and a multigeneration study (Sasser et al., 1989b) in rats were used to identify effect levels. Data from the 90-day study identified a LOAEL of 1.0 mg/kg/day based upon gastric lesions. The accompanying NOAEL from this study was 0.5 mg/kg/day. The multigeneration study represents a chronic exposure situation relative to reproductive/developmental effects but would be considered subchronic duration for systemic effects in the adult animals. It must be noted that the absence of reproductive/developmental effects does not necessarily eliminate the possibility of more sensitive effects in alternate targets. The highest dose (0.6 mg/kg/day) from the multigeneration study of Sasser et al. (1989b) appears to represent the most valid NOAEL and would be the best value for deriving an RfD. However, because of the discontinuous exposure and variable dosing protocol, a time-weighted average dose must be calculated. This adjustment will provide a NOAEL adjusted for discontinuous exposure (NOAELadj) and is based on the following: rats were dosed at 0.6 mg/kg/day × 5 days/7 days for 13 weeks (91 days) = 0.43 mg/kg/day for 13 weeks; females dosed daily (0.6 mg/kg/day) during gestation (21 days) = 0.6 mg/kg/day for 3 weeks, and females dosed at least 4 days per week at 0.6 mg/kg/day during lactation (21 days) = 0.34 mg/kg/day for 3 weeks. The time-weighted average (TWA) dose for this 133-day period is calculated as
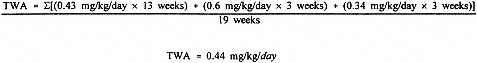
This NOAEL is slightly lower than the NOAEL of 0.5 mg/kg/day from the Sasser et al. (1989a) study and, therefore, is being used as the basis for the RfD for lewisite. The selection of this NOAEL is supported by the available data set with the exception of the rabbit data, the validity of which uncertain. The RfD for lewisite is calculated as follows:

where
The derivation of an RfD for lewisite necessitated addressing several issues regarding the available data set: 1) interpretation of toxicity data from gavage/gastric intubation studies and 2) identification of the critical effect. The available data for lewisite toxicity is limited to gavage and gastric intubation administration studies. Although these routes of administration allow for more precise control of the administered dose as opposed to drinking water or feeding studies, in the case of lewisite (or any highly corrosive agent), they impart substantial caveats in data interpretation. Firstly, the use of a sesame oil vehicle and the gavage/gastric intubation administration result in the gastric mucosae being exposed to a bolus of material in a vehicle that limits normal dispersion of the test article in the stomach. The decrease
in dispersion causes an increase in contact time between the corrosive agent and a limited surface mucosal area, thereby increasing the potential for inflammatory responses observed in the described studies. Furthermore, the presence of an oil vehicle will likely affect the physicochemical interactions at the chemical/tissue interface by altering the solubility and distribution of the chemical. Secondly, within the context of the RfD, intake of a corrosive agent as a bolus/oil suspension would not be toxicologically or physiologically analogous to exposure to the chemical agent via environmental media such as water. The critical effect of orally administered lewisite in animals appears to involve gastric lesions (rats and rabbits), possible developmental effects (rabbits), respiratory tract inflammation responses (rats and rabbits), and increased mortality (rats and rabbits). The increased mortality and respiratory tract inflammation responses reported in the available studies appear to be associated more so with dosing errors or simple reflux of the corrosive, irritating lewisite. The reflux and subsequent respiratory tract response would be highly unlikely in an environmental exposure situation (i.e., drinking water contamination). Furthermore, the studies did not provide data affirming the respiratory responses to be a function of systemically mediated lewisite toxicity. Therefore, an RfD based upon available data is tenuous and difficult to verify.
The proposed RfD for lewisite underwent preliminary review (July 10–12, 1996) by the Material/Chemical Risk Assessment (MCRA) Working Group of the Environmental Risk Assessment Program (ERAP). The MCRA Working Group of ERAP represents multiagency (EPA, DoD, and DOE) input by individuals experienced in deriving and validating toxicity values. The MCRA Working Group agreed that the critical toxic effect observed in the lewisite studies (forestomach lesions) appears to be an artifact of administration, and that the overall database for lewisite is not robust. Although it was recognized that the structure of lewisite might imply toxic activity differing from inorganic arsenic, it was the consensus of the MCRA Working Group that the lewisite RfD be considered not verifiable due to data deficiencies, and that the existing RfD for inorganic arsenic (3E-04 mg/kg/day) be used as a surrogate. This is considered a valid and justifiable approach inasmuch as the inorganic arsenic RfD and the proposed lewisite RfD are similar (3E-04 vs 1E-04 mg/kg/day, respectively), and the fact that lewisite in environmental media will be degraded to inorganic arsenic.
5.CARCINOGENICITY ASSESSMENT FOR LEWISITE
Data are inadequate to quantitatively assess the potential carcinogenicity of lewisite. There are inadequate human and inadequate animal data regarding the carcinogenic potential of lewisite. Genotoxicity data are equivocal or negative.
It was recommended by the MCRA Working Group that for risk assessments, the carcinogenic potential of lewisite degradation products be considered.
6.REFERENCES CITED
Auerbach, C. and J. M. Robson, letter to the Editor. 1946. Chemical production of mutations. Nature 157:302.
Auerbach, C. and J. M. Robson. 1947. Tests of chemical substances for mutagenic action. Proc. R. Soc. Edinburgh 62B:284–291.
Bucci, T., R. Parker, J.C. Dacre and K.H. Denny. 1993. Dominant lethal study of lewisite in male rats. NCTR Technical Report, Experiment No. 6579.
Cameron, G.R., H.M. Carleton and R.H.D. Short. 1946. Pathological changes induced by lewisite and allied compounds. J. Pathol. Bacteriol. 58:411–422.
CDC. 1988. Notice for Final Recommendations for Protecting Human Health and Safety Against Potential Adverse Effects of Long-term Exposure at low doses of agents GA, GB, VX, Mustard Agent (H, HD, T), and Lewisite (L). Fed. Register 53(50): 8504–8507.
Clark, D.N. 1989. Review of Reactions of Chemical Agents in Water. AD-A213 287, Defense Technical Information Center.
DA (U.S. Department of the Army). 1974. Chemical Agent Data Sheets, vol. 1. Edgewood Arsenal Special Report, EO-SR 74001. Defense Tech, Inform. Center, Alexandria, VA.
DA (U.S. Department of the Army). 1988. Final Programmatic Environmental Impact Statement for the Chemical Stockpile Disposal Program, Aberdeen Proving Ground, MD, Office of the Program Executive Officer, Program Manager for Chemical Demilitarization.
Daniels, J.L. 1990. Lewisite. In: Daniels, J.L., Ed. Evaluation of Military Field-Water Quality. Chapter 6, Volume 4, Part 2. Interim Standards for Selected Threat Agents and Risks from Exceeding These Standards. Lawrence Livermore National Laboratory, UCRL-21008.
De Bruin, A. 1976. "Biochemical toxicology of environmental agents." Chapter 24. Sulfhydryl groups and glutathione metabolism. Elsevier. New York, pp. 910–935.
Epstein, J. 1956. Nerve gas in public water. Public Health Rep. 71: 955–962.
Goldman, M. and J.C. Dacre. 1989. Lewisite: its chemistry, toxicology, and biological effects. Reviews Environ. Contam. Toxicol. 110:75–115.
Hackett, P.L., R.L. Rommereim, F.G. Burton, R.L. Buschbom and L.B. Sasser. 1987. Teratology Studies on Lewisite and Sulfur Mustard Agents: Effects of Sulfur Mustard in Rats and Rabbits. Final Report. AD A187495. Pacific Northwest Laboratory, Richland, WA. for the U.S. Army Medical Research and Development Command, Fort Detrick, MD.
HEAST (Health Effects Assessment Summary Table). 1993. "Inorganic arsenic." Office of Research and Development, Office of Emergency and Remedial Response, U.S. EPA. OHEA ECAO-CIN-821.
IARC (International Agency for Research on Cancer). 1987. Overall Evaluations of Carcinogenicity: An Updating of IARC Monographs. Vols. 1 to 42. IARC Monographs of the Evaluation of Carcinogenic Risks to Humans. Suppl. 7. IARC: Lyon, France. pp. 100–106; 373–376.
Inada, S., K. Hiragun, K. Seo and T. Yamura. 1978. Multiple Bowen's disease observed in former workers of poison gas factory in Japan, with special reference to mustard gas exposure. J. Dermatol. Tokyo 5:49–60.
IOM (Institute of Medicine), Committee to Survey the Health Effects of Mustard Gas and Lewisite, Division of Health Promotion and Disease Prevention . 1993. Veterans at Risk; The Health Effects of Mustard Gas and Lewisite, C.M. Pechura and D.P. Rall, eds. National Academy Press, Washington, DC.
Jostes, R.F., Jr., L.B. Sasser and R.J. Rausch. 1989. Toxicology studies on lewisite and sulfur mustard agents: Genetic toxicity of lewisite (L) in Chinese hamster ovary cells. Final Report, Pacific Northwest Laboratory Report, PNL-6922, Richland, WA.
Krause, H. and E.I. Grussendorf. 1978. Syntopy of Bowen's disease and lewisite scar. Hautarzt 29: 490–493.
Leitch, J.L., T.H. Ginsberg and M.E. Price. 1941. MD(EA) Memorandum report 18. Purification of water contaminated with lewisite. A toxicological study of water containing 10 ppm and 16 ppm of lewisite. Medical Research Division, Edgewood Arsenal, MD. (cited in Daniels, 1990)
MacNaughton, M.G. and J.H. Brewer. 1994. Environmental Chemistry and Fate of Chemical Warfare Agents. SwRI Project 01-5864, Southwest Research Institute, San Antonio, TX.
Morrill, L.G., L.W. Reed and K.S.K. Chinn. 1985. Toxic Chemicals in the Soil Environment. Volume 2. Interaction of Some Toxic Chemicals/Chemical Warfare Agents and Soils. Oklahoma State University TECOM Project 2-CO-210-049, Stillwater, OK. Available from DTIC, AD-A158 215.
Rosenblatt, D.H., T.A. Miller, J.C. Dacre, I. Muul and D.R. Cogley. 1975. Problem Definition Studies on Potential Environmental Pollutants. II. Physical, Chemical, Toxicological, and Biological Properties of 16 Substances. Tech. Report 7509, AD AO30428. U.S. Army Medical Bioengineering Research and Development Laboratory, Fort Detrick, MD.
Sasser, L.B., J.A. Cushing, D.R. Kalkwarf, P.W. Mellick and R.L. Buschbom. 1989a. Toxicology studies on lewisite and sulfur mustard agents: Subchronic toxicity study of lewisite in rats. Final Report, Pacific Northwest Laboratory Report, PNL-6860, Richland, WA.
Sasser, L.B., J.A. Cushing, D.R. Kalkwarf, P.W. Mellick and R.L. Buschbom. 1989b. Toxicology studies of lewisite and sulfur mustard agents: Two-generation reproduction study of lewisite in rats. Final Report, Pacific Northwest Laboratory Report, PNL-6978, Richland, WA.
Small, M.J. 1984. Compounds Formed from the Chemical Decontamination of HD, GB, and VX and Their Environmental Fate. Technical Report 8304, AD A149515, US Army Medical Bioengineering Research and Development Laboratory, Fort Detrick, Frederick, MD.
Snider, T.H., M.G. Wientjes, R.L. Joiner and G.L. Fisher. 1990. Arsenic distribution in rabbits after lewisite administration and treatment with British anti-lewisite (BAL). Fundam. Appl. Toxicol. 14:262–272.
Solana, R. 1992. "Toxicology of Lewisite." Presentation before the Committee to Survey the Health Effects of Mustard Gas and Lewisite, Institute of Medicine, National Research Council, Washington, D.C. June 11, 1992.
Sollman, T.H. 1957. Lewisite. In: Sollman, T.H., Ed., Manual of Pharmacology and its Applications to Therapeutics and Toxicology, 8th ed. W.B. Saunders Co.: Philadelphia. pp. 192–193. (cited in Watson and Griffin, 1992)
Stewart, D.L., E.J. Sass, L.K. Fritz and L.B. Sasser. 1989. Toxicology studies on lewisite and sulfur mustard agents: Mutagenicity of lewisite in the Salmonella histidine reversion assay. Final Report, Pacific Northwest Laboratory Report, PNL-6872, Richland, WA.
Trammell, G.L. 1992. Toxicodynamics of organoarsenic chemical warfare agents. In: Somani, S.M., Ed. Chemical Warfare Agents. Academic Press, Inc.: New York, pp.255–270.
U.S. EPA. 1984. Health Effects Assessment for Arsenic. Office of Health and Environmental Assessment, Environmental Criteria and Assessment Office. Cincinnati, OH. PB86-13319, EPA/540/1–86/120.
U.S. EPA. 1988. Methodology for Evaluating Potential Carcinogenicity in Support of Reportable Quantity Adjustments Pursuant to CERCLA Section 102. OHEA-C-073, April, 1988.
U.S. EPA. 1989. Drinking water regulations and health advisories. Office of Drinking Water, Washington, D.C.
U.S. EPA (U.S. Environmental Protection Agency). 1991. General Quantitative Risk Assessment Guidelines for Noncancer Health Effects. ECAO-CIN-538. Environmental Criteria and Assessment Office, Office of Health and Environmental Assessment, Cincinnati, OH.
Wardell, E.L. 1941. Lewisite (M-1): 1940 Summary of physiologic and toxicologic data. Report. No. EATR 285. Chemical Warfare Service, Edgewood Arsenal, MD (ADB959553L).
Watson, A.P. and G.D. Griffin. 1992. Toxicity of vesicant agents scheduled for destruction by the chemical stockpile disposal program. Environ. Health Perspect. 98:259–280.
WHO (World Health Organization). 1981. Arsenic. Environmental Health Criteria Volume 18. IPCS International Program on Chemical Safety, WHO, Geneva.
Yamakido, M., Y. Nishimoto, T. Shigenobu, K. Onari, C.Satoh, K. Goriki and M. Fujita. 1985. Study of the genetic effects of sulfur mustard gas on former workers on Okuno-jima poison gas factory and their offspring. Hiroshima J. Med. Sci. 24:311–322.




















