Major International Programs in Ocean Sciences: Ocean Chemistry
Peter G. Brewer
Monterey Bay Aquarium Research Institute
INTRODUCTION
As I searched through the correspondence for this meeting I discovered that the topic I have been asked to speak on appeared to change. It appeared variously as the topic of ocean chemistry within "large and small science programs," "large oceanographic programs," and "major international programs." This does send a message; after all the so-called "large programs'' are identifiable as discrete from "small science" aren't they? And if the programs were large, well then they were probably international, and vice versa. Is all this true? I cannot give an objective answer, for I have been so intimately involved in the continuum of large and small, national and international programs in ocean chemistry of the National Science Foundation (NSF) over the last 35 years (indeed I am an alumnus of the famed NSF "rotator" program) that subjective perception rules. It is my thesis that the large ocean chemistry programs sponsored by NSF have evolved enormously in method and style over time. Two key program officers, Neil Andersen and Rodger Baier, together with a steady stream of visiting rotators, have had enormous impact on the field over this period.
Please then permit me to give a purely subjective personal account of this period and my own recollections of human effort and scientific achievement. Although some programs may therefore escape comment here, I suspect that my experiences are typical. In my view we need a mixture of large and small—large simply because of the immense scale of the processes we seek to observe, and because human beings are fundamentally captured by the grandeur of the ocean enterprise. And because the best large programs are simply the true ensemble effort of many creative individuals, who have their roots in a first-class small laboratory with theory, and experiment, and all the ferment, creativity, and loyalty that such groups and programs stimulate within their circle.
A companion paper on the overall progress of ocean chemistry is provided by John Farrington earlier in this volume.
EARLY DAYS
I first went to sea as a graduate student from Liverpool University, in 1962, on a small boat in the Irish Sea. There I heard rumors of an expedition to the Indian Ocean, which seemed wonderfully exotic and unknown. With a grant from the Royal Society I was drafted to serve on the RRS Discovery on this two-year effort. With some encouragement from John Riley and training in salinity-nutrient-oxygen analyses, I helped perform thousands of measurements on the 1963-1964 International Indian Ocean Expedition. I heard legends of the International Geophysical Year (IGY) while at sea, and it was there that I first made contact of sorts with the National Science Foundation, in the Seychelles, when we met with the RV Anton Bruun. David Menzel grabbed me and, in the midst of a very boisterous party, grilled me about the controls on nutrient ratios in different monsoon seasons. I suspected much later that he was taking an early look at denitrification in the enormous suboxic regions, which we now know proceeds to an extraordinary extent. I was curious at what might happen to this vast collection of hydrographic data; no one told a graduate student, and I began to suspect that some of the senior scientists just didn't. know. Years later I had in my hands a beautiful atlas compiled by Klaus Wyrtki, published, of course, by NSF. Capturing the power of large data sets is something that is done infinitely better today.
Although the expedition was dubbed International, and it clearly was, I was very struck by the fact that each nation was locked in its own ship and that at-sea contact with other nations' scientists, in the form of direct joint experiments, appeared to be small. We appeared to be "national by facility," but international by concept and desire. Indeed it was clear to me that a large and charming international guild existed and that information was traded here with extraordi
nary speed and generosity, possibly more so on the social occasions wrapped around the formal meetings. This is still true today. The International Indian Ocean Expedition was one of the last of the old-style efforts, where the expedition was coherent in place, but consisted of a mixture of many unrelated scientific activities, from net hauls to seismology.
On September 11, 1964, on the trip home, the Discovery stopped in the middle of the Red Sea for one last hydrographic station. John Swallow (UK) and Rocky Miller (U.S.) who was funded by NSF, had independently noted a warm, saline anomaly and acoustic reflecting layers in the deep water of the Red Sea, and had shared the data. John wanted to track it down. I was assigned the task of (pencil) plotting the data called out from the echo sounder, and we found a definite depression at the site. I then drove the steam winch, hung the bottles, lowered the hydro cast as close to the bottom as we could, carried the samples, and logged the data. The results were extraordinary, with a bottom temperature of 44°C and saturation with salt at >300 grams per kilogram seawater! I was proud of my hard work in the hot sun, after 8 long months at sea, and was very surprised to see a large fraction of the sample being taken and stored in a big plastic bottle that I knew nothing of. "What is that for?" I asked, and was told it was for Harmon Craig at Scripps. I knew nothing of Harmon then, but I did have a first glimpse of how far one could push this international thing.
THE PRE-GEOSECS PERIOD
In 1966 I was offered a job at Woods Hole by John Hunt, who liked the work we had done on the Red Sea brines, and by Derek Spencer, who was building an energetic new chemistry initiative. I knew nothing of the way U.S. science was conducted, but on my first venture into work I observed a plaque on the building commemorating its construction with funds provided by NSF. That summer I went to sea on the Atlantis II and found a similar plaque there too. This seemed to everyone to be quite normal, but such generosity made a great impression on me.
The results of the Red Sea hot brines discovery, soon extended by the group on Atlantis II finding a collection of still hotter, more chemically extreme solutions, with vast metal deposits, occupied much of the ocean chemistry community in the late 1960s, and it received strong NSF support. Dave Ross and Egon Degens led the effort and produced a very fine book (Degens and Ross, 1969). The total saturation with halite made these hot brine pools completely sterile, and it was not until much later, in 1979, that exotic animal communities were discovered in association with hot vents, near the Galapagos, on the East Pacific Rise. Interestingly, if one takes the earlier Red Sea data and simply strips off the NaCl component, the residual chemistry is almost identical to the mid-ocean ridge venting fluids, that is, in showing a dramatic loss of magnesium and sulfate, and strong enrichment in iron and manganese, from water-rock interaction. The fundamental science of what drove the fluids, and the mechanism by which seawater could be altered so dramatically, were very much on our minds. For the first time I began to appreciate why my early sample had been shipped to Scripps so quickly, when I saw the stable isotope data from Craig's lab and realized the constraints it provided.
The Red Sea cruises and the Black Sea cruise in 1969 (Degens and Ross, 1974) were excellent examples of medium-scale ocean science. They had finite goals and a well-constrained geographic area, but they were clearly much larger in scope than a single-investigator laboratory could handle. The papers from these efforts were first class and had many international contributions.
I was advised that I needed to branch out, and in 1967 I wrote my first proposal to NSF, requesting funds to adapt a new fluoride electrode into deep-ocean instrumentation. Funds were awarded, and we quickly wrote a paper on (Mg-F)+ ion-pairing that provided the first experimental test of theoretical models. In some small way we were making a contribution to the building of the elegant thermodynamic model of seawater that we now take for granted. Almost all of this fundamental work on solution physical chemistry was supported by NSF, and all models of the ocean uptake of fossil fuel CO2 today depend critically on this important thermodynamic framework.
A cruise was scheduled, which yielded no unusual results, but it was on this trip, on a warm night on the fantail of the RV Chain in harbor in the Azores, that I first heard, from Derek Spencer, of the plans for a global set of geochemical sections. It seemed to be a very attractive idea. Hank Stommel (Stommel and Arons, 1960) had been persuaded by the early evidence (see Broecker et al., 1961, for the earliest discussion) from Wally Broecker and colleagues, for 14C dating of water masses and of the possibility of putting new time constraints on the mean rate of global ocean circulation by use of the radioactive clock. The new International Decade of Ocean Exploration (IDOE) initiative was arising and would be housed within NSF. This presented a fresh and important opportunity to make a case, and the excitement we felt at revealing the picture of the circulation of the greatest fluid on Earth, painted anew in chemical colors, was palpable.
THE GEOSECS PERIOD: 1968-1978
Building the Program
The object of GEOSECS, the Geochemical Ocean Sections Program, was to trace the picture of the abyssal circulation, using the power of radiotracers to accomplish this, with major cruises in the Atlantic, Pacific, and Indian Oceans. But only just beyond this goal lay much uncertainty. There were many potential tracers in addition to natural 14C; the suite of 226Ra and 228Ra; 222Rn for gas exchange rates and bottom boundary layers; 3H and 14C from the nuclear
tests; and the elusive potential of 32Si. Did they all have to be measured? What were the potential gains? What sampling pattern and density were required? How might the results be incorporated into physical models? These questions, first posed within the GEOSECS context (Craig, 1972), have only been answered with any rigor in the last few years. They are fundamentally hard topics, and it is a tribute to the NSF of those days that it ventured into (literally) such uncharted waters.
Moreover each of these tracers had chemical reactivity as well as radioisotopic decay. This had to be constrained too, and for 14C it meant attacking the full ocean CO2 system, while for the radium isotopes the chemical analogue of barium was selected. Each of these efforts had its advocates, and once the science case had been made within the GEOSECS steering committee, it would petition the NSFIDOE for funds.
It was plain at the outset that the GEOSECS program would be fundamentally different in style and scale than anything before. It was also very confusing. At least three major institutions were big players: Scripps, where Harmon Craig had persuaded Arnold Bainbridge to set up the GEOSECS Operations Group that was to craft the advanced instrumentation and staff the technical support activity; Lamont, where Wally Broecker had pioneered many of the radiochemical tracer techniques and gas exchange rate concepts; and Woods Hole, which was to provide the RV Knorr for the first, Atlantic, expedition and where Derek Spencer created the coordinating center. Karl Turekian at Yale and Gote Ostlund at Miami provided wisdom and refereed the sometimes amazing disputes that arose. Incidents involving fire extinguishers, epoxy, and roller derby are best not mentioned here. At each institute there were young scientists eager to be involved, but all had different views on what would be needed and on how to make a personal scientific effort within this large enterprise.
The problems were typical. Big programs need to be staffed with first-class scientists who will remain with the program for years. First-class scientists cannot be cogs in a big wheel, but are very inventive people of rapidly evolving interests who need to create their own identity and establish their own careers. How to balance these conflicting needs often lies at the heart of ambivalent feelings about large programs. Although the decade was dubbed International, it was not clear what this meant for a particular program, but in the case of GEOSECS several individuals in other countries (Yoshio Horibe, Devendra Lal, Wolfgang Roether, Brian Clarke, and Roger Chesselet) made extraordinary personal efforts.
So far as I can tell, the IDOE programs were not successful in making use of SCOR (the Scientific Committee for Oceanic Research) or the IOC (Intergovernmental Oceanographic Commission), the formal ICSU (International Council for Science) and UN-affiliated international bodies, respectively, to carry out their planning or execution, in spite of a strong effort to do so. Lou Brown was brought into NSF to serve as the internationalist, and he remains at NSF today. In practice the dominant new factor was the forcing by NSF of domestic interinstitutional, not international, expeditions and programs. This radical intrusion into the sovereignty over their ships enjoyed by the major institutions was a source of great discomfort to traditionalists, but it opened the door wide to young and ambitious scientists.
The Expeditions
The Atlantic Ocean—The GEOSECS Atlantic Expedition in 1972, the Pacific Ocean Expedition in 1974, and the Indian Ocean Expedition in 1978, all presented unique challenges. The Atlantic cruise was preceded by at least two test stations, or cruises, that showed somewhat alarming results (Craig and Weiss, 1970). Measurements of the CO2 system properties made by different techniques gave discordant results, calling into question the basis for using the 14C tracer. It was shown that the precision of the ? 14C measurement, achieved by Gote Ostlund and Minze Stuiver, would be ±4 per mille, giving an age resolution ? 30 years. The product of total CO2 and 14C was required, and the supposedly easier total CO2 measurement should not degrade the signal. But it did, and the confusion was to last for several years. It is a tribute to the drive of the leaders, and the courage of NSF, that the expedition went forward.
This courage was soon tested. The Knorr left Woods Hole for a nine-month cruise on July 18, 1972—and lost the entire, horribly expensive, conductivity-temperature-depth probe/profiler (CTD)-rosette sampling package on the very first station. Apparently a locking pin had not been set in place. In spite of heroic efforts by Arnold Bainbridge, not all the advanced analytical systems worked, arid it would be months before the CO2 system was fully operational. Not everyone trusted the new systems to work at all; Joe Reid had insisted that a separate conventional Natnsen bottle cast be done as backup at every station, and this " as laboriously carded out. The cruise tracks are shown in Figure 1.
Tensions soon arose. The work was long and hard, the cost of supporting the expedition was high, and in contrast to small-or medium-scale science, there was no natural break to stop and write papers. It began, to some hostile critics, to look like a large, expensive, general data gathering hydrographic exercise, possibly similar to the International Indian Ocean Expedition a decade before. A review was held at NSF, and the case for going ahead with the Pacific cruise was made. "What," Admiral Owens asked, "would be the consequence of not funding the Pacific cruise?" Amid the uproar he had made his point: this was NSF research, managed by the Foundation which did have the last word. And there were expectations for individual scientific accomplishment.
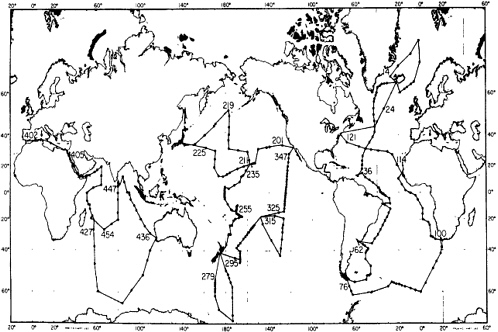
Figure 1 GEOSECS program cruise tracks, 1972-1978. Reprinted from Craig and Turekian (1980) with permission from Elsevier Science.
Hank Stommel noted "a profound sense of beauty" in seeing for the first time the tracer signal of the Noah Atlantic deep water overflows as they began the abyssal tour, and an intense flurry of activity resulted in a strong collection of papers (Craig, 1974). Feenan Jennings at NSF made sure that the program proceeded.
The Pacific Ocean—The GEOSECS Pacific expedition, from August 1973 to June 1974, on the RV Melville, continued the pattern. The Nansen bottle cast requirement was, thankfully, dropped, but the discordant CO2 data problem, latent in the Atlantic cruise results, was now much worse. Nonetheless the classic picture of the chemical response to "aging" of our global circulating fluid was emerging beautifully, and the first glimpse of a global CO2 picture was tantalizingly close.
A Damoclean list appeared above the chief scientist's bunk of the cruise legs on which major equipment was lost; "Bomber" Takahashi led the list since he had had the bad weather legs, for which we were all grateful. The effort to measure the cosmogenic isotope 32 Si, requiring the processing of a thousand liters of seawater through smelly manganese-loaded fibers, was particularly messy. And the early results were showing very little signal. We were to find much later that the half-life had been in error by a factor of four!
NSF realized at some point that this was an enterprise of historic scale and decided to memorialize it on film. A contract was awarded, and a very new cinematographer was flown to Tahiti. The movie is still fun to watch, but it was his personal comment afterwards that shook me. "My God!" he said, "I didn't know the work was that hard!" A plane crash in Samoa sadly resulted in death and injury for the team.
The Indian Ocean—The strains of multiyear devotion to such an all-consuming effort were beginning to show, and by the end of the Pacific cruise, time was needed to regroup, analyze samples, upgrade equipment, and repair relationships with NSF, which, through an evolving stream of program managers, had kept close watch on progress. The urge to focus on showing success and building scientific knowledge, through work on the Atlantic and Pacific results, was getting in the way of creating the Indian Ocean expedition. This illustrates a common problem of large programs—the balance between keeping the technical skills and facility in readiness, and taking definite individual time for research.
In my view it is very much to their credit that NSF program managers have always been wise and pragmatic about this.
The four Indian Ocean legs, from December 1977 to April 1978, were thus a much smaller effort. Taro Takahashi found that the CO2 problem grew yet worse, so that the measured pCO2, and that calculated from the measured alkalinity and total CO2, differed by well over 30 ppm (parts per million). The urge to tackle directly the growing fossil fuel CO2 problem was now very strong, and this stood in the way. The elegant CO2 model by Hans Oeschger and colleagues in Switzerland had appeared two years earlier and had stimulated renewed interest.
New interests were also arising; the "particle reactive tracers" such as thorium and 210Pb were proving more tractable than anyone had thought—not for the original problem of the abyssal circulation rate, but for insights into how the ocean biogeochemical cycle worked. The program was evolving, and important breakthroughs in trace-metal geochemistry, organic geochemistry, and observing the rain of particles to the seafloor were occurring.
GEOSECS Synthesis
The general release of data from the shipboard program was keenly sought, but those close to the measurements were always aware that things could be improved. More problematic still were the results from the shore-based laboratories. These were closely held by the principal investigators (PIs) so as to maximize their advantage in publication; yet for such a conspicuous program there was widespread desire for full disclosure. The data release problem is commonly dealt with by NSF today, but it was the pattern that was created during the GEOSECS era that laid the rules. Pressure, official (the purse string) and peer, was brought to bear on PIs, and the results emerged.
The GEOSECS period resulted in all manner of fundamental insights. I wrote a paper on modifying the equation of state, thus eliminating an ambiguity in connecting the interocean abyssal flow (Brewer and Bradshaw, 1975). Wally Broecker carried on an amazing and sustained attack on the use of the fundamental nutrient relationships to decode water masses, mixing, and chemistry (beginning with Broecker, 1974; see Broecker and Peng, 1982, for a masterful analysis). And eventually the long haul of collecting, stripping, and measuring the radiocarbon signal was completed. Stuiver et al. (1983) published a remarkably simple and elegant paper compiling the 14C results (Figure 2). They reported that "the mean replacement times for the Pacific, Indian, and Atlantic ocean deep waters (more than 1500 meters deep) [are] approximately 510, 250, and 275 years respectively. The deep waters of the entire world ocean are replaced on average every 500 years." These ages were much shorter than first expected: the promise of a radiocarbon solution to the "age" problem was fulfilled; the task of shedding more light on geophysical fluid dynamics by the tracer approach proved to be far more complex. Of course such a paper was principally a necessary and welcome formality. Thanks to the wise NSF data release policy, the GEOSECS results had already been in use around the world, for all kinds of innovative uses, for many years.
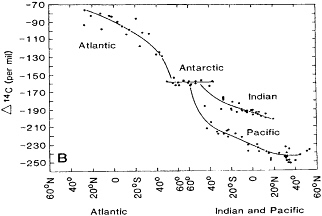
Figure 2 The ?14C values of the cores of North Atlantic, Pacific, and Indian Ocean deep waters. The oldest waters are encountered near 40°N in the Pacific Ocean. Reprinted from Stuiver et al. (1983) with permission from the American Association for the Advancement of Science.
Follow On
In 1978, John Steele called me down to his office at the Woods Hole Oceanographic Institution (WHOI), to meet with John Ryther and Hank Stommel. He wanted to see some fresh starts, and he was concerned about the ending of GEOSECS. He particularly wished to see WHOI tackle the CO2 problem in some way. Hank referred him to a short NAS report, written with Jules Charney, on the anticipated thermal changes; I volunteered to look at the GEOSECS data to find the oceanic chemical signal. Since I had served as co-chief scientist on GEOSECS Atlantic Leg 6, I simply went to those data and wrote a provocative paper on the procedure for detecting the fossil fuel CO2 signal above the very large natural background.
At the same time, Gote Ostlund in Miami was fretting about the lack of a GEOSECS follow-on. The classic dilemma with large programs is the problem of continuity versus innovation; a superb observing system bad been created and refined, and a talented team of people, particularly the Operations Group under Arnold Bainbridge at Scripps, existed. I had heard that Gore was to hold a meeting, with Department of Energy support, to discuss this and I called him to ask if I could attend. I gave the fossil fuel CO2 paper and pointed out that, due to the early GEOSECS technical problems, we had no Atlantic data north of 20°N in the critical deep water formation regions. Others pointed out the new information from the chemical tracers in the region, and we
conceived of a program to measure intensively the invading wave of chemical tracers from the industrial activities of man. The project was soon dubbed "Transient Tracers in the Ocean" (TTO), and of course, we went to NSF for support.
THE TTO PROGRAM
While the TTO program evolved from the GEOSECS experience, it had marked differences, driven both by PI desire for hands-on research and NSF desire for the accountability of individual components to peer review. It was smaller in space and time, the support from the Operations Group was cut in half, and a much more efficient set of observing protocols was adopted. The unseen hand of the scientific marketplace was at work, and our big program follow-on was now to be staffed by an ensemble cast. Within NSF the latent problem of whether this was chemistry (the technique) or physics (a major application) had to be dealt with, and an impasse occurred. Physical Oceanography program manager Curtis Collins at NSF was to rise to the challenge and ably represent the program.
The program was national, but as with GEOSECS, a very strong informal international flavor was simply assumed to exist. I recall driving to Lamont for one meeting with Canadian, German, Japanese, and English participants, which seemed quite normal.
The planning ran into two problems very quickly: facing up to the undiagnosed error in the GEOSECS CO2 results, now nine years old, could no longer be postponed. NSF, quite properly, would not let a new program go ahead without it. And the design of a cruise track that would attempt to cover a very large area of the North Atlantic in one snapshot, proved challenging. In the midst of this, Arnold Bainbridge, the talented, gracious hero of GEOSECS, suddenly died. He was only 48 years old. Years of stress and failure to take care of a chronic health problem had taken a dreadful toll. The shock was enormous.
The death of Arnold Bainbridge left a huge hole and much confusion. When his team went through his office, to put affairs in order and recover original files, they found a drawer full of carefully labeled tapes archiving all the programs that we were using. The problem was that all the labels simply read, "Test"! We were lost.
A meeting was held at Lamont to review the CO2 measurement and data recovery problem, and Bob Williams kindly loaned me a very large binder of FORTRAN printout, which probably contained the answer somewhere. I digested it on a plane flight to Seattle, and by the end of the trip, red eyed, I had found the few lines of code that seemed to count. Arnold had been creative with his chemistry coding and had not told any of us! Al Bradshaw and I painstakingly pulled things apart, and ran some tests (Bradshaw et al., 1981); yes, we could rewrite the equations, and yes, a coding error had occurred during the Atlantic to Pacific transition. We were learning hard lessons—that big programs can be vulnerable. But we could put the problem to rest and advise NSF that publication of the GEOSECS atlases, long stalled by this problem, could proceed. It fell to Taro Takahashi to compile the data (Takahashi et al., 1981), and the classic picture that resulted is shown in Figure 3.
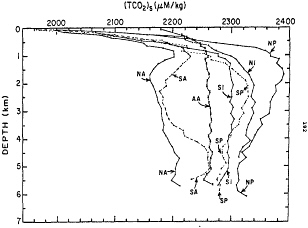
Figure 3 Total CO2 results from the GEOSECS program, showing the progressive enrichment due to respiration and carbonate dissolution accompanying the deep circulation. Reprinted from Takahaski et al. (1981) in SCOPE 16, Carbon Cycle Monitoring, edited by Bert Bolin, with permission from SCOPE, John Wiley & Sons, Ltd., UK.
A test cruise in 1980, and a wonderful year in 1981, saw a large-scale attack on the tracer chemistry of the North Atlantic Ocean. Some 250 stations were occupied (Figure 4), and no equipment was lost. Richard Gammon made the first, exciting measurements of the chlorofluorocarbon tracers. A marked freshening of the North Atlantic was found, which became part of the "Great Salinity Anomaly." The tracer signals showed beautifully the evolution of ocean water masses in the nine years since GEOSECS. Tritium-helium dating of water mass ventilation came of age, thanks to the superb efforts of Bill Jenkins. And chemical coherence within the CO2 system was attained, thanks in large part to the (on-shore) presence of Dave Keeling who provided a limited data set of unassailable integrity. The program provided a superb benchmark for carbon-cycle science.
INSIDE THE FOUNDATION
In the fall of 1981 I took a two-year leave from Woods Hole to serve as program director for Marine Chemistry at NSF. Neil Andersen had been appointed to the Intergovernmental Oceanographic Commission in Paris, and I was his "rotator" replacement. Most of my colleagues were shocked at the move; I had a wonderful time—eventually, and thanks above all to my program colleague Rodger Baier. I learned from Rodger that NSF program managers were not dull; they could play the piano cross-handed while lying down and
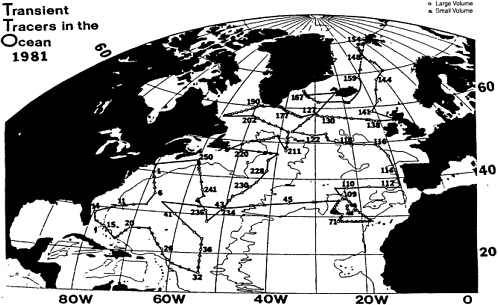
FIGURE 4 Cruise tracks for the Transient Tracers in the Ocean (TTO) North Atlantic Program, 1981.
balance a $10 million budget perfectly on a tiny calculator bought with "global change" in an airport in Tunisia.
My first impression was one of a strange trading market, where almost nothing was done by one program alone, but I saw that I had nonetheless inherited a superbly balanced program. Next I realized how fuzzy our scientist's picture was of how decisions were made. And I learned that nothing counted for more than a clear scientific question to address an unsolved problem; this made a confusing job easy. There were big programs I had never heard of and small programs of all kinds. The folding of the former IDOE big programs into the Division of Ocean Sciences had just occurred, and I began to realize that creating a new big program in this environment would be very difficult indeed. It was the new funds and separateness of IDOE that had allowed the big programs that had shaped our scientific lives to succeed. They also provided the organizational framework to make full use of our ships and yielded dense, well-populated data sets that constrained the ocean in ways not possible by other means.
I attended hearings and learned more about the need to address scientific problems fundamentally important for society. I signed off with pleasure for the publication of the long-awaited GEOSECS atlases. And I served, with Bill Nierenberg and Roger Revelle, on a very special NAS study of "Changing Climate." For 30 years Roger had kept his focus on the CO2-climate problem. He had educated A1 Gore at Harvard, and the political world was beginning to catch up to the issue.
We had at NSF an excellent group of dedicated people: Grant Gross, Bob Wall, Curt Collins, Don Heinrichs, Bruce Malfait, Larry Clark, and rotators Mike Reeve and Rana Fine. All three rotators were to return to academia at about the same time in 1983. We met for lunch to discuss the inevitable exit interview, which we decided to do as a team. It was clear to us that the pattern we had observed, of frequent emergency requests from on high on a Tuesday afternoon for a new long-range plan by Thursday moming ("latest"), barely tolerated by the veterans, was unsustainable. Bob Wall listened, and soon after, a much more vigorous, NSF-initiated planning process took shape in several forms. In some ways it was a natural response to the vacuum created by the demise of a separate IDOE program.
THE JOINT GLOBAL OCEAN FLUX STUDY
The program era that followed was to prove to be fundamentally different. NSF was about to lead, with considerable courage, the new "global change" programs larger in scale and complexity, longer in planning, international in scope,
and far more visible to policy makers. None of us anticipated the work involved.
The Global Ocean Flux Study (GOFS)
I returned to WHOI in late 1983. The World Ocean Circulation Experiment (WOCE) program was then taking shape under Carl Wunsch's leadership. In many ways it seemed a natural ally to the TTO program that had proceeded to the Equatorial, and South, Atlantic. But the latent conflicts between physics and chemistry had not disappeared: how many tracers were really necessary? Could a few sparsely placed samples yield adequate constraints? How solid were the boundary conditions? A rather fierce debate took place. The NAS Ocean Studies Board, with NSF support, provided the forum for this.
John Steele observed the emergence of WOCE, with roots in a desire to use an altimetric satellite combined with a global hydrographic survey, in order to study the global circulation. He was concerned that no program of similar scale existed to constrain the biogeochemical cycles of the ocean and that the promise of an ocean color satellite, hinted at by the Coastal Zone Color Scanner (CZCS) on Nimbus-7, might not be realized unless we took action. The descriptions of the major ocean biogeochemical cycles, which support life in the sea, rested on largely untested ground. He used the Ocean Studies Board to organize a major meeting, held in September 1984, at the Woods Hole Study Center.
The meeting (NRC, 1984) itself was plainly important, but confusing. Mixed together were satellites, primary productivity, higher organisms, sediment traps, radioisotopes, benthic instruments, and the sediment record. Linkages of this kind had been drawn in "horrendograms" by Francis Bretherton as visual drama but executing a coherent study was another matter alltogether. The CO2 story was barely mentioned—would WOCE take care of this? John Steele, Jim Baker, Wally Broecker, Jim McCarthy, and Carl Wunsch kept a careful eye on proceedings. Ken Bruland had been selected to head the Planning Committee. The individual papers were good, but nothing seemed to gel—the topic was so broad, and almost no one had experience dealing with the National Aeronautics and Space Administration (NASA) and satellites. What to do next?
It was Neil Andersen who stepped forward and quietly asked a small group (Ken Bruland, Peter Jumars, Jim McCarthy, and me) to attend a meeting in Washington, ostensibly to edit the report. We met at the NAS building a week or two later. There Jim Baker walked us through the problem; getting a new start in NASA for an ocean color observing satellite would not be easy at all. And the political scene was fundamentally changed by the privatization passion of the current administration, so that commercial possibilities must be factored in. We did edit the GOFS report, and I urged that a far more prominent role for directly observing the controls on the oceanic carbon cycle be included.
A most difficult period then followed. Many participants had naively assumed that simply issuing a report would guarantee funds! The breadth of subject matter left room for a very large number of potential participants. And the review nature of the report, without any early crafting of tactics, left no road map with which to proceed. I was asked to chair the group and instantly felt these problems. A proposal to proceed was submitted through the National Research Council (NRC), and the first "pitch" was made to Burt Edelson at NASA. It went well.
The first step had to be consensus building, and a tense set of small working meetings followed through 1985, touching on each of the subthemes in the GOFS report. I became the sole member of a "Planning Office," helped enormously by the astonishing rise of electronic mail (pioneered by Omnet for the ocean science community). Great credit at this point must go to Neil Andersen, who saw the end point of a powerful program through the clutter of early discussions and carefully guided science along. Again, the Ocean Studies Board meetings served as the debating ground.
In October 1986, plans were more advanced, and I attended a major WOCE meeting, again at NAS. There I addressed the science behind the ocean carbon cycle and received a very enthusiastic audience response (my presentation materials were promptly "borrowed" by a complete stranger!). We had by then conceived within GOFS planning of a three-part attack on the problem: establishment of time-series stations at Bermuda and Hawaii to observe seasonal cycles and secular trends, a set of carefully crafted process studies to illuminate the controlling functions, and a global survey of the CO2 field. It was this latter component that we wished to see accomplished as a collateral program with the WOCE global hydrographic survey, for it would be the critical glue that would scientifically link the two principal ocean observing programs. WOCE was measuring 14C distributions, building on the GEOSECS legacy, and our point was that the full CO 2 system, with its embedded biogeochemical content, naturally followed. We could not afford two global surveys.
But enthusiasm and practicality do clash. There were basic problems of space and funds, let alone the interdisciplinary science. A blunt compromise was quickly reached; WOCE would provide bunk and laboratory space and access to samples. And GOFS would provide trained people, instruments, funds, and data, and would represent the program to appropriate bodies. It was a deal.
It was at once clear that this forced some new steps. WOCE and the global survey were now formally international. GOFS was still national. Within 24 hours a proposal was drafted to the Scientific Committee on Oceanic Research to request its attention to this program and to propose a true international effort. It was immediately hand carried to the SCOR General Meeting in Tasmania and well received.
The first SCOR-sponsored international meeting was held in Paris, at ICSU headquarters, in February 1987. It was
chaired by Jim Baker and carefully observed by Gerold Siedler (Kiel) as president of SCOR. Two key pieces of information came to light at this critical time. We had received the challenge to produce a satellite global chlorophyll image from compositing the CZCS fragments. Gene Feldman rose to the occasion and produced a beautiful image; we saw it for the first time in the luggage area at De Gaul airport in a gray dawn light. It made an enormous impression.
We had earlier received a scientific challenge for the proposed global CO2 survey. In essence it was, "Show us that you can treat ocean CO2 data in the same rigorous manner that we treat the transport of heat." This was very reasonable. By placing ocean CO2 and heat transport in the same observational and theoretical framework, we could link the climate and greenhouse gas signals much more directly. But, as we have seen, the history of such measurement was fraught with difficulty. David Dyrssen (Goteborg) and I had drafted a position paper for the meeting on this very topic and had computed the CO2 and nutrient fluxes across 25°N in the Atlantic Ocean. The problem was far more tractable than we had believed, and new concepts of constraining the mass balance by incorporating some adventurous organic carbon measurements had to be called upon.
It was Jim Baker who suggested that the program henceforth be called JGOFS (not Japanese, but Joint, he quipped). It took. Bernt Zeitschel became the first chair and the first JGOFS International Expedition took place in the North Atlantic in 1988.
This was my first experience at attempting a truly international effort, and I think we all found that it wasn't easy. Neil Andersen's courage, tenacity, and international experience were to serve us well throughout this period. Special mention must also go to Elizabeth Tidmarsh (now Elizabeth Gross) as executive secretary of SCOR for superb efforts in implementing the international form of the program.
Understanding the role of the ocean in the global CO2 equation is not easy, even for many chemists. And here we had a diverse international collection of scientists of several different disciplines, many of whom were now being asked by their government agencies for informed comment on this topic as interest in greenhouse gas policy grew. We soon found that huge differences of opinion occurred. The ocean uptake of fossil fuel CO2 from the atmosphere is not controlled by biological activity, but is an inorganic phenomenon. But the background ocean CO2 level, which the rising trend is imprinted on, is. We are not writing the industrial signal on a blank ocean page. This detail was lost on many, and several highly contentious meetings took place. Years later, we find that a large international population of ocean scientists is now fluent in these issues, and this is a very good thing. The JGOFS program is still in place today, and the results are superb.
The transition from observation and diagnosis of the carbon cycle to active intervention by changing industrial policy, and ocean CO 2 manipulation by disposal and/or fertilization, is about to occur and I have no doubt that NSF will provide the leadership for the scientific underpinnings needed.
IMPRESSIONS TODAY
What can we learn from the big program versus small program theme of this session? Firstly, the style of so-called big programs has changed enormously over the years—from the miscellany of the Indian Ocean expedition, to the large dedicated staff and many-year theme of GEOSECS, to the ensemble cast of TTO, to the remarkable coalescence of individual efforts within JGOFS to attack a very broad problem in a structured way. At each step NSF has shown leadership and creativity in crafting these efforts. And it has enabled the discovery of the fundamental pattern and time scale of ocean circulation, the invading chemical signal of the twentieth century, the chemistry of strange seas, and the fundamental basis for biogeochemical balance as we approach a warmer world. Big programs are not impersonal, but are unusually intense experiences for dozens of small groups. They are a critical part of our ocean science community.
While I have concentrated here on personal experiences, I suspect that others, in parallel programs, have similar tales to tell. The balance of small and large programs comes naturally; theory, instruments, methods, all typically come from small efforts, and the big programs cannot do without this. The best large programs embrace theory, create "small" initiatives, and provide superb opportunities for a very large number of scientists. The future is a bit more worrisome. With the desire to detect global change there is a call for very large-scale operational programs, with data continuity and massive modeling as the goal, rather than a set of evolving questions. Fortunately, there is a new class of medium-scale projects emerging, there are satellites for global observations, and there are exciting new possibilities of sensing and manipulating ocean chemistry in entirely novel ways. NSF is not a mission agency, and I hope that over the next 50 years of ocean discovery the Foundation will keep the healthy large-small ocean program balance in place.
ACKNOWLEDGEMENTS
This paper is dedicated to all my friends at NSF, over many years. It is supported by a grant to MBARI from the David and Lucille Packard Foundation.
REFERENCES
Bradshaw, A.L., P.G. Brewer, D.K. Shafer, and R.T Williams. 1981. Measurements of total carbon dioxide and alkalinity by potentiometric titration in the GEOSECS program. Earth Planet. Sci. Lett. 55: 99-115.
Brewer, P.G., and A. Bradshaw. 1975. The effect of the non-ideal composition of sea water on salinity and density. J. Mar. Res. 33: 157-175.
Broecker, W.S. 1974. "NO," A conservative water-mass tracer. Earth Planet. Sci. Lett. 23:100-107.
Broecker, W.S., and T.-H. Peng. 1982. Tracers in the Sea. Eldigio Press. 690 pp.
Broecker, W.S., R.D. Gerard, M. Ewing, and B.C. Heezen. 1961. Geochemistry and physics of ocean circulation. Pp. 301-322 in M. Sears (ed.), Oceanography. American Association for the Advancement of Science, Publication #67.
Craig, H. 1972. The GEOSECS program: 1970-1971. Earth Planet. Sci. Lett. 16:47-49.
Craig, H. 1974. The GEOSECS program: 1972-1973. Earth Planet. Sci. Lett. 23:63-64.
Craig, H., and K.K. Turekian. 1980. The GEOSECS program: 1976-1979. Earth Planet. Sci. Lett. 49:263-265.
Craig, H., and R.F. Weiss. 1970. The GEOSECS 1969 intercalibration station: Introduction and hydrographic features, and total CO2-O2 relationships, with 9 supporting papers. J. Geophys. Res. 75:7641.
Degens, E.T., and D.A. Ross (eds.). 1969. Hot Brines and Heavy Metal Deposits in the Red Sea. Springer-Verlag, New York. 600 pp.
Degens, E.T., and D.A. Ross (eds.). 1974. The Black Sea—Geology, Chemistry, and Biology. American Association of Petroleum Geologists, Memoir 20. Tulsa, Oklahoma. 633 pp.
National Research Council. 1984. Global Ocean Flux Study. National Academy Press, Washington, D.C. 360 pp.
Stommel, H., and A.B. Arons. 1960. On the abyssal circulation of the world ocean, II. An idealized model of the circulation pattern and amplitude in oceanic basins. Deep-Sea Res. 6:217-233.
Stuiver, M., P.D. Quay, and H.G. Ostlund. 1983. Abyssal water carbon-14 distribution and the age of the world oceans. Science 219:849-851.
Takahashi, T., W.S. Broecker, and A.E. Bainbridge. 1981. The alkalinity and total carbon dioxide concentration in the world oceans. In J. Bolin (ed.), Carbon Cycle Modelling, SCOPE Volume 16, J. Wiley & Sons, New York.











