6
Risk Characterization
INTAKE of low concentrations of copper in the diet can result in deficiency, and high concentrations can result in toxicity. In the general population, there is a range of acceptable intakes that will meet copper requirements and pose no risk of toxicity (Figure 6-1). Generally, copper intake through diet appears to fall within this range for the average, normal, healthy individual. Typically, only a small fraction of an individual's intake of copper derives from drinking water; thus, drinking water should not be relied upon as an important source to meet daily copper requirements. On the other hand, drinking corrosive waters held in copper plumbing can result in copper excess, and the potential for copper toxicity is a concern in that case.
This chapter considers copper concentrations in drinking water that might produce copper excess and provides guidance on the establishment of the maximum contaminant level goal (MCLG). For those individuals with abnormal copper homeostasis, at vulnerable ages, or with already high copper concentrations from occupational exposures, the range of acceptable copper intake through water and diet can be narrow. This chapter also evaluates the prevalence of sensitive populations and the degree to which copper in drinking water might contribute to copper excess in individuals in those populations.
COPPER DEFICIENCY
Severe copper deficiency, characterized by bone abnormalities, severe anemia, compromised immune function, loss of skin, and growth retarda-

FIGURE 6-1 Range of acceptable copper intakes for the general population.
tion, is rarely observed clinically in the United States. Nonetheless, although nationwide surveys suggest that average intake concentrations within the population are at recommended concentrations, a substantial fraction of the population has intakes below currently recommended concentrations (Table 6-1). The significance of those low intakes remains to be determined.
Some populations are a particular concern:
-
Pre-term infants have a lower prenatal accumulation of copper stores and thus can be at increased risk for developing copper deficiency during early infancy.
-
Copper deficiency can be induced by select mineral supplements, particularly zinc.
Individuals taking zinc supplements in excess of the Recommended Daily Allowance (RDA) on a chronic basis might be at particular risk.
-
Large numbers of the elderly appear to have dietary copper intakes below the recommended copper intake.
-
A number of disease conditions, including diabetes and hypertension, are associated with low extrahepatic-tissue copper concentrations.
-
Genetic disorders, such as occipital horn syndrome, might confer an increased risk for copper deficiency.
Several groups appear to be at risk for deficiency, but a substantial fraction of the population have intakes at or above the recommended level (Table 6-1). Therefore, the committee does not recommend redressing copper deficiency via the water supply and notes that the MCLG should not be established on the basis of copper deficiency.
TABLE 6-1 Copper Intake (mg/day)a from Food and Supplements versus the Estimated Safe and Adequate Daily Dietary Intake (ESADDI)
|
|
|
Percentile |
ESADDIb or recommended levels (Mg/d) |
|||||||
|
Age |
Sex |
5 |
10 |
25 |
50 |
75 |
90 |
95 |
99 |
|
|
2-6 mon |
M&F |
0.3 |
0.4 |
0.5 |
0.7 |
0.9 |
1.1 |
1.2 |
1.6 |
0.4 to 0.6 recommended |
|
7-11 mon |
M&F |
0.3 |
0.4 |
0.5 |
0.7 |
0.9 |
1.2 |
1.3 |
1.7 |
0.6 to 0.7 recommended |
|
1-3 yr |
M&F |
0.3 |
0.4 |
0.5 |
0.7 |
1.0 |
1.3 |
1.7 |
2.9 |
Recommended for 7-10-year-old children: 1 to 2 |
|
4-8 yr |
M&F |
0.59 |
0.67 |
0.80 |
0.95 |
1.14 |
1.36 |
1.61 |
3.06 |
|
|
9-13 yr |
F |
0.64 |
0.72 |
0.86 |
1.04 |
1.26 |
1.54 |
1.84 |
3.23 |
|
|
|
M |
0.88 |
0.94 |
1.05 |
1.21 |
1.41 |
1.61 |
1.78 |
3.13 |
|
|
14-18 yr |
F |
0.64 |
0.75 |
0.89 |
1.08 |
1.32 |
1.64 |
1.96 |
3.32 |
|
|
|
M |
0.79 |
0.89 |
1.11 |
1.42 |
1.80 |
2.28 |
2.71 |
3.56 |
|
|
19-30 yr |
F |
0.77 |
0.83 |
0.95 |
1.12 |
1.38 |
1.82 |
3.03 |
3.84 |
ESADDI for adults: 1.5 to 3c |
|
|
M |
1.37 |
1.43 |
1.56 |
1.69 |
1.86 |
2.12 |
3.55 |
4.44 |
|
|
31-50 yr |
F |
0.72 |
0.81 |
0.95 |
1.17 |
1.52 |
2.32 |
3.09 |
4.19 |
|
|
|
M |
0.89 |
1.03 |
1.29 |
1.61 |
2.09 |
2.93 |
3.67 |
4.87 |
|
|
51-70 yr |
F |
0.61 |
0.68 |
0.84 |
1.07 |
1.48 |
2.92 |
3.25 |
4.22 |
|
|
|
M |
0.75 |
0.87 |
1.09 |
1.43 |
1.98 |
3.00 |
3.65 |
5.02 |
|
|
71 + yr |
F |
0.58 |
0.65 |
0.80 |
1.02 |
1.37 |
2.94 |
3.21 |
3.79 |
|
|
|
M |
0.72 |
0.83 |
0.99 |
1.26 |
1.66 |
2.89 |
3.41 |
4.61 |
|
|
Pregnant |
F |
0.71 |
0.82 |
1.07 |
1.62 |
3.11 |
4.03 |
4.39 |
5.56 |
|
|
a Intake figures developed by Environ International Corporation and Iowa State University Department of Statistics from the NHANES III nationwide survey. Breast-feeding infants and children, and eight individuals reporting greater than 150 mg/day of copper from supplements excluded from the analysis. b ESADDI values and recommended concentrations from the Food and Nutrition Board (FNB) (NRC 1989). The Institute of Medicine's (IOM) FNB is reviewing these values, and the reader should consult current IOM references for updated values. c For comparison, the acceptable range of intake values from IPCS (1998) is given as 1 to "several but not many mg per day." "Several" is defined as more than 2 to 3 mg/day. |
||||||||||
COPPER TOXICITY FROM SINGLE OR SHORT-TERM EXPOSURE
A wide range of copper toxicities have been observed subsequent to accidental or iatrogenic poisonings or suicide attempts: hepatic and renal failure, cirrhosis, hemolysis, vomiting, melena, hypotension, cardiovascular collapse, stupor, and coma (Chapter 5). Less severe acute copper toxicity, which can occur from corrosive water standing in copper plumbing and beverages or acidic foods stored or prepared in copper containers, is associated with nausea, vomiting, and diarrhea. Acute exposures at which gastrointestinal (GI) effects first appear are lower than those producing hepatotoxicity and other more severe acute effects.
Copper ions are generally more bioavailable in water than in food (Chapter 2). Because acute irritation of the GI tract is caused by the ionic form of copper, it is reasonable to suggest that GI irritation is more likely to be produced by drinking water than food. Consistent with that suggestion, the majority of reports on copper-induced GI irritation concern the ingestion of fluids high in this element.
Concentrations and doses associated with acute GI effects are detailed in Chapter 5. Human data are consistent with GI symptoms, such as nausea, abdominal pain, and vomiting, arising in sensitive individuals from drinking water and other fluids with copper concentrations greater than or equal to 3 mg/L (see Pizarro et al. 1999). Nausea typically occurs at lower concentrations than vomiting. In that study, some individuals were affected at 3 mg/L, but the majority reported no symptoms at 5 mg/L, indicating a range of sensitivity in the population. Nausea was noted as the sensitive effect in a larger experimental study (ICA, unpublished material, Oct. 13, 1999), and a range of sensitivity was also seen.
The MCLG is to be set at a concentration at which no known or expected adverse health effects occur and for which there is an adequate margin of safety. For noncancer end points, the approach typically applied to achieve that concentration is to determine a no-observed-adverse-effect level (NOAEL), by reviewing the health-effects literature and then dividing that level by dose adjustment and uncertainty factors (NRC 1977, 1994; IPCS 1994; EPA 1998). The adjustment factors applied account for human heterogeneity, study quality, study duration, and severity of effect. When based on the lowest-observed-adverse-effect level (LOAEL), an additional factor is applied to adjust to a level at which effects would not be expected to be observed. However, it is recognized that application of the standard regulatory approach for selection of safety factors for essential nutrients can result in levels below essential requirements.
The EPA (1991) established an MCLG for copper of 1.3 mg/L. The 1991 MCLG, explained in EPA (1994), is based on an analysis provided by EPA
(1987). The MCLG is based on a report (Wyllie 1957) of an episode of acute GI symptoms resulting from mixing alcoholic drinks in a copper-contaminated cocktail shaker. From a dose reconstruction, the author estimated that the dose associated with the lowest amount of cocktail consumed (half a glass, or 0.75 fluid ounces) resulting in symptoms was 5.3 mg. The level of 1.3 mg/L was recommended by EPA (1987) because it satisfied the nutritional requirements (noted by EPA (1987) to be 2–3 mg per day for adults and 1.5 to 2.5 mg per day for children) and because consumption of 2 L per day would result in intakes below the LOAEL (by a factor of 2). The EPA derivation thus uses total amount of daily copper as the dose metric for acute effects and a safety factor of 2.
Studies that have become available over the past decade have improved the basis for the establishment of the MCLG for copper. The recently published human experimental study of GI effects by Pizarro et al. (1999) indicates symptoms arising from copper exposure at approximately 3 mg/L of drinking water. The findings of the study are consistent with those from case reports, and because the study was controlled, it is preferred over them. With respect to the appropriate dose metric (discussed in Chapter 5) in studies of copper induction of the emetic response (e.g., Saito et al. 1998; Makale and King 1992), the response appears to be receptor mediated and a function of the effects of copper ion on the lining of the stomach. In human and experimental animals, the response occurs within a very short period after liquid is consumed, typically within a few minutes (Wang and Borison 1951; Pizarro et al. 1999). Thus in assessing the acute effects from exposure to copper in tap water, the water concentration appears to be of greater relevance than the amount of copper consumed over the day. Although additional work is needed to establish the relationship between concentration, volume consumed, and emetic response, the committee considers drinking-water concentration to be an appropriate dose metric for evaluating the MCLG with respect to the acute GI effects of copper.
In selecting adjustment uncertainty factors for setting the MCLG, issues to consider are the observations that (1) the effect is not severe and life threatening, and (2) the data supporting 3 mg/L as the concentration above which effects begin to appear are from controlled studies in humans and case reports, although much of the data are imprecise and limited. Limitations include the lack of measurement of confounders and biases and the small number of subjects in the published experimental study. Another consideration is the impact of taste on GI symptoms. The taste threshold for copper varies among individuals and depends on an individual's acuity. In controlled studies, Cohen et al. (1960), found that 50% of 15 to 20 taste panelists detected copper at concentrations of 6.6 mg/L and
above in distilled water and 12.7 mg/L and above in spring water. They estimated the most sensitive 5% could taste copper at concentrations greater than 2.6 mg/L in distilled water and 5 mg/L in spring water. Thus, levels associated with the GI response and taste overlap, and taste is a potential confounder in copper studies.
Two additional issues need to be considered in establishing the overall adjustment and uncertainty factors for copper. First, the LOAELs from the controlled human studies appear to be at the lower part of the dose-response curve, where the majority of the population is nonresponsive. Second, copper is an essential micronutrient. Although the committee recommends against basing the MCLG on nutritional requirements, the fact that the compound is essential should limit the size of the overall factor selected.
Ultimately, the selection of an adjustment factor for the acute GI symptoms observed-effect level of 3 mg/L is a scientific and a policy issue. On the basis of the data on GI effects from acute and subacute exposures, IPCS (1998) established as the upper limit of the acceptable range of intake for total copper a value of ''several but not many mg per day." "Several" is defined as more than 2 to 3 mg per day. WHO plans to re-evaluate copper in drinking water based on the IPCS (1998) analysis (Galal-Gorchev and Herrman 1996).
COPPER TOXICITY FROM CHRONIC EXPOSURE
The primary systemic effect of concern associated with chronic excess copper is liver toxicity (Chapter 5). In animal models, liver toxicity has been demonstrated in several species and there are case reports of liver disease in humans ingesting excessive concentrations of copper over a long period of time. In addition to liver pathology, Wilson disease suggests several other systemic end points for chronic copper toxicity. In theory, extrahepatic toxicity is subsequent to the accumulation of copper in the liver and results after cytosolic binding sites in the liver become saturated (Zucker and Gollan 1996). After that point, copper appears to be redistributed from the cytosol to lysosomes, and it can be released into systemic circulation. Copper can then accumulate in select extrahepatic sites, such as the kidney and brain. The neurological effects seen in Wilson-disease patients have not been observed in rodent studies of copper toxicity (see Chapter 5). Nonetheless, from Wilson disease and cases reports of accidental and intentional copper poisonings it can be concluded that the liver is a sensitive end point. An additional sensitive end point to consider that is unrelated to hepatic toxicity is GI irritation.
Sensitive Populations
Severe disorders of copper homeostasis, which occur with Wilson disease, result in hepatic toxicity and other copper toxicity, primarily involving the central nervous system. The prevalence of Wilson disease is small, reported to be roughly 1:40,000 live births (Chapter 6). However, because that estimate is based on autopsy data, it might be an underestimate. The actual prevalence of Wilson disease is likely to be considerably greater, by perhaps a factor of 4 or more. Those diagnosed with this serious illness are generally under a clinician's care. The Wilson-disease patient has limited ability to excrete copper, and copper intake through food and water is curtailed. Certain foods, such as liver and shellfish, that are high in copper should be avoided, and when the copper content in drinking-water sources is high (e.g., more than 0.1 mg/L), alternative sources are typically recommended. Thus, copper concentrations in drinking water for Wilson-disease patients are not managed through the MCLG process. However, in other groups, such as infants with altered copper metabolism, toxicity can occur when individuals in these groups consume drinking water with relatively high copper concentrations. These groups should be considered in establishing the MCLG.
Carriers of the Wilson-Disease Gene and Other Genetically Sensitive Groups
It is likely that a copper sensitivity gene contributes to the hepatic copper toxicity observed in infants and young children ingesting increased amounts of copper in milk and water. The current evidence is that manifestations of Tyrolean infantile cirrhosis (TIC), Indian childhood cirrhosis (ICC) and idiopathic copper toxicosis (ICT) involve both heredity and high copper intake (Muller et al. 1996 and 1998; Tanner 1998). It is a reasonable hypothesis that chronic ingestion of moderately increased amounts of copper produces disease in copper-susceptible genotypes.
Heterozygous carriers of the Wilson-disease gene might represent a susceptible group for copper hepatotoxicity. As evidence, under current environmental conditions in the United States, the heterozygous carriers accumulate copper and have abnormally high concentrations in the liver and urine (see Chapter 4). They can be defective in copper handling in the liver as evidenced by 64Cu incorporation into ceruloplasmin (Brewer and Yuzbasiyan-Gurkan 1992). In addition, unidentified copper sensitivity genes might be responsible for the observed childhood copper toxicity syndromes (Muller et al., 1998; Tanner, 1998).
A heterozygote carrier rate of slightly greater than 1% corresponds to a
prevalence rate of 1:40,000 for those homozygous for the Wilson-disease gene. The actual value might be considerably higher (on the order of 2%) if, as expected, the actual prevalence of Wilson disease is underestimated by a factor of approximately 4. Although Wilson heterozygote carriers likely differ in sensitivity, other genetic mutations might also increase copper retention. Thus, at least 1% of the population might be susceptible for increased copper retention on the basis of genetic susceptibility. Provided that increased copper retention confers increased risk of liver toxicity, the committee concludes that groups of this size should be taken into account in establishing the MCLG for chronic exposures.
Infants
Infants might represent a susceptible group for two reasons. First, newborns have approximately 3 times as much hepatic copper as adults. Those concentrations steadily decrease in early life and achieve adult concentrations after about 6 months (Keen 1996). Infants respond to higher copper intake by increasing fecal losses and decreasing percentage absorption, but they are not as efficient as adults at doing so, and cases of excess copper associated with genetic predisposition (e.g., TIC, and ICT) are most frequently observed in young children. Second, on a body-weight basis, infants drink considerably more water than adults, particularly those that are fed formula. The NHANES III (Table 6-1) and Continuing Survey of Food Intakes of Individuals (CSFII) indicate that intake of copper for the majority of infants is at adequate intake concentrations and above. Infants at higher percentile dietary intakes might be at a heightened risk for copper toxicity if intake of copper in drinking water is excessive. High exposures can especially result from use of first draw-water in preparing infant formula. Evidence from review of ICC and TIC cases, however, indicates a genetic predisposition to liver toxicity from elevated copper ingestion in infants and indicates that all infants are not equally sensitive (Muller et al. 1996; Tanner 1998).
Chronic Liver Disease
Copper accumulation in the liver is also associated with chronic cholestasis. Individuals with primary biliary cirrhosis, intrahepatic cholestasis in childhood, and extrahepatic biliary obstruction might be at increased risk. The extent to which the increased copper affects the already compromised liver is unknown.
Glucose-6-Phosphate Dehydrogenase Deficiency
Individuals with glucose-6-phosphate dehydrogenase deficiency (G6PD) have been hypothesized to be at increased risk for excess copper, because, in vitro, G6PD-deficient red blood cells are more susceptible than normal to hemolysis and damage from copper. However, as discussed in Chapter 5, copper bound to ceruloplasmin is not available for red-blood-cell toxicity. The relatively small amount of low-molecular-weight bound copper available is not likely to alter the survival of G6PD-deficient red blood cells. Thus, there is insufficient evidence that this group represents a susceptible group for chronic toxicity to copper from drinking water.
Overall, the fraction of the population with inherent sensitivity to copper due to genetic make-up, age, or disease state might be 1% or greater.
Implications for the MCLG
Copper is an essential nutrient subject to tight homeostatic control. Applying the standard regulatory approach for selecting safety factors and animal effect levels for essential elements and vitamins can result in guidance inconsistent with known nutritional requirements. Copper is one such case. Concentrations associated with effects observed in long-term animal studies are given in Table 6-2, along with adjustment and uncertainty factors traditionally applied. It is noteworthy that for the rat and rabbit, the LOAEL or NOAEL divided by the traditional adjustment and uncertainty factors results in concentrations below the dietary intake concentrations identified as safe and adequate for adults (1.5 to 3 mg/day, corresponding to roughly 20 to 40 µg/kg per day) and below the intake concentration of 75 µg/kg per day recommended for infants (NRC 1989). They also fall below the value identified by WHO (1996) as a probable average adult-based requirement (11 µg/kg-day) and safe minimum mean copper intake of populations (20 µg/kg-day) and below the values identified by WHO as normative requirements for infants (0–3 months, 50–80 µg/kg per day; 3–6 months, 40–70 µg/kg per day; 6–12 months, 40 µg/kg per day). The detailed pharmacokinetic and mechanistic information needed to perform a more refined safety assessment based on data from chronic exposure animal studies is not available. The chronic exposure animal studies provide qualitative support for findings in humans sensitive to copper. However, they provide little guidance for the establishment of an MCLG for copper.
Human case reports and series suggest a range of copper intakes associated with liver toxicity in sensitive individuals. Table 6-3 lists reports in the literature of increased copper exposure in cases of ICT, ICC, and TIC
TABLE 6-2 Copper-Effect Doses Observed in Long-Term Animal Studies
|
Species |
Effect |
LOALEa (mg Cu/kg-d) |
Traditional adjustment factorb |
Reference |
|
Mouse |
Reduction in life span |
42.5 |
1,000 |
Massie and Aiello 1984 |
|
Rat (90-d) |
Hepatic; increase in SGOT activity |
7.9 |
1,000–10,000 |
Epstein et al. 1982 |
|
Rat (92-d) |
Renal Effects |
16 |
10,000 |
NTP 1993 |
|
Rabbits |
Marked hepatic toxicity |
10 |
1,000 |
Tachibana 1952 |
|
a Effects were either observed at all doses or the only dose studied. Thus, NOAELs were not observed in the studies cited in the table. b Typically, a factor of 10 is used to extrapolate from a dose associated with an effect (i.e., the LOAEL) to predict a NOAEL; a factor of 10 is used to extrapolate from animals to humans; a factor of 10 is used to account for variability within the human population; and a factor of 10 is used to extrapolate studies of insufficient duration to chronic duration. Abbreviations: LOAEL, lowest-observed-adverse-effect level; NOAEL, no-observed-adverse-effect level. |
||||
and a case of self-exposure in an individual otherwise thought to be healthy until the time that signs of liver toxicity began to appear. Those reports are subject to imprecision in exposure ascertainment, but overall suggest that when formulas are made from drinking water containing 3 mg/L and above, genetically sensitive infants might be at increased risk of liver toxicity. It is difficult to ascertain copper concentrations in water and intake associated with toxicity because of varying copper concentrations due in large part to the flushing of the household system as water is used throughout the day. Although these estimates are far from exact, they nonetheless provide an indication of intakes that might cause hepatotoxicity to sensitive individuals upon chronic exposure.
The one case of chronic poisoning through self-exposure to copper supplements (O'Donohue et al., 1993) corresponds to a dose of 0.4–0.9 mg/kg per day, which is similar to crude estimates of dose for cases of ICC and TIC obtained by experimentally simulating copper storage and heating of formula milk (Table 6-3).
Division of the doses reported to induce hepatic toxicity in humans by traditional uncertainty and adjustment factors would result in a suggested maximal concentration of copper below those considered to be essential.
TABLE 6-3 Case Reports and Series of Toxicity Following Chronic Exposure to Copper
|
Effect and Subject |
Exposure Circumstance and Copper Measurement |
Reference |
|
Young adult male; micronodular cirrhosis and liver failure; Wilson homozygosity and heterozygosity ruled out |
Mineral supplementation of 30 mg/d for 2 yr, followed by 60 mg/d for 1 yr; estimated dose is 0.4 to 0.9 mg/kg per d |
O'Donohue et al. 1993 |
|
Idiopathic copper toxicity in predominantly non-breast fed infants: |
|
|
|
7-mon-old German girl |
Tap water, 0.4 to 5.5 mg/L |
Muller-Hocker et al. 1987; Muller et al. 1998 |
|
5- and 9-mon-old siblings |
Tap water, 2.2 to 3.4 mg/L |
Muller-Hocker et al. 1988 |
|
10-mon-old, rural southern Ireland |
Cold tap water, 3.9 mg/L; hot, 8 mg/L; copper blackened the inside of kettles |
Baker et al. 1995 |
|
13-mon-old German male |
Tap water, 12–29 mg/L after standing in pipes |
Bent and Bohm 1995 |
|
14-mon-old Australian boy |
First-draw cold water, 6.75 mg/L; hot water, 9.4 mg/L |
Walker-Smith and Blomfield 1973 |
|
15-mon-old female, rural southeastern Ireland |
Well-water heated and stored in copper container; hot water measured 6.3 mg/L; cold water, 2.3 mg/L |
Baker et al. 1995 |
|
Indian childhood cirrhosis |
Heating and storing infant's milk in untinned copper and brass containers; simulated under experimental conditions copper at 6 mg/L in milk in copper containers, and associated dose, ≈ 0.9 mg/kg-d |
O'Neill and Tanner 1989 |
|
Tyrolean childhood cirrhosis |
Infant feeding milk prepared in untinned copper containers, in experimental simulations, 10–63 mg/L |
Muller et al. 1996, 1998 |
For example, a LOAEL associated with such a serious end point as liver failure might be divided by a factor of 3 to 10 to adjust for the severity of the end point, a factor of 10 to adjust an effect level to a level associated with no effect, and an additional factor between 2 and 10 to adjust for human heterogeneity. For conditions in which the observation is for a susceptible group a factor less than 10 might be used.
Although further research is needed to better define drinking-water doses associated with copper toxicity in those with genetic susceptibility syndromes, such as ICT and ICC, drinking-water concentrations of 3 mg/L and above are associated with hepatotoxicity in case reports (Table 6-3). However, that observation is for case reports for which cumulative intake of copper from water and other sources is not known.
The MCLG is a health goal set at concentrations at which no known or expected adverse health effects occur and the margin of safety is adequate (Chapter 1). However, it is difficult to define an adequate margin of safety for copper toxicity. The standard method for performing a safety assessment for chronic toxicity end points cannot be applied, primarily because the method does not account for certain features related to copper essentiality, tight homeostatic control, and the relatively narrow range of acceptable intake levels. Also, the dose of copper that causes liver toxicity in sensitive humans is uncertain. Nonetheless, it is important to evaluate the potential for liver toxicity from chronic exposure to copper in drinking water when considering changes to the MCLG.
CHRONIC COPPER EXPOSURE THROUGH TAP WATER
Comprehensive nationwide survey data for copper in drinking water are not available, and therefore estimates of copper intake via water cannot be estimated accurately. Clues as to the potential for copper overexposure via tap water come from federal reporting requirements. Under federal law, water systems are required to be sampled for copper in first-draw water (i.e., after water has been "motionless" for at least 6 hr) at the cold-water tap at locations in the water system vulnerable to copper contamination (EPA 1991, 1994). When the 90th percentile of samples taken exceeds 1.3 mg/L, the water purveyor is to report that percentile value to the states, which in turn are required to compile and report such values to the U.S. Environmental Protection Agency.
Figure 6-2 presents the 90th percentile copper concentrations that water purveyors reported for their systems from 1991 to 1999 (E. Ohanian, EPA, personal commun., Nov. 23, 1999). The 7,307 values reported correspond to roughly 4,500 individual water systems. With a few exceptions, water systems reporting values greater than 5 mg/L are small, serving

FIGURE 6-2 Ninetieth percentile copper concentrations for water systems reported to EPA under federal reporting requirements from 1991 to 1999.
Source: Data from E. Ohanian, EPA, personal commun., Nov. 23, 1999.
3,300 or fewer people. The majority of those serve nonresidential consumers, such as those at recreational facilities and schools. By law, corrective action might be required for a number of those systems. Nonetheless, the reported 90th percentile concentrations for numerous systems, some of which serve small communities, are notably high, suggesting the potential for copper overexposure.
DIETARY CONTRIBUTION AND TOTAL COPPER INTAKE
Formula-Fed Infants
Formula-fed infants, particularly those receiving most sustenance from powdered formulas, are a group of particular concern. Virtually all fluid of young infants on a powdered formula diet can come from tap water, and the powder formulation is designed to provide the copper requirement.
Using information on energy requirements, copper intake via powder formula can be estimated. Infant formulas sold in the United States generally contain 75 µg of copper per 100 kcal of energy supplied by the powder. Energy expenditure and growth studies provide data on daily energy intake of infants, and data compiled (Whitehead 1995) are presented in
Figure 6-3. In infants, the energy requirement on a body-weight basis (e.g. kilocalorie per kilogram of body weight) appears to be greatest during the neonatal period and drops steadily until roughly age 6 months, when growth slows and the requirement becomes fairly constant. From those data, the average energy requirement during the first 6 months of life is roughly 100 kcal/kg per day. Copper intake associated with that energy requirement is 75 µg/kg per day (75 µg/kg per day = 100 kcal/kg-day × 75 µg/100 kcal) from the formula powder alone. Daily copper intake from infant formula made from powder and copper-contaminated tap water can be estimated. At the current MCLG of 1.3 mg/L, average daily copper intake during the first 6 months of life is esti mated to be 267 µg/kg per day for the average powder-formula-fed infant.
WHO (1996), after considering copper concentrations not associated with detrimental effects in adult humans, set a value of 150 µg/kg-day as the upper limit of the safe range for mean copper intake for infants.1 The
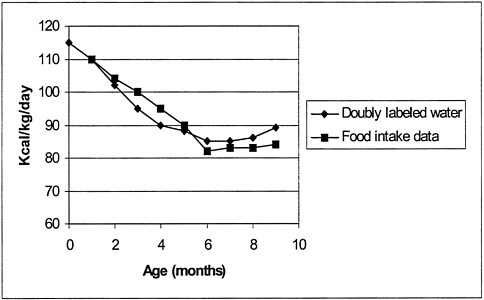
Figure 6-3 Energy requirements for infants predicted from doubly labeled water (following technique of Coward et al. 1979) and food-intake data.
Source: Adapted from Whitehead 1995.
above value is roughly half the intake for infants on a powdered formula diet made with water containing copper at the MCLG. The copper concentration in tap water corresponding to the WHO upper limit is 0.44 mg/L for the average infant fed for the first 6 months of life on standard powder formula. Formula-fed infants consuming water contaminated at 6 mg/L would approach doses of 1 mg/kg per day, a dose associated with cases of liver toxicity in genetically sensitive infants by some researchers (Table 6-3) and approximately a factor of 10 of doses with observed effects in chronic exposure animal studies.
Consumption of water at such a high concentration corresponds to a plausible worst-case scenario for copper intake in which the day's formula is made with first-draw water. The values are calculated for the median infant. Breast-milk consumption (Hofvander et al. 1982; Dewey et al. 1991) and energy requirement (Prentice et al. 1988; Whitehead 1995) studies suggest moderate variation in intake, even when normalized to body size. Those studies suggest that the more voracious infants can consume at rates 30–50% higher than the average.
General Population
From the results of nationwide dietary surveys, copper intake from food can be estimated for different age groups and for the general population (Table 6-4). Dietary survey information can also be used to evaluate water consumption habits and variations in different age groups (Table 6-5). Assuming fixed concentrations of copper, possible intake of copper through water can be evaluated. Total copper intake through food and water can then be evaluated. Figure 6-4 illustrates total copper intake at different concentrations of copper in water. Again, at a concentration of 3 mg/L, relatively high copper intake via water can result for some segments of the population.
CONCLUSIONS
-
As with any essential nutrient, low copper intake can result in pathological changes, and high intake can result in toxicity.
-
GI symptoms, such as nausea and vomiting, associated with acute exposures can be seen in sensitive individuals at drinking-water concentrations of copper greater than 3 mg/L.
-
Systemic toxicity is not expected with an acute exposure to copper at concentrations found in drinking water. That is true even for first-draw water (less than 30 mg/L).
TABLE 6-4 Copper Intake (µg/kg-day)a from Food and Supplements
-
Those at increased risk for chronic liver toxicity are individuals on fluid diets (e.g., formula-fed infants), or with genetically determined differences in copper metabolism, or a combination of these factors.
-
In the case of sensitive populations, a substantial increase in copper intake from water increases the risk for hepatotoxicity. Therefore, in considering changes in the MCLG, the extent to which the copper in water can contribute to the overall dietary copper intake of an individual must be considered.
-
Copper drinking-water concentrations of 3 mg/L and greater have been associated with cases of systemic copper toxicity, suggesting that increasing the MCLG to 3 mg/L and greater might cause an increase in liver disease in genetically susceptible populations.
TABLE 6-5 Daily Tap-Water Consumption (mL/kg)a
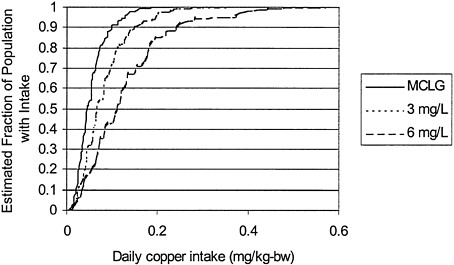
FIGURE 6-4 Total copper intake from water, diet and dietary supplements. Estimates are given for copper concentrations in drinking water at the current MCLG (1.3 mg/L; solid line), 3 mg/L (dotted line), and 6 mg/L (dashed line). Copper concentrations are assumed to be constant.
-
It has been estimated that about 1% of the U.S. population have polymorphisms that increase copper retention, and might increase their risk for copper toxicosis.
RECOMMENDATIONS
-
The MCLG in water should be based on the toxic effects of copper, rather than on copper deficiency.
-
Issues that should be considered in establishing adjustment and uncertainty factors for acute effects are that copper is an essential micronutrient, that the GI effects are not severe or life-threatening, that the effect level is based on human studies and case reports, and that the effect level appears to be at the lower part of the dose-response curve, where the majority of the population is nonresponsive.
-
Given the potential risk for liver toxicity in individuals with polymorphisms, it is recommended that the MCLG for copper should not be increased at this time.
-
Additional information on total copper doses received from drinking water is necessary before the importance of systemic chronic toxicity can be fully evaluated in susceptible populations.
-
Better quantification of the frequency and characterization of copper-sensitive populations should be undertaken.
-
When the above information is obtained, the MCLG for copper should be re-evaluated.
REFERENCES
Baker, A., S. Gormally, R. Saxena, D. Baldwin, B. Drumm, J. Bonham, B. Portmann, A.P. Mowat. 1995. Copper-associated liver disease in childhood. J. Hepatol. 23(5):538–543.
Bent, S. and K. Bohm 1995. Copper-induced liver cirrhosis in a 13-month old boy [in German]. Gesundheitswesen 57(10):667–669.
Brewer, G.J. and V. Yuzbasiyan-Gurkan. 1992. Wilson disease. Medicine 71(3):139–164.
Cohen, J.M., L.J. Kamphake, E.K. Harris, R.L. Woodward. 1960. Taste threshold concentrations of metals in drinking water. J. Am. Water. Works Assoc. 52(5):660–670.
Coward, W.A., M.B. Sawyer, R.G. Whitehead, A.M. Prentice, and J. Evans. 1979. New method for measuring milk intakes in breast-fed babies. Lancet 2(8132):13–4.
Dewey, K.G., M.J. Heinig, L.A. Nommsen and B. Lonnerdal. 1991. Adequacy of energy intake among breast-fed infants in the DARLING study: Relationships to growth velocity, morbidity, and activity levels. J. Pediatr. 119(4):538–547.
EPA (U.S. Environmental Protection Agency). 1987. Drinking Water Criteria Document of Copper. Environmental Criteria and Assessment Office, Office of Health and Environmental Assessment, U.S. Environmental Protection Agency. Cincinnati, OH. February.
EPA (U.S. Environmental Protection Agency). 1994. Drinking water maximum contaminant level goals and national primary drinking water regulations for lead and copper. Fed. Regist. 59(125):33860–33864.
EPA (U.S. Environmental Protection Agency). 1991. Monitoring requirements for lead and copper in tap water. Fed. Regist. 56(110):26555–26557.
EPA (U.S. Environmental Protection Agency). 1998. Draft water quality criteria methodology: human health. Fed. Regist. 63(157):43756–43828.
Epstein, O., R. Spisni, S. Parbhoo, B. Woods, and T. Dormandy. 1982. The effect of oral copper loading and portasystemic shunting on the distribution of copper in the liver, brain, kidney, and cornea of the rat. Am. J. Clin. Nutr. 35(3):551–5.
Galal-Gorchev, H. and Herrman, J.L. 1996. Letter to A.C. Kolbye, Jr., editor of Regulatory and Pharmacology, on the evaluation of copper by the Joint FAO/WHO Expert Committee on Food Additives from WHO. Sept. 12, 1996.
Hofvander, Y., U. Hagman, C. Hillervik, and S. Sjolin. 1982. The amount of milk consumed by 1–3 months old breast- or bottle-fed infants. Acta Paediatr. Scand. 71(6):953–958.
IPCS (International Programme on Chemical Safety). 1994. Assessing Human Health Risk of Chemicals: Derivation of Guidance Values for
Health Based Exposure Limits. Environmental Health Criteria No. 170. Geneva, Switzerland: World Health Organization.
IPCS (International Programme on Chemical Safety). 1998. Copper. Environmental Health Criteria 200. Geneva, Switzerland: World Health Organization.
Keen, C.L. 1996. Teratogenic effects of essential trace metals: deficiencies and excesses. Pp. 977–1001 in Toxicology of Metals, L.W. Chang, L. Magos, and T. Suzuki, eds. New York: CRC Press.
Makale, M.T., and G.L. King. 1992. Surgical and pharmacological dissociation of cardiovascular and emetic responses to intragastric CuSO4 . Am. J. Physiol. 263(2 Pt 2):R284–R291.
Massie, H.R. and V.R. Aiello. 1984. Excessive intake of copper: influence on longevity and cadmium accumulation in mice. Mech. Ageing. Dev. 26(2–3):195–203.
Müller, T., W. Müller, H. Feichtinger. 1998. Idiopathic copper toxicosis. Am. J. Clin. Nutr. 67(suppl):1082S–1086S.
Müller, T., H. Feichtinger, H. Berger, W. Müller. 1996. Endemic Tyrolean infantile cirrhosis: an ecogenetic disorder. Lancet 347(9005):877–880.
Müller-Höcker, J., U. Meyer, B. Wiebecke, G. Hubner. 1988. Copper storage disease of the liver and chronic dietary copper intoxication in two further German infants mimicking Indian childhood cirrhosis. Path. Res. Pract. 183(1):39–45.
Müller-Höcker, J., M. Weiss, U. Meyer, P. Schramel, B. Wiebecke, B.H. Belohradsky, G. Habner. 1987. Fatal copper storage disease of the liver in a German infant resembling Indian childhood cirrhosis. Virchows. Arch. A. Pathol. Anat. Histopathol. 411(4):379–385.
NRC (National Research Council). 1977. Drinking Water and Health. Washington, D.C.: National Academy of Sciences.
NRC (National Research Council). 1989. Recommended Dietary Allowances, 10th Ed. Washington, DC.: National Academy Press.
NRC (National Research Council). 1994. Science and Judgment in Risk Assessment. Washington, D.C.: National Academy Press.
NTP (National Toxicology Program). 1993. NTP Technical Report on Toxicity Studies of Cupric Sulfate (CAS No. 7758-99-8) Administered in Drinking Water and Feed to F344/N Rats and B6C3F1 Mice. NTIS PB94-120870.
O'Donohue, J., M.A. Reid, A. Varghese, B. Portmann, R. Williams. 1993. Micronodular cirrhosis and acute liver failure due to chronic copper self-intoxication. Eur. J. Gastroenterol. Hepatol. 5:561–562.
O'Neill, N.C., and M.S. Tanner. 1989. Uptake of copper from brass vessels by bovine milk and its relevance to Indian childhood cirrhosis. J. Pediatr. Gastroenterol. Nutr. 9(2):167–172.
Pizarro, F.M., R. Olivares, P. Uauy, P. Contreras, A. Rebelo, and V. Gidi.
1999. Acute GI effects of graded levels of copper in drinking water. Environ. Health Perspect. 107(2):117–121.
Prentice, A.M., A. Lucas, L. Vasquez-Velasquez, P.S. Davies, and R.G. Whitehead. 1988. Are current dietary guidelines for young children a prescription for overfeeding? Lancet 2(8619):1066–9.
Saito, R., Y. Suehiro, H. Ariumi, K. Migita, N. Hori, T. Hashiguchi, M. Sakai, M. Saeki, Y. Takano, and H. Kamiya. 1998. Anti-emetic effects of a novel NK-1 receptor antagonist HSP-117 in ferrets. Neurosci. Lett. 254(3):169–172.
Tachibana, K. 1952. Pathological transition and functional vicissitude of liver during formation of cirrhosis by copper. Nagoya J. Med Sci. 15:108–114.
Tanner, M.S. 1998. Role of copper in Indian childhood cirrhosis. Am. J. Clin. Nutr. 67(5 Suppl.):1074S–1081S.
Walker-Smith, J. and J. Blomfield. 1973. Wilson's disease or chronic copper poisoning? Arch. Dis. Child. 48(6):476–479.
Wang, S.C. and H.I. Borison. 1951. Copper sulphate emesis: study of afferent pathways from the GI tract. Am. J. Physiol. 164:520–526.
Whitehead, R.G. 1995. For how long is exclusive breast-feeding adequate to satisfy the dietary energy needs of the average young baby? Pediatr. Res. 37(2):239–243.
WHO (World Health Organization). 1996. Trace Elements in Human Nutrition and Health. Geneva: World Health Organization.
Wyllie, J. 1957. Copper poisoning at a cocktail party. Am. J. Publ. Health 47:617.
Zucker, S. and J.L. Gollan. 1996. Wilson's disease and hepatic copper toxicity. Pp. 1405–1438 in Hepatology. A Textbook of Liver Disease, Vol. 2, 3rd Ed., D. Zakim and T.D. Boyer, eds. Philadelphia: W.B. Saunders.





















