CHAPTER 30
CAN TECHNOLOGY AID SPECIES PRESERVATION?
WILLIAM CONWAY
Director, New York Zoological Society, Bronx, New York
In the preservation of biological diversity, the use of technology is a last resort. When the preservation of ecosystems falters, their fragments may have to be cared for piece by piece.
FOUR OBSTACLES TO SPECIES SURVIVAL
A species of limited distribution faces at least four obstacles. First, there may not be sufficient habitat and the possibility of obtaining numerous large new nature preserves is remote. Even protecting some areas already designated as preserves is not proving possible, and no land whatsoever will be set aside for large numbers of species. Second, many of the preserved habitats will be in pieces too small and too subject to change to sustain unmanaged, genetically and demographically viable populations of the animals and plants they seek to protect (Soulé and Wilcox, 1980). Third, although the majority of wild species must persist outside of wildlife preserves, large land vertebrates and great aggregations that conflict with humans will be mostly confined to refuges and those outside will require continual monitoring, protection, and help. Finally, human populations will continue to grow for some time, inexorably reducing resources available to other species, while human land-use patterns, cultural attitudes, and economic practices will continue to shift and change (Myers, 1979).
PROBLEM AND APPLICATION
Despite the factors mentioned above, the loss of a wild population is not always the result of irreversible habitat change. It can come about for transient economic
and cultural reasons, such as overhunting of the American bison, Arabian oryx, white rhinoceros, and American beaver. Or it may happen for correctable environmental reasons, such as introduced pesticides (the peregrine falcon and bald eagle) and introduced predators and competitors (the Hood island giant tortoise and Howe Island wood rail). And the loss may also last for unpredictably long periods, as with the Pere David’s deer, Mongolian wild horse, European wisent, perhaps the Siberian tiger, brown-eared pheasant, Mauritius pink pigeon, and Guam rail; it could even be forever. But populations of all these species and many more have been increased in one situation or another by intervention strategies (see Table 30–1).
Where a wild population’s ability to survive is lost, especially where the threat and destruction may be temporary, e.g., for the American bison, peregrine falcon
TABLE 30–1 Ex Situ Care and Biotechnology. Each Technique Has Been Utilized with the Species Listed Below It on a Long-Term or Experimental Basis
|
Intervention Technique |
Species |
|
Short-term propagation and reintroduction |
Golden lion tamarin, cheetah, wolf, red wolf, American and European bison, Arabian oryx, onager, Andean condor, bald eagle, peregrine, Hawaiian goose, Lord Howe Island wood rail, Guam rail, European eagle owl, Guam kingfisher, Galapagos giant tortoise, Galapagos land iguana, Ash Meadows Amargosa pupfish |
|
Long-term propagation |
Lion-tailed macaque, Siberian tiger, Pere David’s deer, European bison, Przewalski horse, brown-eared pheasant, Edward’s pheasant, Bali myna, white-naped crane, addax, slender-horned gazelle, scimitar-horned oryx, gaur, Gérevy’s zebra, Puerto Rican horned toad, Chinese alligator, Mauritius pink pigeon, Madagascar radiated tortoise, Aruba Island rattlesnake |
|
Relocation, transplantation |
Koala, mongoose lemur, aye-aye, brown lemur, chimpanzee, gorilla, squirrel monkey, wooly monkey, spider monkey, common marmoset, black rhinoceros, white rhinoceros, red deer, white-tailed deer, mule deer, moose, Tule elk, bighorn sheep, musk-ox, pronghorn antelope, roan antelope, mountain goat, African elephant, more than 400 species of birds, many reptiles and amphibians |
|
Fostering, cross-fostering |
Peregrine, bald eagle, whooping crane, masked quail, polar bear (captive), many species of waterfowl, pigeons, cranes (in nature and captivity), and passerine birds in captivity |
|
Artificial incubation |
Gharial, Siamese crocodile, Chinese alligator, green turtle, ridley, hooded crane, whooping crane, white-naped crane, and many other birds, reptiles, amphibians, and fishes |
|
Artificial rearing |
Hundreds of species of most vertebrate groups |
|
Artificial insemination |
Alligator, ocellated turkey, brown-eared pheasant, whooping crane, squirrel monkey, yellow baboon, giant panda, guanaco, Speke’s gazelle, gemsbok, bighorn sheep |
|
Embryo transfer |
Gaur, bongo, eland, common zebra, Przewalski horse, cottontop marmoset, yellow baboon |
(see Cade, Chapter 32), bald eagle, Arabian oryx, or Ash Meadows Amargosa pupfish, we need new scientific comprehension and a responsive technology. We must be able to relocate, sustain, or store a threatened population, to start and stop its propagation, and to reintroduce or remove it.
Because many populations of species in nature are becoming fragmented and isolated from each other, the emigration and immigration necessary for them to find unrelated mates of the right age and gender are becoming impossible. In such instances, intervention technologies will be necessary to effect the required movements. In such small populations, localized catastrophes, disease, sex and age imbalances, and even inbreeding can threaten viability (Schonewald-Cox et al., 1983). In response, technology may make it possible to remove or insert individuals into populations or even embryos or zygotes with needed characteristics into individuals (see Dresser, Chapter 34).
Where conservation biologists identify threatened but critical coevolutionary links, especially those between keystone species essential to ecosystem stability and diversity, sustaining these links for a time by scientific management of predators, competitors, even of environmental chemistry, microclimate, and with reintroductions, may be our only option for preservation.
LIMITATIONS OF SCIENCE AND TECHNOLOGY
But if such technological treatments and repairs are possible, why can not science and technology simply save biodiversity? Perhaps, as H.L.Mencken is reported to have said in a different context: “For every complex problem there is a simple answer and it is wrong.”
Most losses of biological diversity, to say nothing of lost ecological services, are quite beyond human ability to repair. Too many very intricately interdependent species are being lost too rapidly with too many unpredictable consequences for others. Besides, sustaining species in a freezer, in a captive population, or in small fragmented refuges provides little to the Earth in the way of basic ecological services. However, intensive care and biotechnology can preserve some diversity that would otherwise be lost. But the greatest dimension of such preservation is depressingly slight compared with that which can be or could have been sustained in adequately designed and protected nature preserves and by understanding accommodation outside preserves.
NUMBERS VERSUS TECHNOLOGY
It is the numbers, whether they be those of the great variety of creatures requiring help or those representing the scarcity of biologists and dollars to help them, that discourage prospects for sustaining a sizeable proportion of living creatures solely through technology—despite our most ardent wishes or most arrogant imaginings. There are perhaps 400,000 species of plants. In Chapter 31, Ashton discusses their preservation in gardens and arboreta. But there may be 30 million kinds of invertebrates, mostly insects. Despite their importance, the overwhelming number of
specialized invertebrates makes it logistically impossible for technology to contribute to the preservation of a significant representation for restoration programs.
There are only about 41,000 vertebrates. Of these, 19,000 or so are fishes, about 9,000 are birds, 6,000 are reptiles, 3,100 are amphibians, and 4,300 are mammals (E.O.Wilson, personal communication, 1987). For those that have been or will be totally displaced from nature, we have only a few specialized propagation centers and the world’s zoos.
Zoos currently house about 540,000 mammals, birds, reptiles, and amphibians—an almost trivial number in relation to original wild populations but significant in the impact on human interest. The number is roughly equal to 1% of the domestic cats in American households, 10% of the cats and dogs euthanized annually in the United States, or about 25% of the deer taken by U.S. sportsmen each year. Zoos are popular but have little room. The spaces for animals in the world’s zoos could all fit comfortably within the District of Columbia. Even if half these spaces were suitable for propagation of vanishing animals, the individual numbers of each species necessary to keep viable populations would make it impractical for zoos to sustain more than 900 species very long and probably far fewer in conventional breeding programs (Conway, 1986). But it will not always be necessary to sustain a population for a long time or to do so conventionally. Zoos, revised and improved, can come to have a special role in species preservation, for they represent a unique devotion of local human resources to the care of foreign wildlife.
In the past few years, the world’s zoos have bred more than 19% of all the living mammals and more than 9% of the birds. Thus far, criteria of genetic uniqueness as well as the practicality of care have guided long-term propagation programs. In the future, more attention must be given to ecological criteria with an eye toward the future needs of restoration programs, to species that naturally occur at low density, to the great predators and large ungulates, and to the primates. A foundation for such help rests in growing international collaboration and, in the United States, in unequalled programs of coordinated animal data gathering in the International Species Inventory System and species management in the Species Survival Plan of the American Association of Zoological Parks and Aquariums.
Zoos are breeding orangutans and Chinese alligators, Bali mynas, pink pigeons, and Puerto Rican horned frogs, addax, slender-horned gazelles, wattled cranes, and black lemurs. They have pioneered rare embryo transfers between animal species, artificial rearing techniques, cross-fostering between species, and necessary long-term contraceptives for population management—a host of fundamental technological tools essential to the prospects of helping species in extremis.
Even so, extensive scientific and technological advances would be necessary to appreciably expand the space available for the care of species losing their homes. But where cold storage of sperm and embryos is possible, a herd of wild cattle or antelope can be cared for in a space no larger than a soda straw, moved without risk of trauma, and stored indefinitely—if we are satisfied to have our wild cattle and antelopes in soda straws. Yet, we must consider the alternatives and their time scales.
Unfortunately, practically all that we know of animal reproductive physiology has been worked out with a few domestic and laboratory species. The technology
of sperm and embryo storage in use with domestic cattle is the product of 20 years of research, millions of dollars, and thousands of specimens—a critical matrix for investigation and discovery. A vast amount of research would have to be undertaken before a technology of embryo and sperm storage and transfer as reliable as that in use with cattle could be available for wild species without domestic analogs. But where is the economic incentive for such research? Where are the animals? The apparently simple techniques of artificial insemination, for example, have been successful with scarcely 20 wild species of mammals. Nonetheless, development of the scientific understanding necessary to long-term propagation is a technological fulcrum for many intensive species care programs. In Chapter 33, Seal discusses some of the challenges.
TECHNOLOGY IS EXPENSIVE
Unhappily, high cost is characteristic of high-tech applications, and whereas the capability and the money to apply advanced technologies to preservation is located mostly in wealthy northern countries, the largest problems of species loss are in poor tropical countries. Money used for high-tech intervention strategies obviously can not be used to preserve habitat. For less-developed countries, habitat preservation is the only realistic strategy, unless help comes from outside.
Whatever the help, no available amounts of money can ensure the protection of many species in nature, even vertebrates, such as the addax and scimitar-horned oryx from the Sahel or Guam’s kingfisher and rail. Ex situ care and biotechnology are their only hope. Besides, support from different sources is usually restricted to different purposes. Except in local education, research, and propagation programs in zoos, for example, municipal funds are usually unavailable to international species preservation. In such differentiation of source, competition for funds between preservation options can be diminished. After all, ex situ care and technology are used only after it is evident that conventional conservation efforts could fail.
BUYING TIME
Can technology be used to ensure continuing evolution? Both intensive management and habitat reduction reduce the chances of directional habitat-responsive evolution. And in small unmanaged populations, genetic drift is much more powerful than natural selection. But before worrying about whether species must continue to evolve to survive, please reflect upon the time scale of concern. The profoundly immediate problem is to save as many species as possible through the next 150 years.
It seems inevitable that most large land vertebrates and many plants eventually will survive only as wards of humans, scientifically managed or cared for, even reestablished, at some point. Because of an overall decline of diversity, those species that persist through the feeble efforts of science and technology will become proportionately more important. Saving 200 of 2,000 mammal species seems more important than saving 200 of 4,300. Furthermore, it is the larger forms, among animals if not plants, that will most likely profit from these intervention strategies;
creatures that are not only spiritual symbols for the ongoing promotion of conservation but evolutionary conservative; more irreplaceable than the merely irreplaceable. Preservation science and technology must become an active branch of conservation biology, because future habitat restoration, if any, will depend upon its progress.
Thus, technology is not a panacea for the disease of extinction. It is a palliative—a topical treatment with which to buy time, to preserve options for a few populations and species judged of special value. In the final analysis, it is no more important than the species it sustains, which would otherwise be lost forever, and no less.
REFERENCES
Conway, W. 1986. The practical difficulties and financial implications of endangered species breeding programmes. Int. Zoo Yearb. 24/25:210–219.
Myers, N. 1979. The Sinking Ark. Pergamon Press, Oxford. 307 pp.
Schonewald-Cox, C.M., S.Chambers, B.MacBryde, and W.L.Thomas, eds. 1983. Genetics and Conservation. Benjamin/Cummings, London. 722 pp.
Soulé, M.E., and B.A.Wilcox, eds. 1980. Conservation Biology: An Evolutionary-Ecological Perspective. Sinauer Associates, Sunderland, Mass. 395 pp.
CHAPTER 31
CONSERVATION OF BIOLOGICAL DIVERSITY IN BOTANICAL GARDENS
PETER S.ASHTON
Director, Arnold Arboretum, Harvard University, Cambridge, Massachusetts
Conservation is already, and very appropriately, recognized as being a major activity for botanical gardens in both their research and educational programs. In this field, arboreta and botanical gardens have a particular and important potential, which I discuss in this chapter.
In nature, plants frequently exist in small populations. Examples include many rare endemics, such as those of mountain peaks and many in the Mediterranean dry sclerophyll scrublands, especially in the Cape Province of South Africa and in Southwest Australia, and those of certain rain forests. Over the relatively short time we realistically have had to work as conservation managers, extremely small stands have been found to persist in nature. Higher plants, being sedentary, are often highly site-specific. This facilitates the development of logical plans for demarcating minimal areas for in situ conservation based on ecological knowledge and principles of island biogeography. On the whole, the most favorable sites are a few environmentally heterogeneous reserves of sufficient size to minimize edge effects (e.g., changes in species composition at the periphery caused by in- and out-migrations from adjacent unprotected lands). Ideally, these would be loosely connected by small stepping stones or corridors to allow for the exchange of genes (Diamond, 1975). Identification and immediate protection of sites of high conservation value must be our highest priority in the absence of even the grossest information upon which to base plans, including basic inventory as well as distributional and ecological data on many of the richest biota. This underlines the vital necessity of increasing inventory and ecological information as a prerequisite to developing any logical plan for conservation. In practice, of course, the luxury of regional planning often does not exist. The conservationist only succeeds in
raising awareness when the plant is reduced to endangerment in one or a few isolated localities or, at best, is offered a patchwork of lands for which the farmer and the planner have failed to find other uses. As development proceeds and natural habitats become increasingly fragmented, extinction accelerates (Wilcox and Murphy, 1985). The most endangered floras are those of the arable lands; the current distribution of preserves takes little account of this.
Even when the luxury of time for planning does exist and centers of species richness and endemism can be identified and conserved, many locally endemic plant species refuse to follow the rules and occur in isolated areas where, overall, conservation priorities are low. Even under ideal circumstances, though, decreases in and fragmentation of natural areas is certain to lead, as predicted by the theory of island biogeography, to substantial increases in extinction rates, though it is uncertain whether these rates would be on the scale calculated for large animals (e.g., Schonewald-Cox, 1983; Soulé et al., 1979; Simberloff and Abele, 1984). This is therefore a case for some form of selective program of ex situ conservation, that is, conservation through cultivation.
Too little is known of the ecology of any plant species, let alone those that are rare and endangered, to consider the transfer of species from one natural community to another. But in addition to their immobility, plants have many practical advantages over animals for ex situ conservation.
Plants are generally easy to propagate asexually through a variety of methods, including division of the rootstock, cuttings, and tissue culture. For conservation of heritable character traits, experience with a variety of crop plants has shown that gene cloning is a realistic possibility. Propagants, vegetative or seed, can be collected with minimal disturbance to the wild population and are cheap and easy to transplant.
In comparison to animals, management of ex situ plant populations is relatively simple and inexpensive. Plants do not require caging, and in practice, genotypes can often be maintained for long periods, though probably not permanently, through propagation or forced rejuvenation (e.g., see Rackham, 1976, on the effect of pollarding on trees recorded over half a millennium). Furthermore, plants need less constant care than animals. The bisexuality of the majority of higher plants implies broadly that minimum population sizes for maintenance of heterosis can be half those of populations comprising two sexes. In addition, their modular construction allows considerable phenotypic plasticity. Their habitat requirements can generally be reasonably accommodated ex situ provided that competition is excluded. They do not manifest demanding behavioral traits and are rarely dangerous to humans. Added to these attributes, plants are both attractive and unobtrusive. They are more often scented than smelly, and they are generally perceived by humanity as benign. In short, they are welcomed adornments to the human environment.
Is is certainly true, then, that the rapidly increasing demand for ex situ conservation, occasioned by the inexorable destruction of natural habitats, presents botanical gardens with both a challenge and an opportunity, the likes of which have not arisen for more than a century.
METHODS OF EX SITU CONSERVATION
In comparison to conservation of animals, flowering plants have a disadvantage in the extraordinary diversity of their reproductive systems and, notably, their sexual differentiation. These differences are considered to be major determinants of genetic patterns within populations. The kind of reproductive system influences the minimal viable population sizes needed for conservation (Wilcox and Murphy, 1985) both in situ and ex situ and controlled pollination strategies for stock regeneration. In brief, self-pollinating and vegetatively reproducing species will vary more genetically between than within breeding populations (Allard, 1960). But among outbreeders, especially dioecious species, the reverse will hold true, though differences in gene frequency between reproductively isolated populations will increase over time. In self-pollinating species, representative samples of a wide range of breeding populations should be sampled, but individual samples need be represented by comparatively few individuals. For outbreeders, individual populations should be well sampled, but fewer representatives of different populations will generally be necessary.
Methods of ex situ conservation now available can conveniently be classified according to the part of the plant that is conserved—the whole organism, seed, tissues, or genetic material in culture. When kept in ex situ living collections, whole plants have educational value and can be displayed, and for species that take a long time to reach reproductive maturity, mature specimens on hand are advantageous for research. A relative disadvantage of whole plants is their higher maintenance costs in comparison to other means of ex situ plant conservation. For example, there are high requirements for space, especially for trees. Conversely, annuals require frequent, controlled pollination and reestablishment, unless they are inbreeders or methods of vegetative propagation are available. Whole plants conserved in gardens and plantations often readily hybridize with related taxa. Controlled pollination is therefore obligatory for regeneration from seed among outbreeders.
When grown in single-species plantations, whole plants are more susceptible to communicable diseases than when scattered, as in nature, in a matrix of other species. On the other hand, plants will as a rule prosper outside their natural range in the absence of coevolved pathogens. This explains why crop plants, particularly long-lived tropical species such as Hevea rubber, have flourished best in plantations outside their region of origin. Clearly, this biological reality causes political problems, which must be faced and overcome if ex situ conservation is to succeed and its subjects are to be exploited to benefit humans. It also identifies a conflict between the needs for conservation per se and the need for display or education when demonstration of indigenous flora has high priority. In contrast to masses in plantations, however, specimen plants will not incur this danger.
With present technology, the preferred method of ex situ conservation is through storage as seed. The principal advantage of seed banks is their economy of space and the larger sample sizes that then become possible and, in countries with high labor costs, their low labor demands. The principal practical disadvantage is their
reliance on a dependable power supply, the need for meticulous monitoring of germinability over time, and the need for periodic regeneration under conditions that minimize selection among the residual seed stock. Added to this is the fact that research in seed storage has overwhelmingly been done with crop plants. Many of these are plants of early succession, a habitat in which many species possess seed dormancy. Many plants in nature will more often and even predominantly possess seeds that lack dormancy and will prove recalcitrant to induction of dormancy with current methods. It will not pass unnoticed that many long-lived species, often large at maturity, also predominate in these same habitats.
Tissue cultures, especially of meristems, can maintain genotypes unaltered over long periods (Henshaw, 1975; Wilkins and Dodds, 1983). They also provide an economic means of suspending, at least temporarily, changes in gene frequency in cultivated populations (in this case represented by cells or tissue in culture). Mutations will continue to occur, particularly in cell and callus cultures, but are less frequent than in whole plants.
Each taxon has it own requirements for successful establishment and regeneration in tissue culture. At present, the basic mechanisms are poorly understood, so that successful techniques must be developed through tedious trial and error. Nevertheless, experience is accumulating. It now seems possible that established techniques are only applicable to half the taxa of higher plants at most, although response can be predicted from evolutionary relationships (Einset, 1985).
Because tissue cultures using current media generally appear to be chromosomally unstable over long periods, they too must be viewed as evolving populations, albeit in a microenvironment in vitro. Nonetheless, tissue cultures provide an invaluable alternative for conservation of multiple lines of taxa not easily conserved ex situ on this scale by other measures.
Currently, then, all these methods of ex situ preservation of live plant material require periodic regeneration and sexual reproduction of the stock. The latter presupposes knowledge of the breeding system and pattern of genetic variability of the species concerned.
Although cryogenic storage of seed may in the future provide a solution for long-term preservation of natural patterns of genetic variation within population samples in vitro, DNA libraries are probably the most stable form in which genetic information can be stored. Gene libraries provide a means to conserve DNA sequences, even whole genomes, for research. The genetic material thus conserved can be introduced to other extant genotypes, but whole individuals cannot be regenerated independently. Although gene libraries are subject to the same problems of population genetics as other methods of ex situ conservation, at present they can more easily be stored indefinitely. This can be done by a variety of techniques available for reducing metabolic activity or, if desired, rendering them fully inert.
Some may consider these topics to be remote from the mission of a botanical garden or arboretum. Where, though, if not in a botanical garden, are these issues related to education and to research on and management of plant diversity to be addressed? In fact, with adequate funding botanical gardens and arboreta are well suited to take on this responsibility. After all, botanical gardens are museums without walls generally occupying, if not public space, then prime space from which
the public can hardly be excluded. They are magnets for the curious as well as for seekers of tranquility. Most important, their mission is the cultivation and study of the diversity of plants. Furnished as a rule with a reference herbarium and a library as well as living plant material, they alone have the breadth of facilities required for the research needed to back up and refine the management of conserved plant populations in the wild.
THE GENETIC CONSEQUENCES OF EX SITU CONSERVATION
Although for practical reasons, including nomenclatural custom, we fall into the habit of giving priority to species conservation, the genetic nature of the biological species remains elusive for many kinds of organisms, including bacteria and other microorganisms and a large percentage of plant groups. The genetic mechanisms underlying the maintenance, increase, and loss of biological diversity proceed at the scale of the breeding population. For conservation planning, the unit of the breeding population is therefore clearly more desirable on biological grounds.
For very rare and local endemic species, this should not prove as serious a problem as among widespread and variable species, because, ipso facto, they are generally confined to one or a few populations and restricted habitat ranges in which out-breeding opportunities are already severely limited. Where widespread, variable species are to be conserved, a strong case can be made for preserving infraspecific variation (Antonovics, in press), particularly when eventual reintroduction into the wild is visualized. The restriction of useful heritable attributes to specific populations and races is well known among progenitors and landraces of crop plants and has been demonstrated in unique populations of wild taxa (Bradshaw, 1984). Wherever possible, it is important to distinguish genetic from environmental effects by observation of variation among plants grown in garden plots (Briggs and Walters, 1984).
It is generally agreed that most genes do not vary at the population, or even species, level. Most variable alleles are sufficiently abundant to be adequately sampled and conserved without danger of chance extinction through random drift in artificial populations as small as 50 (Marshall and Brown, 1975) to 100 (Frankel and Soulé, 1981) randomly selected individuals. However, the importance of rare, variable genes in the long-term survival of species is unknown. It can be expected that a few genes rare in one population, or ecotype, will be common in another. Selection varies quantitatively and qualitatively within the life of a population. Some of these changes will be unidirectional and are the stuff of evolution. Alleles will therefore vary in fitness over time scales exceeding the life cycle of the plant in conservation. Samples for conservation in gardens need to be taken from a range of populations, therefore, if genetic variability is to be adequately represented.
Yet more important, the characteristics by which we distinguish races and even some individual populations (Clausen and Hiesey, 1958) may often represent differences in gene complexes rather than in individual gene loci. Such variation between populations in the representation of gene complexes can occur over re-
markably short distances (Antonovics and Bradshaw, 1970; Hamrick, 1983), although they can be remarkably constant within populations (Clay and Antonovics, 1985; Morishima et al., 1984). We know little about the heritability of such complexes and nothing concerning their reconstruction.
Thus, a serious problem is that natural selection cannot be simulated ex situ. If the artificial population is established from seed, the progeny have been released from natural selection from the start. Whether ex situ conservation takes the form of plants, seed, or vegetative propagules, including tissue culture, genetic rejuvenation through controlled cross-pollination of regenerated plants is periodically obligatory with present technology. New unselected gene combinations are inevitably introduced every time. However much care is taken in seed collection by controlled pollinations, natural selection cannot be simulated, particularly if it is mediated by interspecific competition. Some artificial selection is therefore unavoidable, being imposed through the methods adopted for pollination, germination, and continuing storage ex situ.
Ex situ conservation of small samples therefore can be expected to lead inevitably to unpredictable genetic change. Hybridization between different population samples grown for more than one generation in isolation, and more particularly between samples from different populations, will increase the rate of these changes. The proportion of fit genotypes in progeny intended for reintroduction into the wild can thereby be greatly reduced, especially if the original wild donor populations are already small and in decline, and hence already suffering an accumulation of deleterious gene combinations.
For these reasons, we still lack the knowledge to establish genetically representative population samples in cultivation. Nor have there been more than a handful of experimental reintroductions into the wild from cultivated cultures, and none that I know of plants derived from stock that has been in cultivation for a long time.
For all practical intents, therefore, ex situ conservation of species leads inexorably, and perhaps irrevocably, to domestication. But ex situ samples of breeding populations can effectively serve as means to conserve unique or important characteristics rather than species as such. This has been recognized by forest geneticists whose ex situ conservation programs for “provenances” represent just this (Zobel, 1978). Among crop plants, unique genes and gene combinations, which may be as important to the survival of endangered species as to the protection of domestic crops, can presently be recovered if lost from a culture only by resorting to wild populations. Ex situ conservation should also serve the reverse function: reintroduction of desirable alleles and gene combinations that become lost in wild populations. When extinction in the wild becomes a reality, the means to control artificial selection is greatly reduced, and the means to reintroduce alleles lost in culture is eliminated. Ex situ conservation must often be deemed preferable to loss nevertheless (Raven, 1976). But it is imperative for long-term conservation that managers of ex situ population samples define, as carefully as information allows, the characteristics they are intending to conserve: all alleles in the subsample or only some, and, if some, then which? Examples might be the diagnostic charac-
teristics of a taxon or utilitarian attributes. Once a clear set of priorities has been defined, the role of what in effect will become a form of gene library will be considerable.
The role of interspecific competition in speciation has never been rigorously demonstrated in animal, let alone plant, populations. In plant populations it remains unclear whether species-specific natural selection is of common occurrence. Shugart and West (1981) indicated through mathematical modeling that the species composition of the southern U.S. hardwood forest could arise purely as a consequence of the hazards of island biogeography—the balance of immigration and extinction over time. If correct, their view would certainly make the task of ex situ conservation a whole lot easier. But many examples of mutual interdependence do exist. Oaks cannot be conserved without the rich symbiotic fungal flora on their roots on which they depend and which differs quantitatively, and sometimes qualitatively, between species. In some cases, such plants and their symbionts will prove exceptionally difficult to conserve in perpetuity ex situ. Furthermore, it is these keystone mutualists and mobile links whose minimum viable population areas are being used as a principal criterion for assessing the minimum area of in situ preserves (Wilcox and Murphy, 1985).
SOME PRACTICAL ISSUES
It takes an outbreeding species at least 200 generations to diversify to the point of speciation (Stebbins, 1950). Some outbreeding forest trees do not start flowering for 50 years, even when in cultivation (Ng, 1966). We need to think in evolutionary time scales if it is really our intention to conserve species as opposed to alleles or gene complexes ex situ (Frankel and Soulé, 1981). It is questionable whether seed dormancy can be induced for comparable periods in many species. What is the likelihood of a major power cut (perhaps a war?) during such a period? Could not most endangered species be conserved in situ more cheaply by careful planning and management? How is the cost of in situ conservation to be estimated?
The record to date for ex situ culture does not give grounds for optimism. As Raven (1981, p. 56) stated so eloquently:
Unfortunately, such collections are often dismantled or simply deteriorate after the specialists who built them up are no longer active at the respective institutions…. Although they are often of very great value internationally, they may if they are not actively utilized come to be viewed as a drain upon the limited resources of the institution where they are housed. Even when financial considerations are not limiting, it is difficult to provide for such collections the meticulous and sustained care that is essential for their survival without the attention of a specialist who is deeply concerned with them.
Similarly, Peeters and Williams (1984) estimated that of the 2 million accessions of plant germplasm in seed banks worldwide, 65% lack even basic data on source; 80% lack data on useful characteristics, including methods of propagation; and 95% lack any evaluation data such as responses to germinability tests. There are extensive data only on 1%. It goes without saying that a substantial proportion of the accessions not tested for germinability may be dead.
WHAT THEN IS THE SPECIFIC ROLE FOR BOTANICAL GARDENS IN CONSERVATION?
Ex situ conservation is a refuge of last resort: a high-risk refuge, perhaps of no escape. The immediate role of botanical gardens in the ex situ culture of rare and endangered species lies in research and education rather than in conservation per se. This role is absolutely vital if we are to have knowledge about plant populations on the edge of extinction that provides a sufficient basis for their management.
This point is brought home most poignantly by consideration of the humid tropics, where species diversity is at its greatest. There, long-lived perennials are in the majority, making cultivation easier, but few plants possess seed dormancy and many are of formidable size. Knowledge of the cultural requirements of the minority is nonexistent. It is obvious that ex situ conservation cannot be an end in itself under such circumstances.
Yet the accessibility of the plants in cultivation presents all manner of research opportunities not possible with remote and dispersed wild populations as well as opportunities for education and for increasing public awareness that would not otherwise exist. The necessity to focus on strictest priorities leads to the identification of very specific roles in conservation for tropical botanical gardens. One of particular current importance is conservation of the myriad local landraces of tropical cultivated plants from cereals and vegetables, in many of which seed dormancy can be induced, to fruit trees, which must be grown in plantations. The National Botanical Garden of Indonesia is already a pioneer in many aspects of tropical conservation research based on such plantings. Landraces do not exist in nature: their habitat is therefore ephemeral over evolutionary time anyway and is therefore rapidly disappearing as world agriculture becomes increasingly market-oriented and industrialized.
The greatest use is made of gene banks by workers at the bank site itself. In 1981, for example, 4,376 samples were distributed to outside users worldwide from the 57,027 seed accessions at the International Rice Research Institute gene bank, whereas 29,056 samples were requested by Institute staff (IRRI, 1983). This further underlines the unique opportunities that botanical gardens have for promoting interdisciplinary research and instruction. There certainly will be a rapid increase in research on, and public understanding and support of, rare and endangered species once population samples are conserved ex situ in botanical gardens that have active programs of education and research.
The basic data on methods of seed storage can be best accumulated where there is ready access to captive populations. Many other aspects of reproductive biology will be advanced most rapidly where there are botanical gardens with captive populations as well as good laboratory and library facilities and research staff on site. All aspects of research that can be advanced through comparative study of closely related species, generally of different provenances, are aided when plants are grown side by side and at one accessible location. Even when the work must be carried out with populations in situ, as in demographic and population genetic research, much time and expense are saved if captive populations are easily accessible for use in the refinement of field and laboratory techniques prior to im-
plementation (Ashton, 1984). The standard procedures for diagnosing the genetic component in phenotypic variation within and between populations, through culture of subsample populations under uniform garden conditions, would be an essential adjunct of ex situ cultivation methodology and one for which the botanical garden is best equipped. For all these purposes, the long-term maintenance of genetic integrity of the captive population, and indeed the long-term sustenance of the captive populations, are lesser priorities. The aim of the research is always to improve the techniques for managing wild populations in situ. Again, ex situ conservation is an ancillary to in situ conservation, not an alternative. Of course, the fact remains that plant species are becoming extinct in the wild, and some can and should be conserved in botanical gardens.
Purpose must then be strictly defined as a prerequisite to management planning. The result will likely be a program for conservation of chosen heritable attributes rather than of species or even demes.
Fundamental to the role of botanical gardens in conservation research is the development of meticulous, consistent, accessible, and hence computer-based record systems. Not only are details of source and information on the ecology of the wild population essential if this research is to be successfully applied, but plant material ex situ must be carefully inventoried together with comprehensive records of all treatments and the ensuing responses (Frankel and Soulé, 1981). Results of previous research must be retained with the records of ex situ living accessions, together with methods adopted and, preferably, copies of unprocessed results for evaluation and reanalysis by later workers intending to build on them.
REFERENCES
Allard, R.W. 1960. Principles of Plant Breeding. Wiley, New York. 485 pp.
Antonovics, J. In press. Genetically based measures of uniqueness. In G.Orians, W.Kunin, G. Brown, and J.Swierzbienski, eds. Proceedings of the Lake Wilderness Conference on Genetic Resources. University of Washington, Seattle, Wash.
Antonovics, J., and A.D.Bradshaw. 1970. Evolution in closely adjacent plant populations. VIII. Climal patterns at a mine boundary. Heredity 25:349–362.
Ashton, P.S. 1984. Botanic gardens and experimental grounds. Pp. 39–48 in V.H.Heywood and D.M.Moore, eds. Current Concepts in Plant Taxonomy. Academic Press, London.
Bradshaw, A.D. 1984. The ecological significance of genetic variation between populations. Pp. 213–228 in R.Dirzo and J.Sarukhan, eds. Perspectives in Plant Population Ecology. Sinauer Press, Sunderland, Mass.
Briggs, D., and S.M.Walters. 1984. Plant Variation and Evolution. Second edition. Cambridge University Press, New York. 412 pp.
Clausen, J., and W.M.Hiesey. 1958. Experimental Studies on the Nature of Species. IV. Genetic Structure of Ecological Races. Publication No. 615. Carnegie Institution of Washington, Washington, D.C.
Clay, K., and J.Antonovics. 1985. Quantitative variation of progeny from chasmogamous and cleistogamous flavors in the grass Danthonia specata. Evolution 39:335–368.
Diamond, J.M. 1975. The island dilemma: Lessons of modern biogeographic studies for the design of natural reserves. Bio. Conserv. 7:129–146.
Einset, J.W. 1985. Chemicals that regulate plants. Arnoldia 45:28–34.
Frankel, O.H., and M.E.Soulé. 1981. Conservation and Evolution. Cambridge University Press, New York. 327 pp.
Hamrick, J.L. 1983. The distribution of genetic variation within and among plant populations. Pp. 335–348 in C.M Schonewald-Cox, S.M.Chambers, B.MacBryde, and W.L.Thomas, eds. Genetics
and Conservation. A Reference for Managing Wild Animal and Plant Populations. Benjamin/Cummings, Menlo Park, Calif.
Henshaw, G.G. 1975. Technical aspects of tissue culture storage for genetic conservation. Pp. 349–357 in O.H.Frankel and J.G.Hawkes, eds. Crop Genetic Resources for Today and Tomorrow. International Biological Programme 2. Cambridge University Press, New York.
IRRI (International Rice Research Institute). 1983. Annual Report for 1981. Los Banos, Philippines.
Marshall, D.R., and A.H.D.Brown. 1975. Optimum sampling strategies in genetic conservation. Pp. 53–80 in O.H.Frankel and J.G.Hawkes, eds. Crop Genetic Resources for Today and Tomorrow. International Biological Programme 2. Cambridge University Press, New York.
Morishima, H., Y.Sano, and H.J.Oka. 1984. Differentiation of perennial and annual types due to habitat conditions in the wild rice Oryza perennis. Plant Syst. Evol. 144:119–135.
Ng, F.S.P. 1966. Age at first flowing in dipterocarps. Malay. Forester 29:290–295.
Paabo, S. 1985. Molecular cloning of ancient Egyptian mummy DNA. Nature 314:644–645.
Peeters, J.P., and J.T.Williams. 1984. Towards better use of gene-banks with special reference to information. Plant Genet. Resour. News (FAO) 60:22–32.
Rackham, O. 1976. Trees and Woodland in the British Landscape. Dent, London. 204 pp.
Raven, P.H. 1976. Ethics and attitudes. Pp. 155–179 in J.B.Simmons, R.I.Beyer, P.E.Brandham, G.L.Lucas, and V.T.H.Parry, eds. Conservation of Threatened Plants. Plenum, New York.
Raven, P.H. 1981. Research in botanical gardens. Bot. Jahrb. 102:52–72.
Schonewald-Cox, C.M. 1983. Conclusions: Guidelines to management: A beginning attempt. Pp. 414–445 in C.M.Schonewald-Cox, S.M.Chambers, B.MacBryde, and W.L.Thomas, eds. Genetics and Conservation. A Reference for Managing Wild Animal and Plant Populations. Benjamin/Cummings, Menlo Park, Calif.
Senner, J.W. 1980. Inbreeding, depression and the survival of 300 populations. Pp. 209–224 in M.E.Soulé and B.A.Wilcox, eds. Conservation Biology: Evolutionary-Ecological Perspective. Sinauer Associates, Sunderland, Mass.
Shugart, H.H., Jr., and D.C.West. 1981. Long-term dynamics of forest ecosystems. Amer. Sci. 69:647–652.
Simberloff, G., and L.G.Abele. 1984. Conservation and obfuscation: Subdivision of reserves. Oikos 42:399–401.
Soulé, M.E., B.A.Wilcox, and C.Holtby. 1979. Benign neglect: A model of faunal collapse in the game reserves of East Africa. Biol. Conserv. 15:259–272.
Stebbins, G.L. 1950. Variation and Evolution in Plants. Columbia University Press, New York. 643 pp.
Wilcox, B.A., and D.D.Murphy. 1985. Conservation strategy: The effects of fragmentation on extinction. Amer. Nat. 125(6):879–887.
Wilkins, C.P., and J.H.Dodds. 1983. The application of tissue culture techniques to plant genetic conservation. Sci. Prog. 68:259–284.
Zobel, B. 1978. Gene conservation—as viewed by a forest tree breeder. Forest Ecol. Manage. 1:339–344.
CHAPTER 32
USING SCIENCE AND TECHNOLOGY TO REESTABLISH SPECIES LOST IN NATURE
TOM J.CADE
Director of Raptor Research, Laboratory of Ornithology, Cornell University, Ithaca, New York
Science and technology are hardly new to species conservation. Marco Polo related how the great Kublai Khan had an appreciation for their use to increase game-bird populations on his hawking preserves 700 years ago (Komroff, 1926), and the origin of wildlife management goes even further back in human culture to the early empirical practices of aboriginal peoples. The Kutchin Indians were still burning climax spruce forest to create willow browse for moose in the Yukon River Valley when I first visited the region in 1951, and primitive hunters practiced similar manipulations of habitat to favor big game species in many parts of the world.
In 1933, Aldo Leopold wrote the following lines in the preface to his classic textbook Game Management: “game can be restored by the creative use of the same tools which have heretofore destroyed it—axe, plow, cow, fire, and gun. A favorable alignment of these forces sometimes came about in pioneer days by accident. The result was a temporary wealth of game far greater than the red man ever saw. Management is their purposeful and continuing alignment” (Leopold, 1933, p. vii).
Some of the authors in this section of the book write about the development of highly advanced techniques involving cryogenic processes, embryo transplants, artificial insemination, and other sophisticated clinical procedures. Although these new techniques are certainly needed, we have not yet made the most of many of the older technologies that can be applied to species conservation, and I want to emphasize the creative use of some old methods as important tools for conservation.
Two that I have used are the captive propagation of wild species and the reintroduction of individuals produced in captivity into vacant habitats. I work with
the diurnal birds of prey—some 290 species of condors, vultures, eagles, hawks, and falcons, but especially with falcons, principally the peregrine falcon (Falco peregrinus). Some of these raptors, such as the California condor (Gymnogyps californianus) and the Mauritius kestrel (Falco punctatus), are among the most endangered species in the world, and all seem to have attracted increasing human interest in recent years.
Given the long history of human involvement with birds of prey in the sport of falconry and as tribal and national totems, it is curious that the propagation of these species in captivity is a quite recent activity. The first peregrine falcon known to be raised from captive parents was produced as recently as 1942, and even as late as 1965 only about 23 species of diurnal raptors had been successfully bred in captivity, mostly on a casual or accidental basis (Cade, 1986a). In the early 1970s it was still widely believed that systematic large-scale propagation of raptors was an impossible feat, owing, I suppose, to the wild and fierce nature of such birds and to their apparent need for vast expanses of air space in which to perform their spectacular aerial courtship displays prior to mating and nesting.
The situation has changed markedly in the last 15 years. When it became evident in the late 1960s that populations of many species of raptors in north temperate regions had suffered major declines as a result of exposure to DDT and related pesticides or to other forms of environmental degradation, a nascent interest developed, especially among falconers, in perfecting techniques of captive breeding for some of these species. Of particular interest was the peregrine falcon, whose numbers had been severely reduced by pesticides over most of Europe and North America. This species has always been highly esteemed by naturalists and falconers alike for its near perfection as a flying machine.
In a summary of the worldwide effort to propagate raptors for the Fourth World Conference on Breeding Endangered Species in Captivity held in 1984, I came up with a list of 83 species that had reproduced in captivity—most of them since 1965 (Cade, 1986a). At least three species now need to be added to the list—the pondicherry vulture (Sarcogyps calvus), the white-tailed hawk (Buteo albicaudatus), and the crested eagle (Morphnus guianensis) of the Neotropics (ISIS, Captive Population Information, 31 December 1985). At present, more than 25% of all falconiform species have been bred in captivity. Some species have produced thousands of progeny; certainly the number of peregrine falcons raised in captivity exceeds 5,000 worldwide, and the American kestrel (Falco sparverius) is not far behind. At least 12 species have produced more than 100 progeny in captivity since 1975. It is safe to conclude that most if not all diurnal birds of prey can be bred in captivity given sufficient knowledge of their needs and sufficient resources to carry out the work.
What is the explanation for these rather sudden breakthroughs in the breeding of birds of prey in captivity? First, raptor breeders are true zealots with a single-mindedness of purpose to succeed against all odds and at any personal sacrifice. Although they come from all walks of life, most have in common the fact that they are falconers steeped in 3,000 years of accumulated knowledge about handling and training hawks and falcons and possess a special rapport with their birds that gives them an intuitive feel for what is needed in any particular situation.
A second part of the explanation has to do with the rapid and free exchange of information among raptor breeders throughout the world (Cade, 1986a). This exchange has followed the very best tradition of open scientific inquiry, unlike the secretive activities of some aviculturists.
Finally, successful raptor propagation owes much to the application of basic scientific information on avian reproductive physiology and breeding behavior and ecology. One quick example makes the point.
One of the chief bottlenecks hindering the mass production of raptors in captivity has been the frequent failure to achieve fertilization of eggs because of incompatibilities between mates. Thus, breeders must often resort to artificial insemination of egg-laying females. Obtaining usable quantities of semen from male raptors by the standard poultry method of physical massage and forced ejaculation is difficult and yields only small volumes of material over a short time (Weaver and Cade, 1983).
Since the work of the German ethologists Oskar Heinroth and Konrad Lorenz, it has been known that under certain conditions birds reared in isolation from members of their own species will become sexually imprinted on their human keepers and will actually attempt to mate with them. A fascinating body of scientific literature has developed on this subject (Brown, 1975; Immelmann, 1972; Lorenz, 1937). Raptor breeders have capitalized on this information and on their own experiences with hand-reared hawks and falcons raised in isolation to develop human-imprinted raptors into highly effective semen donors for artificial insemination. The birds are usually trained to copulate and ejaculate on a special hat worn by the human companion. Such birds produce copious volumes of high-quality semen for 2 to 3 months and have been great assets in increasing the percentage of fertile eggs laid in raptor breeding projects (Weaver and Cade, 1983).
Other examples of how basic scientific research has provided information useful to propagators involve incubation, photoperiod, hormone physiology, and psychobehavioral factors such as courtship displays, nest-site stimulation, and other factors, which Lack (1937) referred to as “psychological” in nature (Cade, 1980, 1986b).
REINTRODUCTIONS
There have been more than 1,670 attempts to establish avian species in outdoor environments, according to the excellent summary by Long (1981). At least 425 (25%) of these have been successful in establishing breeding populations. For example, in attempts to establish 119 species in North America up to 1980, success was achieved for 39 (37%) species. Comparable figures for Europe are 69 attempted and 27 (31%) established; for the Hawaiian Islands, 162 attempted and 45 (28%) established; for New Zealand, 133 and 38 (29%); and for Australia, 96 and 32 (33%).
Most of these have been introductions of exotic species into a new range. Some of these exotics have caused ecological or economic problems, but others appear to have filled vacant niches without seriously disrupting other species in the ecosystem (Long, 1981). There is much to be learned from careful study of these
exotic introductions—both the successful and failed examples. We need to develop enough ecological foresight to predict and to effect benign exotic introduction as an additional technique of conservation. For example, the Mauritius kestrel badly needs a new home, and the neighboring Indian Ocean island of Runion could well be the place. This possibility needs study.
Reintroduction—the restoration of a species to its native habitat—has only recently been attempted, and there are fewer successful examples. In North America, one of the earliest successes was the restocking of some western ranges with American bison (Bison bison) derived from captive-bred stock held at the Bronx Zoo.
Much public attention has focused lately on the possibility of successful captive propagation and eventual reintroduction of the California condor—the most endangered bird in North America. It is perhaps less widely appreciated by the general public that successful reintroductions have already been achieved with several birds of prey. Since the 1950s efforts have been under way in the Federal Republic of Germany and in Sweden to re introduce the eagle owl (Bubo bubo) into vacant range, and new breeding populations have been established in both countries through the release of captive-bred owls. Breeding populations of the white-tailed sea eagle (Haliaeetus albicilla) and goshawk (Accipiter gentilis) have been reestablished in Great Britain, and the griffon vulture (Gyps fulvus) has been restored to the Massif Central in France (Terrasse, 1985). Also as a result of reintroduction, several pairs of bald eagles (Haliaeetus leucocephalus) and, more recently, ospreys (Pandion haliaetus) are now nesting in formerly vacant haunts in the United States. Through the release of young birds produced mostly by falconers, a small nesting population of Harris’ hawks (Parabuteo unicinctus) has been restored to the lower Colorado River valley of California and Arizona, where the species had disappeared around 1970, primarily because of habitat destruction. (See Cade, 1986b, for details and documentation.)
The best-known case is that of the peregrine falcon. The most widely distributed, naturally occurring avian species in the world, the peregrine suffered drastic reductions in numbers in both Europe and North America in the 1950s and 1960s, primarily because of the effects of organochlorine pesticides such as DDT and dieldrin. Since then there have been major international efforts to restore this species, in part by restricting the use of persistent organochlorine pesticides and by captive breeding and reintroduction. Successes have been achieved from both approaches.
In regions where some breeding pairs were able to persist, the falcon populations recovered dramatically on their own once restrictions were placed on use of these chemicals (Cade et al., in press). Great Britain is the best studied region where natural recovery has occurred (Ratcliffe, 1984). By 1963, the nesting population there had been reduced to 44% of its estimated pre-World War II size: there were some 350 surviving pairs. Once the use of dieldrin and DDT decreased in the latter part of the 1960s, the population began to increase, and today there are more than 1,000 occupied breeding territories in the British Isles. The species is, in fact, more numerous there now than at any time in this century.
In other regions where the species was more severely reduced or entirely extirpated as a breeding bird, restocking or reintroduction through the release of captive-
reared falcons has been the main method for recovery. As a result of this work, small breeding populations have now been reestablished in the Federal Republic of Germany, Sweden, the eastern United States, the upper Mississippi River region in Wisconsin and Minnesota, the Rocky Mountain states, California, and several places in Canada (Cade, 1986b; Cade et al., in press). Three methods of release have been used: hacking, an old technique borrowed from falconry by which young are placed outdoors and cared for until they can fend for themselves; fostering, by placing captive-reared young in the nests of wild peregrines; and cross-fostering, by placing them in the nests of other species. The best results have been achieved with hacking and fostering (Cade et al., in press).
The eastern U.S. recovery program can serve as an example of the effort required for one of these regional reintroductions. The original nesting population of peregrines in the United States east of the Mississippi River in 1942 was estimated to be around 350 pairs on average each year (Hickey, 1942); some 250 eyries are actually known to have existed. By the early 1960s, this breeding population had completely disappeared, and no peregrines are known to have reproduced again in the eastern part of the country until 1980, when the first members of the reintroduced population fledged their young.
The Peregrine Fund began its captive breeding program at Cornell University in 1970. By 1974, there were sufficient numbers of young being raised to begin experimental releases. The following year, with substantial support from the U.S. Fish and Wildlife Service, we were able to release 16 young by hacking, and the number increased each year until we were putting out an average of 100 birds annually from 1981 onward (Barclay and Cade, 1983). At the end of the 1986 season we had released more than 850 captive-produced peregrines in 13 eastern states and the District of Columbia.
Falcons that survived their first winter immediately began establishing spring and summer territories, often at or near locales where they had been released. The first pairs formed in 1977 and 1978; the first eggs were laid at a nesting tower in New Jersey in 1979, but they were depredated by crows. In 1980, three pairs bred and raised young, and the nesting population has been increasing each year since. This past season, 43 pairs were known to be occupying territories from North Carolina to Montreal, Quebec; 30 pairs actually laid eggs, and 24 of them produced 52 young. There have been 83 nestings since 1979, and 71 of them produced 180 young.
The reintroduced breeding population has been doubling in size about every 2 years (Figure 32–1). If that rate of increase can be continued, by 1991 we will have achieved the official recovery goal of 175 nesting pairs established by the U.S. Fish and Wildlife Service in its recovery plan for the eastern United States. The complete restoration of the peregrine falcon in North America is now a predictable result of continuing to apply the technology developed for its recovery.
CAPTIVE BREEDING IN WILDLIFE MANAGEMENT
The rearing of captive animals and their use for restocking and reintroduction have not been favored techniques of modern wildlife management. Leopold (1933)
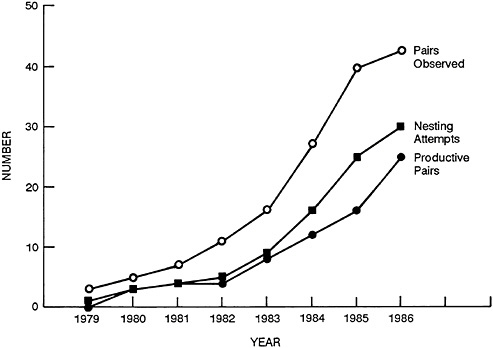
FIGURE 32–1 Growth of reestablished eastern peregrine population.
dismissed these subjects with 5 pages on “artificial propagation” and 7 pages on “transplantation” in a 481-page textbook. He stressed the cost of such artificial techniques and their aesthetic limitations. Leopold began what can be called the “ecological tradition” in wildlife management with its emphasis on habitat—the preservation and manipulation of all the environmental factors that are necessary to support wildlife populations as being more important than direct manipulation of the populations themselves. This approach has continued to the present day and certainly is the best policy wherever it can be pursued.
There have been good reasons for skepticism about propagation and release. Many captive stocks have not been reared in ways that are conducive to their survival after release in the wild (Cade, 1980). There have been many costly failures in attempts to restock or transplant animals into outdoor habitats and, particularly, to introduce exotic game birds by mass releases with little or no conditioning of the birds for their new environment (Bohl and Bump, 1970). One example is the repeated failure of many state game agencies to establish Japanese quail (Coturnix coturnix) in the United States. Hundreds of thousands of these birds were released—more than 350,000 from 1956 through 1958 alone—but no breeding populations ever became established (Long, 1981).
There are two main reasons for such failures. First is the use of genetic stocks or species that are poorly or not at all adapted to the environments into which they are introduced. Second is the lack of understanding of the behavioral, physiological, and ecological processes required for successful establishment (Cade,
1986b). Given the poor record of success with many species, especially when captive-produced individuals have been used, the recent reintroductions of raptors have been more successful than many biologists would have predicted.
These results with raptors and some other species in the last 10 years show that given enough biological knowledge about a species and enough time, effort, and money to apply that knowledge, reintroduction can be made to work. It is, however, an expensive and labor-intensive procedure, especially for captive-produced birds. It requires a tremendous amount of cooperation among numerous private individuals, government agencies, conservation organizations, corporations, and so on. Because so many different interests are usually involved, especially with an endangered species, it almost always becomes highly politicized.
The eastern peregrine recovery program is a case in point. Since 1970, the Peregrine Fund at Cornell (Figure 32–2) has spent about $2.8 million to propagate and release peregrine falcons in the eastern United States, but this figure does not include the expenses of cooperating agencies. The total cost has probably been between $3 and $3.5 million, perhaps more. More than 325 people—mostly temporary summertime helpers—have worked directly for The Peregrine Fund, not to mention dozens of federal and state agency personnel who have also helped. Although the U.S. Fish and Wildlife Service, the federal agency that administers the Endangered Species Act, is nominally in charge of the program, The Peregrine Fund has also worked closely with the U.S. Forest Service, National Park Service, and to some extent with other federal agencies, as well as with 13 state wildlife
FIGURE 32–2 Human-imprinted male peregrine feeding young at the Cornell University “Hawk Barn.” Photo courtesy of The Peregrine Fund.
departments and, occasionally, with county and municipal authorities. Last year no fewer than 50 government agencies and offices, conservation organizations, universities, and businesses of one sort or another were directly involved in some aspect of the eastern program. Financial support has come from numerous conservation organizations, foundations, corporations, several thousand private individuals, and federal and state agencies.
Remember, this is only one of the four major regional recovery programs for the peregrine in the United States not to mention others in Canada and elsewhere. Similar accounts could also be given for the restoration of the Atlantic salmon (Salmo solar) in New England, Kemp’s ridley sea turtle (Lepidochelys kempi) on the Texas coast, the whooping crane (Grus americana), the California condor, the Puerto Rican parrot (Amazona vittata), the golden lion tamarin (Leontopithecus rosalia) in Brazil, the Arabian oryx (Oryx leucoryx) in Oman, or the cheetah (Acinoynx jubatus) in South Africa. Each of these restoration programs is a huge enterprise, drawing on the skills and financial resources of many different individuals and institutions. For example, Snyder et al. (1987) give a detailed account of the work to restore the Puerto Rican parrot.
This new trend to rely more on technologies such as captive breeding and reintroduction for the restoration of species has become necessary because we human beings have not paid heed to the ecological imperative to preserve nature. We have not followed the philosophy of Leopold and others like him to keep the land and waters as fit habitats for wild animals and plants. And we are not likely to change our ways, as Norman Myers and many other authors emphasize in this book. Most human beings continue to be captivated more by what engineers do than by what ecologists say. As a consequence we are left with engineering solutions to environmental problems that have been largely created by engineers in the first place. Thus, I am led back to my opening quotation from Leopold: wildlife “can be restored by the creative use of the same tools which have heretofore destroyed it….”
As natural environments become more fragmented and degraded in their capacity to support a diversity of living forms, many once common species will be reduced to isolated remnants, existing as small populations in marginally suitable habitats separated by vast expanses of uninhabitable area. Some species—especially large ones and predatory ones—will be unable to maintain populations indefinitely under these conditions, owing to genetic, demographic, or ecological problems. Such populations can be sustained in part through captive propagation and reintroduction or by translocating individuals from one island of habitat to another (Cade, 1986b). I believe we will see an increasing need for these sorts of techniques in the coming decades as the biosphere undergoes the final change from unmanaged wilderness ecosystems to managed nature preserves, megazoos, and zooparks, which will be the final refuges for some species in outdoor environments (Conway, 1983).
Although I have stressed the cost and labor-intensive nature of propagation and reintroduction programs, I hasten to add that they really are not very expensive relative to many of the other things we human beings are willing to spend our public and private wealth to obtain. Consider the hundreds of billions of dollars this nation is being urged to spend on star wars technology, or the billions spent
annually on war-making machines. Consider the $10 million one person was willing to pay for an untrained race horse, and single purses of $15 million—win or lose—for a world championship heavyweight prize fight, or the billions of private dollars being spent to support cocaine and heroin addictions. When one takes account of the trillions of dollars human beings are willing to spend on foolish or trivial things, the cost of saving endangered species and endangered habitats pales to insignificance. All the work needed to restore the world’s threatened birds of prey could be accomplished with $5 million a year, about what it costs to make one armored tank.
Conservationists spend entirely too much time talking to each other. We do not need to convince each other of the importance of what we are trying to do. We need to convince the vast majority of other folks! We need less talk in any case and more action.
We do not need more science and more technology nearly as much as we need more innovative application of the knowledge and techniques of wildlife management already available. We do not have more action to preserve and to restore threatened species and their environments, first of all, because conservationists have not been persuasive enough to obtain the money needed to do the work. But another and final reason is the excessive bureaucratic incumbrance associated with the work. The number of state and federal regulations impinging on conservation projects these days, the number of permits required for one action or another, the number of agencies that have to be mollified, the number of committees that have to meet—all sap strength from the effort and further dilute the already limited funds.
All endangered species programs suffer from these ills. The California condor must be the premier example, but the peregrine falcon is a close second. To ensure the survival of these and other endangered species, programs must be clearly focused and the responsibilities for their management carefully allocated to those who are best qualified to work with each species.
REFERENCES
Barclay, J.H., and T.J.Cade. 1983. Restoration of the peregrine falcon in the eastern United States. Bird Conserv. 1:8–40.
Bohl, W.H., and G.Bump. 1970. Summary of foreign game bird liberations 1960 to 1968 and propagation 1966 to 1968. Special Scientific Report—Wildlife, No. 130. U.S. Fish and Wildlife Service, Department of the Interior, Washington, D.C. 61 pp.
Brown, J.L. 1975. The evolution of behavior. W.W. Norton, New York. 761 pp.
Cade, T.J. 1980. The husbandry of falcons for return to the wild. Int. Zoo Yearb. 20:23–35.
Cade, T.J. 1986a. Propagating diurnal raptors in captivity; a review. Int. Zoo Yearb. 24/25:1–20.
Cade, T.J. 1986b. Reintroduction as a method of conservation. Pp. 72–84 in S.E.Senner, C.N. White, and J.R.Parrish, eds. Symposium on Raptor Conservation in the Next Fifty Years, October 1984. Hawk Mountain Sanctuary Association. Raptor Research Reports No. 5. Raptor Research Foundation, Inc., Provo, Utah.
Cade, T.J., C.M.White, J.H.Enderson, and C.G.Thelander, eds. In press. Peregrine Falcon Populations, Their Management and Recovery. The Peregrine Fund, Inc., Boise, Idaho.
Conway, W. 1983. Captive birds and conservation. Pp. 23–36 in A.H.Bush and G.A.Clark, Jr., eds. Perspectives in Ornithology. Cambridge University Press, Cambridge.
Hickey, J.J. 1942. Eastern population of the duck hawk. Auk 59(2):176–204.
Immelmann, K. 1972. The influence of early experience upon the development of social behavior in estrildine finches. Int. Ornithol. Congr. 15:291–313.
Komroff, M. ed. 1926. Travels of Marco Polo. Boni and Liveright, New York. 369 pp.
Lack, D. 1937. The psychological factor in bird distribution. Br. Birds 31:130–136.
Leopold, A.S. 1933. Game Management. Charles Scribner’s Sons, New York. 481 pp.
Long, J.L. 1981. Introduced birds of the world. Universe Books, New York. 528 pp.
Lorenz, K.Z. 1937. The companion in the bird’s world. Auk 54:245–273.
Ratcliffe, D.A. 1984. The Peregrine breeding population of the United Kingdom in 1981. Bird Study 31(1):1–18.
Snyder, N.F.R., J.W.Wiley, and C.B.Kepler. 1987. The Parrots of Luquillo: Natural History and Conservation of the Puerto Rican Parrot. The Western Foundation of Vertebrate Zoology, Los Angeles. 384 pp.
Terrasse, M. 1985. Le grande aventure de Vautors. Fonds D’Intervention Pour Les Rapaces 11:16–18.
Weaver, J.D., and T.J.Cade, eds. 1983. Falcon propagation, a manual on captive breeding. The Peregrine Fund, Inc., Ithaca, N.Y. 93 pp.
CHAPTER 33
INTENSIVE TECHNOLOGY IN THE CARE OF EX SITU POPULATIONS OF VANISHING SPECIES
ULYSSES S.SEAL
Veterans Administration Hospital, Minneapolis, Minnesota
I was tempted, for this brief survey, to think solely in terms of the use of object-oriented technology in captive breeding programs for endangered species. Instead, reflection upon recent activities suggested a focus on information, software, decision analysis, and social psychology.
The use of high technology in ex situ management of endangered species is part of a rescue operation undertaken in response to piecemeal habitat management and ecosystem collapses. With new techniques, we may be able to provide a temporary haven for some of the vertebrate components of these habitats. It is rather like a lifeboat operation. It is our wish, as professionals, to do the best possible job to assist in ensuring the survival of those species that happen to gain a seat.
The word technology encompasses applied science, means of achieving a practical purpose, and the totality of the means used to provide objects necessary for human sustenance and comfort. It’s ancient Indo-European root is tekh(s), means weaving or building (Shipley, 1984).
Over the past 20 years, major contributions to successful captive management programs include the following, which are presented in more or less temporal order:
-
nutrition and prepared diets
-
chemical immobilization and anesthesia
-
vaccinations and antibiotics
-
individual animal identification and records combined with a central data base, the International Species Inventory System (ISIS)
-
reproductive control (contraception) and enhancement
-
population biology and molecular genetics
-
information technology and microcomputers
-
decision analysis and crisis management
SCOPE OF EX SITU MANAGEMENT PROBLEM
The U.S. Congress passed the Endangered Species Act in 1973. Since then, more than 910 animal and plant species worldwide have been listed as threatened or endangered by the U.S. Fish and Wildlife Service. Of these, 410 occur in the United States or its territories. The famed Red Data Books, published by the International Union for the Conservation of Nature and Natural Resources (IUCN), list about 840 animal species and subspecies worldwide as endangered.
Soulé (1986) has suggested that about 1,500 nonfish vertebrate species are likely to become extinct by the year 2050. Inclusion of fish will likely double this number. This implies a growing extinction rate of 10 to 30 vertebrate species per year without consideration of subspecies and populations.
Current guidelines from population biologists indicate that it is desirable to maintain captive populations of 100 to 300 individuals of a species (depending upon the number of founders, i.e., individuals whose offspring form captive populations, and generation time), if it is to be viable and if it is to retain about 80 to 90% of its genetic diversity for 100 to 200 years (Frankel and Soulé, 1981; Rails and Ballou, 1986). These numbers suggest that if all institutional spaces for mammals and birds were used for Species Survival Plan (SSP) programs, then only about 300 birds and mammal species could be managed in North American institutions. This is less than 10% of the number of species now exhibited in North America. Expansion of the programs to fill all the space in all the world’s potentially cooperating exhibit institutions might increase the number to about 900 species. This is 20 to 25% of the number of species exhibited. Thus there is pressure to increase the efficiency of captive management programs and to find alternative means for captive preservation of genetic resources.
NUTRITION
Prepared diets, carefully tuned to the needs of particular groups of species, are available as commercial products, greatly reducing the variations inherent in local supply and introduced by individual preparers. Now attention is directed to special requirements of individual or particular collections, for example, to thwart reproductive failure or failure of neonates to thrive or survive and to meet the special needs of hand-reared animals.
ANIMAL RESTRAINT AND CAPTURE
Remotely administered chemical immobilization of captive and free-ranging animals is a major innovation that has revolutionized our ability to work with wild animals and has become acceptably safe for routine use by experienced personnel (Harthoorn, 1976). Animals can be captured in the wild for treatment, relocation, data collection, and fitting of radio collars (Mech et al., 1984). Modern delivery techniques range from dart guns to blow pipes. The most widely used drugs in
North America are ketamine, etorphine, and xylazine. We are very close to having ideal drugs and combinations of drugs for a wide range of vertebrate species (Seal and Kreeger, in press).
BIOMEDICAL RESOURCES
Individual animal treat1ment, preventive medicine, systematic vaccination regimens, antibiotics, and commercially prepared diets have resulted in a general improvement in the health, well-being, reproduction, and survival of most species maintained in captivity. The advantages gained by increased survival and longevity include reduced demand on captive habitat resources for maintaining a population, longer generations, and a slower loss of genetic diversity through time.
The wild population of 100 to 120 black-footed ferrets (Mustela nigripes) rediscovered in 1981 in Meeteetse, Wyoming, was driven to near extinction (approximately 12 animals) in 1 year by successive epidemics of plague striking the prairie dogs and of canine distemper decimating the ferrets. Now the ferrets survive only in captivity. Management of disease in field populations is being explored through the use of vaccines presented in baits as, for example, rabies vaccines administered to fox populations and, in the near future, parvovirus vaccines for wolves. Individual animal treatment programs have been applied to wild populations of bighorn sheep, mountain goats, elk, and, recently, mountain gorillas. They have also been attempted for black-footed ferrets.
IDENTIFICATION AND DATA COLLECTION
Individual identification remains a difficult problem in captive and free-ranging populations. Animal marking technology, now fairly sophisticated, requires the handling of individuals. This technique presents difficulties for some captive species. Transponders, although appealing in principle, have not been widely used because the animals must be approached closely for scanning.
Telemetry used to locate individual animals has proven to be an essential tool for studies of many species in free-ranging populations and in release programs for condors, Arabian oryx, tamarins, and others. Innovations to the simplest technology for free-ranging species have been remote detection of death, detection of parturition, detection under ground and under water, and continuous monitoring of position with automated recording.
Recapture of individual animals using recently developed radio-operated dart collars is now possible. Dart collars contain syringes loaded with anesthetic drugs that can be injected into the free-ranging animal on command by a radio signal (Mech et al., 1984). These devices are also planned for use in several carnivore release programs to allow elective recapture of dispersing animals.
REPRODUCTION
Discussions of the possible contribution of ex situ captive breeding programs to conservation refer hopefully to the near future of 100 to 200 years by which time methods of reproductive enhancement and long-term gamete or embryo storage
techniques will allow large numbers of a species to be placed on hold in vials or straws. The cost of developing such technologies for cattle, turkeys, and humans has been in the millions of dollars over several decades, but only minimally effective transfer to other species has been accomplished to date. Cryopreservation techniques have been developed only for sperm in egg-laying species—primarily birds; invertebrates have not been given serious attention. Thus embryo and ovum preservation techniques are not yet available for all nonmammalian vertebrate classes and all invertebrates.
Reversible contraception for regulation of zoo mammal populations has been in use for 10 years (Seal et al., 1976). In females, oral preparations are used occasionally; more commonly, individuals are given hormonal implants lasting 2 to 5 years. Male contraception without substantial behavioral disruption or introduction of novel chemicals with uncertain long-term effects remains a major challenge.
Additional manipulations of reproduction and behavior in vertebrates are being achieved by altering photoperiod and by administering the pineal hormone melatonin. In selected cases, these techniques will double the number of annual reproductive periods in species that reproduce seasonally.
POPULATION BIOLOGY AND MOLECULAR GENETICS
The key problem is how to manage small, captive, and fragmented wild-living populations—both demographically and genetically—to ensure their viability and retention of genetic diversity through time (Soulé, 1986). The current flux in the concept of “species” has led to the suggestion that molecular, morphological, and geographic distribution data be examined to assess the genetic structure of the population and to determine whether separate population units should be considered for captive conservation management (Ralls and Ballou, 1986). Molecular techniques are also being used to measure heterozygosity in founders and captive-bred populations, to validate pedigree assignments, and to evaluate the origins of founder stock.
Molecular methods include karyotyping, many forms of electrophoresis, immunological comparison of proteins, sequencing of proteins and DNA, and comparison of DNA restriction fragment-length polymorphisms (O’Brien et al., 1985). It is possible to identify individuals and familial relationships with several of these techniques. The current large-scale automation of these techniques will allow the complete sequencing of any genome.
These methods have had a significant impact on the captive management of three species in particular: orangutans, with the establishment of separate subspecies from Borneo and Sumatra by karyotype and two serum proteins; Asiatic lions, with the electrophoretic demonstration of African lion founders in the lineage of nearly all the captive population; and tigers, with the molecular evidence failing to support the traditional separation into separate subspecies.
INFORMATION TECHNOLOGY
Digital technology is the foundation of our new age of information. Integrated circuits, computing software, and photonics are the key technologies for manip-
ulating digits. Single-chip circuits can now hold about 1 million components; an upper limit of 10 to 100 million should be reached by the year 2000. Integrated circuits are the source of computing power.
This trend combined with access to unlimited, inexpensive random-access memory (RAM) and the availability of gigabyte storage devices with 20-millisecond access times permit full desktop access to all the world’s printed information. We need to place high priority on facilitating access to the world’s conservation data bases stored on magnetic, compact disk/read-only memory (CD-ROM), or laser disk media. Thought must now be given to management of the transfer, loan, or sale of these data bases.
The Conservation Monitoring Center of IUCN’s Species Survival Commission (SSC) collects data worldwide on the status of species in the wild. This information is published in the Red Data Books (see Chapter 29) and is also available on selected printouts. These data are now also becoming available on line or on magnetic media—an urgent need.
The International Species Inventory System (ISIS) provides a continuously updated, centralized data base of census, demographic, pedigree, and laboratory data on wild animals kept in zoos. It has established agreed-upon minimum data standards for animal record keeping, and it reports to all participants at least annually.
International and regional studbooks have been compiled by zoo biologists for about 80 rare and endangered species. Currently, IUCN and the International Union of Directors of Zoological Gardens (IUDZG) require that the data in those books be placed in the ISIS data base for species of captive population management interest.
COMPUTER SOFTWARE AND ISIS
The pressure to develop suitable software is enormous and creates a major bottleneck, hampering progess in developing species survival plans. Programming productivity is very low and has not improved in pace with the development of technology. Serious efforts are under way to develop software application generators that will respond to English language requests and yield programming on a conversational level.
The development of rule-based systems for well-circumscribed problem solving (so-called expert systems) is well under way in the biomedical community, but no applications have yet appeared in the conservation or zoo communities. Training will be accomplished through interactive use of video disk and computer technology.
Contributions to the ISIS data base are now being made by 218 zoological institutions in 14 countries (Flesness et al., 1984). Recently, coverage has expanded substantially in Europe and India and is beginning in Australia. The U.S. Fish and Wildlife Service has agreed to sponsor participation by zoos in India.
Relevant ISIS summary census data are being sent annually to all SSC special groups. Software for in-house inventory records has been developed. This package, ARKS, has now been ordered by more than 60 zoological institutions in 7 countries.
A companion medical record system, MedARKS, is under construction. Phase 1, on anesthesia, will be released this year. ISIS is beginning to work on single species analysis and management software, i.e., studbook software.
New International Studbook keepers are now required to enter their data into the ISIS data base. The American Association of Zoological Parks and Aquariums (AAZPA) requires its studbook keepers to assist in maintaining their species data on ISIS. The European Species Survival Plan coordinators are also required to enter their species data into ISIS.
IN SITU AND EX SITU METHODS
Conservation of species is clearly best served by in situ methods based on biological community and habitat. It is equally clear that the pace of reduction in population numbers and complete loss of the larger vertebrate species—the charismatic megavertebrates—is frequently exceeding our ability to achieve their in situ survival. However, the habitat of many of these species is still present, e.g., for the Arabian oryx, Przewalski’s horse, and black-footed ferret.
The application of ex situ methods and biotechnology may provide a buffer against final extinction. Ex situ methods also promote the use of the “rarity crisis mentality” to stimulate awareness and action on habitat protection schemes. Early and repeated reintroduction programs, as for the peregrine falcon (Falco peregrinus; see Chapter 32), then can encourage continued visibility and protection of the chosen habitat fragments. It will probably also be necessary to manage the species in these fragments for many years to come. Protection may thus be afforded to a wider range of biological diversity, including the plants and invertebrates in these refugia, which may eventually serve as sources for expansion.
ETHICS AND VALUE SYSTEMS
The clearest challenge is one of ethics. None of the technologies and strategies described above will ensure the continuing evolution of these species as part of a biological community. They are temporary expedients at best. Protection of biological diversity by resorting to aesthetic, economic, and other utilitarian values can always be discounted. Any single species can be discounted to near zero value by economic reasoning. The incremental ecological value of a single species, as a part of the interconnectedness of biological communities, is usually small except for keystone species. Thus many species or individual habitat units will be lost before their loss is felt. This characteristic of current human thinking requires catastrophe and crisis to capture awareness and motivate action. Thus much of our time as conservation biologists is spent in crisis management, negotiations, and hasty efforts to assemble sufficient data to retrieve yet another favored species. It is essential that humans be encouraged to expand their ethical awareness to encompass other species and the living land as a loan from our children which must be retained.
REFERENCES
Flesness, N.R., G.Garnatz, and U.S. Seal. 1984. ISIS—An international specimen information system. Pp. 103–112 in R.Allkin and F.A.Bisby, eds. Databases in Systematics. Academic Press, New York.
Frankel, O.H., and M.E.Soulé. 1981. Conservation and Evolution. Cambridge University Press, New York. 327 pp.
Harthoorn, A.M. 1976. The Chemical Capture of Animals. Bailliere Tindall, London. 416 pp.
Mech, L.D., R.C.Chapman, W.W.Cochran, L.Simmons, and U.S. Seal. 1984. Radio-triggered anesthetic-dart collar for recapturing large mammals. Wildl. Soc. Bull. 12:69–74.
O’Brien, S.J., M.E.Roelke, L.Marker, A.Newman, C.W.Winkler, D.Meltzer, L.Colly, J. Everman, M.Bush, and D.E.Wildt. 1985. Genetic basis for species vulnerability in the cheetah. Science 227:1428–1434.
Ralls, L., and J.D.Ballou, eds. 1986. Genetic management of captive populations. Zoo Biol. 5(2):84–238.
Seal, U.S., and T.J.Kreeger. In press. Chemical immobilization of furbearers. In M.Novak and J.A.Baker, eds. Wild Furbearer Management and Conservation in North America.
Seal, U.S., R.Barton, L.Mather, K.Olberding, E.D.Plotka, and C.W.Gray. 1976. Hormonal contraception in captive female lions (Panthera leo). Zoo Anim. Med. 7:12–20.
Shipley, J.T. 1984. The Origins of English Words. A Discursive Dictionary of Indo-European Roots. Johns Hopkins University Press, Baltimore. 636 pp.
Soulé, M.E. 1986. Conservation Biology: The Science of Scarcity and Diversity. Sinauer Associates, Sunderland, Mass. 584 pp.
CHAPTER 34
CRYOBIOLOGY, EMBRYO TRANSFER, AND ARTIFICIAL INSEMINATION IN EX SITU ANIMAL CONSERVATION PROGRAMS
BETSY L.DRESSER
Director of Research, Cincinnati Wildlife Research Federation, Cincinnati, Ohio
As a prelude to this discussion of the ex situ technology that is or could be available to maintain biological diversity in conservation programs, we must first explore why we should be interested in this technology at all. The greatest threat to the preservation of genetic diversity of wild animals comes from the destruction and degradation of their habitats (Croner, 1984). Some experts are estimating that if today’s pace of habitat destruction continues, 1 million species of plants and animals may become extinct by the end of this century (AAZPA, 1983). Continued destruction of tropical rain forests, the most species-rich land environment on Earth, may result in mass extinctions that would permanently impoverish the planet (see Part 3 of this book: Diversity at Risk: Tropical Forests).
Other threats to species come from the destruction of animals and plants for food and trade. The Global 2000 Report to the President of the United States (Barney, 1982) states that the extinctions projected for the coming decades will be largely generated by humans and on a scale that renders the gradual process of natural extinction trivial by comparison. Extinction is a catastrophe, not only aesthetically or because of the effect it has on the ecological balance, but also because it deprives mankind of part of Nature’s potential. Each species forms a genetic reservoir (genome) that may be of value in agriculture, medicine, or industry (Daniel, 1981). Forty percent of our present-day medicines are derived from wild plants and animals (Wolkomir, 1983; see also Farnsworth, Chapter 9 of this volume). It is impossible to know what other additional resources lay waiting to be tapped.
Animal and plant germplasm should not be preserved merely for altruistic reasons
but also for reasons that are of direct benefit to mankind. It is not known which plants and animals may prove useful in the future. It is known, however, that certain animals have proven invaluable to our knowledge of diseases and human health. For example, armadillos (Dasypus novemcinctus) have taught us much about leprosy as they are the only animal model to acquire this disease when injected with Mycobacterium leprae experimentally (Storrs et al., 1980). The small cotton-top tamarins (Saguinus oedipus) from South America have the highest incidence of colon cancer of any animal and have proven to be an excellent species for the study of this fatal disease (Lushbaugh et al., 1984). Studies of spontaneous diabetes mellitus in the South African hamster (Mystromys albicaudatus) (Stuhlman, 1979) and AIDS (acquired immunodeficiency syndrome) in the macaque (Macaca cyclopis, M. mulatta, or both) may eventually lead to cures in humans (Letvin and King, 1984). Hepatitis B virus was discovered in a group of North American woodchucks (Marmota monax) by a zoo pathologist at the Penrose Research Laboratories in Philadelphia (Snyder et al., 1982). The study of the livers of these animals has given human medicine the greatest insight into the etiology of this often seen liver tumor. There are many more examples. Because of the diversity of species, there can be and, in fact, is a broader base of general medical knowledge that is benefiting mankind.
One bright spot in the otherwise gloomy picture of animal extinction is the new interest that has been generated in the field of wildlife reproduction. Fueled by the groundwork laid by the agricultural industry, zoo researchers have begun to study the reproductive processes of wild animals and to use technology such as embryo transfer and artificial insemination to help improve the reproductive potential of wild animals. Zoos worldwide have begun to pursue newfound roles as conservators rather than merely displayers of wildlife.
One of the biggest problems plaguing animals in today’s zoos is the loss of genetic diversity due to inbreeding. This is particularly true with species whose numbers have been so depleted that they cause a genetic bottleneck. For example, the douc langur (Pygathrix nemaeus), a primate from Southeast Asia, is now virtually extinct in the wild, and there are not enough in captivity to ensure the genetic diversity necessary to keep the species alive (Gorman, 1980). With no new blood, so to speak, they can pass only a limited array of genes to their offspring, and as a result, they gradually become genetic carbon copies lacking the built-in adaptability to environmental change that would otherwise occur through natural selection in a genetically diverse group. Genetic diversity is a key to species survival. A species must have enough variation within its genome to enable it to adapt to environmental changes.
Inbreeding has taken its toll in the zoo world, resulting in problems such as decreased fertility, high juvenile mortality (known as inbreeding depression), and birth defects. In 1973, to help fight inbreeding, Ulysses Seal and Dale Mackey created ISIS (the International Species Inventory System), which is based at the Minnesota Zoo (see Chapter 33). ISIS computers catalog animal information from nearly 200 zoological institutions worldwide, including genealogy information for individual animals. One of the best functions of ISIS is that of a computerized
matchmaker for participating zoos who wish to swap bloodlines. As zoo animal sperm and embryo banks become a reality, it is hoped that frozen embryo and semen samples can also be cataloged in the ISIS computer as an aid to zoos interested in transporting germplasm rather than animals themselves for the introduction of new bloodlines.
In consideration of the above concerns, this chapter discusses reproductive technology as it applies to the long-term preservation of animal germplasm and maintenance of biological diversity. Most ongoing wild animal research is directed toward the improvement of genetic and species diversity. Scientists have realized that the development of advanced reproductive technology, such as embryo transfer, gamete cryopreservation, and artificial insemination, may represent the real key to the future for many species who are currently threatened by extinction. They have also realized that this technology will do a great deal to improve and maintain the genetic diversity within captive populations. However, much of the application of this technology to wild species is still in its infancy.
EMBRYO TRANSFER
Embryo transfer is a technique by which fertilized ova and early embryos are recovered from the reproductive tract of a donor female, the genetic mother, and are transferred into the tract of a recipient female, the foster mother, in whom the embryos develop into full-term fetuses and live young. The first successful transfer of mammalian embryos was performed in the rabbit by Heape in 1891. His observations stimulated relatively little further research until about 1950. Since then, there has been an explosion of research in this area. Numerous published reviews and textbooks describe both the methods and the fundamental principles on which the technique of embryo transfer rests [see reviews by Betteridge (1977), Mapletoft (1984), Seidel (1981), and Sreenan (1983), and texts by Adams (1982), Cole and Cupps (1977), and Daniel (1978)]. Although the specifics of the methods depend upon the species used, the general principles are the same whether performed in laboratory animals such as mice and rabbits, in large domestic animals such as horses and cattle, or in wild species such as baboons and antelope.
Embryo transfer in two species, mice and cattle, has become absolutely routine. It is not an exaggeration to state that tens of thousands of living mice and cattle have been produced by embryo transfer. In general terms, the transfer of embryos of other species has been modeled on techniques devised for these two species. Although there are fewer live young, probably thousands of live rabbits, pigs, and horses have been produced by embryo transfer. The application of embryo transfer to wild species is a relatively recent event. Its history can be highlighted as follows:
|
1975 |
The first successful nonhuman primate surgical embryo transfer in a baboon (Papio cynocephalus) (Kraemer et al., 1976). |
|
1976 |
The first successful wildlife surgical interspecies embryo transfer between mouflon (wild sheep; Ovis musimon) and domestic sheep (Ovis aries) (Bunch et al., 1977). |
|
1981 |
Second successful surgical transfer of an embryo from a wild species into a domestic species—gaur (Bos gaurus) to Holstein (Bos taurus) (Stover et al., 1981). |
|
1983 |
First successful nonsurgical embryo transfer performed with an eland antelope (Tragelaphus oryx) (Dresser et al., 1984a). |
|
1983 |
First successful nonsurgical embryo transfer with the eland antelope (Tragelaphus oryx) involving a previously frozen embryo (Kramer et al., 1983). |
|
1984 |
First successful primate interspecies embryo transfer—macaque (Macaca fascicularis) to rhesus (M. mulatta)—following in vitro fertilization (Balmaceda et al., 1986). |
|
1984 |
First frozen embryo transfers in nonlaboratory species of primates accomplished in the common marmoset (Callithrix jacchus) (Hearn and Summers, 1986). |
|
1984 |
First successful nonsurgical interspecies embryo transfer between two different species of wild animals—bongo (Tragelaphus euryceros) to eland (Tragelaphus oryx). These bongo antelope embryos were brought from Los Angeles to Cincinnati and transferred fresh, 12 hours after collection (Dresser et al., 1984b). Embryos from a wild species had never before been transported long distances. |
|
1984 |
Nonsurgical interspecies embryo transfer from Grant’s zebra (Equs burchelli) to horse (E. caballus) (Bennett and Foster, 1985; Foster and Bennett, 1984). |
|
1984 |
First long-term frozen embryo transfer in a wild species: an eland (Tragelaphus oryx) embryo previously frozen for 1.5 years successfully transferred nonsurgically to an eland surrogate (Dresser, 1986). |
|
1984 |
Interspecies embryo transfer from Przewalski’s horse (Equs przewalski) to New Forest pony (Equs caballus) (Kydd et al., 1985). |
|
1985 |
First successful embryo transfer in Dall sheep (Ovis dalli) (K.Mehren, Metro Toronto Zoo, personal communication, 1986). |
|
1987 |
First nonsurgical embryo transfer between guar (Bos gaurus), an endangered species, and Bos taurus, a domestic Holstein, by Dresser and colleagues. |
Synchronization of the donor and recipient animals in embryo transfer can be accomplished through precisely timed injections of prostaglandins, such as Lutalyse and Estrumate. These hormone analogs serve to stimulate the ovaries to begin a new cycle. Superovulation of the donor is accomplished through the injection of fertility hormones such as follicle-stimulating hormone (FSH). Superovulation has been fairly successful with the ungulates, but optimal drugs and dosages have yet to be refined for most other species. As many as 31 embryos have been collected from one FSH-stimulated eland cow (Dresser, 1983). On the other hand, fertility drugs seem to have little or no effect on the equids (such as zebras and Przewalski’s horse) (Hearn and Summers, 1986). Clearly, the hormone regimen that produces the optimal superovulation response within a given species seems to be fairly individualized and much work is needed in this particular area.
In felines, the superovulation of donors and the synchronization of donors and recipients is complicated by the fact that most cats are induced or reflex ovulators, meaning that they ordinarily do not ovulate without the stimulation of copulation. Human chorionic gonadotropin (HCG) has been administered to domestic cats to cause ovulation to occur (sometimes in conjunction with stimulation by a vasectomized male) (Bowen, 1977). Researchers in several institutions have been working on embryo transfer in domestic cats with limited success (Dresser et al., 1987; Goodrowe et al., 1986; Kraemer et al., 1979), but little work has been done thus far on wild cats (Bowen et al., 1982; Reed et al., 1981). It is hoped that domestic cats may be able to serve as surrogates for incubating embryos from small endangered

Gaur calf born to Holstein surrogate on 5/25/87 at Kings Island Wild Animal Habitat. This was the first nonsurgical interspecies embryo transfer between an endangered species and a domestic species. Photo by Dr. Betsy L.Dresser.
wild cats such as the black-footed cat (Felis nigripes). A great deal more work needs to be done in this area to determine the best regimens for stimulating cat’s ovaries to produce more than the usual number of follicles and to cause the ovulation of these follicles and, thus, to obtain embryos.
The development of embryo transfer techniques is essential if genetic diversity within captive populations is to be maximized. The ability to introduce new bloodlines into a captive population through the transfer of nonlocal embryos into surrogates, would be far preferable to the transport of adult animals for breeding purposes, or to the depletion of wild herds to add new breeding stock to captive populations. In addition, it is a goal of many zoo researchers to develop interspecies embryo transfers to the point at which embryos can be collected from endangered species and transferred to surrogates of a more common species, thereby greatly increasing the reproductive potential of the donor species.
Other important benefits result from embryo transfer. For example, it has been found that disease transmission between different populations can be dramatically reduced by embryo transfer (see Hare, 1985, for review). This happens because the intact embryo collected from a diseased mother is almost always free of the microbial or viral disease agent and does not transmit the disease to the foster mother. Alternatively, the surrogate mother may confer passive immunity to her
offspring that develop from transferred embryos. This may occur either through the placental blood supply or via the colostrum, the first milk. That is, it has been found that cattle of a given breed may quickly succumb to local diseases when imported into a new location. However, live calves produced by transfer of embryos from a foreign breed into another (native or endemic) domestic breed will usually be as disease-resistant as this domestic breed. This accident of biology should have important consequences for the transplant of both domestic and wild species from one location to another.
Interspecies embryo transfer has enjoyed limited success in wild animals, but much more research needs to be done in this area. Intergeneric, e.g., eland to cow (Dresser et al., 1982a) or water buffalo to cow (Drost, 1983), as opposed to interspecies, e.g., tiger to lion (Reed et al., 1981), embryo transfers have never been successful. It seems that most embryos can develop to the early blastocyst stage in the oviducts of unrelated species (Daniel, 1981), but further development requires a much closer relationship between donor and recipient and similarity in time and type of implantation, placenta formation, rate of ovum transport, length of gestation, birth weight, and both neonatal and maternal postpartum behavior. Several techniques have been tried to help overcome the surrogate mother’s immune response and prevent the rejection of the foreign embryo in an interspecies embryo transfer. Recently, a domestic horse mare gave birth to a donkey foal after she had been injected with donkey white blood cells (Antczak, 1985). Prior to this treatment, all other donkey embryo transfers into domestic horses had failed. It is not yet clear why the procedure was successful.
Although various attempts have been made to collect embryos from nonhuman primates at various zoos and primate centers over the years, very few of these procedures have been reported in the scientific literature. For a review of the existing literature on superovulation and ova collection attempts in the more common nonhuman primates, see Bavister et al. (1985), Clayton and Kuehl (1984), Hodgen (1983), and Kraemer et al. (1979).
There has been dramatic progress in embryo micromanipulation in little over 6 years, especially as it applies to domestic animals. The research of Willadsen (1979, 1980) and Willadsen et al. (1981) is most notable in this regard.
In the most important application of micromanipulation to exotic animals, an embryo is microsurgically bisected into two or more pieces, thereby producing genetically identical twins or triplets. This technique, although not yet successful with exotic species, could help to quickly increase the numbers of endangered or rare animals.
Another result of micromanipulation has been the production of chimeras, which are embryos that are a product of combining two embryos at a relatively early stage of development. It is possible to prevent the surrogate uterus from recognizing the foreign embryo by combining different blastomeres. The younger cells tend to form the trophoblast that gives rise to the placenta, and the older cells tend to form the inner cell mass that will form the fetus. Chimeric embryos can be constructed so that the cells that constitute the trophoblast belong to the surrogate species and the cells of the inner cell mass belong to the donor species. A reupholstered embryo
such as this can effectively trick the surrogate’s uterus into thinking it is carrying a native embryo (Vietmeyer, 1984).
Interspecies embryo transfer and inner cell mass transfer should not be confused with hybridization of species. Offspring contain only genetic material of the original species. Many people now recognize that the method of chimera production might be utilized to rescue endangered species. It might be possible to construct chimeras consisting of an embryo from an endangered species plus an embryo of a common, but related species. The common species might then carry the fetus of the endangered species to term.
In summary, then, embryo transfer has become a widely used technique to produce live young animals, especially of domestic species. Although it is as yet still a novel procedure in wild species, continued research will inevitably make transfer of wild animal embryos as successful as the transfer of domestic animal embryos.
CRYOPRESERVATION OF EMBRYOS
The first successful freezing of mammalian embryos was reported in 1972 (Whittingham et al., 1972; Wilmut, 1972). Since those reports, more than 300 articles and 100 abstracts have been published on this one subject. There have also been three full meetings (Ciba Foundation, 1977; Muhlbock, 1976; Zeilmaker, 1981) and numerous symposia devoted exclusively to the freezing of mammalian embryos. Numerous reviews have also been published (Lehn-Jensen, 1981; Leibo, 1977, 1981; Maurer, 1978; Rall et al., 1982; Renard, 1982).
Freezing of mouse, rabbit, and bovine embryos has now become a routine procedure. Altogether, embryos of 11 mammalian species have now been successfully frozen. Again, success means that live young have been born from frozen embryos. To date, the species that have been successfully preserved include the mouse (Whittingham et al., 1972), cow (Wilmut and Rowson, 1973), rabbit (Bank and Maurer, 1974), sheep (Whittingham et al., 1972), rat (Whittingham, 1975), goat (Bilton and Moore, 1976), horse (Yamamoto et al., 1982), human (Trounson and Mohr, 1983), baboon (Pope et al., 1984), antelope (Dresser et al., 1984a), and cat (Dresser et al., 1987).
The freezing of mouse embryos has become so routine that banks of tens of hundreds of thousands of mouse embryos have been frozen to preserve valuable genetic stocks for extended times. For some species, most notably the mouse, rabbit, and cow, preservation by freezing has reached such a high level of sophistication and reliability that approximately 80 to 90% of frozen embryos will develop in vitro when thawed and cultured. Moreover, the procedures to freeze embryos have become increasingly simplified. Regardless of the methods used, it can be reasonably estimated that thousands, if not tens of thousands, of live young of domestic species have been produced from frozen-thawed embryos.
Again, because of extremely limited experimental material, only a few attempts have been made to transfer previously frozen wild animal embryos into recipients.
The procedure has yielded some limited success (Cherfas, 1984; Dresser et al., 1984a; Hearn and Summers, 1986; Kramer et al., 1983).
Cryopreservation will undoubtedly prove to be an important adjunct to reproductive research in nondomestic animals. Geneticists Thomas Foote and Ulysses Seal have determined that a population of 250 properly managed animals of a particular species can theoretically preserve 95% of the original genetic diversity of the group after 50 generations (400 years) (Myers, 1984). The world’s zoos, however, have limited facilities and often cannot accommodate large numbers of animals of each species they maintain. Success in the area of cryopreservation will allow zoo professionals to overcome the limited space in zoos and wild animal preserves by maintaining the bulk of the desired genetic diversity in liquid nitrogen freezers. Embryos containing new bloodlines could be recovered in the wild and brought back to zoos in the frozen state to improve the bloodlines of captive populations without depleting the wild herds. U.S. government restrictions currently prohibit the importation of embryos from other countries, but scientists have been actively lobbying for a change in these restrictions. It appears that a change may be possible in the future.
ARTIFICIAL INSEMINATION
Artificial insemination is the introduction of semen into the vagina or cervix by artificial means. This procedure was supposedly used by the Arabs in ancient times, but the first documented success in the modern world occurred in 1784 with the artificial insemination of a dog (Betteridge, 1981). In the 1930s, artificial insemination of livestock was used extensively in Russia. Arthur Walton demonstrated its potential as an effective method to transport genes in the 1920s and 1930s by shipping fresh rabbit, sheep, and bull semen from England to other European countries (Betteridge, 1981). The ability to successfully freeze semen resulted from the discovery of the cryoprotective action of glycerol by Polge, Smith, and Parkes in 1949 (Betteridge, 1981). Artificial insemination is very common in the agricultural industry today. Foote (1981) estimated that close to 90 million head of cattle were produced worldwide in 1977 by artificial insemination with previously frozen semen samples. Artificial insemination has had limited success in wild animals thus far, especially with certain species of mammals and birds. Success has been attained for the following species:
|
Birds Reptiles |
||
|
Cranes (several species) |
Waterfowl (ducks) |
Tortoises1 |
|
Albino cockatiel |
Pheasants |
|
|
Raptors |
|
|
|
1Frozen semen. |
||
Much more semen has been collected from wild species than has actually been evaluated and used. The first successful artificial insemination of a wild species with previously frozen semen occurred in 1973 with the wolf (Seager, 1981). This was followed by the successful insemination of a gorilla (Douglass and Gould, 1981). There has been a great deal of time and effort spent trying to artificially inseminate wild-caught felidae (cats); most attempts have resulted in failure (Dresser et al., 1982b). The London Zoo finally produced a puma in 1980 through surgical artificial insemination with a fresh semen sample (Moore et al., 1981). This was followed in 1981 by a successful nonsurgical artificial insemination of a Persian leopard with fresh semen at the Cincinnati Zoo (Dresser et al., 1982b).
There are three methods of semen collection: manual stimulation of the male reproductive tract, use of an artificial vagina, and electroejaculation (Cherfas, 1984). Electroejaculation was invented by two French workers, Jonet and Cassou, and is by far the most common collection mode for wild animals. Electroejaculation works by inserting a lubricated probe into the rectum of an anesthetized animal. This conveys mild pulsating electrical stimuli to the nerves of the reproductive tract, resulting in ejaculation. There is some question about the fertility of sperm collected through electroejaculation, but there is also a question about how viable a semen sample must be to be effective. For example, the successful gorilla insemination at the Memphis Zoo in 1981 was accomplished with a previously frozen sample whose motility was 10% and judged to be poor at the time of insemination (Douglass and Gould, 1981).
A great many problems are associated with artificial insemination as it applies to wild animals. First of all, it necessitates the use of anesthesia, which is always a risk, for both semen collection from the male and insemination of the female. In addition, as mentioned above for electroejaculation, the fertility of semen obtained from artificial collection techniques is sometimes questionable when compared to that produced in a natural ejaculation. Sperm usually begin to die as soon as they are collected. Even if a fresh sample is used for insemination, it is likely to have undergone a certain amount of sperm loss.
SEMEN CRYOPRESERVATION
The freezing process is somewhat detrimental to sperm, and it is very unlikely for sperm to come out of a thaw as motile as they were going into the freeze (Cochran et al., 1985). Much work needs to be done in the area of semen cryopreservation for exotic animals. To date, sperm from at least 200 different species has been frozen, but very little of it has actually been thawed and tested (Seager, 1981). The ultimate test is production of offspring. From semen that has been tested, cryobiologists have found that sperm from each species needs to be extended and frozen under slightly different conditions to produce the optimal results.
Extenders used to preserve the collected semen basically consist of a buffered solution that contains a cryoprotectant (e.g., glycerol), antibiotics, and either egg yolk or milk. Many variations have been tried in the basic recipe for semen extender. The agricultural industry has found that the optimal extender for a given species seems to be very species-specific and that is turning out to be true with wild animals also. Perhaps some clues for semen preservation can be found in the natural world from studying certain female reptiles who have the potential for keeping sperm viable within their bodies for up to 6 years after mating (Cherfas, 1984).
Other problems with artificial insemination include difficulty in predicting the optimal time for inseminating the female and the fact that artificial insemination cannot occur as frequently as a female would have been inseminated naturally for the duration of her estrous cycle.
On the positive side, artificial insemination can be a great boon toward improving the genetic diversity of a captive population of animals. As with embryo transfer, the risk and expense of transporting semen is far less than that of transporting a male animal for breeding purposes. Artificial insemination could also be used to overcome quarantine restrictions and the risks of disease. Often, the strict agricultural legislation has made transport of zoo animals more difficult and costly than is perhaps necessary.
FUTURE PROSPECTS
Since preservation of genetic material from a species is one of the keys to ensuring diversity, the development of reproductive technology for exotic species, such as cryopreservation of gametes, embryo transfer, and artificial insemination, should be emphasized and supported. Ex situ animal conservation programs that are dependent upon the long-term preservation of genetic variation should apply this technology as it becomes available because of the increasing realization that captive breeding programs are essential to prevent many species from becoming extinct. Loss of genetic diversity could also limit the potential of a population to adapt to new environments when reintroduced to the wild.
A large amount of basic research is urgently needed before application of new technology will be routine for maintaining captive populations. It is hoped that the urgency will be recognized by many more scientists worldwide than at present, and that ex situ conservation programs will become the nuclei of genetic material for dwindling populations of wild animals. Extinction for some may be softened by the frozen zoo concept, which may turn out to be the single most important reproductive technology developed for exotic animals during this decade. Its effects will reach centuries into the future for many species.
REFERENCES
AAZPA (American Association of Zoological Parks and Aquariums). 1983. Species Survival Plan Handbook Publication. Oglebay Park, Wheeling, W. Va. 24 pp.
Adams, C.E., ed. 1982. Mammalian Egg Transfer. C. R. C. Press Inc., Boca Raton, Fla.
Antczak, D.F. 1985. Cute cross-species birth. Sci. News 128:8.
Balmaceda, J., T.Heitman, A.Garcia, C.Pauerstein, and R.Pool. 1986. Embryo cryopreservation in cynamologus monkey. Fertil. Steril. 45(3):403–406.
Bank, H., and R.R.Maurer. 1974. Survival of frozen rabbit embryos. Exp. Cell Res. 89:188–196.
Barney, G.O. (Study Director). 1982. The Global 2000 Report to the President of the United States. Vol. 1: The Summary Report—Special Edition with the Environmental Projections and the Government’s Global Model. Council on Environmental Quality, U.S. Government Printing Office, Washington, D.C. 360 pp.
Bavister, B.D., K.Collins, and S.Eisele. 1985. Non-surgical embryo transfer in the rhesus monkey. Theriogenology 23:177.
Bennett, S.D., and W.R.Foster. 1985. Successful transfer of a zebra embryo to a domestic horse. Equine Vet. J. Suppl. 3:53–62.
Betteridge, K.J. 1977. Embryo Transfer in Farm Animals. Monograph 16. Canada Department of Agriculture, Ottawa. 92 pp.
Betteridge, K.J. 1981. An historical look at embryo transfer. J. Reprod. Fertil. 62:1–13.
Bilton, R.J., and R.W.Moore. 1976. In vitro culture, storage, and transfer of goat embryos. Aust. J. Biol. Sci. 29:125–129.
Bowen, M.J., C.C.Platz, Jr., C.D.Brown, and D.C.Kraemer. 1982. Successful artificial insemination and embryo collection in the African lion (Panthera leo). Am. Assoc. Zoo Vet. 57–59.
Bowen, R.A. 1977. Fertilization in vitro of feline ova by spermatozoa from the ductus deferens. Biol. Reprod. 17:144–147.
Bunch, T.D., W.C.Foote, and B.Whitaker. 1977. Interspecies ovum transfer to propagate wild sheep. J. Wildl. Manage. 41(4):726–730.
Cherfas, J. 1984. Test-tube babies in the zoo. New Sci. 16–19.
Ciba Foundation. 1977. The Freezing of Mammalian Embryos. Symposium 52. Elsevier Excerpta Medica, Amsterdam. 330 pp.
Clayton, O., and T.J.Kuehl. 1984. The first successful in vitro fertilization and embryo transfer in a non-human primate. Theriogenology 21:228. (Abstract)
Cochran, R.C., J.K.Judy, C.F.Parker, and D.M.Hallford. 1985. Prefreezing and post-thaw semen characteristics of five ram breeds collected by electroejaculation. Theriogenology 23:431–440.
Cole, H.H., and P.T.Cupps, eds. 1977. Reproduction in Domestic Animals. Academic Press, New York. 665 pp.
Croner, S. 1984. An Introduction to the World Conservation Strategy. Prepared for the International Union for Conservation of Nature and Natural Resources (IUCN) by its Commission on Education. IUCN, Gland, Switzerland; United Nations Environmental Program, Nairobi, Kenya. 28 pp.
Daniel, J.C., Jr., ed. 1978. Methods in Mammalian Reproduction. Academic Press, New York. 566 pp.
Daniel, J.C. 1981. Preserving the genome of endangered species. SWARA—E. Afr. Wildl. Soc. 4:16–18.
Douglass, E.M., and K.G.Gould. 1981. Artificial insemination in lowland gorilla (Gorilla g. gorilla). Pp. 128–130 in Annual Proceedings—American Association of Zoo Veterinarians. Hills Division, Riviana Foods, Topeka, Kans.
Dresser, B.L. 1983. Embryos of the African eland (Taurotragus oryx). Pp. 9–11 in Proceedings of the Owners and Managers Workshop of the IXth Annual Meeting of the International Embryo Transfer Society. International Embryo Transfer Society, Fort Collins, Colo.
Dresser, B.L. 1986. Embryo transfer in exotic bovids. Int. Zoo Yearb. 24/25:138–142.
Dresser, B.L., L.Kramer, C.E.Pope, R.D.Dahlhausen, and C.Blauser. 1982a. Superovulation of African eland (Taurotragus oryx) and interspecies embryo transfer to holstein cattle. Theriogenology 17:86. (Abstract)
Dresser, B.L., L.Kramer, B.Reece, and P.T.Russell. 1982b. Induction of ovulation and successful artificial insemination in a Persian leopard (Panthera pardus saxicolor). J. Zoo Biol. 1(1):55–57.
Dresser, B.L., L.Kramer, R.D.Dahlhausen, C.E.Pope, and R.D.Baker. 1984a. Cryopreservation followed by successful transfer of African eland antelope (Tragelaphus oryx) embryos. Pp. 191–193 in Proceedings of the 10th International Congress on Animal Reproduction and Artificial Insemination. Standing Committee of the International Congress of Animal Reproduction and Artificial Insemination, University of Illinois at Urbana-Champaign, Ill.
Dresser, B.L., C.E.Pope, L.Kramer, G.Kuehn, R.D.Dahlhausen, E.J.Maruska, B.Reece, and W.D.Thomas. 1984b. Successful transcontinental and interspecies embryo transfer from bongo an-
telope (Tragelaphus euryceros) at the Los Angeles Zoo to eland (Tragelaphus oryx) and bongo at the Cincinnati Zoo. Pp. 166–168 in Annual Proceedings of the American Association of Zoological Parks and Aquariums (AAZPA). American Association of Zoological Parks and Aquariums, Wheeling, W. Va.
Dresser, B.L., C.S.Sehlhorst, G.Keller, L.W.Kramer, and R.W.Reece. 1987. Artificial insemination and embryo transfer in the Felidae. Pp. 287–293 in Tigers of the World: The Biology, Biopolitics, Management and Conservation of an Endangered Species. Noyes Publications, Park Ridge, N.J.
Drost, M. 1983. Reciprocal embryo transfer between water buffaloes and cattle. Pp. 63–71 in Proceedings of the Annual Meetings of the Society for Theriogenology. Society for Theriogenology, Hastings, Nebr.
Foote, R.H. 1981. The artificial insemination industry. Pp. 13–39 in B.G.Brackett, G.E.Seidel, and S.M.Seidel, eds. New Technologies in Animal Breeding. Academic Press, New York.
Foster, W.R., and S.D.Bennett. 1984. Non-surgical embryo transfer between a grant zebra and domestic quarterhorse: A tool for conservation. P. 181 in Annual Proceedings of the American Association of Zoo Veterinarians. Hills Division, Riviana Foods, Topeka, Kans.
Goodrowe, K.L., J.G.Howard, and D.E.Wildt. 1986. Embryo recovery and quality in the domestic cat: Natural versus induced estrus. Theriogenology 25:156. (Abstract)
Gorman, J. 1980. Brave new zoo. Discover 1:46–49.
Hare, W.C.D. 1985. Diseases Transmissable by Semen and Embryo Transfer. Office Internationale des Epizost, Paris. 36 pp.
Heape, W. 1891. Preliminary note on the transplantation and growth of mammalian ova within a uterine foster-mother. Proc. R. Soc. London 48:457–458.
Hearn, J.P., and P.M.Summers. 1986. Experimental manipulation of embryo implantation in the marmoset monkey and exotic equids. Theriogenology 25:3–11.
Hodgen, G.D. 1983. Surrogate embryo transfer combined with estrogen-progesterone therapy in monkeys. J. Am. Med. Assoc. 250:2167–2171.
Kraemer, D.C., G.T.Moore, and M.A.Kramen. 1976. Baboon infant produced by embryo transfer. Science 192:1246–1247.
Kraemer, D.C., B.L.Flow, M.D.Schriver, G.M.Kinney, and J.W.Pennycook. 1979. Embryo transfer in the nonhuman primate, feline and canine. Theriogenology 11:51–62.
Kramer, L., B.L.Dresser, C.E.Pope, R.D.Dahlhausen, and R.D.Baker. 1983. The nonsurgical transfer of frozen-thawed eland (Tragelaphus oryx) embryos. Pp. 104–105 in Annual Proceedings—American Association of Zoo Veterinarians. Hills Division, Riviana Foods, Topeka, Kans.
Kydd, J., M.S.Boyle, W.R.Allen, A.Shephard, and P.M.Summers. 1985. Transfer of exotic equine embryos to domestic horses and donkeys. Equine Vet. J. Suppl. 3:80–83.
Lehn-Jensen, H. 1981. Deep freezing of cow embryos, a review. Nord. Vet. Med. 32:523–532.
Leibo, S.P. 1977. Preservation of mammalian cells and embryos by freezing. Pp. 311–344 in D. Simatos, D.M.Strong, and J.M.Turc, eds. Cryoimmunologie: Colloque, Dijon, 17–18 Juin 1976. Organis Editions de l’Institut National de la Sant et de la Recherche Medicale, Paris.
Leibo, S.P. 1981. Physiological basis of the freezing of mammalian embryos. Pp. 353–379 in M.E. Gershwin and B.Merchant, eds. Immunologic Defects in Laboratory Animals, Vol. 2. Plenum, New York.
Letvin, N.L., and N.W.King. 1984. Human disease: Acquired immune deficiency syndrome. Animal model: Acquired immune deficiency syndrome-like disease in non-human primates. Comp. Pathol. Bull. 16:5–6.
Lushbaugh, C.C., G.L.Humason, and N.K.Clapp. 1984. Spontaneous colorectal adenocarcinoma in cotton-topped tamarins. Comp. Pathol. Bull. 15:2, 4.
Mapletoft, R.F. 1984. Embryo transfer technology for the enhancement of animal reproduction. Biotechnology 2:149–160.
Mauer, R.R. 1978. Freezing mammalian embryos. A review of the techniques. Theriogenology 9:45–68.
Moore, H.D.M., R.C.Bonney, and D.M.Jones. 1981. Induction of oestrus and successful artificial insemination in the cougar, Felis concolor. Pp. 141–142 in Annual Proceedings—American Association of Zoo Veterinarians. Hills Division, Riviana Foods, Topeka, Kans.
Muhlbock, O., ed. 1976. Basic Aspects of Freeze Preservation of Mouse Strains. Gustav Fisher, Stuttgart, Federal Republic of Germany.
Myers, N. 1984. Cats in crisis! Int. Wildl. 14 (Nov-Dec):42–52.
Pope, C.E., V.Z.Pope, and L.R.Beck. 1984. Live birth following cryopreservation and transfer of a baboon embryo. Fertil. Steril. 42:143–145.
Rall, W.F., D.S.Reid, and C.Polge. 1982. Physical and temporal factors involved in the death of embryos that contain ice. Pp. 303–305 in F.Franks, ed. Biophysics of Water. Proceedings of a working conference held at Cirton College, Cambridge, June 29–July 3, 1981. John Wiley and Sons, New York.
Reed, G., B.Dresser, B.Reece, L.Kramer, P.Russell, K.Pindell, and P.Berringer. 1981. Superovulation and artificial insemination of Bengal tigers (Panthera tigris) and an interspecies embryo transfer to the African lion (Panthera leo). Pp. 136–138 in Annual Proceedings—American Association of Zoo Veterinarians. Hills Division, Riviana Foods, Topeka, Kans.
Renard, J.P., Y.Menezo, M.C.Pheron. 1982. Effect of lipid supplement on the viability of D7–D8 Ovine blastocysts after freezing and thawing (Abstract). In Embryo Transfer in Mammals, 2nd International Congress, Annecy. Foundation Marcel Merieux, Lyon, France.
Seager, S.W.J. 1981. A review of artificial methods of breeding in captive wild species. Dodo, Jersey Wildl. Preserv. Trust, 18:79–83.
Seidel, G.E., Jr. 1981. Superovulation and embryo transfer in cattle. Science 211:351–358.
Snyder, R.L., G.Tyler, and J.Summers. 1982. Chronic hepatitis and hepatocellular carcinoma associated with woodchuck hepatitis virus. Am. J. Pathol. 107:422–425.
Sreenan, J.M. 1983. Embryo transfer procedure and its use as a research technique. Vet. Rec. 112:494–500.
Storrs, E.E., C.H.Binford, and G.Migaki. 1980. Experimental lepromatous leprosy in nine-banded armadillos (Dasypus novemcinctus Linn). Am. J. Pathol. 92:813–816.
Stover, J., J.Evans, and E.P.Dolensek. 1981. Interspecies embryo transfer from the gaur to domestic holstein. Pp. 122–124 in Annual Proceedings—American Association of Zoo Veterinarians. Hills Division, Riviana Foods, Topeka, Kans.
Stuhlman, R.A. 1979. Diabetes mellitus. Animal model: Spontaneous diabetes mellitus in Mystromys albicaudatus. Am. J. Pathol. 94:685–688.
Trounson, A.O., and L.Mohr. 1983. Human pregnancy following cryopreservation, thawing and transfer of an eight-cell embryo. Nature 305:707–709.
Vietmeyer, N. 1984. From horses come zebras. Int. Wildl. 14(Nov-Dec):12–13.
Whittingham, D.G. 1975. Survival of rat embryos after freezing and thawing. J. Reprod. Fertil. 43:575–578.
Whittingham, D.G., S.P.Leibo, and P.Mazur. 1972. Survival of mouse embryos frozen to −196° and −269°C. Science 178:411–414.
Willadsen, S.M. 1979. A method for culture of micromanipulated sheep embryos and its use to produce monozygotic twins. Nature 277:298–300.
Willadsen, S.M. 1980. The viability of early cleavage stages containing half of the normal number of blastomeres in the sheep. J. Reprod. Fertil. 59:357–362.
Willadsen, S.M., C.Polge, L.E.A.Rowson, and R.M.Moor. 1974. Preservation of sheep embryos and liquid nitrogen. Cryobiology 11:560. (Abstract)
Willadsen, S.M., H.Lehn-Jensen, C.D.Fehilly, and R.Newcomb. 1981. The production of monozygotic twins on preselected parentage by micromanipulation of nonsurgically collected cow embyros. Theriogenology 15:23–30.
Wilmut, I. 1972. The effect of cooling rate, warming rate, cryoprotective agent and stage of development on survival of mouse embryos during freezing and thawing. Life Sci. 11:1071–1079.
Wilmut, I., and L.E.A.Rowson. 1973. Experiments on the low-temperature preservation of the cow embryos. Vet. Rec. 92:686–690.
Wolkomir, R. 1983. Draining the gene pool. Natl. Wildl. 21(Oct-Nov):24–28.
Yamamoto, Y., N.Oguri, Y.Tsutsumi, and Y.Hachinohe. 1982. Experiments in the freezing and storage of equine embryos. J. Reprod. Fertil. Suppl. 32:399–403.
Zeilmaker, G.H., ed. 1981. Frozen Storage of Laboratory Animals. Proceedings of a workshop at Harwell, U.K., May 6th–9th, 1980. Gustav Fisher, Stuttgart, Federal Republic of Germany. 193 pp.