14
Training Grants in the Chemical and Biological Sciences
Ronald T. Borchardt
University of Kansas
INTRODUCTION
In the 21st century, we must realize that academic programs, like companies, generate products (i.e., graduates, knowledge, technology) and have multiple clients (e.g., other academic departments, government, industry) that use these products. Academic programs also must continually update their information about their clients and their clients’ needs. For example, U.S. corporations in the 1980s and 1990s have undergone dramatic changes in how they conduct business. In addition to introducing new technologies into the processes of discovery, development, manufacturing, marketing, and distribution of products, U.S. corporations now require their employees to perform within highly integrated teams. Participants in these multidisciplinary teams need not only to be technically competent but also to have the interpersonal skills required to function in a team environment. Academic programs, particularly chemistry departments, have been slow to recognize that it is no longer sufficient to train a graduate student to be the world’s expert on a narrowly focused topic. Individuals with narrowly focused areas of expertise are often unable to function effectively on a multidisciplinary team. While depth of knowledge and experience in a particular field is still the most important component of good graduate education, students also must acquire scientific breadth and the interpersonal skills necessary to function as a team member.
In the 1980s and 1990s, academic units in the biological and pharmaceutical sciences recognized the need to change and effectively used predoctoral training grants provided by the National Institutes of Health (NIH) to implement this change. Multidisciplinary predoctoral training grants provide an excellent mechanism to bridge the gaps that often exist between academic units (e.g., departments of chemistry and biology and schools of pharmacy and engineering). These training grants also can serve to broaden the educational experiences of the trainees and to refine their interpersonal skills. However, establishing good multidisciplinary training programs requires that the students’ interests take precedence over the interests of the faculty and the academic unit. This means that faculty and/or academic leaders (e.g., department chairpersons) must be willing to step forward and think out of the box about graduate education. They must also be willing to be implementers of change in their home institutions.
As an example of programmatic change within an academic unit and of the role of NIH predoctoral training grants in facilitating this change, this article focuses on graduate education in the Department of Pharmaceutical Chemistry at the University of Kansas over the past 30 years (Figures 14.1 through 14.3). In particular, it underscores changes implemented in the mid-1980s to generate Ph.D. scientists who could compete effectively for jobs in the emerging biotechnology industry.
HISTORY OF PHARMACEUTICAL CHEMISTRY GRADUATE EDUCATION AT THE UNIVERSITY OF KANSAS
Pre-Biotechnology Revolution (1967 to 1990)
The Department of Pharmaceutical Chemistry (also called pharmaceutics at some universities) at the University of Kansas was founded in 1967 by the late Professor Takeru Higuchi. Professor Higuchi, who was trained as a physical chemist, entered the field of pharmaceutical chemistry in the late 1940s when he joined the faculty of the School of Pharmacy at the University of Wisconsin. After moving to the University of Kansas, Professor Higuchi built the predoctoral training and research programs in the Department of Pharmaceutical Chemistry around the philosophy that success in understanding drug actions, controlling drug delivery across biological barriers and to drug receptors, developing stable drug formulations, and creating methods for analysis of drug substances required a thorough knowledge of the basic principles of analytical, physical, physical organic, and organic chemistry. Because the pharmaceutical industry at that time was oriented largely toward the discovery and development of small-molecule-based drug candidates, Professor Higuchi’s philosophy was well received, and the department built strong relationships with this industry user of its products. These products included Ph.D. scientists as well as knowledge and new technologies focused on small molecules (see Figure 14.1).
In the 1980s, the pharmaceutical industry began to undergo dramatic changes resulting from the biotechnology revolution. The introduction of this new technology made it possible for the first time to produce large quantities of macromolecules (e.g., proteins) as drug candidates. This change began to affect our graduate program in the mid-1980s when some of these macromolecules reached preclinical development. Interestingly, this change in the industry coincided with my transfer from the Department of Biochemistry to the Department of Pharmaceutical Chemistry to replace Professor Higuchi, who had retired as chairperson. While I fully respected and supported Professor Higuchi’s educational philosophy, I also recognized the impact that biotechnology was having on the pharmaceutical industry and the need to implement change in our graduate program to accommodate the changes occurring in industry. The need for this change became more evident when biotechnology companies began to hire our Ph.D. graduates to participate in the preclinical development of macromolecule-based drug candidates. We quickly realized (based on feedback from our alumni) that our Ph.D. graduates were not properly trained to perform this development function. During the next five years (from approximately 1985 to 1990), biotechnology companies continued to hire our Ph.D. graduates but were forced to provide on-the-job training related to macromolecules for these new scientists (see Figure 14.2).
As chairperson of the department, I was then confronted with the delicate problem of implementing change in a program that was highly respected both nationally and internationally. I choose the words “delicate problem” because, while our department needed to begin to train Ph.D. scientists who could function effectively as members of development teams for macromolecule-based drug candidates in biotechnology companies, we needed at the same time to continue to produce Ph.D. scientists who could function effectively as members of development teams for small-molecule-based drug candidates in more traditional pharmaceutical companies.
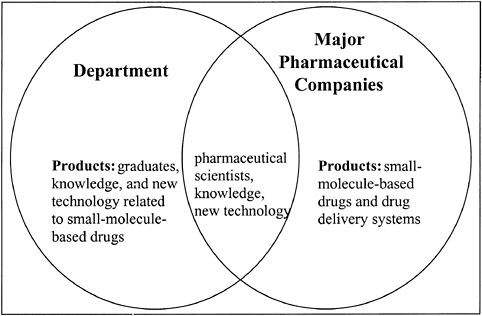
FIGURE 14.1 Pharmaceutical chemistry graduate education at the University of Kansas: 1967 to 1985.
Customarily, academic chairpersons have handled this type of problem by simply hiring additional faculty. While our department was able to add a few new faculty members in the late 1980s, the numbers were not sufficient to produce a dramatic change in our graduate program. The change that was needed involved placing increased research and educational emphasis on molecular biology, biochemistry, biophysical chemistry, and bioengineering as these disciplines apply to pharmaceutical chemistry. This shift was essential because Ph.D. scientists involved in the development of macromolecule-based drug candidates required different training than did Ph.D. scientists involved in the development of small-molecule-based drug candidates (see Figure 14.3).
To gain this training, it became necessary to expand beyond our department’s graduate program to create a multidisciplinary training program in pharmaceutical biotechnology. We recruited a subset of the faculty in the Department of Pharmaceutical Chemistry and then carefully selected additional faculty from the Departments of Chemistry, Biomolecular Sciences, and Engineering on our campus (Figure 14.4). In addition, we recruited scientists from biotechnology and major pharmaceutical companies. These scientists were selected because they had expertise in areas not available from our faculty (e.g., bioprocess engineering) and/or because they could serve as mentors for trainees during their industrial internships.
To facilitate the development of this multidisciplinary program in pharmaceutical biotechnology, the University of Kansas successfully competed for a predoctoral training grant from the National Institute of General Medical Sciences (NIGMS) in 1989.
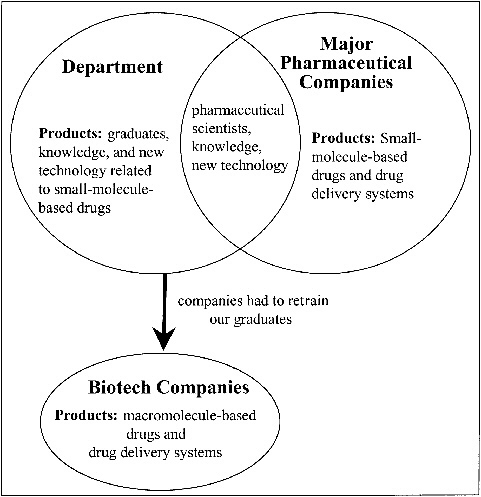
FIGURE 14.2 Pharmaceutical chemistry graduate education at the University of Kansas: 1985 to 1990.
Post-Biotechnology Revolution (1990 to Present)
This multidisciplinary training program in pharmaceutical biotechnology has as its primary objective “the training of pharmaceutical scientists who have the expertise to assist in the preclinical and clinical discovery/development of macromolecule-based drug candidates.” The program is administered by a steering committee composed of faculty representatives from the participating academic units (i.e., Departments of Pharmaceutical Chemistry, Chemistry, Molecular Biosciences, and Chemical Engineer-
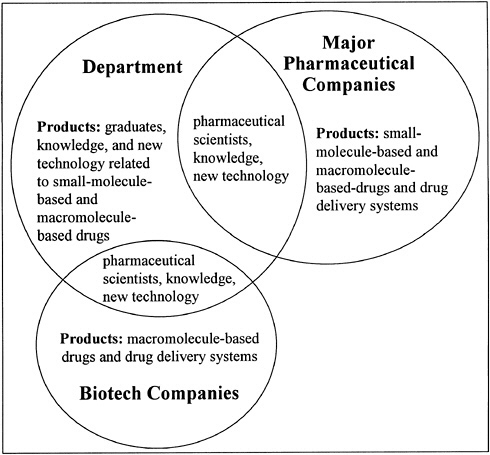
FIGURE 14.3 Pharmaceutical chemistry graduate education at the University of Kansas: 1990 to present.
ing). The trainees for this program are selected from graduate students enrolled in the participating departments who have completed at least one year of graduate education at the University of Kansas. The trainees continue as graduate students in their respective departments. They fulfill the requirements of their “home” graduate program and, ultimately, their Ph.D. degrees are awarded in these disciplines. However, as trainees in the Pharmaceutical Biotechnology Program, the students also must complete several additional requirements, including completion of “core” courses and courses in their area of specialization, participation in a biotechnology seminar and a biotechnology journal club, and completion of an industrial internship. Each of these requirements is described below in detail.
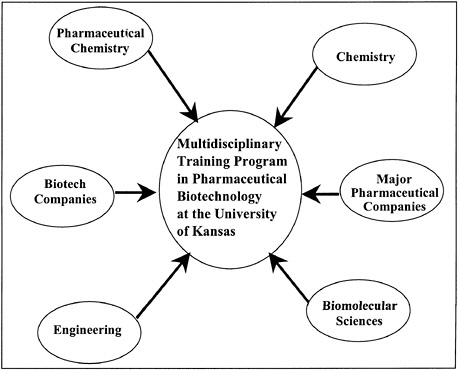
FIGURE 14.4 Participants in the multidisciplinary training program in pharmaceutical biotechnology at the University of Kansas.
Trainees in the Pharmaceutical Biotechnology Program are required to complete three “core” courses, one of which is in molecular biology. From this course, the students learn the language of this discipline, which helps them function effectively in multidisciplinary drug discovery development teams in biotechnology companies. The students also complete a course entitled “Advanced Pharmaceutical Biotechnology,” which increases their scientific breadth through exposure to all aspects of the discovery and development of macromolecules as drugs. Topics covered in the course include techniques used to clone and express proteins, scale-up of expression systems for proteins for manufacturing, characterization of the pharmacological and toxicological properties of the proteins, incorporation of proteins into appropriate dosage forms, and regulatory issues related to approval of the drug product by the Food and Drug Administration. Students are also required to complete a seminar course, “Issues in Scientific Integrity,” which addresses the multiple roles of scientists as researchers, authors, grantees, reviewers, inventors, employers/employees, teachers/students, and citizens. Trainees also complete two additional courses in their chosen areas of specialization (i.e., delivery, formulation, or analysis). These
courses are designed to provide the scientific depth that Ph.D. scientists need to successfully execute their technical role as a member of a multidisciplinary drug development team in a biotechnology company.
To facilitate communication among the trainees and the faculty members and to ensure exposure of the trainees to the external scientific community, the students participate in a monthly seminar series entitled “Pharmaceutical Aspects of Biotechnology.” Many of the seminar speakers are scientists from biotechnology and major pharmaceutical companies. These individuals provide the students with practical examples of the development of macromolecules as drug candidates. This seminar series also serves to expose the students to new scientific findings in the broad field of biotechnology.
Further exposure of students to the newer findings in biotechnology is provided through a biotechnology journal club. Each semester, a new topic (e.g., gene delivery, mass spectrometry in biotechnology) is selected, and the students are required to present and discuss papers relevant to this subject. This journal club has helped to provide additional breadth and depth in the students’ training.
Finally, the trainees must complete an industrial internship that is at least three months in duration. Typically, students undertake this requirement after they have completed their departmental Ph.D. preliminary examinations and are well into their dissertation research. Ideally, the internship allows them to continue some aspect of their dissertation research in an industrial laboratory or to complete a project that can be incorporated into their dissertation. Students often use the internship to learn new techniques or gain access to equipment that will enhance completion of their dissertation research. The success of this type of industrial internship is highly dependent on the involvement of knowledgeable faculty members who can identify appropriate mentors and projects in industry. These faculty members should also be actively involved in supervision of the student’s research in industry; ideally, the work should stem from a collaboration between a faculty member and an industrial scientist. Obviously, appropriate intellectual property agreements between the university and the company must be established to protect the freedom of the student ultimately to publish the data generated during the internship both in a dissertation and in scientific journals.
Through careful planning and coordination, approximately 50 students have completed industrial internships under the Pharmaceutical Biotechnology Program at the University of Kansas. All have been highly successful, particularly from the students’ perspectives. The students not only have been able to do cutting-edge research in pharmaceutical biotechnology that has resulted in high-quality publications but also have gained exposure to the inner workings of biotechnology companies that has influenced their career choices. Faculty have also benefited, since the collaborations with industrial scientists that began through student internships have frequently continued long after the students’ graduation.
In conclusion, the University of Kansas, through effective strategic planning and the support of an NIGMS predoctoral training grant, has been able to modify its graduate program in pharmaceutical chemistry to include aspects of biotechnology. This change was accomplished with the cooperation of the faculty from several academic departments, which allowed the creation of a multidisciplinary training program in pharmaceutical biotechnology. This program was structured in such a way that it did not detract from the graduate programs in the participating departments but, instead, helped to strengthen the departmental programs and make them more attractive to applicants.
The Future
To keep pace with ongoing changes in the pharmaceutical industry, the Department of Pharmaceutical Chemistry and the Pharmaceutical Biotechnology Program at the University of Kansas must
continue to plan strategically and to evolve when necessary to continue to be competitive in selling their products (pharmaceutical scientists, knowledge, technologies) to their clients (pharmaceutical and biotechnology companies). The most significant recent changes include the introduction of new technologies into drug discovery (i.e., high-throughput screening, combinatorial chemistry, bioinformatics, genomics, proteomics), the integration of drug discovery and development, and the globalization of drug discovery and development. Through industrial internships, both students in the Department of Pharmaceutical Chemistry and trainees in the Pharmaceutical Biotechnology Program are being exposed to the technology revolution as well as to “integration initiatives” recently undertaken by the pharmaceutical and biotechnology industries. However, finding ways to expose our students to the globalization of these industries has been more difficult.
To increase the exposure of our graduate students to science at an international level and to facilitate increased interactions between our students and students and faculty from international universities, we established a not-for-profit organization, Globalization of Pharmaceutics Education Network, Inc. (GPEN, Inc.), in 1996. This organization now includes 25 universities—6 in the United States, 4 in Japan, 1 in Australia, and 14 in Europe. GPEN, Inc., sponsors a meeting every 2 years; the first was held at the University of Kansas in 1996, and the second took place at Galenische Pharmazie ETH in Zurich, Switzerland, in 1998. The third GPEN meeting will be held at the University of Uppsala in Sweden in 2000. These 3-day meetings focus largely on presentations by graduate students and discussions about graduate education in the participating universities (2 days). One day is set aside for short courses presented by the faculty on timely topics not necessarily covered in the curricula of the participating universities. Industry representatives are invited to take part, but their participation is limited to that of observers.
In the future, GPEN, Inc., plans to facilitate the exchange of students and faculty between participating universities. Such exchange programs would further globalize the education of graduate students in pharmaceutics, so that they are prepared to function in the international and emerging multinational pharmaceutical companies.
CONCLUSIONS
From my experiences as a mentor of graduate students, as a department chairperson, and as a director of a predoctoral training grant, I offer the following conclusions. While these conclusions appear to be specific to pharmaceutical chemistry, I believe that they are also generally applicable to most areas of chemistry.
-
Changes in the pharmaceutical/biotechnology industry in the 1980s and 1990s have been rapid and dramatic, and additional changes are likely to occur in the future.
-
The skill base required of an ideal pharmaceutical scientist in today’s industry (Figure 14.5) is very different from that required of a scientist in the 1970s. The skill base required in 2010 will be very different from that required today.
-
Pharmaceutical scientists must continue to be highly skilled in their areas of expertise in order to be productive and respected members of project teams.
-
Pharmaceutical scientists today must be broadly trained so that they can communicate with scientists in other disciplines and work effectively on project teams.
-
Training grants provide an excellent mechanism to facilitate change in academic programs and to build multidisciplinary training programs that can expand the scientific breadth of the students as well as enrich their scientific depth.
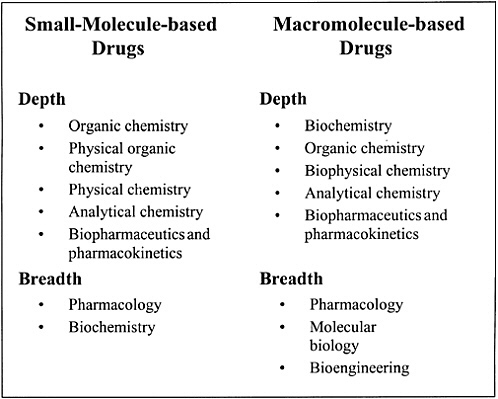
FIGURE 14.5 Scientific depth and breadth needed by scientists who develop small molecule-based drugs or macromolecule-based drugs.
-
Academic institutions have been slow to recognize the changes in the pharmaceutical industry and even slower to adjust their graduate programs to accommodate these changes.
The future strength of the scientific establishment in the United States is highly dependent on the willingness and the ability of academic units to change to accommodate the needs of their clients. Such change can arise only from effective strategic planning that focuses on the future and not the past.
RELATED READING
Ronald T. Borchardt, “Are Graduate Programs Training Pharmaceutical Scientists to Function Effectively in the New, Highly Integrated and Globalized Pharmaceutical Industry?” Pharmaceutical Research 14, 554-555, 1997.
Price Waterhouse Coopers, “Pharma 2005: An Industrial Revolution in R&D,” Price Waterhouse Coopers, United Kingdom, 1998.
R. Hirschmann, “Introduction,” pp. 1-5 in Integration of Pharmaceutical Discovery and Development, R.T. Borchardt, R.M. Freidinger, T.K. Sawyer, and P.L. Smith, eds., Plenum, New York, 1998.
DISCUSSION
Iwao Ojima, State University of New York at Stony Brook: I appreciate the kind of perspective that you brought and would like to get your insight about departments that are closely related to chemistry. In addition to the department of chemistry, you have at the University of Kansas a department of medicinal chemistry and pharmaceutical chemistry. So, I think you are in a position to observe and interact with those who are closely related. Can you comment on changes that have occurred in those two departments?
Ronald T. Borchardt: Let me begin by saying that we have a number of training grants at the University of Kansas, each in a different area. My presentation focused on the training grant in pharmaceutical biotechnology with the “core’ department for this training grant being pharmaceutical chemistry. We also have a training grant on campus in the area of medicinal chemistry/pharmacology with the core departments being medicinal chemistry and pharmacology and toxicology. There is also a chemistry/ biology interface training grant in which the core departments are chemistry and molecular biosciences.
Iwao Ojima: I am asking this question because I tried to hire a famous Russian physicist into our chemistry department, but I encountered a mind-set in the department againt the idea of hiring a physicist. I was shocked by this attitude. Do you know anything about the change in the chemistry department in Kansas?
Ronald T. Borchardt: One thing that I learned from being a department chair for 15 years was how to be somewhat diplomatic; therefore, I think it would be inappropriate for me to comment specifically about our chemistry department. However, I would say that our department, like many other chemistry departments, has gone through significant change in the past 20 years resulting from faculty retirements and the recruitment of young faculty members who are more receptive to multidisciplinary research and training programs.
P. Wyn Jennings, National Science Foundation: I want to comment on the multi-institutional idea that was raised previously for science and technology centers. I am not trying to evangelize with regard to our Integrative Graduate Education and Research Traineeship (IGERT) program, but I want to tell you that we do entertain multi-institutional proposals. Our experiences at this point—and we have several, somewhere between two and seven institutions involved in a single IGERT—is that an enlivened student is one who has studied for awhile in one institution, has gone away for 3 months to a year at another institution, and then has come back to the original institution. This individual is enlivened and infectious with enthusiasm. With regard to the international scene along this line, the IGERT program is negotiating a relationship with the megatraining centers in Germany and would like to be a participant likewise in programs elsewhere in the European Union. We are negotiating with them, and we are also involved with CNRS and the French. With regard to the need for foreign languages in different
disciplines, I saw one experiment at the graduate student level in which three students from three different disciplines discussed the same topic in one seminar. It was extremely exciting.
Ronald T. Borchardt: With respect to industrial internships, our students have found them to be very positive experiences. They return from these industrial internships much more enthusiastic and focused on completing their Ph.D. degree. I think that this change in attitude arises because they realize what they are going to do when they graduate and are excited about the future.
Another comment about industrial internships is that the university must put in place an agreement with the company that completely protects the student. Whatever the student is doing in industry must be nonproprietary.
J. Michael White, University of Texas at Austin: I appreciate the care with which you stated the nature of these external collaborations. We need to be careful about just sending students off for a summer, because it can be extraordinarily counterproductive.
Ronald T. Borchardt: In our program, we carefully pick the company and scientist/mentor in the company so that we develop a true research collaboration for the student. Industrial scientists have many exciting ideas, but often they don’t have time to pursue them. They are very excited about having the opportunity to mentor a graduate student.
James Nowick, University of California, Irvine: Let me start by commending you on the program that you have put together, as well as your presentation of it. Clearly, a lot of thought went into getting all of these areas together, supplementing the program when things were missing, and also responding to the needs of the industry. Irvine has a very zealous program of synthesis and natural-products-oriented synthesis. I would say that 90 percent or more of our students would have failed in being able to answer business-related questions. Yet, our students are gobbled up, particularly the ones from the straight synthesis background, by the pharmaceutical industry. It seems to me that there are multiple models for successfully training students. I would describe yours as more of a professional-school model; maybe the one for our synthetic organic chemists is more of an academic-department model, and they both seem to work. In our case, the students who come out and go on in the pharmaceutical industry quickly pick up the lingo and the issues about why the natural product they have synthesized isn’t active. I would hate to think that our students, because of the fantastic depth they get, are wholly deficient in their training.
Ronald T. Borchardt: I find that graduates of chemistry departments are in general very, very bright, and they quickly learn what is important in the pharmaceutical industry. However, I must say that I was bothered by some of the comments I heard from the graduate students earlier in the workshop, i.e., that some chemistry mentors will not let their students out of the laboratory to attend seminars or courses in biochemistry or molecular biology. If that faculty member were in my department, there would be holy hell to be paid, because I think that students have to be given the opportunity, and even encouraged, when they are in the university to explore outside their major areas of study. To function effectively in the pharmaceutical industry, they need not only depth in their training but also breadth.
Robert L. Lichter, The Camille & Henry Dreyfus Foundation: Thank you for anticipating my question, but maybe you could elaborate. I have heard of a number of people who have traveled around exploring the notion of externships in one or another form, whether in industry or somewhere else,
respond adversely to this idea. They see it as diverting students from the purposes for which they are in graduate school. I am wondering how either of you have dealt with it, to what extent have you run into it, and what the buy-in is for faculty—all of these kinds of ancillary issues.
Ronald T. Borchardt: When we developed the idea of internships in our training program, we decided that we were going to have our cake and eat it, too, in the sense that we are going to structure these experiences as research collaborations. As a result, these internships have ended up not extending the tenure of the students in our graduate program. In fact, I would say in some cases it may have expedited their graduate tenure, because they have gone to industry and done things that would have been difficult or impossible in the university.
Now, the other aspect of this, which I think is important, and which hasn’t been brought up, is the fact that many of our internships have resulted in the establishment of faculty-scientist collaborations that have persisted long after the student has graduated. So, these student internships give the younger faculty the opportunity to get to know people in industry and better understand what their students will be doing when they graduate.
I have yet to see a serious downside to these internships. If you talked to the 50+ students in our department who have participated in internships in the past 10 years, they would all say that they had very positive experiences. Our faculty fully support internships. It turns out that in most cases I am the one who orchestrated the internships because I have the industrial contacts, but that is my responsibility as the program director of this training grant.
J. Michael White: You mentioned the word “contacts.” I didn’t discuss industrial interactions with our center, but what I think is valuable to remember is that those contacts are really important, and they need to be made at a variety of levels, especially in large organizations. There are opportunities for them at all kinds of places. You can get into Glaxo-Wellcome, Motorola, and so on, but you have to sustain those contacts over a long enough period of time so that these companies are willing to engage in projects with you.
Ronald T. Borchardt: I think the key is that the industrial contact must be at the scientist level. If you start with a manager or vice president, he or she can arrange an “industrial experience” for the student, but it will not necessarily be in the form of a research collaboration.
However, I must add that involvement of the managers and the vice president is important, because they are the people who can provide the funding. Funding for the student can come in various forms. Some pharmaceutical companies prefer to hire the student as a part-time employee. In this case, you must guard against the student’s choice being driven by financial considerations. Sometimes companies give us unrestricted grants to supplement a student’s stipend. This supplement is used to cover living expenses while the student is completing the internship.
Victor Vandell, Louisiana State University: I am a proponent of multidisciplinary training, and I am also a product of that, as my background is in organic and analytical chemistry. I think there is a resistance toward that type of approach. I would like to bring up an ongoing problem to show that multidisciplinary training is important. I have been hearing about the discounting of chemists in industry relative to chemical engineers. It bothers me that I could have a Ph.D. in chemistry and be placed on the same level, if not below the level, of a person with a bachelor’s in chemical engineering. I perceive this as a future threat to chemists in industry. If we don’t do something to start incorporating some type of overlap in our training of chemists so that they can take some chemical engineering
courses or get more industrial experience to better compete, some industries might be leaning toward the ideology that they can actually replace us with chemical engineers. I want to know if anybody else perceives this as a threat.
Ronald T. Borchardt: I think that is a question that would be more appropriately directed to the chemistry faculty here than to me. I would make one real quick comment. As the pharmaceutical industry moves more to combinatorial chemistry and high-throughput screening, engineers become more and more important. Setting up robots to do the chemical syntheses and the biology testing is an engineering problem; thus, I would see an increasing need for graduates of engineering schools in the pharmaceutical industry.
Wayne Rohrbaugh, Ashland Chemical Company: If we can get out of the pharmaceutical focus for a moment, I think that there is a clear distinction between the contributions of a Ph.D. chemist and a chemical engineer in industry. Starting salaries, if that is what you are using as a benchmark, are primarily dictated by the law of supply and demand, and that is clearly the only thing that is going on there.
Participant: In other words, chemistry departments should go to the chemical engineering departments and talk about developing a course that takes students through the entire process of discovery through scale-up production. I think that would be of interest to chemical engineers as well as to chemists.
Robert Humphreys, National Starch and Chemical Company: I think that you would want to do that for the same reason that we would want to have a center, which is that people develop a common language. Chemical engineers and chemists do not speak a common language.
Wayne Rohrbaugh: I want to get away from the pharmaceutical focus a little bit because we have been looking at it so much. I have been sitting here for a day and a half, and I have been surprised at how introspective this group is. I have heard only tangential references to such things as the global economy, the impact of world market democratization, who your customers are, whether or not you have ever surveyed your customers and, by the way, your customers are industry, the chemical manufacturing industry in this country. According to the November 15, 1999, issue of Chemical & Engineering News, there are more than two times as many Ph.D. chemists in industry as in academia. If you look at fulltime faculty and subtract out the effect of postdocs, it is closer to three times as many Ph.D.s in industry as there are in academia. So, the customer is missing.
Another thing I didn’t hear anything about was the competitiveness of the U.S. chemical enterprise in the next 20 years. Are we going to be in existence in the next 20 years? What is the contribution of the chemical manufacturing industry to the gross domestic product of the United States? I can tell you it is quite substantial. I heard no description here whatsoever of the demographics of your placement of graduates as a function of industry sector. There is a very heavy emphasis on pharmaceutical research in this country. It is being supported by funding from the National Institutes of Health which is understandable, but the vast majority of National Science Foundation funding, and substantial amounts of Department of Energy funding are biotechnology related. And that accounts for 30 percent of the total research and development funding for the chemical enterprise in this country. The other 70 percent comes from industry. Clearly, the pharmaceutical industry is an important contributor to the gross domestic product, but it is only a fraction of the total chemical manufacturing contribution. I think you are neglecting the rest of us.
How many pharmaceutical biotechnology Ph.D.s will be needed in the next 10 to 20 years? Has that study been made? How many are you producing? What is happening with the law of supply and demand? Are those people going to be employable? Someone mentioned that he had to adjust his career many times; first he became a biochemist and then a pharmaceutical chemist. Was it by choice? How many of you in here started out as chemistry majors wanting to become biochemists? And how many are becoming biochemists? I think there is a fundamental bias right now for biotechnology, as induced from outside resources, the places to which you are going traditionally for funding. I am saying that the U.S. chemical industry is there also to provide funding. If we could break down the barriers of the university front office, we could work with you.
The majority of the U.S. chemical manufacturing industry, from my perspective at least, is not biotechnology related. It feels somewhat disenfranchised by the U.S. educational system, and we are becoming more enamored with European graduate schools. I want to make these comments and hear if we can get any discussion about them. Perhaps you should look at some of these data in the future in these kinds of discussions so that you can see where your product is going, not necessarily today, but in the next 20 years, and determine if you are prepared to supply that pipeline.
Ronald T. Borchardt: Let me take a minute to defend the content of my presentation. You must understand that I come from a department whose primary customer is the pharmaceutical industry. So, I think your question is probably more appropriately directed to the representatives of the chemistry departments in the audience.
Robert Humphreys: I want to pick up a little on what Ron said, particularly with respect to the idea of having a strategic plan. This forces you to develop a good model that other universities can look at, because the plan makes you look at who and where your customers are and, in this particular case, it happens to be pharmaceutical companies. You have to understand where the companies are going and what is driving their actions, in other words, understand your customers and in some cases maybe understand your customers’ customers.
There are a lot of universities in this country and in the world competing with each other. National Starch spends a lot of money at universities outside the United States and for some very good reasons. This competition sets up a sort of war, just like that seen in industry, where the universities try to differentiate themselves. For example, if I look at Kansas, the first thing I think of is Dorothy from The Wizard of Oz. You are out there in the middle of the country and far from a pharmaceutical company. Yet, you have a need to differentiate yourself so when decisions are being made by a pharmaceutical company about whom to work for or work with and where its money should go, one thing that comes to mind right away is Kansas. Right?
Apparently you have been very successful based on the group of customers that you put up on the projector screen. You went through a strategic analysis and understood your customers in order to be successful in developing a plan and implementing it, and you differentiated yourself accordingly. This is the kind of model I think would be important for other universities that are thinking about how to attract and maintain a good base of industrial funding.
Another example of a university that has differentiated itself is Southern Mississippi, which went into the area of water-soluble polymers when everyone else was focusing on materials science. They have done a very good job of advertising it and attracting industrial funding for that reason. So, if this is the kind of thing you are interested in, I think that is a good model. My question would be what prompted you to go after them like that?
Ronald T. Borchardt: During my tenure as a department chair, I became a strong advocate of strategic planning. Our planning process started in 1984 when the faculty participated in a retreat. At that retreat, we asked ourselves, Who are our customers? What services are we providing to our customers? How are their worlds changing? This exercise was a very healthy experience. From this retreat, we developed a strategic plan that we now update annually.
Lynn Melton, University of Texas at Dallas: I am delighted by the compliments you are getting on your programs. I would like to remind people about the doctor of chemistry program at the University of Texas at Dallas, which has been in operation since 1983. We have had a 90 percent direct placement into industry and probably 60 industrial practicum students by now with 50 graduates. We asked our industrial friends what their jobs were like, and then we designed a curriculum to produce students who would be successful in the chemical process industry. We would be delighted to have any of you come on campus and find out whether our ideas are exportable to you. All students have an industrial internship of 9 to 12 months, paid for by industry, in which they are mentored as an industrial problem solver and solve problems to the benefit of the company. A lot of this material is available to you. We have taken it through the pilot stage and will be glad to share it.
Kathleen C. Taylor, General Motors: I think, Ron, you have managed to press the industrial hot button in this last session. I want to congratulate you on bringing up the globalization issue and the initiative that you have started. I think it is one of the most important things I have heard about at this meeting. I was interested to hear that foreign universities seem to be able to find a way to send students to work with you for these symposia. My question is, what have you learned in working with those institutions that is different, pro and con, that we should try to capture in our work in view of trying to bring globalization into the educational process?
Ronald T. Borchardt: I think the real question is what our students have learned, because this program is focused on providing new experiences for our graduate students.
Ernest L. Eliel, University of North Carolina: I am a schizophrenic in that I am both an elitist and an anti-elitist. Most of the time in these meetings, I put on my anti-elitist hat. I must now put on my elitist hat. In looking around the room, I have noticed that there are about a dozen universities in this country that have well-known chemistry departments that are not represented. I agree that one of the important functions of a university is to serve the student, and serving the student includes preparing him or her for a job. But there is another function of the university, which I have not heard about, and that is to advance the frontiers of knowledge. I think we must never forget that this is one of the purposes of the university and that we cannot be exclusively the handmaidens of industry. Universities have a divided role, and in our department, we have faced up to this divided role.
We have some faculty members who are collaborating with industries and even have their own businesses. Some also have very big interdisciplinary grants and turn out students in applied science fields who are readily hired. Others are doing synthesis, and their students are also readily hired. We also have some members on our faculty who are doing fundamental research whose impact cannot be seen perhaps for 10 or 20 years. If we stop doing fundamental research or research that does not yield an immediate return, I think we should close all of our universities and turn our attention to trade schools.
Ronald T. Borchardt: I fully agree with what you just said.















