Merging Living Cells and Microsystems Engineering
MEHMET TONER
Harvard Medical School
Massachusetts General Hospital
Boston, Massachusetts
Microfabrication techniques that have revolutionized the electronics industry are about to revolutionize the pharmaceutical, biotechnology, and biomedical device industries. Photolithography, etching techniques, and deposition methods can create large numbers of microscopic features on silicon or glass substrates. Among these features are biochemical reaction chambers, bioseparation channels, arrays of biological molecules, integrated microelectronics, micropumps and microvalves to control fluid movement, and many other components. These features can also be combined to create fully integrated devices that perform sample preparation, separation, detection and/or analysis, as well as drug delivery and in situ mechanical sensors. The two leading applications of microfabrication in biology are (1) “genes-on-a-chip” to monitor the expression level of potentially all genes in humans or various model systems and organisms simultaneously and (2) “lab-on-a-chip”-type devices to perform biochemistry in microchambers (Voldman et al., 1999).
Equally exciting is that the biomedical application of microfabricated devices is no longer limited to nonliving systems, such as genes-on-a-chip or lab-on-a-chip. Recent advances in understanding cellular behavior in microenvironments are paving the way toward the development or living microdevices. Cellular behavior is significantly influenced or dictated by the characteristics of the local environment: the presence and location of other cells; the chemistry, composition, and texture of the extracellular matrix; and the composition and temporal variation of soluble factors. As Figure 1 shows, the merger of living cells and microdevices involves exquisite control over these three fundamental determinants of cellular behavior.
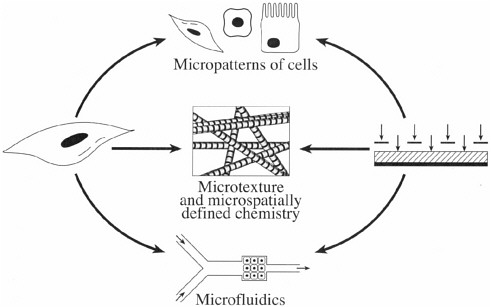
FIGURE 1 The merger of living cells and microsystems engineering involves control of three key ingredients of cellular environment, namely, cells, extracellular matrix, and soluble factors.
MICROSYSTEMS ENGINEERING IN BIOMEDICAL SCIENCES
This section is a brief overview of microsystem tools as applied in biology, especially in living systems. Emerging living cell-based devices are expected to become key technologies in twenty-first century medicine with a broad range of applications, from diagnostic, tissue engineered products to cell-based, high-throughput drug screening tools to basic cell biology tools. Achieving these challenging goals will depend heavily on processing techniques at the micron scale.
Microfabrication techniques are based on photolithography, which enables the creation of precise physical structures with micron scale dimensions (Voldman et al., 1999). Combined with the deposition of metal films, micromachining, etching, and bonding, microfabrication has been widely used for a large number of microdevices, some of them, such as semiconductors, very complex. In some of the early applications of microfabrication in biology, in fact, these processes were used to produce microtextured surfaces to determine the response of cells to well defined micron-scale disturbances in their environment, as well as to confine cells to micron to millimeter regions to probe cellular motility.
Despite the powerful advantages of traditional tools of microfabrication for the development of biological and medical microdevices, the usefulness of these techniques when merged with living cells is limited because of the nonbiological nature of many of the processing steps associated with photolithography. To address these limitations, over the last decade a number of critical advances have been made in the use of microfabrication in cellular systems. Collectively, these advances have dramatically redefined the landscape of microfabrication in biology and medicine and have resulted in the emergence of a new field called “bioMEMS” (bio-microelectromechanical systems).
Because living cells respond to their environment, the chemical nature of the surface is of the utmost importance in how cells behave in contact with artificial surfaces or scaffolds. Surface chemistry techniques have been developed to tailor the interaction of cells with surfaces (Kleinfeld et al., 1988). Self-assembled monolayers (SAMs) with various end-group functionalities to which cells attach provide an excellent tool for creating engineered surfaces for living cells. Various functional groups, such as extracellular matrix proteins (including specific peptides sequences), are used to control the preferential attachment of cells to surfaces. Conversely, fluoroalkylsilane SAMs provide surfaces that are nonadhesive to cells and proteins. These techniques have quickly become ubiquitous in bioMEMS and have greatly influenced our ability to create cellular micropatterns (Whitesides et al., 2001).
A major breakthrough in bioMEMS has been the development of so-called “soft lithography,” which uses poly(dimethylsiloxane) (PDMS) to generate micropatterns (Whitesides et al., 2001). PDMS is essentially a transparent rubber that is ideally suited for biological applications because it is chemically unreactive. Soft lithography is based on a PDMS negative replica mold lifted-off from a microfabricated master wafer. The PDMS replica is then dipped in an ink solution containing SAMs and microstamped onto gold or silver-coated surfaces to transfer the SAMs to the regions where the PDMS stamp contacts the substrate. Because SAMs with many different functional end-groups can be generated easily, this approach enables engineering of surfaces at the micron scale with a large assortment of chemistries containing adhesive and nonadhesive microdomains. These engineered surfaces provide excellent platforms for cell attachment and culture.
In another application, the microstructured PDMS master can be brought together with a large variety of surfaces to seal points of contact hermetically; this is possible because of the highly conformal nature and hydrophobicity of PDMS. Using this technique, complex microfluidic systems can be designed. The microfluidic chambers can then be used either to perform surface chemistry on exposed areas or to pattern cells directly on surfaces (Folch and Toner, 2000). The generation of three-dimensional microfluidic structures provides flexibility and variety in the types of cellular systems that can be generated using these tools (Whitesides et al., 2001).
Another variation of PDMS technology involves curing PDMS in microfluidic networks to generate very thin self-sealing stencils that contain complex geometric arrangements of holes. When these stencils are contacted with a surface, the exposed regions of the surface can thereafter be modified, either through various surface chemistry techniques or by directly attaching living cells. The biologically compatible nature of PDMS and its ease of fabrication (e.g., much less stringent clean-room requirements than photolithography) have made soft lithography a very widely used and, in most instances, the preferred approach in the development of cellular microsystems.
SOME CURRENT AND FUTURE APPLICATIONS OF LIVING MICRODEVICES
The ability to integrate cells with microdevices is important for controlling cellular interactions on a subcellular level, for obtaining highly parallel, statistically meaningful readouts over large cell populations, and for miniaturizing instrumentation for minimally invasive, portable, fast, inexpensive devices. The benefits of integrated, miniaturized systems are high surface-area-to-volume ratio, high-throughput screening capabilities, smaller required volumes of reagents and samples, integration with electronics, and potential automation with a consequent increase in reliability and decrease in cost. The rich assortment of living microsystems that can be built using bioMEMS techniques have important applications in fundamental cell biological studies, tissue engineering, cell separation and culture devices, and drug discovery.
By precisely controlling the shape and type of the extracellular matrix of cells using micropatterned islands of cell adhesive and nonadhesive molecules, it is now possible to investigate how cells respond to their environments and determine what controls cellular phenotype. Micropatterning techniques can also be used to control the position of multiple cell types with respect to each other to investigate how homotypic and heterotypic interactions between cells regulate differentiation and the functioning of engineered tissues (Folch and Toner, 2000).
Microfabrication enables the creation of complex tissue-engineered products that can be used to replace or augment failing organs and tissues. The number and variety of microfabricated tissue-engineered products have increased rapidly in the last several years: microporous biocapsules to provide immunological protection to pancreatic islets; micropatterned hepatocytes and mesenchymal cells as key components of liver assist devices; microtextures basement-membrane-type structures to create skin with proper surface texture; and microspatial control of the distribution of vitronection or other extracellular matrix proteins to promote the formation of mineralized bone-like tissue in vivo (Bhatia and Chen, 1999).
With recent advances in stem cell biology, it has become clear that the cell source for many, if not all, tissue-engineered products will be obtained from
stem cells. The primary challenge for stem cell biology is to determine biological and physical cues leading to the differentiation of these cells into functional tissue units. Microfabricated cell-culture devices will provide tools for engineering well controlled cellular environments, including microtexture, chemistry, soluble stimuli, and physical forces, to help in generating information about the conditions that lead to the differentiation of stem cells into functional tissue units.
Microfabricated structures are also used to measure individual cell mechanics in a population of cells as they pass through microbarriers. The same concepts can also be used to separate cells, based on size and shape or based on the expression of various surface receptors, by coating the microbarriers with different cell-attachment molecules. Another recent innovative application of microfluidic systems is the development of the so-called “reproductive chip” (Glasgow et al., 2001). As this technology evolves, it will become possible to move mammalian oocytes and embryos through various microchannels to perform a multitude of processing steps, such as microinjection of a sperm for fertilization of an oocyte or drilling of the zona pellucida to enhance embryo hatching, all without the need for cumbersome handling and manipulation procedures in a clinical setting.
Biosensors that incorporate living cells have the added advantage of rapidly monitoring the presence of toxic molecules or environmental pollutants, including biowarfare and chemical warfare agents (Pancrazio et al., 1999). Micropatterning provides the capability of establishing a network of cells, such as neurons, to investigate the effects of pollutants on the collective behavior of cells. Cell-based biosensors enable monitoring of the physiological behavior of an analyte of interest.
As we enter the postgenome era during which a massive amount of data concerning the intricate genetic machinery has been assembled, we are poised to decipher the complexity of hierarchical interactions that keep an organism at homeostasis. The best approach to accomplish this goal is a holistic, rather than reductionist, approach that monitors the behavior of cells and their genes in real time under multiple conditions, such as mimicking disease, trauma, development, and so forth. Cell chips are being developed to monitor simultaneously the behavior of living cells exposed to a multitude of environmental conditions using microfluidic and micromixing techniques. The same systems could be used to monitor the metabolic activity of cells. Thus, it is foreseeable that scientists will soon be able to observe simultaneously gene expression levels and the metabolism of massively parallel arrays of cells.
Massively parallel cell-based sensors will also be used to screen large quantities of drugs rapidly, which will dramatically expedite the drug discovery process, which will rely much less on animal models (Kapur et al., 1999). These systems will inevitably involve genetically engineered cells to monitor gene expression profiles in response to exposure to various drugs and toxins. They
will also include multiple cell types to investigate complex interactions between cells from different tissues. The ability to use living cells will also dramatically elevate the biological content of the screening. A large library of molecular probes is already available for monitoring cell function, gene expression, protein levels, and subcellular structures. It is foreseeable that a good part of preclinical and clinical trials will soon be performed using so-called “mouse-on-a-chip” or “human-on-a-chip”-type devices. These sophisticated microdevices will contain microengineered tissue units coupled to each other by complex microfluid-handling networks. Microfluidic mixing systems will also precisely regulate the composition and concentration of drugs to be tested and reduce the cost of drug development in the pharmaceutical industry.
This concise overview describes some of the key advances and challenges related to the coming merger of microfabrication technology and living cells and a sampling of a broad range of exciting opportunities and promises in biology and medicine.
REFERENCES
Bhatia, S.N., and C.S. Chen. 1999. Tissue engineering at the micro-scale. Biomedical Microdevices 2:131–144.
Folch, A., and M. Toner. 2000. Microengineering of cellular interactions. Annual Review of Biomedical Engineering 2:227–256.
Glasgow, I.K., H.C. Zeringue, D.J. Beebe, S.-J. Choi, J. Lyman, N.G. Chan, and M.B. Wheeler. 2001. Handling individual embryos using microfluidics. IEEE Transactions on Biomedical Engineering 48:570–578.
Kapur R., K.A. Giuliano, M. Campana, T. Adams, K. Olson, D. Jung, M. Mrksich, C. Vasudevan, and D.L. Taylor. 1999. Streamlining the drug discovery process by integrating miniaturization, high-throughput screening, high-content screening, and automation on the CellChip™ system. Biomedical Microdevices 2:99–109.
Kleinfeld D., K.H. Kahler, and P.E. Hockberger. 1988. Controlled outgrowth of dissociated neurons on patterned substrates. Journal of Neuroscience 8:4098–4120.
Pancrazio, J.J., J.P. Whelan, D.A. Borkholder, W. Ma, and D.A. Stenger. 1999. Development and application of cell-based biosensors. Annals of Biomedical Engineering 27:697–711.
Voldman, J., M.L. Gray, and M.A. Schmidt. 1999. Microfabrication in biology and medicine. Annual Review of Biomedical Engineering 1:401–425.
Whitesides, G.M., E. Ostuni, S. Takayama, X. Jiang, and D.E. Ingber. 2001. Soft lithography in biology and biochemistry. Annual Review of Biomedical Engineering 3:335–373.






