Appendix H
Overview of the Vijan Colorectal Cancer Screening Model Sandeep Vijan, M.D., M.S.
SLIDE 1

SLIDE 1 NOTES: We used this model in a paper published in 2001 (Vijan et al., 2001), but since that time we have made improvements which will be described here.
I would like to acknowledge Erica Wong, who assisted me on early stages of this project and Rodney Hayward and Tim Hofer, who provided guidance along the way.
SLIDE 2

SLIDE 2 NOTES: Our model uses a Markov cohort structure that produces expected endpoints of total lifetime costs, life expectancy, and colorectal cancer incidence and mortality. It is designed to simulate the natural history of colorectal cancer based on the experience of the U.S. population. We did not stratify the analysis by sex or race.
SLIDE 3
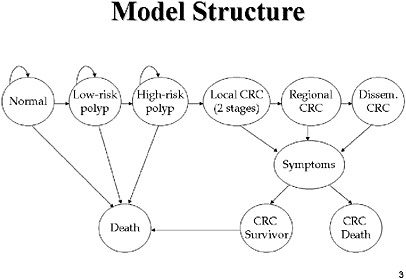
SLIDE 3 NOTES: The graph shown here shows a simplified view of how we modeled the natural history of CRC in the absence of screening. Patients are distributed initially into cohorts based on prevalence data from the literature. A percentage of the population is assigned to each of the different states at age 50. In the absence of screening, all CRCs are detected through the presentation of signs and symptoms. What happens after detection—whether the patient dies from CRC or from some other cause—depends on the stage at which the symptoms developed.
SLIDE 4

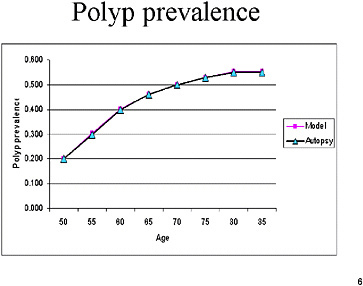
SLIDE NOTES 4: As with other models presented today, the prevalence of polyps is based on autopsy studies. We assumed a fixed time of transition from polyp to cancer of 10 years, based on the sigmoidoscopy studies that have shown a 10-year degree of protection from a sigmoidoscopy.
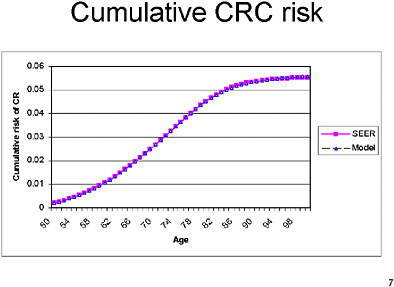
Cancer incidence in this model was based on SEER data from the early 1990s. We chose that period because data from earlier decades might not reflect changes in risk factors and natural history over time. However, incidence data from the 1990’s might be affected by the more frequent use of CRC screening in the 1990’s. In our published work we assumed that 75 percent of cancers arise from polyps that are visible on colonoscopy. The rest arise either de novo or through flat adenomas which are not seen. More recently, however, we have changed our assumptions to 100 percent arising from polyps. That change has a major impact on the relative cost-effectiveness of different strategies, because strategies that are better at detecting cancers than at detecting pre-cancerous polyps (e.g., FOBT) do not compare as favorably with strategies that can detect pre-cancerous polyps.
We assume a fixed dwelling time for a localized CRC of 2 years, and for a regional lesion of 1 year. We also assume that all individuals with disseminated cancer are symptomatic in the year they transition to that stage.
Our mortality rates are taken from life tables for the same time period as the incidence data. Our life-expectancy for the population is therefore lower, in the absence of screening than other models. Over the past 10 years, life expectancy in the US has
SLIDE 8

SLIDE 8 NOTES: We model compliance as an all-or-nothing phenomenon. That is, a person either adheres perfectly to the screening strategy or does not participate at all. In our most recent version of the model (used for the exercises in this workshop) we assumed that follow-up colonoscopy after a positive screening test is independent of the initial screening test. At present, we believe the best estimate of follow-up compliance is 75%.
We have found that that there are major impacts on the cost-effectiveness of strategies involving sigmoidoscopy and FOBT if adherence to follow-up is assumed to be less than 100 percent. For example, as shown above, assuming 75% compliance with follow-up among individuals who comply with the screening test reduces the effectiveness of FS and FOBT screening by 81 percent compared with perfect compliance. That is less than the reduction in effectiveness when compliance with follow-up is set at 25%.
SLIDE 9

SLIDE 9 NOTES: In our published study, we assumed that all adenomas discovered by sigmoidoscopy would go to follow-up by colonoscopy and ultimately to surveillance. However, in the present version we assume that only those greater than or equal to 10 mm would go on to follow-up and surveillance. That change has an enormous impact on the cost of this intervention.
SLIDE 11

SLIDE 11 NOTES: The Panel on Cost-Effectiveness in Medicine (Gold et al., 1996) recommends that cost-effectiveness analyses take a societal perspective. As with all of the models described in the current workshop, we took a third-party payer approach with respect to measuring costs. That means that we did not include the value of lost work time (productivity costs) and other costs (e.g., transportation) associated with obtaining screening, follow-up and surveillance examinations. The productivity costs associated with colonoscopy are likely to be disproportionately higher, since that test requires a separate driver to transport patients to and from the facility because the patient must be sedated for the procedure.
SLIDE 12

SLIDE 12 NOTE: Our main validation exercise was to compare the results of our model against those for the Minnesota FOBT trial (Mandel et al., 1993; Mandel et al., 1999). When we applied the test and population characteristics in that study, we predicted a 39% reduction in CRC mortality over a 13-year follow-up. Our model predicted a greater decrease in the incidence of colorectal cancer than occurred in the Minnesota trial, which may account for the differences in mortality that we found (Mandel et al., 2000). However, our results fall within the 95 percent confidence interval for the Minnesota study’s observed mortality reduction.
Compared with retrospective cohort studies that examined flexible sigmoidoscopy, we predicted lower mortality on the whole. We are currently examining why our model differed from those studies.
SLIDE 13

SLIDE 13 NOTES: No notes.
REFERENCES
Gold MR, Siebel JE, Russell LB, Weinstein MC, Editors. 1996. Cost-Effectiveness in Health and Medicine. New York: Oxford University Press.
Mandel JS, Bond JH, Church TR, Snover DC, Bradley GM, Schuman LM, Ederer F. 1993. Reducing mortality from colorectal cancer by screening for fecal occult blood. N Engl J Med. 328(19): 1365–1371.
Mandel JS, Church TR, Bond JH, Ederer F, Geisser MS, Mongin SJ, Snover DC, Schuman LM. 2000. The effect of fecal occult-blood screening on the incidence of colorectal cancer. N Engl J Med. 343(22):1603–1607.
Mandel JS, Church TR, Ederer F, Bond JH. 1999. Colorectal cancer mortality: Effectiveness of biennial screening for fecal occult blood. J Natl Cancer Inst. 91(5):434–437.
Vijan S, Hwang EW, Hofer TP, Hayward RA. 2001. Which colon cancer screening test? A comparison of costs, effectiveness, and compliance. Am J Med. 111(8):593–601.