SLIDE 2

SLIDE 2 NOTES: I was asked to answer, or try to answer, 5 questions related to compliance with colorectal cancer (CRC) screening. This presentation represents an update on reviews I have conducted with colleagues in the recent past (Peterson and Vernon, 2000; Vernon, 1997). The answers to some of the questions, such as the first two, are straightforward, and we have a reasonable amount of published data to draw on in answering those questions.
The 3rd question requires some extrapolation from existing data because it hasn’t been studied directly. I will share some thoughts about how we might arrive at a best guess, but I think this question warrants discussion.
There aren’t much published data to address the 4th question for any cancer screening test. But I will summarize the available data.
And to answer the 5th question, I plan to turn the podium over to my colleague Dr. Seeff, who has led a study by the CDC that may provide insight to help us answer this important question.
SLIDE 3
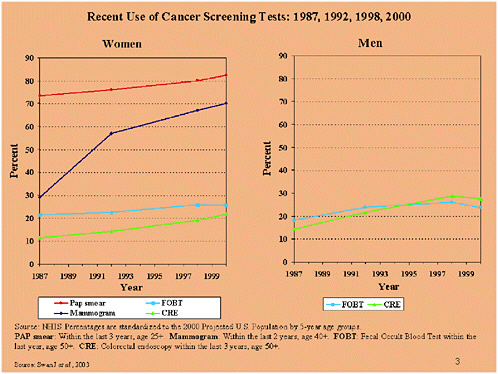
SLIDE 3 NOTES: What is known about the levels of, and patterns over time in, participation in CRC screening in the U.S. population?
There are 2 national data sources that have collected self-report data on use of CRC screening tests—the National Health Interview Survey (NHIS) and the Behavioral Risk Factor Surveillance System (BRFSS). These two surveys use different data collection methods. The NHIS is a face-to-face interview, and the BRFSS is a telephone interview. There also are differences in the question wording and the time intervals asked about, both within and between surveys. Another survey conducted by the NCI called the Health Information and Trends Survey (HINTS) was completed by the NCI and the data are forthcoming. So we are beginning to get a more complete and up-to-date picture of CRC screening test use.
Data on CRC test use have been collected by the NHIS since 1987. The data show that, compared with other types of cancer screening tests, tests for colorectal cancer are infrequently used among both men and women, and that their use is increasing only gradually. In 2000, less than 30% of the population had had a fecal occult blood test or endoscopy within the time interval recommended by the American Cancer Society.
At present, the available survey data do not permit a detailed look at the patterns of screening over time and across individuals. For example, whether a 60 percent screening rate in the population in each year represents the same individuals over time, or different
individuals, is critical to assessing the impact of compliance on screening costs and effectiveness. One survey currently in analysis at NCI may give some hints, because it asks individuals not only about the most recent screening encounter, but also about the next most recent. And the BFRSS may ask more refined questions about such patterns in future surveys.
SLIDE 4
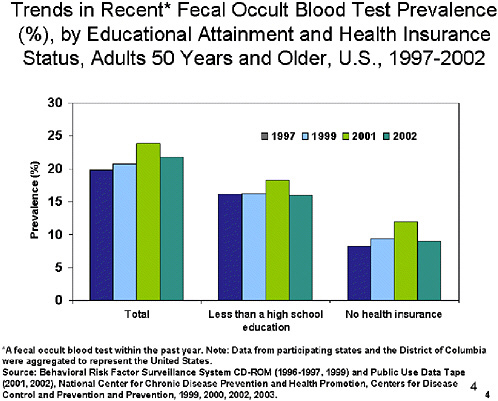
SLIDE NOTES 4: Data from the other national survey, the BRFSS, cover the time period 1997 to 2002. Despite an increase in FOBT use among both men and women from 1999 to 2002, prevalence remains low.
This slide also includes data that, in part, address the 2nd question: “What factors affect adherence to different kinds of technologies?”
Adults with less than a high school education are less likely to report a recent FOBT. And the prevalence for adults with no health insurance is approximately 10 percentage points lower than the prevalence for all adults—less than 10% in every years except 2001.
SLIDE 5
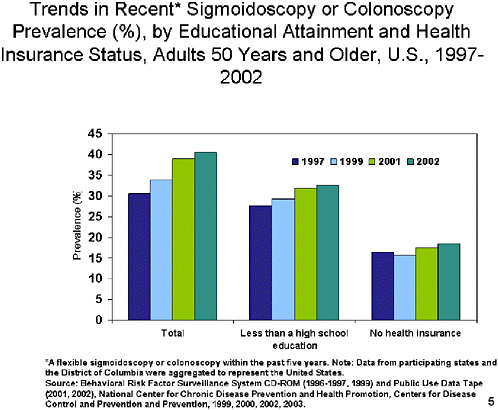
SLIDE 5 NOTES: The prevalence of sigmoidoscopy (SIG) or colonoscopy within the past five years, while low, increased from 30% to 40% between 1997 and 2002. The prevalence and the rate of increase are higher than for recent FOBT.
As with FOBT, adults with less than a high school education were less likely to report SIG or colonoscopy than all adults. And the prevalence for adults with no health insurance is approximately 15 percentage points lower than the prevalence for all adults, and is less than 20% in all years.
SLIDE 6

SLIDE 6 NOTES: This slide summarizes the results of research on factors that are associated with having a CRCS test. Factors are similar for FOBT, SIG, and COL. One of the messages here, and in the prior 2 slides, is that access to medical care—such as having health insurance, a healthcare provider, or frequent contact with the medical care system—is an important factor in increasing the chances of being screened for CRC.
These factors are similar to those for getting mammography screening and Pap testing.
Another factor that has not been looked at in national surveys but that is strongly associated with adherence to cancer screening recommendations, including CRC screening, is having a physician recommend the test. Data from the National Cancer Institute’s Health Information and Trends Survey (HINTS) of 2003 are currently being analyzed, but it appears that a small proportion of patients report having their physicians recommend colorectal cancer screening. So, there is room for improvement on that score.
SLIDE 7
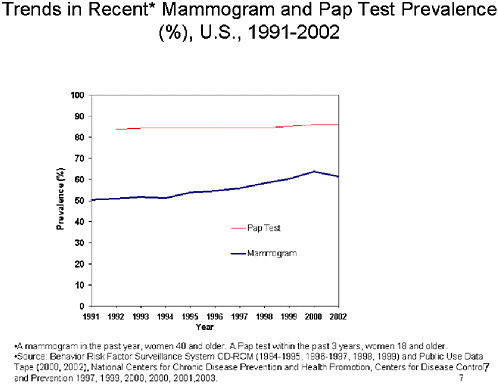
SLIDE 7 NOTES: What is our “best guess” about the level of compliance that might be expected with new screening technologies such as DNA and virtual colonoscopy?
I think that the best way to address this question is by looking at the prevalence and trends in prevalence for the use of other cancer screening tests and associated technologies.
Of all cancer screening tests for which we have data, Pap testing prevalence is the highest. Rates have been steady at ~85% since 1992. The prevalence of women reporting a recent mammogram has increased steadily since 1991 and was 62% in 2002.
SLIDE 8
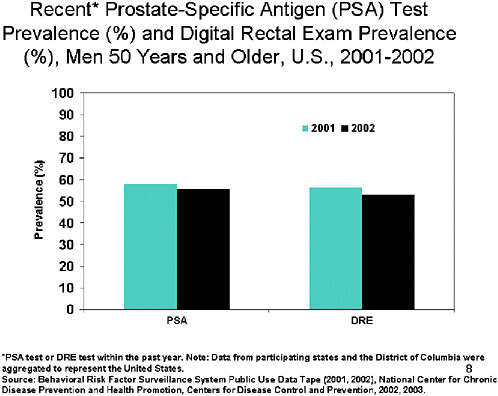
SLIDE 8 NOTES: Although PSA is not a recommended for population-based screening, the data are of interest because the technology used. In 2002, the percentage of men who had a PSA test within the past year was 55% while the percentage of men who had a DRE within the past year was 53%.
The question I would pose from the data viewed collectively is: What makes Pap testing different from the other tests, particularly DRE and PSA? They all are relatively inexpensive, require contact with a physician, can be done easily in the context of a primary care visit, and they aren’t very different in terms of their invasiveness. One obvious difference is that the data on Pap testing are for women while the data from DRE and PSA are for men. We know that women are more likely to have contact with the medical care system than men.
Another possible reason is that PSA is not a test that is recommended by the US Preventive Services Task force for population-based screening. Physicians have been encouraged to discuss PSA testing with their patients to allow an informed decision about whether to engage in it. But, from my own ongoing research, it appears that physicians are not generally engaging in such discussions. So, I believe it may not be done much because it has not disseminated into practice as a routine test.
Another observation from these data is that rates of recent mammography aren’t as high as we would like from a public health perspective—they are around 60%. Mammography is a more inconvenient procedure than DRE, PSA or Pap testing because it typically requires a referral which often means coming back for another appointment. While not invasive, it’s more uncomfortable than DRE, PSA and Pap tests. And it’s potentially more expensive.
What, then, is reasonable to expect for CRC screening tests and procedures? FOBT is similar in some respects to DRE, PSA, and Pap testing in that it is relatively inexpensive. It is different in that it may or may not require contact with a physician to acquire a test kit. SIG and COL are similar to mammography in that they typically require a referral, require more planning, and are arguably even more uncomfortable or unpleasant than mammography, although they are more invasive.
If we had a test that was inexpensive, easy for physicians to do, and acceptable to patients, what might be possible? I would argue that something close to 80% is possible. If we don’t have such a test for awhile, what might be some other issues that affect CRC screening prevalence, particularly for colonoscopy?
SLIDE 9

SLIDE 9 NOTES: There are also issues associated with physicians that are most interesting. Work by Dr. Klabunde at NCI is currently conducting show that physicians’ perceptions of their recommending tests or performing tests on patients are exceedingly high (Klabunde et al., 2003). Roughly 60 to 70 percent of physicians say that they are recommending or performing colon cancer screening tests on their average-risk patients. Those are not consistent, as you can see, with the prevalence estimates for these tests. So, there is some disconnect that merits more exploration.
SLIDE 10

SLIDE 10 NOTES: All studies were regional; there are no national data on follow-up. The majority of studies reviewed by Dr. Yabroff and her colleagues were conducted in clinics, programs, or health care systems that had mechanisms for tracking follow-up care (Yabroff et al., 2003; Bastani et al., under review). The actual prevalence of follow-up may be lower outside of organized systems of care or for the uninsured.
A study by Myers and colleagues (Myers et al., 2001) compared measurement of complete diagnostic evaluation after a positive FOBT by 4 methods—external chart review, internal chart audit, administrative data review, and a combination of chart review and administrative data notes—and found that rates ranged from 49 to 79%. Regardless of how measured, prevalence is less than optimal and may compromise the effectiveness of mass screening programs. We do not know the impact of delayed, incomplete, or no follow-up on stage of disease at diagnosis because population-based data on follow-up care are not routinely collected.
Dr. Yabroff and her colleagues conclude that: “Increasing the availability of screening follow-up data will be an important area for future evaluations of the quality of care provided in screening programs.”
To my knowledge, there are no published data of post-polypectomy surveillance.
SLIDE 11

SLIDE 11 NOTES: This is a compelling question that needs more research. At present, there is little evidence available to report.
REFERENCES
Bastani R, Yabroff KR, Myers R, Glenn B. Interventions to improve follow-up of abnormal findings in cancer screening. Cancer, under review.
Hawley ST, Vernon SW, Levin B, Vallejo B. Prevalence of colorectal cancer screening in a large medical organization. Cancer Epidemiol Biomarkers Prev, in press.
Klabunde CN, Frame PS, Meadow A, Jones E, Nadel M, Vernon SW. 2003. A national survey of primary care physicians’ colorectal cancer screening recommendations and practices. Prev Med. 36(3):352–362.
Myers RE, et al. 2001. Measuring complete diagnostic evaluation in colorectal cancer screening. Cancer Detect Prev. 25(2):174–82.
Peterson SK, Vernon SW. 2000. A review of patient and physician adherence to colorectal cancer screening guidelines. Seminars in Colon & Rectal Surgery. 11:58–72.
Swan J, Breen N, Coates RJ, Rimer BK, Lee NC. 2003. Progress in cancer screening practices in the United States: Results from the 2000 National Health Interview Survey. Cancer. Vol. 97(6):1528–1540.
Vernon SW. 1997. Participation in colorectal cancer screening: a review. J Natl Cancer Inst. 89(19):1406–1422.
Yabroff KR, Washington KS, Jacobs A, Neilson E, Mandelblatt JS. 2003. Is the promise of cancer screening programs being compromised? Quality of follow-up care after abnormal screening results. Med Care Res Rev. 60(3):294–331.













