1
Introduction
The purpose of this study was to respond to a congressional requirement in the Anti-Drug Abuse Act of 1988 (P.L. 100-690) that the Secretary of Health and Human Services request the National Academy of Sciences “to conduct a review with respect to the research activities of the National Institutes of Health (NIH) and the Alcohol, Drug Abuse, and Mental Health Administration (ADAMHA)” and related agencies, such as the Health Resources and Services Administration (HRSA) and the Centers for Disease Control (CDC). Specifically, the committee was asked for:
-
an evaluation of the appropriateness of administering health service programs in conjunction with the administration of biomedical and behavioral research; and
-
a determination of the extent of duplication among selected research programs of NIH and ADAMHA.
It is important to point out that the study was confined to the examination of agencies within the Public Health Service (PHS); it did not examine in depth the current organizational structure of the Department of Health and Human Services (DHHS).
To answer the above questions, the committee was asked in the contract for the study to develop and establish criteria and/or measures for determining the following:
-
the effects of administering service programs in conjunction with research and research-related activities;
-
the extent and effects of duplication, replication, or complementarity among the research activities of NIH and ADAMHA (to be determined through analysis of the scientific process and develop-
-
ment of a classification scheme for research activities and health service programs that allows comparisons across agencies along a number of parameters);
-
the administrative, program, and policy relationships among service programs and research/research-related activities of ADAMHA and its institutes, and the extent to which patterns of communication and leadership activities are attributable to co-administration or to other factors; and
-
the “appropriateness” of these effects, given both the statutory mission of the programs and the changing requirements of public policy, scientific opportunity, and economic conditions.
In its early discussions, the committee arrived at a consensus that the outcome of the study ought not to be a “yes” or “no” decision about the value of co-administration but instead that the study should result in a series of pros and cons about co-administration as it exists at different levels of the PHS (the institute, bureau, or office level; the agency level; and the level of the Office of the Assistant Secretary). The committee interpreted the congressional request as a question about the efficiency of different organizational structures for administration of research and service programs in the PHS, given existing resources. Although many in the fields and disciplines related to basic and clinical research are convinced that there is a need to maximize resources for research, the committee agreed that the means of achieving maximal funding for biomedical research was not the question being asked by Congress.
The committee identified three important issues related to the organization of biomedical and behavioral research and services development and demonstration programs that the study would evaluate in a variety of ways:
-
Do research and services development and demonstration programs benefit or suffer (as measured by funding levels, by analyses of planning and priority-setting activities, and by productivity) by being allied under a single administrative authority?
-
What are the effects of dual missions on the effectiveness of senior agency and institute executives?
-
Do particular organizational forms promote or hinder the transfer of basic research into clinical practice?
DEFINITION OF KEY TERMS USED IN THE STUDY
It is important to distinguish among three concepts that are often confused with each other—linkage, co-location, and co-administration. Also important to the understanding of this report is the distinction between categorical and functional organization:
-
Linkage is a very general term that has emerged from the health services delivery programs. It relates to the processes necessary for the development of systems of care. When raised to the level of federal agencies, however, the concept of linkage seems to focus on choices made by two or more organizations or organizational units to engage (either directly or indirectly) in some sort of mutual activity.
-
Co-location is a term that has been in use among state governments to describe situations in which two or more programs are located in a single organizational unit or in the same physical location. The concept of co-location does not imply a programmatic or budgetary relationship between programs.
-
Co-administration, as defined in this study, includes administrative responsibility and authority for decision making that affects funding and priorities for federal research and service programs. Co-administration can exist at any level at which such responsibility is present, for example, the Office of the Secretary of Health and Human Services, the Office of the Assistant Secretary for Health, or in agencies, institutes, or programs.
-
Categorical organization is defined as organization by field (for example, mental health, alcohol abuse, drug abuse) such as exists in ADAMHA, with subfields that include administration and coordination of research, services, and prevention programs.
-
Functional organization is defined as organization by functional task areas (for example, research, services, or prevention), such as exists in the division of responsibilities among NIH, HRSA, and CDC, respectively; further differentiation may occur by subfields within research (e.g., biomedical research, health services research) and within services (e.g., preventive services, health care or treatment services).
ORGANIZATION OF THE REPORT
A discussion of the organization and mission of the Public Health Service and its agencies follows this introduction, including new
organizational developments that occurred in the spring of this year and resulted in the formation of the Administration for Children and Families. This new office reports directly to the Secretary of Health and Human Services and is composed of the previous Family Support Administration and Administration for Children, Youth, and Families, as well as parts of the Maternal and Child Health Program previously administered by HRSA.
Chapter 2 presents a brief history of the PHS and the agencies involved in the study, as well as a history of previous studies of the organization of ADAMHA. Chapter 3 discusses in detail the methodology the committee used to collect and evaluate data and information for the study. Chapter 3 also presents summaries of each of the background papers prepared for the committee's deliberations.
Chapter 4 presents in depth the findings and recommendations of the committee related to co-administration of research and service programs in the PHS. Chapter 5 presents the findings and recommendations of the committee concerning the issue of programmatic and project duplication, replication, and complementarity of research and service activities in the PHS.
The report also includes four appendixes: Appendix A lists all abbreviations used in the report; Appendix B describes the nomenclature used in analyses of research and services programs and projects; Appendix C lists the membership of the task forces established by the committee in the course of the study; and lists the individuals interviewed in the course of the study.
ORGANIZATION AND MANAGEMENT OF THE PUBLIC HEALTH SERVICE
This study has focused on two quite different organizational structures that exist side by side within the PHS: one that separates tasks (managing and funding research, service demonstrations and service system development, and prevention programs) by function into three distinct agencies, and another that incorporates all three tasks into a single agency with functionally organized units. The study also has looked at some important differences in the history and mission of the agencies and institutes being studied. It has then attempted to describe and analyze the relationship between these differences in history, mission, organizational structure, and leadership and the capacity to perform the tasks for which the PHS and, indeed, DHHS are authorized.
DHHS, a cabinet-level department of the federal government, is comprised of the Office of the Secretary and five operating divisions (Figure 1-1 ). This study focuses on agencies within one of these figure divisions, namely the PHS. The PHS was established by the Public Health Service Act of 1944 “to promote the coordination of research, investigations, experiments, demonstrations, and studies relating to the causes, diagnosis, treatment, control, and prevention of physical and mental diseases and impairments of man.” 1 The PHS is comprised of eight agencies and the Office of the Assistant Secretary for Health (OASH), which provides executive direction to the eight agencies (Figure 1-2 ). The Assistant Secretary for Health is responsible to Congress and to the Secretary of Health and Human Services
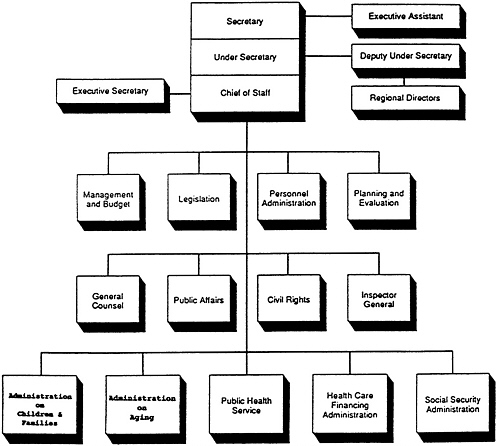
FIGURE 1-1 Organizational structure of the Office of the Secretary, Department of Health and Human Services (DHHS). Also located administratively in DHHS, but reporting to the President, is the Office of Consumer Affairs, which handles consumer-related policy and programs in the federal government.
SOURCE: General Accounting Office Report to Congress, February 1988 (GAO/ARD90-54).
for the performance of the PHS, its agencies, and its programs in meeting national health objectives.
The five PHS agencies relevant to this study are those that administer and fund health research, prevention, or services programs for the nation. Four of these agencies are organized primarily by function:
-
the National Institutes of Health (NIH), which funds, administers, and conducts biomedical research;
-
the Health Resources and Services Administration (HRSA), which administers services programs for underserved populations;
-
the Centers for Disease Control (CDC), which is responsible for disease prevention and health promotion programs; and
-
the Agency for Health Care Policy and Research (AHCPR), which funds, administers, and conducts health services and outcomes research and evaluation.
The fifth relevant agency is the Alcohol, Drug Abuse, and Mental Health Administration (ADAMHA), which administers a full range of programs including research, prevention, and services for the areas of mental health, alcohol abuse, and drug abuse. Figure 1-3 shows the relative shares of these agencies (as well as those of the Food and Drug Administration [FDA] and the Indian Health Service [IHS]) in the total DHHS budget.
NIH, ADAMHA, and AHCPR are established in law and granted specific authorities by Congress through amendments to the Public Health Service Act. HRSA and CDC are administratively created through authority vested in the Secretary of Health and Human Services. Whether legislatively or administratively created, every federal agency is required to publish a statement of its mission, functions, and organization in the Federal Register. Because each agency's activities are legally limited to what is contained in its functional statement, major agency-level policy changes generally require new legislation. 2 Similarly, some institutes and offices within agencies are established through acts of Congress, while others are administratively created. Those that are administratively created can be changed, merged, or dismantled at will by the Secretary of Health and Human Services, but legislation is required to alter the mission and authority of mandated offices or institutes. However, all offices and institutes are directly affected by Congress, which establishes funding levels for agencies through appropriations for all PHS programs. Thus, agencies could have authorization for activities, yet receive inadequate appropriations to carry them out. On the other

FIGURE 1-3 Relative size of Public Health Service agencies (in millions of dollars). Public Health Service total—$18,027 million. Based on fiscal year 1992 President's Budget. The Agency for Toxic Substances and Disease Registry receives its funding through a reimbursement agreement with the Environmental Protection Agency Super-fund. Abbreviations: NIH, National Institutes of Health; FDA, Food and Drug Administration; IHS, Indian Health Service; AHCPR, Agency for Health Care Policy Research; ADAMHA, Alcohol, Drug Abuse, and Mental Health Administration; CDC, Centers for Disease Control; HRSA, Health Resources and Services Administration; OASH, Office of the Assistant Secretary for Health.
SOURCE: Overview of the Major Components of the Public Health Service, March 1990, Office of the Assistant Secretary for Health.
hand, the infusion of funds by Congress for new or expanded programs could alter the direction or focus of an agency or institute.
ORGANIZATION AND MISSION OF THE FIVE PHS AGENCIES
Office of the Assistant Secretary for Health
OASH is organized to support the Assistant Secretary's broad mandate to provide direction for a national health program. 3 OASH
activities include all aspects of planning and development of federal health policy. OASH coordinates and implements policies and programs that cut across agency lines (within and outside of the PHS) such as infant mortality, acquired immune deficiency syndrome (AIDS), minority health, and health promotion and disease prevention, including vaccination programs. OASH also administers some federal grant programs (Family Planning, Adolescent Family Life, and Population Research) and manages the Commissioned Corps (the Surgeon General's office is located in OASH).
National Institutes of Health
The primary responsibility of NIH is to provide federal support to research that stimulates the development of the science base for the generation of new knowledge to prevent, detect, diagnose, and treat disease and disability. The largest of the PHS agencies, NIH is organized into 13 research institutes, primarily by disease or population categories (cancer, heart disease, aging, etc.), plus the National Library of Medicine and seven research and support divisions (Figure 1-4 ). NIH institutes are considered to be semiautonomous: they are coordinated through the Office of the Director, but programmatic decisions and operations are carried out at the institute level.
NIH and its institutes pursue their mission through intramural and extramural research programs, through the support of research centers, and through training programs to enlarge the pool of skilled investigators. Some, but not all, NIH institutes are authorized to conduct research demonstrations. However, NIH does not provide health services other than therapeutic measures and care incidental to research. It has no regulatory responsibilities other than recommending standards for the use of animal and human subjects in federally supported health research and in recombinant DNA research.
Alcohol, Drug Abuse, and Mental Health Administration
ADAMHA holds lead responsibility for the federal government's efforts to seek scientific solutions to the causes, treatment, and prevention of alcohol, drug abuse, and mental health (ADM) disorders. While the research mandate is very similar to the mandate of NIH, ADAMHA's mission also includes authorization to conduct activities
to help improve the nation's capacity to treat and prevent ADM disorders. In addition to basic and clinical biomedical research and research training programs, ADAMHA administers the following prevention and service activities:
-
service demonstration programs related to the prevention and treatment of ADM disorders;
-
administration of block and formula grants to states for ADM services;
-
collection of data on incidence and prevalence of ADM disorders and the national response; and
-
provision of assistance and information about ADM disorders to other federal agencies, states, health care providers, and public and private organizations.
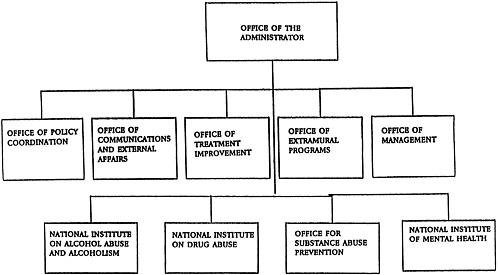
Figure 1-5 shows the major organizational components of ADAMHA. The Office of the Administrator supervises and coordinates ADAMHA's institutes and offices, which include the National Institute of Mental Health (NIMH), the National Institute on Alcohol Abuse and Alcoholism (NIAAA), the National Institute on Drug Abuse (NIDA), the Office for Substance Abuse Prevention (OSAP), and the Office for Treatment Improvement (OTI). Much like the NIH institutes, the three ADAMHA institutes support intramural and extramural basic and clinical research, research training, and research demonstration projects. In addition, NIMH continues to administer some services funding programs. In contrast, the activities of the newer ADAMHA offices, OSAP (established in 1986) and OTI (established in 1989), concentrate on the development and administration of programs for substance abuse prevention and treatment services, and information dissemination. Although the ADAMHA institutes and OSAP are separately authorized in legislation, OTI was administratively created and has yet to be ratified by Congress.
Health Resources and Services Administration
HRSA supports programs that provide health care services to a variety of medically underserved populations, with the goal of improving access, equity, and quality of care. HRSA's range of programs include both the direct and indirect funding of services and the training and placement of health care professionals in underserved
areas. In addition, HRSA administers programs of national importance, such as AIDS service delivery and education, rural health services, and a national organ transplantation network to improve procurement and allocation of organs on a nationwide basis.
Centers for Disease Control
CDC is the federal agency responsible for protecting the public by preventing unnecessary diseases, disability, and premature death and by promoting healthy lifestyles. Included in CDC's mission are the study and prevention of chronic and infectious diseases, the reduction of injury, and the reduction of risk factors that are controllable. CDC also serves as the lead PHS agency in responding to public health emergencies, and it maintains national surveillance and statistics. Like the NIH institutes, which are organized to focus on disease categories or populations, the centers organizational pattern affords CDC the opportunity to organize its internal structure around specific disease prevention objectives or objectives targeting improved health status.
Agency for Health Care Policy Research
AHCPR, the newest PHS agency, was established by the Omnibus Budget Reconciliation Act of 1989. It builds on and succeeds the National Center for Health Services Research and Health Care Technology Assessment (NCHSR/HCTA) as the primary source of federal support for research on medical treatment effectiveness, general health services, and health care technology. Because provision of health care services includes both private and public providers, AHCPR's research focuses on both the private and the public sector. New knowledge generated through intramural and extramural research programs will be used to inform and assist health care decision makers.
NOTES
1. Public Health Service Act of 1944, P.L. 78-410.
2. Administrative Procedures Act, Title V, U.S.C.