2
Application Needs and Trends
The operating requirements of gas-turbine engines dictate the future development of coatings. This chapter discusses two areas of that influence: application needs and substrate material trends. A cross section of a typical modern gas-turbine engine is shown in figure 2-1. As a general rule, components are only coated if system durability and reliability can be improved. For instance, within the last several years, high-temperature coatings have been extended to additional hot-section components in order to reduce surface degradation.
APPLICATION NEEDS
The need for coatings has evolved since they were first applied to high-pressure turbine airfoils in the early 1960s. Coatings were initially used to protect hot structure against degradation by high-temperature oxidation and hot corrosion (These processes are described in chapter 4). Coatings have become increasingly critical as advances in turbine technologies have made possible substrate and gas-path temperatures that approach or even exceed the melting point of the substrate in localized regions.
The duty cycles imposed on gas-turbine engines vary greatly depending on application. Gas-turbine engines are used in a variety of transportation, power-generation, and mechanical-drive applications, listed in figures 2-2 and 2-3. Operating life expectancy is principally determined by balancing the two major factors that contribute to wear: on/off cycle requirements and time-at-temperature. Greater demand on cyclic capability generally reduces the time a system can operate at high power (i.e., the interaction of thermomechanical fatigue and high-temperature creep of materials determine hot-section life expectancy). Other considerations, such as environmental factors (e.g., hot corrosion and high-temperature oxidation), also lower life expectancy.
While engine mission profiles vary with customer requirements, the typical hours of operation expected per engine start are depicted for each generic class of operation in table 2-1. The number of engine start/stop cycles and operating time at high temperature are the basic factors that determine when critical components are to be overhauled. The condition of hot-section coatings is an important indicator of the extent of distress suffered by an engine. Thus, the coatings are regularly inspected. The reliability and life expectancy of high-temperature coatings are major factors in maximizing the service life of a gas-turbine engine.
Aircraft Turbine Needs
Two government programs, the High-Speed Civil Transport program led by NASA and the Integrated High-Performance Turbine Engine Technology program led by DOD, are aimed at developing prototype turbines within the next ten years that will operate at firing temperatures at least 150°C (270°F) higher than the current generation of high-performance gas turbines. Figure 2-4 depicts the increase in firing temperatures over the past 15 years and the nominal goals for these programs. The committee believes that incremental improvements in the durability of substrate materials will not meet the goals of these two government programs. Success
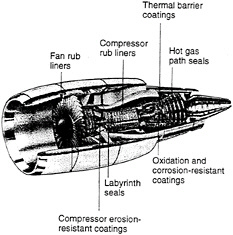
Figure 2-1 Cross section of a typical modern gas turbine engine.
Source: Hillery (1989). Copyright General Electric Company; used with permission of the General Electric Company.
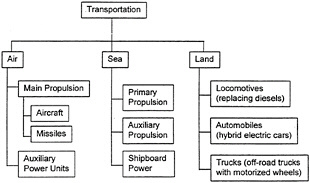
Figure 2-2 Overview of transportation gas-turbine use.
will require a much more rapid improvement in the operating temperature of materials, as depicted in figure 2-4. Incremental development of TBC technology, however, may allow the current generation of materials to achieve a portion of the desired increase in firing temperature.
In addition to a hot-section temperature increase, both of the advanced engine projects also seek to reduce emissions dramatically, particularly NOX. These emission requirements affect the engineering design of the combustor and cannot be met without advances in combustor materials and coating technology. For example, release of NOX and SOX could potentially be controlled by the engineering of ingredients in the coating that act as catalysts for the conversion of these pollutants immediately after their formation.
Land-Based Turbine Needs
Industrial gas turbines for power generation are distinguished from aircraft gas turbines by many characteristics that include larger size, lower rotational speeds, longer life expectancy, longer time between overhauls, high time at full power, few on/off duty cycles, lower quality and lower heating-value fuels, wide variety of fixed locations (e.g., arctic to tropic, oceans to deserts, clean to polluted), and few weight penalties. The needs of large power-generation equipment essentially determine the available technology for land-based turbines (NRC, 1986).
The Advanced Turbine Systems (ATS) program, funded as part of the fossil energy research program of DOE, seeks to develop higher-efficiency land-based turbines that burn
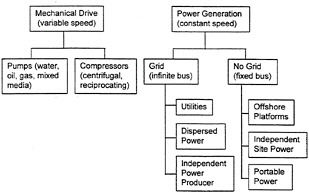
Figure 2-3 Overview of power-generation and mechanical-drive gas-turbine use.
TABLE 2-1 Typical Duty Cycles for Various Gas-Turbine Engines
|
|
Typical Mission Time per Engine Start (hours) |
||
|
Duty Cycle |
Minimum |
Average |
Maximum |
|
Airline/Transport |
1 |
2 |
8 |
|
Military/Fightera |
1 |
2 |
4 |
|
Mechanical Drive |
40 |
2,000 |
8,000 |
|
Base Power Generators |
500 |
2,000 |
8,000 |
|
Peak Power Generators |
2 |
4 |
12 |
|
a The mission profile for military aircraft has greater thermal transient and low-cycle fatigue because of periods of supersonic cruise and combat/avoidance maneuvers. |
|||
natural gas but that are also capable of burning coal-or biomass-derived fuels (NRC, 1995a). Firing temperatures at least 150°C (270°F) higher than the current generation of gas turbines and a combined cycle efficiency of 60 percent are desired, while maintaining the durability levels of current power-generation systems.
Fuel type is probably the most important variable affecting the selection of a coating system (NRC, 1995a). Pipeline-quality natural gas contains virtually no hydrogen sulfide and very low sulfur content, unlike unprocessed natural gas. Coal gasification produces a raw syngas consisting mainly of carbon monoxide and hydrogen, along with substantial quantities of hydrogen sulfide, ammonia, and hydrogen chloride, and a few parts per million of alkali metals (NRC, 1986). To meet environmental requirements, commercial gasification systems include clean-up systems that remove virtually all of the hydrogen sulfide, ammonia, and hydrogen chloride; in general, the alkali metals are also removed. However, it is possible that in the event of a degradation of the clean-up systems, some of these harmful materials may enter the turbine. The complex chemical and electrochemical reactions that occur at high temperatures and the susceptibility of these reactions to small changes in the coating and gaseous environment make coatings for hot-section components a critical enabling technology. Coating degradation by hot-corrosion mechanisms is the primary concern. One goal of the ATS program is to seek lower emission levels, similar to those discussed above for aeronautical turbines.
Marine Turbine Needs
On the ocean, gas turbines provide primary drive, auxiliary drive, and shipboard power. A new type of marine vehicle,
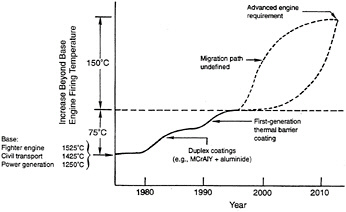
Figure 2-4 Approximate increases in firing temperature capabilities from 1980 to 2010.
the all-electric ship, uses a gas turbine located at mid-ship to power the motors that drive the aft propellers. Because of their low operating cost, gas turbines are expected to become more common on ships.
Although high-quality liquid fuels are used, marine gas turbine engines operate in an environment heavily laden with salt (sodium chloride) and are often close to other ships that burn low-quality fuels with high-sulfur concentrations, such as Bunker C. Contaminants from these exhaust gases are drawn into the ship's intake air. Thus, hot corrosion is the primary nemesis of gas turbines on ships. The coatings primarily developed for marine environments are aimed at increasing the resistance of hot-section components to hot corrosion.
CURRENT COMPONENT DESIGN AND BASE MATERIALS
The operating conditions that a component is expected to encounter drives the component functional design and the resulting choice of material. The materials that have been and continue to be used are nickel-base and cobalt-base superalloys. A partial list of these alloys is given in table 2-2 (Gabb and Dreshfield, 1986; Backman and Williams, 1992; Stringer and Viswanathan, 1993). These chemically complex alloys are highly developed materials with a combination of high temperature strength, inherent environmental resistance, toughness, and fatigue resistance that are not currently available in other alloy system (Sims, 1986). Numerous compositions and processing routes have been developed that can be tailored to specific application needs.
Nickel-base superalloys have had more extensive development than cobalt alloys. Nickel-base superalloys enjoy high strengths at elevated temperatures through the precipitation of a coherent phase: gamma-prime. In addition, these alloys are strengthened by the more common mechanisms of incoherent phases and solid solutions. The result is a family of chemically complex alloys that are capable of operating in aggressive environments at temperatures greater than 90 percent of their incipient melting temperature. These features have made nickel-base superalloys the favored materials for use in rotating turbine components, such as blades. These alloys are also widely used in static structure, such as case, vane, and combustor applications.
The dramatic progress in investment-casting technology ranks as the single most important process development for nickel-base superalloys. Progress in this technology has enabled engineers to cast directionally solidified and single-crystal airfoils that include complex, cast-in, internal air-cooling passages. By using sophisticated cooling techniques with single-crystal alloys, researchers have attained operating temperatures 80°C (144°F) higher than those of conventional, equiaxed castings.
Cobalt-base superalloys are not as developed as nickelbase superalloys because coherent phase strengthening has not been demonstrated for these materials. Although cobaltbase superalloys have less strength at comparable temperatures, they tend to have higher melting points than comparable, conventionally cast nickel-base superalloys and can therefore function at somewhat higher temperatures. Cobalt-base superalloys are more easily repaired by welding than are the advanced nickel-base superalloys and also have better corrosion resistance because of higher chromium levels. For these reasons, the cobalt-base superalloys are used for higher-temperature applications in which high strength is not the primary issue (i.e., vanes and combustor liners). These alloys also prove valuable in certain-but not all-hot-corrosion conditions. However, the advanced nickel-base alloys developed for single-crystal technology have significantly higher melting temperatures than the conventional nickel-base alloys. As a result, single-crystal nickel-base superalloy vanes have replaced polycrystalline cobalt vanes in some applications.
Design engineers seek to mitigate the impact of operating conditions on component integrity by two approaches, both of which have become absolutely essential to the design of hot-section components. The first approach relies directly on coating technology to extend the service life of both nickelbase and cobalt-base superalloys, as discussed in chapter 3.
In the second approach, coatings contribute indirectly, but just as significantly, to extending service life. This method relies on component cooling to decrease the steady-state component temperatures; coatings play a key role in such cooling. For aeronautical turbines, noncombusted air diverted from the compressor is used to cool critical components, such as airfoils (Sims, 1991). Cooling through the use of steam or other means is also possible when engine weight is not a concern. Several methods working in tandem make the most efficient use of the cooling air. These include convection cooling and impingement cooling on the back sides (cold side) of hot surfaces and film cooling on the hot sides that face the combustion gases (figure 2-5). In film cooling, air flowing out through small holes in the component wall provides a boundary layer of air cooler than the combustion gas, thus cooling the component. These film-cooling holes may become blocked during the application of a coating, however. Loss of cooling air may lead to local melting and serious degradation of the component.
ADVANCED MATERIALS FOR ENGINE COMPONENTS
Nickel-base and cobalt-base superalloys have served admirably in gas turbines for over 50 years, and the attractive properties of this class of materials guarantee that superalloys will continue to serve in turbine engines well into the future (Sims, 1986). However, current superalloys operate at 90 percent
TABLE 2-2 Nickel-and Cobalt-Base Alloys Used in Land-Based and Aircraft Gas Turbines
|
Combustor Liners and Transition Pieces: Sheet Metal Applications |
|||||||||||||||||
|
Alloy |
Ni |
Co |
Cr |
Al |
Mo |
W |
Ti |
Fe |
Cb |
C |
Si |
Mn |
Zr |
Ta |
Other |
Type |
TSOLIDUS(ºC) |
|
Hastalloy X |
Bal. |
— |
22 |
— |
9 |
— |
— |
20 |
— |
0.15 |
— |
— |
— |
— |
— |
sheet |
1260 |
|
Haynes 188 |
22 |
Bal. |
22 |
— |
— |
14 |
— |
3 max |
— |
— |
— |
— |
— |
— |
— |
sheet |
1300 |
|
TD-Nickel |
Bal. |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
2.0 ThO2 |
sheet |
1453 |
|
Inconel 617 |
Bal. |
12.5 |
22 |
1 |
9 |
— |
— |
— |
— |
0.07 |
0.5 |
0.5 |
— |
— |
— |
sheet |
1330 |
|
RA 333 |
Bal. |
3 |
25 |
— |
3 |
3 |
— |
18 |
— |
0.05 |
1.25 |
1.5 |
— |
— |
sheet |
|
|
|
Stator Vanes and Nozzles |
|||||||||||||||||
|
Alloy |
Ni |
Co |
Cr |
Al |
Mo |
W |
Ti |
Fe |
Cb |
C |
Si |
Mn |
Zr |
Ta |
Other |
Type |
TSOLIDUS(°C) |
|
X-40 |
10.5 |
Bal. |
25.5 |
— |
— |
7.5 |
|
— |
— |
0.5 |
— |
— |
0.2 |
— |
— |
CC |
1341 |
|
Mar-M 509 |
10 |
Bal. |
23.5 |
— |
— |
7 |
0.2 |
— |
— |
0.6 |
— |
— |
0.45 |
3.5 |
— |
CC |
1288 |
|
F-75 |
— |
Bal. |
27.5 |
— |
5.5 |
— |
— |
— |
— |
0.22 |
— |
— |
— |
— |
0.12N |
CC |
|
|
GTD 222 |
Bal. |
19 |
22.5 |
1.2 |
— |
2 |
2.3 |
— |
0.8 |
0.1 |
— |
— |
— |
1 |
— |
CC |
|
|
IN713C/HC |
Bal |
— |
13.5 |
6 |
4.5 |
— |
0.85 |
— |
2.3 |
0.12 |
— |
— |
0.1 |
— |
— |
CC |
1260 |
|
IN713C/LC |
Bal |
— |
13.5 |
6 |
4.5 |
— |
0.85 |
— |
2.3 |
0.06 |
— |
— |
0.1 |
— |
— |
CC |
1288 |
|
IN738C/HC |
Bal |
8.5 |
16 |
3.4 |
1.7 |
2.6 |
3.45 |
— |
0.9 |
0.17 |
0.3 |
0.2 |
0.1 |
1.75 |
0.012B |
CC |
1232 |
|
IN738C/LC |
Bal |
8.5 |
16 |
3.45 |
1.7 |
2.6 |
3.6 |
— |
0.9 |
0.06 |
0.3 |
0.2 |
0.1 |
1.7 |
0.012B |
CC |
1232 |
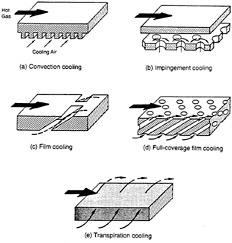
Figure 2-5 Turbine blade cooling methods. Source: Glassman (1975).
of their incipient melting temperatures in some applications. This is both an unprecedented achievement and a graphic illustration of the inherent limitation of these materials. The requirement for more efficient engines in the future will also require even higher operating temperatures and, therefore, higher component temperatures. Thus, the targeted temperatures will inevitably exceed the capabilities of current superalloys.
Variants of current superalloys, such as eutectics and superalloy matrix composites, can fill a need for increased creep resistance and higher specific strength. These materials can also help reduce component weight and, when used as turbine blades, lower the loads on turbine disks. However, their potential for withstanding higher temperatures is still limited by their melting points. Thus, designers are investigating alternative materials to fill the higher-temperature roles. These materials generally fall into three classes: ceramics, intermetallics, and refractory materials (Stoloff and Sims, 1986; Meetham, 1988; Backman and Williams, 1992; Stringer and Viswanathan, 1993). The remainder of this chapter discusses the environmental degradation issues that are faced for each class of these materials. Coatings for these materials are presented in chapter 3.
Ceramics
Ceramics are generally thought to hold considerable promise because they possess many of the properties desired for higher-temperature replacements of superalloys: very high melting points, high-temperature strength, low density, and some increased resistance to aggressive environments (table 2-3). The primary shortcoming of monolithic ceramics is their lack of acceptable low-temperature ductility and toughness. The toughness issue is being addressed by the development of ceramic composite systems.
Ceramics currently under development may serve as combustors in both aircraft and land-based turbine engines in the Advanced Turbine Systems and the Integrated High-Performance Turbine Engine Technology programs. Designers are also exploring the use of ceramics for turbine blades, an application that could become the biggest economic payoff of these materials. Silicon carbide (SiC) and silicon nitride (Si3N4) rank as the prime candidates for making ceramic components because of their high-temperature strength and relative resistance to thermal shock.
Although the development of the requisite mechanical properties of ceramic components continues, it is becoming increasingly clear that these materials must be coated. Both silicon carbide and silicon nitride oxidize to form silicon oxide surface scales, which may limit their high-temperature use unless adequate protective coatings can be applied. Under the operating conditions envisioned for these materials, surface recession caused by active oxidation (SiO formation instead of SiO2) is a significant problem (Jacobson, 1992). Even limited exposure to sodium-containing environments greatly accelerates active oxidation (Jacobson et al., 1990). Coatings for a ceramic component will almost surely have to be ceramic as well, which poses the problem of having to precisely match coefficients of thermal expansion since ceramics do not possess the ductility to accommodate even slight mismatches in thermal expansion. In summary, coatings are needed to act as both thermal barriers to reduce the base-material temperature (and thereby reduce the oxidation rate) and as chemical barriers to slow oxidation.
Oxide-base ceramics are not as attractive for high-temperature structural applications since their strength and toughness are not as high as silicon-base materials (Stoloff and Sims, 1986). Coatings are less important for these materials, however, since oxidation is clearly not a concern, although some (e.g., zirconia and alumina) are susceptible to hot corrosion.
Intermetallics
Intermetallic compounds, especially those involving aluminum, have been heavily researched over the last several years. Compounds of nickel (Ni), titanium (Ti), cobalt (Co), and iron (Fe) with aluminum (Al) have all shown promise, but most studies have focused on nickel aluminides and titanium aluminides. These compounds have higher melting temperatures and lower densities than the superalloys (table 2-4). Thus, they have the possibility of achieving both higher temperatures and higher specific strengths than superalloys. Yet even these alloys will require coatings to excel at high temperatures.
TABLE 2-3 Properties of Ceramic Materials that are Candidates for Hot-Section Use
|
Material |
TMELTING (°C) |
CTE (10-6 °C-1) to =1000°C |
Young's Modulus @ 21°C (GPa) |
Density (g/cc) |
Fracture Toughnessa @ 21°C (MPa lm) |
Flexural Strength" @ 21°C (MPa) |
Flexural Strength @ 1200°C(MPa) |
|
SiC |
4.0c |
410d |
3.2e |
3 |
400 |
276-690c |
|
|
Si3N4 |
3.0c |
310d |
3.2e |
4.0-5.5 |
500-800 |
550-830c |
|
|
Composite Si3N4 20 vol. % SiC whiskers |
— |
— |
— |
— |
6.5-7.5 |
700-950 |
— |
|
|
|
|
|
|
|
|
— |
|
Al203 |
2045e |
9.0f |
360d |
3.97 |
2.5-4.0 |
300-400 |
— |
|
Composite A1203 20 vol% SiC whiskers |
— |
— |
— |
— |
7.5-9.0 |
650-800 |
— |
|
|
|
|
|
|
|
|
— |
|
Zr02 |
˜2700e |
10.0f |
200d |
˜5.73 |
— |
— |
— |
|
fully stabilized |
— |
— |
— |
— |
2.3 |
100-300 |
— |
|
partially stabilized |
— |
— |
— |
— |
15-18 |
>600 |
— |
|
TZP |
— |
— |
— |
— |
5-16 |
>1000 |
— |
|
B 1900 Superalloyd |
1330 |
15.8 |
214 |
8.0 |
˜100g |
970 |
270 (UTS @ 1100°C) |
|
a Tiegs et al. (1992). b Silicon-base ceramics tend to decompose and sublimate at high temperature. Therefore, there is no definable melting point for atmospheric pressures. An "optimistic maximum use temperature," based on oxidation behavior, is 1650°C (Fox, 1992). c Larsen et al. (1985). d CRC (1985). e International Nickel Company (1977). f Kingery et al. (1976). g Actual values for B 1900 were not available. This represents typical superalloy toughness values for comparison with ceramics. NOTE: Ceramic properties are very sensitive to processing; therefore, the values given are representative of a range of values for each property. |
|||||||
Researchers have also begun investigating intermetallics as matrix materials for composites, adding to the specific strength capabilities of the intermetallics. Furthermore, alloys with higher aluminum content (e.g., NiAl, TiAl, and TiAl3) have good inherent resistance to oxidation. The oxidation resistance of NiAl is particularly excellent; NiAl is the main component in oxidation-resistant aluminide coatings for nickel-base superalloys.
Although researchers have considered intermetallics for structural applications for over 20 years, most of the studies on these materials have been conducted during the last decade (Dimiduk et al., 1992). Therefore, intermetallics remain relatively immature materials that have a significant number of-technical barriers to be overcome before they could be used as hot-section materials. Obstacles include low creep strength at high temperature and poor ductility at low temperatures(Dimiduk et al., 1992). Despite these drawbacks, a number of research programs have improved the properties of these materials, and intermetallics now offer the promise of less-demanding, lower-temperature applications. Indeed, studies appear to have shifted from higher-temperature (roughly between superalloy and ceramic capabilities) to lower-temperature applications where these materials have a chance of replacing the denser superalloys. As was the case for superalloys, intermetallics may only attain the required mechanical properties at the expense of some of their environmental durability. Thus, intermetallics developed for hot-section uses will require coatings to mitigate a variety o f environmentally induced problems.
Titanium aluminides that have low aluminum content (i.e., less than 40 atomic percentage) tend to grow brittle during exposure to high temperatures in air. The high-aluminum-content titanium aluminides (TiAl) apparently do not have as severe an embrittlement problem and have a higher resistance to oxidation than the low-aluminum titanium aluminides (Ti3Al; McKee and Huang, 1990). Unfortunately, the relatively good oxidation resistance of TiAl does not extend to the higher temperatures required in the hot section. Thus, TiAl will probably require a coating to perform adequately in the hot section. The oxidation-resistant TiAl3 is brittle at low temperatures and is not currently a promising structural material (Smile et al., 1990).
Applications for NiAl are still somewhat tentative since alloys having the requisite mechanical properties for hot-section use are not currently available. Near-stoichiometric NiAl has excellent resistance to oxidation and therefore should not need an oxidation-resistant coating. However, the poor strength of pure NiAl at high temperatures suggests that the
TABLE 2-4 Selected Properties of High-Temperature-Capable Intermetallics Compared to a Conventionally Cast and a Single-Crystal Superalloy
|
Material |
TSOLIDUS (°C)a |
Young's Modulus (GPa) |
Density (g/cc) |
Fracture Toughness @ 21 °C (MPa vm) |
|
Ti3Alb |
1600 |
145 |
4.3 |
25 |
|
TiAlb |
1460 |
176 |
3.8 |
25 |
|
NiAla |
1640 |
300 |
5.9 |
— |
|
NiAl-eutecticb |
— |
193 |
5.9 |
12-15 |
|
MoSi2b |
1870-2030 |
379 |
6.5 |
4-5 |
|
FeAla |
1250 |
260 |
5.6 |
— |
|
CoAla |
1650 |
300 |
6.1 |
— |
|
B 1900c |
1274 |
214 |
8.2 |
»100 |
|
Single-crystal superalloya |
— |
125 |
8.3 |
35 |
|
a Stoloff and Sims (1986). b Dimiduk et al. (1992). c International Nickel Company (1977). |
||||
material will need to be alloyed for use in a hot section. Alloying will most likely cause some loss of oxidation resistance. Coatings may thus be required to provide requisite oxidation resistance.
Other intermetallics under investigation for high-temperature use include MoSi2, FeAl, and CoAl. It is expected that the same concerns about hot-section oxidation resistance and environmentally induced problems will apply to FeAl and CoAl. Although its high-temperature oxidation resistance is excellent (Vasudevan and Petrovic, 1992), MoSi2 has the added environmental-resistance problem of pesting. 1
Refractory Metals
Scientists have gathered extensive data over the last few decades on the properties and environmental resistance of the refractory materials (Stoloff and Sims, 1986). The continued interest in these materials stems from the attractive combination of high melting point, similar to (and in some cases, higher than) ceramic materials, with some ductility at low temperatures (table 2-5). Most of the research has been conducted on niobium, molybdenum, tantalum, and tungsten because of their relatively low cost compared with other refractory metals. Niobium is the most attractive of the refractories since it has a density slightly lower than nickel while still having a melting temperature approximately 1000°C (1800°F) higher than nickel. In fact, the niobiumbase alloy C-103 has been successfully used in unmanned aircraft (Stoloff and Sims, 1986). Although a diffusion coating of (molybdenum/tungsten)(silicon/germanium)2 has reportedly protected niobium (Mueller et al., 1991), the extremely high oxidation rates and tendencies toward embrittlement of the refractory alloys would result in extremely rapid degradation if the coating failed. Consequently, these materials are not currently used in long-lived, hot-section components.
Recent work on refractory alloys has also focused on developing resistance to environmental attack. These metals have little inherent resistance to environmental attack, which could result in a coating flaw leading to the degradation and subsequent failure of a component. This work has included additions of aluminum and titanium, both of which tend to slow oxidation, and additions of sufficient aluminum to form refractory metal aluminides (Stoloff and Sims, 1986). These efforts have not produced alloys useful as hot-section components in air-breathing engines, however. If these efforts ultimately prove successful, a coating will probably still be necessary to provide the long-term environmental resistance required for all gas-turbine engines.
TABLE 2-5 Properties of Refractory Metals and an Alloya Compared to B 1900 Nickel-Base Superalloy
|
Material |
TSOLIDUS (°C) |
CTE x 106°(C) |
Density (g/cc) |
Young's Modulus @ 25°C (GPa) |
UTS (MPa) |
|
Niobium |
2468 |
8.3 |
8.6 |
97 |
— |
|
Alloy B88 (Nb-28W-2Hf) |
— |
— |
¬11.6 |
— |
>>462 @ 1315°C |
|
Tantalum |
2996 |
6.7 |
16.6 |
185 |
— |
|
Molybdenum |
2610 |
5.8 |
10.2 |
325 |
— |
|
Tungsten |
3410 |
4.6 |
19.3 |
400 |
— |
|
B 1900b |
1274 |
15.8 |
8.0 |
214 |
270 @ 1100°C |
|
a Stoloff and Sims (1986). b International Nickel Company (1977). |
|||||









