2
Breast Cancer: Biology and Medicine
It has been estimated that in 1996 184,000 new cases of invasive breast cancer would be detected in the United States (ACS, 1995). Women born in the United States have, on average, a one in eight (12.6%) chance of developing breast cancer during their lifetime (Kosary et al., 1996). An overview of the current status of breast cancer research is provided in this chapter to serve as a background for this report and to provide the context in which the committee developed recommendations for future research directions. Included in this chapter are brief descriptions of the biology and genetics of the disease, its epidemiological features, current and potential treatment, and prevention strategies.
INCIDENCE AND MORTALITY
Despite the explosion of new knowledge about breast cancer from a variety of disciplines, it is still the most common malignancy among women and the second leading cause of cancer death among women; it was predicted that some 44,300 women would die from breast cancer in 1996 (ACS, 1995). The incidence of breast cancer has climbed at a rate of 1% to 2% per year during the past several decades (Harris et al., 1992a; Miller et al., 1993). Between 1982 and 1986, the incidence increased by approximately 4% per year (Harris et al., 1992a), and continued to increase through 1987, followed by a decline during the next 2 years. The majority of the recent increase has been caused by increased detection of early stage and in situ disease which is likely related in part to increased use of mammography (Harris et al., 1992a; Miller et al., 1993).
Incidence and mortality rates have varied by race and age. In 1992, the age-adjusted incidences in Caucasian and African-American women were 113.1 versus 101.0 cases per 100,000 women, respectively (Kosary et al., 1996). For Caucasian women, mortality rates changed little in the 1970s and 1980s, declined slightly after 1990, and are currently lower than for African-American women. Mortality rates for African-American women have increased steadily since the 1970s. In 1992, the age-adjusted mortality rates for Caucasian and African-American women were 26.0 and 31.2 deaths per 100,000 women, respectively, (Kosary et al., 1996). Table 2-1 outlines age-specific incidence and mortality rates of invasive breast cancer in Caucasian and African-American women in the United States between 1988 and 1992. It is significant to note that between the ages of 20 and 44, African-American women have both a higher incidence and a higher mortality rate than Caucasian women.
TABLE 2-1. Age-Specific Incidence of Breast Cancer and Mortality Rates of Women by Race in the United States, 1988-1992a
In general, women in developing countries have a lower incidence of breast cancer than women in industrial developed countries (Pisani, 1992). However, women who have migrated from areas of low incidence to areas of high incidence, such as Japanese emigrants to Hawaii or California, show a rise in breast cancer incidence over consecutive generations (Ziegler et al., 1993). Variation in incidence rates by ethnic groups appears to be a reflection of incidence rates in the country of origin, the length of residence in the country of immigration (if relevant), and the degree of acculturation. Incidence rates for invasive breast cancer among racial and ethnic groups in the United States during the period 1988–1992 are shown in Table 2-2.
TABLE 2-2. Racial/Ethnic Patterns of Invasive Breast Cancer in the United States, 1988–1992a
STAGES OF BREAST CANCER DEVELOPMENT
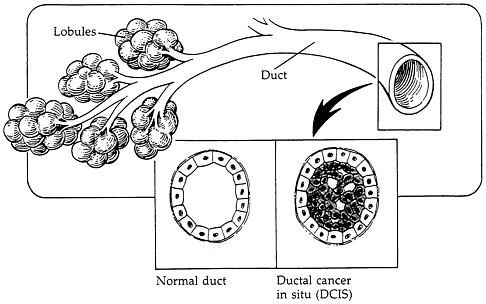
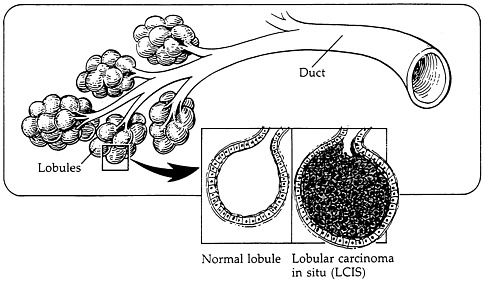
The breast is composed of lobes (lobules) of lactiferous (milk-producing) glands, and ducts (hollow tubes) set in fat tissue that exit at the nipple (Figure 2-1). Most breast cancers (approximately 80%) occur in the ductal region, while the remaining 20% seem to originate in the lobules (Figure 2-2a and 2-2b). Although there is some disagreement as to when histological changes can be defined as cancer, it is generally accepted that atypical hyperplasia is a precancerous entity. Ductal carcinoma in situ and lobular carcinoma in situ (which originates in the lobules) are referred to as noninvasive because the cells do not infiltrate the surrounding tissues (a process referred to as invasion). What

FIGURE 2-1. Female breast. SOURCE: Love, 1995, p. 35. Reproduced with permission.
causes the cascade of events that converts normal breast cells into malignant cells is not known, but it is generally thought to involve a complex interaction of endogenous (e.g., genetic and hormonal) and exogenous (e.g., dietary and other environmental) factors affecting multiple genetic changes in the involved cells. The four stages of transformation from a noncancerous condition to a cancerous condition are depicted in Figure 2-3.
In some instances, microscopic metastasis (i.e., spread beyond the breast) is present at the time of diagnosis, even when the primary tumor is small. This knowledge has resulted in changes in the local treatment of breast cancer. Recent advances in breast-conserving surgery and radiotherapy produce survival rates equivalent to those after total mastectomy, and adjuvant systemic therapy prolongs the disease-free interval and overall survival (Early Breast Cancer Trialists' Collaborative Group, 1992; Harris et al., 1992b). However, current treatments—surgery, radiation, chemotherapy, and hormonal therapy—are not completely effective and exact a substantial physical and emotional toll on the women who are treated with these agents.
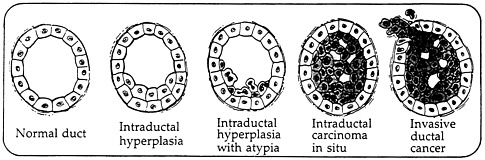
FIGURE 2-3. Four stages of transformation. SOURCE: Love, 1995, p. 220. Reproduced with permission.
Because breast cancer is a heterogeneous condition, with variations in the natural history of the disease and the response to therapy, not all women require the most aggressive therapies. For example, only a small subset of cases of lobular carcinoma in situ progress to invasive cancer; and most women whose axillary lymph nodes are clear of metastases at the time of diagnosis will not develop distant metastases, although 10%–40% will (Harris et al., 1992b). It is currently not possible to determine with certainty the best therapeutic regimen for a particular woman, nor is it possible to determine which individual woman will have a recurrence of her disease.
BREAST CANCER GENETICS
Breast cancer is due to multiple genetic changes affecting numerous genes. Three different types of genes are involved in the process that leads to malignancy: (1) oncogenes—genes that lead to malignancy only if activated by mutation, enhancement of expression, or amplification; (2) tumor suppressor genes—genes whose function has to be lost by either mutation or deletion to lead to malignancy; and (3) modifiers—genes that are involved in DNA repair. (Note: There could be other mechanisms.) Oncogenes and tumor suppressor genes are involved in the control of cell proliferation, programmed cell death (apoptosis), and differentiation.
During the past few years, breast cancer has been associated with the expression of several genes:
-
Amplification of MYC, a gene affecting cellular proliferation, has been detected in 20%–30% of breast cancers studied (Berns et al., 1995);
-
Overexpression of BCL1 or PRAD1, a gene involved in cell cycle control, has been observed in approximately 20%–30% of breast cancers studied (Zukerberg et al., 1995);
-
Overexpression or mutation of erbB2/neu/HER2, a gene coding for a growth factor receptor, has been detected in approximately 30% of breast cancers studied (Berns et al., 1995);
-
Overexpression of BCL2, a gene whose protein is thought to prevent apoptosis and thus be capable of producing increased cell survival, has been observed in advanced breast cancer (Silvestrini et al., 1994);
-
TSG101, a recently discovered tumor suppressor gene, may be more specific, as it was found mutated in nearly 50% of breast cancers studied (Li et al., 1997).
Many of these genetic changes seem to occur during tumor progression and to be involved in the initiation of the malignant process. There are also genes, however, that may confer a higher risk of developing the disease—possibly including the ATM gene, which is mutated in ataxia telangiectasia patients (Savitsky et al., 1995). Individuals with Li-Fraumeni syndrome have inherited mutations of the p53 tumor suppressor gene, and have a high probability of developing breast cancer (Malkin et al., 1990).
Since 1993 two new predisposing genes have been identified and characterized: BRCA1 (Miki et al., 1994) and BRCA2 (Wooster et al., 1995). These genes appear to be responsible for a significant fraction of inherited breast and ovarian cancers studied. However, inherited mutations in these genes may be involved in the pathogenesis of only about 5% of all breast cancers. Studies to date suggest that somatic mutations in these genes do not appear to play a major role in the genesis of sporadic disease. Scientists agree that until the causes of breast cancer are understood, its prevention or eradication is unlikely.
OTHER RISK FACTORS
Despite extensive epidemiological studies spanning several decades, no single dominant etiology for breast cancer has emerged, as it has for lung cancer (i.e., cigarette smoking) (Kelsey and Gammon, 1990; Harris et al., 1992a). The etiology of breast cancer is likely to be quite complex, involving multiple endogenous and exogenous factors. Known risk factors for breast cancer explain approximately 20% of cases. The risk of developing breast cancer is increased by early age of menarche and late age at menopause. The risk of breast cancer is higher among nulliparous than parous women. The risk of breast cancer is directly proportional to the age of the woman at her first full-term pregnancy. Women with proliferative benign breast lesions (i.e., atypical fibroplasia) are also at an increased risk for developing breast cancer. Ionizing radiation can result in increased risk of breast cancer to women exposed as children and
teenagers, but few American women are exposed to the doses required to cause disease.
Women with first-degree relatives (i.e., mother, sister, daughter) diagnosed with breast cancer are themselves about twice as likely to develop breast cancer compared with women who do not have a family history of the disease (Colditz et al., 1993). The discovery of the genes BRCA1 and BRCA2 in affected families greatly enhanced studies of genetic risk and enabled predictions more precise than ever before (Serova et al., 1996).
BREAST IMAGING, TREATMENT, AND PREVENTION
The past decade has been a time of both great optimism and frustration in breast cancer research. The optimism stems in part from the emerging insights into the basic genetic and biochemical mechanisms of breast cancer. Studies in genetics, cellular biology, and molecular biology are providing glimpses into the intricate mechanism that determines when a cell is to grow, differentiate, or die, and how the genes involved in cancer disrupt this delicate and complex process.
Cancer researchers can envision the emergence, however indistinct, of precise targets for the treatment of breast cancer. If the complex mechanisms of breast cancer development can be elucidated, then it may be possible to move away from our current use of relatively toxic and ineffective treatment to more precise interventions that can eliminate the cancer and spare normal cells. Molecular tools may also permit the detection of breast cancer long before it is palpable or visible with mammography, thus enabling local intervention to bring about cure. Greater understanding of molecular biology may also make prevention strategies possible, since individuals at risk can then be targeted to receive systemic interventions that arrest the development of breast cancer.
Breast Imaging
It is generally accepted that the earlier breast cancer is detected, the greater the chance of long-term survival. However, by the time a breast cancer is palpable, 6–8 years may have elapsed since the first cancer cell developed, and micrometastases may be present (Hall, 1986; DHHS, 1996a). The goal of breast imaging is to facilitate the detection and diagnosis of small lesions, thereby permitting the use of less invasive treatment options.
Although the benefits derived from screening mammography among women less than age 50 years remain controversial (Elwood et al., 1993; Kerlikowske, 1996; NIH, 1997; ACS, 1997), mammography has been shown to reduce the mortality associated with breast cancer among women over 50 (Strax, 1990). Access to mammography, however, is a problem in women who
are either members of racial or ethnic minority groups, of low-income, or older age (Horton et al., 1992; Smith et al., 1992). Overall, fewer than half the women (42% of Caucasian and 34% of African-American women) who participated in a recent survey had regular screening mammograms (Romans, 1992).
Screening mammography is limited by its sensitivity and specificity, leading to false negatives and false positives. It is also limited because it causes discomfort and women may avoid it for this reason. Further research is needed in breast imaging and other screening methods, especially those generally acceptable to women. Current investigation in breast imaging includes digital mammography, radionuclide imaging (positron emission tomography [PET] scanning, scintimammography), magnetic resonance imaging (MRI), computerized tomography (CT scans), and virtual reality imaging utilizing combined images produced from MRI and CT scans.
Treatment
Although much progress has been made in determining the molecular and genetic events resulting in the development of breast cancer, progress has been slow in disease treatment. The major focus of treatment continues to involve conventional systemic therapies, such as chemotherapy and hormonal therapy, that are applied to all women in a nonspecific way. Advances in this area have been promising, and include the development of a new class of chemotherapeutic agents (e.g., taxanes—Paclitaxel and Docetaxel), and the integration of laboratory advances in monoclonal antibody production to the clinical arena (e.g., erbB2/neu/HER2 antibody).
Advances in supportive therapies that ameliorate the toxicity of chemotherapy have facilitated studies of dose intensity and its effects on breast cancer treatment. A correlation between intensity of chemotherapy and therapeutic response has been shown to some degree for patients with breast cancer (Hryniuk and Levine, 1986; Stewart et al., 1994; Wood et al., 1994); however, the risk/benefit ratio of more aggressive treatment has not been determined. Ongoing randomized trials are focusing on the efficacy of various dose intensity schedules, using chemotherapeutic agents sequentially or concurrently, or utilizing high doses of chemotherapy that require bone marrow rescue. The results of these studies are not yet available, and the potential toxicity may outweigh the benefit of aggressive treatment.
In general, the systemic treatment of breast cancer continues to progress slowly, most likely a reflection of our lack of understanding of the natural history of the disease and prognostic indicators, and ways in which specific agents could be targeted toward specific cancers and women at high risk. The need for further research to develop more efficacious and less toxic systemic
therapies that specifically target women who stand to benefit from a particular therapy is urgent.
Surgery and radiation therapy continue to be the major treatments for local and regional disease control. While clinical research continues to refine both specialties (e.g., sentinel lymph node biopsy studies and three-dimensional radiation treatment planning), major changes in the application of either specialty would likely be linked to research achievements in other areas.
Prevention
Progress in the field of molecular genetics has resulted in the ability to identify women who possess an inherited risk of developing breast cancer (i.e., germ-line mutations in BRCA1 and BRCA2). However, the medical community has not yet defined a rational therapeutic intervention for these women.
Investigational trials involving other high-risk groups are ongoing. Tamoxifen has been used extensively in clinical trials involving women with advanced and early-stage breast cancer, and has been observed to reduce the incidence of second primary breast cancers in the unaffected breast (Early Breast Cancer Trialists' Collaborative Group, 1992). This use of tamoxifen in a large, cooperative group trial is the first attempt to alter the molecular development of breast cancer using a preventive strategy. Other investigational drugs, including retinoids, limonene, and other monoterpenes, are also being examined for their potential to prevent breast cancer. The Women's Health Initiative and other randomized trials are examining the efficacy of dietary change in the prevention of breast cancer.
SOCIAL AND PSYCHOLOGICAL ASPECTS
The diagnosis of breast cancer and its treatment frequently take a significant emotional, social, and economic toll on patients and their families and on the quality of their lives. A substantial capacity for measuring the functional consequences and quality of life impact of the disease and its treatment now exists (McDowell and Newell, 1987; Stewart and Ware, 1992). Many of the available assessment methods provide a valuable opportunity to elicit the patient's or family members' evaluation of outcomes. Studies are needed to develop a better understanding of the full range of the disease and treatment outcomes and to identify the groups which are most vulnerable to breast cancer's adverse consequences (Barofsky and Sugarbaker, 1990). Research is also required to identify the social and psychological determinants of disease and treatment outcomes, better understand how patients and their families cope with issues of survivorship and recurrence, and determine how best to organize
continuing care and other supportive services. All research must endeavor to include more women who are older, poor, and members of racial and ethnic minority groups.
Although access to high-quality services must be available to all women, access problems persist, especially among low-income and minority women. Studies are necessary to address these inequities in access with particular emphasis on addressing both the institutional and individual barriers.
As tests for mutations in the BRCA1 and BRCA2 genes are becoming available, clinicians, for the first time, will be able to predict an individual's risk of breast cancer. This new capability has multiple ethical, legal, and psychosocial consequences that are not yet fully understood (Brower, 1997).