6
RISK MANAGEMENT AND THE WORLD TRADING SYSTEM: REGULATING INTERNATIONAL TRADE DISTORTIONS CAUSED BY NATIONAL SANITARY AND PHYTOSANITARY POLICIES
DAVID G. VICTOR
Council on Foreign Relations, New York
The Sanitary and Phytosanitary (SPS) Agreement, part of the 1994 accords that established the World Trade Organization (WTO), promotes international trade by requiring countries to base their sanitary (human and animal safety) and phytosanitary (plant safety) measures on international standards. However, it allows countries wide latitude to deviate from international standards when choosing their level of SPS protection, provided that (1) countries base their deviations on scientific risk assessment, (2) countries avoid discrimination by requiring comparable levels of SPS protection in comparable situations, and (3) countries not implement SPS measures that are more restrictive of trade than necessary to achieve the level of SPS protection that they seek. In this paper I review and assess the major provisions of the SPS Agreement (Appendix A), the international SPS standard-setting bodies, and the disciplines that govern allowable deviations from those international standards. I also examine the three WTO disputes that have helped to interpret the provisions of the SPS Agreement: the European Community's (EC)1 ban on meat produced using growth hormones, Australia's ban on imports of fresh and frozen salmon from Canada, and Japan's fumigation testing requirements for imported fruits and nuts.
Although disputes have not led to full interpretation of the major provisions of the agreement, it appears that the SPS Agreement has not led to the ''harmonizing down" of SPS protection that many opponents of free trade have feared. Instead, the wide latitude permitted by the SPS Agreement has allowed national diversity in SPS measures to thrive while also reducing barriers to trade. International standards have not become a straitjacket—rather, they have had remarkably little impact on national SPS protection policies. (The main exceptions are in countries, especially in the developing world, that have not already adopted elaborate SPS protection policies; for those countries, international standards fill gaps and raise—not lower—the level of SPS protection.) The main impact of the agreement appears to be in harmonizing the process by which nations set SPS policies—notably, it is promoting greater use of risk assessment at the national level. More extensive assessment of risks may actually yield greater diversity in national SPS policies. In the paper I also suggest that the novel mechanisms for providing expert advice to WTO dispute panels have been highly effective and have greatly reduced the problems of "advocacy science" that often plague the use of risk assessment in other judicial proceedings. The story—apparent success in imposing international discipline that promotes trade while accommodating national diversity—may be a useful guide for solving similar problems that are the mainstay of the "trade and environment" debate.
INTRODUCTION
One measure of the success of the postwar trading system is that tariff trade barriers have declined sharply. But the reduction in tariffs has exposed the many nontariff barriers that remain, and in many cases governments have kept protectionism in place by simply shifting from tariff to nontariff measures. Included in the broad category of nontariff barriers are differences in technical standards such as labeling requirements and environmental regulations. The focus in this paper is on one subset of these technical barriers: measures for sanitary (animal, including human) and phytosanitary (plant) protection.
SPS measures often have huge effects on trade; yet managing them is not easy. SPS measures vary across and within nations because preferences and circumstances vary. Some nations seek tight protection while others readily consume riskier foods; some pristine environments are vulnerable to pest infestations and require elaborate quarantines for imported products, but other countries are already overrun with pests. The political and technical challenge for advocates of free trade is to accommodate such differences while stripping away SPS measures that are merely disguised protectionism.
In this paper I examine the effectiveness of the 1994 WTO Agreement on the Application of Sanitary and Phytosanitary Measures (SPS Agreement), which is the most significant global effort to reduce trade distortions caused by differences in national SPS protection policies. I examine the major elements of the SPS Agreement and the three international SPS standard-setting processes that are explicitly mentioned in the SPS Agreement. I briefly consider two other
WTO agreements—the General Agreement on Tariffs and Trade 1994 (GATT) and the Agreement on Technical Barriers to Trade (TBT) that are often invoked, along with the SPS Agreement, in studies that examine how the international trading system attempts to accommodate differences in national regulations. I review the major elements and decisions of the three WTO disputes that have concerned SPS measures, which help reveal how the WTO system is interpreting the SPS Agreement. And I identify major conclusions that can be drawn about the operation of this system. Throughout, the goal is not only to assess the SPS Agreement but also to explore the policy question that arises wherever expanding the scope of free trade rules intrudes into national policy: Can international rules and institutions impose discipline on national policy without requiring harmonization to international standards? That question arises frequently—especially in the debate over "trade and environment"—and the SPS Agreement demonstrates a slightly positive answer.
THE SPS AGREEMENT: MAJOR ELEMENTS
The basic obligations for members of the world trading regime have not changed since the first GATT agreement in 1947: Members must give equal treatment to exports from all members, and members are barred from discriminating between locally produced and imported products. Exceptions were allowed for tariffs on specific products, that were "bound" at specific levels. Numerous other "general exceptions" were also allowed for many national policy purposes, such as protection of human, animal, or plant life or the conservation of exhaustible natural resources. But those general exceptions—listed in the famous Article XX—were described only briefly. A system of "dispute panels'' emerged to handle conflicts. In principle, the dispute panel system could have clarified the scope of Article XX. But in practice any GATT member could block adoption of a GATT panel report; and the panel system was often inactive, erratic in operation, and ineffective in major cases.2 Enforcement that did exist was mainly through reciprocity imposed by GATT members themselves. But the blunt instrument of unilateral reciprocity was poorly suited for working out and applying the complex legal interpretations that would be needed to make Article XX workable. In the early decades of the GATT, tariffs were the largest barriers to trade. The main result from each of the first six rounds of negotiations to strengthen the GATT was to revise the list of tariff bindings and reduce the tariff impact on trade. Nontariff measures remained in shadow.
For the past 30 years, attention to nontariff measures has grown. The 1979 Tokyo Round agreements, which resulted from the seventh round of negotiations, included a separate "standards code" that imposed discipline on technical barriers to trade. But the code, like the GATT agreement, was backed by little enforcement; although all GATT members were bound by the GATT's
core rules, they were largely free to pick and choose among "code" rules. The result of the Tokyo Round's "GATT a la carte," most experts agree, had little effect on lowering technical barriers to trade.
The failures of earlier efforts were addressed head-on in the most recent (eighth) Uruguay Round of negotiations. By 1986, the year that the Uruguay Round began, nearly 90 percent of U.S. food imports were affected by nontariff barriers to trade, up from only half in 1966 (Tutwiler, 1991, cited in Vogel, 1995).3 Exporters had a growing interest in taming these barriers.
The main legal products of the Uruguay Round were adopted in 1994. They were an updated version of the GATT (1994) along with 14 other agreements on textiles, subsidies, technical barriers to trade, SPS measures, and other topics. The Uruguay Round also produced a stronger and more judicial dispute-resolution procedure in which three-person panels hear and decide disputes and a standing Appellate Body hears appeals, and produced a mechanism that reviews trade policy in all member countries on a regular basis. Together, these agreements form a single, integrated package of obligations that constitutes the core obligations of a new international organization: The World Trade Organization.4 Countries were no longer free to pick and choose their free trade commitments.
The most important element of the WTO concerning SPS protection is the Agreement on the Application of Sanitary and Phytosanitary Measures (SPS Agreement). The agreement's central purpose is to promote international trade by limiting the use of SPS measures as disguised barriers to trade. The agreement's basic rights and obligations (Article 2) underscore that WTO members have the right to impose SPS measures as necessary "for the protection of human, animal or plant life or health" (Articles 2.1 and 2.2). But members may not arbitrarily or unjustifiably discriminate between members; nor may members use SPS measures as disguised barriers to trade (Article 2.3). These basic rights and obligations are quite general, and thus efforts to interpret them have focused on the more detailed provisions of the SPS Agreement (in particular Article 5, which is detailed below).
In addition to restraining the SPS policies that countries may develop on their own, the SPS Agreement urges members to implement international standards. The agreement's preamble underscores the goal: "Desiring to further
the use of harmonized sanitary and phytosanitary measures between Members, on the basis of international standards, guidelines and recommendations developed by the relevant international organizations. . . ." The agreement declares that "Members shall base their sanitary and phytosanitary measures on international standards, guidelines or recommendations. . . ." (Article 3.1). When a member imposes SPS measures that conform with international standards, guidelines, or recommendations, those measures will automatically be "presumed to be consistent with the relevant provisions of this Agreement. . . ." (Article 3.2). However, countries may introduce measures that are stricter than international standards "if there is a scientific justification, or as a consequence of the level of [SPS] protection a Member determines to be appropriate in accordance with the relevant provisions . . . of Article 5."5
Thus WTO members face a choice. A member may simply implement international standards,6 where they exist, or deviate from those standards. To examine how the agreement affects the SPS measures that countries implement, it is thus necessary to examine both outcomes: (1) how international standards are established, and (2) the exceptions that permit a country7 to deviate from
those international standards. I address these in reverse order because the exceptions are the most elaborate portion of the SPS Agreement and all of the disputes involving the SPS Agreement have focused on how to interpret the exceptions. If a country implements an international standard, it is automatically in compliance with the SPS Agreement, and thus all the WTO disputes concern instances where either international standards are absent, or a member has chosen not to implement existing standards.
Before turning to international standards and exceptions, it is important to note that the SPS Agreement includes several important obligations that extend the agreement's influence beyond simply the setting of SPS levels and measures. In principle, the SPS Agreement also allows exporters broad latitude when determining the SPS measures that are needed to meet the level of SPS protection that importers demand. The agreement requires that importers accept the SPS measures of exporters . . .
. . . as equivalent, even if these measures differ from their own or from those used by other Members trading in the same product, if the exporting Member objectively demonstrates to the importing Member that its measures achieve the importing Member's appropriate level of [SPS] protection (Article 4.1).8
Assuming that exporters have an interest in identifying the least trade-restrictive measure, this "equivalence" requirement could automatically ensure that SPS rules are not more discriminatory than necessary; "equivalence" could also open markets without requiring actual harmonization. In another context—the creation of the EC's single market—similar concepts (e.g., "mutual recognition") created a strong market-opening dynamic by allowing legal production from any European country into any other European national market. The agreement also requires that countries make their SPS policies transparent both through publication and creation of national "enquiry points" that can answer any reasonable question about that country's SPS rules (Articles 5.8 and 7, and Annex B). If that system operates properly then exporters will find it easier to comply with an importer's SPS rules, which should promote trade. Transparency is also essential to making use of the equivalence requirement described above. In addition, the agreement creates an international SPS Committee that meets on a regular basis to consider relevant topics and periodically review the performance of the SPS Agreement (Article 12). That committee is expected to adopt guidelines on SPS-related issues that could help in the interpretation of the agreement, although, to date, its impact on trade patterns has been minimal.
|
|
implemented by nongovernmental organizations (especially private firms, industrial associations and scientific laboratories), with government acting only a supervisor (see SPS Agreement, Article 13). |
The agreement allows the least developed countries to delay implementation of the agreement for five years (Article 14), allows other extensions, and empowers the SPS Committee to grant temporary extensions and relief from the agreement's obligations in cases of hardship.
The Exceptions
One of the most controversial aspects of the debate over opening trade has been the fear that free trade will force all countries to harmonize their national standards into a straitjacket of international standards. Donning the straitjacket, skeptics argue, could force nations to adopt stricter SPS measures than they would otherwise want. That might force societies to spend resources on SPS protection that they could have devoted to other purposes such as economic development. Or the straitjacket could force countries that already have tight SPS measures to relax them, leading perhaps to downward harmonization if international standards merely mirror the lowest common denominator. The latter has been the most controversial because existing SPS measures are generally much tighter in the advanced industrialized countries, which is also where most of the public interest groups active on SPS issues are located. Harmonization, they fear, will require compromising hard-won rules that protect consumers and the environment (Silverglade, 1998; Jacobsen, 1997).9
Because of this heated debate, fully under way when the WTO agreements were negotiated, the SPS Agreement permits countries to adopt SPS protection policies that are stricter or weaker than international standards. Rather than requiring harmonization, the SPS Agreement imposes discipline on both the level of SPS protection that countries seek and the measures they impose to attain those levels. The agreement and disputes over interpretation of the agreement have underscored that any country may set the level of SPS protection that it determines to be "appropriate." (This "appropriate level" is also often termed in the literature on risk management as the "level of acceptable risk.") The SPS Agreement does impose some discipline on the level of SPS protection, but it imposes more elaborate discipline on the measures that countries use to achieve that level. Below, I address the disciplines imposed on SPS levels and measures that are stricter than the international standards, and then I discuss measures that are weaker.
SPS Levels and Measures That Are Stricter Than the International Standard
The SPS Agreement is mainly intended to discipline SPS measures that cause an unjustified barrier or restriction on trade because they are stricter than international standards. Indeed, Article 3.3 (cited above) explicitly carves out an
exception to the goal of harmonization for SPS measures that are stricter than international standards. Article 3.3 requires that a member must be able to provide "scientific justification" for choosing a higher level of SPS protection. Similarly, Article 2.2 requires that members base their SPS measures on "scientific principles." These general requirements are quite broad and thus, in practice, the decisions of the Panels and Appellate Body in the three WTO disputes related to the SPS Agreement have turned to Article 5 for a more detailed description of "scientific" determination of SPS levels and measures.10
Article 5 requires that SPS measures be "based on an assessment, as appropriate to the circumstances, of the risks to human, animal or plant life or health, taking into account risk assessment techniques developed by the relevant international organizations" (Article 5.1, emphasis added). It requires that members take into account available scientific evidence (Article 5.2). When performing risk assessments, countries must account for economic factors such as potential loss in production or sales if a pest or disease enters the country as well as the cost effectiveness of different measures that could limit such risks (Article 5.3).
Article 5 also underscores that the agreement does not address every aspect of SPS protection. Rather, it concerns principally those SPS policies that affect trade. It urges countries to minimize the negative trade effects of SPS measures (Article 5.4). It requires that countries avoid "arbitrary or unjustifiable distinctions" in their levels of SPS protection "if such distinctions result in discrimination or a disguised restriction on international trade" (Article 5.5, emphasis added). Article 5.6 requires that countries not impose SPS measures that are "more trade-restrictive than required to achieve [the level of SPS protection that the member deems appropriate]." A footnote to Article 5.6 declares that a measure would be inconsistent with Article 5.6 if an alternative is found that passes each of the following three tests: (a) it is "reasonably available," (b) it achieves the member's appropriate level of SPS protection, and (c) it is "significantly less restrictive to trade than the SPS measure contested.'' Article 5.7 allows countries to adopt SPS measures even in the absence of good scientific information, provided that they also establish a process to obtain the information needed for a proper risk assessment.
These critical provisions in Article 5 essentially yield four rules that countries must follow when they impose SPS measures that deviate from international standards (or when no international standards exist):
-
The country must obtain a risk assessment (Articles 5.1, 5.2, 5.3, and 5.7).11
-
The SPS measures imposed must be "based on" that risk assessment (Articles 5.1 and 5.7).
-
The country must not discriminate or create disguised trade barriers by requiring different levels of SPS protection in comparable situations (Article 5.5).
-
The measures must not be more restrictive of trade than necessary to reach the level of SPS protection that the country desires (Article 5.6).
As shown below, the exact meaning of these four requirements is not obvious. However, Article 5 is the linchpin of the SPS Agreement—it puts discipline on SPS protection policies that countries adopt without requiring the politically impossible task of harmonization.
There is a revealing silence in Article 5 and other related provisions of the SPS Agreement.12 Article 5 is mainly concerned with ensuring that countries base their SPS measures on risk assessment and that they not adopt measures that are more restrictive of trade than necessary. It is largely silent on the level of SPS protection that a country seeks. Indeed, as mentioned above, several provisions of the SPS Agreement underscore that countries are free to set their own level of SPS protection, even if that level of protection is different from the level that would be afforded by international standards (e.g., Articles 2.1 and 3.3). The only provision in the SPS Agreement that specifically constrains the level of SPS protection that a country may set is Article 5.5, which requires that countries seek comparable levels of SPS protection in comparable situations.13
Thus, to determine whether a country's level of SPS protection is legitimate one must look inside the country itself—at whether the country consistently seeks a particular level of SPS protection. It is possible to interpret the requirements that SPS measures be based on a risk assessment (Articles 5.1, 5.2, 5.3, and 5.7) as also a requirement that a country's SPS levels also be based on risk assessment. Indeed, how can one assess the risks of SPS measures without assessing the risks associated with the level of protection as well? Levels and measures are two sides of the same coin.14 This remains a hotly contested issue because it concerns perhaps the most politically sensitive aspect of the SPS Agreement—whether it will encroach on a nation's sovereign right to determine its own SPS protection level.
SPS Measures That Are Weaker Than the International Standard
The other type of exception to harmonization is the reverse of the first: Nations may adopt SPS measures that are less strict than international standards. The requirement in Article 5 that standards be based on risk assessment and take into account available scientific evidence applies whether standards are stricter or looser.15
So far, none of the formal WTO disputes has addressed SPS measures that are less strict than international standards. Two reasons probably explain why the problem has not arisen: (1) the issue is most prominent in developing countries, many of which are still in transition to full implementation of the SPS Agreement; and (2) for many products, weak SPS measures are much less of a threat to free trade than strong measures. But it is conceivable that this type of exception will come under closer scrutiny and tighter discipline in the future. For manufactured goods, such as processed foods, there is often a substantial premium in efficiency for producers that can export to a market governed by a single standard. Lax standards, even if applied equally to local and imported products, could favor local producers and harm imports that are produced according to more expensive standards that prevail in the rest of the world
market. Using this argument, an alliance of global exporters and environmentalists may discover that the SPS Agreement is a very powerful tool—it could pry open local markets that are "distorted" by weak SPS standards and force a higher level of SPS protection. Whether the SPS Agreement is used in this capacity remains to be seen; such cases probably will be rare, not least because demonstrating the existence of a trade effect is difficult and bringing disputes is costly.
INTERNATIONAL STANDARDS
Although most of the SPS Agreement is focused on exceptions, its principal objective—stated in the preamble—is to promote harmonization of national standards.16 The SPS Agreement explicitly urges countries to adopt the standards set in three international processes: the Codex Alimentarius Commission (food safety), the International Office of Epizootics (animal safety), and the various organizations and processes that operate under the International Plant Protection Convention (plant safety). It also empowers the SPS Committee to identify other appropriate standards, guidelines, and recommendations.
In this section I discuss how these three intergovernmental processes set standards. Most attention is given to the Codex process because that has been the most active in actually setting standards and has, by far, attracted the most political attention because the safety of food for human consumption is the most politicized aspect of the SPS Agreement.
The Codex Alimentarius Commission17
In the aftermath of the World War II, the European nations created several institutions that were designed to promote trade and cooperation. Their architects hoped that the resulting economic integration would widen and deepen—by focusing on making money, Europeans would form a binding political union that would avert future war. The institutions included the European Coal and Steel Community (a predecessor of today's European Union) and the Codex Alimentarius Europaeus, established in 1958 to help harmonize methods for testing food safety in Europe. At the same time the
World Health Organization (WHO) and the Food and Agriculture Organization (FAO), spurred by the European dairy industry, created a committee to harmonize milk standards and thus open trade in milk and milk products. In 1962 WHO and FAO loosely merged these activities into the Codex Alimentarius Commission.
The commission's mandate was to develop and adopt food standards that would allow firms and countries to realize their self-interest: world trade in safe food products. From the outset the emphasis was on participation and consultation, especially with industry; engagement, the Codex architects hoped, would lead these stakeholders to harmonize their activities without the need for international enforcement (which was anyway not an available option). Thus, Codex standards are developed by committees of government representatives and stakeholders through an eight-step cycle shown in Figure 6-1. Technical committees evaluate evidence and elaborate standards, which are then subjected to the approval of the full Codex Alimentarius Commission, which meets every two years. That process of elaboration and approval typically occurs twice (steps 3–5 and 6–8 are a spin cycle), with the goal of ensuring wide input and consensus. Participation in the committee and commission meetings has been open to any stakeholder; yet only rarely have consumer and other public interest groups attended the committee meetings where standards are elaborated. The process is driven by industry, and the vast majority of Codex standards attract essentially no attention from other interest groups.
The commission adopts three types of standards: (1) commodity standards, which define what qualifies as a particular commodity (e.g., what is a "canned peach" or "natural mineral water"); (2) residue standards, which define acceptable levels of pesticides and food additives; and (3) codes of conduct and other guidelines that recommend, for example, good practices in the use of veterinary drugs or methods for risk assessment. To date the commission has adopted about 3,000 standards. Here I briefly review three aspects of those standards—how they are created, the role of risk assessment, and the sources of expert advice that are needed to weigh risks. I focus on commodity and residue standards. The other type of Codex norm—codes of conduct and guidelines—have been intended to augment application of the core standards rather than as principal standards themselves. In some cases, these looser guidelines have been adopted when agreement was not possible on a commodity or residue standard. However, if the SPS Agreement is interpreted broadly then these looser norms will have potentially binding application—that matter of legal interpretation has not been resolved or tested in any WTO disputes.18
The process of setting commodity standards has given practically no attention to risk assessment because most of the work of the Codex commodity committees focuses on the physical attributes of the commodity that, indirectly,
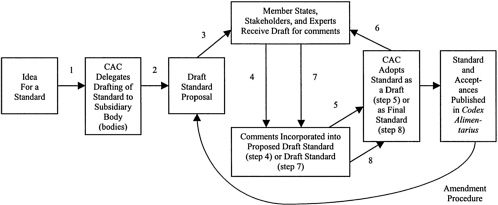
FIGURE 6-1. Elaboration of Food Safety Standards and Other Guidelines by the Codex Alimentarius Commission (CAC) and its Subsidiary Bodies. Major milestones (in boxes) and the eight steps by which standards are proposed (steps 1, 2), proposed drafts are reviewed and revised (steps 3,4), adopted (step 5), reviewed and revised again (steps 6, 7), and finally adopted as a Codex standard (step 8). Following adoption at step 8, member governments are requested to indicate whether they will accept the new standard. The standards and acceptances are published together as the Codex Alimentarius . A similar process is followed for the elaboration of advisory texts, such as codes of practice, except that they are not open for formal acceptance. In cases where the need for a standard is urgent or only one round of review and comments by member governments and stakeholders will be necessary, the commission can choose to omit steps 4–6 and operate an accelerated procedure. The commission can also amend an existing standard, typically sending it back to step 2. Progression throughout the steps is not necessarily linear; the commission often returns a standard to a previous step (e.g., to allow more time for comments and revision).
determine food risks; moreover, there are no specific Codex procedures for setting an "acceptable level of risk." Rather, commodity standards are intended to codify what is considered to be good practice for supplying safe food. Thus, de facto, risk assessment—where it exists—enters the Codex commodity standards from the "bottom up" through existing industry practice and standards. The committee members themselves provide the needed expertise—committees are populated mainly by government regulators and industry representatives who are best able to define characteristics of a safe canned plum or frozen pea. In practice, this organic and decentralized process led to haphazard commodity standards. Some commodity standards included excessive detail about the attributes of foods that were not necessary for food safety and, instead, merely entrenched existing industrial practices. To remedy this problem, a major review and revamping of Codex commodity standards is under way. The goal of that review is to simplify the standards and focus them on safety-related attributes of food products. However, the revamping is not intended to determine particular risk levels or risk assessment procedures that would govern the standard-setting process. So far, none of the WTO disputes related to SPS measures has involved a Codex commodity standard.
Many observers have raised the fear that commodity standards are vulnerable to "regulatory capture"—standards set to benefit the standard setters rather than the public interest. Indeed, the Codex history gives several suspicious examples. The standards for bee honey effectively barred many non-European honeys from the European market, although there was little basis for doing so on grounds of food safety alone. The worldwide standard for natural mineral water, adopted in 1997, requires that natural mineral waters be bottled at the source, which favors European producers who have long done so according to European law, and prohibits the use of antimicrobial agents that could make water safer. It penalizes American, Japanese, and other producers, many of whom truck or pipe their water prior to bottling and often treat it to ensure its safety. Yet there is not much justification in terms of food safety for the requirement. Piping and trucking do not intrinsically yield dirty water.19 The incorporation of the Codex into the WTO gives standards binding force and may increase the danger that commodity standards will be used for industrial promotion and not only for securing food safety. However, the danger has been longstanding, and incorporation into the WTO has brought other changes that
reduce that tendency—in particular, because the Codex is now applied in world trade, regional Codex standards have been eliminated.20 A coalition in favor of protectionism is easier to organize when participants are restricted to a like-minded region. Indeed, both the bee honey and natural mineral water standards emerged from regional European standards.21 Moreover, the "equivalence" provisions of the SPS Agreement (Article 4) will in principle allow exporters to circumvent international standards by using commodity standards that yield an equivalent level of SPS protection.
The Codex standard-setting processes for residues have made much more extensive use of risk assessment. Unlike commodity standards, which define all of the major attributes of a particular commodity, residue standards are simply a value for an acceptable residue (the "maximum residue limit" or MRL) of a food additive or contaminant for a particular food. The standards are set by identifying an acceptable daily intake (ADI) of the residue or food additive in question. Typically ADIs are established by identifying an animal that best mimics the most dangerous possible human response to the residue or food additive and determining the "no effect" level in that animal. What is meant by "no effect" and how it translates to human effects has not been rigorously defined or quantified. The ADI for humans is set by adjusting for the mass, diet, and lifetime of a typical human being compared with the test animal. (In the case of the bovine growth hormones, which is used as an example here because that WTO case involved a Codex residue standard, the typical human is 60–70 kg and the diet is generously assumed to be 500 g of bovine meat per day over an entire lifetime.) The ADI also includes a large safety factor. (In the bovine growth hormone case, the ADIs are 100 times lower than they would be without the safety factor.) A MRL is then calculated that would ensure that the ADI is not exceeded. If guidelines for "good practice" in food production—for example, guidelines for the use of veterinary drugs, which apply to the use of bovine growth hormones—would yield residues that exceed the MRL, then those guidelines are brought into line. In essence, the Codex system adjusts both the "good practice'' standards that govern how pesticides, drugs, and food additives are applied during food production as well as the residue standards that govern when the food products themselves are considered safe.22 In the case of bovine growth hormones, one expert testified that the MRLs adopted by the Codex Alimentarius Commission would result in a cancer risk of between 0 and
about one in a million; 23 but that was an estimate because the Codex system does not have a standard level of risk that guides its standard-setting activities.
Determining ADIs and MRLs is a highly technical process. Experts are needed to review the raw data from scientific studies and to calculate ADIs and MRLs. The Codex system has drawn on the recommendations of two joint WHO/FAO committees that are independent of and external to the Codex system: the Joint Meeting on Pesticide Residues (JMPR) and the Joint Expert Committee on Food Additives (JECFA). Both provide advice not only to Codex but also to many other activities of WHO, FAO and the United Nations system. In the Codex, JMPR and JECFA recommendations are used mainly by the three committees that set residue standards (i.e., MRLs): the Committee on Pesticide Residues, the Committee on Food Additives and Contaminants, and the Committee on Residues of Veterinary Drugs in Foods.
For all three types of Codex standards the working committees make recommendations, which they forward to the full Codex Alimentarius Commission for decision. To speed its work, the commission allows for simple majority voting when adopting a standard.
Prior to 1994—when the WTO agreements were finalized—the mere adoption of a Codex standard had no international legal consequences for Codex members. Thus it was rare for Codex standards to require a vote because a country could simply ignore an unfavorable standard. Indeed, standards were not binding unless the Codex member gave its formal "acceptance." The acceptance process allowed countries to pick and choose which standards they wanted to apply rigorously within their nations. For pesticide residue or food additive MRL standards, a country faced a simple binary choice: accept or not. For more complicated commodity standards, countries could accept the standard "with specific deviations," which gave them the opportunity to unilaterally tune the commodity standard to their own local conditions and preferences.
The combination of extensive consultation in standard setting, simple majority decision making, and the acceptance process makes it difficult to assess what impact Codex standards have actually had on national food safety standards and trade. The only hard data come from acceptances, which are not impressive. Table 6-1 shows that by 1993—on the eve of incorporation into the WTO—only 12 percent of the Codex standards had been accepted. Moreover, the pattern of commodity standard acceptances suggests that international standards followed rather than shaped national standards: in industrialized countries, which typically already had elaborate national commodity standards in place when international Codex norms were developed, nearly all acceptances were "with specific deviations."24 Deviations allowed them to tune international
Table 6-1. Acceptances of the Codex Alimentarius Commodity Standards (163 standards × 138 countries = 22,494 possible acceptances)
|
Acceptances |
Developing Countries (114 in 1993) |
OECD Countries (24 in 1993) |
Total |
|
Actual acceptances |
2,175 |
559 |
2,734 |
|
Possible acceptances |
18,582 |
3,912 |
22,494 |
|
Acceptance rate |
12% |
14% |
12% |
|
Type of Acceptance |
|||
|
Full |
1,215 (56%) |
100 (18%) |
1,315 |
|
With specific deviations |
228 (10%) |
252 (45%) |
480 |
|
Free distribution |
732 |
207 |
939 |
|
TOTAL |
2,175 (100%) |
559 (100%) |
2,734 |
|
Source: Compiled by author from 1989 acceptances, Vol. 14 of Codex Alimentarius Commission; updated 1991 and 1993. |
|||
standards to meet existing local standards; when the needed deviation was large the country could choose simply not to accept the international standard.
Voluntary standards and the acceptance procedure were designed to give states and stakeholders maximum control over which standards they adopted, which, in turn, dampened potential conflicts. Today, after the incorporation of Codex into the WTO, standards are no longer viewed as completely voluntary. Moreover, for purposes of the SPS Agreement, a standard is now considered "adopted" when it has been approved by the Codex Alimentarius Commission. The requirement of acceptance, which previously was the way that countries ensured that no Codex standard would be imposed against its wishes, no longer plays a role. For example, in the meat hormones case the Codex Commission had adopted standards for five of the six hormones in the dispute. 25 The EC did not accept the Codex MRL standards, but that nonacceptance was irrelevant to the requirement in the SPS Agreement that the EC base its national standards on international standards (and to provide justification where it did not).
Because of majority voting rules, in principle, the result may be a large number of standards adopted against a country's wishes. Moreover, the large number of Codex advisory texts and guidelines now also potentially have binding application through the SPS Agreement. What began as a voluntary body has been transformed into a very different purpose. Conflicts that should
have affected the standard-setting process—such as different views on the acceptable level of risk for products, food additives, and residues of veterinary drugs and pesticides—were latent in the Codex system but have now developed fully. Indeed, in recent years—especially in the two commission sessions that have been held since the SPS Agreement entered into force (1995 and 1997)—the commission's work is increasingly mired in controversy because it is now viewed as more relevant.
The International Office of Epizootics
The Office International des Epizooties (OIE) is an intergovernmental body established in 1924 with the purpose of protecting animal health. It serves as the umbrella for numerous commissions that prepare codes, protection strategies, and manuals. Some commissions work on specific diseases (e.g., fish diseases or foot-and-mouth disease), others work on problems of specific geographical regions. The OIE periodically revises the International Animal Health Code (OIE, 1998) which applies to mammals, birds, and bees; it is also the model for a separate International Aquatic Animal Health Code (OIE, 1997).
Both codes include the requirement that countries analyze and manage risks of diseases that are transmitted across borders via international trade and give special attention to adopting measures for controlling diseases that have minimum adverse effects on trade. As with the SPS Agreement itself, the codes also require that countries make their risk analysis transparent and be able to justify their import decisions. In short, the codes provide a basis for establishing quarantines and other sanitary measures and for adjusting the severity of the measures according to the economic risks. However, the requirements strictly apply only to diseases listed in each code; the lists are incomplete and thus offer only a starting point—countries are free to identify other diseases and regulate risks associated with them as well.
In addition to the codes, the OIE also produces guidelines for disease testing and surveillance programs and serves as a clearinghouse for current information on particular diseases (e.g., outbreaks). The work of these commissions is approved by the International Committee, the OIE's main decision-making body. The OIE is also the umbrella for numerous other collaborations that help to develop reference standards; various working groups promote debate that could lead to standards in areas such as biotechnology and wildlife. As of March 1998, 151 countries were members of the OIE.
International Plant Protection Convention
The International Plant Protection Convention (IPPC) entered into force in 1952 and was amended in 1979. It is intended to promote international coordination of measures necessary to limit the spread of plant diseases. The IPPC obliges countries to identify, assess, and manage risks to plants, including
risks from plant pests that are carried through international trade. "Guidelines for Pest Risk Analysis," developed within the framework of the IPPC, provide detailed information on how to assess and manage pest risks and require that countries develop import restrictions for protecting plant safety in conjunction with a broader plan for risk management.
The IPPC requires nations to create official plant protection organizations that perform inspections, conduct research, and disseminate information. Most countries would have such organizations in place even without the IPPC. As with the SPS Agreement, it requires that countries adopt phytosanitary measures only to the extent necessary for phytosanitary protection. Countries must use the least restrictive trade measures, avoid unnecessary delays during inspection and quarantine, and ensure that phytosanitary measures are transparent.26 The IPPC probably aids coordination of national plant protection policies—although some of that would occur anyway among those countries that want to coordinate—but it has not engaged in detailed standard setting to the degree of the Codex Alimentarius Commission or the OIE.
Summary
Of the three international standard-setting bodies explicitly mentioned in the SPS Agreement, Codex has been extremely active in setting standards for particular SPS hazards. The other two—OIE and IPPC—create mainly procedural obligations to conduct risk assessment and adopt SPS measures that are not excessively restrictive of trade, but those obligations are also enshrined in the SPS Agreement. All three also codify norms of good practice that include the requirement to base SPS standards on risk assessments. But those norms are quite broad. As I show below, they play little role in the detailed process of deciding whether a nation has complied with the SPS Agreement. Even in the Codex Alimentarius Commission—where the long experience in setting standards would suggest also long experience in applying risk assessment in formulating those standards—the actual practice of risk assessment is neither
transparent nor codified into institutional procedures. Indeed, the lack of codification is perhaps one reason why agreement has been possible. Risks are assessed and standards are set mainly through a bottom-up process that mirrors the risk-averse practice in advanced industrial nations.
OTHER WTO AGREEMENTS: GATT 1994 AND THE TBT AGREEMENT
Two other WTO agreements are often cited in the debate over how to manage technical barriers to trade and thus might be relevant for managing SPS measures. It is worth pausing to consider these two agreements and to explain why they are not centrally important to how risk assessment and discipline have been applied to SPS measures, which is the topic of this paper. Moreover, it is worth explaining why experience with managing nontariff trade barriers under the SPS Agreement should be applied only with great caution to the management of other technical barriers to trade.
First is the General Agreement on Tariffs and Trade 1994 (GATT 1994), which consists of the original 1947 GATT agreement and some revisions (e.g., new tariff schedules). It defines the basic obligations for members of the world trading regime and allows the Article XX "general exceptions" for various national policy purposes. Among the general exceptions is one for SPS purposes (Article XX(b)). However, all of the exceptions remain poorly elaborated and tested. Furthermore, the SPS Agreement declares that if members' SPS measures conform with the agreement that the measures "shall be presumed to be in accordance with the obligations of the Members under the provisions of GATT 1994 which relate to the use of sanitary or phytosanitary measures, in particular the provisions of Article XX(b)." Thus, what matters when determining the discipline on SPS measures is the SPS Agreement, not Article XX(b) of GATT 1994.
Second is the WTO Agreement on Technical Barriers to Trade (TBT Agreement). The TBT Agreement requires that WTO members not use "technical regulations" to discriminate against products imported from other members.27 The objectives that technical regulations serve must be based on sound science, including risk assessment. The measures employed must not be more trade restrictive than necessary to achieve the objective. The TBT Agreement also urges that, where possible, technical regulations should be based on international standards. Thus the TBT Agreement addresses the generic problems that are the subject of this paper: ensuring that nontariff trade restrictions are not merely disguised protectionism and promoting the use of risk
assessment and international standards in establishing those restrictions. But the TBT Agreement explicitly states that it does not apply to SPS measures (Article 1.5).
Thus, in practice, neither the GATT 1994 nor the TBT Agreement plays a significant role in governing SPS measures. Nor does the experience with SPS measures examined in this paper directly answer the critical question for Article XX: What threshold must be passed for a trade barrier to be valid as an exception listed in Article XX? Insofar as there is growing clarity in which SPS measures might be considered as compatible with the WTO, it is due mainly to the SPS Agreement. None of the other Article XX exceptions is governed by such a detailed separate agreement or understanding. How to interpret other Article XX exceptions—for example, protection of public morals (Article XX(a)) or exhaustible resources (Article XX(g))—still remains shrouded in mystery.
Great care must also be taken if lessons from this study are applied to non-SPS technical barriers to trade. The TBT Agreement applies a discipline to all technical barriers that is similar to the discipline imposed by the SPS Agreement. In particular, it requires that WTO members not impose technical regulations that are more restrictive of trade than necessary "to fulfill a legitimate objective" (Article 2.2); it requires members to use international standards "as a basis" for their technical regulations (Article 2.4). It establishes procedures that members must follow when they deviate from international standards (Articles 2.9–2.12, Article 4, and Annex 3). It requires members to make their technical regulations transparent (Articles 2.9–2.12) and urges members to treat other members' technical regulations as equivalent (Article 2.7). Thus, many of the same issues arise in both the SPS and the TBT agreements. However, there are important differences between the agreements—the SPS Agreement is narrower in scope. The TBT establishes a broad "Code of Good Practice'' for national standard-setting bodies, whereas the SPS Agreement relies on a mixture of specific obligations for national SPS regulatory bodies as well as numerous international guidelines developed by international standard-setting bodies. The TBT Agreement gives close attention to procedures that governments use when implementing technical regulations, whereas the SPS Agreement gives little explicit attention to implementation. The TBT Agreement also requires central governments to ensure that local governments and nongovernmental organizations comply with the agreement, whereas the SPS Agreement does not explicitly address these different layers of regulatory bodies.
Thus, this study attempts to offer insight into only the SPS-related aspects of trade. Some SPS issues also include TBT elements. For example, the dispute over the EC's ban on bovine growth hormones might be resolved by requiring labels on hormone-laced beef and inspection systems to ensure label accuracy. If so, the validity of the label system might be challenged as a technical barrier to trade that is inconsistent with the TBT Agreement. The present study is addressed only to the SPS-related aspects of such disputes.
THE SYSTEM AT WORK: THREE CASES
A full-blown assessment of how the SPS Agreement has affected the use of SPS measures should focus country by country, and measure by measure. That is impractical. The number of trade measures that could be affected by SPS disciplines is potentially huge. So far, only a small fraction has been subjected to international scrutiny. Many changes to national SPS policies will be time-consuming to implement; yet only four years have passed since the WTO agreements went into effect on 1 January 1995.
Thus, the approach here is to examine the three WTO dispute settlement cases that have concerned SPS measures: the EC's ban on imports of bovine meat produced with growth hormones ("EC meat hormones"), 28 Australia's ban on imports of fresh and frozen salmon ("Australian salmon")29 and Japan's ban on imports of numerous varieties of fruits and nuts ("Japanese fruits and nuts'') (WTO, 1998e, 1999). These cases reveal how the SPS Agreement has been interpreted to date and thus are the most instructive means available for beginning to assess the impact of the SPS Agreement.
Prior to the WTO, the dispute settlement procedure had few teeth and was, in essence, voluntary. Any GATT member could block adoption of a dispute panel report and thus block the formal remedies that might help to achieve compliance with trade rules and resolve the dispute. In practice the system was not completely anarchic, but nonetheless it was severely hobbled. The WTO system is more elaborate, has stronger tools at its disposal, is governed by strict timetables that help keep disputes from dragging out over years, and is less vulnerable to dissent. The WTO's Dispute Settlement Body (DSB) manages the process that begins with consultations and other efforts to resolve the dispute. If they fail then the DSB convenes a panel of three experts to hear the arguments of the parties and third parties, consult experts, interpret the relevant WTO obligations, and issue a report with rulings. Either party may appeal the rulings; three members of the standing seven-person Appellate Body review such appeals and issue a report with final rulings. The DSB must adopt Panel and Appellate Body reports; only a consensus of WTO members may block
adoption. Once the final report is adopted, the offending country must comply within a "reasonable period of time."30
Formal disputes are important not only because they often address important trade barriers themselves but also because they create interpretations of the law, focus expectations on how the WTO system will handle possible future disputes, and deter other violations. If disputes demonstrate clear discipline and a credible threat to dismantle trade barriers then countries will be more likely to remove illegitimate SPS measures on their own. Indeed, there is substantial evidence that the extended effect is significant—beyond the three measures that have been the subject of formal disputes, the SPS Agreement has been a "broader catalyst" that has induced some nations to remove illegitimate SPS measures (Roberts, 1998). Moreover, as with any properly functioning enforcement system, well-handled disputes can deter countries from imposing illegitimate SPS measures in the future. These extended and deterrent effects could be extremely important multipliers of the effect of individual disputes, but they are also difficult to assess.
The discussion here presents the basic facts and arguments in the cases.31 In the subsection "Analysis of the System at Work" below, I suggest the major issues and conclusions that should be drawn when examining the whole system: the SPS Agreement, the international standard-setting bodies, and these three cases.
The cases that have been brought to date, as shown below, are relatively clear violations of the SPS Agreement—thus the proper interpretations of central obligations of the SPS Agreement remain fuzzy. The situation may remain that way for a long time. The "transaction costs" of interpreting the SPS Agreement through cases are extremely high. Complainants, defendants, and third parties must prepare complicated arguments; panel members, WTO secretariat staff, and experts must sift through the evidence; the resulting panel decision typically occupies several hundred singled-spaced printed pages. Thus, the system may be inclined to the handling of winner cases in which the challenging member country is relatively sure it will prevail, or highly symbolic cases in which the challenging member country is politically unable to avoid a dispute. 32
EC Meat Hormones33
The first case concerns an EC Directive, imposed in 1981 and strengthened in 1988 and 1996, to ban imports of meat from farm animals that had been administered natural or synthetic hormones. Exceptions were allowed for hormones that are used for therapeutic purposes but not for hormones used to promote growth in cows. American, Canadian, and other beef producers used hormones to accelerate growth while reducing costs and yielding higher quality (leaner) meat. The United States had challenged the EC ban under the Tokyo Round "code" on technical barriers to trade, but the EC had blocked formation of an expert panel to examine the dispute. The conflict festered and became symbolic of why the voluntary Tokyo Round codes and nonmandatory dispute settlement were incapable of imposing discipline on nontariff barriers to trade.
At issue was whether the EC ban, which concerned six hormones, was compatible with the SPS Agreement. In 1995 the Codex Alimentarius Commission had adopted standards for five of the six hormones in the dispute. The standards were based on the work of the Codex Committee on Veterinary Drugs in Foods and the recommendation of JECFA, which had reviewed the scientific evidence related to hormones twice. The Codex standards did not impose MRLs for the three natural hormones in question (oestradiol-17<Symbol,SR,SY>b, progesterone, and testosterone) because naturally produced residues would far exceed the additional residue caused by "good-practice" use of these hormones for promoting growth in cows. For the other two synthetic hormones (trenbolone acetate and zeranol, which mimic the biological activity of natural hormones), the MRLs adopted were far less strict than the level that would be expected if good veterinary practices were followed. There were no Codex standards for melengestrol acetate (MGA), a synthetic hormone administered as a feed additive that was included in the EC ban.
The EC argued that the SPS Agreement explicitly allows WTO members to adopt standards that are stricter than international norms if those standards are based on an assessment of risks. Every risk assessment of these hormones had shown that growth hormones applied according to good veterinary practices would result in no significant harm to humans—those assessments included two major reviews by JECFA (1988 and 1989) and at least two reviews commissioned by the EC itself. 34 The EC argued that, although those studies
|
33 |
For more on the origins of this dispute see Vogel (1995, Chapter 5); for more on the WTO aspects of the dispute see Charnovitz (1997), and Roberts (1998). |
|
34 |
32nd JECFA Report, published in 1988 (1988 JECFA Report); 34th JECFA Report, published 1989 (1989 JECFA Report); Report of the Scientific Group on |
suggested that there was no objective risk, numerous highly publicized incidents since the early 1980s during which hormones entered European food markets had made European consumers wary of beef.35 A ban, the EC argued, was necessary to restore confidence in the market.36
The WTO Dispute Panel ruled against the EC on three grounds. First, it argued that the EC's measure was illegal because more-permissive international standards existed for five of the hormones. The Dispute Panel interpreted Article 3.1 of the SPS Agreement, which declares that "Members shall base their sanitary or phytosanitary measures on international standards" as a requirement that SPS measures conform with international standards.37 In perhaps its single most important ruling on SPS-related issues, the WTO Appellate Body explicitly overturned this interpretation, preferring instead the more common-sense definition of "based on:" A measure can be based on international standards without conforming with those standards. Instead of conformity, the Appellate Body pointed to Article 3's fundamental purpose: to promote the use of international standards while allowing countries to deviate from those standards if those deviations conform with Article 5 which pertains to the use of risk assessment (WTO (1998d), paras. 160–177). This approach of the Appellate Body, although obviously more consistent with the purpose of the SPS Agreement than the narrow interpretation imposed by the Dispute Panel, was nonetheless a watershed—it removed a legal interpretation that could have resulted in international standards becoming the feared straitjacket.
Second, the Dispute Panel also ruled that the EC measure was not based on a risk assessment as required in Article 5.1. The Appellate Body agreed. The Panel and Appellate Body found this for five of the hormones that the EC had obtained assessments of some risks. Among these assessments, only a 1982 report of the EC Scientific Veterinary Committee (the Lamming Report) and two reports by JECFA (1988 and 1989) qualified as adequate risk assessments.38 The Appellate Body
underscored that risk assessments need not be based entirely on research in the physical sciences; nor must risk assessments examine only quantitative risks. However, the EC measure failed because the EC had not applied risk assessment techniques to the particular risks that the EC claimed were the basis of its SPS measures (an import ban). The EC had argued, for example, that misuse of hormones as growth promoters could cause excessive risks and thus all use of hormones for growth promotion must be banned, but the Appellate Body concluded that the EC had not actually presented an assessment of such risks (WTO, 1998d, paras. 207–208). Hence the conclusion that the EC measures were not based on a risk assessment. Moreover, the Appellate Body decided that not only is there a procedural requirement to obtain a risk assessment, but in addition: "The requirement that an SPS measure be 'based on' a risk assessment is a substantive requirement that there be a rational relationship between the measure and the risk assessment" (emphasis added).39 The fact that all of the valid risk assessments showed that "good practice" application of growth hormones was safe—and the failure to examine the risks that the EC claimed could result in harm to consumers—meant that the EC measure failed the "rational relationship" test. But the exact contours of that test remain unexplained.
For the other hormone (MGA), no valid risk assessment existed and thus, by definition, the EC measure was not "based on" a risk assessment (WTO, 1998d, para. 201).40
Third, the Panel found that the EC had violated Article 5.5 of the SPS Agreement by demanding different levels of SPS protection in comparable situations. Notably, the EC allowed carbadox and olaquindox to be used as antimicrobial feed additives that promoted the growth of pigs; yet the EC banned the use of hormones as growth promoters in cows although the hormones resulted in similar (or lower) risks to humans. The Appellate Body overturned that decision by declaring that the SPS level required by a country would be incompatible with Article 5.5 if it failed each of the following three tests: (1) the country did not require comparable levels of protection in comparable situations, (2) the failure to apply comparable measures in comparable situations is arbitrary and unjustifiable, and (3) such measures result
in discrimination or a disguised restriction on international trade. 41 The Appellate Body found that the EC had, indeed, applied different SPS levels in comparable situations and thus failed the first test. 42 The EC ban also failed the second test because the EC could not justify this difference in treatment. But the Appellate Body argued that the third test—whether "arbitrary or unjustifiable" differences in SPS levels harmed trade—was most important, and the complainants provided insufficient evidence that the EC measure failed that test. Allowing carbadox and olaquindox as feed additives on the one hand while barring hormones for promoting growth in cows on the other was not by itself evidence of a disguised barrier to trade. Erecting a trade barrier was not the purpose of the EC rules that created this incongruous situation—in the words of the Appellate Body the "architecture and structure" of the EC Directives was not discriminatory or a disguised restriction on trade. The EC applied the same level of SPS protection (with a ban on hormones as growth promoters) equally to imports and domestic production. Nor had the United States or Canada submitted adequate evidence that the different treatment had resulted in "discrimination or a disguised restriction on international trade."43
In sum, the Panel viewed the SPS Agreement as requiring strict adherence to international standards and sharply limiting a nation's right to determine its SPS levels and measures. The Appellate Body, which is more attuned to the political and social context in which the SPS Agreement and the WTO operate, gave importers much greater autonomy in setting SPS policy. Whereas the Panel found three main reasons to rule against the EC, the Appellate Body endorsed only one—the EC's failure to base its SPS measures on a risk assessment.44
Australian Salmon
This dispute, the second involving SPS measures to result in a Panel decision, concerned an Australian regulation dating from 1975 that bans imports of fresh or frozen salmon in order to prevent 24 fishborne diseases from spreading into Australia's pristine environment. Many of the diseases could adversely affect trout, which are vital to Australian sport fishing and tourism as well as a small trout aquaculture industry. And the diseases could also harm the Atlantic salmon aquaculture farms, first established in 1986 in Tasmania, that export salmon to world markets and also sell their product on the local Australian market. To combat the threat, Australia required heat treatment for all imports from regions where fish might become infected with the diseases.
The OIE listed two of these diseases in the International Aquatic Animal Health Code category of fish diseases that are particularly dangerous threats for spreading. Such transmissible diseases "are considered to be of socio-economic and/or public health importance within countries and that are significant in the international trade of aquatic animals and aquatic animal products" (OIE, 1997, Section 1.1). The OIE also listed four of the diseases in a category of fish diseases that are less well understood but potentially dangerous. For diseases on either list, OIE "Guidelines for Risk Assessment" require countries to undertake analysis to examine the "disease risks associated with the importation" and to tailor particular import controls to the real-world situations in the country.45 The remaining diseases were not listed by OIE and thus no special OIE guidelines were applicable.46
Canada, a major exporter of fresh and frozen salmon, challenged Australia's regulation. Canada did not dispute that Australia had the right to preserve a pristine environment—that is, in the jargon of the SPS Agreement, Australia had the right to determine its own "appropriate level of SPS protection." But, Canada argued, the quarantine was arbitrary because Australia did not apply similarly strict quarantine measures against other disease risks. Australia had allowed imports of frozen herring bait fish and live ornamental fish that could much more easily transmit many of the 24 diseases into Australian waters, but it barred Canadian salmon. Bait fish are, by design, disposed directly into waters where diseases could easily pass to other fish. Ornamental fish often escape their ponds and aquaria; when they die they may be disposed without care for the risk of transmitting diseases to other fish in Australian waters. In contrast, headless and eviscerated fresh or frozen salmon from Canada had low incidence of the diseases and could transmit the disease into the Australian fish population only through a long and implausible chain of events.47 Numerous risk assessments supported the Canadian argument. As the EC argued in the meat hormones case, Australia maintained that, although the risks were low, it could not be certain that headless eviscerated fish would not spread disease.
The Panel and Appellate Body ruled against the Australian measure largely on three grounds. First, the Appellate Body determined that Australia's ban on imports of fresh and frozen Canadian salmon was not based on an assessment of risks. In doing so, the Appellate Body established a three-pronged test for what would qualify as a risk assessment: (1) identification of the diseases and possible biological and economic consequences of their entry or spreading; (2) evaluation of the likelihood of entry, establishment, or spreading; and (3) evaluation of the impact of SPS measures on the likelihood of entry, establishment, or spreading of the diseases.48 Australia's "1996 Final Report," which established the ban on imports of fresh and frozen salmon, met the first requirement. But the Appellate Body said that Australia had failed the other two tests. This finding overturned the Panel, which had ruled that the 1996 Final Report did constitute a "risk assessment." The Panel had followed the cue of the earlier Appellate Body report on EC meat hormones, which suggested that the requirement of the SPS Agreement be "based on an assessment," which implied
that WTO members could include many diverse factors. But the Panel had wrongly assumed that this permissive standard also meant a low threshold for what qualified as a risk assessment. The Panel concluded that the 1996 Final Report "to some extent evaluates" the risks and risk reduction factors and thus qualifies as a risk assessment, but the Appellate Body established a stronger test for compliance.
Second, the Panel and Appellate Body found that the import ban on fresh and frozen salmon was a disguised restriction on trade. Both the Panel and the Appellate Body stressed that Australia was free to determine its own level of SPS protection; however, they found that Australia did not apply that high level of protection in other comparable situations. By allowing imports of bait and ornamental fish, Australia exposed itself to greater risks than from salmon imports; not treating these comparable risks in comparable ways revealed that the salmon import ban was a disguised restriction on trade. To reach this decision the Panel applied the three-step test that the Appellate Body had developed in the EC meat hormones case: (1) it decided that the situation of disease risks from salmon imports was comparable with the disease risks from ornamental and bait fish because they involved similar diseases, media, and modes of propagation; (2) such different treatment for salmon and other disease risks was "arbitrary or unjustifiable;" and (3) the different treatment for salmon resulted in a disguised restriction on international trade. The Appellate Body agreed. Whereas the third element of the test failed in the EC meat hormones cases, the evidence was much stronger in the salmon case. The evidence included the fact that the draft of Australia's salmon rules would have permitted the importation of ocean-caught Pacific salmon under certain conditions; but the final rule—issued after stakeholders such as the Australian salmon industry had commented but based on substantially the same risk assessment information—barred imports. That factor, compounded by many other "warning signals," led the Panel and Appellate Body to decide that the import ban was, indeed, a disguised restriction on trade.49
Third, the panel decided that the particular SPS measure required by Australia—heat treatment of salmon prior to export to Australia—was more trade restrictive than necessary and thus violated Article 5.6 of the SPS Agreement. Heat treatment, in effect, barred Canadian salmon from a lucrative segment of the market because heat treatment, by definition, converted fresh or fresh-frozen fish into less valuable heat-treated fish. (Moreover, some experts consulted by the Panel suggested that heat treatment might actually raise the
disease risks because elevated temperatures were not high enough to kill all pathogens and could cause some to grow more rapidly.) An alternative sanitary measure—requiring the beheading and evisceration of fish—would yield a similar level of SPS protection for Australia with a much less deleterious impact on Canada's exports. The Appellate Body appeared to be inclined to agree with the Panel, but it overturned this aspect of the ruling. The Appellate Body argued that the SPS measure at issue was not heat treatment but rather the import ban on fresh and frozen salmon from Canada. (Because of that ban, the only means available to Canada to supply salmon to the Australian market was heat treatment.) The Appellate Body overturned the Panel because it could not determine Australia's "appropriate level of protection" and, therefore, could not determine whether the Australian measures were excessively restrictive on trade. The Appellate Body underscored that "determination of the appropriate level of protection . . . as a prerogative of the Member concerned [Australia]. . . ."50
Japanese Fruits and Nuts
The final case concerns a Japanese regulation that had the effect of requiring exporters of various fruits and nuts to submit each new variety they intended to export to Japan to an extensive regime to verify that fumigation with methyl bromide would effectively kill the eggs and larvae of coddling moths.51 The case focused on four species (apples, cherries, nectarines, and walnuts), although it potentially had application to others. The required treatment varied not only with the characteristics of the fruit or nut but also the season of harvest because coddling moths exist in different forms (e.g., eggs, larvae, adults) in different seasons. Different varieties have different harvest times, and thus Japan argued that test results for one variety were not applicable to another.52 The United States challenged the requirement as not based on an assessment of risks; it also argued that the varietal testing requirement imposed excessive costs and delays and thus was more trade restrictive than required. The United States
contested only the measures that Japan had applied; it explicitly did not question Japan's right to determine its ''appropriate level of SPS protection"—that is, for Japan to ensure that its pristine islands remain free of the coddling moth (WTO, 1998e, para. 827). 53
The Panel found that Japan's testing requirements were inconsistent with the SPS Agreement for three reasons. First, the varietal testing requirement was not based on a risk assessment. (The failure to employ risk assessment also violated the IPPC's requirement to base plant protection measures on risk assessments. However, in practice, the IPPC's requirements were redundant of the SPS Agreement's obligation to base measures on risk assessment; thus the IPPC played no significant role in this dispute.) In particular, the Panel concluded that "it has not been sufficiently demonstrated that there is a rational or objective relationship between the varietal testing requirement and the scientific evidence submitted to the Panel" (WTO, 1998e, para. 827). Japan claimed that its goal was to ensure that new varieties would impose no danger of coddling moth infestation that was greater than the infinitesimal risk of infestation from varieties that had already undergone extensive testing. Each variety must be tested individually, Japan argued, because there may be a chance (although extremely small) that differences between varieties of fruits and nuts could lead to ineffective treatments that would let a coddling moth slip through. However, the Panel found that " . . . so far not a single instance has occurred in Japan or any other country, where the treatment approved for one variety of a product has had to be modified to ensure an effective treatment for another variety of the same product (WTO, 1998e, para. 872)." Moreover, the United States as well as experts advising the Panel had shown that varietal differences did not influence the efficacy of quarantine methods, and Japan had not presented adequate evidence to the contrary (WTO, 1998e, para. 827).54
The Appellate Body endorsed the conclusion that the Japanese testing requirement was not based on a risk assessment; echoing Article 2.2 of the SPS Agreement, the Appellate Body found that the testing requirement was maintained "without sufficient scientific evidence" (WTO, 1999, para. 76, and SPS Agreement Article 2.2). However, as in the hormones and salmon cases, the
Appellate Body also avoided creating any standard for "sufficient" or "rational relationship;" instead, they found "[w]hether there is a rational relationship between an SPS measure and the scientific evidence is to be determined on a case-by-case basis and will depend upon the particular circumstances of the case, including the characteristics of the measure at issue and the quality and quantity of the scientific evidence'' (WTO, 1999, para. 84).
Japan argued that Article 5.7 allowed countries to adopt stringent measures when "relevant scientific evidence is insufficient." The Panel underscored that Article 5.7 is an exception to the general risk assessment obligations of the SPS Agreement (i.e., Articles 2.2 and 5.1) that applies only to provisional measures. The language of Article 5.7 itself suggests that such provisional measures must meet four cumulative requirements:
-
the measure is imposed where "relevant scientific information is insufficient,"
-
the measure is adopted "on the basis of available pertinent information,"
-
the member must "seek to obtain the additional information necessary for a more objective assessment of risk," and
-
the member must "review the . . . phytosanitary measure accordingly within a reasonable period of time" (SPS Agreement, Article 5.7).
The Panel concluded that Japan had failed on at least both the third and the fourth requirements (WTO, 1998e, paras. 8.49–8.60).
Second, the Panel also found that the varietal testing requirement was more trade restrictive than necessary and thus violated Article 5.6 of the SPS Agreement. Because there is no significant difference in the efficacy of fumigation techniques across different varieties of the same product, alternative measures—such as setting fumigation requirements on the basis of the easily measured "sorption level" of new varieties, rather than a full retesting of each variety—would be less restrictive of trade yet still achieve the level of SPS protection that Japan requires (WTO, 1998e, paras. 8.70-8.104).55 The Appellate Body overturned this ruling because it was based on evidence marshaled by the Panel itself and thus the Panel had overstepped its authority; 56 the United States had not, first, presented a prima facie case that a measure based on determination of sorption levels would have met the requirements of Article 5.6 and thus been less trade restrictive than the Japanese varietal testing scheme (WTO, 1999, paras. 123–131). 57
Finally, the Panel and Appellate Body found that Japan had violated the requirement to make its SPS measures transparent, especially the requirement in Article 7 that countries publish their SPS measures. The Japanese varietal testing requirement was based on numerous de facto rules that were not easily understood by outsiders, which made it difficult for exporters to understand and comply with the requirements of the Japanese market.
ANALYSIS OF THE SYSTEM AT WORK
Here, I examine the legal requirements of the SPS Agreement and the first three SPS cases from the perspective of several major conclusions that can be drawn at this early stage of implementation.
First, however, it is important to underscore that the legal interpretation of the agreement is still evolving. The SPS Agreement was written carefully to allow countries to determine their own "appropriate level" of SPS protection and to determine which SPS measures are necessary to meet that level. It would not have been politically possible to adopt an SPS Agreement that forced strict harmonization of either the level or measures for SPS protection. Nor would strict harmonization be necessary to avoid most of the trade distortions caused by SPS measures. The agreement thus aims to promote trade by imposing strict disciplines on the process by which members set SPS protection levels and measures that affect international trade. Judging the success of the SPS Agreement thus requires determining whether and how those disciplines have been implemented, rather than whether particular world standards have been adopted or even whether nations have adopted the same SPS levels and measures.
The three WTO disputes have helped to clarify the obligations in the SPS Agreement, but the disputes have also left many areas still uncertain. One, perhaps the most important, is the central obligation of the SPS Agreement: SPS measures must be "based on scientific principles," in particular an "assessment . . . of the risks" (Articles 2.2 and 5.1, respectively). All three cases underscore that there is a difference between risk assessment and risk management—the former is a scientific process that examines the magnitude and distribution of possible risks, and the latter employs risk assessment as well as many other factors in determining and attaining the appropriate level of risk. The hormones case underscores that the SPS Agreement does not mandate a particular quantified relationship between risk assessment and risk management. Indeed, the Appellate Body explicitly underscored that when WTO members
determine their SPS measures (i.e., when they manage risks) that many nonquantitative factors (e.g., consumer confidence) may enter the equation.58 But in the hormones case the SPS measures were ruled invalid because the EC appeared to have made no use of risk assessment when determining its SPS measures. Indeed, in that case and the other two cases, WTO panels or the Appellate Body showed that risk assessments conducted by the offending governments themselves did not support the imposition of such extreme trade controls.59 In all three cases the defendants nonetheless supported their actions because, they argued, there could be some risk if the imported products were not subjected to stringent SPS measures. The WTO panels and Appellate Body rejected that argument because science can never prove that an action will result in absolutely zero risk, and thus policy-making requires the assessment of risks. The Appellate Body in the meat hormones case set the standard which has since prevailed: There must be a "rational relationship" between risk assessment and a country's SPS measures.60 But the Appellate Body defined neither "rational" nor "relationship." The standard is both procedural and substantive, but the more important substantive element is unclear. By allowing many nonquantitative factors to enter the equation, the Appellate Body appears to have set a low threshold for meeting this standard, but how low is it? In the Japanese fruits and nuts case the Appellate Body shed no further light when it underscored that this determination must be made on a case-by-case basis (World Trade Organization, 1999, and text above).
Second, it is also unclear how to interpret the requirement in the SPS Agreement which bars countries from setting SPS protection levels that "arbitrarily or unjustifiably discriminate between Members where identical or
similar conditions prevail . . ." (Article 2.3). That requirement is echoed in Article 5.5, which requires that countries apply measures that yield a comparable level of protection under comparable situations. 61 The SPS Committee is charged with developing guidelines to distinguish comparable from noncomparable situations, but that will not be easy. In two of the three disputes to date, it has been relatively easy to identify a comparable situation where allowable SPS risks were substantially different. In the EC hormones case, carbadox and olaquindox were allowed for swine production, but natural and synthetic hormones were not allowed for beef production; yet both substances have been linked to a similar health effect (carcinogenicity); some experts consulted by the WTO Dispute Panel even suggested that such feed additives could directly harm workers who handled feeds.62 In the salmon case, Australia allowed imports of frozen herring bait and live ornamental fish that harbored many of the same diseases that Australia feared would arrive on imported salmon; yet fresh and frozen salmon were effectively barred, which protected the nascent Australian aquaculture industry.63 These easy cases have not given much clarity to what is comparable, except to underscore that comparable will not be interpreted in the narrowest possible manner as a requirement to impose comparable levels of SPS protection only when exactly the same sources and types of risks are at stake.64
A third area of important ambiguity concerns the criteria for judging the trade effects of SPS measures. The SPS Agreement is not a catchall requirement that countries must base all SPS levels and measures on science and risk assessment. Rather, it seeks only to bar SPS levels and measures that cause
unjustifiable distortions of trade.65 However, there is no agreed standard for how large a distortion of trade is necessary for a level or measure to qualify for the discipline of the SPS Agreement. With globalization, practically every national measure has an effect on trade, which would suggest that all SPS levels and measures could be subjected to the discipline of the SPS Agreement. However, in the EC meat hormones case, the Appellate Body appeared to decide that trade distortions must be clear and severe. It invalidated comparison between the meat hormone ban and the EC's rules that permit use of carcinogenic feed additives (carbadox and olaquindox) as "comparable situations" because the trade distortions caused by this difference in SPS protection levels were unclear and not demonstrably large (see footnote 41 and text). Furthermore, Article 5.6 of the SPS Agreement requires that members ensure that SPS measures "are not more trade-restrictive than required" (Article 5.6). Yet there is no clear standard for what is "more trade restrictive."
As the literature on national risk management shows, often risk trade-offs occur on many dimensions and are not simple. As the SPS system develops, there may be attention to trade-offs between national risks and the costs and benefits to international trade. Should a nation be urged or required to lower its SPS protection slightly, if that would allow use of much less distortionary SPS measures? Logically there is only a small step from the existing SPS Agreement—which gives countries the right to determine their "appropriate level of SPS protection"—to one that requires nations to consider international trade effects when determining SPS protection levels. Politically, it would be a huge leap.
The problem of making fine determinations in the allowable degree of trade distortion is unlikely to generate many WTO disputes for one simple reason: There must be a strong and apparent trade effect for a complaining country to justify the cost of raising and prosecuting a dispute. Vague, indirect, and secondary effects could be numerous, but they are difficult to demonstrate and are typically diffused across many actors. In contrast, identifiable and concentrated costs have a larger effect in mobilizing harmed exporters to seek change, especially if the exporters are already organized and have ready access to the agents (governments) that can prosecute the case.
Mindful of these three areas of continued uncertainty, I now turn to major conclusions that can be drawn about how the WTO system has imposed discipline on SPS measures to date.
No "Race Toward the Bottom" or "Downward Harmonization"
Putting discipline on SPS measures that affects trade is but one example of the interaction between the international trading system and national regulation. Freeing trade requires reducing barriers, including nontariff barriers such as national regulations, which has led many observers to fear that free trade could cause a "race toward the bottom." They fear that governments, keen to promote their exporting industries, will dismantle environmental regulations. Many have also feared that promoting the use of international standards in order to yield a level playing field will require "downward harmonization" toward the lowest common denominator. Both fears are important to examine, not only for a proper assessment of the SPS Agreement's effect on national policy but also more generally because they are central issues in the "trade and environment" debate.66 The experience with the SPS Agreement can thus help illuminate whether and how similar fears can be addressed when other areas of national policy collide with the international goal of promoting trade by restricting nontariff barriers to trade.
The experience to date gives no support for either fear. There is no evidence of a "race to the bottom." However, it is unlikely that such a dynamic would develop in the case of SPS protection. The greatest fear with the race to the bottom is that free markets will create a strong incentive for producers to adopt less costly processes that make exports more competitive but have deleterious effects, such as greater pollution, in the locality of production. For SPS issues, however, the importing country retains the power to set its own level of protection, and that choice does not affect environmental quality in the exporting country. Direct competitive pressure to relax that level is largely nonexistent.
Fear of downward harmonization is potentially more valid. Indeed, the main critiques of the SPS Agreement from public interest groups have claimed that the agreement will lead to downward harmonization. Detractors further fear
the transfer of decision-making authority to international standard-setting bodies such as the Codex Alimentarius Commission, which they argue are undemocratic and captured by industrial interests (Silverglade, 1998; Jacobsen, 1997). Public interest groups rightly worry that their voices will not be heard when the Codex determines standards—with few exceptions, they have been poorly represented at Codex meetings, especially technical meetings where most of the detailed work of designing standards occurs (Victor, 1998). However, downward harmonization has not been much evident in practice. Legally, there is no requirement for harmonization. Rather, the SPS Agreement itself clearly states that it is for countries themselves to determine their own "appropriate level" of risk and to adopt SPS measures as necessary to meet that level. The political sensitivity of the WTO system—especially the Appellate Body—to this public fear of harmonization is apparent as it makes sure that WTO agreements are not interpreted as a guise for dismantling legitimate regulation. The decisions in all three SPS-related cases have carefully underscored the sovereign right of nations to determine their own level of SPS protection. The WTO Dispute Panel in the first case—on the EC meat hormones ban—interpreted the SPS Agreement narrowly and suggested that the inconsistency between the EC measure and the Codex standards was evidence that the EC measure was incompatible with the SPS Agreement. If that Panel interpretation had held, then there would indeed be reason to fear that the SPS Agreement would force strict harmonization with international standards, perhaps resulting in some downward harmonization. But the Appellate Body explicitly overturned the Panel's reasoning as too narrow; instead of strictly requiring conformity with international standards, the Appellate Body decided that countries had considerable latitude in setting SPS levels and measures that were different from international standards. The Appellate Body also set high hurdles for judging that a country's SPS levels and measures were incompatible with the SPS Agreement. The final decision remained the same—the EC ban was declared incompatible—but the reasoning was much more permissive of deviations from international standards.67
The three disputes might also be cited as evidence that the existence of the SPS Agreement and dispute resolution, at least in these three cases, has resulted in lower SPS protection. Indeed, in all three cases the SPS measures were struck down. However, in all three cases alternative measures offered a similarly high level of SPS protection with a much lower impediment to trade. Nonetheless, perhaps allowing any marketing of hormone-grown beef in Europe, for example, represents a reduction in SPS protection due to the SPS Agreement. However, if the science is sound—and on this there is a remarkably broad consensus among scientists—then allowing the beef on the market represents no objective reduction in SPS protection.
The fact that all three of the SPS disputes to date have resulted in SPS measures being barred is not evidence that the system is instinctively opposed to strict national SPS protection. Rather, it is the product of a dispute resolution system that is both at its early stages and costly to invoke. In such a system we should expect that all or nearly all disputes raised will be settled in the complainant's favor.
What is perhaps more surprising is that more of these disputes are not settled prior to a Panel decision. Part of the explanation is that dragging out a dispute can afford an additional two to three years of protectionism. A more important explanation may be found in the EC meat hormones case. The ban on hormones is a highly politicized issue in Europe, exacerbated by the controversy over mad cow disease and genetically modified foods. A hot issue for nearly two decades, the coalition of consumer and protectionist interests are deeply entrenched. And the interests became more entrenched with time. The bans did not begin as protectionist measures but rather the responses of some EC governments to uncertain science and consumer outrage. But, over time, beef producers realized that the ban served their interests as well as did managers of the EC agriculture subsidy system, which would be even more bloated if beef production were more efficient. Politically the EC and member governments could not afford to succumb easily to international pressure.
Especially where food is concerned, these highly politicized issues will be commonplace. But a special challenge for the WTO system will be for governments to ready their societies to accept defeat and implement reform. It probably has made matters worse that the EC painted itself further into a corner by continuing to argue that European consumers required the ban to restore confidence in the market and then fanning consumer fears of the same. It is plausible that the EC has learned the lesson that this is a bad strategy because it makes the eventual fall longer and harder. Today, in the handling of import bans for genetically modified foods (which are not currently the subject of a WTO dispute, but are being debated in the Codex Alimentarius Commission and have been the subject of numerous risk assessments), the EC has taken a stance that is more favorable to international trade. Instead of outright bans, it favors tight regulation coupled with labeling.68 Governments have also learned how to play the "SPS game" so as not to run afoul of trade laws. The SPS Agreement allows strict provisional measures when risks are uncertain, provided that they also make an effort to reduce uncertainties (Article 5.7). A form of the "precautionary principle," this provision allows governments to defend strict measures. The EC hormones case could not use that defense because final rules
had been adopted before the WTO dispute was filed. Look now for governments to adapt few "final" rules when their SPS measures could be contested.
Internal Alignment of National SPS Policy Processes and Risk Levels
Thus the SPS Agreement has not required weakening of SPS measures that countries apply to protect humans, animals, and plants. But it may have a different, much larger effect on how countries manage risks. In all three of the disputes one of the critical complaints has been that import bans were arbitrary—challengers argued that the importers had required less restrictive SPS protection in other comparable situations. Although not all of those complaints were successful, the intense focus on ensuring comparable treatment has put all members of the world trading system on notice that they must be able to justify SPS regulations that were previously regarded as purely internal policy matters. Exporters have at their disposal a powerful mechanism—the SPS Agreement backed by the strong WTO dispute resolution system—with which they can attack SPS measures that are inconsistent with the rest of a nation's SPS protection scheme.
If countries are under constant pressure to justify that they adopt comparable SPS measures in comparable situations, then they are likely to give much greater attention to internal alignment of risk assessment and management policies—in other words, they are more likely to ensure that they impose comparable levels of risk management in comparable situations. They are also more likely to ensure that the particular measures they impose are based on risk assessment. The consequences of these external pressures will include much greater application of risk assessment and more transparent national SPS rules. That could be a boon for those who advocate the making of public policy according to sober assessment of risks.
(The requirement for greater use of risk assessment may make it transparent that, in the extreme, the concept of a "comparable situation" logically requires a very broad interpretation. How does one square policies that tolerate high levels of risks in one area—such as from air pollution—with de minimus standards for many food hazards? These are perennial questions in risk management, but the SPS Agreement may now bring them to the WTO.)
However, the impact of much greater transparency and internal alignment of risk management on trade is not obvious. Greater transparency should facilitate trade by making it easier for importers to identify and comply with applicable rules. Greater transparency will also make it easier for exporters to declare that they have imposed SPS measures on their exports that achieve the same level of SPS protection that the importing country achieves with (possibly different) SPS measures that it requires. If so, Article 4 of the SPS Agreement (Equivalence) requires the importer to let the goods flow. In democratic societies, more transparency may also make governments less likely to adopt rules that would be embarrassing and vulnerable to attack. The requirement that
SPS measures not be more trade restrictive than necessary should also facilitate trade.
But greater use of risk assessment, by itself, could have mixed effects on trade. Internal alignment of risks will eliminate grossly protective SPS measures, which should open trade.69 But it may also result in countries tightening some SPS measures to ensure that overall national SPS policy is in alignment. One of Australia's main responses to the argument that allowing imports of potentially disease-carrying live ornamental fish was incompatible with their ban on imports of fresh and frozen salmon was to point out that it was reviewing the rules that govern imports of ornamental fish (and other potential carriers) (WTO, 1998b, para. 4. 190). Similarly, the EC's response to the inconsistency between allowing the use of known carcinogens (carbadox and olaquindox) while prohibiting hormones used for growth promotion was to underscore that the carcinogens were under review and might be regulated more tightly (WTO, 1998d, para. 234).
Thus, internal alignment could raise or lower SPS protection, and more analysis is needed to uncover which effects will occur. The net effect of internal alignment may greatly increase "good government" and yet have remarkably little effect on trade. More assessment should make it easier to identify SPS measures that achieve a given level of protection with fewer restrictions on trade. But more assessment may also aid the development of alliances between advocates of tight SPS protection and others who want to restrict trade, which could lead to more SPS-related trade barriers. More attention to SPS issues worldwide should lead to an international learning process focused on risk management. However, that process might result in learning about some SPS measures that are deemed "legitimate" under the SPS Agreement and are also particularly effective protectionist trade barriers—for example, quarantine measures imposed by countries that are free of particular diseases allow the importer to impose "equal treatment" on both local and foreign production while effectively barring imports. Nations may also learn that protectionist measures will not run afoul of the WTO if they also serve plausible SPS protection goals. In the EC hormones case, for example, the Appellate Body ultimately did not declare that the inconsistency between allowing the use of known carcinogens and banning growth hormones was incompatible with the SPS Agreement. The Appellate Body maintained that the inconsistency was "arbitrary or unjustifiable," but proof had not been offered to show that the difference failed the requirement of Article 2.3 of the SPS Agreement: ''sanitary . . . measures shall not be applied in a manner which would constitute a disguised restriction on international trade." The hormone ban had multiple effects—some legitimate
and others protectionist—but there were plausible reasons for the measure. For example, the Appellate Body found that "depth and extent of the anxieties experienced within the European Communities concerning . . . the carcinogenicity of hormones [and] . . . scandals relating to black-marketing and smuggling of prohibited veterinary drugs . . ." were legitimate reasons for regulatory action (WTO, 1998d, para. 245). Moreover, the Appellate Body also judged as reasonable the fact that the EC had banned hormones across the community—although only a few EC members had taken such action on their own—because the goal of a barrier-free internal market required a common measure across the EC. Whereas the Panel had focused on the protectionist reasons for the inconsistency, the Appellate Body underscored the legitimate ones. By this reasoning, the Appellate Body thus set a high standard for complainants to meet when they attempt to prove that a measure has protectionist aims. The lesson learned by protectionists is certainly to shroud a protectionist measure with legitimate food safety concerns. Expect bootleggers to seek out (or invent) Baptists to help press their interests.
More generally, increased attention to evaluating risks could result in a greater number and diversity of SPS measures. As societies have become more aware of risks and better able to afford risk management, they have demanded more stringent social regulation. Within this context, international rules that force countries to look more closely at their SPS policies are likely to yield more SPS measures by accelerating the tendency for countries to impose SPS measures. And the SPS measures that countries do adopt are more likely to be tuned to local conditions and interests if they are explicitly based on risk assessment. It is thus plausible—perhaps even likely—that the result of greater attention to SPS measures will be greater diversity in SPS levels and measures, not harmonization.
More systematic analysis is needed to determine the trade effects of the SPS Agreement's effort to harmonize the process of SPS protection—in particular, the requirement that members employ risk assessment. It should not be assumed that in all conditions a binding requirement to base SPS measures on risk assessments will lead to more trade or even to more harmonization.
International Standards: Little Impact
Although the central purpose of the SPS Agreement is to promote harmonization of SPS measures, the three cases suggest that international standards have not had much impact. That finding is surely biased by the cases examined here—dispute panels are likely to hear only those cases for which national SPS measures do not conform with international standards since cases where there is conformity are explicitly in compliance with the SPS Agreement and thus yield no viable dispute. Thus, perhaps international standards are having a large unseen effect; systematic research on that possible effect is needed.
In each of the three WTO cases, international standards were referenced in the resolution of the disputes. But none of the outcomes from the disputes actually required the existence of an international standard. The EC hormones case made most extensive use of international standards, but that was because the Codex system—in particular JECFA (which is formally external to Codex)—had given extensive attention to the hormones under review. Even so, the dispute panels did not rely exclusively on the JECFA reviews. Rather, the Panel (advised by experts it had retained) looked at the entire scientific literature, which included several non-JECFA reviews of hormone risks. The JECFA reviews were helpful and set a clear benchmark for quality scientific assessment, but the other scientific reviews came to the same conclusions. Moreover, by overturning the narrow interpretation of the SPS Agreement as requiring conformity with international standards, the Appellate Body underscored that international standards were at best a starting point for countries that wanted to deviate from them. Indeed, the existence of international standards was irrelevant for the main line of legal reasoning that decided the EC meat hormones case—the failure for the EC to establish a "rational relationship" between risk assessment and the measures it imposed. The lack of any international standard for one of the six hormones (MGA) did not excuse the EC from the obligation to base even its ban of that hormone on a risk assessment (see footnote 39 and text above).
The minimal influence of international standards is even more evident in the Australian salmon and the Japanese fruits and nuts cases. In those cases the OIE and IPPC, respectively, had few, if any, standards that were directly applicable to the issues in the disputes. Only a few of the fish diseases were on the lists of diseases in the OIE's International Aquatic Animal Health Code, and thus for only those did OIE offer specific guidelines for imposing trade restrictions. For the other diseases, OIE was largely silent. Both OIE and the IPPC promulgated general standards for risk assessment that could be applicable in those cases where more specific international methods and standards did not exist, but those guidelines were so broad as to be essentially irrelevant to the resolution of these two cases.
This suggestion—that international standards have had much less importance than expected—may hold in the future as well, but for an additional reason. Greater internal alignment of risk management procedures (see above) need not result in alignment according to international standards. Indeed, because the SPS Agreement allows liberal deviations from those standards, more systematic national risk assessment and greater public debate over acceptable risks are likely to result in many more deviations. The history of the Codex Alimentarius acceptances (Table 6-1) lends support to this argument. Developing countries lodged more "full acceptances" of Codex commodity standards because they did not have many SPS measures already in place. But industrialized countries—especially those with the most advanced SPS protection systems—employed principally "acceptances with specific deviations." This suggests that international standards are a fluid that can fill gaps (when countries let the fluid flow), rather than a solid block that crushes
deviation. Moreover, this history suggests that the natural progression of risk management may be toward diversity, not harmonization.
In a decade from now it may be clear whether the assessment suggested here has proved robust—that the SPS Agreement is having a large effect on the process of national risk management without much increasing the degree of harmonization with international standards. That experience may help build consensus around the view that the solution to nontariff barriers to trade is discipline, not harmonization. Whether discipline alone will be enough to achieve adequately free trade remains to be seen. But it could be adequate to blunt opposition to trade liberalization that has, in part, been based on the fear that free trade requires harmonization.
International Standards: From Artificial Consensus to Conflict
All three of the international standard-setting bodies examined in this paper were created for different purposes and then grafted to the WTO system. The discussion here focuses on the Codex Alimentarius Commission—the most important of the three. Codex was originally created to provide a forum that would facilitate coordination and create standards that countries would implement voluntarily. The SPS Agreement has changed that dramatically by making the standards legally binding and enforceable.
The change in status is bound to lead to greater conflict. The consensus and ease of operation that prevailed in the past was artificial—it existed because the standard-setting bodies were not particularly relevant and entirely optional. Today the stakeholders believe that the standards are much more relevant than before. They might be wrong (see above), but it is perceptions that matter. The newfound importance of these international bodies is requiring much greater attention to decision-making procedures and also exposing standards organizations to greater conflict.
The requirement in the SPS Agreement that SPS measures be based on risk assessment unless they are based on international standards will underscore the need for international standards themselves to be based on risk assessment. Yet, to date, none of these organizations has applied a systematic policy for determining acceptable levels of risk. The Codex is in the midst of a systemwide reassessment of risk-related concepts and procedures. But progress has been very slow. Before the conclusion of the SPS Agreement, the Codex Alimentarius Commission contained no principles or definitions related to the application of risk assessment and risk management (Codex Alimentarius Commission, 1993). Today it has several general statements on the role of science and risk assessment, and efforts are under way to expand treatment of risk in setting Codex standards.70 However, the Codex risk principles and
definitions are only broad statements that appear unlikely to have much practical impact on the work of Codex or on how cases under the SPS Agreement might be handled. It is essential that Codex continue development in this area, but it is also likely that debates over the risk principles will underscore that in the past the Codex standards were easily adopted because there had not been a rigorous attempt to align the risk levels across all standards. Trying to do so would have revealed that gaining agreement on a single world level of acceptable risk—which, in turn, could determine Codex standards—is difficult or impossible.71
To date, the Codex standards have largely reflected risk management procedures in the advanced industrial countries. They are developed with extensive input from industry, mainly (but not exclusively) in the advanced industrialized countries. The industry's interest is to ensure that international standards are consistent with national practices. Thus they seek international standards that mirror those at home. The result is that the agenda and standards in the Codex are determined heavily by the SPS policies in the advanced industrialized countries. Similarly, the large safety margins and the desire to set standards at the "no effect" level reflect the goal of the advanced industrialized countries, which is to set food safety risks as close to zero as is practical. It remains unclear whether that view of the proper risk level is shared among all Codex members. Until the application of Codex standards through the SPS Agreement, few Codex members had paid close attention to the exact safety levels that the Codex system assured.
Rising conflict in standard-setting bodies should not be lamented. It is the by-product of a shift from a voluntary (often ineffective) system of standards to a scheme that may have more binding impact. That shift has made some players less willing to sacrifice their interests for the sake of agreement. Previously, compromise was less painful because the Codex Alimentarius procedures, especially the provisions for acceptance, were rife with opportunity to opt out of inconvenient commitments. The impression that international standards are now more relevant has also entrained new actors—such as consumer protection organizations—into the process, and with new voices and interests it has proved more difficult to reach consensus or even majority agreement.
But it is worthwhile to ask whether the strategy adopted has been the most effective. In the case of the GATT, it was the GATT members themselves that made the shift from the weaker GATT 1947 framework to the integrated WTO system. They changed not only the legal framework but also the stringency of commitments and the mechanism for enforcement. In the case of the Codex, however, the change in de facto legal status has arrived on its doorstep from the
outside; internal Codex procedures and mechanisms were not reformed at the same time. With Codex standards now much more relevant, is the huge committee system with majority voting the most efficient way to adopt standards? If, in the future, other standards organizations are brought within a binding framework, is there anything that can be done to prepare the way?72
Finally, although conflict can be productive it does risk greatly slowing the work of international standard-setting bodies because nations (or at least a majority of them in each case) are less willing to adopt standards that could be relevant. The failure of the Codex Alimentarius Commission to adopt a standard for bovine somatotropin—for which scientific evidence of safety is comparable with that of growth hormones—suggests that, on politically charged topics, the standard-setting bodies may become bogged in deadlock. The result will be a lack of standards (or broad and meaningless guidelines that are equally useless). That deadlock may not matter for the disciplines of the SPS Agreement because the requirement to base national SPS levels and measures on risk assessment (Article 5) remains in place, even when international standards do not exist.
However, on balance, the deadlock on standards probably hurts the free trade agenda. The one area where international standards have been consistently influential is when filling gaps—in areas of food law and in nations not already covered by standards. As markets open, the number of gaps—especially in countries where administrative capacity is low—will grow, at least in the short term until countries catch up with the process of national risk assessment and management. International standards could thus play an especially important role in opening trade to new markets, new products, and new methods of SPS protection. Examples currently on the agenda of the WTO include genetically modified organisms, labeling, and a scheme for more consistent implementation of SPS measures known as Hazard Analysis and Critical Control Points. If nations are gridlocked in Codex because they fear binding application in the WTO, they will not have adequate international standards to guide their efforts to address new SPS threats and new opportunities for improved SPS protection.
Expert Advice
The experience with SPS discipline offers several encouraging stories for the perennial problem of incorporating expert advice into the processes of risk assessment and risk management. This problem has often been poorly handled within countries, especially when the need for expert advice intersects with
adversarial legal proceedings. Often the result is a duel between expert advocates that buries and confuses "the truth."
The experience reviewed in this paper confirms that independent expert panels, in contrast with dueling experts, can be a highly effective device for synthesizing complex technical information. The Codex committees that debate the proper level for MRLs would have found it very difficult to agree on standards without the advice of the JECFA and JMPR expert committees. Experts have also played a vital role in the resolution of the three WTO disputes. In each dispute, the WTO dispute panels enlisted experts to answer technical questions. 73 Although it is difficult to assess what would have happened if the dispute panels had not had access to their own experts, it is clear that the experts' assessments formed a prominent part of the record and decision-making process in each case. Approximately one-fourth of each Panel report consists of the transcript from the consultation with expert panelists. Each of the three cases depended critically on expert interpretations of scientific evidence as well as evaluations of the adequacy of risk assessments. The central issue for the Panel in the meat hormones case was to determine the merit of the EC's claim that there were consumer risks associated with using hormones for growth promotion. In the salmon case, two critical judgments relied heavily on technical information—whether headless and eviscerated fish posed disease risks, and whether other imports (e.g., frozen bait herring) posed comparable risks. In the Japanese fruits and nuts case, the critical issue was the highly technical conflict over the efficacy of fumigation techniques.
The SPS Agreement may also change the type of information that is demanded of experts and sought through the risk assessment process. All three of the WTO panel cases made extensive use of risk assessments. But each case required not only the "normal" elements of risk assessment—an evaluation of the risks to humans, animals, or plants in the importing territory—but also an assessment of how different measures to manage those risks would affect trade. As a growing number of national SPS measures come under scrutiny for their consistency with the SPS Agreement, this "trade impact assessment" aspect of risk assessment will probably become commonplace.
SUMMARY
In this paper I have reviewed the provisions of the 1994 SPS Agreement and the first three WTO disputes related to the application of the SPS Agreement. I have argued that large areas of interpretation remain open.
However, the disputes have allowed some interpretation of all the major obligations of the SPS Agreement. In particular,
-
The SPS Agreement urges countries to apply international standards, but it explicitly allows countries to deviate from those standards if they can justify the deviation. However, SPS policies that deviate from international standards must be based on risk assessment. In the Australian salmon case, the Appellate Body clarified the "three prongs" of a satisfactory risk assessment and demonstrated why the risk assessment in the Australian salmon ban was not adequate.
-
Adequate risk assessments did exist in the two other cases (EC meat hormones and the Japanese fruits); however, in both of these cases the Panels and the Appellate Body deemed the SPS measures illegitimate because there was no "rational relationship" between the risk assessment and the measures applied.
-
The SPS Agreement also requires that countries apply comparable SPS policies in comparable situations. In the EC meat hormones case the Appellate Body created a three-part test that clarified this obligation. Applying this test, the Appellate Body found that the ban was "arbitrary or unjustifiable," but it did not declare the ban invalid because there was insufficient evidence that the ban also constituted a disguised restriction on international trade (SPS Agreement, Article 5.5). In the Australian salmon case the evidence for discrimination was stronger, and Australia's ban was declared invalid.74
-
The SPS Agreement also requires that countries apply the least trade-restrictive measures. In two cases (Australian salmon and Japanese fruits and nuts), WTO panels ruled that other measures were available that would achieve the same level of SPS protection and were substantially less restrictive of trade. The basic logic of those findings has been upheld, although the Appellate Body overturned each on other grounds.75
-
The SPS Agreement requires that countries make their SPS policies transparent so as to facilitate compliance and trade (Article 5.8 and especially Article 7). In the Japanese fruits and nuts case, the Panel found that the transparency provisions had been violated because importers found it difficult to understand the de facto rules that governed imports into Japan, and Japan had not published the relevant material in a way that was accessible to outsiders.
Thus, nearly all the major obligations of the SPS Agreement have been addressed, at least partially, in disputes: the urging of nations to apply international standards, the requirement to base SPS measures on risk assessment, the requirement to apply comparable policies in comparable situations, the requirement to use the least trade-restrictive measures available, and the requirement that nations make their SPS policies transparent. The only major requirement that has not been addressed in a dispute concerns "equivalence" (Article 4), which obliges each member to accept the SPS measures of other members as equivalent if the exporter can demonstrate that its SPS measures achieve the level of SPS protection required by the importer. This could prove to be the agreement's largest effect on trade, but working out what is meant by "equivalence" will be complicated.
Although many areas of interpretation remain gray, the legal text of the SPS Agreement and the cases to date have underscored that nations have wide latitude in setting their SPS protection levels and measures. Thus, far from imposing a strict harmonization between national and international standards—which was the main fear of the agreement's detractors—the agreement actually allows diversity to flourish. The agreement is likely to result in increased use of risk assessment, especially in nations where risk assessment has been used only rarely or never, and to promote debate within nations about proper SPS risk management. More informed and extensive debate will likely lead to even greater diversity in SPS levels and measures.
The evidence suggests that harmonization of SPS levels and measures is not under way. However, harmonization of national SPS procedures , such as the requirement for risk assessment, is evident. Procedural harmonization without the strict requirement for harmonization of levels and measures may help to mute the backlash against globalization that, in part, is animated by the fear that national sovereignty is being lost to undemocratic international standard-setting bodies. That procedural harmonization is largely the result of how the Appellate Body—a standing body that serves as a steward of the WTO system and is politically more astute than the panels, which are convened for a particular case and then disbanded—has interpreted the SPS Agreement. In particular, the Appellate Body overturned the legal reasoning of the Dispute Panel in the hormones case, which had maintained that national SPS measures must conform with international standards. The Appellate Body's interpretation probably gives nations more latitude than the creators of the SPS Agreement had originally intended. Advocates of international rule of law probably lament that outcome, but it seems to have been politically wise.
Even procedural harmonization has been difficult to implement. At this writing, the United States and Europe are in the midst of a trade war over meat hormones—a case that Europe lost not because it was forced to don the straitjacket of international standards but because it could not demonstrate that there was any "rational relationship" between risk assessments and the standards it had adopted. To stay on an even keel, the WTO may need additional devices to serve as a safety valve when international rules come into direct conflict with entrenched national interests. With the WTO agreements in 1994, overnight
members of the world trading system shifted from a system in which there was essentially complete freedom to adopt numerous national SPS measures that affected trade to one where freedom is constrained by the SPS Agreement. The transformation has not been easy and is not complete; if not managed with political sensitivity to the domestic backlash it could readily derail.
REFERENCES
Boardman, R. 1986. Residues of pesticides: the Codex system. Chapter 4 in Pesticides in World Agriculture: The Politics of International Regulation . New York: St. Martin's Press.
Charnovitz, S. 1997. The World Trade Organization, meat hormones, and food safety. International Trade Reporter 14(41):1781–1787.
Codex Alimentarius Commission. 1993. Procedural Manual. Rome: Joint FAO and WHO Foods Standards Program. Rome: FAO.
Codex Alimentarius Commission. 1997. Report of the 22nd Session, Geneva, 23–28 June. Rome: FAO.
Codex Committee on General Principles. 1998. Risk Analysis: Working Principles for Risk Analysis. CS/GP 98/4, Thirteenth Session, Paris, France, 11–15 May 1998. Rome: FAO.
Hudec, R.E. 1993. Enforcing International Trade Law: The Evolution of the Modern GATT Legal System. Salem, Mass.: Butterworth Legal Publishers.
Jacobson, M.F. 1997. Consideration of Codex Alimentarius standards, advance notice of proposed rulemaking. Comments of the Center for Science in the Public Interest. Docket 97N-0218. U.S. Department of Health and Human Services and Food and Drug Administration, Washington, D.C.
Kay, D.A. 1976. The International Regulation of Pesticide Residues in Food. Washington, D.C.: American Society of International Law.
Leive, D.M. 1976. International Regulatory Regimes: Case Studies in Health, Meteorology and Food, 2 vols. Lexington, Mass.: Lexington Books for the American Society of International Law.
Maddox, J. 1995. Contention over growth promoters. Nature 378:553.
OIE (Office International des Epizooties). 1997. International Aquatic Animal Health Code, 2nd ed. Paris: OIE.
OIE (Office International des Epizooties). 1998. International Animal Health Code, 7th ed. Paris: OIE.
Roberts, D. 1998. Preliminary assessment of the effects of the WTO Agreement on sanitary and phytosanitary trade regulations. Journal of International Economic Law 1(3):377–405.
Roberts, D., and K. DeRemer. 1997. Overview of Foreign Technical Barriers to U.S. Agricultural Exports. ERS Staff Paper No. 8705. Economic Research Service, Commercial Agriculture Division. Washington, D.C.: U.S. Department of Agriculture.
Silverglade, B.A. 1998. The impact of international trade agreements on U.S. food safety and labeling standards. Food and Drug Law Journal 53:537–541.
Tutwiler, A. 1991. Food Safety, the Environment and Agriculture Trade: The Links . Discussion Papers, Series No. 7. Washington, D.C.: International Policy Council on Agriculture, Food Trade.
Victor, D.G. 1998. The operation and effectiveness of the Codex Alimentarius Commission. In Effective Multilateral Regulation of Industrial Activity: Institutions for Policing and Adjusting Binding and Nonbinding Legal Commitments. Ph.D. thesis, Department of Political Science, Massachusetts Institute of Technology, Cambridge.
Vogel, D. 1995. Trading Up: Consumer and Environmental Regulation in a Global Economy. Cambridge, Mass.: Harvard University Press.
WTO (World Trade Organization). 1997a. EC Measures Concerning Meat and Meat Products (Hormones), Complaint by Canada. Report of the Panel, WT/DS48/R/CAN. Geneva: WTO.
WTO (World Trade Organization). 1997b. EC Measures Concerning Meat and Meat Products (Hormones), Complaint by the United States. Report of the Panel, WT/DS26/R/USA. Geneva: WTO.
WTO (World Trade Organization). 1998a. Australia—Measures Affecting Importation of Salmon. Report of the Appellate Body, WT/DS18/AB/R. Geneva: WTO.
WTO (World Trade Organization). 1998b. Australia--Measures Affecting Importation of Salmon. Report of the Panel, WT/DS18/R. Geneva: WTO.
WTO (World Trade Organization). 1998c. EC Measures Concerning Meat and Meat Products (Hormones). Arbitration under Article 21.3(c) of the Understanding on Rules and Procedures Governing the Settlement of Disputes, WT/DS26/15, WT/DS48/13. Geneva: WTO.
WTO (World Trade Organization). 1998d. EC Measures Concerning Meat and Meat Products (Hormones). Report of the Appellate Body, WT/DS26/AB/R, WT/DS48/AB/R. Geneva: WTO.
WTO (World Trade Organization). 1998e. Japan--Measures Affecting Agriculture Products. Report of the Panel, WT/DS76/R. Geneva: WTO.
WTO (World Trade Organization). 1999. Japan--Measures Affecting Agriculture Products. Report of the Appellate Body, WT/DS76/AB/R. Geneva: WTO.




















































