Scientific Issues of Pain and Distress
G. F. Gebhart
Professor and Head, Department of Pharmacology
University of Iowa
Iowa City, Iowa
I am pleased to have the opportunity to follow Dr. Bayne's presentation because she anticipated many of the concerns and things I want to discuss. My charge was to comment on whether the principles and definitions that were written and presented in the 1992 NRC report Recognition and Alleviation of Pain and Distress in Laboratory Animals are still applicable today and still have merit. I believe they do, and I will discuss issues of pain (including hyperalgesia) and distress. I also will raise the concept of preemptive strategies with respect to anticipation of postoperative pain.
PAIN VERSUS NOCICEPTION
Pain
The widely accepted definition of pain was developed by a taxonomy task force of the International Association for the Study of Pain: “Pain is an unpleasant sensory and emotional experience that is associated with actual or potential tissue damage or described in such terms.” A key feature of this definition is that it goes on to say, “pain is always subjective. ” This aspect of the definition reflects on the issue Dr. Bayne raised when she commented about interpretation of animal behavior and appearance by an observer based on feelings of the observer. We naturally have the tendency, when we observe an animal, to use our own past experiences to interpret and comment on what we perceive or believe to be the animal's status relative to discomfort, pain, or distress. It is very difficult, if not impossible, for our past personal experiences to be meaningfully applied to an
animal. Training and experience in studying and observing animal behavior are required to interpret what we observe in nonhuman animals.
Nociception
The important distinction between pain and nociception must also be made. Nociception is the term introduced almost 100 years ago by the great physiologist Sherrington (1906) to make clear the distinction between detection of a noxious event or a potentially harmful event and the psychological and other responses to it. Sherrington and others before him understood that pain was not a simple sensation, but rather was a complex experience, only a part of which was sensory in nature. Accordingly, it is most accurate to describe what we study as pain in nonhuman animals as nociception. However, although nonhuman animals cannot express in words the psychological and emotional consequences of a noxious stimulus or event, none of us in this audience would hesitate to apply the term “pain” to that circumstance. This fundamental distinction between pain and nociception emphasizes the importance of interpretation of animal behavior by an experienced individual to assess the presence and intensity of pain and distress.
Having said this, it is nevertheless imperative to acknowledge that unless it is established to the contrary, we should assume that those procedures that produce pain in us might also produce pain in animals. This is an entirely appropriate guideline, bearing in mind the caveats that I discussed and the comments Dr. Bayne made just before me.
Noxious Stimuli
I want to consider briefly the nociceptive apparatus (i.e., basic anatomy and physiology). It is important to do this because our concerns about pain in animals are not related to acute pain associated with procedures that are of short duration and/or simple analgesiometric tests, as Dr. Bayne mentioned, such as a tail flick test or a hot plate test. Rather, we are more concerned about the consequences of procedures (surgical procedures included) that may be associated with longer lasting pain that may cause distress.
In Figure 1, a piece of skin that is innervated by a variety of sensory receptors called nociceptors is illustrated. Nociceptors respond only to stimuli that damage tissue or have the potential to damage tissue, and there are several kinds of nociceptors identified in Figure 1. When a nociceptor is activated by a mechanical, thermal, or chemical stimulus, the stimulus energy is transduced by the nociceptor to an electrical event (action potential) and the information is conveyed along nerve axons to the spinal cord, a part of the central nervous system. Input from nociceptors is transferred in the spinal cord dorsal horn to spinal neuron cell bodies whose axons ascend to supraspinal (brain) sites. Nociceptive information is thus distributed to multiple brain sites that give rise to both simple and complex responses to the peripheral noxious event.
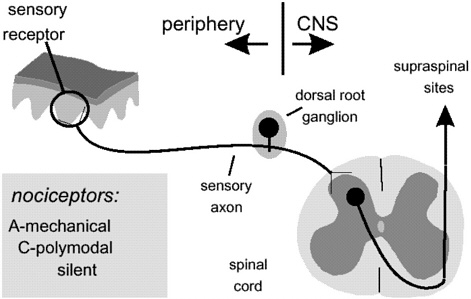
Figure 1 The receptors in the periphery that respond to noxious stimuli, here illustrated for a piece of skin, are termed nociceptors. There are three principal nociceptors: (1) Aδ mechanical nociceptors, which respond to noxious mechanical stimuli that damage or threaten to damage tissue; (2) C-polymodal nociceptors, which respond to noxious mechanical, noxious thermal (>44°C), and noxious chemical stimuli; and (3) silent (or sleeping) nociceptors, which do not respond to acute noxious stimulation of uninjured tissue but become active after tissue is injured. Information from nociceptors is conveyed by sensory axons, whose cell bodies are in the dorsal root ganglion, to the spinal cord where they synapse onto second-order spinal cord neurons, which transmit the information to supraspinal sites (e.g., the thalamus in the brain).
CLASSIFICATIONS OF PAIN
There are many ways to categorize pain. For example, pain can be classified in terms of duration. I think to classify pain in terms of its duration only is inappropriate; that is, to categorize it as acute or short-lasting as opposed to chronic and long-lasting is entirely arbitrary and not particularly helpful.
Protective Pain
I consider the diagram in Figure 2 to be a useful way to consider pain. The diagram suggests that we consider whether pain is “normal” in the sense that it
evokes appropriate protective reflexes and/or behaviors. An example of an acute, protective, nociceptive reflex is withdrawing your finger from a heated surface you touch unexpectedly. If you analyze the event, you will note that you actually removed your finger from the heated surface before you perceived the event as painful. Another more common type of protective pain is that associated with tissue inflammation and tissue repair. One common example is postsurgical pain, which is longer lasting than the pain produced when one touches a heated surface.
Nonprotective Pain
There is also pain that I consider abnormal because it serves no protective value. Examples of these types of pains in humans are those associated with terminal cancer, nerve injury, and after a stroke. Some of the pain syndromes
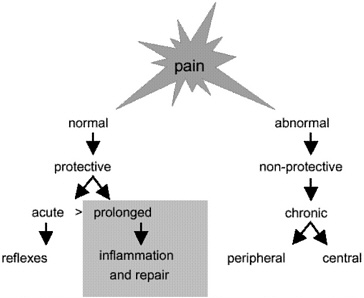
Figure 2 Pain that might be considered “normal” typically serves a protective function. Acute noxious stimulation, such as unexpectedly touching a hot surface or pricking a finger with a needle, evokes a nociceptive withdrawal reflex that, for example, prevents us from further burning our finger. After tissue injury associated with inflammation and repair processes, the injured tissue exhibits enhanced sensitivity to stimulation, termed hyperalgesia (see text and Figure 3). The enhanced sensitivity (e.g., to pressure applied to a sprained ankle or at the site of a surgical incision) is also normal and protective, preventing us from further damaging already injured tissue. Pain that might be considered “abnormal” provides no protective function. Nonprotective pains include those associated with cancers, peripheral and central nervous system damage (e.g., following a stroke), and others often classified as chronic. These pains certainly tell us that something is wrong, but they serve no protective function.
produced are bizarre and very long-lasting; some cannot be controlled and have no protective value to the organism whatsoever.
TISSUE INJURY
Sensitization
What are the consequences of tissue injury that might be associated with a surgical procedure? There is sensitization of the nociceptors illustrated in Figure 1. By sensitization is meant that the nociceptors change their behavior. They become more sensitive to stimuli that are applied to them. This factor contributes to what is termed hyperalgesia, which is discussed below.
There is also an awakening of so-called silent or sleeping nociceptors. Silent nociceptors exist in all of us and invest all of the tissues of our body. These silent nociceptors apparently play no normal physiological role in nociception (or pain), but when tissue is injured or damaged, silent nociceptors become active and begin to contribute information to the nervous system that was never previously contributed.
This activity is one way in which tissue injury or damage helps you protect yourself from further damage. As a consequence of these two events (i.e., nociceptor sensitization and awakening of silent nociceptors), exaggerated sensations can be provoked when you apply a stimulus to or near the site of injury.
Hyperalgesia
In Figure 3, the consequence of tissue injury and the operational definition of hyperalgesia are diagrammed. Plotted vertically on the Y-axis is pain sensation arbitrarily from 0 to 100; and along the horizontal axis is stimulus intensity, ranging from innocuous, nonpainful intensities into noxious or painful intensities. The normal range of pain-produced behaviors is described by the line labeled normal, which represents a normal psychophysical stimulus-response function. If I were to test everyone in the audience with a thermal stimulus or a mechanical stimulus of varying intensity, as the intensity of the stimulus increased, each of us would report increasing pain sensation associated with increasing intensity of stimulation. This stimulus-response function can be shown to exist for individual central nervous system neurons; individual afferent fibers innervating skin, muscle, joints, or viscera; and the behavior of nociceptors. This result applies throughout the realm of biology, if you will, from the single neuron to the integrated response that I would obtain from you when I ask, “Does this hurt? ” and “How much does it hurt?” When tissue is injured or insulted in some way, the normal psychophysical function shifts to the left. The extent to which it shifts is dependent on the magnitude of the insult or the injury. I have illustrated in Figure 3 an example. In the normal individual, the noxious stimulus intensity
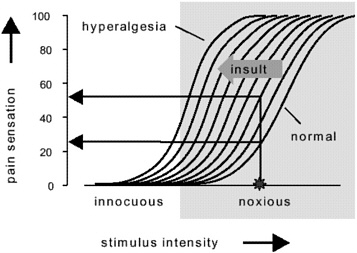
Figure 3 Illustration of how tissue insult affects responses to applied stimuli. Normally, there is a predictable response to stimulation that results in the sensation of pain only when the intensity of stimulation is in the noxious range. The line labeled “normal” represents a typical psychophysical stimulus-response function. In this illustration, a noxious stimulus produces a pain sensation of approximately 25 in uninjured tissue. After tissue insult, that same intensity of stimulation produces significantly greater pain (approximately 55 on the arbitrary vertical scale), which is referred to as hyperalgesia. Hyperalgesia is thus an enhanced sensitivity to noxious stimuli after tissue is injured. Note also that in some cases normally innocuous, nonpainful stimuli can produce pain after tissue injury, a component of hyperalgesia termed allodynia. Allodynia is most common after nerve injury.
identified by the symbol on the horizontal axis produces some amount of pain whereas in the presence of tissue insult or injury, the same intensity of noxious stimulation produces greater pain.
The two types of hyperalgesia are termed primary and secondary and are subserved by different mechanisms. Primary hyperalgesia is associated with a change in the behavior of nociceptors at the site of the injury. Secondary hyperalgesia is associated with a change in the behavior of neurons in the central nervous system. Obviously, the nervous system is not static; it is very dynamic and can be changed by tissue injury and insult. When we injure tissue in the periphery, a host of chemicals contribute to nociceptor sensitization and thus to primary hyperalgesia. These mediators (e.g., prostaglandins, amines, cytokines, kinins, peptides) provide targets for pharmacological intervention to prevent sensitization and thus to prevent the development of hyperalgesia.
When information from nociceptors arrives in the spinal cord, because it is
contributed to by sensitized nociceptors and awakened silent nociceptors and is thus much more intense than normal, it increases the excitability of central neurons. This change in excitability is a consequence of increased release of a variety of other mediators and modulators (e.g., excitatory amino acids and peptides) released from nociceptor terminals in the spinal cord. These mediators contribute to an increase in the excitability of central neurons and to development of secondary hyperalgesia.
STRESS AND DISTRESS
Definition of Terms
The preceding description brings me to a discussion of some of the definitions in Recognition and Alleviation of Pain and Distress in Laboratory Animals (NRC 1992), which Dr. Bayne mentioned in the context of behavior. Stress is defined as the effect produced by external events or internal factors called stressors, which induce an alteration in an animal 's biological equilibrium. I submit to you that getting up and speaking before an audience and not being able to get your computer to work is stressful. Stress is a normal component of life and does not produce distress unless it persists and leads to maladaptive behavior.
Distress is an aversive state in which an animal is unable to adapt completely to stressors and the resulting distress is manifested as a maladaptive behavior. The working definition of distress that Dr. DeHaven presented today, as I heard it, sounded more to me like a definition of stress than a definition of distress. One critical component of any definition of distress, because we cannot objectively measure or quantify distress, is behavior. As Dr. Bayne also indicated, we do not have reliable physiological measures that will tell us always that an animal is or is not stressed or distressed. We have no measures that provide reliable information to us about distress with the exception of the behaviors that we observe in these animals. Importantly, behavior must be assessed by individuals who are trained and knowledgeable about species' typical behavior and who understand what is normal and what is maladaptive.
Reduction with Preemptive Analgesia
One final point I want to make relates to preemptive analgesia. This strategy is based on the knowledge that there will be changes in the behavior of nociceptors and central neurons when tissue is injured. Thus, a preemptive strategy is particularly applicable to surgeries in animals. The strategy involves, for example, administration of opioids before the anesthesia is started and before an incision is made in an attempt to prevent the sensitization of nociceptors. The intent of this strategy is to prevent the development of central hyperexcitability and, as a con-
sequence, reduce postsurgical hyperalgesia and reduce postsurgical stress and potential distress.
CONCLUSION
With that, I will close by saying that the definitions and principles presented in the 1992 NRC publication Recognition and Alleviation of Pain and Distress in Laboratory Animals are still valid and useful. I would comment that the working definition of distress proposed by Dr. DeHaven could be improved by more clearly including behavior, specifically maladaptive behavior, in the definition to clearly distinguish stress from distress.
REFERENCES
NRC [National Research Council]. 1992. Recognition and Alleviation of Pain and Distress in Laboratory Animals . Washington, D.C.: National Academy Press.
Sherrington C.S. 1906. The Integrative Action of the Nervous System. New Haven, Conn.: Yale University Press.
QUESTIONS AND ANSWERS
DR. KARAS (Alicia Karas, Tufts University): In my experience at a veterinary teaching hospital (Tufts University, Boston, MA), analgesics are commonly given to animals before surgery begins, and it does work. Interestingly, that observation was made several years ago at an American Society of Anesthesiology meeting, when the big-breaking media point was preemptive analgesia. One of my technicians expressed surprise when she heard about it on the Today Show because her experiences with preemptive analgesia included having animals recover quietly and with less distress.
DR. GEBHART: Thank you for sharing that experience. I suggest that many people who have tried this strategy and have reported that it does not work have probably underdosed the animals. It is necessary to use a relatively large dose of drug to actually counteraffect the development of sensitization of nociceptors. When you do that, and I am glad your clinical impressions bear this out, you do see improved postsurgical behaviors in these animals. They appear to be less stressed as a consequence; they are up and about sooner, and they seem to recover much better.
DR. DE HAVEN (Ron DeHaven, USDA): In an effort to clarify the working definition that I used, distress is a state in which an animal cannot escape from or adapt to the external or internal stressors or conditions it experiences resulting in negative effects upon its well-being. In my discussion, I did go on to say that there are some things that may cause distress, and those things are boredom, anxiety, fear, pain. So although some stressors may lead to distress, that sequence
was not intended to be part of the working definition. That list simply identified some stressors that can cause distress. My question is whether you were talking about some of those examples or were referring to the working definition that may inappropriately describe the distress and better describe stress.
DR. GEBHART: I was responding principally to the examples that were given as contributing to distress that, to me, contribute to stress. If they are prolonged, obviously, and the animal then cannot adapt to those stressors in the environment and begins to develop maladaptive behaviors, I would agree that every one of those stressors could lead to distress. I would suggest that “negative effects” be more clearly defined in terms of behavior, specifically maladaptive behavior.
DR. BAYNE (Kathryn Bayne, AAALAC): Dr. DeHaven, I believe you said that stress would have a negative impact on animal well-being. How is that negative impact going to be judged? Is the criterion going to be the expression of atypical behaviors or (what Dr. Gebhart and I are suggesting) that there is some evidence of maladaptation on the part of the animal?
DR. DE HAVEN: That is what we are here for, for you to answer that question for us. However, I seriously think there needs to be some evidence of negative effects upon the animal's well-being, and how we assess whether there have been those negative effects becomes critical.
DR. BAYNE: Yes, but I believe some people are going to assume atypical equals lack of well-being, instead of maladaptation reflecting a lack of well-being. Although you are not using either of those terms, the allusion to “negative impact on well-being” requires use of a method to assess the negative impact.
DR. HAYWOOD (J.R. Haywood, University of Texas Health Science Center): I believe we will have to involve stress physiology people to really start working on the development of measurements. However, we will have to address that at a later time. It is very interesting that when we start taking pharmacological approaches, we are going to be affecting all parts of the system and the response system.









