9
Investigating Flow and Transport in the Fractured Vadose Zone Using Environmental Tracers
Fred M. Phillips1
ABSTRACT
Environmental tracers have been applied to a variety of settings that are closely related to fractured vadose zone hydrology, such as structured agricultural soils, unstable wetting fronts, macropores in watersheds, and caves. Results from these tracer applications can give insight into hydrological processes in more traditionally defined fractured vadose zones. The most generally useful tracers appear to be the stable isotopes of oxygen and hydrogen in the water molecule, tritium, halides, and chlorine-36, but there are a wide variety of potentially applicable environmental tracers. Environmental tracers can provide information on the integrated response of actual hydrological systems to real-world boundary conditions over long time scales. Environmental tracers should be considered a primary means of investigating fractured vadose zones in all studies where understanding the integrated system response is an important objective.
INTRODUCTION
In the late 1960s the issue of groundwater contamination, along with other environmental problems, began to receive worldwide attention. One example of this concern was the Chalk aquifer in southern England, a resource of particular national importance inasmuch as it provides approximately 15 percent of the
|
1 |
Department of Earth and Environmental Science, New Mexico Tech, Socorro |
country's water supply (Foster, 1975). Several lines of evidence indicated that this aquifer was particularly vulnerable to pollution. Extensive nitrate contamination had been detected under farmed areas. Bacterial contamination had also been detected, even under areas with thick vadose zones. Also, it was well known that areas underlain by chalk usually generated little or no runoff, even after intense rain. This evidence was interpreted to indicate that pollutants could be rapidly transported to the water table through interconnected channels in the heavily fractured chalk.
The United Kingdom Atomic Energy Authority decided to take advantage of the pulse of elevated tritium then raining out after the recent series of United States and Soviet atmospheric nuclear weapons tests in order to better understand solute transport through the Chalk vadose zone. The results of Smith et al. (1970) were surprisingly at variance with the previous understanding. The measured Chalk vadose zone profiles showed that the tritium peak had penetrated only about 5 m in the past decade (Figure 9-1), indicating slow, relatively uniform infiltration rates between 0.5 and 1 m yr−1. This apparent rate of transport was supported by the results of follow-up studies. Instead of elucidating the contaminant transport problem, the new data only created confusion. How could bacteria be advected to the water table in a matter of hours or days, but tritium take 25 years?
The insight of Foster (1975) solved the dilemma within a few years. Downward transport was almost entirely within the pervasive open fractures. The matrix of the Chalk had very high microporosity, but very low permeability, and hence transport (largely lateral) was mainly by diffusion. The transient pulse of tritium was rapidly diffused into the matrix blocks and retained, while bacteria were too large to enter the matrix pores. The nitrate input was relatively steady and predated that of the tritium by many years, and hence was close to equilibrium with the matrix, permitting unimpeded transport to the water table. This conceptual model was borne out by subsequent studies (Barker and Foster, 1981; Foster and Smith-Carrington, 1980).
The Chalk tritium anomaly was one of the earliest studies in which the importance of matrix diffusion was recognized. It was entirely independent of the better-known discovery of the same phenomenon while assessing fractured, saturated rock as a potential setting for high-level nuclear waste repositories (Neretnieks, 1980). It illustrates well the power of environmental tracers for assessing solute transport in fractured rock, as well as the dangers of inadequate conceptual models causing misinterpretation of the data.
SCOPE
This chapter will focus on the application of environmental tracers to understanding the hydrology of fractured vadose zones. Environmental tracers are defined as solutes that are introduced into the hydrological cycle by either natural
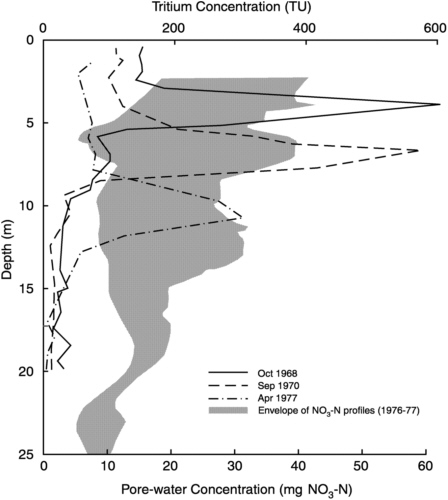
FIGURE 9-1 Profiles of tritium and nitrate concentration with depth, measured at sites in the Chalk of southern England. Tritium is a transient pulse moving downward, whereas nitrate is close to steady state. Data from Foster and Smith-Carrington (1980) and Barker and Foster (1981).
processes or as an inadvertent result of human activities. Deliberately introduced tracers are of great utility for hydrological investigations in both the laboratory and the field, but these are most commonly employed in conjunction with hydraulic testing and are best considered as a component of such tests. There exists a large body of literature on the use of environmental tracers in nonfractured vadose zones, but a comprehensive review of this topic is beyond the scope of
this volume. Two fractured vadose zone sites, Apache Leap and Yucca Mountain, have undergone extensive environmental tracer investigations, but these are treated individually elsewhere in this volume and hence will not be described in detail in this chapter.
COMPARATIVE STUDIES
Environmental tracers have received comparatively little attention in the hydrological literature as a means of understanding flow and transport in fractured rock. For example, the recent National Research Council report Rock Fractures and Fluid Flow: Contemporary Understanding and Applications (National Research Council, 1996) contained within 500 pages of text only 3 pages dealing with environmental tracers, and these described the results of a single case study. Environmental tracers were not mentioned as a possible avenue for better understanding of fractured-rock hydrology. In contrast, such tracers have been extensively used for related studies in other disciplines. It is worthwhile to examine what techniques or findings can be transferred to the fractured vadose-zone problem.
Structured Agricultural Soils
Farmers have been conducting environmental tracer tests in the vadose zone on a grand scale for many years. A variety of agricultural chemicals of environmental concern are routinely applied to fields and then allowed to infiltrate, under either irrigation or natural precipitation. These chemicals have commonly been observed to migrate to the groundwater at rates much faster than would be predicted by uniform one-dimensional flow (Jury and Flühler, 1992). Numerous field tracer investigations (Bowman and Rice, 1986; Flury et al., 1994; Jaques et al., 1998; Johnston et al., 1998; Jørgensen et al., 1998) have revealed that this fast transport can often be attributed to preferential flow along high-permeability pathways, commonly referred to as “structured soils” in agriculture.
Agricultural soils are typically at the low end of the scale in terms of permeability contrast between fast pathways and matrix. In most studies there has been little attempt to sample the fast pathways directly; instead, high-spatial-resolution sampling of the subsoil is employed and inferences about pathways are drawn based on the measured breakthrough curves. In some cases identification of the fast pathways may be attempted by adding dyes and excavating after the experiment is complete.
One significant advantage of the agricultural soil experiments is that the shallow depths and loose nature of the soil permit much more detailed sampling than is usually possible for fractured consolidated rock. The relatively low permeability contrasts and high matrix permeability permit detection of preferential flow by sampling the matrix, rather than needing to directly sample fracture flow,
which is much more difficult. One important result of the agricultural soil studies is that, when sampled at this kind of resolution, nearly all soils show significant indications of preferential flow. Easily visible macropores or other fast paths are not required to produce preferential flow. The breakthrough curves observed in these tracer studies are typically highly skewed with very long tails (Figure 9-2). Continuum models usually adequately reproduce these curves (e.g., Bowman and Rice, 1986; Jaques et al., 1998).
Unstable Wetting Fronts
When large amounts of water infiltrate into dry soils, unstable wetting fronts may develop if the soil has water-repellent surfaces or if the surficial layer is coarse (Glass et al., 1989; Hendrickx et al., 1993). The wetting front changes from a relatively uniform, planar shape to one with elongated fingers that drain the high water-content layer at the soil surface. The flow through such fingers is somewhat analogous to fracture flow. The fingers may, under some circumstances, be persistent, in the sense that during repeated infiltration and drying cycles the
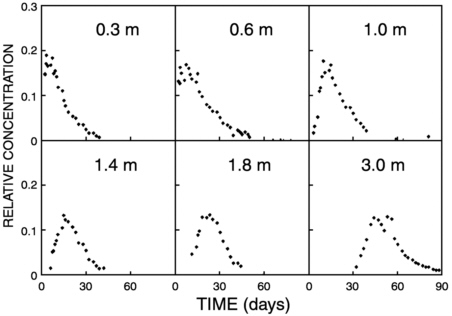
FIGURE 9-2 Breakthrough curves of normalized bromide concentration as a function of time, for different depths, under a semiweekly irrigation regime. Data are from Bowman and Rice (1986). The strongly skewed and tailing breakthrough curves are characteristic of solute transport in strongly structured soils.
same finger paths will be reoccupied (Glass et al., 1989; Ritsema et al., 1998). In real soils there is undoubtedly a continuum between true unstable wetting front propagation (which can be observed even in a completely homogeneous soil) and preferential flow, which requires a high-permeability pathway.
Most studies of unstable wetting fronts in soils have measured water content rather than employing tracers because the contrast between the low initial water content of the soils (which is required for the fingers to form) and the high water content after the fingers have penetrated makes this measurement simple, as does the ease of excavation of the soil. However, anionic tracers have been applied to a limited extent to verify solute transport predictions in the presence of unstable wetting (Van Dam et al., 1990). The extent to which wetting fronts will become unstable in consolidated rocks is at present uncertain, but it does seem likely that under conditions of high water application rates to rock surfaces, preferential paths will develop and may tend to be reoccupied during subsequent wettings. Given the difficulty of excavating consolidated rock and of measuring water content within such rock, tracers may provide an attractive alternative for evaluating related flow effects in more typical fractured rocks.
Watershed Hydrology
Environmental tracers have provided critical evidence for the importance of fracture-analogous flow in the generation of runoff from watersheds. At one time the sudden response in streamflow that closely follows precipitation events was thought to result largely from overland flow of the rainfall. Studies employing δ18O and δ2H in the water molecule, and chloride, demonstrated that in many cases a large fraction of the storm runoff was not from the current precipitation event, but was in fact water that had been stored in the subsurface system for some period of time (McDonnell et al., 1991; Rodhe, 1981; Sklash, 1990; Sklash et al., 1976). Rapid infiltration of precipitation through macropores causes a pressure response of the water table that results in an increase in stream discharge through groundwater discharge into the streambed. This type of lumped system response is surely a close analog to the behavior of many fractured vadose zone/aquifer systems.
Numerous watershed studies have used environmental tracers to directly study preferential flow phenomena (Hammermeister, 1982; Mulholland et al., 1990; Leany et al., 1993). One of the most striking examples of flow partitioning between matrix and fast paths has been provided by Newman et al. (1998). In this study of a hillslope in a ponderosa pine forest in New Mexico, flow interceptors were emplaced on top of the B horizon (dominated by clay) and the C horizon (bedrock), and the interflow generated from these horizons was compared with matrix water sampled by coring. The matrix water in the B horizon had chloride concentrations in the range of 200 to >300 mg L−1, while water collected in the interceptors during the same period had concentrations ranging from 2 to 30 mg
L−1. The very high matrix chloride concentrations resulted from transpiration of matrix water during prolonged periods of low precipitation. The dramatic contrast in concentrations indicates that the macropores were acting almost independently of the matrix. However, after high moisture contents were maintained for more than a week, the chloride concentration of the macropore flow began to rise as solute exchange between the pores and matrix became appreciable (Figure 9-3). This response nicely illustrates the highly transient nature of the matrix/macropore interaction that also characterizes more typical fractured rock in the vadose zone.
Cave Hydrology
It is the desire of every vadose fractured rock hydrologist to have a tunnel drilled beneath the research site in order to actually see and sample in detail the
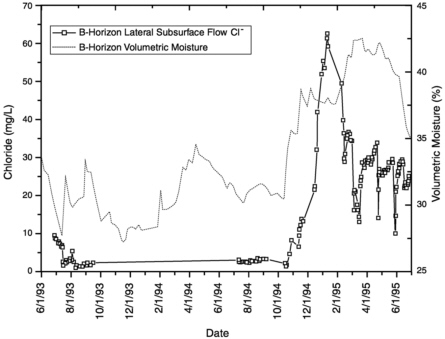
FIGURE 9-3 Changes in soil moisture content and chloride concentration of lateral subsurface flow from the B horizon with time in a semiarid hillslope in New Mexico, from Newman, B. D., A. R. Campbell, and B. P. Wilcox, 1998. Lateral subsurface flow pathways in a semiarid ponderosa pine hillslope. Water Resources Research 34: 3485-3496. Copyright by American Geophysical Union. The increase in chloride concentration beginning in November 1994 illustrates increased exchange between macropores and matrix as moisture content rises.
fractured vadose zone. This has been accomplished (generally at great expense) at a few sites, notably Yucca Mountain. However, an extensive natural network of such tunnels already exists, and their hydrology has been investigated for many years. They are called caves.
Sampling drips into caves provides a direct means of accessing flow passing through fractured and solutioned vadose zones (Even et al., 1986; Harmon, 1979; Ingraham et al., 1990; Yonge et al., 1985). Much effort has been directed toward tracing the transit time from the land surface to the cave depth. These times vary greatly: two weeks for a cave in Kentucky (Harmon, 1979), several months for caves in England (Atkinson et al., 1985), decades at Carlsbad Caverns, New Mexico (Chapman et al., 1992). Both stable isotopes and tritium have proved useful tracers in these systems. Some cave drips show the isotopic signature of nearly every precipitation event, others show no temporal variation at all, indicating either very long flow paths along which infiltrating waters are homogenized by mixing, or large water volumes stored in the matrix (Chapman et al., 1992; Yonge et al., 1985). Transit times and compositions can vary markedly between fractures separated by only a short distance (Even et al., 1986).
Cave seeps represent an underutilized resource for understanding the hydrology of fractured vadose zones. To date, results of these studies have been treated largely as aids to the paleoenvironmental interpretation of cave precipitates, and in relation to the hydrology of particular localities. A more comprehensive and systematic investigation of environmental tracers in cave seeps could add greatly to our understanding of flow and transport in fractured vadose zones.
SUITABLE TRACERS
A very wide variety of environmental tracers have been applied to hydrological problems. Some of these are particularly suitable for work in the fractured vadose zone and others are not. The following tracers appear to be among the most suitable.
Stable Isotopes of Oxygen and Hydrogen in the Water Molecule
Deuterium (2H) and oxygen-18 (18O) are stable isotopes that are incorporated into the water molecule itself. They can thus be considered in some senses to be the “perfect tracer” for water movement. In natural, near-surface hydrological systems these heavy isotopes are fractionated relative to the more abundant light forms of their elements mainly by two influences: evaporation and variations in the isotopic composition of precipitation. Evaporation enriches the heavy isotopes. Fractionation in precipitation depends mainly on temperature (Dansgaard, 1964), and thus carries a seasonal signature. Precipitation falling in the summer tends to be isotopically heavy, that falling in the winter light. These fractionations can be applied to various types of tracing, such as determining
transit times from the surface to a point at depth by comparing time trends of composition with the seasonal precipitation cycle (Saxena, 1984), or distinguishing long-residence matrix water from recent fracture water by the heavy evaporation signature in the former. For example, Thoma et al. (1979) were able to discern the annual cycle in δ2H of recharge through a sand dune in Bordeaux down to 25 m depth. In addition, because temperature decreases with increasing elevation, the lighter isotopic composition of higher-altitude precipitation can be used to help determine the source of recharge (Scholl et al., 1996). The application of stable isotopes to hydrological problems has been reviewed by IAEA (1981) and Coplen et al. (1999).
Tritium
Tritium (3H) is an unstable isotope of hydrogen with a half-life of 12.45 years. It is naturally produced at fairly low levels in the atmosphere by cosmic radiation. A very large amount of tritium, illustrated in Figure 9-4, was also
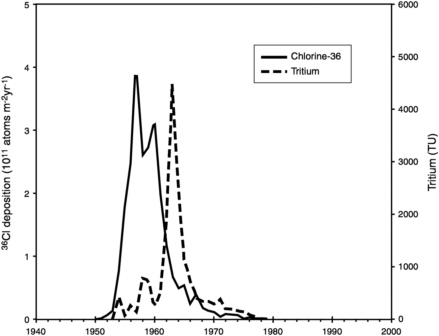
FIGURE 9-4 Deposition of 36Cl and 3H in the Arctic region from nuclear weapons testing. Tritium fallout (not decay-corrected) was reconstructed by Östlund (1982), and 36Cl was measured in Greenland ice by Synal et al. (1990).
released into the atmosphere during the atmospheric thermonuclear weapons testing period (1954-1964). Radioactive decay, rainout, and dilution have by now reduced this pulse down nearly to prebomb levels. During the atmospheric testing period, and for about two decades subsequent to it, this pulse served as a very useful transient tracer (IAEA, 1983c). At present, however, radioactive decay and mixing in the subsurface have reduced the tritium levels of even peak-fallout-period infiltration to the point where it is often difficult to distinguish such water from more recent infiltration. This ambiguity will only increase as the bomb pulse decays.
In saturated zone settings this problem can be remedied by measuring the concentrations of both 3H and its decay product, 3He (Cook and Solomon, 1997). The tritium content at the time of recharge can be inferred from the added concentrations of parent and daughter, and the time since recharge from the ratio of the two. This technique is not generally applicable to fractured vadose zones, however, since the 3He can readily escape by volatilization. As a result, the utility of tritium as a vadose zone tracer is likely to decline in the future.
Tritium is presently most useful in vadose zones where residence times are long. In such settings, relatively high tritium concentrations may be indicative of fast flow paths (e.g., fractures), whereas low tritium indicates matrix water. In this case it may not be necessary to distinguish the bomb-pulse tritium; the knowledge that measurable tritium corresponds to residence times shorter than 50 years and at- or below-detection tritium to longer residence times may be sufficient. In short-residence-time vadose zones (shallow ones or those with uniformly high permeability and large recharge rates), the tritium content is likely to be spatially uniform and yield little information on flow paths.
Halides
Chloride, bromide, and iodide share similar geochemical characteristics in hydrologic systems. In the reduced, anionic form (i.e., Cl−, Br−, and I−) they do not tend to form insoluble minerals and are repelled from negatively charged mineral surfaces, and hence act conservatively in groundwater and soil water. They tend to be left behind when water is removed by evaporation or transpiration, and thus increases in concentration can help to indicate the proportion of water that has infiltrated relative to that lost to the atmosphere (Allison et al., 1994). This evapotranspirative concentration can thus also “tag” long-residence-time matrix water, compared to freshly infiltrated fracture or macropore water (Newman et al., 1998; Scanlon, 1992).
Cl−/Br− ratios can serve to distinguish atmospheric salts carried by surface infiltration from subsurface salts (Davis et al., 1998). This information can be of importance when the fractured rock matrix consists of high-chloride lithologies such as rhyolite or basalt. Shifts in the Cl/Br ratio of porewater may then provide another qualitative indicator of flow path and residence time. In addition, Cl/Br
ratios may be useful to infer the nature of the infiltration process. Because bromide is somewhat less conservative than chloride due to a greater propensity for reaction with plants and other organic matter, the Cl/Br ratio of infiltrating water tends to increase with residence time in the soil zone (Davis et al., 1998). Iodine is even less conservative than bromine because of its stronger affinity for organic matter.
Chlorine-36
Chlorine-36 is a radioactive isotope of Cl with a half-life of 301 ka (Bentley et al., 1986). It is produced at relatively high levels in the atmosphere by cosmic ray reactions on Ar, and in the subsurface at much lower levels from reaction of stable Cl with neutrons derived from U and Th decay. The atmospheric deposition of 36Cl has fluctuated at several time scales. Variations at the ~104 year scale have been observed by Plummer et al. (1997) and Baumgartner et al. (1998). A major pulse of 36Cl was released during the late 1950s by atmospheric nuclear weapons testing (Elmore et al., 1982; Zerle et al., 1997), shown in Figure 9-4. The systematics of 36Cl applications to hydrology have recently been reviewed by Phillips (1999).
The most straightforward application of 36Cl to fractured-rock hydrology is mapping the depth of penetration of the bomb-pulse 36Cl. Due to decay of the bomb-pulse 3H, this radionuclide is now in many cases the most easily distinguishable signal in the vadose zone (Bentley et al., 1982). The most notable use of this tracer thus far in fractured vadose zones has been at Yucca Mountain (Fabryka-Martin et al., 1997). As with tritium, 36Cl is most applicable to long-residence-time vadose zones. Cook and Walker (1996) have noted that in arid soils where the bomb-pulse 36Cl has not penetrated below the root zone, its depth cannot be used to infer the groundwater recharge rate. Nevertheless, this information on depth of penetration may be very important for assessing potential contaminant transport.
Other Tracers
Many tracers that have proved very useful in saturated-zone studies over short time scales are not generally applicable to studying the liquid phase of vadose-zone problems because they are too volatile. These include 85Kr and 3He in the 3H/3He pair (Cook and Solomon, 1997) and chlorofluorocarbons, refrigerant compounds that have built up in the atmosphere over the past four decades (Busenberg and Plummer, 1991). These volatile tracers may either be lost from vadose-zone water, or diffuse into the water, through the gas phase in the vadose zone. Carbon-14 is another common hydrological tracer that is too volatile for vadose-zone applications. [Note that, although these volatile tracers are not generally applicable to tracing liquid water movement in the vadose zone, they may be useful for studying gas-phase behavior (Thorstenson et al., 1998)].
A wide range of chemical compounds may form suitable tracers, depending on circumstances. Anionic chemicals are particularly useful because they usually do not interact strongly with the solid phase. Nitrate has been frequently applied in this way because it is a common agricultural contaminant (Barker and Foster, 1981; Johnston et al., 1998). The stable isotopes of many elements can also be used as tracers. Among the most commonly employed are isotopes of carbon (Bar-Matthews et al., 1996), sulfur (IAEA, 1983a), nitrogen (Böhlke and Denver, 1995), and boron (Leenhouts et al., 1998). Strontium isotopes have been applied to reconstructing ancient flow regimes through the fractured vadose zone at Yucca Mountain (Johnson and DePaolo, 1994). Although the principles are well established, the application of these isotopic techniques to problems of fractured vadose zone hydrology has been very limited.
LIMITATIONS AND ADVANTAGES
Environmental tracers are particularly suitable for answering some kinds of questions, but not others. These are briefly summarized below.
Limitations
-
Investigating perturbed flow systems. Environmental tracers typically yield information on flow systems under fluctuating natural boundary conditions. They are thus not optimal for investigating situations where the circumstances vary significantly from the natural system. For example, environmental tracers would probably give little information about the infiltration of gasoline beneath a leaking storage tank. Similarly, studies of environmental tracers beneath an undisturbed fractured desert vadose zone would be of minimal value in predicting the migration of herbicides beneath a golf course established in the same area.
-
Detailed mechanistic studies. Many studies are directed toward establishing a fundamental mechanistic understanding of flow and transport (e.g., relation between fracture roughness and flow rate, or fracture coatings and matrix imbibition). One motivation for this type of study is that numerical models are frequently constructed using such mechanistic equations as the building blocks. Environmental tracers are generally of limited value for such investigations because the tracer signals are the result of the combined influence of a wide range of environmental variables that include the mechanistic flow and transport effects, but that are integrated over a range of spatial scales and also include effects of fluctuations in the environmental boundary conditions. For this type of goal, either laboratory experiments or field experiments in which a portion of the system can be isolated and stressed in a controlled fashion are generally preferable alternatives.
-
Natural limitations. In some circumstances, suitable environmental tracer signals may not be present at detectable levels in the system, or may be too subtle
-
to be of much use. In other cases, the difficulty of obtaining high quality samples for environmental tracers in fractured vadose zones may make the cost prohibitive. In these situations, artificial tracer experiments will often be the best alternative.
Advantages
-
Integration of natural processes. In many cases the desired goal is to predict flow and transport through fractured vadose zones under conditions approximating the natural situation. This prediction is usually attempted using numerical models that have been developed and calibrated using a limited number of short-term observations and artificial hydraulic and tracer tests. In this circumstance, the extent to which a model assembled from such components can successfully predict the behavior of an integrated system under fluctuating natural boundary conditions is frequently uncertain. Testing against environmental isotope data that represent an integrated response of the system can serve to greatly increase confidence in the modeling results.
-
Long temporal or spatial scales. Predictions over long time scales are frequently desired, particularly for waste disposal problems. The validity of extrapolating model simulations far into the future is often questioned. Environmental tracers can often provide information on solute transport over time scales that are much longer than would ever be feasible for artificial experiments. Analogously, tracers can provide direct information on transport times through large hydrological systems where obtaining an adequate database of model parameter values is difficult.
-
Minimal disturbance of natural systems. In some cases, extensive drilling and hydraulic and tracer testing cannot be performed, either to prevent compromising sites that are supposed to be “leak tight,” or to avoid damaging environmentally sensitive areas. In such cases it may still be possible to obtain samples from natural sources that can be analyzed for environmental tracers.
The bottom line is that environmental tracers can provide a form of “ground truth” with regard to the natural behavior of complex fractured flow systems that is not obtainable in any other way. Unless the system is under conditions that are far from natural, or no useful environmental tracer can be identified, environmental tracers should always be considered a primary investigation technique.
EXAMPLES
Flow Beneath Mont-Blanc
Determining the pattern of flow within a large mountain is a formidable task, and it is difficult to imagine how it could be accomplished using only conventional physical hydrology methods. However, Fontes et al. (1978) were able to
obtain a good understanding of the flow regime beneath Mount-Blanc in the French/Italian Alps merely by sampling water outflows into the 12-km-long tunnel beneath the mountain and analyzing them for environmental tracers. Part of their results is shown in Figure 9-5. The close correspondence between the profile of δ18O and the topographic profile of the mountain above the tunnel is good evidence for nearly vertical downward flow (the lighter isotope values under the peaks reflect colder condensation temperatures with higher altitude). The data preclude arguments that permanent freezing conditions at the highest altitudes do not permit recharge. The anomalous interval of heavy δ18O between
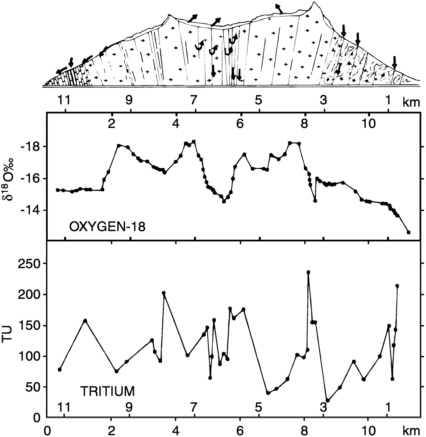
FIGURE 9-5 Variation of δ18O and tritium with distance along the Mont-Blanc tunnel, from Fontes et al. (1978). Note the correspondence between overlying topography and δ18O values, but lack of correlation with tritium. Reproduced with permission from the International Atomic Energy Agency.
5 and 6 km was shown to be due to evaporation of the water during transit through the fracture system, demonstrating that the fractures are not in a fully saturated state. The variable, but frequently high tritium values indicate that there is little interconnection between fractures (and that they do not draw from any common reservoir), and that while some fractures are transmitting infiltration to the tunnel level in a matter of months, others take more than a decade. These conclusions are similar to those obtained by Rauert and Stichler (1974) from a tracer investigation of the 7-km-long Tuxer Hauptkamm tunnel in Austria, except that the tritium concentrations were very low throughout the central section of that tunnel, indicating uniformly slower rates of downward flow.
Hydraulics of Fractured Tuff in New Mexico
Los Alamos National Laboratory recently drilled several boreholes through the Bandelier Tuff at the laboratory site. The fractured rhyolite tuff caps long, fingerlike mesas that project eastward from the flanks of the Jemez Mountains. Deep canyons bound the mesas, and the water table is over 100 m below the land surface. Vertical profiles of stable-isotope composition and chloride concentration were measured with depth and are illustrated in Figure 9-6. The strongly enriched δ18O and very high chloride concentrations in the top 30 m are striking. They are accompanied by very low water contents in the rhyolite matrix. Newman and Birdsell (1997) attributed this profile to geothermally induced convection of air through the mesas. Air moves inward through outcrops of highly permeable basal-surge pumice deposits that are exposed on the canyon walls. Geothermal heating in the centers of the mesas causes the air to rise through fractures that pervade the low-permeability tuff, carrying water vapor with it. Oxygen-18 and chloride are concentrated in the residual pore water. Geothermally stimulated convection of air is undoubtedly a common phenomenon in fractured vadose zones (Weeks, 1987), and this example illustrates how environmental tracers can identify circumstances in which it exerts a profound influence on vadose-zone hydraulics and porewater chemistry.
Tension Fractures in Desert Vadose Zones
In general, the hydraulics of vadose zones of the American Southwest do not appear to be strongly affected by the presence of vertical fractures or other vertical macropores. One exception to this is narrow vertical fractures, sometimes extending as much as several hundred meters deep and many kilometers in length, that spontaneously appear in piedmont deposits or basin fills. In some cases these fissures seem to be a response to groundwater pumping, but in others the cause is not apparent (Larson and Péwé, 1986). Although these fissures are initially only a few millimeters wide, diversion of surface flows can erode the upper portions into chasms over 10 m in both width and depth.
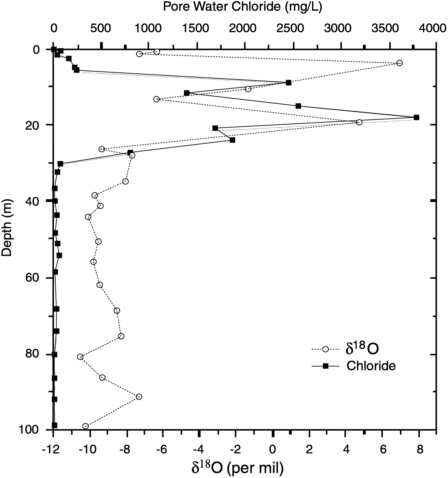
FIGURE 9-6 Variation of δ18O and chloride concentration with depth in fractured rhyolite tuff at Los Alamos, New Mexico (Newman and Birdsell, 1997; B. Newman, Los Alamos National Laboratory, personal communication, 1999). Enriched portion of profile above 30 m overlies a pyroclastic unit that allows air circulation through the fractured vadose zone.
Dramatic erosion of the fissure tops is evidence that large amounts of water have been diverted from ephemeral surface drainages or overland flow to depth. How does the drainage of water into the fissure affect the subsurface hydrology? Scanlon (1992) and Scanlon et al. (1997) have addressed this issue by measurement of chloride, 36Cl, and stable isotopes of oxygen and hydrogen (as well as physical hydrologic parameters) in the vicinity of such fissures. Results are illustrated in Figure 9-7 and Figure 9-8. Variations in water content were small and difficult
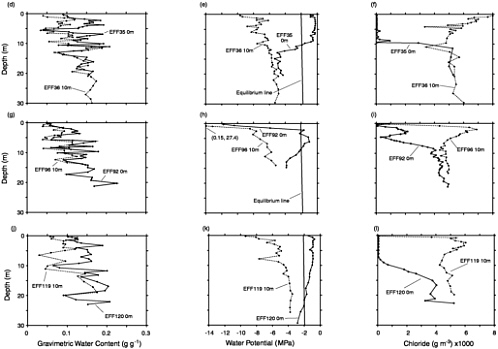
FIGURE 9-7 Profiles of water content, soil-water potential, and chloride concentration in boreholes adjacent to an earth fissure at Eagle Flat, Texas. From Journal of Hydrology, 203, Scanlon, B. R., R. S. Goldsmith, and J. G. Paine, Analysis of focused unsaturated flow beneath fissures in the Chihuahuan Desert, Texas, USA. Pp. 58-78, 1997, with permission from Elsevier Science. Lengths indicated on the figures (“0 m” and “10 m”) refer to distances of the boreholes from the fissure. All boreholes immediately adjacent to the fissure (0 m) show low chloride concentrations and less negative soil-water potentials than at 10 m, due to interception of runoff by the fissure.
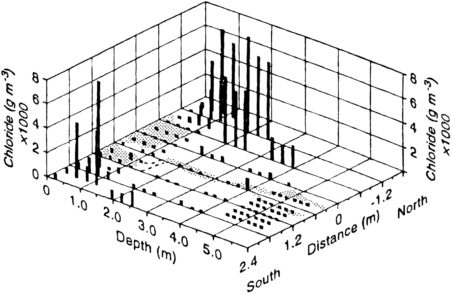
FIGURE 9-8 Plot of chloride concentration as a function of depth and distance away from an earth fissure at Eagle Flat, Texas. From Journal of Hydrology, 203, Scanlon, B. R., R. S. Goldsmith, and J. G. Paine, Analysis of focused unsaturated flow beneath fissures in the Chihuahuan Desert, Texas, USA. Pp. 58-78, 1997, with permission from Elsevier Science. Low chloride concentrations adjacent to the fissure delineate the zone of leaching by runoff captured by the fissure. The configuration of the fissure is indicated by the stippled pattern.
to interpret. Chloride concentration measurements, however, showed a pronounced leached zone surrounding the fissures. The zone was typically 5 to 10 m in width and extended downward 10 to 20 m. This pattern shows that there is a strong tendency for imbibition of the fracture water into the soil matrix. The rate of imbibition is apparently rapid enough that most fracture flow is depleted within 20 m of the surface, a significant finding with respect to contaminant transport and aquifer recharge.
Flow Through Unsaturated Fractured Rhyolite at Yucca Mountain, Nevada
Yucca Mountain is at present the nation's only site under investigation for its suitability as a potential high-level nuclear waste repository. An 8-km-long U-shaped tunnel called the Exploratory Studies Facility (ESF) has been drilled beneath the mountain to provide access to the interior of the mountain for hydrogeologic and engineering research. The thickness of rhyolite tuff over the
tunnel is approximately 300 m, although it is thinner close to the portals. The rhyolite consists of interbedded dense welded tuff and less welded pyroclastic deposits. Fractures in the welded units were recognized as potential fast paths for flow from the surface. The more porous and less cohesive pyroclastic strata were anticipated to provide a capillary barrier to fast-path flow.
Such hypotheses are very difficult to test at the mountain scale using conventional methods. Measurement of dissolved 36Cl, however, can provide a tracer for infiltration on time scales ranging from decades to millennia. Rock samples were collected during drilling of the tunnel (no liquid water seeps were found). Sample points were located both by systematic collection at 200-m intervals and by “feature-based” sampling of observed faults and fractures (Fabryka-Martin et al., 1997, 1998). Many samples showed 36Cl/Cl ratios that were elevated above the contemporary value of ~500 × 10−15 (Figure 9-9). Most of these, however, were below the limit of ~1,200 × 10−15 that has been demonstrated by measurements on
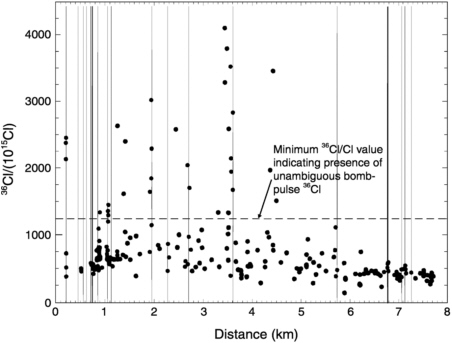
FIGURE 9-9 Distribution of 36Cl/Cl ratios as a function of distance along the Exploratory Studies Facility tunnel beneath Yucca Mountain (Fabryka-Martin et al., 1998. Copyright 1998 by the American Nuclear Society, La Grange Park, Illinois.). Vertical lines illustrate the position of faults within the tunnel. The current meteoric 36Cl/Cl ratio is ~500 × 10−15. Values higher than 1,200 × 10−15 indicate presence of bomb-pulse 36Cl that infiltrated from the surface in the past 50 years.
fossil rat urine to be the atmospheric deposition value during the late Pleistocene (Plummer et al., 1997). All of the systematic samples were below 1,200 × 10−15. Nine locations, however, contained ratios above 1,200 × 10−15, and as high as 4,000 × 10−15, that are most probably ascribed to infiltration of bomb-fallout 36Cl. All except two of these were associated with known faults.
These results indicate a remarkable variability in vertical transport rates. Infiltration of the 36Cl in the samples with ratios between 500 × 10−15 and 1,200 × 10−15 was probably prior to ~15,000 years ago. Infiltration of the 36Cl in the samples with ratios greater than 1,200 × 10−15 was probably less than 50 years ago. The fast paths are almost uniformly associated with faults, indicating that the displacements have ruptured the less-welded units and greatly increased their fracture connectivity and vertical permeability. The presence of presumably Pleistocene-age 36Cl in fractured, but not faulted, sections of the ESF indicates that the less-welded units greatly retard downward fracture transport where the pyroclastics are not faulted.
This environmental tracer study has yielded critically important information on the hydrology of this fractured vadose zone. It illustrates well the ability of environmental tracers to provide information at a wide range of time scales, under fluctuating natural boundary conditions. The results should be of great utility in either evaluating or designing a high-level nuclear waste repository at Yucca Mountain.
SUMMARY
Environmental tracers have been applied for many years to a variety of hydrologically related flow-and-transport problems. Many of these are variants on the fractured-vadose setting, including structured agricultural soils, unstable wetting fronts, macropores in watersheds, and caves. Actual applications to classical fractured vadose zones have not been numerous in the published literature, but this situation is more likely to reflect a general under-investigation of this environment rather than a lack of suitable tracers. The most generally applicable tracers would appear to be the stable isotopes of O and H in the water molecule, tritium, halides, and 36Cl.
The major advantage of environmental tracers is that they provide information on the integrated response of actual hydrological systems to real-world boundary conditions, over long time scales. They are thus particularly useful for problems where long-time or large-scale responses are important. They are also useful for formulating conceptual models of fractured vadose zones, calibrating models, and building confidence in them. They are less useful for detailed mechanistic investigations aimed at understanding the response of small portions of the fractured vadose system to carefully controlled stresses.
Environmental tracers should be considered a primary means of investigating fractured vadose zones in all studies where understanding the integrated
system response is an important objective. Fractured vadose zones are complex hydrological environments where understanding and predicting flow and transport is inherently a very difficult task. Environmental tracers must be incorporated along with mechanistic studies and numerical modeling in order to produce results that can be applied and interpreted with confidence.
REFERENCES
Allison, G. B., G. W. Gee, and S. W. Tyler, 1994. Vadose-zone techniques for estimating groundwater recharge in add and semiarid regions. Soil Sci. Soc. Am. J. 58: 6-14.
Atkinson, T. C., J. W. Hess, and R. S. Harmon, 1985. Stable isotope variations in recharge to a karst aquifer, Yorkshire Dales, England: Preliminary work. Ann. Soc. Géol. Belg 108: 225.
Bar-Matthews, M., A. Ayalon, A. Matthews, E. Sass, and L. Halicz, 1996. Carbon and oxygen isotope study of the active carbonate-water system in a karstic Mediterranean cave: Implications for paleoclimate research in semiarid regions. Geochim. Cosmochim. Acta 60: 337-347.
Barker, J. A., and S. S. D. Foster, 1981. A diffusion-exchange model for solute movement in fissured porous rock. Quart. J. Eng. Geol. (London) 1: 17-24.
Baumgartner, S., J. Beer, J. Masarik, G. Wagner, L. Meynadier, and H.-A. Synal, 1998. Geomagnetic modulation of the 36Cl flux in the GRIP ice core, Greenland. Science 279: 1330-1332.
Bentley, H. W., F. M. Phillips, S. N. Davis, S. Gifford, D. Elmore, L. E. Tubbs, H. E. Gove, 1982. Thermonuclear 36Cl pulse in natural water. Nature 300: 737-740.
Bentley, H. W., F. M. Phillips, and S. N. Davis, 1986. Chlorine-36 in the terrestrial environment. In: Handbook of Environmental Isotope Geochemistry, Vol. 2B (P. Fritz and J.-C. Fontes, eds.). Elsevier, Amsterdam, pp. 427-480.
Böhlke, J. K., and J. M. Denver, 1995. Combined use of groundwater dating, chemical, and isotopic analyses to resolve the history and fate of nitrate contamination in two agricultural watersheds, Atlantic coastal plain, Maryland. Water Resour. Res. 31: 2319-2340.
Bowman, R. S. and R. C. Rice, 1986. Transport of conservative tracers in the field under intermittent flood irrigation: Water Resour. Res. 22: 1531-1536.
Busenberg, E., and L. N. Plummer, 1991. Chlorofluorocarbons (CCl3F and CCl2F2): Use as an age dating tool and hydrologic tracer in shallow ground-water systems. U.S. Geol. Surv., Water Resour. Invest. Rept. 91-4034.
Chapman, J. B., N. L. Ingraham, and J. W. Hess, 1992. Isotopic investigation of infiltration and unsaturated flow processes at Carlsbad Cavern, New Mexico. J. Hydrol. 133: 343-363.
Cook, P. G., and D. K. Solomon, 1997. Recent advances in dating young groundwater: Chlorofluorocarbons, 3H/3He and 85Kr. J. Hydrol. 191: 245-265.
Cook, P. G., and G. Walker, 1996. Evaluation of the use of 3H and 36Cl to estimate groundwater recharge in arid and semi-arid environments . Symposium on Isotopes in Water Resources Management, Vienna, International Atomic Energy Agency, pp. 397-403.
Coplen, T. B., A. L. Herczeg, and C. Barnes, 1999. Isotope engineering: Using stable isotopes of the water molecule to solve practical problems. In: Environmental Tracers in Subsurface Hydrology (P. Cook and A. L. Herczeg, eds.). Kluwer Academic, Boston, pp. 79-110.
Dansgaard, W., 1964. Stable isotopes in precipitation. Tellus 16: 436-468.
Davis, S. N., D. O. Whittemore, and J. Fabryka-Martin, 1998. Uses of chloride/bromide ratios in studies of potable water. Ground Water 36: 338-350.
Elmore, D., L. E. Tubbs, D. Newman, X. Z. Ma, R. Finkel, K. Nishiizumi, J. Beer, H. Oeschger, and M. Andree, 1982. The 36Cl bomb pulse measured in a shallow ice core from Dye 3, Greenland . Nature 300: 735-737.
Even, H., I. Carmi, M. Magaritz, and R. Gerson, 1986. Timing the transport of water through the upper vadose zone in a karstic system above a cave in Israel. Earth Surf. Processes Landforms 11: 181-191.
Fabryka-Martin, J. T., A. V. Wolfsberg, P. R. Dixon, S. S. Levy, J. A. Musgrave, and H. J. Turin, 1997. Summary report of chlorine-36 studies: Sampling, analysis, and simulation of chlorine-36 in the Exploratory Studies Facility. Los Alamos National Laboratory, LA-13352-MS, Los Alamos.
Fabryka-Martin J. T., A. V. Wolfsberg, S. S. Levy, J. L. Roach, S. T. Winters, L. E. Wolfsberg, D. Elmore and P. Sharma, 1998. Distribution of fast hydrologic paths in the unsaturated zone at Yucca Mountain. 8th Annual International High-Level Radioactive Waste Management Conference, Las Vegas, Nevada. American Nuclear Society, La Grange Park, Illinois, pp. 93-96.
Flury, M., H. Flühler, W. A. Jury, and J. Leuenberger, 1994. Susceptibility of soils to preferential flow of water: A field study . Water Resour. Res. 30: 1945-1954.
Fontes, J.-C., G. C. Bortolami, and G. M. Zuppi, 1978. Hydrologie isotopique du massif du MontBlanc: In: Isotope Hydrology (Proc. Symp., Neuherberg, 19-23 June). International Atomic Energy Agency, Vienna, Vol. 1, pp. 411-440.
Foster, S. S. D., 1975. The chalk groundwater tritium anomaly: A possible explanation: J. Hydrol. 25: 159-165.
Foster, S. S. D., and A. Smith-Carrington, 1980. The interpretation of tritium in the chalk unsaturatedzone: J. Hydrol. 46: 343-364.
Glass, R. J., T. S. Steenhuis, and J.-Y. Parlange, 1989. Mechanism for finger persistence in homogeneous, unsaturated porous media: Theory and verification. Soil Sci. 148: 60-70.
Hammermeister, D. P., 1982. Perched water tables on hillsides in western Oregon. II. Preferential downslope movement of water and anions. Water Resour. Res. 46: 819-826.
Harmon, R. S., 1979. An isotopic study of groundwater seepage in the central Kentucky karst. Water Resour. Res. 15: 476-480.
Hendrickx, J. M. H., L. W. Dekker, and O. H. Boersma, 1993. Unstable wetting fronts in water repellent field soils. J. Env. Qual. 22: 109-118.
IAEA (International Atomic Energy Agency), 1981. Stable isotope hydrology: Deuterium and oxygen-18 in the water cycle . Technical Report Series No. 210, Vienna, 337 pp.
IAEA (International Atomic Energy Agency), 1983a. Aqueous sulfur. Ch. 8.4 in: Isotope techniques in the hydrogeological assessment of potential sites for the disposal of high level radioactive wastes. Technical Report Series No. 228, Vienna, pp. 76-86.
IAEA (International Atomic Energy Agency), 1983b. Isotope techniques in the hydrogeological assessment of potential sites for the disposal of high level radioactive wastes: IAEA Technical Report Series No. 228, 151 pp.
IAEA (International Atomic Energy Agency), 1983c. Tritium. Ch. 7 in: Isotope techniques in the hydrogeological assessment of potential sites for the disposal of high level radioactive wastes. Technical Report Series No. 228, Vienna, pp. 57-61.
Ingraham, N. L., J. B. Chapman, and J. W. Hess, 1990. Stable isotopes in cave pool systems: Carlsbad Caverns, New Mexico . Chem. Geol. (Isot. Geosci. Sect.) 86: 65-74.
Jaques, D., D. J. Kim, J. Diels, J. Vanderborght, H. Vereecken, and J. Feyen, 1998. Analysis of steady-state chloride transport through two heterogenous field soils. Water Resour. Res. 34: 2539-2550.
Johnson, T. M., and D. J. DePaolo, 1994. Interpretation of isotopic data in groundwater-rock systems: Model development and application to Sr isotope data from Yucca Mountain . Water Resour. Res. 30: 1571-1588.
Johnston, C. T., P. G. Cook, S. K. Frape, L. N. Plummer, E. Busenberg, and R. J. Blackport, 1998. Ground water age and nitrate distribution within a glacial aquifer beneath a thick unsaturated zone. Ground Water 36: 171-180.
Jørgensen, P. R., L. D. McKay, and N. H. Spliid, 1998. Evaluation of chloride and pesticide transport in a fractured clayey till using large undisturbed columns and numerical modeling. Water Resour. Res. 34: 539-554.
Jury, W. A., and H. Flühler, 1992. Transport of chemicals through soil: Mechanisms, models, and field applications. Adv. Agron. 47: 141-201.
Larson, M. K., and T. L. Péwé, 1986. Origin of land subsidence and earth fissuring, northeast Phoenix, Arizona. Assoc. Eng. Geol. Bull. 23: 139-165.
Leany, F. W. J., K. R. J. Smettem, and D. J. Chittleborough, 1993. Estimating the contribution of preferential flow to subsurface runoff from a hillslope using deuterium and chloride. J. Hydrol. 147: 83-103.
Leenhouts, J. M., R. L. Bassett, and T. I. Maddock, 1998. Utilization of intrinsic boron isotopes as co-migrating tracers for identifying potential nitrate contamination sources. Ground Water 36: 240.
McDonnell, J. M., M. K. Stewart, and I. F. Owens, 1991. Effect of catchment-scale subsurface mixing on stream isotopic response . Water Resour. Res. 27: 3065-3073.
Mulholland, P. J., G. V. Wilson, and P. M. Jardine, 1990. Hydrogeological response of a forested watershed to storms: Effects of preferential flow along shallow and deep pathways. Water Resour. Res. 26: 3021-3036.
National Research Council, 1996. Rock Fractures and Fluid Flow: Contemporary Understanding and Applications . National Academy Press, Washington, D.C., 551 pp.
Neretnieks, I., 1980. Diffusion in the rock matrix: An important factor in radionuclide retardation. J. Geophys. Res. 85: 4379-4397.
Newman, B. D., and K. H. Birdsell, 1997. Characterization of deep evaporation in mesas at Los Alamos, New Mexico, using environmental tracers and numerical modeling. Eos 78(46): F318.
Newman, B. D., A. R. Campbell, and B. P. Wilcox, 1998. Lateral subsurface flow pathways in a semiarid ponderosa pine hillslope . Water Resour. Res. 34: 3485-3496.
Östlund, H. G., 1982. The residence time of the freshwater component in the Arctic Ocean . J. Geophys. Res. 87: 2035-2043.
Phillips, F. M., 1999. Chlorine-36. In: Environmental Tracers in Subsurface Hydrology (P. G. Cook and A. L. Herczeg, eds.). Kluwer Academic, Boston, pp. 299-348.
Plummer, M. A., F. M. Phillips, J. Fabryka-Martin, H. J. Turin, P. E. Wigand, and P. Sharma, 1997. Chlorine-36 in fossil rat urine: An archive of cosmogenic nuclide deposition over the past 40,000 years. Science 277: 538-541.
Rauert, W., and W. Stichler, 1974. Groundwater investigations with environmental isotopes. In: Isotope Techniques in Groundwater Hydrology. International Atomic Energy Agency, Vienna, Vol. 2, pp. 431-443.
Ritsema, C. J., L. W. Dekker, J. L. Nieber, and T. S. Steenhuis, 1998. Modeling and field evidence of finger formation and finger recurrence in a water repellent sandy soil. Water Resour. Res. 34: 555-567.
Rodhe, A., 1981. Spring flood: Meltwater or groundwater? Nordic Hydrol. 12: 21-30.
Saxena, R. K., 1984. Seasonal variations of oxygen-18 in soil moisture and estimation of recharge in esker and moraine formations. Nordic Hydrol. 15: 235-242.
Scanlon, B. R., 1992. Moisture and solute flux along preferred pathways characterized by fissured sediments in desert soils. J. Cont. Hydrol. 10: 19-46.
Scanlon, B. R., R. S. Goldsmith, and J. G. Paine, 1997. Analysis of focused unsaturated flow beneath fissures in the Chihuahuan Desert, Texas, USA. J. Hydrol. 203: 58-78.
Scholl, M. A., S. E. Ingebritsen, C. J. Janik, and J. P. Kauahikaua, 1996. Use of precipitation and groundwater isotopes to interpret regional hydrology on a tropical volcanic island: Kilauea volcano area, Hawaii . Water Resour. Res. 32: 3525-3538.
Sklash, M. G., 1990. Environmental isotope studies of storm and snowmelt runoff generation . In: Process Studies in Hillslope Hydrology (M. G. Anderson and T. P. Burt, eds.). John Wiley, New York, pp. 401-435.
Sklash, M. G., R. N. Farvolden, and P. Fritz, 1976. A conceptual model of watershed response to rainfall, developed through the use of oxygen-18 as a natural tracer. Can. J. Earth Sci. 13: 271-283.
Smith, D. B., P. L. Wearn, H. J. Richards, and P. C. Rowe, 1970. Water movement in the unsaturated zone of high and low permeability strata by measuring natural tritium. In: Isotope Hydrology 1970, International Atomic Energy Agency, Vienna, pp. 73-87.
Synal, H.-A., J. Beer, G. Bonani, M. Suter, and W. Wölfli, 1990. Atmospheric transport of bomb-produced 36Cl. Nucl. Instrum. Meth. Phys. Res. B52: 483-488.
Thoma, G., N. Esser, C. Sonntag, W. Weiss, J. Rudolf, and P. Leveque, 1979. New technique of insitu soil moisture sampling for environmental isotope analysis applied at Pilat sand dune near Bordeaux: In: Isotopes in Hydrology. International Atomic Energy Agency, Vienna, pp. 753-768.
Thorstenson, D. C., E. P. Weeks, H. Haas, E. Busenberg, L. N. Plummer, and C. A. Peters, 1998. Chemistry of unsaturated zone gases sampled in open boreholes at the crest of Yucca Mountain, Nevada. Data and basic concepts of chemical and physical processes in the mountain. Water Resour. Res. 34: 1507-1530.
Van Dam, J. C., J. M. H. Hendrickx, H. C. van Ommen, M. H. Bannik, M. T. van Genuchten, and L. W. Dekker, 1990. Water and solute movement in a coarse-textured water-repellent field soil. J. Hydrol. 120: 359-379.
Weeks, E. P., 1987. Effect of topography on gas flow in unsaturated fractured rock: Concepts and observations. In: Flow and Transport Through Unsaturated Fractured Rock. Geophysical Monograph Series, Volume 42(D. D. Evans and T. J. Nicholson, eds.) American Geophysical Union, Washington, D.C., pp. 165-170.
Yonge, C. J., D. C. Ford, J. Gray, and H. P. Schwarcz, 1985. Stable isotope studies of cave seepage water. Chem. Geol. (Isot. Geosci. Sect.) 58: 97-105.
Zerle, L., T. Faestermann, K. Knie, G. Korschinek, and E. Nolte, 1997. The 41Ca bomb pulse and atmospheric transport of radionuclides. J. Geophys. Res. 102: 19517-19527.
























