Page 107
6
Health and Biomedical Sciences
Like all aspects of the modern world, medicine has been transformed by discoveries in physics during the past century. Modern medicine is unthinkable without modern chemistry, and modern chemistry could never have existed without the fundamental understanding of atoms and molecules provided by the revolution in physics that started just about 100 years ago. A series of advances in physics itself has also had a decisive and direct impact on medical science as we know it today, starting with the discovery of x rays, for which Roentgen was awarded the Nobel Prize in 1901, and the discovery of radioactivity, for which Becquerel and the Curies received the Nobel Prize in 1903. Today nearly all the therapeutic and diagnostic tools of a modern hospital have their origin in basic physics research.
THERAPY
Ionizing radiation remains one of the three options for treating cancer. Either alone, or in combination with surgery and chemotherapy, ionizing radiation is used as therapy for most malignancies. Over the years, the use of ionizing radiation has become more sophisticated, and it is highly effective for certain types of tumors. Recently, for example, the radiation transport absorption coefficients and results from the simulation programs used by nuclear weapons designers have been applied to the problem of optimizing radiation dose in cancer treatment. The result is a new dose calculation system that for the first time can model the varying materials and densities in the body in combination with the radiation beam delivery system.
The breakthrough brings to the field of radiotherapy a new level of accuracy in the ability to predict where radiation deposits energy in the body, opening the way to more accurate prescriptions for radiation therapy, more aggressive treatment of tumors, and substantially lower risk to normal tissue. For example, this new approach will allow more aggressive treat-
Page 108
ment of prostate cancers without endangering the bladder and spinal tissue adjacent to the prostate gland.
Lasers are a much more recent invention, but their therapeutic use is rapidly increasing. Focal tissue coagulation by laser is the standard nonsurgical treatment for a detached retina, and the reforming of the corneal surface shape (laser keratotomy) with laser radiation is a very effective treatment for nearsightedness that is now becoming standard. The use of local laser heating is a microsurgical technique that is finding application in a variety of fields.
Although of more limited use, ultrasound has also found an important place as a nonsurgical treatment for kidney stones and for the cleaning of surgical instruments.
Another revolution in therapy, based on the use of fiber optics, has been developing over the past several decades. Before physicists developed ordered fiber-optic bundles, visualization of internal body surfaces was limited by the requirement that light must follow straight lines; this meant, for instance, that only small portions of the gastrointestinal tract and the airways could be seen by a physician. With fiber-optics and video imaging, the gastroenterologist and otolaryngologist can now see much of the inner surface of humans. A more recent trend has been to use fiber-optics imaging to permit surgery through tiny incisions. Many appendectomies and other surgical procedures—even including heart surgery—are now carried out in this much less invasive way. Because damage caused by large incisions and the exposure of internal organs is minimized, these remote surgical procedures can be less dangerous and patients can recover more quickly and with less discomfort.
New materials have also become available to replace damaged or missing body components. Silicone is used in reconstructive surgery to provide internal support for soft tissues, and new, hard plastic materials are transforming dentistry by permitting better reconstruction of damaged teeth. These materials not only are strong but also bond well to the tooth surface and expand with heat like the natural tooth; this means that a much wider range of tooth damage can be repaired, and that the repairs are much more durable than in the past.
DIAGNOSIS
The use of ionizing radiation has been important therapeutically for more than half a century, but physics also plays an ever more important role in diagnosis. Because such small quantities of radioactive material can be
Page 109
detected, radioactive labels have become very important in a variety of diagnostic contexts. Labeling red blood cells with radioactive isotopes of chromium permits the lifetime of these cells to be measured, and this method can be used to determine if anemia is the result of decreased production or increased destruction of the red blood cells. The radioimmune assay—Yalow received the Nobel Prize in 1977 for the development of this technique—makes use of antibodies that have been made radioactive; these antibodies detect minute quantities of hormones and other chemicals by binding to them and providing a radioactive tag that can be used to detect the presence of the molecule. Because signaling molecules, like hormones, are present at minute concentrations in the blood and other bodily fluids, other methods for detecting their presence are too insensitive. Some organs specifically take up certain atoms or chemical compounds (the uptake of iodine by the thyroid gland is the best-known example), and this fact has enabled physicians to assess organ function or identify the presence of damage by monitoring the uptake of these substances that have been tagged with radioactivity.
Probably the most striking advance in medical diagnostics has been the development of remarkable imaging techniques. Our ability to look inside the living body noninvasively started with Roentgen's discovery of x rays about a century ago. X-ray machines soon became common and were the standard—and only—method for diagnostic imaging until the recent explosion in imaging technology. As powerful computers became available, the data contained in images could be manipulated to extract the available information about three-dimensional structures present in two-dimensional images made from different views. Cormak received the 1979 Nobel Prize for developing computerized tomography, the now-standard method for extracting such three-dimensional information from two-dimensional projections.
Versions of this tomographic method have been extended to other types of imaging. Magnetic resonance imaging (MRI), formerly called nuclear magnetic resonance imaging, is based on the discovery of magnetic resonance in nuclei, for which Rabi received the 1944 Nobel Prize in physics. Rare only a few years ago, MRI is now standard and practiced in virtually all medical centers (see sidebar “Computerized Tomography”). Because of the remarkable resolution of this method—brain structures in the millimeter size range are clearly imaged—MRI and its more recent modifications, such as magnetic resonance angiography (for viewing blood vessels in the heart), have transformed many areas of medical practice, from neurology to the surgical specialties.
Page 110
COMPUTERIZED TOMOGRAPHY
The modern descendants of the venerable x-ray machine use computer technology to greatly improve the quality of the images produced. Computerized tomography (CT) takes information from two or more slightly different views of the same object and reconstructs a three-dimensional image of the object that can be rotated and sliced by computer. This technique is now applied to x-ray images and also to novel imaging methods such as MRI (magnetic resonance imaging) and SPECT (single-photon-emission computerized tomography) imaging. On the top is a pair of slices through an image of a head made by MRI, and below a similar pair of slices of an image of the same head produced using SPECT. Atoms such as hydrogen (in water) have magnetic properties, and these subatomic magnets can be aligned by a very strong magnetic field; how fast the tiny magnets line up depends on their surroundings, and this is exploited by MRI to produce images of tissue in the living body. Certain radioactive agents that have been injected into the bloodstream, like technetium-99, emit photons, and so brightness of the “light” emitted is used by SPECT to tell how much of the agent is present and therefore how much blood is flowing to that region. Because organs increase blood flow according to their needs—the brain does this for very small regions devoted to particular functions, such as language—the colors that appear provide a picture of the brain's activity at each location. Both of the methods that gave these images have become standard ways for evaluating organ structure and function, illustrated here by images of our most complex organ, in health and disease. 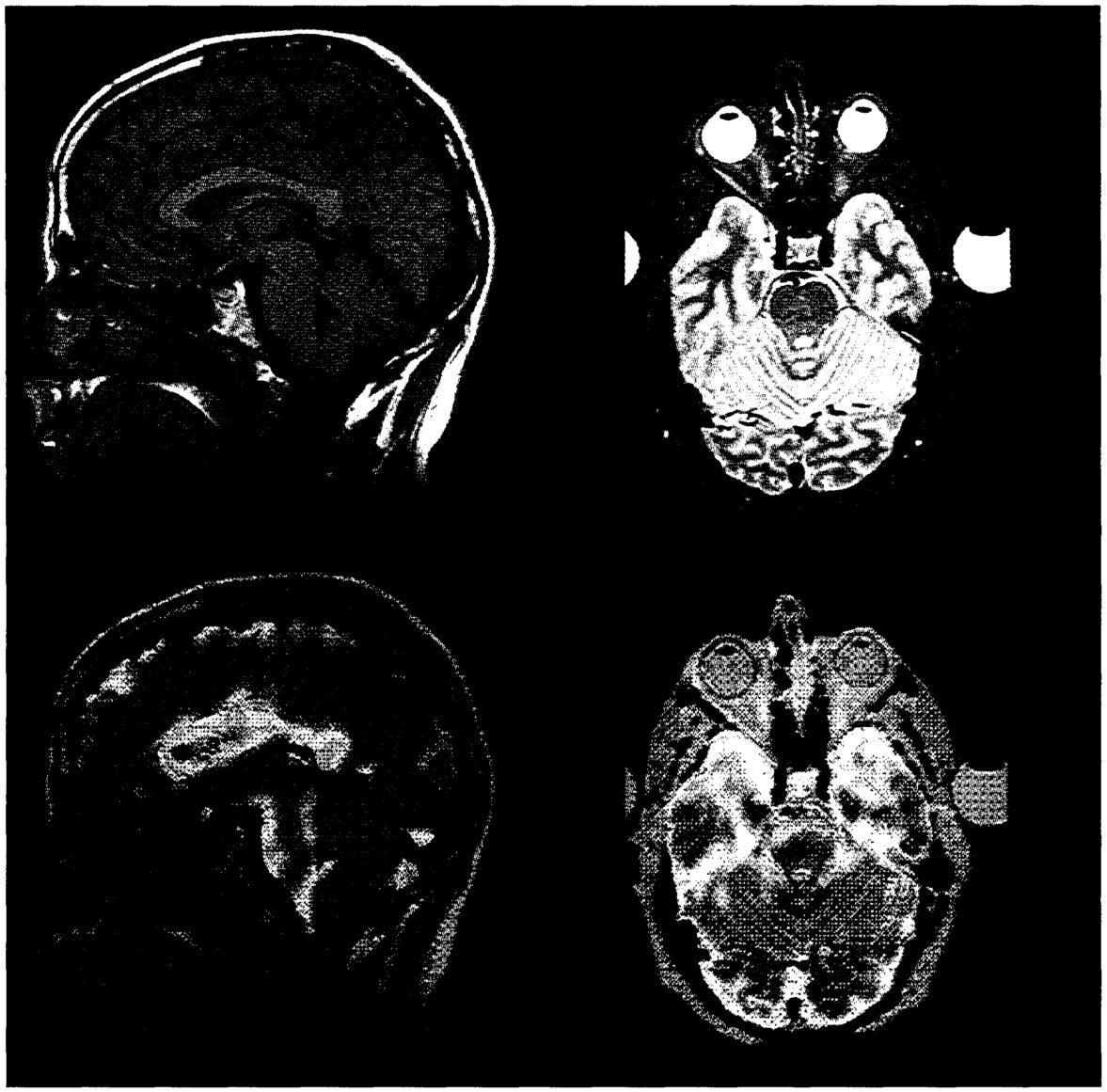
~ enlarge ~ |
Page 111
Writing recently in the Washington Post, former NIH director Harold Varmus pointed to MRI to underscore the important contributions of the physical sciences to health care:
Medical advances may seem like wizardry. But pull back the curtain, and sitting at the lever is a high energy physicist, a combinational chemist or an engineer. Magnetic resonance imaging is an excellent example. Perhaps the last century's greatest advance in diagnosis, MRI is the product of atomic, nuclear and high-energy physics, quantum chemistry, computer science, cryogenics, solid state physics and applied medicine.” 1
Related techniques developed by the physics community, such as functional magnetic resonance imaging (fMRI) (see sidebar “Functional Magnetic Resonance Imaging”) and positron emission tomography (PET), are now being applied to understand the functioning of the human brain. Researchers are using these high-resolution scanning techniques, which are sensitive to local brain metabolism or even to local concentration of complex psychoactive biochemical molecules that are thought to regulate brain function, to diagnose and guide treatment for a number of mental illnesses and for some forms of drug addiction.
For example, brain scans have identified depressed metabolic rates and regions of abnormal biochemical concentrations in those regions of the brain associated with impulse control. Researchers have correlated these deficiencies with patients who have been diagnosed with attention deficit disorder (ADD). This opens the possibility of both a direct diagnosis of this condition and a way to monitor the effect of medications such as Ritalin on the brain function of patients with ADD.
Similarly, brain scans are being used to identify local metabolic or biochemical anomalies in patients with Alzheimer's disease and with addictions to cocaine and other substances. These scans also offer the possibility of directly identifying the underlying disorder and a way of monitoring directly the impact of medications and treatment.
The revolution in imaging made possible by the availability of powerful computers extends to the use of ultrasound. In many areas of medical practice, particularly obstetrics, ultrasound images are standard and can be used to follow changes over time, such as fetal growth, with a resolution sufficient to detect structural abnormalities or determine fetal sex.
Advances in imaging also extend to light microscopy. Zernike's invention of phase-contrast microscopy (for which he received the Nobel Prize in
1 Harold Varmus. 2000. “Squeeze on Science,” Washington Post, October 4, p. A33.
Page 112
FUNCTIONAL MAGNETIC RESONANCE IMAGING
Functional magnetic resonance imaging (fMRI) works by mapping tiny magnetic disturbances caused by changes in the amount of oxygen present in tissue. Since the brain very precisely regulates the blood supply to support the high metabolic demands of active neurons, changes in fMRI signals reflect changes in neural activity. In the image below, fMRI-measured neural activity is projected onto a three-dimensional rendering of a subject's brain. For visualization purposes, the cortex has been computationally inflated so the depths of the brain's folds are revealed; the view is slightly from the back so that some of the right side can be seen. The boundaries of the various cortical regions that process visual information (V1, V2, V3A, MT) are defined by experiments that map the cortical representation of the visual world. The colored pixels within the visual area boundaries represent the results of an experiment measuring the effects of visual spatial attention. During the experiment, a subject had to detect a stimulus that appeared on either the left or the right side of the visual field. The subject was told to attend alternately to the left and the right, but neither the stimulus nor the subject's eye position changed. In the image, the red pixels represent activity modulated during the attend-right phase of the experiment, and the green pixels, activity modulated during the attend-left phase. The red region labeled MT is the cortical area specialized for detecting movement. 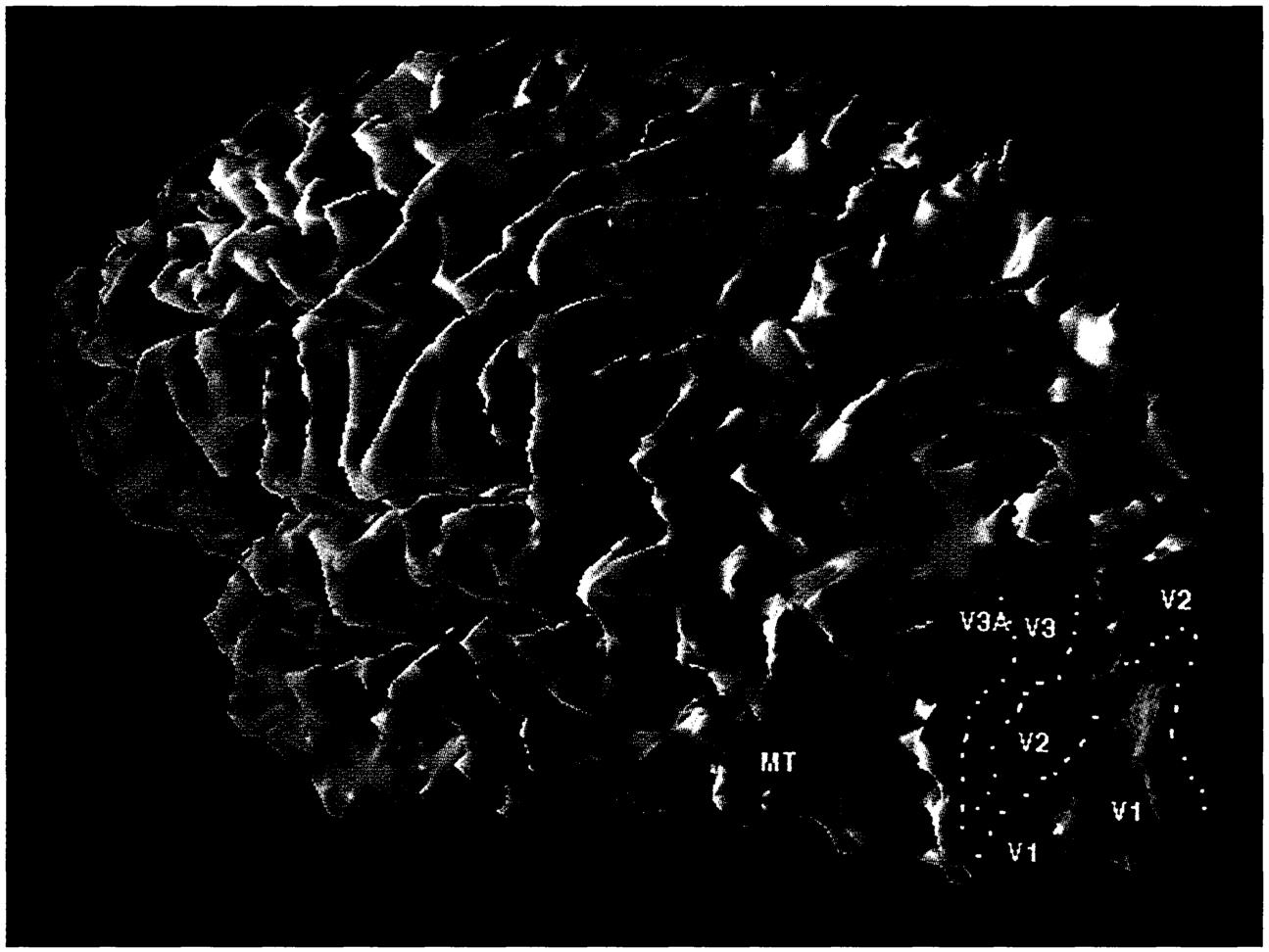
~ enlarge ~ |
1953) was the first in a series of improvements and extensions of traditional light microscopy. Differential interference contrast, videomicroscopy with computerized manipulation of the image, infrared microscopy, laser scan-
Page 113
ning microscopy, and two-photon microscopy are recently developed microscopy techniques that have become standard for research and are beginning to be used for medical diagnosis.
UNDERSTANDING THE BODY
The basis for medical practice resides in an understanding of how the body functions. Physics has contributed to this knowledge in essential ways through biophysical research and through the development of enabling technologies for the biological sciences. At the level of molecules, structure and function are inseparable. Proteins are giant molecules that provide cells with their internal mechanical support and are responsible for carrying out or enabling all of the chemical processes at the core of life. Over the past several decades, biophysicists have determined the exact atomic structure of hundreds of proteins by x-ray crystallography (the technique for which the Braggs won the Nobel Prize). More recently, synchrotron radiation, produced at several of the large particle accelerator facilities (Lawrence received the Nobel Prize in physics in 1939 for inventing the first of these accelerators, the cyclotron), has been used increasingly in place of traditional x-ray machines to determine protein structures more rapidly and accurately. Because of this vast amount of work, we now understand just how many proteins carry out their jobs.
Understanding the structure of complex biological molecules is also the key to developing new pharmaceuticals by an approach that is more rational than the hit-and-miss approach of the past. Viruses are an important example. A virus enters a cell by penetrating the cell membrane after docking to a surface protein, a process that can occur only when the shape of the virus fits the cell's protein as a key would fit a lock. Determining the underlying structure of a virus such as the common cold virus or HIV opens the possibility of developing pharmaceutical molecules that compete with these viruses for the docking sites and thus prevent viral penetration into the cell.
Most drugs work by binding to unique sites on specific proteins and modifying how the protein does its job. The search for molecules that can bind selectively to proteins and alter their function is very difficult and inefficient if done by trial and error. It is the application of physics techniques such as x-ray crystallography that allows determining the structure of viruses and identifying candidate sites on a virus to mimic with a therapeutic molecule. This rational drug design is now a major aspect of research at the many synchrotron radiation facilities that have been developed by the physics community in the past decade. A number of large pharmaceutical
Page 114
OPTICAL TWEEZERS
Novel techniques, most of them invented in the last several decades, are being used increasingly to study individual molecules of importance to biology. A good example is measuring the “horsepower” of kinesin molecules, illustrated in the figure. Kinesin is a molecular motor, one of dozens of different sorts of specialized protein motors that are responsible for all forms of biological movement in all living organisms, from bacteria to humans. The most familiar manifestation of molecular motors at work is our ability to generate force with our muscles, but the beating of our heart, the tension of blood vessels that sets our blood pressure, the division of one cell into two during growth, and the movement of molecular cargoes from one place in a cell to another are all jobs that depend on a variety of molecular motors. Kinesin is a motor that moves along tiny “railroad tracks” called microtubules to carry important molecular structures from one place in a cell to another. The strength of single motors was recently measured at Stanford University. Physicists there used optical “tweezers,” a method that generates a force on small objects by laser light focused at a tiny point. The kinesin motor was attached to a small bead and then permitted to “walk” along its track, a microtubule. The force generated by the kinesin could be measured by seeing how hard the tweezers had to pull back on the bead to keep the motor from moving along the microtubule. Experiments of this sort are revealing how chemical energy, ultimately derived from the food we eat, is turned into forces that we use to affect our environment, both external and internal. 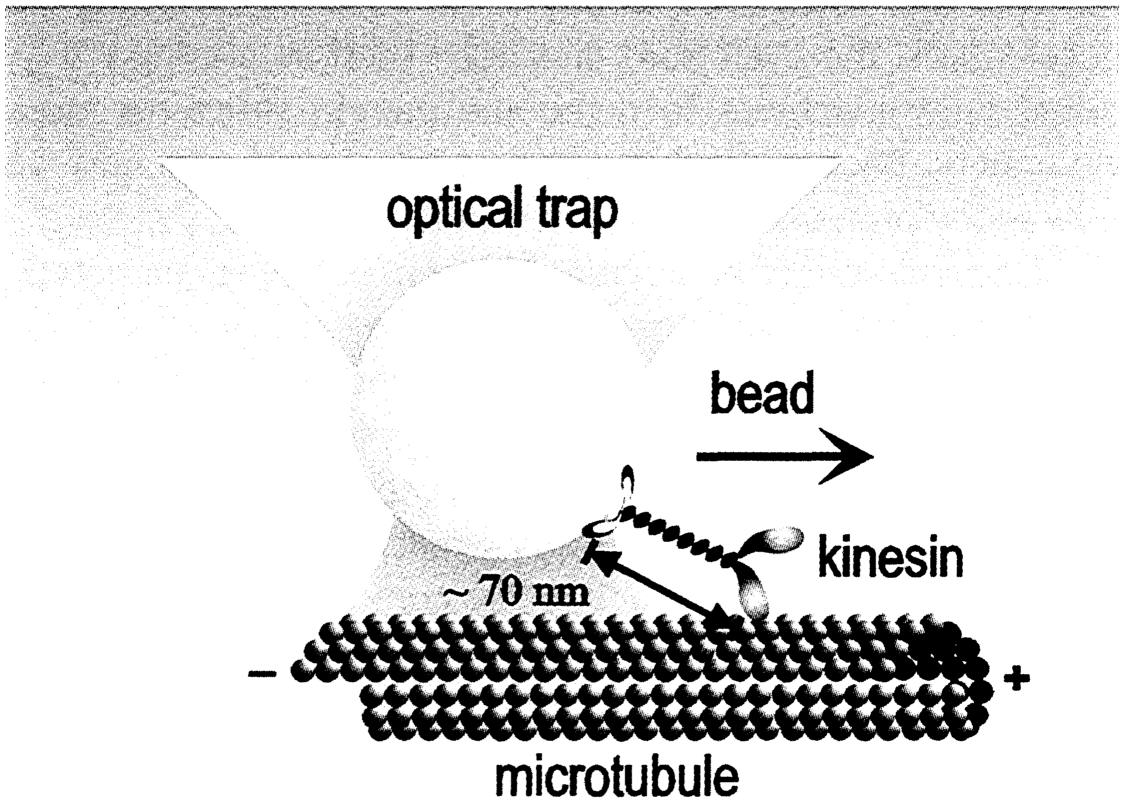
~ enlarge ~ |
Page 115
companies and the NIH have invested many million of dollars in instrumentation at these facilities to support this kind of research.
Knowing the structure of proteins is essential for understanding their function, but we must also learn how parts of proteins move as they carry out their tasks. Biophysicists have combined the methods of modern molecular biology with sophisticated physical techniques to learn just how many types of proteins do their job. All cells contain tiny molecular motors—these motors are a large family of proteins—that help the cell to move or that move material within the cell from one place to another. The most familiar manifestation of these motors at work is muscle contraction and the pumping of the heart, but molecular motors are essential for the jobs performed by all cells, from the dividing of cells to produce replacements for cells that die to the carting of essential chemicals to where they are needed in the brain (see sidebar “Optical Tweezers”). Unraveling how these motors work has required developing clever physical methods to measure the motions of single protein molecules over distances that are smaller than can be seen with microscopes.
The beating of the heart and all functions of the brain require another kind of protein that produces electrical signals in cells. The electrocardiogram and the electroencephalogram are manifestations of the operation of these proteins and are used for the diagnosis of heart and neurological diseases. Biophysicists have also learned how these proteins generate their electrical signals, and this knowledge has given us many therapies, ranging from drugs to treat abnormal heartbeats to the treatment of epilepsy.
SUMMARY
In small ways and large, from the treatment of nearsightedness to the diagnosis of neurological diseases, applications of the discoveries of physics have revolutionized medical practice. Although this transformation began 100 years ago, it has greatly accelerated in the past several decades as our increased ability to process information has been applied to a variety of physical phenomena. In the 21st century, physics will continue to provide the foundation for striking advances in biomedicine and health. The increasingly interdisciplinary nature of physics research is focusing more of the field's intellectual and technical resources on addressing opportunities in biology and medicine, both to increase our understanding of basic mechanisms and to provide novel technologies for research and application. In the coming decades we can confidently expect that physics will continue to contribute to the nation's health and, in fact, that these contributions will increase.









