3
Lifestyle Behaviors Contributing to the Burden of Cancer1
Much of the burden of cancer in the United States can be traced to modifiable health behaviors that increase one’s risk of disease. This chapter reviews evidence on the contributions of five major risk factors to cancer incidence and mortality: tobacco use, physical inactivity, overweight and obesity, poor diet, and alcohol use. Although other lifestyle factors such as sun exposure, sexual practices, and exposure to infected blood also contribute substantially to cancer incidence, this chapter focuses only on the factors that are risks for common cancers and that also have large impacts on the incidences of other major diseases such as cardiovascular disease, stroke, diabetes, and osteoporosis. The benefits of intervention are far greater when reductions in the incidence of other chronic diseases add to reductions in the incidence of cancer achieved through lifestyle changes (Colditz and Gortmaker, 1995).
This chapter summarizes the epidemiological evidence with respect to cancer incidence for each of the five selected risk factors by using the criteria outlined by the World Cancer Research Fund and the American Institute of Cancer Research. For each epidemiological association the level of evidence is categorized as convincing, probable, or possible (Box 3.1).
Much of the literature reviewed in this chapter has been published from research conducted with data from one of the several large prospective cohort studies described in Box 3.2. Such studies involve the identification
|
1 |
The chapter is based on the background paper prepared by Graham A. Colditz, Catherine Tomeo Ryan, Charles H. Dart III, Geetanjali Datta, Laurie Fisher, and Beverly Rockhill (www.iom.edu/ncpb). |
|
BOX 3.1 Definition of Levels of Evidence for Epidemiological Associations
|
||||||||||
of a group of individuals who share a common experience within a defined time period and monitoring of those individuals forward in time for the development of disease (Mausner and Kramer, 1985). These studies have been essential to providing an understanding of the links between health-related behaviors and health outcomes. Other evidence comes from case-control studies in which investigators identify a group of patients with a particular cancer (cases) and a group of patients without cancer (controls) and then compare the histories of the cases and controls to determine the extent to which each was exposed to the intervention of interest.
TOBACCO
Tobacco is the scourge of public health. In the United States alone, tobacco use, primarily in the form of cigarette smoking, causes more than 440,000 premature deaths from cancer and other causes each year and is responsible for approximately 30 percent of all cancer-related deaths (ACS, 2002b). Worldwide, the numbers are even more staggering. The World Health Organization estimated that in 1998 there would be approximately 4 million deaths linked to tobacco use worldwide (World Health Organization, 1999). By 2020, this number is expected to at least double.
|
BOX 3.2 Frequently Referenced Prospective Cohort Studies American Cancer Society’s Cancer Prevention Studies Cancer Prevention Study I (CPS I) A prospective study of approximately one million men and women. Enrollment started in 1959 and follow-up ran through 1972, with mortality being the primary outcome measure (ACS, 2001). Enrollment was household-based, and all members of a household age 30 or older were included in the study if at least one household member was age 45 or older. Upon entering the study, participants completed questionnaires assessing lifestyle factors (e.g., occupation, diet, and tobacco use), reproductive factors (in women), personal and family history of cancer, as well as anthropometric and demographic information. After this, questionnaires were sent to participants at regular intervals to update tobacco use and assess vital status. Cancer Prevention Study II (CPS II) A prospective study of approximately 1.2 million men and women (ACS, 2001). Started in 1982 and still ongoing, the primary aim of CPS II is to assess the effect that lifestyle and environmental factors have on cancer development. As with CPS I, enrollment in CPS II was household-based and included all household members age 30 or older if at least one member was age 45 or older. A range of lifestyle and other factors were assessed by questionnaire at study enrollment. The study’s main outcome measure is mortality, which is assessed biennially through links with the National Death Index. Health Professionals Follow-Up Study A prospective study of approximately 52,000 male health professionals—including dentists, optometrists, osteopaths, podiatrists, pharmacists, and veterinarians (Harvard School of Public Health, 2001). Started in 1986 in men ages 40–75, it is still ongoing. The primary aim of the Health Professionals Follow-Up Study is to assess the effect of lifestyle on the risk of chronic disease, particularly cancer and cardiovascular disease. Data are gathered largely through biennial questionnaires that ask participants detailed questions about their disease status as well as about their lifestyle and personal characteristics. Male British Doctors Study A prospective mortality study of 34,000 male British doctors, which started in 1951 and is still ongoing (Doll et al., 1994). The study’s initial aim was to build a body of evidence linking tobacco use to chronic disease. While the questionnaire at enrollment focused largely on tobacco use, subsequent follow-up questionnaires added questions related to alcohol use, aspirin use, disease status, and certain personal characteristics. Nurses’ Health Study A prospective study of approximately 120,000 female registered nurses, which is primarily designed to assess the effect of lifestyle on the risk of chronic disease, particularly cancer and cardiovascular disease (Colditz, 1995). Started in 1976 in women ages 30–55, the Nurses’ Health Study is still ongoing. Data are gathered primarily through biennial questionnaires that ask participants detailed questions about their disease status as well as their lifestyle and personal characteristics. To provide a more accurate accounting of the intake of certain minerals, participants provided toenail samples in 1982. In 1989, participants provided blood samples to allow the study of potential disease biomarkers. |
Since the first Surgeon General’s report on smoking and health was released in 1964, a causal link has been confirmed between smoking and several cancers (U.S. Department of Health, Education, and Welfare, 1964). The data available at that time were able to support a causal link between smoking and only two cancers: lung cancer and laryngeal cancer. Over the following 35 years, however, enough evidence has accrued to now support smoking as a cause of eight cancers: lung, oral, pharyngeal, laryngeal, esophageal, bladder, kidney, and pancreatic cancer. A review of the current literature provides sufficient evidence to also implicate tobacco as a cause of additional cancers, including cancers of the colon, stomach, and cervix, and leukemia. Smoking has also been identified as a probable cause of liver cancer and has been associated with an increase in the risk of developing aggressive, more deadly forms of prostate cancer (Table 3.1). Smoking is not causally related to several types of cancer, such as breast or brain cancer.
Impact of Cessation of Tobacco Use on Cancer Incidence
Although the prevention of smoking is the best approach to avoiding tobacco-related diseases, there are substantial health benefits for smokers who quit. Scientific data on the benefits of smoking cessation were reviewed in detail in a 1990 Surgeon General’s report (US DHHS, 1990). In the last quarter century, half of all living Americans who have ever smoked have now quit. The 1990 Surgeon General’s report concluded that smoking cessation has major and immediate health benefits for men and women of all ages. Former smokers live longer than continuing smokers; for instance, persons who quit smoking before age 50 have, on average, half the risk of dying in the next 15 years than continuing smokers. This reduction in mortality comes from a reduction in the risk of nearly all smoking-related
TABLE 3.1 Increase in Risk of Incident Cancer Associated with Smoking
|
|
Relative Risk (RR) |
|
|
Level of Evidence |
Moderate (RR 1.35–1.99) |
Large (RR 2.0+) |
|
Convincing |
Colon Stomach Leukemia Cervical |
Lung Oral Pharyngeal Laryngeal Esophageal Bladder Kidney |
|
Pancreatic |
Probable Prostate (mortality) |
Liver |
diseases. Among former smokers, the decline in the risk of death compared with that for individuals who continue smoking begins shortly after quitting and continues for at least 10 to 15 years. The health benefits of smoking cessation far exceed the risk from the weight gain, which is approximately 2.3 kilograms (kg) (5 pounds [lb]), on average, that may follow quitting.
Tobacco Use and Lung Cancer
Voluminous and convincing epidemiological evidence links smoking to lung cancer, the leading cause of cancer death in both men and women in the United States. An estimated 169,400 new cases of lung cancer are diagnosed annually, with an estimated 154,900 deaths each year (ACS, 2002a). More than 80 percent of lung cancer cases are attributable to smoking. As outlined in detail in the Surgeon General’s reports on smoking and health, cigarette smoking is the strongest risk factor for lung cancer, increasing the risk of the disease by at least 10-fold and as much as 20-fold, depending on smoking habits and history (US DHHS, 1989; Thun, Dally-Lally et al., 1997). There is a consistent, strong, and specific link between smoking and lung cancer, with a dose-response relationship seen with the number of cigarettes smoked, the deepness of inhalation of cigarette smoke, and the duration of smoking (US DHHS, 1989).
There is convincing evidence from numerous case-control and cohort studies that former smokers have a lower risk of lung cancer than current smokers (US DHHS, 1990). The benefits begin 2 to 3 years after quitting, and the risk steadily drops over the next 10 years. Although the risk of lung cancer is drastically reduced after quitting, the risk of lung cancer in former smokers never quite returns to the risk for those who have never smoked.
The overall pattern seen in the study of U.S. veterans mimics that of most other large cohort studies of smoking cessation and lung cancer risk (Rogot and Murray, 1980; US DHHS, 1990). In the U.S. Veterans Study, the lung cancer mortality rate in current smokers divided by the rate in nonsmokers (i.e., the mortality rate ratio) was 11.3. In the first 4 years after quitting, this ratio increased to nearly 19 for former smokers and then slowly decreased: 7.7 after 5 to 9 years, 4.7 after 10 to 14 years, 4.8 after 15 to 19 years, and 2.1 after 20 or more years. The increase in lung cancer risk in the first few years after quitting seen in many studies likely reflects the tendency of smokers to quit because of symptoms of disease rather than a true increase in risk linked to cessation per se.
Numerous studies have also found that smoking cessation benefits nearly all smokers, regardless of sex, age, and level of habit. The American Cancer Society’s Cancer Prevention Study I and Cancer Prevention Study II have demonstrated that both men and women benefit from cessation, as do both light smokers (those who smoke 1 to 20 cigarettes/day) and heavy
smokers (those who smoke 21 or more cigarettes/day). In addition, an analysis of national smoking trends in the United Kingdom found substantial benefits for smokers who stopped at a wide range of ages (Peto et al., 2000). Those smokers who stopped before middle age benefited the most, avoiding 90 percent of the excess risk of lung cancer linked to tobacco use. However, those who stopped at ages 50 and 60 years also benefited substantially.
Tobacco Use and Oral, Pharyngeal, Laryngeal, and Esophageal Cancers
Tobacco use is a well-established cause of cancer of the oral cavity, pharynx, larynx, and esophagus (US DHHS, 1989). Oropharyngeal cancer alone—including cancer of the lip, tongue, oral cavity, and oropharynx—is diagnosed in approximately 28,900 people each year in the United States and accounts for about 7,400 deaths annually (ACS, 2002a). Once oropharyngeal cancer was as much as six times more common in men than in women, but men are now only about twice as likely as women to develop the disease. This is due in large part to the disappearing disparity in smoking rates between the two sexes (CDC, 1999b). Although disparity in the incidence of oropharyngeal cancer by sex is waning, the disparity in the incidence by race or ethnicity persists. African Americans not only have a greater risk than whites of developing oropharyngeal cancer, but they also have a much less favorable prognosis after they receive a diagnosis. Only about a third of African Americans survive 5 years after diagnosis, compared to more than half of whites (Ries et al., 2000a).
Numerous cohort and case-control studies have found that the risks of both the incidence of and the mortality from these types of cancers are significantly increased in smokers compared with those in nonsmokers, and many studies have demonstrated a dose-response relationship. The long-running cohort study of British male doctors found rates of mortality from esophageal cancer among the heaviest smokers to be 15 times that among nonsmokers (Doll et al., 1994). Among all current smokers combined, the increased rate of mortality was over seven times that among nonsmokers. In a study of U.S. veterans, current smokers experienced 10 times the risk of mortality from laryngeal cancer than those who never smoked, a risk similar to that seen in the American Cancer Society’s Cancer Prevention Study II (Kahn, 1966; US DHHS, 1990). In addition, a case-control study conducted by the National Cancer Institute found that the risk of oropharyngeal cancer was increased two to three times among those who ever smoked compared with that among those who never smoked, and the risk increased with the amount and duration of smoking. Women who smoked 40 or more cigarettes per day had six times the risk of oropharyngeal cancer as someone who never smoked. Men who smoked 40 or more cigarettes per
day had three times the risk. Overall, the increased incidence of oropharyngeal cancer in smokers compared with that in nonsmokers across studies ranges from 3 to 13 (Schottenfeld and Fraumeni, 1996).
The most likely causal pathway between tobacco use and these cancers is the direct contact of the carcinogens in tobacco and its smoke with the tissues of the oral cavity, larynx, and esophagus. Yet, although smoking alone is an independent risk factor for oral, laryngeal, and esophageal cancer, alcohol consumption greatly exacerbates smoking’s effect on risk (Blot et al., 1988). It is estimated that alcohol and tobacco use together account for approximately 75 percent of oral cancers in the United States (Blot et al., 1988).
There is convincing evidence that former smokers have lower risks of cancers of the oral cavity, pharynx, larynx, and esophagus than current smokers. For oropharyngeal cancer, the risk for former smokers compared with that for current smokers decreases steadily with the number of years since cessation (after the first few years), with some studies demonstrating that the risk of oropharyngeal cancer actually returns to that for never smokers 10 or more years after quitting (Blot et al., 1988). As with lung cancer and smoking cessation, there is also good evidence that the excess risk of oropharyngeal cancer decreases with younger age at cessation (US DHHS, 1990). For laryngeal and esophageal cancer, numerous studies have demonstrated significant drops in risk for former smokers compared with that for current smokers beginning 3 to 4 years after cessation (US DHHS, 1990).
Tobacco Use and Bladder Cancer
Approximately 56,500 cases of bladder cancer are diagnosed each year (ACS, 2002a). Whites have the highest incidence rates, about twice the rate of Hispanics and African Americans (Miller et al., 1996). Significant disparity in the incidence of bladder cancer also exists by sex, as men are about three to four times more likely to develop the disease than women (Miller et al., 1996).
There is convincing evidence that smoking is a cause of bladder cancer. More than 30 case-control and 10 prospective cohort studies support a strong link between smoking and the disease (Silverman et al., 1992). Overall, moderate to heavy smokers tend to have two to five times the risk of nonsmokers, and there is a strong dose-response relationship between smoking and the risk of bladder cancer, with the risk increasing with the duration and amount of smoking. Specifically, the 40-year analysis of British male doctors found that the rate of mortality from bladder cancer was three times greater among heavy smokers (those who smoke 25 or more cigarettes a day) than among nonsmokers (Doll et al., 1994). The most likely causal pathway between tobacco smoking and bladder cancer is the expo-
sure of bladder tissue to the carcinogenic by-products of tobacco metabolism that are excreted in urine. It is estimated that a third of all bladder cancers in women and half of all bladder cancers in men are caused by cigarette smoking (Silverman et al., 1992).
There is convincing evidence that smoking cessation can lower the risk of bladder cancer in former smokers compared with that in current smokers. A recent pooled analysis of 11 case-control studies conducted by the International Agency for Research on Cancer found that the risk of bladder cancer in former smokers compared with that in current smokers began to decrease almost immediately after smoking cessation and continued to do so with the duration of cessation (Brennan et al., 2000). The risk dropped 35 percent 1 to 4 years after quitting and dropped more than 60 percent 25 years after quitting, but it never reached that for someone who had never smoked.
Tobacco Use and Kidney Cancer
Although kidney cancer is less common than bladder cancer, there is convincing evidence from case-control and prospective cohort studies that smoking causes kidney cancer. Approximately 31,800 cases of kidney cancer are diagnosed each year (ACS, 2002a). Men are approximately twice as likely as women to develop the disease (Miller et al., 1996) African Americans, whites, and Hispanics each appear to develop kidney cancer at similar rates (Miller et al., 1996). As reviewed in the 1989 Surgeon General’s report, the relative risk for kidney cancer associated with smoking ranges from one to five and exhibits a dose-response relationship with the number of cigarettes smoked (U.S Department of Health and Human Services, 1989). More recent studies bolster these results. A large study with a cohort of U.S. veterans found that men who smoked more than 40 cigarettes a day had double the risk of kidney cancer compared with that for nonsmokers (McLaughlin et al. 1995a). The International Renal Cell Cancer Study, a large case-control study, also found that the risk increased with both the number of cigarettes smoked and the duration of smoking (McLaughlin et al. 1995b). The causal pathway is likely similar to that for bladder cancer: exposure of kidney tissue to the carcinogenic by-products of tobacco metabolism that end up in urine.
For kidney cancer (including cancer of the renal pelvis), smoking cessation does not seem to provide as much of a benefit as it does for bladder cancer. The study of U.S. veterans found only slight differences in the risk of kidney cancer between former and current smokers (Kahn, 1966), and the International Renal Cell Cancer Study found that after more than 15 years of cessation, the risk of kidney cancer in former smokers was only 15 to 25 percent lower than among current smokers (McLaughlin et al., 1995b).
Tobacco Use and Pancreatic Cancer
Pancreatic cancer is the most deadly of the major cancers, with 5-year survival rates after diagnosis only about 5 percent (ACS, 2002a). Approximately 30,300 cases are diagnosed each year in the United States (ACS, 2002a). The disease is more common in men than in women and is about 50 percent more common in African Americans than in whites (Miller et al., 1996). Smoking was described as a contributory factor to pancreatic cancer in the 1989 Surgeon General’s report, but the evidence now supports smoking as a cause of the disease (US DHHS, 1989). Both case-control and prospective cohort studies have not only linked smoking to an increased risk of pancreatic cancer but also demonstrated a dose-response relationship. Overall, smokers appear to have two to three times the risk of getting pancreatic cancer as nonsmokers. Although the causal pathway between smoking and pancreatic cancer is unknown, it has been proposed that tobacco carcinogens or their by-products cause mutations in pancreatic cells linked to carcinogenesis.
There is convincing evidence that individuals who quit smoking experience significantly decreased risks of pancreatic cancer. Unlike many other cancers linked to smoking, the risk of pancreatic cancer in former smokers begins to drop in the first couple of years after cessation (Anderson et al., 1996). A large prospective study of men found that the risk of the disease dropped nearly 50 percent within 2 years of stopping, and after less than 10 years, the risk nearly returned to that for someone who had never smoked (Fuchs et al., 1996).
Tobacco Use and Colon Cancer
The American Cancer Society estimates that there were 148,300 new cases of colorectal cancer in 2002 and that 56,600 Americans died of the disease (ACS, 2002a). An association between smoking and colorectal cancer was not discussed in previous Surgeon General’s reports on smoking and health because of a lack of data, but there is now convincing new evidence for a causal association between smoking and an increased risk of colon cancer, as well as colon polyps, among both men and women (Giovannucci and Martinez, 1996). A Swedish prospective study of nearly 57,000 men and women found a 60 percent increased risk of colon cancer in smokers compared with that in nonsmokers after 11 to 20 years of follow-up (Knekt et al., 1998). The Nurses’ Health Study and the Health Professionals Follow-Up Study demonstrated significant elevations in risk in long-term smokers as well (Giovannucci et al., 1994a,b). Men who had been smoking for at least 35 years had nearly double the risk of colon cancer compared with that for nonsmokers, as did women who had smoked for 45 years. The long latency period between the initiation of smoking and
the elevation in risk, coupled with the consistent relationship seen between smoking and colorectal polyps, suggests that tobacco may be an initiator of colorectal carcinogenesis.
There is an increasing amount of evidence that smoking cessation can lower the risk of colon cancer, although this evidence was not included in the 1990 Surgeon General’s report mentioned above. Data from the American Cancer Society’s Cancer Prevention Study II indicate that the risk of colon cancer drops with the number of years of smoking cessation as well as a younger age at the time of cessation (Chao et al., 2000).
Tobacco Use and Stomach Cancer
Stomach cancer is one of the most common cancers worldwide. Although it is much less common in the United States, nearly 21,600 cases of stomach cancer are diagnosed and there are approximately 12,400 deaths from stomach cancer each year (ACS, 2002a). In general, Asian populations in the United States have the highest rates of incidence of the disease; the exception, however, is Filipinos, who, along with whites, have the lowest rates of stomach cancer (Miller et al., 1996). African Americans and Hispanics tend to have higher rates than whites but lower rates than most Asian groups.
As early as 1982, the Surgeon General’s reports on smoking and health have documented a positive association between smoking and stomach cancer (US DHHS, 1982). Both prospective and case-control studies have found a link between the two, with a number demonstrating a dose-response relationship (Tredaniel et al., 1997). Although the results of case-control studies have been somewhat variable, prospective cohort studies have consistently found an increased risk of stomach cancer linked to smoking. One meta-analysis of 40 studies—case-control and cohort studies combined—found that, overall, smoking increased the risk of stomach cancer by 50 to 60 percent (Tredaniel et al., 1997). When men and women were assessed separately, male smokers had a greater risk of stomach cancer from smoking than female smokers. A number of possible causal pathways exist between smoking and stomach cancer. Smoking has been linked to Helicobacter pylori infection, a major risk factor for stomach cancer worldwide, as well as decreased levels in serum of certain micronutrients such as carotenoids and vitamin C that may help protect against the disease. It is estimated that 11 percent of stomach cancers worldwide can be attributed to tobacco smoking (Tredaniel et al., 1997).
A growing body of evidence supports a lower risk of stomach cancer in former smokers compared with that in current smokers. The risk of stomach cancer decreases with increasing years of cessation, with the risk nearing that for those who have never smoked after approximately 20 years of cessation (Tredaniel et al., 1997).
Tobacco Use and Cervical Cancer
Worldwide, cervical cancer is one of the most common cancers in women. In the United States, both the incidence of cervical cancer and the rate of mortality from the disease have been declining steadily since the middle of the last century. Since the early 1970s alone, the incidence and mortality for cervical cancer have dropped by approximately 40 percent (Miller et al., 1996), a decrease most likely due to increased screening by Pap tests and the use of effective treatment. Cervical cancer is now known to be caused in large part by chronic infection of the cervix by some sub-types of human papilloma virus (HPV). Cervical cancer is more common in African-American women than white women. Between 1988 and 1992, incidence rates according to the Surveillance, Epidemiology, and End Results (SEER) Program were 13.2/100,000 for African-American women and 8.7/100,000 for white women. Vietnamese women had the highest rates in the United States (43/100,000), and Japanese women had the lowest (5.8/100,000) (Miller et al., 1996).
One challenge with studying smoking and cervical cancer is potential confounding by other risk factors linked with low socioeconomic status. In particular, human papillomavirus (HPV) infection and high levels of sexual activity each increase the risk of cervical cancer and are each also more common in smokers than in nonsmokers. Separation of the effect of smoking from these other risk factors is key to uncovering the true relationship between smoking and cervical cancer.
The Surgeon General’s report Women and Smoking summarizes the evidence linking smoking with cervical cancer (US DHHS, 2001a). Overall, case-control studies that have not controlled for HPV infection status demonstrate a twofold increase in the risk of cervical cancer in smokers compared with that in those who have never smoked, and the risk increases with the duration of smoking. For women who have smoked for more than 20 years, the risk of cervical cancer is threefold that for women who have never smoked (Daling et al., 1996). Results have been mixed in studies that have controlled for HPV infection status. Some have found that smoking raises the risk of cervical cancer, regardless of HPV infection status (Ylitalo et al., 1999; Daling et al., 1996), and others have found that smoking increases the risk of cervical cancer only among women who are not infected with HPV (Bosch et al., 1992; Eluf-Neto et al., 1994; Munoz et al., 1993).
There is evidence of a probable inverse association between smoking cessation and cervical cancer. Taken as a whole, there are good data that former smokers experience a lower risk of cervical cancer than current smokers (US DHHS, 1990). It is unclear how the amount of time since quitting affects the risk of cervical cancer in former smokers.
Tobacco Use and Liver Cancer
Approximately 16,600 cases of liver cancer (including cancer of the bile ducts) are diagnosed each year in the United States (ACS, 2002a). Men have about twice the incidence rate of women, and African Americans and Hispanics have about twice the incidence rate of whites (Miller et al., 1996).
There is evidence of a probable positive association between smoking and liver cancer. Although some studies on this topic have not adequately controlled for alcohol intake and viral hepatitis infection—two key factors that can potentially confound the relationship—the evidence supports a positive relationship with cigarette smoking and suggests a dose-response association. Overall, the relative risk of liver cancer associated with smoking appears to range from 1.5 to 7, with the highest association observed among those without hepatitis (Trichopoulos et al. 1980; Lam et al., 1982; Tu et al., 1985; Trichopoulos et al., 1987; Hsing et al., 1990a; Tanaka et al., 1992; McLaughlin et al., 1995a). However, the results of all studies have not been consistent (Doll et al., 1994). There is only limited evidence that the risk of liver cancer is lower in former smokers than in current smokers (US DHHS, 1990).
Tobacco Use and Leukemia
Approximately 30,800 cases of leukemia are diagnosed each year in the United States, half of which are classified as acute leukemia and half of which are classified as chronic (ACS, 2002a). The large majority of leukemias occur in adults, and men are about 50 percent more likely to develop the disease than women. Whites have the highest rates in the United States, with certain Asian populations—Chinese, Japanese, and Koreans—having the lowest rates (Miller et al., 1996).
The current weight of evidence supports a causal association between smoking and acute leukemia, mainly of the myeloid type. A 1993 meta-analysis of 15 studies found that the data accumulated from prospective and case-control studies support relative risks of 1.3 and 1.1, respectively (Brownson et al., 1993). A dose-response was also seen with the number of cigarettes smoked. Overall, having ever smoked seems to increase the risk of leukemia by 30 to 50 percent. For smokers who smoke more than a pack a day, risk appears to increase about twofold. The chemical benzene is one of the likely causal links between cigarettes and leukemia. In experiments with both humans and animals, benzene has been shown to promote cancerous changes in white blood cells (Korte, Hertz-Picciotto et al., 2000). It has been estimated that 14 percent of all leukemia cases in the United States may be attributable to cigarette smoking (Brownson et al., 1993).
The results of studies on the benefits of smoking cessation on the risk of leukemia are mixed. The American Cancer Society’s Cancer Prevention
Study I and Cancer Prevention Study II both found small decreases in the risk of leukemia in former smokers compared with that in current smokers (Garfinkel and Boffetta, 1990), but other studies did not (Doll et al., 1994; McLaughlin et al., 1995a).
Tobacco Use and Prostate Cancer
More than 189,000 men are diagnosed with prostate cancer each year in the United States, making it the most common cancer in men (ACS, 2002a). Although studies do not currently support a link between smoking and the incidence of prostate cancer, a growing body of evidence links smoking with mortality from prostate cancer. A number of large, prospective cohort studies have documented a link between smoking and mortality from prostate cancer, with some demonstrating a dose-response relationship with the amount smoked (Hsing et al., 1990b; Hsing et al., 1991; Coughlin et al., 1996; Rodriguez et al., 1997; Giovannucci et al., 1999). Overall, relative risks for mortality from prostate cancer seem to range from 1.3 to 2.0 for smokers compared with nonsmokers. Unlike many other cancers, however, recent smoking seems to be more important than total lifetime exposure to cigarette smoke. Although the exact causal pathway between smoking and mortality from prostate cancer is unknown, it may be that the increased levels of testosterone (and other adrenal hormones) seen in the serum of smokers may stimulate the growth of prostate cancer cells (Gann et al., 1996).
Tobacco and Endometrial Cancer
Endometrial cancer is a common cancer in U.S. women, with approximately 39,300 cases diagnosed each year (ACS, 2002a). Current evidence documents that smokers have a lower risk of endometrial cancer compared with the risk for nonsmokers, most likely mediated through weight and hormone levels (US DHHS, 1989). Increased serum estrogen levels and overweight (primarily postmenopause) are linked to an increased risk of endometrial cancer. On average, smokers weigh less than nonsmokers and so have lower serum estrogen levels, which may reduce the risk of endometrial cancer. Although this one inverse association with smoking is well documented, the decreased risk of endometrial cancer is far outweighed by the increased risk of the many other more common cancers as well as other common chronic diseases.
Environmental Tobacco Smoke
In 1993 environmental tobacco smoke was categorized as a Group A carcinogen by the Environmental Protection Agency. Exposure to environ-
mental tobacco smoke significantly increases the risk of lung cancer and other diseases, such as heart disease and respiratory illness in children. One meta-analysis of 5 cohort and 34 case-control studies found that nonsmoking women who lived with a smoker had an approximately 25 percent greater risk of developing lung cancer than non-smoking women who lived with a nonsmoker (Hackshaw et al., 1997). A dose-response relationship with the number of cigarettes that the husband smoked per day was also seen. Passive exposure to tobacco smoke has also been defined as a cause of heart disease among women (US DHHS, 2001a).
Cigars and Pipes
Cigarettes are by far the most common delivery device for tobacco in the United States. However, a substantial number of people also use tobacco in pipes and cigars. Although they are seemingly less dangerous and less addictive than cigarettes, an analysis of the large cohort in the American Cancer Society’s Cancer Prevention Study II found that cigar smoking by men substantially increases their risk of lung, oral, and laryngeal cancer. Lung cancer risk was increased about fivefold, and the risk of pancreas and bladder cancers about threefold (Shapiro et al., 2000). An analysis of a large cohort of U.S. veterans found that cigar and pipe use increased the risk of liver cancer approximately threefold (Hsing et al., 1990b). In addition, pipe smoking is causally related to lip cancer and increases the risk of lung cancer, although to a lesser degree than cigarette smoking (U.S. Department of Health, Education, and Welfare, 1964; Lange et al., 1992; Boffetta et al., 1999).
Smokeless Tobacco
Although the rate of use of smokeless tobacco is generally low nationwide, it is quite common in certain subpopulations, especially young adult males of low socioeconomic status living in rural areas (Nelson et al., 1996; Bell et al., 2000). The relationship between the use of smokeless tobacco and cancer of the oral cavity was reviewed in detail in a 1986 Surgeon General’s Report, The Health Consequences of Using Smokeless Tobacco (Advisory Committee to the Surgeon General, 1986). There is convincing evidence that smokeless tobacco causes cancer of the oral cavity.
Disparities in Use of Tobacco
Nearly one-quarter (23.5 percent) of the US population are current cigarette smokers (Table 3.2). Of the 46.5 million Americans who are, the percentage of users varies by age, race, ethnicity, gender, and education (Table 3.2). Younger adults are more likely to smoke than adults 65 years
TABLE 3.2 Current Cigarette Use, Adults 18 Years or Older, United States, 1999
|
Characteristic |
% Men |
% Women |
% Total |
|
Age group (years) |
|||
|
18 to 24 |
29.5 |
26.3 |
27.9 |
|
25 to 44 |
29.6 |
25.1 |
27.3 |
|
45 to 64 |
25.8 |
21.0 |
23.3 |
|
65 or older |
10.5 |
10.7 |
10.6 |
|
Race/Ethnicity |
|||
|
White (non-Hispanic) |
25.5 |
23.1 |
24.3 |
|
Black (non-Hispanic) |
28.7 |
20.8 |
24.3 |
|
Hispanic |
24.1 |
12.3 |
18.1 |
|
American Indian/Alaska Nativea |
40.9 |
40.8 |
40.8 |
|
Asian/Pacific Islander |
24.3 |
7.1 |
15.1 |
|
Education (Years)b |
|||
|
8 or fewer |
24.7 |
12.8 |
18.3 |
|
9 to 11 |
42.4 |
33.5 |
37.7 |
|
12 |
30.2 |
23.2 |
26.3 |
|
13 to 15 |
27.6 |
23.3 |
25.3 |
|
16 |
14.0 |
11.9 |
13.0 |
|
More than 16 |
9.1 |
7.8 |
8.5 |
|
Total |
25.7 |
21.5 |
23.5 |
|
aEstimates should be interpreted with caution because of the small sample sizes. bPersons aged 25 years or older. NOTE: Data from the National Health Interview Survey, 1999. (Cigarette use is defined as having reported smoking at least 100 cigarettes during a lifetime or having smoked every day or some days at the time of the interview.) SOURCE: ACS, 2002b. |
|||
or older. More than a third of high school students are current smokers (Table 3.3). Although cigarette smoking increased among high school students in the 1990s and is the most prevalent form of tobacco use among this age group, smokeless tobacco and cigars are becoming more commonly used (Table 3.3) (ACS, 2002b).
Among racial and ethnic groups, American Indians and Alaska Natives are most likely to use cigarettes (41 percent) and Asian and Pacific Islanders are the least likely to use cigarettes (15 percent). Whites, African Americans, and Hispanics fell in between and had similar smoking rates (approximately 18–24 percent). Once quite pronounced, the disparity in smoking rates by sex is now relatively small. In 1999, 26 percent of men were current smokers, as compared to 21 percent of women (Table 3.2) (ACS, 2002b).
Smoking rates are also closely tied to income. By both sex and race or ethnicity, people with lower incomes are more likely to smoke than people
TABLE 3.3 Tobacco Use, High School Students, United States, 1999
|
|
Cigarette use |
|||
|
Characteristic |
Currenta % |
Frequentb % |
Current Smokeless Tobaccoc % |
Current Cigar Used % |
|
Gender |
||||
|
Male |
34.7 |
17.9 |
14.2 |
25.4 |
|
Female |
34.9 |
15.6 |
1.3 |
9.9 |
|
Race/Ethnicity |
||||
|
White, non-Hispanic |
38.6 |
20.2 |
10.4 |
18.8 |
|
Male |
38.2 |
20.9 |
18.8 |
28.3 |
|
Female |
39.1 |
19.4 |
1.5 |
8.6 |
|
Black, non-Hispanic |
19.7 |
7.0 |
1.3 |
13.7 |
|
Male |
21.8 |
9.1 |
2.5 |
16.0 |
|
Female |
17.7 |
5.0 |
0.2 |
11.6 |
|
Hispanic |
32.7 |
10.4 |
3.9 |
16.7 |
|
Male |
34.0 |
12.5 |
6.1 |
21.9 |
|
Female |
31.5 |
8.5 |
1.8 |
11.6 |
|
Grade |
||||
|
9 |
27.6 |
11.2 |
6.8 |
13.7 |
|
10 |
34.7 |
15.2 |
7.1 |
17.8 |
|
11 |
36.0 |
18.7 |
8.4 |
18.2 |
|
12 |
42.8 |
23.1 |
8.9 |
22.0 |
|
Total |
34.8 |
16.8 |
7.8 |
17.7 |
|
aSmoked cigarettes on one or more of the 30 days preceding the survey. bSmoked cigarettes on 20 or more of the 30 days preceding the survey. cUsed chewing tobacco or snuff on one or more of the 30 days preceding the survey. dSmoked cigars on one or more of the 30 days preceding the survey. NOTE: Data from the Youth Risk Behavior Surveillance System, 1999. (A current user has smoked cigarettes, cigars, or used tobacco respectively, on one or more of the 30 days preceding the survey. A frequent smoker has smoked at least 20 of the 30 days preceding the survey.) SOURCE: ACS, 2002b. |
||||
with higher incomes (National Center for Health Statistics, 1998). Low-income African-American men and women are about twice as likely to be smokers as African Americans with middle or high incomes. A similar, although slightly lower, disparity in smoking rates exists between income groups among whites. In addition to income level, overall socioeconomic status is associated with smoking among adults, adolescents, and children (Choiniere et al., 2000; Lewis et al., 2001; Lowry et al., 1996; Ross, 2000; Samet et al., 1992; Zhu et al., 1996; Whitlock et al., 1997b; Sussman and Dent, 2000). This relationship is especially apparent among vulnerable populations. For example, pregnant women of lower socioeconomic status are much more likely (3.7 times) to smoke than their counterparts of higher
socioeconomic status (King et al., 1993b; Najman et al., 1998). When education level alone is examined, individuals with only 9 to 11 years of education are about three times more likely to smoke than those with a college education (Table 3.2).
Occupation is strongly linked to rates of smoking as well, even after controlling for age, income, gender, and race or ethnicity (Nelson et al., 1994; Leigh, 1996; Bang and Kim, 2001). According to analyses of smoking by occupation and industry using the third National Health and Nutrition Examination Survey (1988 to 1994), the prevalence of cigarette smoking was highest among material-moving occupations, construction laborers, and vehicle mechanics and repairers. The lowest smoking prevalence was found among teachers. Among industry groups, the construction industry had the highest prevalence of cigarette smoking (Bang and Kim, 2001). Although disparities by occupation have persisted for many years, recent data suggest that the gap in smoking rates may be widening between blue-collar workers and white-collar workers (Nelson et al., 1994). Blue-collar workers tend to be heavier smokers and, therefore, more nicotine-dependent (Covey et al., 1992; Bang and Kim, 2001). Data from the 1997 National Health Interview Survey show that 27.5 percent of blue-collar smokers smoke 25 or more cigarettes per day, whereas only 18 percent of white-collar smokers do (Bang and Kim, 2001).
As with tobacco use, there are disparities in smoking cessation by race or ethnicity as well as other socio-demographic characteristics. When the disparity is assessed by race or ethnicity, whites have a higher rate of smoking cessation than African Americans. The 1989 Behavioral Risk Factor Surveillance System found that whites had a quit rate of 47 percent and African Americans had a quit rate of 39 percent (US DHHS, 1998). In 1994–1995, the percentage of adult African-American ever smokers who had quit was 35.4 percent (US DHHS, 1998). These numbers, however, do not reflect an aversion to quitting. The 1999 Behavioral Risk Factor Surveillance System found that about 59 percent of African Americans and Hispanics had quit for at least 1 day in the previous 12 months, as compared to 49 percent of whites (CDC, Division of Adult and Community Health, 1999). That African Americans have a harder time successfully quitting smoking may reflect a number of issues, such as a lack of access to appropriate smoking cessation programs as well as heightened nicotine dependence (US DHHS, 1990; Caraballo et al., 1998).
Education level is closely tied to rates of cessation of tobacco use. Typically, the higher the level of education that is attained, the higher the rate of smoking cessation (US DHHS, 1990). The quit rate for individuals with less than 12 years of education was 35 percent; for those with 12 years of education, it was 38 percent; for those with 13 to 15 years of education, it was 44 percent; and for those with 16 or more years of education, it was 57 percent.
PHYSICAL ACTIVITY
Physical activity has numerous mental and physical health benefits, including reductions in the risks of premature mortality, cardiovascular diseases, hypertension, diabetes, depression, osteoporosis, and cancer (US DHHS, 1996). Important health benefits include a reduced risk of colon, breast, and endometrial cancer (Table 3.4). In addition, physical activity is an important complement to dietary management for the avoidance of weight gain, and is essential for weight maintenance after intentional weight loss (National Heart, Lung, and Blood Institute, 1998). The International Agency for Research on Cancer (IARC) has estimated that at least 11 to 15 percent of breast and colorectal cancer may be attributable to inadequate physical activity (International Agency for Research on Cancer, 2002). Overall, sedentary lifestyles have been linked to 23 percent of deaths from major chronic diseases (Hahn et al., 1990), including 5 percent of deaths from cancer (Colditz et al., 1996). Fortunately, the negative effects of a sedentary lifestyle are reversible: evidence shows that increasing one’s level of physical activity, even after years of inactivity, can reduce the risk of mortality (Paffenbarger et al., 1993). In addition, the level of activity necessary to reduce risk is not necessarily vigorous. Moderate activity, for example, walking briskly for 30 minutes a day, will lower an individual’s risk of premature death, heart disease, stroke, diabetes, and colon cancer (US DHHS, 1996; Hu et al., 1999; Manson et al., 1999; Hu et al., 2000; International Agency for Research on Cancer, 2002). Because more than half of American adults do not participate in moderate or vigorous leisure-time physical activity (40 percent are not active at all) and because physical activity levels continue to decline in the United States (ACS, 2002b; US DHHS, 1996), the message that significant health benefits are achievable with moderate amounts of physical activity is especially important.
A large number of epidemiological studies have examined the relationship between physical activity and various types of cancers (US DHHS, 1996; McTiernan, et al. 1998; Marrett et al., 2000; International Agency
TABLE 3.4 Reduction in Risk of Incident Cancer Associated with Physical Activity
|
|
Relative Risk (RR) |
|
|
Level of Evidence |
Small (RR 0.76–0.90) |
Moderate (RR 0.51–0.75) |
|
Convincing |
Breast |
Colon |
|
Probable |
Endometrium |
|
|
Possible |
|
|
for research on Cancer, 2002). Over time, the designs of these studies have become progressively more sophisticated in terms of the measurement of physical activity and accounting for potential confounding factors. The studies summarized below were able to remove people with preexisting disease from the analysis and then examine the effect of physical activity over time in healthy populations.
Physical Activity and Colon Cancer
Among both men and women, high levels of physical activity may decrease the risk of colon cancer by as much as 50 percent (Colditz et al., 1997; Marrett et al., 2000). Although studies have not consistently used a standard measure of activity or similarly defined a “high” level of activity, a dose-response relationship between physical activity and colon cancer has consistently been observed across studies with a variety of designs and in many different populations (Garabrant et al., 1984; Slattery et al., 1988; Gerhardsson de Verdier et al., 1990; Whittemore et al., 1990; Giovannucci et al., 1995a; Martinez et al., 1997). In addition, physical activity appears to lower the risk of large adenomatous polyps, which suggests that it may act early in the adenoma-carcinoma sequence (Giovannucci et al., 1995a; Giovannucci et al., 1996; Kahn et al., 1998). The relation is seen across levels of obesity, suggesting a benefit of physical activity for reduction in cancer incidence in addition to weight reduction.
Maintaining high levels of physical activity throughout life appears to impart the greatest protection (Lee et al., 1989; Kune et al., 1990). In a study of Harvard alumni, men who were at least moderately active at two assessments were 48 percent less likely to develop colon cancer than men who were inactive at both assessments (Lee et al., 1991). However, this does not mean that those who have been sedentary in the past cannot reap the benefits if they become active. The same study showed that among men who were sedentary at the initial assessment, those who increased their activity during the 11- to 15-year follow-up period were 13 percent less likely to develop colon cancer than those who remained sedentary during the same time period (Lee et al., 1991). Data from at least two prospective studies indicated that both men and women can lower their risk of colon cancer simply by engaging in moderate physical activity such as brisk walking or stair climbing for an hour a day (Giovannucci et al., 1995a; Martinez et al., 1997). In those studies the risk decreased with increasing walking pace among those whose only recreational activity was walking.
Several mechanisms have been proposed to explain this. Physical activity may decrease gastrointestinal transit time, thereby minimizing contact of the gastrointestinal tract with potential carcinogens in the stool (McTiernan et al., 1998), and it may also reduce circulating levels of insulin, a growth factor for colonic epithelial cells (McKeown-Eyssen, 1994; Gio-
vannucci, 1995b). Additional hypotheses suggest that physical activity alters prostaglandin levels, improves immune function, and modifies bile acid metabolism (Martinez et al., 1999).
Physical Activity and Breast Cancer
A woman’s risk of breast cancer depends largely on the amount of estrogen circulating in her body (Willett et al., 2000; Hankinson et al., 1995). Because physical activity is thought to lower the cumulative level of lifetime exposure to circulating estrogens, researchers have long speculated that it might lower the risk of breast cancer. Numerous studies have examined this possibility, and although their results are not entirely consistent, the majority of studies support an inverse relationship between physical activity and breast cancer (Gammon et al., 1998; McTiernan et al., 1998; Marrett et al., 2000). Just as with colon cancer, the physical activity benefit for breast cancer is mediated both by its effect on body weight and by other mechanisms independent of bodyweight.
Physical activity in childhood may also affect breast cancer risk (Frisch et al., 1985; Bernstein et al., 1994; Colditz and Frazier, 1995). By interacting with the adrenaline system to reduce levels of circulating estrogens, physical activity may result in delayed menarche or a delay in the onset of regular ovulatory menstrual cycles, factors known to be associated with lower lifelong risk for breast cancer (Willett et al., 2000).
Physical Activity and Lung Cancer
Several studies have examined the impact of physical activity on lung cancer risk, and most have suggested a protective effect (World Cancer Research Fund and American Institute for Cancer Research, 1997). Researchers cannot completely control for the confounding effects of cigarette smoking, however. Since active people are less likely to smoke than inactive people and nonsmokers are far less likely to develop lung cancer than smokers, it is not clear whether active people are at lower risk of the disease because they are physically active or because they do not smoke. Most studies have attempted to control for smoking, but the potential for residual confounding cannot be ruled out, as smoking is such a strong causal factor in lung cancer and is associated with many health behaviors, including physical activity (International Agency for Research on Cancer, 2002).
Physical Activity and Prostate Cancer
As with breast cancer, physical activity has been hypothesized to lower the risk of prostate cancer by altering hormone levels. Despite this clear biological hypothesis and a number of studies on the topic, the relationship
between physical activity and prostate cancer has remained unclear (World Cancer Research Fund and American Institute for Cancer Research, 1997; International Agency for Research on Cancer, 2002). Some studies have produced null results, whereas others report an inverse association and still others a positive association. Of the studies that have reported an inverse association, most have shown only a modest protective effect and no dose-response (Paffenbarger et al., 1987; Albanes et al., 1989; Paffenbarger et al., 1992). In addition, several of these studies showed significant relationships only among certain subgroups of men, defined by either age (Lee et al., 1992; Oliveria et al., 1996) or very high levels of activity (Lee et al., 1992; Giovannucci et al., 1998a).
OBESITY
Excess body fat causes numerous medical conditions, including cardiovascular diseases and some major cancers (U. S. Department of Agriculture and U. S. Department of Health and Human Services, 1995; International Agency for Research on Cancer, 2002), and is a growing problem in many countries. A linear relation exists between adiposity and most health conditions, although for mortality the shape of the relations has been debated, in part because of excess mortality among the leanest individuals. This relation between mortality and leanness is confounded in part by cigarette smoking and in part by reverse causation: the major illnesses such as cancer that predispose an individual to death first lead to weight loss and the artifactual appearance of increased mortality risk among lean individuals.
Consistent with the mortality burden described above, the study of cancer incidence shows many of the relations relating increasing adiposity or obesity to the onset of cancer (Table 3.5). Follow-up of 28,129 men and women in Sweden showed a 33 percent excess risk of cancer among obese individuals than among nonobese individuals (Wolk et al., 2001). Obesity contributes to the incidence of cancers through several possible mecha-
TABLE 3.5 Increase in Risk of Incident Cancer Associated with Obesity
|
|
Relative Risk (RR) |
|
|
Level of Evidence |
Moderate (RR 1.35–1.99) |
Large (RR 2.0+) |
|
Convincing |
Colon |
Breast Endometrial Kidney Esophageal |
|
Possible |
Prostate (mortality) |
|
nisms. These may include female hormones for breast and uterine cancer, insulin pathways for colon cancer, and various other explanations for cancer at other sites. The IARC estimates that about 10 percent of breast cancer and colorectal cancer may be attributable to overweight and obesity, and between 25 and 40 percent of kidney, esophageal, and endometrial cancer may be attributed to obesity.
Obesity and Breast Cancer
Consistent relationships have been seen between obesity and postmenopausal breast cancer, reflecting the role of adipose cells in the production of estrogen after menopause (Hunter and Willett, 1993; Hankinson et al., 1995; World Cancer Research Fund and American Institute for Cancer Research, 1997). The strongest relation may be seen among women who have never used postmenopausal hormones (Huang et al., 1997). This may be due in part to the predisposition of lean women and those with low circulating hormone levels to use postmenopausal hormones at the time of menopause, or it may be due to the fact that elevations in estrogen, regardless of the source, increase breast cancer risk similarly. Obesity has been associated with reduced risk for premenopausal breast cancer (International Agency for Research on Cancer, 2002), a relationship likely due to the adverse effects of obesity on ovarian function in premenopausal women.
Obesity and Endometrial Cancer
Women who are overweight are at increased risk of developing endometrial cancer. The risk is two to three times higher in obese women than in lean women, and the risk is more clearly evident in older women (Tornberg and Carstensen, 1994). This association is likely due to higher circulating estrogen levels after menopause among women who are obese.
Obesity and Prostate Cancer
The evidence for an association between obesity and prostate cancer is quite mixed. Although obesity may not increase risk of prostate cancer incidence, there is some evidence to suggest a higher risk of death after the diagnosis of prostate cancer among obese men (Andersson et al., 1997). Obesity may predispose an individual to more aggressive tumors or, alternatively, may stimulate growth or metastases of prostate cancer after it has developed. Several possible mechanisms through which obesity may influence the prostate gland involve hormones, including the insulin-like growth factor pathway, insulin itself, and, potentially, androgens.
SOURCE: Corbis Corporation.
Obesity and Kidney Cancer
Obesity is directly related to the risk of kidney cancer among both men and women (McLaughlin et al., 1996). Virtually every study that has examined this relation has observed a positive association, although it is usually stronger among women than among men. The mechanism for the effect of obesity on the risk of kidney cancer is not clear, though insulin or insulin-like growth factors may be involved.
Obesity and Colon Cancer
Obesity and central adiposity have been related to colon cancer in numerous studies. The association is seen in both sexes, although it has usually been observed to be weaker among women. The mechanism by which obesity contributes to the development of colon cancer has not been fully documented. However, strong evidence supports a relation between excess weight, glycemic control, and colon cancer. Obesity is related to increased insulin resistance and hyperinsulinemia, which in turn are related to proliferation of colon cells in laboratory studies (McKeown-Eyssen, 1994; Giovannucci, 1995b; Kono et al., 1998). Further evidence supporting this mechanism comes from studies of the insulin-like growth factor pathway, which have demonstrated that insulin-like growth factor is directly related to colon cancer risk (Ma et al., 1999). Higher levels of estrogen in obese women after menopause may serve to lower colon cancer risk somewhat, which could explain the weaker association with obesity among women.
Obesity and Esophageal Cancer
Obesity is a known cause of gastroesophageal reflux, a risk factor for esophageal adenocarcinoma. In a population-based case-control study performed in Sweden, a strong direct relation between obesity and esophageal cancer was observed, with a relative risk of esophageal cancer of 16 for obese adults compared with the risk for lean adults. Other studies have observed relative risks of about 3–5 (Brown et al., 1995; Vaughan et al., 1995; Ji et al., 1997; Chow et al., 1998).
Trends in Obesity
In the United States, the age-adjusted prevalence of obesity increased from 15 percent in the late 1970s to 27 percent in 1999. The most recent estimates (1999) from the National Health and Nutrition Examination Survey (NHANES) show that 61 percent of the adult population (20–74 years) exceed the healthy weight range (BMI > 25 kg/m2 ), 34 percent are overweight (BMI 25–29 kg/m2), and 27 percent are obese (BMI = 30 kg/m2) (Figure 3.1) (US DHHS, 2001b).
According to the NHANES III (1988–1994) men are more likely than women to be either overweight or obese (59 vs. 51 percent), but women are
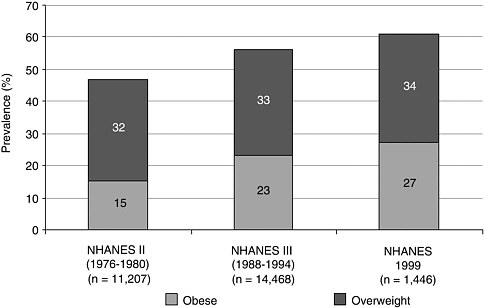
FIGURE 3.1 Age-adjusted prevalence of overweight (BMI 25–29 kg/m2) and obesity (BMI = 30 kg/m2) in U.S. adults aged 20–74.
SOURCE: US DHHS, 2001b.
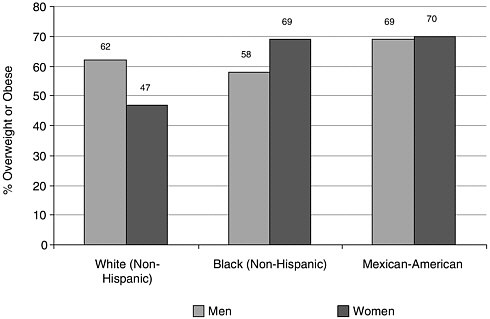
FIGURE 3.2 Age-adjusted prevalence of overweight or obesity (BMI = 25 kg/m2) by sex and race/ethnicity.
SOURCE: US DHHS, 2001b.
more likely than men to be obese (25 vs. 20 percent) (Flegal et al., 1998; ACS, 2001).
Obesity or overweight is higher among non-Hispanic African-American women and Mexican-American women than among non-Hispanic white women. Among men, the prevalence of obesity or overweight is somewhat higher among Mexican-Americans (Figure 3.2).
Setting Weight Guidelines
One essential part of effective policy planning is basing action on consistent messages. Yet, despite solid epidemiological evidence that weight affects the risk of cancer and other chronic diseases—including heart disease, stroke, and diabetes—setting weight guidelines has long been problematic, and recommendations have varied over time.
BMI is used in all these guidelines as a measure of adiposity. It is calculated as weight divided by height squared. Growing evidence suggests that BMI reflects adiposity well through middle age but may be less clearly related to adiposity at older ages, when lean muscle mass may decrease and mass is redistributed to the abdomen.
Debate continues regarding the possible use of waist circumference or waist circumference-to-hip-circumference ratio as a measure of adiposity.
Neither is yet standard clinical practice, and use of a ratio of two measures (the waist circumference-to-hip-circumference ratio) has additional problems, as it includes more complex measurements and the possibility of errors in both measures. “Girth grow,” or increasing waist circumference with age, appears to predict the risk of diabetes, heart disease, and stroke.
In setting the 1995 weight guidelines (U.S. Department of Agriculture and US DHHS, 1995), the Dietary Guidelines Advisory Committee concluded that mortality risk increased significantly among persons with BMIs of 25 kg/m2 or higher (Lee and Paffenbarger, 1992; Rimm et al., 1995; Willett et al., 1995), whereas a linear increase in the risk of being diagnosed with diabetes, hypertension, and coronary heart disease was observed with increasing BMI, even among individuals with BMIs well below 25 kg/m2 (Chan et al., 1994; Colditz et al., 1995; Willett et al., 1995). Because of the importance of total mortality as a summary measure of the public health impact of obesity, the Dietary Guidelines Advisory Committee concluded that a BMI of 25 kg/m2 represents a reasonable upper limit for healthy weight. This cutoff point is consistent with those recommended by a steering committee of the American Institute of Nutrition (Kuller et al., 1993) and an expert committee of the World Health Organization (World Health Organization, 1995).
DIET
There has been an explosion of prospective studies on diet and chronic disease in recent years, and this has greatly furthered understanding of the etiology of cancer (Willett, 1998). Building on international correlation studies and retrospective case-control studies, prospective cohort studies offer the potential to evaluate diet-disease relationships free from recall bias and to correct for measurement error. This section summarizes major areas of advance according to components of the American diet.
Fruits and Vegetables
The strongest evidence of a relationship between diet and cancer has long been related to the benefit of the consumption of least five servings of fruits and vegetables per day. The majority of this evidence has arisen from case-control studies, while the prospective data have been less consistent (Table 3.6). Fruits and vegetables lower the risk of hypertension, coronary heart disease, and ischemic stroke, in addition to containing vitamins and minerals that may protect against cancer and a host of other conditions (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Numerous mechanisms have been proposed to explain the possible
TABLE 3.6 Reduction in Risk of Incident Cancer Associated with Fruit and Vegetable Intake
|
|
Relative Risk (RR) |
||
|
Level of Evidence |
Small (RR 0.76–0.90) |
Moderate (RR 0.51–0.75) |
Large (RR < 0.50) |
|
Convincing |
|
|
|
|
Probable |
|
Pancreas Bladder Lung Colon |
Mouth Pharynx Larynx Esophagus Stomach |
|
Possible |
Breast Prostate Endometrium Ovary |
Thyroid Cervical Liver Kidney |
|
inverse relationship between fruit and vegetable consumption and the risk of various cancers. Most have focused on the effects of specific agents contained in fruits and vegetables, such as carotenoids, selenium, folic acid, fiber, and vitamins C and E. However, Steinmetz and Potter (1991) have suggested that fruits and vegetables contain an anticarcinogenic cocktail of substances, including both recognized nutrients and nonnutritive constituents. Together, these substances inhibit the formation of carcinogens, act as substrates for the endogenous production of anticarcinogens, reduce the capacity of transformed cells to proliferate, and act as antioxidants.
Fruit and Vegetable Consumption and Cancers of the Mouth, Pharynx, Larynx, and Esophagus
Cancers of the mouth, pharnyx, larynx, and esophagus are caused primarily by alcohol and tobacco smoking. To a lesser extent, they may also be influenced by fruit and vegetable consumption. Most of the evidence for this comes from case-control studies, which have consistently shown a moderate to large reduction in risk among those who consume at least one serving of fruits and vegetables a day (World Cancer Research Fund and American Institute for Cancer Research, 1997). Very few prospective data support these findings, however; and the possibility of residual confounding by tobacco and alcohol use and/or recall bias cannot be eliminated.
Fruit and Vegetable Consumption and Stomach Cancer
Of the many cancers that have been studied in association with fruit and vegetable consumption, stomach cancer has been examined most ex-
tensively. More than 30 case-control studies have been conducted, with the majority supporting an inverse relationship (World Cancer Research Fund and American Institute for Cancer Research, 1997). However, as with most cancers, the data from prospective studies are less consistent. Although 8 of the 11 prospective studies suggested that the consumption of fruits and vegetables (either total intake or specific groups) might protect against stomach cancer (Chyou et al., 1990; Hirayama, 1990; Nomura et al., 1990; Zheng et al., 1995; Galanis et al., 1998; Terry et al., 1998; Botterweck et al., 1998; Dorant et al., 1996), only 5 reported a statistically significant association (Chyou et al., 1990; Hirayama, 1990; Galanis et al., 1998; Terry et al., 1998; Dorant et al., 1996). Two additional studies reported null findings (Kneller et al., 1991; Guo et al., 1994), and one suggested a nonsignificant positive relationship (Kato et al., 1992).
Fruit and Vegetable Consumption and Pancreatic Cancer
Of the 15 case-control studies that have been conducted on fruit and vegetable consumption and the risk of pancreatic cancer, 14 have provided evidence of an inverse association (Ohba et al., 1996; World Cancer Research Fund and American Institute for Cancer Research,1997; Mori et al., 1999). These studies, including those that controlled for smoking, consistently suggest a moderate protective effect for both fruits and vegetables. However, data from the four cohort studies on the topic have been far less consistent (Howe and Burch, 1996; World Cancer Research Fund and American Institute for Cancer Research, 1997). Although none of the findings have been statistically significant, they have suggested both inverse and positive associations (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Although case-control studies typically yield less conclusive evidence than cohort studies because of the greater potential for recall bias and residual confounding, cohort studies on this topic are also limited because of the rarity of the disease. Three of the four cohort studies on fruit and vegetable consumption and pancreatic cancer included only 40 to 65 incident cases or deaths, and so their results must be interpreted cautiously (Howe and Burch, 1996).
Fruit and Vegetable Consumption and Bladder Cancer
There is evidence of a probable inverse association between fruit and vegetable consumption and the risk of bladder cancer. Nearly all studies on the topic have adequately controlled for smoking, the primary cause of bladder cancer, and most suggest that both fruits and vegetables offer some degree of protection against bladder cancer (World Cancer Research Fund and American Institute for Cancer Research, 1997).
A recent meta-analysis suggests that fruits might have a greater impact on risk than vegetables (Steinmaus et al., 2000). In that analysis, the risk of bladder cancer was increased 40 percent among those with low levels of consumption of fruits and 16 percent among those with low levels of consumption of vegetables.
Fruit and Vegetable Consumption and Lung Cancer
An inverse association between high levels of consumption of fruits and vegetables and the risk of lung cancer has been observed in both case-control studies and large cohort studies (World Cancer Research Fund and American Institute for Cancer Research, 1997). Most cohort studies have shown, after controlling for smoking status, that a high level of consumption of fruits and vegetables is associated with substantially lower risk of lung cancer (World Cancer Research Fund and American Institute for Cancer Research, 1997; Yong et al., 1997; Knekt et al., 1999; Voorrips et al., 2000b). However, one analysis, which combines data from two large cohort studies, suggests only a weak association (Feskanich et al., 2000).
Much effort has been expended to identify the specific components of fruits and vegetables that might be responsible for the possible reduction in lung cancer risk. The majority of studies have focused on provitamin A carotenoids, particularly beta-carotene, because of their antioxidant properties and the importance of vitamin A in cell differentiation. However, clinical trials of high-dose beta-carotene supplements failed to produce the expected reduction in lung cancer risk (Alpha-Tocopherol Beta-Carotene Prevention Study Group, 1994; Hennekens et al., 1996; Omenn et al., 1996).
Fruit and Vegetable Consumption and Breast Cancer
The relationship between fruit and vegetable consumption and the risk of breast cancer has been evaluated extensively in case-control studies, with the majority suggesting either no association or a modest inverse association. However, in a large reanalysis combining the primary data from eight cohort studies, Smith-Warner et al. (2001) found no association between total levels of fruit and vegetable consumption and the risk of breast cancer. Similar null results were observed both for individual fruits and vegetables and for various botanical groups.
Fruit and Vegetable Consumption and Colon Cancer
The relationship between fruit and vegetable consumption and the risk of colon cancer has been examined in many case-control studies, most of which have suggested a protective effect for higher levels of
vegetable intake (World Cancer Research Fund and American Institute for Cancer Research, 1997). In those studies, green vegetables and cruciferous vegetables (any of a family of vegetables including cabbage, turnip, and mustard) seemed particularly beneficial. However, the prospective cohort studies to examine this relationship have yielded less conclusive findings (Phillips and Snowdon, 1985; Singh and Fraser, 1998; Thun et al., 1992; Voorrips et al., 2000a).
In one study with a large cohort, Thun and colleagues (1992) compared the risk of fatal colon cancer among those in the quintile with the highest levels of vegetable consumption and those in the quintile with the lowest levels of vegetable consumption. Statistically significant reductions were observed for women but not men. In another prospective study, overall fruit and vegetable intake was weakly associated with colon cancer risk, but raw vegetables, green leafy vegetables, and cruciferous vegetables appeared to be more protective for both men and women (Steinmetz et al., 1994). Michels and colleagues (2000) combined data from two large cohort studies and observed no association between overall levels of fruit and vegetable consumption and the risk of colon cancer. Similar null findings were observed for various categories of fruits and vegetables, including green leafy vegetables, citrus fruits, and cruciferous vegetables.
Vegetables contain so many beneficial vitamins and nutrients that it is difficult to identify which ones might be responsible for the possible association with colon cancer. Fiber has received the most attention so far, mainly because the proposed mechanism is so intuitive: fiber can help push waste through the colon more quickly, so that the colon is less likely to come into contact with any carcinogens. However, data from two randomized controlled trials recently failed to support a link between increased fiber intake over three years and the risk of recurrence of colon polyps, the precursor lesion for colon cancer (Alberts et al., 2000; Schatzkin et al., 2000).
Fruit and Vegetable Consumption and Ovarian Cancer
A high level of consumption of vegetables may reduce the risk of ovarian cancer, although few studies have addressed the issue (World Cancer Research Fund and American Institute for Cancer Research, 1997). In the only two cohort studies conducted on this topic to date, researchers did not observe an association between the overall levels of fruit and vegetable consumption and the risk of ovarian cancer (Kushi et al., 1999; Fairfield et al., 2001). In both of those studies, however, and in a large case-control study (Risch et al., 1994), there was a suggestion of modest reduction in risk with higher levels of vegetable consumption.
Fruit and Vegetable Consumption and Endometrial Cancer
The association between fruit and vegetable consumption and the risk of endometrial cancer has been evaluated in several case-control studies, with results suggesting the possibility of a modest inverse association (World Cancer Research Fund and American Institute for Cancer Research, 1997). In the only cohort study to address this relationship to date, researchers found the suggestion of an increased risk of endometrial cancer among those with very low levels of vegetable consumption (Terry et al., 1999).
Fruit and Vegetable Consumption and Prostate Cancer
Although the effects of fruit and vegetable consumption on the risk of prostate cancer have been examined in nearly 20 studies, the data are quite inconsistent. The majority of studies suggest that overall levels of fruit and vegetable consumption have little effect, if any, on the risk of prostate cancer. However, individual fruits and vegetables may provide a greater reduction of risk, with tomatoes being the most promising (Giovannucci, 1999). Four cohort studies have reported on tomato consumption and prostate cancer risk, and all of them demonstrated a 40 to 50 percent reduction in risk among men who consumed large amounts of tomatoes. The carotenoid lycopene is hypothesized to be responsible for a protective effect.
Fruit and Vegetable Consumption and Thyroid Cancer
Several case-control studies have reported an inverse association between fruit and vegetable consumption and the risk of thyroid cancer (World Cancer Research Fund and American Institute for Cancer Research, 1997). In all of the studies, cruciferous vegetables appeared to be particularly beneficial. However, this has not been examined in cohort studies, given the rarity of the disease.
Fruit and Vegetable Consumption and Cervical Cancer
There is evidence of a possible inverse relationship between fruit and vegetable consumption and the risk of cervical cancer. Of the five studies published on the topic, four reported a reduced risk with one or more measures of fruit and vegetable intake (World Cancer Research Fund and American Institute for Cancer Research, 1997). Despite this consistency, few conclusions can be drawn about the relationship because of the potential for confounding. Most of the identified risk factors for cervical cancer—including human papillomavirus infection, cigarette smoking, and infrequent use of Pap screening—are closely related to socioeconomic status,
which is in turn related to dietary intake (Potischman and Brinton, 1996; World Cancer Research Fund and American Institute for Cancer Research, 1997).
Fruit and Vegetable Consumption and Liver Cancer
A high level of consumption of vegetables may lower the risk of liver cancer. In the two cohort studies that have examined this relationship, one demonstrated a sevenfold increase in risk among those who consumed small amounts of fresh vegetables, and the other suggested a protective effect for green-yellow vegetables. Five case-control studies have also been conducted on the topic, three of which suggested a moderate to large inverse association with the consumption of certain types of vegetables.
Fruit and Vegetable Consumption and Kidney Cancer
A number of case-control studies have demonstrated an inverse association between fruit and vegetable consumption and the risk of kidney cancer (Mellemgaard et al., 1996; Wolk et al., 1996; Lindblad et al., 1997; World Cancer Research Fund and American Institute for Cancer Research, 1997; Yuan et al., 1998). In the largest of these studies, the International Renal Cell Cancer Study, Wolk and colleagues (1996) reported a protective effect for several groups of fruits and vegetables, with the strongest association observed for orange and dark green vegetables.
Red Meat
The levels of consumption of red meats such as beef, pork, and lamb have declined dramatically in the United States in the past 40 years, in part because of increasing concern about the effects of meat on health. Although red meat is an excellent source of protein and iron, it also contains large amounts of saturated fat, which can raise the level of cholesterol in the blood. In addition, when cooked at high temperatures, high-protein foods such as red meat may generate carcinogens. Evidence linking the consumption of red meat to a higher incidence of cancer is not yet convincing (Table 3.7).
Red Meat Consumption and Colorectal Cancer
There is probable evidence that the consumption of large amounts of red meat increases the risk of colorectal cancer among both men and women. Although not entirely consistent, numerous studies with both cohort and case-control designs have reported a positive association between the consumption of red meat and colon cancer (World Cancer Research
TABLE 3.7 Increase in Risk of Incident Cancer Associated with High Intakes of Red Meat
|
|
Relative Risk (RR) |
|
|
Level of Evidence |
Moderate (RR 1.35–1.99) |
Large (RR =2.0) |
|
Convincing |
|
|
|
Probable |
|
Colon and rectum |
|
Possible |
Kidney Prostate Pancreas |
|
Fund and American Institute for Cancer Research, 1997). For most studies showing an association, the median relative risk between the categories of high and low levels of red meat consumption is about 2, with the majority falling between 1.5 and 2.5 (Giovannucci and Goldin, 1997).
Several mechanisms have been proposed to explain the relationship between red meat consumption and colon cancer. First, the specific fatty acid content of red meat may be particularly harmful (Giovannucci, 1995a). However, the fatty acids in beef are not unique and overlap substantially with those in dairy fat, which is not related to colon cancer. Second, fat from red meat may be less readily digested or absorbed in the small intestine than fat from other sources; therefore, more of it may reach the large bowel (Giovannucci and Willett, 1994). Third, initiators or promoters may be formed when red meat is cooked, particularly at high temperatures (Schiffman et al., 1989; Gerhardsson de Verdier et al., 1991). Finally, high levels of consumption of red meat may increase the concentrations of fecal iron, which could influence the risk of colon cancer via the generation of hydroxyl radicals (Giovannucci and Willett, 1994; Sesink et al., 1999; Sesink et al., 2000).
Red Meat Consumption and Prostate Cancer
The relationship between red meat consumption and the risk of prostate cancer has been examined in a number of cohort and case-control studies, with results suggesting a positive association (World Cancer Research Fund and American Institute for Cancer Research, 1997). In the largest cohort study to address this issue, the risk of advanced prostate cancer was more than twice as high among men who consumed red meat frequently than among those who consumed it rarely (Giovannucci et al., 1993a). Other studies have shown similar results, although positive findings have generally been limited to populations whose diets include large
amounts of red meat (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Red Meat Consumption and Pancreatic Cancer
The overall evidence on red meat consumption and the risk of pancreatic cancer is somewhat inconsistent but generally suggestive of a positive relationship. Although three cohort studies have demonstrated an increased risk for those with higher levels of meat consumption (World Cancer Research Fund and American Institute for Cancer Research 1997), only one examined the specific effects of red meat. That study reported a twofold increase among men with higher levels of red meat consumption (Zheng et al., 1993).
Case-control studies have gone one step further by evaluating the effects of specific types of red meat, including beef and pork, as well as processed meats, like bacon and sausage. Those studies have consistently shown a positive association between pork consumption and pancreatic cancer risk, although they have yielded inconsistent results for other meats (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Red Meat Consumption and Kidney Cancer
The association between red meat consumption and the risk of kidney cancer has been evaluated in several case-control studies, with results suggesting the possibility of a positive association (World Cancer Research Fund and American Institute for Cancer Research, 1997). In the largest case-control study conducted to date, Wolk and colleagues (1996) reported no association between red meat consumption and the risk of kidney cancer. However, the means of meat preparation and the degree of “doneness” were both related to risk. Compared with baked and roasted meats, the consumption of fried and sautéed meats was associated with a 44 percent increase in risk, and the consumption of meats that are cooked well done, charred, or burnt was associated with a 24 percent increase in risk.
Whole-Grain Foods
Grains such as wheat, rice, corn, barley, and rye form the basis of most diets worldwide. Some grain products, like wheat bread or brown rice, remain in the whole-grain form, whereas others, like white bread or white rice, are more refined. Because most of the fiber, vitamins, and minerals are removed from grains during the refining process, whole-grain foods tend to be more nutrient-rich than refined foods (World Cancer Research Fund and American Institute for Cancer Research, 1997). The relationship between
dietary whole-grain and cancer should be placed in context of the established cardiovascular benefits of whole-grain foods, which have been shown to lower the risk of both coronary heart disease and ischemic stroke (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Whole-Grain Food Consumption and Stomach Cancer
The data on overall levels of grain consumption and the risk of stomach cancer are too inconsistent to judge the nature of the relationship. However, studies that have isolated the effects of whole-grain foods (compared with the effects of both whole grains and refined grains) suggest that whole-grain foods offer moderate levels of protection. In a summary of seven case-control studies, Jacobs and colleagues (1995) reported an inverse association for all but one study. In three of the studies, the relative risk was 0.4 for a high level of consumption compared with that for a low level of consumption. In addition, several of the studies showed either no association or a positive association between the consumption of refined grains and stomach cancer. Taken collectively, these studies suggest that the effect of grains depends on the extent to which the grains have been refined (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Fiber Consumption and Colon Cancer
Recent trials of the effects of increased levels of fiber consumption on the prevention of polyp recurrence among patients who have had polyps removed have shown no reduction in the recurrence of polyps during follow-up of some 3 years. In one trial, a supplement with wheat bran with high levels of fiber (13.5 grams [g]/day) was compared with a supplement with low levels of fiber (2 g/day) (Alberts et al., 2000). In the other study participants were counseled to increase their levels of fiber, fruit, and vegetable consumption, and the comparison group received usual care in the form of a standard brochure on healthy eating (Schatzkin et al., 2000). In both randomized trials the percentage of subjects with recurrent polyps was almost identical in each group. Although the three-year time frame for the preventive intervention may not be long enough, parallel data from the prospective Nurses’ Health Study also showed no relation between fiber intake and the risk of colon cancer during 16 years of follow-up (Fuchs et al., 1999).
Milk and Dairy Products
The effects of dairy products on cancer risk have not been well studied, and most links between the two have been made only indirectly (World Cancer Research Fund and American Institute for Cancer Research, 1997).
The available evidence suggests that milk and other dairy products may raise the risk of prostate cancer, kidney, and ovarian cancer, but may lower the risk of colon cancer.
Milk and Dairy Product Consumption and Prostate Cancer
Case-control studies generally support a positive association between the consumption of dairy products and prostate cancer risk, whereas cohort studies are less consistent (World Cancer Research Fund and American Institute for Cancer Research 1997). A higher level of milk consumption was related to an increased risk of prostate cancer in three of the seven cohort studies on dairy products (Snowdon et al., 1984; Le Marchand et al., 1994; Schuurman et al., 1999). In addition, one study suggested an increased risk with higher levels of consumption of butter, margarine, and cheese (Severson et al., 1989). Taken collectively, the evidence suggests the possibility of a moderate positive association between the consumption of dairy products and prostate cancer risk. Although this relationship has been attributed to the high fat contents of many dairy products, recent evidence suggests that calcium may be the responsible agent (Chan et al., 1998). Calcium has been shown to cause a compensatory decrease in circulating levels of 1,25-dihydroxy vitamin D, which may in turn raise the risk of prostate cancer (Giovannucci, 1998).
Milk and Dairy Product Consumption and Kidney Cancer
Evidence of a possible relationship between the consumption of dairy products and kidney cancer comes entirely from case-control studies (World Cancer Research Fund and American Institute for Cancer Research, 1997). Of nine such studies, seven have shown an increased risk among those with high levels of milk consumption (Mellemgaard et al., 1996; Wolk et al., 1996; World Cancer Research Fund and American Institute for Cancer Research, 1997). Odds ratios tend to be in the range of 1.3 to 1.8, with some studies suggesting even larger effects. Proposed mechanisms involve both animal protein and animal fat (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Milk and Dairy Product Consumption and Ovarian Cancer
Lactose is the primary sugar found in milk and other dairy products. When lactose is consumed and digested, it is broken down into two components, galactose and glucose, which are then broken down even further. If galactose is not metabolized properly, it can accumulate in the ovaries, affecting ovarian function and hormone levels. This may, in turn, contribute to the development of ovarian tumors. Two large cohort studies, the Nurses’ Health
Study and the Iowa Women’s Health Study, have examined the association of lactose consumption on ovarian cancer. Both studies found a modest increase in risk among women who consume large amounts of lactose.
NUTRIENTS
The link between whole foods and cancer is only one aspect of the diet-cancer relationship, but it is generally the most practical link when it comes to policy decisions and consumer action. Overall, people buy, prepare, and eat whole foods rather than specific nutrients. For this reason, this report focuses on the whole-food aspects of diet and their impact on cancer risk. There is a growing body of evidence that specific macronutrients or micronutrients could explain the associations between foods and cancer risk.
Macronutrients
Fat, carbohydrates, fiber, and protein have each been linked with cancer in some way (Table 3.8). A number of studies support a possible association between a high total fat intake and an increased risk of lung, colorectal, and prostate cancer (World Cancer Research Fund and American Institute for Cancer Research, 1997). The findings across studies are not consistent, however. Analyses based on prospective data tend to show very little, if any, association between total fat intake and cancer risk when total energy intake and other potential confounders are controlled for. A large combined analysis of breast cancer and dietary fats showed no association across a wide range of fat intakes (Hunter et al., 1996). The hypothesis that reducing intake of fats will reduce breast cancer risk is now being tested in the Women’s Health Initiative, a large intervention study in the United States (http://www.nhlbi.nih.gov/whi/). More plausible is a link between different types of fat and certain cancers. Saturated or animal fat, trans-unsaturated fat, and cholesterol have been found in some studies to increase the risk of cancer, whereas monounsaturated fat has been found in some studies to lower the risk of breast cancer (World Cancer Research Fund and American Institute for Cancer Research, 1997; Chiu et al., 1996; Willett, 1999; Zhang et al., 1999b). The consumption of large amounts of starch has been linked to a possible increase in stomach cancer risk and a possible decrease in colorectal cancer risk (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Micronutrients
The link between micronutrients and cancer has been studied extensively, and although a number of possible associations have been identified, only a few have enough evidence to qualify as probable associations (Table 3.9).
TABLE 3.8 Risk of Incident Cancer Associated with Selected Macronutrients
|
|
Association of Type of Cancer for the Following Level of Evidence: |
||
|
Macronutrients |
Convincing |
Probable |
Possible |
|
Fat and cholesterol |
|
|
|
|
Total fat |
|
|
Positive association: • Lung cancer • Colorectal cancer • Prostate cancer |
|
Saturated/animal fat |
|
|
Positive association: • Endometrial cancer • Lung cancer • Colorectal cancer • Prostate cancer • Non-Hodgkin’s lymphoma |
|
trans-Unsaturated fat |
|
|
Positive association: • Non-Hodgkin’s lymphoma |
|
Monounsaturated fat |
|
|
Inverse association: • Breast cancer |
|
Cholesterol |
|
|
Positive association: • Lung cancer • Pancreatic cancer |
|
Carbohydrates |
|
|
|
|
Starch |
|
|
Inverse association: • Colorectal cancer |
|
|
|
|
Positive association: • Stomach cancer |
|
Fiber |
|
|
Inverse association: • Endometrial cancer • Pancreatic cancer • Colorectal cancer |
|
Protein |
|
|
Inverse association: • Breast cancer (survival) |
|
Total energy intake |
|
|
Positive association: • Pancreatic cancer |
The most well-established link is between folate and colorectal cancer. A number of studies have found that as the level of folate consumption increases, the risk of colorectal cancer (as well as polyps) decreases (Freudenheim et al., 1991; Giovannucci et al., 1993b; Giovannucci et al., 1998b; Willett, 2000). The Nurses’ Health Study found that the consumption of large amounts of folate from fruits and vegetables was sufficient to lower
TABLE 3.9 Risk of Incident Cancer Associated with Selected Micronutrients
|
|
Association of Type of Cancer for the Following Level of Evidence: |
||
|
Macronutrients |
Convincing |
Probable |
Possible |
|
Vitamin A and carotenoids |
|
Inverse association: • Lung cancer |
Inverse association: • Breast cancer • Esophageal cancer • Stomach cancer • Colorectal cancer • Cervical cancer |
|
Folate |
|
Inverse association: • Colorectal cancer |
Inverse association: • Breast cancer |
|
Vitamin D |
|
Inverse association: • Colorectal cancer |
|
|
Vitamin E |
|
|
Inverse association: • Lung cancer • Cervical cancer • Colorectal cancer |
|
Calcium |
|
|
Inverse association: • Colorectal cancer |
|
|
|
|
Positive association: • Prostate cancer • Kidney cancer • Ovary cancer |
|
Selenium |
|
|
Inverse association: • Lung cancer • Prostate cancer • Colorectal cancer |
the colorectal cancer risk and that supplementation with a multivitamin that contained folate offered additional reductions (Giovannucci et al., 1998b). The underlying biological role of folate and its interaction with the MTHFR gene offers a plausible biological pathway for a causal relation between low levels of folate consumption and colon cancer.
The association of vitamin A and carotenoids with cancer risk has also been studied extensively, with the strongest evidence supporting a probable inverse relation with breast and lung cancers. Currently, however, it seems at most only a small reduction in the risk of breast cancer is associated with the consumption of large amounts of carotenoids from food sources (Zhang et al., 1999a). In addition, a number of observational studies support an association between the consumption of large amounts of carotenoids from food and a lower risk of lung cancer. Randomized trials of beta-carotene supplements found either no effect or an increased risk of lung cancer (Alpha-Tocopherol Beta Carotene Cancer Prevention Study Group, 1994;
Hennekens et al., 1996; Omenn et al., 1996), suggesting that a protective effect of fruits and vegetables is not explained by beta carotene alone. A surprise finding of the ATBC study was the lower prostate cancer risk observed in the vitamin E arm. Whether supplemental vitamin E might reduce prostate cancer risk is now being tested in the SELECT trial (http://www.cancer.gov/newscenter/SELECT).
Studies with animals have suggested that the consumption of larger amounts of selenium reduces the risk of various tumors, and ecological studies show an inverse relation between selenium and cancers of the breast and colon (Clark, 1985). A randomized trial to test the ability of a selenium supplement to reduce skin cancer risk showed no effect for skin cancer, but a significant reduction in the rates of mortality from cancers of the lung, colon, and prostate (Clark et al., 1996). Evidence against a large and rapid impact of selenium on cancer risk comes from a fortification intervention that was implemented in Finland. Because of the low selenium content of the soil in Finland (leading to low food selenium levels), selenium was applied with fertilizer in the mid-1980s. Blood selenium levels rose rapidly following this ecological intervention, but there has been no apparent decline in incidence or mortality rates for prostate or colon cancer (Willett, 1999). Whether supplemental selenium might reduce prostate cancer risk is now being tested, along with vitamin E, in the SELECT trial (http://www.cancer.gov/newscenter/SELECT).
Glycemic Index
The relation between macronutrients in diet and the physiological response to foods is complex. During digestion, the body breaks down dietary glucose into either sugar or starch so that it can be absorbed into the bloodstream. The resulting spike in blood glucose levels leads to a compensatory increase in blood insulin levels. Because insulin allows glucose to be taken up by the tissues, the blood glucose level drops in response to the high insulin levels. Once the blood glucose levels return to normal, insulin is released into the blood at a much lower rate.
The impact that a food has on these patterns of circulating blood glucose and insulin is estimated by its glycemic index. Foods with high glycemic indices are digested and absorbed more quickly, causing a rapid influx of glucose into the bloodstream, causing the insulin level to rise rapidly, while foods with a low glycemic index are converted to glucose more slowly and thus lead to a smaller spike in insulin levels. Foods with high glycemic indices may increase the risk of diabetes and coronary heart disease (Salmeron et al., 1997a; Salmeron et al., 1997b; Liu et al., 2000).
Whether foods with a high glycemic index also have an impact on the risk of cancer is unclear. If hyperinsulinemia is important in carcinogenesis in the colon, however, then the glycemic load from foods may be an impor-
tant risk factor for colon cancer. An increasing number of epidemiological studies have found that the consumption of large amounts of sugar and foods with a high glycemic index is associated with an increased risk of colon cancer (Giovannucci, 1995b). On the basis of this growing evidence relating insulin, glycemic load, and colon cancer risk, further research is needed to determine if the insulin pathway might also contribute to other major malignancies.
Disparities in Dietary Intake
The proportion of adults consuming the recommended five servings of fruits and vegetables a day has been estimated to be no more than 32 percent (Krebs-Smith et al., 1995a; Krebs-Smith et al., 1995b; Thompson et al., 1999; Li et al., 2000). Although these estimates are clearly low for the entire population, there is also considerable variation across subgroups defined by sex, race or ethnicity, education, and income. The levels of fruit and vegetable consumption are lower among men than among women (Subar et al., 1995; Kamimoto et al., 1999). One study showed that the level of fruit and vegetable consumption was lowest among Hispanics (Thompson et al., 1999), another found higher levels of consumption among whites than African Americans ages 55 and older (Kamimoto et al., 1999), and other studies have found lower levels of consumption of fruits and vegetables among individuals with lower incomes and lower levels of educational attainment (Subar et al., 1995; Thompson et al., 1999).
Although the level of consumption of red meat has decreased substantially in the population as a whole (U.S. Department of Agriculture, 1999), lower-income households have experienced less of a reduction than higher-income households (Interagency Board for Nutrition Monitoring and Related Research, 1993). In general, dietary trends in the last 60 years in the United States have shown a complex relationship with social class. Among those with higher socioeconomic status, diets have improved in regards to cancer and heart disease risk, whereas among those at lower socioeconomic classes, diets have worsened in the last half of the 20th century (Popkin et al., 1996).
ALCOHOL
Alcohol consumption has both beneficial and harmful effects on health. A U-shaped relationship between mortality and consumption of alcohol was described as early as 1926 (Pearl, 1926). A number of studies since then have shown that persons who drink heavily have higher rates of death from certain types of acute death, such as poisoning (Anderson, 1995), injuries (Andreasson et al., 1988; CDC, 1995), violence (Andreasson et al., 1988), and suicide (Andreasson et al., 1988), whereas the long-term use of alcohol increases the risk of death from cirrhosis (Norton et al., 1987),
hemorrhagic stroke (Donahue et al., 1986), and certain cancers (International Agency for Research on Cancer, 1988). Alcohol is listed as one of the top identifiable contributors to death in the United States (McGinnis and Foege, 1993), after tobacco and diet and activity patterns; and of all the threats to human health, alcohol probably causes the widest range of diseases and injuries (Rose, 1992).
In 1990, approximately 5 percent of all deaths (about 100,000 deaths) were attributable to alcohol consumption, as were 15 percent of potential years of life lost before age 65 (Rose, 1992). The consequences of alcohol consumption extend beyond death rates. Alcohol affects nerve cells in the brain and impairs cognitive function, both temporarily and permanently, over the long term (Meyer, Terayama et al., 1998), and it contributes to the destruction of personal and social relationships (Brookoff et al., 1997). An estimated 18 million people in the United States suffer from alcohol dependence, and an estimated 76 million people are affected by the consequences of alcohol abuse through either their own habit or that of someone close to them (McGinnis and Foege, 1993). Nearly a third of all U.S. adults engage in risky drinking patterns and thus need some kind of intervention, whether it is advice to cut down or referral for further evaluation and treatment for suspected abuse or dependence (Dawson, 2002).
In contrast to these harmful effects, however, there is substantial evidence for a beneficial effect of the consumption of moderate amounts of alcohol on the risk of coronary heart disease and thrombotic stroke. The risk of death from these cardiovascular causes is reduced, on average, by about 20 to 40 percent by the consumption of moderate amounts of alcohol (Doll, 1997; Thun et al., 1997). Because cardiovascular disease is the leading cause of death among those of middle and old ages, a reduction in the risk of mortality from cardiovascular disease with moderate alcohol use will translate into a reduction in the total risk of mortality in many populations. Clearly, there is a need to balance the many risks and benefits of alcohol use.
The Impact of Alcohol on Cancer Incidence
Because alcohol use tends to be associated with cigarette use and other high-risk behaviors, its independent effects on cancer have long been questioned. However, in 1988, on the basis of abundant epidemiological evidence, the International Agency for Research on Cancer concluded that alcohol is, in fact, a Group A carcinogen and an independent risk factor for cancers of the upper aerodigestive tract and liver (World Cancer Research Fund and American Institute for Cancer Research, 1997). Since publication of that report, a large body of evidence has confirmed that alcohol use also increases the risk of breast cancer (Longnecker et al., 1988; Willett et al., 2000) and, probably, colon cancer (Tomeo et al., 1999) (Table 3.10).
Although numerous studies have compared the effects of beer, wine,
TABLE 3.10 Increase in Risk of Incident Cancer Associated with Alcohol Use
|
|
Relative Risk (RR) |
|
|
Level of Evidence |
Moderate (RR 1.35–1.99) |
Large (RR >2.0) |
|
Convincing |
Breast |
|
|
|
|
Mouth Pharynx Larynx Esophagus Livera |
|
Probable |
Colon and rectum |
|
|
aAlthough light and moderate levels of alcohol consumption do not increase the risk of liver cancer, heavy drinking increases the risk substantially. |
||
and liquor, the type of alcohol consumed does not appear to influence the cancer risk as much as does the amount consumed (World Cancer Research Fund and American Institute for Cancer Research, 1997). For cancers of the upper aerodigestive tract, breast, colon, and rectum, there is a dose-response relationship, such that even low or moderate levels of consumption increase risk slightly. For liver cancer, the most important factor is heavy and persistent use, such as that defined by alcoholism, which can cause chronic liver injury.
Alcohol Consumption and Cancers of the Mouth, Pharynx, Larynx, and Esophagus
There is convincing epidemiological evidence from both cohort and case-control studies that alcohol consumption increases the risk of cancers of the mouth, pharynx, larynx, and esophagus (World Cancer Research Fund and American Institute for Cancer Research, 1997; Kjaerheim et al., 1998). All of the cohort studies that have examined this association, including those that controlled for smoking, have demonstrated a substantially increased risk. In addition, a dose-response relationship has been observed in the majority of case-control studies, including most of those that controlled for smoking. Alcohol also appears to act synergistically with tobacco in causing these cancers (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Several mechanisms have been proposed to explain the carcinogenic effect of alcohol on cancers of the upper aerodigestive tracts, although none are proven (World Cancer Research Fund and American Institute for Cancer Research, 1997). Alcohol may directly influence carcinogenesis in the cells that it touches; it may alter the permeabilities of cell membranes or modify cellular metabolism, thus making these cells more vulnerable to chemical carcinogens;
it may contain toxic substances, such as nitrosamines and polycyclic aromatic hydrocarbons, that alter DNA; and alcohol may in other ways compromise nutritional status and increase susceptibility to cancer (World Cancer Research Fund and American Institute for Cancer Research, 1997).
Alcohol Consumption and Liver Cancer
More than 40 studies have evaluated the relationship between high levels of alcohol intake and liver cancer. Although these studies did not use a standard definition of heavy drinking or even the same reference groups (nondrinkers versus the entire population), a positive association has been observed across a variety of study designs and populations (World Cancer Research Fund and American Institute for Cancer Research, 1997). Unlike most other cancers, liver cancer is associated only with the heavy persistent type of drinking characteristic of alcoholism, with its related chronic liver damage. Neither light nor moderate drinking increases liver cancer risk.
Alcohol Consumption and Breast Cancer
More than 25 studies have shown that alcohol increases breast cancer risk, most likely by raising the level of estrogen in the bloodstream or making the breast more vulnerable to carcinogens (Smith-Warner et al., 1998). A randomized feeding trial indicated that estrogen levels are increased when women consume even low levels of alcohol on a regular basis (Reichman et al., 1993).
The dose-response relationship between alcohol and breast cancer risk is best demonstrated in results from an analysis of data pooled from six prospective studies (Smith-Warner et al., 1998). In that analysis, women’s risk of breast cancer rose by 7 percent for every 10-g increase in daily alcohol consumption. The association did not differ substantially by type of alcoholic beverage consumed. Because breast tissue may be particularly vulnerable during adolescence and early adulthood, researchers have speculated that alcohol consumption during these time periods might be more harmful than alcohol consumption later in life (Colditz and Frazier, 1995), but to date, studies on this topic have been inconsistent (Harvey et al., 1987; Young, 1989; Garland et al., 1999).
Alcohol Consumption and Colorectal Cancer
Although the results are not entirely consistent, the majority of studies support an association between alcohol intake and an increased risk of colorectal cancer in both men and women (Giovannucci and Willett, 1994). Moreover, alcohol consumption is related to an increased risk of colorectal
adenomas (Giovannucci et al., 1993b; Kahn et al., 1998). A dose-response relationship has been observed across several studies, and some evidence suggests that even moderate drinkers (those who consume one drink a day) are at higher risk of colorectal cancer than nondrinkers (World Cancer Research Fund and American Institute for Cancer Research, 1997).
The effects of alcohol may be exacerbated by low levels of folate (Freudenheim et al., 1991). In the Health Professionals Follow-Up Study, an increased risk of colon cancer was observed only among those current and past drinkers who consumed lower levels of methionine or folate in their diets (Giovannucci et al., 1995b). Because alcohol is an antagonist of folate-related methyl group metabolism, it may cause an imbalance in DNA methylation, which may then contribute to colorectal carcinogenesis.
Alcohol Consumption and Lung Cancer
Although a number of cohort studies have examined the association between alcohol consumption and lung cancer, few have adequately adjusted for the effects of smoking. Those that have adjusted adequately have yielded inconsistent findings. Four cohort studies have shown a positive association (Klatsky et al., 1981; Kvåle et al., 1983; Pollack et al., 1984; Potter et al., 1992), and four have yielded null results (Gordon and Kannel, 1984; Bandera et al., 1997; Woodson et al., 1999; Breslow et al., 2000).
Disparities in Alcohol Use
Data on alcohol consumption in the United States indicate that the top 20 percent of drinkers consume more than 85 percent of all alcohol consumed (Greenfield and Rogers, 1999). Men and young adults ages 18 to 29 years are disproportionately represented among those with the highest alcohol consumption levels. The National Health Interview Survey (1998) indicates that at each age, the proportion of current drinkers is highest among non-Hispanic white men, followed by African-American men, and then Hispanic men (National Center for Health Statistics, 2000). Among women, the proportion of current drinkers is also highest among non-Hispanic white women at each age.
SUMMARY AND CONCLUSIONS
There is overwhelming evidence that lifestyle factors affect cancer risk. As detailed throughout this chapter, current epidemiological evidence links the major behavioral risk factors with several major cancers. Tobacco use causes cancers of the lung, oropharynx, larynx, esophagus, bladder, kidney, and pancreas and contributes to the risk of leukemia as well as cancers of the
colon, stomach, cervix, and liver. Recent declines in the rates of lung cancer in the United States among men—first among those who are younger and then among those who are older successively over time—and among women ages 40 to 59 reflect past declines in the use of cigarettes (Wingo et al., 1999). The cessation of tobacco use reduces the excess risk of nearly all of these cancers over time. Obesity increases the risk of breast, endometrial, colorectal, kidney, and esophageal cancer. Regular physical activity lowers the risk of cancers of the colon and breast cancer, and, possibly, endometrial cancer as well. Regular alcohol intake increases the risk of cancers of the oropharynx, larynx, esophagus, breast, liver, colon, and rectum. Diet has been linked to several cancers, with the most consistent evidence linking the consumption of large amounts fruits and vegetables with a lower risk cancer.
Numerous researchers have also estimated that positive changes in these lifestyle factors can reduce a good proportion of the national cancer burden (see Chapter 2). Although the specific methods and the results of these analyses vary, they are all remarkably consistent in pointing to the potential benefits of reducing tobacco use, improving diet, increasing physical activity, maintaining a healthy body weight, keeping alcohol consumption at low to moderate levels, and getting screened regularly for cancer.














































