2
Background
Anthrax is primarily a disease of animals, and historically, humans have generally contracted the disease through contact with infected animals or contaminated animal products. The disease had become extremely un-common in any form in the United States until the intentional mailings of anthrax spores caused an outbreak in the autumn of 2001 that resulted in five deaths from the inhalational form of the disease.
Anthrax vaccines for use in animals were first developed in the late 19th century. Work on vaccines suitable for human use gained urgency in the 1940s, with fears that anthrax would be used as a biological warfare agent. The current vaccine, Anthrax Vaccine Adsorbed (AVA), was licensed in 1970 and was recommended for use by a small population of textile mill workers, veterinarians, laboratory scientists, and other workers with occupational risk of exposure to anthrax. In the 1990s, increased concern about the use of biological weapons led the Department of Defense (DoD) to begin vaccination of U.S. military personnel. Some troops were given anthrax vaccine in the 1991 Gulf War, and a large program to vaccinate all service members was begun in 1998. By 2001 a limited vaccine supply, the result of delays in federal approval for release of newly manufactured vaccine lots, had significantly slowed plans to vaccinate all military personnel. After the deliberate distribution of anthrax spores in bioterrorist incidents in the autumn of 2001, the vaccine was offered as part of the treatment for as many as 10,000 of the civilians who had been exposed.
This chapter summarizes the basic pathophysiology of anthrax, reviews the history of anthrax vaccine development, and outlines the concerns that
have emerged on the part of some people about the adverse health out-comes that might be associated with use of the vaccine.
THE DISEASE
Anthrax is caused by infection with Bacillus anthracis, a gram-positive, nonmotile, spore-forming organism (Brachman and Friedlander, 1999; Dixon et al., 1999). Exposure to the spores of this one organism can cause three different forms of disease—cutaneous, gastrointestinal, or inhalational anthrax—depending on the site of infection. Cutaneous anthrax is the most common and the most treatable form; inhalational anthrax is rare but poses a much greater risk of death.
Epidemiology
Anthrax is found worldwide and is transmitted primarily through spores that are highly resistant to heat, drought, and many disinfectants (Dixon et al., 1999). It is primarily a disease of wild and domestic animals, especially herbivores such as cattle, sheep, and goats. Animals can be infected through exposure to spores in contaminated grazing areas, contaminated feed, or infected carcasses (Friedlander, 2000). Humans in agricultural settings can be infected through contact with infected animals or contaminated animal products. Human infections also occur in industrial settings where contaminated animal products such as wool, hair, hides, or meat are processed. Most human cases in either agricultural or industrial settings are cutaneous. Inhalational anthrax is generally seen only in industrial settings because conditions where a sufficiently large number of spores are aerosolized in an enclosed area do not generally occur naturally (Brachman and Friedlander, 1999). Person-to-person transmission is not known to occur with inhalational anthrax and has rarely been reported with other forms of the disease (Friedlander, 2000).
The worldwide incidence of anthrax in humans is difficult to determine, but the annual number of cases in the 1980s and 1990s is estimated to have been about 2,000, down from an estimated 20,000 to 100,000 cases in 1958 (Brachman and Friedlander, 1999). During the 19th century, “wool-sorters’ disease,” or inhalational anthrax, was fairly common among workers handling animal hides, hairs, or wools. Approximately 200 cases were reported in the United States before 1900 (Plotkin et al., 1960). Only 18 cases of inhalational anthrax were reported in the United States in the 20th century, despite evidence of extensive exposure of workers in goat hair-processing mills to aerosolized spores (Inglesby et al., 1999). In 1957, five cases of inhalational anthrax, four of them fatal, occurred at a goat hair-processing mill in New Hampshire. Vaccination did not become man-
datory for these workers until the 1960s and so cannot account for the low rate of inhalational disease (Inglesby et al., 1999).
Other forms of anthrax are also rare in the United States. Until the bioterrorist events in the autumn of 2001, a total of 238 anthrax cases had been reported since 1955; of those, 95 percent were cutaneous infections (Brachman and Friedlander, 1999; CDC, 2001a). In 2000, one reported case of cutaneous anthrax occurred (CDC, 2001a) and possible cases of gastrointestinal infection were associated with the consumption of contaminated meat (CDC, 2000b).
In the autumn of 2001 the United States experienced an outbreak of anthrax due to bioterrorism. Exposure to letters containing B. anthracis spores sent through the U.S. mail resulted in seven confirmed and five suspected cutaneous cases and 11 confirmed inhalational cases (CDC, 2001e). The victims included postal workers (Gallagher and Strober, 2001), employees of print and broadcast media organizations, and at least one infant (Roche et al., 2001).
Anthrax has also been part of biological warfare programs in some countries. In 1979 in Sverdlovsk, Russia, an apparently accidental release of aerosolized spores from a military facility resulted in 68 deaths among 79 individuals with reported cases of inhalational anthrax (Meselson et al., 1994).
Clinical Features
The outbreak of inhalational and cutaneous anthrax in the United States during the autumn of 2001 produced far more clinical and public health experience with the disease than had occurred in many decades. Both the outbreak and the outcomes of individual cases showed considerable differences from previous classic descriptions. The anthrax spores appeared to have been processed intentionally to enhance their most dangerous properties. They were finely milled and rendered nonpolar to maintain the very small particle size necessary for inhalation and to promote prolonged aerosolization. Naturally occurring spores tend to adhere quickly to each other and to surfaces.
Improvements in both the speed of diagnosis and clinical management resulted in the survival of at least some of those who contracted inhalational anthrax, which would not have been expected on the basis of earlier experience (Brown, 2001). Analysis of this new information on the clinical course of disease was continuing as the committee completed this report. In particular, both the inoculum associated with infection in different individuals and the duration of antibiotic treatment necessary for survival after infection remain uncertain. Although every effort was made to include
current information, some of the information presented here will likely be incomplete or out of date when the report becomes available.
Cutaneous Anthrax
Cutaneous anthrax results from the introduction of spores through the skin, generally at the site of a minor injury. After an incubation period, typically 2 to 5 days, a small pruritic (itchy) papule appears (Brachman and Friedlander, 1999). In 1 to 2 days, the site develops one or more vesicles filled with clear or serous fluid containing numerous anthrax bacilli. Within a week, the vesicle erodes, leaving a necrotic ulcer with a characteristic black eschar at the center (see Figure 2-1). The lesion then heals in another 2 to 3 weeks. Edema may develop around a lesion and can become extensive in some cases. Other symptoms including malaise, low-grade fever, and swelling of adjacent lymph glands may occur, but anthrax lesions are generally painless unless a secondary infection is present. Without antibiotic treatment, up to 20 percent of cutaneous anthrax infections are fatal, but with treatment the fatality rate is less than 1 percent (CDC, 2000c). A recent case report describes bioterrorist-related cutaneous anthrax in an infant (Freedman et al., 2002).
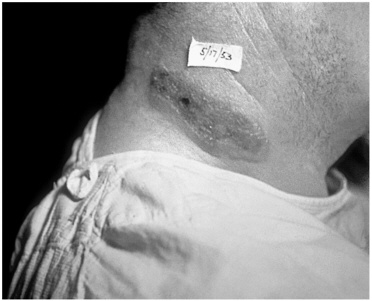
FIGURE 2-1 Cutaneous anthrax lesion.
SOURCE: Public Health Image Library, CDC.
Gastrointestinal Anthrax
Eating meat contaminated with anthrax spores can result in gastrointestinal or oropharyngeal infection. Pathological examinations show ulcerations with edema and mucosal necrosis in the affected area (Dixon et al., 1999). A gastrointestinal infection initially produces nausea, vomiting, and fever, followed by often severe abdominal pain, bloody diarrhea, and ascites. As these symptoms are common to other acute abdominal conditions, gastrointestinal anthrax can be difficult to identify. Blood loss and fluid and electrolyte imbalances can lead to shock, and death may follow intestinal perforation or anthrax toxemia (Dixon et al., 1999). From 25 to 75 percent of cases may be fatal (Brachman and Friedlander, 1999). An oropharyngeal infection, which is milder than the gastrointestinal form, produces fever, visible ulcers, and local edema and swollen lymph glands that can interfere with swallowing and breathing (Dixon et al., 1999).
Inhalational Anthrax
Inhalational anthrax has been a rare disease because there are limited circumstances when spores whose particle sizes are sufficiently small (less than 5 micrometers [µm]) to be inhaled deep into the lung are suspended in air. When anthrax spores are inhaled, they are deposited in the alveolar spaces, where they are taken up by macrophages and transported to the mediastinal and peribronchial lymph nodes. In the lymph nodes, spores germinate to become vegetative, multiply, and cause hemorrhagic mediastinitis. They also move into the bloodstream and spread throughout the body. At Sverdlovsk, the modal incubation period from exposure to the onset of symptoms was 10 days, but some cases developed up to 6 weeks after the reported exposure (Dixon et al., 1999; Inglesby et al., 1999; Meselson et al., 1994).
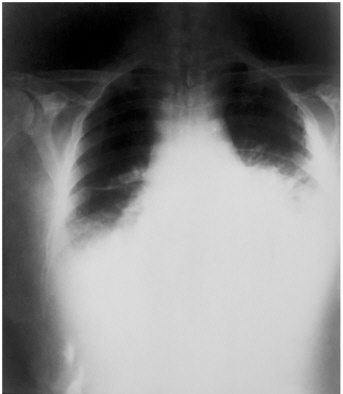
The initial symptoms of inhalational anthrax, which resemble those of influenza and other common upper respiratory infections, include malaise, fatigue, and cough. In days, severe respiratory distress develops, with dyspnea, cyanosis, and strident cough. Other symptoms may include fever, chills, and subcutaneous edema of the chest and neck (Brachman and Friedlander, 1999). Radiographic examination of the chest usually shows a characteristic widening of the mediastinum and pleural effusions (see Figure 2-2). Shock may develop, and hemorrhagic meningitis (see Figure 2-3) may occur in about 50 percent of cases (Brachman and Friedlander, 1999). Death usually occurs within 24 hours following the onset of acute symptoms. At Sverdlovsk, the mean interval from the onset of initial symptoms to death was 3 days (Dixon et al., 1999). On the basis of previously limited observations, inhalational anthrax was fatal in 80 to 90 percent of cases
(CDC, 2000c). Aggressive treatment early enough in the course of the disease appears to improve the rate of survival.
A review of the first 10 reported patients with inhalational anthrax resulting from the bioterrorism release of anthrax spores in the autumn of 2001 indicated that all 10 had abnormal chest X rays (Jernigan et al., 2001). Abnormalities included infiltrates, pleural effusion, and mediastinal widening. Mediastinal lymphadenopathy was observed in seven patients. Extensive sweating was a prominent feature in these 10 patients, although it had not been emphasized in reports of earlier cases. A brief period of improvement after the earliest symptoms noted in earlier cases was not seen in these patients. The median incubation period from exposure to the onset of symptoms was 4 days in the six patients for whom the time of exposure was known. Six of the 10 patients described in the paper survived with multidrug antibiotic regimens and aggressive supportive care (e.g., drainage of pleural effusions). Additional case reports describe clinical features of the tenth and eleventh cases of bioterrorism-related inhalational anthrax (Barakat et al., 2002; Mina et al., 2002).
Treatment
Penicillin, doxycycline, and ciprofloxacin are the primary antibiotics recommended for treatment of all forms of anthrax (CDC, 2000c), although there are no formal studies of clinical treatment of inhalational anthrax in humans (Inglesby et al., 1999). Both doxycycline and ciprofloxacin were recommended as initial therapy for infections associated with the recent bioterrorist attack in the United States (CDC, 2001c). Mild cutaneous cases can be treated effectively with oral medication, but treatment does not alter the course of the skin lesion. Other forms of anthrax and more serious cutaneous cases must be treated with intravenous antibiotics. Additional supportive therapy may be needed to prevent septic shock, to maintain fluid and electrolyte balance, and to maintain a patent airway. If inhalational exposure to anthrax spores is known or suspected but symptoms have not developed, the Centers for Disease Control and Prevention (CDC) recommends a 60-day course of antibiotic treatment to protect against delayed germination of spores (CDC, 2001b). If available, at least three doses of anthrax vaccine can be administered.
Pathogenesis
When anthrax spores are introduced into the body by any route, they are taken up by macrophages and germinate into vegetative bacteria with an antiphagocytic capsule that deters the host’s immune response to the organism. The vegetative bacteria multiply and secrete toxins that produce
local edema and necrosis. If bacteria are carried to regional lymph nodes, they multiply further and produce additional edema and necrosis and enter the bloodstream to produce a systemic infection (Brachman and Friedlander, 1999; Dixon et al., 1999).
The virulence of B. anthracis derives from the production of three toxin proteins and the capsule. The toxin proteins, encoded on the pXO1 plasmid, are protective antigen (PA), edema factor (EF), and lethal factor (LF). To produce active toxins, PA must bind to cellular receptors and then bind with either EF or LF (see Figure 2-4). The human cellular receptor for PA has recently been identified and characterized and named anthrax toxin receptor (Bradley et al., 2001). The resulting edema toxin or lethal toxin can then enter the cell. The effects of edema toxin appear to result from EF, an adenylate cyclase that increases intracellular levels of cyclic adenosine monophosphate, which upsets water homeostasis (Dixon et al., 1999). Edema toxin may also impair neutrophil function. LF is a zinc metalloprotease that cleaves two mitogen-activated protein kinase kinases. The mechanism by which it leads to death of the host remains unknown but
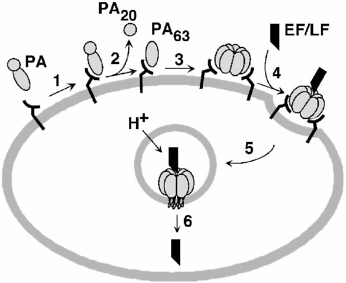
FIGURE 2-4 Model of anthrax toxin action. (1) PA binds to cellular receptor. (2–3) The protein is cleaved and activated to form a heptameric prepore. (4) LF, EF, or both bind to the heptamer, and the resulting complex is taken into an acidic compartment in the cell through endocytosis. (5–6) The acidic pH initiates the heptamer to pierce the membrane of the cell and translocate LF, EF, or both into the cytosol, where the toxins lead to damage. [Reprinted, with permission, from Biochemistry 38:10432–10441 (1999). Copyright 1999 by American Chemical Society.]
may involve suppression of the inflammatory response (Pellizzari et al., 1999, Erwin et al., 2001).
A second plasmid, pXO2, contains a gene that encodes the synthesis of a polyglutamyl capsule that inhibits phagocytosis of the vegetative bacteria.
Full virulence requires both plasmids. Attenuated spore vaccines have been developed with bacterial strains missing one or both plasmids. The livestock vaccine currently in use in the United States and other countries, known as the Sterne vaccine, is derived from a noncapsulated B. anthracis variant that lacks the pXO2 plasmid. The vaccine currently licensed for human use in the United States, AVA, however, is a cell-free filtrate containing PA as the principal immunogen.
ANTHRAX VACCINE DEVELOPMENT
Early work on the development of a vaccine against anthrax in animals was carried out in the 1880s by W. S. Greenfield and by Louis Pasteur (Turnbull, 1991). What became known as Pasteur’s vaccine used an encapsulating nontoxigenic strain of B. anthracis administered to animals in two doses that differed in their degrees of heat attenuation (Turnbull, 1991, 2000). A live spore vaccine developed by Sterne in the 1930s eventually supplanted Pasteur’s vaccine and remains in use for livestock in many parts of the world. It is credited with marked reductions in the incidence of anthrax cases or in the absence of anthrax cases in vaccinated herds, reducing the devastating impact of the disease in both animals and humans in areas where vaccination is carried out. Analogous vaccines were developed in the former Soviet Union and China, where they are used for humans as well as in animals (Turnbull, 2000). Limited data regarding the safety and efficacy of the vaccine used in humans in the former Soviet Union have been reported (Demicheli et al., 1998; Shlyakhov and Rubenstein, 1994).
Live spore vaccines, such as the Sterne vaccine, have been associated with residual virulence that leads to occasional casualties in livestock and thus have not been considered appropriate for human use in the West. Therefore, when interest in the potential use of anthrax as a biological warfare agent arose after World War II, work to develop inactivated vaccines began in both the United States, at Fort Detrick, Maryland, and the United Kingdom, at Porton Down (Turnbull, 2000).
In the United States, the research to develop an anthrax vaccine used B. anthracis cultures in synthetic medium without proteins or other macromolecules (Turnbull, 2000). A production system was described in 1954 (Wright et al., 1954), including a chemically defined growth medium and a method of concentrating, stabilizing, and partially purifying protective antigen by precipitation. The safety and efficacy of this vaccine were evaluated in a controlled trial conducted between 1955 and 1959 at goat hair-
processing mills in the eastern United States (Brachman et al., 1962). The initial production method was soon modified for scale-up, with changes in the culture conditions, product purification method, and strain of the organism used (Auerbach and Wright, 1955; Puziss and Wright, 1963; Wright and Puziss, 1957; Wright et al., 1962).
The current vaccine, AVA,1 was licensed in 1970 for manufacture by the Michigan Department of Public Health. Both the production plant and the product line were eventually sold to a private company, BioPort, which at the time of this report was the sole U.S. manufacturer of an anthrax vaccine. The product license for AVA calls for subcutaneous administration of a basic series of six doses of 0.5 milliliters each. After administration of the initial dose, subsequent doses are administered at 2 weeks, 4 weeks, 6 months, 12 months, and 18 months. Annual booster doses are required.
USE OF ANTHRAX VACCINE
As was noted above, B. anthracis is a widespread organism, and anthrax disease has long been endemic in many agricultural areas. The disease was not, until quite recently, considered threatening outside certain industrial or agricultural settings that allowed exposure. The licensed anthrax vaccine had routinely been administered to the limited population of U.S. workers with occupational exposure to anthrax bacteria or spores. It is estimated that 68,000 doses of AVA were distributed between 1974 and 1989 (Ellenberg, 1999).
In the United States in recent years, however, AVA has been used primarily by the military to protect troops from weaponized B. anthracis (Brachman and Friedlander, 1999; Mazzuchi et al., 2000). At the time of the Gulf War, there were fears that Iraq had produced weapons containing anthrax spores. More than 300,000 doses of AVA were distributed during Operation Desert Storm, probably to more than 150,000 service members (Army Information Paper, 1991).
Although no biological weapons were used against U.S. or other coalition forces during the Gulf War, subsequent inspections of Iraq in 1995 and 1996 by the United Nations Special Commission and the International Atomic Energy Agency revealed that 8,000 liters of anthrax spore suspension had been produced and that during the Gulf War in 1991 Iraq had 200 bombs and 25 ballistic missiles containing biological agents (Henderson, 1999; Zilinskas, 1997). More recent information indicates that Iraq has
been renovating biological, chemical, and nuclear warfare research sites 3 years after barring international inspectors (Bohlen, 2001). In 1997, as a result of the concerns about biological weapons, then Secretary of Defense William Cohen initiated a plan to vaccinate all U.S. service members against anthrax. Immunizations began in March 1998 under DoD’s Anthrax Vaccine Immunization Program (AVIP). As of November 29, 2001, 522,529 service members had received 2,098,544 of doses of AVA (http://www.anthrax.osd.mil/Flash_interface/default.html, accessed January 11, 2002).
Implementation of AVIP has been slowed by a limited supply of vaccine. Renovations were begun at the manufacturing plant in 1998, and BioPort, the sole manufacturer, did not receive approval from the Food and Drug Administration (FDA) for release of newly manufactured vaccine until January 31, 2002. DoD has been able to continue immunizations, despite the limited supply of vaccine, but not at the rate first planned. In July 2000, in November 2000, and again in June 2001, DoD slowed the anthrax immunization program, focusing only on troops thought to be at greatest potential risk (http://www.anthrax.osd.mil, accessed September 5, 2000; Marshall, 2000).
In the autumn of 2001, more than 30,000 civilians were potentially exposed to anthrax in bioterrorist incidents involving the distribution of highly infectious spores through the U.S. mail (CDC, 2001d,e). Beginning in December 2001, CDC began offering vaccination with AVA as a treatment option for selected exposed civilians. This therapeutic use of the vaccine following exposure was not included under the official vaccine license and is being monitored under the provisions of an Investigational New Drug application. As of February 25, 2002, the latest data available at the time that this report was completed, 192 people had begun receiving doses of AVA (Ashford, 2002).
The committee emphasizes that this report is addressed to DoD and focuses on the licensed use of AVA for immunization before exposure to anthrax spores.
CONCERNS ABOUT USE OF AVA
AVIP and the product AVA have become focal points of great concern on the part of at least segments of the military and interested public. A few service members have refused the vaccine, at the risk of court-martial, because of their perception that it is particularly dangerous.2 Among the
concerns are complaints among Gulf War veterans of chronic multisystem clinical conditions that still lack a definable relationship to the anthrax vaccine or to other events in their Gulf War experiences (IOM, 2000b).
The U.S. Congress has responded to these concerns, and hearings have been held in the U.S. House of Representatives and the U.S. Senate.3 The hearings typically included several current or former service members (or family members of service personnel) who had raised concerns about the adverse events that they had experienced or observed or about the responses of military health care providers to these concerns. The witness list usually also included officials from DoD or a branch of the military service, FDA, and sometimes the manufacturer of AVA. As noted in Appendix C, the IOM Committee to Assess the Safety and Efficacy of the Anthrax Vaccine also held a public hearing to gather information from people with concerns about the vaccine. The IOM committee benefited from the perspective provided by the speakers, many of whom also provided testimony during congressional hearings. The witnesses described persistent and debilitating symptoms ranging from fever, headache, and malaise to swelling, joint pain, and tinnitus, which they ascribed to the anthrax vaccine. Several witnesses also described specific serious conditions including hypogonadism; Stevens-Johnson syndrome, which affected their vision as well as their skin; and aplastic anemia, which proved fatal. In addition, many witnesses observed that when they reported their symptoms to medical personnel, the health care providers seemed to be unaware of the Vaccine Adverse Event Reporting System (VAERS) or unwilling to file a report with VAERS and often seemed to doubt that the vaccine could have caused their symptoms.
AVAILABLE DATA ON AVA
In its letter report of March 2000, An Assessment of the Safety of the Anthrax Vaccine, the IOM Committee on Health Effects Associated with Exposures During the Gulf War expressed regret over the lack of information about the vaccine in the peer-reviewed published literature (IOM,
2000a). It listed an array of studies that were unpublished or ongoing that could contribute to the body of information on which conclusions regarding health effects could be based.
As the study presented in this report began, representatives of DoD provided assurances to IOM that all relevant information from DoD would be made readily available to the committee and that efforts would be made to publish the data from completed studies. DoD and its investigators have followed through on these assurances. Most of the studies that have been carried out by DoD investigators to assess the safety and efficacy of the anthrax vaccine have now been written up as manuscripts and submitted for publication. Additional studies have been published since the letter report was released (e.g., CDC, 2000a; Gunzenhauser et al., 2001; Pittman et al., 2001, 2002; Rehme et al., 2002). In addition, one of the most important contributions to the committee’s evaluation was in the form of analyses of data from military databases carried out at the committee’s request. In accordance with its charge, the earlier IOM committee (Committee on Health Effects Associated with Exposures During the Gulf War) reviewed only the published, peer-reviewed literature to reach its conclusions about safety. The current committee had a different purpose and as a result chose to review all the studies it was aware of and for which adequate descriptions of the study methods, data analyses, and results were made available. These studies are systematically reviewed in the chapters that follow.
Several previous IOM committees evaluating possible causal associations between vaccines or other exposures and specific health outcomes have chosen to describe their findings with a weight-of-evidence approach (IOM, 1991, 1994, 2000b). Their findings placed associations between the exposure of interest and the health outcome into categories such as sufficient evidence of a causal relationship, sufficient evidence of an association, limited or suggestive evidence of an association, inadequate or insufficient evidence to determine whether an association does or does not exist, and limited or suggestive evidence of no association. The current committee chose not to use that approach because it was not asked to evaluate exposure to AVA as a cause of specific health outcomes. Rather, the committee was asked to provide an overall evaluation of the anthrax vaccine’s safety. In addition, its charge included addressing various aspects of the efficacy of AVA, as well as manufacturing issues, two topics for which a weight-of-evidence approach is not readily applicable.
REFERENCES
Army Information Paper (from an Army staff officer). 1991. Numbers of service members vaccinated during Operation Desert Storm. [Online]. Available: http://www.gulflink.osd.mil/va/va_refs/n46en087/0215_027_0000001.htm [accessed January 18, 2002].
Ashford D. 2002. Information on the number of participants enrolled in vaccine IND application. E-mail to Joellenbeck L, Institute of Medicine, Washington, D.C., February 25.
Auerbach BA, Wright GG. 1955. Studies on immunity in anthrax. VI. Immunizing activity of protective antigen against various strains of Bacillus anthracis. Journal of Immunology 75:129–133.
Barakat LA, Quentzel HL, Jernigan JA, Kirschke DL, Griffith K, Spear SM, Kelley K, Barden D, Mayo D, Stephens DS, Popovic T, Marston C, Zaki SR, Guarner J, Shieh WJ, Carver HW 2nd, Meyer RF, Swerdlow DL, Mast EE, Hadler JL. 2002. Fatal inhalational anthrax in a 94-year-old Connecticut woman. JAMA 287(7):863–868.
Bohlen C. 2001, December 20. Vanishing Taliban, a British force on deck, spilled Iraqi secrets. New York Times. p. B1.
Brachman PS, Friedlander AM. 1999. Anthrax. In: Plotkin SA, Orenstein WA, eds. Vaccines, 3rd ed. Philadelphia, Pa.: W. B. Saunders Co. Pp. 629–637.
Brachman PS, Gold H, Plotkin S, Fekety FR, Werrin M, Ingraham NR. 1962. Field evaluation of a human anthrax vaccine. American Journal of Public Health 52:632–645.
Bradley KA, Mogridge J, Mourez M, Collier RJ, Young JA. 2001. Identification of the cellular receptor for anthrax toxin. Nature 414(6860):225–229.
Brown K. 2001. Anthrax: a “sure killer” yields to medicine. Science 294(5548):1813–1814.
CDC (Centers for Disease Control and Prevention). 2000a. Surveillance for adverse events associated with anthrax vaccination—U.S. Department of Defense, 1998–2000. MMWR (Morbidity and Mortality Weekly Report) 49(16):341–345.
CDC. 2000b. Human ingestion of Bacillus anthracis–contaminated meat—Minnesota, August 2000. MMWR (Morbidity and Mortality Weekly Report) 49(36):813–816.
CDC. 2000c. Use of anthrax vaccine in the United States: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR (Morbidity and Mortality Weekly Report) 49(RR-15):1–20.
CDC. 2001a. Human anthrax associated with an epizootic among livestock—North Dakota, 2000. MMWR (Morbidity and Mortality Weekly Report) 50(32):677–680.
CDC. 2001b. Update: investigation of bioterrorism-related anthrax and interim guidelines for exposure management and antimicrobial therapy, October 2001. MMWR (Morbidity and Mortality Weekly Report) 50(42):909–919.
CDC. 2001c. CDC health advisory: updated information about how to recognize and handle a suspicious package or envelope. [Online]. Available: http://www.bt.cdc.gov/DocumentsApp/Anthrax/10312001/han50.asp [accessed January 18, 2002].
CDC. 2001d. Update: investigation of bioterrorism-related anthrax and adverse events from antimicrobial prophylaxis. MMWR (Morbidity and Mortality Weekly Report) 50(44): 973–976.
CDC. 2001e. Update: investigation of bioterrorism-related inhalational anthrax—Connecticut, 2001. MMWR (Morbidity and Mortality Weekly Report) 50(47):1049–1051.
Dixon TC, Meselson M, Guillemin J, Hanna PC. 1999. Anthrax. New England Journal of Medicine 341(11):815–826.
Demicheli V, Rivetti D, Deeks JJ, Jefferson T, Pratt M. 1998. The effectiveness and safety of vaccines against human anthrax: a systematic review. Vaccine 16(9-10):880–884.
Ellenberg SS. 1999. Vaccine Adverse Events Reporting System. Statement at the July 21, 1999, Hearing of the Subcommittee on National Security, Veterans Affairs, and International Relations, Committee on Government Reform, U.S. House of Representatives, Washington, D.C.
Erwin JL, DaSilva LM, Bavari S, Little SF, Friedlander AM, Chanh TC. 2001. Macrophage-derived cell lines do not express proinflammatory cytokines after exposure to Bacillus anthracis lethal toxin. Infection and Immunity 69(2):1175–1177.
Freedman A, Afonja O, Chang MW, Mostashari F, Blaser M, Perez-Perez G, Lazarus H, Schacht R, Guttenberg J, Traister M, Borkowsky W. 2002. Cutaneous anthrax associated with microangiopathic hemolytic anemia and coagulopathy in a 7-month-old infant. JAMA 287(7):869–874.
Friedlander AM. 2000. Anthrax: clinical features, pathogenesis, and potential biological warfare threat. Current Clinical Topics in Infectious Diseases 20:335–349.
Gallagher TC, Strober BE. 2001. Cutaneous Bacillus anthracis infection. New England Journal of Medicine 345(22):1646–1647.
Gunzenhauser JD, Cook JE, Parker ME, Wright I. 2001. Acute side effects of anthrax vaccine in ROTC cadets participating in advanced camp, Fort Lewis, 2000. MSMR (Medical Surveillance Monthly Report) 7(5):4–14.
Henderson DA. 1999. The looming threat of bioterrorism. Science 283(5406):1279–1282.
Inglesby TV, Henderson DA, Bartlett JG, Ascher MS, Eitzen E, Friedlander AM, Hauer J, McDade J, Osterholm MT, O’Toole T, Parker G, Perl TM, Russell PK, Tonat K. 1999. Anthrax as a biological weapon: medical and public health management. JAMA 281(18): 1735–1745.
IOM (Institute of Medicine). 1991. Howson CP, Howe CJ, Fineberg HV, eds. Adverse Effects of Pertussis and Rubella Vaccines. Washington, D.C.: National Academy Press.
IOM. 1994. Stratton KE, Howe CJ, Johnston RB, eds. Adverse Events Associated with Childhood Vaccines: Evidence Bearing on Causality. Washington, D.C.: National Academy Press.
IOM. 2000a. An Assessment of the Safety of the Anthrax Vaccine: A Letter Report. Washington, D.C.: National Academy Press.
IOM. Fulco CE, Liverman CT, Sox HC, eds. 2000b. Gulf War and Health. Washington, D.C.: National Academy Press.
Jernigan JA, Stephens DS, Ashford DA, Omenaca C, Topiel MS, Galbraith M, Tapper M, Fisk TL, Zaki S, Popovic T, Meyer RF, Quinn CP, Harper SA, Fridkin SK, Sejvar JJ, Shepard CW, McConnell M, Guarner J, Shieh WJ, Malecki JM, Gerberding JL, Hughes JM, Perkins BA. 2001. Bioterrorism-related inhalational anthrax: the first 10 cases reported in the United States. Emerging Infectious Diseases 7(6):933–944.
Marshall E. 2000. Bioterrorism. DOD retreats on plan for anthrax vaccine. Science 289(5478): 382–383.
Mazzuchi JF, Claypool RG, Hyams KC, Trump D, Riddle J, Patterson RE, Bailey S. 2000. Protecting the health of U.S. military forces: a national obligation. Aviation, Space, and Environmental Medicine 71(3):260–265.
Meselson M, Guillemin J, Hugh-Jones M, Langmuir A, Popova I, Shelokov A, Yampolskaya O. 1994. The Sverdlovsk anthrax outbreak of 1979. Science 266(5188):1202–1208.
Mina B, Dym JP, Kuepper F, Tso R, Arrastia C, Kaplounova I, Faraj H, Kwapniewski A, Krol CM, Grosser M, Glick J, Fochios S, Remolina A, Vasovic L, Moses J, Robin T, DeVita M, Tapper ML. 2002. Fatal inhalational anthrax with unknown source of exposure in a 61-year-old woman in New York City. JAMA 287(7):858–862.
Pellizzari R, Guidi-Rontani C, Vitale G, Mock M, Montecucco C. 1999. Anthrax lethal factor cleaves MKK3 in macrophages and inhibits the LPS/IFN gamma-induced release of NO and TNFalpha. FEBS Letters 462(1-2):199–204.
Pittman PR, Gibbs PH, Cannon TL, Friedlander AM. 2001. Anthrax vaccine: short-term safety experience in humans. Vaccine 20(5–6):972–978.
Pittman PR, Kim-Ahn G, Pifat DY, Coon K, Gibbs P, Little S, Pace-Templeton J, Myers R, Parker GW, Friedlander AM. 2002. Anthrax vaccine: safety and immunogenicity of a dose-reduction, route comparison study in humans. Vaccine 20(9–10):1412–1420.
Plotkin SA, Brachman PS, Utell M, Bumford FH, Atchison MM. 1960. An epidemic of inhalation anthrax, the first in the twentieth century. American Journal of Medicine 29:992–1001.
Puziss M, Wright GG. 1963. Studies on immunity in anthrax. X. Gel adsorbed protective antigen for immunization of man. Journal of Bacteriology 85:230–236.
Rehme PA, Wiliams R, Grabenstein JD. 2002. Ambulatory medical visits among anthrax vaccinated and unvaccinated personnel after return from Southwest Asia. Military Medicine 167:205–210.
Roche KJ, Chang MW, Lazarus H. 2001. Cutaneous anthrax infection. New England Journal of Medicine 345(22):1611.
Shlyakhov EN, Rubinstein E. 1994. Human live anthrax vaccine in the former USSR. Vaccine 12(8):727–730.
Turnbull PC. 1991. Anthrax vaccines: past, present and future. Vaccine 9(8):533–539.
Turnbull PCB. 2000. Current status of immunization against anthrax: old vaccines may be here to stay for a while. Current Opinion in Infectious Diseases 13(2):113–120.
Wright GG, Puziss M. 1957. Elaboration of protective antigen of Bacillus anthracis under anaerobic conditions. Nature 179:916–917.
Wright GG, Hedberg MA, Slein JB. 1954. Studies on immunity in anthrax. III. Elaboration of protective antigen in a chemically-defined, non-protein medium. Journal of Immunology 72:263–269.
Wright GG, Puziss M, Neely WB. 1962. Studies on immunity in anthrax. IX. Effect of variations in cultural conditions on elaboration of protective antigen by strains of Bacillus anthracis . Journal of Immunology 83:515–522.
Zilinskas RA. 1997. Iraq’s biological weapons: the past as future? JAMA 278(5):418–424.