Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
EFects of Beta'Adrenergic Agonists on Growth and Carcass Characteristics of Animals LARRY A. MUIR Until recently, few mechanisms were known through which a drug could promote the growth performance or improve the carcass characteristics of livestock and poul- try (Muir, 1985~. Antimicrobial agents, such as antibiotics and antibacterials, improve growth performance of livestock and poultry by killing or inhibiting the growth of micro- organisms (Muir et al., 1977~. Estrogenic agents improve growth performance and carcass characteristics of cattle and sheep, but the specific mechanism is not well understood (Burroughs et al., 1954; Dinus- son et al., 1950; Muir et al., 1983~. Proges- tational agents improve the growth perform- ance of cyclic heifers by inhibiting estrus and therefore its adverse affects, such as hyperactivity and reduced feed consump- tion (Davis, 1969~. Androgenic agents im- prove growth performance and carcass char- acteristics of cattle and swine, especially females, supposedly through a direct, re- ceptor-mediated action on skeletal muscle cells (Heitzman, 19804. In addition, exoge- nous growth hormone administration re- portedly improves growth performance and carcass characteristics of livestock (Machlin, 1972; Wagner and Veenhuizen, 19784. 184 Now a new mechanism has been found through which the growth performance and carcass characteristics of all poultry and livestock species are dramatically improved (Baker et al., 1984; Beermann et al., 1986; Dalrymple et al., 1984; Moser et al., 1986; Muir et al., 1985; Ricks et al., 1984~. This mechanism involves the activation by beta- adrenergic agonists (beta-agonists) of spe- cific beta-adrenoceptors on the surface of adipocytes and skeletal muscle cells. This paper describes what is known about beta- agonists and the mechanisms through which they work. WHAT ARE BETA-AGONISTS? Beta-agonists are structural analogs of the catecholamines epinephrine and norepi- nephrine. Epinephrine and norepinephrine are very similar in structure, and both bind to four different cell surface receptors called adrenoceptors (specifically, the alpha l, alphas, betel, and betas receptors). Of special in- terest are the effects of beta-agonists on adipose and muscle tissues. The adipose tissue of most species contains beta-recep- tors that, when activated, stimulate lipoly
BETA-ADRENERGIC AGONISTS sis. Most muscle tissue contains primarily betas or betas receptors, which, when ac- tivated, cause a specific muscular function. Skeletal muscle is known to have betas receptors, but their response function is not well understood. The structures of the beta-agonists that will be discussed in this paper isoproter- enol, clenbuterol, cimaterol, L-640,033, add BRL3513~are shown in Figure 1. Isopro- tereno} is a very potent beta/beta agonist that is not orally active but is very effective in vitro. Clenbutero} and cimaterol (Amer- ican Cyanamid) ant! L-640,033 (Merck) are orally active beta-agonists that have been shown to stimulate animal growth and change carcass characteristics (Dalrymple et al., 1984; Muir et al., 1985; Ricks et al., 19844. BRL35135 (Beecham) is an orally active HO HO~CH-CH2-NH-CH (CH3)2 C1 a>=\ OH / \ 1 H2N - \ /^ CH-CH2-NH-C (CH3)3 C1 OH H H9N~ C CH2-NH- IC-CH2-CH2-C6H5 C - N i4: OH H2N~CH-CH2-NH-CH (CH3)2 i=: OH C\H3 ~ ~ CH-CH2-NH-C-CH2~ OCH2-CO2-CH3 185 beta-agonist that has been shown to stim- ulate lipolysis (Arch et al., 1983, 1984~. EFFECTS OF BETA-AGONISTS ON GROWTH PERFORMANCE AND CARCASS CHARACTERISTICS Numerous growth trials have been con- ducted with different beta-agonists at vary- ing dose levels in poultry, swine, sheep, and, to a lesser extent, cattle (Baker et al., 1984; Beermann et al., 1986; Dalrymple et al., 1984; Moser et al., 1986; Muir et al., 1985; Ricks et al., 1984~. The results of these trials are summarized in Tables 1 and 2. In general, beta-agonists work best when used during the finisher period, regardless of species. Optimum responses are obtained when these drugs are administered right to Isoproterenol CIonbuterol L - 40,033 Cimaterol BRL35135 FIGURE 1 Structures of the beta-adrenergic agonists isoproterenol, clenbuterol, L-640,033, cimaterol, and BBL35135.
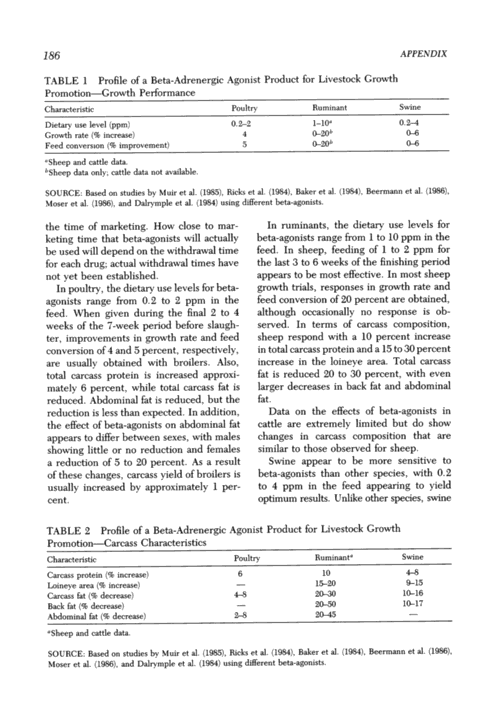
186 TABLE 1 Profile of a Beta-Adrenergic Agonist Product for Livestock Growth Promotion Growth Performance APPENDIX Characteristic Poultry RuminantSwine Dietary use level (ppm) 0.~2 l_l0a0.2 - Growth rate (% increase) 4 ~20b0~ Feed conversion (% improvement) 5 0-2050 6 a Sheep and cattle data. bSheep data only; cattle data not available. SOURCE: Based on studies by Muir et al. I_,, . _ . ~,, _ Moser et al. (1986), and Dalrymple et al. (1984) using different beta-agonists. the time of marketing. How close to mar- keting time that beta-agonists will actually be used will Repent] on the withdrawal time for each cirug; actual withdrawal times have not yet been established. In poultry, the dietary use levels for beta- agonists range from 0.2 to 2 ppm in the feed. When given during the final 2 to 4 weeks of the 7-week period before slaugh- ter, improvements in growth rate and feed conversion of 4 ant] 5 percent, respectively, are usually obtainer] with broilers. Also, total carcass protein is increased approxi- mately 6 percent, while total carcass fat is reduced. Abdominal fat is reduced, but the reduction is less than expected. In addition, the eject of beta-agonists on abclominal fat appears to differ between sexes, with males showing little or no reduction and females a reduction of 5 to 20 percent. As a result of these changes, carcass yield of broilers is usually increased by approximately 1 per cent. (1985). Ricks et al. (1984). Baker et al. (1984), Beermann et al. (1986), In ruminants, the dietary use levels for beta-agonists range from 1 to 10 ppm in the feed. In sheep, feeding of 1 to 2 ppm for the last 3 to 6 weeks of the finishing period appears to be most elective. In most sheep growth trials, responses in growth rate and feed conversion of 20 percent are obtained, although occasionally no response is ob- servecI. In terms of carcass composition, sheep respond] with a 10 percent increase in total carcass protein and a 15 to 30 percent increase in the loineye area. Total carcass fat is reduced 20 to 30 percent, with even larger decreases in back fat and abdominal fat. Data on the effects of beta-agonists in cattle are extremely limiter! but do show changes in carcass composition that are similar to those observer] for sheep. Swine appear to be more sensitive to beta-agonists than other species, with 0.2 to 4 ppm in the feed appearing to yield optimum results. Unlike other species, swine TABLE 2 Profile of a Beta-Adrenergic Agonist Product for Livestock Growth Promotion Carcass Characteristics Characteristic Poultry Ruminanta Swine Carcass protein (% increase) 6 10 4-8 Loineye area (% increase) 15-20 9-15 Carcass fat (% decrease) 4-8 20~0 10-16 Back fat (% decrease) 2~50 10-17 Abdominal fat (% decrease) 2-8 20~5 aSheep and cattle data. SOURCE: Based on studies by Muir et al. (1985), Ricks et al. (1984), Baker et al. (1984), Beermann et al. (1986), Moser et al. (1986), and Dalrymple et al. (1984) using different beta-agonists.
BETA-ADRENERG1C AGONISTS have failed in most reported trials to respond with improved growth rate or feed conver- sion. The studies in which improved growth performance was observed have used short- duration treatment (4 weeks or less). Swine do show very consistent improvement in carcass characteristics when medicated with beta-agonists. Total carcass protein is in- creased 4 to 8 percent, and loineye area muscle protein is increaser] 9 to 15 percent. Total carcass fat and back fat are reduced 10 to 17 percent. In addition to food animal species, beta- agonists are also very effective in laboratory animals. For example, clenbuterol has been shown to improve the growth performance and shift the carcass composition of young, rapidly growing male rats (Table 3; Rickes et al., 1985). Apparently, a dose of 10 ppm in the feed produces the maximum re- sponse: a 9 percent improvement in weight gain, a 10 percent improvement in feed conversion, a 9 percent increase in total carcass protein, and a 20 percent reduction in total carcass fat. These responses to beta- agonists in the rat are very similar to those observer! in food-producing animals. Thus, the rat appears to be an excellent model for studying beta-agonists as growth promoters. Beta-agonists have been examined for TABLE 3 Effects of a Beta-Adrenergic Agonist, Clenbuterol, on Growth Performance and Carcass Composition of the Rat (percent change over control) Clenbuterol, ppm in diet Characteristic 21050 Weight gain (g/day) Feed intake (g/day) Feed conversion (g feed/g gain) Carcass protein (g/carcass) 5.1*8.5**9.2** Carcass fat (g/carcass) - 8.5*- 19.9**- 23.3** 4.8* 9.6* 0.4 -3.4 8.5* 1.7 -3.6 - 10.7* - 7.2* *P < 0.05 compared with control. **P < 0.01 compared with control. SOURCE: Ricks et al. (1985). 187 their effects on milk production by dairy cows. Cows producing 17 to 18 kg of milk per day were medicated with the beta- agonists formoterol, zinterol, or Z1170. The beta-agonists were fed at 20 mg per head per day for 10 clays. Milk production on days 5 to 10 of treatment was not different from that of controls or from milk production before or 5 days after treatment. In addition, the composition of the milk was not altered. These data suggest that beta-agonists, unlike growth hormone, apparently are not able to stimulate milk production, even though both beta-agonists and growth hormone ap- pear to function through a repartitioning of nutrients. EFFECTS OF BETA-AGONISTS ON LIPID METABOLISM Free fatty acid (FFA) synthesis is the conversion of glucose, acetate, or both to free fatty acids. Lipogenesis is the sum of FFA synthesis and the esterification of FFAs to triglycerides (TGs). Lipolysis is the break- clown of TGs to FFAs and glycerol. The rate of glycerol production can be used to estimate lipolysis because the glycerol pro- ducecl during lipolysis cannot be reused for FFA esterification since adipocytes lack the necessary enzyme for phosphorylation of glycerol (phosphokinase). A scheme for the regulation of lipolysis by beta-agonists through specific adrenoceptors is shown in Figure 2. The activation of the beta-receptor on the outer surface of the aclipocyte plasma membrane activates the chain of events that eventually leads to the breakdown of stored triglycerides to FFAs and glycerol. Many beta-agonists effectively reduce lipic! accumulation in adipose tissue. The mech- anisms through which they act were studied at Merck in an in vitro system (Duquette and Muir, 1984~. Adipose tissue was taken from an animal source, for example, rat epicliclymal or perirenal fat, using the pro- ceclure of Rodbell (1964~. Aclipocytes were incubated with treatment for 2 hours at
188 APPENDIX Adipose Plasma Membrane Adrenergic Recept pAC~t~de; a Adrenergic Receptor | Inhibiting Peps - Guanino Nucleo~dde ,4N Phosphodl~terase c IMP ' 5'AMP l ATP Guanine Nucleotide Protein Klnase Active Hormone Inactive Hormone Sensitive Upase ~ Sensitive Upa" TrIglycerld" ~ · - Free Fatty Acids Glycerol FIGURE 2 Scheme for the regulation of lipolysis by beta-agonists through specific adrenoceptors. Source: Adapted from I. A. Garcia-Sarnz and J. N. Fain. 1982. Regulation of adipose tissue metabolism by catechol- amines: Roles of alphas, alphas, and beta-adrenoceptors. Trends Phar- macol. Sci. 3:201. 37°C. Lipolysis was estimated by measuring glycerol production. Glycerol was measured by a fluorometric mollification of the enzy- matic method of Wieland (1974). Lipoge- nesis was estimated by measuring the in- corporation of i4C-acetate into fatty acids from TGs. Isoprotereno} is very effective for stimu- lating lipolysis (glycerol release) in rat a(li- pose tissue in vitro. At a concentration of 0.001 ,uM, isoprotereno} had no effect on the basal rate of glycerol release; maximum stimulation occurred at 1 ,uM. The half- maximal effect dose was 0.017 ,uM. The idea that beta-agonists stimulate lipolysis through a specific beta-receptor is sup- portec! by the observation that the stimu- lation of lipolysis by isoproterenol can be blocked by beta-antagonists, such as pro- pranolol or betaxolol. Isoprotereno} was also studied for its effects on lipogenesis (~4C-acetate incorpo- ration into FFAs in TGs) in rat adipose tissue in vitro (Duquette and Muir, 1985~. Insulin was used in this study to increase lipogenesis and to simulate in viva condi- tions. Isoprotereno! at concentrations of 0. 01, 0. OS, and 0.25 EM reduced i4C-acetate incorporation into TGs in a close-related manner. In addition, when the TGs were hydrolyzes! and the resulting FFAs and glycerol were tested for i4C activity, the results showed that the effect of isoproter- enol was primarily to reduce the incorpo- ration of i4C-acetate into FFAs with only minor effects on glycerol. These observa- tions support the validity of this test system for estimating lipogenesis. These in vitro test systems for lipolysis and lipogenesis were used to compare the activities of four different beta-agonists. Iso- proterenol, clenbuterol, L-640,033, and BRL35135 were dose-titratec! in rat adipose tissue to study their effects on lipolysis and lipogenesis (Duquette ant] Muir, 1985). All four inhibited lipogenesis and stimulated
BETA-ADRENERGIC AGONISTS lipolysis. The intrinsic activity of all beta- agonists was similar for both actions. This means that the maximum effects of the beta- agonists, inclepenclent of dose, were of sim- ilar magnitude for each eject. Three of the four beta-agonists were 5 to 10 times more potent as inhibitors of lipogenesis than as stimulators of lipolysis; BRL35135 was equally potent for both. A comparison of the poten- cies of these drugs for inhibition of lipoge- nesis, as measured by 50 percent effective concentration, were isoproterenol > BRL35135 > L-640,033 > clenbuterol. Sim- ilar comparisons for stimulation of linolvsis animal were BRL35135 > isoproterenol > L-640,033 > clenbuterol. These observations suggest that in the animal a drug's efficacy for reducing belly lipids may be even more depenclent on that drug's activity for block- ing lipogenesis than for stimulating lipolysis. They also indicate that there is considerable variation between beta-agonists in their po- tencies for blocking lipogenesis and stimu- lating lipolysis. The ability of aclipocytes from different species to initiate lipolysis in re- sponse to similar concentrations of isopro- terenol was investigated by Muir et al. (1985~. Adipocytes from sheep, pigs, and rats all responder! with increases of 380 to 2,300 percent over their controls. Aclipo- cytes from chickens failer! to respond. Because chicken adipocytes failed to re- spond to isoproterenol, a more cletailec] study was conductecI. The beta-agonists iso- proterenol, clenbuterol, and L-640,033 and the positive control glucagon were all tested at concentrations of 0. 01, 0. 05, 0. 25, 1. 25, and 6.25 EM for effects on lipolysis in isolated chicken adipocytes (Muir et al., 1985~. As expected, the chicken adipocytes responded to glucagon with increased li- polysis. However, none of the three beta- agonists stimulated lipolysis at any of the concentrations tested. Since the chicken, unlike the other spe- cies studied, synthesizes FFAs in the liver, the ejects of beta-agonists on lipogenesis 189 in isolated chicken hepatocytes were inves- tigated. Isoproterenol, clenbuterol, and L- 640,033 and the positive control glucagon were tested on chicken hepatocytes in a design identical to the one describer! for chicken adipocytes (Muir et al., 1985~. As expected, glucagon caused a close-related inhibition in i4C-acetate incorporation into FFAs in TGs (that is, inhibition of lipoge- nesis). All three beta-agonists also caused a dose-relatec] inhibition of lipogenesis. Thus, beta-agonists block body fat accumulation in chickens by inhibiting lipogenesis in the liver, but they are not able to stimulate lipolysis in adipose tissue. In summary, beta-agonists have been shown to stimulate lipolysis and inhibit lipogenesis in the adipose tissue of the rat (Muir et al., 1985). In adipose tissue from sheep and swine, beta-agonists can stimu- late lipolysis, but no data are available regarding their effects on lipogenesis in these species. Beta-agonists are ineffective in chicken adipocytes. However, they in- hibit lipogenesis in chicken hepatocytes, the primary site for fatty acid synthesis in poultry. EFFECTS OF BETA-AGONISTS ON SKELETAL MUSCLE PROTEIN METABOLISM Skeletal muscle celDs have beta-receptors, ant! beta-agonists increase skeletal muscle pro- tein in animals. Ibus, beta-agonists might exert a direct effect on skeletal muscle cells, or their effect could be indirect through changes in plasma hormone concentrations or nutrient partitioning. Herbert et al. (1986) measurer] the effects of clenbutero! on urinary nitrogen excretion by sheep by infusing clenbutero! together with fee(lstuffs directly into the abom- asum. Within 6 hours after the initiation of infusion, nitrogen excretion dropped about 25 percent anc] remained clepresse ;1 over the entire 7-(lay test. These results suggest that clenbu- terol causer! an immediate improvement in nitrogen retention.
190 Muscle protein accumulation is the net balance of protein synthesis minus protein degradation. A drug like clenbutero} that dramatically increases muscle protein ac- cumulation might be expected to act by altering the rate of muscle protein synthesis, degradation, or both. This was examined by Reeds et al. (1986), who fed clenbuterol at O or 2 ppm in the diet to young male rats. The rates of protein synthesis in two skeletal muscles the gastrocnemius and soleus- were estimates] by the method of Garlick et al. (1980~. Rats were injected! with a large dose of labeled phenylalanine ant] killed 10 minutes later. The rate of protein synthesis was estimated from the amount of labeled phenylalanine in the muscle protein and from the specific activity of the free phen- ylalanine. Protein deposition was calculated from the slope of the line of the log of the protein content versus time. Protein content was estimates! from bocly weight. The rate of protein degradation was estimated from the differences between rates of synthesis and deposition. The effects of clenbuterol on muscle protein mass and rates of protein synthesis and degradation were determiner! on days 4, 11, ant! 21 of treatment. After only 4 days of clenbuterol treat- ment, the protein masses of the gastrocne- mius and soleus muscles were increased 17.7 and 50.6 percent, respectively, over the controls. These larger muscle protein masses were maintained throughout the 21- ciay test period. In both the gastrocnemius and soleus muscles, the rate of protein degradation was decreased on day 4 by 55 percent, with no change in the rate of protein synthesis. By clay 11, this decrease in the rate of protein degraciation relative to the controls was still evident, but the magnitude of the decrease was slightly less (39 en c] 25 percent for the gastrocnemius and soleus muscles, respectively). The rates of protein synthesis were still the same as for the controls. At clay 21, the rates of protein degradation were still reduced (20 related manner (Rickes et al., 1985~. Feed and 30 percent, respectively), but the rates consumption was increased only at the two APPENDIX of protein synthesis had decreased 20 per- cent relative to the controls. These observations suggest that clenbu- tero} increases skeletal muscle protein in the rat by reducing the rate of muscle protein degradation. Apparently, after mus- cle protein mass is increased by a certain amount, the rate of protein synthesis is reduced. At this point, the rate of muscle protein accumulation is reduced to normal, but the extra muscle protein mass is main- tained. SEPARATION OF LIPID AND PROTEIN EFFECTS OF BETA-AGONISTS The information presented thus far clem- onstrates that beta-agonists reduce the con- tent of lipids in the carcass, increase the accumulation of skeletal muscle protein, and improve the growth rate and feed conver- sion of the animal. To understand the mode of action of beta-agonists, studies were un- dertaken to determine whether the growth and feet] conversion responses were asso- ciated with the ejects of the beta-agonists on lipid metabolism, protein metabolism, or both. L-640,033 is an excellent growth-pro- moting beta-agonist. BRL35135 is a potent inhibitor of lipogenesis and a potent stim- ulator of lipolysis, but it does not appear to affect muscle protein metabolism. Both beta- agonists were evaluated in similar rat growth trials. In each trial, 110 young mate rats (10 per treatment and 20 controls) were feel the beta-agonist at 0, 0.25, 1.0, 2.5, 5.0, 10, 15, 25, or 50 ppm for 2 weeks. Clenbuterol at 10 ppm was used as a positive control. Rate of gain, feed intake, and feed conver- sion were determined. In addition, the weights of the gastrocnemius muscle and epididymal fat pads were measured to assess the ejects of the drugs on skeletal muscle protein and carcass fat. L-640,033 increased rate of gain in a dose
BETA-ADRENERGIC AGONISTS highest doses (25 and 50 ppm). Feed con- version was improved 4 to 6 percent, in- dependent of dose. Also, L-640,033 in- creased gastrocnemius muscle weight and decreased epididymal fat pad weight, both in dose-related manners (Rickes et al., 1985~. Both responses were similar to those ob- served with Clenbuterol at 10 ppm. BRL35135 had no effect on either rate of gain or feed conversion at any of the doses tested. Clenbuterol, the positive control, increased rate of gain and improved feed conversion. As expected, BRL35135 re- duced epididymal fat pad weight in a dose- related manner, with the maximum effect at 5 ppm. The reduction in epididymal fat pad weight with BRL35135 was significantly greater than that with clenbuterol. BRL35135 did not increase gastrocnemius muscle weight, while Clenbuterol increased it 17 percent. Thus, BRL35135 reduced carcass lipids in the rat without increasing skeletal muscle protein or improving rate of gain or feed conversion. These observations suggest that improve- ments in growth rate and feed conversion obtained with beta-agonists are associated with the effects of the drugs on skeletal muscle protein metabolism and not with their effects on lipid metabolism. Thus, while the ability of a beta-agonist to reduce carcass fat is an important benefit, this activity does not appear to be related to any growth-promoting activity. EFFECTS OF BETA-AGONISTS ON PLASMA HORMONES The effect of beta-agonists on plasma hormone levels is an important considera- tion when assessing the possible modes of action of these drugs in promoting growth. Thus, a study was carried out in which 50 young male rats (10 per treatment and 20 controls) were fed Clenbuterol at 10 ppm, BRL35135 at 15 ppm, or L-640,033 at 15 ppm for 2 weeks (L. A. Muir, unpublished data). At necropsy, gastrocnemius muscle 191 and epididymal fat pad weights were meas- ured to assess the effects of the beta-agonists on skeletal muscle protein and carcass lipids. In addition, blood samples were obtained and assayed for plasma growth hormone, insulin, somatomedin-C (SM-C), and glu- cose. All three beta-agonists produced the ex- pected changes in gastrocnemius muscle and epididymal fat pad weights. Clenbuterol and L-640,033 increased gastrocnemius muscle weight and decreased epididymal fat pad weights. BRL35135 decreased epi- didymal fat pad weight, but did not alter gastrocnemius muscle weight. Plasma in- sulin levels were decreased approximately 30 percent by Clenbuterol and L-640,033, but were not decreased by BRL35135. Sim- ilar effects were observed for SM-C. Plasma growth hormone was decreased by all three drugs, especially BRL35135, but the re- sponses were so variable that none of these growth hormone reductions was statistically significant. Clenbuterol decreased plasma glucose, while the other beta-agonists had no effect on glucose. Beermann et al. (1985) reported that cimaterol fed to lambs for 12 weeks reduced plasma insulin and elevated plasma T4 (thy- roid hormone thyroxine) but did not alter plasma levels of T3 (thyroid hormone tri- iodothyronine), cortisol, or prolactin. SUMMARY Beta-adrenergic agonists are analogs of the catecholamines epinephrine and nor- epinephrine. They appear to work through specific beta-adrenoceptors on the surface of adipocytes and skeletal muscle cells. Beta- adrenergic agonists that are known to pro- mote growth, such as clenbuterol, cima- terol, and L-640,033, improve the growth rate and feed conversion of sheep and poul- try. Effects on swine are more variable, while definitive data on cattle are not yet available. These drugs have also been shown to decrease total carcass fat and to increase
192 total carcass protein in all four animal spe- cies. Many beta-adrenergic agonists reduce carcass lipids by stimulating lipolysis and blocking lipogenesis in adipose tissue. The exception occurs in poultry, where these drugs inhibit lipogenesis in the liver but do not stimulate lipolysis in adipose tissue. Less is known about the effects of beta- adrenergic agonists on protein metabolism in skeletal muscle. However, recent studies suggest that some of these drugs increase skeletal muscle protein accretion by reduc- ing the rate of protein degradation without altering the rate of protein synthesis. Stud- ies in rats comparing the growth-promoting and carcass-altering effects of two beta- acirenergic agonists, L-640,033 and BRL35135, indicate that improvements in growth rate and feed conversion with beta- adrenergic agonists are associates! with im- proved protein accretion rather than altered lipid metabolism. Finally, growth-promot- ing beta-adrenergic agonists were found to reduce plasma levels of insulin and soma- tomedin-C in the rat but did not elevate plasma growth hormone levels. These ob- servations support the concept that growth- promoting beta-adrenergic agonists work directly through skeletal muscle cell recep- tors and not indirectly through the elevation of plasma growth hormone or insulin con- centrations. In addition, beta-adrenergic ag- onists that reduce carcass lipids appear to work directly through beta-adrenoceptors on the surface of aclipocytes in livestock and hepatocytes in poultry. ACKNOWLEDGMENT The author wishes to acknowledge the work of Paul Duquette, Eric Rickes, and Sandra Wien, who contributed to numerous aspects of Me beta-agonist research at Merck, and Dr. Y. T. Yang, whose ideas and re- search finclings supported our beta-agonists research program. APPENDIX REFERENCES Arch, J. R. S., M. A. D. Phil, and A. T. Ainsworth. 1983. Thermogenic and antiobesity activity of a novel beta-adrenoceptor agonist (BRL26830A) in mice and rats. Am. J. Clin. Nutr. 38:549. Arch, J. R. S., A. T. Ainsworth, M. A. Cawthorne, V. Piercy, M. V. Sennitt, V. E. Thody, C. Wilson, and S. Wilson. 1984. Atypical beta-adrenoceptor on brown adipocytes as target for anti-obesity drugs. Nature 309:163. Baker, P. K., R. H. Dalrymple, D. L. Ingle, and C. A. Ricks. 1984. Use of a beta-adrenergic agonist to alter muscle and fat deposition in lambs. J. Anim. Sci. 59:1256. Beermann, D. H., W. R. Butler, D. E. Hogue, R. H. Dalrymple, and C. A. Ricks. 1985. Plasma metabolic hormone, glucose, and free fatty acid concentrations in lambs fed the repartitioning agent, cimaterol (CL 263,780~. J. Anim. Sci. 61(Suppl. 1~:254 (Abstr.). Beermann, D. H., D. E. Hogue, V. K. Fishell, R. H. Dalrymple, and C. A. Ricks. 1986. Effects of cima- terol and fishmeal on performance, carcass charac- teristics and skeletal muscle growth in lambs. J. Anim. Sci. 62:370. Burroughs, W., C. C. Culbertson, J. Kastelic, E. Cheng, and W. H. Hale. 1954. The effects of trace amounts of diethylstilbesterol in rations of fattening steers. Science 120:66. Dalrymple, R. H., P. K. Baker, P. E. Gingher, D. L. Ingle, J. M. Pensack, and C. A. Ricks. 1984. A repartitioning agent to improve performance and carcass composition of broilers. Poultry Sci. 63:2376. Davis, L. W. 1969. MGA- A new concept in heifer feeding. Pp. 72-82 in Proceedings of the 24th Kansas Formula Feed Conference, Manhattan: Kansas State University of Agricultural and Applied Sciences. Dinusson, W. E., F. N. Andrews, and W. M. Beeson. 1950. The effects of stilbesterol, testosterone, thyroid alteration and spaying on the growth and fattening of beef heifers. J. Anim. Sci. 9:321. Duquette, P. F., and L. A. Muir. 1984. Effects of ovine growth hormone and other anterior pituitary hormones on lipolyis of rat and ovine adipose tissue in vitro. J. Anim. Sci. 58:1191. Duquette, P. F., and L. A. Muir. 1985. Effect of the beta-adrenergic agonists isoproterenol, clenbuterol, L-640,033 and BRL35135 on lipolysis and lipogenesis in rat adipose tissue in vitro. J. Anim. Sci. 61(Suppl. 1~:265 (Abstr.). Garcia-Sarnz, J. A., and J. N. Fain. 1982. Regulation of adipose tissue metabolism by catecholamines: Roles of alphas, alphas and beta-adrenoceptors. Trends Pharmacol. Sci. 3:201. Garlick, P. S., M. A. McNurlan, and V. R. Preedy. 1980. A rapid and convenient technique for meas
BETA-ADRENERGIC AGONISTS uring the rate of protein synthesis in tissues by injection of"3H" phenylalanine. Biochem. J. 192:719. Heitzman, R. J. 1980. Manipulation of protein metab- olism, with special reference to anabolic agent. Pp. 193-203 in Protein Deposition in Animals, P. J. Buttery and D. B. Lindsay, eds. Boston: Butter- worth. Herbert, F., F. D. DeB. Hovell, and P. J. Reeds. 1986. Some preliminary observations on the im- mediate effects of clenbuterol on heart rate, body temperature and nitrogen retention in lambs wholly nourished by intragastric infusion. Br. J. Nutr. 56:483 (Abstain. Machlin, L. J. 1972. Effect of porcine growth hormone on growth and carcass composition of the pig. J. Anim. Sci. 35:794. Moser, R. L., R. H. Dalrymple, S. G. Cornelius, J. E. Pettigrew, and C. E. Allen. 1986. Effect of cimaterol (CL 263,780) as a repartitioning agent in the diet for finishing pigs. J. Anim. Sci. 62:21. Muir, L. A. 1985. Mode of action of exogenous sub- stances on animal growth an overview. J. Anim. Sci. 61(Suppl. 2):154. Muir, L. A., M. W. Stutz, and G. E. Smith. 1977. Feed additives. Pp. 27-37 in Livestock Feeds and Feeding, D. C. Church, ed. Corvallis, Oreg.: ORB Books. Muir, L. A., S. Wien, P. F. Duquette, E. L. Rickes, and E. H. Cordes. 1983. Effects of exogenous growth hormone and diethylstilbesterol on growth and car 193 cass composition of growing lambs. J. Anim. Sci. 56:1315. Muir, L. A., S. Wien, P. F. Duquette, and G. Olson. 1985. Effect of the beta-adrenergic agonist L-640,033 on lipid metabolism, growth and carcass character- istics of female broiler chickens. J. Anim. Sci. 61(Suppl. 1~:263 (Abstr. ). Reeds, P. J., S. M. Hay, P. M. Dorwood, and R. M. Palmer. 1986. Stimulation of muscle growth by clenbuterol: Lack of effect on muscle protein bio- synthesis. Br. J. Nutr. 56:249. Rickes, E. L., L. A. Muir, and P. F. Duquette. 1985. Effect of the beta-adrenergic agonist L-640,033 on growth and carcass composition of growing male rats. J. Anim. Sci. 61(Suppl. 1):264 (Abstr. ). Ricks, C. A., R. H. Dalrymple, P. K. Baker, and D. L. Ingle. 1984. Use of beta-agonist to alter fat and muscle deposition in steers. J. Anim. Sci. 59:1247. Rodbell, M. 1964. Metabolism of isolated fat cells. I. Effects of hormone on glucose metabolism and lipolysis. J. Biol. Chem. 239:375. Wagner, J. F., and E. L. Veenhuizen. 1978. Growth performance, carcass deposition and plasma hor- mone levels in wether lambs when treated with growth hormone and thyroprotein. J. Anim. Sci. 47(Suppl. 1):397. Wieland, O. 1974. Glycerol UV-method. Pp. 140( 1409 in Methods of Enzymatic Analysis, H. U. Bergmeyer, ed. New York: Academic Press.
Anabolic EFects of Porcine Somatotropin on Pig Growth TERRY D. ETHERTON Animal agriculture must develop ways to enhance the growth performance of animals raised for meat production in order to pro- vide consumers with a product that is leaner and, therefore, more nutritious. Because leaner meat products will be sought by consumers concerned about the relation between the consumption of saturated fatty acids and the incidence of coronary heart disease, strategies to increase growth rate and improve feed efficiency (ratio of feed consumed to bocly weight gained) will eco- nomically benefit producers. The central question is, what research options are avail- able now and in the foreseeable future that may provide effective ways to manipulate meat animal growth performance? This paper focuses on the concept that an elevation of blood concentrations of growth hormone (GH, or somatotropin) in meat animals markedly increases growth rate, improves feed efficiency, and dramatically increases muscle mass while decreasing adi- pose tissue (fat) mass (Chung et al., 1985; Etherton et al., 1986a, 1986b, 1987; Mach- lin, 1972~. Table 1 shows the extent to which growth hormone can affect the growth per- formance of pigs. The stimulatory effects of GH on growth performance have created great interest in developing a GH-based product for practical use in animal agricul- ture. In fact, it is likely that such a product will be available for use within the next 2 to 3 years. The mechanisms by which GH works are discussed in the following section, since a better understancling of them may leac! to ways to improve the effectiveness of GH or of alternative strategies for enhancing growth performance. Growth hormone is a protein that is synthesized in the anterior pituitary gland of mammals. It plays a central role in stimulating normal growth and is both an- abolic ant! catabolic in that it stimulates growth rate ant! muscle accretion ant! con- currently decreases adipose tissue growth (Etherton et al., 1986b, 1987~. The positive effects of GH on growth rate are indirect, being mecliated largely by the GH-clepend- ent insulin-like growth factor I (IGF-I, or somatomedin-C) (Etherton ant! Kensinger, 1984~. The effects of GH on adipose tissue growth and metabolism are direct, not being mediated by IGF-I (Walton and Etherton, 1986; Walton et al., 1986, 1987a). Observations by Etherton and coworkers 194
ANABOLIC EFFECTS OF PORCINE SOMATOTROPIN TABLE 1 Effects of Porcine Growth Hormone on Growth Performance (in percent) Dosage of Porcine Growth Hormonea (,ug/kg body wt) Performance Factor 35 70 Carcass lipid Muscle Feed/gain -29 +21 -12 -32 +26 -21 a Dose of porcine GH given daily per kilogram of body weight for 77 days by intramuscular injection. Values are percentage response versus that for control pigs. For example, carcass lipid decreased 68 percent, mus- cle mass increased 24 percent, and the amount of feed consumed per unit body weight gain decreased 24 percent in pigs given 140 ,ug/kg body wt. SOURCE: Adapted from T. Etherton, C. Evock, C. Chung, P. Walton, M. Sillence, K. Magri, and R. Ivy. 1986. Stimulation of pig growth performance by long- term treatment with pituitary porcine growth hormone (pGH) and a recombinant pGH. J. Anim. Sci. 63(Suppl. 1~:219. that GH enhances growth performance sug- gest that any strategy that increases the blood levels of GH should be a viable approach for manipulating growth perform- ance. However, the extent to which GH must be elevated and for how long is still not clear. Since administration of exogenous GH effectively stimulates growth perform- ance, it is reasonable to ask whether an increase in the secretion of endogenous GH can lead to similar anabolic effects. Implicit in this question is that tools are available that allow GH secretion to be modulated. In 1982, a hypothalamic nentide that stimulates GH release was purified from tumors of patients with acromegaly (Guil- lemin et al., 1982; Rivier et al., 1982~. Subsequently, the growth hormone releas- ing factor (GRF) from porcine ant! bovine hypothalami was purified and sequencer! (BohIen et al., 1983; Esch et al., 1983~. GRF is a potent, specific stimulant of GH release in pigs and cattle (Etherton et al., 1986b; Moseley et al., 1984~. Aside from 195 studies done in our laboratory (Etherton et al., 1986b), there are few published data on the effects of long-term administration of GRF on growth performance of meat ani- mals. Our studies have shown positive ef- fects; however, the magnitude has been less 140 than that observed for GH-treated nips -68 +24 -24 (Etherton et al., 1986b). Because so little is known about the optimal dose and temporal profile of GRF administration, it is prema- ture to conclude that GRF is less effective than GH treatment. Nonetheless, it has been establisher] that chronic GRF treat- ment does not result in pituitary refracto- riness to the stimulatory effects of GRF. This indicates that the capacity of the pi- tuitary to respond to GRF is not (liminished over time, thereby suggesting that GRF treatment is a viable candidate for promot- ing growth. Furthermore, GRF analogs have been synthesized that are considerably more potent than the 44-amino-aci:1 pepticle syn- thesized in the hypothalamus and that there- fore may be more effective in promoting growth than the naturally occurring peptide. The counter-regulatory peptide to GRF is somatostatin, which inhibits GH release. The paper by Schelling and Byers in this volume discusses neutralization of somato- statin as a possible means to enhance growth performance. It is likely that concurrent stimulation of GRF and inhibition of so- matostatin would enhance growth perform- ance more than either approach alone. The elevation of blood GH by exogenous GH treatment leacls to a cascade of biological events that account for the increase in growth performance. One such event is an increase in the level of somatomedin (IGF- I) in the blood. Given that many of the somatogenic effects of GH appear to be mediated by IGF-I, it is reasonable to speculate that administration of exogenous IGF-I may be a feasible way to enhance growth performance. But because sufficient quantities of recombinant IGF-I are scarce, this hypothesis has not yet been tested in meat animals.
196 At least two points must be considered regarding IGF-I treatment as a potential growth promoter. First, it is transported by a specific carrier protein (Martin and Baxter, 1986; Zapf et al., 1975) that also affects bioavailability of the hormone to the target tissue. For example, free (unbound) IGF-I . . . . . . . . ~ - 1S an 1nsu in mimic in Bovine and porcine adipose tissue in terms of stimulating lipid metabolism (Etherton and Evock, 1986; Walton et al., 1987a), but addition of the carrier protein blocks these effects in rat and pig adipose tissue (Walton et al., 1987b). Because the circulating carrier protein is normally relatively saturated with IGF-I in serum from pigs (P. Walton and T. Ether- ton, unpublished data), exogenously admin- istered IGF-I would be mostly free in the blood and act as an insulin mimic, resulting in hypoglycemia. Guler et al. (1986) have demonstrated this in mini-pigs treated with a bolus dose of IGF-I. More recent studies with pigs in our laboratory with recombinant human IGF-I have confirmed this (R. Go- pinath and T. Etherton, unpublished data). However, the effects of IGF-I on growth performance in meat animals when the dose is below that which causes hypoglycemia and is given in a sustained manner still must be determined. A central question that arises is whether chronic treatment of ani- mals with IGF-I stimulates expression of the gene that encodes for the carrier protein. If not, then exogenous treatment of meat animals with IGF-I may not be an effective approach to growth manipulation since hy- poglycemia would ensue. Also, when pigs are treated with GH, the increase in cir- culating IGF-I concentration is associated with a concurrent increase in carrier protein (P. Walton and T. Etherton, unpublished data). The second point to consider in IGF-I treatment pertains to the effects of GH on adipose tissue metabolism. In cultured por- cine and bovine adipose tissue, GH antag- onizes insulin action (Etherton and Evock, 1986; Walton et al., 1986~. In viva, treat- ment of pigs with GH decreases the rate of APPENDIX synthesis of fatty acids and markedly blunts the sensitivity and responsiveness of adi- pocytes to insulin and free IGF-I (Walton et al., 1987a). Therefore, it seems unlikely that IGF-I treatment will decrease adipose tissue growth when the effects of the free hormone are stimulatory in the tissue and, hence, anabolic. It has been routinely found that in pigs treated with GH (Etherton et al., 1986b, 1987), adipose tissue growth is decreased rather than increased. Thus, the marked increase in IGF-I concentration in pigs treated with GH is not associated with an increase in adipose tissue growth. This suggests that the inhibition of the insulin- like effects of free IGF-I by the binding protein observed in vitro in pig adipose tissue also occurs in viva (P. Walton and T. Etherton, unpublished data). And because the sensitivity of pig adipocytes to free IGF- I is blunted by GH in viva, it appears that even if there were an increase in free IGF- I in blood or tissue, the adipocytes would be less responsive to the insulin-like effects of free IGF-I. The unique characteristic of GH that appears to account for the remarkable im- provement in feed efficiency of GH-treated animals is that it decreases adipose tissue growth. In the pig, this is associated with a decrease in the rate of fatty acid synthesis. This adaptation by the a(lipocytes results in a redirection of the nutrients from adipose tissue to other target tissues (such as mus- cle). This change in nutrient utilization not only accounts for the decrease in adipose tissue growth but also offers a possible explanation for how muscle growth is in- creased. In all our studies, we have con- sistently noted that blood urea nitrogen (BUN) is decreased in a GH-dependent manner. This indicates that hepatic amino acid oxidation is decreased, which, in turn, infers that delivery of amino acids from peripheral tissues (such as muscle) is de- creased. The glucose carbon normally oes- tined for deposition in adipose tissue may spare amino acids from being oxidized in muscle, thereby providing more amino acids
ANABOLIC EFFECTS OF PORCINE SOMATOTROPIN for protein synthesis. Also, in tissue culture, IGF-I inhibits myofibrillar protein ~legra- ciation and increases the rate of protein synthesis (Ewton and Florini, 1986~. Col- lectively, these observations illustrate the remarkable coordination of nutrient parti- tioning that occurs among the various tissues in an animal. It is only now that we are beginning to appreciate the extent to which nutrient partitioning can be manipulated. For some time endocrinologists have rec- ognized that circulating protein hormones play a role in regulating the number and affinity of their respective receptors in the target cell. For example, treatment of rats with insulin has been shown to result in a decrease (clown regulation) of insulin bind- ing. Thus, a point of regulation occurs when the target cell recognizes the hormone. It may well be that differences exist among meat animals and that this contributes to the variations seen in growth performance. We thought that chronic treatment of pigs with GH might induce down regulation of GH binding and result in a decrease in tissue sensitivity. To assess this we meas- ured GH binding to membranes prepared from livers from pigs treater! with different doses of GH for 35 clays. In contrast to our original speculation, GH treatment in- creased binding (Chung ant] Etherton, 1986), suggesting that tissue sensitivity to GH increases after treatment. If this is indeed! the case, then future research must focus on the mechanisms that regulate GH re- ceptor number, since this information may lead to alternative strategies for increasing growth performance. In particular, it will be important to increase our understanding of how the GH receptor produces signals that alter cell function so markedly ant] how this differs among the different target tissues (for example, muscle, liver, fat). PROSPECTS FOR INCREASING GROWTH PERFORMANCE Growth hormone clearly increases meat animal growth performance, but before a 197 GH-based product is developed for appli- cation in animal agriculture, two questions must be answered. First, will recombinant DNA technology enable sufficient quantities of the protein to be producer] at a cost that will not limit product development? Little concrete information has been published on this topic; however, it is our belief that this is no longer a problem. Second, and more important, how will GH be administered at the farm level? Daily injections are imprac- tical for large-scale production. Thus, a delivery system must be developed where GH is administerecl in a vehicle that pro- vides for controller] delivery of the protein over a sustainer} period! of time (e.g., 30 days). At present, this is the limiting step in the development of a GH-based product, although intense research in the area is ongoing. It is possible that alternative means can be developed for enhancing growth per- formance, but it has been questioned whether there is really any need for them "after" GH. We contend that there is incleec] such need. For one thing, strategies may be developed that improve the effectiveness of GH or that are synergistic to GH. In fact, there are data from studies that indicate that such approaches are possible. It has been found that blocking adrenal function significantly enhances responsiveness to GH in rats treated chronically with GH and trilostane (Sillence et al., 1987~. Trilostane is a specific inhibitor of glucocorticoid hor- mone synthesis. It remains to be established whether trilostane also enhances GH po- tency in pigs. Several conceptual approaches to manip- ulating growth performance have been pre- sented, and it is likely that one or more will be developer] into a product that dramati- cally affects animal agriculture. It is also reasonable to assume that other strategies will evolve as our unclerstan(ling of the biological mechanisms that regulate growth and nutrient partitioning increases. For in- stance, certain monoclonal antibodies to GH, when complexed to the hormones,
198 increase its bioactivity in mice (Holder et al., 19854. An exciting era is evolving in animal agriculture. We now have available the means to alter growth performance in a way that is beneficial to both the producer and the consumer. It should be emphasized that attempts to manipulate meat animal growth perform- ance are not uncler way solely to produce larger animals that grow more rapidly but, rather, to enhance the efficiency of growth performance. Thus, it is remarkable that GH has its greatest ejects on feed efficiency and carcass composition. In terms of the former, it is reasonable to conclude that the savings in feed costs could be on the order of $1 billion to $3 billion a year in the United States alone. This, along with the dramatic beneficial changes observed in carcass com- position, sufficiently illustrates the need for this technology and its benefit to animal agriculture and society. REFERENCES Bohlen, P., F. Esch, P. Brazeau, N. Ling, and R. Guillemin. 1983. Isolation and characterization of the porcine hypothalamic growth releasing factor. Biochem. Biophys. Res. Commun. 116:726. Chung, C. S., and T. Etherton. 1986. Characterization of porcine growth hormone (pGlI) binding to porcine liver microsomes: Chronic administration of pGH induces pGH binding. Endocrinology 119:780. Chung, C. S., T. Etherton, and J. Wiggins. 1985. Stimulation of swine growth by porcine growth hormone. J. Anim. Sci. 60:118. Esch, F., P. Bohlen, N. Ling, P. Brazeau, and R. Guillemin. 1983. Isolation and characterization of the bovine hypothalamic growth hormone releasing factor. Biochem. Biophys. Res. Commun. 117:772. Etherton, T., and C. Evock. 1986. Stimulation of lipogenesis in bovine adipose tissue by insulin and insulin-like growth factor. J. Anim. Sci. 62:357. Etherton, T., and R. Kensinger. 1984. Endocrine regulation of fetal and postnatal meat animal growth. J. Anim. Sci. 59:511. Etherton, T., C. Evock, C. Chung, P. Walton, M. Sillence, K. Magri, and R. Ivy. 1986a. Stimulation of pig growth performance by long-term treatment with pituitary porcine growth hormone (pGH) and a recombinant pGH. J. Anim. Sci. 63(Suppl. 1):219. APPENDIX Etherton, T., J. Wiggins, C. Chung, C. Evock, J. Rebhun, and P. Walton. 1986b. Stimulation of pig growth performance by porcine growth hormone and growth hormone-releasing factor. J. Anim. Sci. 63:1389. Etherton, T., J. Wiggins, C. Evock, C. Chung, J. Rebhun, P. Walton, and N. Steele. 1987. Stimulation of pig growth performance by porcine growth hor- mone: Determination of the dose-response relation- ship. J. Anim. Sci. 64:433. Ewton, D., and J. Florini. 1986. Binding to and actions of somatomedins on myoblasts and myotubes: Dem- onstration of actions mediated by the type I receptor. Paper presented at the 68th annual meeting of The Endocrine Society. P. 167. Bethesda, Md.: The Endocrine Society. Guillemin, R., P. Brazeau, P. Bohlen, F. Esch, N. Ling, and W. Wehrenbert. 1982. Growth hormone- releasing factor from a human pancreatic tumor that caused acromegaly. Science 218:585. Guler, H., P. Zenobi, J. Zapf, E. Scheiwiller, J. Merryweather, C. Scandella, W. Marki, and E. Froesch. 1986. IGF-I and II and recombinant human (RH) IGF-I are hypoglycemic in the rat, mini-pig, and men. Paper presented at the 68th annual meet- ing of The Endocrine Society. P. 129. Bethesda, Md.: The Endocrine Society. Holder, A., R. Aston, M. Preece, and J. Ivanyi. 1985. Monoclonal antibody-mediated enhancement of growth hormone activity in viva. J. Endocrinol. 107:R9. Machlin, L. 1972. Effect of porcine growth hormone on growth and carcass composition of the pig. J. Anim. Sci. 35:794. Martin, J., and R. Baxter. 1986. Insulin-like growth factor-binding protein from plasma: Purification and characterization. J. Biol. Chem. 261:8754. Moseley, W., L. Krabill, A. Friedman, and R. Olsen. 1984. Growth hormone response of steers injected with synthetic human pancreatic growth hormone- releasing factor. J. Anim. Sci. 58:430. Rivier, J., J. Spiess, M. Thorner, and W. Vale. 1982. Characterization of a growth hormone-releasing fac- tor from a human pancreatic islet tumor. Nature 300:276. Sillence, M. N., T. D. Etherton, and K. A. Magri. 1987. Growth response of normal female rats to porcine growth hormone is improved by trilostane. Paper presented at the 69th Annual Meeting of The Endocrine Society. Bethesda, Md.: The Endocrine Society. Walton, P., and T. Etherton. 1986. Stimulation of lipogenesis by insulin in swine adipose tissue: An- tagonism by porcine growth hormone. J. Anim. Sci. 62:1584. Walton, P., T. Etherton, and C. Evock. 1986. Antag
ANABOLIC EFFECTS OF PORCINE SOMATOTROPIN onism of insulin action in cultured pig adipose tissue by pituitary and recombinant porcine growth hor- mone: Potentiation by hydrocortisone. Endocrinol- ogy 118:2577. Walton, P., T. Etherton, and C. Chung. 1987a. Ex- ogenous pituitary and recombinant porcine growth hormones induce insulin and insulin-like growth factor I resistance in pig adipose tissue. Dom. Anim. Endocrinol. 4:183. 199 Walton, P. E., R. Gopinath, B. D. Burleigh, and T. D. Etherton. 1987b. An acid-stable subunit of por- cine serum IGF binding protein specifically blocks biological action of IGF-I on adipose tissue. I. Anim. Sci. 65(Suppl. 1):274. Zapf, J., M. Waldvogel, and E. Groesch. 1975. Binding of nonsuppressible insulin-like activity to human serum. Evidence for a carrier protein. Arch. Biochem. Biophys. 1687:638.
Immunization of Beef Cattle Against Somatostatin G. T. SCHELLING and F. M. BYERS The roles and interactions of somatostatin, growth hormone releasing factor, somato- tropin, ant] somatomedin are of great bio- logical interest and appear to offer consid- erable potential for the modification of grown and production in food-producing animals. It is now clear that single hormones do not function independently to regulate growth. Rather, a number of events or cascading effects come into play to generate animal growth responses. Somatotropin (ST) has been recognized as an important growth regulating factor for some time. Li (1973) clemonstrated its an- abolic nature relative to protein synthesis, and Raben (1973) demonstrated its catabolic nature through its ability to stimulate lipol- ysis. Daughaclay et al. (1972) indicated that somatomeclin (S M) was a factor incluced by plasma ST, and Thorner et al. (1982) isolated several growth hormone releasing factor (GRF) peptides that incluced ST release. The liver release of SM may require insulin (Schalch et al., 1979), and thyroxine may be requires! at the tissue level for SM to be elective (Froesch et al., 1976~. The rela- tionships among these agents became clearer when Vale et al. (1976) demonstrated that somatostatin (SS) inhibited ST release (Fig 200 ure 11. It is also recognized that SS inhibits thyrotropin (VaIe et al., 1976~; insulin and glucagon (Mortimer et al., 1974~; and gas- trin, gastric inhibitory peptide, pepsin se- cretion, motilin, and vasoactive intestinal polypepti(le (C. N. Bloom et al., 1974; S. R. Bloom et al., 1974~. The current general state of knowledge indicates the potential to stimulate growth by exogenously provid- ing a(lditional GRF, ST, or SM. This paper explores the potential for achieving stimu- latec! growth by immunologically alleviating the inhibitory effects of SS. Antibody bind- ing of SS would reduce available SS and therefore shift the balance of control toward GRF, which presumably would result in greater ST production (Figure 21. The other inhibitory roles of SS cannot be neglected, however, since these influences could ben- efit the animal through enhanced metabo- lism or improver! nutrient uptake and uti- lization. METHODS AND RESULTS Passive Immunization Against Somatostatin Immunological approaches to generate antibodies for binding biologically active
IMMUNIZATION OF BEEF CATTLE f somatostatin 1 Inhibitory I ~omatotropln I Growth Hormone Stimulatory '' Releasing Factor 201 | Insulin 1 Dependent -I I Stimulatory I ~ Stimulat°tY | Cellular | | Thyroxine | Dependent FIGURE 1 General hormonal relationships leading to increased growth. molecules have been used as an analytical laboratory technique for some time. Sub- sequent studies extended passive immuni- zation to animal studies. Arimura et al. (1975) generated SS-specific antiserum to stucly the eject of inhibiting SS on serum ST concentrations. The passive immuniza- tion of rats resulted] in increased ST levels (Ferland et al., 1976), and rats injected with SS antiserum had a surge in plasma ST that continued for a 6-hour sampling period] (Tannenbaum et al., 1978~. Arimura and Fishback (1981) administered! SS antiserum to rats daily for 3 clays and found that the mean serum ST of the treated rats was 76 ng/ml, while that of the control rats was 16.8 ng/ml. Lawrence et al. (1985) conducted long- term passive immunization studies with rats to study ST concentrations an(l animal re- sponses. After SS antiserum was produced in goats, rats were prepared with indwelling jugular vein catheters to facilitate the daily SS antiserum administration for 21 (lays. During this time, serum ST levels and performance criteria were measurer] weekly. An ST response was evident and peaked by day 10, when the immunizes! animals showed Effect of Deactivation of Somatostatin on Growth Hormone _~N Immunologic Deactivation /\ | Growth l | Hormone | Muscle Tissue Growth - FIGURE 2 Imbalance created by deactivation of somatostatin. . j Release Factor
202 an ST level of 184.9 nglml as compared with 7.6 ng/ml for the controls. However, the ST response diminished with time, and the levels in the test rats were similar to those in the controls by clay 20. Table 1 indicates the weekly gains of the animals. There was a significant (P < 0.05) gain response during the first week, but the response gradually climinished over the next 2 weeks. This loss of response with time may have been clue to secondary immunological responses, since whole serum was administered to the rats as the source of antibodies. Active Immunization Against Somatostatin Hormonal Responses Active immunity techniques appear to provide the best approach for taking advan- tage of the potential to immunize against SS. They involve establishing immunity to SS in the target animal by immunizing it with SS conjugates] to an appropriate im- munostimulating molecule (Figure 3~. The animal should falsely recognize SS as a foreign molecule ant] begin the active pro- duction of antibodies that would bind the SS producer] by the animal, thereby reduc- ing available SS. Somato) ecstatic J Immune | Response Stimulator Immunizations Against Somatostatin Combine Iniect (Manufacture) (Vaccinate) APPENDIX TABLE 1 Average Weekly Gain of Rats Passively Immunized Against Somatostatin (in grams) Treatment Week 1 Week 2 Week 3 Control 6.0 6.8 Immunized 7. 4a 6.8 5.5 5.7 a Different from the control (P < 0.05). SOURCE: M. E. Lawrence, G. T. Schelling, T. W. Welsh, F. M. Byers, and L. W. Greene. 1985. Effect of passive immunization against Somatostatin on plasma growth hormone and performance of growing rats. J. Anim. Sci. 61(Suppl. 1):47. To study hormonal responses, Varner et al. (1980) actively immunized lambs against SS with an egg albumin-conjugated SS. The episodic nature of ST was thoroughly stud- iec3 by taking frequent blood samples. Mean baseline and overall mean ST concentrations were significantly greater in the treated lambs (Table 2~. The frequency of ST peaks was not influenced by the treatment, and peak amplitude was not statistically different because of high variation. A number of other studies have not in- dicate(1 a statistically significant ST response to active immunization in sheep (Chaplin et al., 1984; Galbraith et al., 1985; Spencer and Williamson, 1981~. Similar studies with growing cattle (Lawrence et al., 1986) Immunize (Inactivate Somatostatin) FIGURE 3 Generation of antigen and subsequent Somatostatin antibody production.
IMMUNIZATION OF BEEF CATTLE TABLE 2 Somatotropin Response to Active Immunization of Lambs Against Somatostatin Variable Control Immunized P Baseline 4.0 6.8 <0. 05 Amplitude (ng/ml) 10.0 18.1 NSa Overall mean (ng/ml) 4.2 7.1 <0.05 Frequency (spikes/12 hours) 1.7 2.0 NS aNS = not significant. SOURCE: M. A. Varner, S. L. Davis, and J. J. Reeves. 1980. Temporal concentrations of growth hormone, thyrotropin, insulin and glucagon in sheep immunized against somatostatin. Endocrinology 106:1027. weighing 250 or 350 kg not only showed no positive ST response, but trends were to- ward lower ST levels with active immuni- zation. Although some studies have indi- cated the ability of metabolically challenged immunized sheep to- yield a greater ST response (Spencer et al., 1983b; Varner et al., 1980), GRF challenge studies with ac- tively immunized growing beef cattle (D. C. Kenison, G. T. Schelling, F. M. Byers, and L. W. Greene, unpublished data) re- sultecl in only slightly higher ST responses. While ST level and challenge studies do not provide convincing evidence of consis- tent ST responses to active immunization, subtle ST changes of biological importance should not be ruled out, since thorough studies of the measurement of ST turnover have not yet been reported. Increased ST turnover rate in actively immunized animals could be responsible for the inconsistent responses in the other ST parameters re- ported. A general ST response is supported by reports of consistent SM responses in sheep actively immunized against SS (Spen- cer and Williamson, 1981; Spencer et al., 1983b). An SM response would presumably be mediated through ST. Growth Responses Spencer and Williamson (1981) were the first to show that lambs actively immunized against somatostatin gainer! weight more rapidly than lambs not actively immunized. 203 Lambs immunized at 3 weeks of age and at regular intervals thereafter with SS conju- gatec! to human serum globulin gainer] weight faster than their twin counterparts injected with human serum globulin alone. This finding was in contrast to the earlier results of Varner et al. (1980~. The lack of a growth response in the work of Varner et al. (1980) was probably due to the nearly mature sheep used in that study. The experiment was designed to specifically investigate hor- monal responses, and young, rapidly grow- ing animals were not used. Recent work by Spencer et al. (1983b), which is more cle- tailed in nature, resulted in similar positive gain responses with growing lambs immu- nized against SS. Chaplin et al. (1984) immunized lambs at 3 weeks of age and every 11 days thereafter for 103 clays. The treated lambs gained weight 16.6 percent faster than the control animals. Other lamb studies by Bass et al. (1983), Spencer et al. (1985), and Spencer and Hallett (1985) have confirmed a rate of gain response with immunization against SS. Lawrence et al. (1986) conducted im- munization studies with 26 young, rapidly growing, Charolais crossbred steers weigh- ing 147 kg. Each treated steer was injected with protein-conjugated SS every 2 weeks during a 154-day study, and each control steer was injected with the conjugated pro- tein alone. Blood samples, taken every 2 weeks, indicated the development of a sig
204 nificant SS antibody titer in each treated steer. A response in gain was observed after 56 days and was maintained (P ~ 0.001) throughout the 154-day study (Table 3~. The treated cattle gained weight 17.6 percent faster than did the control cattle over the 154-day study. There was a positive corre- lation (P c 0.001) between SS antibody titer and average daily gain of the immunized steers. Other work also indicates a gain response in beef castle. G. T. Schelling, F. M. Byers, and L. W. Greene (unpublished data) stud- iec! 28 crossbred beef steers on ryegrass pasture. Fourteen were immunized against SS at the beginning of a 105-day trial and then twice during the trial. The control steers received placebo injections at the same time intervals. Table 4 shows that the treated cattle gained 115 kg compared with 103 kg for the control cattle, for an 11.6 percent gain response (P < 0.05) during the study. Another study, with a limited num- ber of young dairy calves (Vicini et al., 1986), indicated that immunized calves gained more weight. The efficiency of feed utilization is of utmost importance from the production standpoint. The feed efficiency associated with rate of gain was studied in some of the previously cited gain research. Spencer et al. (1983a) fed early-weaned lambs until slaughter to study feed efficiency. Beginning APPENDIX at 3 weeks of age, the animals were im- munized at regular intervals and showed a 14 percent improvement in feed efficiency. The lambs also gained weight faster, and there was a 20 percent reduction in time to reach slaughter weight. Other lamb studies indicated feed efficiency improvements of 11 percent (Spencer and Hallett, 1985) and 27 percent (Spencer, 1986~. In the beef cattle study of Lawrence et al. (1986), steers were fed a completely mixed feed that met or exceeded the nu- trient requirements established by the Na- tional Research Council. The control and treated feed efficiencies (ratio of feed/gain) were 6.11 and 5.42, respectively, for a 12.7 percent improvement (P < 0.01) for the immunized steers (Table 3~. Composition of Growth A limited amount of data has been re- ported on the composition of growth in immunized animals. Spencer et al. (1983a) reported that lambs immunized against SS had an increased stature, as measured by shoulder height. This was subsequently con- firmed by the observation of elongated bones during postmortem carcass dissection. Work by Spencer (1986) has demonstrated the increase in carcass weight and soft tissue that would be expected from lambs that gained weight faster and were heavier. The TABLE 3 Eject of Active Immunization Against Somatostatin in Growing Cattle Variable Control Immunized Initial body weight (kg) 147 147 Final body weight (kg) 329 361 Average daily gain (kg) 1.19 1.40a Average daily gain response (%) 17.6 Feed efficiency (feed/gain) 6.11 5.42a Feed efficiency response (%) - 12.7 aDifferent from control (P < 0.001). SOURCE: M. E. Lawrence, G. T. Schelling, F. M. Byers, and L. W. Greene. 1986. Improvement of growth and feed efficiency in cattle by active immunization against somatostatin. J. Anim. Sci. 63(Suppl. 1):215.
IMMUNIZATION OF BEEF CATTLE TABLE 4 Eject of Active Immunization Against Somatostatin in Growing Cattle on Pasture Variable Control Immunized Initial body weight (kg) 235 235 Final body weight (kg) 338 350a Gain (kg) 103 llSa Average daily gain (kg) 0.98 1. O9a Average daily gain response (%) 11.6 aDi~erent from control (P < 0.05). SOURCE: G. T. Schelling, F. M. Byers, and L. W. Greene (unpublished data). work indicated no significant difference in the proportion of muscle (54 versus 53 percent) or fat (29 versus 31 percent) in control ant] treated lambs, even though the treated lamb carcasses were heavier (14.4 versus 17.1 kid. This suggests that treated 205 creased rate of passage. Subsequent work (Fadlalla et al., 1985) indicated an effect of passive immunization on the rate of passage. The intravenous administration of antibod ies to SS resulted in an increase in the GIT retention time of chromic oxide, a GIT animals would be leaner at a weight equal marker. Further elucidation of the net to that of the controls. There is a limited digestion and absorption effects of SS mod amount of research (Spencer and Hallett, 1985) with lambs slaughterer] at the same weight to support the concept of leaner carcasses. _. ~ Somatostatin Effects at the Gastrointestinal Tract The role of SS at the level of the gastroin- testinal tract (GIT) was recognized early. S. R. Bloom et al. (1974) reporter! effects of SS on motilin levels and gastric emptying. Boden et al. (1976) reported effects on duodenal motility. Other studies (Reichlin, 1987) have reporter! the influences of SS at the GIT level. Recent work by Gyr et al. (1986) has suggester] that circulating plasma SS is a regulator of pancreatic function. Thus, control at the GIT level is probably more than just an eject of locally produced SS, and therefore, circulating antibodies could play an important role there. Several studies have inclicated an effect of SS on the rate of passage through the GIT of sheep. Faichney and Barry (1984) reporter] that the administration of SS in ification is not available at this time. Somatostatin Effects on Other Hormones Eviclence is emerging to indicate that immunization against SS may result in a fairly consistent increase in SM (Spencer, 19854. The general positive relationships among GH, SM, and growth are becoming more clearly formulate(l (Etherton and Ken- singer, 1984~. Thus, the concept of a growth response resulting from immunization against SS must also consider SM. The work of Plotsky and Vale (1985), which suggests a direct SS relationship with GRF release, must also be considered in evaluating all factors influencing growth. While the inhib- itory relationships of SS to insulin and glucagon (Mortimer et al., 1974) and to thyrotropin (Vale et al., 1976) are clear, the importance of these relationships in the stimulation of growth by immunization is less evident. These possible associations make an understanding of the biology of the situation even more difficult.
206 CONCLUSIONS The original simple concept of immuniz- ing against SS to reduce its general availa- bility and thereby allow for increased ST release has turned into something very complex. It now appears that the relation- ships between SS and ST, other hormones, and GIT agents must also be considered. While direct ST involvement should by no means be ruled out as a mechanism of action at this time, it is clear that the striking serum ST concentration increases suggested by earlier rat studies do not occur in the active immunization of ruminants. It must be kept in mind that subtle ST responses, such as turnover, are clifficult to detect and could very well be causing important bio- logical influences. Consiclerable work will be required to elucidate the mechanisms of action. It is likely that significant progress watt be made in this area in the near future because of current interest and the availa- bility of resource materials to study growth regulation. The immunization approach exhibits ex- cellent potential, but its development will be challenging. The inherent complexity of the immunological response and our super- ficial understanding of hormonal relation- ships will present a formidable task. How- ever, the application of"natural" biology to enable the animal to produce its own growth regulators in an appropriate pattern has an element of safety that should be readily accepted by the wary consumer. This ap- proach could be an important new way to promote growth not only in beef cattle but in other species as well. REFERENCES Arimura, A., and J. B. Fishback. 1981. Somatostatin: Regulation ofsecretion. Neuroendocrinology33:246. Arimura, A., H. Sato, D. Coy, and A. Schally. 1975. Radioimmunoassay for growth hormone release in- hibitinghormone. Proc. Soc. Exp. Biol. Med. 148:784. Bass, J. J., P. D. Gluckman, R. I. Fairclough, A. J. Peterson, and S. R. Davis. 1983. Effect of immu nization against somatostatin and feed quality on APPENDIX growth controlling hormones and body composition. Proc. End. Soc. Australia 26(Suppl. 2):30. Bloom, C. N., D. H. Coy, and A. V. Schally. 1974. Inhibition of gastrin and gastric-acid secretion by growth hormone release inhibiting hormone. Lancet 2:1106. Bloom, S. R., D. N. Ralphs, G. M. Besser, R. Hall, D. H. Coy, A. J. Kastin, and A. V. Schally. 1974. Effect of somatostatin on motilin levels and gastric emptying. Gut 16:834. Boden, G., H. Jacoby, and A. Staus. 1976. Somatostatin interacts with basal and carbachol stimulated antral and duodenal motility. Gastroenterology 70:961. Chaplin, R. K., D. E. Kerr, and B. Laarveld. 1984. Somatostatin immunization and growth in lambs. Can. J. Anim. Sci. 64(Suppl.):312. Daughaday, W. H., K. Hall, M. S. Raben, W. D. Salmon, Jr., J. L. Van den Brande, and J. J. Van Wyk. 1972. Somatomedin: Proposed designation for sulphation factor. Nature 235:107. Etherton, T. D., and R. S. Kensinger. 1984. Endocrine regulation of fetal and postnatal meat animal growth. J. Anim. Sci. 59:511. Fadlalla, A. M., G. S. G. Spencer, and D. Lister. 1985. The effect of passive immunization against somatostatin on marker retention time in lambs. J. Anim. Sci. 61:234. Faichney, G. J., and T. N. Barry. 1984. Intravenous somatostatin infusion affects gastro-intestinal tract function in sheep. Can. J. Anim. Sci. 64(Suppl.):93. Ferland, I., F. Labrie, M. Jobin, A. Arimura, and A. V. Schally. 1976. Physiological role of somatostatin in the control of growth hormone and thyrotropin secretion. Biochem. Biophys. Res. Commun. 68:149. Froesch, E. R., J. Zapf, T. K. Audhya, E. Benporath, B. J. Segen, and K. B. Gibson. 1976. Non-suppres- sible insulin-like activity and thyroid hormones: Major pituitary-dependent sulphation factors for chick embryo cartilage. Proc. Natl. Acad. Sci. USA 73:2904. Galbraith, H., S. Wigzell, J. R. Scaife, and G. D. Henderson. 1985. Growth and metabolic response of rapidly growing male castrate lambs to immuni- zation against somatostatin. Anim. Prod. 40:523. Gyr, K., C. Beglinger, E. Kohler, and U. Keller. 1986. Circulating somatostatin (S-14) inhibits both endo- and exocrine pancreatic function in man. International Conference on Somatostatin. P. 78, Abstract No. II-43. Serono Symposia, Washington, D.C. Lawrence, M. E., G. T. Schelling, T. W. Welsh, F. M. Byers, and L. W. Greene. 1985. Effect of passive immunization against somatostatin on plasma growth hormone and performance of growing rats. J. Anim. Sci. 61(Suppl. 1):47. Lawrence, M. E., G. T. Schelling, F. M. Byers, and L. W. Greene. 1986. Improvement of growth and feed efficiency in cattle by active immunization against somatostatin. J. Anim. Sci. 63(Suppl. 1):215.
IMMUNIZATION OF BEEF CATTLE Li, C. H. 1973. Growth hormone: Purification and biochemical characterization. P. 257 in Methods in Investigative and Diagnostic Endocrinology, S. A. Benson and R. S. Yalow, eds. Amsterdam: North- Holland. Mortimer, C. H., D. Carr, T. Lind, S. R. Bloom, C. N. Mallinson, A. V. Schally, W. M. G. Tunbridge, L. Yeomans, D. H. Coy, A. Kastin, G. M. Besser, and R. Hall. 1974. Effects of growth hormone release inhibiting hormone on circulating glucagon, insulin and growth hormone in normal, diabetic, acromegaly and hypopituitary patients. Lancet 1:697. Plotsky, P. M., and W. Vale. 1985. Patterns of growth hormone-releasing factor and somatostatin secretion into the hypophysial-portal circulation of the rat. Science 230:461. Raben, M. S. 1973. Physiology: Hormonal effects. P. 257 in Methods in Investigative and Diagnostic Endocrinology, S. A. Benson and R. S. Yalow, eds. Amsterdam: North-Holland. Reichlin, S., ed. 1987. Somatostatin, basic and clinical studies. Pp. 221-290. New York: Plenum Press. Schalch, D. S., U. E. Heinrich, B. Draznin, C. J. Johnson, and L. L. Miller. 1979. Role of liver in regulating somatomedin activity: Hormonal effect on the synthesis and release of insulin-like growth factor and its carrier protein by the isolated perfused rat liver. Endocrinology 104:1143. Spencer, G. S. G. 1985. Hormonal systems regulating growth. A review. Livestock Prod. Sci. 12:31. Spencer, G. S. G. 1986. Immuno-neutralization of somatostatin and its effects on animal production. Dom. Anim. Endocrinol. 3:55. Spencer, G. S. G., and K. G. Hallett. 1985. Immu- nization against somatostatin in a commercial breed of sheep and its effect on growth and efficiency. Anim. Prod. 40:523. Spencer, G. S. G., and E. D. Williamson. 1981. 207 Increased growth in lambs following auto-immuni- zation against somatostatin. Anim. Prod. 32:376. Spencer, G. S. G., G. J. Garssen, and P. L. Bergstrom. 1983a. A novel approach to growth promotion using auto-immunization against somatostatin. II. Effects on appetite, carcass composition and food utilization in lambs. Livestock Prod. Sci. 10:409. Spencer, G. S. G., G. J. Garssen, and I. C. Hart. 1983b. A novel approach to growth promotion using auto-immunization against somatostatin. I. Effects on growth and hormone levels in lambs. Livestock Prod. Sci. 10:25. Spencer, G. S. G., K. G. Hallett, and A. M. Fadlalla. 1985. A novel approach to growth promotion using auto-immunization against somatostatin. III. Effects in a commercial breed of sheep. Livestock Prod. Sci. 13:43. Tannenbaum, G. S., J. Epelbaum, E. Colle, P. Bra- zeau, and J. B. Martin. 1978. Dissociation of effects of somatostatin antiserum on growth hormone and insulin secretion. Metabolism 27:1263. Thorner, M. D., R. L. Perryman, M. J. Cronin, M. Draznin, A. Johanson, A. D. Rogol, J. Jane, L. Rudolf, E. Horvath, K. Kovacs, and W. Vale. 1982. Acromegaly with somatotroph hyperplasia: Success- ful treatment by restriction of a pancreatic tumor secreting a GH-releasing factor. Clin. Res. 30:555. Vale, W., P. Brazeau, C. Rivier, M. Brown, B. Boss, J. Rivier, R. Burgus, N. Ling, and R. Guillemin. 1976. Somatostatin. Rec. Prog. Hormone Res. 31:365. Varner, M. A., S. L. Davis, and J. J. Reeves. 1980. Temporal concentrations of growth hormone, thy- rotropin, insulin and glucagon in sheep immunized against somatostatin. Endocrinology 106:1027. Vicini, J. L., J. H. Clark, W. L. Hurley, and J. M. Bahr. 1986. Effect of immunization against somato- statin on growth of young dairy calves. J. Anim. Sci. 63(Suppl. 1):242.
Lactation Biology and Methods of Increasing Efficiency RONALD C. GOREWIT Breakthroughs in biotechnology have macle it possible to manipulate physiological mechanisms controlling the growth ant! de- velopment of organ systems, as well as processes such as milk secretion that are of economic importance. Recombinant de- rived bovine growth hormone (bSTH) has a dramatic eject on increasing milk procluc- tion and mammary growth in dairy cattle. This paper briefly summarizes the biology of lactation, using the dairy cow as a model. It then reviews a number of experiments in which the dramatic influence of bSTH on milk secretion ant] mammary gland devel- opment has been shown and alternative methods for increasing the efficiency of mammary gland function have been exam- ined. MAMMARY ANATOMY The cow has four mammary glands grouped into a structure called an udder, which is locater] in the inguinal region of the cow's body. Milk is secretes] by alveolar epithelial cells, which are grouper! into small clusters called lobules. Lobules are surrounded by connective tissue capsules (Schmicit, 1971; 208 Turner, 1939, 1952), often referred to as stroma. Groups of lobules form larger struc- tures called lobes and are considered to be the parenchymal elements. The entire mammary gland is composed of lobules and lobes. The lobules are drained by small ducts. The smaller ducts or capillary milk ducts are lined with a single layer of epi- thelial cells and are capable of secretion. The ducts get progressively larger until they reach a cistern, which is connected to a teat by which milk is released upon suckling or machine milking (Schmi(lt, 1971; Turner, 1939, 1952~. A group of cells called basket cells or myoepithelial cells surround each alveolus (Schmidt, 1971; Pitelka and Hamamoto, 1983~. The neurohypophysial hormone ox- ytocin causes the myoepithelial cells to contract, forcing newly synthesized milk into the lumen of the alveoli. Most milk present between milkings is in the epithelial cells and alveolar lumen. At six months of fetal age, the calf has mammary glands consisting of a small teat with a meatus, a teat cistern, a glancl cistern, and a primitive duct system. Very little further development takes place before birth.
LACTATION BIOLOGY A small amount of growth in secretory tissue occurs from birth to puberty, most of which is due to deposition of adipose tissue. With each recurring estrous cycle after puberty, some further mammary glanc! cle- velopment takes place (Sinha ant! Tucker, 1969~. Estrogen produced during each es- trous cycle is primarily responsible for duc- tal growth ant] progesterone for develop- ment ofthe secretory tissue. When pregnancy occurs, a market] increase in mammary glanc} growth takes place (Hammond, 1927~. Most of the ductal growth occurs during the first part of pregnancy, and the lobulo- alveolar system takes form during the mid- dIe and later parts of pregnancy. A wave of mitosis occurs shortly before or after par- turition (Paape ant] Tucker, 1969; Tucker and Reece, 1963a,b,]~. A slight amount of growth may occur until the peak of lactation, and some evidence indicates that growth may continue throughout certain stages of lactation (Trauig, 1967~. At some point dur- ing lactation, however, cells are clestroyed or eliminated and involution (regression) of the gland begins. Much is being learner! about the hor- monal requirements for mammary growth as more precise tools for quantifying growth are clevelopecI. Nuclear magnetic resonance (NMR) imaging and computerized tomog- raphy (CT scan) technology will help pin- point hormonal requirements for mammary glanc] growth at various physiological stages of the animal s development. Experimental results to ciate indicate that estrogen, progesterone, prolactin, and so- matotropin are required for mammary gland development in the intact animal (Forsyth, 1983~. The placenta may also influence mammary glanc! development during preg- nancy. Hormones with mammogenic, lac- togenic, and luteotrophic stimulating prop- erties have been found in a variety of animal placentas. These proteins are the placental lactogens, which have been characterized biochemically in numerous animals includ- ing cattle (Forsyth, 1983~. Insulin and the 209 adrenal corticoi(ls appear to be more (Erectly involved in maintaining a normal metabolic state in hypophysectomized animals, but they have direct ejects on mammary glanc] proliferation in in vitro systems (Forsyth, 1983; Tucker, 19741. Epithelial cells must divide in organ culture (this process is under the control of insulin) and proliferate in the presence of cortisol in order to synthesize casein in response to prolactin (Anclerson, 1974, Lockwood et al., 1967; Rivera and Bern, 1961~. INITIATION OF LACTATION (LACTOGENESIS) Colostrum or first milk accumulates in the epithelial cells before parturition (Me- pham, 1983; Schmidt, 1971~. It is concen- tratec! with antibodies and passively im- munizes the young to a variety of antigenic factors. In the rat, lactose does not appear in milk until parturition. The initiation of lactation, caller] lactogenesis, is character- ized by increases in the RNA/DNA ratio, the number of ribosomes, the endoplasmic reticulum, and the number of mitochondria per cell. Histological changes are primarily associated with changes due to milk accu- mulation within the lumen of the alveolus. The anterior pituitary produces hormones essential for lactogenesis. Prolactin causes localized initiation of milk secretion when injected into the rabbit mammary gland (Forsyth, 1983; Schmidt, 1971; Tucker, 1974), but corticoids are required for lactogenesis in most animals (Forsyth, 1983; Tucker, 1974~. Insulin and cortiso} are the minimal hor- mone requirements for maintaining viable mammary gland tissue explants in vitro (Forsyth, 1983~. As mentioned previously, the cells must first divide in order to syn- thesize casein. For prolactin ant] human placental lactogen to stimulate casein syn- thesis, cell division must take place in the presence of insulin and cortiso} (Forsyth, 1983~.
210 The hormonal control of lactogenesis is not completely cleaned. Most theories cen- ter on either a rise in the blood levels of prolactin and adrenal glucocorticoids at the time of parturition or a drop in progesterone or glucocorticoid binding globulin (tran- scortin) (Forsyth, 1983; Schwalm and Tucker, 1978; Tucker, 1974~. Transcortin is proposed to bins] the adrenal corticoids, thus inhib- iting their biological activity (Tucker, 19744. Several years ago, it was shown that oro- gesterone inhibited the synthesis of alpha- lactalbumin, a protein essential in forming lactose synthetase. Lactose synthetase is postulated to be a rate-limiting enzyme for lactose biosynthesis. It is thought that es- trogen levels increase before parturition, which stimulates prolactin secretion. At this time, prostaglanclin F2a causes regression of the corpus luteum, which normally main- tains pregnancy (Forsyth, 1983; Schwalm and Tucker, 1978; Tucker, 1974~. Proges- terone concentrations fall, leading to the unmasking of glucocorticoid receptors. Es- trogen continues to increase, thereby in- creasing prolactin concentrations (Forsyth, 1983; Schwalm and Tucker, 1978; Tucker, 1974~. Prolactin may up-regulate its own receptors, and the biosynthetic processes (lactose and casein production) involved in milk synthesis are activated (Forsyth, 1983; Schwalm and Tucker, 1978; Tucker, 1974~. CONTROL OF PROLACTIN PRODUCTION Prolactin secretion, release by the pitui- tary glance, or both are under control of the hypothalamus (Schmidt, 1971; Tucker, 1974~. The hypothalamus produces a chemical compound caller! prolactin inhibiting factor (PIF) that normally inhibits prolactin secre- tion or release from the pituitary. Com- pouncls such as reserpine, epinephrine, ant! other biogenic amines and acetylcholine decrease PIF and therefore increase blood concentrations of prolactin. Thyrotropin re- leasing ~ hormone, estra(liol, triioclothyro APPENDIX nine (T3), ant] tetraiodothyronine (T4) stim- ulate prolactin release (Forsyth, 1983; Schmidt, 1971; Tucker, 1974~. Stress, milk- ing or suckling, high temperatures, and light will also influence prolactin secretion (For- syth, 1983; Tucker, 1974, 1985a). MAINTENANCE OF LACTATION (GALACTOPOIESIS) Galactopoiesis is dependent on removal of milk en cl a suckling or milking stimulus in all animals. Milk synthesis will not con- tinue if the product is not electively re- moved. Prolactin is released at milking along with adrenocorticotrophic hormone (ACTH) and oxytocin. Complete restoration of milk production in hypophysectomized goats has been obtained with injection of prolactin, bSTH, T3, insulin, and corticosteroids (Cowie, 1969~. Much work has been done on the feeding ant! injection of hormones to intact animals, especially ruminants. Anterior pituitary ex- tracts have been found to increase milk secretion in (lairy cows, primarily because ofthe bSTH content ofthe extracts (Forsyth, 1983; Schmidt, 1971; Tucker, 1985a). Sev- eral workers have shown that bSTH isolated from the pituitary increases milk production in cows (Tucker, 1985a). Recombinant-(le- rived bSTH has been shown to increase milk production in short- and long-term experiments with no deleterious ejects. These experiments are discussed later. Pituitary prolactin is not galactopoietic in dairy cattle (Plaut et al., 1985~. Large doses of ACTH or adrenal corticoids inhibit lac- tation in the rat and ruminant (Tucker, 1985a). Feecling of thyroprotein, T4, or T3 in- creases milk production in lactating cows (Meites, 1961; Thomas and Moore, 1953; Thomas et al., 1957~. Thyroprotein feeding increases milk production for 2 to 4 months and also results in a transitory increase in butter fat percentage. It appears that feed- ing thyroprotein causes an increased need
LACTATION BIOLOGY for nutrients; a loss of body weight; and an increase in heart rate, respiration rate, and body temperature (Meites, 1961~. Several workers have shown that long-term treat- ment increases milk production in early lactation (Schmidt, 1971~. However, there is a rapid decline in production in later lactation, resulting in overall lower produc- tion than is normal for the cow. No cletri- mental health effects have been seen with prolonged thyroprotein treatment, pro- vided the nutritional needs of the animal are always considered. MILK EJECTION Removal of milk from the mammary gland is depenclent on a functional milk ejection reflex. This is a neurohormonal-depenclent process. The ejection of milk results from a nervous stimulus that an animal associates with milking or suckling activity, such as manual massage ofthe unpiler teats, suckling, or sight and smell ofthe calf(Tucker, 1985a). The milking machine itselfcan also stimulate the reflex (Gorewit et al., 1983~. The neural stimulus reaches the central nervous system and causes the posterior lobe to release oxytocin (Tucker, 1985a). Oxytocin reaches the mammary gland and myoepithelial cells, and contraction of these cells forces milk from the alveoli into the (luct system. Milk then flows freely into the larger ducts and cisterns. Adrenalin inhibits milk ejection primarily by reducing blood flow to the gland so that sufficient concentrations of oxytocin cannot reach the receptors on the myoepithelial cells (Gorewit and Aromanclo, 1985~. The posterior pituitary hormones are pro- ducec3 in the hypothalamus ant! travel to the posterior pituitary where they are stored until release (Tucker, 1985a). The para- ventricular nuclei are particularly involved with oxytocin production. However, the supraoptic nuclei can also release oxytocin. Oxytocin is a peptide containing eight amino acids. It has a molecular weight of 211 about 1,000 daltons. Vasopressin is a similar molecule and has some oxytocic properties, but oxytocin appears to have five to six times more activity in causing milk ejection than does vasopressin. Also, considerably more oxytocin than vasopressin is released during milk ejection (Schmidt, 1971; Tucker, 1985a). A very significant advancement in lacta- tion research was made when a nonextrac- tion raclioimmunoassay for measuring oxy- tocin was developed at Cornell University (Gorewit, 1979~. This allowed researchers to define more precisely the physiology of milk ejection. Milking or suckling of the cow causes a marked increase in blood oxytocin concentrations (Gorewit, 1979; Go- rewit en c! Aromando, 1985; Gorewit et al., 1983; Wachs et al., 1984a,b). The level of oxytocin cirops to resting concentrations within 20 minutes of release. The synthesis, metabolic clearance rate, and half-life of oxytocin have been determiner] for the cow and change during the lactational cycle (Wachs et al., 1984a,b). Oxytocin exerts a vasoactive influence on the bovine mammary glanc! (Gorewit et al., 19884. During the normal milking process, there is a 30 to 50 percent increase in mammary blood flow, which is primarily caused by oxytocin release (Gorewit et al., in press). This increased blood flow may have a rejuvenating effect on the metabol- ically active cells or aid in the expulsion of milk via pressure differentials between the myoepithelial cells and the capillaries sur- rounding them. Myoepithelial cells contain specific re- ceptors for oxytocin and are under direct hormonal control (Forsyth, 1983; Solon ant] Swartz, 1973; Zhao and Gorewit, 1986, 1987~. The motor innervation of the mam- mary gland per se plays no part in normal milk ejection. Myoepithelial cells have been isolated and grown in primary cell culture (Gorewit and McOsker, 1983) on both plas- tic and collagen matrices, but they appear to remain more differentiated on collagen (Gorewit and Rizzo, 1986).
212 MAMMARY INVOLUTION Regression or involution of the mammary gland takes place after the peak of lactation and after cessation of milking or suckling (Lenfers, 1907; Schmidt, 1971~. The invo- lutionary process is more drastically affected by cessation of milk removal than it is after peak lactation (Schmidt, 1971; Schmidt et al., 1962~. Involution is thought to be due to decreases in cell numbers, the decline in rate of milk synthesis of remaining cells, or both. There are characteristic changes in mam- mary histology during the involutionary process, including decreases in the size of the alveoli, the number of alveoli per lobule, the total number of alveoli and lobular volume, and the number of cells per alveolus (Schmidt, 1971~. Complete lobules disin- tegrate in parts of the mammary gland during advanced involution, and by the end of involution, the gland resembles that of the virginal state. However, the essential lobular structure of the gland is still rec- ognizable (Schmidt, 1971~. Mammary gland engorgement occurs af- ter cessation of milking or suckling and causes irreversible changes in the cytoplasm of the secretory epithelial cells (Schmidt, 1971~. The changes are thought to be due to interference with the blood supply to the mammary epithelium. Alveoli eventually rupture and secretion moves into the inter- cellular spaces. Phagocytes then begin to enzymatically break down milk compo- nents. Changes similar to those that occur normally during lactation are believed to take place during involution, but the entire process is not so abrupt. Biochemical changes also occur in the mammary gland during involution. There is a tremendous decrease in the secretory activity of the cell (Schmidt, 1971; Tucker and Reece, 1963b). A decrease in the res- piratory quotient, a decrease in oxygen consumption, and an accumulation of lactic acid in the tissue take place. Oxidative APPENDIX phosphorylation within the mitochondria is uncoupled within 12 to 24 hours after the young have been removed (Schmidt, 1971; Schmidt et al., 1962~. The suckling stimulus plays an important role in galactopoiesis, since it has been shown that involution is retarded by the suckling of ligated mammary glands (Schmidt, 1971~. Suckling stimuli also maintain the nucleic acid content of the lactating mam- mary gland (Thatcher and Tucker, 1966; Tucker, 1964; Tucker and Reece, 1963c). The suckling stimulus without milk removal, however, neither prevents a decrease in cell loss nor maintains the protein synthetic activity of the cells (Schmidt, 1971~. Prolac- tin injections also retard involution (Hooker and Williams, 1941; Schmidt, 1971; Wil- liams, 1945~. Oxytocin injections retard involution in the rat after cessation of milking (Schmidt, 19714. This may be because of periodic milk ejection caused by oxytocin, which releases milk from the alveoli into the ducts and stromal tissue where it can be absorbed, thereby allowing further milk synthesis to occur. Prolactin will not maintain the full biochemical integrity of the cell unless the secretory products are removed (Schmidt, 1971~. MILK SECRETION RATE Milk secretion rate is important in the dairy industry. It influences the required frequency for milking of cows and the ac- ceptable intervals between milkings. In part, milk secretion rate depends on the pressure that accumulates within the mammary gland. When milk accumulates within the mam- mary gland for a long enough period of time, pressure is built up to a sufficient level to inhibit .senretion and milk is resorbed by the blood Schmidt, 1971~. A marked in- crease in pressure occurs after 1 hour of milking. Residual milk or complementary milk moves from the alveoli into the teat and gland cisterns. Thereafter, a gradual
LACTATION BIOLOGY increase in the pressure occurs owing to the movement of milk from the alveoli to the teat and glanc] cisterns. The rate of milk secretion is linear for about 10 to 12 hours after the last milking, after which it cle- creases slightly. It continues to decrease until it finally reaches zero about 35 hours after the last milking (Schmidt, 1971; Tucker et al., 1961~. 213 yield are increased body weight, advancing age, increased plane of nutrition, fall and winter calving, moderate or cool environ mental temperatures, ant! good body con dition at calving. Factors that tent] to de crease milk yield are advancing lactation, advanced stage of gestation, short dry pe riocl, spring and summer calving, high en vironmental temperatures and humidity, Part of the early increase in intramam- diseases that affect the udder or feed intake mary pressure is clue to residual milk that of the cow, ant! a clecreasec! plane of nutri is left in the udder after normal milking. lion (Schmidt, 1971~. The amount of resiclual milk can be as great During a normal lactation of the dairy as 15 to 30 percent (Gorewit and Sagi, 1984; cow, the milk yield starts out at a high level, Gorewit et al., 1983; Schmidt, 1971~. This peaks 3 to 6 weeks after calving, and then percentage is higher in low-proclucing cows gradually declines towarc] the end of lacta than high-proclucing cows (Schmidt, 1971~. lion. The milk fat ant] protein percentages Increasing the time interval between are inversely related to the milk yield (Dav milkings can increase milk production. De- ies et al., 1983; Mepham, 1983; Schmidt, creasing the milking interval from 2 to 1 1971~. The percentage composition starts will lower milk production in cows bv as out at a moderate level, decreases to a low level during peak lactation, and then gracI ually increases toward the end of lactation (Mepham, 1983; Schmidt, 1971; Tucker and Reece, 1963c). Certain changes in the cow's diet ad versely influence milk fat percentage. Most of these are relater! to a high-concentrate, low-roughage diet that contains a low amount of fiber. The reason for milk fat depression is unclear. The clepressec] milk fat percent age is accompanied by a change in rumen fermentation. There is a decrease in rumen acetate production, an increase in rumen propionate production, and a (lecrease in rumen pH. Feeding sodium or potassium bicarbonate, magnesium bicarbonate, mag nesium oxide, and calcium hydroxide partly prevents the milk fat depression caused by restricted] roughage intake (Davies et al., 1983; Schmidt, 1971~. much as 40 percent (Schmidt, 1971~. FACTORS AFFECTING THE YIELD AND COMPOSITION OF MILK Mflk fat, lactose, and proteins are syn- thesized in the cells from precursors ab- sorbed from blood (Davies et al., 1983; Schmidt, 1971~. These components are re- leased in the milk by apocrine, merocrine, or holocrine secretion (Tucker, 1974~. Water, minerals, and vitamin components of milk enter the lumen of the alveolus primarily through diffusion (Masher and Keennan, 1983; Schmidt, 1971), although some may be bounc] to other compounds. Mammary gland blood flow rate is highly correlates] to milk production. In fact, about 500 volumes of blood] flow through the cow's unpiler for each volume of milk produced (Mepham, 1983; Schmidt, 1971~. The ratio of blood] flow to milk yield is higher in lower milk producing goats and in animals in late lactation. Many physiological and environmental factors can influence milk secretion. For cows, factors related to increases in milk BIOCHEMISTRY OF MILK SECRETION The mammary gland secretory epithelial cells break down substrates to provide en- ergy for synthetic processes within the mam- mary gland. From these substrates, the
214 gland synthesizes milk components such as fat, lactose, and protein. The cells regulate milk composition by controlling amounts of constituents such as water, vitamins, ant! minerals that are not synthesized in the mammary gland. The major blooc] precursors for milk syn- thesis are glucose, acetate, beta-hydroxy- butyrate, triglycericle fatty acids, and amino acids (Kuhn, 1983; Smith et al., 1983~. The two major energy sources are glucose and acetate. Other compounds are absorbed and usual by the mammary gland, but they do not contribute significantly to the quanti- tative aspects of milk composition, even though their qualitative aspects are ex- tremely important. Compounds that are broken down by oxidation in mitochondria are used for en- ergy in milk synthesis. Approximately 90 percent of the adenosine triphosphate (ATP) is former! via the electron transport system. The terminal phosphate group of ATP is transferred to a specific acceptor molecule. The acceptor molecule has its energy con- tent raised to a level at which it can partic- ipate in the energy-requiring processes within the cell, such as the synthesis of triglycer- ides, lactose, and proteins. The liberated adenosine diphosphate (AD P) molecule can be reused for the generation of ATP (Smith et al., 1983~. Energy in the mammary epithelial cell is generated by one of three pathways (Smith et al., 1983~. The EmbJen-Meyerhof gly- colytic pathway (regrades glucose and other hexose molecules into two lactic acids with the generation of ATP at the substrate level of phosphorylation. The pyruvate produced can enter the citric acid cycle. The citric acid cycle is the final common pathway for mammary metabolism. It can also utilize acety! coenzyme A (acetyI-CoA) from fatty acid metabolism and carbon skeletons from amino acid metabolism. A third pathway operating in the mammary gland is the pentose phosphate shunt. Its primary func- tion in the mammary cell is to provide APPENDIX hydrogen ions for reductive stages of fatty acid synthesis. Breakdown products of the pentose phosphate shunt are also involved in nucleic acid synthesis. The pentose phos- phate shunt is the major pathway for glucose oxidation in the epithelial cell. A sucl(len increase in enzyme activity occurs Luring parturition in the rat but has not been describecl for the cow. Milk Protein Synthesis There are a number of proteins that are specific to milk. Among the major ones are casein, alpha-lactalbumin, and beta-lacto- globulin. Most of the milk proteins are synthesized within the mammary gland. Gamma-casein, blooc! serum albumin, and the immune globulins are absorbed as pre- formed proteins from the blood (Mercier and Gaye, 1983~. Most of the protein synthesized by the epithelial cells is synthesized from amino acids that are absorbed from the blood- stream. This has been determined by meas- uring arteriovenous differences across the mammary glanc] and relating uptake by the mammary glancI to the composition of amino acids in milk protein (Mercier and Gaye, 19835. Radioisotopes have been used to follow the pathways of incorporation of la- beled elements of amino aci(ls into the proteins in the milk. The peptides in blood plasma provide less than 10 percent of the amino acids in milk protein (Mercier and Gaye, 1983). Plasma proteins may provide a small portion of the essential amino acids of milk protein synthesized in the mammary gland. However, less than 10 percent of these proteins come from plasma proteins. The essential amino acids are absorbed from the bloodstream, and most of the nonessen- tial amino acids are also absorbed from blood. The mammary gland is capable of producing nonessential amino acids from other amino acids (Mercier and Gaye, 1983~. For example, ornithine can be converted to proline by the secretory epithelial cell.
LACTATION BIOLOGY Carbohydrates and volatile fatty acids can act as substrates for nonessential amino acids. The protein synthetic reaction mecha- nisms appear to be comparable to those found in most other protein-synthesizing cells. Milk Fat Synthesis acetate and beta-hydroxybutyrate, triglyc- ericles of the chylomicra, ant] low-clensity lipoproteins from blood serve as major ore- cursors for milk lipids (Dils, 19834. The beta-hydroxybutyrate is also used for fatty acid synthesis (Dils, 1983~. The ruminant mammary gland cannot effectively utilize acetyl-CoA former] from glucose in the mi- tochondria. The short-chain fatty acids from C4 to Ci4 ant] some palmitic acid are synthesizer] within the mammary gland from acetate clerivec! as absorbed acetate in the ruminant or from glucose in the nonruminant (Dils, 1983~. About 30 percent of palmitic acid is derived from acetate, and the remainder comes from triglycericles of blood. Stearic and oleic acids come primarily from plasma triglycerides. Stearic acid is absorbed in greater quantities from bloom! than is oleic acid, yet oleic acid is the most prevalent Cog fatty acic! in milk. Stearate can be converted to oleic acid by the bovine, ca- prine, and swine mammary glan(ls. Oleic acid cannot be saturated to form stearate. Free glycerol from the bloodstream pro- vicles less than 10 percent of the glycerol portion of milk triglycerides. Most glycerol comes from glycerol-3-phosphate from gly- colysis and the remainder from lipoprotein glycerides (Dils, 1983~. The malonyl-CoA pathway appears to be the predominant route of fatty acid synthe 215 sis. Two pathways for esterification of fatty acids may be present in the mammary gland: the phosphaticlic acid pathway, which is the most common, an(l formation of 1,2-(liglyc- ericle from acylation of 2-monn~lvo~riri (Dils, 19831. Lactose, Minerals, and Vitamins c, , ~ ~ _ The major sugar of milk is lactose. Lactose Fat is the most variable component of is a disaccharide made up of a glucose and milk. Both the percentage composition of agalactose molecule. The primary precursor fat and the fatty acid composition of triglyc- of lactose is glucose (Davies et al., 1983; Brides within and among species vary. Most Kuhn, 1983; Smith et al., 1983~. The glucose milk fat is made up oftrig;lycerides. Glucose. molecule is phosphorylated to form glucose 6-phosphate, which is then converter] into glucose-l-phosphate. The glucose-l-phos phate, in the presence of uridine triphos phate, forms uricline diphosphate (UDP) glucose, which is converted to UDP galac tose. The UDP galactose is united with free glucose to form lactose with the liberation of UDP. The last step is catalyzed by the enzyme lactose synthetase. This is a unique enzyme that is composed of two subunits: the common galactosyl transferase and the milk protein alpha-lactalbumin. The major mineral constituents of milk are calcium, phosphorus, potassium, chlo rine, sodium, and magnesium. Potassium, chlorine, and sodium are in soluble form. Phosphates, citrates, and casein bind var ious minerals such as calcium and sodium. The buffering capacity of milk is due to citrates, phosphates, and bicarbonates, along with the proteins in the milk. Serum calcium is in equilibrium with bone calcium, making it clifficult to increase the calcium content ofthe milk by increasing the calcium content of the fee(l. Inorganic phosphate of blood serum is the precursor of milk phosphates. There is considerable evidence that healthy cows have constant amounts of lactose, po tassium, sodium, ant! chlorine in their milk (Schmicit, 1971~. The exact amount appears characteristic for each cow. There is a close inverse relationship between lactose con tent of milk and the molar sum of sodium
216 and potassium contents, as well as between lactose and potassium contents of milk. Water moves into the milk with casein to maintain osmotic equilibrium with the blood. Consequently, secretion of lactose, potas- sium, sodium, and chlorine controls the milk volume (Peaker, 1983~. Milk contains trace amounts of some other minerals and can take up large amounts of iodine when excessive amounts are fed. Vitamins are absorbed by the mammary gland from the bloodstream. Generally, the vitamin content of milk can be increaser! by increasing the vitamin content of blood supplying the mammary gland. The rumi- nant animal clepends on feet] supply ancl exposure to sunlight for its fat-soluble vi- tamins A, D, and E. Beta-carotene is con- verted to vitamin A in the intestinal mucosa of the ruminant. Vitamin D in the milk of cows comes from activation of ergosterol in feed or from the animal s exposure to sun- light. This activates 7-dehyclrocholesterol in the skin of the animal. Milk contains vita- mins E ant! K. The B vitamins are synthesized by mi- croflora in the rumen. Consequently, changes in the B vitamin contents of the diet clo not change the contents in milk except for riboflavin. The ascorbic acid (vitamin C) content of cow milk cannot be changed by dietary content, since it is synthesized in the ruminant s body. EFFECTS OF HORMONES ON MILK SECRETION AND MAMMARY DEVELOPMENT Milk Secretion Asimov and Krouze (1937) injected! dairy cows with anterior pituitary extracts and found significant increases in milk procluc- tion. British workers established that growth hormone was the active hormone respon- sible for these increases (Young, 1947~. Almost a (lecade later, Brumby and Hancock (1955) reported results of treating lactating APPENDIX cows for 12 weeks with pituitary growth hormone. Twin cows received daily injec- tions of somatotropin and producer] approx- imately 50 percent more milk than their twin mates. Their live weights were un- changed. Machlin (1973) studier] the effect of growth hormone on milk production and feet! uti- lization in dairy cows. Growth hormone injections increased milk production and appeared to increase feed efficiency. More recently, researchers at Cornell University ant] the National Institute for Research in Dairying demonstrated that growth hor- mone increased significantly both milk pro- duction and feed utilization in high-produc- ing, genetically superior cows (Hart et al., 1985; Peel et al., 1981~. Bauman et al. (1982) were the first re- search group to administer recombinant methionyl bovine growth hormone to high- producing dairy cows. The recombinant growth hormone was as effective as pitui- tary-(lerivecl growth hormone in enhancing milk production. It is now possible to apply this breakthrough in biotechnology to wide- spread use on commercial dairy farms. Since 1981, several studies have com- pare(1 the effectiveness of natural pituitary growth hormone versus recombinant growth hormone in increasing milk production and feet] utilization as well as the effectiveness of various routes and methods of hormone administration. In high-yielding Holstein cows, milk pro- cluction has been consistently increased from 2 to 5 kg/day in early, mid, and late lactation by administration of bSTH (Bauman et al., 1982; Hart et al., 1985; Peel et al., 1981, 1982, 1983; Richard et al., 19854. Additional nutrients in the form of glucose and casein (Pee! et al., 1982) or lipids (Lough et al., 1984) have hac] no additive effect compared to bSTH treatment alone. There is a cur- vilinear relationship between milk produc- tion en cl close of growth hormone (Eppard et al., 1985~. However, the pattern in which somatotropin is administered does not ap
LACTATION BIOLOGY pear to affect the increases in milk produc- tion observed (Fronk et al., 1983~. In most short-term trials, feed intake did not increase upon administration of bSTH, despite significant increases in milk pro- cluction. To cIate, no changes in digestibility or the efficiency with which energy is used for maintenance of milk synthesis during growth hormone administration have been observed. Long-term studies have been done with both natural pituitary growth hormone and recombinant derived hormone (Bauman et al., 1985; Brumby ant] Hancock, 1955; Fronk et al., 1983; Machlin, 1973~. Production increases were very similar in studies car- riec! out by Brumby and Hancock (1955) ant] Machlin (1973) for 12 and 10 weeks, respectively, using pituitary-derived soma- totropin. Studies by Bauman et al. (1985) were carried out for 27 weeks. High-yielding Holstein cows received treatments of 0 (control), 13.5, 27.0, and 40.5 mg/day of recombinant-derived bovine growth hor- mone or 27.0 mg/day of pituitary-clerivec] growth hormone. They showed increases in milk production ranging from 16 to 41 percent over the control production. Injec- tions were carried out for 188 clays com- mencing around day 84 of lactation. There were no significant differences in live weight gains even though feet] intake increased. Feed intake gradually increased to support the higher milk production. Cows treated with natural anterior pituitary growth hor- mone responded with substantial increases in milk production. Pee! et al. (1985) treater! five sets of monozygotic twins in which the animals' sole diet was ryegrass ant] clover pasture. They were treater] with natural pituitary- derived bSTH. Like the trials carried out at Cornell, milk production increased ant! so die! feed intake to support the increased production. The improved milk production seen in the two long-term studies was re- lated to the greater efficiency with which 217 feed was utilized for milk secretion (kg of milk/kg of feed). This was primarily related to dilution of feed costs for maintenance at the higher production levels. Effects of Somatotropin on Mammary Growth Somatotropin has been shown to be es- sential for pubertal mammary clevelopment (Forsyth, 1983; Tucker, 1985a). The lack of available growth hormone necessary to carry out studies on large animals, such as cattle, has made it difficult to study the hormone's effects on enhancing normal rates of mam- mary growth at puberty in these animals. Recently, Sejrsen et al. (1986) have exam- ined the effects of daily administration of exogenous Somatotropin on mammary de- velopment in dairy heifers. Nine pairs of identical twins were used. One twin re- ceivec3 a daily subcutaneous injection of Somatotropin (20 IU) and the other received excipient. Treatments commenced at 8.0 months of age (179 kg live weight) and continued for 15.6 weeks. At that time, heifers were slaughtered and mammary de- velopment was evaluated. Somatotropin re- sultec] in an increase in mammary paren- chyma and decreases in extraparenchymal tissue and weight of the mammary glands. Increases in parenchyma were 56 percent as determinecl by computer assisted x-ray tomography and 18 percent as determined by dissection. Chemical composition (39 percent water, 7 percent protein, and 54 percent fat) and histological and cytological appearance of the mammary gland paren- chyma were not affected by the treatment. Treatment with exogenous Somatotropin around puberty enhanced the growth rate of mammary parenchymal tissue. This in- crease in tissue might well lead to increased future milk production in these animals. It is likely that Somatotropin does not directly stimulate milk secretion and mam- mary growth. Receptors for bSTH have not been found in mammary tissue from dairy
218 cows. Moreover, the infusion of bSTH di- rectly into the mammary artery has not stimulated milk production in sheep. So- matotropin is thought to activate the pro- duction of somatomedins or insulin-like growth factors (IGFs). The role of somato- meclins is well documenter] for growth but not for lactation. Davis et al. (1984) and Peel et al. (1985) have shown that somatotropin administra- tion increases IGF. IGF-I and IGF-II re- ceptors have been reported in bovine and porcine mammary tissue (Gregor and Bur- leigh, 1985; Pee} and Bauman, 19871. There is no doubt that growth factors play a pivotal role in mammary glanc] function and clevel- opment. GENETICALLY SUPERIOR COWS AND bSTH-TREATED COWS There are many similarities between ge- netically superior cows and cows treated with growth hormone (Pee} and Bauman, 1987~. Both consume more feed and pref- erentially partition nutrients to the mam- mary gland for milk synthesis. Somatotropin treatment floes not change the digestibility of feed maintenance requirements of ani- mals or the efficiency with which milk is synthesized. The gross feed efficiency is improved, however. This is because main- tenance represents a smaller proportion of consumed nutrients in both the genetically superior cow and the cow that has higher milk yields during bSTH treatment. In genetically superior cows, there is a greater use of body reserves in early lacta- tion. In bSTH-treated cows, increaser! mo- bilization of nutrient reserves to support increased milk yields in the first weeks of hormone treatment occurs. Genetically su- perior cows are thought to contain large quantities of secretory tissue, even though the synthetic activity per secretory cell is not known. Improved management is necessary to APPENDIX optimize reproductive performance in ge- netically superior cows. In cows receiving somatotropin, it is not known what effect the increased milk production will have on reproductive performance. It is likely that there will be no difficulty in well-managed herds. ALTERNATE METHODS FOR INCREASING TtIE EFFICIENCY OF LACTATION Optimizing Milk Removal For copious milk secretion to continue, the milk must be effectively extracted from the udder. -Oxytocin is the principal hor- monal factor responsible for milk ejection. Under normal conditions, 15 to 30 percent of the milk procluced by the udder can be left in the unpiler after milking. Moreover, there appears to be a negative feedback system wherein the milk remaining in the ud(ler can influence further secretion rates. If milk ejection can be optimized to reduce this residual milk, then milk yields can be increase(1 significantly. Growth Factors and Other Biological Compounds Animals can be made to produce milk more efficiently by treatment with specific agents such as somatomedins, neurotrans- mitters, or specific growth factors. Mono- clonal antibodies directed toward key en- zymes controlling milk secretion could be utilized. Once the mechanisms underlying autocrine regulation of growth are deter- minecl, the chemical factors can be isolate(l, identifie(l, and procluced by genetic engi- neering. Genes for specific growth factors can also be incorporated into appropriate somatic cells of animals. These cells in turn can be transplanted back into the animal's body after their genetic code has been altered to produce the desired growth fac tor.
LACTATION BIOLOGY Hormone Receptors One of the key steps in the action of a hormone is the binding of that hormone to its receptor within or on the cell. Increases in animal efficiency may come about in the future if the amplification steps to subse- quent hormonal binding of the ligand with its receptor can be regulated. Methods that will increase the affinities of the ligand for its receptor so that the desired physiological response will take place can be clevelopecl. Alternatively, unclesirec! or negative control mechanisms brought about by interactions of hormones or growth factors with their receptors can be moclulatecI. Controlling Involution The mammary glanc! undergoes a natural period of regression or involution. During this time, either the rate of milk synthesis diminishes or the cells begin to age and die. The mechanisms of senescence in the mam- mary glance must be examiner! so that lac- tation can be extender] indefinitely, thereby increasing the efficiency of milk secretion. Controlling the Environment Cows are very sensitive to environmental stresses such as heat ant] humidity. Fortu- nately, great progress has been made in improving the management practices of farms in areas where heat and humidity are a problem. Hormones critical for the control of lactation are sensitive to temperature, humidity, and photoperiod (Tucker, 1985b). Development of methods to optimize the environmental conditions to promote secre- tion of galactopoietic factors would certainly lead to increased animal efficiency. Genetic Selection Dairy scientists conducting research in animal breeding have made great progress in improving lactational efficiency. How 219 ever, it takes a long time for improvements to be realized through the genetic selection process, and scientists are constantly search- ing for physiological factors that can be correlates] immecliately to the animal's fu- ture ability to produce milk. Several workers have shown that serum concentrations of growth hormone are associated with milk production (Barnes et al., 1985; Flux et al., 1984; Hart et al., 1978~. Substances like bSTH that can be directly correlated with milk production could be used effectively for the genetic selection of animals. SUMMARY The mammary gland is a very complex organ system typically classifier] as an exo- crine or cluct gland. It is an "accessory organ" to the reproductive system and is specializes] in both structure and function. Most of the major development of the gland occurs after the start of pregnancy. Blood hormones cluring pregnancy play a major role in controlling the extent of mam- mary gland development. Lactogenesis is also uncler the influence of hormones. The mammary glancl, therefore, provides a tis- sue system in which the action of a hormone can be isolated and the precise role of the hormone inducing the synthesis process can be stuclied. Milk secretion is controllecl by several factors. The total amount of milk procluced and its composition can be altered by changes in the hormonal, environmental, and nutri- tional status of the animal. Growth hormone and thyroactive compounds are galacto- poietic. However, for milk secretion to continue, the secretory products must be effectively removed. This requires a func- tional milk ejection reflex. Milk removal requires neural stimulation through suck- ling or milking procedures. The nerve stim- ulus induces the release of the hormone oxytocin, which causes the myoepithelial cells surrounding the milk-producing alveoli to contract, thus forcing the milk from the
220 alveoli into the ducts. If milk is not evacu- ated, milk synthesis decreases and eventu- ally stops with the complete involution of the secretory tissue. Thus, milk secretion requires the interplay of hormonal, nutri- tional, and neurohormonal processes in ad- clition to evacuation of the milk by suckling or milking. A very unique aspect of the mammary gland is that a gradual drop in the level of milk production occurs after the peak level of secretion. This happens regarcIless of the nutritional regime of the animal or the intensity of the suckling stimulus. This is a normal process similar to senescence or aging and reacts to the complete cessation of secretion. Normal secretion can only resume after another pregnancy, after which the secretory tissue has developed in the udder. With the recent breakthroughs in molec- ular biology, it is now possible to increase mammary growth and the efficiency of milk production in dairy animals. Bovine soma- totropin increases mammary growth in pre- pubertal calves. This increase in growth could leacI to greater milk production per individual animal during lactation. Increases in milk production of up to 40 percent have been clemonstrate<1 with exogenous admin- istration of bSTH. Cows adjust their nu- trient intake upward to support this in crease. Additional ways of increasing the effi- ciency of lactation could be achiever! by treating animals with specific agents such as somatomeclins, antibodies, or specific growth factors. Once the mechanisms un- clerlying autocrine regulation of growth are cleterminecI, the chemical factors involved can be isolated, identifiecl, and produced by genetic engineering. Genes for these substances can also be incorporated directly into the host's cells. Numbers of hormonal receptor sites and amplification steps sub- sequent to binding may be manipulated to alter the efficiency of milk production. Milk secretion will be optimized further after it APPENDIX is more thoroughly understood how to in- crease the efficacy of milk ejection. In- creasec! milk production can be achieved per animal by developing methods that retard mammary involution, thereby lengthening the period of lactation. Environmental factors can adversely in- fluence milk production. When those factors that optimize milk secretion are controlled, the efficiency of lactation is increased. If strong correlations between blood hormone concentrations and total milk production can be ma(le, there is a potential for se- lecting animals for high milk production. The milk of the dairy cow is a very important economic commodity because of its nutritional nature and the products that are procluced from it and its by-products. With the increasing size of the world pop- ulation and the decreasing numbers of dairy farms, it is imperative that we seek ways to increase animal production by increasing the animal's own efficiencies. REFERENCES Anderson, R. R. 1974. Endocrinological control. P. 97 in Lactation: A Comprehensive Treatise, Vol. 4, B. L. Larson, ed. New York: Academic Press. Asimov, G. J., and N. K. Krouze. 1937. The lactogenic preparations from the anterior pituitary and the increase of milk yield in cows. J. Dairy Sci. 20:289. Barnes, M. A., G. W. Kazmer, R. M. Akers, and R. E. Pearson. 1985. Influence of selection for milk yield on endogenous hormones and metabolites in Holstein heifers and cows. J. Anim. Sci. 60:271. Bauman, D. E., M. J. DeGeeter, C. J. Peel, G. M. Lanza, R. C. Gorewit, and R. W. Hammond. 1982. Effects of recombinantly derived bovine growth hormone (bGH) on lactational performance of high yielding dairy cows. J. Dairy Sci. 65(Suppl. 11:121 (Abstr. ). Bauman, D. E., P. J. Eppard, M. J. DeGeeter, and G. M. Lanza. 1985. Responses of high-producing dairy cows to long-term treatment with pituitary somatotropin and recombinant somatotropin. J. Dairy Sci. 68:1352. Brumby, P. J., and J. Hancock. 1955. The galacto- poietic role of growth hormone in dairy cattle. N. Z. J. Sci. Technol. 36A:417. Cowie, A. T. 1969. General hormonal factors involved in lactogenesis. P. 157 in Lactogenesis: The Initiation
LACTATION BIOLOGY of Milk Secretion at Parturition, M. Reynolds and S. I. Folley, eds. Philadelphia: University of Penn- sylvania Press. Davies, D. T., C. Holt, and W. W. Christie. 1983. The composition of milk. In Biochemistry of Lacta- tion, T. B. Mepham, ed. Amsterdam: Elsevier. Davis, S. R., P. D. Gluckman, and I. C. Hart. 1984. Effects of growth hormone and thyroxine treatment of lactating cows on milk production and plasma concentrations of IGF-I and IGF-II. Proc. Endocri- nol. Soc. Aust. 27(Suppl. 1): 16 (Abstr. ). Dils, R. R. 1983. Milk fat synthesis. P. 142 in Bio- chemistry of Lactation, T. B. Mepham, ed. Amster- dam: Elsevier. Eppard, P. J., D. E. Bauman, and S. N. McCutcheon. 1985. Effect of dose of bovine growth hormone on lactation of dairy cows. J. Dairy Sci. 68:1109. Flux, D. S., D. D. S. McKenzie, and G. F. Wilson. 1984. Plasma metabolite and hormone concentra- tions in Friesian cows of differing genetic merit measured at two feeding levels. Anim. Prod. 38:377. Forsyth, I. A. 1983. The endocrinology of lactation. P. 309 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Fronk, T. J., C. J. Peel, D. E. Bauman, and R. C. Gorewit. 1983. Comparison of different patterns of exogenous growth hormone administration on milk production in Holstein cows. J. Anim. Sci. 57:699. Gorewit, R. C. 1979. Method for determining oxytocin concentration in unextracted sera, characterization in lactating cattle. Proc. Soc. Exp. Biol. Med. 160:80. Gorewit, R. C., and M. C. Aromando. 1985. Mecha- nisms involved in the adrenalin-induced blockade of milk ejection in dairy cattle. Proc. Soc. Exp. Biol. Med. 180:340. Gorewit, R. C., and J. McOsker. 1983. Isolation, separation and pure culture of mammary secretory and myoepithelial cells from lactating cows. J. Dairy Sci. 66(Suppl. 1~: 106 (Abstr. ). Gorewit, R. C., and N. Rizzo. 1986. Ultrastructural features of mammary gland myoepithelial cells cul- tured on plastic and mammary gland collagen sub- strates. J. Dairy Sci. 69(Suppl. 1):204 (Abstr. ). Gorewit, R. C., and R. Sagi. 1984. Effects of exogenous oxytocin on production and milking variables of cows. J. Dairy Sci. 67:2050. Gorewit, R. C., R. Sagi, E. A. Wachs, and W. G. Merrill. 1983. Current concepts on the role of oxytocin in milk ejection. J. Dairy Sci. 66:2236. Gorewit, R. C., M. C. Aromando, and D. G. Bristol. In press. Measuring mammary gland blood flow using a transit-time ultrasonic flow probe. J. Appl. Physiol. Gregor, P., and B. D. Burleigh. 1985. Presence of high affinity somatomedin/insulin-like growth factor receptors in porcine mammary gland. Endocrinology 116(Suppl. 1):223 (Abstr. ). 221 Hammond, J. 1927. The Physiology of Reproduction in the Cow. London: Cambridge University Press. Hart, I. C., J. A. Bines, S. V. Morant, and J. L. Ridley. 1978. Endocrine control of the levels of hormones (prolactin, growth hormone, thyroxine and insulin) and metabolites in the plasma of high- and low-yielding cattle at various stages of lactation. J. Endocrinol. 77:333. Hart, I. C., J. A. Bines, S. James, and S. V. Morant. 1985. The effect of injecting or infusing low doses of bovine growth hormone on milk yield, milk composition and the quantity of hormone in the milk serum of cows. Anim. Prod. 40:243. Hooker, C. W., and W. F. Williams. 1941. Retardation of mammary involution in mice by injection of lactogenic hormone. Endocrinology 28:42. Kuhn, N. J. 1983. The biosynthesis of lactose. P. 159 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Lenfers, P. 1907. Zur Histolgie der Milchdruse des Rindes. Z. Fleisch- Milchhygiene 17:340. Lockwood, D. H., F. E. Stockdale, and Y. J. Topper. 1967. Hormone-dependent differentiation of mam- mary gland: Sequence of action of hormones in relation to cell cycle. Science 156:945. Lough, D. S., L. D. Muller, R. S. Kensinger, T. F. Sweeney, L. C. Griel, and T. D. Etherton. 1984. Effect of dietary fat and exogenous growth hormone on the performance of early lactation Holstein cows. J. Dairy Sci. 67(Suppl. 1):115 (Abstr.~. Machlin, L. J. 1973. Effect of growth hormone on milk production and feed utilization in dairy cows. J. Dairy Sci. 56:575. Mather, I. H., and T. W. Keennan. 1983. Function of endomembranes and the cell surface in the se- cretion of organic milk constituents. P. 231 in Bio- chemistry of Lactation, T. B. Mepham, ed. Amster- dam: Elsevier. Meites, J. 1961. Farm animals: Hormonal induction of lactation and galactopoiesis. Ch. 8 in Milk: The Mammary Gland and Its Secretion, Vol. 1, S. K. Kon and A. T. Cowie, eds. New York: Academic Press. Mepham, T. B. 1983. Physiological aspects of lactation. P. 13 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Mercier, J. C., and P. Gaye. 1983. Milk protein synthesis. P. 177 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Paape, M. J., and H. A. Tucker. 1969. Influence of length of dry period on subsequent lactation in the rat. J. Dairy Sci. 52:518. Peaker, M. 1983. Secretion of ions and water. P. 292 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Peel, C. J., and D. E. Bauman. 1987. Somatotropin and lactation. J. Dairy Sci. 70:474.
222 Peel, C. J., D. E. Bauman, R. C. Gorewit, and C. J. Sniffen. 1981. Effect of exogenous growth hormone on lactational performance in high-yielding dairy cows. J. Nutr. 111:1662. Peel, C. J., T. J. Fronk, D. E. Bauman, and R. C. Gorewit. 1983. Effect of exogenous growth hormone in early and late lactation on lactational performance in dairy cows. J. Dairy Sci. 66:776. Peel, C. J., T. J. Fronk, D. E. Bauman, and R. C. Gorewit. 1982. Lactational response to exogenous growth hormone and abomasal infusion of a glucose- sodium caseinate mixture in high-yielding dairy cows. J. Nutr. 112:1770. Peel, C. J., L. D. Sandles, K. J. Quelch, and A. C. Herington. 1985. The effects of long term adminis- tration of bovine growth hormone on the lactational performance of identical twin dairy cows. Anim. Prod. 41:135. Pitelka, D. R., and S. T. Hamamoto. 1983. Ultrastruc- ture of the mammary secretory cell. P. 249 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Plaut, K., D. E. Bauman, and N. Agergaard. 1985. Effects of exogenous prolactin on lactational per- formance of dairy cows. J. Dairy Sci. 68(Suppl. 1~: 169 (Abstr.). Richard, A. L., S. N. McCutcheon, and D. E. Bauman. 1985. Responses of dairy cows to exogenous bovine growth hormone administered during early lactation. J. Dairy Sci. 68:2385. Rivera, E. M., and H. A. Bern. 1961. Influence of insulin on maintenance and secretory stimulation of mouse mammary tissues by hormones in organ- culture. Endocrinology 69:340. Schmidt, G. H. 1971. Biology of Lactation. San Fran- cisco: W. H. Freeman. Schmidt, G. H., R. T. Chatterton, and W. Hansel. 1962. Histological changes during involution of the mammary gland of ovariectomized and intact lactat- ing goats. J. Dairy Sci. 45:1380. Schwalm, J. W., and H. A. Tucker. 1978. Glucocor- ticoids in mammary secretions and blood serum during reproduction and lactation and distributions of glucocorticoids, progesterone and estrogens in fractions of milk. J. Dairy Sci. 61:550. Sejrsen, K., J. Foldager, M. T. Sorensen, R. M. Akers, and D. E. Bauman. 1986. Effect of exogenous bovine somatotropin on pubertal development in heifers. J. Dairy Sci. 69:1528. Sinha, Y. N., and H. A. Tucker. 1969. Mammary development and pituitary prolactin level of heifers from birth through puberty and during the estrous cycle. J. Dairy Sci. 52:507. Smith, G. H., B. Crabtree, and R. A. Smith. 1983. Energy metabolism in the mammary gland. P. 121 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. APPENDIX Soloff, M. S., and T. L. Swartz. 1973. Characterization of a proposed oxytocin receptor in rat mammary gland. J. Biol. Chem. 248:6471. Thatcher, W. W., and H. A. Tucker. 1966. Effects of intense nursing stimulation on lactation. J. Anim. Sci. 25:932 (Abstr.~. Thomas, J. W., and L. A. Moore. 1953. Thyroprotein feeding to dairy cows during successive lactations. Dairy Sci. 36:657. Thomas, J. W., D. V. Kopland, E. A. Keyes, and L. A. Moore. 1957. A study of the short-term use of iodinated casein for milk production. J. Dairy Sci. 40:128. TrauIg, H. H. 1967. Cell proliferation in the mammary gland during late pregnancy and lactation. Anat. Rec. 157:189. Tucker, H. A. 1964. Influence of number of suckling young on nucleic acid content of lactating rat mam- mary gland. Proc. Soc. Exp. Biol. Med. 116:218. Tucker, H. A. 1974. General endocrinological control of lactation. P. 277 in Lactation: A Comprehensive Treatise, Vol. 4, B. L. Larson, ed. New York: Academic Press. Tucker, H. A. 1985a. Endocrine and neural control of the mammary gland. P. 39 in Lactogenesis: The Initiation of Milk Secretion at Parturition, M. Rey- nolds and S. I. Folley, eds. Philadelphia: University of Pennsylvania Press. Tucker, H. A. 1985b. P. 211 in Recent Advances in Animal Nutrition, W. Haresign and D. J. A. Cole, eds. Storeham, Mass.: Butterworth. Tucker, H. A., and R. P. Reece. 1963a. Nucleic acid content of mammary glands of pregnant rats. Proc. Soc. Exp. Biol. Med. 112:370. Tucker, H. A., and R. P. Reece. 1963b. Nucleic acid content of mammary glands of lactating rats. Proc. Soc. Exp. Biol. Med. 112:409. Tucker, H. A., and R. P. Reece. 1963c. Nucleic acid content of mammary glands of rats lactating 41 and 61 days. Proc. Soc. Exp. Biol. Med. 112:688. Tucker, H. A., and R. P. Reece. 1963d. Nucleic acid content of rat mammary glands during post-lacta- tional involution. Proc. Soc. Exp. Biol. Med. 112:1002. Tucker, H. A., R. P. Reece, and R. E. Mather. 1961. Udder capacity estimates as affected by rate of milk secretion and intramammary pressure. J. Dairy Sci. 44:1725. Turner, C. W. 1939. The Comparative Anatomy of the Mammary Gland. Columbia, Mo.: University of Missouri Press. Turner, C. W. 1952. The Mammary Gland, Vol. 1, The Anatomy of the Udder of Cattle and Domestic Animals. Columbia, Mo.: Lucas. Wachs, E. A., R. C. Gorewit, and W. B. Currie. 1984a. Half-life, clearance, and production rate for oxytocin in cattle during lactation and mammary involution. I. Dom. Anim. Endocrinol. 1:121.
^~o~ ~oLocy Chubs, E. AL B. C. Corew~, and W. B. Currie. I984b. Oxytocin concentrations of cattle in response to milking stimuli tbrou~ lactation and involution. H. Dom. Dim. End. LI4I. Madams, W. L. I94S. We enact of lactogenic hormone on post parturient unsuckled mummed glands of the mouse. AnaL Rec. 93:I7I. Young, F. C. 1947. Experiments sUmulabon (g~ac- topoies~) of lotion. Br. Wed. BuN. S:I55. boa Zhao, X., and B. C. Corewit 1986. Cb~acterization of oxytocin receptors in mummed tissue Gom 1~- tabng and nonlact~ing cows. ~ Dad ScL 69(Suppl. D: I65 (Abstr. ). ohm, X~ and B. C. Corewit 1987. Oxytocin receptors in bovine mummed Ussue. ~ Recept. Res. 7:72 741.
Factors Affecting the Composition of Milk from Dairy Cows J. G. LINN Milk composition is economically impor- tant to milk producers and processors and nutritionally important to consumers. It has been known for years that variations in milk composition occur; however, the composi- tion of milk marketed nationally has been rather constant over the last 15 years, av- eraging 3.6 percent fat, 3.2 percent protein, and 4.7 percent lactose (Young et al., 1986~. This is probably partly because of the prom- inence of the Holstein breed and the pricing of milk based on fat concentration. The introduction of milk pricing on a component basis and the perception by consumers that animal fats are unhealthy have created new interest in how milk components can be altered to accommodate these emerging markets. The paper starts with a brief over- view of the biosynthesis of milk components since changes in these reflect changes in the mammary gland synthesis or secretion of the component. Factors affecting milk composition such as breed, genetic variation within breed, health, environment, man- agement practices, and diet are then re- viewed. 224 LIPIDS Biosynthesis The synthesis of milk fat has been recently reviewed (Dils, 1983, 1986; Larson, 1985; Rook and Thomas, 1980). The following discussion highlights relevant stages of bio- synthesis where fat composition can be altered. The predominant fat in milk is triacylglyc- erol, which contains fatty acids of short- (C4-C~o), intermediate- (C~2-C~6), or Tong- chain (Cat) length. The short-chain acids are synthesized within the mammary gland from acetate and beta-hydroxybutyrate; long-chain acids are almost exclusively derived from blood plasma fatty acids of dietary origin; and intermediate-chain acids arise from both sources. In broad terms, about 50 percent of the fatty acids in milk are synthesized in the mammary gland and the other 50 per- cent are derived directly from blood. Fatty acids are synthesized in the mam- mary gland via the malonyl coenzyme A (malonyI-CoA) pathway. Blood beta-hydrox- ybutyrate is primarily used for the first four
COMPOSITION OF MILK FROM DAIRY COWS carbons initiating fatty acid synthesis. Chain length occurs two carbons at a time, with acetate being the major carbon donor. Once formed, fatty acyI-CoAs may combine with glycerol or diacy~glycero! or monoacylglyc- ero! to form triacy~glycerol. Placement of the fatty acids on the glycerol molecule is not random. Short-chain fatty acids are essentially in the 3 position, ant! Cog acids are on either the 1 or 3 position. Triacylglycerols are synthesizer! in the endoplasmic reticulum. As they are formed, they are rapidly incorporated into lipid- covered droplets. The droplets then migrate toward the apical membrane ofthe secretory cell, where they become encapsulated by the membrane, pinched off, and released into the lumen. Factors Affecting Milk Fat Content BreedlGenetics Between and within breecls, fat varies the most and lactose the least (WooUford et al., 1986~. Gaunt (1980) reported cattle in the United States tent! to have the lowest per- centage of milk fat. This may be partly because of environmental factors, but some genetic variation within a breec] in different countries must exist. The repeatability from one lactation to another for the percentage of constituents in milk is quite high, an average of 0.67 (Gaunt, 1980~. Repeatability of milk fat percentage for Holsteins is 0. 76. Other breeds appear to have a similar repeatabil ity. Jerseys have the highest heritability for milk fat percentage (0. 71), with other breeds ranging from 0.51 to 0.57. The small vari- ation between ratios of one milk constituent to another, particularly fat to protein, sug- gests little hope for drastic changes in milk yield and milk composition (Gaunt, 1973; Wflcox, 1978~. Heritabflities of solids-not- fat (SNF) to fat and protein to fat ratios are highest for Ayrshire followed by Jersey, 225 Guernsey, Brown Swiss, and Holstein. Dif- ferences in heritabflities of breeds other than Holstein may be overestimated be- cause of a small sample population. Genetic correlations between milk com- position percentages are high and positive, averaging 0.74. However, milk yield and composition percentages are negatively cor- related, -0.3 for milk yield and fat per- centage (Gaunt, 1980~. Thus, it is very difficult to improve milk yield and milk percentage composition simultaneously. Selection in Holstein cattle for the single trait of milk fat percentage would decrease milk yield by 287 pounds but increase fat percentage by 0.19 percent per generation. Selection for milk yield only increases milk yield by 607 pounds and decreases fat per- centage by 0.036 percent. Selection for milk fat yield is the most effective method for increasing fat percentage (+0.058 percent) and milk yield ~ + 443 pounds) (Gaunt, 1980~. E nvironment/Management A (lecrease in milk fat percentage of 0.2 percent over five lactations has been re- ported by Rogers ant! Stewart (19824. Fat yields would be expected to increase, since the increase in milk yields with age more than offsets the drop in fat percentage. Milk fat percentages vary with stage of lactation. The highest percentages are usu- ally fount] in colostrum, followed by a (le- cline during the first 2 months of lactation, then a slow increase as lactation progresses. Davies et al. (1983) reported distinct changes in the fatty acid content of milk over the lactation cycle. During the first half, the proportions of short- and intermecliate-chain fatty aci(ls increase, and the proportion of long-chain fatty acids decreases. No further changes occur during the last half of lacta- tion. Some of these changes are influenced by environment, diet, and rates of fatty acid synthesis in the mammary gland. Seasonal variations in milk fat percentages are well recognized, with summer months
226 averaging 0.4 percentage units less . . . . than winter months (lenness, 19851. The higher environmental temperatures during the summer also affect milk fatty acid compo- sition. Milk fat in the summer tends to be lower in palmitic acid relative to stearic and octadecanoic acids than milk fat from the same cows during the winter (Christie, 19794. Some of the changes in milk fat percentage and composition with tempera- ture change can be related to changes in blood plasma lipids, but these observations are also confounded by dietary changes. Milam et al. (1986) observed no change in milk fat percentage when heat-stresse(l cows were given water at 10 or 28°C. The fat percentage of milk increases con- tinuously during the milking process, with the lowest fat milk drawn first and the highest fat milk drawn last. The increase in fat percentage throughout the milking proc- ess is due to the clustering of fat globules trapped in the alveoli (lenness, 1985~. Thus, if cows are not milker] out completely, fat percentage will be Tower than normal, but, at the next milking, fat content will be higher than normal. Furthermore, when milking intervals are unequal, the highest fat percentage is obtained after the shortest interval (Wheelock, 19801. Milk fatty acid composition is not affected by milking in- terval or time of day milking (Christie, 1979~. The effect of milking three versus two times a day on milk fat percentage has varied, with some researchers reporting no change (Amos et al., 1985; DePeters et al., 1985; Gisi et al., 1986) ant! others reporting decreases (Allen et al., 1986; Gist et al., 1986~. HealthlPhysiology Mastitis (inflammation of the udder) gen- erally causes a decline in milk fat percentage and a change in milk fat composition (Kitchen, 1981; Needs and Anderson, 1984; Schultz, 1977~. The decrease in fat percentage, how- ever, is less (about 10 percent) than that APPENDIX observer! for lactose or casein (about 15 percent). Reported changes in milk fat com- position from mastitis have varied. There is general agreement on increases in amounts of free fatty acids and short-chain fatty acids, but both increases (Needs and Anderson, 1984) and (decreases (Kitchen, 1981; Schultz, 1977) in phospholipid ant! long-chain fatty acids have been reported. The effects of hormones on milk fat per- centage are not well known (Bauman and Elliot, 1983; Tucker, 1985~. It has been demonstrated that adrenaline and nora- drenaline increase lipolytic activity in adi- pose tissue, but their effect on milk fat is unknown. Administration of exogenous growth hormone has resulted both in no change (Bauman et al., 1985; Peel et al., 1985) and in changes (Epparcl et al., 1985) in milk fat percentage en c] composition. At low closes (5 and 10 IU/day), growth hor- mone lowered fat percentage with no change in fat composition, but at high closes (50 and 100 IU/day), milk fat percentage was increased ant] milk fat contained more en- clogenous fatty acicis (Eppard et al., 1985~. Growth hormone affected both synthesis of fatty acids in the mammary gland and uptake of preformed fatty acids from the blood, depending on dose level and energy balance of the cow. Sutton (1980) reported that the use of thyroprotein, 1,3-butanediol, and glucocorticoids have generally not increased milk fat percentage. Nutrition Diets for today's high-producing dairy cows are typically higher in energy from readily fermentable carbohydrates than fats. Feeding of these diets often causes a con- dition known as low-milk-fat syndrome. Characteristics of low-milk-fat syndrome are a reduction in milk fat percentage (as much as 60 percent) and changes in milk fat composition (an increase in C18 polyunsat- urated and monounsaturated acids and de- creases in C~60 and C~80 fatty acids) (Banks
COMPOSITION OF MILK FROM DAIRY COWS et al., 1983; Christie, 19791. Causes of low- milk-fat syndrome probably involve both an alteration in rumen fermentation and avail- abflity of enclogenous fatty acid sources (Christie, 1979~. Feeding of readily fer- mentable carbohydrates depresses fiber digestion and pH in the rumen and thus decreases acetic ant] butyric acid production and increases propionic acic] production. Increased propionic acid concentrations in the rumen lead to increased lactic acid and glucose production, which, in turn, stimu- lates insulin production, reducing free fatty acic] release from adipose tissue. Thus, the main precursors of milk fat (acetic and butyric acids cierivecT from rumen fermen- tation, long-chain fatty acids of dietary ori- gin, and acetic acid and long-chain fatty acids from endogenous sources) can be af- fectec! by diet through changes in rumen fermentation or addition of fats for direct absorption and inclusion into milk fat. Rumen fermentation. Milk fat percent- age is relater! positively to rumen molar percentages of acetic and butyric acids and negatively to that of propionic acid. Davis (1978) reported that rumen molar percent- age of propionate must be above 25 before a highly significant negative relationship between milk fat percentage ant! propionate exists. Sutton (1980) estimated that 60 per- cent of the variations observed in milk fat percentage can be accounted for by changes in the molar proportion of propionate in the rumen. A positive relationship exists between the molar ratio of acetate to propionate and milk fat percentage. A linear increase in milk fat percentage occurs as the ratio of acetate to propionate increases up to 2.2 (Davis, 1978~. Above a ratio of 2.2 there is little change in milk fat percentage. Thus, (liets that in- crease propionate production have the greatest effect on milk fat percentage. Numerous dietary factors affect rumen fermentation (Sutton, 1980~. Those most commonly associated with changes in the 227 acetate to propionate ratio are forage to concentrate ratio, type of carbohydrate in the diet, physical form of the (lies, process- ing of ingredients, additives, and the fre- quency en c] method of offering feed. The following discussion summarizes the influ- ence ofthese factors on rumen fermentation, acetate to propionate ratio, and change in milk fat percentage. The general effect of decreasing the forage to concentrate ratio on rumen fermentation is to decrease pH, increase propionic acid production, and reduce fiber (ligestion. Thus, as forage declines, milk fat percentage falls proportionately; however, milk fat yields may increase (Sutton, 19801. The critical forage to concentrate ratio appears to be about 40:60, beyond which adclitional con- centrate (lrastically lowers milk fat percent- age (Coppock, 1985; Sutton, 1985~. How- ever, Sutton (1980) reported that the actual level of forage needed in a diet to maintain normal milk fat percentage may be affected by total feed intake. At high levels of intake, more forage is needed than at low-intake levels to maintain the same milk fat per- centage. Recent work by Shaver et al. (1986) has shown similar results, with milk fat percentages being higher in milk from cows fed a 60:40 forage to grain diet at 2.93 percent of body weight than at 3.75 percent of body weight. Declines in milk fat per- centage with high-grain feeding are accom- panied by a change in milk fatty acid com- position from saturated fatty acids to more unsaturated acids, especially those contain- ing 16 carbons or less (Banks et al., 1983; Sutton, 1980~. The type of forage and its effect on milk fat percentage are influenced by forage particle size, maturity, and fiber content of the forage. It has been known for a while that finely ground forages reduce milk fat percentage. Finely ground forages appar- ently result in higher levels of propionate being produced during rumen fermentation than forages of adequate particle size (Sut- ton, 1980~. Recent work by Woodford et al.
228 (1986) has shown that a mean forage particle length of 0.64 cm or more is needed to keep rumen molar percentage of propionate be- low 25 and milk fat above 3.6 percent. Mertens (1985) recommended a minimum of 28 percent neutral detergent fiber and about 18 percent acid detergent fiber in diets to maximize milk procluction and fat percentage. The daily amount of neutral detergent fiber needed was estimates] to be 1.2 percent of body weight. Stage of forage maturity is an important factor in the supply of adequate fiber in the diet. More immature alfalfa hay was re- quired in the cliet to obtain maximum pro- duction of 4 percent fat-corrected milk than when micI- or late-bloom alfalfa hay was fed (Kawas et al., 1983~. Recent work (Hansen et al., 1984) has shown that an interaction between forage species ant! concentrate level in the diet affects milk fat percentage. Bro- megrass supported a higher milk fat per- centage at higher concentrate feeding than did alfalfa. No difference between the two forage sources was observed at lower con- centrate levels. Carbohydrate source can influence rumen fermentation ant! consequently milk fat per- centage. Sutton (1985) reported that the lower ruminal clegradability of corn com- pared with that of barley would result in the production of milk with a higher fat percentage. Recent work (DePeters ant! Taylor, 1985) has confirmed that barley- based concentrates tend to depress fiber digestibility, resulting in lower ruminal ace- tate to propionate ratios and lower milk fat percentages than those with corn-based con- centrates. The higher digestion of barley in the rumen produces more propionate ant! results in less starch being presented to the lower digestive tract for conversion to glu- cose than with corn. However, the increased production of propionate in the rumen from barley appeared to stimulate milk yield more than glucose derivecl directly from corn in the lower (ligestive tract. The mech- anism~s) by which these two (differences in APPENDIX nutrient supply affect milk fat is not well known. Processing of grains such as grind- ing, rolling, heating, steam flaking, and pelleting increases digestion of the starch in the rumen en c! produces effects similar to those reporter] above for barley (Sutton, 1980~. Increasing butyric acid production in the rumen shouIc! also help to maintain or increase milk fat percentages. Sutton (1980) suggested that beet pulp is a promoter of butyric acid production in the rumen. Other carbohydrates such as whey (Casper ant] Schingoethe, 1986; Schingoethe, 1976), su- crose, and lactose (Sutton, 1980) have been evaluated as sources of soluble carbohydrate to prevent milk fat depression. The pattern of feeding, often referrer! to as beetling strategy, was found to have little if any benefit in terms of increasing milk fat percentage under normal conditions (Linn and Otterby, 1984~. However, under feed- ing regimes where fat-depressing conditions are likely, increasing the frequency of of- fering concentrates to six or more times per day appears to stabilize the rumen environ- ment (Bragg et al., 1986) and increase milk fat percentage (Sutton, 1980, 19851. The mixing of all feed ingredients before feeding does not affect milk fat yield or percentage any differently than if the ingre- dients were fed separately (Holler et al., 1977; Marshall and Voigt, 1975; Owen, 1981~. Thomas and Chamberlain (1984) sum- marize;1 the effects of infusion of specific nutrients into cows on changes in milk constituents. Intraruminal infusions of acetic acid consistently increase milk yielcI, lactose yielcl, en cl milk fat yield, whereas infusions of propionate re(luce milk fat yield. Glucose infusions, either intraabomasal or intrave- nous, increase milk yield ant] decrease milk constituent percentages. Infusions of pro- tein or amino acids (Schwab et al., 1976) have had variable or no effect on milk fat percentage. The effects of dietary protein on milk fat
COMPOSITION OF MILK FROM DAIRY COWS percentage are variable but generally small when diets within normally accepted ranges of nutrients have been fed (Sutton, 1980; Thomas ant! Chamberlain, 1984~. Changes in fat percentage result from changes in milk yield rather than from a direct effect of dietary protein source or amount. Insuf- ficient amounts of rumen-clegradable pro- tein may lower milk fat percentage because of a lack of ruminal ammonia for optimal microbial digestion of fiber and other feed- stuffs. Additives such as buffers and methionine hyciroxy analog have been used to promote increases in milk fat percentage. Cows in early lactation fee! high-concentrate diets were shown to benefit from the inclusion of the methionine hydroxy analog in their rations (Lun~quist et al., 1983~. Feeding of 25 grams of methionine hyclroxy analog daily during the first 120 days of lactation in- creased milk fat 0.35 percentage units. Buffers are compounds used to raise ru- men pH through the neutralization of vol- atile fatty acids. However, other modes of action have been indicated for the group of compounds commonly alluded to as buffers (soclium bicarbonate, potassium bicarbon- ate, limestone, magnesium oxide, and ben- tonite) (Chalupa and Schneider, 1985~. In general, the bicarbonates have been effec- tive in maintaining or increasing milk fat percentages of cows fed high-grain diets, especially when corn silage was the main forage source (Chalupa and Schneider, 1985; Davis, 1978; Sutton, 1980~. Magnesium ox- ide has also been shown to help prevent milk fat percentage depression; however, it appears that the mechanism of action is through transfer of lipid into the mammary gland from blood rather than through a change in rumen fermentation (Chalupa and Schneider, 1985~. Added fats. Dietary fats can alter milk fat composition in a number of ways (Chris- tie, 1979~. One route is for fatty acids to be unaltered cluring digestion and absorption 229 and therefore appear in milk fat directly. Another route is for the rumen microorga- nisms to hydrogenate the fatty acicI, which can then appear in milk fat in this form or be further modified by desaturation before appearing in milk fat. Dietary fatty acids can appear in milk fat in the same form in which they were fed or be completely changed to another form before entering milk. In acldition, the amount of particular fatty acids in the diet can alter lipid metabolism in the animal through mammary glanc] uptake problems or enzyme inhibitions. Dietary long-chain fatty acids can affect rumen fer- mentation and thus alter the amount of volatile fatty acids (acetic, propionic, and butyric acids) available for fat synthesis in the mammary gland. The use of fats and oils in the (1iets of dairy cows has received considerable atten- tion (Fogerty ant! Johnson, 1980; Linn, 1983; Palmquist and Jenkins, 1980; Storry, 1980; Storry and Brumby, 19804. Numerous lipid sources, from natural to manufactured, have been evaluated. Their effects on milk yield and composition depend on type of fat, characteristics of the diet into which they are incorporated, rate and form fecI, and method of feecling. Only a brief sum- mary of changes in milk fat percentage and composition is reported here. The changes in milk fat percentage and composition observed with the use of fat in diets of dairy cows are a reflection of the change in output of different fatty acids from the mammary gland; short- and medium- chain fatty acids (C4 to Ci4) are synthesized in the mammary gland, the Cog fatty aci(ls come from the diet, and the Can fatty acids come from both synthesis and clietaIy sources. Although dietary fats and oils may alter milk fat composition, the output of total milk fat depends on the balance of increased dietary transfer and decreased synthesis. However, there is probably a minimum content of short-chain fatty acids necessary to maintain melting points at body temperatures (Chris- tie, 1979~.
230 Both protected and unprotected fats and oils have been fed to dairy cows. Some of the unprotected fat or oil sources reported in the literature are tallow, yellow grease, vegetable oils, blends of animal-vegetable fats, and whole oilseeds (soybeans, sunflow- ers, cottonseed, and rapeseed). The com- mon protected! fat sources, so called because they are unavailable in the rumen and therefore do not alter rumen fermentation, fee! are tallow and vegetable oils. Common methods of protection are formaldehyde- protein coating (Storry and Brumby, 1980) and formation of insoluble calcium salts of the fat Jenkins and Palmquist, 1984~. In general, the addition of unprotected fat to dairy diets results in variable effects on milk yield and milk fat composition. The addition offats, oils, or long-chain fatty acids depresses the synthesis of C4 to Cal fatty acids in the mammary gland. This most likely results from an alteration in rumen fermentation rather than an inhibition of mammary gland acetyl-CoA carboxylase ac- tivity (Banks et al., 1983; Storry, 1980; Thomas, 1980~. The effect on rumen fer- mentation is most pronounced with unsat- uratec! fatty acid beetling. Long-chain fatty acid sources (more than 20 carbons) such as fish oils and Seterculia seed fats have a specific inhibitory action on the uptake of preformed fatty acids by the mammary gland. The changes in milk fat composition that occur with fat feeding are predominantly in the triglyceride fraction, with very little change occurring in the phospholipid and fat membrane fractions (Storry, 1980~. Protected polyunsaturated fatty acids ap- pear to be the most promising for consist- ently increasing milk fat percentage and altering milk fat composition. Protected oil- seeds or oils rich in linoleic acid (sunflower, corn, and soybean) produce large, rapid increases in the linoleic acic] content of milk fat when fed. The increases in linoleic acid content are generally associated with de- clines in myristic, palmitic, and oleic acids. Transfer of linoleic acid from protected APPENDIX supplements to milk is reported to be be- tween 20 and 40 percent (Christie, 1979; Fogerty and Johnson, 19804. Feeding of protected saturated fats, the most common source being tallow, generally invokes the same response in increase of milk fat percentage as feeding of protected polyunsaturated fats. However, protected hydrogenated soybean oil has clecreasec! the milk fat percentage (Banks et al., 1983~. Protected tallow increases the amounts of C4' CI6l, CI8:0, and Cash fatty acids found in milk fat (Christie, 1979~. Similar results were reported for unprotected tallow. PROTEIN The total (cru(le) protein content of milk is determined by analyzing milk for nitrogen and multiplying by a factor of 6.38. The total protein percentage of milk is generally considerecl to be about 3.5, of which 94 to 95 percent is in the form of true protein (Davies et al., 1983; lenness, 1985~. Casein accounts for approximately 80 percent of the true protein, and milk serum or whey proteins account for about 20 percent. Urea is the largest single nonprotein nitrogen (NPN) component, accounting for approxi- mately 50 percent of the total NPN (Wolf: schoon-Pombo and Klostermeyer, 1981~. Milk proteins fall into several families of polypeptide chains, for which a systematic nomenclature system has been defined (Eigel et al., 1984~. Casein proteins are characterized by ester-bound phosphate, high praline con- tents, and few or no cysteine residues and are precipitable from milk at pH 4.6 and 20°C. The main casein types in milk are alpha-, beta-, gamma-, and kappa-caseins. Whey proteins are distinguished from casein by remaining in solution upon precipitation of casein proteins. The major whey proteins are beta-lactoglobulin and alpha-lactalbumin. Serum albumin, immunoglobulins, proteose peptones, lactoferrin, and transferrin repre- sent a smaller proportion of the whey protein fraction (Davies et al., 1983; lenness, 1985; Kuzdzal-Savoie et al., 1980~.
COMPOSITION OF MILK FROM DAIRY COWS Biosynthesis The synthesis of milk proteins has been extensively reviewer! (Larson, 1979, 1985; Mercier ancl Gaye, 1983~. In general, pro- tein synthesis in mammary alveolar cells is similar to other protein synthesis systems in which DNA controls protein synthesis. Messenger RNA carries the encoded DNA message from the nucleus to the ribosomes located in the rough encloplasmic reticulum (RER) and cytoplasm. Ribosomes are com- posec3 of ribosomal RNA and several pro- teins combiner! into a ribonucleoprotein complex, which, in conjunction with trans- fer RNA, combines amino acids into peptide chains. As the polypeptide chains are elon- gatecl to form proteins, they pass out of the HER, through the lumen, and into the region of the Golgi apparatus where they accumulate and polymerize into different milk protein molecules. Casein must be phosphorylated, bound with calcium, and stabilized by calcium phosphate linkages and other ionic bonds before being released from the vesicles. The presence of alpha- lactalbumin in the region of the Golgi ap- paratus promotes synthesis of lactose. The secretory vesicles containing essentially nonfat milk constituents leave the cell by moving to the apical surface and fusing with the plasma membrane and discharging the vesicular contents into the cell lumen. Most of the proteins present in milk are synthesized in the mammary gland, al- though some immunoglobulins and albu- mins are transferred from the blood (Larson, 19791. Blood leukocytes can also cross mam- mary barriers either by passing between secretory cells or by pushing secretory cells directly into the lumen. Urea diffuses freely across mammary cells, so there is a high correlation between blood plasma and milk urea concentrations (Thomas, 1980~. The synthesis of milk protein requires that both essential and nonessential amino acids be supplied to the mammary gland (Clark et al., 1978; Mepham, 1982~. Uptake 231 of free amino acids from the blood by the mammary gland can occur via several trans- port systems (Baumrucker, 1985~. Mepham (1982) has classifier] essential and nonessen- tial amino acids into three groups according to uptake by the mammary gland. Group I essential amino acids (methionine, histi- dine, phenylalanine, tyrosine, and trypto- phan) are taken up in amounts just sufficient to meet milk protein synthesis needs. Group II essential amino acids (valine, leucine, isoleucine, arginine, lysine, and threonine) are taken up in excess. However, some data (Thomas, 1983) suggest that lysine and pos- sibly leucine, isoleucine, and threonine should also be included in group I. Group III is the nonessential amino acicls. The amounts taken up vary with animal, time, and availability. In addition to free amino acid uptake from blood, there is evidence that rec] blood cells and the recycling of amino acids also contribute to the cellular amino acid pool (Baumrucker, 1985~.13reak- down of red blood cell glutathionine can make a significant contribution to the amount of cysteine, glycine, and glutamic acic! avail- able in the cell. Recycling of casein proteins is reporter! to account for at least 7 percent of the protein synthetic capacity in the mammary gland. Factors Affecting Milk Protein Content Bree~lGenetics Breeds differ in total milk protein per- centage and type of milk protein produced. Jersey and Guernsey cattle have the highest percentages of total protein, casein, and whey. Variability of the major protein frac- tions within breeds has also been reporter] (Roller) et al., 1956), with Holstein milk containing less of the major caseins and more gamma-casein than milk from other breecis. Genetic variants have been dem- onstrated for the milk protein groups, and breed differences have been found for the frequency of occurrence of these variants (Gaunt, 19801.
232 Genetic selection would increase the per- centage of protein in milk 0.075 percentage units but decrease milk yield 231 pounds. loins selection for milk yield, protein, and fat is recommencled if the desired result is increased yield of protein and fat (Gaunt, 1980; Van VIeck, 1978; Wilcox, 1978~. Gaunt (1980) estimates] that it would take about 11 generations for milk protein percentages to equal milk fat percentages if protein yield with no change in fat percentage were used as the selection criterion. EnvironmentlManagement ness, 1985; Ng-Kwai-Hang et al., 1982; Rogers ant] Stewart, 19821. Milk protein percentage declines in cows older than 3 years, with a 0.4 percentage unit drop being reported over five lactations (Rogers and Stewart, 1982~. This decline appears to be primarily in the casein fraction; however, changes in whey protein fractions have also been reported (Kroeker et al., 1985~. Sug- gestec! reasons for the change are deterio- ration of unpiler tissue, selective culling for high production, and increased incidences of mastitis. The increase in immunoglobu- lins with advancing age reported by Kroeker et al. (1985) supports the latter suggestion. Stage of lactation has a considerable in- fluence on milk protein concentration (Dav- ies et al., 1983; Ng-Kwai-Hang et al., 1982, 1985; Rogers and Stewart, 1982~. At the beginning of lactation, colostrum is excep- tionally rich in protein containing large quantities of immunoglobulins and about twice the levels of casein, beta-lactoglobu- lin, ancl alpha-lactalbumin fount] in micI- lactation milk. Total protein amounts fall rapidly during the first few days of transition from colostrum to normal milk ant! reach a minimum about 5 to 10 weeks into lactation, corresponding inversely to maximum milk yield. Thereafter, the amount of protein tends either to increase gradually as lacta APPENDlX lion progresses or to rise sharply when the cow becomes pregnant. Milk protein percentage (Ng-Kwai-Hang et al., 1982) and yield (Keown et al., 1986) are higher during fall and winter than spring and summer. However, stage of lactation and feeding practices confound these ob servations as cows on spring pasture have elevated milk protein concentrations (Rog ers and Stewart, 1982~. Whey proteins have been found to have no definite seasonal variations (Kroeker et al., 1985~. High en vironmental temperatures, above 29°C, have been suggester! to depress milk protein percentage, but cows offered cold water Age has a significant e~ecton milk protein(10°C) cluring heat stress do not show in neroenta~f, an] nomno.sition in onw.s (~en-creased milk protein concentrations over cows offered 28°C water (Milam et al., 1986~. Variations in milking procedure or fre quency have a minor effect, if any, on milk protein percentage. Milk protein or SNF percentages clo not change during the milk ing process (lenness, 1985~. Extended milk ing intervals do not change milk protein or SNF percentages until intervals exceed 16 hours (Rogers and Stewart, 1982~. Increas ing milking frequency from twice to three times daily for more than 15,000 Holstein cows dill not change the percentage of SNF (Gist et al., 1986~. Similar results were reported by Amos et al. (1985) and DePeters et al. (1985~. HealthlPhysiology Mastitis has very little effect on total milk protein percentage; however, it drastically alters the composition of milk protein (Kitchen, 1981; Schultz, 1977~. The general effect of mastitis is to impair milk synthesis and loosen the connections between cells, thereby increasing permeability of blood! constituents Jenness, 1985; Wheelock, 1980~. Milk proteins synthesized in the mammary gland (caseins, beta-lactogIobulin, and al pha-lactalbumin) decrease (Kitchen, 1981; Schultz, 1977), whereas blood serum pro teins (whey proteins) increase (Kitchen, 1981;
COMPOSITION OF MILK FROM DAIRY COWS Kroeker et al., 1985; Poutrel et al., 1983; Schultz, 1977~. Grappin et al. (1981) re- ported a whey protein to total protein ratio increase of 2.08 percent and a casein to total protein ratio decrease of 1.85 percent for every 1 log unit increase in somatic cell count. The same change in somatic cell count was reporter! by Ng-Kwai-Hang et al. (1982) to decrease the ratio of casein to total protein by 2.79 percent. The hormone requirement for milk syn- thesis and secretion is prolactin, acirenocor- ticotrophic hormone, and estrogens and the relative absence of progesterone. Of partic- ular importance to milk protein synthesis is prolactin (Tucker, 1985~. Current studies (Bauman et al.. 1985; Peel et al., 1985) on administration of exogenous growth hor- mone have generally shown increases in milk yield without significant changes in composition. However, Eppard et al. (1985) observed a slight decrease in milk protein percentage and an increase in alpha-lactal- bumin as a percentage of total milk protein with increasing dosage levels (0 to 100 IU/ day) of bovine growth hormone. Nutrition Dietary crude protein affects milk yield and consequently milk protein yield more than milk protein percentage (Emery, 1978; Kaufman, 1980; Thomas, 1980, 1983~. A small effect of dietary crude protein con- centration on milk protein percentage was reported by Emery (1978~: a 0.02 percentage unit increase in milk protein with every 1 percentage unit increase in clietary crude protein between 9 and 17 percent. More recently, Cragle et al. (1986) reported an increase of 0.1273 Meal in milk protein energy content per 1 Mcal gross energy increase in feed protein. Neither of these studies, however, considered source of di- etary crude protein or change in milk pro- tein composition. Thus, the increases in milk protein observed may have been in milk NPN and not true milk protein. Ele 233 vate(1 milk protein concentrations from cows fed cliets high in rumen-clegradable protein or NPN most likely will be from increased milk urea or NP~ levels (Oltner et al., 1985; Thomas, 1980~. On the other hand, diets low in rurnen-degraciable protein or bal- anced for optimal microbial protein synthe- sis should increase supplies of amino acids available to the mammary gland for protein synthesis, and thus, more true milk protein should be proclucec! (Kaufman, 1980; Old- ham, 1984; Thomas, 1980~. However, the proportions between true milk proteins (cas- eins, beta-lactoglobulin, and alpha-lactal- bumin) (lo not appear to change with in- creases or decreases in milk protein synthesis (Thomas, 1983~. In experiments where protein (usually casein) has been abomasally infused to in- crease amino acid supplies to the tissue, increases in milk protein percentage along with milk yield have been reporter! (Clark, 1975; Clark et al., 1977~. Abomasal infusions of amino acid mixtures also increased milk protein percentage, with methionine and lysine accounting for more than 68 percent of the observed increase (Schwab et al., 1976~. Baser! on these responses, it could be concluded that increasing the intestinal supply of amino acids through increaser! rumen protein synthesis or low rumen- degradable protein sources would increase milk protein percentage and probably milk yield. However, on a practical feeding basis, milk protein responses to dietary proteins with clifferent rum en ciegradabilities have been quite variable but generally of no effect. A number of studies (Crawford and Hoover, 1984; Crooker et al., 1983; Forester et al., 1983; Henderson et al., 1985; Holter et al., 1985; Kung ant! Huber, 1983; Lund- quist et al., 1986) reported no increases in milk protein when protected proteins were fed. Madsen's (1982) study, however, re- ported significant increases. Again, none of the studies cited determined milk protein composition except that of LuncIquist et al. (1986), which showed no change in milk
234 NPN content within equal dietary crude protein percentages due to feeding formal- dehyde-treated soybean meal compared with feeding an untreated soybean meal. Kaufman (1980) summarized the effects of dietary protein supply on milk protein concentration. Insufficient amounts of die- tary protein will reduce milk protein con- centrations, but the reduction is minimized when low rumen-degradable protein sup- plements are fed. Increasing dietary crude protein supply has little effect on milk protein percentage. The amount of energy consumed, ctens~ty of energy in the flier, and the source of energy in the diet all influence milk protein percentage and yield. Cragle et al. (1986) compared 59 percent versus 49 percent concentrate feeding and found that cows fed rations containing 59 percent concentrate procluced an average of 11 percent more milk, 13 percent more protein, 3 percent more fat, and 11 percent more lactose than cows fed 49 percent concentrate rations. Of the increase in milk protein, 85 percent was attributed to increased yield and only 15 percent to increased percentage in the milk. Emery (1978) reported that milk protein percentage increases 0.015 percent for each Meal of additional net energy fed from 9 to 40 Mcal/ciay and that the increaser! protein percentage was usually accompanied by an increased milk yield. Mild energy malnu- trition has been reported to slightly reduce milk protein percentage; however, uncler severe energy malnutrition, milk protein percentage is unaltered but yields (decrease drastically (Thomas, 1980, 1983~. Rogers and Stewart (1982) reviewed the effects of various forage sources in the diet on milk composition. Cows grazing early spring pastures were reported to have in- creasec3 milk protein percentages. How- ever, the confounding of energy, protein, and condition of the cow in most forage studies where milk composition is reported prohibits the (lrawing of definite conclu- s~ons. APPENDIX Thomas (1980, 1983) discussed the notion that increasing propionic acid in the rumen through increased concentrate feeding or reduced forage particle size affects milk protein percentage. To summarize, there is a strong positive correlation between rumen production of propionic acid and milk pro- tein; however, the exact mechanism is un- known. One suggestion is that propionate increases glutamic acid availability to the mammary gland and, through its role in amino acid transamination, enhances syn- thesis of nonessential amino acids. A second hypothesis is that propionate through in- sulin couIcl enhance plasma concentrations of glutamine and alanine. Propionate could also enhance glutamate output from the liver by increasing its synthesis or reducing usage in gluconeogenesis. Intake of energy can also be increased through inclusion of fats or oils in the diet. Beetling protected lipids, vegetable fats, or vegetable oils to lactating cows depressed milk protein percentage, whereas animal fats had no effect or minimal effect on milk protein percentages (Linn, 1983; Palmquist and Jenkins, 1980~. Dunkley et al. (1977) indicated that the depressing effect was on the casein fraction. Although the exact de- pressing mechanism is unknown, it may be through altered glucose metabolism (Palmquist and Jenkins, 1980), changes in rumen metabolism Jenkins and Palmquist, 1984), or both. Thus, the source of increased dietary energy (carbohydrate versus lipid) fed to lactating cows has a significant eject on milk protein percentage changes. CARBOHYDRATES The predominant carbohydrate in milk is the disaccharide lactose. It is composed of one molecule of glucose and one molecule of galactose joined in a 1-4 carbon linkage as beta-galactoside. The principal biological function of lactose in milk is the regulation of water content and, thus, the regulation
COMPOSITION OF MILK FROM DAIRY COWS of osmotic content (Davies et al., 1983; lenness, 1985~. Because of this function, lactose is the most constant constituent in milk, averaging 4.6 percent. Carbohydrates other than lactose that are found in milk are monosaccharides, sugar phosphates, nucleoticle sugars, free neutral and acid oligosaccharides, ant] glycosyl groups of peptizes and proteins (lenness, 19854. Free glucose and galactose and the sugar alcohol myo-inositol are also present in milk. However, the amounts of these carbohy- drate fractions are minor compared with that of lactose. Biosynthesis Glucose is the primary substrate for lac- tose synthesis, with 85 percent of the carbon secreted in lactose derived from blood glu- cose (Thomas and Chamberlain, 1984~. Lac- tose synthesis is initiated in the Golgi ap- paratus and continues in the vesicles with an influx of water and ionic constituents that causes the vesicles to swell as they pass toward the cell surface. Glucose and uridine cliphosphate (UDP)-galactose, derived from glucose, combine to form lactose under the action of the enzyme lactose synthetase. The milk protein beta-lactalbumin must be present for glucose and UDP-galactose to combine. Thus, beta-lactalbumin appears to be a prime regulator of lactose synthesis (Kuhn, 1983; Larson, 1985~. Entry of water into the vesicle is linked with lactose syn- thesis to maintain osmotic equilibrium with surrounding fluids. Thus, the rate of lactose synthesis regulates water secretion and con- sequently milk yield. MINERALS Factors Affecting Milk Mineral Content The mineral content of milk is derived from minerals fount] in circulating body fluids. The factors influencing mineral con- tent of milk are discusser] below. 235 BreedlGenetics Cerbulis and Farrel (1976) reported the ash, calcium, phosphorus, and magnesium contents of milk from different breeds of dairy cattle. The average ash content varied from 0.74 percent for Holsteins to 0.83 percent for Jerseys. The highest calcium and phosphorus contents in milk were re- ported for lerseys. EnvironmentlManagement It is well documented that the mineral composition of colostrum is higher than that of milk. Calcium, phosphorus, potassium, and chloride concentrations follow the same lactation curves as fat and protein-that is, high in colostrum, lowest at peak milk yield, and then gradually increasing as lactation progresses (Iyengar, 1982; Jenness, 1985~. Milk inorganic phosphorus levels were shown to be higher in first lactation cows than in multiparous cows, and milk phosphate lev- els were lowest during the summer (Forar et al., 1982). HealthlPhysiology Mastitis increases the percentages of so- dium and chloride in milk and decreases the percentage of potassium (Kitchen, 1981; Peaker and Faulkner, 1983; Schultz, 1977~. Bacterial infection of the udder results in damage to the ductal and secretory epithe- lium and increases the permeability of blood capillaries. Thus, sodium-and chloride, which are higher in blood, pour into the lumen of the alveolus, and in order to maintain os- molarity, potassium is decrease propor- tionally. Fernando et al. (1985) reported the decline in lactose and potassium and in- crease in sodium and chloride in mastitic milk was most prominent in strippings of milk after milking. The percentages of calcium and phospho- rus in milk decline with mastitis infections (Kitchen, 1981; Schultz, 1977~. Most likely
236 this reflects lower casein levels, since both ions are complexed with casein micelles. Contradictory evidence exists regarding the effect of mastitic infections on levels of magnesium. Trace elements may increase slightly in mastitic milk (TalIamy and Ran- dolph, 19704. Administration of exogenous growth hor- mone has relatively little effect on the per- centages of minerals in milk, but yields of minerals increased with increasing milk pro- cluction (Epparc] et al., 1985~. Nutrition Normal dietary regimes have little influ- ence on the mineral composition of milk, especially the macromineral constituents. Forar et al. (1982) fed two levels of phos- phorus (0.31 and 0.54 percent) and two levels of calcium (1.0 and 1.8 percent) in four diets to lactating cows and found no differences in milk inorganic phosphorus percentages. Diets depressing milk fat per- centage have been shown to lower the percentage of citrate and soluble calcium in milk (Davies et al., 1983~. Changes in milk phosphorus en cl calcium percentages woulc! not be expected, since very few of these ions are in the free form in milk. Dietary factors affecting citrate ant! casein contents of milk would be expected to correspond with small changes in calcium, since calcium is complexed and secreted with these sub stances. Fettman et al. (1984) observed decreases in milk chloride percentage when cows were fed chloricle-deficient rations during early lactation. Milk potassium percentage de- clinec] along with chloride levels, reflecting altered mineral metabolism in chloricle-de- ficient cows. A recent report (Schneider et al., 1986) evaluating dietary sodium and potassium effects on heat-stressed cows found no change in milk potassium percentages based on quantity of potassium fed or source of sodium fed. However, cows offered shacle had higher milk potassium percentages than APPENDIX cows given no shade. Percentages of sodium in milk were lowered significantly by feed- ing cows sodium bicarbonate and only slightly by feeding salt or high levels of potassium, as compared with results for control cows. The lower milk sodium percentages corre- spondec3 with lower plasma sodium levels in cows fee} sodium bicarbonate. Milk iodine levels have been shown to increase with increased beetling of iodine. Franke et al. (1983) observed progressive increases in milk iodine concentrations dur- ing lactation when as little as 4 ppm of organic iodine were a(lded to the (lies. Larson et al. (1983) found cow age, season of calving, milk production, and health sta- tus to have no effect on the concentration of iodine in milk. Iyengar (1982) reported that iocline, man- ganese, molybdenum, selenium, zinc, and cobalt concentrations in milk could be al- tered by dietary means. However, very limited research has been directed toward! this end. Most of the changes that have been observed are the result of marked dietary changes. The effects of slightly un- clerfeeding or overfeeding a required dietary mineral or the effects of mineral interactions on the mineral composition of milk are not well known. OTHER MILK SOLUBLES There are many other components in milk in adclition to those already discussed. They can be categorized as either natural or contaminant. They appear in milk both from leakage during the normal secretory process and by actual secretion. Whatever the mode of entry, their concentrations can vary con- siderably, but their significance and purpose remain largely unknown. The natural compounds that have been detected in milk are gases, alcohol, alde- hydes, ketones, carboxylic acicI, sulfur-con- taining compounds, nucleotide material, hormones, phosphate esters, glucose, ace- tate, and citrate. Many of these are products
COMPOSITION OF MILK FROM DAIRY COWS of intermediary metabolism ofthe mammary gland (Davies et al., 1983; lenness, 1985; Peaker and Faulkner, 1983~. The reasons for changes in the concentrations of these compounds in milk are unknown. An exception to the above is the com- pounc] citrate. The concentration of citrate in milk is moclifiable and is important from a milk-processing standpoint. Alteration of citrate concentrations changes the amount of free calcium in the soluble phase of milk, which, in turn, affects the precipitation of milk proteins. Milk citrate concentrations are highly correlates] with fat percentage, ant] therefore diets that lower milk fat per- centage also decrease the citrate content of milk (Faulkner ant] Peaker, 1982~. Stage of lactation and season of the year also affect milk citrate levels. It appears that the source of milk citrate is from synthesis within secretory cells and that secretion into milk is similar to that of lactose and casein (Faulk- ner and Peaker, 1982~. The other category of components-con- taminants inclucles compounds that are not normally found in milk but that enter acciclentally or by design. Included here are chemicals, pesticides, herbicides, fungi- cides, heavy metals, and drugs. These items are mentioned as a reminder that milk can contain compounds other than those of nu- tritional importance to humans and that maintaining a nutritious, wholesome milk supply is of utmost importance. MANIPULATING MILK CONSTITUENT~SUMMARY AND CONCLUSIONS Variations in milk composition arise from differences in relative rates of synthesis ant] secretion of milk components by the mam- mary gland. The processes involved for lactose, protein, and fat synthesis and se- cretion are independent but regulated through nutrient or substrate availability ant! hormonal control of nutrient utilization. Thus, genetics, which mediates hormonal 237 effects, and diet, which regulates nutrient availability, are the major factors affecting milk composition. The most variable milk constituent is fat. Consiclerable variation exists between and within dairy cattle breeds. Genetic selection for fat percentage can change fat content of -milk but will also affect other constituents since there is a high correlation between the percentage of components in milk. Ge- netic selection for fat content would! alter the quantity of fat produced but not the composition of the fat. The best hope for altering composition is through diet. Changes in fat percentage and composition can be accomplished by altering the flier to produce changes in fermentation patterns or the composition of fat absorbe(1 from the diges- tive tract. Diets that increase the proportion of propionate in the rumen depress milk fat percentage, but changes in fat composition are minimal, including slight increases in Cog polyunsaturated fatty acids and slight decreases in C~60 and C~80 fatty acids. In- clusion of fats in the diet, particularly ru- men-protected fats, is the most effective way to alter milk fat composition. Significant increases in long-chain fatty acids can be achieved by inclucling-fat containing these acids in the diet. However, the amount and composition of fats in the diet Heel] to be controlled to avoid impairment of digestion of other (lietary constituents in the rumen. Unsaturated fatty acids are hydrogenated extensively in the rumen. Milk protein percentage and composition can be manipulates] through genetic selec- tion. Variations in the casein, beta-lacto- globulin, and alpha-lactalbumin fractions are known to exist. Heritability estimates of protein percentage range from 0.3 to 0.7. Increasing milk protein percentage through genetic selection is feasible; however, in- creasing milk protein yield! through selec- tion is more desirable. The percentage of true proteins in milk cannot be manipulated through feecling. Total protein percentage in milk can be lowered by including fats in
238 the diet or raised in relation to milk fat percentage by feeding high-concentrate diets. Dietary protein percentage has a minimal eject on milk protein percentage when it is within practical feeding ranges. Levels of other nutritional components of milk lactose, vitamins, and minerals are rather constant and not subject to large changes through genetic or nutritional ma- nipulation. The manipulation of milk components through changes in dairy management prac- tices, breeding, feecling, health, environ- ment, and general management appears to be rather limited. Milk fat percentage and composition can be changed through feed- ing, whereas milk protein percentage is best changed through genetics. Any changes will be slow in coming and minor compared to those achieved through processing and man- ufacturing. The goal of milk producers should be to modify composition as much as pos- sible to meet market demand but to em- phasize maximum yield of components in high-quality, wholesome milk. REFERENCES Allen, D. B., E. J. DePeters, and R. C. Laben. 1986. Three times a day milking: Effects on milk produc- tion, reproductive efficiency and udder health. J. Dairy Sci. 69:1441. Amos, H. E., T. Kiser, and M. Loewenstein. 1985. Influence of milking frequency on reproductive and productive efficiencies of cows. J. Dairy Sci. 68:732. ~ J Banks, W., J. L. Clapperton, and W. Steele. 1983. Dietary manipulation of the content and fatty acid composition of milk fat. Proc. Nutr. Soc. 42:399. Bauman, D. E., and J. M. Elliot. 1983. Control of nutrient partitioning in lactating ruminants. Ch. 14 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Bauman, D. E., P. J. Eppard, M. J. DeGeeter, and G. M. Lanza. 1985. Responses of high-producing dairy cows to long term treatment with pituitary somatotropin and recombinant somatotropin. J. Dairy Sci. 68:1352. Baumrucker, C. R. 1985. Amino acid transport systems in bovine mammary tissue. J. Dairy Sci. 68:2436. Bragg, D. St. A., M. R. Murphy, and C. L. Davis. 1986. Effect of source of carbohydrate and frequency of feeding on rumen parameters in dairy steers. J. Dairy Sci. 69:392. APPENDIX Casper, D. P., and D. J. Schingoethe. 1986. Evaluation of urea and dried whey in diets of cows during early lactation. J. Dairy Sci. 69:1346. Cerbulis, J., and H. M. Farrell, Jr. 1976. Composition of the milk of dairy cattle. II. Ash, calcium, mag- nesium and phosphorus. J. Dairy Sci. 59:589. Chalupa, W., and P. L. Schneider. 1985. Buffers for dairy cattle. 20th Annual Proceedings of the 1985 Pacific Northwest Animal Nutrition Conference, sponsored by the Pacific Northwest Feed Manufac- turers. October 1985, Boise, Idaho. Christie, W. W. 1979. The effects of diet and other factors on the lipid composition of ruminant tissues and milk. Prog. Lipid Res. 17:245. Clark, J. H. 1975. Lactational responses to post-ruminal administration of proteins and amino acids. J. Dairy Sci. 58:1178. Clark, J. H., H. R. Spires, R. G. Derrig, and M. R. Bennik. 1977. Milk production, nitrogen utilization and glucose synthesis in lactating cows infused post- ruminally with sodium caseinate and glucose. J. Nutr. 107:631. Clark, J. H., H. R. Spires, and C. L. Davis. 1978. Uptake and metabolism of nitrogenous compounds by the lactating mammary gland. Fed. Proc. 37:1233. Coppock, C. E. 1985. Energy nutrition and metabolism of the lactating cow. J. Dairy Sci. 68:3403. Cragle, R. G., M. R. Murphy, S. W. Williams, and J. H. Clark. 1986. Effects of altering milk production and composition by feeding on multiple component milk pricing system. J. Dairy Sci. 69:282. Crawford, R. J., Jr., and W. H. Hoover. 1984. Effects of particle size and formaldehyde treatment of soy- bean meal on milk production and composition for dairy cows. J. Dairy Sci. 67:1945. Crooker, B. A., J. H. Clark, and R. D. Shanks. 1983. Effects of formaldehyde treated soybean meal on milk yield, milk composition and nutrient digesti- bility in the dairy cow. J. Dairy Sci. 66:492. Davies, D. T., C. Holt, and W. W. Christie. 1983. The composition of milk. Ch. 5 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Davis, C. L. 1978. The use of buffers in the rations of lactating dairy cows. In Proceedings of the Begula- tion of Acid-Base Balance, sponsored by the Uni- versity of Arizona and Church and Dwight Co., Inc., W. lI. Hale and P. Meinhardt, eds. Tuscon: Uni- versity of Arizona. DePeters, E. J., and S. J. Taylor. 1985. Effects of feeding corn or barley on composition of milk and diet digestibility. J. Dairy Sci. 68:2027. DePeters, E. J., N. E. Smith, and J. Acedo-Rico. 1985. Three or two times daily milking of older cows and first lactation cows for entire lactations. J. Dairy Sci. 68:123. Dils, R. R. 1983. Milk fat synthesis. Ch. 5 in Bio- chemistry of Lactation, T. B. Mepham, ed. Amster- dam: Elsevier.
COMPOSITION OF MILK FROM DAIRY COWS Dils, R. R. 1986. Comparative aspects of milk fat ~^~ synthesis. J. Dairy Sci. 69:904. Dunkley, W. L., N. E. Smith, and A. A. Franke. 1977. Effects of feeding protected tallow on com- position of milk and milk fat. J. Dairy Sci. 60:1863. Eigel, W. N., J. E. Butler, C. A. Ernstrom, H. M. Farrell, Jr., V. R. Harwalker, R. Jenness, and R. McL. Whitney. 1984. Nomenclature of proteins of cow's milk: Fifth revision. J. Dairy Sci. 67:1599. Emery, R. S. 1978. Feeding for increased milk protein. J. Dairy Sci. 61:825. Eppard, P. J., D. E. Bauman, J. Bitman, D. L. Wood, R. M. Akers, and W. A. House. 1985. Effect of dose of bovine growth hormone on milk composition: Alpha-lactalbumin, fatty acids, and mineral ele- ments. J. Dairy Sci. 68:3047. Faulkner, A., and M. Peaker. 1982. Secretion of citrate into milk. J. Dairy Res. 49:159. Fernando, R. S., S. L. Spahr, and E. H. faster. 1985. Comparison of electrical conductivity of milk with other indirect methods for detection of subclinical mastitis. J. Dairy Sci. 68:449. Fettman, M. J., L. E. Chase, J. Bentinck-Smith, C. E. Coppock, and S. A. Zinn. 1984. Nutritional chloride deficiency in early lactation Holstein cows. J. Dairy Sci. 67:2321. Fogerty, A. C., and A. R. Johnson. 1980. Influence of nutritional factors on the yield and content of milk fat: Protected polyunsaturated fat in the diet. Int. Dairy Fed. Bull. Doc. 125:96. Forar, F. L., R. L. Kincaid, R. L. Preston, and J. K. Hillers. 1982. Variation of inorganic phosphate in blood plasma and milk lactating cows. J. Dairy Sci. 65:760. Forester, R. J., D. G. Grieve, J. G. Buchanan-Smith, and G. K. MacLeod. 1983. Effect of dietary protein degradability on cows in early lactation. J. Dairy Sci. 66:1653. Franke, A. A., J. C. Bruhn, and R. B. Osland. 1983. Factors affecting iodine concentration of milk of individual cows. J. Dairy Sci. 66:997. Gaunt, S. N. 1973. Genetic and environmental changes in milk consumption. J. Dairy Sci. 56:270. Gaunt, S. N. 1980. Genetic variation in the yields and contents of milk constituents. Int. Dairy Fed. Bull. Doc. 125:73. Gisi, D. D., E. J. DePeters, and C. L. Pelissier. 1986. Three times daily milking of cows in California dairy herds. J. Dairy Sci. 69:863. Grappin, R., V. S. Packard, and R. E. Ginn. 1981. Variability and interrelationship of various herd milk components. J. Food Prot. 44:69. Hansen, W. P., D. E. Otterby, J. D. Donker, R. G. Lundquist, and J. G. Linn. 1984. Influence of grain concentrations, forage type, and methionine hydroxy analog on lactational performance of dairy cattle. J. Dairy Sci. 67(Suppl. 1):99 (Abstr. ). Henderson, S. J., H. E. Amos, and J. J. Evans. 1985. 239 Influence of dietary crude protein concentration and degradability on milk production, composition, and ruminal protein metabolism. J. Dairy Sci. 68:2227. Holter, J. B., W. E. Urban, Jr., H. H. Hayes, and H. A. Davis. 1977. Utilization of diet components fed blended or separately to lactating cows. J. Dairy Sci. 60:1288. Holter, J. B., W. E. Hylton, and C. K. Bozak. 1985. Varying protein content and nitrogen solubility for pluriparious, lactating Holstein cows: Lactation per- formance and profitability. J. Dairy Sci. 68:1984. Iyengar, G. V. 1982. Elemental Composition of Human and Animal Milk: A Review. International Atomic Energy Agency Technical Document 269. Vienna: International Atomic Energy Agency. Jenkins, T. C., and D. L. Palmquist. 1984. Effect of fatty acids of calcium soaps on rumen and total nutrient digestibility of dairy rations. J. Dairy Sci. 67:978. Jenness, R. 1985. Biochemical and nutritional aspects of milk and colostrum. Ch. 5 in Lactation, B. L. Larson, ed. Ames: Iowa State University Press. Kaufman, W. 1980. Protein degradation and synthesis within the reticulorumen in relation to milk protein synthesis. Intl. Dairy Fed. Bull. Doc. 125:152. Kawas, J. R., N. A. Jorgensen, A. R. Hardie, and J. L. Danelon. 1983. Change in feeding value of alfalfa hay with stage of maturity and concentrate level. J. Dairy Sci. 66:181 (Abstr. ). Keown, J. F., R. W. Everett, N. B. Empet, and L. H. Wadell. 1986. Lactation curves. J. Dairy Sci. 69:769. Kitchen, B. J. 1981. Bovine mastitis: Milk composi- tional changes and related diagnostic tests. J. Dairy Res. 48:167. Kroeker, E. M., K. F. Ng-Kwai-Hang, J. F. Hayes, and J. E. Mosley. 1985. Effect of beta-lactoglobulin variant and environmental factors on variation in the detailed composition of bovine milk serum proteins. J. Dairy Sci. 68:1637. Kuhn, N. J. 1983. The biosynthesis of lactose. Ch. 6 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Kung, L., Jr., and J. T. Huber. 1983. Performance of high producing cows in early lactation fed protein of varying amounts, sources, and degradability. J. Dairy Sci. 66:227. Kuzdzal-Savole, S., W. Manson, and J. H. Moore. 1980. The constituents of cow's milk. Int. Dairy Fed. Bull. Doc. 125:4-13. Larson, B. L. 1979. Biosynthesis and secretion of milk protein: A review. J. Dairy Res. 46:161 Larson, B. L. 1985. Biosynthesis and cellular secretion of milk. Ch. 4 in Lactation, B. L. Larson, ed. Ames: Iowa State University Press. Larson, L. L., S. E. Wallen, F. G. Owen, and S. R. Lowry. 1983. Relation of age, season, production and health indices to iodine and beta-carotene con
240 centrations in cow's milk. J. Dairy Sci. 66:2257. Linn, J. G. 1983. The addition of fats to diets of lactating dairy cows: A review. In Proceedings of a Feed Fat Seminar, sponsored by Central Bi-Prod- ucts. Redwood Falls, Minn. Linn, J. G., and D. E. Otterby. 1984. Feeding strategies in dairy nutrition. Pp. 1~22 in Proceed- ings of the 45th Minnesota Nutrition Conference. St. Paul, Minn.: University of Minnesota Press. Lundquist, R. L., J. G. Linn, and D. E. Otterby. 1983. Influence of dietary energy and protein on yield and composition of milk from cows fed methi- onine hydroxy analog. J. Dairy Sci. 66:475 Lundquist, R. L., D. E. Otterby, and J. G. Linn. 1986. Influence of formaldehyde-treated soybean meal on milk production. J. Dairy Sci. 69:1337. Madsen, J. 1982. The effect of formaldehyde-treated protein and urea on milk yield and composition in dairy cows. Acta Agric. Scand. 32:389. Marshall, S. P., and A. R. Voigt. 1975. Complete rations for dairy cattle. I. Methods of preparation and roughage-to-concentrate ratios of blended ra- tions with corn silage. J. Dairy Sci. 58:891. Mepham, T. B. 1982. Amino acid utilization by lac- tating mammary gland. J. Dairy Sci. 65:287. Mercier, J.-C., and P. Gaye. 1983. Milk protein synthesis. Ch. 7 in Biochemistry of Lactation, T. B. Mepham, ed. Amsterdam: Elsevier. Mertens, D. R. 1985. Effect of fiber on feed quality for dairy cows. Pp. 209-224 in Proceedings of the 46th Minnesota Nutrition Conference. St. Paul, Minn.: University of Minnesota Press. Milam, K. Z., C. E. Coppock, J. W. West, J. K. Lanham, D. H. Nave, J. M. Labore, R. A. Stermer, and C. F. Brasington. 1986. Effects of drinking water temperature on production responses in lactating Holstein cows in summer. J. Dairy Sci. 69:1013. Needs, E. C., and M. Anderson. 1984. Lipid com- position of milk from cows with experimentally induced mastitis. J. Dairy Res. 51:239. Ng-Kwai-Hang, K. F., J. F. Hayes, J. E. Moxley, and H. G. Monardes. 1982. Environmental influences on protein content and composition of bovine milk. J. Dairy Sci. 65:1993. Ng-Kwai-Hang, K. F., J. F. Hayes, J. E. Moxley, and H. G. Monardes. 1985. Percentages of protein and nonprotein nitrogen with varying fat and somatic cells in bovine milk. J. Dairy Sci. 68:1257. Oldham, J. D. 1984. Amino acid metabolism in rum- inants. Pp. 137-151 in Proceedings of the Cornell Nutrition Conference, sponsored by Cornell Uni- versity and American Feed Manufacturers Associa- tion. Ithaca, N.Y.: Cornell University. Oltner, R., M. Emanuelson, and H. Wiktorsson. 1985. Urea concentrations in milk in relation to milk yield, live weight, lactation number and amount and com- position of feed given to dairy cows. Livestock Prod. Sci. 12:47. APPENDIX Owen, J. B. 1981. Complete-diet feeding of dairy cows. In Recent Development in Ruminant Nutri- tion, W. Haresign and D. J. A. Cole, eds. London: Butterworth. Palmquist, D. L., and T. C. Jenkins. 1980. Fat in lactation rations: Review. J. Dairy Sci. 63:1 Peaker, M., and A. Faulkner. 1983. Soluble milk constituents. Proc. Nutr. Soc. 42:419. Peel, C. J., L. D. Sandles, K. J. Quelch, and A. C. Herington. 1985. The effects of long-term adminis- tration of bovine growth hormone on the lactational performance of identical-twin dairy cows. Anim. Prod. 41:135. Poutrel, B., J. P. Caffin, and P. Rainard. 1983. Phys- iological and pathological factors influencing bovine serum albumin content in milk. J. Dairy Sci. 66:535. Rogers, G. L., and J. A. Stewart. 1982. The effects of some nutritional and nonnutritional factors on milk protein concentration and yield. Aust. J. Dairy Technol. 37:26. Rolleri, G. D., B. L. Larson, and R. W. Touchberry. 1956. Protein production in the bovine. Breed and individual variations in the specific protein constit- uents of milk. J. Dairy Sci. 39:1683. Rook, J. A. F., and P. C. Thomas. 1980. Principles involved in manipulating the yields and concentra- tions of constituents in milk. Int. Dairy Fed. Bull. Doc. 125:66. Schingoethe, D. J. 1976. Whey utilization in animal feeding: A summary and evaluation. J. Dairy Sci. 59:556. Schneider, P. L., D. K. Beede, and C. J. Wilcox. 1986. Responses of lactating cows to dietary sodium source and quantity and potassium quantity during heat stress. J. Dairy Sci. 69:99 Schultz, L. H. 1977. Somatic cell in milk physiolog- ical aspects and relationship to amount and compo- sition of milk. J. Food Prot. 40:125. Schwab, C. G., L. D. Satter, and A. B. Clay. 1976. Response of lactating cows to abomasal infusions of amino acids. J. Dairy Sci. 59:1254. Shaver, R. D., A. J. Nytes, L. D. Satter, and N. A. Jorgensen. 1986. Influence of amount of feed intake and forage physical form on digestion and passage of prebloom alfalfa hay in dairy cows. J. Dairy Sci. 69:1545. Storry, J. E. 1980. Influence of nutritional factors on the yield and content of milk: Nonprotected fat in the diet. Int. Dairy Fed. Bull. Doc. 125:88. Storry, J. E., and P. E. Brumby. 1980. Influence of nutritional factors on yield and content of milk: Protected nonpolyunsaturated fat in the diet. Int. Dairy Fed. Bull. Doc. 125:105. Sutton, J.D. 1980. Influence of nutritional factors on the yield and content of milk fat: Dietary components other than fat. Int. Dairy Fed. Bull. Doc. 125:126. Sutton J. D. 1985. Digestion and absorption of energy substrates in the lactating cow. J. Dairy Sci. 68:3376.
COMPOSITION OF MILK FROM DAIRY COWS Tallamy, P. T., and H. E. Randolph. 1970. Influence of mastitis on properties of milk. V. Total and free concentrations of major minerals in skim milk. J. Dairy Sci. 53:1386. Thomas, P. C. 1980. Influence of nutrition on the yield and content of protein in milk: Dietary protein and energy supply. Int. Dairy Fed. Bull. Doe. 125:142. Thomas, P. C. 1983. Milk protein. Proc. Nutr. Soc. 42:407. Thomas, P. C., and D. G. Chamberlain. 1984. Ma- nipulation of milk composition to meet market needs. Ch. 14 in Recent Advances in Animal Nutrition, W. Haresign and D. J. A. Cole, eds. London: Butter- worth. Tucker, H. A. 1985. Endocrine and neural control of the mammary gland. Ch. 2 in Lactation, B. L. Larson, ed. Ames: Iowa State University Press. Van Vleck, L. D. 1978. Breeding for increased milk protein. J. Dairy Sci. 61:815. Wheelock, J. V. 1980. Influence of physiological factors 241 on the yields and contents of milk constituents. Int. Dairy Fed. Bull. Doc. 125:83. Wilcox, C. J. 1978. Genetic considerations of economic importance: Milk yield, composition and quality. Ch. 2 in Large Dairy Herd Management, C. J. Wilcox, H. H. Van Horn, B. Harris, Jr., H. H. Head, S. P. Marshall, W. W. Thatcher, D. W. Webb, and J. M. Wing, eds. Gainesville: University Presses of Florida. WolEschoon-Pombo, A., and H. Klostermeyer. 1981. The NPN-fraction of cow milk. I. Amount and composition. Milchwissenschaft 36:598. Woodford, J. A., N. A. Jorgensen, and G. P. Barring- ton. 1986. Impact of dietary fiber and physical form on performance of lactating dairy cows. J. Dairy Sci. 69:1035. Young, C. W., J. K. Millers, and A. E. Freeman. 1986. Production, consumption, and pricing of milk and its components. J. Dairy Sci. 69:272.