4
Advances in Risk Assessment Since the Establishment of the Part 503 Rule
The committee’s review of the risk assessment used to support the Part 503 rule was carried out in the context of current and emerging practice in risk assessment. The committee determined that its review of the risk assessment should communicate the committee’s interpretation of how the risk-assessment process has evolved from the time the Part 503 rule was issued until present. Of particular interest to the committee were documents from EPA and the National Research Council (NRC) that propose and encourage methods that differ substantially from the methods used in the Part 503 risk assessment. This chapter provides a foundation and context for the following chapters.
This chapter first describes new approaches and considerations in risk assessment since the Part 503 rule (Standards for Use or Disposal of Sewage Sludge) was established in 1993 (40 CFR Part 503). It focuses on the changing priorities of cancer versus noncancer end points, acute versus chronic end points, probabilistic risk-assessment approaches, and the need to address aggregate exposures and cumulative risk. A brief description is then given of the changes in risk-assessment approaches of EPA over this period.
THE RISK-ASSESSMENT PROCESS
Risk assessment is a process for identifying potential adverse consequences along with their severity and likelihood. In contrast to other tools
used for environmental evaluation and policy, the principal objective of the risk assessment and risk management approach is not to eliminate all risk but to quantify the risk and provide risk managers with tools to balance the level of risk against the cost of risk reduction, against competing risks, or against risks that are generally accepted as trivial or acceptable. Controlling the exposure of human populations to environmental contaminants in biosolids using a risk-based approach requires a definition of both an appropriate metric for assessing the impacts of contaminants on human health and a defensible process for assigning value to the predicted impacts. The end product of a risk-based approach to environmental management is either to identify an acceptable level of exposure or to prescribe the technical controls or political process needed to attain acceptable risk. Intervention can be achieved through technical or political controls.
Components of the Risk-Analysis Process
The NRC (1982, 1994) has divided and continues to divide the practice of risk analysis into two substantially different processes—risk assessment and risk management. Along with these processes are concurrent efforts to communicate and evaluate risk (NRC 1989, 1996). This section explores the evolution of the risk-assessment process over the last decade by considering the component steps in the process.
Risk assessment is the process of selecting and quantifying the adverse consequences that result from an action, such as application of biosolids to soils, or from inaction. A risk assessment begins with efforts to identify the potential hazards associated with a chemical or microbial agent and its use or occurrence. Hazard identification addresses the potential for harm but not the likelihood of harm. Risk characterization establishes the significance of an identified hazard by quantifying the likelihood and severity of exposure scenarios linked to that hazard. As applied to toxic agents, risk characterization has five principal elements: (1) quantification of sources and environmental concentrations in exposure media; (2) quantification of exposure to the target population and distribution of the dose among the population; (3) characterization of a dose-response function for all potential toxic agents that have been identified; (4) estimates of the number of people affected and severity of consequences expected within the population at risk; and (5) an assessment of the magnitude and sources of uncertainty that limit the precision of the estimate of consequences.
Risk management is the process of weighing policy alternatives and selecting the appropriate societal or institutional response. Risk management is
used to integrate the results of a risk characterization with social, economic, and political valuation to reach a decision. The goal of the risk-management process is to establish the significance of the estimated risk, compare the costs of reducing this risk with the benefits gained, compare the estimated risks with the societal benefits derived from incurring the risk, and carry out the political and institutional process of reducing risk.
Linking the risk-assessment and risk-management processes are the concurrent efforts to evaluate and communicate risk. Risk evaluation is the process by which the risk-characterization and risk-management processes are reconciled with individual and societal valuations of risk (NRC 1996). A key step in this link is effective risk communication. According to the NRC (1989), risk communication has become more difficult in recent decades and common misconceptions often hamper communication efforts. In considering these issues, the NRC (1989) emphasizes that solving the problems of risk communication is as much about improving procedures as improving the content of risk messages.
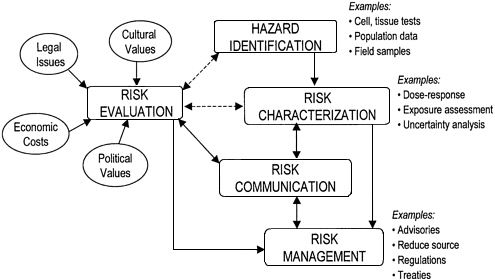
Figure 4–1 provides a view of how the risk-analysis process might proceed for assessing the health impacts of pollutants in biosolids. Each of the major steps in this process involves one or more actions that are listed to the right of each major step.
Confronting Uncertainty and Variability
An important and often ignored final step in the risk characterization process is the characterization of uncertainties. Important sources of uncertainty and variability in risk assessments involve the data and models used. With incomplete data and models used to characterize contaminant transport representing heterogeneous geographic and climate regions, the variability and uncertainty associated with the resulting risk estimates are large.
In evaluations of uncertainty in risk assessment, Morgan et al. (1990) and Finkel (1990) distinguish among parameter uncertainty, model uncertainty, decision-rule uncertainty, and natural variability in any of the parameters and call for separate treatment of the different types of uncertainty. Probabilistic methods such as Monte Carlo analysis are available to evaluate uncertainty in parameters. According to Finkel (1990), model uncertainty derives from a number of actions, including the use of simplifications that might exclude relevant variables from the analysis; the use of surrogate variables that might not be appropriate for the variable of interest; the appearance of abnormal conditions that might occur in nature but that might not be appropriate in the model; and the use of incorrect model forms. Morgan et al. (1990) noted that
relatively little research has been done on uncertainty or disagreement about what form of model to use. Decision-rule uncertainty applies to risk management and arises whenever ambiguity or controversy exists about quantifying or comparing social objectives. According to Finkel (1990, p. 16), “to take any actions using the outputs of a risk assessment, including the decision not to take action, one must be prepared to make a series of potentially controversial value judgments.”
An important source of uncertainty in risk characterization is the development and application of dose-response models. Among the many issues that complicate the process of establishing a dose-response function is the variation in human susceptibility. In large heterogeneous populations, there are large variations in susceptibility to toxic effects. Those variations are due in part to variations in genetic predisposition to certain disease states, variations in age, and large variations in physical stresses and other chemical or non-chemical exposures that might be extant in the system of interest.
NEW APPROACHES AND CONSIDERATIONS IN RISK ASSESSMENT
This section reviews new approaches to risk assessment that were developed since the Part 503 rule was issued. A summary of key documents from the NRC, the Presidential/Congressional Commission on Risk Assessment and Risk Management, and EPA are provided. Then, consideration is given to how those documents have altered the standard practice in each of the key steps of the risk-assessment process.
Recent Reports Define New Directions in Risk Assessment
Among the reports that have had particular impact are two reports issued by the NRC. The first report, titled Science and Judgment in Risk Assessment, provided an update on the process of risk assessment and management (NRC 1994). This report made seventy-five specific recommendations, but among its overarching recommendations are those to address explicitly uncertainty and variability in risk assessment, to address multimedia exposures and cumulative intake through multiple exposure pathways, to and foster more interaction among risk assessors and risk managers. The second report, titled Understanding Risk, Informing Decisions in a Democratic Society (NRC 1996), used several case studies to evaluate the emerging trends in risk-assessment methodology.
The Presidential/Congressional Commission on Risk Assessment and Risk Management was created through the 1990 Clean Air Act amendments to make recommendations for improving the risk-assessment and risk-management process. In 1997, the commission issued Framework for Environmental Health Risk Management. The report emphasizes how to present a risk assessment and how to work with community concerns in an iterative fashion. It identifies a clear need to modify the traditional approaches used to assess and reduce risks. Traditional approaches rely on a chemical-by-chemical, medium-by-medium, risk-by-risk strategy. The report states the need to focus less attention on refining assumption-laden mathematical estimates of the small risks associated with exposures to specific chemicals and the need to focus instead on the overall goal of reducing risk and improving health status. There is strong emphasis on stakeholder participation. Stakeholders are groups who are potentially affected by the risk, groups who will manage the risk, and groups who will be affected by efforts to manage the source of the risk. Involving stakeholders throughout the risk-assessment process provides opportunities to gather information and to bridge gaps in understanding, language, values, and perspectives.
Over the last decade, EPA issued a number of reports that are having an impact on the framework and process of regulatory risk assessment. Of particular note are the 1992 Habicht memo, which provides guidance to EPA managers on risk characterization (Habicht 1992); a journal report on benchmark dose (Barnes et al. 1995), which provides guidance for a more harmonized approach for addressing cancer and noncancer health end points; and the proposed guidelines for carcinogen risk assessment (EPA 1996a). The Habicht memo emphasizes the need to avoid point estimates of risk and to provide instead details on the scientific basis of decisions, including clear statement of assumptions and uncertainties. Barnes et al. (1995) recommend the use of the benchmark-dose approach as an alternative to using the no-observed-adverse-effect level. EPA’s proposed guidelines for carcinogen risk assessment put more emphasis on “margin of exposure” (relative to a benchmark dose), weight of evidence, and the use of uncertainty factors in the risk characterization process. Also of note is EPA’s (1997a) Exposure Factors Handbook, which provides a large compendium of information on human activities that relate to exposure—including time-activity data, exposure duration, consumption of homegrown food, and water ingestion.
In addition, there is an ongoing effort to address aggregate exposures to the same substances from multiple sources and pathways and cumulative exposures and risk from mixtures. The 1996 Food Quality Protection Act (FQPA) explicitly calls for addressing aggregate exposure and cumulative risk in setting standards for pesticide residues in food.
From a risk assessment perspective, this report will clearly establish that biosolids are a complex mixture of chemical and biological agents, the exact composition of which can change from time to time and place to place. Moreover, it will never be possible to account for all the components of the mixture, although the stable components are well characterized. As discussed in detail in various sections of this report, considerable effort has been devoted to an enumeration of the hazardous constituents of biosolids. During the course of its study, the committee found that it remains necessary to conduct risk assessments on biosolids based on their component parts.
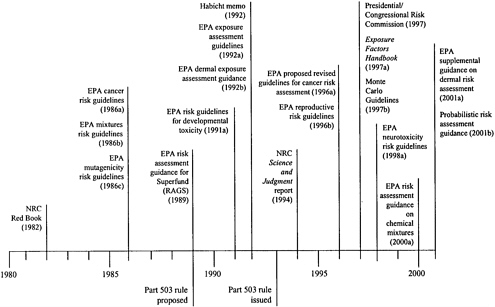
Figure 4–2 provides a time line showing when a number of significant risk-guidance documents have been issued relative to the year when the Part 503 rule was issued.
Advances in Hazard Identification
Since EPA issued cancer and mutagenicity risk-assessment guidelines in 1986 (EPA 1986a,c), the types and reliability of methods used to identify potential hazard have advanced. In the 1986 guidelines, the stated goal of a hazard assessment was to provide a review of the relevant biological and chemical information on an agent that might pose cancer or other health hazards. At that time, the recommended elements of the hazard identification included (1) a summary of an agent’s physical-chemical properties and routes and patterns of exposure; and (2) a review of toxic effects, structure-activity indicators of toxicity, metabolic and pharmacokinetic properties, short-term animal and cell tests, long-term animal tests, and human studies. These elements have remained the core components of hazard identification, but the arsenal of methods, the reliability of techniques, and the relative emphasis on the various hazard identification elements have changed over the past decade. In particular, risk assessors can now make use of better markers of genetic damage (toxicogenomics) for rapid assessment, improved structure-activity relationships (SAR), and improved quantitative structure-activity relationships (QSAR). However, to date, these emerging methods have seen only limited use in regulatory risk assessment. Health-effects research has focused more on early indicators of outcome, making it possible to shorten the time between exposure and observation of an effect Use of measures of exposure as hazard indicators (e.g., Hertwich et al. 2001) has increased, and more-sophisticated measures of hazard such as the human toxicity potential have been developed. Human toxicity potential includes emissions, exposure potential, and toxic hazard indicators in a single measure of potential harm. It has been used as a cumulative-exposure screening tool for multiple chemical agents.
Public-health and environmental concerns about biosolids foster a need for hazard assessments that can address multiple and complex issues. Among these issues are health hazards from chemical mixtures and pathogens, as well as concerns about specific categories of chemical hazard, such as metals, persistent organic pollutants (POPs), and high-production-volume chemicals (HPVs). Recent advances in hazard assessment provide EPA with better tools for those issues. Community issues are not adequately addressed in the current risk-assessment paradigm (e.g., property intrusions, odor, and truck traffic). Other issues have been addressed in EPA programs but have not been explicitly addressed in the risk-management goals of the biosolids program. Those include potential health effects from added diesel exhaust and potential environmental effects from added nitrogen burdens, runoff, damage to endangered species habitat, and conversion of inorganic mercury to organic mercury in situ and in water bodies following runoff.
Advances in the Dose-Response Characterization Process
A number of important changes have been proposed and, in some cases, applied to dose-response characterization over the last decade. In 1993, the NRC considered the scientific basis, inference assumptions, regulatory uses, and research needs in risk assessment and focused on two dose-response issues—the use of maximum tolerated dose in animal bioassays and the use of two-stage models of carcinogenesis (NRC 1993). The report presented options for revising those default procedures. Recent EPA documents (EPA 1996a, 2001a) proposed that dose-response characterization be handled differently from that proposed in the 1986 risk-assessment guidelines (EPA 1986a). According to the 1986 guidelines, risk for carcinogens is modeled using potency—the increase of risk per unit increase of dose or exposure. Risk for noncarcinogens is addressed using a hazard index—the ratio of the predicted dose to the reference dose. More recently, efforts have been made to harmonize those two approaches by using a margin of exposure (MOE) to characterize risk for both carcinogens and noncarcinogens. MOE is the ratio of a dose derived from a tumor bioassay, epidemiologic study, or biologic marker study to an actual or projected human exposure.
Changes in Dose-Response Methods
Several proposals within and outside EPA have been made to modify the standard approach for building dose-response models on the basis of animal or human data. The most important and comprehensive proposal is EPA’s
1996 proposed revisions to its carcinogen risk-assessment guidelines (EPA 1996a). These guidelines, which are still undergoing review and revision within EPA, propose a different weight-of-evidence classification and the option of using an MOE in place of potency to estimate risk. Risk-assessment literature has provided proposals for the use of time-to-tumor models (Krewski et al. 1983), Bayesian methods for constructing and revising dose-response models (Taylor et al. 1993; Evans et al. 1994; Wilson 2001), and meta-analysis.
EPA’s Proposed 1996 Carcinogen Risk-Assessment Guidelines
In 1996, EPA issued its proposed Guidelines for Carcinogen Risk Assessment (EPA 1996a) for a 120-day public review and comment period. EPA issued the guidelines as a replacement for the 1986 Guidelines for Carcinogen Risk Assessment (EPA 1986a). The revised guidelines were issued in part to address changes in the understanding of the variety of ways in which carcinogens can operate. For example, because many laboratories now use test protocols aimed at mode of action, the 1996 proposed guidelines provide a framework that allows for incorporation of all relevant biological information and flexibility to consider future scientific advances.
In contrast to the single default dose-response relationship (the linearized multistage model for extrapolating risk from upper-bound confidence intervals) used in the 1986 cancer guidelines, the 1996 guidelines provide several options for constructing the dose-response relationship. Biologically based extrapolation, that is, extrapolation from animals to humans based on a similar underlying mechanism of action, is the preferred approach for quantifying risk. However, because data for the parameters used in such models are not likely to be available for most chemicals, the 1996 guidelines allow for alternative quantitative methods, including several default approaches. In the default approaches, dose-response assessment is a two-step process. In the first step, response data are modeled in the range of observation; in the second step, a determination is made of the point of departure (benchmark) or the range of extrapolation below the range of observation. In addition to modeling tumor data, the new guidelines call for the use and modeling of other kinds of responses if they are considered measures of carcinogenic risk. Three default approaches—linear, nonlinear, or both—are provided. Curve fitting in the observed range provides the effective dose corresponding to the lower 95% limit on a dose associated with a 10% response (LED10). The LED10 is then used as a point of departure for extrapolation to the origin as the linear default or for an MOE as the nonlinear default The LED10 is the standard point of departure, but other departure points can be used when the data justify it.
Other modifications of interest in the 1996 guidelines include the following:
-
Emphasis is placed on all biological information rather than only tumor findings in the hazard-assessment phase of risk assessment.
-
Mode of action is emphasized to reduce the uncertainty in describing the likelihood of harm and in determining the dose-response approaches.
-
A weight-of-evidence narrative replaces the current alphanumeric classification categories (A, B1, B2, C, D, E) from the 1986 cancer guidelines. The narrative summarizes the key evidence, describes the agent’s mode of action, characterizes the conditions of hazard expression, and recommends appropriate dose-response approaches. The overall conclusion on the likelihood of human carcinogenicity is given by route of exposure. Only three descriptors for classifying human carcinogenic potential are now available—known/likely, cannot be determined, and not likely.
-
In contrast to the 1986 guidelines that provide very little guidance for risk characterization, the 1996 guidelines provide direction on how the overall conclusion and the confidence of risk are presented for the risk manager and call for assumptions and uncertainties to be clearly explained.
Time-to-Tumor Models
Because dose-response functions for many chemical substances are derived from lifetime animal-feeding studies, results apply to lifetime risk of cancer. The most common dose-response model derived from such toxicological experiments describes the lifetime change in cancer incidence with dose. However, the stage theory of cancer and other diseases emphasizes that many harmful exposures can be more accurately characterized as reducing the time to tumor induction rather than increasing the lifetime risk of tumor (Armitage and Doll 1954). In a time-to-tumor dose-response model, important information is disclosed by the time it takes for a fraction of the test subjects to get tumors (Krewski et al. 1983). Some animal bioassay data indicate when individual bioassay animals died before scheduled terminal sacrifice and whether they died with or without tumors. In some human populations, time to tumor or other disease is also available. Use of time-to-tumor data in the analysis of the tumor dose-response relationship provides a credible estimate of the potency of the carcinogen by incorporating considerable information. These models are not common but have much potential when data are substantial.
Use of Subjective Statistics: Bayesian Methods
Bayesian analysis is an important tool now widely used in many domains, including some parts of risk analysis (Taylor et al. 1993; Evans et al. 1994). It provides the foundation for the technical field of decision analysis. Bayesian approaches have begun to be applied to assessments of exposure for human health and environmental risks. In 2000, Resources for the Future (RFF) in conjunction with EPA and other organizations held a workshop to discuss ways in which Bayesian approaches could be useful in improving techniques for estimating exposure-response functions. Participants in the workshop agreed that wider use of Bayesian approaches can improve human health risk-assessment practices (Wilson 2001). The areas judged to have the most significant opportunities include estimating exposure-response functions; inferring causality, especially when interpreting results of epidemiological studies; and performing complex exposure assessments.
Use of Meta-Analysis in Place of Single-Species Data Sets
In the evaluation of chemical compounds for carcinogenic risk, regulatory agencies have traditionally fit a low-dose linear dose-response model to data from rodent bioassays. Recently, there is much interest in incorporating additional scientific information on the properties of the chemical under investigation into the risk-assessment process, including biological mechanisms of cancer induction. However, few attempts have been made to investigate the overall relationship between the shape of dose-response curves and mutagenicity.
Assessment of Mixtures
In 1986, EPA issued risk-assessment guidelines for chemical mixtures (EPA 1986b). This framework described three approaches to conduct a quantitative risk assessment for the potential health effects associated with exposure to chemical mixtures. First, when data are available on the health impacts of the mixture of concern or similar mixtures, these data should be used in formulating the risk models. When data are not available on the actual mixture or similar mixture of concern, data from risk assessments of individual components are then used to estimate the risk of the mixture of concern by applying a dose-additivity model (second approach) for systemic toxicants and
a response-additivity model (third approach) for carcinogens. Both of these models assume that no interaction occurs among chemicals. The two most accepted dose-additivity models are the hazard-index (HI) model and the toxicity-equivalency-factor (TEF) model. The response-additivity model is used primarily in cancer risk assessment of chemical mixtures; it is assumed that the components in the mixture act independently on the same target site but by different mechanisms of action, thus the toxicological responses to each component in the mixture are summed.
A significant advance in chemical-mixture risk assessment was the newly developed interaction-based method in which Mumtaz and Durkin (1992) used binary interaction data to modify the dose-additive HI. Recently, EPA (2000a) issued a revised guidance document for chemical mixtures as a supplement to the original guidelines of 1986. The document Supplementary Guidance for Conducting Health Risk Assessments for Chemical Mixtures provides details on the nature of mixtures and the procedures to use for data analyses. It also describes recent scientific advances in the area of chemical-mixture risk assessment, including methods for using whole-mixture data on a toxicologically similar mixture, methods for incorporating information on toxicological interactions into an HI (modified from the original method developed by Mumtaz and Durkin [1992]), procedures for including carcinogen interactions in mixture risk characterization, and generalized procedures for assessing mixtures of similar chemicals.
The incompleteness of the classic risk-assessment process as applied to biosolids can be illustrated by reference to the EPA guidance document (EPA 2000a), which details EPA’s current thinking on the mixture issue. A complex mixture is defined as “a mixture containing so many components that any estimation of its toxicity based on its components’ toxicities contains too much uncertainty and error to be useful. The chemical composition may vary over time or with different conditions under which the mixture is produced. Complex mixture components may be generated simultaneously as by-products from a single source or process, intentionally produced as a commercial product, or may coexist because of disposal practices. Risk assessments of complex mixtures are preferably based on toxicity and exposure data on the complex mixture” (EPA 2000a). Chapter 3 shows that health risk data on the complete mixture are insufficient in the case of biosolids to provide the basis for a risk assessment. Hence, assessors are dependent on a component-based assessment strategy that, while not containing “too much uncertainty and error to be useful,” will be incomplete as a basis for defining a strictly prospective strategy for risk management (EPA 2000a).
Advances in the Exposure Characterization Process
There have been a number of important changes in the exposure characterization process over the past decade. Among the changes of note are increasing focus on indoor and residential environments; methods for monitoring biological agents in exposure media (air, water, and soil); a movement away from simple bounding estimates to probabilistic assessments that include explicit treatment of uncertainty and variability; and the use of multimedia and multiple-pathway exposure assessments. In the sections below, the committee highlights the changes in exposure assessment methods that have particular relevance to biosolids risk assessments. A review and evaluation of specific exposure pathways in the Part 503 rule risk assessment are provided in Chapter 5.
Ten years ago it was common to conduct an exposure assessment using simple models that define a maximum exposed individual (MEI). The MEI was one who obtained all of his or her air, water, and/or food from an area contaminated by the pollutant of interest over a lifetime. The implicit and unquantified overestimate of exposure in the MEI as well as the failure of the MEI to capture all exposure pathways led to a search for alternative schemes. At first, there was an effort to define a highly exposed individual (HEI) as someone who had a plausibly high exposure but less exposure than the MEI. However, the HEI was found to have many of the same limitations as the MEI. Current practice is to use a reasonable maximum exposure (RME) receptor. EPA (1989) specifies that calculation of the RME requires a combination of average and upper-bound values for various exposure parameters, so that the final exposure estimate will represent an upper bound exposure that could reasonably be expected to occur. This is commonly interpreted to be a 90th to 95th percentile of exposures for each pathway. Due to its inconsistent combination of upper percentile and mean values, the RME approach can be arbitrary and fail to fully account for population exposure variability. Nevertheless, the use of RME in place of HEI has fostered the increasing use of probabilistic methods in exposure assessments (EPA 2001b). In its recent assessment of exposures to dioxins in biosolids, EPA partially makes use of a probabilistic risk-assessment approach (EPA 2001c).
Increased Focus on Indoor and Residential Environments
One theme that is clear in the literature on exposure assessment is the importance of the indoor environment and residential factors in understand-
ing human exposure to many agents. Indoor and residential scenarios received little attention in the Part 503 rule risk assessment, but those issues have received much greater attention in risk-assessment practice over the last decade.
Assessments of the human health impact of airborne pollutants revealed the importance of cumulative exposure to microenvironments, such as indoor air, and of household sources, such as consumer products, combustion, appliances, and tracked-in soil. Efforts to better understand urban air pollutants, such as particulate matter, revealed the importance of increased indoor concentrations of certain pollutants (Melia et al. 1978; Dockery and Spengler 1981; Spengler et al. 1983). Subsequent studies, most notably EPA’s Total Exposure Assessment Methodology (TEAM) studies, demonstrated that for a variety of contaminants, residential indoor air is often a more significant source of exposure than outdoor air (Pellizzari et al. 1986; Thomas et al. 1993; Wallace 1993).
Methods for Monitoring Biological Agents in Exposure Media
Although the issue of exposure to and risk from pathogens is addressed in Chapter 6, it is of note here that methods available for monitoring exposure to pathogens have improved greatly in the last decade. Traditional detection of microorganisms is performed using microscopy, culture, biochemistry, or immunoassay. Microscopy is used to detect total microbial populations in a given sample without regard to the physiological state of the organism; both viable and nonviable organisms can be detected. Culture-based assay is limited to detection of those organisms that will proliferate under the growth conditions of the analysis design. Biochemical and immunological-based analyses have improved the identification and enumeration of specific microbial contaminants in environmental samples. Improved detection and identification of microorganisms have been achieved using advanced biotechnology-based methodologies, including polymerase chain reaction (PCR) amplification, microchips, molecular beacons, electrochemiluminescence, biosensors, mass spectrometry, and flow cytometry.
Explicit Treatment of Uncertainty and Variability
Estimating potential human exposures and source-to-dose relationships for harmful substances in biosolids involves the use of models and large amounts of data. Because these data and models must be used to predict individual behaviors, engineered system performance, contaminant transport, human contact and uptake, and dose among large and often heterogeneous
populations, variability and uncertainty associated with these predictions are large.
Over the last decade, explicit assessment of sensitivity and uncertainty has become common practice in many risk assessments. This practice has been driven in large part by the ready availability of software for uncertainty and sensitivity analysis, improvements in computers that make it possible to run large numbers of repeated simulations, and the availability of Monte Carlo guidance from EPA (1997b). Also supporting this process is the wider availability of summary statistics for exposure factors, available in references such as the EPA (1997a) Exposure Factors Handbook.
One of the key issues in uncertainty analysis that has been addressed over the last decade is how to distinguish between the relative contribution of true uncertainty and that of interindividual variability (heterogeneity) to characterize the predicted population risk (Bogen and Spear 1987; NRC 1994). Uncertainty or model-specification error (e.g., statistical estimation error) can be modeled using a random variable, but the characteristics of this variable are often subjective. In contrast, variability refers to quantities that are distributed empirically within a defined population. Such factors as food ingestion rates, exposure duration, and expected lifetime are considered as variable but not uncertain. The recognition of the difference between uncertainty and variability has resulted in efforts to carry out assessments in which both uncertainty and variability are characterized in the final results.
The Habicht memo (1992) seems to have encouraged the growth in efforts to address uncertainty. The recent Exposure Factors Handbook (EPA 1997a), the Monte Carlo guidance document (EPA 1997b), and the recent report on policy for use of probabilistic risk assessment (EPA 1997c) reveal that EPA has and will continue to support and encourage more explicit treatment of uncertainty and variability. In its 1997 Monte Carlo guidelines and its Superfund guidance for conducting probabilistic risk assessment, EPA identified a tiered scheme for updating and calibrating a model as more data become available (EPA 1997b, 2001b). As a first step in this scheme, the variance of all input values should be clearly stated, and the impact of these variances on the final estimates of risk should be assessed using sensitivity analysis. Here, it helps to provide a clear summary and justification of the assumptions used for each aspect of a model. In addition, it should be stated whether these assumptions are likely to result in representative values or conservative (upper bound) estimates. The next step in this scheme is the use of variance propagation methods (including but not necessarily limited to Monte Carlo methods) to map how the overall precision of risk estimates is tied to the variability and uncertainty associated with model choice, inputs, and scenarios.
The risk assessment for the Part 503 rule does not provide a clear analysis of uncertainties and their potential impacts on the assessment of risks. A
quantitative analysis would allow identification of critical parameters that have a strong influence on the outcome of the calculations of risk. However, the limits of time and resources at EPA mean that choices must be made when planning whether and how to update risk assessments and collect site-specific data in support of the risk assessment calculations. In making revisions to the biosolids risk assessment, EPA must strike a balance between expending resources to carry out site-specific data collection and expending resources to model and assess risk using existing information.
Multimedia and Multiple-Pathway Exposure Assessments
Efforts to assess human exposure to contaminants from multiple environmental media have been evolving over the past several decades. Knowledge of potential environmental pathways is an important component of a health risk assessment for biosolids. The need to assess human exposure to global fallout in the 1950s resulted in the development of a framework that included transport of contaminants through air, soil, surface water, vegetation, and food chains. More recently, reported concentrations of semivolatile organic compounds and mercury species in water, vegetation, soil, and food products have increased interest in more accurate characterizations of chemical transport on a local, regional, and global scale. In response to the need for better characterization, a number of multimedia transport and transformation models for organic chemicals and metal species have appeared. Multimedia models are also being developed for pathogens. Over the past decade or so, relatively detailed single-domain transport and transformation models have been developed to model aspects of chemical transport and transformation within a single medium or domain (e.g., groundwater models, vadose zone models, surface-water mixing models, and air-dispersion and transformation models).
Multimedia, multipathway assessments have fostered increasing interest about indirect exposure pathways. But only limited efforts have been made to develop source-to-dose relationships using multimedia models. Moreover, these complex source-to-dose models are difficult to validate. The increasing sophistication of mass-transfer models has as yet had almost no impact on human exposure models. None of the exposure models available to date provides an integrated simulation of major transport processes and indoor and outdoor relationships for toxic substances in air, water, food, and soil.
The Food Quality Protection Act (FQPA) of 1996 draws attention to the need for methods to assess aggregate intake of agents with similar target organs.
Biological Markers
Outside of occupational settings or specific research studies, most current exposure-sampling strategies do not rely on biological markers. Although there are reasonable biomarker methods for several metals (e.g., mercury, arsenic, cadmium, chromium) and some organic compounds, the lack of reliable and nonintrusive biomarkers continues to limit their widespread use in exposure tracking studies. For example, the Centers for Disease Control and Prevention (CDC) is exploring biomarkers for classes of organophosphate (OP) pesticides. In some occupational settings, biological markers (e.g., for lead) are part of the surveillance process. It is feasible that a set of biomarkers could be created using less invasive methods (e.g., urine, saliva, and hair sampling). Urinary biomarkers have worked well for some metals, tobacco smoke, and some other pollutants. As new biomarkers are developed and existing ones improved, emerging sampling strategies will rely more on them. It is conceivable that in the future EPA will be able to evaluate more DNA adducts, possibly even after exposure of embedded personal DNA worn by individuals as a monitor. For many contaminants of concern in biosolids, biomarker approaches may be both feasible and informative. However, for the near future, it is not likely that biomarkers will be of great value for monitoring exposures near biosolids-application sites.
Challenges to the Risk-Characterization Process for Biosolids
The emphasis here is on how the process of risk characterization is changing and how those changes impact the Part 503 rule. Particular challenges to the risk-characterization process are to better link risk assessment to risk management, consider risk perception and risk valuation more explicitly, and provide better risk communication between risk assessors and affected populations.
To examine the Part 503 rule risk assessment in the context of the evolving risk-assessment paradigm, EPA must consider the objectives of the Part 503 rule risk assessment: Was it to convince the community that it is safe? Was it to justify what is being done or what has been decided? Was it to organize information on exposures and health effects to communicate what is known and what the information gaps and key uncertainties are?
One key risk characterization and management issue that emerged during the committee discussions was whether quality-of-life issues that have the potential to affect health, such as odors, should be considered a factor in
setting standards for land application of biosolids. In particular, could minimizing odors be an effective way to manage some potential risks?
Acceptance of a risk assessment by regulators and community groups often requires surveillance and monitoring to ensure that the assumptions used in the risk assessment are in place. Many of the chemical substances in municipal waste streams are also in biosolids. The chemicals in municipal solid-waste landfills are monitored. Should the same chemicals be monitored following biosolids application? Answers to these questions help to put the risk assessment in both a scientific and political context. That is, once the objectives of the risk assessment are established, what and whose decisions are being informed by the assessment and the level of scientific confidence needed can be identified.
Characterizing Exposures to Children as a Subpopulation
Organizations such as EPA and the National Institutes for Health are giving special consideration to children’s risks from exposure to environmental contaminants. In 1996, EPA’s Office of the Administrator issued Environmental Health Threats to Children (EPA 1996c) and set an agenda that called for consideration of children’s risks in all EPA actions. The report also emphasized the need for more research to support children’s risk assessments. Children are considered a special subpopulation because their health risks can differ from those of adults because of their immature physiology, metabolism, and differing levels of exposure due to factors such as greater food consumption per unit of body weight and outdoor play activities.
Differing levels of exposure for children are typically considered in risk assessments, but the underlying toxicity database often does not specifically address effects on children. Such limitations in toxicity data are typically addressed by application of uncertainty factors to protect susceptible populations, such as children. Additional research would allow an assessment of the adequacy of such uncertainty factors.
Participation of the Affected Populations
Local opposition to land application of biosolids appears to be growing, in part because regulators, such as EPA, have failed to systematically address concerns and experiences of residents near land-application sites. Because no process is in place to register complaints, EPA might be unaware of complaints lodged with a local or state agency. Public meetings held by the com-
mittee have identified residents near land-application sites and biosolids appliers who believe that they have suffered health impacts and believe that they have been excluded from having input in the risk-assessment process. Health complaints include irritation of the eyes, nose, and throat; headaches; nausea; cough; chest tightness; congestion; shortness of breath; drowsiness; skin lesions; and mood disorders (Schiffman et al. 2000; Shields1). The committee was not charged with the task of evaluating the legitimacy of the complaints, nor of determining whether application of sewage biosolids is related to the complaints. However, it notes that the primary concerns of neighbors to land-application sites and the alleged health impacts associated with land application of biosolids have not been addressed in the risk assessments upon which the Part 503 rule is based.
A critical aspect of the risk-assessment process is ensuring that those assessing risks are asking the right questions. Potentially affected people often have knowledge to contribute to the accurate characterization of exposures and to the assessment of risks. When such knowledge is not tapped, the outcome of the process can be flawed, rejected by stakeholders, or both. Tapping local knowledge is necessary but not sufficient to characterize risks. Some risks, such as secondary exposures or effects with long periods of latency, might not be apparent to those exposed.
The risk assessment in support of the Part 503 rule was the product of agency and academic experts, including individuals with long-term associations with land applications and awareness of community concerns. As required under federal law, EPA took public comment on the proposed regulations. Nevertheless, there was no evidence of efforts to engage people living adjacent to sites where biosolids are being or could be applied at the level recommended by the Presidential/Congressional Commission on Risk Assessment and Risk Management (1997). EPA guidance, such as the supplement to Risk Assessment Guidance for Superfund, Part A (EPA 1999a), provides information to improve community involvement in the Superfund risk-assessment process. Specifically, this document identifies where community input can augment and improve EPA’s estimates of exposure and risk and illustrates why community involvement is valuable during the human health risk-assessment process.
Link Between Risk Assessment and Management of Land-Application Sites
Risk assessments are conducted with the assumption that specific management practices are in place and remain in force. If these practices are not followed, the estimated risks can differ from those estimated under the assumed management practices. The risk assessment for the Part 503 rule was conducted with the assumption that specific management practices are followed. For example, complete incorporation of biosolids into the soil is assumed in assessing runoff impacts. For many sites, however, surface application to pastures is normal practice and is allowed under the Part 503 rule. Surface application provides the potential for erosion and off-site movement of biosolids and their constituents in a form much different from that assumed in the risk assessment.
The risk assessment for the Part 503 rule included the assumption that specific management practices are followed. However, because the rule does not explicitly require some of these practices, it is difficult to confirm the extent to which site operators employ these management practices. Some are measures that may be useful in minimizing risks; however, most are not requirements under the Part 503 rule.
It should be recognized that even in cases in which specific management practices are clearly delineated and required under regulations, there can be cases in which management practices are not followed through oversight, negligence, or willful noncompliance. Efforts to make risk assessment more realistic are challenged by the issue of dealing with the likelihood of noncompliance. For example, risks of home-use pesticides are assessed assuming that label directions are followed; yet experience shows that a significant number of users disregard such directions. In the case of land application of biosolids, concerns have been raised about the ability of EPA to enforce the Part 503 rule (EPA 2000b). When there are such alleged violations as applying biosolids within buffer zones and grazing of livestock on land less than 30 days after Class B biosolids were applied, any risk assessment that ignores the likelihood of those violations will not be applicable where those conditions exist. No information is available on the frequency and severity of violations of management requirements. Moreover, the committee is not aware of any risk assessment that was carried out under the assumption that one or more violations had occurred. An assessment of the risks both with and without the specified management practices would indicate the significance of noncompliance. This would provide information to be used in risk-management decision-making. Without a system that provides for registration, investigation, enforcement, and documentation of complaints concerning management
practices, EPA will not be able to compile relevant data on the level of compliance with biosolids management requirements in the Part 503 rule.
Odors present a challenge to risk assessors and managers. Until recently, odors were assumed to be an aesthetic issue. Odor control, however, is an important focus of recommendations for good practice (NBP 2001), and Schiffman et al. (2000) have suggested that odors can affect health. Odors and disease vectors as health issues are clearly within the scope of EPA. Less clear is whether EPA may address quality-of-life issues such as enjoyment of property where odors or flies might be objectionable but not an unacceptable health risk.
CHANGES IN RISK-ASSESSMENT APPROACHES IN EPA OFFICES
A number of EPA offices and programs are involved in developing risk-assessment protocols for chemical releases to ambient air, indoor air, surface water, soil, and groundwater. The methods developed in these programs and the evolution of risk-assessment methods within these offices and programs over the past 10 years provide benchmarks against which the relevance and reliability of the Part 503 rule risk assessments can be evaluated. The committee recognizes that other government agencies, such as the CDC, the U.S. Department of Agriculture, and the National Institute of Environmental Health Sciences, have also been involved in research of risk-assessment methods and in developing risk-assessment protocols. In some cases, those agencies have had a direct interest in biosolids risk. Nevertheless, the committee believes that it is beyond the scope of this report to explore the evolution of the risk-assessment process in all U.S. government agencies. Moreover, because EPA has lead responsibility for biosolids risk and works closely with other agencies on issues of risk assessment, the committee decided to focus on the offices of EPA in its review of risk-assessment methods in the U.S. government.
Office of Research and Development (ORD)
EPA’s ORD is the principal scientific and research arm of EPA. It conducts research and fosters the use of science and technology in fulfilling EPA’s mission. ORD’s two major programs involved in developing guidance on risk assessment are the National Center for Environmental Assessment and the National Exposure Research Laboratory. A brief description of some
of the major risk-assessment developments in each of these programs is provided below.
National Center for Environmental Assessment (NCEA)
NCEA serves as the national resource center for the overall process of human health and ecological risk assessments. It develops methods that reduce uncertainties in risk assessments (e.g., dose-response models and exposure models), conducts assessment of contaminants and sites of national significance, and provides guidance and support to risk assessors. Two major program areas with important developments since the risk assessments were conducted for the Part 503 rule are exposure assessment and cancer assessment.
Exposure Assessment
In 1992, EPA promulgated a new set of exposure-assessment guidelines to replace the 1986 version (EPA 1992a). The new guidelines explicitly consider the need to estimate the distribution of exposures among individuals and populations and discuss the need to incorporate uncertainty and variability analysis into exposure assessments. The guidelines discuss the roles of both analytic measurement and mathematical modeling in estimating concentrations and durations of exposure. They do not recommend specific models but suggest that models match the objectives of the particular exposure assessment being conducted and that they have the accuracy needed to achieve those objectives. They also call for detailed explication of the choices and assumptions that often must be made when faced with incomplete data and insufficient resources.
In 1997, NCEA published a support document to the guidelines called the Exposure Factors Handbook (EPA 1997a). It contains a summary of human behaviors and characteristics that affect exposure to environmental contaminants and recommends values to use for these factors. A new exposure factors handbook dealing specifically with children is in development. EPA gives special consideration to children, because they can be more heavily exposed to environmental contaminants than adults. EPA released an external review draft of the handbook in June 2000 (EPA 2000c).
NCEA has also developed a guidance document on how to conduct dermal exposure assessments (EPA 1992b). The dermal route of exposure is not understood as well as the other major routes of exposure (ingestion and inhalation). NCEA’s guidelines discuss the principles of dermal absorption
from exposures to water, soil, and vapor media and presents methods for applying those principles to human exposure assessment The guidelines were developed primarily for evaluations of waste-disposal sites or contaminated soils but are applicable to land-applied biosolids. The Office of Solid Waste and Emergency Response has also developed guidance for dermal risk assessment (EPA 2001a).
Guidance is also being developed for approaches to modeling health risks from indirect exposures to environmental contaminants. For example, Methodology for Assessing Health Risks Associated with Multiple Pathways of Exposure to Combustor Emissions (EPA 1998b) presents procedures for estimating exposures resulting from atmospheric pollutants emitted from stationary combustors, transferred through the atmosphere, and deposited on environmental media and biota. It discusses ways to estimate indirect exposures that could result from uptake and transfer from atmospheric agents through the terrestrial or aquatic food chains. This example also illustrates the need for conducting multimedia and multiple-pathway exposure assessments.
Cancer Risk Assessment
In 1996, NCEA proposed a revision to the 1986 EPA Guidelines for Carcinogen Risk Assessment to reflect new developments in understanding carcinogenesis (EPA 1996a). Revisions have been made since that proposal, and work on the guidelines is still in progress (EPA 1999b). The proposed revisions include placing greater emphasis on analyzing all the biological information on an agent rather than analyzing only the tumor data; understanding an agent’s mode of action; taking a weight-of-evidence approach to drawing conclusions about hazard; and providing guidance on assessing risks to children. When finalized, the guidelines will provide an analytical framework that will allow the incorporation of all relevant biological information, recognize a variety of situations regarding cancer hazard, and be flexible enough to allow consideration of future scientific advances.
National Exposure Research Laboratory (NERL)
NERL is EPA’s resource for guidance on exposure assessment for all environmental stressors (e.g., chemicals, biological agents, and radiation). NERL conducts research on stressor sources; pollutant transport, transformation, and exposure; and source-to-receptor predictive exposure models. NERL is also involved in the development of innovative exposure-assessment technologies.
National Exposure Surveys
One of NERL’s major efforts is to address the need to reduce uncertainty and variability in exposure assessments and the need to develop realistic exposure scenarios and assumptions. A key determinant of exposure variability is human activity. Between October 1992 and September 1994, NERL conducted the National Human Activity Pattern Survey (NHAPS) to collect data on activity patterns of subjects over a 24-hour period. The survey was intended to provide comprehensive exposure information over broad geographical and temporal scales that can be used for detailed exposure studies targeted to specific populations in the United States. Detailed tables of the survey results have been compiled (EPA 1996d), and some of the data were incorporated into the Exposure Factors Handbook (EPA 1997a).
NHAPS provides a broad description of individual activities for distinct combinations of location and time (macroactivity [e.g., amount of time spent in an enclosed vehicle]). For specific risk assessments, activity patterns can be analyzed in even greater detail using microactivity models, which can be used to describe specific contacts with exposure media (e.g., frequency of a child’s hand contact with soil and mouth). Exposures from residential environments have been given greater attention in recent years.
Another survey that was undertaken is the National Human Exposure Assessment Survey (NHEXAS). This survey was designed to evaluate comprehensive human exposure to multiple chemicals on a community and regional scale. The first phase of the survey involved measuring concentrations of chemicals in various exposure media (e.g., air, food, drinking water, soil, and dust) and in biological samples (e.g., blood and urine), and administering questionnaires to identify possible sources of exposure to chemicals. The sample collection and laboratory analyses were completed in 1998, and statistical analyses of the data are being performed. As the database is developed, it will be possible to use the data as a baseline to determine whether specific populations are exposed to increased levels of environmental contaminants.
Pharmacokinetic Models and Biomarker Data
NERL’s Exposure Methods and Monitoring Branch develops indicators of human exposure to environmental stressors. One set of indicators that provides a direct measure of exposure is biomarker data sets. Biomarkers are indicators, specific to a contaminant, of variation in cellular or biochemical components or processes, structure, or function that are measurable in biological systems or samples. When used with pharmacokinetic data and informa-
tion on the interval between exposure and collection of the biomarker information, biomarker data can be used to reduce uncertainties about exposure.
The study of pharmacokinetics provides an understanding of a chemical’s absorption, distribution, metabolism, and excretion that occurs between the time a chemical enters the body and when it leaves. Pharmacokinetic models are a mathematical representation of those processes and can be used to describe the quantitative differences between an exposure dose, a delivered dose, and, when possible, a biologically active dose at the target organ. EPA’s strategic plan for evaluating data from NHEXAS (EPA 2000d) discusses the need to consider pharmacokinetic models and parameters in evaluating the time course and associations between exposure and dose.
Office of Air and Radiation (OAR)
EPA’s OAR is responsible for national programs, technical policies, and regulations for controlling air pollution and radiation exposure. Currently, there are OAR programs to address pollution prevention, indoor and outdoor air quality, industrial air pollution, pollution from vehicles and engines, radon, acid rain, stratospheric ozone depletion, and radiation protection. Of particular interest for considering applications of risk-assessment policy are the Radiation Protection Division, Indoor Air Quality Programs, and the Office of Air Quality Planning and Standards within OAR.
Radiation Protection Division
The Radiation Protection Programs within the Radiation Protection Division provide the methods and scientific basis for EPA’s radiation exposure, dose, and risk assessments. These assessments in turn support the development of EPA policy, guidance, and rule-makings concerning radiation protection and risk management. Among other functions, the Radiation Protection Program develops radionuclide fate and transport models, dose and risk models, and dose and risk coefficients.
Indoor Air-Quality Programs
Because of the importance of understanding the sources and pathways of exposure in indoor environments, EPA has established and promoted indoor air-quality programs over the past decade. These programs deal with indoor
exposures to contaminants originating from both outdoor and indoor sources. Among the sources of indoor pollution addressed by EPA are combustion sources, such as oil, gas, kerosene, coal, and wood-combustion and tobacco products; building materials and furnishings, such as wet or damp carpet and cabinetry or furniture made of certain pressed-wood products; household cleaning and maintenance products; central heating and cooling systems and humidification devices; and outdoor sources, such as radon, pesticides, and outdoor air pollution. Of particular interest to the issue of biosolids risk assessment is the potential for indoor exposures to pathogens.
Office of Air Quality Planning and Standards (OAQPS)
EPA’s OAQPS directs national efforts to meet air-quality goals, particularly for smog, air toxics, carbon monoxide, lead, particulate matter (soot and dust), sulfur dioxide, and nitrogen dioxide. OAQPS is responsible for implementing major provisions of the Clean Air Act, including those related to visibility, permitting, and emissions standards for a wide variety of industrial facilities. Of particular interest in risk assessment is the OAQPS effort to develop methods to assess human exposure and health risks for particulate matter (PM) and multimedia pollutants released in urban air sheds. As part of that effort, OAQPS has formulated advanced and novel methods for addressing multimedia pollutants. Those methods are being incorporated into the OAQPS total risk integrated model (TRIM). TRIM provides a multimedia fate analysis and multipathway exposure assessment for toxic air pollutants and aerosols (PM).
OAQPS is also working on the National Air Toxics Assessment (NATA), a program to assess the cumulative exposures of the U.S. population to toxic air pollutants through a combination of monitoring and models.
The OAQPS effort to assess PM exposure has particular relevance to biosolids risk. PM exposure from biosolids application is raised as a concern of local communities and some public-health officials. From biosolids-application sites, PM is produced by numerous sources, including diesel emissions, traffic, and dust suspensions. A related issue is rafting—pathogens catching a ride on dust particles. Whether and how allergen proteins are transported from site to receptor is still poorly understood.
Office of Solid Waste and Emergency Response (OSWER)
OSWER provides policy, guidance, and direction for EPA’s solid-waste and emergency-response programs. Within OSWER, the Office of Solid
Waste (OSW) develops guidelines for the land disposal of municipal and hazardous waste and the Office of Underground Storage Tanks (OUST) develops guidance for limiting the risks from leaks of underground storage tanks. OSWER provides technical assistance to all levels of government to establish safe practices in waste management. OSWER is also home to the Superfund program, which addresses health concerns of communities with abandoned and active hazardous waste sites and accidental oil and chemical releases. Superfund also encourages innovative technologies to address contaminated soil and groundwater.
Office of Solid Waste (OSW)
OSW is responsible for setting limits on the concentrations of chemicals that can be placed in municipal landfills. Limits are set through a risk-assessment process that identifies and evaluates multiple exposure pathways. OSW has identified a number of potential exposure pathways linked to landfills and uses multimedia risk assessments to link human exposure and health risk to chemicals in the landfill waste. The assessment is a forward-calculating analysis that evaluates the risks of multiple exposure pathways to human and ecological receptors. One of the pathways that the OSW landfill risk assessments addresses is the advection of chemicals out of the landfill due to forced convection that results from methane and carbon dioxide generation in the waste pile.
Office of Underground Storage Tanks (OUST)
OUST was created in 1985 to carry out a congressional mandate to develop and implement a regulatory program for underground storage tank (UST) systems. OUST works with EPA regional offices and state and local UST programs to promote the use of risk-based decision-making. In OUST, risk-based decision-making (RBDM) is a process by which decisions are made about contaminated sites using a site-specific assessment of the risk each site poses to human health and the environment In cooperation with the American Society for Testing and Materials (ASTM), OUST is evaluating whether its RBDM programs are achieving their stated agency management goals.
Office of Emergency and Remedial Response (OERR)
The EPA Superfund program is administered by the OERR. After a
hazardous waste site is listed on the National Priorities List, risk assessment has an important role in the characterization and cleanup of Superfund sites. OERR provides general tools and specific tools to assist in the major steps of the risk-assessment process. In 1989, Risk Assessment Guidance for Superfund (RAGS), Part A, was issued (EPA 1989). This document provides recommended algorithms and data for calculating potential exposures to chemical contaminants found at Superfund sites. In contrast to the OUST risk methods, RAGS are more generic in providing uniform national risk-assessment defaults. Additional RAGS documents were issued in 1991 in Part B (EPA 1991b), which provides guidance on using EPA toxicity values and exposure information to derive risk-based preliminary remediation goals, and Part C (EPA 1991c), which provides guidance on the human health risk evaluations of remedial alternatives. In 1998, OERR issued Part D (EPA 1998c), and in 1999, it issued a supplement to Part A (EPA 1999a). This document is of interest to biosolids risk assessors, because the supplement provides information to improve community involvement in the Superfund risk-assessment process. Specifically, the supplement suggests ways for Superfund staff and community members to work together during the early stages of Superfund cleanup; identifies where community input can augment and improve EPA’s estimates of exposure and risk; recommends questions that the site team should ask the community; and illustrates why community involvement is valuable during the human health risk assessment at Superfund sites. A review draft of Part E provides dermal risk assessment guidance (EPA 2001a). OERR has also developed probabilistic risk assessment guidance for Superfund (EPA 2001b).
Office of Water (OW)
EPA’s OW is responsible for all national water-quality activities, including the regulation of surface water and groundwater supplies to protect human health and the environment. OW is responsible for implementing the Clean Water Act, Safe Drinking Water Act, and portions of other environmental laws and treaties that apply to water quality. Several organizations make up the OW, including the Office of Wetlands, Oceans, and Watersheds; the Office of Science and Technology; the Office of Wastewater Management (which oversees EPA’s biosolids program); and the Office of Ground Water and Drinking Water.
A major task of OW is to set drinking-water standards. Risk assessment provides a key input to this process. Since 1986, OW has more than tripled the number of contaminants for which it has published drinking-water stan-
dards, bringing the total to 94. A current challenge for OW in its effort to minimize health risks from water supplies is to find the appropriate balance between the risks from naturally occurring microbial pathogens and the chemical by-products of disinfection processes used to remove the pathogens. It is important to provide protection from these microbial pathogens while ensuring decreasing health risks to the population from disinfection by-products.
As part of its effort to protect watersheds, OW has established the total maximum daily load (TMDL) program. A TMDL is a calculation of the maximum amount of a pollutant that a body of water can receive and still meet state water-quality requirements. TMDLs are determined in part by considering multiple sources of pollutants (from point, nonpoint, and background sources, including atmospheric deposition), seasonal variations, and margins of safety. The calculations of these programs provide benchmarks for the continuing evaluation of biosolids standards.
Office of Prevention, Pesticides and Toxic Substances (OPPTS)
EPA’s OPPTS develops national strategies for toxic substance control and promotes pollution prevention and the public’s right to know about chemical risks. OPPTS has an important role in protecting public health and the environment from potential risk from toxic chemicals and pesticides. OPPTS is dealing with issues such as endocrine disruptors and lead poisoning prevention.
Within OPPTS, the Office of Pesticide Programs (OPP) regulates the use of all pesticides in the United States and establishes maximum concentrations for pesticide residues in food. As part of this effort, OPP is expanding access to information on risk-assessment and risk-management actions to help to increase transparency of decision-making and facilitate consultation with the public and affected stakeholders. OPP has a mandate under the FQPA of 1996 to address aggregate exposure and cumulative risk from multiple sources of pesticide exposure. To address that issue, OPP developed a framework for conducting cumulative risk assessments for organophosphates and other pesticides that have a common mechanism of toxicity (that act in the same way in the body). Through its cumulative risk-assessment framework, OPP will be able to consider whether the risks posed by a group of pesticides that act the same way in the body meet the FQPA safety standard of “reasonable certainty of no harm.” As part of that framework, OPP is developing new methods to assess cumulative risk, to assess residential exposure, and to aggregate exposures from all nonoccupational sources.
FINDINGS AND RECOMMENDATIONS
The Part 503 rule risk assessments were carried out more than a decade ago. In this chapter, the committee considered the likely impact of changes in risk-assessment practice in general and in various EPA offices in particular on the risk-assessment process for biosolids. The committee found that the development of methods in the broader academic community and the evolution of risk-assessment methods within various EPA offices and programs provide important benchmarks for the committee’s assessment of the relevance and reliability of the Part 503 rule risk assessments. Of particular note are updates to the risk-assessment framework recommended by the NRC, the Presidential/Congressional Commission on Risk Assessment, and various EPA offices.
The risk-assessment methods and policies practiced and advocated at EPA have changed significantly, although not at the pace recommended by the NRC and the risk commission. As a result, the Part 503 rule, which has not been modified to account for any new methods and policies, is now inconsistent with current NRC recommendations and EPA policies within various offices. Particularly relevant examples of the inconsistency are the absence of stakeholder participation and the lack of explicit treatment of uncertainty and variability.
Recommendation: Because of the significant changes in risk-assessment methods and policies over the last decade, EPA should revise and update the Part 503 rule risk assessments. Important developments include recognition of the need to include stakeholders throughout the risk-assessment process, improvements in measuring and predicting adverse health effects, advances in measuring and predicting exposure, explicit treatment of uncertainty and variability, and improvements in describing and communicating risk. EPA should consider how the updated risk assessments would change the risk-management process. A similar approach can be taken with the issue of biological agent risks.
In recent years, health-effects research has made use of large-scale studies of human health end points at multiple sites. Health-effects research has also focused on early indicators of outcome, making it possible to shorten the time between the exposure and the observation of an effect. In addition, more use has been made of meta-analysis, better modeling of dose-response relationships, and more sophisticated regression models. These improvements make possible more site-specific assessments of the impacts of biosolids land-application practices.
Managing exposure of human populations to environmental contaminants using a risk-based approach requires an accurate metric for the impacts of
contaminants on human health and a reliable process for monitoring and recording the exposures within populations assumed to be at risk. Over the past decade, the practitioners of exposure assessment have made important improvements in methods to measure and model source-to-dose relationships. These improvements have been made through greater use of time-activity surveys, personal monitors, and biomarkers of exposure, and they have made it possible to confirm some of the exposures predicted in risk assessments.
Recommendation: Many of the measures of risk used in developing the Part 503 rule guidelines cannot be monitored. Because of that inability to monitor, the committee acknowledges that EPA must perform theoretical risk assessments. Nevertheless, there is a continuing need to provide some measures of performance that can be monitored (e.g., concentrations of selected chemicals in exposure media, such as indoor air, house dust, or tap water of residences near land-application sites; and exposure biomarkers in the blood or urine of nearby residents). Recent improvements in health surveillance and exposure monitoring provide new opportunities for EPA to develop more explicit and measurable metrics of performance for biosolids land-application practices.
Advancements in monitoring health outcomes and exposure have resulted in improvements in the description and communication of risk. In particular, improved exposure assessments have led to better exposure classification in health-effects studies. Better descriptions of risk are available, using benchmark dose and margin of exposure to communicate hazard and risk in place of risk of death, hazard quotients, or exposure-potency product relationships. There have also been improved methods for prioritizing compounds using measures of risk.
Recommendation: In making revisions to the Part 503 rule risk assessment, EPA must strike a balance between expending resources to carry out site-specific data collection and expending resources to model and assess risk using existing information. In light of improvements in exposure and health monitoring, the committee encourages EPA to consider options carefully for collecting new data in support of risk-assessment assumptions before resorting to another risk assessment that relies only on existing data, models, and default assumptions. Among the data that would be of value are data on proximity of receptors to land-application sites; surveys of activities that could increase direct and indirect exposures; and samples of biosolids, air, vegetation, runoff, groundwater, and soil in environments surrounding land-application sites. In addition, EPA should conduct site-specific surveys of performance (e.g., monitor the extent to which rates and depth of application are consistent with risk-assessment assumptions) and scientifically relevant studies of health complaints.
Risk assessments make use of a number of assumptions to define chemical loading in biosolids that pose no undue risk to surrounding populations. Implicit in this process is the premise that these assumptions and the associated demographic and operational conditions will persist. However, there are no guidelines to ensure that these conditions persist.
Recommendation: Because there are no guidelines to ensure that conditions assumed in the risk assessment actually transpire, the committee recommends that the Part 503 rule provide guidance for periodic reassessments that will be used to ensure that the demographic and operational conditions of biosolids land application are consistent with the assumptions of the applicable risk assessment.
REFERENCES
Armitage, P., and R.Doll. 1954. The age distribution of cancer and a multistage theory of carcinogenesis. Br. J. Cancer 8(March):1–12.
Barnes, D.G., G.P.Daston, J.S.Evans, A.M.Jarabek, R.J.Kavlock, C.A.Kimmel, C.Park, and H.L.Spitzer. 1995. Benchmark Dose Workshop: Criteria for use of a benchmark dose to estimate a reference dose. Regul. Toxicol. Pharmacol. 21(2):296–306.
Bogen, K.T., and R.C.Spear. 1987. Integrating uncertainty and interindividual variability in environmental risk assessment. Risk Anal. 7(4):427–436.
Dockery, D.W., and J.D.Spengler. 1981. Indoor-outdoor relationships of respirable sulfates and particles. Atmos. Environ. 15(3):335–343.
EPA (U.S. Environmental Protection Agency). 1986a. The Risk Assessment Guidelines of 1986. EPA/600/8–87/045. Office of Health and Environmental Assessment, U.S. Environmental Protection Agency, Washington, DC. August 1986.
EPA (U.S. Environmental Protection Agency). 1986b. Guidelines for the Health Risk Assessment of Chemical Mixtures. EPA/630/R-98/002. Risk Assessment Forum, U.S. Environmental Protection Agency, Washington, DC. Fed. Regist. 51(185):34014–34025. (September 24, 1986). [Online]. Available: http://www.epa.gov/ncea/raf/rafguid.htm [December 27, 2001].
EPA (U.S. Environmental Protection Agency). 1986c. Guidelines for Mutagenicity Risk Assessment. EPA/630/R-98/003. Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC. September 1986.
EPA (U.S. Environmental Protection Agency). 1989. Risk Assessment Guidance for Superfund, Vol. 1. Human Health Evaluation Manual (Part A). Interim Final. EPA/540/1–89/002. Office of Emergency and Remedial Response, U.S. Environmental Protection Agency, Washington, DC. December 1989.
EPA (U.S. Environmental Protection Agency). 1991a. Part V. Guidelines for Developmental Toxicity Risk Assessment. Notice. EPA/600/R/91/001. Fed. Regist.
56(234):63798–63826. (December 5, 1991).
EPA (U.S. Environmental Protection Agency). 1991b. Risk Assessment Guidance for Superfund: Vol. 1. Human Health Evaluation Manual (Part B, Development of Risk-Based Preliminary Remediation Goals). Interim. EPA/540/R-92/003. Office of Emergency and Remedial Response, U.S. Environmental Protection Agency, Washington, D.C. December 1991.
EPA (U.S. Environmental Protection Agency). 1991c. Risk Assessment Guidance for Superfund: Vol. 1. Human Health Evaluation Maual (Part C, Risk Evaluation of Remedial Alternatives). Interim. EPA/540/R-92/004. Office of Emergency and Remedial Response, U.S. Environmental Protection Agency, Washington, DC. December 1991.
EPA (U.S. Environmental Protection Agency). 1992a. Guidelines for Exposure Assessment. EPA/600Z-92/001. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency. Washington, DC. Fed. Regist. 57(May 29):22888–22938. [Online]. Available: http://www.epa.gov/nceawww1/exposure.htm [August 1, 2001].
EPA (U.S. Environmental Protection Agency). 1992b. Dermal Exposure Assessment: Principles and Applications. Interim Report. EPA/600/8–91/011B. Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC. [Online]. Available: http://www.epa.gov/ncea/dermal.htm [August 10, 2001].
EPA (U.S. Environmental Protection Agency). 1996a. Proposed Guidelines for Carcinogen Risk Assessment. EPA/600/P-92/003C. Fed. Regist. 61(79):17960–18011. (April 23, 1996). [Online]. Available: http://www.epa.gov/nceawww1/raf/cra_prop.htm [August 1, 2001].
EPA (U.S. Environmental Protection Agency). 1996b. Guidelines for Reproductive Toxicity Risk Assessment. EPA/630/R-96/009. Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC. October 1996.
EPA (U.S. Environmental Protection Agency). 1996c. Environmental Health Threats to Children. EPA175-F-96–001. Office of the Administrator, U.S. Environmental Protection Agency. September 1996. [Online]. Available: http://www.epa.gov/epadocs/child.htm [August 1, 2001].
EPA (U.S. Environmental Protection Agency). 1996d. Descriptive Statistics Tables from a Detailed Analysis of the National Human Activity Pattern Survey (NHAPS) Data. EPA/600/R-96/148. Report prepared by A.M.Tsang and N.E.Klepeis, Lockhead Martin Environmental Service Company, Las Vegas, NV, for National Exposure Research Laboratory, Office of Research and Development, U.S. Environmental Protection Agency, Las Vegas, NV. July 1996.
EPA (U.S. Environmental Protection Agency). 1997a. Exposure Factors Handbook. Vol. I, II, III. EPA/600/P-95/002Fa-c. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency. [Online]. Available: http://www.epa.gov/ncea/exposfac.htm [July 31, 2001].
EPA (U.S. Environmental Protection Agency). 1997b. Guiding Principles for Monte
Carlo Analysis. EPA/630/R-97/001. March 1997. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency. [Online]. Available: http://www.epa.gov/ncea/monteabs.htm [April 2, 2001].
EPA (U.S. Environmental Protection Agency). 1997c. Policy for Use of Probabilitistic Analysis in Risk Assessment at the U.S. Environmental Protection Agency. May 15, 1997. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency. [Online]. Available: http://www.epa.gov/ncea/mcpolicy.htm [April 2, 2001].
EPA (U.S. Environmental Protection Agency). 1998a. Guidelines for Neurotoxicity Assessment. EPA/630/R-95/001F. Risk Assessment Forum, U.S. Environmental Protection Agency, Washington, DC. April 1998.
EPA (U.S. Environmental Protection Agency). 1998b. Methodology for Assessing Health Risks Associated with Multiple Pathways of Exposure to Combustor Emissions. EPA 600/R-98/137. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, OH. [Online]. Available: http://www.epa.gov/nceawww1/combust.htm [December 26, 2001].
EPA (U.S. Environmental Protection Agency). 1998c. Risk Assessment Guidance for Superfund: Volume I, Human Health Evaluation Manual (Part D, Standardized Planning, Reporting, and Review of Superfund Risk Assessments). Interim. OSWER 9285.7–01D. Office of Emergency and Remedial Response, U.S. Environmental Protection Agency, Washington, D.C.
EPA (U.S. Environmental Protection Agency). 1999a. Risk Assessment Guidance for Superfund: Vol. 1. Human Health Evaluation Manual, Supplement to Part A: Community Involvement in Superfund Risk Assessments. EPA 540-R-98–042. Office of Solid Waste and Emergency Response, U.S. Environmental Protection Agency, Washington, DC. March 1999. [Online]. Available: http://www.epa.gov/superfund/programs/risk/ragsa/ci-ra.htm [December 26, 2001].
EPA (U.S. Environmental Protection Agency). 1999b. Guidelines for Carcinogen Risk Assessment. NCEA-F-0644. Review Draft, July 1999. National Center for Environmental Assessment, Risk Assessment Forum, U.S. Environmental Protection Agency, Washington, DC. [Online]. Available: http://www.epa.gov/ncea/raf/crasab.htm [September 13, 2001].
EPA (U.S. Environmental Protection Agency). 2000a. Supplementary Guidance for Conducting Health Risk Assessment of Chemical Mixtures. EPA/630/R-00/002. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC. August 2000. [Online]. Available: http://www.epa.gov/nceawww1/raf/chem_mix.htm [December 26, 2001].
EPA (U.S. Environmental Protection Agency). 2000b. Water. Biosolids Management and Enforcement. Audit Report No. 2000-P-10. Office of Inspector General. March 20, 2000. [Online]. Available: http://www.epa.gov/oigearth/audit/list300/00P0010.pdf [December 20, 2001].
EPA (U.S. Environmental Protection Agency). 2000c. Child-Specific Exposure
Factors Handbook. (External Review Draft). NCEA-W-0853. National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC. June 2000. [Online]. Available: http://www.epa.gov/nceawww1/csefh2.htm [December 26, 2001].
EPA (U.S. Environmental Protection Agency). 2000d. Strategic Plan for the Analysis of the National Human Exposure Assessment Survey (NHEXAS) Pilot Study Data. EPA/600/R-00/049. National Exposure Research Laboratory and National Center for Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency, Washington, DC. November 2000. [Online]. Available: http://www.epa.gov/NERL/research/nhexas/strategy.pdf [August 6, 2001].
EPA (U.S. Environmental Protection Agency). 2001a. Risk Assessment Guidance for Superfund, Vol. 1. Human Health Evaluation Manual (Part E, Supplemental Guidance for Dermal Risk Assessment). Interim Review Draft. EPA/540/R/99/005. OSWER 9285.7–02EP. PB99–963312. Office of Emergency and Remedial Response, U.S. Environmental Protection Agency, Washington, DC. September 2001. [Online]. Available: http://www.epa.gov/superfund/programs/risk/ragse/introduction.pdf [March 12, 2002].
EPA (U.S. Environmental Protection Agency). 2001b. Risk Assessment Guidance for Superfund: Vol. 3.- Part A. Process for Conducting Probabilistic Risk Assessment. EPA 530-R-02–002. PB2002 963302. Office of Emergency and Remedial Response, U.S. Environmental Protection Agency, Washington, DC. December 2001. [Online]. Available: http://www.epa.gov/oerrpage/superfund/programs/risk/rags3a/pdf/contpref.pdf [May 21, 2002].
EPA (U.S. Environmental Protection Agency). 2001c. Exposure Analysis for Dioxins, Dibenzofurans, and CoPlanar Polychlorinated Biphenyls in Sewage Sludge Technical Background Document. Draft. EPA Contract No. 68-W6–0053. RTI 7600-OP3.040. Prepared by Center for Environmental Analysis, Research Triangle Institute, Research Triangle Park, NC, for the Office of Water, U.S. Environmental Protection Agency, Washington, DC. November 30, 2001. [Online]. Available: http://www.epa.gov/waterscience/biosolids/riskasdraft.pdf [February 26, 2002].
Evans, J.S., J.D.Graham, G.M.Gray, and R.L.Sielken Jr. 1994. A distributional approach to characterizing low-dose cancer risk. Risk Anal. 14(1):25–34.
Finkel, A.M. 1990. Confronting Uncertainty in Risk Management: A Guide for Decision-Makers. Washington, DC: Center for Risk Management, Resources for the Future.
Habicht, H.F. 1992. Guidance on Risk Characterization for Risk Managers and Risk Assessors, EPA Memorandum from EPA Deputy Administrator F.Henry Habict, III, to Assistant Administrators and Regional Adminstrators. (Publish as pages 351–374 in Science and Judgment in Risk Assessment. 1994. Washington, DC: National Academy Press)
Hertwich, E.G., S.F.Mateles, W.S.Pease, and T.E.McKone. 2001. Human toxicity potentials for life-cycle analysis and toxics release inventory risk screening. Environ. Toxicol. Chem. 20(4):928–939.
Krewski, D., K.S.Crump, J.Farmer, D.W.Gaylor, R.Howe, C.Portier, D.Salsburg, R.L.Sielken, and J.Van Ryzin. 1983. A comparison of statistical methods for low dose extrapolation utilizing time-to-tumor data. Fundam. Appl. Toxicol. 3(3):140–160.
Melia, R.J.W., C.D.V.Florey, S.C.Darby, E.D.Palmes, and B.D.Goldstein. 1978. Differences in NO2 levels in kitchens with gas or electric cookers. Atmos. Environ. 12(6–7):1379–1381.
Morgan, G.M., M.Henrion, and M.Small. 1990. Uncertainty: A Guide to Dealing with Uncertainty on Quantitative Risk and Policy Analysis. Cambridge, UK: Cambridge University Press.
Mumtaz, M.M., and P.R.Durkin. 1992. A weight-of-evidence approach for assessing interactions in chemical mixtures. Toxicol. Ind. Health 8(6):377–406.
NBP (National Biosolids Partnership). 2001. Manual of Good Practice for Biosolids. Interim Final Draft. March 13, 2001. [Online]. Available: http://biosolids.policy.net/proactive/newsroom/release.vtml?id=20961 [January 18, 2002].
NRC (National Research Council). 1982. Risk and Decision Making: Perspectives and Research. Washington, DC: National Academy Press.
NRC (National Research Council). 1989. Improving Risk Communication. Washington, DC: National Academy Press.
NRC (National Research Council). 1993. Issues in Risk Assessment. Washington, DC: National Academy Press.
NRC (National Research Council). 1994. Science and Judgement in Risk Assessment. Washington, DC: National Academy Press.
NRC (National Research Council). 1996. Understanding Risk, Informing Decisions in a Democratic Society. Washington, DC: National Academy Press.
Pellizzari, E.D., T.D.Hartwell, R.L.Perritt, C.M.Sparacino, L.S.Sheldon, H.S.Zelon, R.W.Whitmore, J.J.Breen, and L.A.Wallace. 1986. Comparison of indoor and outdoor residential levels of volatile organic chemicals in five U.S. geographic areas. Environ. Int. 12(6):619–623.
Presidential/Congressional Commission on Risk Assessment and Risk Management. 1997. Framework for Environmental Health Risk Management, Final Report, Vol. 1. Washington, DC: The Commission.
Schiffman, S.S., J.M.Walker, P.Dalton, T.S.Lorig, J.H.Raymer, D.Shusterman, and C.M.Williams. 2000. Potential health effects of odor from animal operations, wastewater treatment, and recycling of byproducts. J. Agromed. 7(1):7–81.
Spengler, J.D., C.P.Duffy, R.Letz, T.W.Tibbets, and B.G.Ferris Jr. 1983. Nitrogen dioxide inside and outside 137 homes and implications for ambient air quality standards and health effects research. Environ. Sci. Technol. 17(3):164–168.
Taylor, A.C., J.S.Evans, and T.E.McKone. 1993. The value of animal test information in environmental control decisions. Risk Anal. 13(4):403–412.
Thomas, K.W., E.D.Pellizzari, C.A.Clayton, R.L.Perritt, R.N.Dietz, R.W.Goodrich, W.C.Nelson, and L.A.Wallace. 1993. Temporal variability of benzene exposures for residents in several New Jersey homes with attached garages or tobacco smoke. J. Expo. Anal. Environ. Epidemiol. 3(1):49–73.
Wallace, L.A. 1993. A decade of studies of human exposure: What have we learned? Risk Anal. 13(2):135–139.
Wilson, J.D. 2001. Advanced Methods for Dose-Response Assessment: Bayesian Approaches. Final Report based on a workshop held September 18–20, 2000. Discussion Paper 01–15. Resources for the Future, Washington, DC.