4
Tools
This chapter describes the physical, chemical, and biological tools that have been used to evaluate bioavailability, and it assesses their scientific basis. In general, understanding contaminant bioavailability from soils and sediments requires studying the processes illustrated in Figure 1-1. A first-order need is to identify the contaminant of concern and determine its form, concentration, and distribution (which can correlate with understanding bioavailability process A). These characteristics can be inferred from the soil or sediment matrix or determined directly with operational or mechanistic measurements. Some analytical techniques like spectroscopy can directly address where and how a chemical is associated with sediment or soil, while techniques like extractions operationally address form. Biological tools typically consider entry of the contaminant into the living organism (D in Figure 1-1) without directly measuring processes A–C. Of course, processes A, B or C might be manipulated or measured by other means, with biological tools then being used to evaluate an organism’s responses to those manipulations or measurements. One class of biological tools addresses complex responses like toxicity (E in Figure 1-1), for which bioavailability is only one of several possible influences. This chapter does not discuss tools applicable to processes B and C, like fate and transport models, as there are numerous other reports dealing with fate and transport. Rather, the tests that are part of this chapter mainly deal with bioavailability processes A, D, and E; such tests usually assume a constant transport condition.
SUMMARY TABLES
In illustrating the range of physical, chemical, and biological approaches that have been used to evaluate bioavailability processes, this chapter reflects the existing state of knowledge. It is not meant to be an exhaustive list from which one can choose the ultimate tool, nor should it be read as a list of approved approaches for explicitly considering bioavailability. The state of the science is such that little consensus exists about optimal approaches. Among the tests reviewed here, some are appropriate for some situations, but most are not generally applicable to a wide spectrum of situations.
Table 4-1 summarizes the characteristics of the tools covered in the chapter, including what process the tool studies, the approximate cost, and the status of the tool in terms of its future use. It is important to recognize that most tools are still in development and few are fully validated by a body of work relating their predictions to independent measures from nature. Almost all of the tools are broadly applicable to both soils and sediments. Where a test is specific to one or the other, it is mentioned in the description of that test, rather than in the table.
Table 4-2 specifies some generic strengths and limitations of each method and thereby illustrates that every method has tradeoffs. The criteria used for Table 4-2 are:
-
Application to the field. Some methods can be employed in complex natural settings (score 3), some can be used on materials collected from the field (score 2), and some require experimental manipulations such as contaminant spiking (score 1).
-
Application to solid phase. A method that directly addresses processes in the solid phase of sediments or soils, such as a method that evaluates contaminant form in the solid, would score 3. In contrast, a method that requires measurement of the properties of an extract scores 1. A biological test that addresses the solid phase in situ scores higher (a field bioaccumulation survey) than a method that takes the solid phase out of context for the evaluation (a lab sediment bioassay), which scores higher than a test that uses an extract (pore water, Microtox or elutriate bioassay).
-
Single vs. lumped processes. Methods that measure a single process are most likely to illustrate a specific mechanism at work. For example, some physical-chemical methods directly evaluate metal form, while other methods measure one mechanism instrumental to bioavailability such as initial biouptake. These score 3. Speciation can be inferred from some methods, as can biouptake from methods like whole organism bioaccumulation (score 2). Other methods that measure a mixture of processes are more operational and less mechanistic (score 1). For example, extractions remove contaminants from an unknown suite of forms without quantifying any processes. Biological methods like toxicity tests are influenced by biouptake plus other processes that influence toxicity.
TABLE 4-1 Characteristics of Tools for Measuring Bioavailability
|
Tool |
Process Studieda |
Costb |
Statusc |
|
Physical/chemical characterization of the solid phase |
|||
|
General characteristics • Organic carbon content • Particle/grain size • pH • CEC • Humic and fulvic acid content |
Chemical characteristics of the solid (except particle size which is a physical characteristic). |
$ |
Standard protocols available |
|
Specific structures • Characterization of carbonaceous and other solid phases using NMR, petrography, EA, IR/FTIR |
Molecular characteristics of solid substrate. |
$$ to $$$ |
Currently research grade for contaminated site application |
|
Specific forms of contaminant bound to solids • XRD and SEM • XAS • μL2MS • SIMS • NMR • EPR • XPS |
Association and dissociation processes, including the roles of surface morphology, oxidation state, and compound or element location. |
XRD, SEM—$$ All others—$$$ |
XRD, SEM—Standard protocols available; all others are research grade |
|
Extraction of soils and sediments for inorganic contaminants |
|||
|
Extracts that change the solid phase • Conventional • Sequential • TCLP, SPLP |
Dissociation from the solid phase. Sequential extracts attempt to differentiate between forms of elements associated with different components or phases of the particle. |
$ |
Some extracts in use and in regulations and thus standardized, but sequential extracts at research stage or in development |
|
Passive approaches • Passive extracts • Pore water measurements with ASV or ion-specific electrode • Exchangeable resins |
Passive extracts determine dissociation from the solid phase. ASV and electrodes measure pore water concentrations. Exchange resins measure dissociation from the solid phase and physical flux to aqueous phase. |
$(but need ICP-MS for exchangeable resins) |
Research grade, no standard protocols developed; exchangeable resins better developed for sediments |
|
Tool |
Process Studieda |
Costb |
Statusc |
|
In vitro tests to mimic human intake for both organics and inorganics |
Dissociation from the solid phase that mimics physiological fluids. |
$ to $$ |
Variable: validated for lead, but in various stages of development for others |
|
Extraction and other tests of soils and sediments for organic contaminants |
|||
|
Fluid-phase extractions • Mild solvents • SWE • Supercritical CO2 extraction • PTD |
Dissociation from the solid phase. |
$ |
Mild solvents have standardized protocols; supercritical CO2 and SWE are in development |
|
Solid phase and membrane-based extractions • Tenax • C-18 • SPME • SPMD • DGT |
Dissociation from the solid phase and physical flux to aqueous phase by capturing desorbed contaminant on highly sorptive matrix or gel device. |
$ |
Standard protocols for using these methods for measuring contaminants in water; for soils and sediments, all of these techniques are in development |
|
Other desorption tests • Gas purge • Desorption kinetics and activation energy |
Dissociation from the solid phase. |
$$ to $$$$ |
In development |
|
Normalizations |
|||
|
Organic and inorganic correlations • Ratios and models • AVS/SEM • EqP |
EqP and AVS/SEM assume reactants control dissociation from the solid phase; other ratios are determined empirically from regressions in field data. |
$ |
Research grade; varies with evaluator |
|
Biological approaches to measuring uptake |
|||
|
Assimilation efficiency |
Biological uptake across the gut wall. |
$$ |
Research grade |
|
Mineralization/assimilation assays for microorganisms |
Integrated measure of bacterial uptake and metabolic degradation. |
$ to $$ |
Research grade |
|
Tool |
Process Studieda |
Costb |
Statusc |
|
Bioassays: cell cultures and isolated organs/tissues |
Biological uptake into cell or organ. |
$$ |
Research grade |
|
Bioassays: whole organism bioaccumulation • Plants • Invertebrates • Fish • Birds and mammals |
Biological uptake into whole organism.Various endpoints are measured, including tissue, blood,etc. |
Plants—$ to $$ Earthworm test—$$ Mammals—$$$ |
Standard protocols forplants, invertebrates, and birds; research grade when plants and other animals used as surrogates |
|
Field survey: whole organism bioaccumulation |
Biological uptake into whole organism in field. |
$$ |
Research grade |
|
Biological approaches to measuring organismal response and toxicity |
|||
|
Reporter systems |
Integrated measure of dissociation from the solid phase,bacterial uptake, and effect on gene expression and subsequent events. |
$$$ |
Research grade |
|
Biomarkers |
Integrated measure of uptake and response at a subcellular level. |
$ to $$$$ (gene expression) |
In development |
|
Toxicity tests: spiked • Plant • Invertebrate • Fish • Mammal, bird |
Integrated measure of uptake and toxic effects. |
$ to $$ |
Standard protocols available for fresh and saltwater sediments |
|
Toxicity tests: site- specific materials |
Site-specific integrated measure of uptake and toxic effects. |
$ to $$$$ |
Standard protocol available |
|
Microbial community bioassays |
Integrated measure of uptake, toxic effects, and community interactions. |
$$ |
Research grade |
|
Ecosystem level mesocosms |
Integrated measure of many processes including ecosystem level processes like food web transfer. |
$$$$$ |
Standard protocols available |
|
Tool |
Process Studieda |
Costb |
Statusc |
|
Environmental exposure studies |
Integrated measure of many processes including measurable effects in humans. |
$$$$$ |
Research grade |
|
aProcess Studied: How does this tool address the physical, chemical or biological processes that influence bioavailability? bCost: $ to $$$$$: Costs in approximate order of magnitude, with $ equal to $100s. cStatus: standardized protocol, research grade, or in development. |
|||
-
Immediacy or relevance to entry of contaminant into living cell (biouptake). Entry of a contaminant into a living cell across a biological membrane is the process most immediately relevant to determining bioavailability. Some biological methods involve direct determination of transport or biouptake (score 3). Some measure many processes including biouptake, or a process tangential to biouptake like toxicity, or they mimic biouptake as with certain extractions (score 2). Some physical-chemical methods are unrelated to biouptake (score 1).
-
Ability to generalize. Although site-specific tests are essential to managing an individual site, methods that allow predictions (or development of predictive capabilities) without measuring all processes are ultimately a desirable approach. Methods that are predictive, like some models or some tests that determine a mechanism that can be unambiguously compared from site-to-site, score highest in this category (score 3). Methods that are predictive but not yet of proven reliability score 2. Approaches that are of value at a site but do little to explain how the bioavailability processes at that site are comparable to other sites score 1.
-
Relevance to regulation. The relevance of a method to the pressing concerns at a site has led to the use of certain tests for regulatory purposes (like toxicity tests or direct evaluations of human health). Also, methods that are simple and practical to employ, or methods that yield a single value, are most likely to have been applied in the regulatory setting. Thus, methods that managers or decision-makers can interpret or have interpreted as directly relevant to their needs score 3. Methods that have seldom been used in a regulatory setting or have limited potential for such use score 1.
-
Usefulness as a research tool. Relevance as a research tool is just as important as relevance to regulation because of the great need for better understanding the processes that govern bioavailability. Methods that are of use in explaining processes in specific circumstances or in mechanistic detail score highest (score 3), even if they are of limited use in applications. Methods that are of use in a correlative fashion in experimental studies score 2. Methods of limited use in research score 1.
TABLE 4-2 Rankings of Bioavailability Tools According to Seven Criteria
|
Technique |
Application to the Field |
Application to Solid Phase |
Single vs. Lumped Processes |
|
Physical/chemical characterization of the solid phase |
|||
|
General characteristics • Organic carbon content • Particle/grain size • pH • CEC • HA/FA |
2 Can test field samples in the laboratory. |
3 Directly relevant to solid phase in situ; necessary to understand solid phase reactions. |
2 Measures are the outcome of lumped processes, but can be used to interpret single processes. |
|
Specific structures • Characterization of carbonaceous and other solid phases using NMR, petrography, EA, IR/FTIR. |
2 Can test field samples in the laboratory. |
3 Directly measures the solid phase. |
2 Determines nature of the phase but not contaminant-phase interactions. |
|
Specific forms of contaminant bound to solids • XRD and SEM • XAS • μL2MS • SIMS • NMR • EPR • XPS |
2 Some methods hard to use on natural particles. Detection limits of equipment can cause problems in natural settings. |
3 Directly applicable to solid phase. |
3 Uniquely suited to identify mechanisms of association. |
|
Extraction of soils and sediments for inorganic contaminants |
|||
|
Extracts that change the solid phase • Conventional • Sequential • TCLP, SPLP |
2 Can extract field soils and sediments, but must remove from field for test. |
2 Concentration extracted is qualitatively or operationally related to associations (form) in the solid phase. |
1 Operational measure that lumps different association/dissociation processes. |
|
Immediately Relevant to Entry into Living Cell |
Ability to Generalize |
Relevance to Regulation |
Usefulness as a Research Tool |
|
|
|||
|
1 Characteristics alone are not predictive of biouptake, but are necessary for inferences about other measures and models. |
2 Leads to generalization, but by themselves such measures are not predictive of bioavailability processes. |
2 Regulators sometimes use such information in normalizations. |
3 Essential to understanding contaminant form and links to biouptake in situ. |
|
1 Characteristics alone are not predictive of biouptake, but are necessary for inferences about other measures and models. |
2 Leads to generalization, but by themselves such measures are not predictive of bioavailability processes. |
1 Seldom used for soil/sediment criteria. May be useful eventually. |
3 Potential for contributing to mechanistic understanding. |
|
1 Requires inference about link between specific form and biouptake. |
2 Will eventually be essential to generalizing about bioavailability processes. |
1 Complicated and consequently of limited use in regulatory environment. |
3 Potential to understand what controls bioavailability processes. |
|
2 Extracted concentrations are linked to biouptake by correlation. Best developed for use in particular conditions (e.g., restricted soil series; nutrient deficiency). |
2 Generalizations are correlative and some are useful in the appropriate context. |
2 Some extracts are in regulatory guidelines, mainly for use as screening tool (e.g., TCLP). Used where groundwater is focal point. Not for sequential extracts. |
2 Better accepted for soils. Contentious for use in sediments. Relationships are correlative rather than mechanistic. |
|
Technique |
Application to the Field |
Application to Solid Phase |
Single vs. Lumped Processes |
|
Passive approaches • Passive extracts • ASV • Pore water measurements with ASV or ion-specific electrode • Exchangeable resins |
2 Extracts miss in situ influences because you must remove materials from field setting. In situ pore water measurements are difficult to make and thus limited. |
2 Passive extracts mimic solid phase exchange reactions, at equilibrium. Measures in pore water determine actual outcome of solid phase reactions and dissolved speciation. |
1 Extracts and resins are operational measures that lump different association/dissociation processes. Pore water concentrations are the outcome of several processes. |
|
In vitro tests to mimic human intake for both organics and inorganics |
2 Can use field soils and sediments, but must remove from field for test. |
2 Extract the solid phase with simulated physiological fluid. |
1 Operational measure that lumps multiple processes. |
|
Extraction and other tests of soils and sediments for organic contaminants |
|||
|
Fluid-phase extractions • Mild solvents • SWE • Supercritical CO2 extraction • PTD |
2 Can extract field sediments, but must remove from field for test. |
2 Extracts mimic solid phase exchange reactions. |
1 Operational measure that lumps multiple processes. |
|
Solid phase and membrane-based extractions • Tenax • C-18 • SPME • SPMD • DGT |
2–3 Can use field soils and sediments, but must remove from field for test. May be able to use SPME, SPMD, DGT in situ. |
3 Directly applicable to the solid phase or slurry. |
1 Operational measure that lumps multiple processes. |
|
Other desorption tests • Gas purge • Desorption kinetics and activation energy |
2 Can use field samples, but difficult to sustain in field setting. |
3 Directly applicable to the solid phase or slurry. |
2 Single vs. lumped processes can be decoupled by careful experimental design and working with component materials. |
|
Immediately Relevant to Entry into Living Cell |
Ability to Generalize |
Relevance to Regulation |
Usefulness as a Research Tool |
|
2 Extracted concentrations are linked to biouptake by correlation. Pore water concentrations are linked by inference that unassociated form is taken up; most useful for plant uptake. |
2 The best methods (like DGT) correlate with bioavailability, but there is uncertain reliability of generalizations. |
3 Used in some instances as trigger values for soils. Some sediment guidelines use porewater concentrations. |
2 Used in research, although relationships are correlative rather than mechanistic. |
|
2 Infers that what can be extracted will be taken up by organism (biomimetic). |
1 Site-by-site test. Limited for generalization. |
3 Simplicity makes it attractive to regulators. |
2 Operational aspects limit use in research. |
|
|
|||
|
2 Infers that what can be extracted will be taken up by organism (biomimetic). |
1 Reliability of generalizations is unproven. |
1 Regulators seldom use such information for soil/sediment criteria; may be useful eventually. |
2 Operational aspects limit use in research. |
|
2 Biomimetic but still an inferential link to biouptake. |
2 Reliability of generalizations about bioavailability is unproven; work in progress. |
1 Regulators seldom use such information for soil/sediment criteria; may be useful eventually. |
3 Potential to measure processes important to biouptake (e.g., can get rates of release). |
|
1 Inferential link to biouptake by correlation or mechanistic model. |
3 Generalizations possible with careful experimentation on component materials from different sites. |
2 Potential to reveal the relationship between aqueous and solid phase concentrations and soil quality criteria. |
3 Can lead to greater mechanistic understanding. |
|
Technique |
Application to the Field |
Application to Solid Phase |
Single vs. Lumped Processes |
|
Normalizations |
|||
|
Organic and inorganic correlations • Ratios and models • AVS/SEM • EqP |
2 Extracted contaminant normalized to in situ conditions. Difficult to mimic field setting. |
2 Designed to describe associations with the solid phase that are relevant to biouptake (e.g., those that control exchange). |
2 Ratioing assumes specific processes are described, and infers that they define biouptake. |
|
Biological approaches to measuring uptake |
|||
|
Assimilation efficiency |
1 Can use natural samples, but requires spiking and loss of in situ influences. |
2 Direct intake from solid. Allows inferences about natural solids that are ingested. |
3 Mechanistic. Determination of single process (biouptake). |
|
Mineralization/assimilation assays for microorganisms |
1 Requires sample removal, and sometimes spiking. |
1 Requires contaminant transfer to aqueous phase. |
1 Measures the composite effect of several processes. |
|
Bioassays: cell cultures and isolated organs/tissues |
2 Can use field soils and sediments, but must remove from field for test. |
1–2 Some techniques can use solid phase material while others require extracts. |
3 Mechanistic. Determination of single process (biouptake). |
|
Bioassays: whole organism bioaccumulation • Plants • Invertebrates • Fish • Birds • Mammals (all exposure routes) |
1–2 Can use field soils and sediments, but must remove from field for test. May have to spike dermal tests. |
2 Solid phase materials can be tested directly. |
2 Whole organism bioaccumulation integrates influences of several biological processes, but is indicative of biouptake. |
|
Immediately Relevant to Entry into Living Cell |
Ability to Generalize |
Relevance to Regulation |
Usefulness as a Research Tool |
|
|
|||
|
2 Inferential link to biouptake or toxicity via correlation. Some tests assume pore water is the only route of intake. |
2 Generalizations can be made, but uncertainties add controversy. |
2 Accumulation ratios are used (EqP and AVS/SEM) or proposed for use in regulations because of simplicity. |
2 Useful for want of better method, but operational simplifications limit use in understanding bioavailability processes. |
|
3 Directly measures biouptake. |
2 Can generalize about intake from food types only. |
1 Unused. Potential if used with models, but complex. |
3 Simple and reliable way to study important bioavailability processes other than just intake. Mainly useful for small animals. |
|
2 Requires intracellular activity in bacterium, so must assume a link between biouptake and degradation. |
1 Used for site-specific measures; generalizations difficult to draw. |
2 Unused, but perhaps could be standardized. |
2 Might shed light on the biouptake step for microorganisms. |
|
3 Directly measures biouptake. |
2 When experiments are focused on mechanisms, results can be generalized. |
1 Unused. Potential if used with models, but complex. |
3 Simple and reliable way to study biouptake in vitro. |
|
3 Can be used to directly measure biouptake. |
1 Generalization possible only if data are available for a broad array of sites or situations. |
3 Used directly in risk assessments. |
2 Mostly a tool for empirical measurements, but commonly used as a probe of bioresponse in experimental research. |
|
Technique |
Application to the Field |
Application to Solid Phase |
Single vs. Lumped Processes |
|
Field survey: whole organism bioaccumulation |
3 In situ test. |
2 Integrates exposure from all influential media, including solid phase. |
2 Whole organism bioaccumulation integrates influences of several biological processes, but is indicative of biouptake. |
|
Biological approaches to measuring organismal response and toxicity |
|||
|
Reporter systems |
2 Can use field soils and sediments, but usually must remove from field for test. |
1 Usually does not directly assess the sorbed phase, but an extract. In situ tests may be available soon. |
1 Measures the composite effect of several processes. |
|
Biomarkers |
2 Can use field soils and sediments, but usually (not always) must remove from field for test. In situ tests with invertebrates possible. |
2 Solid phase materials can be tested directly (but generally not in situ. |
2 Measures the composite effect of several processes, but gene expression techniques can be used to interpret single processes. |
|
Toxicity tests: spiked • Plants • Invertebrates • Fish • Birds • Mammals |
1 Simulates exposure. |
1 Indirect application to solid phase. Although solids are used in tests, they are not natural samples. |
1 Measures the composite effect of several processes. |
|
Toxicity tests: site-specific materials |
2 –3 Use sediment or soil from nature; in situ tests increasingly used. |
2 Solid phase materials can be tested directly (some in situ), but response integrates exposure from other media as well. |
1 Measures the composite effect of several processes. |
|
Immediately Relevant to Entry into Living Cell |
Ability to Generalize |
Relevance to Regulation |
Usefulness as a Research Tool |
|
2 Can be used to directly measure biouptake, but as an integrated response to influential biological and physicochemical processes. |
1 Generalization possible only if data are available for a broad array of sites or situations. |
3 Concentrations can be used to regulate exposure, but guidance is often limited, especially for ecosystems. |
2 Mostly a tool for empirical measurements, but commonly used for research applications. |
|
|
|||
|
2 –3 (for bacteria) Must assume a link between biouptake and response being measured by the reporter system. |
1 Site-specific use is most viable. Ultimately may be able to draw generalizations (e.g., relative availability of chemicals). |
2 Standardization and use feasible, and hence the potential for use in regulations. |
2 Research may shed light on relative bioavailability to microorganisms under different conditions. |
|
2 Must assume a link between biouptake and biomarker response. |
1 Site-specific use is most viable. Ultimately may be able to draw generalizations from experimental studies. |
2 Need to evaluate every site; may be extrapolated for some regulations. |
2–3 Historically used as a research tool. More recently used as a site-specific tool in the field. Potential for understanding what controls bioavailability at molecular level. |
|
2 Must assume a link between biouptake and toxic response. |
2 Generalizations most useful for extreme cases. Uncertain generalizations in natural settings. |
3 Simple test commonly used for regulation, although it does not reflect the natural condition. |
2 Primary research tool but not mechanistic. Most effective tests attempt to mimic nature. |
|
2 Must assume a link between biouptake and toxic response. |
1 Site specific tests have limited generalization. Generalizations are correlative. |
3 Used for some regulations. Need to test every site. |
2 A primary research tool but not mechanistic. Has potential for site-specific use as survey tool. |
|
Technique |
Application to the Field |
Application to Solid Phase |
Single vs. Lumped Processes |
|
Microbial community bioassays |
3 Tests microbial communities in situ. |
2 Biological signal associated with solid phase is feasible. |
1 Measures the composite effect of several processes. |
|
Ecosystem level mesocosms |
1–2 Simulates exposure in nature (with field samples—2 or artificial samples—1). |
2 Solid phase materials can be tested directly, but response integrates exposure from other media as well. |
1 Measures the composite effect of several processes. |
|
Environmental exposure studies |
3 Study conducted in natural setting. |
1 Indirect application to the solid phase. |
1 Measures the composite effect of several processes. |
No one method achieves the highest rating in all columns, and none of these methods fail all criteria. Because all approaches involve tradeoffs, there is not a universal method that meets all needs for characterizing the complex processes that determine bioavailability. For example, some approaches focus on understanding physical desorption of the contaminant from the solid phase but implicitly assume that desorption equals bioavailability (ignoring, for example, a dietary component to bioavailability). A number of tests encompass multiple processes in a single measurement such that isolating individual influences is difficult. Others attempt to isolate individual processes and as a consequence may have endpoints that are marginally relevant to bioavailability. The Table 4-1 and 4-2 entries also suggest how the tools would rank for other parameters of interest. For example, the uncertainty of a method’s results can be inferred from its status, the ability to generalize its results, and its relevance to regulations.
An important factor that is not addressed directly in Table 4-2 is the inherent conflict of scale between the methods and processes experienced by an organism. That is, many tools measure outcomes at a scale different from the processes that influence bioavailability. For example, some probes of specific forms determine interactions at the molecular scale or at an individual site, whereas bioavailability in a natural setting will result from integration across a number of sites, not all of
|
Immediately Relevant to Entry into Living Cell |
Ability to Generalize |
Relevance to Regulation |
Usefulness as a Research Tool |
|
1 Must assume many links between biouptake and ecosystem processes. |
1 Site-specific usually. Broad generalizations can be drawn from experiments or correlative studies. |
1 Unlikely regulatory use unless “control ” site available. |
2 Limited potential to elucidate processes, but new methods assess microbial community function and diversity. |
|
1 Must assume many links between biouptake and ecosystem processes. |
2 Can broadly extrapolate to field conditions. Uniquely able to generalize about responses across ecosystem-level processes. |
1 Could provide valuable information but regulators reject complexity. |
3 Very useful research tool if designed carefully. Replication is big challenge. |
|
2 Must assume links between biouptake and other physiological processes. |
1 Site-specific nature limits generalization. |
3 Used in regulation because human population studied. |
2 Limited use as a research tool, but has potential when coupled to exposure models. |
which are necessarily similar to that characterized by the probe. More gross techniques, like extraction-based methodologies, have the opposite problem. They determine form from a sample that may encompass much more sediment or soil than the microhabitat-scale at which many organisms experience their environmental milieu.
The purpose of Tables 4-1 and 4-2 is to show that a variety of tools can be applied to the question of contaminant bioavailability and show what processes the tools address. The categories are meant to guide readers toward potentially practical tools for their individual needs, and to compare the varying attributes of different tools. Of course, many of the categories have an element of subjective judgement, and experts may disagree about the details of some entries. Thus, the tables (particularly Table 4-2) are not meant to provide a quantitative scoring system to compare methods, or to provide precise justifications for choosing one approach to studying bioavailability over another. Rather, they are intended to help understand that tradeoffs are always involved in choosing tools to evaluate any bioavailability question, and to provide some general guidance about what the broadest tradeoffs might be.
The following sections describe and evaluate tools and techniques, many of which remain the state of the science for risk assessment. For each method we
explain the technique and why it is useful for measuring an aspect of bioavailability, weigh advantages and disadvantages with regard to evaluating bioavailability processes, and when possible evaluate performance in terms of reproducibility, repeatability, multi-lab calibration, and other factors. Because of the varied status of each tool (see Table 4-1), the evaluations are not equivalent in that not all of the same information is provided for each tool. In addition, some sections focus on an individual technique (e.g., X-ray spectroscopy), while others cover an entire approach (e.g., sediment bioassays). Thus, the details of each discussion, which focus on strengths and weaknesses, necessarily vary throughout. For example, in some cases methodological problems will be highlighted, while for other tools their potential for practical application will be assessed. In all cases, references are provided to direct the reader to further information about any specific test. Finally, to increase the utility and uniqueness of the tables, not all of the information presented in Tables 4-1 and 4-2 are repeated in the following discussions. Points to consider when choosing tests specifically for use in human health and ecological risk assessment, including criteria for validation, are presented at the end of the chapter.
Although it is difficult to encompass all the methods used to evaluate bioavailability processes, some relatively safe generalizations are possible from the discussions that follow.
-
Although approaches to measuring bioavailability can be quantitative or qualitative, ultimately those approaches that allow quantitative estimation of bioavailability are the most important.
-
Mechanistic approaches (that unambiguously determine the form of a contaminant) have the greatest potential to ultimately result in useful approaches for defining bioavailability processes and narrowing uncertainties. But they are less applicable at present.
-
Regulatory and industry interests prefer simplified approaches that are operational (e.g., extractions), that provide shortcuts to estimate mechanistic processes (e.g., equilibrium partitioning), or that estimate bioavailability indirectly via complex responses (e.g., toxicity bioassays). Such approaches have important practical and scientific tradeoffs. Because some of these approaches lack explanatory capability, have narrow applicability, and have uncertain meaning, they should be employed cautiously in the current regulatory environment so as not to increase uncertainty or the degree to which actions seem arbitrary.
TECHNIQUES TO CHARACTERIZE INTERACTIONS AMONG PHASES
Contaminants occur in soils and sediments as a complex mixture of solid-phase chemical compounds associated with particles of varying size and morphology. Contaminant forms include discrete mineral phases, co-precipitated and
sorbed species associated with solid minerals or organic matter, complex compounds or associations among organic moieties, and dissolved species that may be complexed by a variety of organic and inorganic ligands. The occurrence and relative distribution of contaminants among various phases, and the physical relation between the phases and the soil or sediment, will control a contaminant’s dissolution properties and its bioavailability. The spatial heterogeneity of these complex mixtures in soil and sediment will be reflected in variable bioavailability of an element at a site.
This section discusses methods that can be used to investigate physicochemical forms of solids and contaminants and interactions among forms and phases as well as methods to obtain information on soil and sediment characteristics, which is often necessary to understand form. The methods include those that investigate both the microscale location and association of contaminants within solid matrices and the nature of the contaminant binding. The purpose of these tools is to provide a better mechanistic understanding of the chemical release portion of bioavailability and for interpreting differences in bioavailability of contaminants residing in different environmental matrices.
Physical and Chemical Characterization of the Solid
Although basic solid parameters are probably not sufficient to understand bioavailability processes, they provide critical ancillary information. In the absence of this information, more direct tests of bioavailability are difficult to interpret, making generalizations to other places, circumstances, or times problematic. Over the long term, knowledge of contaminant behavior in the field, combined with comparable basic characterization data of solids from many study sites, will improve our understanding of the factors that control bioavailability. This section describes physical and chemical analyses that can be useful as a routine part of evaluating bioavailability at contaminated sites.
Basic Characterization of Soil or Sediment
The measurements described below provide important contextual information about the solid matrix in which contaminants occur, and they are inexpensive and routinely conducted. Some of the measurements can be used as screening tools to make simplistic estimates of contaminant availability. Methods for conducting these analyses are described in Page (1965), Sparks (1996), and Meyers (1998).
Organic Carbon and Organic Matter Content. Organic carbon content (measured as foc) provides a simple index of solid reactivity to hydrophobic organic contaminants and some metals and is, therefore, an indicator of potentially reduced bioavailability. Organic matter content (fom) provides similar information, except that the entire mass of organic matter including hydrogen, oxy-
gen, sulfur, and nitrogen is determined. Organic carbon content can be used to obtain a reasonably accurate estimate of the sorption distribution coefficient for hydrophobic organic compounds in most modern soil or sediment samples that have ≥ 0.5 percent carbon (wt/wt) (Allen-King et al., 2002). However, the estimate may be poor for some subsurface samples (e.g., those with lower carbon contents or when contaminant concentrations are low). A variety of methods are available to determine foc or fom in a sample. Methods to measure fom typically do not include black carbon, while methods to measure foc that rely on high temperature combustion (see Heron et al., 1997) will include all forms of non-carbonate carbon, from humic materials through black carbon.
Particle Size. This measurement provides a crude indication of the grain or particle surface area and can be used to estimate soil or sediment permeability and to better understand chemical release from the solid. Particle size provides screening information on the rate and magnitude of contaminant desorption, which can control bioavailability. If the desorption mechanism involves diffusion within a particle or grain (such as shown for lead in Figure 3-9), then smaller particle-size would result in a smaller diffusion distance and more rapid uptake and release rate compared to larger sizes. For example, Ball and Roberts (1991) found that the time needed for perchloroethylene to attain sorption equilibrium with large grains was much greater than for smaller grains from the same sandy aquifer. The difference was attributed to diffusion to sorption sites within the grains or particles. When the mechanism is primarily sorption to external grain surfaces, then surface area (per unit mass), and hence reactivity, are generally greater for smaller compared to larger particles. In this case, particle size measurements provide information directly related to the magnitude (but not the rate) of sorption. It should be noted that the magnitude and rate of contaminant desorption cannot be reliably estimated from particle size alone, because other characteristics of the particle and contaminant are important in controlling behavior. However, information on contaminant concentration and desorption rate by particle size may reveal the mechanisms that limit bioavailability on a site-specific basis and provide information that will improve long-term predictions.
Surface Area. Surface area (also frequently termed specific surface area or SSA) provides indirect information on the types of mineral surfaces present and on reactivity, especially for metal ions. Silicate minerals that do not have internal porosity typically have low SSA (≤ ~1 m2/g), while some clay minerals can have much greater surface areas (> ~100 m2/g) (Selker et al., 1999). SSA is frequently determined by developing N2 gas adsorption isotherms using the BrunauerEmmett-Teller method (Gregg and Sing, 1982). The isotherm shape can be used to interpret the micro- and meso-porosity connected to the external particle surface. Mercury porosimetry is another method that can be used to determine SSA and intragranular porosity, as described extensively by Gregg and Sing (1982).
Finally, ethylene glycol monoethyl ether vapor adsorption is frequently used to evaluate the surface area of clay mineral phases.
pH. Many contaminants, particularly cationic metals, tend to be more mobile (and thus bioavailable) in acidic soils. Furthermore, the low pH characteristic of some contaminated sites can cause dissolution of relatively high surface area iron oxyhydroxide grain coatings that in turn results in release of cationic species, including metal co-contaminants. Sorption of weak acids or bases to solids is also generally pH dependent. Neutral compounds tend to be least affected by system pH.
Cation Exchange Capacity. The cation exchange capacity (CEC) is the total charge excess of cations over anions for a soil. It is generally measured by procedures designed to saturate the exchangeable sites with particular “probe” cations (e.g., Mg+2) under controlled ionic strength and pH conditions and is, therefore, somewhat empirical. The CEC provides information on the reactive surface properties that are particularly relevant to sorption of cationic metals. A larger CEC (within the range typically observed for soils) indicates a relatively high content of high CEC mineral phases, such as for smectite or montmorillonitic clay, and is generally associated with greater cationic metal sorption.
Characterization of Carbonaceous Phases
Characterizing the type, chemical composition, and structure of carbonaceous materials can provide information about the extent of hydrophobic organic compound (HOC) sorption. As discussed in Chapter 3, substantial sorption of HOCs onto the more condensed forms of carbonaceous materials and black carbons (chars, soots, coals, and kerogens) may make them much less bioavailable than those associated with natural organic matter coatings on mineral solids. There are multiple techniques to identify forms of carbonaceous materials, including extraction and separation of fulvic and humic acid fractions, nuclear magnetic resonance spectroscopy, petrographic and elemental analysis and pyrolysis, and infrared absorbance.
Separation of Humic, Fulvic, and Humin Fractions. Humic, fulvic, and humin fractions of soil organic matter (SOM) are operationally defined by acid and base extractions (Swift, 1996). Determining these fractions in a soil sample can lead to a better understanding of contaminant sorption because each fraction has a different affinity for HOCs. For example, Njoroge et al. (1998) demonstrated the heterogeneity of soil organic matter with respect to sorption by determining its fulvic acid component. At this field site, it was shown that sorption could be adequately modeled by assuming that the sorbent was a mixture of two materials with different affinities for the HOCs. Differences in sorption magni-
tude and nonlinearity for humic and humin fractions from a peat soil have also been demonstrated (Chiou et al., 2000).
Nuclear Magnetic Resonance. Solid-state 13C nuclear magnetic resonance (NMR) with cross-polarization and magic angle spinning has been used to characterize the relative abundances of different functional groups within soil, and these characteristics have been related to sorption magnitude, desorption rate, and bioavailability. For example, NMR has been used to detect aromatic character (Xing, 1997) and more reduced and condensed organic matter (Lueking et al., 2000), both of which were correlated with greater sorption of polyaromatic hydrocarbons (PAHs). Because the results are qualitative and the technique requires a relatively strong magnet and long times (see Swift, 1996), NMR is useful primarily in research applications.
Petrography and Elemental Analysis. Several techniques to characterize carbonaceous materials have been used extensively as geochemical tools in oil and coal reservoir analysis, including coal petrography, extraction followed by elemental analysis, and pyrolysis techniques. These methods have recently been used to show correlations between HOC sorption and various properties of the carbonaceous material. They are most successful for materials that are relatively rich in carbon and resistant to acid treatment.
Petrographic methods are qualitative to semi-quantitative and are best used to identify the relative proportions of different phases. For example, recent studies have correlated the types of carbonaceous matter in sedimentary rocks or unconsolidated sediments to HOC sorption behavior (Kleineidam et al., 1999; Karapanagioti et al., 2000). Coaly particles had a significant effect on sorption even though they comprised a low proportion of the carbonaceous material.
Elemental analysis has been used to characterize differences in primarily hydrogen (H), oxygen (O), carbon, and nitrogen content, which indicate the degree of condensation and polarity of carbonaceous materials. The carbonaceous material H/O ratio has been correlated to sorption of chlorinated solvents within a series of samples representing a wide range of properties, from fulvic acid to hard coals and shales (Grathwohl, 1990), and for a series of kerogen-containing subsurface sediments (Binger et al., 1999). Because the method is relatively simple, commercially available, and quantitative, it shows promise. However, to date the relationships between the elemental composition of soil carbonaceous matter and sorption magnitude have been tested for only a few combinations of soils/sediments and contaminant compounds.
Infrared Absorbance. Radiation in the infrared (IR) range corresponds to the stretching and bending vibrational frequencies of covalent bonds. Thus, the IR absorbance spectrum (usually measured by Fourier transform IR instruments, FTIR) can provide structural information about an organic molecule. Like NMR,

Scanning electron micrograph of goethite laths developed around a ferrihydrite substrate.
FTIR provides qualitative information regarding complex bonding in natural carbonaceous matter. It has been used primarily to characterize humic substances in soils. Because it responds to bulk sample properties, it is not likely to be well suited to low foc materials or for discerning subtle differences between samples. FTIR microspectroscopy makes it possible to analyze IR absorbance at small spatial scales, such as the sub-grain scale in soil or sediment. This research grade tool has been used to characterize the chemical properties of surfaces in concert with contaminant analysis to better understand the nature of contaminant binding, as further described in Box 4-1.
Probing Contaminants within the Solid Phase
A variety of spectroscopic techniques are available to evaluate the chemical and mineralogical properties of contaminants associated with soils and sediments. Spectroscopy can also provide information about the solid phase itself and thus can be used to complement the techniques described in the preceding discussion. Although some of these spectroscopic methods are commonplace, most are research grade tools.
|
BOX 4-1 Complementary Sediment Characterization and Contaminant Distribution Facilitates an Understanding of Bioavailability Detailed physical and chemical characterization of the solid phase provides information complementary to contaminant concentration and release data regarding mechanisms controlling bioavailability. Recent studies have determined the associations between PAHs and particles of a harbor sediment (Ghosh et al., 2000a; Talley et al., 2002). In these studies, seven different sediment fractions enhanced in particular particle types were obtained by a combination of density and particle-size separations. As shown in Figure 4-1, PAH analysis showed that more than half of the total extracted 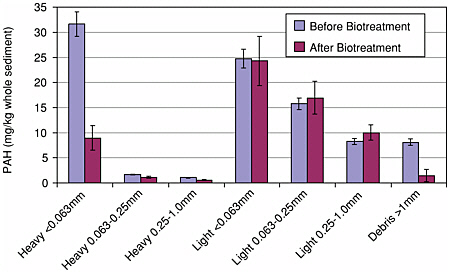 FIGURE 4-1 PAH distribution in sediment size and density fractions before and after bioslurry treatment showing loss of PAHs from the clay–silt fraction (< 0.063 mm) and no change in PAHs from the light coal/wood derived fractions. SOURCE: Reprinted, with permission, from Talley et al. (2002). © (2002) American Chemical Society. |
X-ray Diffraction and Scanning Electron Microscopy
X-ray diffraction (XRD) and scanning electron microscopy (SEM), with associated energy dispersive spectroscopy, have been used extensively to characterize solids. X-ray diffraction defines the crystalline structure of the solids, while scanning electron microscopy provides information on particle size and morphology along with elemental composition, all of which are pertinent to evaluating contaminant bioavailability, particularly for metals. Thus, for example, scanning electron microscopy and electron microprobe analyses can establish not only the chemical composition, size, and morphology of individual soil particles but also the distribution of a particular element (for example, lead) within a soil particle. These data can be used to estimate or model the solubility, and hence the bioavailability, of the mineral assemblage in a particular soil. Indeed, analysis of
|
PAH mass was associated with the “light” (low-density) sediment fractions, although these fractions comprised less than 5 percent of the total sediment by weight. Approximately one third of the total PAH mass was associated with the “heavy” clay–silt size (< 0.063 mm mineral) fraction, and about one tenth was associated with the coarse material (>1 mm). Hence, PAH concentrations on light particles were approximately two orders of magnitude greater than on the heavy particles. Characterization of the particles provided complementary insights into the nature of PAH binding. The particles in the light fractions were identified by petrographic analysis as primarily coal and coal-derived, and were thought to originate from historic coal shipping and processing operations in the harbor. The light fractions also contained particles of wood and vegetative debris. The more dense fractions were composed primarily of silicate minerals. An FTIR analysis of the heavy clay–silt size fraction revealed that its associated organic matter is more polar than the carbonaceous matter in the light fractions. Consistent with other work (Grathwohl, 1990; Karapanagioti et al., 2000), these results demonstrate that the sorption capacity for HOCs of more condensed coal-derived carbonaceous matter is much greater than the sorption capacity of the more polar organic matter coating silicate grains. Based on these results and the results of sequential Tenax extractions, the authors posited that PAHs sorbed to the coal-derived materials were more strongly bound than PAHs sorbed to the heavy clay–silt size fraction, and so less bioavailable. To test the hypothesis, the authors subjected the sediment to bioslurry treatment followed by PAH analysis by density and particle size separation. The findings supported the hypothesis (Figure 4-1, dark bars). The total PAH concentration in the heavy clay–silt size fraction diminished by about 75 percent with bioslurry treatment, while there were no significant changes in the total PAH concentrations in any of the three fractions dominated by coal-derived material. This study demonstrates that using techniques to determine the composition of the solid can provide information complementary to the identification of contaminant associations and provide insights on the mechanisms controlling bioavailability. |
lead-bearing soils has indicated that the chemical forms and sizes of lead-bearing particles control the oral bioavailability of lead (Ruby et al., 1999), and the same appears to be true for arsenic in soils. U.S. Environmental Protection Agency (EPA) Region 8 has participated in the development of a protocol for site-specific assessment of lead and arsenic mineralogy in soil using electron microprobe analysis (CDM, 1994). However, given the complexity of lead and arsenic associations within soil solids, and the multitude of reactions that may lead to their dissociation, data regarding contaminant phases and size alone have not been deemed adequate to estimate bioavailability. X-ray diffraction has two primary limitations: (1) only crystalline solids are detected and (2) detection requires greater than 1 percent of the specific phase. The drawback with scanning electron microscopy is simply that particle morphology and shape do not translate directly into a relationship with bioavailability.
X-ray Absorption Spectroscopy
X-ray absorption spectroscopy (XAS) has recently proven to be a powerful means for obtaining the speciation and structure of elements (such as metals) present in complex media. It has a number of advantages for studying natural materials that include element specificity, the ability to probe local chemical and structural states of an element, and the ability to analyze materials in situ (meaning that a natural water, soil, or sediment sample can be placed directly in the spectrometer without further alteration). XAS probes the local chemistry and structure of a single element throughout a sample, revealing a “view” of the element’s electronic structure and the atoms that coordinate it, as illustrated in Figure 4-2. The oxidation state, types of nearest neighbors, coordination number, bond distances, and orbital symmetries of the x-ray-absorbing element can be accurately determined in an array of media (Eisenberger and Lengeler, 1980). XAS is not useful for detecting trace quantities of a contaminant and is not ideal for most organic contaminants. It is, however, extremely useful for identifying the stability of metal contaminants residing in the solid phase of soils and sediments at part per million levels. Few techniques provide greater information on the chemical environment of a metal within natural materials.
X-ray absorption spectroscopy can be broken into two main subsets—X-ray absorption near edge structure (XANES) and extended X-ray absorption fine
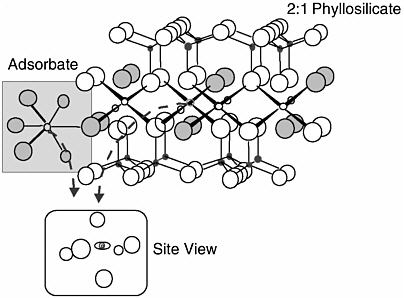
FIGURE 4-2 An illustration of the hypothetical view obtained by X-ray absorption spectroscopy. The eye in the “site view” represents the atom around which the image is centered, which can be either an adsorbate or a mineral atom. The image reveals the configuration of that atom’s nearest neighbors.
structure (EXAFS) spectroscopies. XANES spectroscopy provides an opportunity to ascertain the oxidation state or speciation of elements in soils or sediments—until recently a near impossible task. To date, the oxidation states of uranium, manganese, chromium, iron, selenium and other elements have been elucidated with XANES spectroscopy (see Table 4-3). EXAFS spectroscopy, in contrast, probes the local structure of an element within solids, providing detailed information on its bonding environment.
To date, most studies utilizing EXAFS spectroscopy have been conducted on rather simple, homogeneous systems, such as to elucidate the structure of ions on surfaces common to soils or sediments. This information is essential for determining the stability of a sorbate, which dictates the potential for desorption. Furthermore, such information is needed to develop accurate mechanistic models that can predict the fate of metal ions. Whether a metal ion binds electrostatically or chemically can be discerned with EXAFS. Electrostatic retention was observed for lead on corundum (Bargar et al., 1996), while chemical complexes of numerous ions have been noted on many surfaces (for example, Scheidegger et al., 1997). In the case of chemical binding, the coordination environment (i.e., the interatomic distances and coordination number) of the surface complex can be obtained.
To provide the intensity necessary for performing XAS measurements in a reasonable time period and in solvated systems, high intensity X-ray sources—
TABLE 4-3 Selected XAS Studies of Metal Contaminants within Soils and Sediments
|
Subject |
Reference |
|
Selenite and selenate on goethite |
Hayes et al. (1987) |
|
Np(V) retention on goethite |
Combes et al. (1992) |
|
Cr oxidation states on ferrous hydroxides |
Bidoglio et al. (1993) |
|
Speciation of U in soils and sediments |
Bertsch et al. (1994) |
|
S speciation in marine sediments |
Vairavamurthy et al. (1994) |
|
Se speciation in sediments |
Tokunaga et al. (1996, 1998) |
|
Pb complexes and α-alumina |
Bargar et al. (1996) |
|
Arsenate sorption on ferrihydrite |
Waychunas et al. (1996) |
|
U in soils |
Duff et al. (1997) |
|
Mn forms in lake waters and sediments |
Friedl et al. (1997) |
|
Precipitation of selenocyanate in water |
Manceau and Gallup (1997) |
|
Ni complexes and precipitates on phyllosilicates |
Scheidegger et al. (1997) |
|
Cr in soils |
Szulczewski et al. (1997) |
|
Cu and Pb on soil humic substances |
Xia et al. (1997) |
|
Pb on goethite and alumina |
Bargar et al. (1998) |
|
Zn, Cd, Pb in river sediments |
O’Day et al. (1998) |
|
Zn and Pb in Penicillium |
Sarret et al. (1998) |
|
Cr in soil columns |
Jardine et al. (1999) |
|
Pb in mine tailing |
Ostergren et al. (1999) |
|
Mn in automobile exhaust particulates |
Ressler et al. (2000) |
generally found only at synchrotron facilities—are necessary. High intensity synchrotron X-ray sources permit in situ investigations of most elements (those heavier than boron). Facilities are being developed to analyze lighter elements of biological significance, such as carbon and nitrogen in thin films of water. Because synchrotron X-ray sources are required, it is unlikely that this technique will be a mainstream analytical tool but will rather serve as a means for describing high priority samples or for calibrating more accessible methods. Box 4-2 describes the use of XAS in conjunction with other tests to help determine bioavailability of metals in sediment.
|
BOX 4-2 X-ray Absorption Spectroscopy Provides Molecular Understanding of Metal Bioavailability in Sediment O’Day et al. (2000) assessed metal speciation and bioavailability for contaminated estuarine sediments obtained from the East Outfall Site of the Seaplane Lagoon, at the former Naval Air Station Alameda, located on an island in San Francisco Bay. The researchers assessed a measure of bioavailability proposed by EPA for five metals (cadmium, copper, lead, nickel, and zinc) based on comparison of simultaneously extracted metals (SEM) to acid volatile sulfide (AVS) (see Chapter 2). If ΣSEM/AVS > 1, there is potential bioavailability because of insufficient FeS(s) to precipitate the five toxic metals. Toxicity of lagoon sediment to sand dollar embryos and adult amphipods was compared to SEM and AVS measurements and to the speciation and local molecular bonding of metals in sediment as determined by synchrotron radiation X-ray absorption spectroscopy. The results showed that assumptions about ΣSEM/AVS were not valid for this study. Of six metals studied, only cadmium was present in sediment exclusively as a sulfide phase; chromium and lead were coordinated with oxygen. Toxicity tests with amphipods and invertebrate embryos also did not support ΣSEM/AVS predictions. In surface sediments, this ratio was between 2.7 and 5.25, yet the sediment was nontoxic, while sediments from 30-cm depth gave 100 percent toxicity despite ΣSEM/AVS = 0.54. Toxicity may have been due to either high ammonia or low dissolved oxygen. There was no evidence that FeS(s) was the primary contributor to AVS, as assumed in the AVS method. XAS showed that the metal contaminants were present in reduced sediment as both sulfide and oxide solid phases. Thus, the assumption that metal and iron monosulfides control the partitioning of toxic metals was not substantiated (except for cadmium and to some extent for zinc). For chromium and lead, and possibly for copper and nickel, pore-water concentrations were dependent on sorption and precipitation processes associated with clays, carbonates, and/or oxyhydroxide minerals. This study demonstrated the potential for using multiple bioavailability tests to gain mechanistic understanding of bioavailability processes. The toxicity tests and X-ray absorption spectroscopy did not fully support ΣSEM/AVS predictions, which should warn against using this ratio to infer mechanisms. Spectroscopic techniques can verify contaminant speciation and thus substantiate proposed standard sediment tests to provide a molecular basis for interpretation and extrapolation. |
Laser Desorption and Laser Ionization Mass Spectrometry
In general, spectroscopic assessments of solids provide information on the functional group structure of organic material and the associations of atoms in an organic matrix. However, to date there is a lack of methods that provide direct identification of organic contaminant molecules and their specific locations in soils or sediments. A new technique—microprobe laser desorption/laser ionization mass spectrometry (μL2MS)—offers the opportunity to determine where exactly on solid surfaces organic contaminants reside. μL2MS involves desorption of constituent molecules on a particle using a pulsed IR laser beam followed by selective ionization of the desorbed molecules with a pulsed, tunable ultraviolet laser. The resulting ions are then extracted into a reflectron time-of-flight mass spectrometer. The PAH detection limit of the μL2MS instrument is estimated to be in the sub-attomole range. Depth of penetration of the desorption laser is approximately 0.5–1.0 microns based on test results with PAH embedded in thin resin sections. Currently the resolution of the instrument is a circular spot 40 microns in diameter with the potential to be much smaller, in the range of 10 microns.
This method has been used to measure PAHs on field soils and sediments to determine their relative distribution and locations (Gillette et al., 1999). Along with particle sectioning procedures, it can assess the precise distribution of contaminants within sectioned particles (Ghosh et al., 2000b). Such information allows better understanding of microscale sorption mechanisms and can be used with other measurements to assess how organic contaminant locations and sorbent interactions affect bioavailability. This instrument is a unique research tool and is not available for regular screening of environmental samples.
Secondary Ion Mass Spectrometry
Direct surface analysis of environmental matrices for organic and inorganic contaminants is possible using secondary ion mass spectrometry (SIMS) methods. SIMS works by bombarding a specimen with either an ion or molecular beam; surface layers are then “bumped” off the surface and their speciation is determined by mass spectrometry.
For inorganic analysis, SIMS has principally been used to determine the distribution of elements on surfaces of soils or sediments (e.g., Eick and Fendorf, 1998; O’Day et al., 2000). SIMS has also shown utility for investigating organic contaminants when a molecular beam is used. For example, Ingram et al. (1997) used SIMS to analyze 16 pesticide residues on the surfaces of soil, leaves, grass, and stainless steel. Typical spot sizes for SIMS analysis are 3 to 6 mm2, and minimum detection limits range from 0.03 monolayers (2100 ppm) for tributyl phosphate to 0.005 monolayers (6 ppm) for the pesticide paraquat (Ingram et al., 1996, 1997).
One of the greatest benefits of SIMS is the low detection limit, at least relative to other solid-phase techniques. Generally one can detect species in the range of 10–5 to 10–6 mg contaminant per kg of solid. However, samples must be subjected to a high vacuum environment. Due to the relatively large spot size, SIMS may not be widely applicable to determine sorption mechanisms, but may be a valuable technique to determine surface concentrations on different environmental matrices.
Nuclear Magnetic Resonance
Nuclear magnetic resonance (NMR) gives information about the number and nature of the immediate chemical environment of each type of a target atom, as described earlier in this section. In addition to characterizing organic matter itself, NMR has been used to investigate the binding of organic and inorganic contaminants to soils and sediments. Weissmahr et al. (1997) studied the specific sorption of nitroaromatic compounds including trinitrotoluene, nitrobenzenes, and herbicides onto clay minerals with NMR. 15N-NMR studies also provided direct evidence of covalent binding of aromatic amines to humic substances (Thorn et al., 1996). Solid-state 15N-NMR studies of humic acids extracted from 15N-2,4,6-trinitrotoluene show that the explosive is reduced to aromatic amines, and some of the products are covalently bound to the natural soil organic matter (Achtnich et al., 1999; Knicker et al., 1999). Each of these cases is notable because covalently bound contaminant residues may not be bioavailable. Interactions between PAH molecules and aromatic structures within coals have also been studied using NMR techniques (Sakurovs, 1998). NMR has been used to better understand the binding of inorganic contaminants such as cadmium (Sharps et al., 1993; Otto et al., 2001), aluminum (Casey et al., 1998) and vanadium (Lu et al., 1998) to soils and sediments.
The greatest limitation of NMR is that the nuclei of the target contaminant must have unpaired spin-states to be active. Additionally, the material must have limited quantities of interfering species; iron is a noted problem with NMR and restricts its use on natural materials. Commonly studied nuclei of environmental interest include hydrogen, carbon, cesium, phosphorus, fluorine, and aluminum.
Electron Paramagnetic Resonance
Electron paramagnetic resonance (EPR) spectroscopy has been used extensively for the study of selected elements and molecules. In contrast to NMR, EPR probes the chemical nature of a species through electron spin interactions. For an EPR signal to be produced, the species must have an unpaired electron spin state. By definition, all free radicals fulfill this requirement, as do inorganic contaminants such as Mn(II), Cu(II), and Cr(III). EPR has been used to decipher the chemical state of elements such as manganese and copper within soils and sedi-
ments (McBride, 1982; McBride et al., 1984; Bleam and McBride, 1986). In fact, EPR provided some of the first details on the chemical interactions of transition element contaminants with clay minerals. More recently, EPR has been employed to provide direct information about the molecular-scale environment of xenobiotics in natural porous media (Dumestre et al., 2000). A virtue of EPR spectroscopy is that samples are easily prepared; soil or sediment suspensions can be placed directly in an EPR glass tube and inserted into the spectrometer. The main drawback is that studies are restricted to EPR-active compounds, as mentioned above. Also, a number of interfering species may reside within natural material.
X-ray Photoelectron Spectroscopy
X-ray photoelectron spectroscopy involves bombarding a solid with x-rays of fixed energy and then resolving the kinetic energy of the ejected photoelectrons to provide information on the solid’s oxidation state and bonding (chemical) environment. XPS is useful only for solids (and adsorbed elements) and has been applied to soils and sediments for at least 30 years, albeit not routinely. Because XPS is rich in information and useful for elements that may reside in different oxidation states, it has been used extensively to characterize surfaces of sulfur and manganese solids (Junta-Rosso and Hochella, 1994; Nesbitt et al., 1998a,b) and for defining the chemical state of arsenic on the surface of soils and sediments (Soma et al., 1994). Unfortunately, there are two distinct drawbacks of XPS. The first is the need for a high vacuum environment, which may potentially distort an environmental sample. The second (and more problematic) is the poor detection limit. A specific element of interest generally needs to be present at concentrations greater than 1 percent of the solid phase.
Summary
Mechanistic understanding of physicochemical phenomena controlling bioavailability processes requires knowledge of the geochemical compartments that contain the contaminant, the forms of the contaminant, and interactions of the contaminant within the compartment. New instruments are helping to develop this understanding. For example, NMR and microscale surface mass spectrometric and microscale infrared spectroscopic methods are capable of describing the occurrence and role of black carbon that may serve as an especially strong sorbent for organic contaminants. X-ray absorption near-edge structure and X-ray absorption fine structure spectroscopy can discern the distribution and bonding of metals in solids. Thus, new spectrometric and spectroscopic methods can identify the locations of specific organic compounds in natural materials, while X-ray absorption spectroscopy can provide data on element mineralogy, which is useful in modeling the solubility of mineral assemblages. Owing to the sophisticated,
specific nature of the instruments needed to address these questions, most of these methods will remain research tools. However, detailed examination of selected samples advances mechanistic understanding and thereby furthers the development of validated conceptual models for describing the chemical and kinetic factors controlling contaminant release, transport, and exposure.
PHYSICAL/CHEMICAL EXTRACTION TECHNIQUES FOR MEASURING BIOAVAILABILITY
A wide variety of extraction tests have been proposed for estimating the bioavailability of organic and inorganic compounds to humans and ecological receptors. The tests involve chemical extraction for metal contaminants and extraction using organic solvents or solid phase adsorbents for organic contaminants. These techniques attempt to provide a site-specific measure of the bioavailable fraction of a contaminant as opposed to the total extractable contaminant based on a rigorous extraction procedure, and they are meant to be simple and reliable. For human exposures, these tests have generally been physiologically based (i.e., relying on knowledge of the mechanism by which the chemical would become solubilized and available for absorption). Extraction tests are generally not considered valid until they have been shown to correlate with an inherently biological measure of bioavailability. The fact that many have not yet been validated reflects the difficulty and expense of measuring the bioavailability of xenobiotics in humans, ecological receptors, or an appropriate surrogate.
Extraction Tests for Inorganic Contaminants in Soils
Extraction tests for inorganics in soils have long been used, particularly for agricultural applications. Thus, most of the tests discussed below were initially developed to mimic plant uptake of metals so that plant tissue analysis would not be needed to determine a soil’s ability to provide nutrients. These tests were designed to be easily reproducible, rapid, and relatively inexpensive (O’Conner, 1988). Soil tests were initially developed to predict nutrient deficiencies in soil, and they were calibrated with plant response across different plant species and soil types. In general, it has been possible to determine critical extract levels for certain elements and crops within soil series, but not across all soil series (e.g., Cox, 1968; Lindsay and Norvell, 1978). Extraction methods will undoubtedly need to vary by soil type.
Because the vast majority of extractions were developed to predict metal deficiencies, they tend to be fairly aggressive in order to mimic plant behavior. Traditional extracts, which vary with soil type, generally contain organic chelates and/or acids to solubilize labile pools of soil nutrients. For example, to test for phytoavailable zinc, diethylenetriaminepentacetic acid (DTPA or DTPA-AB) is used as an extract in neutral to calcareous soils, the Mehlich-I or III method is
used in acidic southeastern soils, and the dilute hydrochloric acid method is used for neutral and acidic soils in the north central United States (Reed and Martens, 1996). Predicting plant uptake of elements that are present at potentially phytotoxic concentrations requires a different approach, because plants are generally not aggressively manipulating the rhizosphere to solubilize these elements. Several passive extracts have been developed to predict plant behavior under these conditions. Figure 4-3 shows a conceptual diagram of four extraction strategies for inorganic compounds, examples of which are given below.
Passive Approaches
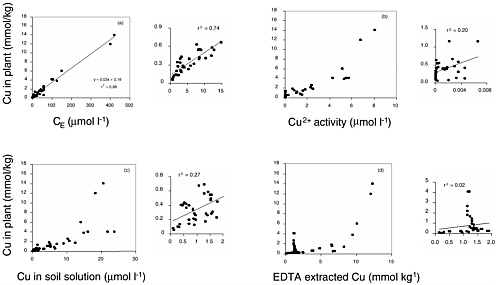
Because plant uptake of metals from soils occurs only via soil solution, measurement of contaminant concentration in soil solution gives an instantaneous view of the bioavailable fraction of the contaminant. Passive approaches, including passive extractions, pore water measurements, and some exchange resins, evaluate the concentration of contaminants that are present in soil solution or are readily soluble (that is, the portion held electrostatically on soil exchange sites). Metals in soil solution will be present as hydrated ions, ion pairs, chelated complexes, and complexed on colloidal material (Helmke, 1999). The most specific approach is to measure the concentration of free ions in solution. Indeed, there are indications that only the free ionic species of an element in soil solution is accessible to plants (Parker et al., 1995). Although there are cases demonstrating the uptake of metals present as ion pairs or chelated complexes, this is likely to be much less significant than uptake of free ions (Bell et al., 1991; Smolders and McLaughlin, 1996). Because these approaches do not involve altering the solid phase, they tend to be useful across a wide range of soil series.
Passive Extractions. Of the passive extracts, water and neutral salt extracts are the most widely used, simplest, and best correlated with plant uptake. A range of neutral salt extracts have been used including Ca(NO3)2, CaCl2, SrNO3, NaNO3, NH4NO3, and MgCl2 (McLaughlin et al., 2000). The technique involves collecting soils, adding either water or a neutral salt solution to the soil, shaking, and filtering. The filtrate is generally taken to be representative of the soil solution and readily soluble fraction, and unless otherwise specified, is analyzed via either atomic adsorption or inductively coupled plasma spectroscopy.
Although these extracts are very straightforward, there are some minor complications. For example, when water is used as the extractant, the normal salt concentrations and ionic strength of the soil solution will be diluted, and this will effect the partitioning of metals between exchange sites and solution. In a similar vein, the use of a neutral salt extract may affect changes in metal partitioning that would not otherwise occur under natural conditions—the extent of which varies depending on the salt chosen.
In spite of these complications, studies have generally shown that neutral salt
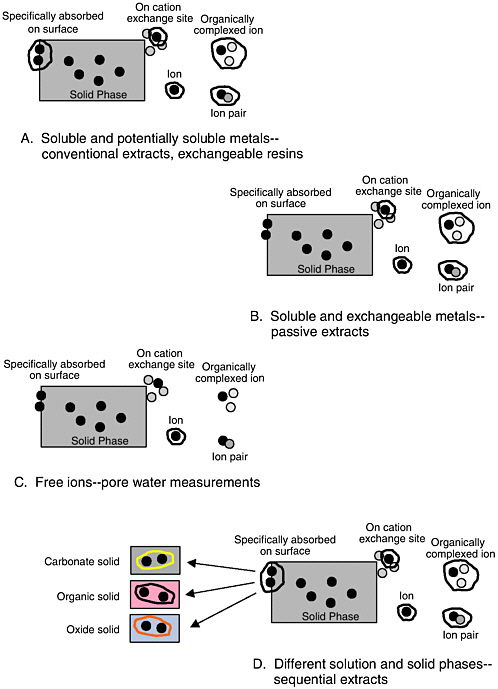
FIGURE 4-3 Different strategies for extraction tests to remove inorganics from soil and sediment. Conventional extracts and exchangeable resins (A) attempt to quantify all metals that are soluble or have the potential to be available. Less aggressive extracts (B) such as dilute salt extracts are directed towards the soluble or exchangeable fraction. Methods have also been developed to measure the concentration of ionic species in solution (C). Finally, sequential extraction methods (D) attempt to differentiate between solution and different forms of precipitated ions in soils and sediments.
extracts are more predictive of plant metal concentration than total metals in contaminated soils (Symeonides and McRae, 1977; Hani, 1996; Zhang et al., 2001). Lebourg et al. (1996) reviewed 20 years of research comparing soil extracts to plant uptake of cadmium, chromium, copper, lead, nickel, and zinc and concluded that unbuffered salt solutions were the most appropriate way to estimate the transfer potential of these elements from soil to plant, as well as to define guide values for risk assessment. Sauerbeck and Styperek (1984) reported r2 values between 0.66 and 0.8 for the correlation between CaCl2-extractable soil cadmium and plant cadmium for five crops grown in pot studies using three different soils. Correlation of plant uptake with total soil cadmium was much lower (between 0.01 and 0.22). These extracts show the best correlation over a wide gradient of contamination. They are much less effective at low contamination levels when soil solution is not the primary contaminant source for plants.
Passive extracts can also be used to predict metal bioavailability to other soil organisms. Janssen et al. (1997) found that earthworm bioaccumulation of arsenic, cadmium, copper, and lead was correlated with metal in CaCl2 extracts from contaminated soils (although the correlations were not particularly strong— r2 = 0.39 for CaCl2-extractable arsenic, compared to a correlation of earthworm bioaccumulation with total arsenic concentration of 0.27). Conder et al. (2001) similarly reported that reductions in Ca(NO3)2-extractable zinc in a smelter contaminated soil were correlated with reductions in earthworm mortality.
Pore Water Measurements. Where uptake of the free metal ion from the aqueous phase is the dominant exposure pathway (as it is for metal uptake into plants—see Chapter 3), pore water measurements can be valuable. Soil solution can be directly measured by centrifuging moist soil or through the use of soil solution samplers. Centrifuging soils requires large volumes of soil to generate sufficient solution for analysis, and thus is not practical for many applications. Soil solution samplers have been recently developed and show promise. These are robust enough for use in pot studies and have also been used in field situations with excellent correlation to plant tissue concentration, although their detection limits can be poor (Doberman et al., 1994; Knight et al., 1998; Farley and Fitter, 1999; Zhang et al., 2001).
Ion specific electrodes are straightforward to use and are relatively rapid and inexpensive. However, for many elements, the electrodes are prone to interference and do not have sufficient sensitivity to be useful for environmental samples. At the present time, copper is the only element that has a sufficiently low detection limit in combination with a lack of interference to be viable for environmental samples. Solution activities of Cu2+ have been measured with a copper electrode and compared to plant uptake for three plant species (Sauvé et al., 1996).
Anodic stripping voltammetry (ASV) has been used to operationally define free ion concentrations for a range of elements, including copper, cadmium, lead, nickel, selenium, and zinc, with detection limits of approximately 10–9–10–10 M

Plant growth response, as measured in these pot studies, is often used to validate extractions tests for inorganics in soil.
(Shuman, 1996; McBride, 1998; Sauvé et al., 1998). The assumption underlying this method is that only the ionic species from easily disassociated ion pairs or very weak organic complexes can be concentrated on the electrode. ASV has been used to correlate the free ion concentration of cadmium, copper, lead, and zinc in soil solution to a range of soil parameters, including pH, total organic carbon, and total metal (del Castilho et al., 1993; Sauvé et al., 1997, 1998, 2000). Unfortunately, attempts to correlate ASV results to plant uptake are absent from the literature. Validation with plant uptake in field studies will be required before the value of this method can be assessed.
Exchange Resins. Exchange resins have been used extensively to quantify free ion activities, solution fractions, and labile pool concentrations of metals in soils. These different fractions are assessed by varying the particular resin used, the ratio of resin to soil solution, and the equilibrium time. The basic procedure involves circulating soil solution, or soil solution extracted with water or a dilute salt, through an acceptor solution that contains a resin that has been impregnated with a particular cation. The amount of metal exchanged onto the resin is then operationally defined as corresponding to a portion of the total present in soil solution. For detailed discussion of individual tests see Cox et al. (1984), Fitch and Helmke (1989), Jing and Logan (1991), del Castilho et al. (1993), Lee and Zhang (1993), and Holm et al. (1995).
Currently, there is no standardized technique for measuring the free ion in soil solution using resins, making interpretation problematic (Skogely and Doberman, 1996). In addition, most studies use different techniques to measure labile concentrations (e.g., different soil:solution ratios, different equilibrium times, and different types of resins). In some cases, extraction tests using exchange
resins have been shown to correlate well with plant uptake, as in the case of cadmium (Jing and Logan, 1991; Lee and Zheng, 1993). This technique has the potential to be robust across a range of soil series. With standardization, exchange resins could become a widely applicable tool to measure bioavailability.
One concern with passive extracts and pore water measurements is that the measured information (the instantaneously labile pool of contaminants) may not be sufficient to evaluate the potential for contaminants to become more available with time. Exchange resins have the potential to measure both the solution and potentially soluble fraction of total soil metals. Strong resins can be used to remove metals from soil solution, after which metals in the solid phase will replenish the solution concentration. If the time course of this replenishment can be quantified, it may indicate the fraction of total metal that will become bioavailable over time. Thus, such resins could be used to indicate both the immediate and potential bioavailability of a contaminant. A technique recently developed for this purpose involves the use of a diffusive gradient in thin films (DGT) (Zhang et al., 1998; Hooda et al., 1999). As discussed in Box 4-3, DGT has the potential to serve as a single resin type for a wide range of contaminants and soils. A new version of the resin technique, DGT was initially developed for use in sediments and water and has only recently been used for soils (Davison and Zhang, 1994; Zhang and Davison, 1995; Hooda et al., 1999). It has not been robustly tested, standard protocols have not been developed for its use, and it has not been validated across different levels of contamination or different soil series.
Conventional Extractions
More aggressive extractions have also been used to define the bioavailable fraction of total metals in contaminated soils. These extractions, including DTPA, Mehlich I, II and III, 0.1 M HCl, and EDTA (disodium ethylenediaminetetraacetate), were developed to predict nutrient deficiencies and so alter the solid phase of the soil as a plant might (Reed and Martens, 1996). They are not robust across soil series. These extractions correlate much more poorly with plant tissue concentrations than do soil solution-based extracts (e.g., Taylor et al., 1992; Brown et al., 1994; Pichtel and Salt, 1998), and thus are not recommended where potential contaminant toxicity is the primary concern.
Sequential Extractions
Passive extracts measure only the instantaneous bioavailable fraction of the contaminant and provide no information on the solid-bound contaminant. Thus, a number of sequential extracts have been developed to quantify the distribution of metals in various solid phases for both soils and sediments (Tessier et al., 1979; Emmerich et al., 1982; Quevauviller et al., 1993; Berti and Cunningham, 1997). Each successive treatment is more drastic in chemical action, or of a different
|
BOX 4-3 An Exchange Resin Technique for Measuring Metal Bioavailability in Soils: DGT DGT devices measure metal bioavailability by being placed directly on the surface of moist soil. The device has a filter that permits diffusion of ions through two gels, the second of which has a resin that absorbs ions, setting up a diffusion gradient. Thus, the resin is separated from the soil by an ion permeable membrane. By monitoring both changes in solution concentration and the amount of metal adsorbed on the resin, metal distribution and flux for particular soils can be evaluated. The method assumes that changes in solution concentration (Csoln) are resupplied from the labile particulate phase (CLP) at a resupply rate defined by the constant (k1) for a particular soil and contaminant (Zhang et al., 2001). Zhang et al. (2001) assessed the phytoavailable fraction of copper in 29 naturally copper-contaminated soils using DGT and other tools. Copper uptake by Lepidium heterophyllum Benth. (pepperwort) was compared to (1) free Cu2+ in soil solution measured by an ion specific electrode, (2) total copper in soil solution collected with a soil moisture sampler, (3) EDTA-extractable copper, and (4) the effective copper concentration (CuE) as measured using DGT (Figure 4-4). Copper measured by DGT was most closely correlated to plant uptake (r2 = 0.98), with total copper in the soil solution being the next best indicator (r2 = 0.85) (Davison et al., 2000). The performance of the DGT resin was superior to the others, particularly at the low end of copper uptake by plants. DGT is currently under development. Hooda et al. (1999) showed differences in available metal measured based on soil moisture, incubation time, and thin film thickness. While metal flux into the resin was linear in a soil:water slurry, it was not linear in a saturated soil. In addition, trace metal fluxes across different soil moisture concentra |
nature, than the previous one, such that each solid phase is defined as having different potential bioavailability (see Figure 4-3). Metals in soil solution or exchangeable metals are considered the most labile and bioavailable. Organically bound metals and inorganically bound metals are progressively less labile. The residual fraction extracted using strong acids is seen as unavailable (Sposito et al., 1982).
Sequential extraction has been employed for numerous purposes. For example, it was used to assess the differential uptake of metals by plants grown in biosolids-amended soils (Chang et al., 1984a; Sims and Kline, 1991; Berti and Jacobs, 1996; Sloan et al., 1997; Basta and Gradwohl, 2000). In some cases, these studies have been part of the larger goal of evaluating the success of in situ soil
amendments to stabilize metals (Berti and Cunningham, 1997). Changes in metal partitioning over time have also been evaluated with sequential extraction. Almas et al. (1999) added cadmium and zinc isotopes to naturally enriched soils, let them incubate for a year, and then used a sequential extraction procedure to evaluate changes in metal distribution. They observed an approximate 50 percent reduction in the mobile fraction with a migration of the isotopes to more inert fractions over time.
Although sequential extracts can reveal changes in how metals are bound to solids, these procedures may be more appropriate for qualitative rather than quantitative determination of metal partitioning. Both soils and sediments consist of heterogeneous layered aggregates; even with an extractant targeted for a spe-
cific phase, it is likely that some phases are leached progressively instead of fully by their selective extractant (Jenne, 1977). Redox-sensitive elements can be gratuitously reduced or oxidized during the procedure, leading to experimental artifacts (Gruebel et al., 1988). In addition, both reprecipitation and readsorption can occur as metals released from heterogeneous phases interact with soluble and insoluble components in the extract (Belzille et al., 1989; Apte and Bately, 1995; Ahnstrom and Parker, 1999; Bunzl et al., 1999). As Nirel and Morel (1990) emphasizes, sequential extraction procedures do not provide actual particulate speciation, and the conditions used during the extraction procedures (strong reagents and fast kinetics) are difficult to extrapolate to naturally occurring processes (weak reagents, slow kinetics).
Despite these concerns, efforts to standardize sequential extractions are under way, as evidenced by the Measurements and Testing Programme of the European Commission (Quevauviller, 1995) and the National Institute for Standards and Testing (Ho and Evans, 1997). Although changes in the operationally defined fractions of metals in soil may provide some indication of changes in bioavailability, at the current time sequential extractions are best used in comparative experiments. The qualitative nature of this information makes its use for development of regulations potentially problematic.
Regulatory Leaching Tests
Simple regulatory leaching tests, such as the Toxicity Characteristic Leaching Procedure (TCLP; EPA Method 1311) or the Synthetic Precipitation Leaching Procedure (SPLP; EPA Method 1312), have been used at times to determine the bioavailability of metals from soil. However, while these tests are useful for evaluating the mobility of contaminants in soils, they have minimal relevance for estimating biological absorption. The TCLP test involves leaching a soil sample in an acetic acid solution at a pH of 2.9 or 5.0 (depending on whether the waste is characterized as alkaline or non-alkaline), and was designed to simulate leaching in a landfill environment. The SPLP test involves leaching in a fluid that is pH 4.2 or 5.0 depending on whether the site in question is east or west of the Mississippi river, respectively, to simulate leaching in rainwater. Because neither of these tests bears a mechanistic resemblance to the processes that would control the uptake of contaminants in soil by ecological or human receptors, it is not reasonable to expect that they would be predictive of bioavailability, nor is their use for this purpose recommended. For example, TCLP tests for arsenic on 13 samples of soil and mine waste compared to the EPA Region 8 young swine model tests (for the same set of substrates) indicated no correlation between these two methods (Rodriquez et al., 1999). However, their use as a first level, commercially available test to evaluate mobility in soils may be appropriate for particular situations.
Extraction Tests for Inorganic Contaminants in Sediments
Extraction tests for inorganic contaminants have a parallel history in soil science and aquatic geochemistry, and most of the tests used for soils are applicable for sediments as well. Thus, for example, pore water measurements in sediments can be used to estimate bioavailability in those cases where the major exposure pathway involves metals in the aqueous phase. An example is tributyltin; dissolved forms of the compound that have leached directly from vessel hull paints are more available that solid-bound forms, such that regulatory thresholds for tributyltin in Puget Sound sediments have been based on correlation between interstitial water and invertebrate tissue concentrations (Michelsen et al., 1998). In other cases, however, more complex tools are required because of the aggregated and complex nature of multi-ligand particulate material with which metals interact. Chemical methods that would selectively extract specific forms of metals once seemed an attractive approach. Thus, batch or more commonly sequential extractions with appropriate reagents were devised for aquatic sediments (Jenne, 1977; Jenne and Luoma, 1977), following the theoretical constructs laid out for soils. As with soils, the goal of sequential extractions of sediments is to leach successive fractions of metal selectively from the sample; that is, at each step to extract a metal completely from a given phase while leaving more or less intact the same metal bound to other phases.
The extractants used fall into the typical classes of chemical behavior, including inert electrolytes, weak acids, reducing agents, complexing agents, and oxidizing agents (Campbell and Tessier, 1989). In general, the outcome for extractions of sediments has been much the same as for soils. A few approaches are somewhat selective, but most are not (see reviews by Campbell and Tessier, 1989; Luoma, 1989). Sequential or batch extractions are probably the least selective for cadmium, copper, and zinc, but may be more effective for establishing forms of mercury (Davis et al., 1997), selenium (Cutter, 1985) or chromium (EPA Method 7195, SW846) because of the unique chemistry of these elements and because analytical methods exist for differentiating oxidation states of these elements once they are extracted. Verifying the specificity of extractions has been difficult because few methods exist to demonstrate metal form in complex natural sediments other than extractions (i. e., comparative verification is difficult). X-ray adsorption spectroscopy, which can determine both oxidation state (XANES) and mineral form (EXAFS) of a range of elements, may at some future date be useful in this regard.
Many extractions of sediment purport to remove that fraction of metal that organisms remove from sediments, either by empirically reflecting sediment– water exchangeability or imitating digestive removal. Reviews of the numerous attempts to establish such correlations conclude that no one universal extractant procedure can closely define the availability of all metals (Luoma, 1989; Apte and Bately, 1995), although some successful correlations are found in certain
instances with specific trace elements. For example, extractions may improve understanding of available concentrations of a trace element by excluding the most recalcitrant and unavailable forms (for example with selenium—Schlekat and Luoma, 2000). Mimicry of digestion is also a possible avenue wherein extractants might offer some value in explaining uptake. Recent studies have used extractions with the digestive fluids of invertebrates (the “biomimetic” approach discussed below for humans) to successfully explain bioavailability of at least some metals (Mayer et al., 1996; Chen and Mayer, 1999). Extraction by weak hydrochloric acid seems to greatly improve predicted silver availability to bivalves (Luoma, 1996), although the mechanistic reasons are not clear. Nonetheless, as discussed in Box 1-2, simple correlations between extracted concentrations and bioaccumulation are rare when applied across diverse sediments.
Extraction Tests for Inorganics that Mimic Human Exposure
Extraction tests that are intended to predict the extent of oral bioavailability of inorganic elements in humans have been available for several decades and first appeared in the field of nutrition. In the late 1970s and 1980s, several research groups were developing in vitro extraction tests that simulated the function and chemistry of the human gastrointestinal tract to predict the amount of iron in food substances that would be bioavailable upon ingestion (Bezwoda et al., 1978; Miller and Schricker, 1982; Reddy et al., 1988). Several of these groups also attempted to “validate” their in vitro extraction tests against iron bioavailability results observed in swine. These tests were the forerunners of the extraction tests currently used to estimate the oral bioavailability of toxic metals in soil. To date, there has been extensive work to develop and validate an in vitro extraction test for lead, a moderate amount of work on arsenic and mercury, and a small amount of work on beryllium, cadmium, chromium, and manganese.
These in vitro extraction tests simulate dissolution in a fasting gastric environment because lead and cadmium, and most likely other inorganics as well, are more bioavailable under fasting than fed conditions (James et al., 1985; Maddaloni et al., 1998). The gastric phase may be followed by a small intestinal simulation of near neutral pH that contains various enzymes and acids (e.g., pancreatic enzymes and bile acids). (See Oomen et al., 2002, for a recent comparison of five different digestion models.) Obviously, such tests are only capable of simulating dissolution of metals from soil in the gastrointestinal tract (bioaccessibility) and do not simulate the process of absorption across the intestinal epithelium. Therefore, if absorption is the rate-limiting bioavailability process, rather than the rate or extent of dissolution from soil, then these tests will not be capable of predicting oral bioavailability. However, for lead and arsenic in soil, the extent of dissolution in the acidic stomach environment appears to be the determining factor for oral bioavailability, based on comparison to bioavailability
studies for lead in rats (Ruby et al., 1996) and swine (Medlin, 1997) and for arsenic in swine (Rodriguez et al., 1999).
In addition to predicting oral bioavailability, in vitro extraction tests have been used to examine the effects of gastrointestinal tract chemistry on the availability and chemistry of metals in soil. For example, the tests described above have been used to examine the effects of pH and gastrointestinal fluid composition on lead and arsenic bioaccessibility, as well as the effect of gastric fluid pH and chemistry on the rate and extent of hexavalent chromium reduction.
In vitro tests are often used to estimate exposure parameters for human health risk assessment, and because they are relatively new, some validation has been conducted to promote regulatory acceptance. This has involved comparison of the extraction test results to those from an in vivo model for a set of samples that is large enough (on the order of 10–20) to develop a statistically significant correlation. The cost of generating this amount of in vivo data is the primary limitation to the development and validation of this type of assessment tool. To date, only an in vitro extraction test for lead has received such validation (against the EPA Region 8 young swine model for lead bioavailability), while an extraction test for arsenic is in the process of validation (against the young swine model and a primate model). At this time, no in vitro to in vivo comparisons are available for beryllium, cadmium, chromium, or mercury in soil.
Extraction tests using real or synthetic sweat have been used to evaluate the fraction of chromium that might dissolve at the skin surface and become available for dermal absorption. Horowitz and Finley (1993) determined that 0.1 percent of Cr(VI) and 0.3 percent of total chromium in soil samples contaminated with chromite ore processing residue would be extracted by human sweat. Using similar soils (i.e., those containing chromite ore processing residue from the same site), Wainman et al. (1994) concluded that synthetic sweat could reliably be used to estimate dermal exposure to chromium in soil. Given the expense of measuring dermal Cr(VI) absorption using animal studies, or in vitro studies using human cadaver skin, it is possible that such extraction studies in real or synthetic sweat will receive further consideration in the future.
Extraction Tests for Organics
Analytical methods for measuring concentrations of organic chemicals in soil or sediment have historically entailed vigorous extraction with low polarity organic solvents to remove all or as much hydrophobic organic contaminant as possible. Common techniques include Soxhlet extraction, XAD extraction, and solid-phase extraction microcolumn. As one would expect, the bioavailability of contaminants in soil and sediments is overestimated by such analytical methods. For example, exposure estimates based on Soxhlet extracts of organic compounds do not accurately reflect the concentrations of compounds available for uptake, as
determined by the number of bacterial mutations in treated soil (Alexander and Alexander, 2000).
Several milder extraction tests have been developed that correlate with bioavailable organic compound concentrations in soil and sediment and may serve as surrogate assays for bioavailability. These chemical tests are being developed for use in site-specific assessment because they are generally less time-and resource-dependent than biological assessments. A limitation of most of the tests described in this section is that they are typically applied ex situ and may not be truly site specific because of biases inherent in sample collection and disruption.
Figure 4-5 shows a conceptual diagram of four extraction strategies, examples of which are given below. Extraction tests can be differentiated by (1) the need to make a slurry of the soil or sediment sample versus using the sample directly, (2) use of a semipermeable membrane, and (3) use of a liquid or solid
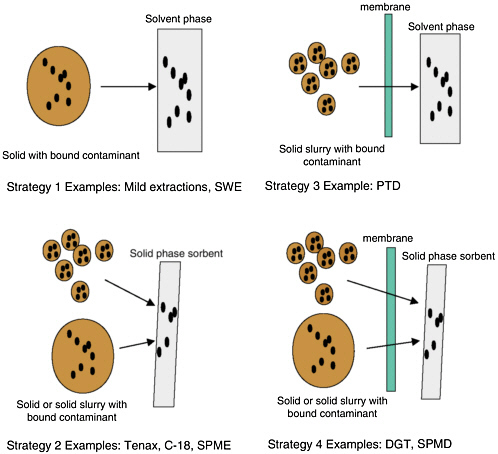
FIGURE 4-5 Four different strategies for extraction tests to remove organics from soil and sediment.
adsorbing phase. First, techniques that require a soil or sediment slurry measure contaminant concentration in the pore water of samples, and thus are an indication of aqueous phase concentrations in the natural environment. They cannot be used in situ. A semipermeable membrane allows one to differentiate between contaminants that have desorbed from the soil or sediment to the aqueous phase and those that are still bound to colloids. Techniques that do not use semipermeable membranes tend to measure higher contaminant concentrations than those that do. Finally, the adsorbing phase is typically something that might mimic a biological system (biomimetic). As discussed above for extraction tests for inorganics, techniques using solid-phase adsorbents (e.g., exchange resins) can measure both the instantaneous bioavailable contaminant fraction as well as the fraction potentially available over time (i.e., the rate of desorption).
As with inorganic contaminants, there is no ideal extraction strategy for organic contaminants bound to soils and sediment. Rather, the techniques must be chosen to reflect the conditions present at the site, particularly soil or sediment type and the feeding behavior and uptake mechanisms of the receptors. Ideally, experiments should be conducted within and between different soils and sediments (to determine the effects of the soil or sediment matrix on experimental results) and the tools should be validated by comparison to a bioassay.
Mild Extractions with Various Solvents
As with the passive extractions conducted for inorganics, there are several mild extractions that can be used to determine the available fraction of an organic contaminant in a soil or sediment sample. The techniques generally involve collecting a soil or sediment sample, adding the solvent, shaking and filtering, and analyzing the contaminant in the solvent phase.
Moderately Polar Organic Solvents. Extraction with moderately polar organic solvents, such as butanol, methanol, n-propanol, or ethyl acetate, has been used to estimate the availability of PAHs and a few pesticides in soil, and validation by comparison with biological assays has been achieved. For example, in several studies with freshly added and aged chemicals in soil, a good correlation was observed between the fraction of contaminant extractable using a moderately polar solvent and the fraction taken up by earthworms (Kelsey et al., 1997; Tang and Alexander, 1999). Similar experiments have shown good correlations between extractable fractions and other measures of bioavailability, such as biodegradability or soil mutagenicity assessed by a bacterial genotoxicity assay (Alexander and Alexander, 2000; Liste and Alexander, 2002). Comparisons suggest that the strength of the correlation to bioavailability varies by solvent, at a minimum (e.g., Reid et al., 2000). While these tests have shown promising results for assessing how bioavailability changes with contaminant aging in a particular soil, they demonstrate little consistency among different soils (Chung and
Alexander, 1998). Because these methods are also relatively simple, a better understanding of the attributes of soils that control or limit consistency are needed before such tests could be employed for wide-spread use. Additionally, these methods have not been widely tested on sediments.
Supercritical Fluid Extraction. Supercritical fluid extraction using CO2 from 40 to 150ºC has been developed for sequential extraction of polychlorinated biphenyls (PCBs) associated with field contaminated soils and sediments (Bjorklund et al., 1999). In this method, the sample is placed in a pressure- and temperature-controlled extraction chamber through which supercritical CO2 is flushed. By increasing the temperature of the supercritical fluid, the solvency power of the extractant is increased in a step-wise fashion (similar to sequential chemical extractions described earlier). Hawthorne and Grabanski (2000) used sequentially stronger supercritical fluid extraction conditions to selectively extract PAHs associated with “fast” (or “rapidly desorbing”), “moderate,” “slow,” and “very slow” sites on the soil collected before and during one year of field bioremediation of a manufactured gas plant site soil. They found that supercritical fluid extraction under the mildest conditions (120 bar, 50ºC) gave good quantitative agreement with removals achieved after one year of bioremediation for PAH compounds ranging from two to six rings. This is a promising method to assess quickly the easily available contaminant fraction in soils (as in Hawthorne et al., 2000a), but it needs to be verified on several other soil and sediment samples.
Subcritical Water Extraction. Subcritical water extraction (SWE) involves varying temperature and pressure to change the polarity of the water solvent. Thus, at low temperature, water extracts polar organics, while at higher temperature, water extracts moderately polar and nonpolar organics. By varying the temperature of extraction, quantitative recoveries of a range of compounds of varying polarities (e.g., PCBs, PAHs, alkylbenzenes, aromatic amines) have been achieved from soils and sediments that compare well to exhaustive solvent extraction methods (e.g., Hageman et al., 1996; Hawthorne et al., 1998, 2000b). In at least one study, temperature was the most influential experimental factor affecting extraction efficiency and kinetics (Krieger et al., 2000). At lower water temperatures (more mild conditions), this method has also shown diminishing contaminant recovery with aging in soil. As such, this relatively simple method shows promise in estimating bioavailability. At this time, there have been no reported correlations of subcritical water extraction results to bioassays.
Extractions with a Solid-Phase Sorbent
Solid phase extraction is a technique that can assess both the rate and extent of desorption of a sorbed organic compound because the solid phase (e.g., an exchange resin) acts as a contaminant sink. These approaches are termed “bio-
mimetic” because they mimic uptake from the solid or pore water directly to the organism.
Slurries with Sorbents. Most widely employed are variants on solid-phase extractions that use soil or sediment slurries (Yeom et al., 1996; Cornelissen et al., 1997a; Gustafson and Dickhut, 1997; Macrae and Hall, 1998; Morrison et al., 2000; Krauss and Wilcke, 2001). In these assays, a strong sorbent for the target compound is intimately mixed with the soil or sediment and water in a batch reactor. An adequate amount of sorbent is provided to ensure that the aqueous phase concentration of the target compound is maintained near zero to ensure a maximum driving force for desorption from the contaminated soil or sediment. Polymeric resins such as XAD-2, XAD-4, and Tenax TA (a 2,6-diphenyl-p-phenylene oxide based polymer) or C-18-coated materials such as Empore™ discs are typical sorbents for extracting HOCs from soil or sediment. After exposure, the sorptive phase is physically removed from the sample, and extracted to determine the total contaminant concentration. Because only the sorptive phase-associated concentrations are measured, this method quantifies the fraction of contaminant that was physically transferred into aqueous phase from the soil or sediment, under the assumption that partitioning to the sorptive phase is rapid. A potential artifact of these measurements is the possible inclusion of colloidal-associated contaminants if the colloids are incompletely separated from the resins during the separation step. The test result can be expressed as a rate (flux or kinetics of contaminant desorption from solid phase), but more commonly it is expressed as a mass (amount of a contaminant that can enter the aqueous phase after a defined incubation time and conditions).
Correlations between the contaminant mass removed by these desorption techniques and other bioavailability assays have been developed. In addition, results from many studies have revealed the existence of fast- and slow-release fractions of contaminant in the soil or sediment (Cornelissen et al., 1997a; Ghosh et al., 1999; Opdyke and Loehr, 1999). For example, strong correlations were observed between rapidly desorbed fractions of DDT, DDE, and DDD (as determined by C-18 extraction) from soils in which pesticides were freshly added or aged and earthworm assimilation (Tang et al., 1999; Morrison et al., 2000). In the study by Tang et al. (1999), worms assimilated 3 to 66 percent of the compounds in test samples with freshly added pesticides or residues persistent in the field for 49 years, which correlated well with the amount of pesticides taken up by C-18 disks. Similarly, the total mass of biodegradable PAH that desorbed during the “fast” phase as determined by Tenax TA extraction closely matched that which was bioavailable as determined by bioremediation via soil-slurry or land-farm treatment (Cornelissen et al., 1997a). Although these correlations suggest the technique can represent bioavailability to some organisms, limited validation has been performed. No validation on the purported correlation to bioconcentration in higher organisms has been reported.
These tests provide more information about the timing of contaminant release from the soil and sediment than mild extraction tests. However, the long time frames involved to obtain the information, particularly for the most hydrophobic compounds studied, can be a disadvantage (although C-18 disks require shorter equilibration periods and simpler extraction procedures). Furthermore, the distinction between the “fast” and “slow” release periods is empirical. Thus, these methods may be most useful for estimation of the contaminant fraction within the soil that is bioavailable within a reasonably short period of time, comparable to the study duration. Finally, these assays cannot be employed in situ, but require some type of solid–liquid slurrying.
Solid Phase Microextraction. Solid phase microextraction (SPME) is a relatively recent analytical development that is seeing increased use for aqueous, air, and soil analysis of volatile organic compounds (VOCs) and semi-volatile organic compounds (SVOCs) (Eisert and Pawliszyn, 1997; Penalver et al., 1999; Alpendurada, 2000). The SPME fibers consist of a sorbent coating (several types are available) on a support. As the fiber (approximately 1-cm long) is exposed to the sample, compounds are sorbed. Following exposure, the entire fiber is desorbed in the injection port of a gas chromatograph for analysis. Thus, SPME does not employ slurries of soil or sediment samples. Because of the small sizes and masses involved in the analysis compared to other procedures, and the now greater variety and selectivity of sorbent coatings, SPME has potential to be used as a biomimetic technique. Headspace solid phase microextraction is a modification that has been proposed as a sample concentration and preparation technique for the analysis of volatile and semi-volatile contaminants in soil (Havenga and Rohwer, 1999; Llompart et al., 1999). Inter-laboratory validation studies to determine the presence of different organic compounds at ppt levels demonstrated use of the technique for quantitative analysis (Alpendurada, 2000).
Solid phase microextraction requires no solvents, and it permits sample transfers and analyses with little modification of chromatographic equipment. Thus, cost can be kept at a minimum. The sample sizes and the extraction times needed to reach equilibrium are small compared to C-18 slurry methods. Moreover, the SPME technique is simple, amenable to automation, and suitable for field and onsite application. However, the method may suffer from the fact that samples are not “cleaned up” as with traditional chromatographic methods, resulting in interference. Thus, it may be useful only for certain chemicals in certain settings. Further, SPME does not give information on concentration but rather on the mass of compound collected over some prescribed interval. This suggests the need for rigorous QA/QC protocols along with proper calibration. There are no published studies that relate the rate or extent of uptake to a SPME fiber directly to a bioassay, although bioaccumulation tests using terrestrial organisms (i.e., enchytraeids and earthworms) are now being conducted. Initial results suggest that SPME fibers may be appropriate for measuring bioavailability of chemicals
with a log Kow up to 6.0 in soil (De Maagd and Staeb, 2002; Heslie et al., 2002; van der Wal et al., 2002).
Aqueous or Solvent Extractions using a Membrane
Polyethylene Tube Dialysis. An ex situ method that separates the contaminant sink from the solid–liquid matrix is polyethylene tube dialysis (PTD), wherein the slurry is placed inside a polyethylene tubing suspended in a strong organic extractant (e.g., pentane) and tumbled for 24 hours (Macrae and Hall, 1998). After exposure, the solvent is removed, concentrated, cleaned and analyzed by gas chromatography. Because contaminants associated with soils or sediments cannot pass through the tubing associated with the technique, only dissolved forms of the chemical are measured. This is an important concern for accurately measuring very hydrophobic compounds (certain pesticides, PAHs, and PCBs) which are sorbed strongly onto colloids.
Because less material is extracted than in Tenax TA methods, the detection limit of PTD for PAHs is approximately an order of magnitude greater (Macrae and Hall, 1998). Limited direct comparison of membrane-less and membrane-containing extraction assays has indicated that the former, expectedly, results in larger contaminant recoveries (Macrae and Hall, 1998). Nonetheless, there are situations where membrane-containing extraction assays may extract more contamination than membrane-less tests. For example, the availability of PAHs in marine sediments was greater when measured using polyethylene tube dialysis than Tenax TA methods or a semipermeable membrane device (SPMD, described below) (Macrae and Hall, 1998). The higher availability measurements from PTD were because some of the pentane used in the extraction passed through the tubing and acted as a co-solvent, facilitating desorption of the PAH from the sediment.
Polyethylene tube dialysis is useful when only small samples are available. This is a relatively inexpensive tool for initial evaluation or screening of contaminated soils and sediments. However, the technique imposes conditions that are hardly reflective of conditions in situ. Of the Tenax, SPMD, and PTD methods, comparison has shown that the Tenax extraction method was the least expensive, recovered the larger molecular weight compounds more efficiently, and worked well for heterogeneous environmental matrices (Macrae and Hall, 1998). There are no studies that compare extractability by PTD directly to a bioassay.
Membrane-Based Desorption Tests using a Solid-Phase Sorbent
Of slightly increased complexity are desorption assays wherein the solid sorptive phase sink is separated from the solid–liquid phase extractant by another phase (usually some kind of membrane). (The DGT technique described earlier for extracting inorganics from soil and sediment is an example.) Although such
techniques can be used with either a moist solid sample or a solid slurry, in practice most techniques have been developed for application to moist soil samples and sediments. Because of the need to cross a membrane, only truly soluble (i.e., non-colloid-associated) contaminants are measured in these assays.
Semipermeable Membrane Devices. SPMDs are patented devices used to measure the bioavailability of hydrophobic contaminants in the aquatic environment (Huckins et al., 1990, 1993, 1996; Lebo et al., 1992). The device is made from nonporous polyethylene tubing coated with a thinly spread layer of triolein. Hydrophobic contaminants become concentrated in the lipid relative to the water phase according to their respective partitioning coefficient as they would in organismal lipids or tissues. This method allows exposures to be determined without having to account for the variation between individual test organisms or the metabolism or depuration rates in organisms.
Semipermeable membrane devices may be employed in the field at the test site where affected organisms have been located. PAHs with up to five rings have been successfully recovered from marine sediment using this approach (Macrae and Hall, 1998). However, other methods such as the Tenax TA extraction gave higher recoveries for five- and six-member PAHs (Macrae and Hall, 1998). This was attributed to a slow transfer rate of large PAHs across the polyethylene tubing in the SPMD. For lower molecular weight compounds, SPMD was found to be superior to Tenax TA but more expensive.
As with other techniques using a solid-phase-supported sorbent, SPMD should be useful for estimating the fraction of contaminants in soil or sediment samples that will be bioavailable in a reasonable amount of time. However, sorption kinetics to these phases are relatively slow so that weeks may be required before pseudo-equilibrium is reached. Relatively large sample volumes may be necessary to obtain a sufficient level of analytical sensitivity.
The most important limitation of SPMD (or any solid-phase technique) is its dependence on partitioning alone to simulate biological exposure. Partitioning has a first order influence, but as discussed in Chapter 2 has often been shown to not be fully predictive of bioavailability to many organisms.
Both SPMD and DGT can be employed in situ, and concentration measures with both techniques were in good agreement with actual aqueous concentrations (Macrae and Hall, 1998; Zhang et al., 1998). With appropriate mathematical modeling, results from DGT or SPMD permit an estimation of the contaminant flux (Zhang et al., 1995, 1998). To date, there are no studies that compare extractability of organics by SPMD or DGT directly to results from a bioassay.
Other Extraction Techniques
Gas Purge. Gas purging of soil or sediment is one of the early techniques to measure the availability and desorption kinetics of semi-volatile organic com-
pounds from soils and sediments (Larsson, 1983; Hassett and Milicic, 1985; Karickhoff and Morris, 1985; Larsson, 1985; Gong et al., 1998). In this method, clean air is purged through a soil or sediment slurry in water, and the off gas is passed through organic traps that are sampled at specific intervals. Data from these tests show the mass fraction of organic compound released versus time. The fractional mass released may plateau with time, and from this it is possible to infer what fraction of the organic contaminant is relatively available.
The main advantage of the gas purge is that the method avoids a solid-separation step and thus eliminates any analytical bias in measuring aqueous concentrations due to colloids that may be present in aqueous samples. Although this method ensures measurement of released organic compound, quantification of mass transfer processes or extrapolation to in situ conditions is difficult because both solid–liquid and gas–liquid transfers are involved. As an example of this tool’s use, Wu and Gschwend (1986) used data from gas purge of chlorobenzenes from a solid slurry to model rate of release and particle size effects.
Enthalpy and Activation Energy of Desorption. The effects of temperature on sorption can provide information about sorption mechanisms and help explain why an organic compound is more or less tightly bound. The effects of temperature may be explored under isothermal or nonisothermal conditions. Desorption rate tests conducted at different temperatures yield kinetic rate constants from which the temperature dependence of the rate constant may be described by an activation energy (Cornelissen et al., 1997b; Ghosh et al., 1999). High activation energies would be associated with slow release, as in activated diffusion. Activation energies may also be determined by assessment of compound release under continuously increasing temperature conditions, as in automated thermal program desorption techniques (Ghosh et al., 2001). These measurements allow inferences about factors controlling the rate of release and the solid’s geochemistry. For example, in sediment significant differences were noted in activation energies for PAH release from clay–silt (37-41 kJ/mol) versus coal-derived particles (115-139 kJ/mol) (Ghosh et al., 2001). High PAH desorption activation energies from coal-derived particles were associated with low lability compared to PAHs on clays and silt. The relationships among rate of release, activation energy, the solid’s geochemistry, and the compound’s lability were linked in one study for PAHs in sediment and shown to correlate with biodegradability and bioaccumulation (Talley et al., 2002). Such tests are still largely in the development phase. Box 4-4 describes how desorption tests at different temperatures provide some insight on the nature of organic compound interaction with solid sorbent.
Extraction Tests for Organics that Mimic Human Exposure
In vitro extractions for predicting the oral bioavailability of hydrophobic organic compounds in soil to humans also exist, although they are less well
|
BOX 4-4 Temperature Desorption Techniques According to the van’t Hoff relationship, increasing temperatures result in decreasing equilibrium distribution coefficients for an exothermic process. For heterogeneous surfaces containing sites with different energies, sorption to higher energy sites is often characterized by larger negative enthalpies (or greater heat of sorption). This means that an increase in temperature decreases the amount sorbed, and more so at lower sorption values (Werth and Reinhard, 1997a). Thus, if the heat of sorption is positive at low sorption values and decreases with increasing sorption values, then different temperature isotherms converge with increasing sorption values. Werth and Reinhard (1997a) invoked these considerations to infer whether trichloroethylene (TCE) (1) partitions into natural organic matter or sorbs on water-wet mineral surfaces, which would show small changes with temperature, or (2) adsorbs in hydrophobic micropores that would show very large heats of sorption and significant differences at different temperatures. They demonstrated that temperature did not significantly affect sorption onto sand, aquifer sediment, or soil, suggesting that sorption onto mineral surfaces or partitioning into organic matter controlled equilibrium. However, for a clay and silt a large heat of sorption was calculated for low equilibrium concentrations, indicating that sorption in this region was occurring in micropores. In follow-on work, the authors concluded that the fraction of the TCE mass contributing to slow desorption was attributed to activated diffusion in micropores (Werth and Reinhard, 1997b). Young and Weber (1997), using supercritical fluid techniques to examine desorption, concluded that relatively weak nonspecific forces governed the binding of phenanthrene to three soils. |
developed than those for metals. The mechanism by which organic compounds become bioaccessible from soil appears to be primarily a matter of lipid chemistry in the gastrointestinal tract rather than simply dissolution in the acidic gastric environment. Thus, in vitro extraction tests for organics in soil use the framework established for in vitro extractions for metals with additional chemical components to simulate the lipid and protein chemistry of the gastrointestinal tract.
In the human gastrointestinal system, ingested lipids are hydrolyzed into absorbable forms (fatty acids and monoglycerols) by gastric and pancreatic lipases (Hernell et al., 1990). These fatty acids combine with bile salts to form mixed micelles—a core of hydrophobic lipids surrounded by a shell of lipoproteins. In the small intestine, bile salts form the outer layer of these micelles, which can traverse the mucine layer adjacent to the intestinal wall and then be absorbed across the intestinal epithelium. It is believed that these bile salt micelles in the small intestine provide a lipid sink into which HOCs can partition, and that the HOCs are then absorbed across the intestinal mucosa along with the micelle (Hack and Selenka, 1996; Guha et al., 1998; Holman, 2000; Oomen et al., 2000a). For this reason, bioaccessibility tests for HOCs in soil have all included
some form of bile salt micelle. Different lipid sources have been used to form bile salt micelles, including powdered whole milk (Hack and Selenka, 1996; Wittsiepe et al., 2001), a mixture of oleic acid, monoolein, diolein, and lecithin (Holman, 2000), and oleic acid alone (Oomen, 2000b; Ruby et al., 2002). Various HOCs also appear to partition into protein phases during simulated human digestion (Hack and Selenka, 1996; Oomen et al., 2000a). Therefore, a representative protein such as bovine serum albumin has been added to some extraction systems. Finally, mucin (a viscous mixture of glycoproteins and enzymes present in the mammalian stomach and intestines) has been used in several in vitro extraction systems because it has been observed to increase the fraction of HOCs liberated from soil (Hack and Selenka, 1996).
To date, in vitro extraction systems have been applied to PCBs (Hack and Selenka, 1996; Oomen et al., 2000a), PAHs (Hack and Selenka, 1996; Holman, 2000), polychlorinated dibenzodioxins/furans (Rotard et al., 1995; Wittsiepe et al., 2001, Ruby et al., 2002), and lindane (Oomen et al., 2000a) in soil and solid wastes. Results from these studies indicate that the fraction of different HOCs extracted is variable, and depends greatly on the composition of the test fluid. For example, for both PCBs and PAHs, extractability from soil is relatively low when only the gastric phase of the extraction is employed, but it increases dramatically when the small intestinal phase of the extraction is added (Hack and Selenka, 1996). For all of these HOCs, the inclusion of bile salts and a lipid source to the extraction test greatly increased the fraction of the HOC liberated from the soil or waste. In general, the more thorough extraction tests gave bioaccessibility results of 20 to 60 percent for PCBs, PAHs, and polychlorinated dibenzodioxins/furans from soils and solid wastes. Because there have been no published comparisons between these in vitro data and those from in vivo studies for the same soil substrates, the predictive value of these in vitro tests is unknown. The lack of in vivo studies for HOCs in soil is not surprising given their cost and difficulty. Substantial effort will be required to validate these in vitro extraction tests for HOCs in soil.
At this time, no work has been reported on the development of extraction tests to estimate the dermal bioavailability of organic compounds in soil or sediment.
Normalization Techniques
Normalization of extracted contaminant concentrations with measured soil or sediment characteristics is a long-standing approach that has been used to move beyond the simple assumptions of extractions and to incorporate chemical and biological complexities. In this approach, contaminant concentrations in sediments or soils are arithmetically “corrected” via geochemical or biological factors thought to influence bioavailability. Many normalizations were developed from observing correlations between factors during field studies and as such do
not have a complete mechanistic underpinning. Others are based on a more theoretical construct.
Soil scientists have long combined results from extractions with correlative normalizations to empirically develop predictive equations for metal availability to plants (Pickering, 1981). For aquatic sediments, Luoma and Davis (1983) suggested that contaminants generally associate more completely with a geochemical component of the sediments as the number of binding sites of that component increases in the sediment. If that component–contaminant association reduces bioavailability, then higher component concentrations (e.g., iron oxide) result in reduced bioavailability. Some impressive empirical correlations with bioavailability have been obtained using this general approach. For example, correlation was demonstrated between lead bioaccumulated by bivalves and lead:iron ratios in oxidized surface sediments across 17 English estuaries (Luoma and Bryan, 1978). Tessier et al. (1984) showed a similar relationship for a variety of Quebec lakes. Other successful normalizations were found for arsenic:iron ratios in sediments and arsenic uptake by bivalves in English estuaries (Langston, 1980) and mercury:carbon ratios in a variety of environments (Langston, 1982; Breteler et al., 1981).
The studies cited above included broad concentration gradients, careful biological sampling, and complex geochemical conditions across the data set (i.e., they were not the result of co-variances or simple conditions). Nevertheless, this approach has not been extensively employed in recent years. One reason is that the approach is not mechanistic, although mechanistic explanations have been offered (Luoma, 1989). In addition, there is a lack of models or direct analytical techniques to relate metal form to the normalized concentration. Indeed, the quantitative relationship between bioavailability and normalized concentration is unique for each metal–species–environment combination, making development of generalized models difficult. Finally, factors affecting bioavailability are more complex than accounted for by simple normalization for many metals or animal species (e.g., Luoma and Bryan, 1982; Amyot et al., 1994).
Normalization techniques also were developed as shortcuts or surrogate measures to represent expected outcomes of a mechanistic theory like equilibrium partitioning (DiToro et al., 1991). Equilibrium partitioning (EqP) theory uses hydrophobicity and normalization to organic carbon in sediment to predict porewater contaminant concentrations, assuming bioavailability is controlled by pore waters alone (DiToro et al., 1991). Normalization to lipid content of the organism(s) can also improve such relationships (e.g., Lake et al., 1996). Bioassays that manipulated organic carbon in sediments provided the experimental substantiation of the theory (Swartz et al., 1990). But field studies have begun to indicate that bioaccumulation of organic chemicals is not necessarily predictable from EqP alone (Pereira et al., 1988; Swackhamer and Hites, 1988). Dissolved organic matter (DOC) is one factor important in deviations from EqP-predicted distributions of organic chemicals in soil and sediment (DeWitt et al., 1992;
Suedel et al., 1993). Complexities affecting the form of the chemical, including the age of the association, also cause deviations (Meador et al., 1995). Apparently, this approach is most valid if the biological processes involved are relatively simple. In complex food webs, though, additional considerations are necessary to predict bioavailability to upper trophic level animals (Kidd et al., 1995).
Perhaps the best-known normalization technique for defining metal bioavailability is based on redox condition for metals and EqP theory. As discussed in Chapter 2, DiToro et al. (1990, 1991) suggested that normalizing metal concentration in sediment by acid volatile sulfide (AVS) might explain metal bioavailability from sediments. This approach was initially tested using bioassays and manipulations of natural sediments. Recent analyses of the early approaches suggest co-variance among total metal concentration and AVS effects might confound some of those tests (Lee et al., 2000). Additional limitations of this technique have been noted in Chapter 2 and Box 4-2. The AVS normalization technique has not been tested using a field correlative approach—that is, correlating AVS measurements from field samples with bioaccumulation of chemicals in organisms residing at the field site.
Analysis of Extraction Techniques
Operational extractions, empirically and theoretically developed normalizations, and models like equilibrium partitioning have each provided some of the ingredients needed to explain when, where, and how contaminants in soils and sediments become bioavailable. It is important when using these extractions to understand their strengths and limitations, many of which have been touched upon in this section.
Perhaps most importantly, extractions should be verified by comparing the chemical predictions with responses in biological indicators. There is an extensive body of data comparing certain extracts with plant uptake, with substantial observed correlation over a large gradient of contaminant concentrations and soil types. A much smaller body of information exists for other biological endpoints (with earthworm bioaccumulation tests being a commonly used bioassay). Several different extraction procedures have shown correlations with metal bioaccumulation in experimental organisms (Luoma and Jenne, 1977; Pickering, 1981; Fisher and Telyssie, 1986). However, not all of the validation efforts reported to date have been successful, calling into question the reliance on extraction procedures to measure bioavailability. Certainly no one universal extraction procedure has been shown to consistently correlate with tissue concentrations in plants or animals across complicated environmental conditions.
The greatest validation with bioassays has come for those extractions designed for specific metals or chemicals and specific endpoints. For example, extraction of silver from oxidized sediments with 0.5–1 N HCl appears to consistently improve correlation of sediment-bound silver with bioaccumulated silver
(Luoma et al., 1991). When such extracts are used in situations for which they were not developed, they are generally unreliable. For example, one of the extractants employed most widely and successfully in studies of plant-available copper from calcareous soils is 0.004 M DTPA (Lindsay and Norvell, 1978). Correlations between the extraction results and plant uptake have been found to be insignificant among soil series (Pickering, 1981), primarily due to inappropriate application of the test. The use and misuse of the DTPA test for plant-available nutrients and contaminants are described in Box 4-5.
Most extractions account for contaminant release from the solid surface to pore water. Thus, they are most successful (i.e., predictive) when biological uptake is dominated by a pore-water pathway (e.g., plant uptake of metals). Extractions cannot account for other, more complicated uptake mechanisms that control an organism’s overall dose (Landrum et al., 1992; Luoma et al., 1992; Luoma and Fisher, 1995). Contaminants are distributed among solution, suspended particles, sediments, pore waters, and specific (living and non-living) food sources within all of these. Each species’ exposure to those contaminants is determined by how the species “samples” this complex milieu, and by the accessibility of pollutants within each compartment of the milieu. Digestion is a flexible, adaptive, multi-faceted living process that differs among species and can change within a single species with life history or environmental conditions. Thus, to the extent that dietary uptake of a contaminant is important, simulation of that process by single chemical extraction will be close to impossible. This limitation also extends to extraction tests that use a solid-phase absorbent meant to simulate biological exposure via a first-order partitioning mechanism. In addition to dietary uptake, organisms differ in the rate at which they pass water across their gills, in the ways they are exposed to soil and sediment, pore waters and surface waters, as well as in trophic relationships. These factors have important implications for bioavailability that solid-phase extractants cannot simulate.
Extraction procedures do not (with a few exceptions) remove metals or organic compounds from specific components of soils and sediments, nor can they explain the type or character of the sorbent phase to which an organic sorbate may be sequestered. Thus, they are operational, not mechanistic, methods for estimating contaminant availability. Several obstacles preclude the development of extractions and other tools that can directly determine critical forms of contaminants at the proper scales, concentrations, and conditions. Metal ion activities in solution, for instance, are difficult to determine in pore waters due to problems with detection limits at small scale. This limitation has lead to sediments and pore waters being characterized geochemically on scales much broader than the microhabitat scales experienced by benthic or other organisms (Luoma and Ho, 1993).
Extraction approaches and the use of normalizations coupled with extractions have a mixed history of success (with more success in soil systems than sediment systems). Many prove limited in their abilities to consistently predict
|
BOX 4-5 Misuse of DTPA Soil Test The DTPA soil extraction was designed to predict micronutrient deficiencies in neutral to calcareous soils (Lindsay and Norvell, 1978). When used appropriately, the test is effective, reproducible, and accurate. In calcareous soils, micronutrients such as zinc, iron, manganese, and copper will generally be sparingly soluble, and nutrient deficiencies are common. As a result, plants may aggressively manipulate the rhizosphere to access required but insoluble micronutrients. Graminaceous plants (grass species) secrete phytosiderophores into the soil solution. These compounds chelate iron as well as other cations in solution, thereby reducing the concentration of free metal ions. In response to this change in equilibria, a portion of the cations in the labile solid phase will come into solution and be available for complexation. The transpiration stream will transport the chelated cations to the rhizosphere where they can be absorbed by plants (Marschner, 1995). The DTPA extraction was developed to mimic the behavior of plant roots under these circumstances. In developing the extract, Lindsay and Norvell tested a range of pH levels, time intervals, and solution molarities to determine which were best correlated with plant behavior (as shown in pot studies using 77 soils from Colorado). Both corn and sorghum were used to calibrate the extraction. Since its development, the DTPA soil extraction is one of the most widely used to predict micronutrient availability in soil systems (Loeppert and Inskeep, 1996). It has also been used for regulatory purposes; the Wyoming Department of Environmental Quality has set a suitable soil selenium level as < 0.1 mg kg–1 as measured by the ammonium bicarbonate-DTPA extract (WDEQ-LQD, 1984). Because of this success, the DTPA extract has been used to assess micronutrient availability under a range of soil conditions as well as for a range of elements (e.g., Amacher, 1996; Li and Shuman, 1997). Outside of its intended use, however, results from the extract often do not correlate with plant uptake data (e.g., Bidwell and Dowdy, 1987; O’Connor, 1988; Kuo, 1990; Miner et al., 1997). This can be the result of many factors. The soil test was developed for neutral soils with a potential for deficiencies and to mimic the behavior of graminaceous species. When it is used to predict uptake by other species, the potential for poor correlation with plant data increases. When the extract is used for soils with different properties, results may also not relate to plant uptake. Using the DTPA test to measure the plant available fraction of contaminants in disturbed soils may be inappropriate for two reasons: the extract may be overly aggressive in relation to plant behavior in these environments, and the chelate may become saturated and not truly measure the full extractable pool. Of these, the first is the most significant. In one study where the soil:solution ratio was corrected to compensate for high solution metal concentrations, there was no relationship between plant uptake and DTPA extractable metals (Li et al., 2000). Dilute salt extractable metals were a much better predictor of plant uptake. Similarly, Brown et al. (1994) found that dilute salt or water extractable metals were a much more accurate measure of plant available metals than DTPA in smelter-contaminated soils. Less aggressive extracts appear to be more appropriate for cases where excess, rather than deficient, concentrations of metals are expected. |
bioavailability processes when applied across a wide range of conditions. The best that can usually be hoped for is, for example, prediction of 50 percent or so of the variance in bioaccumulation in the field (see Box 1-2). Given the heterogeneity of soils and sediments and multitude of exposure pathways present to a given organism, it is unrealistic to expect extraction tools to fully account for such variability. Rather, such tests should be viewed as qualitative measures of reactivity that may be useful as screening tools. Given the cost of bioassays, the use of extractions is likely to increase in the human health risk assessment arena, particularly for metals where some in vitro extraction tests have been validated.
BIOLOGICALLY BASED TECHNIQUES FOR MEASURING BIOAVAILABILITY
The role of physical and chemical processes is well recognized in bioavailability discussions, but biological processes also play important roles. Biological techniques are employed to study influential biological processes themselves, and as probes to study physical and chemical processes. In a controlled experiment, almost any technique that measures a biological response to contaminant exposure is suitable. However, interpreting the results from such experiments is not always straightforward. This is because biological processes other than the one under investigation can confound the results, making generalizations among experiments or about natural settings a challenge.
Tests that measure biological responses at levels of organization closest to contaminant transport across the membrane, of which assimilation efficiency is perhaps the best example, are easy to interpret from a mechanistic standpoint compared to responses that take place at more complex levels of organization (see Figure 4-6). Gross rates of contaminant biouptake (across the gills or the gut) provide a direct and unambiguous evaluation of bioavailability process D. Whole organism bioaccumulation tests are more complicated in that they reflect not just movement across the membrane, but also how the organism encounters its environment and species-specific internal processing mechanisms like digestion. However, depending on the length of the exposure and the organism under study, these internal processes may be minimized. The “uptake bioassays” discussed in this chapter include those that measure the initial biouptake of a contaminant across a biological membrane (bioavailability process D) as well as longer-term bioaccumulation tests.
Other tests that measure more complicated biological responses or groups of processes reveal less about uptake and accumulation but are valuable for studying toxic effects. For example, biochemical responses to exposure at the cellular level can be measured with biomarkers such as P450. While P450 levels might be unambiguously related to contaminant transport across a biological membrane if all else is controlled, in a natural setting elevated P450 can result from exposure
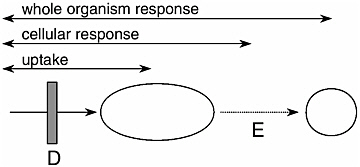
FIGURE 4-6 Biologically based tests measure responses at different levels of organization.
to any of several possible stressors. Toxicity tests (acute and sublethal) are widely used both in the lab and in situ to evaluate bioavailability because they are practical, they depict responses of high relevance, and they are particularly useful for helping to understand the effect of contaminant mixtures. Because the number of potentially confounding factors grows beyond those relevant to whole organism bioaccumulation (e.g., detoxification of chemicals can differ widely among species or even among environments), toxicity tests are not optimal mechanistic indicators of bioavailability processes (as defined in Chapter 1).
The section begins with tests that measure uptake (synonymous with absorption) only, including tests used for human health risk assessment purposes and those used primarily for ecological receptors. Determining absorption of chemicals in the digestive tract is one way to evaluate process D in Figure 1-1 (although as noted in Chapter 1, human health bioassays measure absorption into systemic circulation, which includes some E bioavailability processes). Absorption itself can be the subject of study, or absorption efficiency can be used as a probe to test site-specific soil or sediment properties or draw generalizations about effects of bioavailability processes A through C. Methodologies for determining absorption exist for both the mammalian model (using cell cultures, organ studies, and feeding studies of whole animals) and for lower-order organisms (using assimilation efficiency). After assimilation efficiency, the tests described encompass additional processes, including cellular responses and toxic effects within an organism. They are organized by scale, starting with molecular approaches, organismal approaches, and finally ecosystem-level approaches. Within the discussion of organismal approaches, uptake and effects-based tests are discussed for each organism type. The conditions present during these studies can vary widely, from laboratory tests using synthetic or field samples, to in situ bioassays with caged animals, to field studies with only natural elements.
Adsorption by Mammals: Human Health Bioassays
The tools discussed in this section are used primarily to gain insight on the bioavailability of chemicals in humans. They are focused on the bioavailability processes related to absorption of chemicals via direct ingestion and dermal contact. Although many of these techniques have been developed in the context of drug bioavailability, they hold potential for measuring the bioavailability of environmental contaminants from soils or sediments, but have not yet been used for this purpose.
The best source of information on bioavailability of environmental contaminants to humans would come from studies of humans, but there are very few of these studies. Experimental and ethical constraints dealing with the kinds of dosing and sampling that are possible with human subjects greatly limit clinical research on bioavailability of environmental contaminants. Consequently, most of the bioavailability information used in human health risk assessments must come from laboratory animals serving as surrogates (including pigs, rats, mice, monkeys, rabbits, and dogs) or from experimental model systems. The discussion begins with the simplest in vitro models, progressing to more complex mimics of human physiology, and finishing with approaches for deriving information from humans.
Cell Culture Studies
The simplest biological systems to study absorption (specifically bioavailability process D in Figure 1-1) in humans are cell cultures.
Gastrointestinal Absorption. The most established example of a cell culture system to understand absorption processes is the Caco-2 cell, which is a human colon adenocarcinoma cell line. In culture, Caco-2 cells differentiate to form a monolayer with brush borders resembling the apical surface of enterocytes (Pinto et al., 1983). They have been used extensively to study nutrient uptake from food and to explore intestinal absorption of drugs. They have not been used to any great extent to study intestinal absorption of soil or sediment contaminants for a number of reasons as discussed below, although theoretically there is no reason why they could not be adapted to do so.
During experiments using Caco-2 cells, the cells are allowed to form monolayers, substances of interest are added to the cell culture, and the rate of uptake into the Caco-2 cells is measured. The advantages of cell cultures are that they are easily manipulated and are well suited to the study of membrane transport mechanisms and of interactions among substances that affect substance uptake into enterocytes. Caco-2 cells have, for example, provided valuable information on the effect of dietary composition on iron absorption (Glahn et al., 1998). The disadvantage of Caco-2 cells is that they can provide information on only one
facet of bioavailability—absorption in the enterocyte. Absorption that occurs elsewhere, such as the gastric mucosa, and bioavailability processes occurring before and after enterocyte uptake are not addressed. In addition, absorption may not reflect the amount of chemical reaching the systemic circulation for chemicals that undergo presystemic elimination (see Chapter 1).
Cell culture methods have undergone rudimentary validation. Au and Reddy (2000) reported excellent correlation between iron uptake ratios in human subjects and Caco-2 cells. Studies comparing uptake in Caco-2 cells with permeability1 in the jejunum of the small intestine measured in humans found reasonably good agreement for rapidly absorbed chemicals, but up to 1,000-fold lower permeability in Caco-2 cells than in humans for poorly absorbed chemicals (Lennernas, 1998). These observations indicate that Caco-2 cells may have limited value as quantitative indicators of absorption. Caco-2 cells have been used primarily as a research tool, and formal studies of reproducibility and inter-laboratory consistency have not been conducted.
Dermal Absorption. For dermal absorption, a number of commercial “human skin equivalent” products have been developed. These are based on stratified epithelial cells in culture with an architecture resembling normal human epidermis (Ponec et al., 2000). Examples include Epiderm, Episkin, Skin2, and SkinEthic. These tools have been used primarily in developing in vitro tests for skin irritancy and, as part of their development, tests of intra- and interbatch consistency of morphology (e.g., Boelsma et al., 2000) and intra- and inter-laboratory irritation tests have been conducted (e.g., Fentem et al., 2001). The possibility of using these skin tests for examining dermal absorption of chemicals has only recently been explored (Zghoul et al., 2001). The potential advantages of using “skin equivalent” cultures to study absorption of chemicals include lower cost than in vivo studies and the ability to avoid use of human and animal subjects. However, there are virtually no data available at present with which to judge the predictive capabilities of this tool.
Isolated Tissues and Organs
Gastrointestinal Absorption. Gastrointestinal absorption has been measured in isolated segments of the gastrointestinal tract of laboratory animals. There are two basic approaches. One involves excision of a segment of the gut and perfusion with a specialized apparatus, such as when a rat jejunal segment is mounted in an Ussing Chamber. Uptake by passive diffusion or transport can be readily measured. Rank order of permeabilities in this system among several
drugs was observed to be similar to that in jejunal permeability studies in human volunteers, although the permeability rates from the rat jejunum segment were lower, particularly for transported substances (Lennernas, 1998). Lower permeabilities may result from the absence of blood flow in the excised segment, which could lead to lower concentration gradients across the jejunum and perhaps deficiencies in cofactors needed for optimum function of transporters.
The second approach is to surgically isolate, but not remove, a segment of gut. The procedure is performed with the laboratory animal under anesthesia. Substances to be measured are introduced into the lumen of the surgically isolated segment. In order to measure the absorption rate, the vasculature serving the gut segment may be sampled. Alternatively, the vasculature may be cannulated and perfused with an artificial medium that is periodically sampled. The rat is often used, but other animals including catfish have been employed (Kleinow et al., 1998). This method has the same advantages as the excised segment model described above but it more closely approximates the intact gut. In a study of rat jejunum in situ, permeabilities were higher than measurements in excised segments (as above), but still less than observations in human subjects (Lennernas, 1998).
Both isolated gut models have the advantage of measuring absorption in gut tissue that is morphologically intact. Absorption processes affecting movement of the chemical from the gut lumen to the serosal side of the gut tissue are measured, offering a greater integration of events than is possible with cells in culture. Also, xenobiotic metabolism enzymes in intestinal epithelium remain active for some period of time, permitting an examination of the nature and extent of biotransformation that occurs during absorption. Ease of manipulation of these model systems makes them very useful research tools. As with other in vitro tools, they afford the ability to measure large numbers of samples or combinations of chemicals in a short period of time. However, the validation studies performed to date suggest that these models probably underestimate absorption rates in the intact human gut, at least for many chemicals. Further, as discussed above, absorption rate across the gut may not be the most important determinant of bioavailability, particularly for chemicals that undergo presystemic elimination in the liver. Therefore, the absorption rates may not correlate with absolute bioavailability in vivo (although this is not a concern when conducting relative bioavailability studies). There are no examples of this system being used to measure uptake from contaminated soil or sediment.
Dermal Absorption. The use of skin tissue from humans or animals to study dermal absorption in vitro is very common. The basic design consists of either human or animal skin placed in a chamber, with the skin dividing the chamber into donor and receptor compartments. The dermal dose is place on the donor side, and fluid in the receptor compartment is tested over time for the appearance of the chemical. The receptor fluid, which is usually saline or an aqueous buffer,
may remain in the chamber throughout the experiment (static design), or receptor fluid may be circulated through the chamber (flow-through design). Results may be expressed as a rate of chemical movement across the skin barrier or in terms of a percent of the amount of chemical applied on the donor side reaching the receptor side over a specified period of time.
In vitro skin tissue offers a more convenient means by which to study the dermal bioavailability of chemicals than in vivo tools. Properly excised skin can maintain the anatomical barrier of the stratum corneum. There is evidence that fresh skin tissue may also retain, at least temporarily, xenobiotic metabolism activities of skin in situ, allowing the opportunity to study metabolism of chemicals during the dermal route. Studies comparing dermal absorption of chemicals in human subjects with results from human skin in vitro have generally found good agreement. For example, Bronaugh and Franz (1986) found that dermal absorption of benzoic acid, caffeine, and testosterone applied to the skin of human volunteers was comparable to absorption in vitro through human skin, both in terms of percent dose absorbed per hour and total percent absorbed.
There is considerable interest in using skin from species that could act as surrogates for humans. The monkey and pig as well as the hairless mouse have been used, although there are examples of chemicals for which concordance was poor. Perhaps the most striking example of this is paraquat, with a permeability in the guinea pig and mouse 268- and 1,461-times that in human skin, respectively (Bronaugh and Collier, 1993).
There have been a limited number of in vitro studies conducted with contaminants bound to soil or sediment. Wester and Maibach (1998) measured the percutaneous absorption of DDT, benzo(a)pyrene, chlordane, and pentachlorophenol from soils after application to in vitro skin tissue. Both the chemical concentration in the receptor fluid and that remaining in the skin at the end of the experiment were lower when the soil-bound chemical was applied vs. the pure chemical. Similarly, the concentration of PCBs (Aroclors 1242 and 1254) in receptor fluid and skin was diminished when administered in soil as compared with a mineral oil vehicle, and absorption of arsenic, cadmium, and mercury from soils was less than from water. These studies can provide quantitative information on the extent of absorption from soils useful in human health risk assessment. There are a number of factors that must be considered in applying this information. Percutaneous absorption of a chemical from soil, both in vitro and in vivo, may not be linear over time. This raises questions about how the extent of absorption observed in vitro over one period of time should be applied to environmental exposures that may occur over a different period of time. Also, there is some uncertainty as to how the dose retained in the skin during in vitro studies should be regarded. In the in vitro studies of dermal absorption described above, the percent of dose remaining in the skin at the end of the experiment was greater than the percent of dose in the receptor fluid for most of the chemicals studied, both organics and metals. It is not clear if the percent remaining in the skin will
ultimately reach the systemic circulation in situ (as some may be lost through skin exfoliation). If so, it should be considered as contributing toward systemic bioavailability. Greater information regarding the fate of metals absorbed through the skin is needed to resolve this. Finally, studies of the reproducibility and repeatability of in vitro skin absorption studies have not been conducted in the context of assessing the bioavailability of chemicals from soils or sediments.
Whole Animal Approaches
Several approaches are available for measuring both oral and dermal bioavailability in laboratory animals. These tests are sometimes used to validate the physical and chemical tools discussed earlier, or to provide complementary evidence about bioavailability processes in a system. The best approach for a particular situation depends upon the objective (i.e., whether measurement of absolute or relative bioavailability is sought—see Chapter 2), the toxicokinetics of the chemical (e.g., rate and major pathway(s) of excretion), analytical capabilities, and time and financial constraints on the study.
Gastrointestinal Absorption: Blood or Plasma Measurements. Chemicals absorbed and reaching the systemic circulation can be measured in blood or plasma. The systemically absorbed dose is usually determined from the concentrations in blood or plasma over time after a measured dose of the chemical is administered to the animal. For this technique to be effective, the time frame of measurement must cover all of the absorption and most of the elimination of the chemical from blood. Blood or plasma concentrations are plotted against time, and the area under the concentration versus time profile (AUC) is calculated.
In order to determine absolute oral bioavailability, the AUC following oral administration (AUCoral) is compared with the AUC after intravenous administration (AUCiv), the latter representing the AUC expected if the entire oral dose reaches the systemic circulation. The equation below represents the calculation of absolute bioavailability (Fabsolute) based on a single oral dose:
Notice that the equation includes terms for the oral and intravenous dose (Doral and Div). This allows the AUCs to be corrected for dose if different doses are used for the two routes. This might be required, for example, if the intravenous dose is limited by poor aqueous solubility of the chemical or pronounced acute toxicity. The use of different doses assumes that the AUC is directly proportional to dose (i.e., linear pharmacokinetics), at least within the range of the doses being compared. This may not always be the case, particularly if the chemical is subject to
saturable absorption or metabolic processes. If the pharmacokinetics are not linear, the use of different doses can result in substantial error in measurement of bioavailability. This method also assumes that the clearance of the chemical is the same following oral and intravenous administration, which for most chemicals is not an unreasonable assumption.
An analogous approach can be used to assess relative bioavailability. In this case, bioavailability under differing sets of conditions (e.g., oral bioavailability of a chemical from a soil matrix versus from water) can be obtained from the ratio of the their AUCs, with one designated as the reference for comparison (“condition A”, in the equation below).
As with the measurement of absolute bioavailability, doses of different size can be used, but only if they are in the linear pharmacokinetic range.
In addition to providing information on the extent of absorption of a chemical, blood or plasma data provide the best information on the rate of absorption. Although the method can theoretically be applied to virtually any chemical, this approach is best suited for chemicals eliminated from blood in a matter of hours to a few days. Also, reliable AUC measurements require several blood or plasma samples with chemical concentrations that are measurable. Animal subjects must be large enough to provide the number of samples and blood volume dictated by the experimental design and the sensitivity of available analytical methods. This limits the utility of small animals for these studies, and often makes the testing of environmentally relevant doses of chemicals difficult.
Gastrointestinal Absorption: Urine Measurements. Many chemicals are excreted extensively in urine following their absorption, and analysis of the urine can provide an indication of absorbed dose. Typically, the animal subject is given a measured dose of the chemical, and urine is collected over time. The appropriate urine collection period depends on the elimination rate of the chemical but is usually extended until the chemical reaches undetectable or background concentration in urine. Based on the concentration of chemical in urine samples and their volumes, the cumulative amount excreted is calculated.
The absolute oral bioavailability of a chemical can be calculated from the amount excreted following an oral dose (Aurine(oral) in the equation below) divided by the amount excreted after an intravenous dose (Aurine(iv)). Analogous to the approach using blood or plasma data, the intravenous dose is intended to represent the amount excreted in urine if the entire oral dose is absorbed. If doses of different sizes are used, the excreted amounts can be corrected for dose, if it is known or can be assumed that the amounts excreted are linearly related to dose.
Sometimes, urinary excretion data are used to draw inferences on absolute bioavailability without benefit of a comparison with an intravenous dose. The amount excreted in urine provides an indication of absorbed dose only if other routes of excretion (e.g., biliary, pulmonary) are negligible and elimination of the dose of chemical is complete. Because these conditions are rarely satisfied fully, bioavailability is usually underestimated by this method. Urinary excretion data can also be used to assess relative bioavailability by comparing the excreted amount under two different dosing conditions (see equation below).
This technique is less invasive than blood or plasma measurements and can provide reliable bioavailability measurements for chemicals excreted primarily in urine. This approach should not be used if urinary excretion accounts for less than 20 percent of the dose. Also, accurate measurement of bioavailability requires complete urine collection, not just discrete urine samples, which may be difficult in some circumstances.
Gastrointestinal Absorption: Fecal Measurements. Fecal excretion represents the inverse of oral bioavailability. A chemical that is not absorbed following oral exposure will ultimately be excreted in feces. Therefore, measurement of fecal concentration can be used as an indication of the extent of absorption. Measurement of oral bioavailability involves collection of feces following single or multiple doses of the chemical. The collection interval must be sufficiently long to accommodate the gastrointestinal transit of the dose. Also, some chemicals do not reach the systemic circulation, but are instead excreted in the feces as the epithelial lining is sloughed into the lumen of the gastrointestinal tract. The collection of the unabsorbed dose must take into consideration the time course for these events.
Absolute oral bioavailability can be estimated by comparing fecal excretion of the chemical following both oral and intravenous doses. The intravenous dose is important because it provides information on the extent of biliary excretion of the chemical and diffusion of the chemical from systemic circulation into the gut. Both contribute to chemical in the feces, but represent absorbed, rather than unabsorbed, chemical. Some investigators have suggested that an intraperitoneal dose of the chemical (obviously relevant for animal studies, but not humans) can be used for the same purpose. The amount of chemical excreted in the feces after an oral dose (Afeces(oral) in the equation below), corrected for these confounding
inputs (Afeces(iv)), can then be compared with the dose to obtain an estimate of oral bioavailability.
If biliary excretion is known or assumed to be negligible, then fecal excretion data from oral dosing alone can be used to approximate the oral bioavailability. However, to the extent that this assumption is in error, the approximation will underestimate the actual bioavailability. It is also important to recognize that this method estimates absorption into the portal circulation, which is not necessarily equivalent to systemic absorption. For chemicals with substantial hepatic first-pass metabolism that detoxifies them in the liver, fecal excretion will overestimate systemic bioavailability. If the extent of pre-systemic elimination by the liver is known or can be estimated, this can be used to correct the apparent oral bioavailability based on fecal excretion to reflect systemic bioavailability.
This approach is generally less invasive than methods based on blood or plasma but requires quantitative collection of feces. For chemicals that are extensively absorbed, have substantial pre-systemic elimination by the liver, or prominent excretion in bile, fecal excretion data may not be a reliable bioavailability tool.
Gastrointestinal Absorption: Tissue Measurements. Tissue concentrations may be used in combination with measurements of excreta to assess absorbed chemicals using a mass-balance approach. Mass-balance approaches require measuring the chemical in various tissues in the body to determine the total internal dose. Unabsorbed dose and the amount of dose excreted are also measured, such that the entire dose can be accounted for. From these measurements, the amount absorbed can be calculated. Measurement of absolute oral bioavailability can be accomplished without the need for a comparison intravenous dose, but the mass-balance approach is analytically intensive and obviously unsuitable for measurements in humans.
Alternatively, tissue concentrations alone can be used in some situations to assess oral bioavailability. This approach assumes that the concentration of chemical in tissues is directly proportional to the absorbed dose. It is best suited to measurement of relative bioavailability, and is similar to the feeding tests described later for birds and mammals. Animal subjects may be administered the chemical in one or multiple doses. At specified times, animals are euthanized, and the concentration in one or more tissues is measured. Relative bioavailability is determined from the ratio of the tissue concentrations between the different types of oral doses (Ctissue(conditionA) and Ctissue(conditionB) in the equation below). If the oral doses compared are of different size, the tissue concentrations can be
corrected for dose, provided that the relationship between dose and tissue concentration is linear.
The tissue(s) selected for analysis may represent a target organ for toxicity or, more commonly, a tissue to which the chemical preferentially distributes. This facilitates accurate measurement of concentration, particularly in studies using small animals where the size of the tissue sample available for analysis may be limited.
Tissue ratios offer the advantage of an internal measurement of systemic bioavailability, and they may be more suitable than blood or plasma measurements for chemicals with protracted elimination phases. However, they provide little or no information on absorption rate, and the tissue(s) to be measured and the timing of measurements must be carefully considered to avoid misleading results. Also, use of this approach requires the assumption that distribution and clearance of the chemical are equivalent under the two dosing conditions.
***
The principal advantage of whole-animal oral absorption studies is that they measure bioavailability in its most clinically relevant form, that is, the absorption of chemicals from the gastrointestinal tract and into the systemic circulation. This integrates all of the relevant biological components related to systemic absorption, including presystemic elimination if present. By using the animals as surrogates for humans, these studies avoid the experimental and ethical problems associated with the use of human subjects. Currently, certain in vivo bioavailability studies conducted with an appropriate species are considered the “gold standard” for developing bioavailability information suitable for use in quantitative human health risk assessments, and they are often used to validate other bioavailability tools. For example, the young swine model for lead bioavailability has been used to validate in vitro extraction tests. The principal disadvantages of whole animal bioavailability studies are their expense and the time required to conduct them.
The assumption that certain species serve as valid models for human absorption comes primarily from studies in the pharmaceutical industry rather than direct animal-to-human comparisons for environmental contaminants. As discussed below in the section on clinical studies, there are almost no definitive data on the absorption of environmental contaminants in human subjects to serve as the basis for comparison. However, the extensive use of animal models in preclinical drug development for a variety of different kinds of chemicals offers some assurance that data derived in appropriate animal subjects is relevant to humans.
Dermal Absorption. In theory, the same approaches used to assess oral bioavailability can be used to test bioavailability for the dermal route (except, of course, measurement of fecal excretion). The principal difficulty in applying these methods to dermal bioavailability involves analytical sensitivity. Simply put, the doses absorbed through the skin in typical dermal uptake experiments are often too small to measure in blood, urine, or tissues. In order to maximize sensitivity of measurement, many dermal absorption studies use radiolabeled chemicals. This has been employed successfully in the measurement of dermal absorption of chemicals in soils. However, the use of radiolabeled compounds precludes testing bioavailability of a chemical in soil samples other than those prepared in the laboratory. As discussed elsewhere in this report, such soils may or may not reflect bioavailability of contaminated soils found in the environment.
To assess dermal bioavailability, a measured dose is placed on the skin. For experiments involving laboratory animals, the skin is shaved unless the animal is hairless (e.g., the nude mouse). A measured dose is applied to the skin, either in a liquid vehicle or solid matrix. The dose is left in place for a prescribed period (often 24 hours), and the amount absorbed is assessed in a variety of ways (that can include measurement of blood, urine, or tissue concentrations). Interpretation of results is analogous to that described above for oral bioavailability studies.
Another approach is to estimate absorption by measuring disappearance of the dose from the skin surface. After the exposure period, the applied dermal dose is removed and measured. Removal can consist of simply washing the skin and collecting the wash and rinse solutions for measurement, or may be more aggressive in the form of tape stripping. Strips of cellophane tape are successively applied to the skin in the dose area and removed, taking with them cells of the stratum corneum containing unabsorbed chemical. This may be repeated 20 or 30 times, and the amount of chemical on the tape strippings is then determined. If the chemical is radiolabeled, the amount remaining on the skin may be determined by placing a detector over the skin area and quantitating remaining radioactivity. Regardless of the procedure used to measure dose remaining on the skin, it is assumed that the amount of applied dose not recovered was absorbed.
The choice of where to apply the dermal dose is an important consideration. Generally, the dose is placed on an area that is most convenient for the investigator and offers the least potential for interference from the animal (e.g., from scratching or licking). In animals as in humans, the dermal permeability can vary with location on the body (see Chapter 3) and the results obtained from one area of placement may not be representative of dermal permeability elsewhere. Also, the choice of animal model is important. As discussed earlier under in vitro methods, the monkey and pig appear to be the best models for human dermal absorption, but are more expensive and can be more difficult to handle than smaller animals such as rats and mice. The disadvantage to using rats and mice is that their dermal permeability is usually much greater than human skin, and results obtained may therefore overpredict dermal absorption. One approach to
overcome this has been the use of a skin flap model in which human skin is grafted to a suitable animal host such as the nude mouse. The grafted skin maintains the morphological features of human skin, and the model can be used for up to six months.
The dermal absorption of different radiolabeled contaminants mixed with soil has been measured in rhesus monkeys (Wester and Maibach, 1998). Compared with delivery in acetone, dermal absorption of chlordane and pentachlorophenol was slightly reduced. More significant reductions were observed for DDT and benzo(a)pyrene. A reduction of more than 30 percent was observed for Aroclor 1242 when administered in soil compared to acetone, but essentially the same extent of absorption was observed for Aroclor 1254. A 30 percent reduction was consistently observed for both Aroclors in soil when compared with application of the doses in mineral oil.
The advantages and disadvantages of the use of animal surrogates for studying dermal absorption are the same as those described above for gastrointestinal absorption. However, the need to use radiolabeled compounds in most dermal studies in order to achieve adequate measurement sensitivity limits such studies to non-human subjects.
Clinical Studies
Oral, dermal, and even inhalation bioavailability studies are regularly conducted on humans by pharmaceutical researchers in the context of drug development. The employed methods parallel those described above in the section on whole animal studies. The conceptual approaches and techniques are the same, except of course that tissue sampling is precluded. Some specialized procedures, such as the technique for isolated segment permeability studies in the gut (Lennernas, 1998) can be fairly invasive.
In contrast, there has been almost no clinical study of the bioavailability of environmental contaminants in soils and sediments. There are several impediments to this type of research. It may be difficult to convince potential subjects and Institutional Review Boards that it is appropriate to intentionally expose healthy humans to environmental contaminants, even if assurances can be provided that the doses will be well below those associated with adverse health effects. Another impediment is analytical sensitivity. Bioavailability studies in pharmaceutical research can be performed with substantial doses of the chemical because the objective is to determine absorption under conditions of clinical use where effects from the drug are expected. For environmental contaminants, the doses must very low to avoid any possibility of effects. This means that the analytical methods must be able to reliably measure very low concentrations. There also must be some means to distinguish low administered concentrations from “background” levels in the body resulting from the subjects’ incidental environmental exposure to the chemical. One approach might be to use
radiolabeled chemical, but there are potential risks to the subject from exposure to radioactive material, and this would restrict the soils and sediments tested to those created in the laboratory rather than samples from contaminated sites.
One study of lead bioavailability from soil has taken advantage of the presence of naturally occurring stable isotopes of lead to avoid these problems (Maddaloni et al., 1998; see Box 4-6). In measuring the stable isotopes by mass spectrometry, adequate sensitivity could be achieved to follow gastrointestinal absorption of relatively small doses of lead in soil (approximately 200 μg). Also, using differences in stable isotope ratios that exist naturally, they were able to identify lead in the blood originating from the soil sample as opposed to lead from other sources. Unfortunately, the unique circumstances for lead (i.e., the existence of naturally occurring stable isotopes in different ratios in soils and individuals from different areas) make this approach difficult to reproduce for other chemicals.
Assimilation Efficiency
The equivalent of determining absorption in humans is also done in invertebrates and fish. A tool that quantitatively integrates processes in the gut that affect uptake is assimilation or absorption efficiency. Assimilation efficiency is defined as the fraction of contaminant absorbed by the gut (measured either in vitro for larger animals or in the whole organism for smaller animals) relative to the amount ingested (per gm weight food, per gm organism, per day). It is a direct measurement of biouptake in that it determines how much of the ingested contaminant is transported across a biological membrane. When assimilation efficiency is combined with feeding rate and concentration in the ingested material, the final concentration of a contaminant taken up can be modeled (Luoma et al., 1992; Wang et al., 1996a; Luoma and Fisher, 1997).
Assimilation efficiency is most applicable to benthos and water column dwelling organisms that ingest bed sediments or suspended materials that move in and out of sediments. Although the importance of this pathway to overall exposure has been somewhat controversial, a body of work supports the view that diet is consistently responsible for half or more of contaminant uptake by most organisms that ingest sediments, provided the experimental conditions are typical of nature (Landrum et al., 1992; Kidd et al., 1995; Reinfelder et al., 1998).
Because assimilation efficiency is a direct measurement of a biological mechanism (Luoma and Fisher, 1997), it is unambiguously comparable among species, contaminants, and environmental conditions. Thus, comparing assimilation efficiencies by a deposit feeder among sediments of different character is a way to determine how those sediment characteristics affect bioavailability. Assimilation efficiency is especially valuable for invertebrates because it is relatively simple to use, such that experiments can be conducted quickly.
|
BOX 4-6 Measurement of the Bioavailability of Lead from Soil in Humans Maddaloni et al. (1998) utilized the stable isotopes of lead that exist naturally to measure absolute bioavailability of lead from soils in humans. Lead (Pb) has four stable isotopes—204Pb, 206Pb, 207Pb, and 208Pb. Three of these isotopes (206Pb, 207Pb, and 208Pb) are produced continually by radioactive decay, and consequently the ratio of these isotopes varies from location to location with the geologic age of the lead deposit. Given the geographic differences in lead isotope ratios in soils, it is not surprising that lead isotope ratios in human blood can also vary with location. If individuals ingest soils with a very different lead isotope ratio than the one that exists in their body, the change in ratio after ingestion can be used to estimate the amount of lead absorbed from the soil. This study sought information on lead bioavailability from the soil at the Bunker Hill, Idaho Superfund site. The ratio of 206Pb/207Pb in this soil was 1.057. Twelve adult volunteers were selected from the New York area, all with a 206Pb/207Pb ratio > 1.190. The subjects were divided into two groups. One group received a soil dose (250 μg Pb per 70 kg body weight) after an overnight fast, while the second group received the same dose immediately after a standardized, high-fat breakfast. Blood and urine samples were collected from each subject over a 30-hour period after the dose. After the soil dose, blood lead concentrations increased while the 206Pb/207Pb ratio decreased. From the change in blood concentration and isotope ratio, the percent of the lead dose in the blood compartment of the subjects could be calculated. Among fasted subjects, this averaged 14.4 ± 4.5 percent of the administered dose. Data from a previous study indicated that at 24 hours after an intravenous 203Pb dose to human volunteers, 55 percent of the dose remained in the blood compartment. This indicated that the average absolute bioavailability of lead from the soil samples was 26.2 ± 8.1 percent (14.4 ÷ 0.55 = 26.2). For non-fasted subjects, the absolute bioavailability was much lower, averaging only 2.5 percent. Figure 4-7 shows how the isotope dilution technique was able to distinguish blood concentrations resulting from the soil lead dose from total blood lead concentrations, as well as differences between fasted (A) and fed (B) subjects. |
Until the last ten years, assimilation efficiencies were poorly known for aquatic organisms, although the concept has long been employed to study contaminant uptake in higher animals. Decho and Luoma (1994) and Wang et al. (1996a) first showed that repeatable assimilation efficiencies can be determined for sediment-bound contaminants under a variety of conditions and for a variety of species. There are few impediments to determining assimilation efficiency in any species that ingests sediments if the animal can be fed in the laboratory. The traditional approach in mammalian physiology, nutritional physiology, and more recently with invertebrates is to use radionuclides in pulse-chase experiments (Reinfelder and Fisher, 1991; Decho and Luoma, 1991, 1994; Luoma et al., 1992). Particles can be fed directly to the experimental animals or in any feasible matrix or configuration. Experimental conditions can be set up to eliminate the confounding influence of pore water contamination or desorbed contaminant and thus isolate the contribution of ingested material to overall bioavailability. In
vitro studies with excised gut are possible, especially with larger organisms; in vivo studies with whole organisms are used most frequently with invertebrates.
Recent studies show that assimilation efficiencies can vary widely among food types and among species, and some unexpected results have emerged. For example, bivalves appear able to absorb a substantial fraction (20 percent or greater) of what might be considered recalcitrant forms of trace elements from sediments. Both a deposit feeding bivalve Macoma balthica and the mussel Mytilus edulis absorbed 15–30 percent of sulfide-associated cadmium and silver from ingested particles (Lee et al., 2000). As discussed in Chapter 3, the form or source of the contaminant in the sediment or suspended material that deposit-feeding or detritus-feeding animals ingest also has a strong influence on assimilation efficiency.
A limitation to using assimilation efficiency to measure bioavailability is its dependence on radioisotopes. Exchange between the radioisotope and the stable

Laboratory set-up for measuring assimilation efficiency of clams.
chemical and whether the isotope fully reflects the chemical forms present in the sediment are critical considerations. (The same limitation applies to any spiking experiment.) High specific activity radioisotopes are sometimes difficult to obtain for some contaminants, and handling radioisotopes requires special precautions. Stable isotopes might eventually replace radioisotopes but will not have the
advantages of using gamma isotopes (for which non-destructive determinations are possible).
Models of exposure and bioaccumulation can incorporate assimilation efficiencies, such that the potential uncertainty associated with different sediment types can be determined. This may be especially useful in forecasting potential bioaccumulation under different conditions. (If applied to a specific field setting, however, the assimilation efficiency–model approach would still involve characterizing the sediment character in that setting.) As discussed later in this chapter, Dynamic Bioaccumulation Models (DYMBAM) use assimilation efficiency and seem to provide good estimates of uptake when compared to field results. These models may be an attractive next step (beyond the more empirical methods used to forecast bioaccumulation from sediments like BSAF) for quantifying the implications of considering bioavailability processes (Luoma and Presser, 2001).
Molecular Approaches
The remaining tests in this section have endpoints beyond initial biouptake and include molecular, cellular, and organismal responses to exposure to contamination. Because the success of bioavailability screening may hinge on the speed of the determination and on the reliability of the results, molecular tests are expected to have a significant impact on bioavailability assessment in the future. It is envisioned that bioavailability assessment protocols will require sufficient throughput capacity to be able to handle a relatively high number of samples and, hence, will be of relatively low fidelity. Moreover, such assays also will need to be economical to conduct. Molecular tools hold promise for providing such rapid and accurate assays of bioavailability that are modest in terms of expense (Dodi et al., 1999; Pennie, 2000). Indeed, the past decade has seen an explosion in the number and variety of techniques available for molecular analysis of exposure and toxicity and thus bioavailability, including molecular reporter systems and molecular biomarkers.
Molecular Reporter Systems using Bacteria
Whole cell bioreporters measure the intracellular response of a microorganism (typically bacteria) to an extracellular chemical or physical signal, and as such may provide an indirect measure of contaminant concentration and bioavailability. The attractiveness of a bioreporter system derives from the ease with which the signal, typically bioluminescence (light emission), can be measured. Further, because the response for many chemical-specific bioreporters is contingent on the contaminant molecule passing the microorganisms’ membranes, these assays may be relevant to other ecological receptors.
Most bioreporters are based on a variant DNA construct that is inserted into a bacterial strain, which will then be put in contact with a contaminated sample.
The DNA construct links genes that respond to the presence of a contaminant with a “reporter” gene whose output can be easily detected. The luxCDAB or luxAB cassette, a set of genes derived from the marine eubacterium Vibrio fisheri that results in luminescence, is frequently used (Meighen, 1991). Other commonly used reporter genes include luc, which encodes firefly luciferase; lacZ, which encodes β-galactosidase; and inaZ, which encodes the ice nucleation protein (Loper and Lindow, 1994). After an appropriate incubation time between the organism and the sample, the signal is recorded with a luminometer or other device (Reid et al., 1998; McGrath et al., 1999; Shaw et al., 2000). A very recent reporter gene that will see increasing use in biosensor development because of its ease of detection and minimal metabolic cost to the host cells encodes for the green fluorescent protein (GFP), originally isolated from the jellyfish Aequorea victoria (Tsien, 1998; Cha et al., 1999; Hansen and Sørensen, 2000; Joyner and Lindow, 2000; Stiner and Halvorson, 2002).
Depending on the DNA construct, bioreporters can have either narrow or intentionally broad contaminant specificity. In the former case, the reporter is designed to respond to a sole organic or inorganic chemical species such that response to a secondary species is considered undesirable. Most of these reporter systems are based on promoters and transcriptional regulatory proteins that respond to specific chemical species (e.g., Hg(II), arsenic, antimony, zinc, copper, cadmium, lead, toluene, and naphthalene) (Selifonova et al., 1993; Heitzer et al., 1992, 1994; Taurianen et al., 1998; Stiner and Halverson, 2002). For example, some bioreporters have been designed such that bioluminescence is switched on in the presence of the target contaminant and measured quantitatively. Insertion of the lux reporter system into P. fluorescens HK44 was reported to successfully provide real-time data of naphthalene bioavailability, degradative activity, and optimal degradative conditions (Heitzer et al., 1992).
If broader contaminant specificity is desired, the reporter is designed to capture the physiological response against a wide range of contaminants. These DNA constructs are either based on the use of constitutive promoter elements where luminescence can be seen as an overall measure of cellular activity (Rattray et al., 1990; Chaudri et al., 2000), or on promoter elements that are expressed systemically in response to cellular stress, e.g., the promoter of recN (van der Lelie et al., 1997) and various heat shock stress protein promoter elements (Cha et al., 1999). The original Microtox™ bioassay belongs to the latter group, because it measures the luminescent response of the marine Vibrio fisheri containing luxCDAB under control of its wild-type regulation (Meighen, 1991). Hence, toxicity or inhibition to the microorganism is inferred from a reduction in luminescence intensity against a control treatment (ASTM method D-5660-95). In this case, reduction in luminescent response is believed to serve as a general indicator of both inorganic and organic toxicant stress. GFP-based systems under control of constitutive promoters can be employed similarly to measure generic cytotoxic effects (Rabbow et al., 2002).
Although bioreporters have the potential of providing real-time data of compound availability, the short lifetime of the bacterial cells is not always optimal for developing biosensors capable of long-term, online monitoring of vapor and aqueous phase contaminants. Another key challenge for biosensors is their limited sensitivity, which is in the 1 nM range for some reported metals, toluene, and naphthalene (Selifonova et al., 1993; Taurianen et al., 1998; Stiner and Halverson, 2002), but may be orders of magnitude higher for others (Willardson et al., 1998). In addition, bioreporters can have limited genetic stability (Heitzer et al., 1992, 1994; Ripp et al., 2000), their results can be confounded by the effect of non-specificity (many metal-specific and arene-specific sensors suffer from this), there is substantial background response, and there can be strong matrix effects on signal response (Selifonova et al., 1993; Neilson et al., 1999). In general, luminescence- and fluorescence-based biosensors are an inexpensive and rapid technique that may become useful for evaluating the bioavailability of both organics and metals in the soil.
Most bioreporter systems rely on intimate contact between the reporter strain and the environmental matrix. Thus the tests typically employ soil or sediment extracts within which the bioreporting organism is suspended (although pore water that is reflective of in situ conditions might be employed) (e.g., Willardson et al., 1998; Rasmussen et al., 2000). A biosensor system that relies on a very small volume of extractant has been developed for assessing heavy metal toxicity of soils, sediments, and sludge (Bitton et al., 1996; Boularbeh et al., 1996). In the future, the use of fiber-optic devices may permit application in situ (Heitzer et al., 1994). Little validation of molecular bioreporter systems has been performed, except for the heavy metal biosensors for which the response was reasonably well correlated with phytotoxic (bean and tomato) and zootoxic (Eisenia fetida) end points across several different soils (Corbisier, 1999).
Biomarkers
Biomarkers represent responses of living organisms that may indicate exposure to contaminants, predict harm, or themselves be harmful effects (Timbrell, 1998). A biomarker is a biochemical, physiological, or morphological response (usually on a molecular level), but not a population or ecosystem bioindicator (Stegeman et al., 1992). At minimum, a biomarker response discloses that a contaminant (1) is present in the environment, (2) is available to the organism, and (3) has reached the affected tissue or organ in sufficient amounts for a period of time long enough to produce an observed response (Depledge et al., 1993). An impetus for developing biomarker techniques for measuring contaminant bioavailability is that the measurement of parent toxicants or metabolites in biological samples (e.g., urine or blood) is currently limited to about 100 chemicals or classes of related compounds. Less than half of these can be quantitatively related to exposure.
Biomarkers have been categorized as biomarkers of exposure or effect. A biomarker of exposure indicates the presence of a xenobiotic substance or its metabolite(s) or is the product of an interaction between a xenobiotic agent and some target molecule or cell (such as the formation of a macromolecular adduct) (DeCaprio, 1997). Biomarkers of exposure are mainly useful in establishing contaminant dose in both ecological as well as human studies where they provide information about long-term exposure to carcinogens (Waterfield and Timbrell, 2000). Generally, these biomarkers reflect recent exposure, although the half-life of the contaminant must be taken into account. Biomarkers of exposure are the most convenient to determine. For example a contaminant or its metabolites often can be quantified from samples of blood, breast milk, feces, or urine, as well as tissues obtained through biopsy or necropsy (Fossi et al., 1994). While macromolecular adducts do provide some degree of specificity and sensitivity, they are expensive to evaluate and not always quantitatively related to exposure.
Biomarkers of effect indirectly indicate exposure and are defined as any measurable biochemical, physiological, or other alteration within an organism that can be recognized as an established or potential health impairment or disease (Hugget et al., 1992). This includes induction of proteins (e.g., metallothioneins and heat-shock proteins). Markers that are the result of pathological damage can be considered separately from markers that indicate a metabolic lesion. Clinical or behavioral observations can also be considered a separate type of biomarker. Biomarkers of effect vary markedly in their specificity, sensitivity, usefulness, and feasibility. A consideration for the well-documented biomarkers (e.g., metal-lothioneins and stress proteins) is that they are “general” responses induced by exposure to a variety of compounds. This can be an advantage in situations where the total biological response, for example from a mixture of contaminants, is the preferred endpoint.
The use of biomarkers to further mechanistic understanding is made difficult by the fact that biomarkers indicate the cumulative effects of chemical interactions and reflect a temporal and spatial integration of exposures. Ideally, a suite of biomarkers would be needed to observe different classes of chemicals. Biomarkers could be used as early warnings to detect exposure shortly after it has occurred. However, their use over long periods of time may be hard to interpret unless more is known about the duration of the actual response. The specificity of biomarkers decreases with an increasing level of organization such that molecular biomarkers are more specific than organ- or organism-level ones. Finally, it can be hard to relate lab results to the field because of interspecies differences and ecological impacts. As discussed later, gene expression technology holds great promise in complementing more “general” biomarkers to further mechanistic understanding. [Interestingly, even though data are being assembled on gene expression during exposure to specific contaminants (see following section), the number of genes whose activation has been linked to a biomarker is minor. Ideally, activation of a gene or gene cluster would signal a specific response related to the appearance of a biomarker.]
Four prominent examples of molecular biomarkers with potential applicability to bioavailability processes are stress proteins (see Box 4-7), DNA damage, metallothioneins, and cytochrome P450 activity. The more common methods for determining DNA damage are (1) direct measurements of DNA structural damage, (2) assessment of DNA repair, or (3) determination of mutations present (Shugart et al., 1992). Metallothioneins are a class of small proteins that are rich in cysteine, capable of binding metal ions, and inducible by cadmium, copper, mercury, zinc, cobalt, bismuth, nickel, and silver ions (Waterfield, 2000). The P450 cytochromes are a class of hemoproteins present in a wide variety of organisms and in all tissues in mammals, especially the liver. They are inducible by a variety of organic chemicals (De Caprio, 2000). Although several sensitive assays have been developed for quantifying cytochrome P450 induction (Saint-Denis et al., 1999), the appropriate assay conditions and specificity of response must be ascertained for each species.
|
BOX 4-7 Heat Shock Proteins as Biomarkers One of the best-studied biomarkers of exposure is production of stress proteins (Bierkens, 2000). A wide variety of organisms from bacteria to humans produce proteins that provide some protection from cellular damage (Hightower et al., 1985; Hightower, 1993; Morimoto et al., 1995a,b; Hartl, 1996). These proteins, initially described in fruit fly cells during exposures to high temperature (Ritossa, 1962), are termed “heat shock proteins” (hsp). Since the initial discovery, a range of environmental stresses has been shown to induce heat shock proteins; thus, the term “stress protein” consequently has been coined. Environmental contaminants that can induce these proteins include both trace metals (Sanders et al., 1991; Bauman et al., 1993; Williams et al., 1996) and organic compounds (Sanders, 1990). There are several families of heat shock proteins classified by molecular weight: Hsp90, Hsp70, chaperonin, and those of low molecular weights. An increase in the total specific activity of Hsp70 within an organism can be used as a nonspecific indicator of stress, exposure, and potentially bioavailability. Nadeau et al. (2001) demonstrated that stress-induced Hsp70 could be used to monitor exposure of the earthworm species Lumbricus terrestris to various soil contaminants. The midgut and intestinal tissues of L. terrestris revealed expression of an inducible member of the Hsp70 family after heat shock treatment in vitro (positive control) and after exposure to different toxicants in artificial soil. Short-term (24–72 hours) and long-term (14–16 days) exposure to chloroacetamide and pentachlorophenol as well as heavy metals (Pb+2, Gd+2, Cu+2, and Hg+2) in soil induced Hsp70 in the earthworms’ midgut and intestinal tissues. This biomarker appears to be sensitive with a good level of reproducibility despite some individual variations. The use of non-exposed animals transposed into contaminated environments should be highly relevant to bioavailability studies. Stress proteins do have some selectivity (Ait-Aissa et al., 2000), as not all contaminants induce a stress response. However, among those contaminants that do induce expression of Hsp70, the potency of induction was related to the octanol–water partition coefficient. |
Fouchecourt et al. (1999) and Koganti et al. (1998) evaluated the bioavailability of PAHs from ingested soil via the measurement of several different exposure biomarkers in target organisms, including whole body or organ burden of the toxicant, measurement of cytochrome P450-dependent monooxygenase activities, urine levels, and chemical:DNA adduct levels in lungs. The gastrointestinal absorption and systemic bioavailability of PAHs was determined for soil containing complex organic mixtures. The results of the biomarker assay were compared to PAH bioavailability as measured with soil and organic extract of each soil (Soxhlet) to give a relative bioavailability value for each soil type. In another study (Fouchecourt et al., 1998), rats maintained on a litter of PCB-polluted soil were used to assess bioavailability. PCB burdens and activities of microsomal liver and lung cytochrome P450 monooxygenases were the biomarkers assessed. A near dose-response relationship was found between concentrations of PCB in the litter and activity of the monooxygenase EROD in both the liver and lungs. This suggests that EROD activity measurements in both liver and lung of rats maintained on a litter of PCB-polluted soil can be used to assess the bioavailability of PCBs to mammals.
Gene Expression Techniques. Like other biomarkers, gene expression techniques quantify a molecular response to contaminant exposure. In this case, the response is an alteration in gene expression at the level of transcription, detected by making real-time measurements of particular messenger RNAs. The underlying scientific basis is that an organism’s contaminant exposure is manifested in (among other things) creation of unique mRNAs that direct protein manufacture and other cellular responses. Such genomic biomarkers have the potential of acting as a toxicant-specific or at least toxicant class-specific “fingerprint” of chemical bioavailability.
Specific techniques can be divided into two types: high fidelity, low-through-put techniques and low fidelity, high-throughput techniques. Microarrays, subtractive hybridization, and serial analysis of gene expression (SAGE) are high-fidelity assays in that they generate considerable information about an organism’s genetic response to exposure. However, because of the amount of information generated, as well as the number of steps involved, the amount of mRNA required, and the costs, these techniques cannot currently be used for high-through-put screening. More rapid flow-through DNA hybridization array or “genosensor” systems are being developed (Fredrickson et al., 2001) that hold great promise in the area of soil and sediment bioavailability. All these techniques are described in detail in Box 4-8.
Microarrays have generated considerable interest in toxicology and thus indirectly for use in studying bioavailability. Using microarray techniques, it is possible to develop a sensitive and inclusive snapshot of the responses of cells, tissues, and organisms to a contaminant without the time requirements, labor, or subjectivity of more traditional analyses. Validating these techniques, and in-
creasing their practicality for specifically assessing contaminant bioavailability from soils and sediments, should occur in the near future.
Organismal Approaches
A variety of bioassays at the level of individual organisms can be used both to assess bioavailability of contaminants in soils and sediment and to validate the physical and chemical tests discussed earlier in this chapter. Bioassays generally can be divided into two basic categories. First, uptake tests directly reflect how much of a contaminant is in the tissue of an organism. Such tests are commonly conducted in plants, invertebrates, and fish, and sometimes in birds and mammals when these organisms are an ecological receptor of concern. For obvious reasons, uptake tests are generally not feasible in microorganisms. Uptake tests generally do not take into account whether the compound of interest is transformed within the body of the organism. The most common measurement is simply the concentration of the compound in the tissue of interest, and when this is measured after a prolonged exposure, the test is referred to as a bioaccumulation test. The second major type of bioassay is a toxicity test to determine what concentration of a compound brings about some toxic effect, such as suppressed growth or death. Following a brief discussion of microbial mineralization assays, this section is organized by organism, with both uptake and toxicity tests discussed for each.
Mineralization and Assimilation Assays for Microorganisms
Microbial mineralization (and concomitant CO2 evolution) has been applied to assess bioremediation potential of soil-bound HOCs and hence their bioavailability to microorganisms. In this assay, the initial mineralization rate and extent in soil-slurry experiments supplemented with a HOC mineralizing strain are compared with soil-free controls to estimate bioavailability reduction (Guerin and Boyd, 1997; Feng et al., 2000). Such measures for estimating qualitative trends in contaminant availability to microorganisms have long been employed by Alexander and coworkers. Employing 14C-labeled compounds, reduction in degree of mineralization of PAHs and other compounds was linked to bioavailability reduction (e.g., Chung and Alexander, 1998, 1999; Hatzinger and Alexander, 1998; Tang et al., 1999; White et al., 1997, 1999).
Because the method relies on the use of 14C-labeled marker compounds, it is not suitable to measuring mineralization of the relevant aged contaminants, nor can it be conducted in soils or sediments in situ. Further, different bacterial strains may yield different results, suggesting that strains have varying abilities to degrade solid phase-associated compounds (Guerin and Boyd, 1992; Friedrich et al., 2000; Grosser et al., 2000).
A wholly empirical but promising technique that estimates the microbially oxidizable fraction of soil- and sediment-bound hydrophobic organic compounds
|
BOX 4-8 The Pros and Cons of Gene Expression Techniques The basis of gene expression techniques is that an organism’s contaminant exposure is manifested in the creation of unique messenger RNAs. Thus, most of these techniques revolve around detection of specific mRNA species through the use of hybridization techniques. Membrane-based microarray assays (the forerunner of current hybridization microarrays) employ membrane filters onto which are adsorbed thousands of cDNA sequences related to various aspects of cell regulation. (cDNA is complementary DNA formed using messenger RNA as a template and the enzyme reverse transcriptase.) Such membranes are available from several commercial sources at reasonable cost (Cheung et al., 1999). mRNA from two different cells (test and control) thought to respond to contaminant exposure is then hybridized onto the two filters. In this way, the exact genes that are being turned on in response to the exposure can be identified. Glass slide-based microarray technology is an even faster and more efficient microarray setup, for a variety of technical reasons. Subtractive hybridization is another high-throughput technique that allows for the isolation and cloning of mRNA unique to an exposed population. Pennie et al. (2001) have shown that cDNA microarrays allow comprehensive coverage of genes associated with entire pathways (such as oxidative stress, signal transduction, and stress response). Tully et al. (2000) used the CAT-Tox(L) assay system (which utilizes human liver carcinoma cells) to examine patterns of gene expression to several heavy metals. Similarly, Liu et al. (2001) used the Atlas Mouse Stress/Toxicology array to observe alteration of gene expression related to stress, DNA damage, and metabolism in mice following acute arsenic treatments. As an example, DNA arrays containing 148 genes for xenobiotic metabolizing enzymes, DNA repair enzymes, heat shock proteins, cytokines, and housekeeping genes were used to examine gene expression patterns in the livers of mice in response to exposure to cadmium chloride (CdCl2), benzo(a)pyrene (BaP), and TCE (Bartosiewicz et al., 2001a). Each toxicant was found to produce a unique pattern of gene induction or “fingerprint.” Exposure to CdCl2 resulted in marked up-regulation of metallothionine I and II, several of the heat shock-stress response proteins, and early response genes. In contrast, exposure to BaP lead to up-regulation of only metabolizing enzymes Cyp1a1 and Cyp1a2 genes and produced no significant increases in any of the stress response genes or the DNA repair genes present on the array. Exposure to TCE was shown to induce gene expression of the heat shock proteins Hsp 25 and 86 as well as Cyp2a. |
entails gentle persulfate (S2O82–) oxidation (Cuypers et al., 2000). This method assumes the soil organic matter oxidized under the applied conditions is the primary source of readily available HOC. Thermal gravimetry analysis confirmed that oxidation indeed removed the 250–350ºC labile organic matter phase (Bierkens et al., 1998). Results from this assay for soil- and sediment-bound PAHs correlated well with batch biological oxidation after 21 days and Tenax TA extraction after 264 days. Hence, this assay may correlate with prokaryal intracellular availability. However, it should be note that this method is not itself a mineralization assay but rather a physicochemical test that is biomimetic.
|
Bartosiewicz et al. (2001b) expanded on this study by looking at both liver and kidney of mice exposed to five classes of chemicals (PAHs, DNA alkylators, peroxisome proliferators, heavy metals, and oxidative stressors). Each toxicant group gave a similar pattern of gene expression in the liver and kidney, which was dissimilar from that of the other four toxicant groups, with both time and dose being important to class differentiation. Using in vitro techniques, Waring et al. (2001a) investigated whether chemicals with similar mechanisms of toxic action produced similar changes in gene expression. They treated rat hepatocytes with 15 known hepatoxins (carbon tetrachloride, allyl alcohol, aroclor 1254, methotrexate, diquat, carbamazepine, methapyrilene, arsenic, diethylnitrosamine, monocrotaline, dimethyl-formamide, amiodarone, indomethacin, etoposide, and 3-methylcholanthrene) and used microarray technology to characterize alterations in gene expression. Results revealed that gene expressional profiles for toxicants with similar toxic mechanisms formed clusters, suggesting a similar effect on transcription. However, each toxicant produced a unique fingerprint. Along the same lines, Waring et al. (2001b) showed that gene expression changes caused by an agent in vitro reflected those produced in vivo. These findings suggest that microarray analysis with a focused set of genes might be capable of discriminating exposure to, and thus bioavailability of, different toxicants. Other potentially useful gene expression tools include SAGE analysis (Velculescu et al., 1995; Bertelsen and Velculescu, 1998) and differential display (Liang and Pardee, 1992). SAGE quantifies the level of RNA in each individual cell population. With differential display, mRNA samples from several samples can be analyzed at the same time, which is not possible with other techniques. Both SAGE analysis and differential display have the ability to identify previously unknown genes that may be expressed upon exposure. Several researchers have used differential display to identify genes expressed with exposure to certain toxicants (Wang et al., 1996b; Selmin et al., 1996; Kegelmeyer et al., 1997; Donat and Able, 1998; Muhlenkamp and Gill, 1998; Roman and Peterson, 1998; Rodi et al., 1999). Finally, real-time polymerase chain reaction (Higuchi et al., 1993), scintillation proximity (Harris et al., 1996), and branched-DNA (Waring and Ulrich, 2000) are molecular techniques used to follow the responses in one or a few genes that could be constructed as high-throughput, albeit low-fidelity, screens. |
Plant Bioassays
Plant bioassays can be used to measure bioavailability processes for a range of organic and inorganic compounds in soils. Two types of results can be generated. First, plant tissue can be analyzed to determine if the contaminants of concern are present at elevated or potentially toxic levels. It is relatively straightforward to analyze plant tissue for concentrations of toxic inorganic contaminants. The second approach is to measure of the growth and vigor of the plant. If the plant can grow in the presence of a contaminant, then it is possible to conclude that the contaminant is not present in phytotoxic concentrations. For both
types of assays, the results can be used either to determine the bioavailability of contaminants to plants and to organisms that consume the plants, or to estimate bioavailability of the contaminants to other organisms (assuming a correlation between plant and animal uptake can be shown).
This type of testing has been routinely done in agriculture for decades, and has been used to validate many of the extraction tests discussed earlier in the chapter (Leschber et al., 1984; O’Conner, 1988). For example, growth tests are commonly used to better understand the bioavailability of herbicides, and tests that measure plant tissue concentrations are routinely conducted to evaluate plant nutrient status. Tests have most often focused on identifying plant deficiencies of particular elements but are easily adapted to evaluate toxicities (Gettier et al., 1985). Plant uptake has been used to evaluate the effect of soil contamination as well as the ability of in situ treatments to reduce those effects (Pierzynski and Schwab, 1993; Chaney and Ryan, 1994; EPA, 1995; Laperche et al., 1997). When used appropriately, plant tissue analysis can provide a quantitative assessment of bioavailability process D in Figure 1-1.
Appropriate methods for plant sampling and analysis have been outlined for a range of agronomic crops (see Westerman, 1990; Rayment and Higginson, 1992; Kalra, 1998). Field studies are generally conducted for a minimum of two growing seasons, while controlled environment studies often involve multiple harvests to mimic changes over time. Depending on the goals, typical measured responses include visual symptoms of toxicity and deficiency or simply above-ground biomass. Analysis of plant tissue for total elements from samples collected at sites of concern is generally inexpensive with a turn around time of one to two weeks. Bioassays using plants in field studies or in controlled environment studies are considerably more expensive as well as time consuming. There are experimental artifacts in metal uptake results obtained in plants grown in pot versus field studies (deVries and Tiller, 1978) in that plants grown in pots with access to a limited volume of soil will have increased metal concentrations when compared to field studies.
Appropriate use of plant bioassay data must take into account characteristics of the plant species tested, the experimental conditions, the role of the contaminant of concern in plant nutrition, and how the contaminant of concern may have interacted with necessary plant nutrients to cause imbalances. For example, metals spiked to soil as salts are generally much more phytoavailable than comparable concentrations added in municipal biosolids or present in historically contaminated sites (Brown et al., 1998). Pot studies using metal salts may greatly overestimate uptake into plant tissue in situ (Logan and Chaney, 1983; Page et al., 1987; Sauerbeck, 1991). It is also important to understand the mechanism by which the contaminant is most likely to cause negative health effects. In certain cases, contaminants may not be phytotoxic, but they may be accumulated in sufficient concentrations in edible plant tissue to cause negative health effects to consumer populations.
Metals. The utility of plant bioassays for measuring bioavailability of metals is dependent on the particular metal, its route of uptake, and its potential mode of toxicity. For example, zinc, nickel, copper, and manganese toxicities have been reported for plants growing under field conditions. For these elements, the potential toxicity to plants must be considered in any evaluation of bioavailability. For other elements, their concentration in plants does not vary significantly even with changes in soil concentration that span orders of magnitude. For even other elements, consumption of enriched forages is the primary pathway through which these elements can enter the food chain and cause harm. Here, plant tissue concentration is a viable means of measuring bioavailability to higher organisms, even though plant yields may not be impacted. Specific examples of each of these cases follow.
Cadmium, lead, arsenic, chromium, and cobalt are not generally phytotoxic, even in cases of severe soil contamination in the field. Furthermore, lead, arsenic, chromium and cobalt are generally not taken up by plants in readily measurable quantities (Xu and Thornton, 1985; Chaney and Ryan, 1994; McGrath, 1995; Chaney et al., 2000). When these four metals have been found to be toxic to plants, uptake was generally confined to root tissues; thus, measurements of plant shoot concentrations are not useful. For lead, arsenic, chromium and cobalt, plants are not the most sensitive species, and consumption of contaminated plant material is not a relevant exposure pathway for higher organisms.
For other elements, consumption of foodstuffs with elevated metal concentrations can be an important exposure pathway, although the metals are not toxic to plants themselves. For example, consumption of plants containing elevated concentrations of cadmium has resulted in human fatalities (Kobayashi, 1978). Although plant concentrations alone are not sufficient to determine if consumption of cadmium-enriched foodstuffs will result in negative human health effects, they are an important indicator of bioavailability in a soil system (Chaney et al., 1999). In the case of selenium and molybdenum, uptake into the edible portion of plant tissues is generally not sufficient to cause plant toxicities but has lead to toxicities of animals consuming enriched plant tissue (Foy et al., 1978; Bingham et al., 1986; McGrath, 1995). Thus, for cadmium, selenium, and molybdenum, measuring plant uptake from soil is a means to evaluate their bioavailability to higher organisms. It should be remembered when sampling plants as part of ecological risk assessment that wildlife species may feed on different plant parts, which may accumulate contaminants to different degrees.
Plants are considerably more sensitive than other organisms to manganese and particularly zinc. Indeed, phytotoxicity of zinc is one of the primary concerns of excess zinc in soils. It is not surprising, then, that plant uptake of zinc has been identified by EPA as the controlling pathway for setting maximum permissible zinc concentrations for biosolids applied to land (Chaney et al., 2001). Because zinc will kill plants at concentrations lower than those generally associated with negative health effects in animals, plant phytotoxicity effectively prevents trans-
fer of soil zinc through the food chain (Chaney and Ryan, 1994). Plant zinc concentrations are also effectively used to measure changes in bioavailability as a function of soil treatment with different amendments, such that reduction of plant zinc following amendment is accepted as evidence of the reduced bioavailability of the metal (Basta and Sloan, 1999; Brown et al., 2000).
It is important to understand that there is not a single metal concentration that is associated with growth suppression and phytotoxicity across all plant species. For example, concentrations of zinc in plant tissue associated with phytotoxicity vary greatly both within and across species. Twenty varieties of soybean (Glycine max L.) grown on the same high zinc soil, were found to have different uptake as well as yield response (White et al., 1979). Four barley cultivars (Hordeum vulgare L.) grown under identical conditions had plant zinc concentrations ranging from 52 to 126 mg kg–1 (Chang et al., 1984b). Values for toxic concentrations have been reported to range from 200 mg kg–1 (Bingham et al., 1986) to 500– 1500 mg kg–1 (Chaney et al., 2000). For metal concentrations to be effectively used as a measure of bioavailability, it is important that the threshold values of the plant tested are well understood. In addition, toxicities of certain elements are associated with deficiencies of others. For example, zinc, copper, and nickel toxicities can be associated with iron deficiencies (Bingham et al., 1986), while lead and zinc toxicities can also be related to phosphorus deficiencies (Laperche et al., 1997; Brown et al., 1999, 2000). Behavior of plant species in response to nutrient deficiencies varies, and this response can affect the uptake of potentially toxic elements (Marshner, 1998).
Organics. Use of plant bioassays to assess the bioavailability of organics in soils to plants and higher animals is not well documented. Plant germination and growth tests are routinely used to evaluate the efficacy of herbicides in soils, but these organics were developed specifically to be available to certain classes of plants. Plant assays have been used to measure the uptake and toxicity of explosives (Anderson et al., 1999; Gong et al., 1999a,b; Krishan et al., 2000). But for the majority of organic contaminants, use of plant assays is limited and generally not well accepted because plant uptake of such compounds tends to be minimal (perhaps because they are not required for growth) (Chaney et al., 2001). Organics must be present in the transpirational stream to be taken up by plants, which is unlikely for compounds of limited solubility. Hydrophilic compounds cannot easily pass through the lipid portion of the root’s plasma membrane (Marschner, 1995; Burken and Schnoor, 1998), while more hydrophobic compounds can penetrate the membrane but are prohibited from entering the xylem and being translocated to shoot tissue. These compounds are generally bound to the mucigel at the root surface or to the lipid membranes of the root cell walls.
Measurement of organic compounds in plant tissue also presents analytical difficulties. Radiolabeled compounds are often used (Burken and Schnoor, 1997), such that pure compounds as well as metabolites can be detected in plant tissue.
It is not always clear if compounds are metabolized once they have entered the plant tissue, although axenic plants and cultures have been used in an attempt to clarify this process (Hughes et al., 1996; Newman et al., 1997). While these studies have shown that certain species are capable of metabolizing compounds internally, the possible contributions of rhizosphere and endophytic microorganisms must also be emphasized and has generally not been assessed (Banks et al., 1999; Siciliano et al., 2001). Because of these factors, plant bioassays of organic compounds are currently not viable tools for understanding bioavailability processes.
Invertebrate Bioassays
Soil Bioassays. A variety of tests are available to determine the uptake of organic compounds and metals and their toxicity to soil invertebrates (Løkke and van Gestel, 1998). These tests can be conducted with site-specific soils, or spiking studies can be used to predict the potential accumulation from soils that do not currently contain the contaminants of interest. The measured endpoints of soil bioassays include tissue accumulation, altered growth and reproduction, and mortality. Thus, such studies can be either direct measures of biouptake (bioavailability process D) or more indirect measures (that capture bioavailability processes D and E).
Invertebrate tests have been developed for representatives of the major terrestrial groups including protozoa, nematodes (Caenorhabditis), annelids (earthworms), crustaceans (isopods), and various insects (Bierkens et al., 1998; Løkke and van Gestel, 1998). However, studies have focused mainly on earthworms and the springtail (collembolan) Folsomia candida (Cortet et al., 1999). The terrestrial oligochaetes such as Lumbricus terrestris, Eisenia foetida, or E. andrei typically live burrowing through the organic rich upper third of a meter of soils. Moreover, they are well studied, easy to rear, reproduce rapidly, and are an important part of most soil ecosystems, thus making them an excellent candidate for soil bioavailability studies.
Several tests have been devised to assess the effects of contaminants on earthworms, including bioaccumulation, survival (OECD, 1984), reproduction and growth (van Gestel et al., 1989; Gibbs et al., 1996; ISO, 1996; Holmstrup, 2000), avoidance (Yeardley et al., 1996), as well as a range of immunological (Chen et al., 1991) and biochemical (Arnaud et al., 2000) parameters. In the growth and reproduction studies, adult earthworms are weighed prior to being incubated in food-supplemented test soils for varied periods of time (21–56 days). At the end of exposure the worms are collected and reweighed. In addition, the soil is washed through 2- and 1-mm sieves, and the cocoons collected and counted. Such tests have been applied to contaminated soils (Robidoux et al., 2000; van Gestel et al., 2001). The E. foetida 14-day artificial soil test (OECD, 1984) has been adapted to L. terrestris and used in assessing mortality, morbidity, and
whole worm burden of contaminated soils (Callahan, 1991; Chang et al., 1997; Charrois et al., 2001). While typically there is increasing sensitivity in going from survivability to biochemical indicators, there is also increased individual variability, with responses often being attributed to non-toxicological events (e.g., handling) (Arnaud et al., 2000).
The springtail (F. candida) assay is a 28-day soil assay that evaluates survival, growth, and reproduction (ISO, 1999). Growth is determined by comparing pre- and post-exposure weights. Survival and reproduction are determined by “floating” the springtails out of the soil after exposures. An image of the floating specimens is captured, and from this image the numbers of adults and juveniles can be determined. This assay, while more labor intensive, has been applied to contaminated soils (van Gestel et al., 2001).
Nematodes, especially C. elegans, are good biomonitors of soils (Donkin and Dusenbery, 1993). While mortality and to a lesser extent development are the common endpoints with this species (Peredney and Williams, 2000), more recent work has focused on the development of transgenic strains with heat shock proteins (Stringham and Candido, 1994) or metallothioneins (Cioci et al., 2000) linked to the β-galactosidase (lac-Z) reporter gene. As noted by Drobne (1997), a variety of endpoints has been developed for terrestrial isopods (e.g., woodlice).
There are a number of factors to consider before deciding whether to use in situ or laboratory soil invertebrate bioassays. A major advantage offered by laboratory measurements of invertebrate bioaccumulation is the greater control that the investigator can exert over the test. For example, a field-collected soil can be mixed in the laboratory to help reduce variability in concentrations (if such variability is not the focus of the investigation) or create soils with a desired contaminant concentration. Potential disadvantages of laboratory tests are numerous, and mainly concern the representativeness of the test results to field conditions. Laboratory-maintained cultures of invertebrates may differ from indigenous biota in their ability to tolerate or accumulate contaminants, with evidence that some field populations develop resistance and can accumulate toxins to a greater degree than laboratory counterparts (Ron Chekai, personal communication, 2002). A related issue concerns whether lab experiments are conducted long enough for conditions to reach steady state. It appears that in some cases exposure duration on the order of a few weeks to a month is adequate (Edwards and Jeffs, 1973; Stafford and Edwards, 1985). An important consideration with laboratory tests is the extent to which whole, “undiluted” field soils are used rather than field soils diluted with a reference soil of similar physical characteristics (usually collected from or near the site of contamination) or a standard laboratory reference soil. While such dilution provides a greater degree of control, the modifications produced must be taken into account (e.g., mixed soil is more analogous to an emulsion of particles with higher and lower contaminant concentrations rather than a homogenous mass of particles of intermediate contaminant concentration).
Box 4-9 describes the use of bioaccumulation measurements in earthworms to determine the soil or sediment availability ratio, which is a crude measurement of the relative bioavailability of fresh versus aged contaminants in soil.
Sediment Bioassays. Both laboratory and field studies of contaminant bioavailability from sediments are conducted with benthic invertebrates. This information can generally be obtained rapidly and applied directly to predictions of direct exposure or food chain bioaccumulation. A range of tests is available to directly determine the uptake and accumulation of organic compounds and metals and their toxicity to invertebrates, in this case aquatic organisms (Giesy and Hoke, 1989; Burton, 1991). These tests can be conducted with historically contaminated field sediments or field or clean sediments that have been spiked to contain the contaminants of interest. Like the soil invertebrate bioassays, the measured endpoints of sediment bioassays include tissue accumulation as well as functional responses such as altered growth and reproduction and mortality.
|
BOX 4-9 Soil or Sediment Availability Ratios The soil or sediment availability ratio (SARA) method uses organisms to estimate the relative bioavailability of aged versus unaged organic and inorganic chemicals in soil or sediment (Sijm et al., 2000). This is accomplished by exposing organisms to soil or sediment with freshly added chemicals and to soil or sediment with aged chemicals, both at similar total concentrations. The resulting ratio of the concentrations in the organism gives the relative bioavailability. The most frequently used biological system for SARA involves earthworms, particularly Eisenia foetida (Haque and Ebing, 1988; Van Gestel and Ma, 1988; Van Gestel and van Dis, 1988; Belfroid et al., 1995; Kelsey and Alexander, 1997; Chung and Alexander, 1999; Morrison et al., 2000). Using SARA, Belfroid et al. (1995) measured the relative bioavailability of hydrophobic compounds present for more than 20 years in a field-contaminated soil. They demonstrated that the aged compounds had a bioavailability of < 3 percent relative to the same chemical freshly added to soil samples. This method has also been used to show that select PAHs aged for only three months in sediment had similar bioavailability as freshly spiked sediment (Kraaij et al., 1998). This method has substantial potential for the indirect evaluation of bioavailability of chemicals that have been present in the soil or sediment for an extended period of time and on how contaminant association with the solid phase changes over time. It should be recognized, however, that SARA relies on the comparison of organismal concentrations of contaminant from two different scenarios, for which several bioavailability processes may be different. Hence, the inferred “bioavailability” number should be considered a lumped estimate of several bioavailability processes rather than measurement of an individual process like solid partitioning or gastrointestinal absorption. Validation of the technique has not occurred, probably due in part to the difficulties in comparing results from different tests. |

Set-up for measuring bioaccumulation and toxicity of contaminated sediments to invertebrates using material from Hunter’s Point, CA.
Uptake bioassays directly determine the biologically available fraction of compounds associated with sediment (EPA, 2000a,b), that is, they measure the potential for chemicals to partition into benthic biota or other aquatic organisms. These tests are often used to calculate Biotic Sediment Accumulation Factors (BSAF), discussed in Chapter 2. Such bioassays are most appropriate when the contaminants are not toxic to benthic invertebrates but can be bioaccumulated and biomagnified up food chains or through food webs to the point where they may be chronically toxic to higher-order organisms. This is the case for a range of organic compounds such as PCBs and chlorinated dioxins. Benthic invertebrates are tolerant of exposure to these compounds because such invertebrates do not contain the aromatic hydrocarbon receptor through which the critical toxic effects are mediated.
Uptake bioassays can be conducted under either field or laboratory conditions and on field-collected sediments or on spiked sediments (Giesy and Hoke, 1990). Field studies with indigenous invertebrates and in situ studies where caged organisms of a known history are placed in the field are more realistic measures of the biologically available fraction than laboratory studies (Salazar et al., 1995; Chappie and Burton, 2000; Burton et al., 2000). In situ bioassays with benthic invertebrates are particularly relevant to understanding contaminant bioavailability, because these organisms often feed on sediments or sediment-associated meiofauna or algae. Thus, contaminants in their tissues reach steady-state concentrations more quickly than for higher-order organisms, such that studies can be conducted for shorter duration. It is important to determine if the confined animals have sufficient natural food items available to sustain them (likely for most sediment-dwelling organisms); if they do not, an accurate measure of uptake and accumulation of sediment-bound chemicals is not obtained. In addition, when in situ tests are used, it is critical to have reached steady-state tissue con-
centrations of the contaminant of concern, or at least know what proportion of steady state has been obtained.
The greatest limitation of laboratory bioassays using spiked sediment is the questionable ability of the experimental conditions to mimic field conditions. For a variety of reasons (see Chapter 3), chemicals bound in sediments can take a long time to reach a steady state—a situation that may take months or years to simulate using laboratory-spiked sediments. In addition, adding exogenous chemicals to sediments will disrupt the dynamic equilibrium of other chemicals in the sediment and can result in estimates of accumulation that are different from what would be observed for an undisturbed sediment. Similarly, when laboratory studies are conducted with sediments collected from the field, the manner in which sediments are handled can affect the bioavailability of organic compounds and metals in the sediment (EPA, 2001).
Uptake and bioaccumulation of contaminants from sediments has been studied in a range of test designs with a number of species including invertebrates from both freshwater and saltwater environments (EPA, 1994, 2000c). Some of the most useful protocols for the study of benthic invertebrates are for worms, benthic insects, and mollusks such as clams (EPA, 1987a,b). One limitation of both field and laboratory tests that utilize small organisms is obtaining accurate estimates of concentrations and particularly weights or lipid contents for normalization. Also, it is important to be able to separate the material that is actually accumulated into organismal tissue relative to that which is simply adsorbed to the surface. Protocols for conducting both toxicity studies and bioaccumulation studies have been suggested, and some have been adopted by state, provincial, and federal agencies or international bodies (Boese and Lee, 1992). A comprehensive discussion of the theory and issues involved in the standard tests for sediment toxicity testing and determination of bioaccumulation of contaminants from freshwater sediments to benthic invertebrates, as well as specific guidance for test methods, has been provided by EPA and thus is not discussed here (EPA, 1994; Ingersoll et al., 1995; EPA, 2000c). In addition to providing information on the specific tests, EPA (2000c) gives compound- and element-specific bioaccumulation information for a range of species. Specific guidance has been produced jointly by EPA and U.S. Army Corps of Engineers for evaluating dredged materials for both toxicity and uptake and bioaccumulation in invertebrates in marine (USACE and EPA, 1991) and freshwater environments (EPA and USACE, 1998). The strengths and limits of applying such tests to nature have also been discussed in Luoma (1996).
A subset of sediment bioassays are uptake and toxicity tests involving sediment interstitial water (pore water) (Lee, 1978; Giesy et al., 1988, 1990; Ankley et al., 1989; Hoke et al., 1992, 1993). For certain organisms, contaminant concentrations in pore water are more closely related to bioavailability than are concentrations in bulk sediments (Ankley et al., 1992a; Bonnell et al., 1995). Indeed, this is the basis of equilibrium-partitioning methods for predicting bioavailability
of metals and neutral organic compounds (see Chapter 2). Sediment pore water tests are most useful when the duration of the assay is short. Otherwise, the contaminant can become depleted and the tests will underestimate both bioaccumulation and toxicity of bulk sediments (Giesy and Hoke, 1989; Giesy et al., 1990). Obviously, these tests are accurate only for assessing exposure via the pore water pathway.
Another related bioassay is the elutriate test, which was developed to mimic the potential bioavailability (and in some cases toxicity) of contaminants following sediment resuspension in the water column caused by dredging (Palermon and Thackson, 1988; Ludwig, 1989; Bonnet et al., 2000). The test is actually an extraction followed by an uptake or toxicity bioassay on the extracted liquid. Sediments are combined with water in a ratio of approximately 4:1 (Daniels, 1989; Giesy and Hoke, 1990), which leads to fundamental changes in contaminant–sediment binding and hence bioavailability (Harkey et al., 1994). Where compounds are accumulated across membrane surfaces such as gills, this resuspension and the resulting contaminant release into the dissolved phase can increase overall bioaccumulation and toxicity. In cases where primarily particle-bound compounds are taken up (such as with zooplankton), the opposite is generally true (Nalewajko, 1989). Thus, the accuracy of elutriate tests in assessing bioavailability is a function of the compound of interest, the exposure pathway, and the dilution ratio.
Fish Bioassays
Fish have been used in tests of sediment contamination in both field and laboratory studies (Anon, 1978; Ankley et al., 1992b). Accumulation of contaminants from sediments can have direct effects on fish and the predators that eat them, including humans. Thus, fish bioassays can be used in food-chain studies as well as to predict the potential adverse effects these residues could have on the fish themselves. Contaminant concentrations in fish tissues provide an integrated measure of the exposure from all pathways (e.g., ingestion of contaminated water, food, or sediment; dermal contact; and passage across the gills).
In situ studies give the most accurate measurements of bioavailability under field conditions, as long as the study design does not inject biases into the results. Two basic fish bioassays are possible under field conditions. Either wild (feral) fish can be collected at a site, or (because it may be difficult to know if fish are resident or for how long they have been exposed) fish can be caged at a location. There are different measurement endpoints that can be used, including bioaccumulation as measured by tissue concentrations and more functional responses. For example, toxicity—measured by survival or growth—is a standard endpoint. Whether a bioassay that uses accumulation into tissues or a functional measure of response is chosen depends on the particular scenario, including the contaminant of interest. For example, some metals are homeostatically regulated in fish, such
that accumulation of these compounds is a less appropriate measurement of bioavailability. Instead, functional responses that are manifested, for example, on the surface of the gills, might be used.
Field studies of contaminant accumulation and toxicity in fish are complicated by a number of factors that are difficult to control. It is generally impossible to distinguish between different sources of contamination, be it the sediment itself, the water column, and food items, including invertebrates, consumed by the fish. Indeed, the primary route of fish accumulation of many contaminants, especially neutral organic compounds that tend to be persistent in sediments, is through food chain transfers (Jones et al., 2001). Fish are excellent integrators of contamination coming from these multiple sources, but as a consequence fish bioassays in the field are not effective for measuring individual bioavailability processes. Another important logistical limitation to caged fish studies is that unless there is sufficient natural food, the fish need to be fed. If this is the case, the caged fish may not accurately reflect exposure under natural conditions.
Standard protocols have been proposed to determine the accumulation and toxicity of various contaminants under laboratory conditions, including metals and organic compounds from both water (EPA, 1975; ASTM, 1980) and sediments (ASTM, 1988, 1994; Ankley et al., 1992b; EPA Region 5, 1994). For such tests that utilize contaminated water only, a direct measurement of biouptake into fish can be made. The ecological relevance of laboratory tests is not always clear because biouptake may not be the rate-limiting step for overall fish accumulation of contaminants from sediment. However, results from such bioassays are valuable as input into simulation models of contaminant accumulation by fish (which attempt to account for uptake from multiple sources of contamination). Laboratory protocols that include sediment should be designed to capture diffusion and disturbance processes in sediments, which may be more important to overall contaminant accumulation than the biouptake process (Magee, 1991; Ankley et al., 1992b).
Mammal and Bird Bioassays
As part of ecological risk assessments, both mammals and birds have been used to monitor for exposure to residues at contaminated terrestrial sites (Phillips and Rainbow, 1993; Talmage and Walton, 1993). Although there are some differences between birds and mammals, there are enough similarities that they can be considered together when discussing the bioassays available for both toxicity and accumulation (Tank et al., 1993). Vertebrates can be exposed to residues in sediments; however, the issue of bioavailability is generally more relevant for their exposure to terrestrial soils (Pankakoski et al., 1994). As with fish, tissue accumulation or toxicity measurements in mammals or birds are integrative measures of exposure. Sentinel animals can be used either in field situations or in closed laboratory systems where soils are brought from the field.
There are several pathways through which vertebrates can be exposed to contaminants in soil, including direct consumption of plants, prey, and soil. However, except for some species of ducks, direct ingestion of soil by birds is rare, such that few if any bird bioassays have been designed to capture that pathway. Rather, tests are designed around the pathway of plant or prey consumption, and contaminants in food are assumed to be 100 percent bioavailable. (This may or may not be true for all organic compounds and metals, but it seems to be true for most neutral organic residues, such as PCBs and DDTs.) For the food consumption pathway, the availability of contaminants in sediments or soils is measured by determining the bioavailable fraction to the prey rather than the predator, using tests described earlier for invertebrates and fish. For example, if midges living in PCB-contaminated sediments will molt and subsequently be eaten by tree swallows, the normal practice is to determine the bioavailable fraction of the PCBs in the sediments to the insects, not to the birds. It is possible to determine the bioavailable fraction to the predator directly, for example by measuring the concentration of the residue in the bird’s diet and the fraction remaining in the feces.
In some cases, direct ingestion of soil can be an important exposure pathway for which direct measurements of uptake would be useful. For example, birds that have a crop consume grit in the form of small stones or sand (Solomon et al., 2001). Determining the fraction of the contaminant in soil or sediment that would be biologically available under the conditions in the crop can be approached with standard laboratory feeding studies (Romijn et al., 1995). In the case of birds, the tissue of interest for which the contaminant levels are measured is often the eggs (Keith, 1996).
No specific protocols have been developed to determine the chronic toxicity of soil or sediment contamination to birds. However, the protocols that have been developed to conduct dietary toxicity tests (ASTM, 1999) can be applied.
Field studies are another possibility, using wild, feral, or even domestic mammals (Pankakoski et al., 1994) or birds (Tank et al., 1993; Hothem and Welsh, 1994; Baars et al., 1995; Nabel et al., 1995) to monitor for the exposure expected for wild animals at a specific site. It is difficult to determine the contaminant fraction taken up from soils or sediments by measuring the concentrations of residues in wild mammals and birds, given the various sources of contamination to which they may be exposed and a general inability to control for confounding factors. However, if the site use factor2 is well known, wild mammals and birds may be helpful to determining potential exposure. Another approach is to release sentinel organisms such as small mammals or birds of a known dietary type at a site (Custer et al., 1996). For gallinaceous, ground-dwelling birds, the domestic chicken is a useful surrogate species (Schuler et al.,
1997). Sentinel birds can be placed in cages over the area of interest to determine the degree of accumulation that occurs under relatively natural field conditions. Pinioned birds can be released into larger areas, but recovering the birds can be difficult and predation can be a problem. A third approach is the bird box study, which works well for a wide range of species including blue birds, robins, doves, tree swallows, house wrens, and any species that will build a nest in a nest box (Cain and Bunck, 1983; Bunck et al., 1987; Blus et al., 1993; Bishop et al., 1995; Kemler et al., 2000). Nest boxes can be placed in certain regions to determine the overall exposure that is expected to occur to birds of a particular feeding type. This type of monitoring has been used successfully at a number of contaminated sites (Froese et al., 1998), including the use of kestrels to determine bird exposure to polychlorinated dibenzodioxins in soils (Kemler et al., 2000). Such monitoring obviously cannot account for specific bioavailability processes, because the measured bird contaminant burdens are an integration of multiple processes occurring in the area around their nests. However, if concentrations of the contaminant of interest are also measured in soil, sediment, and other dietary items, then bioconcentration factors, which implicitly include a measure of the bioavailable fraction, can be determined (Froese et al., 1998).
Environmental Health Studies
Environmental health studies are designed to evaluate human childhood exposures to a contaminant in the residential environment. Such studies have been conducted for lead at numerous mining sites and to a lesser extent for arsenic at mining and smelting sites. Although these studies are not intended primarily to evaluate the bioavailability of the contaminant to the exposed population, they can yield this type of data all the same. Given the difficulty of studying absolute and relative contaminant bioavailability in children, environmental health studies offer one of the few mechanisms for obtaining this type of data.
To conduct such a study, a cohort of individuals living within an area with elevated soil concentrations of the element of concern must be recruited. For each individual, all known potential exposure sources within the residential environment are sampled, along with a biomarker(s) for exposure to that element. For example, when conducting such a study for arsenic, sampling would include yard soils (particularly from bare areas), house dust, and tap water as the potential exposure sources. Potential biomarkers include total arsenic, speciated arsenic (i.e., As+3, As+5, monomethylarsonic acid, and dimethylarsinic acid), and creatinine concentrations in urine. Exposure from food would be estimated based on diet, since the extent of this exposure is well characterized. A detailed questionnaire would be administered to identify any behavioral or dietary sources of arsenic exposure. Urinary and fecal arsenic concentrations would be monitored to establish arsenic exposure to each individual. These data would then be used as
input to a human health risk assessment model for the affected community. The risk assessment model requires the relative bioavailability of arsenic from each exposure source to be known or estimated. Since the relative bioavailability of arsenic from soil and house dust, along with soil ingestion rate, are two of the least well characterized variables in the model, combinations of these variables can be tested to establish the best fit to observed exposures. Such an approach was performed for childhood exposures to arsenic in the residential soils at the Anaconda, Montana, National Priorities List site (Cohen et al., 1998). The study yielded plausible estimates for relative arsenic bioavailability in local children. This approach to estimating relative arsenic bioavailability in children contains considerable uncertainty, and, having only been performed once, the reproducibility is unknown. In addition, such a study is sufficiently expensive that it would likely be performed only at the most high profile sites. Finally, the study must be specifically designed to yield estimates of uptake from soil and house dust. Of all the environmental health studies for lead and arsenic conducted to date, only the Anaconda study included the data necessary to evaluate contaminant bioavailability from soil.
Ecosystem Level Tests
Microbial Community Assays
There are no reports that explicitly address contaminant “bioavailability” measurement inferred from a response at the level of whole soil microbial community. However, several community-based assays to measure soil microbial activity exist and could potentially be employed to infer contaminant bioavailability. As microorganisms account for up to 90 percent of the soil biomass and contribute a large proportion of essential soil functions, such as cycling of C and N, examining contaminant effects at the level of the microbial community seems critical. This is supported by the fact that microorganisms are in direct and intimate contact with contaminated soil particles and pore water.
Microbial systems can be investigated at two fundamentally different levels: the level of system function and the level of community structure. System function is most often examined by studying elemental (nitrogen or carbon) cycling. Carbon mineralization can be inferred from CO2 evolution and measured as a basal level or in response to the addition of specific carbon substrates of interest (Stenstrom et al., 1998; Lin and Brookes, 1999; Gong et al., 2000; Murray et al., 2000; OECD, 2000a). An extension of this method concerns the examination of the community level utilization of different carbon sources (Degens and Harris, 1997), facilitated with tools such as the commercially available Biolog (Garland and Mills, 1991; Rutgers et al., 1998). Measurement of nitrogen cycling is more complicated and requires selective extraction of nitrogen species from soil matrix and subsequent analysis (Johansson et al., 1998; Kandeler et al., 1999; OECD,
2000b). Rapid and inexpensive qualitative assessments of whole system function can be derived from the activity of several key soil enzymes such as dehydrogenases, amylases, phosphatases, arylsulfatases, and cellulases (Kelly and Tate, 1998; Margesin et al., 2000). Unfortunately, the true ecological relevance of these snapshot measurements is not clear, and microbial activity cannot be easily separated from plant root activity.
Direct measurements of community structure—presumably correlated to activity—can be made (Ibekwe and Kennedy 1998; Waldrop et al., 2000). It has long been recognized that cultivation-based techniques to address community structure vastly underestimate community diversity, yielding biased estimates of community structure (Amann et al., 1995). Hence noncultivation-based molecular techniques are typically adopted. Most of those techniques as applied to soil microbial community inspection either target the lipid profiles (Lindahl et al., 1997; MacNaughton et al., 1999; Zelles, 1999) or nucleic acid fraction (Muyzer et al., 1993; Liu et al., 1997; von Witzingerode et al., 1997; Hill et al., 2000) of the community. When targeted at the nucleic acid fraction, analysis can be done with or without enzymatic amplification of the nucleic acid pool. Although vast progress has been made in development of such techniques, their successful implementation awaits standardization and decisive studies on the link between community diversity and contaminant toxicity (and/or bioavailability).
Mesocosm Tests
This chapter has previously discussed field-scale tests of contaminant uptake and accumulation, such as putting caged fish into aquatic systems or putting penned or pinioned birds into a terrestrial area for a known period of time. While conducting such studies at the site of interest is often desirable, it is not always possible. Also, the inability to control or maintain organisms for long periods of time and the inability to control exogenous factors makes interpreting these results difficult. Therefore, semi-field scale tests, referred to as mesocosms or microcosms depending on their size, have been developed to mimic realistic exposure scenarios (Perez et al., 1977) while allowing some control over complicating factors (Brockway et al., 1979; Craft, 1983; Anon, 1984).
Mesocosms are subsets of ecosystems (Giesy and Odum, 1980). They can be bounded natural systems or completely artificial (gnotobiotic) (Graney et al., 1995). Mesocosms are generally used to study population, community, and ecosystem processes, including responses to stressors and chemical toxicants (Davies and Gamble, 1979). This is done to give more realistic exposures and to allow for the study of interactions that occur in these more complex systems (Addison and Holmes, 1995). For example, volatilization, photolysis, and sorption to inorganic and organic matrices may be fairly site-specific and important to bioavailability processes. Of particular importance is the interaction between biota and their environment (Rodgers, 1983).
A wide range of mesocosm types and sizes has been developed to test for the toxic effects and bioaccumulation of compounds (Giesy, 1980; Odum, 1984). For example, intact soil cores have been used in soil leaching studies to estimate the bioavailability of metal ions (Tolle et al., 1985). Mesocosms are also useful for validation of complex models of bioaccumulation (Larsson, 1984; Anderson et al., 1987; Larsson and Sodergren, 1987; Abbott et al., 1995). Mesocosms serve as an intermediate-scale system in both size and complexity, and thus must be large enough to have certain attributes, but not so large that they cannot be studied as experimental units and replicated. Thus, mesocosms allow for potentially more realistic exposure scenarios (and chemical and physical processes) than could be simulated in smaller bench-top studies. Finally, mesocosms can include complex interactions between and among organisms and their abiotic environment to more closely mimic field conditions. Because such tests are complex, they tend to be useful but expensive to conduct. Currently, mesocosms are neither required nor readily accepted as tools to study bioavailability for regulatory purposes.
Summary
This section has discussed dozens of biological tools available for measuring bioavailability to both ecological (microorganisms, plants, and animals) and human receptors. The tools range from those that measure just one process, such as absorption across a membrane (biouptake), to those that measure the integrated effect of multiple processes. There are tradeoffs between such tests, as clarified in Table 4-2. In particular, those tests that directly measure biouptake, such as isolated organ tests or assimilation efficiency, provide unambiguous results about distinct mechanisms, but they may not capture the complexity of the environmental system nor speak to important effects, like mesocosms and toxicity tests can.
Certain biological tests have been used to validate some of the physical and chemical tools discussed earlier, or they have been used to provide complementary evidence about bioavailability processes in a system. For example, assimilation efficiency used in parallel with spectroscopy could reveal the properties of sediments that control bioavailability process A in Figure 1-1. Finally, many of the tools discussed represent the state of the art or require additional research in order to reach their potential, especially molecular tools such as biomarkers and reporter systems.
TOOLS FOR HUMAN HEALTH AND ECOLOGICAL RISK ASSESSMENT
Exposure assessment is central to assessing risks of chemicals in the environment. The tests described in this chapter can be used to incorporate site-specific information into exposure assessment and to improve general knowledge. In order for the results to be acceptable to risk managers and regulators, the
tools used must be well supported technically. This section discusses the tools commonly chosen to provide information about bioavailability processes in site-specific human health and ecological risk assessment. Of course, the choice of a tool or tools is driven by the purpose of the assessment (e.g., to evaluate effects of specific soil or sediment conditions, to improve direct estimates of absorption processes, or to determine how bioavailability processes affect toxicity). Tests that measure uptake and bioaccumulation, are biomimetic (extractions), or measure toxicity directly or in surrogates are deployed on a regular basis for human health or ecological bioavailability assessment. New tools and modeling approaches are also available, and these are discussed as well. Because no tool or approach is universally the best, tradeoffs among tools (as described in Table 4-2) should be considered.
Prior to engaging in any attempt to measure the bioavailability of contaminants from soils or sediments, it is critical to establish an accurate site conceptual model that describes the relevant exposure pathways, the receptors to whom the exposures are occurring, and the environmental conditions under which the exposures are occurring. This is vital because the available tools for assessing bioavailability processes from soil are receptor-, pathway-, and contaminant-specific; bioavailability data for a chemical for one exposure pathway are not necessarily applicable to another exposure pathway. Because the development of a site conceptual model is generally the first step in any human health or ecological risk assessment, this information may already be available for a particular site. The lack of an accurate site conceptual model can lead to (among other problems) measurement of the wrong endpoint or selection of an inappropriate bioavailability tool.
Ideally, the tools chosen should support mechanistic understanding of bioavailability processes and subsequent model development. Only if this is a common goal will bioavailability assessment progress to the point of being used regularly, consistently, and accurately. This focus on processes suggests that a suite of tools is needed to fully assess bioavailability. Tools that collectively cut across different processes are more valuable than having multiple tools for the same process. As an example of why tools should be chosen to better understand fundamental processes, consider the role of soil properties on contaminant retention. As discussed extensively in Chapter 3, the mechanisms by which contaminants are bound to solids are controlled by a number of soil factors (e.g., organic matter content). Measurement of these factors early in the evaluation can guide the selection of other bioavailability tools and help interpret their results. (See Chapter 5 for additional discussion of this aspect of tools selection.)
A third important consideration is that since human health and ecological risk assessments tend to follow specific procedures, in order for bioavailability information to be useful within a risk assessment it must be in an appropriate form. This form may include (1) values or factors that are used in deterministic or equilibrium exposure equations; (2) kinetic models that take into account the time
of exposure and other factors; or (3) direct measures of chemical concentrations or biological responses to exposure.
Regulatory acceptance of the tools used to generate bioavailability information in risk assessment is expected to be influenced by several factors, including the validity of the method (Menzie et al., 2000). Validation variously refers to the performance of a tool or approach in terms of reproducibility, reliability, and multi-lab calibration. More important is validating the application of the approach to bioavailability processes; that is, it must be proven that the approach responds to changes in bioavailability. Ultimately, approaches that allow quantitative estimation of bioavailability should be validated by correlation with a biological response both experimentally and in the field situation where they are most relevant (see Box 4-10 for an example). An appropriate body of experimental and field study work would:
-
clarify where and when a method yields a definitive response;
-
clarify that the tool can be linked to a biological response of a similar magnitude, and that the linkage stands up across a range of conditions in the type of environment that is being managed;
-
test the prediction of bioavailability using different types of experiments and field studies;
-
clarify which types of species-specific biological responses are best predicted by the approach; and
-
include critiques of the best applications and the limits of the approach, especially compared to alternatives.
A method that is well accepted and validated should be given greater weight than one that is new or experimental.
Human Health
Several tools currently exist to evaluate the relative bioavailability of contaminants from soil to humans, all of which are based on exposure through direct contact. The tools include (1) animal studies that compare the absorption of a particular compound from soil relative to its absorption from a soluble salt (for inorganics) or a readily-absorbed vehicle such as oil (for organics); (2) in vitro test systems that have been developed to measure solubility or absorption of a chemical under a defined set of exposure conditions; and (3) various indirect techniques that evaluate the chemical forms of inorganics in soil or the manner in which organics are sequestered in soil. To date, this last approach has most often been used with certain metals (arsenic, lead, mercury) in soil; however, this information is generally used to elucidate the mechanisms underlying bioavailability rather than as the sole approach to estimating a bioavailability adjustment factor. Although any of these approaches could potentially be used with sedi-
|
BOX 4-10 In Vitro Validation Study for Lead This box describes validation of an in vitro extraction test against the EPA Region 8 young swine model for determining the relative bioavailability of lead in soil. This work was a collaboration among a group of academics, consultants, regulators, and industry personnel to validate a simple test method that could predict relative lead bioavailability from soil and could be used for risk assessment. Initially, the group developed a simplified in vitro test, consisting of a well-mixed, 1-hour extraction (37ºC) in pH 1.5 HCl that was buffered with glycine (0.4 M). Initial testing indicated that this method correlated well with results from the young swine model. A Standard Operating Procedure (SOP) for the in vitro test, and an associated Quality Assurance Project Plan (QAPP), were prepared for the method validation study. The QAPP specified a high frequency of Quality Assurance (QA) sample analysis, including blanks, duplicates, and spikes. Three independent laboratories were selected (National Exposure Research Lab in Las Vegas, Nevada; Bureau of Reclamation, Environmental Research Chemistry Laboratory in Denver, Colorado; and ACZ Laboratories, Inc. in Steamboat Springs, Colorado), and each reviewed the SOP and QAPP. Each laboratory was then sent blind triplicate splits of three samples (nine samples total) and asked to perform the in vitro extraction and report the extracted lead concentrations for each of the nine samples. The resultant data were used to evaluate the precision, accuracy, and reproducibility of each laboratory, and to identify any deficiencies so that corrective actions could be instituted. The actual validation study involved submitting blind triplicate splits to each of the three laboratories from the 19 samples that had been tested for relative lead bioavailability in the young swine model. Results from these analysis indicated that the in vitro extraction method has good inter- and intra-laboratory reproducibility, and it correlates well with results from the young swine model (in vitro to in vivo correlation is linear with an r2 = 0.93; Drexler, 1997). The in vitro extraction method used in the validation study may be found in Kelley et al. (2002), and the results of the validation study have recently been submitted for publication. |
ments, their practical use to date has been limited to soils because this is the medium to which humans are most frequently exposed.
The importance of establishing an accurate site conceptual model during human health risk assessment to guiding the selection of bioavailability tools cannot be overstated. For example, under residential exposure conditions it is generally young children who are the most highly exposed and sensitive population. Their exposures to inorganics in soil are predominantly oral (because of hand-to-mouth activity) and may occur from both soil and house dust (depending on activity patterns and the season). To address such a childhood exposure, it is important to select a tool that measures relative oral bioavailability relevant to children, particularly if they have higher oral absorption rates of the chemical than adults (e.g., as with lead). The young swine model, which was specifically designed to determine the relative oral bioavailability of lead in soil to young
children, is just such a tool. However, were the contaminant of concern an organic compound in soil or house dust, then it is likely that dermal exposures would also be important, and an entirely different assessment methodology might be required. Finally, if it were an adult that was exposed to lead in soil, for example a construction worker exposure scenario, this would be better evaluated using the adult human model for lead uptake from soil.
Table 4-4 provides a summary of the methods that are currently available or are in development for estimating the relative bioavailability of organics and inorganics in soil to humans via oral, dermal, and inhalation exposure routes.
TABLE 4-4 Tools for Estimating the Relative Bioavailability of Soil Compounds to Humans
|
Exposure Pathway/Contaminant |
Currently Availablea |
In Developmentb |
|
Oral/Inorganics |
In vivo: Weanling rat (Pb, Cd) Young swine (Pb, As) Adult primate (As) Adult human (Pb) In vitroc: Pb, CN– |
In vivo: Young swine (Cd, Cr) In vitroc: As, Hg, Cd, Cr |
|
Oral/Organics |
In vivo: Mice (PAHs) Rat (PCBs) Rat, rabbit, and guinea pig (PCDDs/Fs) |
In vivo: Rat (DDT) In vitroc: PAHs, PCBs, OCPs, PCDDs/Fs |
|
Dermal/Inorganics |
In vivo: Monkey (As) Swine (Ni) In vitrod:As, Cd, Cr, Hg, Ni |
|
|
Dermal/Organics |
In vivo: Monkey (PCBs) Mice, rat, guinea pig, and swine (PAHs) In vitro: PAHs, PCBs, OCPs PCDDs/Fs |
|
|
Inhalation |
None |
None |
|
a“Currently available” in vivo tests are indicated for the contaminants to which they have been applied on a regular basis, while for in vitro methods the table indicates contaminants for which tests have received a substantial level of validation, or have been accepted by a regulatory agency. b“In development” indicates methodologies on which development work is actively being conducted. cIn vitro tests for estimating oral bioavailability are based on extraction in simulated gastrointestinal fluids. dIn vitro tests for estimating dermal bioavailability are generally based on penetration of a compound through skin (human or animal) in a special test cell. |
||
Given the critical role of the biological system, in vivo methods are generally preferred as a technical basis for refining risk assessment for human health. In vivo studies, however, require that an acceptable animal model is available or can be developed within the technical, cost, and ethical constraints associated with a particular project. As evident from Table 4-4, the in vivo models for estimating the oral relative bioavailability of inorganics (particularly lead and arsenic) in soil have received considerable attention, and therefore are the most fully developed. The in vitro tests for these elements, which are based on extraction in simulated gastrointestinal fluids, are also relatively well developed. However, as mentioned in Box 4-10, only the in vitro test for lead has been fully validated (against the young swine model).
After oral bioavailability models for specific inorganics, the dermal absorption of hydrophobic organic compounds has received the most attention. Both in vivo and in vitro methods have been developed, although in this case the in vitro tests involve measurement of penetration through actual skin (human or animal) in a special test cell. The dermal absorption of certain metals (Table 4-4) has also received a certain amount of attention, with most of the studies to date having been conducted using in vitro rather than in vivo methods. This reflects the technical difficulty and expense of designing and conducting studies using animal models. Finally, the oral bioavailability of hydrophobic organic compounds in soil is an area where much work remains to be done. Although animal models have been developed for the major classes of organic compounds, all could benefit from further refinement, and no validated in vitro models are available at this time.
To date, no specific test methods, either in vivo or in vitro, have been developed to measure the pulmonary bioavailability of organics or inorganics from soil. This situation reflects the fact that inhalation exposures from soils are generally small relative to the oral and dermal exposure pathways, except under special circumstances.
Ecological Applications
Ecological risk assessment presents complexities over human health risk assessment because of the potential for contaminants to be differentially accumulated by different organisms and transferred up food chains. The tools that are used must be able to discern the fraction of the contaminant that is available for release, for absorption by critical species, and for further passage into the food web. One way to handle this complexity is to consider tools for measuring bioavailability processes in terms of the exposure pathways to which they apply. Bioavailability tools have been considered, developed, or applied to many of the common ecological pathways in soil, such as soil ingestion, dermal contact, and ingestion of plant matter, as well as for pathways that occur in aquatic environments. Table 4-5 lists those tools available for determining the bioavailability of contaminants in soil and sediment to ecological receptors for all major pathways,
TABLE 4-5 Tools for Estimating the Bioavailability of Compounds in Soil or Sediment to Ecological Receptors
|
Exposure Pathway |
Currently Available |
In Development |
|
Soil/Sediment →Invertebrate |
• Equilibrium partitioning and AVS methods • Toxicity tests with benthic and soil invertebrates to measure effects • Laboratory or field tests on benthic and soil invertebrates to measure accumulation of chemicals in their tissues |
• Use liquid or solid extraction media to simulate uptake and accumulation into invertebrates • Critical Body Residues (CBRs) in invertebrates that are predictive of effects |
|
Soil/Sediment →Plants |
• Soil/sediment extraction tests • Toxicity tests with appropriate plant species to measure direct effects on germination and growth • Laboratory or field exposures to contaminated soils/sediments to measure accumulation of chemicals in plants for direct use chain models |
• Test aqueous extracts or elutriates of soils in various plant bioassays including algal tests, seed germination, and root elongation • Utilize the Plant Micronucleus test—Tradescantia—to evaluate potential genotoxic effects |
|
Soil/Sediment →Groundwater →Surface Water Biota |
• Leaching and desorption tests • Direct measurements of chemicals in groundwater, pore water, or surface water |
|
|
Soil/Sediment →Wildlife |
|
• Extraction tests that simulate physiological fluids may be useful for evaluating availability of incidentally ingested soils/sediments • Short-term feeding studies may be useful for evaluating incidental ingestion of soils/sediments • Selected chemical-specific biomarkers |
|
Soil/Sediment →Plant or Invertebrate →Wildlife |
• Depending on wildlife diet, measures of bioaccumulation in plants and animal prey can be used and combined with a model |
|
|
Exposure Pathway |
Currently Available |
In Development |
|
|
• Body burden measurements and measures of chemical metabolites can be used as indicators of exposure and of the availability of chemicals in soils/sediments • Selected chemical-specific biomarkers |
|
|
Soil/Sediment → [invertebrate or plant] → Wildlife → Predatory birds and mammals |
• Depending on wildlife diet, measures of bioaccumulation in plants and animal prey can be used and combined with a model • Body burden measurements and measures of chemical metabolites can be used as indicators of exposure and of the availability of chemicals in soils/sediments • Selected chemical-specific biomarkers |
|
|
Soil → Soil Vapor → Burrowing wildlife |
• Field measurements of soil gas to estimate exposure • Field measures can be used in soil gas models |
|
while the discussion below focuses on those pathways that have received the most attention with respect to bioavailability measurements.
Pathway from Soil or Sediment to Invertebrates or Plants
Invertebrates and plants are in direct contact with soils or sediments and therefore subject to chemical exposures at levels that could be toxic either to themselves or to higher trophic levels via food consumption and predation. For this reason, many tools for measuring contaminant uptake and bioaccumulation into plants and invertebrates as well as toxicity tests have been developed. Bioaccumulation test data have been used to create a variety of models of chemical exposure (such as the equilibrium partitioning and empirical methods discussed in Chapter 2 and other more mechanistic models, as described below).
Toxicity tests (described variously throughout the chapter) are frequently applied to evaluate effects of chemicals and chemical mixtures in soils and sedi-
ments to plants and invertebrates. The drawback of laboratory tests is that they may not reflect field conditions. In situ tests, on the other hand, generally lack the formal standardization and control available in the laboratory. Although the use of toxicity tests in regulatory programs is well accepted for invertebrates and plants, a number of uncertainties are associated with their application that have fostered debate and further research and development (Luoma, 1996). Most of this uncertainty concerns the selection of test species, test duration, appropriate toxicological endpoints, and extrapolations from simple laboratory conditions to the highly variable field conditions. Because toxicity tests reflect the integration of multiple physical, chemical, and biological processes, they are of limited use for gaining a better mechanistic understanding of bioavailability. However, they provide information that is unique from initial biouptake and bioaccumulation tests and they measure endpoints that are often of greater interest to stakeholders and regulatory agencies.
Bioaccumulation tests on sediment or soil invertebrates and plants that measure tissue concentrations of contaminants following exposure are frequently used to assess bioavailability and to provide input data for such simple empirical models as BSAF. As discussed previously in this chapter, such tools have been developed for the laboratory and the field and are in wide use. The major uncertainty associated with these tools stems from species-specific differences in the degree to which organisms accumulate compounds, which may reflect variability in exposure as well as variability in their anatomy and physiology. Therefore, data developed for a limited number of species may not be directly extrapolated to other species. This uncertainty can be somewhat accounted for by selecting a range of species considered representative of groups of other species (i.e., guilds). These measures of exposure can also be combined with effects information based on critical body residues (CBRs) to estimate toxic effects to target organisms, particularly acute effects (Fitzgerald et al., 1996; Lanno et al., 1997). Indeed, there is much interest in coupling tissue residue measurements with CBRs for bioaccumulative compounds to provide a better measure of risk to organisms than can be achieved solely from chemical measurements in soils or sediments.
One of the most important uses of bioaccumulation data has been to try to develop more sophisticated models of uptake that might take into account soil and sediment properties. Many of these models assume a mechanistic underpinning of equilibrium partitioning between the solid phase, pore water, and tissue. For example, Connell and Markwell (1990) initially proposed an equation to model the distribution of nonpolar organic contaminants in three compartments: soil, soil water, and earthworm tissue. Menzie et al. (1992) later modified the soil-to-earthworm bioaccumulation model to include a variable for soil organic content that yielded predictions that were in agreement with site-specific data. Soil characteristics have also been incorporated into models of heavy metal (cadmium, copper, lead, and zinc) accumulation in radish (Raphanus sativus L.), although not all metal distributions were accurately predicted (Davies, 1992).

Sampling sediments in San Fransicisco Bay for clams that bioaccumulate contaminants.
Assuming that contaminants will partition between soil organic matter and organic matter in the root system, Polder et al. (1995) designed a plant uptake model using data for 27 organic compounds, including pesticides and PCBs, and 18 plant species, mostly agriculturally important crops. These models were found to work best when soil organic matter is between 0 and 30 percent. In addition,
accumulation into stem tissues was not well predicted and in some cases only certain root tissues (i.e., root peel but not core) were well represented.
Jager (1998) employed the concept of thermodynamic partitioning between water and lipid phases of tissue to model bioconcentration factors for earthworms. Although tissue concentrations correlated well with aqueous concentrations for chemicals with log Kow values from 2 to 6 (r2 = 0.9), the model overestimated the bioaccumulation factor for earthworms exposed to soil-borne contamination by an average factor of 5.6—a discrepancy explained in part by the differences in the feeding ecology of the various species of earthworm tested.
More complicated models of bioaccumulation were generated by Sample et al. (1998) during a literature survey of 32 studies which examined co-located earthworm and total soil chemical concentrations for nine inorganics (arsenic, cadmium, chromium, copper, manganese, mercury, nickel, lead, and zinc) and two organics (PCBs and tetrachlorodibenzo-p-dioxin). Results from 26 of the studies were used to prepare simple regression models, while the remaining six studies were used to validate predicted values. For PCBs and seven of the nine metallic elements considered (arsenic, cadmium, copper, mercury, manganese, lead, and zinc), the best estimate of tissue concentration in earthworms was given by a natural log-natural log regression with total soil metal concentration. The addition of soil pH data to the regression model did not markedly improve fit, although when soil calcium concentrations were incorporated, a better fit was obtained for cadmium and lead. Tissue concentrations were inaccurately estimated for the transition metals nickel and chromium, by either simple or multiple regression models.
Finally, Saxe et al. (2001) have also suggested a partitioning approach for metals in earthworms that considers both dermal exposure to soil pore water and ingestion of soil particles.
All these examples of simple compartment-type and regression models that incorporate key soil characteristics into the prediction of bioaccumulation are necessarily limited by our lack of knowledge regarding the soil factors most important for influencing bioavailability processes. It may be possible to use such models to bound the reasonable range of bioaccumulation—thereby providing conservative site-specific estimates that are required for screening ecological risk assessment—but additional work is clearly needed in this area.
Pathway from Soil or Sediment to Wildlife via Incidental Ingestion
There are few tools available to measure the bioavailable fraction of chemicals in soils and sediments incidentally ingested by wildlife. Extraction tests that simulate the action of physiological fluids may be useful for evaluating this pathway, although they have not yet been developed for that purpose or tested against actual responses. Some of the more sophisticated methods (e.g., the use of biomarkers in urine or blood, or feeding studies with laboratory animals) are used
only occasionally in site-specific studies, although their results might be generalizable in some circumstances.
Experimental feeding studies with representative species are the most direct way for evaluating bioavailability to wildlife, although they are rarely carried out. Because of variations in the physiology and anatomy of wildlife species, data developed for one species may not apply to another. On the other hand, field studies or surveys have long been useful in identifying bioavailability influences (e.g., Luoma and Bryan, 1978), or how food web relationships influence biotransfer of contaminants that originate from sediments. Use of stable isotopes has proven a relatively inexpensive approach to food web studies (Kidd et al., 1995). However, limitations imposed by logistical complexities and the availability of the proper expertise so far have precluded conducting such work for any but generic purposes.
***
In any pathway where food chain transfer is expected to play a role in exposure, models can be used to predict the exposure expected for each trophic level or individual receptor. One of the most common methods of prediction is the food chain model based on fugacity (and hence on partitioning theory and ultimately thermodynamics) (Ling et al., 1993; Mackay and Paterson, 1991; Nichols et al., 1995). A more mechanistic modeling approach for site-specific exposure assessment for trace elements that takes multiple pathways into account is the Dynamic Multipathway Bioaccumulation Model (DYMBAM) (Wang et al., 1996a; Luoma and Fisher, 1997; Schlekat et al., 2002). This is a relatively new approach similar in principle to generic biokinetic models (Thomann et al., 1995). Yet, DYMBAM is more applicable to specific circumstances because it uses empirically developed physiological rate parameters representative of one or more key native species and environmental data representative of a range of system conditions.
DYMBAM models bioaccumulation as a combination of gross influx and efflux rates, such that key parameters to determine experimentally include influx rates from solution and from food and efflux rates. Influx from solution can be determined with radionuclides in short exposures (e.g., one day) because the goal is to estimate the unidirectional flux. Influx rates from ingestion vary with the food source and so are best determined from the product of assimilation efficiencies (from specific types of food), feeding rate, and concentration. The efflux of inorganic contaminants is characterized by rate constant(s) describing exponential disappearance as function of time (first order isotope-substitution kinetics; Riggs, 1963; Ruzic, 1972; Cutshall, 1974; Luoma et al., 1992). Obtaining the data for such models has been considered onerous in the past (Landrum et al., 1992). However, recent studies show that model behavior can be reasonably constrained (McKim and Nichols, 1994), and manageable methods are available for obtaining species-
specific biological data, especially if whole organism data is the goal (Wang et al., 1996a). If correctly determined, these parameters are directly comparable among species. Thus, in addition to their use in models, these data could lead to better understanding of interspecies differences in bioaccumulation.
DYMBAM is the simplest form of a bioaccumulation model. It lacks, for example, bioenergetic terms or considerations for seasonal gain and loss of lipid that affect both trace element and organic contaminant bioaccumulation (Capuzzo et al., 1989; Cain and Luoma, 1990). Nonetheless, even this simple model approach appears to provide reasonable compatibility with field observations (Luoma, 1976; Luoma et al., 1992; Griscom et al., 2002), although further studies undoubtedly will find ways to improve the model predictions. Data needs for expanding even the simple empirical pathway models are, at present, large. However, as rate constants are defined for common species, and as experiments with different geochemical conditions are related to these mechanistic biological responses, adequate data should become available for site-specific exposure assessments.
One of the goals of developing such models is to help pinpoint those bioavailability tools that should be used in a particular situation. For example, DYMBAM has been used as part of a large framework for modeling selenium fate and transport in the San Francisco Bay (Luoma and Presser, 2001). In addition to DYMBAM predictions of bioaccumulated selenium in marine invertebrates, the framework also incorporates thermodynamic predictions of metal speciation, empirical observations of trophic transfer, and results from toxicity studies. As discussed in Box 4-11, preliminary results suggest that particulate selenium and selenium concentrations in bivalve tissues should be the target of measurement tools.
CONCLUSIONS AND RECOMMENDATIONS
A wide array of approaches can be used to better understand bioavailability processes in soils and sediments. Physical and chemical tests including modern spectroscopic techniques have been developed for determining contaminant form and better understanding contaminant–solid interactions. Simple extraction tests provide operational results about bioavailability. An array of biological approaches are available that vary widely in how they address bioavailability processes. Despite these advances, at the present time, the “tool box” of methods is incomplete. Table 4-1 confirms that few of the tools developed to date are ready for widespread application on any level other than as research tools. The following conclusions and recommendation summarize the future directions that bioavailability tools development should take.
At a given site, a suite of tools is needed to describe bioavailability processes in soils or sediments. No single tool has been developed that can univer-
sally describe or measure bioavailability, and approaches that have attempted this have failed. Thus, a complementary group of tools that characterize different bioavailability processes is a better choice than multiple tools that focus on only one step. Ideally, risk managers should consider processes influencing contaminant concentration, form, or transformation; biological processes affecting uptake; and linkages between internal concentrations and adverse effects in receptors. The complexity of this requirement illustrates the importance of a well-developed site conceptual model and a more comprehensive approach to exposure assessment as compared to a single-value regulatory approach in evaluating contaminant bioavailability. The corollary is that simple tests should be used cautiously. Simplification should only proceed once more mechanistic knowledge has become available, not in lieu of such information.
The suite of bioavailability tools should be comprised of a cluster of standardized and when possible validated protocols (physical, chemical, and biological assays) that are initially applicable at specific sites but with the potential of being general in application. New approaches must continue to be validated via intensive peer review, comparison against actual observations in natural systems or contaminated settings, and pilot testing before they can be applied as unambiguous regulatory tools. Strict validation criteria are necessary to avoid premature or inappropriate application of methods.
There are tradeoffs associated with the use of bioavailability tools such that not all tests for the same bioavailability process are of equal value. Different tests for a single bioavailability process may yield different kinds of information—a fact that must be considered carefully in evaluating the relevance and meaning of various experimental data. In addition, tests that aggregate the effects of multiple processes may have limited relevance to understanding mechanisms (if that is the primary objective), while tests that constrain certain variables for direct elucidation of mechanisms may have less relevance for assessment of overall toxicity.
To avoid misapplying bioavailability tools it is important to understand the environmental setting for which a tool was designed and intended. As illustrated in Box 4-5, some tools have been used in situations for which they were not designed. The long-term success of implementing considerations of bioavailability in the management of environmental hazards depends upon the development of improved models and measurement techniques appropriate to site-specific conditions. Confusion in the regulatory process could result if tools intended for other purposes are misapplied to soil and sediment management.
An intensive effort to develop mechanistic tools and models is critical to future development of bioavailability tools. Many operational tools (e.g., extractions, normalizations, and simple models) have proven ambiguous or shown
|
BOX 4-11 Forecasting Ecological Effects of Selenium from Discharge of Irrigation Drainage to San Francisco-Bay Knowledge of many contaminants, including selenium, has grown sufficiently that evaluations of risk can consider the full complexity of processes that lead from inputs to toxicity. A large drainage system has been proposed that would carry selenium-laden irrigation drainage from the farmlands of the San Joaquin Valley and discharge it into San Francisco Bay. Luoma and Presser (2001) introduced an ecosystem-scale modeling approach to forecast potential effects under different load scenarios associated with the irrigation drainage input. The step-by-step conceptual model is shown schematically and illustratively in Figures 4-8 and 4-9, respectively. At each step, field data, oceanographic principles, and/or simple modeling approaches are used to forecast how selenium is transferred to the next level. Luoma and Presser (2001) forecast environmental concentrations under three different climate (river inflow) conditions. Speciation was inferred from sources and transformations typical of Bay conditions. This then  FIGURE 4-8 Schematic ecosystem selenium model. |
large uncertainties in their estimates of bioavailability when rigorously tested. Such empirical tests cannot be extrapolated to other sites, nor can they be used with confidence to understand permanence or unforeseen conditions. They are poorly correlated across species and ranges of environmental conditions. Development of a suite of mechanistically based tools is the best way to overcome such limitations.
Not every bioavailability process must be evaluated and understood in order to get useful measurements for practical application. Rather, measurements can be made that provide information for a specific process or for an integrated group of processes. These might include macro-scale measurements, such as measures of accumulation in organisms, and micro-scale measurements of key processes such as desorption. Micro-scale measurements can be especially
useful for explaining observed variability in the accumulation of a chemical in organisms.
REFERENCES
Abbott, J. D., D. L. Borton, and S. W. Hinton. 1995. Pilot scale validation of the river/fish bioaccumulation modeling program for nonpolar hydrophobic organic compounds using the model compounds 2,3,7,8-TCDD and 2,3,7,8-TCDF. Environ. Toxicol. Chem. 14:1999-2012.
Achtnich, C., E. Fernandes, J.-M. Bollag, H.-J. Knackmuss, H. and Lenke. 1999. Covalent binding of reduced metabolites of [15N3] TNT to soil organic matter during a bioremediation process analyzed by 15N NMR spectroscopy. Environ. Sci. Technol. 33:4448-4456.
Addison, J. A., and S. B. Holmes. 1995. Comparison of forest soil microcosm and acute toxicity studies for determining effects of fenitrothian on earthworms. Ecotox. Environ. Safety 30:127-133.
Ahnstrom Z. S., and D. R. Parker. 1999. Development and assessment of a sequential extraction procedure for the fractionation of soil cadmium. Soil Sci. Soc. Am. J. 63:1650-1658.
Ait-Aissa, S., J. M. Porcher, A. P. Arrigo, and C. Lambre. 2000. Activation of the hsp70 promoter by environmental inorganic and organic chemicals: relationships with cytotoxicity and lipophilicity. Toxicology 145:147-157.
Alexander, R. R., and M. Alexander. 2000. Bioavailability of genotoxic compounds in soils. Environ. Sci. Technol. 34:1589-1593.
Allen-King, R. M., P. Grathwohl, and W. P. Ball. 2002. New paradigms for modeling paradigms for the sorption of hydrophobic organic chemicals to heterogeneous carbonaceous matter in soils, sediments, and rocks. Advances in Water Resources 25(8-12):985-1016.
Almas, A., B. R. Singh, and B. Salbu. 1999. Mobility of cadmium-109 and zinc-65 in soil influenced by equilibration time, temperature, and organic matter. J. Environ. Qual. 28:1742-1750.
Alpendurada, M. D. 2000. Solid-phase microextraction: a promising technique for sample preparation in environmental analysis. J. Chromatogr. A 889:3-14.
Amacher, M. C. 1996. Nickel, cadmium and lead. Pp. 739-768 In: Methods of soil analysis. Part 3— chemical methods. D. Sparks et al. (eds.). Madison, WI: American Society of Agronomy and the Soil Science Society of America.
Amann, R. I., W. Ludwig, and K. H. Schleifer. 1995. Phylogenetic identification and in situ detection of individual microbial cells without cultivation. Microbiological Reviews 59: 143-169.
American Society for Testing and Materials (ASTM). 1980. Standard practice for conducting acute toxicity tests with fishes, macroinvertebrates and amphibians. ASTM E 729-80. Philadelphia, PA: American Society for Testing and Materials.
ASTM. 1988. Standard guide for conducting bioconcentration tests with fishes and saltwater bivalve molluscs. E1022-84. Philadelphia, PA: American Society for Testing and Materials.
ASTM. 1994. Standard guide for designing biological tests with sediment. Annual ASTM Book of Standards 11.04. Philadelphia, PA: American Society for Testing and Materials.
ASTM. 1999. Standards on biological effects and environmental fate, 2nd edition. Philadelphia, PA: American Society for Testing and Materials. 996 p.
Amyot, M., B. Pinel-Alloul, and P. G. C. Campbell. 1994. Abiotic and seasonal factors influencing trace metal levels (Cd, Cu, Ni, Pb and Zn) in the freshwater amphipod Gammarus fasciatus in two fluvial lakes of the St. Lawrence River. Can. J. Fish. Aquat. Sci. 51:2003-2016.
Anderson, R. F., P. H. Santschi, U. P. Nyffeler, and S. L. Schiff. 1987. Validating the use of radiotracers as analogs of stable metal behavior enclosed aquatic ecosystem experiments. Can. J. Fish. Aquat. Sci. 44:251-259.
Anderson, W. C., R. C. Loehr, and B. P. Smith. 1999. Environmental availability of chlorinated organics, explosives and metals in soils. Annapolis, MD: American Academy of Engineers.
Ankley, G. T., R. A. Hoke, J. P. Giesy and P. V. Winger. 1989. Evaluation of the toxicity of marine sediments and dredge spoils with the Microtox® bioassay. Chemosphere 18:2069-2075.
Ankley, G. T., K. Lodge, D. J. Call, M. D. Balcer, L. T. Brooke, R. D. Johnson, P. M. Cook, R. G. Kreis, Jr., A. R. Carlson, G. J. Niemi, R. A. Hoke, C. W. West, J. P. Giesy, P. D. Jones, and Z. C. Fuyin. 1992a. Integrated assessment of contaminated sediments in the Lower Fox River and Green Bay, Wisconsin. Ecotoxicol. Environ. Safety 23:46-63.
Ankley, G. T., P. M. Cook, A. R. Carlson, D. J. Call, J. A. Swenson, H. F. Corcoran, and R. A. Hoke. 1992b. Bioaccumulation of PCBs from sediments by oligochaetes and fishes: comparison of laboratory and field studies. Can. J. Fish. Aquat. Sci. 49:2080-2085.
Anon. 1978. Aquatic toxicology comes of age. Environ. Sci. Technol. 12(1):23-24.
Anon. 1984. A subsurface flexible plastic enclosure for the in situ study of short term microbiological and chemical dynamics. Limnol. Oceanog. 29:1140-1144.
Apte, S. C., and G. E. Bately. 1995. Trace metal speciation of labile chemical species in natural waters and sediments: non-electrochemical approaches. Pp. 260-306 In: Metal speciation and bioavailability in aquatic systems. A. Tessier and D. Turner (eds.). Chicester: John Wiley and Sons Ltd.
Arnaud, C., M. Saint-Denis, J. F. Narbonne, P. Soler, and D. Ribera. 2000. Influences of different standardised test methods on biochemical responses in the earthworm Eisenia fetida andrei. Soil Biol. Biochem. 32: 67-73.
Au, A. P., and M. B. Reddy. 2000. Caco-2 cells can be used to assess human iron bioavailability from a semipurified meal. J. Nutr. 130:1329-1334.
Baars, A. J., H. Esselink, L. P. Jager, G. A. Posthuma-Trumpie, F. M. Van der Geld, and P. E. F. Zoun. 1995. Biomonitoring heavy metals using the barn owl (Tyto alba guttata): sources of variation especially relating to body condition. Arch. Environ. Contam. Toxicol. 28:471-486.
Ball, W. P., and P. V. Roberts. 1991. Long term sorption of halogenated organic chemicals by aquifer material. 2. Intraparticle diffusion. Environ. Sci. Technol. 25:1237-1249.
Banks, M. K., E. Lee, and A. P. Schwab. 1999. Evaluation of dissipation mechanisms for benzo [a]pyrene in the rhizosphere of tall fescue. J. Environ. Qual. 28:294-298.
Bargar, J. R., G. E. Brown, Jr., and G. A. Parks. 1998. Surface complexation of Pb(II) at the oxide-water interfaces. III. XAFS determination of Pb(II) and Pb(II)-chloro adsorption complexes on goethite and alumina. Geochim. Cosmochim. Acta 62:193-208.
Bargar, J. R., S. N. Towle, G. E. Brown, Jr., G. A. Parks. 1996. Outer-sphere Pb(II) adsorbed at specific surface sites on single crystal α-alumnia. Geochim. Cosmochim. Acta 60:3541-3547.
Bartosiewicz, M. J., D. Jenkins D, S. Penn, J. Emery, and A. Buckpitt. 2001b. Unique gene expression patterns in liver and kidney associated with exposure to chemical toxins. J. Pharmacol. Experiment. Therap. 297:895-905.
Bartosiewicz, M., S. Penn, and A. Buckpitt. 2001a. Applications of gene array in environmental toxicology: fingerprints of gene regulation associated with cadmium chloride, benzo(a)pyrene, and trichloroethylene. Environ. Health Perspect. 109:71-74.
Basta, N. T., and J. J. Sloan. 1999. Bioavailability of heavy metals in strongly acidic soils treated with exceptional quality biosolids. J. Environ. Qual. 28:633-638.
Basta, N., and R. Gradwohl. 2000. Estimation of heavy metal bioavailability in smelter-contaminated soils by a sequential extraction procedure. J. Soil Contam. 9:149-164.
Bauman, J. W., J. Liu, and C. D. Klaassen. 1993. Production of metallothionein and heat-shock proteins in response to metals. Fundamental and Applied Toxicology 21:15-22.
Belfroid, A., W. Seinen, M. van den Berg, J. Hermens, and K. van Gestel. 1995. Uptake, bioavailability and elimination of hydrophobic compounds in earthworms (Eisenia andrei) in field contaminated soil. Environ. Toxicol. Chem. 14:605-612.
Bell, P. F., R. L. Chaney and J. S. Angle. 1991. Free metal activity and total metal concentrations as indices of micronutrient availability to barley (Hordeum vulgare (L.) Klages). Plant and Soil 130:51-62.
Belzille, N., P. Lecomte, and A. Tessier. 1989. Testing readsorption of trace elements during partial chemical extractions of bottom sediments. Environ. Sci. Technol. 23:1015-1020.
Bertelsen, A. H., and V. E. Velculescu. 1998. High-throughput gene expression analysis using SAGE. Drug Discov. Today 3:152-159.
Berti, W. R., and L. W. Jacobs. 1996. Chemistry and phytotoxicity of soil trace elements from repeated sewage sludge applications. J. Environ. Qual. 25:1025-1032.
Berti, W., and S. Cunningham. 1997. In-place inactivation of Pb in Pb-contaminated soils. Environ. Sci. Technol. 31:1359-1364.
Bertsch, P. M., D. B. Hunter, S. R. Sutton, S. Bajt, M. L. Rivers. 1994. In situ chemical speciation of uranium in soils and sediments by micro x-ray absorption spectroscopy. Environ Sci. Technol. 28:980-984.
Bezwoda, W., R. Charlton, T. Bothwell, J. Torrance, and R. Mayet. 1978. The importance of gastric hydrochloric acid in the absorption of nonheme food iron. J. Lab. Clin. Med. 92(1):108-116.
Bidoglio, G., P. N. Gibson, M. O’Gorman, and K. J. Roberts. 1993. X-ray absorption spectroscopy investigation of surface redox transformations of thallium and chromium on colloidal mineral oxides. Geochim. Cosmochim. Acta 57:2389-2394.
Bidwell, A. M., and R. H. Dowdy. 1987. Cadmium and zinc availability to corn following termination of sewage sludge-applications. J. Environ. Qual. 16:438-422.
Bierkens, J. G. E. A. 2000. Applications and pitfalls of stress-proteins in biomonitoring. Toxicology 153:61-72.
Bierkens, J., G. Klein, P. Corbisier, R. Van Den Heuvel, L. Verschaeve, R. Weltens, and G. Schoeters. 1998. Comparative sensitivity of 20 bioassays for soil quality. Chemosphere 37:2935-47.
Binger, C. A., J. P. Martin, R. M. Allen-King, and M. Fowler. 1999. Variability of chlorinated-solvent sorption associated with oxidative weathering of kerogen. J. Contamin. Hydrol. 40:137-158.
Bingham, F. T., F. J. Peryea, and W. M. Jarrell. 1986. Metal toxicity to agricultural crops. Metal Ions Biol. Sys. 20:119-56.
Bishop, C., M. D. Koster, A. A. Chek, D. J. T. Hussell, and K. Jock. 1995. Chlorinated hydrocarbons and mercury in sediments, red-winged blackbirds (Agelaius phoniceus) and tree swallows (Tachycinata bicolor) from wetlands in the Great Lakes-St. Lawrence river Basin. Environ. Toxicol. Chem. 14:491-501.
Bitton, G. E. Garland, I.-C. Kong, J. L. Morel, and B. Koopman. 1996. A direct solid-phase assay specific for heavy metal toxicity. I. Methodology. J. Soil Contam. 5(4):385-394.
Bjorklund, E., S. Bowadt, L. Mathiasson, and S. B. Hawthorne. 1999. Determining PCB sorption/ desorption behavior on sediments using selective supercritical fluid extraction. 1. Desorption from historically contaminated samples. Environ. Sci. Technol. 33:2193-2203.
Bleam, W. F., and M. B. McBride. 1986. The chemistry of adsorbed Cu(II) and Mn(II) in aqueous titanium dioxide suspensions. J. Colloid Interface Sci. 103:124-132.
Blus, L. J., C. J. Henny, D. J. Hoffman, and R. A. Grove. 1993. Accumulation and effects of lead and cadmium on wood ducks near a mining and smelting complex in Idaho. Ecotoxicol. 2:139-154.
Boelsma, E., S. Gibbs, C. Faller, and M. Ponec. 2000. Characterization and comparison of reconstructed skin models: morphological and immunohistochemical evaluation. Acta Derm. Venereol. 80:82-88.
Boese, B. L., and H. Lee II. 1992. Synthesis of methods to predict bioaccumulation of sediment pollutants. ERL-N No. N232. Narragansett, RI: EPA/Environmental Research Laboratory.
Bonnell, M., J. C. Greene, and C. J. Reddy. 1995. Review of whole-organism bioassays: soil, fresh-water sediment and freshwater assessment in Canada. Ecotox. Environ. Safety 30:221-231.
Bonnet C., M. Babut, J.-F. Ferard, L. Martel, and J. Garric. 2000. Assessing the potential toxicity of resuspended sediment. Environ. Toxicol. Chem. 19:1290-1296.
Boularbeh, A., J. L. Morel, G. Bitton, and M. Mench. 1996. A direct solid phase assay specific for heavy-metal toxicity. II. Assessment of heavy-metal immobilization in soils and bioavailability to plants. J. Soil Contam. 5(4):395-404.
Breteler, R. J., I. Valiela, and J. M. Teal. 1981. Bioavailability of mercury in several northeastern US Spartina ecosystems. Estuarine, Coastal Shelf Sci. 12:155-166.
Brockway, D. L., J. Hill, J. R. Maudsley, and R. R. Lassiter. 1979. Development, replacability and modeling of naturally derived micro-cosms. Environ. Studies 13:149-158.
Bronaugh, R. L., and T. J. Franz. 1986. Vehicle effects on percutaneous absorption: in vivo and in vitro comparisons with human skin. Br. J. Dermatol. 115:1-11.
Bronaugh, R. L., and S. W. Collier. 1993. In vitro methods for measuring skin permeation. Pp. 93-111 In: Skin permeation—fundamentals and applications. J. L. Zatz (ed.). Wheaton, IL: Allured Publishing Corp.
Brown, S. L., R. L. Chaney, J. S. Angle, and A. J. Baker. 1994. Phytoremediation potential of Thlaspi caerulescens and Bladder Campion for zinc- and cadmium-contaminated soil. J. Environ. Qual. 23:1151-1157.
Brown, S. L., R. L. Chaney, J. S. Angle, and J. A. Ryan. 1998. The phytoavailability of cadmium to lettuce in long-term biosolids-amended soils. J. Environ. Qual. 27:1071-1078.
Brown, S. L., R. Chaney, and W. Berti. 1999. Field test of amendments to reduce the in situ availability of soil lead. Pp. 506-507 In: Abstracts of the 5th International Conference on Biogeochemistry of Trace Elements, July 11-15. W. W. Wenzel, D. C. Adriano, B. Alloway, H. E. Dorner, C. Keller, N. W. Lepp, M. Mench, R. Naidu, and G. M. Pierzynski (eds.). Vienna, Austria: International Society for Trace Element Research.
Brown, S. L., C. L. Henry, H. Compton, R. L. Chaney, and P. DeVolder. 2000. Using municipal biosolids in combination with other residuals to restore metal-contaminated mining areas. Chap.1 In: Proceedings of a symposium on mining, forest and land restoration: the successful use of residuals/biosolids/organic matter for reclamation activities, Denver, CO, July 17-20, 2000. Denver, CO: Rocky Mountain Water Environment Association.
Bunck, C., R. Prouty, and A., Krynitsky. 1987. Residues of organochlorine pesticides and polychlorinated biphenyls in starlings from the continental United States, 1982. Environ. Monit. Assess. 8:59-75.
Bunzl, K., M. Trautmaunsheimer, and P. Schramel. 1999. Partitioning of heavy metals in a soil contaminated by slag: a redistribution study. J. Environ. Qual. 1168-1173.
Burken, J. G., and J. L. Schnoor. 1997. Uptake and metabolism of atrazine by poplar trees. Environ. Sci. Technol. 31:1399-1406.
Burken, J. G., and J. L. Schnoor. 1998. Predictive relationships for uptake of organic contaminants by hybrid poplar trees. Environ. Sci. Technol. 32:3379-3385.
Burton, G. A., Jr. 1991. Assessing freshwater sediment toxicity. Environ. Toxicol. Chem. 10:1585-1627.
Burton, G. A., Jr, R. Pitt, and S. Clark. 2000. The role of traditional and novel toxicity test methods in assessing stormwater and sediment contamination. Critical Reviews in Environmental Science and Technology 30:413-447.
Cain, B., and C. Bunck. 1983. Residues of organochlorine compound in starlings, 1979. Environ. Monit. Assess. 3:161-172.
Cain, D. J., and S. N. Luoma. 1990. Influence of seasonal growth, age and environmental exposure on Cu and Ag in a bivalve indicator, Macoma balthica, in San Francisco Bay. Marine Ecol. Prog. Ser. 60:45-55.
Callahan, C. A. 1991. On-site methods for assessing chemical impact on the soil environment using earthworms: a case study at the Baird and McGuire Superfund site, Holbrook, Massachusetts. Environ. Toxicol. Chem. 10:817-826.
Campbell, P. G. C., and A. Tessier. 1989. Geochemistry and bioavailability of trace metals in sediments. Chapter 7 In: Aquatic ecotoxicology: fundamental concepts and methodologies, Volume 1. Boca Raton, FL: CRC Press.
Carroll, K. M., Harkness, M. R., Bracco, A. A. and Balcarel, R. R. 1994. Application of a permeant/ polymer diffusional model to the desorption of polychlorinated biphenyls from Hudson River sediments. Environ. Sci. Technol. 28:253-258.
Capuzzo, J. M., J. W. Farrington, P. Rantamaki, C. H. Clifford, B. A. Lancaster, D. F. Leavett, and X. Jia. 1989. The relationship between lipid composition and seasonal differences in the distribution of PCBs in Mytilus edulis L. Mar. Environ. Res. 28:259-264.
Casey, W. H., B. L. Phillips, J. P. Nordin, and D. J. Sullivan. 1998. The rates of exchange of water molecules from Al(III)-methylmalonate complexes: the effect of ring size. Geochim. Cosmochim. Acta 62:2789-2798.
CDM. 1994. Metal speciation report. California Gulch CERCLA site, Leadville, Colorado. Prepared for U.S. Environmental Protection Agency, Region VIII, Denver, Colorado. Denver, CO: Camp Dresser & McKee.
Cha, H. J., R. Srivastava, V. N. Vakharia, G. Rao, and W. E. Bentley. 1999. Green fluorescent protein as a noninvasive stress probe in resting Escherichia coli cells. Appl. Environ. Microbiol. 65:409-14.
Chaney, R. L., and J. A Ryan. 1994. Risk based standards for arsenic, lead and cadmium in urban soils. Frankfurt, Germany: DECHMEA.
Chaney, R. L., J. A. Ryan, Y.-M. Li and S. L. Brown. 1999. Soil cadmium as a threat to human health. Pp. 219-256 In: Cadmium in soils and plants. M. J. McLaughlin and B. R. Singh (eds.). Boston: Kluwer Academic Publishers.
Chaney, R. L., S. L. Brown, J. S. Angle, T. I. Stuczynski, W. L. Daniels, C. L. Henry, G. Siebielec, Y. M. Li, M. Malik, J. A. Ryan, and H. Compton. 2000. In situ remediation/reclamation/ restoration of metals contaminated soils using tailor-made biosolids mixtures. In: Proceedings of a symposium on mining, forest and land restoration: the successful use of residuals/biosolids/ organic matter for reclamation activities, Denver, CO, July 17-20, 2000 . Denver, CO: Rocky Mountain Water Environment Association.
Chaney, R. L, J. A. Ryan, U. Kukier, S. L. Brown, G. Siebielec, M. Malik, and J. S. Angle. 2001. Heavy metal aspects of compost use. In: Compost utilization in horticultural cropping systems. P. Stoffella and B. Kahn (eds.). Boca Raton, FL: Lewis Publishers.
Chang, A. C., A. L. Page, J. E. Warneke, and E. Grgurevic. 1984a. Sequential extraction of soil heavy metals following a sludge application. J. Environ. Qual. 13:33-38.
Chang, A. C., A. L. Page, K. W. Foster, and T. E. Jones. 1984b. A comparison of cadmium and zinc accumulation by four cultivars of barley grown in sludge-amended soils. J. Environ. Qual. 3:409-412.
Chang, L. W., J. R. Meier, and M. K. Smith. 1997. Application of plant and earthworm bioassays to evaluate remediation of a lead-contaminated soil. Arch. Environ. Contam. Toxicol. 32:166-171.
Chappie, D. J., and G. A. Burton, Jr. 2000. Applications of aquatic and sediment toxicity testing in situ. Soil and Sediment Contamination 9:219-245.
Charrois, J. W. A., W. B. McGill, and K. L. Froese. 2001. Acute ecotoxicity of creosote-contaminated soils to Eisenia fetida: a survival-based approach. Environ. Toxicol. Chem. 20:2594-2603.
Chaudri, A. M., K. Lawlor, S. Preston, G. I. Paton, K. Killham, and S. P. McGrath. 2000. Response of a rhizobium-based luminescence biosensor to Zn and Cu in soil solutions from sewage sludge treated soils. Soil Biology and Biochemistry 32:383-388.
Chen, Z., and L. M. Mayer. 1999. Assessment of sedimentary Cu availability: a comparison of biomimetic and AVS approaches. Environ. Sci. Technol. 33:6650-653.
Chen, S. C., L. C. Fitzpactrick, A. J. Goven, B. J. Verables, and E. L. Cooper. 1991. Nitrobule tetrazolium dye reduction by earthworm (Lumbricus Terrestris) coelomcytes: an enzyme assay for nonspecific immunotoxicity of xenobiotics. Environ. Toxicol. Chem. 10:1037-1043.
Cheung, V. G., M. Morley, F. Aguilar, A. Massimi, R. Kucherlapati, and G. Childs. 1999. Making and reading microarrays . Nat. Genet. Suppl. 21:15-19.
Chiou, C. T., D. E. Kile, D. W. Rutherford, G. Y. Sheng, and S. A. Boyd. 2000. Sorption of selected organic compounds from water to a peat soil and its humic-acid and humin fractions: potential sources of the sorption nonlinearity. Environ. Sci. Technol. 34(7):1254-1258.
Chung, N., and M. Alexander. 1998. Differences in sequestration and bioavailability of organic compounds aged in dissimilar soils. Environ. Sci. Technol. 32:855-860.
Chung, N., and M. Alexander. 1999. Effect of concentration on sequestration and bioavailability of two polycyclic aromatic hydrocarbons. Environ. Sci. Technol. 33:3603-3606.
Cioci, L. K., L. Qui, and J. H. Freedman. 2000. Transgenic strains of the nematode Caenorhabditis elegans as biomonitors of metal contamination. Environ. Toxicol. Chem. 19(8):2122-2129.
Cohen, J, B. D. Beck; T. S. Bowers, R. L. Bornschein, and E. J. Calabrese. 1998. An arsenic exposure model: probabilistic validation using empirical data. Human Ecol. Risk Assess. 4:341.
Combes, J. M., C. J. Chisolm-Brause, G. E. Brown, Jr., G. A. Parks, S. D. Conradson, P. G. Eller, I. R. Triay, D. E. Hobart, and A. Meijer. 1992. EXAFS spectroscopic study of neptunium(V) sorption at the α-FeOOH/water interface. Environ. Sci. Technol. 26:376-382.
Conder, J. M., R. P. Lanno, and N. T. Basta. 2001. Assessment of metal availability in a smelter soil using earthworms and chemical extractions. J. Environ. Qual. 30:1231-1237.
Connell, D. W., and R. D. Markwell. 1990. Bioaccumulation in the soil to earthworm systems. Chemosphere 20(1-2):91-100.
Corbisier, P. 1999. The use of whole-cell biosensors to detect bioavailable heavy metals (biomet) and genotoxic compounds (vititox). Ninth European Congress on Biotechnology. July, 1999.
Cornelissen, G., P. C. M. Van Noort, and A. J. Govers. 1997a. Desorption kinetics of chlorobenzenes, PAHs, and PCBs—sediment extraction with tenax and effects of contact time and solute hydrophobicity. Environ. Toxicol. Chem. 16:1351-1357.
Cornelissen, G., P. C. M. Noort, J. R. Parsons, and H. A. J. Govers. 1997b. Temperature dependence of slow adsorption and desorption kinetics of organic compounds in sediments . Environ. Sci. Technol. 31:454-460.
Cortet, J., A. Gomot-De Vauflery, N. Poinsot-Balaguer, L. Gomot, C. Texier, and D. Cluzeau. 1999. The use of invertebrate soil fauna in monitoring pollutant effects. Euro. J. Soil Biol. 35:115-134.
Costa, M. 1997. Toxicity and carcinogenicity of Cr(VI) in animal models and humans. Crit. Rev. Toxicol. 27(5):431–442.
Cox, F. R. 1968. Development of yield response prediction and manganese test interpretation for soybeans. Agron. J. 60:521-524.
Cox, J. A., K. Slonawska, and D. K. Gatchell. 1984. Metal speciation by Donnan dialysis. Anal. Chem. 56:650-653.
Craft, D. 1983. Microsystem sediment-water simulation: a practical technique for predicting reservoir water quality. U.S. Dept. of Interior Bureau of Land Management. Rec-Erc-83-12. 8 Pp.
Custer, T. W., D. W. Sparks, S. A. Sobeich, K. K. Hines, and M. J. Melancon. 1996. Organochlorine accumulation by sentinel mallards at the Winston-Thomas sewage treatment plant, Bloomington, Indiana . Arch. Environ. Contam. Toxicol. 30:163-169.
Cutshall, N. 1974. Turnover of zinc-65 in oysters. Health Physics 26:327-331.
Cutter, G. A. 1985. Determination of selenium speciation in biogenic particles and sediments. Analytical Chemistry 57:2951-2955.
Cuypers, C., T. Grotenhuis, J. Joziasse, and W. Rulkens. 2000. Rapid persulfate predicts PAH bioavailability in soils and sediments. Environ. Sci. Technol. 34:2057-2063.
Daniels, S. A. 1989. An improved elutriation technique for the bioassessment of sediment contaminants. Hydrobiologia 188/189:619-631.
Davies, J. M., and J. C. Gamble. 1979. Experiments with large enclosed ecosystems. Philos. Trans. R. Soc. London Ser. B. 286:523-544.
Davies, B. E. 1992. Inter-relationships between soil properties and the uptake of cadmium, copper, lead and zinc from contaminated soils by radish (Raphanus sativus L.). Water, Air and Soil Pollution 63:331-342.
Davis, A., N. S. Bloom, and S. S. Que Hee. 1997. The environmental geochemistry and bioaccessibility of mercury in soils and sediments: a review. Risk Anal. 17(5):557–569.
Davison, W., and H. Zhang. 1994. In situ speciation measurements of trace components in natural waters using thin-film gels. Nature 367:546-548.
Davison, W., P. Hooda, H. Zhang, and A. C. Edwards. 2000. DGT measured fluxes as surrogates for uptake of metals by plants. Advances in Environ. Res. 3:550-555.
DeCaprio, A. P. 1997. Biomarkers: coming of age for environmental health and risk assessment. Environ. Sci. Technol. 31:1837-1848.
DeCaprio, A. P. 2000. Biomarkers of exposure and susceptibility. Pp. 1875-1898 In: General and applied toxicology, Volume 3, 2nd edition. B. Ballantyne, T. Marrs, and T. Syversen (eds.). London: MacMillan Reference.
Decho, A. W., and S. N. Luoma. 1991. Time-courses in the retention of food material in the bivalves Potamocorbula amurensis and Macoma balthica: significance to the absorption of carbon and chromium. Marine Ecology Progress Series 78:303-314.
Decho, A. W., and S. N. Luoma. 1994. Humic and fulvic acids: sink or source in the availability of metals to the marine bivalves Macoma balthica and potamocorbula amurensis? Marine Ecology Progress Series 108:133-145.
Degens, B. P., and J. A. Harris. 1997. Development of a physiological approach to measuring the catabolic diversity of soil microbial communities. Soil Biology and Biochemistry 29(9-10): 1309-1320.
del Castilho, P., W. J. Chardon, and W. Salomons. 1993. Influence of cattle-manure slurry application on the solubility of cadmium, copper, and zinc in a manured acidic, loamy-sand soil. J. Environ. Qual. 22:689-697.
De Maagd, P., and J. Staeb. 2002. Identifying potential bioaccumulating substances in effluents using SPME-GCMS. Abstr. Annu. Meeting Soc. Environ. Toxicol. Chem., 80-59:263. Pensacola, FL: SETAC Press.
Depledge, M. H., J. J. Amaral-Mendes, B. Daniel, R. S. Halbrok, P. Kloepper-Sams, M. N. Moore, and D. B. Peakall. 1993. The conceptual basis of the biomarker approach. Pp. 19 In: Biomarker, NATO ASI Series, Vol. H68. D. B. Peakall and L. R. Shugart L. R. (eds.). Berlin: Springer.
deVries, M. P. C., and K. G. Tiller. 1978. Sewage sludge as a soil amendment, with special reference to Cd, Cu, Mn, Ni, Pb, and ZnS: comparison of results from experiments conducted inside and outside a greenhouse. Environ. Pollut. 16:213-240.
DeWitt, T. H., R. J. Ozretich, R. C. Swartz, J. O. Lamberson, D. W. Schults, G. R. Ditsworth, J. K. P. Jones, L. Hoselton, and L. M. Smith. 1992. The influence of organic matter quality on the toxicity and partitioning of sediment-associated fluoranthene. Environ. Toxicol. Chem. 11:197-208.
DiToro, D. M., J. D. Mahony, D. J. Hansen, K. J. Scott, M. B. Hicks, S. M. Mayr, and M. S. Redmond. 1990. Toxicity of cadmium in sediments: the role of acid volatile sulfides. Environ. Toxicol. Chem. 9:1487-1502.
DiToro, D. M., C. S. Zarba, D. J. Hansen, W. J. Berry, R. C. Swartz, C. E. Cowan, S. P. Pavlou, H. E. Allen, N. A. Thomas, and P. R. Paquin. 1991. Technical basis for establishing sediment quality criteria for nonionic organic chemicals using equilibrium partitioning. Environ. Toxicol. Chem. 10:1541-1583.
Doberman, A., J. L. Gaunt, H. U. Neue, I. F. Grant, M. A. Adviento, and M. F. Pampolino. 1994. Spatial and temporal variability of ammonium in flooded rice fields. Soil Sci. Soc. Am. J. 58:1708-1717.
Dodi, C. P., R. T. Bunch, S. W. Curtiss, L. D. Kier, M. A. Cabonce, J. C. Davila, M. D. Mitchell, C. L. Alden, and D. L. Morris. 1999. Revolution through genomics in investigative and discovery toxicology. Toxicologic Pathology 27:107-110.
Donat, S., and J. Abel. 1998. Analysis of gene expression in lung and thymus of TCDD treated C57BL/6 mice using differential display RT-PCR. Chemosphere 37:1867-1872.
Donkin, S. G., and D. B. Dusenbery. 1993. A soil toxicity test using the nematode Caenorhabditiselegans and an effective method of recovery. Arch. Environ. Contam. Toxicol. 25(2):145-151.
Drexler, J. 1997. Validation of in vitro testing. Presentation at IBC’s International Congress on human health—bioavailability: quantifying the toxicity of common soil contaminants. Scottsdale, Arizona. December 11–12, 1997.
Drobne, D. 1997. Terrestrial isopods—a good choice for toxicity of pollutants in the terrestrial environment. Environ. Toxicol. Chem. 16:1159-1164.
Duff, M. C., C. Amrhein, P. M. Bertsch, and D. B. Hunter. 1997. The chemistry of uranium in evaporation pond sediment in the San Joaquin Valley, California, USA using X-ray fluorescence and XANES techniques. Geochim. Cosmochim. Acta 61:73-81.
Dumestre, A., M. McBride, and P. Baveye. 2000. Use of EPR to monitor the distribution and availability of organic xenobiotics in model soil systems. Environ. Sci. Technol. 34:1259-1264.
Edwards, C. A., and K. Jeffs. 1973. Rate of uptake of DDT from soil by earthworms. Rothamsted Experimental Station, Harpenden, Hertfordshire.
Eick, M. J., and S. E. Fendorf. 1998. Reaction sequence of nickel sorption on kaolinite. Soil Sci. Soc. Am. J. 62:1257-1267.
Eisenberger, P., and B. Lengeler. 1980. Extended x-ray absorption fine-structure determinations of coordination numbers: limitations. Phys. Rev. B 22:3551-3562.
Eisert, R., and J. Pawliszyn. 1997. New trends in solid-phase microextraction. Crit. Rev. Anal. Chem. 27:103-135.
Emmerich, W. E., L. J. Lund, A. L. Page, and A. C. Chang. 1982. Solid phase forms of heavy metals in sewage sludge-treated soils. J. Environ. Qual. 11:178-181.
Environmental Protection Agency (EPA). 1975. Methods for acute toxicity tests with fish, macroinvertebrates and amphibians. EPA-600/3-75-009. Washington, DC: EPA.
EPA. 1987a. Bioaccumulation monitoring guidance: selection of target species and review of available data, Vol. I. EPA 430/9-86-005. Washington, DC: EPA Office of Marine and Estuarine Protection.
EPA. 1987b. Bioaccumulation monitoring guidance: selection of target species and review of available data: Vol II, Appendix. EPA 430/9-86-006. Washington, DC: EPA Office of Marine and Estuarine Protection.
EPA. 1994. Methods for measuring the toxicity and bioaccumulation of sediment-associated contaminants with freshwater invertebrates. Washington, DC: EPA Office of Research and Development.
EPA. 1995. A guide to the biosolids risk assessments for the EPA Part 503 Rule. EPA 832-B-93-005. Washington, DC: EPA Office of Wastewater Management.
EPA. 2000a. Bioaccumulation testing and interpretation for the purpose of sediment quality assessment-status and needs. EPA-823-R-00-001, I-113. Washington, DC: EPA.
EPA. 2000b. Appendix to bioaccumulation testing and interpretation for the purpose of sediment quality assessment: status and needs: chemical specific summary tables. EPA-823-R-00-002, i-816. Washington, DC: EPA.
EPA. 2000c. Methods for measuring the toxicity and bioaccumulation of sediment-associated contaminants with freshwater invertebrates. EPA/600/R-99/064, 1-192. Washington, DC: EPA Office of Water Office of Science and Technology.
EPA. 2001. Methods for the collection, storage, and manipulation of sediments for chemical and toxicological analysis: technical manual. EPA-823-B-01-002. Washington, DC: EPA Office of Water.
EPA Region 5. 1994. Predicting bioaccumulation of sediment contaminants to fish-draft report. Joint Health Effects Forum/In Place Pollutant Task Force Workgroup on Human Health Risks from Contaminated Sediments.
EPA and U.S. Army Corps of Engineers. 1991. Evaluation of dredged material proposed for ocean disposal—testing manual. EPA 503-8-91-001. Washington, DC: US EPA/USACE.
EPA and U.S. Army Corps of Engineers. 1998. Evaluation of dredged material proposed for discharge in waters of the U.S.: inland testing manual. EPA-823-B-98-004. Washington, DC: EPA/USACE.
Farley, R. A., and A. H. Fitter. 1999. Temporal and spatial variation in soil resources in a deciduous woodland. J. Ecol. 87:688-696.
Feng, Y., J. H. Park, T. C. Voice, and S. A. Boyd. 2000. Bioavailability of soil-sorbed biphenyl to bacteria. Environ. Sci. Technol. 34:1977-1984.
Fentem, J. H., D. Briggs, C. Chesne, G. R. Elliott, J. W. Harbell, J. R. Heylings, P. Portes, R. Roguet, J. J. van de Sandt, and P. A. Botham. 2001. A preevaluation study on in vitro tests for acute skin irritation. Results and evaluation by the Management Team. Toxicol. In Vitro 15:57-93.
Fisher, N. S., and J. L. Telyssie. 1986. Influence of food composition on the biokinetics and tissue distribution of zinc and americium in mussels. Mar. Ecol. Prog. Ser. 28:197-207.
Fitch, A., and P.A. Helmke. 1989. Donnan equilibrium/graphite furnace atomic absorption estimates of soil extract complexation capacities. Anal. Chem. 61:1295-1298.
Fitzgerald, D. G., K. A. Warner, R. P. Lanno, and D. G. Dixon. 1996. Assessing the effects of modifying factors on pentachlorophenol toxicity to earthworms: applications of body residues. Environ. Toxicol. Chem. 15:2299-2304.
Fossi, M. C., C. Leonzio, and D. B. Peakall. 1994. The use of nondestructive biomarkers in the hazard assessments of vertebrate populations. Pp. 3-34 In: Nondestructive biomarkers in vertebrates. Fossi M. C. and Leonzio C. (eds.). Boca Raton, FL: Lewis Publishers (CRC Press).
Fouchécourt, M. O., M. Arnold, P. Berny, B. Videmann, B. Rether, and J. L. Rivière. 1999. Assessment of the bioavailability of PAHs in rats exposed to a polluted soil by natural routs: induction of EROD activity and DNA adducts and PAH burden in both liver and lung. Environ. Res. 80:330-339.
Fouchécourt, M. O., P. Berny, and J. L. Rivière. 1998. Bioavailability of PCBs to male laboratory rats maintained on litters of contaminated soils: PCB burden and induction of alkoxyresorufin 0-dealkylase activities in liver and lung. Arch. Environ. Contam. Toxicol. 35:680-687.
Foy, C. D., R. L. Chaney, and M. C. White. 1978. The physiology of metal toxicity in plants. Ann. Rev. Plant Physiol. 29:511-66.
Fredrickson H. L., E. J. Perkins, T. S. Bridges, R. J. Tonucci, J. K. Fleming, A. Nagel, K. Diedrich, A. Mendez-Tenorio, M. J. Doktycz, and K. L. Beattie. 2001. Towards environmental toxicogenomics—development of a flow-through, high-density DNA hybridization array and its application to ecotoxicity assessment. Science of the Total Environment 274:137-149.
Friedl, G., B. Wehrli, and A. Manceau. 1997. Solid phases in the cycling of manganese in eutrophic lakes: new insights from EXAFS spectroscopy. Geochim. Cosmochim. Acta 61:275-290.
Friedrich, M., R. J. Grosser, E. A. Kern, W. P. Inskeep, and D. M. Ward. 2000. Effect of model sorptive phases on phenanthrene biodegradation: molecular analysis of enrichments and isolates suggests selection based on bioavailability. Appl. Environ. Microbiol. 66(7):2703-10.
Froese, K. L., D. A. Verbrugge, G. T. Ankley, G. J. Niemi, C. P. Larsen, and J. P. Giesy. 1998. Bioaccumulation of polychlorinated biphenyls from sediments to aquatic insects and tree swallow eggs and nestlings in Saginaw Bay, Michigan, USA. Environ. Toxicol. Chem. 17:484-492.
Garland, J. L., and A. L. Mills. 1991. Classification and characterization of heterotrophic microbial communities on the basis of patterns of community-level sole-carbon-source utilization. Appl. Environ. Microbiol. 57: 2351-2359.
Gettier, S. W., D. C. Martens, and S. J. Donohue. 1985. Soybean yield response prediction from soil test and tissue manganese levels. Agron. J. 77:63-67.
Ghosh, U., A. S. Weber, J. N. Jensen, and J. R. Smith. 1999. Congener level PCB desorption kinetics of field-contaminated sediments. J. Soil Contamination 8(5):593-613.
Ghosh, U., R. G. Luthy, J. S. Gillette, and R. N. Zare. 2000a. Microscale location, characterization, and association of polycyclic aromatic hydrocarbons on harbor sediment particles. Environ. Sci. Technol. 34:1729-1736.
Ghosh, U., A. S. Weber, J. N. Jensen, and J. R. Smith. 2000b. Relationship between PCB desorption equilibrium, kinetics, and availability during land biotreatment. Environ. Sci. Technol. 34:2542-2548.
Ghosh, U., J. W. Talley, and R. G. Luthy. 2001. Particle-scale investigation of PAH desorption kinetics and thermodynamics from sediment. Environ. Sci. Technol. 35(17):3468-3475.
Gibbs, M. H., L. F. Wicker, and A. J. Stewart. 1996. A method for assessing sublethal effects of contaminants in soils to the earthworm, Eisenia fetida. Environ. Toxicol. Chem. 15:360-368.
Giesy, J. P. 1980. Microcosms in ecological research. Springfield, VA: U.S. Department of Energy.
Giesy, J. P., and E. P. Odum. 1980. Microcosmology: the theoretical basis. Pp. 1-13 In: Microcosms in ecological research. DOE CONF-781101 . J. P. Giesy (ed.). Oak Ridge, TN: Department of Energy Technical Information Center.
Giesy, J. P., C. S. Rosiu, R. L. Graney, J. L. Newsted, A. Benda, R. G. Kreis, and F. J. Horvath. 1988. Toxicity of Detroit River sediment interstitial water to the bacterium Photobacterium Phosphoreum. J. Great Lakes Res. 14:502-513.
Giesy, J. P., and R. A. Hoke. 1989. Freshwater sediment toxicity bioassessment: rationale for species selection and test design. J. Great Lakes Res. 15:539-569.
Giesy, J. P., and R. A. Hoke. 1990. Freshwater sediment quality criteria: toxicity bioassessment. Pp. 265-348 In: Sediments: the chemistry and toxicology of in-place pollutants. R. Baudo (ed.). Chelsea: Lewis Publishers.
Giesy J. P., C. J. Rosiu, R. L. Graney, and M. G. Henry. 1990. Benthic invertebrate bioassays with toxic sediment and pore water. Environ. Toxicol. Chem. 9:233-248.
Gillette, J. S., R. G. Luthy, S. J. Clemett, and R. N. Zare. 1999. Direct observation of polycyclic aromatic hydrocarbons on geosorbents at the subparticle scale. Environ. Sci. Technol. 33(8): 1185-1192.
Glahn, R. P., C. Lai, J. Hsu, J. F. Thompson, M. Guo, and D. R. Van Campen. 1998. Decreased citrate improves iron availability from infant formula: application of an in vitro digestion/Caco2 cell culture model. J. Nutr. 128:257-264.
Gong, Y., J. V. Depinto, G.-Y. Rhee, and X. Liu. 1998. Desorption rates of two PCB congeners from suspended sediments: experimental results. Water Research 32:2507-2517.
Gong, P., B. M. Wilke, and S. Fleischmann. 1999a. Soil-based phytotoxicity of 2,4,6-trinitrotoluene (TNT) to terrestrial higher plants. Arch. Environ. Contam. Toxicol. 36:152-157.
Gong, P., S. D. Siciliano, C. W. Greer, L. Paquat, J. Hawari, and G. I. Sunahara. 1999b. Effects of bioavailability of 2,4,6-trinitrotoluene in spiked and field-contaminated soils to indigenous microorganisms. Environ. Toxicol. Chem. 18(12):2681-2688.
Gong, P., P. Gasparrini, D. Rho, J. Hawari, S. Thiboutot, G. Ampleman, and G. I. Sunahara. 2000. An in situ respirometric technique to measure pollution-induced microbial community tolerance in soils contaminated with 2,4,6-trinitrotoluene. Ecotoxicology and Environmental Safety 47(1):96-103.
Graney, R. L., J. P. Giesy, and J. R. Clark. 1995. Field studies. Pp. 257-305 In: Fundamentals of aquatic toxicology: effects, environmental fate and risk assessment, 2nd edition. G. M. Rand (ed.). Bristol, PA: Taylor & Francis.
Grathwohl, P. 1990. Influence of organic matter from soils and sediments from various origins on the sorption of some chlorinated aliphatic hydrocarbons: implications on Koc correlations. Environ. Sci. Technol. 24:1687-1693.
Gregg, S. J., and K. S. W. Sing. 1982. Adsorption, surface area and porosity, 2nd edition. New York: Academic Press. 303 pp.
Gruebel, K. A., J. A. Davis, and J. O. Leckie. 1988. The feasibility of using sequential extraction techniques for arsenic and selenium in soils and sediments. Soil Sci. Soc. Am. J. 52:390-397.
Griscom, S. B., N. S. Fisher, and S. N. Luoma. 2002. Kinetic modeling of Ag, Cd and Co bioaccumulation in the clam Macoma balthica: quantifying dietary and dissolved sources. Mar. Ecol. Progress Series 240:127-141.
Grosser, R. J., M. Friedrich, D. M. Ward, and W. P. Inskeep. 2000. Effect of model sorptive phases on phenanthrene biodegradation: different enrichment conditions influence bioavailability and selection of phenanthrene-degrading isolates. Appl. Environ. Microbiol. 66(7):2695-702.
Guerin, W. F., and S. A. Boyd. 1992. Differential bioavailability of soil-sorbed naphthalene to two bacterial species. Appl. Environ. Microbiol. 58(4):1142-52.
Guerin, W. F., and S. A. Boyd. 1997. Bioavailability of naphthalene associated with natural and synthetic sorbents. Water Research 31(6):1504-1512.
Guha, S., P. R. Jaffe, and C. A. Peters. 1998. Bioavailability of mixtures of PAHs partitioned into the micellar phase of a nonionic surfactant. Environ. Sci. Technol. 32(15):2317–2324.
Gustafson, K. E., and R. M. Dickhut. 1997. Distribution of polycyclic aromaric hydrocarbons in southern Chesapeake Bay surface waters: evaluation of three methods for determining freely dissolved water concentration. Environ. Toxicol. Chem. 16:452.
Hack, A., and F. Selenka. 1996. Mobilization of PAH and PCB from contaminated soil using a digestive tract model. Toxicol. Lett. 88:199–210.
Hageman, K. J., L. Mazeas, C. B. Grabanski, D. J. Miller, and S. B. Hawthorne. 1996. Coupled subcritical water extraction with solid-phase microextraction for determining semivolatile organics in environmental solids. Anal. Chem. 68:3892-3898.
Hani, H. 1996. Soil analysis as a tool to predict effects on the environment. Commun. Soil Sci. Plant. Anal. 27:289-306.
Hansen, L. H., and S. J. Sørensen. 2000. Versatile biosensor vectors for detection and quantification of mercury. FEMS Microbiol Lett. 193:123-7.
Haque. A., and W. Ebing. 1988. Uptake and accumulation of pentachlorophenol and sodium pentachlorophenate by earthworms from water and soil. Sci. Total Environ. 68:113-125.
Harkey, G. A., S. J. Klaine, and P. F. Landrum. 1994. Partition coefficients of hydrophobic contaminants in natural water, pure water, and elutriates obtained from dosed sediment: a comparison of methodologies. Chemosphere 28:583-596.
Harris, D. W., M. K. Kenrick, R. J. Pither, J. G. Anson, and D. A. Jones. 1996. Development of a high-volume in situ mRNA hybridization assay for the quantification of gene expression utilizing scintillating microplates . Anal. Biochem. 243:249-256.
Hartl, F. U. 1996. Molecular chaperones in cellular protein folding. Nature 381:571-80.
Hassett, J. P., and E. Milicic. 1985. Determination of equilibrium and rate constants for binding of a polychlorinated biphenyl congener by dissolved humic substances. Environ. Sci. Technol. 19:638-643.
Hatzinger, P. B., and M. Alexander. 1998. Biodegradation of organic compounds sequestered in organic solids or in nanopores within silica particles. Environ. Toxicol. Chem. 16:2215-2221.
Havenga, W. J., and E. R. Rohwer. 1999. Chemical characterization and screening of hydrocarbon pollution in industrial soils by headspace solid-phase microextraction. J. Chromatogr. A 848: 279-295.
Hawthorne, S. B., and C. B. Grabanski. 2000. Correlating selective supercritical fluid extraction with bioremediation behavior of PAHs in a field treatment plot. Environ. Sci. Technol. 34:4103-4110.
Hawthorne, S. B., C. B. Grabanski, K. J. Hageman, and D. J. Miller. 1998. Simple method for estimating polychlorinated biphenyl concentrations on soils and sediments using subcritical water extraction coupled with solid-phase microextraction. J. Chromatogr. A 814:151-160.
Hawthorne, S. B., C. B. Grabanski, E. Martin, and D. J. Miller. 2000a. Comparisons of Soxhlet extraction, pressurized liquid extraction, supercritical fluid extraction and subcritical water extraction for environmental solids: recovery, selectivity and effects on sample matrix. J. Chromatogr. A 892:421-433.
Hawthorne, S. B., S. Trembley, C. L. Moniot, C. B. Grabanski, and D. J. Miller. 2000b. Static subcritical water extraction with simultaneous solid-phase extraction for determining polycyclic aromatic hydrocarbons on environmental solids. J. Chromatogr. A 886:237-244.
Hayes, K. F., A. L. Roe, G. E. Brown, Jr., K. O. Hodgson, J. O. Leckie, G. A. Parks. 1987. In situ X-ray absorption study of surface complexes: selenium oxyanions on α-FeOOH. Science 238:783-786.
Heitzer, A., K. Malachowsky, J. E. Thonnard, P. R. Bienkowski, D. C. White, and G. S. Sayler. 1994. Optical biosensor for environmental on-line monitoring of naphthalene and salicylate bioavailability with an immobilized bioluminescent catabolic reporter bacterium. Appl. Environ. Microbiol. 60:1487-1494.
Heitzer, A., O. F. Webb, J. E. Thonnard, and G. S. Sayler. 1992. Specific and quantitative assessment of naphthalene and salicylate bioavailability by using a bioluminescent catabolic reporter bacterium. Appl. Environ. Microbiol. 58:1839-1846.
Helmke, P. A. 1999. Chemistry of cadmium in soil solution. Pp. 39-64 In: Cadmium in soils and plants. M. J. McLaughlin and B. R. Singh (eds.). Dordrecht, the Netherlands: Kluwer Academic Publishers.
Hernell, O., J. E. Staggers, and M. C. Carey. 1990. Physical-chemical behavior of dietary and biliary lipids during intestinal digestion and absorption. 2. Phase analysis and aggregation states of luminal lipids during duodenal fat digestion in health adult human beings. Biochemistry 29(8): 2041–2056.
Heron, G., M. J. Barcelona, M. L. Andersen, and T. H. Christensen. 1997. Determination of nonvolatile organic carbon in aquifer solids after carbonate removal by sulfurous acid. Ground Water 35(1):6-11.
Heslie, H. A., M. H. S. Kraak, and J. L. M. Hermens. 2002. Biomimetic extraction with SPME to estimate critical body residues and baseline toxicity. Abstr. Annu. Meeting Soc. Environ. Toxicol. Chem. 79-02:246. Pensacola, FL: SETAC Press.
Hightower, L. E. 1993. A brief perspective on the heat-shock response and stress proteins. Marine Environmental Research 35:79-83.
Hightower, L. E., P. T. Guide, S. A. Wholeness, and C. N. White. 1985. Stress responses in avian and mammalian cells. Pp. 197-210 In: Changes in eukaryotic gene expression in response to environmental stress. B. D. Atkinson and D. B. Waldens (eds.). New York: Academic Press.
Higuchi, R., C. Fockler, G. Dollinger, and R. Watson. 1993. Kinetic PCR analysis: real-time monitoring of DNA amplification reactions. Biotechnology 11:1026-1030.
Hill, G. T., N. A. Mitkowski, L. Aldrich-Wolfeb, L. R. Emelea, D. D. Jurkoniea, A. Fickea, S. Maldonado-Ramireza, S. T. Lyncha, and E. B. Nelsona. 2000. Methods for assessing the composition and diversity of soil microbial communities. Applied Soil Ecology 15(1):25-36.
Ho, M. D., and G. J. Evans. 1997. Operational speciation of cadmium, copper, lead and zinc in the NIST standard reference materials 2710 and 2711 (Montana soil) by the BRC sequential extraction procedure and flame atomic adsorption spectrometry. Anal. Comm. 34:363-364.
Hoke, R. A., J. P. Giesy, and R. G. Kreis, Jr. 1992. Sediment pore water toxicity identification in the Lower Fox River and Green Bay, Wisconsin, using the Microtox assay. Ecotoxicol. Environ. Safety 23:343-354.
Hoke, R. A., M. Zabik, J. P. Giesy, and M. Unger. 1993. Toxicity of sediment and sediment pore water from the Grand Calumet River—Indiana Harbor, Indiana area of concern. Ecotoxicol. Environ. Safety 26:86-112.
Holm, P. E., T. H. Christensen, J. C. Tjell, and S. P. McGrath. 1995. Heavy metals in the environment: speciation of cadmium and zinc with application to soil solutions. J. Environ. Qual. 24:183-195.
Holman, H. N. 2000. In vitro gastrointestinal mimetic protocol for measuring bioavailable contaminants. United States Patent No. 6,040,188 (March 21, 2000).
Holmstrup, M. 2000. Field assessment of toxic effects on reproduction in the earthworms Aporrectodea Longa and Aporrectodea Rosea. Environ. Toxicol. Chem. 19:1781-1787.
Hooda, P. S., H. Zhang, W. Davison, and A. C. Edwards. 1999. Measuring bioavailable trace metals by diffusive gradients in thin films (DGT): soil moisture effects on its performance in soil. Eur. J. Soil Sci. 44:97-110.
Horowitz, S. B., and B. L. Finley. 1993. Using human sweat to extract chromium from chromite or processing residue: applications to setting health-based cleanup levels. J. Toxicol. Environ. Health 40:585-599.
Hothem, R. L., and D. Welsh. 1994. Contaminants in eggs of aquatic birds from the grasslands of central California. Arch. Environ. Contam. Toxicol. 27(2):180-185.
Huckins, J. N., M. W. Tubergen, and G. K. Manuweera. 1990. Semipermeable membrane divices containing a model lipid: a new approach to monitoring the bioavailability of lipophilic contaminants and estimating their bioconcentration potential. Chemosphere 20:533.
Huckins, J. N., G. K. Manuweera., J. D. Petty, D. Mackay, and J. A. Lebo. 1993. Lipid-containing semipermeable membrane devices for monitoring organic contaminants in water. Environ. Sci. Technol. 27:2489-2496.
Huckins, J. N., Petty, J. D., Lebo, J. A., Orazio, C. E., Prest, H. F., Tillitt, D. E., Ellis, G. S., Johnson, B. T., Manuweera, G. K. 1996. Semipermeable membrane devices (SPMDs) for the concentration and assessment of bioavailable organic contaminants in aquatic environments. Pp. 625-655 In: Techniques in aquatic toxicology. G. K. Ostrander (ed.). Boca Raton, FL: Lewis Publishers.
Hugget, R. J., R. A. Kimerie, P. M. Mehrle, Jr., and H. L. Bergman. 1992. Biomarkers: biochemical, physiological, and histological markers of anthropogenic stress. Boca Raton, FL: Lewis Publishers.
Hughes, J. B., J. Shanks, M. Vanderford, J. Lauritzen, and R. Bhandra. 1996. Transformation of TNT by aquatic plants and plant tissue cultures. Environ. Sci. Technol. 31:266-271.
Ibekwe, A. M., and A. C. Kennedy. 1998. Phospholipid fatty acid profiles and carbon utilization patterns for analysis of microbial community structure under field and greenhouse conditions. FEMS Microbiology Ecology 26(2):151-163.
Ingersoll, C. G., G. Ankley, D. A. Benoit, E. L. Brunson, G. A. Burton, F. J. Dwyer, R. A. Hoke, P. F. Landrum, T. J. Norberg-King, and P. V. Winger. 1995. Toxicity and bioaccumulation of sediment-associated contaminants using freshwater invertebrates: a review of methods and applications. Environ. Toxicol. Chem. 14:1885-1894.
Ingram, J. C., G. S. Groenwold, A. D. Applehans, D. A. Dahl, and J. F. Delmore. 1996. Detection limits and surface coverage determinations for tributyl phosphate on soils by static SIMS. Analytical Chem. 68:1309-1316.
Ingram, J. C., G. S. Groenwold, A. D. Applehans, J. E. Delmore, J. E. Olson, and D. L. Miller. 1997. Direct surface analysis of pesticides on soil, leaves, grass, and stainless steel by static secondary ion mass spectrometry. Environ. Sci. Technol. 31:402-408.
International Standardization Organization (ISO). 1996. Soil quality—effects of pollutants on earthworms (Eisenia fetida). Part 2: Determination of effects on reproduction. ISO 11268-2. Geneva, Switzerland.
International Standardization Organization (ISO). 1999. Soil quality—inhibition of reproduction of Collembola (Folsomla candida) by soil pollutants. ISO 11267. Geneva, Switzerland.
Jager, T. 1998. Mechanistic approach for estimating bioconcentration of organic chemicals in earthworms (Oligochaeta). Environ. Toxicol. Chem. 17(10):2080-2090.
James, B. R., J. C. Petura, R. J. Vitale, and G. R. Mussoline. 1997. Oxidation-reduction chemistry of chromium: relevance to the regulation and remediation of chromate-contaminated soils. J. Soil Contam. 6(6):569-580.
James, H. M., M. E. Hilburn, and J. A. Blair. 1985. Effects of meals and meal times on uptake of lead from the gastrointestinal tract in humans. Human Toxicol. 4:401-407.
Janssen, R. P. T., L. Posthuma, R. Baerselman, and H. A. Den Hollander. 1997. Equilibrium partitioning of heavy metals in Dutch field soils. II. Prediction of metal accumulation in earthworms. Environ. Toxicol. Chem. 16(12):2479-2488.
Jardine, P. M., S. E. Fendorf, M. A. Mayes, I. L. Larsen, S. C. Brooks, and W. B. Bailey. 1999. Fate and transport of hexavalent chromium in undisturbed heterogeneous soil. Environ. Sci. Technol. 2939-2944.
Jenne, E. A. 1977. Trace element sorption by sediments and soils: sites and processes. Pp. 425-553 In: Molybdenum in the environment. W. R. Chappell and K. K. Peterson (eds.). New York: Marcel Dekker.
Jenne, E. A., and S. N. Luoma. 1977. Forms of trace elements in soils, sediments, and associated waters: an overview of their determination and biological availability. Pp. 110-143 In: Biological implications of metals in the environment. R. E. Wildung and H. Drucker (eds.).
Jing, J., and T. J. Logan. 1991. Chelating resin method for estimation of sludge-cadmium bioavailability. Commun. Soil Sci. Plant Anal. 22:2029-2035.
Johansson, M., M. Pell, and J. Stenström. 1998. Kinetics of substrate-induced respiration (SIR) and denitrification: applications to a soil amended with silver. Ambio 27(1):40-44.
Jones, P. D., K. Kannan, J. L. Newsted, D. E. Tillitt, L. L. Williams, and J. P. Giesy, J. P. 2001. Accumulation of 2,3,7,8-tetrachlorodibenzo-p-dioxin by rainbow trout (Onchorhynchus mykiss) at environmentally relevant dietary concentrations. Environ. Toxicol. Chem. 20(2):344-350.
Joyner, D. C., and S. E. Lindow. 2000. Heterogeneity of iron bioavailability on plants assessed with a whole-cell GFP-based bacterial biosensor. Microbiology 146:2435-45.
Junta-Rosso, J. L., and M. F. Hochella, Jr. 1994. Manganese (II) oxidation at mineral surfaces: a microscopic and spectroscopic study. Geochim. Cosmochim. Acta 58:4985-4999.
Kalra, Y. P. 1998. Handbook of reference methods for plant analysis. Boca Raton, FL: St. Lucie Press.
Kandeler, E., D. Tscherko, and H. Spiegel. 1999. Long-term monitoring of microbial biomass, N mineralisation and enzyme activities of a Chernozem under different tillage management. Biology and Fertility of Soils 28(4):343-351.
Karapanagioti, H., S. Kleineidam, D. A. Sabatini, G. Grathwohl, and B. Ligouis. 2000. Impacts of heterogeneous organic matter on phenanthrene sorption: equilibrium and kinetic studies with aquifer material. Environ. Sci. Technol. 34:406-414.
Karickhoff, S. W., and K. Morris. 1985. Sorption dynamics of hydrophobic pollutants in sediment suspensions. Environ. Toxicol. Chem. 4:469-479.
Kegelmeyer, A. E., C. S. Sprankle, G. J. Horesovsky, and B. E. Butterworth. 1997. Differential display identified changes in mRNA levels in regenerating livers from chloroform-treated mice. Mol. Carcinog. 20:288-297.
Keith, J. O. 1996. Residue analyses: how they were used to assess the hazards of contaminants to wildlife. Pp. 1-48 In: Environmental contaminants in wildlife. W. N. Beyer, G. H. Heinz, and A. W. Redmon-Norwood (eds.). Boca Raton, FL: Lewis Publishers.
Kelley, M. E., S. E. Brauning, R. A. Schoof, and M. V. Ruby. 2002. Assessing oral bioavailability of metals in soil. Columbus, OH: Battelle Press.
Kelly, J. J., and R. L. Tate. 1998. Effects of heavy metal contamination and remediation on soil microbial communities in the vicinity of a zinc smelter . J. Environ. Qual. 27(3):609-617.
Kelsey, J. W., and M. Alexander. 1997. Declining bioavailability and inappropriate estimation of risk of persistent compounds. Environ. Toxicol. Chem. 16:582-585.
Kelsey, J. W., B. D. Kottler, and M. Alexander. 1997. Selective chemical extractants to predict bioavailability of soil-aged organic chemicals. Environ. Sci. Technol. 31:214-217.
Kemler, K., P. D. Jones, and J. P. Giesy. 2000. Risk assessment of 2.3.7.8-tetrachlorodibenzo-p-dioxin equivalents in tissue samples form three species in the Denver metropolitan area. Human and Ecological Risk Assessment 6(2):1087-1099.
Kidd, K. A., D. W. Schindler, D. C. G. Muir, W. L. Lockhart, and R. H. Hesslein. 1995. High concentrations of toxaphene in fishes from a subarctic lake. Science 269:240-242.
Kleineidam, S., H. Rugner, B. Ligouis, and P. Grathwohl. 1999. Organic matter facies and equilibrium sorption of phenanthrene. Environ. Sci. Technol. 33:1637-1644.
Kleinow, K. M., M. O. James, Z. Tong, and C. S. Venugopalan. 1998. Bioavailability and biotransformation of benzo(a)pyrene in an isolated perfused in situ catfish intestinal preparation. Environ. Health Perspect. 106:155-166.
Knicker, H., D. Brunsnagel, O. Drzyzga, E. V. Low, and K. Steinbach. 1999. Characterization of 15N-TNT residues after an anaerobic/aerobic treatment of soil/molasses mixtures by solid-state 15N NMR spectroscopy. 1. Determination and optimization of relevant NMR spectroscopic parameters. Environ. Sci. Technol. 33:343-349.
Knight, B. P., A. M. Chaudri, S. P. McGrath, and K. E. Giller. 1998. Determination of chemical availability of cadmium and zinc using inert soil moisture samplers. Environ. Pollut. 99(3):293-298.
Kobayashi, J. 1978. Pollution by cadmium and the itai-itai disease in Japan. Pp. 199-260 In: Toxicity of heavy metals in the environment. F. W. Oehme (ed.). New York: Marcel Dekker, Inc.
Koganti, A., D. A. Spina, K. Rozett, B.-L. Ma, and E. H. Weyand. 1998. Studies on the applicability of biomarkers in estimating the systemic bioavailability of polynuclear aromatic hydrocarbons from manufactured gas plant tar-contaminated soils. Environ. Sci. Technol. 32:3104-3112.
Kraaij, H., G. Cornelissen, A. Belfroid, D. H. T. M. Sijm, and J. Tolls. 1998. A combined desorption/ bioaccumulation study on PAHs in aged and nonaged sediments. Poster presented at the 18th Annual SETAC meeting, Charlotte, NC.
Krauss, M., and W. Wilcke. 2001. Biomimetic extraction of PAHs and PCBs from soil with octadecyl-modified silica disks to predict their availability to earthworms. Environ. Sci. Technol. 35:3931-3935.
Krieger, M. S., J. L. Wynn, and R. N. Yoder. 2000. Extraction of cloransulam-methyl from soil with subcritical water and supercritical CO2. J. Chromatography 897(1-2):405-413.
Krishnan, G., G. L. Horst, S. Darnell, and W. L. Powers. 2000. Growth and development of smooth broomegras and tall fescue in TNT-contaminated soil. Environ. Pollut. 107:109-116.
Kuo, S. 1990. Cadmium buffering capacity and accumulation in Swiss chard in some sludge-amended soils. Soil Sci. Soc. Am. J. 54:86-91.
Lake, J. L., R. McKinney, F. A. Osterman, and C. A. Lake. 1996. 18C-coated silica particles as a surrogate for benthic uptake of hydrophobic compounds from bedded sediment. Environ. Toxicol. Chem. 15:2284.
Landrum, P. F., H. Lee II, and M. J. Lydy. 1992. Toxicokinetics in aquatic systems: model comparisons and use in hazard assessment. Environ. Toxicol. Chem. 11:1709-1725.
Langston, W. J. 1980. Arsenic in UK estuarine sediments and its availability to benthic organisms. J. Mar. Biol. Assn. UK 60:869-881.
Langston, W. J. 1982. The distribution of mercury in British estuarine sediments and its availability to deposit feeding bivalves. J. Mar. Biol. Assn. UK. 62:667-684.
Lanno, R. P., S. C. LeBlanc, B. L. Knight, R. Tymowski, and D. G. Fitzgerald. 1997. Application of body residues as a tool in the assessment of soil toxicity. Pp. 41-53 In: Advances in earthworm ecotoxicology. S. Separd, J. Bmbridge, M. Holmstrup, and L. Posthuma (eds.). Pensacola, FL: SETAC Press.
Laperche, V., T. J. Logan, P. Gaddam, and S. J. Traina. 1997. Effect of apatite amendments on plant uptake of lead from contaminated soil. Environ. Sci. Technol. 31:2745-2753.
Larsson, P., and A. Sodergren. 1987. Transport of polychlorinated biphenyls (PCBs) in freshwater mesocosms from sediment to water and air. Water, Air and Soil Poll. 36:33-46.
Larsson, P. 1984. Uptake of sediment released PCBs by the eel Angela anguilla in static model-systems. Ecol. Bull. (Stockholm) 36:62-67.
Larsson, P. 1983. Transport of 14C-labeled PCB compounds from sediment to water and from water to air in laboratory model systems. Water Research 17(10):1317-1326.
Larsson, P. 1985. Change in PCB (Clophen A 50) composition when transported from sediment to air in aquatic model systems. Environ. Pollution (Series B) 9:81-94.
Lebo, J. A., J. L. Zajicek, J. N. Huckins, J. D. Petty, and P. H. Peterman. 1992. Use of semipermeable-membrane devices for in situ monitoring of polycyclic aromatic hydrocarbons in aquatic environments. Chemosphere 25:697.
Lebourg, A., T. Sterckeman, H. Ciesielski, and N. Proiz. 1996. Suitability of chemical extraction to assess risks of toxicity induced by soil trace metal bioavailability. Agronomie 16:201-215.
Lee, G. F. 1978. Evaluation of the elutriate test as a method of predicting contaminant release during open-water disposal of dredged sediments and environmental impact of open-water dredged material disposal. Dredged Material Res. Prog. Vol. II. 611p.
Lee, D. Y., and H. C. Zhang. 1993. Chelating resin membrane method for estimation of soil cadmium phytoavailability. Commun. Soil Sci. Plant Anal. 24:685-700.
Lee, B.-G., S. B. Griscom, J.-S. Lee, H. J. Choi, C.-H. Koh, S. N. Luoma, and N. S. Fisher. 2000. Influence of dietary uptake and reactive sulfides on metal bioavailability from aquatic sediments. Science 287:282-284.
Lennernas, H. 1998. Human intestinal permeability. J. Pharm. Sci. 87:403-410.
Leschber, R., R. D. Davis, and P. L. Hermite (eds.). 1984. Chemical methods for assessing bioavailable metals in sludges and soils. Essex, England: Elsevier Applied Science Publishers.
Li, Y. M., R. L. Chaney, G. Siebielec, and B. A. Kerschner. 2000. Response of four turfgrass cultivars to limestone and biosolids-compost amendment of a zinc and cadmium contaminated soil at Palmerton, Pennsylvania. J. Environ. Qual. 29:1440-1447.
Li, Z., and L. M. Shuman. 1997. Mehlich-1 and DTPA-extractable lead in soils in relation to soil properties. Commun. Soil Sci. Plant Anal. 28(3-5):351-363.
Liang, L., and A. B. Pardee. 1992. Differential display of eukaryotic messenger RNA by means of the polymerase chain reaction. Science 257:967-971.
Lin, Q., and P. C. Brookes. 1999. An evaluation of the substrate-induced respiration method. Soil Biology and Biochemistry 31(14):1969-1983.
Lindahl, V., A. Frostegard, L. Bakken, and E. Baath. 1997. Phospholipid fatty acid composition of size fractionated indigenous soil bacteria. Soil Biology and Biochemistry 29(9-10):1565-1569.
Lindsay, W. L., and W. A. Norvell. 1978. Development of a DTPA soil test for zinc, iron, manganese and copper. Soil Sci. Soc. Am. J. 42:421-428.
Ling, H., M. Diamond, and D. Mackay. 1993. Application of the QWASI fugacity/equivalence model to assessing sources and fate of contaminants in Hamilton Harbor. J. Great Lakes Res. 19:582-602.
Liste, H.-H., and M. Alexander. 2002. Butanol extraction to predict bioavailability of PAHs in soil. Chemophere 46:1011-1017.
Liu, J., M. B. Kadiiska, Y. Liu, T. Lu, W. Qu and M. P. Waalkes. 2001. Stress-related gene expression in mice treated with inorganic arsenicals. Toxicol. Sci. 61:314-320.
Liu, W. T., T. L. Marsh, H. Cheng, and L. J. Forney. 1997. Characterization of microbial diversity by determining terminal restriction fragment length polymorphisms of genes encoding 16S rRNA. Appl. Environ. Microbiol. 63(11):4516-22.
Llompart, M., K. Li, and M. Fingas. 1999. Headspace solid phase microextraction (HSSPME) for the determination of volatile and semivolatile pollutants in soils. Talanta 48: 451-459.
Loeppert, R. H., and W. P. Inskeep. 1996. Iron. Pp. 639-664 In: Methods of soil analysis. Part 3— chemical methods. D. Sparks et al. (eds.). Madison, WI: American Society of Agronomy and the Soil Science Society of America.
Logan, R. J., and R. L. Chaney. 1983. Utilization of municipal wastewater and sludge on land— metals. Pp. 235-323 In: Utilization of municipal wastewater and sludge on land. A. L. Page, T. L. Gleason, J. E. Smith, A. K. Iskander and L. E. Sommers (eds.). Riverside, CA: Univ. California.
Løkke, H., and C. A. M. van Gestel. 1998. Handbook of soil invertebrate toxicity tests. Chicester, UK: John Wiley & Sons.
Loper, J. E., and S. E. Lindow. 1994. A biological sensor for iron available to bacteria in their habitats on plants. Appl. Environ. Microbiol. 60:1934-1941.
Lu, X., D. Johnson, and J. Hook. 1998. Reaction of vanadate with aquatic humic substances: an ESR and 51V NMR study. Environ. Sci. Technol. 32: 2257-2263.
Ludwig, D. D. 1989. Evaluation of the standard elutriate test as an estimator of contaminant release at dredging sites. Res. J. Water Pollut. Control Fed. 61:1666-72.
Lueking, A. D., W. L. Huang, S. Soderstrom Schwarz, M. S. Kim, and W. J. Weber. 2000. Relationship of soil organic matter characteristics to organic contaminant sequestration and bioavailability. J. Environ. Qual. 29(1):317-323.
Luoma, S. N. 1976. The uptake and interorgan distribution of mercury in a carnivorous crab. Bulletin of Environmental Contamination and Toxicology 16(6):719-723.
Luoma, S. N. 1977. Physiological characteristics of mercury uptake by two estuarine species. Marine Biology 41:269-273.
Luoma, S. N. 1989. Can we determine the biological availability of sediment-bound trace elements? Hydrobiologia 176/177:379-396.
Luoma, S. N. 1996. The developing framework of marine ecotoxicology: pollutants as a variable in marine ecosystems? J. Exptl. Mar. Biol. Ecol. 200:29-55.
Luoma, S. N., and G. W. Bryan. 1978. Factors controlling availability of sediment-bound lead to the estuarine bivalve Scorbicularia plana. J. Mar. Biol. Assoc. 58:793-802.
Luoma, S. N., and G. W. Bryan. 1982. A statistical study of environmental factors controlling concentrations of heavy metals in the burrowing bivalve Scorbicularia plana and the polychaete Neris diversicolor. Estuarine, Coastal and Shelf Sci. 15:95-108.
Luoma, S. N., and J. A. Davis. 1983. Requirements for modeling trace metal partitioning in oxidized estuarine sediments. Marine Chemistry 12:159-181.
Luoma, S. N., J. Carter, and C. Brown. 1991. Assessing adverse effects of contaminants on benthic species at the San Francisco Toxics Site. Pp. 667-672 In: Toxic Substances Hydrology Program. G. E. Mallard and D. A. Aronson (eds.). WRI 91-4034.
Luoma, S. N., C. Johns, N. S. Fisher, N. A. Steinberg, R. S. Oremland, and J. Reinfelder. 1992. Determination of selenium bioavailability to a benthic bivalve from particulate and solute pathways. Environ. Sci. Technol. 26:485-491.
Luoma, S. N., and N. Fisher. 1995. Uncertainties in assessing contaminant exposure from sediments. Bioavailability in sediment risk assessment. In: Ecological risk assessments of contaminated sediments. G. Biddinger and T. Dillon (eds.). Pellston Workshop, Asilomar, CA.
Luoma, S. N., and N. Fisher. 1997. Uncertainties in assessing contaminant exposure from sediments. Pp. 211-237 In: Ecological risk assessment in contaminated sediments. C. G. Ingersoll, T. Dillon, and G. R. Biddinger (eds.). Pensacola, FL: SETAC Press.
Luoma, S. N., and K. T. Ho. 1993. The appropriate uses of marine and estuarine sediment bioassays. Pp. 193-226 In: The handbook of ecotoxicology. P. Calow (ed.). Oxford: Blackwell Scientific Press.
Luoma, S. N., and E. A. Jenne. 1977. The availability of sediment-bound cobalt, silver, and zinc to a deposit-feeding clam. Pp. 213-230 In: Biological implications of metals in the environment. R. W. Wildung and H. Drucker (eds.).
Luoma, S. N., and T. P. Presser. 2001. Forecasting selenium discharges to the San Francisco Bay– Delta estuary: ecological effects of a proposed San Luis drain extension. U.S. Geological Survey Professional Paper P1646. 217 pp.
Mackay, D., and S. Paterson. 1991. Evaluating the multimedia fate of organic chemicals: a level III fugacity model. Environ. Sci. Technol. 25:427-436.
MacNaughton, S. J., J. R. Stephen, A. D. Venosa, G. A. Davis, Y.-J. Chang, and D. C. White. 1999. Microbial population changes during bioremediation of an experimental oil spill. Appl. Environ. Microbiol. 65(8):3566-3574.
Macrae J. D., and K. J. Hall. 1998. Comparison of methods used to determine the availability of polycyclic aromatic hydrocarbons in marine sediment. Environ. Sci. Technol. 32:3809-3815.
Maddaloni, M., N. LoIacono, W. Manton, C. Blum, J. Drexler, and J. Graziano. 1998. Bioavailability of soilborne lead in adults, by stable isotope dilution . Environ. Health Persp. 106(6):1589-1594.
Magee, P. 1991. Complex factors in hydrocarbon/water soil/water and fish/water partitioning. Science of the Total Environment 109(110):155-178.
Manceau, A., and D. L. Gallup. 1997. Removal of selenocyanate in water by precipitation: characterization of copper-selenium precipitate by x-ray diffraction, infrared, and x-ray absorption spectroscopy. Environ. Sci. Technol. 31:968-976.
Margesin, R., A. Zimmerbauer, and F. Schinner. 2000. Monitoring of bioremediation by soil biological activities. Chemosphere 40(4):339-346.
Marschner, H. 1995. Mineral nutrition of higher plants, second edition. New York: Academic Press.
Marshner, H. 1998. Soil-root interface: biological and biochemical processes. Pp. 191-232 In: Soil chemistry and ecosystem health. P. M. Huang (ed.). Madison, WI: Soil Science Society of America.
Mayer, L. M., Z. Chen, R. H. Findlay, J. Fang, S. Sampson, R. F. L. Self, P. A. Jumars, C. Quetel, and O. F. X. Donard. 1996. Bioavailability of sedimentary contaminants subject to deposit-feeder digestion. Environ. Sci. Technol. 30:2641-2645
McBride, M. 1998. Soluble trace metals in alkaline stabilized sludge products. J. Environ. Qual. 27:578-584.
McBride, M. B., A. R. Fraser, and W. J. McHardy. 1984. Cu2+ interaction with microcystalline gibbsite: evidence for oriented chemisorbed copper ions. Clays and Clay Minerals 32:12-18.
McBride, M. B. 1982. Cu2+-Adsorption characteristics of aluminum hydroxide and oxyhydroxides. Clays and Clay Minerals. 30:21-28.
McGrath, S. P. 1995. Chromium and nickel. Pp. 152–178 In: Heavy metals in soils. B. J. Alloway (ed.). London, England: Blackie Academic and Professional.
McGrath, S. P., B. Knight, K. Killham, S. Preston, and G. I. Paton. 1999. Assessment of the toxicity of metals in soils amended with sewage sludge using a chemical speciation technique and a lux-based biosensor. Environ. Toxicol. Chem. 18(4):659-663.
McKim, J. M., and J. W. Nichols. 1994. Use of physiologically based toxicokinetic models in a mechanistic approach to aquatic toxicology. Pp. 469-521 In: Aquatic toxicology: molecular, biochemical, and cellular perspectives. D. C. Malins and G. K. Ostrander (eds.). Boca Raton, FL: Lewis Publishers.
McLaughlin, M., B. A. Zarcinas, D. P. Stevens, and N. Cook. 2000. Soil testing for heavy metals. Commun. Soil Sci. Plant Anal. 31:1661-1700.
Meador, J. P., J. E. Stein, W. L. Reichert, and U. Varanasi. 1995. Bioaccumulation of polycyclic aromatic hydrocarbons by marine organisms. Rev. Environ. Contam. Toxicol. 143:79-165.
Medlin, A. 1997. An in vitro method for estimating the relative bioavailability of lead in humans. MS Thesis. Department of Geological Sciences, University of Colorado, Boulder, CO.
Meighen, E. A. 1991. Molecular biology of bacterial bioluminescence. Microbiol. Rev. 55:123-142.
Menzie, C. A., D. E. Burmaster, J. S. Freshman, and C. A. Callahan. 1992. Assessment of methods for estimating ecological risk in the terrestrial component: a case study at the Baird & McGurie Superfund site in Holbrook, Massachusetts. Environ. Toxicol. Chem. 11:245-260.
Menzie, C. A., A. M. Burke, D. Grasso, M. Harnois, B. Magee, D. McDonald, C. Montgomery, A. Nichols, J. Pignatello, B. Price, R. Price, J. Rose, J. Shatkin, B. Smets, J. Smith, and S. Svirsky. 2000. An approach for incorporating information on chemical availability in soils into risk assessment and risk-based decision making. Human and Ecological Risk Assessment 6(3):479-510.
Meyers, R. A. 1998. Encyclopedia of environmental analysis and remediation. Wiley encyclopedia series in environmental science. New York: John Wiley. 5400 pp.
Michelsen, T., T. C. Shaw, and S. Stirling. 1998. Testing, reporting, and evaluation of tributyltin data in PSDDA and SMS Programs. Appendix 8A: dredged material evaluation framework: Lower Columbia River Management Area, November 1998. Portland, OR:US Corps of Engineers, Seattle District.
Miller, D. D., and B. R. Schricker. 1982. In vitro estimation of food iron bioavailability. In: Nutritional bioavailability of iron. ACS Symposium Series No. 203. C. Kies (ed.). Washington, DC: American Chemical Society.
Miner, G. S., R. Gutierrez, and L. D. King. 1997. Soil factors affecting plant concentrations of cadmium, copper, and zinc on sludge-amended soils. J. Environ. Qual. 26:989-994.
Morimoto, R. I., D. A. Jurivich, P. E. Kroeger, S. K. Mathur, S. P. Murphy, A. K. Nakai, K. Abravaya, and L. T. Sistonen. 1995a. Regulation of heat shock gene transcription by a family of heat shock factors. Pp. 417-55 In: The biology of heat shock proteins and molecular chaperones. R. I. Morimoto, A. Tissieres, and C. Georgopoulos (eds.). New York: Cold Spring Harbor Laboratory Press.
Morimoto, R. I., A. Tisseriers, and C. Georgopoulos. 1995b. Progress and perspectives on the biology of heat shock proteins and molecular chaperones. Pp. 1-30 In: The biology of heat shock proteins and molecular chaperones. R. I. Morimoto, A. Tissieres, and C. Georgopoulos (eds.). New York: Cold Spring Harbor Laboratory Press.
Morrison, D. E., B. K. Robertson, and M. Alexander. 2000. Bioavailability to earthworms of aged DDT, DDE, DDD, and dieldrin in soil. Environ. Sci. Technol. 34:709-713.
Muhlenkamp, C. R., and S. S. Gill. 1998. A glucose-regulated protein, GRP58, is down-regulated in C57B6 mouse liver after diethylhexyl phthalate exposure. Toxicol. Appl. Pharmacol. 148:101-108.
Murray, P., Y. Ge, and W. H. Hendershot. 2000. Evaluating three trace metal contaminated sites: a field and laboratory investigation. Environ. Pollut. 107(1):127-135.
Muyzer, G., E. C. de Waal, and A. G. Uitterlinden. 1993. Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl. Environ. Microbiol. 59(3):695-700.
Nabel, A., B. Streit, and S. Winter. 1995. Bioaccumulation of selected organo-chlorines in bats and tits: influence of chemistry and biology. Environmental Science and Pollution Research 4:194-199.
Nadeau, D., S. Corneau, I. Plante, G. Morrow, and R. M. Tanguay. 2001. Evaluation for Hsp70 as a biomarker of effect of pollutants on the earthworm Lumbricus terrestris. Cell Stress & Chaper-ones 6:153-163.
Nalewajko, C. E. A. 1989. Effects of contaminated sediments and standard elutriates on primary production in Lake Ontario. Water, Air, and Soil Pollution. 45:275-86.
Neilson, J. W., S. A. Pierce, and R. M. Maier. 1999. Factors influencing expression of luxcdabe and nah genes in Pseudomonas putida RB1353(nah7, putk9) in dynamic systems. Appl. Environ. Microbiol. 65:3473-3482.
Nesbitt, H. W., G. M. Bancroft, A. R. Pratt, and M. J. Scaini. 1998b. Sulfur and iron surface states on fractured pyrite surfaces. Am. Mineral. 83:1067-1076.
Nesbitt, H. W., G. W. Canning, and G. M. Bancroft. 1998a. XPS study of reductive dissolution of 7Å-birnessite by H3AsO3 with constraints on reaction mechanism. Geochim. Cosmochim. Acta 62:2097-2110.
Newman, L. A., S. E. Strand, N. Choe, J. Duffy, G. Ekuan, M. Ruszaj, B. Shurtleff, J. Wilmoth, P. Heilman, and M. Gordon. 1997. Uptake and biotransformation of trichloroethylene by hybrid poplars. Environ. Sci. Technol. 31:1062-1067.
Nichols J. W., C. P. Larsen, M. E. McDonald, G. J. Niemi, and G. T. Ankley. 1995. Bioenergetics-based model for accumulation of PCBs by nesting tree swallows, Tachycineta bicolor. Environ. Sci. Technol. 29:604-612.
Nirel, P. V. M., and F. M. M. Morel. 1990. Pitfalls of sequential extraction. Water Research 24:1055-1056.
Njoroge, B. N. K., W. P. Ball, and R. S. Cherry. 1998. Sorption of 1,2,4-trichlorobenzene and tetrachloroethene within an authogenic soil profile: changes in Koc with soil depth. Journal of Contaminant Hydrology 29(4):347-377.
O’Conner, G. A. 1988. Use and misuse of the DTPA soil test. J. Environ. Qual. 17:715-718.
O’Day, P. A., S. A. Carroll, and G. A. Waychunas. 1998. Rock-water interactions controlling zinc, cadmium, and lead concentrations in surface waters and sediments, U.S. tri-state mining district. 1. Molecular identification using x-ray absorption spectroscopy. Environ. Sci. Technol. 32:943-955.
O’Day, P. A., S. A. Carroll, S. Randall, R. E. Martinelli, S. L. Anderson, J. Jelinski, and J. P. Knezovich. 2000. Metal speciation and bioavailability in contaminated estuary sediments, Alameda Naval Air Station, California. Environ. Sci. Technol. 34:3665-3673.
Odum, E. P., 1984. The mesocosm. BioScience 34:558-562.
Oomen, A. G. 2000b. Determinants of oral bioavailability of soil-borne contaminants. Bilthoven, the Netherlands: Dutch National Institute of Public Health and the Environment.
Oomen, A. G., A. Sips, J. P. Groten, D. Sijm, and J. Tolls. 2000a. Mobilization of PCBs and lindane from soil during in vitro digestion and their distribution among bile salt micelles and proteins of human digestive fluid and the soil. Environ. Sci. Technol. 34(2):297-303.
Oomen, A. G., A. Hack, M. Minekus, E. Zeijdner, C. Cornelis, G. Schoeters, W. Verstraete, T. Van de Wiele, J. Wragg, C. J. M. Rompelberg, A. J. A. M. Sips, and J. H. Van Wijnen. 2002. Comparison of five in vitro digestion models to study the bioaccessibility of soil contaminants. Environ. Sci. Technol. 36(15):3326-3334.
Opdyke, D. R., and R. C. Loehr. 1999. Determination of chemical release rates from soils: experimental design. Environ. Sci. Technol. 33:1193-1199.
Organization for Economic Cooperation and Development (OECD). 1984). Terrestrial plants, growth test. OECD Guideline 208. Paris, France: OECD.
OECD. 2000a. Guidelines for the testing of chemicals—soil microorganisms: carbon transformation test. OECD Guideline 217. Paris, France: OECD.
OECD. 2000b. Guidelines for the testing of chemicals—soil microorganisms: nitrogen transformation test. OECD Guideline 216. Paris, France: OECD.
Ostergren, J. D., G. E. Brown, Jr., G. A. Parks, and T. N. Tingle. 1999. Quantitative speciation of lead in selected mine tailings from Leadville, CO. Environ. Sci. Technol. 33:1627-1636.
Otto, W. H., W. R. Carper, and C. K. Larive. 2001. Measurement of cadmium(II) and calcium(II) complexation by fulvic acids using 113Cd NMR. Environ. Sci. Technol. 35:1463-1468.
Page, A. L. 1965. Methods of soil analysis. Agronomy no. 9. Madison, WI: American Society of Agronomy. 1142 pp.
Page, A. L., T. J. Logan, and J. A. Ryan (eds.). 1987. Land application of sludge-food chain implications. Chelsea, MI: Lewis Publishers Inc.
Palermon, M. R., and E. L. Thackson. 1988. Test for dredged material effluent quality. J. Environ. Engineering ASCE 114(6):1295-1309.
Pankakoski, E., I. Koivisto, H. Hyvarinen, J. Terhivuo, and K. M. Tahka. 1994. Experimental accumulation of lead from soil through earthworms to common shrews. Chemosphere 29:1639-1649.
Parker, D. R., R. L. Chaney and W. A. Norvell. 1995. Chemical equilibrium models: applications to plant nutrition research. Pp. 163-200 In: Chemical equilibrium and reaction models. R. H. Loeppert et al. (eds.). Madison, WI: Soil Sci. Soc. Amer. Spec. Pub. No. 42.
Penalver, A., E. Pocurull, F. Borrull, and R. M. Marce. 1999. Trends in solid-phase microextraction for determining organic pollutants in environmental samples. Trac-trends Anal. Chem. 18:557-568.
Pennie, W. D., N. J. Woodyatt, T. C. Aldridge, and G. Orphanides. 2001. Application of genomics to the definition of the molecular basis for toxicity. Toxicol. Lett. 120:353-358.
Pennie, W. D. 2000. Use of cDNA microarrays to probe and understand the toxicological consequences of altered gene expression. Toxicol. Lett. 112-113:473-477.
Peredney, C. L., and P. L. Williams. 2000. Utility of Caenorhabditis elegans for assessing heavy metal contamination in artificial soil. Arch. Environ. Contamin. Toxicol. 39:113-118.
Pereira, W. E., C. E. Rostad, C. T. Chiou, T. I. Brinton, L. B. Barber, D. K. Demcheck, and C. R. Demas. 1988. Contamination of estuarine water, biota, and sediment by halogenated organic compounds: a field study. Environ. Sci. Technol. 22:772-778.
Perez, K. T., G. M. Morrison, N. F. Lackie, C. A. Oviatt, S. W. Nixon, B. Buck-Ley, and J. F. Heltshe. 1977. The importance of physical and biotic scaling to the experimental simulation of a coastal marine ecosystem. Helgolander Wiss Meeresauters 30:144-162.
Phillips, D., and P. S. Rainbow. 1993. The biomonitoring of organochlorines and hydrocarbons. Pp. 133-178 In: Biomonitoring of trace aquatic contaminants. D. Phillips (ed.). Essex, England: Elsevier Science Publishers Ltd.
Pickering, W. F. 1981. Selective chemical extraction of soil components and bound metal species. CRC Critical Reviews Anal. Chem. 12:233-266.
Pierzynski, G. M., and A. P. Schwab. 1993. Bioavailability of zinc, cadmium, and lead in a metal-contaminated alluvial soil. J. Environ. Qual. 22:247-254.
Pinto, M., S. Robine-Leon, M.-D. Appay, M. Kedinger, N. Triadou, E. Dussaulx, B. LaCroix, P. Simon-Assmann, K. Haffen, J. Fogh, and A. Zweibaum. 1983. Enterocyte-like differentiation and polarization of the human colon carcinoma cell line Caco-2 in culture. Biol. Cell 47:323-330.
Pichtel, J., and C.A. Salt. 1998. Vegetative growth and trace metal accumulation on metalliferous wastes. J. Environ. Qual. 27:618-624.
Polder, M. D., E. M. Hulzebos, and D. T. Jager. 1995. Hazard assessment. Validation of models on uptake of organic chemicals by plant roots. Environ. Toxicol. Chem. 14(9):1615-1623.
Ponec, M., E. Boelsma, A. Weerheim, A. Mulder, J. Bouwstra, and M. Mommaas. 2000. Lipid and ultrastructural characteristics of reconstructed skin models . Int. J. Pharm. 203:211-225.
Quevauviller, P. H., G. Rauret, and B. Griepink. 1993. Single and sequential extraction in sediments and soils. Int. J. Environ. Anal. Chem. 51:231-235.
Quevauviller, P. 1995. Trends in trace element determinations in environmental and food matrices: conclusions of the workshop. Mikrechim. Acta 119:329-338.
Rabbow, E., P. Rettberg, C. Baumstark-Khan, and G. Horneck. 2002. SOS-LUX- and LAC-FLUORO-TEST for the quantification of genotoxic and/or cytotoxic effects of heavy metal salts. Analytica Chimica Acta 456:31-39.
Rasmussen, L. D., S. J. Sorensen, R. R. Turner, and T. Barkay. 2000. Application of a mer-lux biosensor for estimating bioavailable mercury in soil. Soil Biology and Biochemistry 32:639-646.
Rattray, E. A. S., J. I. Prosser, K. Killham, and L. A. Glover. 1990. Luminescence based non-extractive techniques for in situ detection of Escherichia coli in soil. Appl. Environ. Microbiol. 56:3368-3374.
Rayment, G. E., and F. R. Higginson. 1992. Australian laboratory handbook of soil and water chemical methods. Melbourne, Australia: Inkata Press.
Reddy, M. B., E. J. Browder, and G. W. Bates. 1988. Cannulated swine and in vitro approaches to iron bioavailability. Pp. 173–185 In: Essential and toxic trace elements in human health and disease. A. Prasad (ed.). New York: Alan R. Liss, Inc.
Reed, S. T., and D. C. Martens. 1996. Copper and zinc. Pp. 703-722 In: Methods of soil analysis. Part 3—chemical methods. D. Sparks et al (eds.). Madison, WI: American Society of Agronomy and the Soil Science Society of America.
Reid, B. J., K. T. Semple, C. J. Macleod, H. J. Weitz, and G. I. Paton. 1998. Feasibility of using prokaryote biosensors to assess acute toxicity of polycyclic aromatic hydrocarbons. FEMS Microbiol. Letters 169:227-233.
Reid, B. J., K. C. Jones, and K. T. Semple. 2000. Bioavailability of persistent organic pollutants in soils and sediments—a perspective on mechanisms, consequences and assessment. Environ. Pollut. 108:103-112.
Reinfelder, J. R., and N. S. Fisher. 1991. The assimilation of elements ingested by marine copepods. Science 251:794-796.
Reinfelder, J. R., N. S. Fisher, S. N. Luoma, J. W. Nichols, and W.-X. Wang. 1998. Trace element trophic transfer in aquatic organisms: a critique of the kinetic model approach. Sci. Total Environ. 219:117-135.
Ressler, T., J. Wong, J. Roos, and I. L. Smith. 2000. Quantitative speciation of Mn-bearing particulates emitted from autos burning (methycyclopentadienyl)manganese tricarbonyl-added gasolines using XANES spectroscopy. Environ. Sci. Technol. 34:950-958.
Riggs, D. S. 1963. The mathematical approach to physiological problems. Baltimore, MD: Williams and Wilkins Co. 444 pp.
Ripp, S., D. E. Nivens, Y. Ahn, C. Werner, J. Jarrell, IV, J. P. Easter, C. D. Cox, R. S. Burlage, and G. S. Sayler. 2000. Controlled field release of a bioluminescent genetically engineered microorganism for bioremediation process monitoring and control. Environ. Sci. Technol. 34:846-853.
Ritossa, F. 1962. A new puffing pattern induced by temperature shock and DNP in Drosophila. Experientia 18:571-573.
Robidoux, P. Y., C. Svendsen, J. Caumartin, J. Hawari, G. Ampleman, S. Thiboutot, J. M. Weeks, and G. I. Sunahara. 2000. Chronic toxicity of energetic compounds in soil determined using the earthworm (Eisenia andrei) reproduction test. Environ. Toxicol. Chem. 19:1764-1773.
Rodgers, J. H., Jr. 1983. Use of microcosms to study transport, transformation and fate of organics in aquatic systems. Environ. Toxicol. Chem. 2:155-167.
Rodi, D. J., R. W. Janes, H. J. Sanganee, R. A. Holton, B. A. Wallace, and L. Makowski. 1999. Screening of a library of phage-displayed peptides identifies human Bcl-2 as a taxol binding protein. J. Molecular Biol. 285:97-203.
Rodriguez, R. R., N. T. Basta, S. W. Casteel, and L. W. Pace. 1999. An in vitro gastrointestinal method to estimate bioavailable arsenic in contaminated soils and solid media. Environ. Sci. Technol. 33(4):642–649.
Roman, B. L., and R. E. Peterson. 1998. In utero and lactational exposure of the male rat to 2,3,7,8-tetrachlorodibenzo-p-dioxin impairs prostate development. Toxicol. Appl. Pharmacol. 150:240-253.
Romijn, C. A. F. M., R. Grau, J. A. Guth, E. G. Harrison, C. M. Jackson, B. Lefebvre, W. W. Smith, and J. R. Street. 1995. The use of Japanese and bobwhite quail as indicator species in avian toxicity test. Chemosphere 30:1033-1040.
Rotard, W., W. Christman, W. Knoth, and W. Mailahn. 1995. Defining the fraction of PCDD/PCDF that is available for absorption from a red cinder substrate. Z. Umweltchem. Okotox. 7(1):3-9.
Ruby, M. V., A. Davis, R. Schoof, S. Eberle, and C. M. Sellstone. 1996. Estimation of lead and arsenic bioavailability using a physiologically based extraction test. Environ. Sci. Technol. 30(2):422–430.
Ruby, M. V., R. Schoof, W. Brattin, M. Goldade, G. Post, M. Harnois, D. E. Mosby, S. W. Casteel, W. Berti, M. Carpenter, D. Edwards, D. Cragin, and W. Chappell. 1999. Advances in evaluating the oral bioavailability of inorganics in soil for use in human health risk assessment. Environ. Sci. Technol. 33(21):3697-3705.
Ruby, M. V., K. A. Fehling, D. J. Paustenbach, B. Landenberger, and M. Holsapple. 2002. Oral bioaccessibility of dioxins/furans at low concentrations (50–350 ppt TEQ) in soil. Environ. Sci. Technol. 36(22):4905-4911.
Rutgers, M., I. M. vantVerlaat, B. Wind, L. Posthuma, and A. M. Breure. 1998. Rapid method for assessing pollution-induced community tolerance in contaminated soil. Environ. Toxicol. Chem. 17(11):2210-2213.
Ruzic, I. 1972. Two-compartment model of radionuclide accumulation into marine organisms. I. Accumulation from a medium of constant activity. Mar. Biol. 15:105-112.
Saint-Denis, M., J. F. Narbonne, C. Arnaud, E. Thybaud, and D. Ribera. 1999. Biochemical responses of the earthworm Eisenia fetida andrei exposed to contaminated artificial soil: effects of benzo(a)pyrene. Soil Biology and Biochemistry 31:1837-1846.
Sakurovs, R. 1998. Interactions between a bituminous coal and aromatic hydrocarbons at elevated temperatures. Energy & Fuels 12:631-636.
Salazar, M. H., P. B. Duncan, S. M. Salazar, K. A. and Rose. 1995. In situ bioassay using transplanted mussels. 2. Assessing contaminated sediment at a Superfund site in Puget Sound. Pp. 242-262 In: Environmental toxicology and risk assessment. Philadelphia, PA: ASTM.
Sample, B. E., J. J. Beauchamp, R. A. Efroymson, G. W. Suter, II, and T. L. Ashwood. 1998. Development and validation of bioaccumulation models for earthworms. ES/ER/TM-220. Prepared for the U.S. Department of Energy under Contract DE-AC05-84OR21400. Code EW 20. Oak Ridge, TN: Oak Ridge National Laboratory.
Sanders, B. M. 1990. Stress-proteins: potential as multitiered biomarkers. Pp. 165-91 In: Environmental biomarkers. L. Shugart and J. McCarthy (eds.). Boca Raton, FL: Lewis Publishers.
Sanders, B. M., L. S. Martin, W. G. Nelson, D. K. Phelps, and W. Welch. 1991. Relationships between accumulation of a 60 kDa stress protein and scope for growth in Mytilus edulis exposed to a range of copper concentrations. Marine Environmental Research 31:81-97.
Sarret, G., A. Manceau, L. Spadini, J.-C. Roux, J.-L. Hazemann, Y. Soldo, L. Eybert-Berard, and J.-J. Menthonnex. 1998. Structural determination of Zn and Pb binding sites in Penicillium chrysogenum cell walls by EXAFS spectroscopy. Environ. Sci. Technol. 32:1648-1655.
Sauerbeck, D. R. 1991. Plant, element and soil properties governing uptake and availability of heavy metals derived from sewage sludge. Water, Air, and Soil Pollution 57-58:227-237.
Sauerbeck, D. R., and B. Styperek. 1984. Evaluation of chemical methods for assessing the Cd and Zn availability from different soils. Pp. 49-67 In: Chemical methods for assessing bioavailable metals in sludges and soils. R. Leschber, R. D. Davis, and P. L. Hermite (eds.). Essex, England: Elsevier Applied Science Publishers.
Sauvé, S., M. McBride, and W. Hendershot. 1997. Speciation of lead in contaminated soils. Environ. Pollut. 98:149-155.
Sauvé, S., M. McBride, and W. Hendershot. 1998. Soil solution speciation of lead(II): effects of organic matter and pH. Soil Sci. Soc. Am. J. 62:618-621.
Sauvé, S., N. Cook, W. Hendershot, and M. McBride. 1996. Linking plant tissue concentrations and soil copper pools in urban contaminated soils. Environ. Pollut. 94:153-157.
Sauvé, S., W. Hendershot, and H. Allen. 2000. Solid-solution partitioning of metals in contaminated soils: dependence on pH and total metal burden. Environ. Sci. Technol. 34:1125-1131.
Saxe, J., C. A. Impellitteri, W. J. Peijnenburg, and H. Allen. 2001. Novel model describing trace metal concentrations in the earthworm, Eisenia andrei. Environ. Sci. Technol. 35(22):4522-4529.
Scheidegger, A. M., G. M. Lamble, and D. L. Sparks. 1997. Spectroscopic evidence for the formation of mixed-cation hydroxide phases upon metal sorption on clays and aluminum oxides. J. Colloid Interface Sci. 186:118-128.
Schlekat, C. E., and S. N. Luoma. 2000. You are what you eat: incorporating dietary metals uptake into environmental quality guidelines for aquatic ecosystems. SETAC Globe 1(2):38-39.
Schlekat, C. E., B-G. Lee, and S. N. Luoma. 2002. Dietary metals exposure and toxicity to aquatic organisms: implications for ecological risk assessment. Chapter 7 In: Coastal and estuarine risk assessment. M. C. Newman, M. H. Roberts, Jr., and R. C. Hale (eds.). Boca Raton, FL: Lewis Publishers.
Schuler, F., P. Schmid, and C. Schlatter. 1997. The transfer of polychlorinated dibenzo-p-dioxins and dibenzofurans from soil into eggs of foraging chicken. Chemosphere 34:711-718.
Selifonova, O., R. Burlage, and T. Barkay. 1993. Bioluminescent sensors for detection of bioavailable Hg(II) in the environment. Appl. Environ. Microbiol. 59:3083-3090.
Selker, J. S., C. K. Keller, and J. T. McCord. 1999. Vadose zone processes. Boca Raton, FL: Lewis Publishers. 339 pp.
Selmin, O., G. W. Lucier, G. C. Clark, A. M. Tritscher, J. P. Vanden Heuvel, J. A. Gastel, N. J. Walker, T. R. Sutter, and D. A. Bell. 1996. Isolation and characterization of a novel gene induced by 2,3,7,8-tetrachlorodibenzo-p-dioxin in rat liver. Carcinogenesis 17:2609-2615.
Sharps, J. A., G. E. Brown Jr., and J. F. Stebbins. 1993. Kinetics and mechanisms of ligand exchange of Au(III), Zn(II), and Cd(II) chlorides in aqueous solution: an NMR study from 28-98ºC. Geochim. Cosmochim. Acta 57:721-732.
Shaw, L. J., Y. Beaton, L. A. Glover, K. Killham, and A. A. Meharg. 2000. Interactions between soil, toxicant, and a lux-marked bacterium during solid phase-contact toxicity testing. Environ. Toxicol. Chem. 19(5):1247-1252.
Shugart, L., J. Bickman, G. Jackin, G. McMohan, W. Ridley, J. Stein, and S. Steinert. 1992. DNA alterations. Pp. 125-153 In: Biomarkers (biochemical, physiological, and histological markers of anthropogenic stress). R. J. Huggett, R. A. Kimerle, P.M. Mehrle, Jr., and H. L. Bergman (eds.). Boca Raton, FL: SETAC Special Publications/Lewis Publishers.
Shuman, L. M. 1996. Differential pulse voltammetry. Pp. 247-268 In: Methods of soil analysis. Part 3—chemical methods. D. Sparks et al. (eds.). Madison, WI: American Society of Agronomy and the Soil Science Society of America.
Siciliano, S. D., N. Fortin, A. Mihoc, G. Wisse, S. Labelle, D. Beaumier, D. Ouellette, R. Roy, L. G. Whyte, M. K. Banks, P. Schwab, K. Lee, and C. W. Greer. 2001. Selection of specific endophytic bacterial genotypes by plants in response to soil contamination. Appl. Environ. Microbiol. 67:2469-75.
Sijm, D., R. Kraaij, and A. Belford. 2000. Bioavailability in soil or sediment: exposure of different organisms and approaches to study it. Environ. Pollut. 108:113-119.
Sims, J. T, and J. S. Kline. 1991. Chemical fractionation and plant uptake of heavy metals in soils amended with co-composted sewage sludge. J. Environ. Qual. 20:387-395.
Skogley, E. O., and A. Doberman. 1996. Synthetic ion-exchange resins: soil and environmental studies. J. Environ. Qual. 25:13-24.
Sloan, J. J., R. H. Dowdy, M. S. Dolan, and D. R. Linden. 1997. Long-term effects of biosolids applications on heavy metal bioavailability in agricultural soils. J. Environ. Qual. 26:966-974.
Smolders, E., and M. J. McLaughlin. 1996. Effect of Cl on Cd uptake by Swiss chard in nutrient solution. Plant and Soil 179:57-64.
Solomon, K. R., J. P. Giesy, R. J. Kendall, L. B. Best, J. R. Coats, K. R. Dixon, M. J. Hooper, E. E. Kenaga, and M. J. McMurtry. 2001. Chlorpyrifos: ecotoxicological risk assessment for birds and mammals in corn agroecosystems. Human and Ecological Risk Assessment 7(3):497-632.
Soma, M., A. Tanaka, H. Seyama, and K. Satake. 1994. Characterization of arsenic in lake sediments by X-ray photoelectron spectroscopy. Geochim. Cosmochim. Acta 58: 2743-2745.
Sparks, D. L. 1996. Methods of soil analysis. Part 3—chemical methods. Soil Science Society of America Book Series #5. Madison, WI: American Society of Agronomy and the Soil Science Society of America. 1390 pp.
Sposito, G., L. J. Lund, and A. C. Chang. 1982. Trace metal chemistry in arid-zone field soils amended with sewage sludge. I. Fractionation of Ni, Cu, Zn, Cd, and Pb in solid phases. Soil Sci. Soc. Am. J. 46:260-264.
Stafford, E. A., and C.A. Edwards. 1985. Comparison of heavy metal uptake by Eisenia foetida with that of other common earthworms. Final technical report. USARDSG-UK, Rothamsted Experimental Station, Harpenden, Herts AL5 2JQ, U.K. Contract #DAJA45-84-C-0027.
Stegeman, J. J., M. Bronwer, R. T. DiGiulio, L. Forlin, B. A. Fowler, B. M. Sanders, P. A. Van Veld. 1992. Molecular responses to environmental contamination: enzyme and protein as indicators of chemical exposure and effect. Pp. 235-335 In: Biomarkers (biochemical, physiological, and histological markers of anthropogenic stress). R. J. Huggett, R. A. Kimerle, P. M. Mehrle, Jr., and H. L. Bergman (eds.). Boca Raton, FL: SETAC Special Publications, Lewis Publishers.
Stenstrom, J., B. Stenberg, and M. Johanson. 1998. Kinetics of substrate-induced respiration (SIR): theory. Ambio 27(1):35-39.
Stiner, L., and L. J. Halverson. 2002. Development and characterization of a green fluorescent protein-based bacterial biosensor for bioavailable toluene and related compounds. Appl. Environ. Microbiol. 68:1962-71.
Stringham, E. G., and E. P. M. Candido. 1994. Transgenic hsp16-lacZ strains of the soil nematode Caenorhabditis elegans as biological monitors of environmental stress. Environ. Toxicol. Chem. 13:1211-1220.
Suedel, B. C., J. H. Rodgers, Jr., and P. A. Clifford. 1993. Bioavailability of fluoranthene in freshwater sediment toxicity tests. Environ. Toxicol. Chem. 12:155-165.
Swackhamer, D. L., and R. A. Hites. 1988. Occurrence and bioaccumulation of organochlorine compounds in fishes from Siskiwit Lake, Isle Royale, Lake Superior. Environ. Sci. Technol. 22:543-548.
Swartz, R. C., D. W. Schults, T. H. Dewitt, G. R. Ditsworty, and J. O. Lamberson. 1990. Toxicity of fluoranthene in sediment to marine amphipods: a test of the equilibrium partitioning approach to sediment quality criteria. Environ. Toxicol. Chem. 9:1071-1080.
Swift, R. S. 1996. Organic matter characterization. Pp. 1011-1070 In: Methods of Soil Analysis. Part 3—chemical methods. D. L. Sparks (ed.). Madison, WI: American Society of Agronomy and the Soil Science Society of America.
Symeonides, C., and S. G. McRae. 1977. The assessment of plant-available cadmium in soils. J. Environ. Qual. 6:72-77.
Szulczewski, M. D., P. A. Helmke, and W. F. Bleam. 1997. Comparison of XANES analyses and extractions to determine chromium speciation in contaminated soils. Environ. Sci. Technol. 31:2954-2965.
Talley, J. W., U. Ghosh, S. Tucker, J. Furey, and R. G. Luthy. 2002. Particle-scale understanding of the bioavailability of PAHs in sediment. Environ. Sci. Technol. 36:477-483.
Talmage, S. S., and B. T. Walton. 1993. Food chain transfer and potential renal toxicity of mercury to small mammals at a contaminated terrestrial field site. Ecotoxicology 2:243-256.
Tang, J., B. K. Robertson, and M. Alexander. 1999. Chemical extraction methods to estimate bioavailability of DDT, DDE, and DDD in soil. Environ. Sci. Technol. 33(23):4346–4351.
Tang, J., and M. Alexander. 1999. Mild extractability and bioavailability of polycyclic aromatic hydrocarbons in soil. Environ. Toxicol. Chem. 18(12):2711–2714.
Tank, S. L., L. W. Brewer, M. J. Hooper, G. P. I. I. Cobb, and R. J. Kendall. 1993. Survival and pesticide exposure of northern bobwhites (Colinus virginianus) and eastern cottontails (Sylvilagus floridanus) on agricultural fields treated with counter 15G. Environ. Toxicol. Chem. 12:2113-2120.
Taurianen S., M. Karp, W. Chang, and M. Virta. 1998. Luminescent bacterial sensor for cadmium and lead. Biosens. Bioelec. 13:931-938.
Taylor, R. W., I. O. Ibeabuchi, K. R. Sistani, and J. W. Shuford. 1992. Accumulation of some metals by legumes and their extractability from acid mine spoils . J. Environ. Qual. 21:176-180.
Tessier, A., P. G. C. Campbell and M. Bisson. 1979. Sequential extraction procedure for the speciation of trace metals. Anal. Chem. 51:844-851.
Tessier, A., P. G. C. Campbell, J. C. Auclair, and M. Bisson. 1984. Relationships between the partitioning of trace metals in sediments and their accumulation in the tissues of the freshwater mollusc Elliptio complanata in a mining area. Can. J. Fish. Aquat. Sci. 41:1463-1471.
Thomann, R. V., J. D. Mahony, and R. Mueller. 1995. Steady state model of biota sediment accumulation factor for metals in two marine bivalves. Environ. Toxicol. Chem. 14:1989-1998.
Thorn, K. A., P. J. Pettigrew, W. S. Goldenberg, and E. J. Weber. 1996. Covalent binding of aniline to humic substances. 2. 15N-NMR studies of nucleophilic addition reactions. Environ. Sci. Technol. 30:2764-2973.
Timbrell, J. A. 1998. Biomarkers in toxicology. Toxicology 129:1-12.
Tokunaga, T. K., I. J. Pickering, and G. E. Brown, Jr. 1996. Selenium transformations in ponded sediments. Soil Sci. Soc. Am. J. 60:781-790.
Tokunaga, T. K., S. R. Sutton, S. Bajt, P. Nuessle, and G. Shea-McCarthy. 1998. Selenium diffusion and reduction at the water-sediment boundary: micro-XANES spectroscopy of reactive transport. Environ. Sci. Technol. 32:1092-1098.
Tolle, D. A., M. F. Arthur, J. Chesson, and P. Van Voris. 1985. Comparison of pots versus microcosms for predicting agroecosystem effects due to waste amendment. Environ. Toxicol. Chem. 4:501-509.
Tsien, R. Y. 1998. The green fluorescent protein. Ann. Rev. Biochem. 67:509-544.
Tully, D. B., B. J. Collins, J. D. Overstreet, C. S. Smith, G. E. Dinse, M. M. Mumtaz, and R. E. Chapin. 2000. Effects of arsenic, cadmium, chromium, and lead on gene expression regulated by a battery of 13 different promoters in recombinant HepG2 cells. Toxicol. Appl. Pharmacol. 168:79-90.
Vairavamurthy, A., W., Zhou, T. Eglinton, and B. Manowitz. 1994. Sulfonates: a novel class of organic sulfur compounds in marine sediments. Geochim. Cosmochim. Acta 58:4681-4687.
van der Lelie, D., L. Regniers, B. Borremans, A. Provoost, and L. Verschaeve. 1997. The VITOTOX test, an SOS bioluminescence Salmonella typhimurium test to measure genotoxicity kinetics. Mutation Research 389:279-290.
van der Wal, L., T. Jager, R. Fleuren, C. A. M. van Gestel, and J. L. M. Hermens. 2002. Predicting uptake and bioavailability of organic contaminants in terrestrial organisms using SPME in different soils. Abstr. Annu. Meeting Soc. Environ. Toxicol. Chem. 38-10:146. Pensacola, FL: SETAC Press.
van Gestel, C. A. M., and W. Ma. 1988. Toxicity and bioaccumulation of chlorophenols in earthworms, in relation to bioavailability in soil. Ecotox. Environ. Safety 15:289-297.
van Gestel, C. A. M., and W. A. van Dis. 1988. The influence of soil characteristics on the toxicity of 4 chemicals to the earthworm Eisena fetida andrei (Oligachaeta). Biology and Fertility of Soils 6:262-265.
van Gestel, C. A. M., W. A. van Dis, E. M. van Breemen, and P. M. Sparenburg. 1989. Development of a standardized reproduction toxicity test with the earthworm Eisenia fetida andrei using copper, pentachlorophenol, and 2,4-dichloroaniline . Ecotoxicol. Environ. Saf. 18:305-312.
van Gestel, C. A. M., J. van der Waarde Jaap, J. G. M. A. Derksen, E. E. van der Hoek, M. F. X. W. Veul, S. Bouwens, B. Rusch, R. Kronenburg, and G. N. M. Stokman. 2001. The use of acute and chronic bioassays to determine the ecological risk and bioremediation efficiency of oil-polluted soils. Environ. Toxicol. Chem. 20:1438-1449.
Velculescu, V. E., L. Zhang, B. Vogelstein, and K. W. Kinzler. 1995. Serial analysis of gene expression. Science 270:484-487.
von Witzingerode, F., U. B. Göbe, and E. Stackebrandt. 1997. Determination of microbial diversity in environmental samples: pitfalls of PCR based rRNA analysis. FEMS Microbiology Reviews 21:213-229.
VSBo. 1986. Verordnung über Schadstoffgehalt im Boden. Pp. 1-4 In: Swiss ordinance on pollutants in soils No. 814.12, Publ. Eidg. Dricksachen und Materialzentrale, Bern.
Wainman, T., R. E. Hazen, and P. J. Lioy. 1994. The extractability of Cr(VI) from contaminated soil in synthetic sweat. J. Exposure Anal. Environ. Epidemiol. 4(2):171-181.
Waldrop, M. P., T. C. Balser, and M. K. Firestone. 2000. Linking microbial community composition to function in a tropical soil. Soil Biology and Biochemistry 32(13):1837-1846.
Wang, W.-X., N. S. Fisher, and S. N. Luoma. 1996a. Kinetic determinations of trace element bioaccumulation in the mussel, Mytilus edulis. Marine Ecology Prog. Ser. 140:91-113.
Wang, X., P. K. W. Harris, R. G. Ulrich, and R. L. Voorman. 1996b. Identification of dioxin-responsive genes in Hep G2 cells using differential mRNA display RT-PCR. Biochem. Biophys. Res. Commun. 220:784-788.
Waring, J. F., R. Ciurlionis, R. A. Jolly, M. Heindel, and R. G. Ulrich. 2001a. Microarray analysis of hepatotoxins in vitro reveals a correlation between gene expression profiles and mechanisms of toxicity. Toxicol. Lett. 120:359-368.
Waring, J. F., R. A. Jolly, R. Ciurlionis, P. Y. Lum, J. T. Praestgaard, D. C. Morfitt, B. Buratto, C. Roberts, E. Schadt, and R. G. Ulrich. 2001b. Clustering of hepatotoxins based on mechanisms of toxicity using gene expression profiles. Toxicol. Appl. Pharmacol. 175:28-42.
Waring, J. F., and R. G. Ulrich. 2000. The impact of genomics-based technology on drug safety evaluation. Annu. Rev. Pharmacol. Toxicol. 40:335-352.
Waterfield, C. J. 2000. Biomarkers of effect and response. Pp. 1855-1873 In: General and applied toxicology, Volume 3, 2nd edition. B. Ballantyne, T. Marrs, and T. Syversen (eds.). London: MacMillan Reference.
Waterfield, C. J., and J. A. Timbrell. 2000. Biomarkers—an overview. Pp. 1842-1854. In: General and applied toxicology, Volume 3, 2nd edition. B. Ballantyne, T. Marrs, and T. Syversen (eds.). London: MacMillan Reference.
Waychunas, G. A., C. C. Fuller, B. A. Rea, and J. A. Davis. 1996. Wide angle X-ray scattering (WAXS) study of “two-line” ferrihydrite structure: effect of arsenate sorption and counterion variation and comparison with EXAFS results. Geochim. Cosmochim. Acta 60:1765-1781.
WDEQ-LQD. 1984. Guideline No. 1 topsoil and overburden. Cheyenne, WY: Wyoming Department of Environmental Quality, Land Quality Division.
Weissmahr, K. W., S. B. Haderlein, and R. P. Schwarzenbach. 1997. In situ spectroscopic investigations of adsorption mechanisms of nitroaromatic compounds at clay minerals. Environ. Sci. Technol. 31:240-247.
Werth, C. J., and M. Reinhard. 1997a. Effects of temperature on trichloroethylene desorption from silica gel and natural sediments. 1. Isotherms. Environ. Sci. Technol. 31:689-696.
Werth, C. J., and M. Reinhard. 1997b. Effects of temperature on trichloroethylene desorption from silica gel and natural sediments. 2. Kinetics. Environ. Sci. Technol. 31:697-703.
Wester, R. C., and H. I. Maibach. 1998. Percutaneous absorption of hazardous substances from soil and water. Pp. 29-39 In: Dermatotoxicology methods. Washington, DC: Taylor & Frances.
Westerman, R. L. 1990. Soil testing and plant analysis, 3rd edition. Madison, WI: Soil Science Society of America.
White, M. C., A. M. Decker, and R. L. Chaney. 1979. Differential cultivar tolerance in soybean to phytotoxic levels of soil zinc: range of cultivar response. Agron. J. 71:121-126.
White, J. C., M. Alexander, and J. J. Pignatello. 1999. Enhancing the bioavailability of organic compounds sequestered in soil and aquifer solids. Environ. Toxicol. Chem. 18(2):182-187.
White, J. C., J. W. Kelsey, P. B. Hatzinger, and M. Alexander. 1997. Factors affecting sequestration and bioavailability of phenanthrene in soils. Environ. Toxicol. Chem. 16:2040-2045.
Willardson, B. M., J. F. Wilkins, T. A. Rand, J. M. Schupp, K. K. Hill, P. Keim, and P. J. Jackson. 1998. Development and testing of a bacterial biosensor for toluene-based environmental contaminants. Appl. Environ. Microbiol. 64:1006-1012.
Williams, J. H., A. M. Farag, M. A. Stansbury, P. A. Young, H. L. Bergman, and N. S. Petersen. 1996. Accumulation of HSP70 in juvenile and adult rainbow trout gill exposed to metal-contaminated water and/or diet. Environ. Toxicol. Chem. 15:1324-1328.
Wittsiepe, J., P. Schrey, A. Hack, F. Selenka, and M. Wilhelm. 2001. Comparison of different digestive tract models for estimating bioaccessibility of polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/F) from red slag “Kieselrot.” Int. J. Hyg. Environ. Health 203:263–273.
Wu, S., and P. M. Gschwend. 1986. Numerical modeling of sorption kinetics of organic compounds to soils and sediment particles. Water Resources Research 24:1373-1383.
Xia., K., W. Bleam, and P. A. Helmke. 1997. Studies of Cu2+ and Pb2+ binding sites in soil humic substances using X-ray absorption spectroscopy. Geochim. Cosmochim. Acta 61:2211-2222.
Xing, B. S. 1997. The effect of the quality of soil organic matter on sorption of naphthalene. Chemosphere 35(3):633-642.
Xu, J., and I. Thornton. 1985. Arsenic in garden soils and vegetable crops in Cornwall, England: implications for human health. Environ. Geochem. Health 7:131-133.
Yeardley, R. B., J. M. Lazorchak, and L. C. Gast. 1996. The potential of an earthworm avoidance test for evaluation of hazardous waste sites. Environ. Toxicol. Chem. 15:1532-1537.
Yeom, I. T., M. M. Ghosh, C. D. Cox, and K. H. Ahn. 1996. Dissolution of polycyclic aromatic hydrocarbons from weathered contaminated soil. Water Sci. Technol. 34:335-342.
Young, T. M., and W. J. Weber, Jr. 1997. A distributed reactivity model for sorption by soils and sediments. 7. Enthalpy and polarity effects on desorption under supercritical fluid conditions. Environ. Sci. Technol. 31:1692-1696.
Zelles, L. 1999. Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: a review. Biology and Fertility of Soils 29(2):111-129.
Zghoul, N., R. Fuchs, C. M. Lehr, and U. F. Schaefer. 2001. Reconstructed skin equivalents for assessing percutaneous drug absorption from pharmaceutical formulations. ALTEX 18:103-106.
Zhang, H., and W. Davison. 1995. Performance characteristics of diffusion gradients in thin films for the in situ measurement of trace metals in aqueous solution. Anal. Chem. 67:3391-3400.
Zhang, H., F. J. Zhao, B. Sun, W. Davidson, and S. P. McGrath. 2001. A new method to measure effective soil solution concentration predicts copper availability to plants. Environ. Sci. Technol. 35:2602-2607.
Zhang, H., W. D. S. Miller, and W. Tych. 1995. In situ high resolution measurements of fluxes of Ni, Cu, Fe, and Mn and concentrations of Zn and Cd in porewaters by DGT. Geochim. Cosmochim. Acta 59:4181-4192.
Zhang, H., W. Davison, B. Knight, and S. McGrath. 1998. In situ measurements of solution concentrations and fluxes of trace metals in soils using DGT. Environ. Sci. Technol. 32:704-710.