8
Materials by Design1
|
Some Challenges for Chemists and Chemical Engineers
|
GOALS
A material is defined as “the substance or substances out of which a thing is made or composed.”2 Understanding materials therefore necessitates a marriage between understanding substances and knowing how to assemble them into useful structures. Materials with specially tailored properties are at the core of nearly all interesting assemblies that are not living, and many that are. Chemical scientists synthesize, characterize, produce, and construct with materials. Moreover, they do this at length scales from molecules at the nanometer level, to polymers and electronic devices at the submicron scale, and to ceramics and cement in large-scale structures. Revolutionary developments are being made in all aspects of materials science. The materials sector of the chemical sciences is vital, both fundamentally and pragmatically, for all areas of science and technology—as well as for the societal needs in energy, transportation, national defense, and medicine.
The overall goal of materials research within the chemical sciences is to explore, design, and control—through synthesis and processing—the relationships among structure, properties, composition, and processing that determine the useful behavior of all materials. The chemical sciences are especially powerful (as they are in all areas of synthesis and manufacturing) in molecular-level construction of material structures. Though the domain of materials chemistry is rapidly expanding, it remains underdeveloped.
A frontier challenge in the chemical sciences is to investigate the chemistry and properties of single isolated molecules and compare that behavior with the average molecular behavior in an assembly, solution, or condensed phase of molecules. Many parts of the chemical sciences are concerned with the collective properties of materials in condensed phases, which have a variety of intriguing and controllable properties. Today, we recognize fully that the most interesting materials are functional systems, derived from our evolving knowledge of structure-function relationships. Catalysts are superb examples of chemically functional materials, and the area of catalytic materials is an exemplary branch of modern functional materials science. Responsive materials, often consisting of softer materials, give rise to many kinds of functionality, such as sensing and actuation.
Composites are an important class of solid materials, whose history goes back to ancient times. For example, bricks made only of clay were not as strong as those in which straw was mixed with the clay. Now we use clay as the filler in new polymeric composites to enhance their mechanical properties. The ionic con-
ductivity of the polymer electrolytes used in energy storage systems also can be enhanced by addition of tiny clay particles. Another example is found in building construction, where concrete is reinforced with steel rods to producing a composite in which the components mutually reinforce the overall strength. Other recent examples are the graphitic materials used in tennis rackets and golf clubs, where long strands of carbon fibers are combined with resins. The science of the boundaries or interfaces between phases, and aggregates of matter with sizes between the molecular and the macroscopic, has become a vigorous part of the chemical sciences. Constructing nano- and microstructures of any complexity requires joint modules or elements through interfaces.
The goals of molecular understanding, synthetic control, and novel fabrication of various materials are inherently based in the chemical sciences and technology. The goal of applied chemistry and chemical engineering is to convert available substances into useful materials, normally by changing their molecular composition and arrangement, through controlled synthesis, processing, and manufacturing methods.
Chemical scientists seek to understand the properties of materials in which there is organization of the components. Chemistry is the original synthetic nanotechnology (as biology is the original natural nanotechnology); chemists have been designing and executing constructions requiring placements of atoms with subnanometer precision for most of the last century. Chemical engineers are now aiming to do this on larger scales. As self-assembly and nanotechnology move from laboratory demonstrations to more widespread means of fabrication and manufacturing, the variety of materials available to technology and society will grow enormously. New catalytic chemistry and processes, such as the revolution in metallocene catalysts, is an area where chemists and chemical engineers are creating new routes to macromolecular structural control at the nanometer scale.
Methodologies of synthetic chemistry (Chapter 3) must be adapted to achieve the full potential of chemical materials science and technology. This in turn will allow chemists and chemical engineers to characterize the synthesis of supramolecular entities and the three-dimensional character of materials. Our abilities to achieve these goals in new materials synthesis are enhanced by intricate optical, micromechanical, and spectroscopic probes, just as they are by the use of noncovalent bonding, self-assembly, and assembly directed by forces such as fluid mechanical or electric fields. The miniaturization and diversification of synthesis through biological or combinatorial approaches provide unprecedented opportunities. Chemical science should take better and broader advantage of naturally abundant substances to produce building blocks for molecular (or larger) assemblies. Surface science applied to materials—particularly to organic materials—is of growing importance and will expand significantly with the development of new materials for biotechnology, medicine, information technology, and nanotechnology.
To achieve these goals will require, among many other things, a dramatic increase in the interactions among chemists, engineers, biologists, and physicists.
PROGRESS TO DATE
Since early civilization humans have been interested in the properties of the various minerals found in the earth. The discovery that materials we now recognize as iron oxides could be heated with charcoal to produce iron led to wonderful new tools in the Iron Age, while similar transformation of other minerals led to copper, tin, and other metals. Although we think of them as common, few metals are naturally occurring; they are produced by chemical reactions of their naturally occurring compounds. One of the earliest synthetic materials is glass, produced over 5,000 years ago by heating various natural minerals together. Clearly, the discovery, refinement, and creation of materials has arisen from the chemical sciences and processing technology (and sometimes vice versa).
Synthetic Polymers and Self-Assembly
The story of polymers is one that shows enormous effects on human life. Though polymer science revolutionized 20th century life and is now a well-developed academic field, polymer synthesis is still progressing rapidly. Synthetic polymers have often consisted of long chains of identical subunits. Sometimes the synthetic polymer chains have cross-links between the chains (in proteins, cross-links within a chain help determine a specific folded geometry). For many years, copolymers have also been produced to gain the beneficial properties from more than one monomer. Glassy polymers can be blended with rubbery ones to generate desirable mechanical properties. Block copolymers—produced with long runs of one or the other monomer—phase separate on a nanoscopic scale (typically 10 to 50 nm) that is determined by the block molecular weight. These microphase-separated polymers often have remarkably better properties than blends of the two components, and are an early example of using self-assembly to produce new materials.
The architecture of macromolecules is another important synthetic variable. New materials with controlled branching sequences or stereoregularity provide tremendous opportunity for development. New polymerization catalysts and initiators for controlled free-radical polymerization are driving many new materials design, synthesis, and production capabilities. Combined with state-of-the-art characterization by probe microscopy, radiation scattering, and spectroscopy, the field of polymer science is poised for explosive development of novel and important materials. New classes of nonlinear structured polymeric materials have been invented, such as dendrimers. These structures have regularly spaced branch points beginning from a central point—like branches from a tree trunk. New struc-
tures create new possibilities for applications, a direction that will continue to drive materials chemistry.
High-molecular-weight polymers can be useful as solid materials and in solution, and lower molecular weight polymers can make liquids that are unusual in character. Synthetic adhesives illustrate liquid-phase materials that cross-link or polymerize when they set. Water-based paints are another example, liquids with suspended solid polymer particles that form uniform solid films during drying. So-called liquid crystals illustrate another exciting example of complex fluid materials; these are liquid-phase materials made up of anisotropic, usually fairly rigid, molecules of high aspect ratio that have strong electric dipole moments. Such molecules are prone to adopt preferred orientations, especially under the influence of surfaces, electric fields, and flow processes. Control over preferred orientations gives high anisotropic strength of materials and switchable optical properties, making them useful in displays such as those on digital watches and laptop computers.
Multicomponent systems having molecules of macromolecular size and heterogeneous composition can be exquisitely sensitive to the delicate balance of intermolecular forces. The fine interplay among a suite of noncovalent interactions (e.g., steric, electrostatic, electrodynamic, and solvation forces) dictates microstructure and dynamics. Molecular organization and interaction cause collective and cooperative behavior to dictate macroscopic properties. Often the balance of forces is such that self-assembly occurs to generate aggregates, arrays, or other supramolecular structures. Large molecular size enables amplification of a small segmental effect into a large intermolecular effect. Self-assembly can amplify the small forces between small objects to produce large-scale structures useful for macroscopic creations for patterning, sieving, sorting, detecting, or growing materials, biological molecules, or chemicals. Learning to understand and harness intermolecular interactions in multicomponent polymer and composite systems offers huge challenges, as well as opportunities to mimic nature, which has learned to do this in many instances.
Self-assembled monolayers (SAMs) are ordered, two-dimensional crystals or quasi-crystals formed by adsorption and ordering of organic molecules or metal complexes on planar substrates. Development of these monolayers is based on early studies in which chemists learned to attach chemicals to surfaces—for purposes ranging from adhesion to chromatography to electrochemistry—but often without strong ordering in the monolayers. The ordered structures have made it possible to develop a rational surface science of organic materials. They provide the best current example of the power of self-assembly to make possible the design of the properties of materials. They have made routine the control of wetting, adhesion, and corrosion in certain systems, and—through soft lithography—they have provided a new approach to microfabrication that is uniquely chemical in its versatility. They have also greatly advanced the field of biomaterials by making it possible to control the interface between cells and synthetic materials at the molecular level.
|
Soft Lithography Building ever smaller devices has been a dominant trend in microelectronics technology for 50 years. The technology used for this type of fabrication is photolithography. This astonishingly sophisticated technology is a kind of photography: The pattern that is to be a part of the circuit is formed by shining ultraviolet light through a mask (a pattern of chromium on silica), through a reducing lens, onto a thin film of photosensitive polymer (a photoresist) covering the surface of a silicon wafer. After exposure, the exposed polymer differs in its solubility from unexposed material, and a suitable solvent allows the selective dissolution of either exposed or unexposed regions. The exposed regions are then treated (by deposition of metal, etching, or implantation of ions) to make a part of the final device. Photolithography is the basis of one of the technologies that has genuinely changed the world—it has made possible the computing and information revolution. But it is not suited for making every possible type of small structure. As the advantages of “small” have become obvious in microelectronics, researchers have searched for ways to make small channels (for analysis of fluids or for synthesis), small machines (so-called microelectromechanical systems, or MEMS, for accelerometers and display projectors), and small optical systems (for optical communications). Such targets for fabrication all have different requirements in materials and costs, and photolithography is not a “one size fits all” technology. An alternative to photolithography has been developed that is—for many applications in chemistry and biology—more versatile and much less expensive. This technology depends on a “back to the future” strategy that produces micron- and nanometer-scale patterns by stamping, printing, and molding. The element in these methods is a stamp or mold that is fabricated in a transparent, chemically inert elastomer, poly(dimethyl siloxane) (PDMS). Because the stamp can deform, it is called soft; the organic materials that are printed and molded are also called soft matter by physicists—hence the name soft lithography. Patterns of small features are embossed in the surface of the element; when it is “inked” with a suitable ink, it can print lines that are <100 nm in width (that is, the size of a line of 200 gold atoms). As illustrated here, when the recessed regions are filled with a polymer, and the pattern is transferred to a surface, the resolution is <10 nm. If the stamp is sealed to another surface, the patterns become microchannels for analysis of nucleic acids, proteins, or cells. |
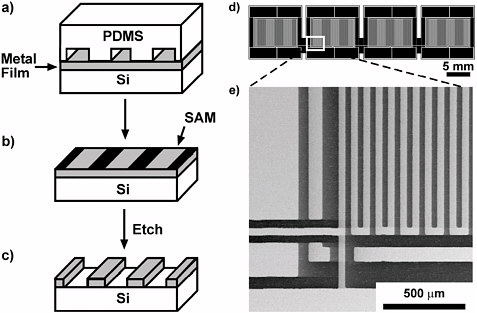 Soft Lithography. Figures a-c illustrate a soft-lithographic technique called microcontact printing. A PDMS stamp with features in bas-relief is coated with an ethanolic solution of octadecanethiol, and placed in contact with the surface of a thin metallic film (30-50 nm) of gold, silver, or palladium. A self-assembled monolayer (SAM) of octadecanethiolate forms on the surface of the metal in the regions where it contacts the PDMS stamp. The stamp is removed and the regions of the metallic film without a SAM are dissolved by wet-chemical etching. Figure d is a schematic diagram of a long, serpentine, palladium wire (2 m) with contact pads that are connected to the wire at every 0.25 m along the length of the wire; it is a part of a sensor for hydrogen. Figure e is a SEM image of a section of the pattern. Drawings a-c courtesy of George M. Whitesides; d-e reprinted with permission from D. B. Wolfe et al., Applied Physics Letters 80, 12, 2222 (2002). Soft lithography is very simple, and it does not require expensive instrumentation or access to clean rooms. It does not give the lateral accuracy of photolithography, but it is much less expensive. The size of the features it can make is not limited by optical diffraction, but rather by van der Waals contacts and by deformations in the polymer used. It has become a tool that is widely used in chemistry to make micro- and nanostructures. It also has helped to open doors to chemists wishing to play an active role in many areas of cell biology, bioanalytical chemistry, microfluidics, optics, and new forms of electronics such as “all-organic” electronics: that is, electronics that does not rely on silicon, but instead uses organic or organometallic compounds as conductors, semiconductors, and insulators. |
Micelles, liposomes, shell-linked particles, and vesicles are all results of the spontaneous self-assembly of amphiphilic molecules to form enclosed or aggregate structures that contain solvophobic regions surrounded by solvent-loving moieties. In all of these structures, opportunities abound to exploit them for chemical separations, controlled release, directed transport, and synthesis. Fundamental studies of these organized systems have increased in the recent decade. The pursuit is often biologically inspired, but in creating mimics we still fall short of the natural systems. Combining this activity with concerted synthetic chemistry and biochemistry provides great potential for the future.
Electronic, Optoelectronic, Photonic, Magnetic, and Superconducting Materials
The properties of modern electronic, optoelectronic, photonic, and magnetic devices provide another story of great science that has affected most of humankind. Electronic devices require special materials: materials that emit light when struck by a beam of electrons for use in television screens and computer monitors, materials to make the semiconductors that are the heart of electronic and microelectronic circuits, and materials that are used in magnetic memory storage devices for computers.
Classical electronic circuits and communication lines are made of metal to conduct electricity. Now we have the prospect of massively communicating by optical signals. The great progress in the use of optical fibers to permit light to travel in and between devices results from major achievements in materials processing. Special surface coatings on the fibers reduce signal degradation; optical switches allow connections with devices communicating through optical fibers. The optical fiber revolution provides very high speed plus the ability to pack much more information into a given transmission.
There is considerable interest in developing new types of magnetic materials, with a particular hope that ferroelectric solids and polymers can be constructed— materials having spontaneous electric polarization that can be reversed by an electric field. Such materials could lead to new low-cost memory devices for computers. The fine control of dispersed magnetic nanostructures will take the storage and tunability of magnetic media to new levels, and novel tunneling microscopy approaches allow measurement of microscopic hysteresis effects in iron nanowires.
One of the most exciting properties of some materials is superconductivity. Some complex metal oxides have the ability to conduct electricity free of any resistance, and thus free of power loss. Many materials are superconducting at very low temperatures (close to absolute zero), but recent work has moved the so-called transition temperature (where superconducting properties appear) to higher and higher values. There are still no superconductors that can operate at room temperature, but this goal is actively pursued. As more current is passed through
a superconductor the transition temperature moves lower; consequently, high electrical current tends to make the materials lose their superconducting ability. The development of a full, predictive theory of high-temperature superconductivity would be a major asset to the realization of practical materials in this area. The materials studied to date are also difficult to process—they are easily corroded or brittle—thus motivating further study of novel processing or assembly techniques. If practical superconductors can be made that will conduct appreciable currents at reasonable temperatures—perhaps even from organic materials—it may become possible to transfer electric power over long distances with high efficiency, and to exploit magnetic levitation for transportation systems.
|
Quantum Computation The first demonstration of continuous electrical tunability of spin coherence (the state and degree of alignment of electronic spins) in semiconductor nanostructures has recently been made. This opens possibilities for the field of quantum computation by permitting properties other than electronic charge—and particularly the quantum property of spin—to be manipulated for computing purposes. Spin, often described by analogy with rotation of the earth, is a quantum property of electrons (and some atomic nuclei) that must have one of two possible values analogous to clockwise or counter-clockwise rotation of a rotating body. While magnetic fields are conventionally used to manipulate spins in familiar magnetic devices like hard-disk drives, this demonstration of electrical control of aligned spins represents a significant step toward making new spin-based technologies. One future technology is quantum computing, where many schemes make use of electron spin states as bits of information analogous to the 0 and 1 of binary computing. Unlike ordinary bits, quantum bits can be any combination of both 0 and 1 simultaneously, corresponding to a continuous range of possible directions. By classical mechanics, magnetic fields can modify the behavior of spins by inducing precession, which is an additional rotation of the spin axis with respect to the magnetic field. While the speed of electron spin precession in a magnetic field is generally fixed by the particular materials used, recent research has shown that both the speed and direction of precession can be continuously adjusted by applying electric fields in specially engineered quantum structures.3 It is more fitting to refer to |
|
these devices as gates rather than switches because they perform continuous tuning of electron spin. Instead of the “off” and “on” options for a switch, a gate operates across a continuum in the same way that lights can be dimmed by a rheostat. Such spin gates are an example of the rapidly developing field of spintronics, which studies electronic devices that are based on electron spin. Spintronics uses magnetic fields to manipulate the distribution of spin coherence, whereas electronics uses electric fields to manipulate charge distribution. This raises the question, What might spintronics do that electronics cannot? In addition to the longer-term goal of quantum computing, spintronics offers the near-term possibility of revolutionizing the way we think about piecing together different technologies. The creation of nanoscale sandwiches of compound semiconductor heterostructures, with gradients of chemical composition that are precisely sculpted, could produce quantum wells with appropriate properties. One can eventually think of a combined device that incorporates logic, storage, and communication for computing—based on a combination of electronic, spintronic, photonic, and optical technologies. Precise production and integrated use of many different materials will be a hallmark of future advanced device technology. The opportunities to develop new structures for computing, quantum computation, and spintronics—together with other areas in molecular electronics—raise important issues about the role of computation in the chemical sciences (Chapter 6). In order for chemical scientists to play a major role in converting clever new ideas for computational devices into full-fledged computers, they will have to become increasingly competent in the architectures, algorithms, and protocols that are necessary for reliable computation. |
Ceramics, Carbon Structures, and Crystal Engineering
Inorganic substances are the components of ceramics, such as those in dinner plates. Ceramics have important industrial uses as well; a typical example is the ceramic insulating materials that are used to suspend power lines. Ceramics are typically poor conductors of heat and electricity, and they perform well at high temperatures. Consequently, they find applications that take advantage of these properties. Some use of ceramics in automobile engines is being developed to achieve improved fuel efficiency at higher-temperature operation. The fragility of current ceramics and the difficulty in machining them (relative to machining
metals) are still problems for such uses. One approach to ceramics with better properties is to overcome their fragility by incorporating them into composites. As chemistry moves from pure materials to organized systems of different materials, composites are leading the way. A challenge for the future is to invent improved structural materials, probably composites based on resins or on ceramics, that are stable at high temperatures and easily machined.
Carbon atoms in pure form can be obtained as materials having two classic types of molecular structure: diamond and graphite. In diamond each carbon atom is linked by equivalent single bonds to four neighboring carbons. The result is a clear very hard material that is used for cutting, in saws with tiny diamonds imbedded in the blades, as tough coatings for metals, and in other industrial uses as well as in jewelry. By contrast, each carbon atom in graphite is linked to only three neighboring carbons, in a sheet, and some of the electrons are in delocalized pi orbitals that permit them to move easily along the sheets. The extensive aromaticity of carbon sheets leads to electronic transitions with energies in the visible light region, so that graphite absorbs throughout the visible region and is a black material. In addition, the mobility of the pi electrons in graphite makes it an electrical conductor, in stark contrast to the insulating properties of diamond.
A new type of structure has recently been discovered in which the sheets of graphite-like carbons are curved. The first example, called fullerene (after the geodesic domes of Buckminster Fuller), has 60 carbons in a sphere. It resembles a soccer ball with its five- and six-sided polygons (in contrast to graphite, which resembles a floor tile pattern, with hexagons only). A Nobel prize was awarded in 1996 to Robert F. Curl, Jr., Harold W. Kroto, and Richard E. Smalley for their discovery of fullerenes. Instead of curling into a sphere, the sheets of carbon with hexagons can also curve into tubes with diameters on the order of 1 nm (often called nanotubes), tiny whiskers that are sometimes quite long. Because of the electrical conductivity of pi electrons, these tubes are also electrically conducting, somewhat like graphite. While they are already used in research instruments to probe microscopic structures, one of the challenges is to use these new structures in miniature devices, or as building blocks for organized chemical structures.
The importance of crystal form often is underappreciated. In many applications—from drugs (in which bioavailability may be determined by crystal form) to explosives (where crystals may differ in stability) and optical devices (where the nonlinear optical properties required for the device are based on a particular crystalline architecture)—the correct crystalline form is essential to obtaining the desired chemical and physical properties of a material. Crystallization has long been an art rather than a science; sometimes the same substances will exhibit polymorphism and adopt different crystalline forms depending on the crystallization conditions. Crystal engineering—the prediction and control of molecular crystal structures based on the constituent molecular structures—is on the verge of becoming a science. The current generation of computers is finally powerful
enough to rationalize the crystal structures of simple, relatively rigid, organic molecules. As computer capability increases, and as the sophistication of the programs used increases, it seems very probable that it will soon be possible to predict the structures of crystals. Learning to template or guide desired organization of molecules will have great utility.
Layered Materials and Surface Modification
The scale of components in complex condensed matter often results in structures having a high surface-area-to-volume ratio. In these systems, interfacial effects can be very important. The interfaces between vapor and condensed phases and between two condensed phases have been well studied over the past four decades. These studies have contributed to technologies from electronic materials and devices, to corrosion passivation, to heterogeneous catalysis. In recent years, the focus has broadened to include the interfaces between vapors, liquids, or solids and self-assembled structures of organic, biological, and polymeric nature.
In a simple material, its surface properties are dictated by the properties of the bulk, which are not necessarily desirable. For example, we may need a bulk material for its strength but want to make a medical device—such as an artificial heart—where the surface must not cause a reaction leading to rejection or blood clotting. This leads to the challenge of learning how to add biocompatible surface layers to materials. This challenge is not yet fully met, but interesting approaches to creating biomimetic functionality on surfaces are rapidly emerging. This field is an example of the transition of chemistry from pure materials to organized systems and materials, in this case the organization being the modification of the surface with a different material for a biofunctional purpose.
The ability to modify surfaces by attaching chemicals to them has for years encouraged scientists to attempt to design surface adhesive and wetting properties. The advent of self-assembled monolayers, including mixtures of molecules in a monolayer, has led to more detailed control and understanding of surface adhesion and wetting. This capability has been extended with the use of novel monolayers to alter liquid-crystalline anchoring processes, surface friction, and biocompatibility. Important applications of this approach have arisen in microfluidics and liquid crystalline displays. Work pioneered by Nuzzo and Allara at Bell Laboratories in the early 1980s with thiol self-assembled monolayers on gold has led to a great deal of research, much of which has been revolutionary.
Thus the study of surfaces has emerged as an important focus in the chemical sciences, and the relationship between surfaces of small systems and their performance has emerged as a major technological issue. Flow in microfluidic systems—for example, in micromechanical systems with potential problems of stiction (sticking and adhesion) and for chemistry on gene chips—depends on the properties of system surfaces. Complex heterogeneous phases with high surface areas—suspensions of colloids and liquid crystals—have developed substantial
technological importance. In certain size ranges, we have seen new and scientifically engaging phenomena, such as electron tunneling through nanometer-thick insulators and diffraction of light in photonic band-gap crystals. New tools and systems—from scanning tunneling microscope and atomic force microscope (STM and AFM) to self-assembled monolayers and carbon nanotubes—have fundamentally changed our ability to characterize and prepare these complex systems. Finally, microelectronics—complex systems of small functional components fabricated in silicon and silicon dioxide, and other materials—have become so important that we must develop the science and technology relevant to future systems of small components, whether based on microelectronics or other technologies. The microelectronics industry is entirely based on chemical processing, using such techniques as chemical vapor deposition (CVD), plasma processing, etching, and electroless deposition.
Nanomaterials
As the analytical, synthetic, and physical characterization techniques of the chemical sciences have advanced, the scale of material control moves to smaller sizes. Nanoscience is the examination of objects—particles, liquid droplets, crystals, fibers—with sizes that are larger than molecules but smaller than structures commonly prepared by photolithographic microfabrication. The definition of nanomaterials is neither sharp nor easy, nor need it be. Single molecules can be considered components of nanosystems (and are considered as such in fields such as molecular electronics and molecular motors). So can objects that have dimensions of >100 nm, even though such objects can be fabricated—albeit with substantial technical difficulty—by photolithography. We will define (somewhat arbitrarily) nanoscience as the study of the preparation, characterization, and use of substances having dimensions in the range of 1 to 100 nm. Many types of chemical systems, such as self-assembled monolayers (with only one dimension small) or carbon nanotubes (buckytubes) (with two dimensions small), are considered nanosystems.
Whether there is currently a nanotechnology is a question of definition. If one asks whether there are (or are soon likely to be) commercial electronic fluidic, photonic, or mechanical devices with critical lateral dimensions less than 20 nm, the answer is “no,” although there may be in 10 to 20 years. There is, however, a range of important technologies—especially involving colloids, emulsions, polymers, ceramic and semiconductor particles, and metallic alloys—that currently exist. But there is no question that the field of nanoscience already exists.
The current, intense interest in “nano” is based on the (correct) perception that the study of nanoscience has exploded. As new tools have become available for the preparation and characterization of systems with these dimensions, the opportunities in the chemical sciences have grown enormously. The attention
also results from the as yet untested proposition that nanoscience will eventually revolutionize existing areas of important technology, especially microelectronics.
There is great interest in the electrical and optical properties of materials confined within small particles known as nanoparticles. These are materials made up of clusters (of atoms or molecules) that are small enough to have material properties very different from the bulk. Most of the atoms or molecules are near the surface and have different environments from those in the interior—indeed, the properties vary with the nanoparticle’s actual size. These are key players in what is hoped to be the nanoscience revolution. There is still very active work to learn how to make nanoscale particles of defined size and composition, to measure their properties, and to understand how their special properties depend on particle size. One vision of this revolution includes the possibility of making tiny machines that can imitate many of the processes we see in single-cell organisms, that possess much of the information content of biological systems, and that have the ability to form tiny computer components and enable the design of much faster computers. However, like truisms of the past, nanoparticles are such an unknown area of chemical materials that predictions of their possible uses will evolve and expand rapidly in the future.
Several techniques are now available for the fabrication of nanostructures. These techniques arise from four approaches, and their simultaneous applicability to a common set of targets is one of the reasons for the excitement in the field. The first set includes the classical techniques developed from microfabrication:
-
electron beam writing, which is the most important, although x-ray and deep UV photolithographies may also contribute;
-
use of scanning probe devices to move individual atoms or to write patterns;
-
preparation of colloids, vesicles, emulsions, buckytubes, and self-assembled monolayers using chemical self-assembly methods (some with histories that date back to the beginning of chemistry);
-
soft lithography and nanoimprint lithography, which use printing, molding, and embossing technologies developed on the macroscopic scale to replicate structures at the nanoscale.
The characterization of simple nanostructures is now possible with remarkable detail, but is highly dependent on access to the tools of measurement science and to scanning probe microscopies.
These methods have made available a set of nanostructured systems that have begun to reveal the characteristics of nanoscale matter. The long list of discoveries in the last decade includes:
-
“quantum box” behavior in colloids of semiconductors precipitated from solution;
-
quantized capacitive charging of metal nanoparticles coated with low dielectric monolayers;
-
a range of electrical properties in carbon nanotubes grown from vapor-phase precursors using metallic catalysts (with the highest observed conductivities comparable to those observed in graphite);
-
high mechanical strength of buckytubes (combined with the above-mentioned electrical properties) that makes them possible candidates for “nanowires”;
-
remarkably regular nanostructures in phase-separated block copolymers;
-
functional transistors prepared in organic semiconductors with 100-nanometer gate widths;
-
membranes containing nanopores with controlled interior functionality;
-
versatile methods of preparing nanostructures that are based on simple ideas taken from printing, writing, molding, and embossing, and that have made it possible to prepare certain nanostructures without use of expensive apparatus.
Two important conclusions have emerged in this field. First, the methods employed for microelectronics—photolithography using UV wavelengths—are unlikely to provide inexpensive access to nanostructures. Second, the techniques of chemistry and chemical engineering—although very early in their development—will be able to provide nanostructures with a wide range of compositions and properties, and at costs that are very low compared to those prepared by e-beam or other “conventional” techniques for fabrication at small dimensions. In particular, chemical affinities should make it possible for tiny structures and devices to self-assemble spontaneously, an appealing idea for large-scale manufacturing.
CHALLENGES AND OPPORTUNITIES FOR THE FUTURE
Many of the challenges of the formation and processing of new materials will be met with advances in the chemical sciences. There are some revolutionary things happening in materials: organic electronics and spintronics, attempting to replace classical silicon electronics, the exploration of single-molecule electronics to achieve the ultimate in size reduction, sophisticated biocompatible materials for tissue engineering, implants, man-machine hybrids, ferromagnetic organic materials, materials with negative index of refraction, nanoelectronics, and functional colloids.
Self-assembly and nanotechnology are advancing rapidly, but the challenge still remains to develop a means of fabrication and manufacturing. The rapid developments in synthetic chemistry produce myriad new polymeric and composite materials. These advances are enhanced by progress in optical, micromechanical, and spectroscopic probes. The miniaturization and diversification of synthesis through biological or combinatorial approaches provide unprecedented
opportunities. The approach to the future should be a holistic one, with synthetic advances moving in concert with assembly and microstructural control. Summarized below are a few of the leaps that can be viewed as important aspirations for the chemical science community.
Templating
The development of templated syntheses—of metallic, ceramic or semiconductor particles, wires using novel synthetic and self-assembling structures such as dendrimers, micelles, and nanotubes—is in its infancy. This is a prime example where synthetic advances in the creation of new lipids, surfactants, and amphiphilic polymers work together with probes of structure and function of infinitesimal wires or particles. New techniques such as scanning microscopy must be developed to follow the electronic and magnetic processes occurring in the small systems. Spectroscopists, microscopists, engineers, and chemists must work together at the frontiers involving techniques developed by those from disparate fields of electronics and biology.
Higher Order Structures
The ability to program synthetic polymers with the correct information to self-assemble, recognize analytes, or provide biological function seems fairly futuristic. However, the close interplay between chemical composition and physical interactions makes this a possibility; new synthetic approaches involving controlled living polymerizations and biological synthetic pathways allow control of molecular composition. Additional research on the balance of physical forces driving self-assembly, recognition, field responsive behavior, and biological compatibility should be closely tied to the synthetic efforts.
New approaches in synthetic chemistry and biochemistry pave the way for tremendous advances in self-assembly. Highly controlled living polymerizations will allow the creation of ever more complex macromolecules having prescribed architectures (branching, stereoregularity) and chemical specificity. The future will hold the opportunity for chemists to make molecules of size and complexity approaching protein structures—and to fold and assemble them. Then, mimicking nature becomes a question of choosing important problems and technologies needing improvement or intervention. By creating the appropriate molecules, patterning them on the appropriate surfaces, and providing them with the appropriate functions, we can think of mimicking the most chemical of senses: taste and smell. These analytic advances, when taken in parallel with the computational and electronics revolution, suggest the possible creation of the robots and gadgets that were previously envisioned only in science fiction.
Semiconductor Processing
We need novel materials to maintain the computer revolution, since we may be reaching the limits of “top down” miniaturization. Instead of etching pieces of silicon to produce electronic circuits that are smaller and smaller, there is the hope that “bottom up” design will work. In other words, self-assembly of molecules or nanoparticles offers the potential for construction of miniature electronic circuits that will be faster and will permit more computer power in a given space. Promising physical approaches involve soft-lithography or crystal or nanoparticle growth, but other processes doubtless will emerge as the field of nanoparticle science evolves.
Chemical scientists will seek new methods of generating nanostructures with a range of materials and processes that rely on ideas common in chemistry—self-assembly, diffusion, phase-separation, catalysis, wetting—to make these structures accessible and inexpensive.
In terms of technology, it is too early to predict what will emerge from nanoscience, although it is clear—for a field as fundamental as this one—that technologies will surely emerge. Candidates for early success include systems of photoluminescent colloids in which the same materials base provides any desired color simply by tailoring the size (for displays); compact disks with <50-nanometer pits (for very dense memory devices that will require near field recording technology); and optical elements for manipulating extreme UV and x-ray light. In the longer term, there will be, at minimum, demonstrations of information processors having key components with nanometer dimensions (perhaps made of organic or organometallic materials) and probes for exploring the interior of the cell.
As self-assembly and nanotechnology move from curiosities and demonstrations to more serious means of fabrication and manufacturing, the need for characterization tools, especially those that can meet the time scales for real-time processing, will grow enormously.
Molecular Electronic Materials
Improving the molecular control of addressable, switchable, or conducting molecules that have extremely high purity, selectivity, or specificity is a goal within reach in the coming decades. This will require the combination of synthetic and processing strategies, such as recognition and controlled binding, to tailor oligomeric materials with finely tuned properties. In this field, the chemical sciences will have to interact creatively with computer science and engineering in order to turn promising molecular switching ideas into practical computer architectures.
Composite and Hybrid Materials
As the ability to control materials moves to molecular dimensions, the expectations for composite materials will grow. Rather than relying on incorporation of macroscopic particles or fibers as discussed above, one can hope to create important marriages between disparate materials that will allow the development of new material properties. Combining this synthetic expertise with physical patterning, self-assembly, deposition, or quenching techniques will lead to the creation of new materials with optimal properties. Organized nanocomposite materials can be important for photonic band gap materials as well as membranes and catalysts with high selectivity.
Surface Modification and Interfaces with Biology and Electronics
The connection between biological function and a useful electrical signal is the capstone of sensor technology that will change medical, environmental, and personal-protection strategies in the coming decades. The link between biology and electronics is through the chemical sciences. The ability to mimic nature and reliably anchor biologically active moieties to a surface is in its infancy. Here another level of complexity and functional integration are possible. Coupling the physical chemical means to manipulate interfaces with synthetic strategies inspired by nature provides powerful opportunities for gains in environmental and medical devices. As an example, the recent understanding of virus phage packaging, combined with the ability to inject DNA into a host or a host mimic, opens the way for molecular scientists to develop new therapies and delivery strategies. Another challenge is the construction of materials with the kind of actuating response found in physiological systems such as muscle—soft materials in which a large-amplitude mechanical response could be produced in response to a small-amplitude stimulus.
Capitalizing on self-assembly and nanoscience will enhance the ability to screen drugs for individual sensitivities. Advances in drug discovery, combinatorial synthesis, and screening with sensors that have the ability to detect multitudes of specific genetic matches—marrying microelectronics and self-assembly—are expected to be near-term breakthroughs. Then the creation of advanced forms of “in the field” or “in the office” tests for chemical risks, pharmaceutical compatibility, or environmental hazards will be possible.
The processing of materials through self-assembly will also have to meet several kinds of challenges that arise from scaling up in physical size and speed, and in complex shapes. Little has been done with complex shapes and nonplanar surfaces. Can processes that work on glass slides or mica or 4-inch silicon wafers be scaled up to very large surface areas, such as continuously moving webs of paper, film, or tape, or surfaces of tubular biomaterials? Building large volumes
of nanostructured, self-assembled materials in three dimensions may be even more challenging than scaling up to large surfaces.
Processing speed presents different challenges. Since self-assembly is a process that moves down a free energy gradient, there is a predetermined end point; but it is not known how to anticipate its speed. Study of the kinetics and dynamics of self-assembly processes will be necessary to bring this field to a level comparable to traditional methods of synthetic chemistry and chemical engineering. Few studies of kinetics of assembly processes have been pursued, in part because following the assembly process presents analytical difficulties. Practical processing rates will certainly have to be much faster than those of typical current research laboratory practice. The free energy landscapes along the paths toward self-assembled products are not fully explored. Local minima and metastable states lurk but are uncharted, and it is often not appreciated when they are occupied or when they are trapping the process far from the desired equilibrium state. The challenges of kinetics and of metastable states raise the question of catalysis and whether routes to assist, accelerate, and guide self-assembly processes can be developed.
Self-assembly processes in nature are sometimes catalyzed by enzymes. Zeolites are, in many ways, the inorganic counterparts of enzymes, with their ability to selectively bind other substances and perform catalysis. Can templates or catalysts be effective in increasing rates and reducing defects in a wide range of nanostructured materials?
One of the most general forms of the surface modification of materials is painting. We have become used to the idea that paints, while serving their important function of preventing corrosion and water damage, need to be renewed on a regular basis, with a significant cost in labor and materials. One less glamorous challenge that could make a significant contribution to modern life is the invention of long-lasting paint, perhaps 100-year paint. In addition, it would also be desirable that this long-lasting paint be easily cleaned, perhaps simply by natural rain, and that it have a mechanism to repair damage to itself. We are used to the idea that our body can repair wounds; it is an important challenge to devise methods by which synthetic materials would also have this wound-healing ability. As one approach, the wound might expose pools of monomer that could spontaneously fill the void and solidify.
Green Materials and Eco-Technology
Other challenges facing materials scientists have to do with the environment. We have traditionally made materials that are as stable as possible, so they will last a long time and not need to be replaced. This longevity has the undesirable consequences of creating waste that requires significant energy to process, or else it clutters the landscape when discarded. As one approach, chemical scientists and engineers need to focus on recycling and materials that can be easily re-
cycled. Another approach is to produce materials that undergo rapid degradation into invisible and harmless substances. Some progress has been made by incorporating ketone groups into polyethylene so that sunlight will break chemical bonds and cause the polymer to disintegrate into tiny particles. However, these materials are not yet economically attractive, and more efforts of this kind are needed.
This is part of the general need, discussed in Chapter 9, to make materials such as insecticides or refrigerants that will degrade in the environment rather than cause problems with bird life or with the ozone layer in the stratosphere. In addition, as the world demand for synthetic materials grows, new renewable resources for chemical feed stocks must be sought, and we must reconsider our current infatuation with burning the petroleum reserves that are important feedstocks (as discussed also in Chapter 10). Zero-effluent processing plants also need to be developed. There are innumerable opportunities for advanced chemical processing of materials to create micro- and nanodevices for environmental and ecological monitoring.
Another challenge is to develop methods to replace the volatile organic solvents that are used in many industrial procedures. One choice is water as a solvent; it is easily repurified, and has a harmless vapor. Another choice is supercritical carbon dioxide, a good solvent for many organic substances. It is not as innocuous as is water, but carbon dioxide can be easily recovered and reused. It is currently used to remove caffeine from coffee, and is being developed as a dry-cleaning solvent to replace organic solvents (Chapter 9).
Analysis and Simulation
Advances in computational capability have raised our ability to model and simulate materials structure and properties to the level at which computer “experiments” can sometimes offer significant guidance to experimentation, or at least provide significant insights into experimental design and interpretation. For self-assembled macromolecular structures, these simulations can be approached from the atomic-molecular scale through the use of molecular dynamics or finite element analysis. Chapter 6 discusses opportunities in computational chemical science and computational materials science.
Molecular dynamics simulations are capable of addressing the self-assembly process at a rudimentary, but often impressive, level. These calculations can be used to study the secondary structure (and some tertiary structure) of large complex molecules. Present computers and codes can handle massive calculations but cannot eliminate concerns that boundary conditions may affect the result. Eventually, continued improvements in computer hardware will provide this added capacity in serial computers; development of parallel computer codes is likely to accomplish the goal more quickly. In addition, the development of realistic, time-efficient potentials will accelerate the useful application of dynamic simulation to the self-assembly process. In addition, principles are needed to guide the selec-
tion of initial configurations; the proper initial conditions accelerate convergence and minimize problems associated with metastable configurations.
Molecular calculations provide approaches to supramolecular structure and to the dynamics of self-assembly by extending atomic-molecular physics. Alternatively, the tools of finite element analysis can be used to approach the simulation of self-assembled film properties. The voxel4 size in finite element analysis needs be small compared to significant variation in structure-property relationships; for self-assembled structures, this implies use of voxels of nanometer dimensions. However, the continuum constitutive relationships utilized for macroscopic-system calculations will be difficult to extend at this scale because nanostructure properties are expected to differ from microstructural properties. In addition, in structures with a high density of boundaries (such as thin multilayer films), poorly understood boundary conditions may contribute to inaccuracies.
Image analysis is an important aspect of many areas of science and engineering, and imaging will play an important role in characterizing self-assembled structures as well as in on-line process control. Development of effective noise identification and suppression, contrast enhancements, visualization, pattern recognition, and correlation algorithms should be co-opted where possible and adapted to the analysis of self-assembled structures.
Models of the self-assembly process also will be important. Because self-assembled structures can be diverse, those models are likely to be highly complex. Sensitivity analysis can be an important approach to the identification and control of critical parameters.
Adaptive and Responsive Materials
An area of great promise for the future is that of materials for which properties change in response to external influences. The most prominent is giant magnetoresistive (GMR) materials—materials for which the electrical conductivity can change by a few percent under the application of an external magnetic field. GMR materials moved from a laboratory curiosity to the dominant technology in computer memories within a decade—a startling example of how a new material can completely change a major industry. The invention of new materials has been an area in which the United States has played a central role; the current emphasis on focused, shorter-term projects has, however, made it very difficult to support a lively activity in new materials, which by definition are far from a final product.
Among the adaptive materials currently on the forefront are liquid crystals, electro- and magneto-rheological (ER/MR) fluids, thermo- and physioreponsive gels, and shape memory alloys. In all but the last case, these systems involve
fluid-solid interfaces, complex or dispersed suspensions, and external fields. Thus, all of the directions outlined above for self-assembly and interfaces come into play when pursuing these “smart fluids.” Improvements in synthesis and spatial resolution of the external fields through soft-lithography (self-assembled monolayers, etc.) will provide new capabilities of control and tuning of these complex fluids.
Tools, Resources, and Infrastructures
The area of complex condensed matter depends crucially on the availability of appropriate tools for both fabrication and characterization. These tools are of “intermediate” size: they are neither a test tube nor a synchrotron. Typical tools— scanning probe microscopes, x-ray photoelectron spectrometers, electron microscopes, clean rooms—cost from $0.1 million to $5 million. They are shared-use facilities, but they must be local to the user group—travel to distance facilities for routine measurements is not practical.
Simulations in this area also require access to high-level computational capabilities. By definition, simulations involve both large numbers of atoms and dynamic behavior; both consume large numbers of CPU cycles. Because the field is so demanding on computer time, analytical theory is very important, even if it yields only approximate solutions.
Many of the tools required for the nanoscience revolution in materials involve sophisticated experiments requiring leadership, training, and infrastructure. User facilities such as high-resolution electron microscopes, synchrotrons and NMR facilities need to be easily accessible and provide adequate support for efficient experimentation.
The nanometer- to micrometer-scale dimensions of supramolecular assemblies present many challenges to rigorous compositional and structural characterization. Development of adequate structure-property relationships for these complex hierarchical systems will require improved measurement methods and techniques. The following areas constitute critical thrusts in instrument development.
-
Many techniques ideally suited for nanostructure characterization unfortunately depend also on the substrate properties. For example, the reflectivity and conductivity of a substrate play an important role in the successful execution of the instrumental method. Hence, substrate-independent techniques are needed so that structure and/or behavior of the material can be investigated in a confined geometry, decoupled from the potentially invasive effect of the substrate-material interface.
-
Feedback provided by on-line monitoring of self-assembling processes will play an increasingly important role in controlling the microscopic and macroscopic architecture of molecular assemblies. Successful adaptation of char-
-
acterization methods for noninvasive in situ monitoring will require miniaturization of many of the analytical devices commonly used for interrogating self-assembled structures. The revolution in microscopic identification and control provides new opportunities for chemists and chemical engineers. Promising routes to achieve this goal include the use of optical fibers in spectroscopic methods and the development of MEMS-based analytical instrumentation.
-
Order and polydispersity are key parameters that characterize many self-assembled systems. However, accurate measurement of particle sizes in concentrated solution-phase systems, and determination of crystallinity for thin-film systems, remain problematic. While inverse methods such as scattering and diffraction provide measures of these properties, often the physical information derived from such data is ambiguous and model dependent. Hence development of improved theory and data analysis methods for extracting real-space information from inverse methods is a priority.
-
Use of diverse techniques provides significant structural information in dispersed and thin-film systems. If significant advances in source intensities (e.g., higher neutron and x-ray fluxes, higher power diode lasers) and photon detector technology (e.g., high quantum efficiency, large bandwidth charge-coupled devices, CCDs) are achieved in the near term, these methods could be extended readily to studies of nanoscale features. Since the experimental and theoretical frameworks for these techniques are well established, extension to smaller length scales should be straightforward.
-
The already critical need for molecular-scale compositional mapping will increase as more complex structures are assembled. Currently, electron microscopy, scanning probe microscopy (SPM) and fluorescence resonance energy transfer (FRET) are the only methods that routinely provide nanometer resolution.
In contrast to the mature instrumental techniques discussed above, a hitherto nonexistent class of techniques will require substantial development effort. The new instruments will be capable of measuring the thermal (e.g., glass transition temperatures for amorphous or semicrystalline polymers and melting temperatures for materials in the crystalline phase), chemical, and mechanical (e.g., viscoelastic) properties of nanoscale films in confined geometries, and their creation will require rethinking of conventional methods that are used for bulk measurements.
WHY ALL THIS IS IMPORTANT
Materials science and engineering is inseparable from chemistry and chemical engineering. The importance of materials is illustrated by the effects they have on the quality of human life—underscored by the way our society uses new
materials and the technology they spawn to define epochs in history. The list of the 20 greatest engineering achievements of the 20th century, compiled in 2000 by the National Academy of Engineering,5 contains many entries (for example, high-performance materials, automobiles, airplanes, electronics, computers, telephones, and fiber optics) that depend essentially on advances in materials science and engineering. From synthesis to processing to commodity manufacturing of materials, the tools of chemical science and engineering will be essential to defining the next century in these terms. New materials with predictable properties will provide worthy and formidable targets for design and synthesis, while processing and manufacturing these new materials will present significant challenging new objectives for chemical process-systems engineering.
The science of materials reflects the way that chemistry has changed—from a field concerned only with atoms and molecules and their properties in isolation, to a field increasingly concerned with organized interactive systems. This change opens opportunities and challenges in fundamental science that will let us ask not just: What are the chemical components of that stuff? We will be asking as well: How do those components interact to produce the properties of that stuff? The answers to such questions will greatly add to our understanding of the chemical universe.


























