2
Discovery
INTRODUCTION
Development of new materials has followed a number of different pathways, depending on both the nature of the problem being pursued and the means of investigation. Breakthroughs in the discovery of new materials have ranged from pure serendipity, to trial-and-error approaches, to design by analogy to existing systems. These methodologies will remain important in the development of materials but as the challenges and requirements for new materials become more complex, the need to design and develop new materials from the molecular scale through the macroscopic final product will become increasingly important. The use of molecular modeling and the engineering of new materials into useable forms or devices are of particular importance.
BREAKTHROUGHS IN MATERIALS DEVELOPMENT
The chemical sciences have made great strides over the past several decades in the development of novel and useful materials. Although the following is not meant to be an exhaustive list of such breakthroughs, these examples point to the range of materials and their applications.
Polymers
Examples such as Teflon serve to show how the chemical sciences have contributed indispensable materials to everyday use. More recently, the development of thermoplastics and/or structural polymers has had an increasing influ
ence on applications ranging from construction to national defense. New paints and coatings, clothing fibers, and photographic films have all benefited from the development of new materials.
There are newer polymeric materials whose commercial impact has yet to be realized. Work on semiconductive and conductive polymers have made great strides, but further work is necessary. Synthesis of amphiphilic dendritic block copolymers that are designed to form ultrathin organic films have also had major advances, but these materials also need further development. Other promising materials, from polymers for drug delivery to tissue engineering, have the potential to benefit the biomedical field but are still in a relatively early stage of development.
Catalysis
Advances in new materials for catalysis cover a wide range of applications. Zeolites and pillared clays have had a huge impact on the petroleum industry. New zeolites with specified properties continue to be developed with various utilities.
Ziegler-Natta catalysts allow the preparation of billions of pounds per year of organic polymers with controlled molecular structures and useful material properties. This method is also useful because it allows the synthesis of polymers that cannot be produced in a practical manner by any other method. Some examples of these are linear unbranched polyethylene and isotactic polypropylene.
In the energy and transportation sector, catalysis has been an especially fruitful area of research. As a result, supported gold catalysts have been developed. In addition, selective oxidation of carbon monoxide has been achieved and a gold-transition-metal oxide has been developed that provides very active NOx reduction as well as hydrocarbon oxidation. Perhaps no more ubiquitous an example of novel catalysis exists than the catalytic converter, which contains a porous ceramic coating embedded with palladium and rhodium. The platinum particles serve to complete the oxidation of hydrocarbons and carbon monoxide to carbon dioxide, while rhodium converts nitrogen oxides to nitrogen and oxygen.
Another important breakthrough in this field includes the development of metallocene catalysts, which are expected to revolutionize the polyethylene and polypropylene markets. The use of supramolecular organic templates containing appropriate surface functionalities to regulate the nucleation and growth of inorganic magnets, semiconductors, and catalysts is significant as well .
Electronics
This broad category has benefited from many breakthroughs in the development of new materials. Perhaps no recent advance has had a greater impact in this area than the creation of chemically amplified photoresist. Photoresist, resins con
taining photochemically active polymers, can be coated on a wafer and irradiated using photons (photolithography), electrons (electron-beam lithography), or X-rays (X-ray lithography). These developments have had considerable impact on computer chip production.
In the field of telecommunications, high-temperature superconductors and ceramic materials containing copper-oxide planes have potential uses in communications shielding. However, the commercial impact of these materials has yet to be demonstrated.
Instrumentation
The development of new instrumentation is essential both in characterizing materials and in exploring their potential applications. Scanning probe microscopes have enabled greater understanding of interfacial phenomena and are particularly important as work on new materials progresses on the nanoscale. Matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF), a mass spectrometry technique that uses laser light to ablate unfragmented polymer molecules mixed with an organic acid matrix into a time-of-flight mass spectrometer, is finding increasing application in the polymer community. Likewise, induction by coupled plasma mass spectroscopy has become a pervasive tool for analysis at materials interfaces.
METHODS OF DISCOVERY
As the areas and applications of materials discovery continue to expand, so too does methodology. The fields of materials chemistry and engineering are drawing on a wide range of resources to develop new compounds and applications.
Design by Analogy
Design by analogy is perhaps the most common method for producing new materials. As the range of available materials continues to grow and the community fully explores the behavior of existing materials, design by analogy will continue to be productive. Research into the development of new inorganic nanoscale materials by analogy with biological systems is just one example of this approach. For example, work in sensor design is progressing on “synthetic cells” that couple reactions much in the same way that living cells utilize metabolite channeling to localize reactions.
Chemistry and materials manufacturing technologies will be required to make structures of increasing complexity, hierarchically organized, and with a precision greater than what is possible today. Biological synthesis of such materials may hold the key to new routes that might be utilized for future technologies
(Sidebar 2.1). For example, the silica shell of a diatom illustrates the complexity of materials synthesis that biological systems can achieve.
Syntheses such as the silica shell of a diatom are remarkable in at least three respects. First, the precision with which the nanoscale structure of these biological materials is formed in many cases exceeds that of present human engineering. Second, the conditions under which these syntheses occur are very mild. These are physiological, low-temperature, ambient pressure processes occurring at neutral pH without the use of caustic chemicals. This is in marked contrast to human manufacturing of silica materials. Synthesis occurs on an enormously large scale, approaching gigatons of silica shells annually. Formation of these materials occurs at temperatures close to 0°C in the polar seas, indicating the presence of unique mechanisms of synthesis. The challenge for scientists is to identify these mechanisms in order to utilize their advantages.
Computer-Assisted Materials Design
A key goal in the development of any new material is to be able to design and construct materials with desirable and predictable properties. In order to meet this goal, researchers seek to design these new materials at the molecular and atomic level. Increasingly, this capability depends on materials design facilitated by computer modeling and simulation. The increase in supercomputing calculating power allows researchers to move design of materials properties from the macroscopic to the quantum-mechanical level.
It is possible to determine the properties of new materials by utilizing density functional theory. As computing power has increased, density functional theory— first postulated in the 1960s—has had an enormous impact on the design of new materials. Greater computing power combined with greater accuracy, now makes it possible to understand complex systems at the molecular level. For example, density functional theory combined with a self-consistent field theory for polymers has been applied to reveal new self-assemblies in which both particles and polymers organize into mesoscopically regular patterns. In particular, researchers have begun to delineate conditions in which polymer nanostructures can drive filler particles to self-assemble into nearly continuous nanowires.1 Computer modeling will continue to have a large impact on both chemical synthesis and theoretical applications as computational power at the desktop level increases.
Combinatorial Synthesis and High-Throughput Screening
Since most processes for synthesis by analogy remain imprecise, high-throughput methods offer another promising alternative to conventional cycles of
|
SIDEBAR 2.1 Nanostructured Biological Materials Reveal New Routes to Synthesisa The mother-of-pearl of the abalone shell, composed of the microlaminate of nacre, is a high-performance composite with a fracture toughness 3,000 times greater than that of the mineral alone. While its structure is only about 1 percent protein by weight, these proteins are responsible for the control of the structure and thus its high performance.b Studying this material presents significant challenges because to get the proteins it is necessary to literally pull biomolecules out of a rock (the limestone of the shell), usually with low yields. About 50 different proteins exist in the mineral composite of nacre. Some of these proteins lie between crystals, and many of them are occluded within crystals. Extraction of occluded proteins from the calcite, which forms a primer layer, and the aragonite that grows upon it shows that these are different families of proteins. In reconstruction experiments in vitro, it was shown that the proteins determine both the nanostructures and the hierarchical organization of the two different mineral layers. The key is to isolate enough of the protein to analyze part of its structure. Once this is accomplished, it becomes possible to clone the DNA that codes for the protein. The protein is then produced by genetic engineering with E. coli bacteria. With much more of the protein in hand, it is possible to study the mechanism of synthesis of the microlaminate. The results from this approach led to a break from the paradigm that had governed the field for a number of years. The prevailing model was that a self-assembling organic template first serves as the surface upon which nucleation of the crystals takes place and that this process is simply repeated many times. Investigators learned that although such a process could make an artificial microlaminate, it does not produce the remarkable enhancement of fracture toughness found in the natural material. Organization of the proteins and minerals forming the mother of pearl of the abalone shell in nature proceeds via multiple mechanisms operating to control structure and assembly, each working at a different length scale. These mechanisms operate contemporaneously. Small proteins interact with the growing crystals by specifically binding to certain step-edges or crystal faces. This determines polymorph selection, atomic lattice orientation, and morphology of the crystals. An unanticipated mechanism of molecular stenciling guides the growth of the crystals from one layer to the next to generate the microlaminate structure. Research into the biosynthesis of these materials indicates that this process is quite complex, far beyond a simple linear synthesis. A genetic switch between one polymorph and the other governs one of the key steps. This switch turns off the synthesis of one family of proteins that |
 FIGURE 2.1 Crystal lattices of aragonite. directs the synthesis of calcite and turns on the synthesis of a second family of proteins that synthesize aragonite. A very different and even more complex mechanism is responsible for the hierarchical organization of the crystal plates. The crystal lattices of aragonite grow in the form of tapered conical stacks of flat plates, resembling the tapered and layered shapes of pagodas (Figure 2.1). Atomic force microscopy revealed that each stack consists of a single crystal, even though protein sheets apparently separate the layers from which it is made. The underlying conclusion is that the protein sheets must be fenestrated, providing a path for communication and growth of the crystals through these sheets. Further work subsequently confirmed that there are nanopores in the protein sheets separating the crystal layers and that these act like molecular stencils. Because the pores are distributed randomly, the growth of the crystals from one layer to the next is randomly offset, which generates the interdigitation of the crystals that contributes to the remarkably enhanced fracture toughness. Elucidation of these mechanisms relied extensively on the development of advanced instrumentation. Six different inventions that modified and improved the capabilities of atomic force scanning probe microscopy were required to successfully solve this system. |
design, evaluation, and redesign. Work in the chemical sciences on the development of new materials is now integrating methodology developed in the pharmaceutical industry. Specifically, materials companies are beginning to use combinatorial synthesis in their research (Sidebar 2.2).
By creating large collections of chemically diverse materials, called libraries, and screening these libraries and materials simultaneously for desirable performance, enormous quantities of information regarding reaction conditions and potential target materials can be generated at a vastly accelerated pace. The challenges of implementing such methods include creating screens for target properties and developing software for interpretation of the data streams that emerge from high-throughput experimental programs.
Reaction scale is a further advantage of the combinatorial process. As opposed to traditional multi-gram reactors, combinatorial reactions can be performed at milligram concentrations. This enables large numbers of experiments to be performed with minimal use of reagents, lab space, and waste generation. This methodology does, however, require diversity of the substructure of reactants and a wide variety of synthetic routes. The importance of process conditions must be considered. In addition, an efficient automation screening and data storage analysis system must be in place.
SUMMARY AND FINDINGS
The roles of “needs-driven” and “discovery-driven” research on materials in the chemical sciences will always be an important consideration. Although it may seem that “materials discovery” must lie in the latter area by definition, there is considerable room in the fields of chemistry and chemical engineering for discovery of materials that have clear links to future technologies. The continued health of materials research will require fundamentally new insights into the behavior of condensed matter (whether or not applications are identified at the outset), as well as developments driven by clearly articulated technological and market needs. Important research programs can mix these two kinds of objectives in many different ways. This broad spectrum of activities is most likely to lead to breakthroughs.
Finding: Materials discovery occurs via many routes. A diverse portfolio of research efforts directed toward the discovery and development of new materials systems is likely to produce significant advances in the field. Renewed and expanded emphasis on synthesis, catalysis, and processing methods will be essential to continuing advances for new materials.
The chemical sciences are distinguished by their emphasis on synthesis. Synthetic chemistry may claim a critical role in the development of new materials and in the creation of entirely new classes of materials. Realization of the techno
logical impact of early advances also required skills in areas such as processing and analysis. Highly efficient and selective catalytic transformations are especially promising. As chemical synthesis of new materials proceeds into the twenty-first century, new methodologies will be adapted and incorporated into the complement of synthetic approaches.
As the chemical sciences begin to expand their use of high-throughput screening for the development of new materials, it is likely that researchers will look to the lessons learned in the pharmaceutical industry in the use of combinatorial synthesis. Discovery efforts must therefore encompass not only new products, but new processes as well.
Finding: Recent developments in parallel synthesis and high-throughput screening of materials offer substantial promise as adjuncts or alternatives to more traditional programs of design, evaluation, and redesign.
Instrumentation has always played an important role in materials research. New tools for fabrication and analysis will continue to move the field forward. However, changes in instrumentation needs are to be expected, driven in part by increased emphasis on nanoscale science and engineering and partly by developments in neighboring fields such as microfluidics and robotics. The materials chemistry and chemical engineering community must position itself to contribute to these developments and to exploit such advances in the discovery and refinement of materials and materials systems. Continuing developments in areas such as scanning tunneling microscopy and atomic force microscopy are examples of instrumentation that will have an impact on nanotechnology research and development (see Sidebar 5.1 for others).
Finding: Exploitation of high-throughput experimental designs will require development of new instrumentation, new methods for analysis of high-volume data streams, and testing of key properties on very small amounts of material.
|
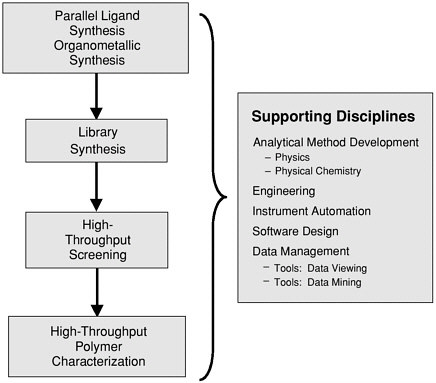
SIDEBAR 2.2 Combinatorial Materials Synthesis: Homogeneous Catalysisa A homogeneous catalyst consists of a metal center with stabilizing ancillary ligands, usually organic ligands, and a vacant coordination site at which the reaction takes place. Performance properties of the catalyst site can be varied dramatically depending on the choice of ligands, the choice of metal, the process conditions, the activation conditions, and the conditions under which the reaction is run. Within the field of polyolefin catalysis, it is currently not possible to predict which new metal-ligand combinations will lead to active catalyst classes and which will have low or no activity. Even for a well-studied catalyst, small changes in the ligand structure can have dramatic and unpredictable effects on catalyst performance. Olefin polymerization catalysts therefore represent good candidates for the application of high-throughput approaches. An integrated high-throughput workflow for the discovery of new catalysts offers enormous advantages over conventional methods of catalyst discovery. A high-throughput screen can identify catalytically active systems rapidly and also identify and reject inactive systems, which is important. Additionally, the ability to generate sufficient quantities of meaningful catalyst performance data can provide comprehensive structure-property relationships. Conventionally, a Ph.D.-level researcher and a technician can only perform one experiment in the morning and one experiment in the afternoon. Using high-throughput methods, a primary screen can typically run a thousand polymerization experiments a day with four people to perform the screening. In the secondary screening, typically a hundred experiments are performed a day. A high-throughput program to discover new polyolefin catalysts requires large numbers of ligands that express suitably diverse electronic and steric properties as well as efficient methods of attaching the ligand to the metal to form the catalyst candidates. Furthermore, efficient activation methods, suitable rapid screening techniques, efficient automation, and a data storage and analysis system are also required (see Figure 2.2). The number of experiments rapidly increases. For instance, consider the situation with 6,000 ligands in the ligand archive and 40 metal precursors. With five complexation routes, five activation conditions, and three process conditions, the number of experiments approaches 20 million! The high-throughput workflow includes the design of libraries using custom-made computer software, automated delivery of metal precursors and ligands into the reactors using a liquid-handling robot, and a rapid primary screen that serves to assess the potential of each metal |