3
Movement Control in the Older Adult
Caroline J. Ketcham and George E. Stelmach
INTRODUCTION
The control of movements is a complex interaction of cognitive and sensorimotor systems. Researchers in movement science aim to understand how an action is produced and what mechanisms are involved in regulating the movement. Motor control declines in older adults include changes in both the peripheral and the central nervous system, which lead to an array of behavioral decrements (Salthouse, 1985; Welford, 1977; Ketcham and Stelmach, 2002). It is well known that as adults age, the execution of movement becomes slow and more variable, and there is emerging evidence that the microstructure of the movement also changes. In this chapter we document most of the major changes that occur in the control and coordination of movement with respect to aging. In the studies reviewed, older adults are classified as over 60 years and are compared with young adults typically between 18 and 30 years of age in a cross-sectional manner (see Schaie, this volume, for a methodological description). Results reported are means derived from age-group comparisons and do not address individual differences.
The review begins with a discussion of processing speed defined by reaction time and presents differences between young and older adults on simple and complex tasks. The following topics include changes that occur in older adults related to the control of movement including: reduced movement speed, movement composition differences, increased variability, reduced force control, and coordination difficulties. Subsequently highlighted are some of the possible sensorimotor changes that
may contribute to slower, more-variable movements and reduced strength observed in older adults. Changes in posture and balance are then discussed, as a stable base of support is necessary to execute precise motor skills as well as being important for mobility of older adults. Finally, an overview of motor learning research as well as a discussion of improvements in motor function with generalized and specific training programs are presented. As is apparent, changes in control and coordination of movement significantly affect the type of activities that older adults can efficiently perform and often determine whether they can live independently. Thus, those involved in enhancing the performance capabilities of these individuals need to have a good understanding of how the aging processes diminish motor performance.
RESPONSE INITIATION
Reaction time is defined as the time required to initiate a movement response following a visual, auditory, or other sensory signal and is thought to reflect the speed of transmission of the central nervous system (Stelmach and Goggin, 1988). Experiments are conducted to measure the time it takes to initiate a response when an imperative stimulus is presented. The imperative stimulus is usually visual, but may be auditory or tactile. Such reactions can be to a single stimulus, multiple stimuli, or may include incompatible responses. In a simple reaction-time task, where one stimulus is given and one response is required, it has been demonstrated that reaction time increases in range from 0.5 ms/yr (5 ms/decade) (Fozard, Vercryssen, Reynolds, Hancock, and Quilter, 1994) to 2 ms/decade (Gottsdanker, 1982). It has been widely shown in the research that the speed of processing information decreases (i.e., the time increases) with advanced age on the order of 26 percent (264 ms in the young—20 years old—versus 327 ms in older adults—60 years old) (Welford, 1984). Similar findings have been reported for auditory and tactile simple reaction times as well (Redfern, Muller, Jennings, and Furman, 2002; Walhovd and Fjell, 2001; Liu, 2001; Walker, Alicandri, Sedney, and Roberts, 1991). This approximately 50-ms increase in simple reaction times is consistent across studies that have examined such changes across the life span (Fozard et al., 1994) as well as those that compare groups of young and older adults on the same reaction-time tasks (Amrhein, Stelmach, and Goggin, 1991; Walker, Philbin, and Fisk, 1997; Stelmach and Goggin, 1988; Cerella, 1985; Cerella, Poon, and Williams, 1980; Bashore, Ridderinkhof, and van der Molen, 1997; Gottsdanker, 1982; Stelmach and Goggin, 1988). See Schaie (in this volume) for a similar discussion of response speed with respect to cognitive changes that occur with advanced age.
The slowing of processing speed in older adults is greater in tasks that require more complicated processing to initiate the appropriate response (Amrhein et al., 1991; Goggin and Stelmach, 1990; Larish and Stelmach, 1982; Stelmach, Goggin, and Garcia-Colera, 1987; Diggles-Buckles and Vercruyssen, 1990; Simon, 1967; Welford, 1977; Bashore et al., 1997; Cerella, 1985; Cerella et al., 1980; Fozard et al., 1994; Melis, Soetens, and van der Molen, 2002; Gottsdanker, 1982; Stelmach and Goggin, 1988). In a choice reaction-time task, subjects are required to select the appropriate response that corresponds to a specific stimulus. These reaction times are typically longer than in simple reaction-time tasks as they include an additional element of selecting the appropriate response. Older adults are 30-60 percent (50–500 ms) slower than young adults in reaction-time tasks with two to four choices (Amrhein et al., 1991; Simon, 1967; Welford, 1984; Jordan and Rabbitt, 1977; Stelmach and Goggin, 1988). Choice reaction time in older adults has been found to increase by 1.6 ms/yr and is amplified as the number of choices increases (Fozard et al., 1994). For example, in a two-, four-, and seven-choice reaction-time task, older adults were 39, 40, and 45 percent slower than young adults, respectively. Furthermore, older adults respond similarly to young adults: When the number of response choices increases, reaction time increases (Hick, 1952); however, the delays in responding are more substantial with multiple response choices.
Some researchers have sought to decompose reaction time into premotor and motor time. Premotor time is defined as the time from the presentation of the stimulus until the onset of muscle activity and is thought to reflect cognitive processes, whereas motor time is the time from muscle activation to the beginning of the movement and reflects efficiency of the motor system. These studies have shown that most of the response delays in older adults are accounted for in the premotor or cognitive period (Clarkson, 1978; Hart, 1980; Spirduso, 1995). Further studies have decomposed the premotor cognitive processes into the time it takes to detect, prepare, and initiate an appropriate response. The majority of these studies have shown that the time utilized for each of these elementary components is prolonged equally in older adults (Simon and Pouraghabagher, 1978; Gottsdanker, 1982; Stelmach and Goggin, 1988; Stelmach et al., 1987, 1988). Collectively, the literature on response speed documents delayed initiation of a response in older adults compared with young adults across an array of simple and complex tasks.
MOVEMENT CONTROL DECREMENTS
Movement Duration
Movement duration is defined as the time from the initiation of the movement to the termination of the movement (Birren, 1974; Salthouse,
1985). Movement time is increased in older adults for a variety of tasks including point-to-point movements (Amrhein et al., 1991; Cerella, 1985; Cooke, Brown, and Cunningham, 1989; Ketcham, Seidler, Van Gemmert, and Stelmach, 2002; Goggin and Meeuwsen, 1992), reaching and grasping movements (Carnahan, Vandervoort, and Swanson, 1998; Bennett and Castiello, 1994), handwriting (Amrhein and Theios, 1993; Dixon, Kurzman, and Friesen, 1993; Contreras-Vidal, Teulings, and Stelmach, 1998), and continuous movements (Greene and Williams, 1996; Pohl, Winstein, and Fisher, 1996; Wishart, Lee, Murdoch, and Hodges, 2000; Ketcham, Dounskaia, and Stelmach, 2001). Movement durations are on the order of 30-60 percent (50-90 ms) longer in older adults compared with young adults in tasks ranging from simple to complex (Welford, 1977); in extreme cases, slowing has been reported as great as 69 percent (421 ms compared with 132 ms in young adults) in a point-to-point movement (Stelmach et al., 1988). Although movement time is an important measure of how the motor system is performing, the effects observed vary greatly depending on the task.
One common approach to assessing movement slowing is to manipulate task difficulty (information to be processed) in a stepwise fashion. Fitts′s law, a well-studied law in motor control research (Fitts, 1954), states that, as the difficulty of the movement increases, the speed of the movement decreases. A typical task would require the subject to move a hand as quickly as possible from a starting position to touch a target with a stylus when a “go” signal is given. The size of the target and the distance from the starting point to the target can be varied. The index of difficulty (ID) is greater for smaller targets and for longer movements. Research has shown that, in such tasks, older adults tend to move slower than young adults at all levels of difficulty but are differentially slower at higher levels of difficulty (Bashore, Osman, and Heffley, 1989; Goggin and Meeuwsen, 1992; Hines, 1979; Ketcham et al., 2002; Salthouse, 1988; Pohl et al., 1996; Walker et al., 1997; Brogmus, 1991; Fozard et al., 1994). For example, Ketcham and colleagues (2002) reported movement durations of a low ID to be 333 and 642 ms in young and older adults, respectively. At the higher ID, movement time of young adults was on average 717 ms compared with 1304 ms for older adults. Pohl and colleagues (1996) reported similar differences on a continuous movement task. Differences in movement times between young and older adults were amplified as task difficulty increased with an 80-ms time difference at the high ID compared with a 29-ms difference at the low ID. Task difficulty according to Fitts′s law can be manipulated in two ways: by a change in either the target size or the distance between the start and the end of the movement. When target size and movement distance are manipulated separately, researchers have demonstrated that older compared with younger adults
are more affected by increases in movement amplitude (a change from 9.6 to 19.2 cm resulted in a 108-ms increase in movement time for young adults versus 293 ms for older adults) but not by decreases in target size (Ketcham et al., 2002; Goggin and Meeuwsen, 1992). Some authors have speculated that these effects are caused by the reduced ability of older adults to produce and maintain forces across the entire spectrum of the movement (Ketcham et al., 2002; Galganski, Fuglevand, and Enoka, 1993; Darling, Cooke, and Brown, 1989), which may have real-world implications on a variety of precision aiming tasks.
Movement Components
Modern data-acquisition techniques make it possible to record and reconstruct movements in real time, which permit investigators to decompose a movement trajectory to gain information on how a movement is controlled and coordinated. Trajectory profiles are processed to yield velocity and acceleration profiles, which are further decomposed into acceleration and deceleration phases as well as parsed into movement substructures. Experiments that have employed these kinematic analyses have provided insights into how the movements produced by older adults differ from those of young adults (Slavin, Phillips, and Bradshaw, 1996). It has been shown that the velocity profiles of young adults are typically bell shaped, where the acceleration phase equals the deceleration phase. In studies that have examined trajectory profiles of young and older adults, it has been observed that for older adults the trajectories are asymmetrical with a longer deceleration phase (Ketcham et al., 2002; Bennett and Castiello, 1994; Brown, 1996; Cooke et al., 1989; Darling et al., 1989; Goggin and Stelmach, 1990; Marteniuk, MacKenzie, Jeannerod, Athenes, and Dugas, 1987; Pratt, Chasteen, and Abrams, 1994).
The deceleration phase has been suggested to contain the portion of movement that is under corrective control because there is sufficient time for sensory feedback to be processed and implemented into the control of the terminal phase of the movement. The deceleration phase in older adults is on the order of 20-40 percent longer than that of young adults (Brown, 1996; Cooke et al., 1989; Pratt et al., 1994; Bennett and Castiello, 1994; Morgan et al., 1994).
In addition to longer deceleration phases, older adults produce movements with 30-70 percent lower peak velocity compared with young adults (Ketcham et al., 2002; Bellgrove, Phillips, Bradshaw, and Gallucci, 1998; Cooke et al., 1989; Goggin and Meeuwsen, 1992; Pratt et al., 1994) (see Figure 3-1). Furthermore, when movement distance increases, older adults do not increase the velocity of their movements to the same degree as young adults (Ketcham et al., 2002; Gutman, Latash, Almeida,
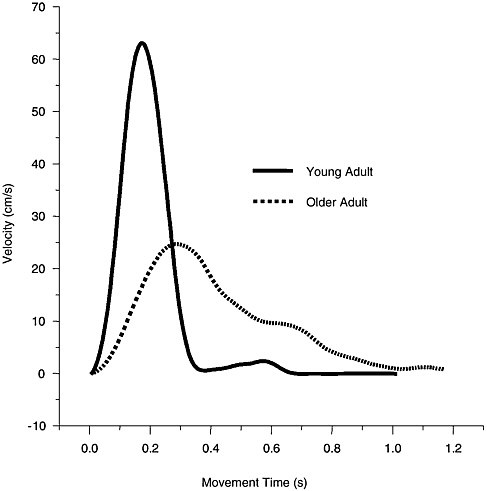
FIGURE 3-1 Example velocity profiles for an older and a young adult on a point-to-point aiming task.
SOURCE: Adapted from Ketcham et al. (2002, p. 56).
and Gottlieb, 1993). For example, Ketcham and colleagues (2002) found that the peak velocity of a shorter-distance movement was 15.9 cm/s in older adults and 29 cm/s in young adults. When movement distance was increased from 9.6 to 19.2 cm, the peak velocity of older adults was 27.6 cm/s whereas for young adults it was 48 cm/s.
Acceleration profiles can be partitioned into movement substructures (primary and secondary submovements) for a more in-depth analysis. The movement optimization model (Meyer, Abrams, Kornblum, Wright, and Smith, 1988) maintains that the primary submovement represents the portion of the movement under preplanned control where the limb is
propelled to the target during the acceleration phase, whereas the secondary submovement represents the feedback-controlled portion of the movement. The closer to the target the primary submovement ends, the more efficient the motor system is thought to be (Meyer et al., 1988). Overall, research has demonstrated that older adults cover 10-70 percent less distance with their primary submovement compared with young adults, depending on the task (Bellgrove et al., 1998; Darling et al., 1989; Hsu, Huang, Tsuang, and Sun, 1997; Ketcham et al., 2002; Pratt et al., 1994; Walker et al., 1997; Romero, Van Gemmert, Adler, Bekkering, and Stelmach, 2003; Seidler-Dobrin, He, and Stelmach, 1998). Pratt and colleagues (1994) found that older adults covered 50 percent of the distance to the target with the primary submovement compared with young adults who traveled 70 percent of the distance (Figure 3-2). Because the primary submovement ends further from the movement end point, older adults need to make one or more adjustments with the secondary submovement to complete the movement accurately (Goggin and Meeuwsen, 1992; Hsu et al., 1997; Ketcham et al., 2002; Pohl et al., 1996; Pratt et al., 1994; Seidler-Dobrin and Stelmach, 1998; Walker et al., 1997).
Researchers have extended the use of substructure analysis to assess how young and older adults differ in improving their movements with practice. Pratt et al. (1994) and Seidler-Dobrin and Stelmach (1998) demonstrated that both groups improved their movement times with practice, but they did it quite differently. Older adults only slightly increased (50 to
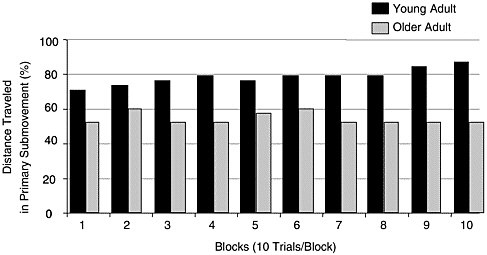
FIGURE 3-2 Percentage of distance traveled in the primary submovement for older and young adults in a point-to-point aiming task over 10 blocks of 10 trials.
SOURCE: Adapted from Pratt et al. (1994, p. 360).
51 percent) the portion of the movement covered with the primary submovement with 100 trials of practice whereas young adults increased the distance covered from 67 to 75 percent. These data, along with other studies that have used kinematic and movement subparsing techniques, have shown that the initial phases of the movement are similar in young and older adults, with older adults producing movements with lower peak velocity outputs. Conversely, these methods have shown marked differences in the terminal phase of the movement, measured by the deceleration phase, proportion of the movement covered in the primary submovement, and the subsequent secondary submovements, suggesting that older adults need to make corrective adjustments to their movement as they approach the target.
Movement Variability
Movement variability refers to an individual′s overall consistency of an executed task across trials. Increased variability may reflect decrements in the motor system in its ability to produce the same movement output repeatedly. There are two types of movement variability: variability of the end point and variability of the components of the movement trajectory.
Over a wide variety of tasks, researchers report higher variability in the trajectory and end-point position of movements of older adults compared with young adults overall and when performance is examined in a more detailed trial-by-trial basis in a rapid aiming task (Brown, 1996; Cooke et al., 1989; Greene and Williams, 1996; Seidler-Dobrin et al., 1998; Ketcham et al., 2002; Darling et al., 1989; Welford, 1984; Abrams, Pratt, and Chasteen, 1998; Warabi, Kase, and Kato, 1984; Tedeschi et al., 1989). Walker et al. (1997) have shown that older adults have higher variability of end-point of their first submovement compared with young adults. For both young and older adults, as acceleration increased, the variability of end-point position also increased—however, at a significantly greater rate for older adults. Pratt and colleagues (1994) documented that older adults had higher end-point variability than young adults. Both young and older adults showed decreased end-point variability after extended practice; however, older adults did not improve as much as young adults.
In addition to end-point analyses, researchers also have examined the variability of the movement trajectory using kinematic analysis techniques. Cooke and colleagues (1989) found that older adults were significantly more variable compared with young adults on measures including movement duration, peak velocity, and the acceleration/deceleration ratio. Furthermore, the variability of acceleration and deceleration increased differentially for older adults compared with young adults as the ampli-
tude of the movement increased (Darling et al., 1989). Pratt et al. (1994) found that older adults showed higher variability in the distance traveled in the primary submovement that did not improve as much as young adults with extensive practice. It has been suggested that the irregularity of the amplitude and timing of muscle output in older adults is responsible for this overall increased variability in the trajectory of movements as well as variability of end-point position (Darling et al., 1989; Cooke et al., 1989; Brown, 1996; Goggin and Meeuwsen, 1992; Ketcham et al., 2002; Greene and Williams, 1996).
Variability of executed movements on a moment-to-moment basis has large implications for daily activities of older adults. For example, if the motor system is quite variable, it is difficult to know whether you may knock over a glass when you reach for it. If you know you will always undershoot the glass, then you can plan for, prepare for, and compensate for that decrement.
Speed and Accuracy
Movements made to functional targets have a known speed-accuracy relationship. As individuals attempt to move faster, there is a point where the response accuracy is compromised. Individuals, based on their ability, often have different speed-accuracy behavioral patterns. The literature has shown that the reaction time and movement time of older adults are slower than those of young adults (see “Response Initiation” and “Movement Duration” above). One common observation of those investigators who have made cross-sectional comparisons is that older adults have a bias for accuracy at the expense of speed (Salthouse, 1985). Older adults are often more conservative with respect to speed than young adults (Salthouse and Somberg, 1982; Ketcham et al., 2002; Walker et al., 1997; Goggin and Meeuwsen, 1992; Darling et al., 1989). The question arises as to whether such differences are caused by changes in the neurophysiological factors or by different cognitive strategies. Do older adults purposely slow down their movements to ensure that they are made with a high level of accuracy? Most of the studies have attributed the observed slowing in movement control to physiological factors with only a few examining directly whether the speed-accuracy trade-offs actually exist (Salthouse, 1985; Bashore et al., 1989). Salthouse (1985) cited two studies that examined age differences in the speed-accuracy trade-off by manipulating the instructions or incentives that the subjects received for emphasizing speed or accuracy, respectively. Salthouse (1985) further states that both of these studies reported that adults of different ages have specific speed-accuracy characteristics, which show slower response speed as target accuracy becomes more precise, with older adults having slower re-
sponse speeds than young adults at the same level of precision. Thus, age differences in relation to speed production, with an accuracy component, do exist independently of the subject′s emphasis on speed or accuracy. Therefore, in any study showing speed differences in cross-sectional age-group comparisons, the speed versus accuracy relationship should be determined. When an individual trades response speed for response accuracy, it is an example of the influence of cognitive processes on motor performance. Such cognitive strategies make it difficult to accurately determine the amount of change across age groups that is due to neurophysiological factors. This has significant implications for those who work with older adults. First, training programs should challenge older adults to move faster while maintaining accuracy. In addition, when assessing capabilities of older adults, it is important to give older adults more time to complete the task as they perform with accuracy levels similar to young adults when given enough time.
Force Control and Regulation
Force control is an elementary component of movement production because smooth and accurate movements require efficient modulation of force outputs. Changes in the regulation of force outputs lead to decrements in the initiation and control of movements. Older compared with young adults have decreased force outputs and inefficient force regulation making it difficult to initiate and execute movements quickly and accurately across a variety of tasks (Brown, 1996; Campbell, McComas, and Petito, 1973; Clamann, 1993; Cooke et al., 1989; Darling et al., 1989; Davies and White, 1983; Doherty, Vandervoort, and Brown, 1993; Galganski et al., 1993; Izquierdo, Aguado, Gonzalez, Lopez, and Hakkinen, 1999; Larsson and Karlsson, 1978; Milner-Brown, Stein, and Yemm, 1973; Milner, Cloutier, Leger, and Franklin, 1995; Roos, Rice, Connelly, and Vandervoort, 1999; Singh et al., 1999; Stelmach, Teasdale, Phillips, and Worringham, 1989). Stelmach and colleagues (1989), using an isometric task, demonstrated that older adults have a reduced range of force production and higher force output variability compared with young adults. In addition, their rate of force production was substantially slower, as it took 20 ms longer to achieve a force level 45 percent of their maximum (15 N). Ng and Kent-Braun (1999) documented similar findings with older adults. They reported 60-N lower peak force output in older adults compared with young adults and a 20-ms-longer time for force production.
It has been shown that older adults produce multiple bursts of force in tasks when they must achieve targeted force levels approaching maximum (Kinoshita and Francis, 1996; Brown, 1996; Galganski et al., 1993).
This is in contrast to young adults who produce a single burst to the targeted force level. Although these irregularities are small and occur over short periods, they do suggest a reason why control and coordination change with advanced age. Changes in force regulation and control have large implications for most functional tasks—for example, turning a door knob or picking up a glass of liquid. These changes may be a result of motor unit reorganization and muscle composition changes; see “Muscle Composition and Muscle Activation Patterns” below (Erim, Beg, Burke, and de Luca, 1999; Galganski et al., 1993; Hakkinen et al., 1996; Yue, Ranganathan, Siemionow, Liu, and Sahgal, 1999; Clamann, 1993; Davies and White, 1983; Milner-Brown et al., 1973).
Coordination
Coordination is the ability to control a number of movement segments or body parts in a refined manner resulting in a well-timed motor output. The ability to control multiple movement components at any one particular time becomes increasingly difficult with advanced age across a variety of movements including aiming, reaching and grasping, drawing, handwriting, and bimanual coordination tasks (Bennett and Castiello, 1994; Carnahan et al., 1998; Teulings and Stelmach, 1993; Greene and Williams, 1996; Swinnen et al., 1998; Wishart et al., 2000; Ketcham et al., 2001). For example in reach-to-grasp tasks, there are transport and grasp components that must be coordinated both spatially and temporally. Researchers have shown that older adults exhibit unstable temporal coupling between these components (Bennett and Castiello, 1994; Carnahan et al., 1998). Conversely, tasks such as drawing or handwriting that require subjects to control multiple joints in a linked segment have demonstrated that the joints involved require more regulation at fast movement speeds (Teulings and Stelmach, 1993; Ketcham et al., 2001). For example, Ketcham and colleagues (2001) found that in a cyclical drawing task older adults begin to distort their movements at 2.0 Hz (two cycles per second) compared with young adults who begin distortions at 2.5 Hz. It appeared that older adults were unable to accurately control the passive properties of linked segments, resulting in slower, more variable movements. Seidler and colleagues (2002) found that aiming movements away from the body, that required shoulder and elbow participation, became less smooth and decoupled as shoulder contribution increased (Figure 3-3). Furthermore, young adults tended to increase activity of opposing muscles as shoulder involvement increased, whereas older adults coactivated these muscles at high levels during single joint elbow movements and reduced coactivation as shoulder involvement increased.
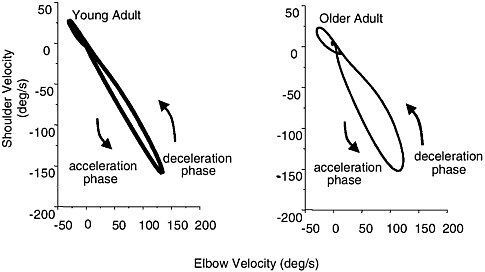
FIGURE 3-3 Coordination of shoulder and elbow joint rotations in a point-to-point aiming task for an example of a young and an older adult.
Bimanual coordination is also a widely researched topic in older adults. In these tasks subjects are asked to produce the same movement with left and right limbs. These movements are typically either in phase, where the two limbs move in the same anatomical direction (homologous muscles activated together), or antiphase, where the two limbs move in the same absolute direction (homologous muscles activated at a 180-degree offset or opposite of each other). In these experiments older adults tend to have difficulty maintaining the more complicated antiphase movements as movement speeds increase (Swinnen et al., 1998; Greene and Williams, 1996; Wishart et al., 2000). Older adults were less accurate at movement speeds of 1.5 and 2.0 Hz compared with young adults, with absolute errors on the order of 20 degrees of relative phase offset. Overall, it has been shown that older adults have increased difficulty controlling and regulating multiple segments to produce smooth motor outputs. Coordination is a part of most tasks of daily living and therefore it is essential to understand breakdowns in control and regulation.
SENSORIMOTOR DECLINES WITH AGE
There are a variety of neurophysiological changes that occur with aging that affect how movements are controlled and regulated in older adults. In this section we focus on declines in proprioception, changes that occur in the composition and activation patterns of muscle, and
changes in flexibility. Although we acknowledge that there are important changes that occur in other anatomical and physiological components, they are beyond the scope of this chapter.
Proprioception
Proprioception is a term used to denote the sense of how body segments are oriented in relation to each other. It depends on receptors in the joints and muscles. Research has shown that older adults have decreased proprioceptive capabilities, which includes joint and muscular sensitivity (Levin and Benton, 1973; Ferrell, Crighton, and Sturrock, 1992; Hurley, Rees, and Newham, 1998; Kaplan, Nixon, Reitz, Rindfleish, and Tucker, 1985; Pai, Rymer, Chang, and Sharma, 1997; Skinner, Barrack, and Cook, 1984; Lord, Rogers, Howland, and Fitzpatrick, 1999). To produce smoothly controlled and regulated movements, the central nervous system must be able to accurately identify movement onsets and determine the exact location of the limb at any given time. A reduced capability to detect the position of the limb has large implications for movement control, and therefore it is important to identify age differences in proprioception. Investigators test proprioceptive capabilities by having individuals reproduce joint positions or recognize joint displacements. Smaller errors reflect better proprioceptive acuity. Research has shown that errors in determining joint positions are approximately three times greater in older adults than in young adults (Petrella, Lattanzio, and Nelson, 1997; Kaplan et al., 1985; Ferrell et al., 1992). Petrella and colleagues (1997) assessed the capability to reproduce joint angles in the knee and found that older adults had errors of ±1.2 degrees compared with young subjects who had errors of only ±0.4 degrees. Skinner and colleagues (1984) have suggested, by comparing subjects across age, that for each additional year the ability to detect changes in joint positions decreases by approximately 0.06 degrees.
In paradigms where subjects are asked to match one limb posture with that of the contralateral limb, errors in older adults are on the order of two times greater than those of young adults (Kaplan et al., 1985; Stelmach and Sirica, 1986; Stelmach and Worringham, 1985). Kaplan et al. (1985) found matching errors of ±4 degrees for young adults and ±7 degrees for older adults. The largest errors in both groups were found for the largest joint angles, with the older adults tending to underestimate the joint angle. The reduced ability in older adults to accurately detect movement or localize a body segment position makes it difficult to produce rapid, well-coordinated movements. This has major functional implications for older adults in a variety of tasks of daily living, from sitting in a chair to reaching for an object.
Muscle Composition and Muscle Activation Patterns
The loss of muscle mass in older adults leads to overall decreases in magnitude of force production (Doherty et al., 1993; Lexell, 1993; Metter et al., 1999; Roos et al., 1997, 1999). It has been shown that the number and the size of muscle fibers decrease in older adults, with the most substantial decrease occurring in fast-twitch fibers, which can be activated quickly for large force outputs, but are unable to sustain force output for long periods of time (Yamada, Masuda, and Okada, 2002). It has been estimated that type II (fast-twitch) muscle fibers decrease by approximately 40 percent in older adults, whereas slow-twitch muscle fibers stay relatively stable across the life span (Aniansson, Hedberg, Henning, and Grimby, 1986; Lexell, 1993; Singh et al., 1999; Yamada et al., 2002). In addition to changes in fiber composition, it has also been shown that there is a reorganization of existing motor units (Doherty et al., 1993; Campbell et al., 1973; Yamada et al., 2002). The existing motor units are reorganized to include more muscle fibers per innervation and subsequently change the way force outputs are achieved. Activation of muscle is more bursty and less smooth than in young adults, resulting in force outputs of large incremental steps (Brown, 1972; McComas, Fawcett, Campbell, and Sica, 1971; Roos et al., 1997, 1999). Moreover, the contractile speed of muscles in older adults is slower than in young adults, which also influences the ability to ramp forces in any given muscle (Davies and White, 1983; Larsson, Li, and Frontera, 1997; Ng and Kent-Braun, 1999; Roos et al., 1997).
For most movements, the underlying muscle activation patterns are organized in a triphasic pattern consisting of two bursts of agonist muscle activity separated by a single burst of antagonistic muscle activity (Berardelli et al., 1996). This triphasic pattern of muscle activity produces a smooth trajectory of a body segment from one position to another, with the first agonist burst initiating the movement, overcoming inertial forces; then the subsequent two bursts decelerate or brake the movement of the limb to the desired position (Berardelli et al., 1996; Brown, 1996; Buneo, Soechting, and Flanders, 1994; Darling et al., 1989). Research has found that older adults do not tightly couple the triphasic agonist-antagonist-agonist activation pattern as young adults do. The timing of the triphasic muscle activity is highly variable, without a clear alternating pattern of agonist-antagonist activation. The antagonist burst is not well defined and occurs abnormally early (Darling et al., 1989). Consequently, older adults often produce movements that have prolonged deceleration patterns or periods of braking of the movement (Berardelli et al., 1996; Brown, 1996; Seidler-Dobrin et al., 1998; Darling et al., 1989).
Joint Characteristics and Flexibility Changes
Movement can be restricted by changes in joint characteristics, including tight ligaments, tendons, and muscle; decreased amounts of cartilage; and thicker consistency or decreased amounts of synovial fluid. With advanced age, the length of muscles around the joints is reduced as a result of lower flexibility of joint structures (Nonaka et al., 2002; Wachtel, Maroudas, and Schneiderman, 1995). The deterioration of these structures is thought to result from changes in the hydration and microstructure of collagen within the joint (Bailey and Mansell, 1997; Wachtel et al., 1995). In addition, the loss of cartilage surface and the chemical characteristic changes of this surface lead to osteoarthritic changes common in older adults (Laver-Rubich and Silbermann, 1985; Beaupre, Stevens, and Carter, 2000; Bernick and Cailliet, 1982). The prevalence of joint diseases such as osteoarthritis is extremely high in older adults, on the order of 80 percent of adults over the age of 65 years (Spirduso, 1995; Reginster, 2002). However, even without diseases of the joints, older adults show substantial loss in the range of motion of their joints due to the anatomical changes in joint structures (Bloem, Allum, Carpenter, Verschuuren, and Honegger, 2002; Ronsky, Nigg, and Fisher, 1995; Nonaka et al., 2002).
Range of movement of a joint refers to the excursion a segment can make before being impeded by bone, tight ligaments and tendons, or muscles. Changes in joint characteristics, muscle composition, and activation characteristics as well as higher levels of disuse in older adults (Raab, Agre, McAdam, and Smith, 1988; Bassey, 1998) lead to overall decreases in flexibility in older adults (Shepard, Berridge, and Montelpare, 1990; Spirduso, 1995). Spirduso (1995) cites a study by Kuo (1990), which tested 3,562 Japanese subjects of ages 25-80 years on trunk flexibility. He reported decreases in range of motion on the order of 20 percent in women and 40 percent in men 55 years and older compared with 25-30-year-old subjects. Decreases in range of motion have been largely reported in the lower limbs, primarily in the hip, knee, and ankle joints of men (Ronsky et al., 1995; Nonaka et al., 2002; Bell and Hoshizaki, 1981; Gehlsen and Whaley, 1990). Decreased flexibility has implications for tasks of daily living as it often determines whether a task, like putting on socks or stockings, safely pulling out into traffic, picking up a dropped object, can be successfully completed (Gehlsen and Whaley, 1990; Nonaka et al., 2002; Shepard et al., 1990; Spirduso, 1995).
CHANGES IN POSTURE AND BALANCE
Most skilled movement involves posture in some manner because there needs to be a stable base of support to perform motor skills such as
pointing, reaching, and grasping. The ability to stabilize posture is important not only for upright stance, but also when performing a variety of upper-extremity movements. Balance and posture control is assessed to understand motor decrements that contribute to instability. Postural stability is commonly measured during quiet stance or following platform perturbations. Older adults have been shown to have deficits during both quiet stance and perturbation-induced sway balance tests (Maki, Holliday, and Fernie, 1990). Older adults exhibited increased sway range (18 percent), sway variability (16 percent), and sway velocity (43 percent) compared with young adults when standing with eyes open (Teasdale, Stelmach, and Breunig, 1991a). Larger variability in postural sway in the absence of vision in older adults is well documented (Woollacott, 1993; Whipple, Wolfson, Derby, Singh, and Tobin, 1993; Peterka and Black, 1990). Older adults had considerable trouble maintaining balance when the eyes were closed (Whipple et al., 1993; Stelmach and Worringham, 1985). Research has also reported that postural sway range and velocity increased when vision was occluded. When vision was occluded and proprioception disturbed, sway substantially increased (65 percent compared with young adults) (Hay, Bard, Fleury, and Teasdale, 1996; Woollacott, 1993; Schieppati, Grasso, Siliotto, and Nardone, 1993; Sheldon, 1963; Teasdale et al., 1991a). Collins and colleagues (1995) studied postural control mechanisms during quiet stance over a 30-s period. They compared early and late sway stability and found that older adults were substantially more unstable than young adult controls in the first 200 ms of the trial when sensory feedback was not available; they became increasingly stable after 200 ms, similar to young adult controls, when sensory feedback had sufficient time to be processed and implemented into control (Collins et al., 1995). Therefore, during quiet stance, disturbances in incoming information greatly affect postural stability in older adults whereas young adults can quickly and effectively recover from such disturbances.
To further test the capability to recover from perturbation, researchers disturb the support surface while a participant is standing. When platform perturbations are introduced that destabilize the body, older adults take longer to initiate corrective or protective actions than young adults. These delays greatly increase the risk of falling as the time period to prevent destabilization is quite short (Tinetti, Speechley, and Ginter, 1988; Stelmach and Worringham, 1985). Furthermore, older adults become unstable even with small perturbations that typically require moderate corrective actions (Maki et al., 1990; Woollacott, Moore, and Hu, 1993; Hu and Woollacott, 1994). Other types of studies found that the combination of visual occlusion and compliant surface conditions resulted in increased variability, velocity, range, and dispersion of postural sway
(Teasdale et al., 1991a, 1991b; Hu and Woollacott, 1994; Maki et al., 1990; Redfern, Moore, and Yarsky, 1997). Overall, data on balance and postural stability in older adults document that older adults show declines in the ability to maintain and recover from disturbances to their upright stance. These decrements result in an increased risk for falls and greatly influence functional tasks of daily living.
SKILL LEARNING
The ability of older adults to learn new skills and relearn previously learned skills is an important area of motor behavior research. Overall, it has been shown that older adults are able to relearn old skills and learn novel skills, but at a much slower rate than young adults (Spirduso, 1995; Murrell, 1970; Salthouse, 1984). Skilled abilities such as coordination, balance, associative learning, and handwriting all improve with extended practice, although not always to the level of young adults (Czaja and Sharit, 1998; Harrington and Haaland, 1992; Lazarus and Haynes, 1997; Strayer and Kramer, 1994; Judge, King, Whipple, Clive, and Wolfson, 1995; Dixon et al., 1993; Woollacott, 1993). Older adults require more time to practice a skill before improvements are shown. For example, Judge and colleagues (1995) found that older adults were able to improve their balance, measured by postural sway range and recovery, when vision and proprioception were distorted but required substantial practice. Similar improvements were found for recovery of balance with a postural perturbation task after substantial practice (Woollacott, 1993). Improvements with practice have also been documented with speed of handwriting. Dixon et al. (1993) found that older adults were almost two times slower than young adults prior to practice, but they improved to being just as fast as the young adults after extensive practice. Although these data document the ability of older adults to relearn a skill, research examining the ability to learn novel tasks will better address the question of motor learning in older adults.
Novel tasks have been studied to assess how older adults are able to learn a new skill. For example, Seidler-Dobrin and Stelmach (1998) and Pratt et al. (1994) had participants perform an aiming task with accuracy requirements. Both of these studies found that, with extensive practice, overall performance (measured by movement time) improved. However, older adults did not improve the distance traveled in the primary submovement, or ballistic phase (see “Movement Components” above) of the movement, whereas young participants did. Others have reported similar overall improvements, but no specific improvements in variables that measure the fundamental components of movement (Cerella et al., 1980; Seidler-Dobrin et al., 1998; Brown, 1996; Darling et al., 1989; Murrell, 1970).
Another example of skill aquisition on a novel task was done by Etnier and Landers (1998), who demonstrated on a mirror star-tracing task that older adults learned at a much slower rate than young adults as measured by time on target. Furthermore, they showed that older adults who had more practice also had better retention. Overall, these data suggest that older subjects are able to learn and improve new skills. Furthermore, the data suggest that improvements may occur in different ways than in young adults, which has implications for motor performance in older adults.
There is some evidence that cognitive strategies, specifically having older adults verbalize a sequence of content-specific cues, improve the speed of learning in older adults (Greenwood, Meeuswsen, and French, 1993; Proteau, Charest, and Chaput, 1994). These strategies help speed up the learning process, make learning more enjoyable, and therefore result in lower attrition rates of older adults in tasks that are novel. In addition there are some studies that show that exercise improves the cognitive abilities (i.e., executive functions) of older adults and subsequently their ability to learn a task (Churchill et al., 2002; Fillit et al., 2002). Churchill et al. (2002) found that executive functions of older adults were maintained or enhanced in individuals with higher levels of fitness. Knowledge about improvements in cognition and movement with exercise has important implications for rehabilitation and training programs for older adults.
TRAINING PROGRAMS
Although there are several studies that have documented age-related decrements in older adults, few have demonstrated how interventions or lifestyle can maintain abilities or even reverse changes observed with advanced age. There is evidence that maintaining an active lifestyle preserves motor functions (Spirduso, 1975; Spirduso and Clifford, 1978; Raab et al., 1988; Fatouros et al., 2002; Gehlsen and Whaley, 1990; Girouard and Hurley, 1995; Drowatzky and Drowatzky, 1999; Morey et al., 1999). Spirduso (1975) and Spirduso and Clifford (1978) performed a study in which they compared active and sedentary young and older adults on reaction and movement time skills. They also addressed whether specificity of skill or overall general fitness influenced reaction and movement time performance by testing an active group, subjects who were either runners or racquet sport players. Spirduso and Clifford also found that older active adults had faster reaction times and movement times than sedentary men of the same age regardless of whether they were runners or racquet sport players. Although these studies have been criticized for not having adequate controls, which may lead to different outcomes in the magnitude of differences, they nevertheless suggest that older adults
who maintain an active lifestyle show positive effects of exercise on the production of movement.
Exercise training (both aerobic and strength) has also been shown to have general beneficial effects on strength and flexibility in older adults. It has been found that exercise training slows the adverse affects of aging even in those who start exercising as late as 80 years of age (Cress et al., 1999). Several studies have shown that light resistance training, stretching, and moderate aerobic exercise have a beneficial effect on strength, balance, flexibility, coordination, and range of motion in older adults (Raab et al., 1988; Fatouros et al., 2002; Gehlsen and Whaley, 1990; Girouard and Hurley, 1995; Drowatzky and Drowatzky, 1999; Morey et al., 1999).
Strength training has been shown to have a specific impact on muscle composition and subsequent motor function. One of the major decrements in older adults is the change in composition of muscle. The decrease in muscle fibers, particularly type II muscle fibers (fast twitch), is largely associated with the lack of use. If the level of exercise training is maintained, the loss of muscle fibers is slowed or does not occur (Rogers and Evans, 1993; Frischknecht, 1998; Fielding, 1995). Weight training increases the number of type II fibers by 20 percent (Drowatzky and Drowatzky, 1999).
It has also been shown that strength training increases maximum torque in plantar flexion movements in the feet, which are important for balance and mobility (Blanpied and Smidt, 1993). Increases in muscle mass and range of motion have been shown to reduce the risk of detrimental falls (Allander, Bjornsson, Olafsson, Sigfusson, and Thorsteinsson, 1974; Hortobagyi and DeVita, 1999; Pendergast, Fisher, and Calkins, 1993). Similar improvements have been found in dynamic balancing of older athletes with increased muscle mass and range of motion (Raty, Impivaara, and Karppi, 2002). Overall, the research shows that maintaining physical activity, including strength and flexibility training, slows the effects of aging on the motor system and may prevent some irreversible injuries or declines.
Other data show more specific improvements in cognitive and motor function with specialized training. Kramer, Hahn, and Gopher (1999) have shown very specific benefits from training over several sessions on a dual-task paradigm. They showed that older adults, compared with young adults, have large time costs, measured by reaction time, when performing tasks that require switching of attention from one task to another. However, with modest practice, older adults were able to reduce the costs of switching between tasks. These improvements were maintained over a 2-month period. These data show that highly specialized training can improve a very specific kind of performance.
Similar specialized training benefits have been shown with balance training interventions, particularly multifactor training (Daubney and Culham, 1999; Hu and Woollacott, 1994; Tang and Woollacott, 1996). Multifactor balance training requires several modalities of sensory information to be processed and integrated simultaneously. A review conducted by Tang and Woollacott (1996) found that multifactor balance training targeted to specific subsystems, working on individual needs, showed the most improvements in balance and postural responses. Training on a set of specific defined deficits in individuals resulted in improvements in stability and recovery from postural disturbances. Shumway-Cooke and colleagues (1997) have demonstrated that balance training programs that include tasks that involve multiple processes increase the attentional demands associated with balance control and become more like real-world experiences in which a person must respond to multiple inputs. Tasks such as maintaining balance while performing rhythmic movements between limbs increase the postural response resources available to individuals and subsequently improve compensatory strategies (Tang and Woollacott, 1998).
Rose and Clark (2000) have reported that a biofeedback-based balance intervention improves balance control in older adults as measured by postural sway. Individuals who participated in biofeedback balance training were able to make quicker corrections to perturbations and able to recover from larger sway dispersions, suggesting more control of their center of gravity. Proprioception and gait training has also been observed to be beneficial to older adults to maintain balance (Gauchard, Jeandel, Tessier, and Perrin, 1999; Galindo-Ciocon, Ciocon, and Galindo, 1995).
Overall, most available data that measure defined motor performance variables suggest that the benefits of intervention training are relatively specific. It needs to be determined whether more generalized intervention strategies such as exercise produce specific improvements in motor function such as speed, accuracy, coordination, and balance control.
Another area that has begun to emerge, but needs to become the forefront of the field, is how technology can assist movement control and accuracy. There have been few studies that have shown the benefits of devices that improve the speed and accuracy of movements, as well as coordination and balance, in older adults. Maki and colleagues (1999) performed a study in which enhanced sensory inserts (raised edge around perimeter of foot) were put in the soles of subjects′ shoes. They found that this intervention improved the efficiency of stabilizing reactions elicited by unpredictable postural perturbations. This device targets improving balance control by enhancing sensation in the soles of individuals′ feet so that postural disturbances can be recognized and corrected before a detrimental outcome occurs. The results may be important in the design of
assistive technologies to reduce instability and risk of falling in older adults. Technological advances in devices that assist older adults should target improving parameters of movement performance that have the largest impact on the skills of daily living. It is important for future technological advancements for older adults to incorporate and capitalize on the intact ability of older adults, while compensating for declines. This will lead to improvements in performance with training as well as help older adults maintain skills in which they are proficient.
REFERENCES
Abrams, R.A., Pratt, J., and Chasteen, A.L. (1998). Aging and movement: Variability of force pulses for saccadic eye movements. Psychology and Aging, 13(3), 387-395.
Allander, E., Bjornsson, O.J., Olafsson, O., Sigfusson, N., and Thorsteinsson, J. (1974). Normal range of joint movements in shoulder, hip, wrist and thumb with special reference to side: A comparison between two populations. International Journal of Epidemiology, 3(3), 253-261.
Amrhein, P.C., and Theios, J. (1993). The time it takes elderly and young individuals to draw pictures and write words. Psychology and Aging, 8(2), 197-206.
Amrhein, P.C., Stelmach, G.E., and Goggin, N.L. (1991). Age differences in the maintenance and restructuring of movement preparation. Psychology and Aging, 6(3), 451-466.
Aniansson, A., Hedberg, M., Henning, G.B., and Grimby, G. (1986). Muscle morphology, enzymatic activity, and muscle strength in elderly men: A follow-up study. Muscle and Nerve, 9(7), 585-591.
Bailey, A.J., and Mansell, J.P. (1997). Do subchondral bone changes exacerbate or precede articular cartilage destruction in osteoarthritis of the elderly? [Review]. Gerontology, 43(5), 296-304.
Bashore, T.R., Osman, A., and Heffley, E.F. (1989). Mental slowing in elderly persons: A cognitive psychophysiological analysis. Psychology and Aging, 4(2), 235-244.
Bashore, T.R., Ridderinkhof, K.R., and van der Molen, M.W. (1997). The decline of cognitive processing speed in old age. Current Directions in Psychological Science, 6(6), 163-169.
Bassey, E.J. (1998). Longitudinal changes in selected physical capabilities: Muscle strength, flexibility and body size. Age and Ageing, 27(Suppl 3), 12-16.
Beaupre, G.S., Stevens, S.S., and Carter, D.R. (2000). Mechanobiology in the development, maintenance, and degeneration of articular cartilage. [Comment]. Journal of Rehabilitation Research and Development, 37(2), 145-151.
Bell, R.D., and Hoshizaki, T.B. (1981). Relationships of age and sex with range of motion of seventeen joint actions in humans. Journal Canadien Des Sciences Appliquees Au Sport, 6(4), 202-206.
Bellgrove, M.A., Phillips, J.G., Bradshaw, J.L., and Gallucci, R.M. (1998). Response (re-) programming in aging: A kinematic analysis. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 53(A3), M222-M227.
Bennett, K.M., and Castiello, U. (1994). Reach to grasp: Changes with age. Journal of Gerontology, 49(B1), P1-P7.
Berardelli, A., Hallett, M., Rothwell, J.C., Agostino, R., Manfredi, M., Thompson, P.D., and Marsden, C.D. (1996). Single-joint rapid arm movements in normal subjects and in patients with motor disorders. Brain, 119, 661-674.
Bernick, S., and Cailliet, R. (1982). Vertebral end-plate changes with aging of human vertebrae. Spine, 7(2), 97-102.
Birren, J.E. (1974). Translations in gerontology: From lab to life. Psychophysiology and speed of response. American Psychologist, 29(11), 808-815.
Blanpied, P., and Smidt, G.L. (1993). The difference in stiffness of the active plantarflexors between young and elderly human females. Journal of Gerontology, 48(2), M58-M63.
Bloem, B.R., Allum, J.H., Carpenter, M.G., Verschuuren, J.J., and Honegger, F. (2002). Triggering of balance corrections and compensatory strategies in a patient with total leg proprioceptive loss. Experimental Brain Research, 142(1), 91-107.
Brogmus, G.E. (1991). Effects of age and sex on speed and accuracy of hand movements and the refinements they suggest for Fitts′ law. Paper presented at the Human Factors Society 35th Annual Meeting, September 2-6, San Francisco, CA.
Brown, S.H. (1996). Control of simple arm movements in the elderly. In A.-M. Ferrandez and N. Teasdale (Eds.), Changes in sensory motor behavior in aging. Advances in psychology (Vol. 114, pp. 27-52). North Holland, Amsterdam: Elsevier Science.
Brown, W.F. (1972). A method for estimating the number of motor units in thenar muscles and the changes in motor unit count with ageing. Journal of Neurology, Neurosurgery and Psychiatry, 35(6), 845-852.
Buneo, C.A., Soechting, J.F., and Flanders, M. (1994). Muscle activation patterns for reaching: The representation of distance and time. Journal of Neurophysiology, 71(4), 1546-1558.
Campbell, M.J., McComas, A.J., and Petito, F. (1973). Physiological changes in ageing muscles. Journal of Neurology, Neurosurgery and Psychiatry, 36(2), 174-182.
Carnahan, H., Vandervoort, A.A., and Swanson, L.R. (1998). The influence of aging and target motion on the control of prehension. Experimental Aging Research, 24(3), 289-306.
Cerella, J. (1985). Information processing rates in the elderly. Psychological Bulletin, 98(1), 67-83.
Cerella, J., Poon, L.W., and Williams, D.M. (1980). Age and the complexity hypothesis. In L. Poon (Ed.), Aging in the 1980′s: Psychological issues. Washington, DC: American Psychological Association.
Churchill, J.D., Galvez, R., Colcombe, S., Swain, R.A., Kramer, A.F., and Greenough, W.T. (2002). Exercise, experience and the aging brain. Neurobiology of Aging, 23(5), 941-955.
Clamann, H.P. (1993). Motor unit recruitment and the gradation of muscle force. Physical Therapy, 73(12), 830-483.
Clarkson, P.M. (1978). The effect of age and activity level on simple and choice fractionated response time. European Journal of Applied Physiology, 40, 17-25.
Collins, J.J., DeLuca, C.J., Burrows, A., and Lipsitz, L.A. (1995). Age-related changes in open-loop and closed-loop postural control mechanisms. Experimental Brain Research, 104, 480-492.
Contreras-Vidal, J.L., Teulings, H.L., and Stelmach, G.E. (1998). Elderly subjects are impaired in spatial coordination in fine motor control. Acta Psychologica, 100(1-2), 25-35.
Cooke, J.D., Brown, S.H., and Cunningham, D.A. (1989). Kinematics of arm movements in elderly humans . Neurobiology of Aging, 10(2), 159-65.
Cress, M.E., Buchner, D.M., Questad, K.A., Esselman, P.C., deLateur, B.J., and Schwartz, R.S. (1999). Exercise: Effects on physical functional performance in independent older adults. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 54(5), M242-248.
Czaja, S.J., and Sharit, J. (1998). Ability-performance relationships as a function of age and task experience for a data entry task. Journal of Experimental Psychology: Applied, 4(4), 332-351.
Darling, W.G., Cooke, J.D., and Brown, S.H. (1989). Control of simple arm movements in elderly humans. Neurobiology of Aging, 10(2), 149-157.
Daubney, M.E., and Culham, E.G. (1999). Lower-extremity muscle force and balance performance in adults aged 65 years and older. Physical Therapy, 79(12), 1177-1185.
Davies, C.T., and White, M.J. (1983). Contractile properties of elderly human triceps surae. Gerontology, 29(1), 19-25.
Diggles-Buckles, V., and Vercruyssen, M. (1990). Age-related slowing, S-R compatibility, and stages of information processing. Paper presented at the Human Factors Society 34th Annual Meeting, October 8-12, Orlando, FL.
Dixon, R.A., Kurzman, D., and Friesen, I.C. (1993). Handwriting performance in younger and older adults: Age, familiarity, and practice effects. Psychology and Aging, 8(3), 360-370.
Doherty, T.J., Vandervoort, A.A., and Brown, W.F. (1993). Effects of ageing on the motor unit: A brief review. Canadian Journal of Applied Physiology, 18(4), 331-358.
Drowatzky, K.L., and Drowatzky, J.N. (1999). Physical training programs for the elderly. Clinical Kinesiology, 53(3), 52-62.
Erim, Z., Beg, M.F., Burke, D.T., and de Luca, C.J. (1999). Effects of aging on motor-unit control properties. Journal of Neurophysiology, 82(5), 2081-2091.
Etnier, J.L., and Landers, D.M. (1998). Motor performance and motor learning as a function of age and fitness. Research Quarterly for Exercise and Sport, 69(2), 136-146.
Fatouros, I.G., Taxildaris, K., Tokmakidis, S.P., Kalapotharakos, V., Aggelousis, N., Athanasopoulos, S., Zeeris, I., and Katrabasas, I. (2002). The effects of strength training, cardiovascular training and their combination on flexibility of inactive older adults. International Journal of Sports Medicine, 23(2), 112-119.
Ferrell, W.R., Crighton, A., and Sturrock, R.D. (1992). Age-dependent changes in position sense in human proximal interphalangeal joints. NeuroReport, 3(3), 259-261.
Fielding, R.A. (1995). The role of progressive resistance training and nutrition in the preservation of lean body mass in the elderly. Journal of the American College of Nutrition, 14 (6), 587-594.
Fillit, H.M., Butler, R.N., O′Connell, A.W., Albert, M.S., Birren, J.E., Cotman, C.W., Greenough, W.T., Gold, P.E., Kramer, A.F., Kuller, L.H., Perls, T.T., Sahagan, B.G., and Tully, T. (2002). Achieving and maintaining cognitive vitality with aging. Mayo Clinic Proceeds, 77(7), 681-696.
Fitts, P.M. (1954). The information capacity of the human motor system in controlling the amplitude of movement. Journal of Experimental Psychology, 47, 381-391.
Fozard, J.L., Vercryssen, M., Reynolds, S.L., Hancock, P.A., and Quilter, R.E. (1994). Age differences and changes in reaction time: The Baltimore Longitudinal Study of Aging. Journal of Gerontology, 49(4), P179-189.
Frischknecht, R. (1998). Effect of training on muscle strength and motor function in the elderly. Reproduction, Nutrition, Development, 38(2), 167-174.
Galganski, M.E., Fuglevand, A.J., and Enoka, R.M. (1993). Reduced control of motor output in a human hand muscle of elderly subjects during submaximal contractions. Journal of Neurophysiology, 69(6), 2108-2115.
Galindo-Ciocon, D.J., Ciocon, J.O., and Galindo, D.J. (1995). Gait training and falls in the elderly. Journal of Gerontological Nursing, 21(6), 10-17.
Gauchard, G.C., Jeandel, C., Tessier, A., and Perrin, P.P. (1999). Beneficial effect of proprioceptive physical activities on balance control in elderly human subjects. Neuroscience Letters, 273(2), 81-84.
Gehlsen, G.M., and Whaley, M.H. (1990). Falls in the elderly: Part II, Balance, strength, and flexibility. Archives of Physical Medicine and Rehabilitation, 71(10), 739-741.
Girouard, C.K., and Hurley, B.F. (1995). Does strength training inhibit gains in range of motion from flexibility training in older adults? Medicine and Science in Sports and Exercise, 27(10), 1444-1449.
Goggin, N.L., and Meeuwsen, H.J. (1992). Age-related differences in the control of spatial aiming movements. Research Quarterly for Exercise and Sport, 63(4), 366-372.
Goggin, N.L., and Stelmach, G.E. (1990). Age-related differences in a kinematic analysis of precued movements. Canadian Journal on Aging, 9(4), 371-385.
Gottsdanker, R. (1982). Age and simple reaction time. Journal of Gerontology, 37(3), 342-348.
Greene, L.S., and Williams, H.G. (1996). Aging and coordination from the dynamic pattern perspective. In A. Ferrandez and N. Teasdale (Eds.), Changes in sensory motor behavior in aging (Vol. 114, pp. 89-131). North Holland, Amsterdam: Elsevier Science.
Greenwood, M., Meeuswsen, H., and French, R. (1993). Effects of cognitive learning strategies, verbal reinforcement, and gender on the performance of closed motor skills in older adults. Activities, Adaptation and Aging, 17(3), 39-53.
Gutman, S.R., Latash, M.L., Almeida, G.L., and Gottlieb, G.L. (1993). Kinematic description of variability of fast movements: Analytical and experimental approaches. Biological Cybernetics, 69(5-6), 485-492.
Hakkinen, K., Kraemer, W.J., Kallinen, M., Linnamo, V., Pastinen, U. M., and Newton, R.U. (1996). Bilateral and unilateral neuromuscular function and muscle cross-sectional area in middle-aged and elderly men and women. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 51(1), B21-29.
Harrington, D.L., and Haaland, K.Y. (1992). Skill learning in the elderly: Diminished implicit and explicit memory for a motor response. Psychology and Aging, 7(3), 425-434.
Hart, B.A. (1980). Fractionated reflex and response times in women by activity level and age. Unpublished doctoral dissertation. University of Massachusetts, Amherst.
Hay, L., Bard, C., Fleury, M., and Teasdale, N. (1996). Availability of visual and proprioceptive afferent messages and postural control in elderly adults. Experimental Brain Research, 108(1), 129-139.
Hick, W. (1952). On the rate of gain of information. Quarterly Journal of Experimental Psychology, 4, 11-26.
Hines, T. (1979). Information feedback, reaction time and error rates in young and old subjects. Experimental Aging Research, 5(3), 207-215.
Hortobagyi, T., and DeVita, P. (1999). Altered movement strategy increases lower extremity stiffness during stepping down in the aged. Journals of Gerontology: Biological Sciences and Medical Sciences, 54(2), B63-70.
Hsu, S.H., Huang, C.C., Tsuang, Y.H., and Sun, J.S. (1997). Age differences in remote pointing performance. Perceptual and Motor Skills, 85(2), 515-527.
Hu, M.H., and Woollacott, M.H. (1994). Multisensory training of standing balance in older adults: II. Kinematic and electromyographic postural responses. Journal of Gerontology, 49(2), M62-M71.
Hurley, M.V., Rees, J., and Newham, D.J. (1998). Quadriceps function, proprioceptive acuity and functional performance in healthy young, middle-aged and elderly subjects. Age and Ageing, 27(1), 55-62.
Izquierdo, M., Aguado, X., Gonzalez, R., Lopez, J.L., and Hakkinen, K. (1999). Maximal and explosive force production capacity and balance performance in men of different ages. European Journal of Applied Physiology and Occupational Physiology, 79(3), 260-267.
Jordan, T.C., and Rabbitt, P.M. (1977). Response times to stimuli of increasing complexity as a function of ageing. British Journal of Psychology, 68(2), 189-201.
Judge, J.O., King, M.B., Whipple, R., Clive, J., and Wolfson, L.I. (1995). Dynamic balance in older persons: Effects of reduced visual and proprioceptive input. Journal of Gerontology, 50A(5), M263-M270.
Kaplan, F.S., Nixon, J.E., Reitz, M., Rindfleish, L., and Tucker, J. (1985). Age-related changes in proprioception and sensation of joint position. Acta Orthopaedica Scandinavica, 56(1), 72-74.
Ketcham, C.J., and Stelmach, G.E. (2002). Motor control of older adults. In D.J. Ekerdt, R.A. Applebaum, K.C. Holden, S.G. Post, K. Rockwood, R. Schulz, R.L. Sprott, and P. Uhlenberg (Eds.), Encyclopedia of aging. New York: Macmillan Reference USA.
Ketcham, C.J., Dounskaia, N., and Stelmach, G.E. (2001). Older adults demonstrate trajectory distortions in multijoint coordination. Society for Neuroscience Abstracts, 27(Program number 834.1).
Ketcham, C.J., Seidler, R.D., Van Gemmert, A.W., and Stelmach, G.E. (2002). Age-related kinematic differences as influenced by task difficulty, target size, and movement amplitude. Journals of Gerontology: Psychological Sciences and Social Sciences, 57(1), P54-64.
Kinoshita, H., and Francis, P.R. (1996). A comparison of prehension force control in young and elderly individuals. European Journal of Applied Physiology and Occupational Physiology, 74(5), 450-460.
Kramer, A.F., Hahn, S., and Gopher, D. (1999). Task coordination and aging: Explorations of executive control processes in the task switching paradigm. Acta Psychologica, 101(2-3), 339-378.
Kuo, G.H. (1990). Physical fitness of the people in Taipei including the aged. In M. Kaneko (Ed.), Fitness for the aged, disabled, and industrial worker (pp. 21-24). Champaign, IL: Human Kinetics.
Larish, D.D., and Stelmach, G.E. (1982). Preprogramming, programming, and reprogramming of aimed hand movements as a function of age. Journal of Motor Behavior, 14(4), 322-340.
Larsson, L., and Karlsson, J. (1978). Isometric and dynamic endurance as a function of age and skeletal muscle characteristics. Acta Physiologica Scandinavica, 104(2), 129-136.
Larsson, L., Li, X., and Frontera, W.R. (1997). Effects of aging on shortening velocity and myosin isoform composition in single human skeletal muscle cells. American Journal of Physiology, 272(Cell Physiology 41), C638-C649.
Laver-Rubich, Z., and Silbermann, M. (1985). Cartilage surface charge: A possible determinant in aging and osteoarthritic processes. Arthritis and Rheumatism, 28(6), 660-670.
Lazarus, J.C., and Haynes, J.M. (1997). Isometric pinch force control and learning in older adults. Experimental Aging Research, 23, 179-200.
Levin, H.S., and Benton, A.L. (1973). Age effects in proprioceptive feedback performance. Gerontologia Clinica, 15(3), 161-169.
Lexell, J. (1993). What is the cause of the ageing atrophy? Assessment of the fiber type composition in whole human muscles. In G.E. Stelmach and V. Homberg (Eds.), Sensorimotor impairment in the elderly (pp. 143-153). North Holland, Amsterdam: Elsevier Science.
Lord, S.R., Rogers, M.W., Howland, A., and Fitzpatrick, R. (1999). Lateral stability, sensorimotor function and falls in older people. Journal of the American Geriatrics Society, 47(9), 1077-1081.
Liu, Y.C. (2001). Comparative study of effects of auditory, visual and multimodality displays on drivers′ performance in advanced traveler information systems. Ergonomics, 44(4), 425-442.
Maki, B.E., Holliday, P.J., and Fernie, G.R. (1990). Aging and postural control: A comparison of spontaneous and induced sway balance tests. Journal of the American Geriatrics Society, 38, 1-9.
Maki, B.E., Perry, S.D., Norrie, R.G., and McIlroy, W.E. (1999). Effect of facilitation of sensation from plantar foot-surface boundaries on postural stabilization in young and older adults. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 54(6), M281-M287.
Marteniuk, R.G., MacKenzie, C.L., Jeannerod, M., Athenes, S., and Dugas, C. (1987). Constraints on human arm movement trajectories. Canadian Journal of Psychology, 41(3), 365-378.
McComas, A.J., Fawcett, P.R., Campbell, M.J., and Sica, R.E. (1971) Electrophysiological estimation of the number of motor units within a human muscle . Journal of Neurology, Neurosurgery and Psychiatry, 34(2), 121-131.
Melis, A., Soetens, E., and van der Molen, M.W. (2002). Process-specific slowing with advancing age: Evidence derived from the analysis of sequential effects. Brain and Cognition, 49, 420-435.
Metter, E.J., Lynch, N., Conwit, R., Lindle, R., Tobin, J., and Hurley, B. (1999). Muscle quality and age: Cross-sectional and longitudinal comparisons. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 54(5), B207-B218.
Meyer, D.E., Abrams, R.A., Kornblum, S., Wright, C.E., and Smith, J.E.K. (1988). Optimality in human motor performance: Ideal control of rapid aimed movements. Psychological Review, 95(3), 340-370.
Milner, T.E., Cloutier, C., Leger, A.B., and Franklin, D.W. (1995). Inability to activate muscles maximally during cocontraction and the effect on joint stiffness. Experimental Brain Research, 107(2), 293-305.
Milner-Brown, H.S., Stein, R.B., and Yemm, R. (1973). The contractile properties of human motor units during voluntary isometric contractions . Journal of Physiology, 228(2), 285-306.
Morey, M.C., Schenkman, M., Studenski, S.A., Chandler, J.M., Crowley, G.M., Sullivan, R.J., Jr, Pieper, C.F., Doyle, M.E., Higginbotham, M.B., Horner, R.D., MacAller, H., Puglisi, C.M., Morris, K.G., and Weinberger, M. (1999). Spinal-flexibility-plus-aerobic versus aerobic-only training: Effect of a randomized clinical trial on function in at-risk older adults. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 54(7), M335-M342.
Morgan, M., Phillips, J.G., Bradshaw, J.L., Mittingley, J.B., Iasek, R., and Bradshaw, J.A. (1994). Age-related motor slowness: Simply strategic? Journal of Gerontology, 49(3), M133-M139.
Murrell, F.H. (1970). The effect of extensive practice on age differences in reation time. Journal of Gerontology, 25, 268-274.
Ng, A.V., and Kent-Braun, J.A. (1999). Slowed muscle contractile properties are not associated with a decreased EMG/force relationship in older humans. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 54(10), B452-B458.
Nonaka, H., Mita, K., Watakabe, M., Akataki, K., Suzuki, N., Okuwa, T., and Yabe, K. (2002). Age-related changes in the interactive mobility of the hip and knee joints: A geometrical analysis. Gait and Posture, 15(3), 236-243.
Pai, Y.C., Rymer, W.Z., Chang, R.W., and Sharma, L. (1997). Effect of age and osteoarthritis on knee proprioception. Arthritis and Rheumatism, 40(12), 2260-2265.
Pendergast, D.R., Fisher, N.M., and Calkins, E. (1993). Cardiovascular, neuromuscular, and metabolic alteration with age leading to fraility. Journal of Gerontology, 48, 61-67.
Peterka, R.J., and Black, F.O. (1990). Age-related changes in human posture control sensory organization tests. Journal of Vestibular Research, 1, 73-85.
Petrella, R.J., Lattanzio, P.J., and Nelson, M.G. (1997). Effect of age and activity on knee joint proprioception. American Journal of Physical Medicine and Rehabilitation, 76(3), 235-241.
Pohl, P.S., Winstein, C.J., and Fisher, B.E. (1996). The locus of age-related movement slowing: Sensory processing in continuous goal-directed aiming. Journals of Gerontology Series B: Psychological Sciences and Social Sciences, 51(2), P94-P102.
Pratt, J., Chasteen, A.L., and Abrams, R.A. (1994). Rapid aimed limb movements: Age differences and practice effects in component submovements. Psychology and Aging, 9(2), 325-334.
Proteau, L., Charest, I., and Chaput, S. (1994). Differential roles with aging of visual and proprioceptive afferent information for fine motor control. Journal of Gerontology, 49B(3), P100-P107.
Raab, D.M., Agre, J.C., McAdam, M., and Smith, E.L. (1988). Light resistance and stretching exercise in elderly women: Effect upon flexibility. Archives of Physical Medicine and Rehabilitation, 69(4), 268-272.
Raty, H.P., Impivaara, O., and Karppi, S.L. (2002). Dynamic balance in former elite male athletes and in community control subjects. Scandinavian Journal of Medicine and Science in Sports, 12(2), 111-116.
Redfern, M.S., Moore, P.L., and Yarsky, C.M. (1997). The influence of flooring on standing balance among older persons. Human Factors, 39(3), 445-455.
Redfern, M.S., Muller, M.L., Jennings, J.R., and Furman, J.M. (2002). Attentional dynamics in postural control during perturbations in young and older adults. Journals of Gerontology: Biological Sciences and Medical Sciences, 57(8), B298-B303.
Reginster, J.Y. (2002). The prevalence and burden of arthritis. Rheumatology, 41 Supp(1), 3-6.
Rogers, M.A., and Evans, W.J. (1993). Changes in skeletal muscle with aging: Effects of exercise training. Exercise and Sport Sciences Reviews, 21, 65-102.
Romero, D.H., Van Gemmert, A.W.A., Adler, C.H., Bekkering, H., and Stelmach, G.E. (2003). Time delays prior to movement alter the drawing kinematics of elderly adults. Human Movement Science, 22(2), 207-220.
Ronsky, J.L., Nigg, B.M., and Fisher, V. (1995). Correlation between physical activity and the gait characteristics and ankle joint flexibility of the elderly. Clinical Biomechanics, 10(1), 41-49.
Roos, M.R., Rice, C.L., and Vandervoort, A.A. (1997). Age-related changes in motor unit function. Muscle and Nerve, 20(6), 679-690.
Roos, M.R., Rice, C.L., Connelly, D.M., and Vandervoort, A.A. (1999). Quadriceps muscle strength, contractile properties, and motor unit firing rates in young and old men. Muscle and Nerve, 22(8), 1094-1103.
Rose, D.J., and Clark, S. (2000). Can the control of bodily orientation be significantly improved in a group of older adults with a history of falls? Journal of the American Geriatrics Society, 48(3), 275-282.
Salthouse, T.A. (1984). Effects of age and skill in typing. Journal of Experimental Psychology: General, 113, 345-371.
Salthouse, T.A. (1985). Speed of behavior and its implications for cognition. In J.E. Birren and K.W. Schaie (Eds.), Handbook of the psychology of aging (2nd ed., pp. 400-426). New York: Van Nostrand Reinhold.
Salthouse, T.A. (1988). Cognitive aspects of motor functioning. Annals of the New York Academy of Sciences, 515, 33-41.
Salthouse, T.A., and Somberg, B.L. (1982). Isolating the age deficit in speeded performance. Journal of Gerontology, 37, 59-63.
Schaie, K.W. (2004). Cognitive aging. In National Research Council, Technology for adaptive aging (pp. 43-63). Steering Committee for the Workshop on Technology for Adaptive Aging. R.W. Pew and S.B. Van Hemel (Eds.). Board on Behavioral, Cognitive, and Sensory Sciences. Division of Behavioral and Social Sciences and Education. Washington, DC: The National Academies Press.
Schieppati, M., Grasso, M., Siliotto, R., and Nardone, A. (1993). Effect of age, chronic diseases and Parkinsonism on postural control. In G.E. Stelmach and V. Homberg (Eds.), Sensorimotor impairments in the elderly (pp. 355-373). Dordrecht, Amsterdam: Kluwer Academic.
Seidler, R.D., Alberts, J.L., and Stelmach, G.E. (2002). Changes in multi-joint performance with age. Motor Control, 6(1), 19-31.
Seidler-Dobrin, R.D., and Stelmach, G.E. (1998). Persistence in visual feedback control by the elderly. Experimental Brain Research, 119(4), 467-474.
Seidler-Dobrin, R.D., He, J., and Stelmach, G.E. (1998). Coactivation to reduce variability in the elderly. Motor Control, 2(4), 314-330.
Sheldon, J.H. (1963). The effect of age on the control of sway. Gerontology Clinical, 5, 129-138.
Shepard, R.J., Berridge, M., and Montelpare, W. (1990). On the generality of the “sit and reach” test: An analysis of flexibility data for an aging population. Research Quarterly Exercise and Sport, 61(4), 326-330.
Shumway-Cook, A., Woollacott, M., Kerns, K.A., and Baldwin, M. (1997). The effects of two types of cognitive tasks on postural stability in older adults with and without a history of falls. Journals of Gerontology Series A: Biological Sciences & Medical Sciences, 52(4), M232-M240.
Simon, J.R. (1967). Choice reaction time as a function of auditory S-R correspondence, age and sex. Ergonomics, 10(6), 559-564.
Simon, J.R., and Pouraghabagher, A.R. (1978). The effect of aging on the stages of processing in a choice reation time task. Journal of Gerontology, 33(4), 553-561.
Singh, M.A., Ding, W., Manfredi, T.J., Solares, G.S., O′Neill, E.F., Clements, K.M., Ryan, N.D., Kehayias, J.J., Fielding, R.A., and Evans, W.J. (1999). Insulin-like growth factor I in skeletal muscle after weight-lifting exercise in frail elders. American Journal of Physiology, 277(Endocrinology and Metabolism 40), E135-E143.
Skinner, H.B., Barrack, R.L., and Cook, S.D. (1984). Age-related decline in proprioception. Clinical Orthopaedics and Related Research, 184, 208-211.
Slavin, M.J., Phillips, J.G., and Bradshaw, J.L. (1996). Visual cues in the handwriting of older adults: A kinematic analysis. Psychology and Aging, 11(3), 521-526.
Spirduso, W.W. (1975). Reaction and movement time as a function of age and physical activity level. Journal of Gerontology, 30(4), 435-440.
Spirduso, W.W. (1995). Physical dimensions of aging. Champaign, IL: Human Kinetics.
Spirduso, W.W., and Clifford, P. (1978). Replication of age and physical activity effects on reaction time and movement time. Journal of Gerontology, 33(1), 26-30.
Stelmach, G.E., and Goggin, N.L. (1988). Psychomotor decline with age. Physical Activity and Aging, 22, 6-17.
Stelmach, G.E., and Sirica, A. (1986). Aging and proprioception. Age, 9, 99-103.
Stelmach, G.E., and Worringham, C.J. (1985). Sensorimotor deficits related to postural stability: Implications for falling in the elderly [Review]. Clinics in Geriatric Medicine, 1(3), 679-694.
Stelmach, G.E., Goggin, N.L., and Amrhein, P.C. (1988). Aging and the restructuring of precued movements. Psychology and Aging, 3(2), 151-157.
Stelmach, G.E., Goggin, N.L., and Garcia-Colera, A. (1987). Movement specification time with age. Experimental Aging Research, 13(1-2), 39-46.
Stelmach, G.E., Teasdale, N., Phillips, J., and Worringham, C.J. (1989). Force production characteristics in Parkinson′s disease. Experimental Brain Research, 76(1), 165-172.
Strayer, D.L., and Kramer, A.F. (1994). Aging and skill acquisition: Learning-performance distinctions. Psychology and Aging, 9(4), 589-605.
Swinnen, S.P., Verschueren, S.M.P., Bogaerts, H., Dounskaia, N., Lee, T.D., Stelmach, G.E., and Serrien, D.J. (1998). Age-related deficits in motor learning and differences in feedback processing during the production of a bimanual corrdination pattern. Cognitive Neuropsychology, 15 (5), 439-466.
Tang, P.F., and Woollacott, M.H. (1996). Balance control in older adults: Training effects on balance control and the integration of balance control into walking. In A.-M. Ferrandez and N. Teasdale (Eds.), Changes in sensory motor behavior in aging (Vol. 114, pp. 339-367). North Holland, Amsterdam: Elsevier Science.
Tang, P.F., and Woollacott, M.H. (1998). Inefficient postural response to unexpected slips during walking in older adults. Journal of Gerontology, 53A(6), M471-M480.
Teasdale, N., Stelmach, G.E., and Breunig, A. (1991a). Postural sway characteristics of the elderly under normal and altered visual and support surface conditions. Journal of Gerontology, 46A(6), B238-B244.
Teasdale, N., Stelmach, G.E., Breunig, A., and Meeuwsen, H.J. (1991b). Age differences in visual sensory integration. Experimental Brain Reseach, 85, 691-696.
Tedeschi, G., Di Costanzo, A., Allocca, S., Quattrone, A., Casucci, G., Russo, L., and Bonavita, V. (1989). Age-dependent changes in visually guided saccadic eye movements. Functional Neurology, 4(4), 363-367.
Teulings, H.L., and Stelmach, G.E. (1993). Signal-to-noise ratio of handwriting size, force, and time: Cues to early markers of Parkinson′s disease. In G.E. Stelmach and V. Homberg (Eds.), Sensorimotor impairment in the elderly (pp. 311-327). Dordrecht, Amsterdam: Kluver Academic.
Tinetti, M.E., Speechley, M., and Ginter, S.F. (1988). Risk factors for falls among elderly persons living in the community. New England Journal of Medicine, 319, 1701-1707.
Wachtel, E., Maroudas, A., and Schneiderman, R. (1995). Age-related changes in collagen packing of human articular cartilage. Biochimica and Biophysica Acta, 1243(2), 239-243.
Walhovd, K.B., and Fjell, A.M. (2001). Two- and three-stimuli auditory oddball ERP tasks and neuropsychological measures in aging. Neuroreport, 12(14), 3149-3153.
Walker, J., Alicandri, E., Sedney, C., and Roberts, K. (1991). In-vehicle navigation devices: Effects on the safety of driver performance. In Vehicle navigation and information systems conference proceedings (pp. 499-525). Warrendale, PA: Society of Automotive Engineers.
Walker, N., Philbin, D.A., and Fisk, A.D. (1997). Age-related differences in movement control: Adjusting submovement structure to optimize performance. Journals of Gerontology Series B: Psychological Sciences and Social Sciences, 52(1), P40-P52.
Warabi, T., Kase, M., and Kato, T. (1984). Effect of aging on the accuracy of visually guided saccadic eye movement. Annals of Neurology, 16(4), 449-454.
Welford, A.T. (1977). Motor performance. In J.E. Birren and K.W. Schaie (Eds.), Handbook for the psychology of aging. New York: Van Nostrand Reinhold.
Welford, A.T. (1984). Between bodily changes and performance: Some possible reasons for slowing with age. Experimental Aging Research, 10(2), 73-88.
Whipple, R., Wolfson, L., Derby, C., Singh, D., and Tobin, J. (1993). Altered sensory function and balance in older persons. The Journal of Gerontology, 48(SI), 71-76.
Wishart, L.R., Lee, T.D., Murdoch, J.E., and Hodges, N.J. (2000). Effects of aging on automatic and effortful processes in bimanual coordination. Journal of Gerontology, 53B(2), P85-P94.
Woollacott, M.H. (1993). Age-related changes in posture and movement. Journal of Gerontology, 48(SI), 56-60.
Woollacott, M.H., Moore, S., and Hu, M.H. (1993). Improvements in balance in the elderly through training in sensory organization abilities. In G.E. Stelmach and V. Homberg (Eds.), Sensorimotor impairment in the elderly (pp. 377-392). Dordrecht, Amsterdam: Kluver Academic.
Yamada, H., Masuda, T., and Okada, M. (2002). Age-related EMG variables during maximum voluntary contraction. Perceptual and Motor Skills, 95(1), 10-14.
Yue, G.H., Ranganathan, V.K., Siemionow, V., Liu, J.Z., and Sahgal, V. (1999). Older adults exhibit a reduced ability to fully activate their biceps brachii muscle. Journals of Gerontology Series A: Biological Sciences and Medical Sciences, 54(5), M249-M253.





























